


























































































































































Below is the uncorrected machine-read text of this chapter, intended to provide our own search engines and external engines with highly rich, chapter-representative searchable text of each book. Because it is UNCORRECTED material, please consider the following text as a useful but insufficient proxy for the authoritative book pages.
A USTIN H. RIESEN Effects of Visual Environment on the Retina The study of transneuronal effects on neural microanatomy has a rather long history, which I will not attempt to repeat here. (It has been brought up to about 2 years ago elsewhere.9) The forward march in this area has been increasing in momentum. The phenomenon of transneuronal degeneration is no longer just a curiosity, as it was re- garded by many in the 1940's, particularly after LeGros Clark called attention to it in his study of the lateral geniculate nucleus of primates. Transneuronal effects are turning out to be very extensive, and much more needs to be learned about their time course. They are clearly both upstream and downstream effects within the nervous system. The ef- fects are not limited to early development, although they are consider- ably faster during infancy. Later, some are rather slow. EFFECTS OF LIGHT DEPRIVA TION ON PROTEINS One of the more dramatic landmarks dealing specifically with changes in the retina was a study by Brattgaard.2 He reared rabbits in darkness to the age of 10 weeks, and showed that the retinal ganglion cells were markedly retarded in development. He found that 3 weeks of light 249
AUSTIN H. RIESEN stimulation after 10 weeks in the dark brought about a recovery of some cells but virtually no recovery of others. There was a great in- crease in variability of RNA content and also in the relatively stable protein content of the cell body, both nucleolar and cytoplasmic. At about the same time, we were rearing chimpanzees in the dark; we found that, if we kept them in the dark too long, the ganglion cells not only lost out in the race for protein, but died and disappeared. The first signs of this effect could be seen by the age of 3 months as a disk pallor of the retina. The leads from Brattgaard's study induced us to look at RNA in dark-reared rats and kittens and in the remaining cells of the chimpanzees' retinal ganglia. We found that impaired protein metabolism is common to the retinal cells of dark-reared rabbits, rats, cats, and chimpanzees. Rates of change from normal RNA levels appear to vary with species, when average val- ues are examined, and with individual cells (as determined by measures of variability within a particular class of retinal cell). Special staining techniques make these cytochemical determinations possible. Using hematoxylin and eosin staining permitted us to determine only that large numbers of ganglion cells in chimpanzees and monkeys eventually disappeared3 and that in cats the mean thickness of the inner plexiform layer of the retina was significantly reduced.7 At 90 days of age, normally reared rats showed more than seven times the concentration of RNA found in dark-reared littermates. The values for retinal ganglion cells were intermediate when the animals were reared in the dark for 90 days and then in normal diurnal lighting conditions for 60 days. We have not determined visual acuity in these animals, but they can discriminate on visual cliff and visual placing tasks. In cats from 3/4 months to over 3 years old, the differences between light- and dark-reared animals in cells of receptor, bipolar, and ganglion layers are similar in amount, but in no instance are they proportionately as great as those cited for the rat. By making photometric estimates of azure-B binding, we found that the cells from dark-reared cats had cyto- plasmic RNA concentrations between 40% and 55% of those in normal cats, and animals given 1 hr of light daily had intermediate levels, about 60-75%. These data do not reflect additional observations that the mean cytoplasmic cross-sectional areas were also significantly reduced in cells of the dark-reared animals; that finding augments the differ- ences in total cytoplasmic RNA. 250
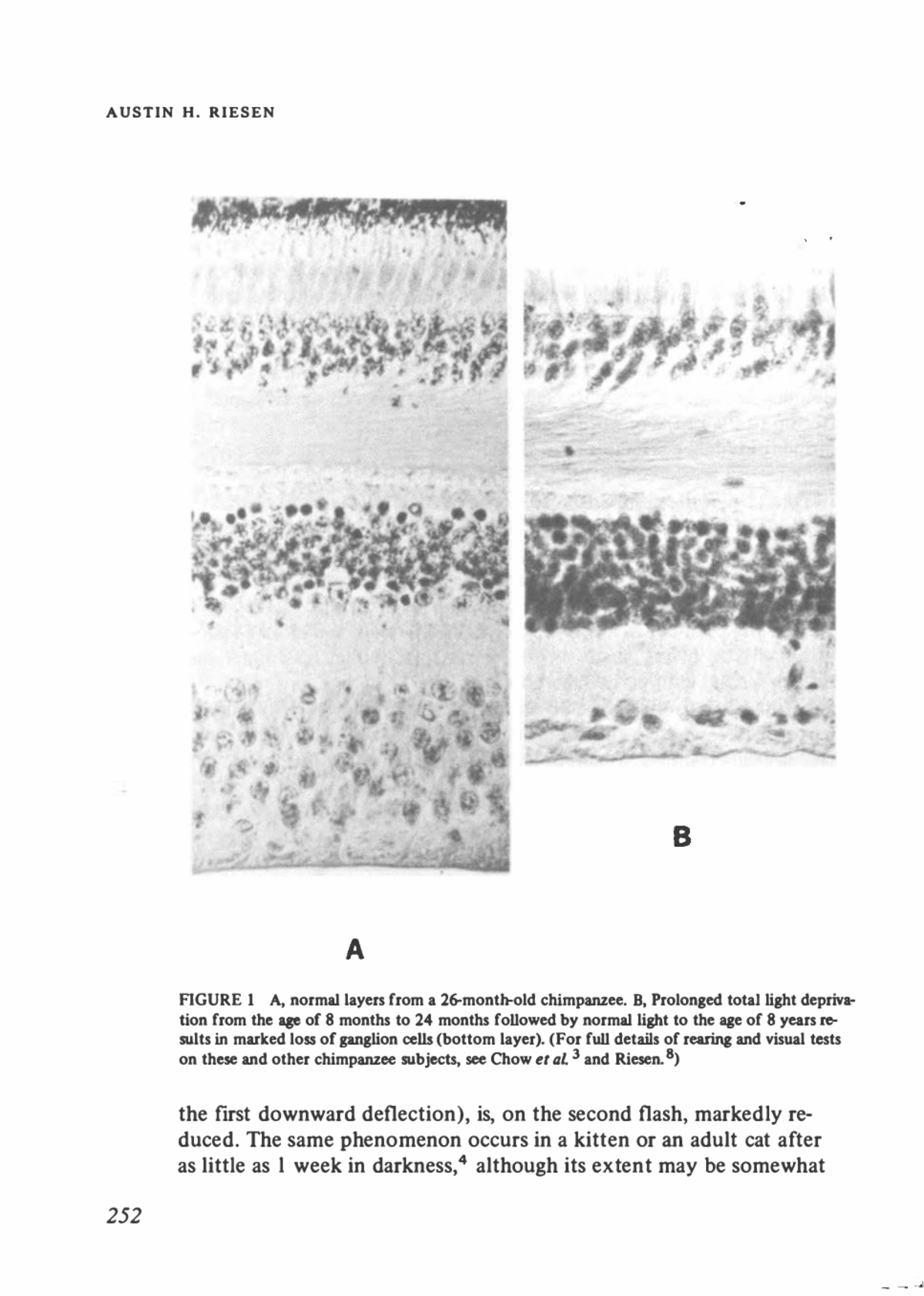
Effects of Visual Environment on the Retina Dark-reared chimpanzees and monkeys and one chimpanzee that was given normal light stimulation from birth to the age of 8 months and then reared in total darkness until the age of 2 years revealed markedly reduced cytoplasmic RNA concentrations in surviving retinal ganglion cells. Depending on the age reached before the primate was placed in the darkroom, the reduction in number of ganglion cells varied from zero to over 90%. When cell loss occurred, visual functions became im- paired,3'8 but reduced RNA concentrations were not necessarily cor- related with losses in visual capacities. The late onset of deprivation, after up to 9 months of normal stimulation, produced more visual im- pairment in higher primates than in the other mammals that we have studied. Figure 1 permits comparison of the parafoveal retina of a control chimpanzee with that of a chimpanzee that was reared normally to the age of 8 months, then lived in total darkness until the age of 24 months, and then lived in normally lighted environments until enucleation at the age of 8 years. The darker appearance of the retina on the right is due to its having been cut in a thicker section (15 ju) than the other (8 ju). More than 90% of the ganglion cells had disappeared in this animal and also in a second chimpanzee reared with 5 min of daily exposure to light from birth to the age of 33 months. These effects were found to be irreversible, whereas shorter intervals of total darkness in other pri- mate subjects resulted in temporary pallor of the optic disk. EFFECTS ON ELECTRICAL ACTIVITY Loss of RNA and cell-volume reduction do not culminate in death of cells in the cat retina. The usual population of ganglion cells is markedly lower in cat, rat, or rabbit than in primates. This may permit the spon- taneous firing of receptor cells, which occurs even in total darkness, to activate ganglion cells frequently enough to ensure their continuing viability. Converging pathways through bipolar fibers would lead to relatively greater use of the individual optic nerve fibers in cats, for ex- ample, than in monkeys, apes, or man. Figure 2 shows electroretinograms (ERG) of a retina that had light deprivation. This represents a monkey that was normally reared and then put in the dark for a relatively short period, about 1 month. The b-wave, the upper deflection (as opposed to the a-wave, represented by 257
AUSTIN H. RIESEN * ' » * ⢠t â¢ft *⢠* ,* £ r. v '. i-â, -^ ^5- ' *. . ^ ^. **"" B FIGURE 1 A, normal layers from a 26-month-old chimpanzee. B, Prolonged total light depriva- tion from the age of 8 months to 24 months followed by normal light to the age of 8 years re- sults in marked loss of ganglion cells (bottom layer). (For full details of rearing and visual tests on these and other chimpanzee subjects, see Chow er al. 3 and Riesen.8) the first downward deflection), is, on the second flash, markedly re- duced. The same phenomenon occurs in a kitten or an adult cat after as little as 1 week in darkness,4 although its extent may be somewhat 252
Effects of Visual Environment on the Retina less. The lower traces are of later responses to flashes given at 2-sec intervals and show that these effects persist. Even when the flashes are spaced rather widelyâas long as 10 sec apartâthe recovery of the b- wave is still incomplete.1 Figure 3 compares some b-wave mean amplitudes in monkeys. The upper curve shows the reduction in the normal monkey's response to the second and third flashes. After 5 weeks in darkness, the b-wave re- sponses of this group of animals to the second and third flashes, on the average, dropped to only one fifth or less of normal. I will not speculate on the mechanism involved, but we do know that lack of stimulation in an adult or mature monkey, as well as in a mature cat, does something to retinal function, as measured by b-wave ampli- tude and its recovery. FIGURE 2 Individual monkey ERG records from surface electrodes, show- ing marked reduction of b-waves in re- sponses after the first in a series of flashes. (Data from R. L. Ramsey.) 253
AUSTIN H. RIESEN 100 0> £ 75- ^50 g c 5 p 25 FIGURE 3 Amplitudes of ERG (fa- wave) responses in normal and light- deprived monkeys. Rhesus monkeys â¢â Normal Oâ 5 wks. no light I 2 3 Successive flashes at 2 sec. intervals BEHA VIORA L COR RE LA TES I would like to indicate some behavioral correlates of lack of visual stimulation in the monkey at birth and shortly thereafter. The data (Figure 4) are from some acuity measurements that Dr. Paul D. Wilson made in our laboratory in Chicago. He raised animals in diffuse light of fluctuating intensity, rather than patterned light; the diffuse light was provided for 2.5 hr each day. On day 20 in this experiment, the animals were first brought into patterned light. Their visual acuity was mea- sured with an optokinetic drum that had stripes of various widths to "pull" the eyes. There was a high degree of variability, but in the first set of three columns (representing three animals), the visual angle that was required to induce eye movements ranged from approximately 20 min to about 170 min on the first day. The subjects improved rather dramatically on the second and third days of patterned light experi- 254
Effects of Visual Environment on the Retina ence, and there was continuing improvement as these 20-day-old mon- keys were reared in a normal environment up to the age of 41 days. The black bars represent normative data taken from a study by Ordy et al.6 Their infant monkeys were tested from the second day after birth, and their improvement corresponds well according to this measure of visual acuity. These results fit the data of Wiesel and Hubel,5'11 showing that it takes patterned light, not diffuse light, to improve the responses of edge- detector units in the visual system. We do not think that this measure of acuity represents retinal improvement itself. Rather, it is a visual-system measure that relies, probably, on midbrain oculomotor organization. Other kinds of behavioral indicators show that patterned light is a 176 ⢠F 160 ⢠144 ⢠i 1 £_Q J o < 112 ' _J |96 ^ F u. 80 o ⢠£ 64 " J D Z z 48 F F 32 i: i 5 :' p ilu s fl r 16 G J 4 r FIRST SECOND THIRD SEVENTH DAY DAY DAY DAY Visually deprived their lirst 20 days of life Normally reared L, I 234 23 jf|G â¢â¢â¢â¢ J FG Bf rt h || rm MB FOURTEENTH TWENTY-FIRST DAY DAY DAYS OF PATTERNED-LIGHT EXPERIENCE FIGURE 4 Improving visual resolving capacities of infant monkeys after 20 days of patterned- light deprivation (gray bars), compared with those of normally reared monkeys (black bars). The normally reared monkeys were studied by Ordy et al.6 Data for the monkeys reared in diffuse light from Riesen et al . 1 ° 255
AUSTIN H. RIESEN critical stimulating factor in the early days of visual development in the higher primates. Table 1 presents data from the rhesus monkeys used in Wilson's experiments.12 The data on neonatally deprived subjects are ranges for 12 animals, of which six were raised in diffuse light for 20 days, and the other six for 60 days. The actual improvement in the particular behavioral measures listed did not differ significantly for the 60-day and 20-day groups, and they paralleled light-reared normal sub- jects that were studied from birth. Some of the differences indicate an advantage for the animals reared in diffuse light. Ocular pursuit of a moving light shows this slight advantage, perhaps: 1-3 days versus 3-12 days of patterned light experience. Inaccurate pursuit of objects is the exception, for this item first appears on days 6-13 in the 20-day-old or 60-day-old pattern-deprived animals, and on days 3-10 in the light- reared subjects. For binocular convergence movements, we used a variety of stimuli for getting the best fixation response possible from a little point of light or a colored object, and then moved the stimulus toward the infant's face. The animals that had matured for 20 days under the diffuse-light conditions did markedly better. They showed convergence for the first time in the second week (days 7-11) after patterned light was initiated. The others did not show this behavior until after the third week (after birth). Accurate reaching to an object starts at about 2 weeks of age in both groups. Starting at 20 days or 60 days provides some advantages Visual Behavior 12 Neonatally Deprived Subjects6 Light-Reared Subjects Consensual pupillary response 1-2C 3-7c Ocular pursuit of light 1-3 3-12 Ocular pursuit of objects 4-12 4-28 Visual placing response 5-16 6-21 Inaccurate reach to object 6-13 3-10 Binocular convergence 7-11 21-35 Accurate reach to object 12-29 14-56 Visual cliff avoidance 11-34 â TABLE 1 Age in Days at Initial Appearance of Visual Behaviors in Monkeys" "Derived from Wilson and Riesen.12 "Patterned light given 2.5 hr daily to neonatally deprived group, beginning on 21st or 61st day after birth. ^Ranges represent days of age for the normally light-reared subjects and days of age minus 20 or 60 for the deprived subjects. 256
Effects of Visual Environment on the Retina that reflect either the motor activity that they have had from birth or maturation, or both. In concluding, I would like to stress one point. This kind of finding, that stimulation is necessary for full development, is not restricted to vision and the retina. There are studies in other sense modalities, and we should take the data as a whole. We have to consider that development, as measured by various growth indicators and by anatomic and electro- physiologic methods, is maximized if there is appropriate stimulation, but that stimulation can also be excessive. There is evidence that 18-24 hr of excessive stimulation will also impair protein content of cells, whether measured by the RNA precursor index, by dry weight, or by total-protein assays. The notion of an optimum is clearly implied in the data, but the determination of the optimum remains for future investigation. REFERENCES 1. Baxter, B. L., and A. H. Riesen. Electroretinogram of the visually deprived cat. Science 134:1626-1627, 1961. 2. Brattgaard, S. O. The importance of adequate stimulation for the chemical composition of retinal ganglion cells during early postnatal development. Acta Radiol. Suppl. 96:1-80, 1952. 3. Chow, K. L., A. H. Riesen, and F. W. Newell. Degeneration of retinal ganglion cells in infant chimpanzees reared in darkness. J. Comp. Neurol. 107:27-42, 1957. 4. Cornwell, A. C., and S. K. Sharpless. Electrophysiological retinal changes and visual deprivation. Vision Res. 8:389-401, 1968. 5. Hubel, D. H., and T. N. Wiesel. Receptive fields of cells in striate cortex of very young, visually inexperienced kittens. J. Neurophysiol. 26:994-1002, 1963. 6. Ordy, J. M., L. C. Massopust, Jr., and L. R. Wolin. Postnatal development of the retina, electroretinogram, and acuity in the rhesus monkey. Exp. Neurol. 5:364- 382, 1962. 7. Rasch, E., H. Swift, A. H. Riesen, and K. L. Chow. Altered structure and com- position of retinal cells in dark-reared mammals. Exp. Cell Res. 25:348-363, 1961. 8. Riesen, A. H. Effects of stimulus deprivation on the development and atrophy of the visual sensory system. Amer. J. Orthopsychiat. 30:23-36, 1960. 9. Riesen, A. H. Sensory deprivation, pp. 117-147. In E. Stellar and J. M. Sprague, Eds. Progress in Physiological Psychology. Volume I. New York: Academic Press, 1966. 285pp. 10. Riesen, A. H., R. L. Ramsey, and P. D. Wilson. Development of visual acuity in rhesus monkeys deprived of patterned light during early infancy. Psychonomic Science 1:33-34, 1964. 257
AUSTIN H. RIESEN 11. Wiesel, T. N., and D. H. Hubel. Effects of visual deprivation on morphology and physiology of cells in the cat's lateral geniculate body. J. Neurophysiol. 26:978- 993, 1963. 12. Wilson, P. D., and A. H. Riesen. Visual development in rhesus monkeys neo- natally deprived of patterned light. J. Comp. Physiol. Psychol. 61:87-95, 1966. DISCUSSION DR. DENENBERG: You indicated that, in the rat, 90 days of visual deprivation produces 10% or 15% of normal RNA. But I recall reports that animals raised in total darkness still do exceptionally well on discrimination-learning tasks. DR. RIESEN: Rats do, up to some age (about 140 days) when they begin to show impairment. That points to the fact that RNA content as a measure does not correlate well with seeing, and I hold no brief for the measured quantities of RNA protein precursors as an index of whether these systems have been organized. I should make one other point here. Once there is a visual system functioning well, one can impose conditions that will eventually result in a ganglion-cell de- pletion, probably even in ganglion-cell death. Our chimpanzees performed better visually about 2 years before we sacrificed them; then, their performance wors- ened up to the time of sacrifice, 4 years after they underwent early deprivation. The early deprivation can, indeed, leave a partially functioning system, and yet eventually lead to death of the system. One animal, Snark, was seeing poorly; we are sure his visual acuity was low when he was 4 years old, and he was around 10 years old when we did the histo- logic examination, by which time he was hardly seeing at all. What the sections showed was a loss of ganglion cells. He also had pallor of the disk, indicating an unhealthy optic nerve, from the time when he was still seeing some things rather well. He was groping around finding things slowly, using what vision he still had, and he had pupillary responses that were still fairly lively. We have data from tests of rather difficult form discrimination in the cat. We reared the animals in darkness for the first 5 months and then light for 5 months, or light for 5 months and then darkness for 5 months. We found that the latter order results in normally rapid, complex form-discrimination learning. We used a block "X" and a block "N" that leaned over. The animals that were reared in the light first learned both form and movement discrimination at 10 months of age as rapidly as any normal cat of the many that we have tested in that situa- tion. There were three animals in that experimental setting, and their scores were in the upper part of the range found for about 12 normally reared cats (see 255
Effects of Visual Environment on the Retina pp. 117-147 in E. Stellar and J. M. Sprague, Eds. Progress in Physiological Psychology. Vol. I. New York: Academic Press, 1966). DR. DOTY: In the chimpanzee experiments, did the degenerative process continue after the animal was restored to normal lighting conditions? DR. RIESEN: Indeed it didâboth in the animal reared in darkness from birth and the animal reared in the light from birth to 8 months and then in darkness for 24 months. The atrophy, as we can tell from ophthalmologic examination, not only did not improve in the light, but it gradually worsened. We could not tell how many ganglion cells were left in that intervening stage, but the restoration of the light did not save the system. DR. VALVERDE: Did you mention that ganglion cells of the retina completely disappeared? DR. RIESEN: In chimpanzees, 90% of the cells disappeared, and we have con- firmed that with a couple of monkeys. In a shorter period, not so many of them disappeared, but there was a reduced count. This same loss of cells will occur in other, related nuclei. I am thinking particularly of lateral geniculate nucleus de- generation, described by Wiesel and Hubel, in the works that I cited. Of course, there have been many studies in which the actual end organ was removed; then, there was slow but progressive degeneration, until, after shrinking, some cells finally disappeared (see the paper in Progress in Physiological Psychology, cited above). DR. LUDLAM: When you raise a diurnal animal, like a monkey, cat, or rabbit, in the dark, other things happen as well. Do these animals eat as well as the others? Are they exercised in the same way as the others? I can envision changes in pro- tein caused by a lack of exercise, improper nutrition, and so forth. DR. RIESEN: We frequently measured activity. Our chimpanzees totaled as much activity for a 24-hr period in the dark as in the light. The distribution of the activity was a little different: they tended to be highly active in the early hours of the morning. They were getting hungrier and hungrier, I suppose, instead of sleeping. They did not develop the usual sleep-wakefulness cycle, but they were as heavy as normally reared animals and they ate as much. We handled, played with, and fed them in the dark. We learned quite early not to isolate these cats or monkeys or chimpanzees. Some animals that we used earlier in our work did not do very well, because we were not providing enough extra inducement for activity. I do not think that this is a nutritional problem in any sense, although there were some indications that calcium metabolism was slightly affected: two of our chimpanzees showed somewhat later ossification-center appearance in the long bones of the body. We were studying various growth indices at the same time, and this is the only effect of that kind. In the past we thought that the skulls of some were more brittle and thinner, but it was highly variable. Others that were dark-reared had normal skull thick- 259
AUSTIN H. RIESEN ness. We do not know what to say about that, except that, in some genetically highly variable animals like the domestic cat, some might require sunlight for utilization of vitamin D in calcification and others require only what we gave them in the food. Many indices were more variable in the dark-reared animals, but enough of them were just right in all measures so that I am no longer very worried about the nutritional variable. DR. ALPERN: I have the impression that there were changes in the electroretino- grams and that histologically you found that the chimpanzees, in contrast with the cats, had particular kinds of ganglion cells. This is not characteristic of elec- troretinograms, in that in a clinical population you find perfectly normal ERG'S in patients, for example, with extensive damage and degeneration in the ganglion-cell layers of the retina. 1 wonder how you would react to the following trivial explanation: If an animal is reared in darkness and has a vigorous pupil re- sponse to the first flash in the stimulus trainâa response that kept the pupil con- tracted much longer than in control animalsâone would expect that the reduced retinal illumination of successive flashes would evoke smaller ERG's. DR. RIESEN: I do not know whether we have any data that would answer that. It is a good possibility. There were RNA losses in the bipolar cells and even in the receptor cell bodies in those eyes. DR. LINDSLEY: You said that the flashes were spaced as much as 10 sec apart; it was not a train of repetitive flashes. DR. RIESEN: No; it is possible to keep an animal's pupil down by giving it a vigor- ous flash of light, and it might stay down that long. Even your pupils, if you keep yourself in the dark for 30 min, would remain contracted that long. We (Science 134:1626-1627,1961; Psychonomic Science 1:33-34, 1964) used homatropine to keep the pupil dilated, as did Cornwell and Sharpless (Vision Res. 8:389-401,1968). 260
F. VALVERDE / A. RUIZ-MARCOS The Effects of Sensory Deprivation on Dendritic Spines in the Visual Cortex of the Mouse: A Mathematical Model of Spine Distribution INTRODUCTION Dendritic spines, a sequence of postsynaptic structures, are small, thorn- like projections on the dendrites of neurons in the mammalian cerebral cortex. They were first discovered by Cajal,3 who believed that they represented normal morphologic formations, although many contempo- raries repeatedly questioned their existence. Electron microscopy not only has demonstrated that dendritic spines are widespread in the cen- tral nervous system (as they are observed to be with the light microscope in Golgi-Cox and methylene blue preparations), but has shown that they are sites of synaptic contact with the same characteristics as those of synapses formed elsewhere with dendritic trunks.8'17'18'25'50 The distribution of fiber terminals in the dendritic pools of cortical neurons reveals some principles of common organization. Some of these principles may be deduced through the study of the distribution of the dendritic spines that represent an accurate imprint of the dendritic syn- aptic coverage. It has been found that the distribution of spines along apical dendrites reflects not only the functional maturation of the cells2,10,11,34 but damage to them14'15-44'45 and possibly the functional state of their afferent fibers.43 267
F. VALVERDE / A. RUIZ-MARCOS In a preliminary study,47 we had found that the mean number of spines per consecutive segment along apical dendrites (the superficial ramifications being excluded) of the layer V pyramidal cells increases exponentially with distance from the cell body. These observations are based on the area striata of the mouse. Further analysis permitted us to partially adjust this exponential relationship in two groups of mice of the same age: controls and mice raised in darkness from birth up to the age of 24 days.43 Since the work of Mann32 and Carlson4 around the turn of the cen- tury, it has been known that significant modifications in retinal nerve cells resulted from prolonged variations in visual stimulation. Recently, Chow et al.,s Weiskrantz,49 and Riesen37 confirmed the existence of important alterations in the mammalian retina after prolonged light deprivation. On the one hand, current studies have proved that modifications of sensory input and environmental conditions can affect the morphology of the cortex and some subcortical structures. Gyllensten21 described statistically significant diminutions in the diameter of cell nuclei and in the quantity of internuclear material. He made these observations in the supragranular layers of the area striata in mice raised in the dark from birth to the age of 1 month. Wiesel and Hubel51 found decreased mean cell areas in the lateral geniculate nuclei of kittens in which one eye was deprived of vision for 3 months. However, they reported that no obvious histologic changes were observed in the retinas, the optic nerves, the superior colliculi, or the visual cortex. Using the Golgi-Cox technique, Coleman and Riesen6 have compared the dendritic fields of the stellate cells of the visual cortex in cats that were reared in the dark with those of their normal siblings. In dark-raised cats, they found shorter den- drites, a reduction in the number of dendrites per cell, and (using SholFs procedure of concentric circles4) a reduction in the number of intersec- tions of dendrites with circles centered around the cell body. On the other hand, Rosenzweig et a/.,38 Bennett et a/.,1 and Diamond et al. n have demonstrated that an enriched environment positively in- creases the brain weight in rats. These authors have suggested that the changes might be due in part to further development of the dendrites. The suggestion was tested by Holloway,27 who found convincing evi- dence that dendritic branching increases in rats raised in environmental complexity. Furthermore, Gyllensten et al.23 have recently described hypertrophy of the supragranular layers of the auditory cortex after 262
The Effects of Sensory Deprivation on Dendritic Spines visual deprivation and have suggested that it might represent a compen- satory mechanism. From the foregoing accounts, it is evident that either a sensory depri- vation or an increase in training can modify the fine structure of the central nervous system. These structural changes can be studied with some of our classical procedures for light microscopy. The purpose of the investigation to be described here was to verify whether the number of spines in the apical dendrites of the large pyramidal cells in the mouse area striata can be modified by decreasing the sensory information that reaches the visual cortex. It was found that the number of spines dimin- ishes in mice enucleated on one side and in mice reared in complete darkness and that in both normal and visually deprived subjects the number of spines along apical dendrites increases exponentially with distance from the cell body. We would like to describe some morphologic details of dendritic spines and their relevant afferent connections, the effects of unilateral enuclea- tion on the number and distribution of dendritic spines, and a mathe- matical model that defines the distribution of spines in the apical den- drites of the layer V pyramidal cells of normal and dark-raised mice. MATERIALS AND METHODS The material for this study consisted of 153 brains stained by the Golgi method48 from a closed colony of black mice derived from an inbred stock. They were collected over a period of 4 years from litters born after early spring matings. The brains used were from animals in three groups: 73 animals raised under normal conditions up to the ages of 10, 14, 19, 21, 24, 36, 48, 78, and 180 days; 58 animals raised in complete darkness, as described elsewhere,43 up to the same ages as the control group; and 22 animals unilaterally enucleated at birth and allowed to survive up to the ages of 24 and 48 days. The spines were counted in about 1,200 complete apical dendrites of pyramidal cells of layer V in the area striata. The apical dendrites were arbitrarily divided into nine consecutive segments 50 ju long, and the spines on each segment were counted. The numbers of spines were plotted and punched on cards. Further processing for study and adjust- ment of the exponential relationship between the mean number of 263
F. VALVERDE / A. RUIZ-MARCOS spines per segment and the distance from the cell body involved the use of IBM 7070 FORTRAN and Autocoder programs written especially for this work. RESULTS Connections of Apical Dendritic Spines in Visual Cortex The synaptic terminals on dendritic spines can be observed easily on Golgi preparations. They appear in the form of short twigs derived from ascending or descending fibers that are closely parallel to the apical den- drites for considerable distances. These parallel fibers were identified as the descending principal axons or their collaterals of the superficial pyramidal cells, or the ascending or descending collaterals of stellate cells.33'34'44 In other cases, a group of fibers approach the dendrite at various angles, leaving a number of clustered terminals over localized portions of the dendrite.44 These two forms of afferent dendritic spine synapses (from parallel fibers and by crossing-over contacts) have been well established in many previous Golgi studies. Figure 1 is a camera lucida drawing of a portion of an apical dendrite of a layer V pyramidal cell at the level of layer IV of the visual cortex. Dendritic spines are clearly visible as small short-side appendages throughout the entire portion of the dendrite. Several fibers (2-6) ap- proach the dendrite to make synaptic contacts on the spines. Fiber 1 is the main descending axon of one superficial pyramidal cell. This axon partly follows the dendrite and appears visible in the lower part of the drawing, where it runs parallel to and contacts the dendritic spines (sp). We have been particularly interested in the establishment of the cir- cuit that relates the specific afferents of area striata with the apical den- drites of layer V pyramidal cells. On the one hand, the specific visual afferents might synapse directly on the dendrites. Several indirect argu- ments pointed this out in previous studies,15'44 but, to our knowledge, this articulation has never been observed. The specific afferents appear difficult to stain with the Golgi method in subjects over 3 weeks old, and only in younger animals is the identification of such fibers clear; but the dendritic spines are not fully developed, and the synaptic forma- tions involved in this connection may be absent. On the other hand, the specific visual afferents synapse on the dendrites of intracortical asso- 264
The Effects of Sensory Deprivation on Dendritic Spines 30 fj MK1T PSR 13 FIGURE 1 Camera lucida drawing of portion of apical dendrite of a layer V pyramidal cell of visual cortex of nor- mal 48-day-old mouse. Golgi method. 265
F. VALVERDE / A. RUIZ-MARCOS elation cells in layers IV and III, which in turn contact the apical den- drites through their short axons. We believe that this multisynaptic articulation is more frequent and most interesting, in that it increases the possibilities of modulatory effects on the pyramidal cells. Figure 2 is a camera lucida drawing of one stellate cell in layer IV of the area striata of a normal mouse 48 days old. The dendrites radiate in two opposite bunches: the ascending one extends widely through layers IV and III, and a thick descending dendrite penetrates into layer V and bifurcates. The dendrites have numerous spines. The axon of this cell (la) descends initially from its origin at the base of the descending den- drite and soon emits numerous collaterals (2a-9a). For example, col- lateral 4a originates in layer V and splits into a fiber that immediately turns horizontally and a branch that contacts the dendrite designated 2 at s. (The dendrite is a portion of the apical shaft of one pyramidal cell of layer V.) Likewise, collateral 6a emerges in layer V with several rela- tively large thickenings contacting the same dendrite 2 at two points s. Collateral 7a ascends for a long distance, traversing layers V, IV, and III; and collateral 9a descends in layer V after sending off a second collateral at a right angle (to the left in the figure). The main axonal fiber, now considerably thinner, pursues an obliquely ascending course (lOa), giv- ing off a small twig for dendrite 1, which is another apical shaft of one pyramidal cell of layer V. Effects of Enudeation on Number of Dendritic Spines: Further Details of Axospinodendritic Contacts In Golgi preparations, the apical dendrites of the pyramidal cells of layer V, which are 500-600n deep, as they ascend through layers V, IV, and III appear densely covered with spines, a series of short side ap- pendages representing postsynaptic structures. The entire apical shaft, excluding the terminal superficial ramifications, can be arbitrarily divided into a number of consecutive segments (usually nine or ten in the mouse's visual cortex) each 50 p. long. Three of them were selected for counting dendritic spines: between 50 and 100n, between 150 and 200/n, and between 250 and 300n from the cell body. The first segment is in layer V, the second in layer IV, and the third in layer III. The spines were counted in 540 selected segments in both areae striatae in mice enucleated of the right eye at birth, and the results were compared with those from normal mice of the same ages. 266
The Effects of Sensory Deprivation on Dendritic Spines MK1T PSR 12.13 FIGURE 2 Camera lucida drawing of stellate cell of layer IV of area striata of normal 48-day- old mouse. Golgi method. (Reprinted with permission from Valverde.44) 267
F. VALVERDE / A. RUIZ-MARCOS In segments at the same distance from the cell body and from animals of the same age and condition, the numbers of spines per segment fol- lowed the normal distribution curve. Each segment contained a number of spines that remained, within statistically reasonable limits, around a mean value that was found to be specific for each distance from the cell body. The mean number of spines per segment varied, however, in ani- mals of different age, diminishing significantly in segments in layer IV from the corresponding area striata of mice enucleated at birth. Correspondingly, in the segment in layer IV from the affected areae striatae of enucleated animals, the diminution of the mean number of dendritic spines is statistically significant with respect to mean values from the same segment of normal animals. The difference is significant in both groups, 24 days old (p < 0.001) and 48 days old (p < 0.005). The difference is not significant when the comparison is between normal values and those obtained in the contralateral (right) area striata. For layer III, the same comparison reveals a difference significant beyond the 2.5% level. There was no difference in the number of spines in seg- ments of layer V between enucleated and normal animals of the same age. Figure 3 is a detailed camera lucida drawing of several portions of apical dendrites of deep pyramidal cells traversing layer IV. The draw- ing was made from the area striata of a normal mouse 48 days old. Figure 4 is a detailed camera lucida drawing of a field similar to that of Figure 3 but corresponding to the left area striata of a 48-day-old mouse enucleated at birth on the right side. Several portions of apical dendrites of deep pyramidal cells traversing layer IV clearly show the effects of enucleation. The number of spines has diminished considerably on den- drites 1-5. Dendrite 6 in Figure 4 is accompanied by a fiber coursing in the same direction, showing several short side appendages that contact the dendrite and spines. This fiber could not be identified, but it prob- ably represents a descending axon or collateral of a superficial pyramidal cell, or an ascending fiber of a stellate cell. Axons of both types of cells have been observed frequently to lie parallel to and contact apical den- drites in this manner. Figure 5 A shows at the level of layer IV part of the apical dendrite of one pyramidal cell of layer V of the right (nonaffected) area striata from a mouse 24 days old, enucleated at birth on the right side. Figure 5B shows at the same level part of another apical dendrite from a pyramidal cell of layer V of the area striata from a normal mouse 48 days old. 268
The Effects of Sensory Deprivation on Dendritic Spines > M150T PSL I4 FIGURE 3 Camera lucida drawing of portions of several apical dendrites of layer V pyramidal cells traversing layer IV of area striata of normal 48-day-old mouse. Details of synaptic contacts between various fibers and the dendrites can be seen. Golgi method. (Reprinted with permission from Valverde.44) 269
F. VALVERDE / A. RUIZ-MARCOS M1UT PSL 14,15 ER FIGURE 4 Camera lucida drawing of portions of several apical dendrites of layer V pyramidal cells traversing layer IV of left area striata of 48-day-old mouse enucleated on right side at birth. Compare with Figure 3; note diminution of spines. Same magnification as in Figure 3. Golgi method. (Reprinted with permission from Valverde.44) 270
The Effects of Sensory Deprivation on Dendritic Spines â 0 FIGURE 5 Mosaic photomicrographic reconstruction of two apical dendrites of layer V pyramidal cells at the level of layer IV of area striata. A, 24-day- old mouse enucleated at birth; normal dendrite in the unaffected area striata with contacts in parallel. B, normal 48-day-old mouse; dendrite with crossing synapses. Golgi method. (Reprinted with permission from Valverde.44) 271
