Below is the uncorrected machine-read text of this chapter, intended to provide our own search engines and external engines with highly rich, chapter-representative searchable text of each book. Because it is UNCORRECTED material, please consider the following text as a useful but insufficient proxy for the authoritative book pages.
8 Hydrogen Bromide1 Acute Exposure Guideline Levels PREFACE Under the authority of the Federal Advisory Committee Act (FACA) P.L. 92-463 of 1972, the National Advisory Committee for Acute Exposure Guide- line Levels for Hazardous Substances (NAC/AEGL Committee) has been estab- lished to identify, review, and interpret relevant toxicologic and other scientific data and develop AEGLs for high-priority, acutely toxic chemicals. AEGLs represent threshold exposure limits for the general public and are applicable to emergency exposure periods ranging from 10 minutes (min) to 8 hours (h). Three levelsâAEGL-1, AEGL-2, and AEGL-3âare developed for each of five exposure periods (10 and 30 min and 1, 4, and 8 h) and are distin- guished by varying degrees of severity of toxic effects. The three AEGLs are defined as follows: AEGL-1 is the airborne concentration (expressed as parts per million or milligrams per cubic meter [ppm or mg/m3]) of a substance above which it is predicted that the general population, including susceptible individuals, could experience notable discomfort, irritation, or certain asymptomatic, nonsensory effects. However, the effects are not disabling and are transient and reversible upon cessation of exposure. 1 This document was prepared by the AEGL Development Team composed of Sylvia Talmage (Oak Ridge National Laboratory), Lisa Ingerman (SRC, Inc.), Chemical Man- ager Susan Ripple (National Advisory Committee [NAC] on Acute Exposure Guideline Levels for Hazardous Substances), and Ernest V. Falke (U.S. Environmental Protection Agency). The NAC reviewed and revised the document and AEGLs as deemed neces- sary. Both the document and the AEGL values were then reviewed by the National Re- search Council (NRC) Committee on Acute Exposure Guideline Levels. The NRC com- mittee has concluded that the AEGLs developed in this document are scientifically valid conclusions based on the data reviewed by the NRC and are consistent with the NRC guidelines reports (NRC 1993, 2001). 429
430 Acute Exposure Guideline Levels AEGL-2 is the airborne concentration (expressed as ppm or mg/m3) of a substance above which it is predicted that the general population, including sus- ceptible individuals, could experience irreversible or other serious, long-lasting adverse health effects or an impaired ability to escape. AEGL-3 is the airborne concentration (expressed as ppm or mg/m3) of a substance above which it is predicted that the general population, including sus- ceptible individuals, could experience life-threatening health effects or death. Airborne concentrations below the AEGL-1 represent exposure concentra- tions that could produce mild and progressively increasing but transient and nondisabling odor, taste, and sensory irritation or certain asymptomatic, nonsen- sory effects. With increasing airborne concentrations above each AEGL, there is a progressive increase in the likelihood of occurrence and the severity of effects described for each corresponding AEGL. Although the AEGL values represent threshold concentrations for the general public, including susceptible subpopula- tions, such as infants, children, the elderly, persons with asthma, and those with other illnesses, it is recognized that individuals, subject to idiosyncratic respons- es, could experience the effects described at concentrations below the corre- sponding AEGL. SUMMARY Hydrogen bromide (HBr) is a colorless, corrosive, and non-flammable gas. HBr fumes strongly in moist air. It is one of the strongest mineral acids, with a reducing action stronger than that of hydrogen chloride (HCl). It is extremely soluble in water, forming a strong acid that is available as 48% or 68% solu- tions. HBr is used both as a reagent and as a catalyst in a variety of organic reac- tions; it is also used in the preparation of numerous bromide compounds. Anhy- drous HBr is shipped in high-pressure steel cylinders. HBr is a severe irritant to the eyes, skin, and nasal passages; high concen- trations may penetrate to the lungs resulting in edema and hemorrhage. Data on irritant effects in humans and lethal and sublethal effects in rats and mice were available for developing AEGL values. Although the database for HBr is sparse, data on the toxicity of HBr relative to that of hydrogen fluoride (HF) and HCl were available for comparison purposes. The databases for HCl and HF are ro- bust. On the basis of lethality data from studies of rats and mice, HF is more potent than HCl and HBr; HCl and HBr have similar potencies (MacEwen and Vernot 1972). At sublethal concentrations, the severity and extent of lesions to the upper respiratory tract were greatest for HF, followed by HCl and then HBr, although the severity and extent of lesions in the anterior most region of the res- piratory tract were similar among the three chemicals (Kusewitt et al. 1989; Stavert et al. 1991). The data also show that all three chemicals are well scrubbed in the upper respiratory passages.
Hydrogen Bromide 431 The AEGL-1 values for HBr are based on a study of six human volunteers exposed at 2, 3, 4, 5, or 6 ppm for several minutes (CT Department of Health, unpublished data, 1955, as cited in ACGIH 2002). No nasal, throat, or ocular irritation was reported at 2 ppm. One subject reported nasal and throat irritation (severity not defined) but no ocular irritation at 3 ppm. Nasal irritation was re- ported by all six subjects at 5 and 6 ppm, but only one reported throat irritation and none reported ocular irritation. The concentration of 3 ppm was considered a no-observed-adverse-effect level (NOAEL) for notable discomfort. This point of departure was divided by an uncertainty factor of 3 to protect sensitive indi- viduals; time-scaling was not performed, because irritation is concentration re- lated and humans adapt to the slight sensory irritation that defines the AEGL-1. A concentration of 1.0 ppm across the AEGL exposure durations is supported by the AEGL-1 values for HF and HCl of 1.0 and 1.8 ppm, respectively (NRC 2004). The AEGL-1 value might be conservative, as only one of six subjects reported any sensory irritation and the value is the same as that of HF, a slightly more toxic chemical. It is also below the AEGL-1 value of 1.8 ppm for HCl, which was based on a no-effect concentration in exercising asthmatics. There are limited data on AEGL-2 effects from exposure to HBr. Stavert et al. (1991) reported severe necrohemorrhagic rhinitis in rats exposed to HBr at 1,300 ppm for 30 min; however, 8% mortality was also reported at that concen- tration. In the absence of suitable data, the AEGL-3 values for HBr were divided by 3 to derive AEGL-2 values. A BMCL05 (benchmark concentration, 95% lower confidence limit with 5% response) of 1,239 ppm was calculated from 1-h lethality data from studies of Sprague-Dawley rats exposed to HBr (MacEwen and Vernot 1972). The BMCL05 is an estimate of the threshold for lethality, and was used as the point of departure for calculating AEGL-3 values for HBr. A total uncertainty factor of 10 was applied: 3 for interspecies differences and 3 for human variability. Those individual factors are considered sufficient because the action of a direct- acting irritant is not expected to vary greatly among species or between individ- uals (NRC 2001). The 60-min point of departure was time-scaled to the other AEGL durations using the equation Cn à t = k. The value of n was 1, on the ba- sis of data for the related compound HCl, for which regression analysis of com- bined rat and mouse LC50 (lethal concentration, 50% lethality) data resulted in a value of 1 for n (see NRC 2004). The AEGL values for HBr are presented in Table 8-1. 1. INTRODUCTION Hydrogen bromide (HBr) is a colorless nonflammable gas that fumes strongly in moist air. It is highly water soluble. HBr is one of the strongest min- eral acids, with a reducing action stronger than that of hydrogen chloride (HCl) (Jackisch 1992). Chemical and physical properties for HBr are presented in Ta- ble 8-2.
