Assessment of Possible Carbon Dioxide Removal and Long-Term Sequestration Systems
This chapter reviews a number of systems to remove carbon dioxide from the atmosphere and reliably store it for long periods of time. Several techniques that integrate carbon capture and sequestration as a single inseparable process are described first: land management strategies, accelerated weathering on land and in the ocean, and ocean iron fertilization. This is followed by a discussion of two methods in which capture and disposal are separate: bioenergy with carbon capture and sequestration (BECCS) and direct air capture and sequestration (DACS). A table summarizing a number of aspects of these systems is presented at the end of this chapter. Other approaches have been suggested; however, the committee focuses here on techniques for which there is sufficient information to make a preliminary assessment.
Afforestation and Reforestation
Currently, global reforestation (the restoration of forest on recently deforested land) and afforestation (the restoration of forest on land that has been deforested for 50 years or more) create substantial carbon sinks, with net annual uptake of about 1 GtCO2 (Baumert et al., 2005). Deforestation, on the other hand, is the single largest source of land use-related greenhouse gas (GHG) emissions and accounts for about 10 percent of total current anthropogenic GHG emissions from all sources (and one-third of total cumulative emissions from all sources). As shown in Table 2.1, net land use emissions averaged 3.3 ± 2.9 GtCO2/yr between 2002 and 2011 and were dominated by tropical deforestation. Land use emissions since 1750 total about 660 GtCO2, which suggests an upper limit to the physical potential of reforestation and afforestation to remove carbon dioxide from the atmosphere. In reality, the number would be much lower because society needs to manage previously forested land to meet the need for food and fiber, and these managed systems typically have lower average carbon stocks than they did prior to conversion.
Until the early twentieth century, the highest rates of deforestation occurred in temperate forests in Asia, Europe, and North America. However, deforestation had essentially stopped in the world’s temperate forests by midcentury. As deforestation slowed in the temperate zone, it increased rapidly in the world’s tropical forests (FAO, 2010). Rates of deforestation in boreal forests tend to be lower than in tropical forests (Ruckstuhl et al., 2008). A critical component of any climate mitigation strategy is to prevent additional tropical deforestation, which as an outside limit could add as much as 1,800 GtCO2 to the atmosphere in cumulative emissions—roughly as much CO2 as from all the fossil fuel use from the preindustrial period until the present (Allen et al., 2009). Emissions from deforestation and land use change are about one-tenth of those from fossil fuels and cement production (see Table 2.1).
The rate at which carbon can be removed from the atmosphere through afforestation and reforestation is determined by a number of factors, including the age of trees, species composition, temperature, geology, precipitation, carbon dioxide concentration, and site history. The Intergovernmental Panel on Climate Change (IPCC) report, Land Use, Land-Use Change, and Forestry (IPCC, 2000), provides average annual net uptake rates associated with afforestation and reforestation activities of 1.5-4.5, 5.5-16, and 15-30 tCO2/ha for boreal, temperate, and tropical forests, respectively. The rate of net uptake typically reaches a maximum in 30-40 years, although the timing depends on biome type and site factors. After this initial phase, the rate of net uptake declines to zero as the forest matures, with the timing depending on forest type and structure (Ryan et al., 1997).
The IPCC Fifth Assessment reports potential carbon sequestration rates of up to 1.5, 9.5, and 14 GtCO2/yr in 2030 for global afforestation and reforestation activities, depending on the mitigation scenario (IPCC, 2014b, Table 11.8); these estimates are slightly higher than other estimates because they include CH4 and N2O in addition to CO2. Brown et al. (1996) estimated a maximum physical potential carbon sequestration rate of 4-6 GtCO2/yr for global afforestation and reforestation activities. Smith and Torn (2013) estimate that removing 3.7 GtCO2/yr through tropical afforestation would require at least 7 Mha/yr of land,1 0.09 Mt/yr of nitrogen, and 0.2 Mt/yr of phosphorus and would result in a 50 percent increase in evapotranspiration from this land; this is a better estimate of a feasible maximum rate of CO2 removal compared to earlier higher estimates. Nitrogen required for both BECCS (discussed below) and afforestation raises an additional concern: 1 percent to 5 percent of nitrogen fertilizer is converted to nitrous oxide, which has a global warming potential up to 300 times greater than CO2 (Crutzen et al., 2008; IPCC, 2013b). In one example in which this was further quantified,
_______________
1 For reference, the state of West Virginia has a total area of 6.3 Mha (U.S. Census Bureau, 2012).
the addition of inorganic fertilizer with subsequent N2O emissions can offset stored CO2 by 75 percent to 310 percent (Brown et al., 2004; Li et al., 2005; Robertson et al., 2000).
There are natural limits to the amount of carbon that can be removed from the atmosphere through reforestation and afforestation. When a forest ecosystem matures, the rate of CO2 uptake is balanced by respiration and the decay of dead organic matter. Based on land availability over the next 100 years, afforestation has been estimated to have a physical potential cumulative global impact of about 380 GtCO2 (Nilsson and Schopfhauser, 1995). Based on past soil carbon losses and the availability of land over the next 50 years, physical potential soil carbon sequestration estimates are between 110 and 180 GtCO2 (Lal, 2004).