F. VALVERDE / A. RUIZ-MARCOS Dendritic spines are more numerous and thickly distributed in older animals. In Figure 5, afferent fibers cross the dendrite at various angles over a small segment, about 20 ju long, which appears densely covered by terminals of these fibers. It seems clear that contacts in parallel (Figure 5A) would have widespread effects over long dendritic portions, whereas crossing synapses (Figure 5B) would affect restricted parts of the dendrite. Repeated observations showed that fibers synapsing i . parallel on dendritic shafts of deep pyramidal cells represent either the main de- scending axon of a superficial pyramidal cell or its collaterals or ascend- ing axonal branches of stellate cells. Synaptic contacts like those in Figure 5B have often been observed in layer IV. We believe that they represent terminations of specific afferent fibers over apical dendrites. In Figure 6, the effects of enucleation at birth on the number of den- dritic spines at the level of layer IV (affected area striata) in apicals of two layer V pyramidal cells (A, 24-day-old mouse; B, 48-day-old mouse) can be clearly observed. Distribution of Apical Dendritic Spines in Visual Cortex of Mice Raised under Normal Conditions: A Mathematical Model The number of dendritic spines was found to increase with distance from the cell body. We have previously defined this relationship for apical dendrites of pyramidal cells of layer V in the area striata of 24- day-old mice.43'47 It can be expressed by the following exponential equation: ye=ym(l-Ke-**), (1) in which ye = mean number of spines per 50-ju segment, ym= maximal number of spines on one segment, e = basis of natural logarithms, B = slope of corresponding regression line, x = distance from cell body, and K = value of (ym - ye )/ym when x = 0. Equation 1 was found to define satisfactorily the distribution of spines along the first seven segments beginning in the pyramidal cell body, but it did not explain the decay of the number of spines occur- 272
The Effects of Sensory Deprivation on Dendritic Spines B FIGURE 6 Mosaic photomicro- graphic reconstruction of two apical dendrites of layer V pyramidal cells at the level of layer IV of area striata. A, 24-day-old mouse enucleated at birth. B, 48-day,old mouse enucleated at birth. Both dendrites belonging to af- fected areae striatae clearly show dimi- nution of spines. Compare with Fig- ure 5. Same magnification as in Fig- ure S. Golgi method. (Reprinted with permission from Valverde.44) 273
F. VALVERDE / A. RUIZ-MARCOS ring distally on the apical dendrite. We have studied the distribution of spines in mice 10, 14, 19, 21, 36, 48, and 180 days old raised under normal conditions and have obtained a sequence of different ye's along the apical dendrites for each age. Figure 7 shows the observed distribu- tion of the mean number of spines per segment in four representative age groups of normal mice. The mean number of spines for the first segment, between the body and 50ju along the apical dendrite, corre- sponds to 25 on the abscissa; the mean number for the segment between 50 and lOOju along the apical dendrite corresponds to 75 on the ab- scissa; and so on. We then confirmed that ye increases distally along the dendrite in all ages studied. But we observed, in the 24-day-old group43 and in older 60 50 I m I 40 30 1 â¢g 20 10 o 10 . 19 - 48 ⢠180 days old hi HL 25 75 125 175 225 275 Distance from cell body (microns) 325 375 425 FIGURE 7 Number and distribution of spines in consecutive 50-/J segments along the apical dendrites of layer V pyramidal cells in the area striata. Sequence of mean values in four representative age groups of normal mice. Curves were fitted by hand. See text for details. 274
The Effects of Sensory Deprivation on Dendritic Spines animals, that the number decreases gradually in more distal segments. The observation of these distributions suggested two factors: the poten- tial factor B responsible for the increase in ye distally along the dendrite and estimated by the slope of the regression line; and the inhibitory fac- tor, IF, antagonistic with 5, gradually hampering ye in such a way that the combination might result in the distributions of Figure 7. It will be seen that IF becomes greater distally along the apical dendrite and also with the age of the animal, effecting the maximal decay of the mean number of dendritic spines in the last segments of 180-day-old mice. Taking into account the specific properties of the exponential func- tion, we have postulated that the observed distributions may be defined by this equation: ye=ym(\-Ke-Bx)e-rK. (2) Thus, Equation 1 would be a particular case of Equation 2 when IF = 0. To test whether Equation 2 defines the observed spine distribution, several programs were written for an IBM 7070. The data were fed into the computer on punched cards. The computer retrieved the mean num- ber of spines (ye) for each dendritic segment, the experimental distribu- tion, and the 95% confidence limits on the basis of a Student's t-test. This program (RV-6801) was the first that gave the basis for further analysis of the spine distribution. Through a specially programmed trial-and-error process (program RV- 6803), the computer found the values of K, B, and IF that best fitted the experimental distribution by regression analysis.46 After obtaining the values of K, B, and IF that gave the highest correlation, the com- puter calculated from Equation 2 the theoretical values of ye and the chi-square values corresponding to the difference between the theoreti- cal and experimental distributions, calculating the goodness of fit by comparing these chi-square values with a series of tabulated chi-square values stored previously. The computer than printed out a graphic for each group of animals of the same age and condition with the experimental and theoretical distribution of the mean number of dendritic spines and their corre- sponding numeric values. The graphic output from this program, repro- duced in Figure 8, corresponds to the distribution of spines along apical dendrites in normal 19-day-old mice. With this program, we have studied routinely the distribution of 275
F. VALVERDE / A. RUIZ-MARCOS 50 40 o 20 10 ym 400.00 B -0.00111 K 1.00718 IF 0.00307 25 75 125 175 22S 275 325 375 425 Distance From C«ll Body in Microns FIGURE 8 Experimental and theoretical distribution of dendritic spines along apical shafts of layer V pyramidal cells of area striata of four normal 19-day-old mice. Total dendrites, 49. Dot, experimental ordinate; asterisk, theoretical ordinate. When difference between theory and experiment is less than one spine per segment, only asterisk is shown. Data prepared from IBM 7070 program RV-6803. Chi value 2.409,8df,p<0.05. spines along apical dendrites of the layer V pyramidal cells of area striata in four groups of mice: Controls: 10, 14, 19, 21, 24, 36, 48, and 180 days old Raised in darkness since birth: 10, 14, 19, 21, 24, 36, 48, and 180 days old Enucleated on right side for study of distribution of dendritic spines in left area striata: 24 and 48 days old Enucleated on right side for study of distribution of dendritic spines in right area striata: 24 and 48 days old In all four groups of striate apical dendrites, the computer obtained highly significant adjustments to the theoretical distribution formu- 276
The Effects of Sensory Deprivation on Dendritic Spines lated by Equation 2 withp values always less than 0.05 (< 0.01 and < 0.005 in several groups). Equation 2 is valid to describe the distribu- tion of spines along apical dendrites in the pyramidal cells of layer V of the area striata of the mouse at the ages and conditions just mentioned, yielding specific values of the coefficients IF, B, and K for each age. We have studied the relationships among the values of these coeffi- cients and the age, T, of the animal, and obtained a series of graphics that suggest the existence of a relationship between IF and B and the age of the animal: IF = IFm ( 1 - IFK . T-1FB ) (3) and B = Bm (\âBK-T-BB), (4) and also suggest a relationship modulated by an exponential function for the values of K: p.T, (5) in which e = basis of natural logarithms, T = age of animal, and IFm,IFK, IFB, B,,, , BK, BB, ]<â, , KK, KB, and P = series of coefficients required for adjustments. To test whether Equations 3-5 would satisfactorily represent the se- quence of values, two programs (RV-6807 and RV-6808) were written for the IBM 7070. An adjustment better than p <0.05 was obtained for all cases. The system of Equations 2-5 constitutes a mathematical model that permits us to determine the distribution of the dendritic spines as a func- tion of age. Through Equations 3-5 it is possible to calculate the values of IF, B, and K corresponding to a given T; then, by appropriate substi- tutions in Equation 2, the distribution in terms of number of spines per segment can be obtained for that animal. Program RV-681 1 resolves the system of equations of the model and prints out a graphic with the spine distribution corresponding to a given age. Figure 9 is an example: after obtaining the data sheet of a predicted distribution corresponding to the 21 -day-old mouse raised in darkness, we counted spines as usual through the microscope on a series of Golgi-stained brains and obtained a spine distribution that fitted the predicted distribution of Figure 9 277
F. VALVERDE / A. RUIZ-MARCOS SO "m " K 1.00860 B 0.00100 IF 0.00353 40 30 20 10 25 75 125 175 225 275 325 375 425 Distance From Cell Body in Microns FIGURE 9 Predicted distribution of dendritic spines along apical shafts of layer V pyramidal cells of area striata corresponding to 21-day-old mice that were raised in darkness. These values fit (/> <0.05) the experi- mental distribution obtained later from microscope countings. Data prepared from IBM 7070 program RV-6811. Determination of Age on the Basis of Distribution of Dendritic Spines With our mathematical model it is possible to solve the inverse problem; i.e., once we know the distribution of dendritic spines and the values of IF, B, and K, we can obtain the age of an animal according to the in- verse of Equations 3-5. We tried several animals to prove the validity of this process.39 In some, we obtained T values differing by 15-20 days from the real ages of the subjects tested, but in others we got fairly good approximations of the true age. For example, the brain of mouse M240, whose age was not previously revealed to us, was stained by the Golgi method. We counted the spines on consecutive segments of 43 apical dendrites. The values for each apical dendrite were punched on individual cards and processed with program RV-6805 to obtain values of IF, B, and K cor- 278
The Effects of Sensory Deprivation on Dendritic Spines responding to the minimal chi-square value. These were substituted in Equations 3-5 to obtain the corresponding T values. Identical T values in a given animal were never obtained. For mouse M240, 21 days old, we obtained a mean T value of 21.77 days. Distribution of Dendritic Spines in Visual Cortex of Enucleated and Dark-Raised Mice Mice enucleated at birth or raised in darkness are subject to a statisti- cally significant diminution of the number of spines, which is most evi- dent at layer IV in enucleated animals and throughout the apical den- drites in dark-raised animals of all the ages we have studied. Figure 10 is a graphic output from program RV-6803 corresponding to the distribution of dendritic spines in the affected area striata (con- 50 40 20 10 ym 400.00 B 0.00115 K 0.99982 IF 0.00365 25 75 125 175 225 275 325 375 425 Distance From Cell Body in Microns FIGURE 10 Experimental and theoretical distribution of dendritic spines along apical shafts of layer V pyramidal cells of affected area striata of four 48-day-old mice enucleated at birth. Total dendrites, 31. See caption of Figure 8. Data prepared from IBM 7070 program RV-6803. Chi value 2.257, 8 df,p <0.05. 279
F. VALVERDE / A. RUIZ-MARCOS tralateral to the enucleated side) of 48-day-old mice enucleated at birth. The distribution can be compared with that obtained for normal mice of the same age (Figure 7). In enucleated mice, we observed somewhat lower mean numbers of spines per segment at classes 125, 175, 225, and 275 corresponding to the level of the apical dendrites traversing layer IV; the differences from the averaged numbers in normal mice were significant. Figure 11 shows the graphic output from the same program corre- sponding to the distribution of dendritic spines in 48-day-old mice raised in darkness. In all age groups of mice raised in darkness that we have studied, highly significant agreements with the theoretical distribu- tion formulated by Equation 2 have been obtained, with p always less than 0.05. The differences from the averaged numbers of spines ob- 50 40 I V) a 30 j 8 6 z 20 10 ym 400.00 B -0.00184 K 1.02462 IF 0.00491 25 75 125 175 225 275 325 375 425 Distance From Cell Body in Microns FIGURE 11 Experimental and theoretical distribution of dendritic spines along apical shafts of layer V pyramidal cells of area striata of nine 48-day-old mice raised in darkness. Total dendrites, 81. See caption of Figure 8. Data prepared from IBM 7070 program RV-6803. Chi value 0.152, 8 df, p <0.005. 250
The Effects of Sensory Deprivation on Dendritic Spines served in normal mice were significant, except for mice whose eyes were not yet open. The distribution of dendritic spines in dark-raised mice, then, may be described by Equation 2. Values of IF, B, and K corresponding with the age of the animal can also be described by Equations 3-5, except func- tion K =f(T), which expresses the exponential modulation observed for normal mice and is not presented here. The mathematical model of the distribution of dendritic spines in dark-raised mice appears to be repre- sented by a set of equations similar to Equations 2-5 for normal mice with a series of coefficients homologous to those corresponding to Equa- tions 2-5.39 Figure 12 is a three-dimensional reconstruction (by Lison's method31) of the distribution of dendritic spines per segment along the apical shafts in relation to age in normal and dark-raised mice from 10 to 180 days old. The reconstruction has been drawn with values of the theoretical distributions given by the computer according to the mathematical model. The distance from the cell body along the apical dendrites is represented by the x-axis; the mean number of spines per 50-ji segment, by the y-axis; and age, by the z-axis. Four sections perpendicular to the z-y plane, corresponding to the distributions at the ages of 10, 19, 48, and 180 days, were used to build these reconstructions. Comparison of the two reconstructions reveals a great difference in mean numbers of spines per segment that is associated with age. There is a sharp increase in the number of spines throughout the apical den- drites in normal mice between 10 and 19 days, which is related to the opening of the eyes. In dark-raised mice, the numbers of spines, except at 10 days, appear always below the corresponding numbers observed in normal mice. The surface of the reconstruction is rather smooth and does not present a crest at the 19-day level. As in normal mice, there is a continuous decrease in the number of spines on the last dendritic seg- ments from the 19-day level, and the number of spines on segments closer to the cell body increases continuously with age, although not as markedly. DISCUSSION Recent observations of Colonnier8 show that little of the dendritic inter- spine surface bears presynaptic formations. His results point out that 281
F. VALVERDE / A. RUIZ-MARCOS Distance from cell body (days) 425 FIGURE 12 Three-dimensional reconstruction of distribution of dendritic spines along apical dendrites and evolution of distribution with age in normal and dark-raised mice. Reconstruction is based on theoretical distributions according to our mathematical model. the vast majority of synaptic contacts are established on the dendritic spines, and consequently these represent almost the total postsynaptic apparatus of most dendrites in the cerebral cortex. Dendritic spines thus are an exact imprint of the presynaptic dendritic load. Their pattern of distribution, number, and arrangement reflect precisely the distribution, number, and arrangement of presynaptic terminations. This interrelation- ship has been found particularly advantageous, in that qualitative and 252
The Effects of Sensory Deprivation on Dendritic Spines quantitative variations from normal spine distribution can specifically indicate the nature and time of visual sensory deprivation, provided that the normal spine frequency and distribution are known. Because the normal spine distribution has been found to be specific for age, quanti- tative mathematical studies of variations can be used to determine age. The mathematical analysis of biologic processes is useful insofar as it clarifies the functional relationships between variables. The results may be presented by equations that form a mathematical model. It should be possible to use a model of a biologic process in interpolating and ex- trapolating to obtain unknown values of variables. This has been one of the purposes of our study of the distribution of dendritic spines and the evolution of the distribution with age in normal and dark-raised mice. Comparative studies of the mammalian cortex have suggested that the brain is organized at macroscopic and cellular levels according to some invariable laws. These laws may appear extremely complicated, but they might also result in very simple patterns of connectivity that become complex in being repeated again and again. The mathematical model that we have introduced39'46 demonstrates that the distribution of den- dritic spines along the apical shafts of layer V pyramidal cells of the area striata in the mouse follows a mathematical law defined by Equa- tion 2, which is valid for all age groups studied. If we now consider that every dendritic spine supports at least one synaptic connection, it is evident that the patterns of connectivity with respect to apical dendrites might be organized in part according to this law. Furthermore, the func- tional relationship between the values of the coefficients IF, B, and K and the time, T, defined by Equations 3-5, or their homologues for dark- raised mice, indicates that the evolution of this process follows particu- lar laws represented by these equations. We have repeatedly found evidence that short axon cells do not con- nect with apical dendrites at random, but according to the pattern of distribution of the apical dendritic spines.46 On the one hand, that means that connections may be established not by chance proximity, but according to particular intraneuronal and interneuronal factors that govern cortical organization. On the other hand, in relation to the dis- tribution of spines along the apical dendrites, we have applied program RV-6803 to the data of spine distribution in man obtained by Marin- Padilla34 and obtained close correlations (p< 0.005) with the theoretical distribution formulated by Equation 2 in all his cases. This is an impor- tant observation because it constitutes evidence that the distribution of 283
F. VALVERDE / A. RUIZ-MARCOS spines along the apical dendrites follows the same laws in man as in the mouse. Our model has been used to predict the distribution of the dendritic spines corresponding to other age groups not previously studied, as well as to find out the age of an experimental animal from the known spine distribution. This last application might be of considerable practical in- terest in a wide field of approaches to the study of brain evolution. The distribution of the dendritic spines in the visual cortex and its variations with the age and condition of the animal have been found highly specific, and it is on this specificity that the validity of the application rests. The results were obtained with fairly good approximation to the real age (p < 0.05) in almost all our examples. Obviously, exactness would be obtained after addition of coefficient values corresponding to more age groups to provide margins for the natural dispersion inherent in all bio- logic processes. In relation to spine loss in enucleated mice, Valverde44 has stated: Minkowski's early observations36 of transneuronal degeneration in the lateral geniculate nucleus after eye removal, first revealed the existence of a functional de- pendence of post-synaptically related neuronal structures, from their afferent fibers. These observations, which were later extended through many well-known studies, led to the general acceptance of the notion that, in cases of close or exclusive depen- dence, the post-synaptic element suffers a process of mild, long lasting and progres- sive degeneration, which may eventually effect a complete loss of the cell. An early report by Donaldson13 on the famous Laura D. Bridgman's case, as well as other reports of cases of human blindness, reviewed by Tsang,42 alleged the exis- tence of cortical atrophy of the visual area. Many of these studies have been dis- credited for the lack of adequate controls, but recent experimental observations in Rodents and Lagomorpha26'28'30-42 proved that removal of the eye has significant effects on the fiber and cell content of the visual area. The evidence so far strongly suggests that transneuronal degeneration is not halted at the first post-synaptic ele- ment, but damage in the main afferent supply may set up a progressive involvement of successive neuronal links of the sensory neuronal chain. Transneuronal degeneration appears, however, difficult to be predicted in terms of severity and time-course. It depends upon at least three main variables: age, species differences, and nuclear formation in question.35 A quantitative relation exists moreover between the amount of reduction of the afferent supply and the resulting degeneration.29 In many instances transneuronal effects do not lead to complete degeneration of the post-synaptic element, but only to production of slight structural changes. In this case we should speak of transneuronal changes rather than degeneration and raise the question, whether or not these changes are similar to structural modifications of neurons resulting from functional demands. 284
The Effects of Sensory Deprivation on Dendritic Spines The diminution of spines observed [in enucleated animals] might represent a transneuronal change of this nature whereby functionless or degenerating specific afferents in layers IV and III would induce the removal of the spines attached nor- mally to these afferents. This assumption is based further on the demonstration that synaptic contacts are not easily broken. Portions of post-synaptic structures remain firmly attached to isolated endings in damaged tissue or in centrifugated prepara- tions. 17.19.20 Colonnier7-9 showed in the cortex that post-synaptic membranes and spines attached to degenerating terminals are also phagocytosed. Histologic alterations in the cortical visual centers of mice raised in complete darkness have been studied by Gyllensten and co-workers.21'22'24 They found decreased mean volume of internuclear material and nuclear size at the age of 1-4 months. The existence of a statistically significant diminution of the mean number of dendritic spines per segment in the apical shafts of layer V pyramidal cells of the visual cortex in dark-raised mice was first reported by Valverde.43 Changes in the morphology of the dendritic spines of young rabbits subjected to visual deprivation for the first 30 days of life have been described by Globus and Scheibel.16 Coleman and Riesen6 showed that stellate cells of layer IV in the visual cortex of cats reared in the dark have smaller dendritic length and fewer dendrites than those of normal animals. All these studies point out, as we have stated,43 that visual sensory deprivation affects the fine struc- ture of the central nervous system and that some structural changes in nerve cells might occur as the result of experience. The problem of spine function has been discussed recently by Scheibel and Scheibel.40 The current point of view is that dendritic spines receive characteristic signal patterns whose spatial and temporal integration might code specifically the function of each cortical neuron. The signifi- cance of the diminution of the number of dendritic spines in some ex- perimental situations (e.g., dark-rearing and enucleation) has been dis- cussed elsewhere, with the spines considered as structures capable of reflecting the functional state and the damage of their afferent fibers.43"45 The distribution of the dendritic spines in dark-raised mice reported here demonstrates that there is a more reduced rate of spine growth after mice open their eyes for all the age groups we have studied. These changes appear to be due entirely to the effects of light deprivation, specifically in the visual cortex; we reported previously43 that no dimi- nution of spines was detected in other cortical areas when mice were reared in darkness. There are two important considerations. First, the distribution of spines along the apical dendrites in dark-raised mice shows mean values 285
F. VALVERDE / A. RUIZ-MARCOS below normal for every dendritic segment, but the characteristic distribu- tion formulated by our mathematical model is maintained for all age groups. This seems to indicate that the supposed somadendritic factor responsible for the distribution of dendritic spines is not affected by visual deprivation. Second, we have not found evidence of partial nor- malization of the number of spines in any group of dark-raised mice. Gyllensten et a/.24 found slight growth and normalization of the volume of internuclear material and the diameters of cell nuclei in mice after long periods in the dark. Finally, we cannot yet identify the intrinsic mechanism that produces the diminution of dendritic spines in dark-raised mice. Two explanations are possible: (1) visual deprivation has a transneuronal, metabolic, or other deleterious effect on the spines, so that some of them would be removed; and (2) dendritic spines would not grow normally in the ab- sence of normal visual inputs. Whatever the effect might be, the theoreti- cal interest of our observations is obvious: they may give new clues to the anatomic plasticity of the brain in relation to behavioral and learning phenomena. The work reported here was supported by U.S. Public Health Service grant TW 00202 03, a grant (Ayuda de Investigation 1968) from the Juan March Foundation to F. Valverde, and a grant from the Sociedad Espafiola de Industrias Quimicas y Farmaceuticas S.A., Division Farmace'utica Lepetit, to F. Valverde and A. Ruiz- Marcos. We are grateful to Mrs. Eva V. Valero for assistance in the preparation of the histologic material and to Miss M. Estrella Esteban, who helped with computer programming. REFERENCES 1. Bennett, E. L., M. C. Diamond, D. Krech, and M. R. Rosenzweig. Chemical and anatomical plasticity of brain. Science 146:610-619, 1964. 2. Cajal, S. R. Les preuves objectives de 1'unite anatomique des cellules nerveuses. Trab. Lab. Invest. Biol. Univ. Madr. 29:1-137, 1934. 3. Cajal, S. R. Sur la structure de 1'ecorce cerebrale de quelques mammiferes. Cellule 7:125-172, 1891. 4. Carlson, A. J. Changes in the Nissl's substance of the ganglion and the bipolar cells of the retina of the Brandt cormorant Phalacrocorax pencillatus during prolonged normal stimulation. Amer. J. Anat. 2:341-347, 1902-1903. 5. Chow, K. L., A. H. Riesen, and F. W. Newell. Degeneration of retinal ganglion cells in infant chimpanzees reared in darkness. J. Comp. Neurol. 107:27-42, 1957. 286
The Effects of Sensory Deprivation on Dendritic Spines 6. Coleman, P. D., and A. H. Riesen. Environmental effects on cortical dendritic fields. I. Rearing in the dark. J. Anat. 102:363-374, 1968. 7. Colonnier, M. Experimental degeneration in the cerebral cortex. J. Anat. 98:47- 53, 1964. 8. Colonnier, M. Synaptic patterns on different cell types in the different laminae of the cat visual cortex. An electron microscope study. Brain Res. 9:268-287, 1968. 9. Colonnier, M. The structural design of the neocortex, pp. 1-23. In J. C. Eccles, Ed. Brain and Conscious Experience: Study week, September 28 to October 4, 1964, of the Pontificia Academia Scientiarum [papers and discussions]. New York: Springer-Verlag, 1966. 591 pp. 10. Conel, J. L. The Postnatal Development of the Human Cerebral Cortex. Volume III. The Cortex of the Three-month Infant. Cambridge, Mass.: Harvard Univer- sity Press, 1947. 158 pp. 11. Conel, J. L. The Postnatal Development of the Human Cerebral Cortex. Volume IV. The Cortex of the Six-month Infant. Cambridge, Mass.: Harvard University Press, 1951. 189pp. 12. Diamond, M. C., D. Krech, and M. R. Rosenzweig. The effects of an enriched environment on the histology of the rat cerebral cortex. J. Comp. Neurol. 123:111-120, 1964. 13. Donaldson, H. H. Anatomical observations on the brain and several sense-organs of the blind deaf-mute, Laura Dewey Bridgman. Amer. J. Psychol. 3:293-342, 1890;4:248-294, 1891. 14. Globus, A., and A. B. Scheibel. Synaptic loci on parietal cortical neurons: termi- nations of corpus callosum fibers. Science 156:1127-1129, 1967. 15. Globus, A., and A. B. Scheibel. Synaptic loci on visual cortical neurons of the rabbit: the specific afferent radiation. Exp. Neurol. 18:116-131, 1967. 16. Globus, A., and A. B. Scheibel. The effect of visual deprivation on cortical neurons: a Golgi study. Exp. Neurol. 19:331-345, 1967. 17. Gray, E. G. Axo-somatic and axo-dendritic synapses of the cerebral cortex: an electron microscope study. J. Anat. 93:420-433, 1959. 18. Gray, E. G. Electron microscopy of synaptic contacts on dendrite spines of the cerebral cortex. Nature 183:1592-1593, 1959. 19. Gray, E. G., and V. P. Whittaker. The isolation of nerve endings from brain: an electron-microscopic study of cell fragments derived by homogenization and centrifugation. J. Anat. 96:79-88, 1962. 20. Gray, E. G., and V. P. Whittaker. The isolation of synaptic vesicles from the central nervous system. J. Physiol. 153:35P-37P, 1960. 21. Gyllensten, L. Postnatal development of the visual cortex in darkness (mice). Acta Morph. Neerl. Scand. 2:331-345, 1958-1959. 22. Gyllensten, L., T. Malmfors, and M. L. Norrlin. Effect of visual deprivation on the optic centers of growing and adult mice. J. Comp. Neurol. 124:149-160, 1965. 23. Gyllensten, L., T. Malmfors, and M. L. Norrlin. Growth alteration in the auditory cortex of visually deprived mice. J. Comp. Neurol. 126:463-469, 1966. 24. Gyllensten, L., T. Malmfors, and M. L. Norrlin-Grettve. Visual and non-visual factors in the centripetal stimulation of postnatal growth of the visual centers in mice. J. Comp. Neurol. 131:549-557, 1967. 25. Hamlyn, L. H. An electron microscope study of pyramidal neurons in the Am- mon's Horn of the rabbit. J. Anat. 97:189-201, 1963. 287
F. VALVERDE / A. RUIZ-MARCOS 26. Hess, A. Optic centers and pathways after eye removal in fetal guinea pigs. J. Comp. Neurol. 109:91-115, 1958. 27. Holloway, R. L., Jr. Dendritic branching: some preliminary results of training and complexity in rat visual cortex. Brain Res. 2:393-396, 1966. 28. Krech, D., M. R. Rosenzweig, and E. L. Bennett. Effects of complex environ- ment and blindness on rat brain. Arch. Neurol. 8:403-412, 1963. 29. Levi-Montalcini, R. The development of the acoustico-vestibular centers in the chick embryo in the absence of the afferent root fibers and of descending fiber tracts. J. Comp. Neurol. 91:209-241, 1949. 30. Lindner, I., and K. Umrath. Veranderungen der Sehsphare I and II in ihrem monokularen und binokularen Teil nach Extirpation eines Auges beim Kaninchen. Deutsch. Z. Nervenheilk. 172:495-525, 1955. 31. Lison, L. Les me'thodes de reconstruction graphique en technique microscopique, pp. 28-42. In A. Policard, Ed. Actualites Scientifiques et Industrielles, Sub-series 553, VI, Histophysiologie. Paris: Hermann, 1937. 42 pp. 32. Mann, G. Histological changes induced in sympathetic, motor, and sensory nerve cells by functional activity: a preliminary note. Originally presented to the Scottish Microbiological Society under the title: "What alterations are produced in nerve calls by work?" 1894. 33. Marin-Padilla, M. Cortical axo-spinodendritic synapses in man: a Golgi study. Brain Res. 8:196-200, 1968. 34. Marin-Padilla, M. Number and distribution of the apical dendritic spines of the layer V pyramidal cells in man. J. Comp. Neurol. 131:475-489, 1967. 35. Matthews, M. R., W. M. Cowan, and T. P. S. Powell. Transneuronal cell degener- ation in the lateral geniculate nucleus of the macaque monkey. J. Anat. 94:145- 169, 1960. 36. Minkowski, M. Uber den Verlauf, die Endigung und die zentrale Representation von gekreuzten und ungekreuzten Sehnervenfasern bei einigen Saugetieren und beim Menschen. Schweiz. Arch. Neurol. Psychiat. 6:201-252 and 7:268-303, 1920. 37. Riesen, A. H. Effects of stimulus deprivation on the development and atrophy of the visual sensory system. Amer. J. Orthopsychiat. 30:23-36, 1960. 38. Rosenzweig, M. R., D. Krech, E. L. Bennett, and M. C. Diamond. Effects of environmental complexity and training on brain chemistry and anatomy: a replication and extension. J. Comp. Physiol. Psychol. 55:429-437, 1962. 39. Ruiz-Marcos, A., and F. Valverde. Mathematical model of the distribution of dendritic spines in the visual cortex of normal and dark raised mice. J. Comp. Neurol. (in press) 40. Scheibel, M. E., and A. B. Scheibel. On the nature of dendritic spines. Report of a workshop. Commun. Behav. Biol. 1:231-265, 1968. 41. Sclioil, D. A. Dendritic organization in the neurons of the visual and motor cortices of the cat. J. Anat. 87:387-406, 1953. 42. Tsang, Y. C. Visual centers in blinded rats. J. Comp. Neurol. 66:211-261, 1937. 43. Valverde, F. Apical dendritic spines of the visual cortex and light deprivation in the mouse. Exp. Brain Res. 3:337-352, 1967. 44. Valverde, F. Structural changes in the area striata of the mouse after enuclea- tion. Exp. Brain Res. 5:274-292, 1968. 45. Valverde, F., and M. E. Esteban. Peristriate cortex of mouse: location and the effects of enucleation on the number of dendritic spines. Brain Res. 9:145-148, 1968. 288
The Effects of Sensory Deprivation on Dendritic Spines 46. Valverde, F., and A. Ruiz-Marcos. Dendritic spines in the visual cortex of the mouse. Introduction to a mathematical model. J. Comp. Neurol. (in press) 47. Valverde, F., and A. Ruiz-Marcos. Light deprivation and the spines of apical dendrites in the visual cortex of the mouse. Anat. Rec. 157:392, 1967. (abstract) 48. Valverde-Garcia, F. Studies on the Piriform Lobe. Cambridge, Mass.: Harvard University Press, 1965. 131 pp. 49. Weiskrantz, L. Sensory deprivation and the cat's optic nervous system. Nature 181:1047-1050, 1958. 50. Whittaker, V. P., and E. G. Gray. The synapse: biology and morphology. Brit. Med. Bull. 18:223-228, 1962. 51. Wiesel, T. N., and D. H. Hubel. Effects of visual deprivation on morphology and physiology of cells in the cat's lateral geniculate body. J. Neurophysiol. 26:978- 993, 1963. DISCUSSION DR. SPERLING: Do you think that some spines are present at first and then fall off as the animal gets older? You have a given number of spines at one age and fewer spines in the same position a little later. What happened to them? DR. VALVERDE: The curves actually crossed the last part of the 180-day-old dis- tribution. We do not know what happened here; it may be that the spines have somehow disappeared. DR. RiESEN: I might point out that the Scheibels (Exp. Neurology 19:331-345, 1967) have reported shriveled spines, some of which they believe will disappear. DR. DOTY: What about the basal dendrites? DR. VALVERDE: We have completed work only on the apical dendrites and are just beginning to work on the basal dendrites, as well as on the dendrites of other neurons. We suspect that our model will fit all spine distribution in most den- drites, but we have to prove that. At present, we have no data on the matter. DR. BUSER: I have found that the anatomic results would fit some electrophysio- logic data of mine. DR. LiNDSLEY: Dr. Valverde, how can you be sure that these fibers are from the lateral geniculate and not from some other source? DR. VALVERDE: This is, of course, very complex in most mammals. The cortex of the mouse is rather simple. Fibers entering area 17 approach layer IV obliquely. They can usually be followed long enough through the white substance to make sure that they do not come from neighboring association cells. There is another distinction. The fibers coming from the lateral geniculate nucleus are very thick 289
F. VALVERDE / A. RUIZ-MARCOS and make complex arborizations, particularly on layers IV and III. Association fibers are much thinner and reach layers II and III primarily. DR. DOTY: It is well known that the primary afferent fibers coming into the striate in primates can be identified on the basis of their coming in on a slant across the cellular columns, whereas all the intercellular arrangements seem to be directly perpendicular to the surface. Inasmuch as you are working with the mathematical treatment of your data, you can anticipate and extrapolate them to infinity. What do they predict for a mouse that lives forever? And, a more serious question, in the synapses of, say, the stellate cells on the pyramidal cells, or of the afferent fibers on either stellate or pyramidal cells, do the endings select a particular portion of the dendrite? Are they limited to only one portion, or might an afferent end on both the basal and the apical dendrites? DR. VALVERDE: FunctionsIF(T) and5(T), two potential functions relating the coefficients IF and B of the principal equation with the age T, asymptotically approach given values, which we defined as IFm and Bm, respectively. Conse- quently, the number of spines tends to stabilize at maturity. We have been working with a procedure for anterograde degeneration for the tracing of pathways, and have found, as on our Golgi material, that specific af- ferents arrive at the cortex undivided; as the fibers pass up to layer IV, they make associations with apical and other dendrites. The basal dendrites of pyramidal cells of layer V are below this level, but we believe that the basal dendrites of the layer III pyramidal cells receive terminals from the specific afferents. It is very difficult, however, to stain these specific afferents, and we have to work this on hundreds of sections. We have not yet observed that specific afferents select par- ticular dendrite portions. DR. DOTY : What about the stellate-cell endings? Do any of them end on both basal and apical dendrites? DR. VALVERDE: Yes. Although in higher mammals there would be more diverse types of cells, in the mouse we have found at least two definite types. There is a type of stellate cell with axons going up to the superficial layers and synapsing on apicals at the level of layers II and HI. The other type makes a very complex axonal arborization, and, in this case, the collaterals of the axon might actually contact both basal and apical dendrites on the same or on other cells. 290