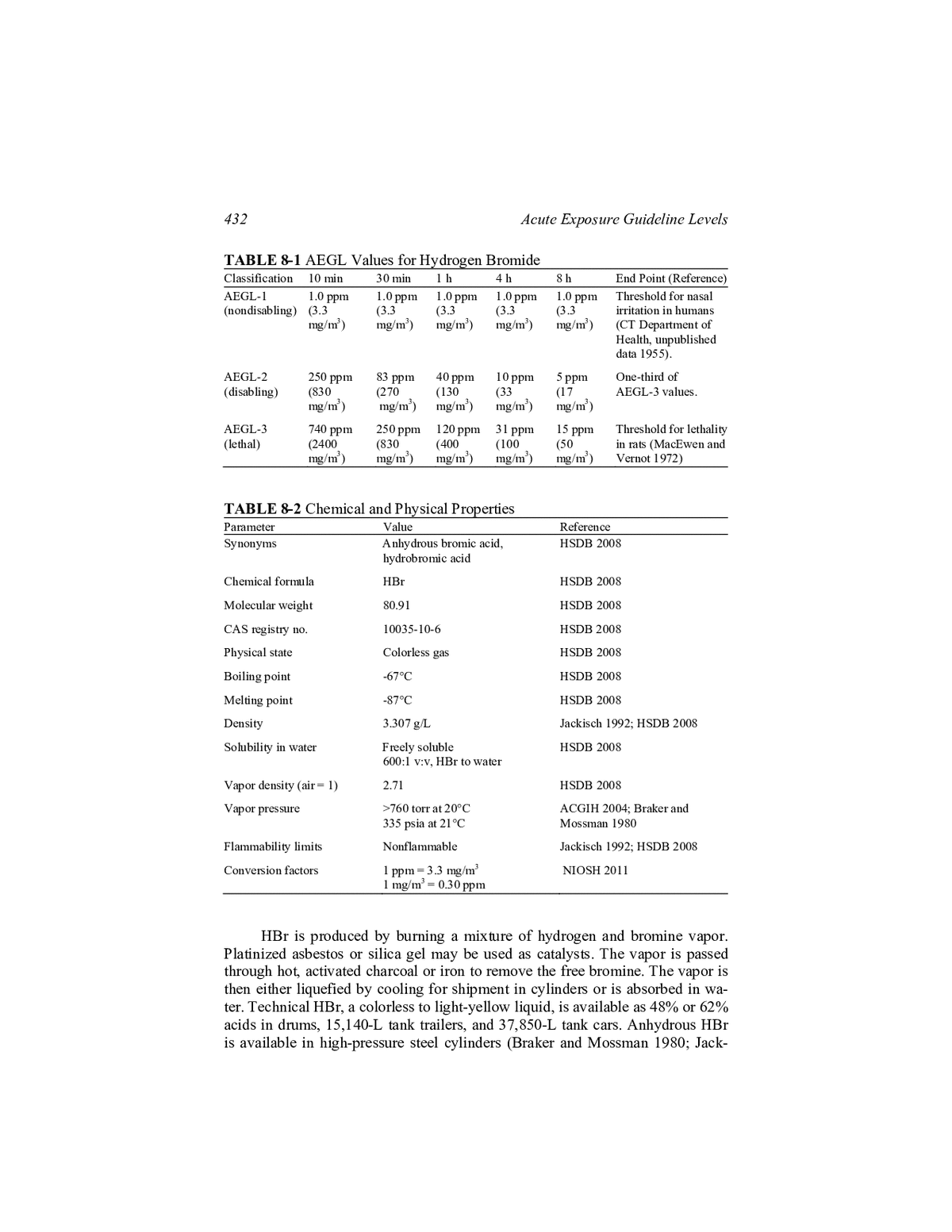
432 Acute Exposure Guideline Levels TABLE 8-1 AEGL Values for Hydrogen Bromide Classification 10 min 30 min 1h 4h 8h End Point (Reference) AEGL-1 1.0 ppm 1.0 ppm 1.0 ppm 1.0 ppm 1.0 ppm Threshold for nasal (nondisabling) (3.3 (3.3 (3.3 (3.3 (3.3 irritation in humans mg/m3) mg/m3) mg/m3) mg/m3) mg/m3) (CT Department of Health, unpublished data 1955). AEGL-2 250 ppm 83 ppm 40 ppm 10 ppm 5 ppm One-third of (disabling) (830 (270 (130 (33 (17 AEGL-3 values. mg/m3) mg/m3) mg/m3) mg/m3) mg/m3) AEGL-3 740 ppm 250 ppm 120 ppm 31 ppm 15 ppm Threshold for lethality (lethal) (2400 (830 (400 (100 (50 in rats (MacEwen and mg/m3) mg/m3) mg/m3) mg/m3) mg/m3) Vernot 1972) TABLE 8-2 Chemical and Physical Properties Parameter Value Reference Synonyms Anhydrous bromic acid, HSDB 2008 hydrobromic acid Chemical formula HBr HSDB 2008 Molecular weight 80.91 HSDB 2008 CAS registry no. 10035-10-6 HSDB 2008 Physical state Colorless gas HSDB 2008 Boiling point -67°C HSDB 2008 Melting point -87°C HSDB 2008 Density 3.307 g/L Jackisch 1992; HSDB 2008 Solubility in water Freely soluble HSDB 2008 600:1 v:v, HBr to water Vapor density (air = 1) 2.71 HSDB 2008 Vapor pressure >760 torr at 20°C ACGIH 2004; Braker and 335 psia at 21°C Mossman 1980 Flammability limits Nonflammable Jackisch 1992; HSDB 2008 3 Conversion factors 1 ppm = 3.3 mg/m NIOSH 2011 1 mg/m3 = 0.30 ppm HBr is produced by burning a mixture of hydrogen and bromine vapor. Platinized asbestos or silica gel may be used as catalysts. The vapor is passed through hot, activated charcoal or iron to remove the free bromine. The vapor is then either liquefied by cooling for shipment in cylinders or is absorbed in wa- ter. Technical HBr, a colorless to light-yellow liquid, is available as 48% or 62% acids in drums, 15,140-L tank trailers, and 37,850-L tank cars. Anhydrous HBr is available in high-pressure steel cylinders (Braker and Mossman 1980; Jack-
Hydrogen Bromide 433 isch 1992). HBr is used in the manufacture of organic and inorganic bromides, hydrobromic acid, as a reducing agent, as a catalyst in controlled oxidation reac- tions, in the alkylation of aromatic compounds, and in the isomerization of con- jugated diolefins (OâNeil et al. 2006). 2. HUMAN TOXICITY DATA 2.1. Acute Lethality No data on concentrations of HBr lethal to humans were found. 2.2. Nonlethal Toxicity Amoore and Hautala (1983) reported an odor threshold of 2 ppm for HBr. HBr liquid and vapor are highly corrosive to tissues. Symptoms of over expo- sure include coughing, choking, burning in the throat, wheezing, and asphyxia. Skin contact may cause severe burns, and contact of the eyes with the liquid or vapor may result in permanent damage (Jackisch 1992). One report by the Connecticut State Department of Health (unpublished data, 1955, as cited in ACGIH 20022) addressed responses of human subjects to HBr vapor. Six volunteers inhaled HBr at concentrations of 2-6 ppm for several minutes (Table 8-3). The odor was detected by all subjects at all concentrations. None of the subjects experienced ocular irritation. Only one subject experienced nasal and throat irritation at 3 ppm. One subject (presumably the same one) ex- perienced throat irritation at all of the higher concentrations, and all subjects experienced nasal irritation at 5 and 6 ppm. Responses ranged from slight, sting- ing sensations to a definite feeling of irritation. Although exposure at 5 ppm caused nasal irritation in all of the subjects, the authors stated that âit was con- sidered unlikely that noticeable disturbances will occur if peak concentrations do not exceed this value for brief periods.â According to Braker and Mossman (1980), hydrogen halides at concentra- tions of approximately 35 ppm cause irritation of the throat after short expo- sures. Concentrations of 1,000-2,000 ppm are lethal to humans from brief expo- sures and concentrations of 1,000-1,300 ppm are dangerous if breathed for 30- 60 min. Those data appear to be from the study by Henderson and Haggard (1943) and apply to HCl. 2 A detailed description of this study was provided in ACGIHâs 2002 documentation of threshold limit values and biological exposure indices for HBr. In its update of the docu- mentation in 2004, ACGIH omitted the study description and did not consider the data in its recommendations. The information from the study by the Connecticut Department of Health is retained here because it provides the only quantitative information on human exposure to HBr.