Excluding deforestation, terrestrial ecosystems currently sequester carbon on a global scale, largely as a result of forest regrowth on lands previously cleared for agricultural use in the Northern Hemisphere and enhanced productivity in response to increasing carbon dioxide concentrations. It is unclear, however, how a changing climate will affect sequestration. If climate change results in widespread forest disease or accelerates the decomposition of carbon stored in soils, terrestrial ecosystems could become a net source rather than a sink of GHGs, further contributing to climate change (USGS, 2011, 2012). However, if climate or land use–induced transitions are more gradual, shifts in carbon stocks may not be large, even in the presence of major species shifts. The spatial scale of any accelerated disturbance regimes (e.g., fire, exotic pests and pathogens, or extreme weather) will determine if rapid loss of sequestered carbon is likely (USGS, 2011, 2012). Either way, the rate of additional sequestration of carbon in terrestrial ecosystems in the Northern Hemisphere will decline as afforested trees mature or are brought under management. Increasing atmospheric CO2 content also affects carbon sequestration. If CO2 fertilization of plants proves to have a substantial effect on carbon sequestration in forests, higher future CO2 concentrations may act to increase the effectiveness of afforestation and reforestation (Bala et al., 2007).
Biological sequestration in forests can be relatively inexpensive. In the United States, the cost of a program of 1.1 GtCO2/yr of forest sequestration has been estimated at $7.5/tCO2 to $22/tCO2 (Stavins and Richards, 2005), and at higher volumes of sequestration, the cost per ton is comparable to other abatement techniques. Another review of forest mitigation opportunities in the United States found that carbon prices from $1/tCO2 to $41/tCO2 generated an economic mitigation potential of 0.5 to 2.7 GtCO2 in total forest carbon (Richards and Stokes, 2004). A study by the U.S. Environmental Protection Agency (USEPA, 2005) suggested that, at $15/tCO2, the mitigation potential of afforestation and forest management in the United States would amount to
0.35 GtCO2/yr over a 100-year time frame. The IPCC Fifth Assessment reports potential carbon sequestration for global afforestation and reforestation activities at costs between $20/tCO2 and $100/tCO2, depending on the scale of the activity (IPCC, 2014b, Table 11.8). A major question is whether the true cost of preserving the forest for millennia is accounted for in the cost estimates, and whether the appropriate liabilities for accidental or intentional release of carbon by fire or future harvesting have been factored into the costs.
Although reforestation and afforestation projects remove CO2 from the atmosphere that would otherwise contribute to global climate change, the net climatic impact of additional forest growth is determined by the combination of carbon-cycle impacts with biogeophysical processes including albedo and hydrological impacts, which are coupled through cloud feedbacks, sensible and latent heat fluxes, and water vapor (Anderson et al., 2011; Bala et al., 2007; Bonan, 2008; Swann et al., 2010, 2012). In tropical forests, increases in tree growth may lead to an increase in evapotranspiration that can warm the atmosphere through the greenhouse effect but cool the atmosphere through enhanced cloudiness and albedo, as well as cool the land surface directly through evaporation. In boreal regions, planting trees on open land that is often covered by snow in wintertime decreases surface albedo, resulting in surface warming (Bonan, 2008). The net climatic effect of additional temperate and high-latitude forest sequestration is unclear. Model simulations by Swann et al. (2010) suggest that an increase in atmospheric water vapor from the growth of high-latitude deciduous forests in the future will have a warming effect 1.5 times larger than that due to changes in surface albedo, offsetting the impact of carbon uptake.
In the near term, the benefits of reducing deforestation are greater than that of reforestation and afforestation. In a study of seven developing countries, half of the cumulative mitigation potential of 23 GtCO2 between 2000 and 2030 could be achieved at a negative cost (Sathaye et al., 2001). Slowing or even ending deforestation is a CO2 mitigation strategy, but it is not considered carbon dioxide removal (CDR) since it does not result in a net decrease in atmospheric CO2.
Carbon Sequestration on Agricultural Lands
The use of land for agricultural production has led to a net transfer of terrestrial carbon to the atmosphere. It is estimated that, over the past 10,000 years, land conversion and land use caused soil carbon to decrease globally by 840 GtCO2 (Lal, 2001). On average, the amount of organic carbon in intensively cultivated soils is much lower than the potential carbon sequestration capacity below ground. Many cultivated soils have
lost 50 to 70 percent of their original organic carbon (IPCC, 2000), and that intensive soil cultivation has the potential to reduce soil carbon by 25 percent to 50 percent after 30-50 years (Johnson, 1992; Post and Kwon, 2000; Wei et al., 2014). Although it is difficult to compensate for the conversion of forests to cultivated lands, in part because most of the carbon in forest ecosystems is above ground, it is possible to manage agricultural lands to partially reverse the loss of carbon in some situations (Lal, 2007). Soil carbon can be increased by growing cover crops,2 leaving crop residues to decay in the field, applying manure or compost, using low- or no-till systems, and employing other land management techniques that increase soil structure and organic matter inputs.