MER TON C. FL OM Early Experience in the Development of Visual Coordination The human visual apparatus is surprisingly well developed at birth, and it proceeds rapidly in its further development. Within hours after birth, the human infant can sustain visual fixation on a stationary target,14 move his eyes conjugately in the direction of an invisible auditory stimu- lus,20 make compensatory eye movements in response to movements of his head,14 and exhibit nystagmus in response to moving black and white bars of various widths.3'6'8'15 Within a few weeks, he can pursue a simple moving target with his eyes.15 And by 7 or 8 weeks, ocular ac- commodation9 and convergence14 can be exhibited. With this early motor development of the eye, there is a coordinate sensory develop- ment that involves a remarkable capacity for human perception and resolution. To what extent does experience affect oculomotor and oculosen- sory development in the human infant? For this large question we have only small answers. In an albino infant, light is diffusely scattered across the retina, which results in lowered visual acuity. If this continual scat- tering of light is not remedied at an early age (for example, by means of an artificial pupil), permanent ocular nystagmus develops.13 Scattering of light and obscuring of the retinal image by congenital cataracts also prevent the normal development of foveal fixation and acuity, even after removal of the cataracts later in life.19 297
MERTON C. FLOM The abnormal experience of continually diffuse imagery, as in albinism and congenital cataracts, is blatantly severe and fortunately rare in hu- mans. A less severe and less rare form of abnormal experience is produced by strabismus ("turned eye"). In this condition, the retinal image of the turned eye receives a continuous flow of visual information different from that received by the fixating eye. Visual deprivation may also result from "suppression" of the turned eye's image. STRABISMUS AND VISUAL ACUITY Some children with strabismus develop in the turned eye an amblyopia, a reduction in visual acuity that cannot be improved with lenses or attributed to disease. The onset of strabismus normally occurs between birth and 5 or 6 years of age (it rarely develops after these ages), and it is relevant to ask whether the acuity of an amblyopic eye is poorer if the onset of the strabismus is earlier or treatment begins later. In 1903, Claude Worth26 reported on 985 children with constant con- vergent strabismus whom he had examined and followed during the previous 10 years. I have reanalyzed Worth's data and have displayed them in a way suited to the present discussion. In Figure 1, Worth's data are presented to show how the time of onset of strabismus and the delay in treatment affected the development of acuity. Displayed on the x-axis is the posttreatment visual acuity, ranging from 20/20 to less than 20/200 acuity and including a category for children who lost the ability of steady monocular fixation. Treat- ment consisted of glasses to neutralize the refractive error, occlusion of the normal eye with a patch or atropine, orthoptic training, and sur- gery on the extraocular muscles. Of the children who had onset of strabismus before 1 year of age and were treated immediately, approximately 90% (y-axis) ultimately ob- tained normal visual acuity of 20/20. If treatment was delayed by as much as 3 months, but no longer, only about 60% or 65% of the chil- dren ultimately obtained normal acuity. If treatment was delayed by more than 3 months, none of them obtained normal acuity, few ob- tained 20/30 or 20/40 acuity, and most had poorer than 20/200 acuity or lost the ability to fixate monocularly with the amblyopic eye. If the onset of strabismus was between 1 and 3 years of age and treatment was delayed by no more than 3 months, about 90% obtained 292
Early Experience in the Development of Visual Coordination 80 40 V) 80 U- O 40 LJ O 60 ff 40 ONSET > 3 YRS. TRE ATMENT ONSET 1-3 YRS. TREATMENT N«72 ONS < I YR. TREATMENT D- ««/o« 20/30 20/60 20/120 20/20 a 20/40 ⢠20/80 azO/200 LOSS OF FIXATION POST - TRE AT M E NT ACUITY FIGURE 1 Data of Claude Worth26 on 985 constant convergent squinters displayed to illustrate the influence of the onset of strabismus and delay in treatment on the develop- ment of visual acuity. 293
MERTON C. FLOM normal acuity. If treatment was delayed by 3-12 months, a reasonable number of the children obtained normal or nearly normal acuity. If treatment was delayed by more than a year, most of the children did not obtain good visual acuity, and approximately 40% lost the ability to obtain monocular fixation. If strabismus occurred after 3 years of age and treatment was delayed by as much as 7 months, about 85% of the children obtained normal acuity. Even if treatment was delayed for 4 years and more, many chil- dren obtained moderate recovery of acuity, and only a small proportion lost the ability to fixate. Worth's results show that normal acuity was not obtained in ambly- opic eyes if treatment was delayed by more than 3 months after onset at the age of less than 1 year, by more than 12 months after onset be- tween 1 and 3 years of age, or by more than 4 years after onset later than 3 years of age. In short, recovery of acuity in amblyopia was re- lated both to the age at onset of strabismus and to the delay of treatment. Unfortunately, both Worth and the subsequent editor (Chavasse) of his book27 made remarks over the years to suggest to others1'4 a criti- cal age of 6-8 years after which recovery of acuity in amblyopia is sup- posedly hopeless. The results of Worth's study do not support the idea of a critical age for recovery in amblyopia. Worth's experimental results seem to be forgotten. Although many doubts have been raised about the validity of Worth's data, for the moment I propose to accept them as valid and to see whether they support the notion of a critical age. Assuming that Worth was able to determine the age of onset precisely and that the time of treatment was relatively independent of the severity of the defect, the data indicate to me no specific age beyond which acuity is irrecoverable. If there is a critical age, it is much earlier than 7 years. Consider Worth's data for the children in whom onset of strabismus occurred before 1 year of age. If treatment was delayed by more than 3 months, practically no children obtained acuity better than 20/200. To emphasize 6-8 years as a critical age is clearly misleading, for Worth's data indicate that treatment begun at 15 months of age is too late for these children. Since Worth's early study, there have been many reports of acuity improvement in amblyopia when treatment was begun at ages up to 60 years. On the basis of research reported in his textbook and in more 294
Early Experience in the Development of Visual Coordination than 10 papers on strabismus and amblyopia, Peter16 concluded ... there is no definite upper age limit in the correction of amblyopia. Until the seventh year, most cases can be restored to normal vision with proper methods; from 7 to 12 the percentage of recoveries is slightly lower, from 12 to 21 years the possibilities are still fair.. .. In an investigation of 91 amblyopes over 5 years old, Scobee17 found that 69% obtained 20/40 or better acuity after treatment. Of 34 ambly- opes whose average age was 8 years, Dowling5 reported that treatment led to 76% obtaining 20/40 or better acuity. Kasser and Feldman12 showed that 31 of 34 adult amblyopes (ages 25-60 years) had improved acuity in the amblyopic eye after treatment. It is relevant to cite here three representative investigators who have used the recently developed "pleoptics" for treating amblyopia. Ban- gerter,2 originator of the term "pleoptics" and director of the world's largest amblyopia treatment center, has reported on some 300 patients as follows (translated from the German): In high grade amblyopia ... the few completely negative cases were limited mainly to children between three and six years of age.. .. The majority of patients who were treated were children between six and twelve years of age; but even among the fifteen and sixteen year olds treatment did not prove hopeless. The percentage of failures in middle grade amblyopia is small. In cases with low- grade amblyopia the treatment was successful almost without exception. The only fact which deserves to be pointed out is that it was possible to obtain essential improvement in a relatively large number of cases beyond the age of childhood. Tommila18 used pleoptics on 137 Finnish amblyopes 6-16 years old and observed that about 60% obtained 20/40 or better acuity and that about one third achieved normal acuity. Girard et a/.7 reported that 96% of 71 amblyopes 6-48 years old showed improvement in acuity after pleoptics; 58% achieved a final acuity of 20/30 or better. The actual success rates in improving acuity in older amblyopes are unimportant here. What is important is the evidence that amblyopia can be improved and even corrected in patients older than 8 years. NORMAL DEVELOPMENT OF ACUITY Let us now consider the development of acuity in the normal infant. Many innovative experiments have been done in the last several years to 295
MERTON C. FLOM determine (or approximate) the visual acuity of the human infant. In 1963, Weymouth21 analyzed most of the infant-acuity data reported to that date. He pointed out that the assembled data are too low, and that the median curve (Figure 2) fitted to these data therefore probably underestimates the rapid development of visual acuity in the human infant. The steep initial rise in the median (middle) curve shows that by 1 year of age, the average child has at least 40% of the acuity he will have as an adult, and by 2 years of age, he has at least 80% of the acuity he will have as an adult. Weymouth argued that the 90th-percentile curve (upper) may actually depict the average corrected (with lenses) acuity for children. On this basis, one could expect the average child to have developed about 73% of his adult acuity by 1 year of age, and about 92% of his adult acuity by 2 years of age. In any case, it is clear that a child's visual acuity develops remarkably rapidly and reaches nearly its maximum by about 2 years of age. Somewhat related to our discussion on the development of acuity and amblyopia in human infants are the experimental results of Hubel and Wiesel.10 They cut loose the right medial rectus muscle of four no 100 >- a: -. V) o ± < -I UJ z (/> 90 80 70 60 50 40 30 20 I 0 0 OMORGAN OSLATAPER â¢WORTH-CHAVASSE ASCOBE E ASCHWARTING VGORMAN el al â¢f FANTZ XDAYTON 0 I 2 34 5 67 8 9 10 II 12 13 14 15 16 17 18 19 20 AGE IN YEARS FIGURE 2 Composite data on visual acuity in children. The central heavy line represents the median acuity; the upper and lower lighter lines represent the 90th and 10th percentiles, re- spectively. (Reprinted with permission from Weymouth.21) 296
Early Experience in the Development of Visual Coordination kittens between 8 and 10 days after birth, thus creating a right diver- gent strabismus. After 3 months to a year, these animals exhibited no behavioral visual defects to suggest an amblyopia in the "turned" eye. This failure to develop an amblyopia in the deviated eye may have re- sulted from the cats' frequently observed fixation with this eye. In any event, the only structural change obtained was a substantial decrease (by one fourth) in the number of striate cortex cells that could be driven from input to both eyes. They concluded that the lack of synergy in the input from the two eyesâas created by strabismusâ caused a disruption in the neural connections that subserve binocular interaction. These results bear more on the problem of abnormal bin- ocular directionalization (e.g., anomalous retinal correspondence) than on the problem of amblyopia. Other experiments by Hubel and Wiesel,11'22-25 in which one eye of young kittens was sutured shut or was covered with a translucent occluder, produced marked histologic changes in the lateral geniculate nucleus, physiologic changes in the striate cortex, and some perceptual impairment. These latter experi- ments with form deprivation in cats speak more relevantly to the problem of the development of acuity in human infants, but care must be taken in extrapolating these results from cat to man. Several points emerge from the preceding discussion. First, the ocu- lomotor and visuosensory (visual acuity) systems are nearly fully devel- oped in the human infant by about 6 and 24 months, respectively. Second, embarrassment of information to one eye during childhood can lead to amblyopia. If the embarrassment occurs during the devel- opmental period of visual acuity (birth to 2 or 3 years), then the ensu- ing amblyopia may include some portion due to impairment of normal development. Embarrassment after this period probably results in a genuine loss of function that was previously present. There is no evi- dence that 6-8 years is a critical age beyond which recovery of acuity in an amblyopic eye is impossible. On the contrary, there is consider- able evidence that many amblyopias can be improved or corrected in late childhood and even in adulthood. If there is a critical age for the treatment of amblyopia, it is more likely to be 1-2 years for children who have experienced visuosensory embarrassment in infancy. I think that we can conclude from the observations discussed that, if it is considered advisable to provide suitable visual experience to as- sist the development of the visual system, this experience should occur quite early in a child's lifeâprobably before 1 or 2 years of age. 297
MERTON C. FLOM REFERENCES 1. Allen, H. F. Make sure your child has two good eyes. Today's Health 34:22-23, 65, 1956. 2. Bangerter, A. Amblyopiebehandlung, pp. 17-18. (2nd ed.) Basel: S. Karger, 1955. 142pp. 3. Dayton, G. O., Jr., M. H. Jones, P. Aiu, R. A. Rawson, B. Steele, and M. Rose. Developmental study of coordinated eye movements in the human infant. I. Visual acuity in the newborn human: a study based on induced optokinetic nystagmus recorded by electro-oculography. Arch. Ophthal. 71:865-870, 1964. 4. Deutsch, P., and R. Deutsch. The hidden threat to children's eyes. Today's Health 43:29-32, 64-65, 1965. 5. Dowling, H. E. Treatment of amblyopia ex anopsia. Arch. Ophthal. 28:369-370, 1942. 6. Fantz, R. L., J. M. Ordy, and M. S. Udelf. Maturation of pattern vision in infants during the first six months. J. Comp. Physiol. Psychol. 55:907-917, 1962. 7. Girard, L. J., M. C. Fletcher, E. Tomlinson, and B. Smith. Results of pleoptic treatment of suppression amblyopia. Amer. Orthopt. J. 12:12-31, 1962. 8. Gorman, J. J., D. G. Cogan, and S. S. Gellis. A device for testing visual acuity in infants. Sightsav. Rev. 29:80-84, 1959. 9. Haynes, H., B. L. White, and R. Held. Visual accommodation in human infants. Science 148:528-530, 1965. 10. Hubel, D. H., and T. N. Wiesel. Binocular interaction in striate cortex of kittens reared with artificial squint. J. Neurophysiol. 28:1041-1059, 1965. 11. Hubel, D. H., and T. N. Wiesel. Receptive fields of cells in striate cortex of very young, visually inexperienced kittens. J. Neurophysiol. 26:994-1002, 1963. 12. Kasser, M. D., and J. B. Feldman. Amblyopia in adults: treatment of those engaged in various industries. Amer. J. Ophthal. 36:1443-1446, 1953. 13. Kestenbaum, A. Clinical Methods of Neuro-ophthalmologic Examination. (2nd rev. and enl. ed.) New York: Grune and Stratton, 1961. 557 pp. 14. Ling, B. C. A genetic study of sustained visual fixation and associated behavior in the human infant from birth to 6 months. J. Genet. Psychol. 61:227-277, 1942. 15. McGinnis, J. M. Eye-movements and optic nystagmus in early infancy. Genet. Psychol. Monogr. 8:321-430, 1930. 16. Peter, L. C. Abstract of discussion, pp. 455-457. In G. P. Guibor. Some pos- sibilities of orthoptic training; a comparative study of control group and treated group of patients with squint, with remarks concerning technic used in orthop- tic training. Arch. Ophthal. 11:433-461, 1934. 17. Scobee, R. G. Esotropia; incidence, etiology, and results of therapy. Amer. J. Ophthal. 34:817-833, 1951. 18. Tommila, V. Results in amblyopia treatment with pleoptophor. Acta Ophthal. 39:439_444? 1961. 19. Von Noorden, G. K., and E. Maumenee. Clinical observations on stimulus- deprivation amblyopia (amblyopia ex anopsia). Amer. J. Ophthal. 65:220-224, 1968. 20. Wertheimer, M. Psychomotor coordination of auditory and visual space at birth. Science 134:1692, 1961. 295
Early Experience in the Development of Visual Coordination 21. Weymouth, F. W. Visual acuity of children, pp. 119-143. In M. J. Hirsch and R. E. Wick, Eds. Vision of Children. Philadelphia: Chilton, 1963. 434 pp. 22. Wiesel, T. N., and D. H. Hubel. Comparison of the effects of unilateral and bilateral eye closure on cortical unit responses in kittens. J. Neurophysiol. 28:1029-1040, 1965. 23. Wiesel, T. N., and D. H. Hubel. Effects of visual deprivation on morphology and physiology of cells in the cat's lateral geniculate body. J. Neurophysiol. 26:978-993, 1963. 24. Wiesel, T. N., and D. H. Hubel. Extent of recovery from the effects of visual deprivation in kittens. J. Neurophysiol. 28:1060-1072, 1965. 25. Wiesel, T. N., and D. H. Hubel. Single-cell responses in striate cortex of kittens deprived of vision in one eye. J. Neurophysiol. 26:1003-1017, 1963. 26. Worth, C. A. Squint: Its Causes, Pathology and Treatment. Philadelphia: The Blakiston Co., 1903. 229 pp. 27. Worth, C. A. Squint: or, The Binocular Reflexes and the Treatment of Strabismus. F. B. Chavasse, Ed. (7th ed.) Philadelphia: The Blakiston Co., 1939. 688 pp. DISCUSSION D R . KAG AN : In Worth's data, do we have a problem of whether the doctor may have decided to treat only easy cases early? Is it possible that the start of treat- ment depended on the severity of the strabismus? If he said, "If it is a hard case I won't treat it," then your conclusion would not be possible. DR. FLOM: I think Worth had a predisposition for early treatment and tried, wherever possible, to give treatment early. Those instances where treatment was not instituted early probably resulted from parental noncooperationâthat is, they did not bring the child back for treatment or moved away and came back several years later. I think that is usually the reason for delayed treatment, but there may be some confounding of this, as you indicated. DR. KAGAN: In either case, the samples of children are not evenly distributed. DR. FLOM : To justify the conclusion that emerges from Worth's results, one must presume that the time of instituting treatment was relatively independent of the severity of the amblyopia. DR. LUDLAM: I think there may be an even greater confounding factor: the identification of the age of onset. It was said back in 1896 that one could believe almost anything except the age of onset, because mothers traditionally think that children got this on their own, and not in association with the mothers. There are all kinds of emotional situations involved, so that parents 299
MERTON C. FLOM traditionally state the age of onset of amblyopia or strabismus as much later than it really is. DR.ALPERN: What is the evidence that albinos fitted with artificial pupils at an early age develop normal acuity? DR. FLOM : I did not mean to suggest that normal acuity will necessarily develop in an albino if he is fitted with an artificial pupil at an early age. I was referring to the work of Kestenbaum, which I cited, wherein the use of pinhole spectacles in an albinotic infant before 4 months of age (the age at which development of the macular area is normally completed) led to reduced nystagmus and improved acuity. Meloan has reported similar results (Amer. J. Optom. 28:435-437, 1951). Dr. Monroe Hirsch (Berkeley) and Dr. Leonard Apt (uctA)have re- cently collaborated in providing pinhole spectacles for an albino infant, but their results have not been published. Artificial-pupil contact lenses (with opaque iris and scleral sections) have been used by Edmunds (Arch. Ophthal. 42:755-767,1949) to increase acuity of older children and adults with albinism. DR. ALPERN: The histologic evidence of albinism is absence of a foveal pit, and it is surprising to me that merely fitting with an artificial pupil will allow normal vision to develop. DR. FLOM: It is true that cases have been reported in which the fovea was ill- formed or lacking, but the absence of a well-formed fovea is not invariable (S. Duke-Elder, System of Ophthalmology. Vol. Ill, Normal and Abnormal Develop- ment, pp. 314-1190, Part II, Congenital Deformities. St. Louis: C. V. Mosby Co., 1964). Kestenbaum's idea is that the use of an artificial pupil in infancy aids the development of the fovea in albinos (ClinicalMethods of Neuro-ophthalmologic Examination, New York, Grune and Stratton, 1961). Edmunds, however, has proposed that overstimulation of the fovea in albinos leads to atrophy of an otherwise normal fovea. DR. DOTY: I have the impression that Hubel and Wiesel did not get an irrecover- able amblyopia except when they used the plastic occluder on one eye. If that is the case, then there is some very important information here of relevance to the human situation. If you produce strabismus in a cat by cutting the muscle, then you do not get amblyopia. But you do get amblyopia in the same species by playing with the process as it affects the central nervous system. This would sug- gest that the amblyopia does not arise simply from noncongruence, but can also arise from some problem in the lack of visual acuity itself. Are there any human cases that are comparable to the Hubel-Wiesel situation, in which the amblyopia arises from a purely physical difficulty with the peripheral muscles, rather than a difficulty in the central control mechanisms? DR. FLOM: I am not sure that the loss of vision that occurs in kittens as a result of light deprivation is amblyopiaâit is a loss of vision. In a population of amblyopic persons, about one third will have strabismus alone, one third a substantial dif- 300
Early Experience in the Development of Visual Coordination ference in refractive error between the two eyes (anisometropia), and one third both (data to be published). That covers approximately 99% of the total sample of amblyopes. It appears that the two associated conditions in amblyopia are a deviant eye, which produces a lack of synergy in the two visual inputs, and one defocused retinal image. Each of these conditions has, of course, its counterpart in the Hubel and Wiesel studies. Defocusing of the retinal image in one eye is anal- ogous to the experimental situation of placing half a Ping-Pong ball over the eye, so that diffuse light but no image falls on the retina. Their experiment10 in which they operated on the medial rectus muscle is related to strabismus. DR. DOTY: Not necessarily, because the origin of that strabismus may be a cen- tral defect or a problem in neuromuscular control. DR. FLOM : There are types of strabismus that are purely peripheral in originâthat is, they are due to abnormalities of the muscles or ligaments. DR. DOTY: What happens in those cases? Are they equally amblyopic? DR. FLOM: I cannot say. I do not think anyone has looked at the distribution of amblyopia in two different classes of strabismus. That would be worth looking at. 301
WILLIAM A. MASON Information Processing and Experiential Deprivation: A Biologic Perspective Man's capability in the living world as a seeker and user of information is undeniably unique. Of all his attributes and skills, in fact, most people are likely to agree that maii's ability to process information most clearly sets him apart from the rest of the animal kingdom, giving par- ticular weight to language and other cognitive skills that no other creature can equal. It is a common belief that the abilities to speak, to read, to plan, and to cipher are distinctively human achievements. However, even if we grant that man has exceptional talents as a seeker and user of information, we must still concede that he is scarcely more than an impressive newcomer on the information- processing scene. The need to know about the world and to act on information received are far older than manâolder, indeed, than be- havior itself, since behavior has emerged in response to these demands. It seems most likely that man's achievements in dealing with informa- tion are built on ancient foundations that he shares with many humbler types. Accordingly, our appreciation of the special skills of the human organism as an information-processing system and our understanding of the disabilities to which this system is liable will be improved if they are considered within a broad biologic perspective. Such, at least, is the viewpoint of this presentation. 302
Information Processing and Experiential Deprivation Other portions of this volume deal with the effects of experience on particular organs and processes in animal subjects. In contrast, my in- terest here is directed toward a broader set of questions. The primary concern will be with the organism as a whole, viewed as an information- processing system. The thesis to be developed can be summarized briefly: All organisms have evolved within an environment that is stable when viewed broadly and yet on closer examination is seen to be sub- ject to continuous change. Behavior has emerged as an important means of detecting and adjusting to change. To the extent that all behavior is organized with respect to environmental regularities, contingencies, and constraints, the structure of behavior is necessarily isomorphic with the structure of the environment. In this sense, all behavior can be consid- ered as a manifestation of a representational process (schema). "In- stincts," viewed as more or less fixed sequences of action in response to particular stimuli (releasers), can be considered to constitute a special case of such representational processes or schemata. Schemata are in- herent, species-characteristic modes of organizing experience, but there has been a general trend in mammalian evolution for schemata to be- come more open, more responsive to variations in environmental input. As would be expected, openness is greatest in species that are the most accomplished and versatile users of information, and it is those species whose behavior most clearly reflects variations in individual experience. In the more advanced species, early environmental restriction will affect schemata in two ways: It will impede their complete development and elaboration by withholding "essential" classes of information to which the individual (as a representative of a particular species) is selectively "tuned," and it will lead to the development of seemingly aberrant or distorted schemata that are actually reflections of (or isomorphic with) the peculiar properties of the rearing environment. ORGANISM AND ENVIRONMENT The basic relationship we will be concerned with, of course, is the relationship between the organism and its environment, and it will be helpful to deal with this question first in rather general terms. This rela- tionship is obviously fundamentalânot only in behavioral research, but in the whole of biologyâbut does not imply that the more complex be- havioral attributes of man can be reduced to terms appropriate to a rat 303
WILLIAM A. MASON or a frog. It does imply, however, that even the most complex behavioral attributes have an evolutionary history and serve a biologic purpose. Man has to cope with the same inexorable demands as the monkey or the mouseâmaintaining proper body temperature and oxygen tension, finding enough to eat, procreating, providing for the young. These are as essential to our own survival as a species as they are to the survival of any other biologic group. What is important is the obvious fact that all of us, man and animal alike, are the products of our environments, and we bear the stamp of our origins on every aspect of our structure and function. The environment implies the kind of organism that it can sus- tain, and the organism implies its appropriate environment.8 The infor- mation that an organism can register and how it gets this information and uses it are determined by the exigencies and constraints of the en- vironment in which it has evolved. One can look upon the organism as a biologic system that exists for the "purpose" of achieving a workable fit between its vital functions and its environment and that continues to exist because it fulfills this purpose with some degree of success. That is the essential idea behind Darwinian fitness. Clearly, however, to speak of the environment in this wayâas though it were a static entityâis merely a convenient fiction. As we all know, the environment is not a "thing," but a process, and change is the rule. No environment remains the same for long, and no two environments are ever precisely alike. There are regularities, to be sure; without a large measure of stability, life would be impossible. But stability is only a matter of degree, and even in the most nearly constant of environments, some element of caprice is always potentially present. All successful or- ganisms possess some ability to cope with the reality of change, some way of altering their functions in accordance with changing circum- stances. The necessity of coping with change has given rise to countless evolutionary innovations, and among these, behavior must certainly be included as one of the most significant. Its peculiar virtue is flex- ibility. "Behavior" implies the capacity to select from the total array of environmental energies particular "relevant" segments (for example, from the electromagnetic spectrum), to encode this energy, and to use the encoded information as the basis for selecting some response. The "apparatus" of behavior thus has as its main business the evaluation of environmental contingencies and the selection and elaboration of appro- priate courses of action. An amoeba's movement toward a weak stimu- 304
Information Processing and Experiential Deprivation lus is a reaction to the "probability" that a suitable food object will be encountered.26 Likewise, a monkey's leap from one tree to another implies a statistical "faith" in the weight-bearing potential of tree limbs, the reliability of distance cues, and so on. From this point of view, all behavior points forward; it has a predictive or probabilistic compo- nent.3'4 Even the simplest-behaving organism is always of necessity playing the oddsâusing information received to initiate some program of action as the most likely to result in success. REPRESENTATIONAL PROCESSES How information is received and organized, the form in which it is stored, and how it comes to be tied in with specific acts are subject to debate. Until recently, the most heated arguments revolved around stimulus-response (S-R) versus cognitive views of learning. Stimulus- response theories grew out of reflex-arc conceptions of learning, es- pecially the work of Pavlov, whereas cognitive theories are most heavily indebted to the organismic views of Gestalt psychology and of Piaget. It is becoming clearer every day, however, that the issues that separate those positions are more often semantic and methodologic than substan- tive. The main virtues of the S-R formula are that it is simple, explicit, and easily translated into operational terms. It is the working language of the scientist at his bench. But no one could claim that the S-R for- mula in its primitive, unadorned form can handle the complexities of intellectual life in the more advanced species. And with every conces- sion to that factâconcessions expressed by postulating hypothetical processes intervening between stimulus and responseâthe S-R position and the cognitive positions move closer together. The shape of the final rapprochement, however, is not our concern. What we are seeking is a way of characterizing the information- processing activities of nonhuman forms that seems to fit the facts. And I believe that we are constrained by the evidence now available to rec- ognize that all information-processing activities occur within the frame- work of existing systems. The sequence consisting of the selection of stimuli, the encoding and storage of information, and the elaboration of specific actions is from beginning to end a highly organized process. How we refer to this processâas instinct, habit, set, expectancy, engram, cognitive map, plan, program, or schemaâis determined largely 305
WILLIAM A. MASON by the complexity of the phenomenon being considered, by our knowl- edge of its history, and by our preference for "analytic" as opposed to "synthetic" terms. It boils down to a matter of scope and personal taste. The term that will be used here is "schema." Schemata are by no means the exclusive property of more advanced animals. On the contrary, they are very widely distributed within the animal kingdom. They play a major role in the performance of biolog- ically essential tasks, such as mating, care of the young, filial responses, reactions to predators, and pursuit of prey; and they provide the basic groundplan according to which behavioral development must proceed. They are the major vehicles for maintaining an acceptable fit between behavior and ecology. The evolution of schemata has occurred within a particular array of environmental energies. They have been subject to the same kind of selection pressures as any other organismic functions; therefore, they necessarily reflect the regularities, contingencies, and constraints that the environment imposes. In this sense, they can be regarded as functional representations of the environment. The frog waiting patiently for his supper affords one example of such an organized representational process in one of the humbler vertebrate forms. The frog is equipped with his schematic fly. To be sure, the schema seems primitive by our standards, because it is such a poor rep- resentation of flies as we know them. For the frog, a fly is any small ob- ject that moves into its visual field, no matter whether it is edible, and the fly-catching program is activated by that movement. Under most circumstances, such a program accomplishes its purpose effectivelyâ flying pebbles, after all, are something of an anomaly in the frog's nat- ural environment. But one of the limitations of the program is that it is unable to recognize flies if they are not moving. This creates a problem for frog fanciers, in that the frog can starve to death surrounded by im- mobile but perfectly nutritious flies, and steps must be taken to animate its diet. The frog is not entirely without resources for modifying its prey-catching schema in the face of new information, however; if a hairy caterpillar is substituted for the fly, the snapping response is in- hibited after a few feeding attempts, at least temporarily.21'26,37 The situation does not appear to be vastly different at the outset for animals much closer to man. The newborn monkey begins life with a schematic mother, which is a far cry from monkey mothers as we know them. In the beginning, the defining properties of "mother" are almost entirely tactile, and virtually any claspable object can serve as a mother 306
Information Processing and Experiential Deprivation substitute.18 In contrast with the frog's schematic fly, however, the monkey's primitive schema undergoes a rapid and progressive enrich- ment as development proceeds. As physical attributes and their arrange- ment in time and space become associated with a single source, "mother" emerges as an entity; she acquires object status. Eventually, she is recog- nized at a distance and from various angles of regard. Her distinctive physical attributes combine with a great number of more dynamic func- tions that help to define the relationship between mother and child. The growing monkey ultimately arrives at a conception of "mother" that in subtlety and complexity must come close to our own. PH YL O GENE TIC CONTRASTS AND TR ENDS It is clear that, as species become more "intelligent," they also become less "instinctive." The contrast between the schemata of the frog and the infant monkey suggests how this relationship may have come about. Presumably, all schemata are innate to the extent that they exist in every normal individual as inherent species-typical dispositions toward organizing experience in particular ways. At the same time, however, all schemata are probably "open" in some degree. Those which are less open are susceptible to little more than minor "tuning" in response to altered environmental input, whereas the more open schemata are sub- ject to extensive modification and may even remain functionally incom- plete in the absence of appropriate input. And, of course, it is precisely the species that we consider most "intelligent" that display the greatest degree of openness. In other words, in the more advanced biologic forms, limited but highly stable systems for finding, encoding, and acting on information have been exchanged for increased behavioral plasticity. Relatively closed and invariant schemata are highly adaptive in a stable environ- ment, and this advantage has been sacrificed to achieve greater flexibil- ity in adjusting to change. "Instincts" (i.e., primordial schemata) have not become lost in evolution, but they have become progressively more open, and the result of this transformation has given rise to what we refer to as "intelligence."22'23'25'41 The advantages of this develop- ment are obvious, and so are some of the risks. Increased openness of schemata amounts to a degeneration or dissolution of the organization of instinctive patterns, and this change will be adaptive only if some 307
WILLIAM A. MASON provision is made for creating behavioral stability by other means. And it is at this point, of course, that an improved capacity to deal with in- formation becomes critical. The functional changes that accompany this improvement are fairly well agreed on, even if the underlying mechanisms are obscure, and I will describe them briefly here (for documentation, see, e.g., Harlow,15 Lashley,23 Maier and Schneirla,26 Nissen,35'36 and Warren43): Increased sensory differentiation. Within any given situation, the range of effective stimuli increases, as does the ability to respond selec- tively to different patterns or configurations of stimuli. There is in- creased effectiveness of complex perceptual relations (such as figure- ground, salience, and congruity) and of the various perceptual illusions. Presumably, the same process is also involved in some types of insight- ful problem-solving and detour behavior. Increased responsiveness to stimulus change. This is expressed in the elaboration of attentional mechanisms (orienting reflexes, scanning, ob- serving responses, visual curiosity) and in the tendency to respond to novel stimuli with a wide range of spatial adjustments resulting in ap- proach (exploration, manipulation, play, aggression) or withdrawal (timidity, fear of novelty, flight). Increased ability for sensory integration. This is expressed in im- proved capabilities for linking together information from different modalities, or from the same modality at different times, for abstract- ing, generalizing, and elaborating rules and strategies on the basis of experience. Increased differentiation of motor acts. Motor patterns become more highly differentiated and particulate (e.g., ability to move each digit independently versus opening and closing hand as a whole) and motor sequences become "looser" (e.g., ability to locomote using a variety of postures and gaits). The organism that emerges from such evolutionary developments embodies a superior information-processing system. It is better equipped to select and analyze current information from the environment, to in- tegrate it with previous information, and to elaborate a plan of action. And it has the motor refinements to translate this plan into concrete achievements. Such an organism is opportunistic and stimulus-seeking. Its behavior has become something more than a mechanism for dealing 308