434 Acute Exposure Guideline Levels TABLE 8-3 Human Responses to Hydrogen Bromide Vapor Number of Subjects (n = 6) Reporting Responses Response 2 ppm 3 ppm 4 ppm 5 ppm 6 ppm Detectable odor 6 6 6 6 6 Nasal irritation 0 1 3 6 6 Throat irritation 0 1 1 1 1 Ocular irritation 0 0 0 0 0 Source: Adapted from ACGIH 2002. 2.3. Neurotoxicity No information on the neurotoxicity of HBr in humans was found. 2.4. Developmental and Reproductive Toxicity No data on the developmental or reproductive effects of HBr in humans was found. 2.5. Genotoxicity No data on the genotoxicity on HBr in humans was found. 2.6. Carcinogenicity No data on the carcinogenicity of HBr in humans was found. 2.7. Summary The only human data on HBr involved six volunteers exposed at 2-6 ppm for several minutes (CT Department of Health, unpublished data, 1955, as cited in ACGIH 2002). All six volunteers detected HBr at 2 ppm, and one individual experienced subjective irritation involving the nose and throat at 3 ppm. At higher concentrations, at least half of subjects experienced nasal and throat irri- tation. No information on neurotoxicity, developmental or reproductive effects, genotoxicity, or carcinogenicity of HBr was found. 3. ANIMAL TOXICITY DATA 3.1. Acute Lethality 3.1.1. Rats As part of a series of inhalation toxicity studies performed at Wright- Patterson Air Force Base, MacEwen and Vernot (1972; also reported in Back et al. 1972 and Vernot et al. 1977) subjected groups of 10 male Sprague-Dawley-
Hydrogen Bromide 435 derived rats to HBr at 2,205-3,822 ppm for 1 h (Table 8-4). Exposures took place in a modified Rochester chamber and concentrations were monitored with a bromide ion-specific electrode. The rats were monitored for mortality for 14 days. The 1-h LC50 was 2,858 ppm (95% confidence limits of 2,581-3,164 ppm) (Table 8-5). Responses of the animals during the exposures were dose-related and had the following a sequence: nasal and ocular irritation, labored breathing, gasping, and convulsions. The fur turned orange-brown during the exposures, and the color intensity was related to the concentration. The authors attributed a smoky haze around the animals during exposure to the reaction of HBr with the fur or moisture on the fur. During the 14-day postexposure period, the surviving animals were prostrate and most lost weight. Delayed deaths were observed. Burns accompanied by autolysis were observed on exposed areas of the skin. Rats exposed at the lowest concentration (2,205 ppm) returned to a normal weight gain by the end of the postexposure period. Gross examination at necrop- sy showed severe pulmonary and hepatic congestion and pulmonary edema in rats exposed at 3,822 ppm. The investigators noted that rats exposed at the lower concentration (not specified) had necrotic lesions on their feet and tails for up to 14 days. Opacity of the cornea, observed immediately following exposure, dis- appeared within 24 h. Groups of 5-8 male Fischer 344 rats were exposed to HBr at approximate- ly 1,300 ppm for 30 min (Stavert et al. 1991). Rats were placed into whole body flow plethysmographs to measure ventilatory rates. Body weight and respirato- ry-tract histology were investigated 24 h later. The mortality rate was 8% (Table 8-5). Rats exposed to HBr experienced an immediate and persistent drop in mi- nute ventilatory rate of 25%. The effect on ventilatory rate was similar with HF exposure, whereas exposure to HCl caused a much smaller decrease in ventila- tion. A small (<10%) reduction in body weight compared to nonexposed rats occurred by 24 h postexposure. TABLE 8-4 Results of One-Hour Inhalation Studies of Hydrogen Bromide in Rats and Mice Species Concentration (ppm) Mortality Ratio Rat 2,205 1/10 2,328 4/10 2,759 4/10 3,253 6/10 3,711 7/10 3,822 10/10 Mouse 507 0/10 875 7/10 1,036 9/10 1,163 10/10 Source: Adapted from MacEwen and Vernot 1972.
436 Acute Exposure Guideline Levels TABLE 8-5 Summary of Acute Lethality Data on Hydrogen Bromide in Rats and Mice Species Concentration (ppm) Exposure Time Effect Reference Rat 1,300 30 min 8% mortality Stavert et al. 1991 2,858 1h LC50 MacEwen and Vernot 1972 Mouse 507 1h No deaths MacEwen and Vernot 1972 814 1h LC50 As part of the same study, Stavert et al. (1991) compared the toxicities of three hydrogen halides (HF, HCl, and HBr) in rats exposed at 1,300 ppm for 30 min. Mortalities were 0% for HF, 6% for HCl, and 8% for HBr. Damage to the respiratory tract was assessed 24 h after the exposure. For all three hydrogen halides, tissue injury was confined to the nasal cavity. Tissue injury in the ante- rior nasal cavity was similar for all three compounds and involved moderate to severe fibrinonecrotic rhinitis. The mucosa and submucosa were necrotic, with necrosis extending to the turbinate bone. Blood clots were observed in nasal blood vessels; hemorrhage, fibrin, and fluid were observed in the nasal passages; and polymorphonuclear cells were observed in the submucosa and in the lumen. The severity of these lesions is summarized in Table 8-6. Exposure to HBr re- sulted in bilateral or unilateral severe necrohemorrhagic rhinitis in the anterior quarter of the nasal cavity, and necrosis of the mucosa and submucosa that ex- tended to the nasal turbinate bone. For HF and HCl, but not HBr, the lesions were also observed in the second anterior quarter of the nasal cavity. After expo- sure to all three halogen halides, the posterior half of the nasal cavity (including the ethmoid region) was essentially normal in appearance, showing that all three chemicals were well scrubbed. No pulmonary or tracheal injury was evident for any of the chemicals. The authors concluded that respiratory-tract injury caused by exposure to the three hydrogen halides was quantitatively similar. There was no change in pulmonary weight. In the same study (Stavert et al. 1991), groups of male Fischer 344 rats were exposed to HBr at 1,300 ppm for 30 min via a tracheal cannula (to simulate mouth breathing). This procedure bypasses the scrubbing of the nasal passages. Within 24 h after exposure, 19% of the rats died. Mean pulmonary weight was not significantly different from that of noncannulated rats or of rats exposed to air. Pulmonary lesions observed in treated animals were not significantly differ- ent from those of the cannulated control group. 3.1.2. Mice MacEwen and Vernot (1972) (see also Back et al. 1972) exposed groups of 10 CF1 (ICR-derived) mice (20-30 g) to HBr at concentrations ranging from 507 to 1,163 ppm for 1 h (Table 8-4). The LC50 was 814 ppm (95% confidence
Hydrogen Bromide 437 limits of 701-947 ppm) (Table 8-5). Responses during exposure were the same as those described for rats (see Section 3.1.1). No deaths occurred in mice ex- posed at 507 ppm, and the mice had a normal weight gain during the 14-day recovery period. Mice surviving the 14-day postexposure period had necrotic lesions of their tails. No other gross pathologic changes were apparent in surviv- ing mice. 3.2. Nonlethal Toxicity As part of the Stavert et al. (1991) study, Kusewitt et al. (1989) reported on exposures to three hydrogen halides at lower concentrations. Fischer 344 rats (number not specified) were exposed to HF, HCl, or HBr at concentrations of 100-1,000 ppm for 30 min and were killed 8 and 24 h later. Tissue damage was restricted to the nasal region and consisted of necrosis and inflammation; the severity of the damage increased with concentration. HF was the most toxic, and that the toxicities of HCl and HBr were similar. Histopathologic examinations and gravimetric measurements revealed no damage to the lungs. No further de- tails were reported in the available abstract. Toxicity data on the related chemical, HCl, are relevant to evaluating the toxicity of HBr. In a study in which the ventilatory rate of rats exposed to HCl at 1,000 ppm for 30 min was increased by the addition of CO2 to the exposure chamber, no deaths occurred and histopathologic lesions were confined to the upper respiratory tract and (Lehnert and Stavert 1991). Barrow et al. (1977) ex- posed groups of four male Swiss-Webster mice to HCl at concentrations of 40, 99, 245, 440, or 943 ppm for 10 min. An RD50 (a 50% decrease in the respirato- ry rate) of 309 ppm was calculated. At 99 ppm, approximately one-third of the RD50, the decrease in respiratory rate was 25-30%. Additional studies summa- rized in NRC (2004) showed that primates were less sensitive to the toxic effects of HCl than rodents. TABLE 8-6 Severity of Lesions in the Anterior Region of the Nasal Cavity of Rats Following Exposure to Hydrogen Fluoride, Hydrogen Chloride, or Hydrogen Bromide at 1,300 ppm for 30 Minutes Necrotic Lesion HF HCl HBr Epithelial 3.3a 3.8a 3.3a Submucosal 2.6a 3.0a 2.6a Bone 0.3 2.4a 1.6a a a Gland 1.8 2.4 1.4a Severity index: 1 = mild, 2 = moderate, 3 = severe, and 4 = very severe (n = 8). a Statistically significant compared to air-exposed controls, p < 0.05. Source: Adapted from Stavert et al. 1991.
