Nanoparticle Drug Delivery and Diagnostics in Health

Imagine a medical patient suffering from cystic fibrosis, a disease caused by a faulty gene in the patient’s cells, or from cancer, a multigene disease. Then imagine the patient being cured by a new class of treatment, gene therapy, which replaces or supplements the patient’s own faulty or diseased genes with new therapeutic nucleic acids, DNA or RNA. These nucleic acids may be encapsulated with carrier molecules such as lipids (fat molecules), which protect them from destruction by the patient’s own immune system. The carrier lipids are designed to deliver their cargo specifically to the patient’s lungs or to a tumor, allowing for small dosages and reducing harmful side effects.
The delivery of drugs where they are needed is not an empty dream; more than 1,800 human clinical trials are ongoing, with over 110 of them employing lipid carriers.12Many of these clinical trials, including those for treating cystic fibrosis as well as cancer, have been aided by the recent discoveries of condensed matter and materials researchers at the University of California, Santa Barbara. These researchers, funded by the National Science Foundation and the National Institutes of Health and using synchrotron facilities funded by the Department of Energy, have uncovered the internal structures of lipid-nucleic acid nanoparticles (NPs)—that is, the arrangement of the lipid and nucleic acid molecules inside and on the surface of the NP. This new knowledge promises to dramatically raise the success rates of clinical trials because it gives scientists crucial insights into how to design new variations of the lipids that are more efficient at carrying the NP to the diseased cells of interest (which depends on lipids with a special chemical structure being present at the surface of the NP) and at releasing the therapeutic nucleic acid cargo molecules upon cell entry (which depends on the lipid and nucleic acid arrangement inside the NP).
Much of the development of new nucleic-acid-based gene therapies is occurring in small start-up companies, whose technology may be traced to research concepts realized at universities and national laboratories in the course of research supported by federal funding agencies. For example, DNA-chewing enzymes in the blood normally destroy DNA, but materials researchers found that the DNA is protected when grafted to gold nanoparticles.13 DNA-functionalized gold nanoparticles provide an alternative route to gene therapy as well as high sensitivity to new protein markers for bladder, kidney, prostate, and other cancers. Small businesses such as Nanosphere, based in Illinois, are developing these new technologies for medical applications.
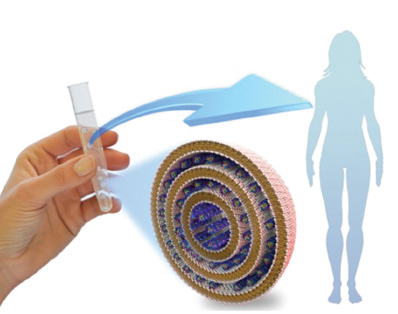
Ongoing clinical trials aim to efficiently deliver therapeutic DNA or RNA molecules for treating a wide range of human diseases. In the figure a human hand is shown holding a vial containing a liquid suspension of lipid-nucleic acid (DNA or RNA) nanoparticles intended for delivery through direct injection into tissue or intravenously or orally. The blow-up of one such lipid-nucleic acid nanoparticle (with a diameter between 100 and 200 nanometers) reveals the internal onion-like structure consisting of lipid bilayers (mustard tails attached to white spheres forming the onion layers) wrapped around therapeutic nucleic acids (DNA or RNA, helically shaped purple molecules). The multi-layered onion structure significantly increases the therapeutic loading capacity (the number of nucleic acids) per particle, thus improving the therapeutic effectiveness of each particle entering a diseased cell. SOURCES: Rädler, J.O., I. Koltover, T. Salditt, and C.R. Safinya, 1997, Structure of DNA–cationic liposome complexes: DNA intercalation in multilamellar membranes in distinct interhelical packing regimes, Science 275: 810-814; Ewert, K.K., A. Zidovska, A. Ahmad, N.F. Bouxsein, et al., 2010, Cationic liposome–nucleic acid complexes for gene delivery and silencing: Pathways and mechanisms for plasmid DNA and siRNA, Topics in Current Chemistry 296: 191-226.
This technology could truly level the field, bringing modern, state-of-the-art diagnostics to every doctor’s office, even in the poorest countries.
To achieve their full potential, gene therapy and DNA-based disease diagnostics face a huge challenge—to deliver nucleic acids to specific diseased cells within the body. Reaching the holy grail of therapeutic efficacy through precise targeting will require continued sustained research efforts by multidisciplinary teams of researchers, including condensed matter and materials scientists.

Lab-on-a-chip devices such as the integrated fluidic circuit (IFC) shown above can be considered the biological equivalent of the integrated circuit. These devices provide a simple, fast, and cheap system for streamlining genetic research. SOURCE: Courtesy of Fluidigm Corporation.
During the same time that these new targeted delivery systems were being developed, there has been a veritable revolution in disease diagnostics that mirrors the revolution in microelectronics. This revolution exploits microfluidic technologies to develop lab-on-a-chip devices that provide miniature and much simpler diagnostic systems for point-of-care deployment. Condensed matter and materials research has been instrumental in the development of these new technologies. For example, a California company, Fluidigm, founded by a Stanford professor, is developing lab-on-a-chip diagnostics based on soft lithography. Even more easily distributable diagnostic devices are being developed by a Harvard start-up, Diagnostics for All; these devices are printed directly on paper, making their cost very low and thus affordable throughout the world.14 The rapid ongoing development of this new technology is expected to lead to a suite of patterned paper devices able to assay for a range of organ functions, as evidenced by the recent introduction of such devices for cost-efficient liver function assays.15 This technology could truly level the field, bringing modern, state-of-the-art diagnostics to every doctor’s office, even in the poorest countries.