Information Processing and Experiential Deprivation with imposed changes in environmental input. Behavior now has the additional role of creating variable input, which is ordered and organ- ized as it comes in. The cumulative effect of the information it receives is the development, enrichment, and elaboration of schemata. The es- sential framework for this development is present from the outset. If this view is correctâif it is true that primitive schemata provide the basic framework within which and on which experience operatesâthen the deprivation experiment can be viewed as one method for investigat- ing this framework. Withholding information early in life does not elim- inate the framework or necessarily prevent its subsequent elaboration. Schemata may continue to develop under conditions in which the impe- tus and constraints that influence normal development are changed radically. The result is not behavioral disorganization, but a form of organization that reflects the particular circumstances of the rearing environment. EFFECTS OF DEPRIVA TION The implications of this view will become clearer when we consider the effects of deprivation on behavioral development. Particular attention will be given to research in which animal subjects, chiefly monkeys and apes, are raised under conditions of general environmental restriction. It is important to recognize that we will be dealing with a form of depriva- tion that is not limited to a particular sensory modality, but with a rad- ical distortion in normal ecology. The animal is removed from its mother at birth and raised alone in a cage that permits it to see the nursery environment and other individuals or in an enclosed cage that curtails even that kind of stimulation. If we compare the rearing environment of such an animal with that of an individual growing up in the wild, the dif- ferences are enormous. Obviously, such deprivation cuts across the en- tire spectrum of normal environmental input. As expected, the behavioral consequences of such a distortion are massive and widespread. Indeed, a primary purpose of much of the work that has been done thus far was to sort out the damage. The aim has been to provide a general overview of the range of behaviors that are affected and to map out some of the more obvious relationships be- tween rearing conditions and development. The emphasis has been problem-oriented and descriptive, rather than process-oriented and 309
WILLIAM A. MASON analytic. Because of this emphasis, we have not gone very far toward a precise understanding of the sources and nature of specific psychologic disabilities. But we are gaining a clearer view of nonhuman organisms as information-processing systems and of the effects of environmental con- ditions on the development and functioning of these systems. It has become clear, for example, that early restrictions on experi- ence produce a predictable syndrome of effects whose major elements have been described for rats, guinea pigs, cats, dogs, monkeysâall mam- mals, in fact, that have been examined. The deprivation syndrome in- cludes not only deficiencies (functional "gaps" in the behavioral reper- toire of deprived animals), but aberrations, behaviors that are not seen in nondeprived animals. There is thus the suggestion of a common mam- malian response to restricted environmental input that transcends dif- ferences among species and minor variations in rearing conditions. Moreover, there is the implication that developing mammals have similar requirements for information and similar propensities for processing information; animals display not only similar deficiencies, but also similar aberrations, when information is withheld. It has also become clear as the result of recent research that variations within the broad framework of the deprivation syndrome tend to fol- low phyletic lines. An animal does not abandon its species-typical modes of processing information, even under extreme conditions. In general, one can say that the closer the phyletic affinity between two individuals, the greater the likelihood that they will resemble each other in their specific behavioral reactions to deprivation, even when the reac- tions are apparently maladaptive and are qualitatively distinct from be- haviors shown by nondeprived members of the species. Such species- typical variations within the deprivation syndrome provide strong support for the view that the selection and processing of input occurs within the framework of existing semiautonomous systems. Stereo typed Mo tor Acts The interplay between environmental input and intrinsic schemata can be illustrated by the stereotyped motor acts that develop in nonhuman primates raised apart from their mothers. The most prominent of these acts are nonnutritive sucking, self-clasping, and body rocking (for re- view see Berkson1). An infant monkey or ape raised alone in a bare cage fails to encounter normal outlets for clinging and sucking and hence comes to direct these responses toward itself. In a limited sense, 310
Information Processing and Experiential Deprivation it develops its maternal schema out of the most accessible and appro- priate material that is available, which happens to be its own body. This interpretation is plausible for thumb-sucking and self-clasping, both of which have normal counterparts in patterns that are directed toward the natural mother. The third pattern, stereotyped rocking, has no obvious parallel in the normal mother-infant relationship, but we have recently obtained presumptive evidence that it, too, can be con- sidered a "substitute" response to the deprivation of stimuli ordinarily provided by the mother. If infant rhesus monkeys are raised with sta- tionary surrogates of the sort used by Harlow, most will develop stereo- typed rocking as a habitual pattern. If they are raised with the same de- vice, except that it is motorized to move up, down, and around within the cage, stereotyped rocking does not develop, presumably because the robot surrogate simulates the kind of input that the natural mother pro- vides as she carries the infant about. In other words, I am suggesting that stereotyped rocking of the rhesus monkey is a manifestation of a primitive schema working itself out on the basis of reduced environ- mental input. The organizing effects of intrinsic schemata on motor patterns are evident not only in the early stages of postnatal life, but throughout development. Isolation-reared monkeys display self-biting and self- stimulation of the genitals, and these self-directed behaviors become progressively more frequent as the animals enter adolescence, the pe- riod in which social aggression and sexual interaction assume prominence in nondeprived monkeys.5 The most compelling evidence of the guiding role of schemata in later development, however, is provided not by ex- perientially deprived animals, but by a mother-reared monkey that was deprived of normal effector mechanisms as the result of a congenital malformation. An infant rhesus monkey with a thalidomide-induced de- formity of the forelimbs not only walked bipedally, a behavior that is infrequent in normal animals but can be seen on occasion, but used its feet to groom its motherâa pattern that to my knowledge has never been observed in an intact monkey, an "invention" in response to very special circumstances.24 Arousal Effects Another reliable consequence of early environmental restriction is a tendency toward fearfulness or heightened emotionality. The behav- ioral expressions of this effect are varied and depend on the particular 311
WILLIAM A. MASON species and the situation in which the animal is observed. Motor dis- charge may take an exaggerated form, as in seizures, tics, whirling fits, or frantic running; or it may be minimal, as in crouching, freezing, or falling into syncope or sleep. The general picture is one of behavioral disorganization or the activation of organized patterns that are inappro- priate to the situation. What is the source of these extreme emotional behaviors, and what implications do they suggest for the information-processing activities of deprived animals? The immediate source, it would seem, is any large increase in stimulation over that to which the animal is accustomed, and the range of stimulation to which the deprived animal is accus- tomed is, by definition, narrow. Seizures, tics, and freezing are most likely to occur in response to novelty or change, presumably because an isolation-reared animal is overwhelmed by the sudden increase in stimulation and has no other means of coping with it. Plausible as this explanation may seem, however, it does not go far enough. In many experiments, the test situation is also new to the con- trols, and initially they do indeed show some signs of fear. But they are not overwhelmed; their reaction is less intense than that of isolation- reared animals, and it diminishes more rapidly. A deprived animal's response is often entirely out of line with the degree of novelty or change. For example, a chimpanzee raised in an enclosed cubicle may wait many days before it makes its first hesitant contact with a small and entirely innocuous object, such as a matchbox or a block of wood. If one were to take every precaution to ensure that "novelty" were equivalent for wild-born and restricted animals, as by constructing an object from exotic materials, there is no reason to doubt that the wild- born animals would still contact it sooner than the restricted chim- panzees.33 The point is that we need know only that one animal was raised in isolation and another was not to predict how their reactions will differ toward any small inanimate object. A detailed knowledge of their individual histories is not necessary and in fact would probably add very little to the accuracy of prediction. Knowing whether or not a spe- cific object has been encountered previously is less important than knowing that there have been many such encounters in the past. It is a question not of whether an object is more novel to one animal than to the other, but of the kinds of reactions that novelty produces. A deprived organism is ill-equipped to deal with unfamiliar input, with complexity, and with change. It is deficient in ability to assimilate 312
Information Processing and Experiential Deprivation new information, an ability that a normal animal has been exercising al- most continuously from early infancy. Excessive arousal in response to novelty or change can thus be considered the result of a breakdown in the processing of information. It is caused by an overload on a system whose development has been curtailed by restrictions on early experience. A similar view has been advanced by Melzack and co-workers.30-32 They propose a neurologic model in which sensory input is conceived as a two-part process. Melzack believes that neurophysiologic data point to two classes of afferent fibers that can be distinguished on the basis of conduction rates. The fast afferent fibers are believed to activate central nervous system processes (phase sequences) subserving memory, atten- tion, and similar functions. When activated, these processes cause a feed- back to lower synaptic levels, where they can inhibit, facilitate or other- wise modify the input patterns of the more slowly conducting afferent fibers; in this fashion, they exert active control over the selection of in- formation. As the result of sensory restriction, the development of these central nervous system processes is impaired. The selection of rel- evant information and the filtering out of irrelevant information are faulty, and the loss of filtering leads to diffuse bombardment of the central nervous system, one consequence of which is excessive arousal. This state of affairs can trap a deprived animal in a vicious circle. Lack of experience engenders excessive arousal, and excessive arousal dis- rupts the activities of the very mechanisms in the central nervous system whose further development is required for orderly and efficient pro- cessing of information. To return to the terminology of earlier sections, an advanced organism that has been able to develop only a limited num- ber and variety of environmental schemata because of restrictions on early experience may be hindered by excessive arousal from overcom- ing this deficiency in later life, even when adequate environmental op- portunities are provided. Recent experience leaves no serious doubt that this is a real possibil- ity. The heightened emotional responsiveness of the experientially de- prived animal is, for all practical purposes, a persistent disability. It can be ameliorated by subsequent treatment, to be sure,11 but it can prob- ably never be entirely overcome. The electroencephalograms of isolation- reared dogs show evidence of high arousal for at least 6 months after they are released from restriction, and behavioral indications of in- creased arousal persist in monkeys and apes throughout years of ex- posure to the normal laboratory environment. 5'6'29'32'39 313
WILLIAM A. MASON Basic Cognitive Skills Certainly the strongest support for the thesis that high arousal exerts a disruptive effect on information processing is the poor performance of restricted animals on learning tasks. Isolation-reared dogs are inferior to controls on spatial-orientation tests, on delayed-response problems (in which an animal is shown food and required to locate it after an im- posed delay), and on the reversal of a previously acquired discrimina- tion response (in which an animal must select a previously unrewarded cue).11'31'42 They are also slower to inhibit a response that no longer leads to reward.31 Similar data have been reported for chimpanzees. Davenport and Rogers6 investigated delayed-response performance in animals raised in isolation for the first 2 years of life. The chimpanzees were 7-9 years old at the time of testing and thus had exposure of at least 5 years to the normal laboratory environment, including experience in group living. Although one might assume that this intervening experience would be sufficient to overcome any early cognitive deficits, the re- sults quite clearly show otherwise. At all levels of delay, the perfor- mance of the restricted chimpanzees was markedly inferior to that of wild-born animals, and the differences on the longer delays persisted throughout testing. Data on response latencies strongly suggest that attentional factors were important. At the longer intervals especially, the restricted animals were slow to respond at the end of the delay period, suggesting that they had difficulty maintaining a consistent orientation toward the relevant aspects of the experimental task. The same subjects were also compared on a discrimination task.7 The procedure followed the learning-set paradigm developed by Harlow,16 in which a series of two-object discrimination problems is presented, each problem for a fixed number of trials. As Harlow showed, the facility with which such problems are solved improves with experience, until eventually the more proficient animals are capable of near-perfect performance on the basis of a single informa- tion trial. This ability is attributed to the formation of a "discrimina- tion learning-set." The chimpanzees were trained on a total of 438 four-trial object-discrimination problems. Although both groups showed unequivocal evidence of developing learning-sets, the restricted group was inferior to the wild-born group. A significant factor in this 314
Information Processing and Experiential Deprivation inferiority was the tendency to persevere in the selection of a previously unrewarded object. Contrasting results have been obtained from similar tests performed with rhesus monkeys. Harlow and associates report no differences in delayed-response or learning-set performance between monkeys raised from birth in open-wire cages and monkeys raised as total isolates for the first 6 or 12 months of life.17'38 It should be emphasized, however, that all the monkey groups were born and bred in laboratory cages (in contrast with the chimpanzee study, which included a wild-born con- trol group), and it may be supposed that none of the rearing conditions was sufficiently "enriched" to provide for full intellectual growth. This possibility seems most likely in the light of recent findings obtained at the Delta Center by Mr. Paul Anastasiou. Two laboratory- reared groups of adolescent rhesus monkeys (raised with moving and stationary surrogates), an adolescent jungle-born group, and an adult jungle-born group were tested on 40 different tasks. The problems were designed to assess a variety of cognitive skills, ranging from rela- tively simple and straightforward visual search to solution of complex puzzle devices. The results demonstrate that both wild-born groups were unequivocally superior to either laboratory group; indeed, the best of the 14 laboratory-reared subjects was essentially no better than the worst of 13 wild-born monkeys. Fuller, on the basis of his studies of dogs, considers that "behavioral deficits following isolation result more from competing emotional re- sponses than from failure of behavioral organization during isolation or from loss of established patterns."11 Although heightened arousal prob- ably played a significant part in the performance deficits that have been considered here, it is well to keep in mind that its precise role remains to be established. Furthermore, whatever its role, it is likely that arousal level is only one of several factors that affect the performance of isolation-reared animals. For example, there is convincing evidence that some deficiencies are the result of withholding specific learning opportunities or classes of information. Specific Functional Relations It is by now firmly established that some level of stimulation is required for the full structural and functional development of the peripheral re- 315
WILLIAM A. MASON ceptors and their associated neural systems. This question is fully re- viewed elsewhere in this volume and will not be pursued here. The pres- ent concern is with an organism that may be assumed to have had a sufficient variety and amount of visual and auditory stimulation to pre- clude gross neuropathology and to have had the opportunity to form basic sensorimotor coordinations of the sort investigated by Held and co-workers.19'20 Given such an organism, we are concerned with the ex- tent to which its ability to process information is influenced by specific input features of the rearing environment. How clearly is information received (or withheld) early in life reflected in later behavioral capabilities? Some of the first evidence suggesting a rather close correspondence between early information input and later performance was obtained with rats. Animals raised in cages with triangles and circles on the walls were shown to be superior to controls in learning to discriminate be- tween these forms as adults.13 This superiority was maintained (and in some experiments even enhanced) if the test stimuli departed slightly from those to which the animal was exposed in its living cage. Transfer effects were no longer obtained, however, when the differences be- tween the familiar stimuli and the test patterns were extreme.9'10'14 It thus seems clear that "casual" exposure to specific shapes during the rearing period will not only facilitate recognition of these patterns when they are later encountered in a discrimination-learning test, but will also lead to the establishment of some form of generalization "gradient." It becomes easier to recognize new stimuli that merely resemble the familiar pattern. The range of stimuli that are responded to as equivalent might be expected to depend on early experience. This question has recently been reviewed by Ganz.12 Ganz distinguishes between the neural or- ganization of information and the control that information exerts over behavior. On the basis of his own careful studies of hue generalization in light-deprived monkeys and the work of other investigators, he con- cludes that some organization is innately imposed on stimulus input. The scaling of sensory dimensions occurs before any stimulus experi- ence. This is inferred from the finding that an animal that has been ex- posed to only one hue value will respond to it on stimulus-generaliza- tion tests more than to other hue values. But the generalization gradient is "flat"; the responses to the familiar hue and to the test stimuli are 316
Information Processing and Experiential Deprivation similar. This is viewed as evidence that the dimension as a whole has not developed effective control over behavior. How is such control acquired? Ganz argues that control is achieved through a history of differential reinforcement within the appropriate stimulus dimension. Thus, an animal must experience reward in the presence of one value of the hue dimension and the absence of reward in the presence of a different value of the same dimension. As the result of such experience, the generalization gradient becomes steeper; the ani- mal differentiates more sharply between the familiar stimulus and the test stimuli. Differential reinforcement does not merely strengthen the response to one value and weaken the response to another, however; it also increases the relevance or salience of the entire stimulus dimen- sion. A further long-term result of experience is that different cues be- come functionally related or the same cue becomes effective across dif- ferent conditions (for example, by being presented now to one eye, now to the other, or by being viewed from different angles of regard). Even- tually, this leads to the development of functional equivalence within and across cues, expressed in equivalent responses ("recognition"), in spite of changes in conditions of presentation and the specific receptor surfaces that are stimulated. Ganz's conceptual approach developed out of research on animals raised under conditions of highly restricted visual input, which, strictly speaking, are outside the population that we are most concerned with here. Nevertheless, I have discussed his position in some detail because it very clearly illustrates the complexities that are encountered in any systematic effort to tease out the subtle interplay between experience and innate organization in the development of schemata by advanced organisms. As far as functional outcomes are concerned, the interpene- tration of "nature" and "nurture" is, for all practical purposes, com- plete. Learning plays an essential part in development. But most learn- ing occurs within the framework of existing schemata. It receives guidance from these structures and is a primary agent in their trans- formation from phylogenetic "plans" into adaptive functional systems. According to this view, one might speculate that the learning process evolved as handmaiden of the instincts and that, even in the most advanced organisms (by which we mean ourselves, of course), it has not yet achieved full emancipation from this ancient position of subservience. 317
WILLIAM A. MASON Much of the evidence that has led me to this view has come out of deprivation experiments with nonhuman primates. Some of the deficits point rather clearly to inadequate opportunities for specific forms of learning in the rearing environment. Such deficits fall into two broad classes: defective integration of sensorimotor patterns and deficiencies in signal functions. Examples of both types of deficit can be found in the social behavior of deprived animals. The sexual performance of socially deprived male rhesus monkeys suggests a failure in sensorimotor integration.28 Most of the components of the complete actâpenile erection, clasping with the hands, clasping with the feet, thrustingâare present, but they are so poorly integrated that intromission is all but impossible. Clearly, the rearing environment has failed to provide the opportunity to form these components into an effective pattern. Accordingly, given a suitable environmental structure for rehearsal and appropriate instigation (and both conditions might be met with a suitably designed inanimate model), one would anticipate that the laboratory-reared animal would show a gradual progression toward the smoothly coordinated pattern seen in normally socialized adult males. Inasmuch as the normal pattern is not present in deprived males, it seems reasonable to infer that learning is a factor in its develop- ment. Insofar as the components of the normal pattern are present even in the socially deprived male, tend to occur together, and, under appro- priate circumstances, eventually become fused into an integrated species-typical pattern, the presence of some guiding structure seems to be impliedâa "template" that must be standard equipment for the rhesus macaque but remains functionally incomplete unless appropri- ate input is provided. Deficiencies in signal functions can be inferred from the observation that isolation-reared monkeys do not show consistent and appropriate responses to the distinctive postures, gestures, facial expressions, and sounds that serve as the vehicles of communication in natural primate societies.27'34 Again, however, such learning seems to operate within the context of pre-existing structures. Some configurations of stimuli and some sequences of stimulation possess primitive functions that apparently precede specific experience. These stimulus patterns are similar to the innate releasing mechanisms (IRM'S) described by the ethologists, but there is no fixed relationship between stimulus and response as implied by the IRM concept. For example, young labor- atory-reared rhesus monkeys display stereotyped affective-social re- 318
Information Processing and Experiential Deprivation sponses toward inanimate objects that they have never before encoun- tered. The frequency of these responses increases as the objects become more animal-like, or more representational. This effect was obtained in infants as young as 1 month, housed individually from birth in a stan- dard nursery environment.2 The response to the optical stimulus of "looming" is another exam- ple of a primitive schema. Looming, created by a sudden increase in the size of circular stimulus, causes persistent fear responses in laboratory- reared infant monkeys (as well as adults).40 The rapid approach of a solid body (which was simulated by the experimental procedure) is an important ecologic sign for the free-ranging monkey, typically associ- ated with a potential hazard, such as a predator or an aggressive member of one's own group. At the same time, however, it is easy to see how the adaptiveness of the looming response would be improved if other input features were also taken into account. Looming can mean the approach of a hungry lion, a docile bullock, or an adult male coming to the rescue. An animal that can distinguish these possibilities and act accordingly clearly appears to be in a better position to survive than an animal that cannot. It is in this process of differentiation and evalua- tion of environmental contingencies that learning can be seen to play one of its most important parts. REFERENCES 1. Berkson, G. Abnormal stereotyped motor acts, pp. 76-94. In J. Zubin and H. F. Hunt, Eds. Comparative PsychopathologyâAnimal and Human. New York: Grune and Stratton, 1967. 350 pp. 2. Bernstein, S., and W. A. Mason. The effects of age and stimulus conditions on the emotional responses of rhesus monkeys: responses to complex stimuli. J. Genet. Psychol. 101:279-298, 1962. 3. Bjorkman, M. Predictive behavior. Some aspects based on an ecological orien- tation. Scand. J. Psychol. 7:43-57, 1966. 4. Brunswik, E. Representative design and probabilistic theory in a functional psychology. Psychol. Rev. 62:193-217, 1955. 5. Cross, H. A., and H. F. Harlow. Prolonged and progressive effects of partial isolation on the behavior of Macaque monkeys. J. Exp. Res. Personality 1:39-49,1965. 6. Davenport, R. K., and C. M. Rogers. Intellectual performance of differentially reared chimpanzees: I. Delayed response. Amer. J. Ment. Defic. 72:674-680, 1968. 7. Davenport, R. K., C. M. Rogers, and E. W. Menzel. Intellectual performance of differentially reared chimpanzees: II. Discrimination-learning set. Amer. J. Ment. Defic. 73:963-969, 1969. 319
WILLIAM A. MASON 8. Foerster, H. von. From stimulus to symbol: the economy of biological compu- tation, pp. 170-181. In W. Buckley, Ed. Modern Systems Research for the Behavioral Scientist. Chicago: Aldine Publishing Co., 1968. 525 pp. 9. Forgus, R. H. The effect of different kinds of form pre-exposure on form discrimination learning. J. Comp. Physiol. Psychol. 51:75-78, 1958. 10. Forgus, R. H. The interaction between form pre-exposure and test require- ments in determining form discrimination. J. Comp. Physiol. Psychol. 51:588-591, 1958. 11. Fuller, J. L. Experiential deprivation and later behavior. Science 158:1645- 1652,1967. 12. Ganz, L. An analysis of generalization behavior in the stimulus-deprived organism, pp. 365-411. In G. Newton and S. Levine, Eds. Early Experience and Behavior. Springfield, 111.: Charles C Thomas, 1968. 785 pp. 13. Gibson, E. J., and R. D. Walk. The effect of prolonged exposure to visually presented patterns on learning to discriminate them. J. Comp. Physiol. Psychol. 49:239-242, 1956. 14. Gibson, E. J., R. D. Walk, H. L. Pick, Jr., and T. J. Tighe. The effect of pro- longed exposure to visual patterns on learning to discriminate similar and different patterns. J. Comp. Physiol. Psychol. 51:584-587, 1958. 15. Harlow, H. F. The evolution of learning, pp. 269-290. In A. Roe and G. G. Simpson, Eds. Behavior and Evolution. New Haven, Conn.: Yale University Press, 1958. 557pp. 16. Harlow, H. F. The formation of learning sets. Psychol. Rev. 56:51-65, 1949. 17. Harlow, H. F., and G. Griffin. Induced mental and social deficits in rhesus monkeys, pp. 87-106. In S. F. Osier and R. E. Cooke, Eds. The Biosocial Basis of Mental Retardation. Baltimore: Johns Hopkins Press, 1965. 151 pp. 18. Harlow, H. F., and R. R. Zimmermann. The development of affectional responses in infant monkeys. Proc. Amer. Phil. Soc. 102:501-509, 1958. 19. Held, R., and J. A. Bauer, Jr. Visually guided reaching in infant monkeys after restricted rearing. Science 155:718-720, 1967. 20. Held, R., and A. Hein. Movement-produced stimulation in the development of visually guided behavior. J. Comp. Physiol. Psychol. 56:872-876, 1963. 21. Kaess, W., and F. Kaess. Perception of apparent motion in the common toad. Science 132:953, 1960. 22. Kuttner, R. An hypothesis on the evolution of intelligence. Psychol. Rep. 6:283-289, 1960. 23. Lashley, K. S. Persistent problems in the evolution of mind. Quart. Rev. Biol. 24:28-42, 1949. 24. Lindburg, D. G. Behavior of infant rhesus monkeys with thalidomide-induced malformations: a pilot study. Psychonomic Sci. 15:55-56, 1969. 25. Lorenz, K. Z., Ed. Evolution and Modification of Behavior. Chicago: University Press, 1965. 121 pp. 26. Maier, N. R. F., and T. C. Schneirla. Principles of Animal Psychology. New York: McGraw-Hill, 1935. 529 pp. 27. Mason, W. A. The effects of social restriction on the behavior of rhesus monkeys: III. Dominance tests. J. Comp. Physiol. Psychol. 54:694-699, 1961. 28. Mason, W. A. The effects of social restriction on the behavior of rhesus monkeys' I. Free social behavior. J. Comp. Physiol. Psychol. 53:582-589, 1960. 29. Mason, W. A., and R. R. Sponholz. Behavior of rhesus monkeys raised in isola- tion. J. Psychiat. Res. 1:299-306, 1963. 320
Information Processing and Experiential Deprivation 30. Melzack, R. Early experience: a neuropsychological approach to heredity- environment interactions, pp. 65-82. In G. Newton and S. Levine, Eds. Early Experience and Behavior. Springfield, 111.: Charles C Thomas, 1968. 785 pp. 31. Melzack, R. Effects of early experience on behavior: experimental and con- ceptual considerations, pp. 271-299. In P. H. Hoch and J. Zubin, Eds. Psychopathology of Perception. New York: Grune and Stratton, 1965. 336 pp. 32. Melzack, R., and S. K. Burns. Neurophysiological effects of early sensory restriction. Exp. Neurol. 13:163-175, 1965. 33. Menzel, E. W., Jr. Patterns of responsiveness in chimpanzees reared through infancy under conditions of environmental restriction. Psychol. Forsch. 27:337-365, 1964. 34. Miller, R. E., W. F. Caul, and I. A. Mirsky. The communication of affects between feral and socially isolated monkeys. J. Personality Soc. Psychol. 7:231-239,1967. 35. Nissen, H. W. Axes of behavioral comparison, pp. 183-205. In A. Roe and G. G. Simpson, Eds. Behavior and Evolution. New Haven, Conn.: Yale University Press, 1958. 557 pp. 36. Nissen, H. W. Phylogenetic comparison, pp. 347-386. In S. S. Stevens, Ed. Handbook of Experimental Psychology. New York: John Wiley & Sons, 1951. 1436 pp. 37. Noble, G. K. The Biology of the Amphibia. New York: McGraw-Hill, 1931. 577 pp. 38. Rowland, G. L. The effects of total isolation upon learning and social behavior in rhesus monkeys. Unpublished doctoral dissertation, University of Wisconsin, 1964. 39. Sackett, G. P. Some persistent effects of different rearing conditions on pre- adult social behavior of monkeys. J. Comp. Physiol. Psychol. 64:363-365, 1967. 40. Schiff, W., J. A. Caviness, and J. J. Gibson. Persistent fear responses in rhesus monkeys to the optical stimulus of "looming." Science 136:982-983, 1962. 41. Stenhouse, D. A general theory for the evolution of intelligent behavior. Nature 208:815, 1965. 42. Thompson, W. R., and W. Heron. The effects of restricting early experience on the problem-solving capacity of dogs. Canad. J. Psychol. 8:17-31, 1954. 43. Warren, J. M. The comparative psychology of learning. Ann. Rev. Psychol. 16:95-118, 1965. (214 ref.) DISCUSSION DR.ALPERN: Asl recall from Harlow's data on rhesus monkeys, there is no evi- dence of any learning deficits in isolation-reared animals. But you do find it with the chimpanzee? 321
WILLIAM A. MASON DR. MASON: Yes. There is no evidence of such an effect in the data on monkeys at the University of Wisconsin. I think Harlow points out, however, that they took great pains to ensure that the animals were adapted to the test situation before formal testing was started. He also indicated that the isolation-reared animals were slower to adjust to the testing procedure and that not all the animals were able to adapt. This means that considerable training and some selection occurred before formal testing began. The animals that completed the preliminary train- ing showed apparently normal intellectual performance. DR. INGRAM: I suggest that the data on Harlow's rhesus monkeys are rather dubi- ous in any event. Broadbent has recently criticized the methodology of the Har- low experiments, and has suggested that many of the conclusions about cogni- tive development are unjustified. Broadbent in particular feels very strongly that cognitive development is impaired, but this has not been shown yet. DR. MASON: I certainly would expect to find a variety of intellectual deficits in deprived monkeys or apes. What I am saying is that, if I did not expect it, then I would be accepting a kind of a faulty psychology, in which descriptively sep- arate endpoints or activities are viewed as representing separate mechanisms, like so many different watertight compartments, with the animal developing intel- lectual abilities here, and social behavior there, and emotional behavior some- where else. I do not believe that psychologic development proceeds in that fashion. Actually, learning, broadly defined, enters into all aspects of behavioral development, just as unlearned patterns do. Normal social development requires learning. It is one aspect of intellectual development, and all the adaptive tasks that an animal must perform provide the occasion and the necessity for the development of "intellectual" skills. DR. GAARDER: I wonder if in some ways your position on dyslexia would tend to support the idea that dyslexia could be, not so much a derivative of many of the things that we have discussed, but rather the result of a somewhat higher process that we do not yet have adequate tools to study. This higher process has to do with the sequencing of a chain of interactions between an organism and its en- vironment, both the nonliving parts of the environment and the human beings that are in the environment. DR. MASON: I think a large element has been left out of our thinking about behavioral development: the ecologic element. Psychologists (ethologists, too, for that matter) lack a precise language for dealing with the microecology. Ultimately, we will have to treat the environment, not as a vast aggregate of stimuli, but in terms of its structure, because it is this structure that is reflected in the evolution of behavior. All deprivation studies deal with the impact of some drastic environmental events on ongoing, adaptive, and highly organized biologic systems. We need to develop as much concern and skill for dealing with those events and the systems that they act on as we have for dealing with behav- ioral outcomes. The tendency at the moment is to select the outcomeâthe 322
Information Processing and Experiential Deprivation performanceâand try to infer the antecedents from that. In the long run, we must do better. We may have to start with some very messy antecedent condi- tions and try to get some idea of their specific relationship to the performance deficits that are produced. Eventually, we may develop sufficient sophistication to deal with the kind of higher-order process referred to by Dr. Gaarder. And the preparation of my paper gave me the opportunity to take a small step in that di- rection. I do not think we will ever get very close to dyslexia by using monkeys, but I do think we may get some general notion from them as to how such higher- order information-processing systems function and what they require from the environment in order to develop effectively. 323