438 Acute Exposure Guideline Levels 3.3. Neurotoxicity No information on the neurotoxicity of HBr in animals was found. 3.4. Developmental and Reproductive Toxicity No information on the developmental or reproductive effects of HBr in an- imals was found. 3.5. Genotoxicity No information on the genotoxicity of HBr in animals was found. 3.6. Chronic Toxicity and Carcinogenicity No information on the chronic toxicity or carcinogenicity of HBr in ani- mals was found. 3.7. Summary Two studies of HBr in animals were available. In the first study (MacE- wen and Vernot 1972), groups of rats and mice were exposed by inhalation to a range of concentrations for 1 h. The 1-h LC50 value was 2,858 in rats and 814 ppm in mice. All tested concentrations resulted in lethality in rats during the 14- day postexposure period. No deaths occurred in mice exposed at 507 ppm for 1 h. In rats exposed at 1,300 ppm for 30 min, mortality was 8% (presumably one of 12 rats) and lesions were confined to the anterior nasal passages (Stavert et al. 1991). Nasal lesions were also observed in rats exposed at up to 1,000 ppm for 30 min (Kusewitt et al. 1989). Animals in the Kusewitt et al. (1989) and Stavert et al. (1991) studies were killed 24 h after exposure. Only one of 10 rats exposed at 2,205 ppm died in the MacEwen and Vernot (1972) study. 4. SPECIAL CONSIDERATIONS 4.1. Metabolism and Disposition No data on the metabolism and deposition of HBr were found. Data on soluble bromides are available from their medical use as oral sedatives, diuret- ics, and antiepileptics. An oral dose of 3 g (30-60 mg/kg for an adult) is consid- ered a âno-ill effectâ dose (Teitelbaum 2001).
Hydrogen Bromide 439 4.2. Mechanism of Toxicity The available studies indicate that the hydrogen halides are severe irritants to the skin, eyes, and respiratory tract, particularly the anterior nasal passages where, depending on concentration, they appear to be effectively scrubbed from the inhaled air. For HBr, deposition in the anterior nasal passages may be at- tributed to its high solubility and reactivity. At high concentrations (e.g., 3,822 ppm for 1 h), penetration into the lungs occurs as evidenced by pulmonary hem- orrhage, edema, and death. Although HBr is absorbed, serious systemic effects are unlikely to occur at concentrations below those that would cause serious respiratory effects. In the studies summarized in Tables 8-4 and 8-5, the tissues of the respiratory tract and exposed dermal surfaces sustained the impact of an acute exposure. Therefore, the concentration of HBr in the inhaled air and not the absorbed dose is the primary determinant of the effects from acute expo- sures. 4.3. Structure-Activity Relationships Differences in size and electron configuration of the various halogen at- oms result in substantial differences with respect to their chemical and physical properties, which in turn affect their toxicologic properties. The atomic weights of fluorine, chlorine, bromine, and iodine are 19, 35.5, 80, and 127, respectively. Data on the relative toxicities of HF, HCl, and HBr on the basis of lethality are available. As can be seen from the data in Table 8-7, three rodent studies using different exposure durations show that HF is more lethal than HCl (Rosenholtz et al. 1963; Higgins et al. 1972; MacEwen and Vernot 1972; Wohlslagel et al. 1976). For both the rat and mouse, HF is also more lethal than HBr (MacEwen and Vernot 1972). Data from the same laboratory (Wohlslagel et al. 1976; MacEwen and Vernot 1972) show that HCl and HBr have similar 1-h LC50 values of 3,124 and 2,858 ppm, respectively. Data on the nonlethal toxicity of the three hydrogen halides (Stavert et al. 1991) suggest that HF, HCl, and HBr are similarly toxic to the nasal cavity following acute exposure. HBr and HF exposure resulted in simi- lar decreases (by about 25%) in the ventilation rate of cannulated rats (simulation of mouth breathing), whereas the decrease associated with HCl exposure was smaller (Stavert et al. 1991). 4.4. Other Relevant Information 4.4.1. Species Variability HBr toxicity data, available for only the rat and mouse, showed that mice are more susceptible than rats. However, when considering lethal concentrations of respiratory irritants (such as HCl), the mouse âmay not be an appropriate model for extrapolation to humans,â because âmice appear to be much more
440 Acute Exposure Guideline Levels TABLE 8-7 Relative Toxicities of Hydrogen Fluoride, Hydrogen Chloride, and Hydrogen Bromide Exposure LC50 Values (ppm) Species Duration HF HCl HBr Reference Rat 5 min 18,200 41,000 â Higgins et al. 1972 Mouse 5 min 6,247 13,750 â Rat 30 min 2,042 4,700 Rosenholtz et al. 1963; MacEwen and Vernot 1972 Mouse 30 min â 2,644 â MacEwen and Vernot 1972 Rat 1h 1,395 3,124 â Wohlslagel et al. 1976 Mouse 1h 342 1,108 â Monkey 1h 1,774 â â MacEwen and Vernot 1970 Rat 1h 1,278 â 2,858 MacEwen and Vernot 1972 Mouse 1h 501 â 814 MacEwen and Vernot 1972 The data of Wohlslagel et al. (1976) and MacEwen and Vernot (1972) were generated in the same laboratory. Therefore, the values for HCl (Wohlslagel et al. 1976) can be com- pared with those for HF and HBr (MacEwen and Vernot 1972). susceptible to the lethal effects of HCl than other rodents or baboons. To some extent, this increased susceptibility may be due to less effective scrubbing of HCl in the upper respiratory tractâ (NRC 1991). The same principle reasonably holds true for HF and HBr. The respiratory rate of mice is also higher than that of rats. The data in Table 8-7 show species that mice are the most susceptibile to HF, followed by the rat and nonhuman primate (rhesus monkey). 4.4.2. Susceptible Populations Individuals with asthma may respond to exposure to respiratory irritants, such as HBr, with increased bronchial responsiveness. No information on the relative susceptibility of asthmatic and normal individuals to HBr was found. In a study with HCl, exposure at1.8 ppm for 45 min was a no-effect level for exer- cising asthmatics (Stevens et al. 1992). Individuals under stress, such as those involved in emergency situations and individuals engaged in physical activity, will likely experience increased penetration of HBr into the lower respiratory tract due to increased minute vol- umes, with the potential for increased irritant response, as compared to individu- als at rest. 4.4.3. Concentration-Exposure Duration Relationship No information on the relationship between concentration and exposure for a single end point was found. When no data are available, time scaling is
Hydrogen Bromide 441 based on the equation Cn à t = k, with default values of n = 3 for extrapolation to shorter exposure durations and n = 1 for extrapolation to longer exposure dura- tions (NRC 2001). However, information on relevant chemicals HF and HCl are available. On the basis of lethality data, the n values for time scaling was 2 for HF and 1 for HCl (NRC 2004). HBr is more similar chemically to HCl than HF. 4.4.4. Concurrent Exposure Issues No information on concurrent exposure issues for HBr was found. 5. DATA ANALYSIS FOR AEGL-1 5.1. Human Data Relevant to AEGL-1 Reliable human data on HBr are available from a study of six volunteers exposed at 2-6 ppm for several minutes (CT Department of Health, unpublished data, 1955, as cited in ACGIH 2002). Nasal irritation was reported by 0, 1, 3, 6, and 6 individuals at 2, 3, 4, 5, and 6 ppm, respectively. Throat irritation did not appear to be concentration dependent and no ocular irritation was reported. Therefore, the threshold for subjective nasal irritation is 3 ppm. 5.2. Animal Data Relevant to AEGL-1 No data on HBr relevant to notable discomfort in animals was found. 5.3. Derivation of AEGL-1 Values The threshold for nasal irritation of 3ppm in human subjects exposed to HBr for several minutes (CT Department of Health, unpublished data, 1955, as cited in ACGIH 2002) was selected as the basis for the AEGL-1 values. That concentration was considered to be a threshold for notable discomfort, as only one individual was affected at that concentration. The 3 ppm point-of-departure was divided by an intraspecies uncertainty factor of 3, because response to sen- sory irritation is not expected to vary greatly among individuals (NRC 2001). A factor of 3 was considered sufficient because the effect of slight irritation is be- low the definition of AEGL-1. In addition, an intraspecies uncertainty factor of 3 was used to derive AEGL values for the related compounds HCl and HF, which have the same mode of action as HBr (NRC 2004). It is reasonable to use the same uncertainty factors for a class of chemicals whose mode of action is the same. Finally, the uncertainty factor used to derive the AEGL-1 values for HBr is believed to be protective of asthmatic individuals on the basis of comparison of the AEGL-1 value for HBr (1.0 ppm) with the AEGL-1 value for HCl (1.8
442 Acute Exposure Guideline Levels ppm); the latter is based on a no-effect level for irritation in exercising asthmat- ics. There is evidence that HBr is of similar toxicity to HCl; thus, the lower AEGL-1 values for HBr are considered to be protective of asthmatics on the basis of data on HCl. Because irritation depends on concentration rather than time, and adapta- tion to slight irritation occurs (Dalton 2001), 1.0 ppm was used for all of the AEGL-1 exposure durations (see Table 8-8). Derivation of the AEGL-1 values for HBr are presented in Appendixes A and D, and a category plot of the toxicity data for HBr in relation to AEGL values is presented in Appendix B. Although the AEGL-1 values for HBr are based on data presented in a secondary source, the values obtained from the data are supported by compari- son with the related compounds HF and HCl. The AEGL-1 values for HF (1.0 ppm) and HCl (1.8 ppm) are equal to or higher than the values obtained for HBr using data from Connecticut State Department of Health (unpublished data, 1955, as cited in ACGIH 2002). Thus, if AEGL-1 values for HBr were obtained by analogy to these related compounds, the same or higher AEGL-1 values would be derived. 6. DATA ANALYSIS FOR AEGL-2 6.1. Human Data Relevant to AEGL-2 No human data on HBr relevant to development of AEGL-2 values were found. 6.2. Animal Data Relevant to AEGL-2 The only study of HBr that addresses effects that meet the definition of an AEGL-2 was a study on hydrogen halides by Stavert et al. (1991). Following inhalation of HBr, HCl, or HF at 1,300 ppm for 30 min, male F-344 rats exhibit- ed severe necrotic lesions of the anterior nasal passages and 8% of the rats died (Stavert et al. 1991). Lesions consisting of necrosis and inflammation were re- stricted to the nasal region; the lungs appeared unaffected. Rats were killed 24 h after exposure and no judgment could be made about whether the lesions were reversible. The authors noted that the nasal lesions were similar in severity and location for all three hydrogen halides when tested at the same concentration. 6.3. Derivation of AEGL-2 Values The Stavert et al. (1991) study was not considered a suitable basis for der- ivation of AEGL-2 values because 8% of the animals died after exposure to HBr at 1,300 ppm. Additionally, the study only tested a single concentration and the number of animals tested was not specified. In the absence of suitable data, the
Hydrogen Bromide 443 TABLE 8-8 AEGL-1 Values for Hydrogen Bromide 10 min 30 min 1h 4h 8h 1.0 ppm 1.0 ppm 1.0 ppm 1.0 ppm 1.0 ppm (3.3 mg/m3) (3.3 mg/m3) (3.3 mg/m3) (3.3 mg/m3) (3.3 mg/m3) AEGL-3 values for HBr were divided by 3 to estimate AEGL-2 values. This approach is supported by the steep concentration-response curve demonstrated in the MacEwen and Vernot (1972) lethality studies. The AEGL-2 values for HBr are presented in Table 8-9, and the calculations are presented in Appendix- es A and D. A category plot of the toxicity data on HBr in relation to AEGL values is presented in Appendix B. 7. DATA ANALYSIS FOR AEGL-3 7.1. Human Data Relevant to AEGL-3 No human data on HBr relevant to development of AEGL-3 values were found. 7.2. Animal Data Relevant to AEGL-3 Lethality data on HBr were available for the rat and mouse. One-hour LC50 values for the rat and mouse were 2,858 and 814 ppm, respectively (MacEwen and Vernot 1972). The data are summarized in Table 8-4. A 30-min exposure to HBr at 1,300 ppm resulted in 8% mortality in rats (Stavert et al. 1991). From the MacEwen and Vernot study in the rat, a 1-h LC01 of 1,350 ppm was calculated by probit analysis. The BMCL05 was 1,239 ppm (see Appendix C) and the BMC01 was 1,456 ppm (data not shown). No deaths occurred in rats exposed at 1,000 ppm for 30 min (Kusewitt et al. 1989) or in mice exposed at 507 ppm for 1 h (MacEwen and Vernot 1972). As noted in Section 4.4.1 (Spe- cies Variability), mice are not considered an appropriate species for setting le- thality values for hydrogen halides, because mice are more susceptible to the lethal effects of HCl than rats or non-human primates (NRC 1991). 7.3. Derivation of AEGL-3 Values The BMCL05 of 1,239 ppm, calculated from 1-h lethality data from studies in Sprague-Dawley rats exposed to HBr (MacEwen and Vernot 1972), is an es- timate of the threshold for lethality and was selected as the point of departure to develop AEGL-3 values for HBr. This value was more conservative than the BMC01 of 1,456 ppm calculated from the same data. A total uncertainty factor of 10 was applied: 3 for interspecies differences and 3 for intraspecies variability.