JEROME KA G AN Continuity in Cognitive Development During the First Year of Life Each generation of psychologists seems to discover a fresh set of phenomena and a sparkling new object of study. The introspectively trained adult was the pet of the academic before and around the begin- ning of this century, and the contents of consciousness the favored theme. The exploring albino rat captured the stage from the brooding adult when the academy decided that public responses and easily in- duced drives were more critical than feelings, sensations, or thoughts. One of the stars of this decade is the human infant, and the theme turns once again to those mental processes called "cognitive structure." The ease with which loyalties to subjects or subject matters are broken is to be expected in a discipline as young as psychology. The behavioral sciences are in the important but necessarily frustrating pe- riod of defining the phenomena they wish to pursue in depth and the concepts they must quantify and place in propositional form. The be- havior of psychologists during the last three quarters of a century is not unlike that of a person who has a blanket thrown over him as he stands in a large room containing a variety of interesting objects. His task is to decide, without removing the opaque cover, what objects are in the room. It would be proper strategy to walk until he hits something, put 324
Continuity in Cognitive Development During the First Year of Life out a hand, and explore the object to see whether a quick determination is possible. If a few minutes of exploration proves fruitless, no worry; there are many other objects in the room that can be explored. Our temporarily blinded subject would pass from object to object, exploring quickly, hoping for a clue that might define one object permanently but always prepared to pass on if an initial probe does not prove rewarding. This horizontal progress is a fair description of psychology's first hundred years. We probe the subjectâan idea or phenomenon until we become frustrated conceptually or methodologically; then we look for another love object. We turned from introspection because we were un- able to solve the subjectivity problem. We grew bored with rats and mazes because we could not solve the reinforcement problem and were unable to explain human thought by turn sequences in alleys. We aban- doned personality study because we failed to develop sensitive proce- dures to measure motives, anxieties, or conflicts. And now we affirm the child. THE CONCERN WITH CHILDHOOD AND INFANCY Interest in the young child, especially the infant, draws its force from divergent sources. The commitment to historical explanation, which is basic to American psychology, has always been a guide to domestic empiricism. A large proportion of studies on rats, cats, mice, and mon- keys have posed developmental questions. Harlow and his colleagues18'19 asked about the later effects of infant experience with terry-cloth mothers. Denenberg7 and Levine28 inquired into the effect of mild handling of week-old mice on adult behavior in an open field. The historical bias, which is not shared by all psychology groups, takes its strength from a strong belief in the persistence of well-learned habits (an heir of Hullian behaviorism) and an optimism that affirms that what was done can be either undone in the same organism or done differently in the next generation. The influence of psychoanalytic theory, although waning rapidly, also directed our interest to the child. As the major theory of personality de- velopment, it made strong statements about the long-term effects of family experience during the first 5 years. Finally, the unprecedented excitement about educational progress in the lower-class child, an excitement that has always been in the back- 325
JEROME KAGAN ground, has suddenly come into focus and produced a heady dose of investigations into the mental processes of young children. There is agreement that one can preview the awesome class differences in school-age children as early as the age of 3, but we do not know the temporal patterning of events during the prenatal months and the first 36 postnatal months that lead to this variation. Although scholarly concern with childhood has a broad foundation, the more limited interest in the infant required a special catalyst, and it was provided by Robert Fantz's demonstration that one could tell with remarkably simple methods what a baby was looking at and perhaps what he preferred to look at.9'10 To everyone's surprise, the very young infant was not perceptually innocent. He stared longer at checkerboards and bull's-eyes than at homogeneous gray patterns9 and more at moving lights than at stationary ones.17 As early as the age of 4 months, he looked longer at a picture of a human face than at nonsense designs.31'32 Moreover, the infant always seems to be seeking variety. PERCEPTUAL LEARNING Does the infant learn anything when he looks at an object, and if he does, what does he learn? These simple questions have generated con- siderable controversy because of an unstated faith that the infant was cognitively different from the adult. If an adult were to scan a photo- graph of Lake Atitlan for the first time he would learn and retain the fact that it has a very irregular perimeter. The adult would not have to feel the photo, swim in the lake, or draw its outline on paper. At a more conceptual level, if an adult is told that an operon is a concept used by molecular geneticists to explain suppression of structural genes, he has only to listen to this statement to learn the meaning of this con- cept. At a behavioral level, Bandura and Menlove2 have shown that a 3- year-old child who is afraid of dogs will approach and play with them after watching a short series of films showing children playing with dogs. These examples are sufficient to warrant faith in the common- sense proposition that an older child or adult can learn a perceptual structure, a conceptual unit, or changes in the hierarchy of overt be- haviors by merely looking or listening. But is this true of infants? Investigators of mental growth in the human infant before the 1960's 326
Continuity in Cognitive Development During the First Year of Life were prejudiced against this view. Tests constructed to assess infant in- telligence emphasized motor accomplishmentsâage of sitting, walking, hand-eye coordination, removal of a napkin from a toy, prehension of a button. The implicit assumption was that the child's level of intellec- tual development covaried with his precocity in sensorimotor acquisi- tions. This reasonable belief was supported by the major cognitive theorists of this century. Jean Piaget36 stated explicitly that the in- fant's first mental structures (schemata) are "interiorizations of overt actions," a view resembling Watson's belief that thought was derived from subvocal speech. Gesell, Piaget, and Watson probably arrived at their faith independently; none was especially devoted to the others' writing during the first half of the century, and there has been minimal contact among the loyal disciples of each of the three leaders. Such remarkable consistency is often a reliable sign of validity. The reasons for this commitment to the value of actions in mental growth are probably different. The early behaviorists were in a meth- odologic rebellion, and it was important for its vitality to forbid ideo- logic deviance. No concepts were permissible that could not be measured publicly. The intelligence testers were probably moved by pragmatic considerations and Gesell's clearly described procedures.15 There was no way to assess what the infant was thinking, but there were ways of mea- suring what he was doing, and the American attraction to reasonable solutions makes it easy to understand why they focused on overt be- haviors. It is difficult to understand Piaget's affinity for this materialistic view of infant intelligence. Piagetian theory is profoundly cognitive, and his epistemologic position would lead one to expect a more mentalistic flavor to infant psychology.35'36 Perhaps his autobiography will solve that puzzle. Existing empiric data suggest that an infant may acquire a mental representation of an event by only looking or listening. An infant who has habituated to a repeated presentation of the same, initially novel, stimulus shows a dramatic change in fixation time when presented with a transformation of the stimulus. This phenomenon can be demon- strated in studies using short-term habituation (McCall and Kagan, un- published data) or exposure of an infant to a stimulus over a period of several weeks.31'32 These data imply that the infant, as well as the older child, establishes cognitive structures as a result of exposure to an event. 327
JEROME KAGAN CONTINUITY IN MENTAL DEVELOPMENT The mechanisms of establishment of cognitive structures are separate from the issue of continuity in level of cognitive development. Do in- fants who at some time show precocious acquisition of a class of struc- tures remain precocious? That is the question the early infant testers wished to answer. Their efforts were ambiguous, because they evaluated behavioral accomplishments in the infant and cognitive accomplish- ments in the older child. Although demonstration of stability or con- tinuity is one prerequisite for a theory of intellectual development, stability is necessarily ambiguous as to cause. Suppose, as our data and those of others indicate, that there is a high correlation between reliable indexes of vocalization in 4-month-old girls and verbal ability at the age of 2 years. One might view this correlation as proof of a constitutional basis of intelligence. The argument would state formally that the same central intellective process caused the frequent vocalizing at 4 months and the high verbal score at 2 years. This phenomenon might be called "endogenous continuity" (i.e., a particular internal process remains stable over time). However, it is possible that an environmental force, such as a mother's reciprocal interaction with her child, produced both the frequent vocalization in infancy and a rich vocabulary and strong motivation for cognitive mastery in the older child. The latter forces produced the high verbal score. There is no necessary causal relation- ship between the vocalization at 4 months and the later verbal profi- ciency. It is possible that each is an independent correlate of different sets of maternal behaviors and that the stability was in the mother's behavior. This phenomenon might be called "exogenous continuity." Value of Continuity Studies The issue of continuity is relevant to three themes in developmental psychology. First, knowledge of responses that show stability facilitates prediction of future behaviors and early diagnosis of psychologic syn- dromes that are harmful and should be treated, as well as of socially valued syndromes that should be protected and encouraged. Second, continuity studies contribute to the validation of major theoretical positions. Knowledge of continuity in behavior allows tests of, for example, whether frustration of oral needs in infancy leads to 328
Continuity in Cognitive Development During the First Year of Life depression in adulthood and whether partial reinforcement of aggression in childhood produces greater resistance to extinction of aggressive be- havior during later childhood. Validation of these theoretical predic- tions, which requires studies of behavior over time, is necessary to evaluate the power of the theoretical schemes. The third rationale for continuity studies is the one that will be discussed here. Studies of continuity facilitate the understanding of the meaning of responses at particular ages. Contemporary psychology does not understand the meaning of some of its simplest phenomena, such as the smile at 4 months, separation anxiety at 10 months, attempts at task mastery at 1 year, and the early attainment of what Piaget calls the "object concept." It is not clear what covert processes each of these public phenomena reflects. Study of the predictive consequences of selected responses may clarify the significance of the act. Indexes of Mental Development A central methodologic problem common to many investigations of mental development is lack of agreement on the behavior patterns that provide a faithful picture of the infant's mental development. The classic conceptual stance assumed that selected motor and sensorimotor responses mirrored the level or rate of cognitive development. These motor patterns are disrupted by brain-stem damage, and anomalies in their development are used as diagnostic signs of lack of integrity of the central nervous system. But the rate of development of these complex motor responses may not be the most sensitive index of the infant's cognitive development ("cognitive" refers to perceptual structures, language, and problem-solving skills). Among infants with no central nervous system damage, there appears to be a minimal relation be- tween age of appearance or quality of these sensorimotor patterns during the first year and standard indexes of language and problem- solving ability during the preschool years.3'8 However, responses that index differential attentiveness to selected events may reflect differential acquisition of cognitive structures related to those events, and they may provide evidence of intra-individual con- tinuity of cognitive dimensions from infancy to early childhood. Dura- tion of orientation toward a visual stimulus ("fixation time") and vocal- ization during stimulus presentation are two reasonable indexes of an infant's degree of attention to an external event. 329
JEROME KAGAN FIXATION TIME The intuitively obvious index of visual attention is the duration of study of an event. Like most acts, fixation time has multiple determi- nants, and the power of each seems to change with age. Physical con- trast, movement, discrepancy from an acquired schema, and density of hypotheses activated to explain a discrepancy all exert some control on fixation time during the first 30 months of life. Movement and Contour Ontogenetically, the first determinant of orientation toward a visual event is a high rate of change in the stimulus itself, typically produced by movement or contour contrast. The infant has an unlearned disposi- tion to attend to events that possess a high rate of change in their phys- ical characteristics. Newborn infants are dramatically more attentive to moving lights than to static ones and to designs with a high degree of black-white contour than to events with minimal contour con- trast.11'17'38 These induced generalizations from molar behavior concur with neurophysiologic studies of ganglion potentials in vertebrate ret- inas. Some ganglion cells respond to onset of illumination, others to offset, and still others to both. A stimulus moving across a visual field stimulates sets of cells for a short period, creating onset and offset dis- charge patterns. Contour edges function as onset stimuli, whereas solid patterns do not; and the change in stimulation created by a sharp con- tour edge or movement elicits specialized firing patterns that can serve as the basis for sustained fixation.25.26 Discrepancy The initial disposition to maintain long fixations on visual stimuli with movement and contour eventually competes with a second, acquired determinant: the degree of discrepancy between an event and the child's acquired schema for that event. Stimuli that deviate moderately from an existing schema release longer fixations than either completely familiar events or completely novel events. Empiric support for the role of schema discrepancy on attention is suggestive, rather than definitive. At 4 months of age, achromatic illustrations of male faces elicit fixation times twice as long as those elicited by random shapes of varying num- 330
Continuity in Cognitive Development During the First Year of Life bers of turns that contain extreme novelty and greater contour contrast than the faces.31'32 Haaf and Bell16 have shown that 3-month-olds study a regular schematic face longer than one that has the same facial components disarranged. Moreover, the infants study partly disarranged schematic faces longer than completely disarranged ones. The stimuli that were similar to faces elicited longer fixations than equally complex stimuli with minimal resemblance to faces. Final support for the dis- crepancy hypothesis comes from a study in which 3-month-old infants were exposed to a novel three-dimensional stimulus at home for a month and then shown the same stimulus and three transformations of it at 4 months of age. Control children viewed all four stimuli for the first time at 4 months. The experimental infants showed shorter fixation times to all four stimuli than the controls. The discrepancy effect emerged for girls, who showed the longest fixation times to the transformations of the standard they viewed at home.31'32 Although the definitive study demonstrating a curvilinear relationship between fixation time and discrepancy from an acquired schema is still to be done, existing data are persuasive of the usefulness of this idea. At the least, they suggest that the degree of familiarity, independent of move- ment and contour qualities, does exert a powerful influence on fixation time. Density of Associations A third determinant of fixation time begins its growth during the sec- ond half of the first year and involves the density or richness of the symbolic associations activated by the child to assimilate experience. The richer the nest of hypotheses available and activated, the longer the fixation. High rate of change, discrepancy, and activation of hypotheses may be additive in their total effect on fixation time. The combined effect of discrepancy and hypotheses is illustrated in a comparative study of 1-, 2-, and 3-year-old children from two cultures. Finley12 showed cul- turally appropriate illustrations of male faces and forms to Cambridge, Massachusetts, middle-class children and Mayan peasant children from Yucatan. Fixation time increased linearly with age, for both groups, as predicted from the principle of increased density of hypotheses. But the largest increases in fixation time occurred in response to a dis- arranged face or form, which is a discrepancy stimulus, rather than to 331
JEROME KAGAN the regular face or form. The hypothesis vector complemented the dis- crepancy vector to produce a significant age-stimulus effect on fixation time.12 The changing pattern of influences on fixation time should result in different patterns of continuity over time, and, as we shall see, that expectation has been verified. VOCALIZATION The determinants of infant vocalization are much less clear. There is a dramatic increase in positive vocalization at about 10-12 weeks of age, accompanied by an equally salient decrease in fretting and irritability.27 Infants often babble when they study an interesting visual stimulus or hear selected auditory events, and it is reasonable to suggest that these vocalizations reflect the excitement that accompanies focused attention. Thus, one early determinant of vocalization may be similar to the dis- crepancy principle that states that partially novel events engage the in- fant's attention and elicit vocalization. Around 1 year of age, it appears that the hypothesis vector may also control vocalization: Finley found linear increases in vocalization in response to human faces and forms across the period from 1 to 3 years of age in both the American and the Mayan children. This report is concerned primarily with the intra-individual stability of fixation time and vocalization in response to representations of human faces during the first year of life. It is believed that the pattern of this stability provides information on continuity of acquired cogni- tive structures associated with human faces. A subordinate theme is the possibility that vocalization during the early months has different meanings for the two sexes. Vocalization may display a sexual dimorphism in the human infant. There has been a remarkable change in our attitude toward the etiology of human psychologic sex differences, in that we now acknowledge that these differences may not all be environmentally determined. Infrahuman studies have demonstrated sex differences in dominance, as well as pre- potent reactions to stress or threat.6'13'19 Recent reviews of the com- parative literature by Hinde20 and Marler and Hamilton30 indicate strong support for sex differences in a variety of response dimensions in mammalian species. It is reasonable, therefore, to expect sex differ- 332
Continuity in Cognitive Development During the First Year of Life ences in initial response hierarchies that would lead infant boys and girls to react differently to the same stimuli or environmental intrusions. PROCEDURES Our total sample consisted initially of 91 boys and 89 girls; all were Caucasian, firstborn, and living with their natural mothers within a 30- mile radius of Cambridge, Massachusetts. The range of educational level of the parents of these children ran from 8 years through a graduate de- gree. Most of the parents of the sample children were volunteers re- cruited by advertisements in local newspapers. Each infant came to the laboratory with its mother when the infant was 4, 8, and 13 months old for a series of assessments. Each 4-month-old infant was placed supine in a crib. When the infant was quiet and content, but alert, he was shown a series of 16 achromatic slides of four different human faces (Figure 1); there were four presen- tations of each of the four faces. After a short recess, the child was shown a series of slides of four different three-dimensional clay faces (41/2 X 6Vi in.) painted flesh color (Figure 2); there were four presenta- tions of each of the four clay faces. For both episodes, the order of presentation was such that each of the four faces appeared once every four slides, and there were two different orders of presentation. Each stimulus was shown for 30 sec, with 15-sec intervals between exposures, during which the visual field was homogeneously white. The stimuli were presented 20 in. from the plane of the child's face and occupied a visual angle of about 20 deg. Two of the variables coded during these two episodes were length of each fixation on the stimulus and duration of vocalization during stim- ulus presentation. These variables were coded by two independent ob- servers watching from opposite sides of the crib, neither of whom could see the stimulus being presented to the child. Interobserver reliabilities were 0.97 for fixation time and 0.71 for duration of vocalization. When the infant returned to the laboratory at 8 months of age, he was seated in a high chair behind a gray enclosure with his mother seated to his right and slightly behind him. A screen was 2 ft in front of the child at his eye level. The first episode was the same set of four achromatic faces that had been presented at 4 months. However, each 333
JEROME KAGAN FIGURE 1 Achromatic faces shown to infants. 334
Continuity in Cognitive Development During the First Year of Life FIGURE 2 Clay faces shown to infants. 335
JEROME KAGAN 3 m 10 c o d d â¢^ '^ 5 S 0 >. m vo O . â o d d H U. CO V) â¢* 00 OJ 3 (S Tt 'a B 0 d d â ts ^« tO «« ^O O ^ .S .2 -H O M o d d U â â CQ V) â¢C .0 â¢a 'â¢" c o b d d M 1 s >, -H TJ, (S (N S H £ I d d â a a (U ⢠-3 3 (N (N oo â¢s E c o b d d c o ll "o i B M o *o d d C/S 00 ~ U- U, I â¢S s o -H 10 rr ro -H \rt s JS -HO -H 0 O O § b do do o d o 31 M §-H V) OO v£) fN O -H -H O O Tj- O .2 I do do o d E â 1 O â¢* UO W3 (N I £ 3 O fS âO -JO *M u C o T T T i M o I H ll (A 04 m \o r^ ^" O I -HO (N (N O O B « 0 â â do do o d .§ 3 f*"i O m fN CO ^ rt -H -H -H O O U. c o do do do O t^ r* vo m *x) T3, ju â¢^ *-t *J >, 0 O -H -H O O CV3 Z o .2 o do d d do ^ ⢠H Pu CO vt c 1 Q a O m o> -H 10 O O O (N m o O 0 '3 u s | ⢠m c o b do do do .£: E U c o o ll â¢^ fS r- rf) vo (N u o i â¢o £ u -HO â < fS O -H o do do do 'f * â - CO 1 .2 I .2 1 .2 1 £J o 1 ill 11 â!! 5 S u â¢i â 1 > â¢S §£^'>,i2f2'^'tl£(2 EgSs'S^MM'SS r. .. w c 0 0 09 i |< 3 II 6 o VV < â¢c a0* H 00 â< 336
Continuity in Cognitive Development During the First Year of Life stimulus was presented for only 15 sec, with 15-sec intervals between exposures. The second episode was a tape recording of four different 20-sec recitations read by a male voice, with 10-sec silent periods be- tween recitations. Two of the recitations contained the same set of meaningful sentences, one set read with high inflection and the other with no inflection. The other two recitations contained the same set of meaningless sentences (nonsense words), one read with high inflection and the other with no inflection. Each stimulus was presented three times in a fixed order that was the same for all the children. The third episode was the same set of clay faces that had been presented at 4 months; the procedure was identical with that used at 4 months. Fixation time and duration of vocalization were again coded during the presentation of the faces. Vocalization time and duration of orienta- tion toward a speaker baffle were coded during the auditory episode. The speaker baffle was 1 ft above and 45 deg to the right of the child's right eye. Many of the children oriented their heads and eyes toward the speaker baffle when the recorded voice was being presented. Each 13-month-old infant was exposed to four episodes in the same apparatus used at 8 months. This report is concerned only with the third of these four episodes, during which the child was shown a set of five different clay faces. Four of the faces were identical with those viewed at 4 and 8 months. The fifth face contained a pair of eyes cor- rectly placed, but no nose or mouth. Each of the five faces was pre- sented three times in three different orders of presentation. Each face was presented for 30 sec, with 15-sec intervals between exposures. The duration of each fixation and vocalization was coded. RESULTS Fixation-Time Stability Two fixation-time variables will be considered: average first fixation and average total fixation across all stimulus presentations in an episode. First fixation times averaged 8 sec at 4 months and 5 sec at 8 and 13 months. Total fixation times averaged 17 sec at 4 months and 8 sec at 8 and 13 months. There were no significant sex differences in average fixa- tion time or variability at any of the ages. Table 1 presents the stability of first and total fixation times across the three ages expressed as the co- 337
JEROME KAGAN efficients of product moment correlations; sample sizes are usually 60 boys and 60 girls. There was minimal intra-individual stability of fixation time from 4 to 8 or from 4 to 13 months for either sex. There was moderate contin- uity from 8 to 13 months for girls, but not for boys. It is relevant to add that both first and total fixation times at 8 months covaried posi- tively with the parents' educational level for girls, but not for boys. When the mean parental educational attainment was divided into four steps, analysis of variance on the total-fixation-time scores revealed a significant variance ratio (F) for girls (F = 3.49; 3, 75 df; p < 0.05 for achromatic faces; F= 2.28; 3, 61 df;p = 0.09 for clay faces). The cor- responding variance ratios for boys were less than 1.0. The greater stability of fixation time for girls was accompanied by evidence that social-class membership was influencing fixation time. The favored in- terpretation of these data is that duration of fixation time at 4 months is primarily a function of degree of discrepancy between the stimulus faces and the child's schema of a face. The primary determinant of fix- ation time at 8 and 13 months, however, is the density of hypotheses activated to assimilate the faces. If middle- and upper-middle-class girls have acquired richer nests of hypotheses to faces, they should have longer fixation times. This interpretation suggests that the stability of fixation time from 8 to 13 months reflects continuity of a dimension that we can call "richness of cognitive structures associated with the human face." This continuity is analogous to the typical correlation of 0.50 between vocabulary levels at the ages of 3 and 6 years. Children who are precocious in the acquisition of hypotheses to faces at 8 months retain this precocity for the next half-year. Vocalization-Time Stability Mean vocalization time was highest at 4 months (average, 2 sec), lowest at 8 months (average, 1.2 sec), and intermediate at 13 months (average, 1.5 sec). Vocalization in response to the faces typically occurred while the infant was looking at the face. Vocalization during the auditory episode at 8 months typically occurred when exposure to the stimulus ended, not during it. The children were usually quiet and attentive while the voice was speaking. When it stopped, babbling began, as though the vocalizations were released by the processing of the sounds. 338
Continuity in Cognitive Development During the First Year of Life There were no significant sex differences in mean vocalization times or variance ratios to any episode. Table 2 presents the product moment correlation coefficients for vo- calization times across the three ages. As with fixation times, there was no stability from 4 to 8 months for either sex. However, vocalization time showed suggestive stability for girls from 4 to 13 months and mod- erate stability from 8 to 13 months. There was no consistency for boys between any two ages. The sex difference in stability of vocalization is not linked to social class; there was no strong relationship between vo- calization and educational level of the parents at any age. Despite the absence of sex differences in duration or variability of vocalization, it is suggested that vocalization reflects processes closely related to atten- tiveness more faithfully in girls than boys. The girls' stability coeffi- cients imply continuity of an attentiveness dimension in girls. Let us consider support for this statement. The 4-month-old girls who vocalized in response to the achromatic faces displayed longer visual orientations to the speaker baffle at 8 months; this relationship did not occur in boys (r â 0.28, p < 0.05 for girls; r = -0.10 for boys; r is the correlation coefficient). This sex dif- ference in the predictive significance of vocalization is seen in bolder relief when the analysis is restricted to infants with long fixation times (i.e., above the median on first fixation at 4 months). This group of long-fixation 4-month-old infants was divided at the median for the vocalization score into those who vocalized 1 sec or more and those who vocalized less than 1 sec in response to the achromatic faces. The high-vocalizing 4-month-old girls oriented to the speaker more often than the low-vocalizing girls (8 versus 2 sec; p < 0.05). The differences for boys were the reverse of those for girls. High-vocalizing boys ori- ented less often toward the speaker than low-vocalizing boys (4 versus 8 sec; p < 0.05). If orientation toward the source of the human voice at 8 months is regarded as a partial index of a tendency to attend to inter- esting auditory stimuli, this disposition is predicted by frequent vocali- zation in the 4-month-old girl, but by infrequent vocalization in the 4-month-old boy. A second source of support for the hypothesis that vocalization is a more faithful index of attentional processes in girls than in boys comes from the covariation between duration of first fixation and vocalization at 4 months. The distribution of first fixation times to each stimulus 339
JEROME KAGAN M J '5 1 o '3 T3 3 V3 O "â O «£ ⢠« '5. I § b d UJ s â¢& gj o a 5 â¢3 ll I 00 o d A 3 to ⢠o °" ^ _ M JJJ O 5 5^> n ⢠< 0 a T 's Crt â¢S B Wl *o O u, !5 if, A _o o d fi -. *^ i s O rf c o o 1 (N f u q S CO ** m I o I 1 S S § R H do do B n 0 a 1 u. ⢠oo r~, O fi -H -H -HO 1j « o d d d cd o D V) CO J3 ^ 3 , E 0 Oi 8' â¢^ ^1 C/5 1 « O O -H -H -H I b c o b o o o o do do S Predictor H Achromat Vocalizati 8 c o ts o r.J r- u 0 O -HO § o do do â¢5 CO 1 5 ^l i!i.i I (A Sfl 1 11 u 1 'B .N 00 .â ° .N '^ C W5 â¢N i i 5 "3 g T3 jj "9 5 â¢? g "« ** \f\ *^ a c o 00 §o J2 3 t2 JS ^ < 0 < â¢Â§ G do OQ vv H a c_ Q. 00 "*i d .C 340
Continuity in Cognitive Development During the First Year of Life was split into thirds, and the distribution of vocalization into halves (i.e., equal to or greater than 1 sec versus less than 1 sec) for each trial. The probability of vocalizing 1 sec or more increased linearly with length of first fixation for girls, but was independent of fixation time for boys. The pattern of intercorrelations at 8 months furnishes additional support for the attentional significance of girls' vocalization. Boys and girls were equally likely to vocalize at the end of each of the auditory recitations at 8 months. But the vocalization was positively associated with magnitude of cardiac deceleration (one index of orientation to an event) on the preceding trial for girls, but not for boys. For example, the correlation between vocalization following the high-meaning-high- inflection stimulus and magnitude of cardiac deceleration during the presentation of the stimulus was 0.36 (p < 0.01) for girls and only 0.01 for boys. The girls who vocalized after voice stimulation showed the largest cardiac decelerations in response to the voice. Inasmuch as cardiac deceleration is one reasonable index of quieting in the service of attention,29-31'32 the girls' babbling seems more clearly in the service of attentional processes than the babbling of boys. A fourth line of evidence is based on the girls' differential vocaliza- tion in response to the faces. In the first presentation of the faces, the girls vocalized more in response to the most regular representation (photo regular) than to the schematic representation (schematic regu- lar), whereas the boys' vocalizations were equivalent (2.2 versus 1.5 sec for girls; 2.5 versus 2.4 sec for boys). Similarly, the girls vocalized twice as long in response to the regular clay face as to the scrambled clay face (3.7 versus 1.9 sec); the boys' scores were almost equal (2.6 versus 2.8 sec). This differential vocalization is supported by an independent inves- tigation of 32 girls and 32 boys, 6 months old, who viewed achromatic faces and geometric forms in the same series. The girls vocalized sig- nificantly more in response to the faces than to the forms; the boys' vocalizations were equal for both classes of stimuli (Lewis and Kagan, unpublished). The special link between early vocalization and indexes of level of cognitive development in girls is supported by the research of others. Infant girls 6-12 months old with high scores on a vocalization index derived from the Bayley Infant Intelligence Scale had high Stanford- Binet IQ scores during the period from 6 to 26 years of age (correla- 341
JEROME KAGAN tions ranged from 0.40 to 0.60 during adolescence and early adult- hood). There was no relationship between the boys' vocalization scores and IQ during the later years.5 Only the girls' early babbling previewed a future level of cognitive development. A final source of evidence is a longitudinal study in London in which 41 boys and 35 girls were seen at the ages of 6 and 18 months and 2, 3, 4, 5, and 8 years.33 At 6 and 18 months, each child was assigned a speech quotient from the Griffith's Infant Scale, which assessed spon- taneous babbling at 6 months and use of words at 18 months. Although there were no sex differences in mean speech quotient at 6 or 18 months, the speech quotient was more stable from 6 to 18 months for girls than for boys (r = 0.51 for girls versus 0.15 for boys). Further- more, the speech quotient at 6 months predicted vocabulary level at 3 years for girls (r = 0.52, p < 0.01) but not for boys (r = -0.01). SUMMARY Despite different procedures, populations, and a priori hypotheses, the data from Cambridge, Yellow Springs, Berkeley, and London are re- markably concordant in suggesting that babbling in response to human faces or voices is more stable for girls than for boys during the first year and a better predictor of future indexes of level of cognitive de- velopment for girls than for boys. There are at least two possible inter- pretations of this empiric generalization. It can be argued from a purely environmental position that mothers who are motivated to accelerate their daughters' mental development are likely to spend a lot of time in face-to-face vocalization with themâmore time than they would with sons, and more time than would mothers who are not overly concerned with their daughters' rate of development. A mother's face-to-face vo- calization should lead to increased levels of babbling in her daughter.37 However, such a mother would also be expected to continue to stim- ulate her daughter and would probably teach her words early and encourage the development of other skills that are measured on intelli- gence tests. The predictive link between early babbling and later cogni- tive abilities would be a function of the continuity of a mother's accel- eration of her daughter's development. The absence of this predictive link between infant vocalization and cognitive development in the boy would require the assumption that accelerating mothers do not prefer- 342
Continuity in Cognitive Development During the First Year of Life entially engage in face-to-face vocalization with their sons. Preliminary data support this assumption. Observations of mother-infant interac- tions in the home reveal that well-educated mothers engage in more dis- tinctive face-to-face vocalization with their daughters than do less- educated mothers, whereas there is no comparable difference among the mothers of sons.24 Moreover, Moss34 has reported that middle-class mothers are more likely to imitate the vocalization of their 3-month-old daughters than that of their sons. A second interpretation has a more biologic flavor and holds that there is a basic sex difference in neuromotor organization. It is possible that vocalization is more prepotent for girls than for boys as a behav- ioral reaction to the arousal occasioned by the processing of interesting stimuli. This more speculative position finds some support in natural observations in primates. Among the langur of northern India, for ex- ample, squeals and screams are observed more frequently among fe- males than among males.21 More important, however, is the generally accepted premise that closely related strains or the sexes within a strain can differ in their typical reactions to states of arousal.6'39 This gen- eralization might hold for male and female infants with respect to early vocalization. Aside from possible sex differences in the significance of vocaliza- tion, it is to be noted that both vocalization and fixation time displayed better stability from 8 to 13 months among girls than among boys. In- deed, there was minimal continuity for the boys. This finding is paral- leled by data on older children, indicating greater long-term stability for girls on a variety of cognitive dimensions, including intelligence quo- tients4 and decision times in problem situations.23 Although it is pos- sible to explain the older girls' greater continuity as a result of social- ization experiences, there may be some biologic basis to this sexual dimorphism in cognitive functioning. Girls manifest greater stability than boys on a variety of physical-growth variables, including onset of ossification centers,14 and Acheson1 has concluded that "in almost every respect the physical development of the female is more stable than that of the male"(p. 497). The biologic processes that mediate girls' more stable physical growth may also be partly responsible for their more impressive stability in psychologic dimensions during in- fancy, as well as later childhood. At the least, the data question the popular myth of female unpredictability. 343
JEROME KAGAN The work reported here was supported by Public Health Service research grant MH 8792 from the National Institute of Mental Health and contract PH 43-65-1009 from the National Institute of Child Health and Human Development and by a grant from the Carnegie Corporation of New York. I thank Robert McCall, Barbara Henker, Michael Ross, Janet Levine, Leslie Rovainen, Judith Jordan, and Cheri Minton for their assistance in this research. REFERENCES 1. Acheson, R. M. Maturation of the skeleton, pp. 465-502. In F. Falkner, Ed. Human Development. Philadelphia: W. B. Saunders Co., 1966. 644 pp. 2. Bandura, A., and F. L. Menlove. Factors determining vicarious extinction of avoidance behavior through symbolic modeling. J. Personality Soc. Psychol. 8:99-108, 1968. 3. Bayley, N. On the growth of intelligence. Amer. Psychol. 10:805-818, 1959. 4. Bayley, N., and E. S. Schaefer. Correlations of maternal and child behaviors with the development of mental abilities: Data compiled from the Berkeley Growth Study. Society for Research in Child Development, Volume 29, No. 6, Series No. 97, 1964. 80pp. 5. Cameron, J., N. Livson, and N. Bayley. Infant vocalizations and their relation- ship to mature intelligence. Science 157:331-333, 1967. 6. Collins, R. L. Inheritance of avoidance conditioning in mice: a diallel study. Science 143:1188-1190, 1964. 7. Denenberg, V. H. Critical periods, stimulus input, and emotional reactivity: a theory of infantile stimulation. Psychol. Rev. 71:335-351, 1964. 8. Escalona, S. K.., and A. Moriarty. Prediction of school age intelligence from infant tests. Child Develop. 32:597-605, 1961. 9. Fantz, R. L. Pattern discrimination and selective attention as determinants of perceptual development from birth, pp. 143-172. In A. H. Kidd and J. L. Rivoire, Eds. Perceptual Development in Children. New York: International Universities Press, 1966. 548 pp. 10. Fantz, R. L. Pattern vision in newborn infants. Science 140:296-297, 1963. 11. Fantz, R. L., and S. Nevis. Pattern preferences and perceptual-cognitive development in early infancy. Merrill-Palmer Quart. 13:77-108, 1967. 12. Finley, G. E. Visual attention, play, and satiation in young children: a cross- cultural study. Doctoral dissertation. Cambridge, Mass.: Harvard University Press, 1967. 13. Fuller, J. L. Experiential deprivation and later behavior. Science 158:1645-1652, 1967. 14. Garn, S. M., F. N. Silverman, and C. G. Rohmann. A rational approach to the assessment of skeletal maturation. Ann. Radiol. 7:297-307, 1964. 15. Gesell, A. L., and C. S. Amatruda. Developmental Diagnosis (2nd ed.). New York: Paul B. Hoeber, 1947. 496 pp. 16. Haaf, R. A., and R. Q. Bell. A facial dimension in visual discrimination by human infants. Child Develop. 38:893-899, 1967. 344
Continuity in Cognitive Development During the First Year of Life 17. Haith, M. M. The response of the human newborn to visual movement. J. Exp. ChUd Psychol. 3:235-243, 1966. 18. Harlow, H. F. The heterosexual affectional system in monkeys. Amer. Psychol. 17:1-9, 1962. 19. Harlow, H. F., and M. K. Harlow. Learning to love. Amer. Sci. 54:244-272, 1966. 20. Hinde, R. A. Animal Behavior: A Synthesis of Ethology and Comparative Psychology. New York: McGraw-Hill, 1966. 534 pp. 21. Jay, P. The common langur of North India, pp. 197-249. In I. DeVore, Ed. Primate Behavior: Field Studies of Monkeys and Apes. New York: Holt, Rinehart and Winston, 1965. 654 pp. 22. Kagan, J. On cultural deprivation. In D. Glass, Ed. Proceedings of a Conference on Biology and Behavior. New York: Rockefeller University Press, (in press) 23. Kagan, J. Reflection-impulsivity and reading ability in primary grade children. Child Develop. 36:609-628, 1965. 24. Kagan, J., J. Levine, and C. Fishman. Sex of child and social class as deter- minants of maternal behavior. Paper presented to the Society for Research in Child Development, March, 1967. 25. Kuffler, S. W. Discharge patterns and functional organization of mammalian retina. J. Neurophysiol. 16:37-68, 1953. 26. Kuffler, S. W. Neurons in the retina: organization, inhibition and excitation problems. Sympos. Quant. Biol. 17:281-292, 1952. 27. Lenneberg, E. H. Biological Foundations of Language. New York: John Wiley &Sons, 1967. 489pp. 28. Levine, S. Maternal and environmental influences on the adrenocortical response to stress in weanling rats. Science 156:258-260, 1967. 29. Lewis, M., J. Kagan, H. Campbell, and J. Kalafat. The cardiac response as a correlate of attention in infants. Child Develop. 37:63-71, 1966. 30. Marler, P. R., and W. J. Hamilton. Mechanisms of Animal Behavior. New York: John Wiley & Sons, 1966. 771 pp. 31. McCall, R. B., and J. Kagan. Attention in the infant: effects of complexity, contour, perimeter and familiarity. Child Develop. 38:939-952, 1967. 32. McCall, R. B., and J. Kagan. Stimulus-schema discrepancy and attention in the infant. J. Exp. Child Psychol. 5:381-390, 1967. 33. Moore, T. Language and intelligence: a longitudinal study of the first eight years. I. Patterns of development in boys and girls. Hum. Develop. 10:88- 106, 1967. 34. Moss, H. A. Sex, age, and state as determinants of mother-infant interaction. Merrill-Palmer Quart. 13:19-36, 1967. 35. Piaget, J. The Construction of Reality in the Child. New York: Basic Books, 1954. 386pp. 36. Piaget, J. The Origins of Intelligence in Children. (2nd ed.) New York: International Universities Press, 1952. 419 pp. 37. Rheingold, H. L., J. L. Gewirtz, and H. Ross. Social conditioning of vocal- ization in the infant. J. Comp. Physiol. Psychol. 52:68-73, 1959. 38. Salapatek, P., and W. Kessen. Visual scanning of triangles by the human new- born. J. Exp. Child Psychol. 3:155-167, 1966. 39. Tollman, J., and J. A. King. The effects of testosterone propionates on aggres- sion in male and female C-57 BL/10 mice. Anim. Behav. 6:147-149, 1956. 345
JEROME KAGAN DISCUSSION DR. KAGAN: I want to add to the first empiric generalization summarized in my paper, dealing with the changing control of fixation time in the young infant. The second generalization, which I hold with a little less confidence than I do the first, is an idiographic issue, rather than a nomothetic one. We noticed both at the Pels Research Institute and in Cambridge a covaria- tion between the style of the child's habituation and his pattern of play, and this story takes us to age 7 and reading ability. We put children at three ages in a free-play situation. (These are the same longitudinal studies from which we gathered the previous data.) At 8 and 13 months, a child is placed in a room about 11 ft square (the mother is asked to sit in a chair and not say anything), is given age-appropriate toys, and is allowed to play for about a quarter of an hour. We code the rate at which the child changes his attentional involvement. Some children pick up a toy, play with it for 10 sec, drop it, pick up another toy, drop it in 5 sec, and so on. Another child picks up a toy and stays with it for a minute. He does not do anything more creative with the toyâhe does the same thing that the other child does; but he does not turn his attention from it so quickly. There appear to be stable individual differences in the rate at which children change attentional involvement. Moreover, there is a correlation for boys, but not for girls, between rate of habituation to visual stimuli at 4 months and pat- tern of play. Specifically, rapid habituators at 4 months show a fast tempo of play at 8 months, with many act changes in their 15-min free-play period, whereas children who are slow habituators at 4 months show long periods of attentional involvement in play. At 27 months of age, the children play in a larger room, decorated as a living room. The mother sits on a couch reading a magazine, and a new set of toys is introduced. We record the duration of each attentional involvement during the half-hour session. Children 27 months old tend to play with a toy for 30 sec be- fore they turn their attention from it, but there are dramatic individual differ- ences. Some children have a modal attentional involvement of 20 sec with no act lasting longer than 100 sec. Some have a mode of 40 sec with many acts that last 2 or 3 min. We then asked whether there was a relationship between tempo of play at 8 months and at 27 months of age. There was for boys, if one looked at the ex- tremes. That is, fast-tempo boys at 8 months tend to be fast-tempo boys at 27 months. 346
Continuity in Cognitive Development During the First Year of Life We believe that tempo of play, like any behavior, is determined by multiple factors. Obviously, the richer the set of hypotheses the child has for a particular object, the longer he will play with it. But, as a working idea, we suggest that there may be a biologic variable that exerts some influence on the tendency for a child to develop into a fast- or slow-tempo person. When we compared nine boys who were slow-tempo versus nine who were fast-tempo at 2*7 months, the slow-tempo boys had slightly shallower habituation curves at 4, 8, and 13 months, with the difference greatest at 4 months. The differential habituation curves to the faces at 4 months suggest that there is a tendency for fast-tempo boys to have been rapid habituators as infants. We should remember that we are talking about the top and bottom 10%, and not the whole population. We have worked for several years on a psychologic dimension in school-age children that we call "reflection-impulsivity." This tendency is assessed in a per- ceptual match-to-standard test in which the child is shown a picture and must pick from a set of six or eight variants the one that matches the standard. Some children offer their first hypothesis in 4 sec, and they are often wrong. Some wait 30 sec and are usually correct. The former are called "impulsive," the lat- ter, "reflective." The correlation between errors and response time on this task is typically negative. The tendency to be reflective or impulsive generalizes across tests and tasks. We also found that this tendency tends to be stable over short (12 weeks) or long (1-2 years) periods. What makes a child reflective or impulsive? Most of the variance, we believe, is due to psychodynamic factors related to anxiety over error. If a child is anx- ious over making a mistake, he is likely to be reflective. If he is not, he is likely to be impulsive. But when we look at extreme children and match them on social class and intelligence, I am persuaded that a few impulsive children would find it difficult to be reflective, and a few of the reflective children would find it diffi- cult to be impulsive. We suggest that fast-habituating, fast-tempo infants have a slightly higher probability of ending up impulsives, rather than reflectives, as a result of biologic variables. We believe, moreover, that impulsive children are more likely to make errors in school tasks when reflection would be an advantage (Child Develop. 36:609- 628, 1965). We selected children who were either reflective or impulsive, but who were of equal intelligence, and gave them a test to see the kinds of read- ing errors they would make. A child was given a card on which were printed "moon," "boom," "soon," and "room." The examiner then said one of the words and asked the child to point to it. Impulsive children made many more errors than reflective children. A year later, we asked the same children to read a paragraph of prose. The results will not be surprising: the impulsive children made many more errors, adding suffixes and usually missing initial graphemes (saying "trick" for "truck" or "wagon" for "witch"). These errors occurred 347
JEROME KAGAN more commonly for the impulsive than for the reflective children. Only one type of error was more frequent for the reflective children. They more fre- quently corrected a mistake spontaneously. The reflective child might say: "The dog ranâno, noâthe cat ran." Some evidence that tempo of play has a biologic component comes from a study of twins done by Christine Reppucci. She saw monozygotic and dizygotic twins in the free-play situation described earlier and also had them look at visual stimuli. The results suggested heritability, for this population, for tempo of play at 8 months and rate of habituation. DR. CAVANAGH: Do your data permit you to say anything about the fluency of these children, their use of language? DR. KAGAN: We thought that there would be a correlation between language flu- ency and impulsiveness, and William Ward of the Educational Testing Service ran a study (unpublished) of the fluency of verbal resources or fluency of ideas. We were hoping that the impulsive children would have greater fluency, but the cor- relation was low and nonsignificant. DR. GUNDERSON: Did you find any understanding about language acquisition? DR. KAGAN: The correlation between verbal resources and impulsivity is 0.15 for boys and insignificant, and about 0.30 for girls. In general, reflectivity is corre- lated with verbal IQ. Others have found that verbal fluency is more highly corre- lated with verbal IQ in girls than in boys. DR. LINDSLEY : Do you associate reflectivity with perseverance, in the sense that a person stays with a thing longer? There is a two- or three-pronged aspect to this question. A study done several years ago on activities that were started and then interrupted tried to find out whether subjects would resume the same ac- tivity after the interruption. There was some evidence of perseverance, and I am sure you have seen that in animal studies when you interrupted the system through the cortex. In relation to active children and what you called the "fast-tempo" group: All of us tend to walk fast or slow, and the temporal factors characteristic of a per- son may be biologically inherent or acquired. I am interested in whether reflec- tivity had an aspect of perseverance or long-term attention, and whether these are advantageous features. It seems to me that we must have people who can at- tend to what they are working on long enough for information exchange to take place, but who need not hang on so long that they cannot break off and pay at- tention to something else. DR. KAGAN: When a school-age child becomes involved in a task for which there is a correct or an incorrect solution, he reflects for a variable period. It seems to me that, although an impulsive child does not care about failure, a reflective child does not like to have a failure on his record and, therefore, perseveres. I do not want to suggest that reflective children's behavior is superior or better in all situ- ations; it depends on the task. I believe that we are viewing in these data an as- 348
Continuity in Cognitive Development During the First Year of Life pect of a variable that has always been present in man's description of man. Hippocrates talked of the sanguine, choleric, and phlegmatic types, and Jung talked about introversion and extroversion. These terms capture a stable char- acteristic of human beings, and we think that the reflection-impulsivity dimen- sion is related to these typologies. DR. YOUNG: Did you find any intelligence differences between the reflective and the impulsive types? DR. KAGAN: Girls show a higher correlation between reflectivity and verbal IQ than boys, and that is a consistent finding. But the correlation is not high enough to suggest that these are similar processes. Dr. Keogh has some interesting exper- iments that would help us to understand these data and that also relate to Dr. Sperry's ideas on cerebral dominance. DR. KEOGH: Most of the traditional tests of spatial orientation have been paper- and-pencil tests. Youngsters are asked to copy a pattern to see whether they can produce something like the original. The child is usually sitting in a relatively sta- tionary position, and the reference points are clearly defined for him. We have been asking children to walk, a three-dimensional activity, to reproduce some rather simple patterns, and it is very interesting to observe their behavior. There was a definite difference between boys and girls in the production of simple and complex forms. We asked our children to pretend they had paint on the bottoms of their feet, and to make a five-pointed star, or to make one in the sand by walking in it, which was something relatively easy for them to do. We noticed in an early study (Keogh and Keogh, Amer. J. Mental Def. 71:1009-1013, 1967) that normal British schoolboys aged 6-9 years showed improvement with age in their ability to make these patterns by walking. Later, in this country, where we were working with 8- and 9-year-old normal youngsters, we asked them to walk patterns under three conditions. One was to walk on the open floor with no ref- erence points defined; they were told in essence, "You can walk anywhere in the room that you want to; make the pattern any size you want, in any position you want." Another was to walk the same pattern on a 9- by 9-ft plain linoleum mat; this condition limited the space to be used and supplied structure to the field. And in the third test, the mat was covered with very fine sand so that the foot- prints left a good pattern. We found with boys that they got better under the three conditions and were best on the sandâthey made very precise figures, with accurate angles and so forth. Some 8- or 9-year-old normal girls walked these patterns under the same conditions as for the boys. There were some differences in pattern orientation, and the boys were significantly better at producing precise figures (Keogh, sub- mitted for publication). There are many reasons why this might be so, but it is probably simply that girls just do not use the available cues. The differences were dramatic, not only in the precision of the patterns, but also in the sort of organizational system 349
JEROME KAGAN used. To be specific, one of the patterns was a triangle inside a circle. Almost without exception, the boys walked a circle, then stopped, sometimes stepping off the mat or the sand, and then made the triangle. We have observed that girls attempted to make the patterns in one continuous line as if it were all one big pattern; they did not seem to break it down into parts. One of our patterns was two circles in a horizontal plane. We have had a num- ber of girls, but fewer boys, who walked a figure "8." We feel we have come across something that is fairly specific in perceptual abilities, namely, a sex differ- ence in spatial perception. All these children can discriminate between the patterns, and all can draw them. We have also found (Keogh, Amer. J. Mental Def., in press) that retarded youngsters are very disoriented. One sometimes wonders how they ever get home, because they really do not seem to know where they are. 350
R OBER T L. FANTZ Visual Perception and Experience in Infancy: Issues and Approaches The intake and processing of information by young infants is a rela- tively new field of study, even though there has been a rapid accelera- tion of research efforts during the last decade. It is too soon to give a clear picture of the visual world of the infant or to attempt to state the basic principles underlying early perceptual development and acquisi- tion of visual information. Certainly, it is too soon to select with confi- dence the findings that will prove to be of most value in understanding the eventual attainment, or lack of attainment, of complex perceptual performances by the child and adult. It will have to suffice to point out some of the issues in the field, to describe some promising approaches to them, and to present a highly selected sampling of data already ob- tained. The material to be covered is heavily influenced by the view- points of one investigator in the field and by the research that has re- cently been done in one laboratoryâthe Perceptual Development Laboratory of Case Western Reserve University. The field of early perceptual development can be conveniently di- vided into four interrelated topics: visual-motor coordination and spa- tial localization, visual resolution and discrimination capacities, visual preferences and other selective responses to visual stimulation, and the retention of visual information and other effects of visual experience. 351
ROBERT L. FANTZ VISUAL-MOTOR COORDINATION Because this topic is covered by other papers of this conference, my re- marks here will be limited to emphasizing the distinction between the first two topics. Visual-motor coordination is the primary determinant of spatial localization and accurate directed responses, whereas visual discrimination capacity is the primary determinant of the intake of vi- sual information, assisted by visual-motor coordination to the degree that it may exist. This distinction raises an issue of historical impor- tance: By what response indicators shall visual perception be measured in the young infant? For many of the early investigators (e.g., Gesell etal.,15 Ling,23 and McGraw25) visual perception was operationally de- fined as the ability to successfully fixate, pursue, and grasp an object. Because these abilities were found to be initially poor or absent and only gradually perfected during the early weeks or months of life, the assumption was that other important aspects of visual perception, es- pecially the ability to distinguish the form and patterning of objects, were similarly slow in developing. But lack of precision in oculomotor coordination or accommodation, or even severe impairment of these functions, does not preclude the ability to see, although it interferes with the most effective use of vision. This may be illustrated by the evi- dence from patients with congenital cataracts, who, soon after opera- tion, sometimes were able to distinguish between (as distinct from nam- ing) different colors and forms, in spite of lacking a lens, having minimal oculomotor practice, and often having spontaneous nystagmus.28'30 Conversely, precise coordination in fixating, pursuing, and grasping an object does not indicate whether the form, size, color, solidity, or any other attribute of the object is discriminable. Good visual-motor co- ordination and spatial localization are as phylogenetically old as the ver- tebrate eye, but the ability to discriminate complex forms has only gradually evolved. An obvious answer to the question of how to measure perception in the infant is that responses and experimental procedures appropriate to the particular aspect of perception under study should be selected. For measuring visual-motor coordination and spatial localization, the pre- cision of the infant's response to a single stimulus target is sufficient. For measuring discrimination or identification in nonverbal subjects, a minimum of two targets is required, along with the usual controls nec- essary for a reliable discrimination experiment. The nature or precision 352
Visual Perception and Experience in Infancy of the response is not critical, as long as the subject's choice between stimuli differing in some dimension can be determined without ambigu- ity. The visual fixation response, in particular, is sufficiently coordinated even in the newborn infant to indicate the choice of one or another target. Recent studies,2-5.l8'19 with more accurate measurement and better-controlled stimulus conditions than most of the earlier work, have generally found the neonate to possess better-developed abilities to fixate, converge, pursue, and accommodate than had been realized. VISUAL DISCRIMINATION Visual-discrimination tests in nonverbal subjects have traditionally used discrimination training or other conditioning procedures. Until recently, these procedures were not thought to be applicable to the infant in the early months, owing to the lack of coordinated, visually directed re- sponses, as well as to the lack of adequate learning abilities. But ad- vances in technology have made feasible the conditioning even of new- born infants (see the following presentation, by Lipsitt) and have opened up wide possibilities for discrimination testing in young infants. In particular, Bower1 was able to demonstrate in the 2-month-old in- fant a number of phenomena of spatial perception, including depth dis- crimination, orientation discrimination, size constancy, and shape con- stancy. He first conditioned, using "peek-a-boo" as reinforcement, a head-turning response to a particular object, and then tested for dis- crimination among this and other objects differing in specified ways. Most of the available information on discrimination capacities, es- pecially pattern discrimination, in the early months of life has come from the visual-preference method. This approach to discrimination testing measures the untrained differential responsiveness to two or more stimulus targets. The consistent tendency of the infant to look longer or more often at a particular target in repeated exposures, with position and other extraneous factors controlled, indicates both the ability to distinguish a stimulus difference and the selection of a par- ticular stimulus for attention. Of course, the absence of a differential response may indicate either inability to discriminate or equal interest among the targets. This limitation has proved not to be serious, in view of the wide range of stimulus attributes that have elicited visual preferences. 353
ROBERT L. FANTZ The question asked in the early visual-preference experiments was how soon the infant began to see patterned stimulation, the source of most visual information. The controversy between those who argued that the infant requires a long period of visual experience and learning and those who insisted that the infant requires a long period of post- natal maturation was eventually resolved by the finding that even the newborn infant can resolve and discriminate patterns.6'10'19'20'27 But there remained three issues: When does the visual system first become capable of function? What are the discrimination capacities at various maturational levels after that beginning point? What are the specific contributions of postnatal experience to the initial primitive level of perception? Research on the last of these will be discussed below. Re- garding the first and second issues, some very recent work26 has pro- vided the most relevant information, as well as incidentally giving the most extensive and most reliable data available on differential visual responses in newborn infants. These results will be summarized here. Miranda26 tested a group of 27 premature infants, less than 38 weeks after conception but after various periods in an incubator, and com- pared them with 27 full-term infants averaging 3l/2 days of age. Our paired-comparison visual-preference technique13 was modified to the requirements of the small and immature subjects. The infant was se- cured in a semireclining position in a form-fitting, adjustable seat and rolled under a stimulus chamber (Figure 1). The chamber was relatively homogeneous inside, except for two stimulus targets (mostly 5-in. squares) placed side by side 1 ft from the infant and about 1 ft apart from center to center. Each of 14 pairs of flat, achromatic targets (Table 1) was exposed for two 10-sec periods with reversed left and right positions on the second period. Vertical striped patterns of three widths were each paired with gray to determine the smallest pattern that would be discriminated from an unpatterned target, as a rough es- timate of visual acuity.14 Listed next are three steps in a "complexity" dimension, using a series of white squares containing 0, 1, 4, or 16 black squares, regularly arranged. In the next three pairs, linear versus curvilinear forms or arrangement were opposed, but with white : black ratio and length of contour equated. The remaining five pairs of as- sorted and often multiple stimulus variations were taken from previous studies indicating differential responses by newborns or infants in the early weeks of life. The reliability of the experimenter, who observed through a tiny hole 354
Visual Perception and Experience in Infancy FIGURE 1 Visual-preference testing apparatus in mock operation. So that the baby could be seen in the picture, the crib was not set in as far as it would be in normal operation. The ex- perimenter observes through a peephole between the targets. The second observer (standing) looks at a mirror above this hole. The recording unit and timer can be seen in the foreground. (From Miranda.26) 355
ROBERT L. FANTZ TABLE 1 Differential Visual Responses of Premature and Full-Term Newborn Infants" No. Infants Showing Longer Fixation of Left or Right Stimulus of Pair6 Stimulus Targets Premature0 Full-termd Acuity gratings ^-in.-striped-gray 22-0 26-0 ^-in.-striped-gray 23-1 23-3 g-in.-striped-gray 13-7 10-11 Complexity pairings, no. of elements 0-1 2-27 3-24 1-4 18-9 15-11 4-16 20-7 16-8 Linear versus round configurations L1-R1 14-7 13-11 L2-R2 12-10 12-11 L3-R3 9-10 13-10 Other stimulus variations* 1A-1B 22-5 19-6 2A-2B 15-5 17-9 3A-3B 21-3 20-2 4A-4B 24-1 25-0 5A-5B 7-11 13-10 "Adapted with permission from Miranda. "Tie scores and incomplete tests are omitted; results in italics are significant (p > 0.05) by two- tailed correlated t-test on time scores transformed for parametric analysis. ''Mean conceptional age, 35l weeks; mean postnatal age, 22i days; gestation varied from 28 to 37 weeks. "Gestation was over 37 weeks, with a mean of 40 weeks; age, not more than 1 week. '' 1 op to bottom pairs shown in Figure 3 as pairs 1,13 (with circles replacing the oval outlines), 17, 8, and 5. between the targets, in recording fixations was checked for most of the subjects by a second observer. This observer looked through a hole in the ceiling of the chamber into a small mirror above the center hole, adjusted to reflect the eyes of the infant, and recorded without knowl- edge of the targets or of the experimenter's recordings. In spite of infe- rior observation conditions for the second observer, the results for the two were in good agreement, in that the same pairs of targets were found either to show or not to show differential fixation, and the me- dian of the discrepancies between observers, disregarding the sign of 556
Visual Perception and Experience in Infancy the difference, was less than 2 sec in 456 tests of 20-sec duration. Table 1 gives one measure of the differential responses: the fre- quency of longer fixation on one or the other target of the pair. Sig- nificant preferences were shown by both premature and full-term groups for six of the pairs: the two larger acuity patterns over gray, one square over plain white in the complexity series, one of a pair of diverse black-and-white patterns (1A versus IB; pair 1 in Figure 3), a large over a similar small pattern (3A versus 3B; pair 17 in Figure 3), and a sharply defined pattern over one with shades of gray (4A versus 4B; pair 8 in Figure 3). Significant preference was shown by the prema- ture infants alone for an additional pair (one over four squares). It is evident from the responses of the premature sample that the var- ious parts of the visual system are capable of function at least a month before the usual time of birth. This conclusion is not altered by the pos- sibility that the longer opportunity for postnatal experience and oculo- motor practice for the prematures had some facilitating effect on the visual performances. However, such an effect, tending to compensate for the shorter period of maturation, is one of the possible explanations for the surprising degree of similarity in the responses of infants that have had 8 or 9 months to develop. It might even appear that the premature infants showed better pattern discrimination for several pairs. But at least in the case of the complexity pairings this is illusory, for the lower differential shown by the full-term infants probably represents the begin- ning of an upward shift in the preferred or "optimal" complexity level. Evidence of such a shift was obtained by a significant negative correla- tion between the degree of preference for the one-square over the four- square pattern and the maturational level (age from conception) of the combined premature and full-term groups. This result hints of the issue, to be discussed later, of whether the age of the infant should be mea- sured from conception or from birth in developmental research. On the stimulus side, it is evident from these results that both pre- mature infants (of the given range of conceptional and postnatal ages) and full-term neonatal infants can discriminate between patterns and plain surfaces of the same size and light reflectance, and that they can discriminate between different patterned surfaces in some cases. The results cannot be considered to indicate the limits of pattern vision in either group, unless the optimal testing conditions and stimulus varia- tions are known. For example, better visual acuity than hitherto found has been suggested for newborn infants by tests using both differential 357
ROBERT L. FANTZ fixation and optokinetic nystagmus responses, but with different ranges of pattern size and other differences in conditions.5-10 These and other results from newborn infants give no conclusive evi- dence of ability to discriminate between two patterns that are different in form or arrangement but equated in "complexity" or "quantity," such as the linear versus round pairs of Table 1. But for slightly older infants, such an ability has been demonstrated repeatedly: differentia- tion between irregular and regular arrangements of squares was suggested within 2 weeks of age,10 between stripes and bull's-eye patterns dur- ing the first month,7 between linear and circular configurations of line segments by 5 weeks, and between lattice and checkerboard arrange- ments of squares by 7 weeks13 (see Figure 3, pairs 3 and 5). Another study12 used a set of four different arrangements of 25 white squares against a dark background, with three of the arrangements being de- rived from a five-by-five matrix and differing only in orientation of the elements. Differences in response time increased with age (Figure 2) and became significant starting with the group at the age of 1-2 months. It may be concluded that within the first 2 months of life the in- fant can discriminate among patterns equated for total area, light: dark ratio, number of elements, and length of contour. On the other hand, it may be concluded that infants can discriminate from birth among pat- terns differing in such measures as number of squares in the pattern, length of contour, and number of "turns" in a random shape.3'19'20'22'24 Older infants have often but not always shown higher preferred- complexity levels. Without getting into the problem of defining "com- plexity" and distinguishing it from other variations in patterning, it can safely be stated that the young infant can discriminate more than one dimension of visual patterns. These various developmental changes in responsiveness to patterns might be explained by improvement in visual acuity, by an increase in the number and variety of discriminable dimensions of patterns, or by changes in the preferred points along the discriminated dimensions. The resolution of finer patterns with increasing age very likely contributes to some changes, but it would not explain demonstrated changes in pref- erence between patterns containing the same width of lines or size of squares; furthermore, the patterns in most studies have all been well above threshold. Regarding the second possible explanation, additional stimulus characteristics undoubtedly become discriminable with age 358
Visual Perception and Experience in Infancy 30 - o < 10 """' ⢠«l OOOOQ aanno DDDDD DDDDn aDDDD aaaan 2 3 AGE IN MONTHS (MEDIAN FOR GROUP) FIGURE 2 Relative visual attention to four arrangements of white squares, averaged for infants in five age groups. (Reprinted with permission from Fantz and Nevis.12) and experience (see Gibson16) and may result in some preference changes. But this explanation is not plausible when the patterns are found to be equally discriminable before and after the change, as in the case of a shift in preference from one to another of two patterns differing in complexity or configuration. Thus, some of the changes cannot be attributed to the development of visual capacities as such, and indicate, instead, the increased attention value of some patterns relative to others. Changes in visual selectivity may have as much por- tent for perceptual development in the infant and child as changes in visual acuity. 359
ROBERT L. FANTZ VISUAL SELECTIVITY Optimal perception of an object requires the turning of the eyes so as to allow foveal examination of the object. This involves picking a small area from a large, intricate, and variegated visual array. At a given mo- ment, this selection might be due to the intrinsic attraction of some stimulus characteristics; it might be a temporary interest in a stimulus due to novelty or movement or proximity; or it might be due to a long- term familiarization or conditioning process. Visual-motor and discrim- ination capacities set the limits of what the infant can perceive, but the nature of the environment and the selectivity of the moment determine what is perceived and hence what is learned about or responded to. The infant's experience is influenced just as surely by a tendency to look four times as long at one stimulus as at another as it is by the presence of one stimulus as opposed to another. Therefore, it is important to know the basis of selection by the infant and how it changes with age. For this purpose, the visual-preference method offers a simple and di- rect approach, although other relevant response indicators, especially for autonomic responses, can measure the "intensity," rather than the duration, of attention. The most general finding for infants of all ages is that a plain, non- moving, unpatterned surface or object of any color or brightness is low in attention value; it is probably more than coincidental that such a stimulus conveys little information. Conclusions as to the character- istics of nonmoving stimuli that are of high attention value depend much more on the age of the subjects and on the particular stimuli available. Among the stimuli that have been used to date, infants dur- ing the early weeks of life have looked most at sharply defined patterns, especially black-and-white patterns. That was brought out most clearly in a longitudinal study designed for other purposesânamely, to show differences in the rate of development of visual preferences between two selected groups of infants.12'13 The 18 pairs of stimulus targets (Figure 3) were chosen as those most likely, on the basis of past re- sults, to elicit definite intrapair changes in preference within the first 6 months of life; the specification of relevant stimulus characteristics was not the aim. Results for some pairs will be given later. At the mo- ment, the comparison of the responses among the 18 pairs is of interest to bring out the relative attention value of broad categories of stimulus targets. It happened that eight of the stimulus pairs included black-and- 360