444 Acute Exposure Guideline Levels TABLE 8-9 AEGL-2 Values for Hydrogen Bromide 10 min 30 min 1h 4h 8h 250 ppm 83 ppm 40 ppm 10 ppm 5 ppm (830 mg/m3) (270 mg/m3) (130 mg/m3) (33 mg/m3) (17 mg/m3) The individual factors are considered to be sufficient because the action of a direct-acting irritant is not expected to vary greatly among species or between individuals (NRC 2001). The 60-min point of departure was time-scaled to the other AEGL-3 dura- tions using the equation Cn à t = k. The value of n was 1, on the basis of data on the related compound HCl, for which regression analysis of combined LC50 data from rats and mice resulted in an estimate of n = 1 (see NRC 2004). The AEGL- 3 values for HBr are presented in Table 8-10. The use of the BMCL05 as the point-of-departure for the AEGL-3 values is supported by the finding that the point-of-departure for the 30-min AEGL-3 is an estimate of the threshold for lethality of 155 ppm and is approximately 10-fold lower than the concentration which resulted in 8% mortality (1,300 ppm) (Stavert et al. 1991). The AEGL-3 calculations are presented in Appendices A and D, and a category plot of the toxicity data on HBr in relation to the AEGL values is presented in Appendix B. 8. SUMMARY OF AEGLS 8.1. AEGL Values and Toxicity End Points The AEGL values for HBr are presented in Table 8-11. The AEGL-1 val- ues were based on the concentration that did not result in nasal irritation in sub- jects exposed to HBr for several minutes. AEGL-2 values were derived by tak- ing one-third the AEGL-3 values, and the AEGL-3 values were based on the BMCL05 estimated from rat lethality data. A comparison of the AEGL values for HBr, HCl, and HF is presented in Table 8-12. The AEGL-1 values for the three hydrogen halides are similar, as are the longer-term AEGL-2 values. The AEGL-3 values for HBr and HCl are similar and the HF values are generally lower; this is consistent with the find- ings presented in Table 8-7, which showed that lethality was observed at lower concentrations of HF, as compared to HBr and HCl. 8.2. Comparison with Other Standards and Guidelines Other standards and guidelines for HBr are presented in Table 8-13. Ex- cept for the Occupational Safety and Health Administrationâs permissible expo- sure limit, ceiling or peak limits rather than 8-h time-weighted averages (TWA) have been derived for the workplace. The AEGL-1 for HBr is below the work- place guidelines. The immediately dangerous to life or health (ILDH) value is
Hydrogen Bromide 445 based on analogy with HCl (NIOSH 1994). The IDLH for HCl is 50 ppm which is 10 times the recommended exposure limit (REL) of the National Institute for Occupational Safety and Health (NIOSH). Therefore, the IDLH for HBr was set at 10 times the NIOSH REL of 3 ppm. The 30-min AEGL-2 is similar to the IDLH. TABLE 8-10 AEGL-3 Values for Hydrogen Bromide 10 min 30 min 1h 4h 8h 740 ppm 250 ppm 120 ppm 31 ppm 15 ppm (2,400 mg/m3) (830 mg/m3) (400 mg/m3) (100 mg/m3) (50 mg/m3) TABLE 8-11 AEGL Values for Hydrogen Bromide Exposure Duration Classification 10 min 30 min 1h 4h 8h AEGL-1 1.0 ppm 1.0 ppm 1.0 ppm 1.0 ppm 1.0 ppm (nondisabling) AEGL-2 250 ppm 83 ppm 40 ppm 10 ppm 5 ppm (disabling) AEGL-3 740 ppm 250 ppm 120 ppm 31 ppm 15 ppm (lethal) TABLE 8-12 AEGL Values for Hydrogen Bromide, Hydrogen Chloride, and Hydrogen Fluoride (ppm) Classification 10 min 30 min 1h 4h 8h AEGL-1 HBr 1.0 1.0 1.0 1.0 1.0 HCl 1.8 1.8 1.8 1.8 1.8 HF 1.0 1.0 1.0 1.0 1.0 AEG-2 HBr 250 83 40 10 5 HCl 100 43 22 11 11 HF 95 34 24 12 12 AEGL-3 HBr 740 250 120 31 15 HCl 620 210 100 26 26 HF 170 62 44 22 22 Source of the HCl and HF values: NRC 2004.
446 Acute Exposure Guideline Levels TABLE 8-13 Standards and Guidelines for Hydrogen Bromide Exposure Duration Guideline 10 min 30 min 1h 4h 8h AEGL-1 1.0 ppm 1.0 ppm 1.0 ppm 1.0 ppm 1.0 ppm AEGL-2 250 ppm 83 ppm 40 ppm 10 ppm 5 ppm AEGL-3 740 ppm 250 ppm 120 ppm 31 ppm 15 ppm b IDLH (NIOSH) â 30 ppm â â â PEL-TWA (OSHA)b â â â â 3 ppm c TLV-C (ACGIH) 2 ppm 2 ppm 2 ppm 2 ppm 2 ppm REL-C (NIOSH)d 3 ppm 3 ppm 3 ppm 3 ppm 3 ppm MAK peak limit 2 ppm (15 min, 4 times/shift) â â â (Germany)e a IDLH (immediately dangerous to life or health, National Institute for Occupational Safe- ty and Health) (NIOSH 1994) represents the maximum concentration from which one could escape within 30 min without any escape-impairing symptoms or any irreversible health effects. b PEL-TWA (permissible exposure limits â time-weighted average, Occupational Health and Safety Administration) (29CFR 1910.1045 [2006]) is defined analogous to the ACGIH TLV-TWA, but is for exposures of no more than 10 h/day, 40 h/wk. c TLV-C (threshold limit value â ceiling, American Conference of Governmental Industri- al Hygienists) (ACGIH 2012) is a limit that should not be exceeded during the working day. d REL-C(recommended exposure limit â ceiling, National Institute for Occupational Safe- ty and Health) (NIOSH 2011) is defined analogous to the ACGIH TLV-ceiling. e MAK Spitzenbegrenzung (peak limit) (German Research Association (DFG 1999) con- stitutes the maximum average concentration to which workers can be exposed for a peri- od of 15 min with no more than four excursions per work shift and with an interval of 1 h between excursions. 8.3. Data Adequacy and Research Needs Only one study of human subjects was available for development of AEGL-1 values (CT Department of Health, unpublished data, 1955, as cited in ACGIH 2002). The study was unpublished and available only in a secondary source. Although the study used short exposure durations, an adequate number of subjects was used, a range of concentrations was tested, and irritant levels were clearly described. Animal data on HBr were available from studies of two species, the rat and mouse. The well-conducted studies with rats from two dif- ferent laboratories (MacEwen and Vernot 1972; Stavert et al. 1991) had reason- able agreement in results. Those studies also addressed the relative toxicities of HBr, HF, and HCl in the rat. Although the data on HBr were sparse, supporting information on related hydrogen halides and information on relative toxicity are
Hydrogen Bromide 447 available; thus, the data were considered adequate to derive AEGL-1 and 3 val- ues for HBr. The database was not considered suitable for AEGL-2 values; the AEGL-3 values were divided by 3 to derive AEGL-2 values for HBr. 9. REFERENCES ACGIH (American Conference of Government and Industrial Hygienists). 2002. Docu- mentation of the Threshold Limit Values (TLVs) for Chemical and Physical Agents and Biological Exposure Indices (BEIs): Hydrogen Bromide. American Conference of Government and Industrial Hygienists, Cincinnati, OH. ACGIH (American Conference of Government and Industrial Hygienists). 2004. Docu- mentation of the Threshold Limit Values for Chemical and Physical Agents and Biological Exposure Indices (BEIs): Hydrogen Bromide. American Conference of Government and Industrial Hygienists, Cincinnati, OH. ACGIH (American Conference of Government and Industrial Hygienists). 2012. Thresh- old Limit Values (TLVs) and Biological Exposure Indices (BEIs) Based on the Documentation of the Threshold Limit Values for Chemical and Physical Agents and Biological Exposure Indices. American Conference of Government and Indus- trial Hygienists, Cincinnati, OH. Amoore, J.E., and E. Hautala. 1983. Odor as an aid to chemical safety: Odor thresholds compared with threshold limit values and volatilities for 214 industrial chemicals in air and water dilution. J. Appl. Toxicol. 3(6):272-289. Back, K.C., A.A. Thomas, and J.D. MacEwen. 1972. Reclassification of Materials Listed as Transportation Health Hazards. Report No. TSA-20-72-3. Aerospace Medical Research Laboratory, Wright Patterson Air Force Base, OH. Barrow, C.S., Y. Alarie, M. Warrick, and M.F. Stock. 1977. Comparison of the sensory irritation response in mice to chlorine and hydrogen chloride. Arch. Environ. Health. 32(2):68-76. Braker, W., and A.L. Mossman. 1980. Matheson Gas Data Book, 6th Ed. Lyndhurst, NJ: Matheson. Dalton, P. 2001. Evaluating the human response to sensory irritation: Implications for setting occupational exposure limits. Am. Ind. Hyg. Assoc. J. 62(6):723-729. DFG (Deutsche Forschungsgemeinschaft). 1999. List of MAK and BAT Values, 1999. Maximum Concentrations and Biological Tolerance Value at the Workplace Re- port No. 35. Weinheim, Federal Republic of Germany: Wiley-VCH. Henderson, Y., and H.W. Haggard. 1943. Hydrochloric acid (Hydrogen chloride). P. 126- 127 in Noxious Gases and the Principles of Respiration Influencing Their Action, 2nd Rev. Ed. New York: Reinhold Publishing. Higgins, E.A., V. Fiorca, A.A. Thomas, and H.V. Davis. 1972. Acute toxicity of brief exposures to HF, HCl, NO2 and HCN with and without CO. Fire Technol. 8(2):120-130. HSDB (Hazardous Substances Data Base). 2008. Hydrogen bromide. TOXNET, Special- ized Information Services, U.S. National Library of Medicine, Bethesda, MD [online]. Available: http://toxnet.nlm.nih.gov [accessed Jan. 10, 2013]. Jackisch, P.F. 1992. Bromine compounds. Pp. 536-560 in Kirk-Othmer Encyclopedia of Chemical Technology, 4th Ed., Vol. 4. New York: John Wiley & Sons. Kusewitt, D.F., D.M. Stavert, G. Ripple, T. Mundie, and B.E. Lehnert. 1989. Relative acute toxicities in the respiratory tract of inhaled hydrogen fluoride, hydrogen bromide, and hydrogen chloride. Toxicologist 9:36 [Abstract No. 144].