Visual Perception and Experience in Infancy FIGURE 3 Pairs of stimulus targets used in the longitudinal study of selected home- and institution- reared infants, numbered according to order of presentation during each weekly test. Results in text are comparisons among the pairs and between the two members of some pairs. (Reprinted with permission from Fantz and Nevis.12) white patterns (this includes repeated testing of pair 1 following pair 12 and it includes pair 8, only one of whose members is black-and-white). Each of the eight pairs was fixated longer than any of the remaining pairs in averages of the seven tests given during the first 2 months of life. The ranking of these pairs, starting with the highest response, was pair 12, 1, 1 (repeated), 8, 17, 14, 3, and 13. The next two highest pairs in average fixation time (pairs 11 and 5) were also sharply defined pat- terns consisting of white elements against a blue background. By far the lowest in attention value was pair 18 (plain white and gray); the next 361
ROBERT L. FANTZ lowest were pair 9 (bright red checks and square), then pair 16 (flicker- ing orange light and complex pattern), and then pair 4 (red patterned boards). On the average, the eight black-and-white patterns were fix- ated significantly longer than the remaining pairs (even excluding pair 18) for each week of age from 2 through 12. However, this differential decreased considerably after 8 weeks; it disappeared by 15 weeks, and was reversed later. The pairs containing facelike patterns were initially about as high in attention value, and dropped almost as much, as the abstract patterns. It would seem that both the complexity and the configuration of a pattern are less critical determinants of visual attention early in life than its contour definition or brightness contrast. Although the sim- plest black-and-white pair of patterns (pair 13) was the lowest ranking of the eight, it was fixated longer than many pairs of targets that would be rated more complex by any definition. After 2 months of age, high pattern definition was not sufficient, although some sharply defined patterns continued to receive long fixation times. Between 4 and 6 months, widely diverse targets were of high attention value; the top- ranking five were pair 7 (mesh and wood object versus rotating red-on- yellow disk), pair 8 (schematic face versus face photograph), pair 15 (solid head model versus photograph), pair 16 (Egyptian art versus flickering orange globe), and pair 2 (brightly colored toys). Visual selectivity and developmental changes were still more marked in the comparison of the lengths of response to two stimulus targets ex- posed together. The selected results presented here are based on reanal- ysis of published longitudinal data on the first 6 months of age from the 10 infants constituting the "home sample,"12'13 combined with data from follow-up tests of the same infants during the second 6 months of life. In Figures 4-9, the graphed fixation-time averages be- fore 16 weeks of age are pooled averages for two successive weekly tests; after that age, they represent single tests given every 2 weeks through 24 weeks of age and every 4 weeks thereafter. The target des- ignations are based on left or right position of a stimulus as shown in Figure 3; testing positions of the two were varied systematically within each test and among successive tests. The results for two pairs of sharply defined, abstract, achromatic patterns are given in Figure 4. There is a general decrease with age in response to these patterns, as indicated above. More striking, however, is the rapid development of preference both for the bull's-eye pattern 362
Visual Perception and Experience in Infancy 35 25 15 PAIR 11 - Left target - Right target o I- I I I I I 35 PAIR 14 - Left target Right target 25 15 5 - I . I . I . I . I 4 8 12 16 20 24 28 32 36 40 44 48 52 AGE, WEEKS FIGURE 4 Fixation times for each target of a pair presented together for a total of 40 sec, averaged for the same 10 infants at each age. Left and right targets are as illustrated in Figure 3. Both pairs were flat, achromatic, sharply defined patterns varying in circular versus linear contours (pair 11) or in irregular versus regular arrangements of squares (pair 14). 363
ROBERT L. FANTZ and for the irregular arrangement of squares, reaching a peak in the third month, and then gradually falling off. Differentials for other pairs were less strong and consistent, even when the stimulus difference was much greater. For example, in Figure 5, the targets preferred during the first month of age and then again after 5 months were (in pah- 7) a slowly rotating disk covered with flu- orescent red and yellow paper and (in pair 16) a flickering 40-watt orange light bulb enclosed in a 6-in. translucent globe. The targets pre- ferred between 2 and 4 months were much higher in informational value but would generally be considered to be uninteresting: a spiral of wire mesh mounted to a board with holes in it and partly painted white (pair 7) and a dark achromatic photograph of intricate Egyptian art (pair 16). In Figure 6 (top), the strong initial preference for a black-and-white schematic face pattern over a face photograph disappeared by 16 weeks of age, most likely owing to the decrease in the early attention value of sharply defined patterns and the subsequent interest in other, more subtle patternsâas was true for pair 16. The irrelevance of the facial re- semblance is suggested by an absence, in the lower graph, of preference for the correct over the scrambled arrangement of a schematic face un- til about 20 weeks of age; in fact, there was some early preference for the scrambled face. These results are not in agreement with those in var- ious studies of institution infants that indicate a general preference for the correct arrangement during the third month.10 However, the differ- ential for this pair has never been as strong or consistent as that repeat- edly obtained with pairs of geometric patterns (cf. Figure 4). Other investigators' results have also varied as to whether or when infants dis- criminate correct from distorted face-like configurations, even though face-like stimuli in general have been of high attention value. It is prob- able that by several months of age the infant is capable of discriminat- ing the various experimental representations of faces both from each other and from real faces and that the presence or absence of differen- tial attention to some representations has little relevance to social rec- ognition and responsiveness. Figure 7 illustrates the visual selectivity found in a number of studies relative to two further types of stimulus variation. The top graph shows the development of increased responsiveness to a solid, contoured ob- ject, along with a corresponding decrease for a flat surface. Although the solid object, to an adult, resembles a sculptured head without 364
Visual Perception and Experience in Infancy 35 - 25 - 15 o K < 25 15 PAIR 7 - Left target - Right target /\ I i I i I , l I I I I I I I PAIR 16 - Left target Right target I . I . I . I . -L i I i l I L 4 8 12 16 20 24 28 32 36 40 44 48 52 AGE, WEEKS FIGURE 5 Fixation times for each target of a pair presented for a total of 40 sec, averaged for the same 10 infants at each age. Targets (from Figure 3) were a complex achromatic object (pair 7, left) versus a plain, brightly colored, rotating disk; and a dark achromatic art reproduc- tion (pair 16, left) versus a blinking orange globe. 365
ROBERT L. FANTZ 35 25 15 _ PAIR 8 - Left target Right target < rr 35 25 15 PAIR 12 - Left target Right target I I I I I I J_ 52 8 12 16 20 24 28 32 AGE, WEEKS 36 40 44 48 FIGURE 6 Fixation times for each target of a pair presented for a total of 40 sec, averaged for the same 10 infants at each age. Targets (from Figure 3) were schematic face pattern (pair 8, left) versus a face photograph; and the same schematic face pattern (pair 12, left) versus a scrambled arrangement of the same features. 366
Visual Perception and Experience in Infancy 35 25 15 <u c t/i 5 35 25 15 5 - PAIR 6 - Left target Right target I . I . I . L PAIR 1 (Repeat) Familiar target Unfamiliar target -L -L -L 12 16 20 24 28 32 AGE, WEEKS 36 40 44 48 52 FIGURE 7 Fixation times for each target of a pair presented together for a total of 40 sec, averaged for the same 10 infants at each age. Targets (from Figure 3) were a solid model of a head, painted flat white (pair 6, left) versus a similar white outline form; and one of two pat- terns (varied from week to week) that had been repeatedly exposed to the infant during the course of a testing session (pair 1, familiar) versus the other pattern, which was relatively novel. 367
ROBERT L. FANTZ painted features, the preference development is closer to that shown for other variations in solidity than that for other face-like pairings. As with several other pairings of solid and flat targets, for pair 6 the solid- ity preference appeared in the early months and was maintained thereafter. The bottom graph of Figure 7 shows the development of a prefer- ence based on specific visual experience, rather than on either intrinsic stimulus characteristics or general types of familiar objects (such as solid objects or face-like patterns). One of a pair of diverse black-and- white patterns was placed in such a position in the stimulus chamber that it would be exposed during the 10 sec or so after each test ex- posureâthat is, while the targets were being reversed or changed. After 22 such exposures, this pattern was tested along with the other pattern of pair 1. The two patterns were used as the "familiar" patterns on al- ternate test weeks for each subject. Although the novelty preference was not strong, compared to many stimulus variations, it was consistent among the infants after 2 months of age and for most of the first year, before it finally disappearedâperhaps due to the decreased interest in flat patterns in general. Results such as these, showing a change in responsiveness to repeat- edly exposed patterns, are of direct relevance to the ability to process and retain visual information. Such results might well have been in- cluded under the heading of "visual experience." But they are equally relevant to visual selectivity, inasmuch as novelty is one category of stimulus determinant of differential attentionâone that happens to be of particular theoretical importance. Surprisingly, response to novelty has been quite difficult to measure reliably and predictably; it has been affected by seemingly minor variations in experimental conditions.8 One of the persistent problems is that of equating patterns in initial attention value, so that effects of recent exposures will not be ob- scured. Another problem is to ensure that the decrease in response is specific to the pattern and therefore indicates pattern recognition. Some studies have not controlled this factor, and their results may in- dicate only a general habituation of response, decreased arousal, or fa- tigue. One solution is to use a paired-comparison procedure and to de- termine the response to the familiar stimulus relative to a novel one of similar attention value.11'13 There are other solutions, however. For example, Caron and Caron4 used the single-stimulus procedure quite effectively. They demonstrated 368
Visual Perception and Experience in Infancy the specificity of the response decrement for a repeated pattern by the maintained high response to novel patterns. The same study showed a smaller response decrement for a complex than for a simple repeated stimulus and showed retention of visual information over a short in- tervalâi.e., a single presentation of the familiar stimulus following three novel stimuli brought a significant drop in fixation time. These and other characteristics relevant to the effects of specific short-term visual exposure have only begun to be explored. Two additional variables, the source of the subjects and the duration of the test exposure, were found to be related to short-term familiariza- tion effects. In the longitudinal study using the stimulus pairs of Figure 3,12'13 two samples of infants were selected so as to be as different as possible in parental background and early environment. Half the infants were offspring of university faculty, the others were institution-reared infants. All infants were healthy, full-term, and of Caucasian parents; the mean birth weights of the two groups were identical. The familiar- ization test, included as part of the weekly preference testing, was as described above; fixation times for the home group were as given in Fig- ure 7. To permit better comparison of the two groups, fixation times were converted to percentages of total fixation time given to the famil- iar pattern in successive testing weeks (Figure 8); 50% indicated chance response. The top graph, based on the entire 40 sec of postfamiliarization ex- posure, shows decreasing response to the familiar pattern with increas- ing age for both groups. This development was more rapid for the home group; the difference in percentage between groups was significant at 12 through 18 weeks of age. In a subsequent analysis of these data by successive 4-sec periods of exposure, it was noticed that in the early months the home infants showed more differentiation between familiar and novel patterns after longer exposure to the two, as though recogni- tion of the familiar pattern (or habituation to it) required some period of examination of both patterns. The institution infants did not in the early months show this change during the exposure. Consequently, the preference curves based on only the last 4 sec of the second 20-sec ex- posure (bottom graph) show more group difference and show a signifi- cant novelty preference by the home group as early as 6 weeks of age. Increased stimulus differentiation after longer examination was not the usual outcome. For most of the pairs, varying in intrinsic stimulus characteristics rather than in previous exposure, at least as much prefer- 369
ROBERT L. FANTZ 70 PAIR 1 - Postfamiliarization exposure Total 40 sec of exposure 50 111 o te. 30 ee. u. o u- .*. Home â Institution J , I , i., __i_ 70 50 PAIR 1 - Postfamiliarization exposure Last 4 sec of exposure 30 Home â â â â Institution __, |_ , I , I I 10 12 AGE, WEEKS 14 16 18 20 FIGURE 8 Pattern familiarization results during the first 6 months for the selected homc- reared infants of earlier graphs compared with 10 institution-reared infants. For each group, fixation times for the two targets were converted to percentages for the target that had been repeatedly exposed earlier in the testing session, relative to the novel one. Percentages are based either on two 20-sec exposures (top graph) or on only the last 4 sec of the last exposure (bottom graph). 370
Visual Perception and Experience in Infancy 70 50 Q. LU o a. a. O U. 70 50 30 PAIR 3 Based on total 40 sec of exposure PAIR 3 I Based on first 4 sec of exposure Home Institution ! , ! , I . i_ 8 10 12 14 WEEKS OF AGE 16 18 20 FIGURE 9 Results comparing selected home infants and institution infants on development of preference for circular over linear arrangement of line segments. Percentages of fixation time are for target pictured on left (Figure 3), based either on two 20-sec exposures (top graph) or on only first 4 sec of the first exposure (bottom graph). 371
ROBERT L. FANTZ ence was shown early in the exposure of a pair as later on, especially by the home group. Consequently, in a few cases, more difference between groups was shown in preference curves based only on the first 4 sec of exposure, as will be illustrated with results for pair 3. The development of preference for a circular over a linear configura- tion of line segments (Figure 9) is one of the best examples of the dif- ference between the groups in the age of preference development. For the total 40 sec of exposure (top graph), the two curves are almost iden- tical in shape but are displaced about 2 weeks in age, indicating earlier development of a basic perceptual response by the selected home-reared group. For the first 4 sec of the first 20-sec exposure (bottom graph), the group difference was still greater: the home infants appeared to be able to discriminate the patterns within a few seconds as well as after long examination, whereas the institution group appeared to require more time for examination to show maximal differential response. The accentuated group difference for the first 4 sec of exposure is only sug- gestive; it was not duplicated for any other pair of targets. These and other less clear-cut results indicate that tracing changes in visual selec- tivity within short exposures, as well as over weeks of age, is a promis- ing approach to the problems of studying information processing and effects of visual exposure in the young infant. In the overall analysis of group differences, including the results given in Figures 8 and 9, the home group of infants showed significantly earlier or greater changes in preference for eight pairs of targets taken separately and for all 18 pairs averaged together.13 The significance of changes in visual selectivity as indicators of rate of perceptual-cognitive development is suggested by this differentiation between groups of in- fants with high expectation of eventual differences in cognitive per- formances, due to either congenital or early environmental differences. This significance is also suggested by the scores on the Griffiths Mental Development Scale (modified for better appropriateness for institution infants) given at 20 weeks of age. These scores for the combined groups showed a correlation of 0.74 with the rate of preference development during earlier months of life. In contrast, the scores during the early months, measuring largely motor development and sensorimotor co- ordination, were not related to the preference changes shown during that period; in particular, the institution infants developed oculomotor coordination as early as the home infants. If early cognitive abilities are to be related to later intelligence, then "perceptual intelligence" appears 372
Visual Perception and Experience in Infancy to be a more appropriate description than "sensorimotor intelligence" for these early abilities. VISUAL EXPERIENCE A general finding of the visual-preference research has been that many of the basic skills and selectivities required for pattern perception are present at birth or within several months of age. Rather than arguing for a purely nativistic or maturational view of perceptual development, this finding instead emphasizes the role of experience by showing that visual exploration and information processing begin at birth and that ample opportunities for a wide variety of experiential effects are there- fore present long before the infant is able to learn about the environ- ment through manual or locomotor explorations. The main issues, then, are to what degree and in what way these opportunities are used. As in the case of studying visual capacities, the available approaches to studying experiential effects are limited by the slow motor develop- ment of the infant, with the additional problem of varying experience in acceptable but sufficiently controlled ways. Recently developed con- ditioning procedures (as described above) can be of value, provided that the experimenter goes beyond the initial shaping of a response and is able to determine the specific stimulus features that have been per- ceived and remembered. For example, in the experiments of Bower,1 the initial conditioning of head-turning to a particular object showed only retention of the response-reinforcement contingencies and dis- crimination between the presence and absence of an object. But the later discrimination tests with varied objects or situations showed that such features of the object as size, distance, shape, and orientation were perceived and remembered for a short time. The experiments described above on decreased attention to a spe- cific, repeatedly exposed pattern provide another approach for study- ing short-term memory, differing mainly in that the response tendency was initially present without conditioning and that it decreased with experience. This experience effect assumes added interest from the fact that the necessary conditions are no doubt frequently present in the everyday life of the infant and from the adaptive consequences that the infant spends more time looking at the relatively novel parts of the environment. 373
ROBERT L. FANTZ Long-term effects of experience are more difficult than short-term effects to study experimentally in human subjects. Changes in visual preferences over some weeks or months of age can indicate long-term effects if there is sufficient evidence for assigning an experiential cause. This was not possible for most of the changes described here, although experience may often have been important. In the area of depth percep- tion, however, several lines of evidence converge to show the importance of experience in the development of the preference for solid over flat objects found to develop around 2 months of age (Figure 7).10'13 In research with infant monkeys,9 it was found that subjects reared in darkness from birth until various ages required a period of living in a lighted environment for the development of preferences for solid over flat objects (comparable with those shown by human infants), but failed to develop consistent preferences if the deprivation period was over 6 weeks. In contrast, preferences for patterned over plain targets were shown initially without visual experience and then were lost with con- tinued deprivation. For varying experience in human infants, enrichment procedures must be substituted for visual deprivation, perhaps starting with infants who happen to be in a relatively stimulus-poor environment.29 In a re- cent unpublished study of institution-reared infants using this approach, we provided 10 weeks of extra patterned visual stimulation in the crib and nursery (including a merry-go-round of varied objects) for every other entering neonate. In the results from weekly preference tests, for each of three pairs of solid versus flat targets the enriched group showed more attention than the control group to the solid object at one or more testing weeks. Few other significant differences in preferences were shown by the two groups, thus suggesting a limited area of effect of the perceptual enrichment. In the same study, data were obtained on another variation in visual experienceâone occurring naturally as a result of various lengths of ges- tation (within the normal range). For infants of the same conceptional age (i.e., length of gestation plus age from birth), an infant born several weeks before term, for example, has had that much more opportunity to see the world and practice visual coordinations than the full-term in- fant; the question is whether at that developmental level the extra ex- perience is effective in some measurable way. The finding was that, for two pairs of solid versus flat targets, the development of a solidity pref- 374
Visual Perception and Experience in Infancy erence was in closer agreement among the infants when their ages from birth were used than when their ages from conception were used, sug- gesting a determining role of early experience, regardless of the matura- tional level. However, many of the changes in pattern preferences were in closer agreement among the groups when ages were figured from the estimated date of conception (based on mother's report of last men- struation), suggesting the importance of degree of neural maturation for those responses. It seems clear that both the quantity and the quality of visual ex- perience determine to a considerable degree the beginnings of depth perception in the early months of life. Methodologically, the various results given above have demonstrated several practical approaches through which specific long-term or short-term effects of visual expe- rience can be investigated, starting from birth. The simple procedure of comparing visual preferences or other discriminative responses at equal conceptional ages, as well as at equal postnatal ages, is particularly promising for distinguishing between experiential and maturational in- fluences in the early months of life. One might argue that the usual practice in behavioral studies of considering only the age from birth implicitly assumes that nothing happens in the last weeks of fetal de- velopment to affect neurologic or behavioral maturity. By several years of age, the proportionally small differences in length of gestation within the normal range might well be ignored; but at birth or at several months of age, this practice sometimes may leave the pivotal variable out of the developmental equation and on other occasions may, through assuming complete experiential determination, throw away the proof of some effects of experience. STA GES OF PER CEPTUAL DE VEL OPMENT In most of the research cited here, visual preferences have served as con- venient behavioral indicators of the early development of visual percep- tion and information processing. From another viewpoint, the visual selectivities themselves are part of the development process; they have considerable influence on what information is taken in for processing and for directing behavior. It is to be assumed, then, that in "normal" development the various changes in visual selectivities will tend to facil- 375
ROBERT L. FANTZ itate, rather than retard, the development process. And yet it is difficult even to guess at the adaptive value of all the changes in selectivity at various ages. It would make good sense if the infant, starting with the selection of stimuli of minimal informational content, would gradually select more complex, more detailed, more subtle, more varied, more novel, and more changing stimuli as his capacities developed and his optimal com- plexity level rose. It might also be expected that this process would cul- minate in the fine discrimination and absorbed attention to patterned stimulation necessary for reading and other difficult perceptual tasks. The findings in the early months in some respects agree with this hypo- thetical view: the infant, although starting well above the minimal com- plexity level, does begin to attend to more complex, subtle, detailed, and novel patterns. But in later months, the attention value of various patterns generally decreases, and attention turns more to solid objects, flashing lights, brightly colored objects, and moving targets (among the targets used to date). By the end of the first year, little interest is shown in flat patterns and little differential attention is shown for differences among such patternsâa development seemingly opposite to the expected direction. Although the accumulation of information by the developing child is a gradual and continuous process, the kinds of information received and the ways in which they are collected can change radically with age and experience. The young infant's absorption in patterns and differentia- tion of variations in patterning, reaching a peak in the third and fourth months of life, perhaps represents a stage in perceptual development in which the motto is "information for its own sake." In this stage, the in- fant spends vast amounts of time in seemingly useless visual explorations and examinations, guided by the selective tendencies of his maturational and experiential level. What is learned in this way is largely a mystery, but it is certain that it goes beyond the perfection of oculomotor skills and discriminative capacities and, most essentially, involves the accumu- lation or assimilation of information from visual patterning. Also, it is safe to assume that this stage is essential for normal progress toward stages of visual information processing. Interference with this stage is a likely factor in the retarding effects of environmental deprivation in infants and of complete deprivation of patterned stimulation in young animals. One closely following stage is that in which visual exploration is a ?7<5
Visual Perception and Experience in Infancy means, rather than an end; looking is a guide to oral, manual, and loco- motor explorations and manipulations of the environment through which very different kinds of information are accumulated or assim- ilated. For this purpose, the patterning of flat surfaces is of little value (unless accompanied by tactile qualities), whereas object solidity and form are highly relevant stimulus characteristics that were found to continue to be of high attention value throughout the first 6 months of life. This apparent development has also been shown to take place in in- fant monkeys,9 which in the early months of life without deprivation showed visual preferences for patterned over plain stimuli and for some patterns over others. Early in the second year of life, when given a se- ries of discrimination-training problems preceded by "free-choice" tests, the animals showed few selective responses to, and subsequently rarely learned to discriminate, painted variations in color, brightness, or pat- terning. However, targets differing tactually as well as visibly (i.e., dif- fering in form, texture, solidity, or size) tended to be differentiated in the free-choice tests; in discrimination training, these pairs all received more correct responses and were usually discriminated to criterion. The monkeys had learned in everyday life to pay attention to palpable, "real" object characteristics and to ignore differences between painted surfaces that were irrelevant for behavior. The variations in patterning or color of flat surfaces were still discriminable in the sense of being within the known capacities of the animals; that had been shown by early visual preferences. But they were not actually discriminated at this stage because they were not noticed. It may be asked whether the early selectivity for patterns and the differentiation among patterns has any adaptive significance, inasmuch as it is replaced by other selectivities some months later. Perhaps the early selectivity for patterns is simply the consequence of the selectivity of the nervous system: the high responsiveness of visual receptors and nerve cells, especially cortical cells, to spatial or temporal patterns and the low responsiveness to uniform stimulation may in the early months be directly translated into oculomotor responsiveness. This explanation is certainly plausible in general, and it could be related to some of the specific pattern selectivities shown by the infant. For example, the tendency to give more attention to regular, linear patterns than to var- ious other configurations during the early weeks of life could be a di- rect behavioral expression of the selective responsiveness of cells in the visual cortex to linear contours.21 Also, subsequent shifts in preference 377
ROBERT L. FANTZ to circular or irregular configurations could signify the dominance of higher levels of neural organization in the direction of attention, but this is only a vague conjecture, made in the absence of other explana- tions of the amazingly strong selectivity for some pattern configura- tions in terms of either adaptive value or experiential origins. However, the more general selectivity for patterns in the young infant can easily be considered to be adaptive in focusing attention on information-rich parts of the surroundings and thus facilitating the process of perceptual learning. Developmental shifts in preference to more complex patterns, to more subtle variations in patterning, and to novel patterns appears to be similarly adaptive. And even after the in- fant seems to lose all interest in patterns per se, the patterning produced by the form, texture, fine details, and contouring of objects, surfaces, and people must be attended to for adaptive responses17; it is to be pre- sumed that this is made possible in part by the early intensive examina- tion of simpler varieties of patterning. That an infant or a monkey ig- nores the patterns on stimulus cards does not mean that a pattern denoting a feelable texture, a movable object, or a walkable surface will be similarly ignored. Eventually, a further stage of development is reached in which patterning unrelated to spatial orientation or object manipulation is again a focus of attention. For monkeys, this stage may occur only after intensive discrimination trainingâtraining not of pattern-vision capacities but of pattern selectivitiesâto counteract the earlier learning to ignore such stimulus variations as irrelevant for everyday behavior. But in children, a similar change occurs, apparently without training or even encouragement. The young child develops an interest in picture books, photographs, representational drawings, abstract designs and scribbles, and eventually printed language. (This is not intended to deny the complexities of learning to read, but merely to suggest that the un- trained interest in patterns on paper may be one essential predisposing factor.) This is a new stage of information processing, rather than a re- version to the earlier visual exploration of patterns, for the child is now much more efficient and retentive in his explorations and gives meaning to much of the informational content of the patterns. Furthermore, the motor skills and interests developed in the preceding stage can have a facilitating effect, as in the scribbling and drawing of the child, as sug- gested by Gibson17: "But scribbling is not simply play,... it is an op- portunity for the educating of visual attention and for learning to per- 378
Visual Perception and Experience in Infancy ceive in new ways" (p. 230). Gibson has also cogently analyzed the perception of pictorial representations as distinct from and secondary to the perception of real objects and surfaces, the former being more difficult and developing later than the latter. (In this light it would not be surprising if the young infant failed to recognize a schematic or photographic representation of a human face, as suggested above.) How the perception of pictures and symbols on paper develops is not well understood. The research of Eleanor Gibson and associates16 on the dif- ferentiation and attention to distinctive features of letters or other graphemes has given some clues to this process and illustrated some of the direct experimental approaches being applied. The improvement in discriminative capacities is clearly part of this developmental process; but alterations and refinements in visual selectivity are also essential for the exercising and use of these capacities. Changes in visual selectivity may in fact be of more critical importance, in that the basic oculomotor co- ordinations, visual acuities, and pattern-discrimination capacities are present long before they are put to use in deciphering markings on paper. And if the new visual selectivities of the young child are neces- sary for facilitation of the normal development of pictorial perception and reading, then aberrations in the development of these selectivities could easily cause retarded development of these complex perceptual achievements; such a possibility is at least worth exploring. A conclusion of the recent research on infant perception, of perhaps more significance than the early presence of pattern-vision capacities, is the strong influence of selective attention to patterns in general and among various patterns during the earliest stage of perceptual develop- ment. The specific selectivities and the way they affect the development process change considerably, but the importance of their influence probably remains throughout all succeeding stages. Previously unpublished research findings reported here were supported by National Science Foundation research grant GB 1874 and U.S. Public Health Service grant HD 00314 from the National Institute of Child Health and Human Development. REFERENCES 1. Bower, T. G. R. The visual world of infants. Sci. Amer. 215(6):80-92, 1966. 2. Brazelton, T. B., M. L. Scholl, and J. S. Robey. Visual responses in the newborn. Pediatrics 37:284-290, 1966. 379
ROBERT L. FANTZ 3. Brennan, W. M., E. W. Ames, and R. W. Moore. Age differences in infants' atten- tion to patterns of different complexities. Science 151:355-356, 1966. 4. Caron, R. F., and A. J. Caron. The effects of repeated exposure and stimulus complexity on visual fixation in infants. Psychonomic Sci. 10:207-208, 1968. 5. Dayton, G. O., Jr., H. H. Jones, P. Aiu, R. A. Rawson, B. Steele, and M. Rose. Developmental study of coordinated eye movements in the human infant. I. Visual acuity in the newborn human: a study based on induced optokinetic nystagmus recorded by electro-oculography. II. An electro-oculographic study of the fixation reflex in the newborn. Arch. Ophthal. 71:865-870; 871-875, 1964. 6. Fantz, R. L. Pattern vision in newborn infants. Science 140:296-297, 1963. 7. Fantz, R. L. Pattern vision in young infants. Psychol. Rec. 8:43-47, 1958. 8. Fantz, R. L. Studying visual perception and the effects of visual exposure in early infancy. In D. Gelfand, Ed. Readings in Child Development and Behavior Modification. Belmont, Calif.: Brooks-Cole Publishing Co. (in press) 9. Fantz, R. L. Visual perception and experience in early infancy: a look at the hidden side of behavior development, pp. 181-224. In H. W. Stevenson, E. H. Hess, and H. L. Rheingold, Eds. Early Behavior: Comparative and Develop- mental Approaches. New York: John Wiley & Sons, 1967. 303 pp. 10. Fantz, R. L. Visual perception from birth as shown by pattern selectivity. Ann. N.Y. Acad. Sci. 118:793-814, 1965. 11. Fantz, R. L. Visual experience in infants: decreased attention to familiar pat- terns relative to novel ones. Science 146:668-670, 1964. 12. Fantz, R. L., and S. Nevis. Pattern preferences and perceptual-cognitive develop- ment in early infancy. Merrill-Palmer Quart. 13:77-108, 1967. 13. Fantz, R. L., and S. Nevis. The predictive value of changes in visual preferences in early infancy, pp. 351-413. In J. Hellmuth, Ed. The Exceptional Infant. Volume I. Seattle: Special Child Publications, 1967. 14. Fantz, R. L., J. M. Ordy, and M. S. Udelf. Maturation of pattern vision in infants during the first six months. J. Comp. Physiol. Psychol. 55:907-917, 1962. 15. Gesell, A. L., F. L. Ilg, and G. E. Bullis. Vision: Its Development in Infant and Child. New York: Paul B. Hoeber, 1949. 329 pp. 16. Gibson, E. J. Principles of Perceptual Learning and Development. New York: Appleton-Century-Crofts, Inc., 1969. 17. Gibson, J. J. The Senses Considered as Perceptual Systems. Boston: Houghton MifflinCo., 1966.335pp. 18. Haynes, H., B. L. White, and R. Held. Visual accommodation in human infants. Science 148:528-530, 1965. 19. Hershenson, M. Visual discrimination in the human newborn. J. Comp. Physiol. Psychol. 58:270-276, 1964. 20. Hershenson, M., H. Munsinger, and W. Kessen. Preference for shapes of inter-. mediate variability in the newborn human. Science 147:630-631, 1965. 21. Hubel, D. H., and T. N. Wiesel. Receptive fields, binocular interaction and functional architecture in the cat's visual cortex. J. Physiol. 160:106-154, 1962. 22. Karmel, B. Z. The effect of age, complexity, and amount of contour on pattern preferences in human infants. J. Exp. Child Psychol. 7:339-354, 1969. 23. Ling, B. C. A genetic study of sustained visual fixation and associated behavior in the human infant from birth to 6 months. J. Genet. Psychol. 61:227-277, 1942. 380