448 Acute Exposure Guideline Levels Lehnert, B.E., and D.M. Stavert. 1991. The Acute Inhalation Toxicity of Pyrolysis Prod- ucts of Halon 1301. Annual Report. AD-A246 031. Los Alamos National Labora- tory, Los Alamos, NM. MacEwen, J.D., and E.H. Vernot. 1970. Toxic Hazards Research Unit Annual Technical Report: 1970. AMRL-TR-70-77, AD 714694. Aerospace Medical Research La- boratory, Wright-Patterson Air Force Base, OH [online]. Available: http://www. dtic.mil/dtic/tr/fulltext/u2/714694.pdf online]. [accessed Apr. 5, 2014]. MacEwen, J.D., and E.H. Vernot. 1972. Toxic Hazards Research Unit Annual Technical Report: 1972. AMRL-TR-72-62, AD 755 358. Aerospace Medical Research La- boratory, Wright-Patterson Air Force Base, OH [online]. Available: http://www. dtic.mil/dtic/tr/fulltext/u2/755358.pdf [accessed Apr.5, 2014]. NIOSH (National Institute for Occupational Safety and Health). 1994. Documentation for Immediately Dangerous to Life or Health Concentrations (IDLHs): Hydrogen bromide. National Institute for Occupational Safety and Health [online]. Available: http://www.cdc.gov/niosh/idlh/10035106.html [accessed Apr. 5, 2014]. NIOSH (National Institute for Occupational Safety and Health). 2011. NIOSH Pocket Guide to Chemical Hazards: Hydrogen bromide. National Institute for Occupa- tional Safety and Health [online]. Available: http://www.cdc.gov/niosh/npg/npgd 0331.html [accessed Apr. 5, 2014]. NRC (National Research Council). 1991. Hydrogen chloride. Pp. 37-52 in Permissible Exposure Levels and Emergency Exposure Guidance Levels for Selected Airborne Contaminants. Washington, DC: National Academy Press. NRC (National Research Council). 1993. Guidelines for Developing Community Emer- gency Exposure Levels for Hazardous Substances. Washington, DC: National Academy Press. NRC (National Research Council). 2001. Standing Operating Procedures for Developing Acute Exposure Guideline Levels for Hazardous Chemicals. Washington, DC: Na- tional Academy Press. NRC (National Research Council). 2004. Acute Exposure Guideline Levels for Selected Airborne Chemicals, Vol. 4. Washington, DC: National Academies Press. OâNeil, M.J., A. Smith, and P.E. Heckelman, eds. 2006. Hydrogen bromide. P. 829 in The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals, 14th ed. Whitehouse Station, NJ: Merck. Rosenholtz, M.J., T.R. Carson, M.H. Weeks, F. Wilinski, D.F. Ford, and F.W. Oberst. 1963. A toxicopathologic study in animals after brief single exposures to hydrogen fluoride. Am. Ind. Hyg. Assoc. J. 24(3):253-261. Stavert, D.M., D.C. Archuleta, M.J. Behr, and B.E. Lehnert. 1991. Relative acute toxici- ties of hydrogen fluoride, hydrogen chloride, and hydrogen bromide in nose- and pseudomouth-breathing rats. Fundam. Appl. Toxicol. 16(4):636-655. Stevens, B. J.Q. Koenig, V. Rebolledo, Q.S. Hanley, and D.S. Covert. 1992. Respiratory effects from the inhalation of hydrogen chloride in young adult asthmatics. J. Oc- cup. Med. 34(9):923-929. Teitelbaum, D.T. 2001. The halogens. Pp. 731-825 in Pattyâs Industrial Hygiene and Toxicology, 5th Ed., Vol. 3. New York: John Wiley & Sons. Vernot, E.H., J.D. MacEwen, C.C. Haun and E.R. Kinkead. 1977. Acute toxicity and skin corrosion data for some organic and inorganic compounds and aqueous solutions. Toxicol. Appl. Pharmacol. 42:417-423. Wohlslagel, J., L.C. DiPasquale and E.H. Vernot. 1976. Toxicity of solid rocket motor ex- haust: effects of HCl, HF, and alumina on rodents. J. Combust. Toxicol. 3:61-69.
Hydrogen Bromide 449 APPENDIX A DERIVATION OF AEGL VALUES FOR HYDROGEN BROMIDE Derivation of AEGL-1 Values Key study: CT Department of Health, unpublished data (1955, as cited in ACGIH 2002) Toxicity end point: Nasal and throat irritation in one of six subjects to HBr at 3 ppm for several minutes Time scaling: No time scaling, because there is adaptation to slight irritation. Uncertainty factors: 3 for intraspecies variability; irritation from a direct-contact irritant should not vary greatly among individuals (NRC 2001). Calculation: 3 ppm ÷ 3 = 1.0 ppm (applied to all AEGL durations) Derivation of AEGL-2 Values Because data on HBr were inadequate, AEGL-2 values were derived by taking one-third of the respective AEGL-3 values. Calculations: 10-min AEGL-2: 740 ppm ÷ 3 = 250 ppm 30-min AEGL-2: 250 ppm ÷ 3 = 83 ppm 1-h AEGL-2: 110 ppm ÷ 3 = 40 ppm 4-h AEGL-2: 31 ppm ÷ 3 = 10 ppm 8-h AEGL-2: 15 ppm ÷ 3 = 5 ppm Derivation of AEGL-3 Values Key study: MacEwen and Vernot (1972) Toxicity end point: Lethality in rats exposed for 1 h, BMCL05 of 1,238.95 ppm.
450 Acute Exposure Guideline Levels Time scaling: Cn à t = k; n = 1 on the basis of lethality data on HCl in rats (1,238.95 ppm ÷ 10) à 60 min = 7,433.7 ppm-min Uncertainty factors: 3 for interspecies differences; a direct-contact irritant is not expected to vary greatly between species (NRC 2001) 3 for intraspecies variability; response to a direct- contact irritant is not expected to vary greatly among humans (NRC 2001) Calculations: 10-min AEGL-3: 7,433.7 ppm-min ÷ 10 min = 740 ppm 30-min AEGL-3: 7,433.7 ppm-min ÷ 30 min = 250 ppm 1-h AEGL-3: 7,433.7 ppm-min ÷ 60 min = 120 ppm 4-h AEGL-3: 7,433.7 ppm-min ÷ 240 min = 31 ppm 8-hAEGL-3: 7,433.7 ppm-min ÷ 480 min = 15 ppm
Hydrog gen Bromide 4451 AP PPENDIX B GORY PLOT FOR CATEG F HYDRO OGEN BROM MIDE FIGUR RE B-1 Category y plot of toxicity data and AEGL L values for hydrrogen bromide.