Visual Perception and Experience in Infancy 24. McCall, R. B., and J. Kagan. Attention in the infant: effects of complexity, con- tour, perimeter, and familiarity. Child Develop. 38:939-952, 1967. 25. McGraw, M. B. The Neuromuscular Maturation of the Human Infant. New York: Columbia University Press, 1943. 140 pp. 26. Miranda, S. B. Visual abilities and pattern preferences of premature infants and full-term neonates. (unpublished manuscript, 1969) 27. Stechler, G. Newborn attention as affected by medication during labor. Science 144:315-317, 1964. 28. von Senden, M. Space and Sight: The Perception of Space and Shape in the Congenitally Blind Before and After Operation. London: Methuen, 1960. 348 pp. 29. White, B. L., and R. Held. Plasticity of sensorimotor development in the human infant, pp. 60-70. In J. F. Rosenblith and W. Allinsworth, Eds. The Causes of Behavior, II: Readings in Child Development and Education Psychology. 2nd Ed. Boston: Allyn and Bacon, Inc., 1966. 608 pp. 30. Zuckerman, C. B., and I. Rock. A reappraisal of the roles of past experience and innate organizing processes in visual perception. Psychol. Bull. 54:269- 296, 1957. 381
LEWIS P. LIPSITT Pattern Perception and Information Seeking in Early Infancy This report concerns some of the research conducted at the Brown University Child Psychology Laboratories on infant learning. It may have some bearing on problems of reading disability, but in any event it is related to visual information processing and early infant experience, which I take to be the primary concerns of this conference. TECHNIQ UE OF STUD Y Fantz devised a most ingenious technique for determining what infants prefer to look at and, by inference, what they can see. His technique is so simple as to pose an embarrassment for the history of the child- development field; one would think that it might have been capitalized on before. Previously, people assessed the visual capacities of infants in terms of what they grasped with their hands. As you know, young in- fants do not grasp for very much, so that early investigators were in- clined to assume that newborn infants do not see anything. Now we know better. Newborn infants do indeed see, and they profit from, or appreciate, and process visual information in important waysâi.e., their memories are affected by what they look at. Fantz's experiments, as 552
Pattern Perception and Information Seeking in Early Infancy well as others, indicate that newborn infants fixate objects and patterns in their environment with their eyes, and they fixate some objects and patterns more often or for longer periods than others. One can tell from the choice of one pattern or figure over another, or from the in- fant's responses to different visual patterns, what he prefers to look at, or at least what he discriminates. A study done by Miranda (a former student of Fantz) has recently shown that even the premature infant can see patterned stimulation.2 Miranda's study shows that conceptional age, in addition to actual age (age since birth), is a determinant of visual selection. To demonstrate that, he compared 27 31/2-day-old, normal full-term babies with 27 pre- mature infants, testing the latter group at less than 38 weeks of concep- tional age. That is, the premature babies were tested, on the average, approximately 1 month before they would have been born as full-term infants. The stimuli were always presented in pairs, and Fantz's tech- nique (which apparently has high reliability) involves observing whether the infant's eyes go to the left or the right. He used all the necessary counterbalancing of left and right figures to preclude erroneous infer- ences from position preferences. PR EM A TURE INFANTS The easiest way to summarize the Miranda data, without distorting the essential results, is to say that 1 month before the expected date of birth, but 3 weeks after birth, the premature babies' pattern preferences were essentially consonant with the visual-fixation behavior displayed by the full-term newborn babies tested at a mean age of 3l/2 days. In general, both the premature and the full-term babies resolved grating stimuli of 1/2 in. and 1/4 in.; they fixated these patterns more frequently and for longer periods than they did a plain gray stimulus. Both the premature and the full-term infants, moreover, tended to prefer the less complex of paired stimuli, but more consistently so for the premature babies of lower conceptional age. FIRST YEAR OF LIFE Fantz reports, on the basis of longitudinal data, that there is a marked change in discriminative capacity and in preference with age. He is 383
LEWIS P. LIPSITT conducting a longitudinal study from the ages of 2 to 52 weeks. Fantz does not posit many explanations for the marked change in preferences that infants display over that period, but he does suggest the possibility that experience is a primary consideration in determining what the child attends to most. He talks, for example, about the attention and interest value of various stimuli, which, according to his findings, seem to change drastically over the first year of life. The general finding within this period is that a plain, unmoving, unpatterned surface or object of any color or brightness is low in attention value at all ages. Younger infants in particular seem to like patternsâi.e., they attend to them moreâand especially black-and-white patterns with well- demarcated boundaries. It is also possible to conclude from the longitudinal study that by the age of 30-60 days a circular figure, such as a bull's-eye, is preferred over a linear figure. That is the age range that Dr. Kagan has described within which some remarkable changes occur (see p. 324). The sche- matic face seems to be preferred at birth over a picture of a real face. The schematic face is fixated more than a photograph of a real face un- til 16 weeks of age, when the child comes to fixate more on the real-life likeness. By 20 weeks of age, the child prefers a schematic face over a jumbled face. A solid face (three-dimensional) is preferred with increas- ing age over two-dimensional figures, and markedly so by 8 weeks of age. By 6 weeks of age, an unfamiliar figure tends to be preferred over a repeatedly exposed figure. HABITUATION I hope that Dr. Kagan's presentation does not leave the impression that habituation and learning processes do not occur in the human infant until he is 30, 60, or 90 days old. A growing body of data indicates that newborn children are remarkable habituators to stimulation of many sorts. Some of the best data in the field are derived from olfactory- stimulus work, and much work has also been done with auditory stimulation. In our own laboratory, we have done a series of experiments in which we delivered to the newborn child such olfactory stimuli as anise oil. The newborn child does indeed respond with a startle-like reaction, and when stimulation by these odorants is repeated, the response de- 384
Pattern Perception and Information Seeking in Early Infancy clines in even as few as 10 trials. The newborn habituates quickly to olfactory stimuli, as well as to other stimuli that we administer in the laboratory. Figure 1 shows a visually aware and alert child being stimu- lated with an odorant on the end of a swab. The infant's response in- cludes respiratory disruption, heart-rate acceleration, and bodily move- ment measured on a stabilimeter. These infants, all within the first 4 days of life, are hooked up to a polygraph from which we record a num- ber of different behaviors. We are also doing habituation experiments with the sucking response. The babies suck an artificial nipple that con- tains a device permitting us to record their rate and intensity of sucking. Presentation of a tone or odorant interrupts a baby's sucking; succes- sive presentations of the same stimulus will produce a diminishing amount of interruption. Once the newborn child is habituated to a given odorant or tone, presentation of a different stimulus without violation of the temporal FIGURE 1 Awake and alert newborn being administered an odorant on cotton swab. 385
LEWIS P. LIPSITT sequencing of the stimuli will cause the response to recoverâi.e., dis- habituation will take place. The newborn can tell us which odorants he is sensitive to both by his initial reactions to stimuli and by his recovery behavior after habituation to stimuli. Thus, the infant "reports" to us when he can discriminate the difference between two stimuli. For ex- ample, if he habituates within 10 trials to the anise oil, and then we change to asafetida, his response will recover and he will thereby tell us that he noted the difference between asafetida and anise oil. (Strength differences of the odorants are controlled through counterbalancing.) In an elaboration of this procedure, we mixed odorants (A and B) and delivered the mixture to the infants. After habituation, we admin- istered A or B separately in the same diluent. In this situation, the child similarly dishabituates, telling us that he can discriminate between odor- ant A or B and the mixture of odorants A and B. We think that we are dealing here, not with a peripheral phenomenon, but rather with a central nervous system function, or sensory integra- tion. We think that the last type of study, in which we induce habitua- tion to mixtures and then administer single components of those mix- tures, brings us closer and closer to a learning process. REINFORCEMENT Kagan's inclination, I think, is toward the view that a child does not become a habituator or learner until perhaps 30-60 days of age. Studies of newborn children show that they learn; the best evidence comes from a series of studies in which Dr. Einar R. Siqueland and I have po- tentiated the sucking response through reinforcing circumstances. Stim- ulating a newborn child on one side of his mouth elicits an ipsilateral rooting reflex. Head-Turning I am getting into head-turning behavior now, which I think is close to eye-turning behavior, which in turn should be related to reading behavior. In Figure 2, it can be seen that the baby's eyes are open and that he has a rather alert appearance. Infants while sucking do not always go to sleep, as we so often read. We hear all about how pacification produces 386
Pattern Perception and Information Seeking in Early Infancy FIGURE 2 Newborn infant sucking on automatic nipple device that enables polygraphic recording of suction. Three wells surrounding the transducer enable administration of different nutrients or tastes. sleep, a general lessening in body tone, and so on, but if the child has recently slept and is now feeding, putting a nipple in his mouth will probably arouse him. It arouses him visually and it arouses him with respect to head-turning activity. Many mothers report that they are able to communicate with their newborn babies best while they are feeding them. The child looks up at its mother's eyes, and while suck- ing looks around at the corners of the room and at other interesting ob- jects in the environment. We ourselves have not studied the looking be- havior of the young child, but we have been observing other response processes, such as head-turning behavior, and I think that head-turning and eye-turning responses are innately related. The situation pictured in Figure 2, incidentally, is one in which we are able to record the child's sucking characteristics polygraphically. I might say as an aside that we also have the opportunity, using this automated technique,1 of feeding 387
LEWIS P. LIPSITT the infant as a consequence of certain types of behavior. We can rein- force him through wells that enable the introduction into his mouth of dextrose solutions or milk in controlled amounts, not only to compare the relative attractiveness of gustatory stimulation, but also to reinforce the child in learning studies. Conditioning In our conditioned-head-turning studies, we capitalize on a response that is in the newborn child's repertoire of congenital responses: the rooting reflex or the turning of the head to the stimulated side. We feed the child when he turns his head after we touch him on the face. The touch induces a head-turning response about 25% of the time. If we sound one tone, touch the baby on the left side of his face, and feed him for turning there, the head-turning response in that direction goes up from 25% to 75%. In the same child and on alternate trials, if we sound another tone and touch in the same place but do not feed him for turning his head (i.e., do not operantly reinforce the response), the frequency of the response will either remain the same or even go down to around 20%. We are thus able to establish discrimination in the new- born child on the basis of tones and touch to the cheeks, feeding differ- entially in the presence of the two tones. An elaboration of that technique involves switching tones A and B. If A was the previously reinforced tone, such that the child was fed for responding to tone A plus touch and has come to respond 75% of the time, we can make A the negative tone and B the positive tone, and we get flip-over behavior. The child who was previously responding more to A than to B (A having been reinforced) can now be made to respond more to B than to A simply through changing the reinforcement con- tingencies.4 Even the newborn child, not to mention the 2-week-old or 2-month-old child, is remarkably receptive, and his behavior changes markedly in consequence of the stimulation provided. In speaking to nurses with considerable experience with newborn babies, I have often been amazed, in view of their extensive opportu- nities to observe infants, that many do not believe that newborn babies can see. When we invite them into our laboratory and show them new- born babies' horizontal and vertical scanning of dangling objects, they are often overwhelmed and begin to wonder about their own visual perception. 388
Pattern Perception and Information Seeking in Early Infancy In addition to our newborn-infant laboratory at the Providence Lying-in Hospital, we have a research facility at St. Vincent's Home for Infants, an orphanage in Rhode Island. Figure 3 shows an apparatus for head-turning experiments at the orphanage. The head-turning is re- corded on a polygraph, so that it is possible to note its direction and ex- tent. The apparatus would be suitable for the study of visual exploration in infants, although we have not used it that way. When we went to work in the infant home, we discovered that the nuns had suspended mobiles over the cribs of infants as young as 5 days of age. That was not part of our experiment; it just happened to be what was (and is) done in that particular institution. The personnel were aware (before we arrived on the scene) of the literature relating to effects of stimulus deprivation upon the development of personality and other psychologic attributes. r i I FIGURE 3 Infant attached to apparatus for recording head-turning. The plastic yoke is attached to a rotary potentiometer allowing polygraphic recording of incidence and magnitude of head-turning activity. 389
LEWIS P. LIPSITT Conjugate Reinforcing with Mobiles Stimulated by the presence of the mobiles there and by some sugges- tions of J. McV. Hunt5 concerning their attractiveness for infants, we did some studies with mobiles. There is a vast amount of literature on operant conditioning in animals and children. Most of the work has used what I would call pellet reinforcementâthe delivery of something that is discrete and can be picked up by the child or popped into his mouth, such as a small piece of candy. But concentration on that type of reinforcement reflects a restricted view of what reinforcement is like in real life. Picture, for example, what goes on between a mother and a nursing child. Whatever reinforcement occurs is not of the pellet variety. When the child is sucking at the breast or bottle, the harder he sucks, the more food he gets; reinforcement is commensurate with the fre- quency and intensity of the sucking. I submit that visual reinforcement in real life is more like that, and not so much like pellet reinforcement. We implemented research involving visual reinforcement of the hanging- mobile type, in which the child controls his own visual input from his surroundings. Now, most parents and nurseries simply suspend the mo- biles above the crib. What would happen if, instead of suspending the visual stimulation before the child, we enabled the child to control his own input? That is really what we all do when we turn our heads and move our eyes. We control our own visual input, presumably in some sort of rational manner having to do with the curiosity or interest value of the stimulus. Carolyn Kent Rovee, a former student in the Brown University Child Laboratories, implemented a study with her own 2-month-old child and with five other children of similar age, using a type of reinforcement such as that just described.3 It is called "conjugate reinforcement"; I think Ogden Lindsley devised the term to signify situations in which the subject is in direct control of the reinforcer, the reinforcer occur- ring immediately and being commensurate in intensity with the activity of the subject. Pressing on an accelerator pedal would be an example of this sort of interaction between a subject and his environment; turning a spigot to obtain water, in greater amounts with increasing turns of the spigot, would be another example. Two observers recorded the re- sponses. They obtained baseline measurements of activity of, for ex- ample, the infants' right arms; if an arm moved a specified minimal distance, that would be called a response. Each baby's right arm was 390
Pattern Perception and Information Seeking in Early Infancy o Q. Q£ o O 30 25 20 15 10 13 20 23 27 OPERANT LEVEL ACQUISITION MINUTES EXTINCTION FIGURE 4 Mean rate of response as a function of conjugate reinforcement condition in 18 infants 9-12 weeks old; leg attached to mobile during acquisition. Each point represents a suc- cessive minute of observation. Each curve represents six subjects. Modified from Rovee and Rovee.3 then tied to the mobile hanging above the crib, so that when the baby moved its arm, the mobile moved. Figure 4 shows some data from Dr. Rovee's experiment. First, an operant level may be seen, consisting simply of a 3-min period, during which she recorded the number of responses from each of the six chil- dren while they were in the presence of the mobile but not operating it. This was followed by a 15-min conjugate reinforcement phase. The effect of this reinforcement on the babies' activity levels was highly significant. Dr. Rovee later introduced an extinction period by detach- ing the cords from the babies' limbs. During extinction, there was a diminution of behavior, and the children gradually became uninterested, or at least less interested in the mobile than they had been before. The figure shows a cumulative recording of a full minute of the limb activity of a child; the child has had a minute to become higher in level of re- 391
LEWIS P. LIPSITT sponse to the manipulable mobile than during baseline measurement, when responses did not affect movement of the mobile. By the time the first minute of the conditioning phase is over, the infant has already had an opportunity to learn that his actions control the mobile. However, there is not a reliable difference between the last point in baseline and the first point in conditioning. The increase in activity between baseline and conditioning over minutes of recording is highly significant. The infant is learning to con- trol the stimulus. Then, in extinction, he learns that he no longer con- trols it. There is actually a very fast dropoff in activity when the baby's behavior no longer has anything to do with the control of the visual stimulation. This is also a reliable difference between the conditioning and the extinction phases. Figure 5 deals with a possible objection to a learning interpretation of the effects shown in the previous figure. It might be argued, for example, that the effect obtained is only appar- ently a learning phenomenon and that it is due to a baby's becoming 50r 40 o CL. et d Of LU 30 20 10 V 136 20 23 27 30 40 42 46 OPERANT LEVEL ACQUISITION EXTINCTION MINUTES RE-ACQ. RE-EXT. FIGURE 5 Mean response rate as a function of conjugate reinforcement condition over 46 min of continuous observation in four infants from previous group; leg attached to mobile during acquisition. Modified from Rovee and Rovee.3 592
Pattern Perception and Information Seeking in Early Infancy CONJUGATE REINFORCEMENT ENTIRE BIRD MOBILE S 65 DAYS OLD: SESSION I NOV. 14, 1967 to IS i CC CO U 6 CC BASELINE CONDITIONINS EXTINCTION RECONDITIONING EXTINCTION SUCCESSIVE 30 SECOND INTERVALS FIGURE 6 Unpublished data of Smith and Lipsitt showing operant responding of infant in conjugate mobile situation. During baseline, infant's right arm was not in mechanical contact with mobile. During conditioning phase, the cord was attached between the baby's arm and the mobile, following which the cord was again detached for an extinction session. Note that the onset of crying elevated responses in the second extinction session. increasingly alert, with exposure, to the mobile (and perhaps to the experimenter), and that the eventual diminution of behavior during extinction is really a fatigue effect. But Rovee was able to keep four of the six subjects in a study of reacquisition after the extinction phase. There was recovery of the conjugate reinforcement effect, and if the subject's limb was released from control of the mobile, his activity then declined. That the infants could be shown to intensify their behavior when in control of their own stimulus input and to reduce their behavior when the response was no longer pertinent to visual input indicates that lively visual stimulation may serve as a reinforcer to enhance learning 393
LEWIS P. LIPSITT behavior. It might also be speculated, conversely, that the infant's op- portunity to control his own visual environment enhances the attrac- tiveness of that stimulation. Following Dr. Rovee's study, I was fortunate to have another stu- dent, Mrs. Leslie Smith, who decided to follow her own baby over a longer period in this type of mobile reinforcement situation. Her data relate to the behavior of this one child successively tested over several months and with different mobiles. The responses of this baby, first seen at 6 weeks of age, may be seen in Figures 6 and 7 in the baseline condition. Following this, Mrs. Smith attached the child's arm or leg to the mobile in a manner similar to the procedure used by Dr. Rovee. Mrs. Smith, however, introduced an automated procedure for counting responses; the lead to the counter is attached to the baby's limb along with the mobile, so that a minimal movement would activate the re- CONJUGATE REINFORCEMENT ENTIRE BIRO MOBILE 5 67 DAYS OLD: SESSION 3 NOV. I6, 1967 BASELINE CONDITIONING EXTINCTION RECONDITIONING EXTINCTION RECONDITIONING EXTINCTION SUCCESSIVE 30 SECOND INTERVALS FIGURE 7 Unpublished data of Smith and Lipsitt. In third training session, infant's right leg responses activated the mobile during conditioning periods. Response is elevated during conditioning, in contrast with performance during baseline and extinction periods. 394
Pattern Perception and Information Seeking in Early Infancy corder. It may be seen that when the mobile was controlled by the child there was an increase in response, then a decline under extinction con- ditions, then an increase and decline again, and so on. The conjugate re- inforcement phenomenon obviously occurs even at 6 weeks of age (rather, it can occur; there are undoubtedly individual differences about which we know very little), and it can be a striking phenomenon. Note that in this extinction phase the baby began crying, following training in control of the stimulus, when his control of it was removed. This cry- ing phenomenon occurred frequently in this child during extinction phases and Mrs. Smith responded, as would most mothers, by making the mobile's movement once again available to the child, whereupon crying stopped. This phenomenon may well be akin to or a precursor of what is called "separation anxiety" in older children. When Mrs. Smith changed the mobile, she found that introduction of a novel mo- bile enhanced the response of the child. On the basis of data obtained later, it seems to us that this was not merely an effect of novelty, which was systematically checked out, but was a true preference of a red mo- bile over a white one (Figure 8). Again, response level went down sharply during extinction, although the child was still in the presence of the red mobile. Conjugate Reinforcing with Sucking We are also seeing infants at various ages in the Hunter Laboratory for conjugate reinforcement experiments devised and conducted by Pro- fessor Siqueland. In these experiments, the child controls his visual in- put through his sucking behavior. The visual stimulation is presented on the screen (Figure 9). The more frequently and more intensively the baby sucks, the more visual stimulation he gets, because the projector is equipped to provide greater illumination with greater intensity or fre- quency of sucking. It can be set in such a way that the child's visual stimulation will be proportional to both the rate and the intensity of sucking. One can use a psychophysical-threshold procedure, whereby the child has to suck with a certain intensity or the projector is not triggered. In such instances, one can operantly reinforce only high- intensity sucks, and not low-intensity sucks. From the behavior of con- trol subjects in this situation, it appears that high-amplitude sucking will wane over time if it is not reinforced. Thus, the control group shows a decrement in sucking behavior relative to the group whose be- 395
LEWIS P. LIPSITT havior produces increased visual stimulation. The parallel between this technique and the conjugate mobile technique is perhaps obvious. Figure 10 presents data from three groups of 10 4-month-old chil- dren in one of Dr. Siqueland's studies. One group had its sucking be- havior reinforced with light onset and light intensification. Another group was a baseline group, which received no visual stimulation for sucking; the babies had pacifiers in their mouths and their sucking be- havior was recorded, but the sucking did not control the visual feed- back. A third group, represented by the dashed line that begins at the bottom, got reinforcement withdrawal with increases in sucking; that is, this group started with the visual stimulus already full on, and suc- cessive or repetitive sucking on the device produced withdrawal or CONJUGATE REINFORCEMENT RED AND WHITE BIRDS 5 70 DAYS OLD' SESSION 6 NOV. 19, I9«7 9 M 20 CONDITIONING W RED BIRD CONDITIONING W WHITE BIRD EXTINCTION RECONDITIONING W RECONDITIONING * RED BIRD WHITE BIRD SUCCESSIVE 30 SECOND INTERVALS FIGURE 8 Unpublished data of Smith and Lipsitt. Infant's responses to two mobiles, one red and one white, are compared in successive sessions. Response levels tend to be higher during presentations of the red stimulus than the white stimulus, and both tend to be higher than performance during extinction conditions. 396
Pattern Perception and Information Seeking in Early Infancy FIGURE 9 Infant operating sucking manipulandum. High-intensity sucks increase illumination of slides shown on screen in front of subject. (After E. R. Siqueland.) diminution of illumination of the visual stimulus commensurate with the high-amplitude sucking. These children, 4 months old, were being operantly reinforced for their sucking behavior, and the behavior was controlled by the "nov- elty" and attractiveness of the stimulus. This technique should enable us to increase our understanding, as did Fantz's innovations, of atten- tional processes in the infant and the preferences that infants have for different stimulation at different ages. IMPLICA TIONS IN PR EM A TURITY We will be able to follow some of these children when they become readers and perhaps see how some of our intervention procedures do work. For example, we have a study in our Child Study Center with 397
LEWIS P. LIPSITT premature children, who are fortuitously subjected to a relatively im- poverished environment, sometimes for as long as the first two months of their lives. The natural state of affairs in the ordinary premature nursery of most hospitals is such that the infants exist in what may be a rather deprived setting compared with the experiences that normal CO ft Ul cc 60 SO 40 30 20 10 oâ SUCKING REINF. 6RP STIMULUS WITHDRAWAL GRP BASELINE GRP BASAL 10 II 12 13 14 15 COND1 COND' EXTINCT2 ONE MINUTE INTERVALS FIGURE 10 Unpublished data from experiment of Siqueland involving three groups of 10 4-month-old infants in whom sucking activity produced different visual consequences. One group received visual reinforcement; the sucking behavior of a second group was associated with stimulus diminution; and a third group served as a control, with no visual changes oc- curring during the recording of sucking behavior. 398
Pattern Perception and Information Seeking in Early Infancy full-term newborn infants have outside an incubator, in contact with adults. There are considerable data available to indicate that the pre- mature child is in developmental jeopardy for some time after he is dis- charged from the premature nursery and is growing up normally. Avail- able documentation indicates that premature children as a group tend to have lower intelligence, be poorer readers, have more difficulty in school, and so on. Statistically speaking, the premature child has handi- caps that seem to persist, and one has to wonder why. Some biologic explanations might be invoked: the viability of the organism was in jeopardy from the start, it suffered respiratory distress, and it did not have the full complement of fetal nurture normally received by the full- term infant. But one has to entertain an alternative hypothesis, namely that part of the deficit results from the understimulation that is made necessary for the first few weeks or months of postnatal life by the bio- logic prematurity. Dr. Siqueland is attempting to provide compensatory stimulus enrichment to premature babies through various means. The children are reinforced for looking, and they get picked up, rocked, and spoken to by motherly nurses; in this way, they are provided with extra visual and other stimulation of a wide variety. It is the intent to follow up these infants for a long period to find out what effects occur over the long run, in contrast with a control group that receives the routine premature care. On follow-up, one of the situations to which the infants are subjected is the sucking visual reinforcement previously described. Thus far, a number of children have come back at 4 months of chronologic age, some of them having been out of the hospital for only a couple of months; among these infants are several sets of twins. When the stim- ulated twins are now tested in the controlled learning situation, those who have had such training seem on the basis of preliminary findings to learn faster in this conjugate reinforcement situation. The stimulated and nonstimulated premature babies are tested "blind" by an assistant who does not know whether they are stimulated or control subjects. First indications are that at least some learning effects are facilitated by early stimulation of the premature children. RELEVANCE TO READING Let me try to draw some conclusions in an attempt to make these data and this program of studies relevant to the concerns of the present con- 399
LEWIS P. LIPSITT ference. It should be apparent that very young infants do indeed learn. It should also be obvious from our conjugate reinforcement data that infants are captivated by visual stimulation, particularly if it is novel. It is important to note, however, that some of our most striking learning effects in infants appear in situations in which the baby himself is acting on the environment to change his visual input. What does all this mean for reading disability? First, the human being is very responsive to stimulation, including visual stimulation, from birth onward, and techniques are now available for extensive and longi- tudinal study of the ways in which newborn and older infants are af- fected by their visual surroundings. Second, it is now inappropriate for us (educators and psychologists alike) to think in terms of merely waiting for maturation of some behavioral processes. We now know that the failure of some kinds of behavior to occur is experientially induced, and it appears that the overemphasis on maturational processes as pri- mary determinants of development has, to some extent, precluded or inhibited our full exploration of the real learning potential of children at different ages. Too often we are inclined to say, when a child does not seem to be learning to read well, that he is not mature enough, that he is not "ready" for reading. That position is often just an escapeâa poor excuse for our lack of competence in devising appropriate tech- niques for producing the desired behavior. We child developmentalists and educators cannot retreat much longer into the maturation argument to account for our failures to implement appropriate techniques to facil- itate education in children of diverse experience. We saw from the presentation of Dr. Flom (p. 291) that by 3 years of age at most, and I would guess by 1 or P/z years (if not before), the child has the appropriate visual-acuity equipment to do a lot of things that were at one time thought impossible for the very young. The argu- ment that 4-year-olds, for example, lack the appropriate visual appara- tus to read has obviously been refuted, but that is exactly the kind of "reading-readiness" argument that has been offered in the past to for- bid earlier exploration of children's reading potential. I do not advocate teaching 1-year-olds to read, and in fact I do not know whether it is pos- sible. Moreover, I have no idea as to whether it would be good to teach a 1-year-old or even a 3-year-old to read; that is quite another issue. But because the early indications are that it is possible to do so, we can no longer resort to the often-voiced argument that it is better to wait for maturation of "reading readiness" to evolve. From the scientific point 400
Pattern Perception and Information Seeking in Early Infancy of view, moreover, the learning components of "reading readiness" should be given further study. Certainly, perceptual pretraining and transfer of the effects of such training to the reading situation consti- tute important areas for exploration. Pretraining with visual stimulation of the sort that I have described here, perhaps along with coordinated auditory stimulation such as occurs in real life, must be exceedingly im- portant in the development of fascination with reading materials on the part of children who read well and who like to read. It is my belief that children who read well and who read a lot find some sort of joy in readingâjoy in controlling their own environmentâ very much as the children in the conjugate reinforcement situation seem to enjoy manipulating their own visual input. Controlling one's own vi- sual environment (and hence perhaps one's visual fantasies and imagery) must be one of the reinforcing characteristics involved in the persistence of reading behavior. If a child is going to enjoy reading, or enjoy the "ingredients" or precursors of readingâsuch as looking at books around his home, looking at the printed word, and looking at picturesâhe has to be reinforced. That is, for curiosity behavior to be perpetuated, it seems to me that some sort of reinforcement process is involved in look- ing at things in the environment, the kinds of things that reading teach- ers get children to observe before they are presented with the printed word. My pointâand perhaps it is far-fetchedâis that the kinds of processes we are dealing with in this research on infants capitalize on the curiosi- ties of the child with respect to visual stimulation and reinforce the child for looking, or for being curious. I think that this is the sort of thing that occurs in the natural process of growing up and that rein- forcement for looking constitutes a precursor for reading behavior. For example, if you punish a child every time he looks at a printed page, he will soon not have much joy in looking at the printed page, and that is probably what some teachers actually do to children during the early phases of reading training. We should note that early readers are good readers. It is possible that both attributes are simply correlates of intelligence and that intelligence is really the primary cause of both attributes; but I think that it is rea- sonable to consider the possibility that early visual and other stimula- tion is an important determinant of all threeâearly reading, proficient reading, and intelligence. Finally, I think that reading disorders call for developmental investi- 401
LEWIS P. LIPSITT gations of learning processes and learning disorders. The antecedents of reading disabilities must have a good deal in common with other developmental and learning attributes. We will know more about de- velopmental processes generally when the learning of reading is better understood, and we will know more about reading aberrations when developmental and learning processes are better understood. Similarly, learning disorders call for the discovery of new types of remedial tech- niques involving learning principles. Even children who have true brain dysfunction have to learn and do learn new modes of behavior. A diag- nosis of brain dysfunction does not preclude the beneficial administra- tion of compensatory training. It is certainly inappropriate to assume that, if a child has central nervous system anomalies, there is nothing for the learning psychologist or the educator to do about it, from either the scientific or the remedial standpoint. Appropriate reading tech- niques are necessary for the dyslexic child no less than for the neuro- logically intact child. REFERENCES 1. DeLucia, C. A. A system for response measurement and reinforcement delivery for infant sucking-behavior research. J. Exp. Child Psychol. 5:518-521, 1967. 2. Miranda, S. B. Visual abilities and pattern preferences of premature infants and full-term neonates. (unpublished manuscript, 1969) 3. Rovee, C. K., and D. T. Rovee. Conjugate reinforcement of infant exploratory behavior. J. Exp. Child Psychol. 8:33-39, 1969. 4. Siqueland, E. R., and L. P. Lipsitt. Conditioned head-turning in human new- borns. J. Exp. Child Psychol. 3:356-376, 1966. 5. Uzgiris, I., and J. McV. Hunt. A longitudinal study of recognition learning. Paper read at meeting of Society for Research in Child Development, Minnea- polis, Minnesota, 1965. 402
THE ROLE OF INFORMATION PROCESSING IN PERCEPTUAL AND READING DISABILITIES