452 TABLE B-1 Data Used in the Category Plot for Hydrogen Bromide Source Species ppm Minutes Category AEGL-1 1.0 10 AEGL AEGL-1 1.0 30 AEGL AEGL-1 1.0 60 AEGL AEGL-1 1.0 240 AEGL AEGL-1 1.0 480 AEGL AEGL-2 250 10 AEGL AEGL-2 83 30 AEGL AEGL-2 40 60 AEGL AEGL-2 10 240 AEGL AEGL-2 5 480 AEGL AEGL-3 740 10 AEGL AEGL-3 250 30 AEGL AEGL-3 120 60 AEGL AEGL-3 31 240 AEGL AEGL-3 15 480 AEGL CT State Dept. Health 1955 Human 2 5 0, no irritation CT State Dept. Health 1955 Human 3 5 1, nasal and throat irritation, 1 subject CT State Dept. Health 1955 Human 4 5 1, nasal and throat irritation, 3 subjects
CT State Dept. Health 1955 Human 5 5 1, nasal and throat irritation, 6 subjects CT State Dept. Health 1955 Human 6 5 1, nasal and throat irritation, 6 subjects MacEwen and Vernot 1972 Rat 2,205 60 SL, 10% mortality MacEwen and Vernot 1972 Rat 2,328 60 SL, 40% mortality MacEwen and Vernot 1972 Rat 2,759 60 SL, 40% mortality MacEwen and Vernot 1972 Rat 3,253 60 SL, 60% mortality MacEwen and Vernot 1972 Rat 3,711 60 SL, 70% mortality MacEwen and Vernot 1972 Rat 3,822 60 3, 100% mortality MacEwen and Vernot 1972 Mouse 507 60 2, no mortality MacEwen and Vernot 1972 Mouse 875 60 SL, 70% mortality MacEwen and Vernot 1972 Mouse 1,036 60 SL, 90% mortality MacEwen and Vernot 1972 Mouse 1,163 60 3, 100% mortality Stavert et al. 1991 Rat 1,300 30 SL, 8% mortality For category: 0 = no effect, 1 = discomfort, 2 = disabling, SL = some lethality, 3 = lethal. 453
454 Acute Exposure Guideline Levels APPENDIX C BENCHMARK CONCENTRATION CALCULATION Hydrogen bromide BMCL05 Probit Model. (Version: 2.8; Date: 02/20/2007) Input Data File: C:\BMDS\HBR05.(d) Gnuplot Plotting File: C:\BMDS\HBR05.plt Mon Dec 17 11:29:37 2007 BMDS MODEL RUN The form of the probability function is: P[response] = Background + (1-Background) * CumNorm(Intercept + Slope*Log (Dose), where CumNorm(.) is the cumulative normal distribution function Dependent variable = COLUMN3 Independent variable = COLUMN1 Slope parameter is not restricted Total number of observations = 7 Total number of records with missing values = 0 Maximum number of iterations = 250 Relative Function Convergence has been set to: 1e-008 Parameter Convergence has been set to: 1e-008 User has chosen the log transformed model Default Initial (and Specified) Parameter Values background = 0 intercept = -29.967 slope = 3.76563 Asymptotic Correlation Matrix of Parameter Estimates (***The model parameter(s) - background have been estimated at a boundary point, or have been specified by the user, and do not appear in the correlation matrix.) Intercept Slope Intercept 1 -1 Slope -1 1 Analysis of Deviance Table Model Log (likelihood) No. Parameters Deviance Test Test d.f. P-value Full model -29.5498 7 Fitted model -32.7425 2 6.38533 5 0.2705 Reduced model -48.2628 1 37.426 6 <.0001 AIC: 69.485
Hydrogen Bromide 455 Parameter Estimates 95.0% Wald Confidence Interval Variable Estimate Standard Error Lower Conf. Limit Upper Conf. Limit Background 0 NA Intercept -27.4619 7.00164 -41.1848 -13.7389 Slope 3.45097 0.877253 1.73158 5.17035 NA: Indicates that this parameter has hit a bound implied by some inequality constraint and thus has no standard error. Goodness of Fit Scaled Dose Estimated Probability Expected Observed Size Residual 0.0000 0.0000 0.000 0 10 0.000 2205.0000 0.1855 1.855 1 10 -0.696 2328.0000 0.2397 2.397 4 10 1.188 2759.0000 0.4518 4.518 4 10 -0.329 3253.0000 0.6727 6.727 6 10 -0.490 3711.0000 0.8164 8.164 7 10 -0.951 3822.0000 0.8422 8.422 10 10 1.369 Chi Sq. = 5.02; DF = 5; P-value = 0.4134 Benchmark Dose Computation Specified effect = 0.05 Risk Type = Extra risk Confidence level = 0.95 BMC = 1774.18 BMCL05 = 1238.95 Probit 1 BMD Lower Bound 0.8 Fraction Affected 0.6 0.4 0.2 0 BMDL BMD 0 500 1000 1500 2000 2500 3000 3500 4000 dose 13:14 12/11 2007 FIGURE C-1 Probit model with 0.95 confidence level.
456 Acute Exposure Guideline Levels APPENDIX D ACUTE EXPOSURE GUIDELINE LEVELS FOR HYDROGEN BROMIDE Derivation Summary AEGL-1 VALUES 10 min 30 min 1h 4h 8h 1.0 ppm 1.0 ppm 1.0 ppm 1.0 ppm 1.0 ppm Key reference: Connecticut State Department of Health. 1955. Unpublished data. Occupational Health Section, CT Department of Health, Hartford, CT (as cited in ACGIH 2002) Test species/Strain/Number: Humans, six subjects Exposure route/Concentrations/Durations: Inhalation; 2, 3, 4, 5, or 6 ppm for several minutes Effects: Odor detectable for all six subjects at all concentrations 2 ppm: No nasal, throat, or ocular irritation. 3 ppm: Nasal and throat irritation in one of six subjects; no ocular irritation. 4 ppm: Nasal irritation in three of six subjects; throat irritation in one of six subjects; no ocular irritation. 5 ppm: Nasal irritation in all six subjects; throat irritation in one of six subjects; no ocular irritation. 6 ppm: Nasal irritation in all six subjects; throat irritation in one of six subjects; no ocular irritation.6 ppm: Nasal irritation in all six subjects; throat irritation in one of six subjects; no ocular irritation. End point/Concentration/Rationale: 3 ppm is considered a threshold for notable discomfort Uncertainty factors/Rationale: Total uncertainty factor: 3 Interspecies: 1, because key study is in human subjects Intraspecies: 3; the response to a direct irritant is not expected to differ greatly among humans (NRC 2001), and the resulting AEGL-1 value appears protective for asthmatics on the basis of data on HCl (NRC 2004). Modifying factor: Not applied Animal-to-human dosimetric adjustment: Not applicable Time scaling: Not applied; humans adapt to the slight sensory irritation. Data adequacy: Old but well-conducted study with human subjects. AEGL-1 value is supported by similar AEGL values for other chemicals in this class, HF and HCl. The databases on HF and HCl are robust.
Hydrogen Bromide 457 AEGL-2 VALUES 10 min 30 min 1h 4h 8h 250 ppm 83 ppm 40 ppm 10 ppm 5 ppm Data adequacy: The database on HBr is inadequate, so AEGL-2 values were derived by dividing the AEGL-3 values by 3. This is supported by the steep concentration-response curve observed in the lethality studies by MacEwen and Vernot (1972). AEGL-3 VALUES 10-min 30-min 1-hr 4-hr 8-hr 740 ppm 250 ppm 120 ppm 31 ppm 15 ppm Key reference: MacEwen, J.D., and E.H. Vernot. 1972. Toxic Hazards Research Unit Annual Technical Report: 1972. AMRL-TR- 72-62. AD 755-358. Aerospace Medical Research Laboratory, Wright-Patterson Air Force Base, OH. Test species/Strain/Number: Rat, Sprague-Dawley, 10 per group Exposure route/Concentrations/Durations: Inhalation; 2,205-3,822 ppm for 1 h Effects: Lethality: 2,205 ppm: 1/10 2.328 ppm: 4/10 2,759 ppm: 4/10 3,253 ppm: 6/10 3,711 ppm: 7/10 3,822 ppm: 10/10 End point/Concentration/Rationale: 1-h BMCL05 of 1,239 ppm Uncertainty factors/Rationale: Total uncertainty factor: 10 Interspecies: 3, a direct-contact irritant is not expected to vary greatly between species (NRC 2001) Intraspecies: 3, response to a direct-contact irritant is not expected to vary greatly among humans (NRC 2001) Modifying factor: Not applied Animal-to-human dosimetric adjustment: Insufficient data Time scaling: Cn à t = k; n = 1 on the basis of rat and mouse lethality data on HCl. Data adequacy: Although there were only two well-conducted studies of HBr in the rat and mouse, the values are consistent with those for the related chemicals, HF and HCl. The databases for HF and HCl are robust.