Cover crops can be grown when a field is not planted with a market crop; they can increase organic matter inputs into the soil and have been found to increase soil carbon sequestration (Freibauer et al., 2004). Cover crops are also reported to decrease emissions of nitrous oxide and leaching losses of soil nitrate in some situations. In Iowa, double cropping, in which a food or feed crop such as maize is grown during its usual growing season, and a second crop is grown as an energy source at other times, was found to offer similar carbon benefits as the use of a conventionally managed sole-crop system while also producing 20 percent more dry biomass for bioenergy (Heggenstaller et al., 2008). The mitigation potential for this type of improved agronomy practice has been estimated to have a range of 0.07 to 0.7 tCO2-eq/yr per hectare, with significantly higher values in warm and moist climates (Smith et al., 2007). Such efforts could be sustained for a decade or so before uptake rates would level off as soil carbon content approached steady state.
Most farmers, both in the United States and globally, plow fields before planting, which increases decomposition rates through the “priming effect” with the net effect of releasing carbon from the soil into the atmosphere until a lower equilibrium is established. Switching to no- or low-till practices has the potential for increased carbon sequestration in soil. Marland et al. (2003) conclude that, for the average U.S. farm, a change from conventional tillage to no-till agriculture will result in net soil carbon sequestration that averages 1.2 tCO2/yr per hectare for the first 20 years with a decline to near zero in the following decades. More recent analyses suggest that no-till agriculture results in some net sequestration of soil carbon, but the amount of carbon stored is much smaller (about a 5 percent increase in soil carbon) and less consistent than previously believed (Baker et al., 2007). Another important point is that the benefits of no-till agriculture may be reversed by reintroducing tilling. Thus, for no-till practices to
_______________
2 Often leguminous crops such as bean, lentil, and alfalfa (Thiessen-Martens et al., 2005).
be used effectively as a form of long-term carbon sequestration, the practice must be maintained without interruption.
The greatest per-hectare emissions of CO2 from agricultural soils have occurred on cropland created by the drainage of wetlands and the lowering of water tables by installation of drainage systems often referred to as “tiling” (Fargione et al., 2008). Prior to cultivation, these lands were rich in organic carbon due to anoxic conditions in hydric soils. Both draining and tiling allow oxygen to enter deeper into these soils, greatly increasing the rate at which organic matter is decomposed to carbon dioxide. Smith et al. (2008) note that raising water tables and converting cropland back to wetlands can lead to “rapid accumulation of soil carbon” but may also increase releases of methane, a potent GHG. The mitigation potential of improved water management activities is estimated to be between −0.6 and 3 tCO2-eq/yr per hectare (Smith et al., 2007).
Although intensively managed annual croplands lose much of their pre-agricultural soil carbon, well-managed pastures retain most of their soil carbon (Guo and Gifford, 2002). The rate at which soil carbon increases in former croplands is reported to be greater when they are planted with diverse mixtures of both grass and legume species (Guo and Gifford, 2002; Tilman et al., 2006). Over a 10-year period, a low-input, high-diversity bioenergy crop grown on low-nutrient-status agricultural soils had a total sequestration rate of 4.4 tCO2/yr per hectare in soil and roots, although the research suggests that this rate might decline to 3.3 tCO2/yr per hectare with time because of slower root mass accumulation (Tilman et al., 2006). This is contrasted with a lack of carbon accumulation in previously agriculturally disturbed soils in New England with up to 120 years of reforestation (Compton and Boone, 2000). Programs that set aside agricultural land can increase net carbon sequestration and provide wetland, stream, river, and lake protection, although indirect land use impacts (i.e., the creation of farmland in other regions or countries to offset the land set aside) should be considered (Plevin et al., 2010).
Most of the estimates in this section are on a per-hectare basis. Of the total 13 billion hectares that make up Earth’s ice-free surface, cropland accounts for ~12 percent, pastureland ~26 percent, forest land ~32 percent, and urban land ~9 percent (Foley et al., 2011). The global technical potential for agricultural land management is 5.2 GtCO2/yr in 2030 (IPCC, 2014b). The carbon removal potential of these techniques will need to be balanced with food production needs and other co-benefits and side effects will need to be factored in. This is also true for biochar, which is another technique for affixing carbon (Box 3.1).
Another possible method of enhancing carbon sequestration is to store biomass, such as crop or forest residues, in the ocean. As described by Strand and Benford (2009) and
BOX 3.1 BIOCHAR
Biochar refers to a broad class of products in which biomass (e.g., trees, grasses, and crop residuals) is combusted at moderately low temperatures (300°C to 600°C) without oxygen through low-temperature pyrolysis. The pyrolysis process allows for the formation of charcoal, a relatively stable form of organic carbon, thereby slowing the inevitable release of CO2 into the atmosphere due to decomposition when compared with adding the organic matter to the soil directly. The residence time of biochar in situ is not well established (Gurwick et al., 2013). Although there has been research associated with the role biochar could play in carbon and nitrogen dynamics, the literature is still limited, and the impacts of utilization on net greenhouse gas emissions are not well defined (Gurwick et al., 2013). Since biochar is seen as largely responsible for reducing emissions by decreasing decomposition of waste plant material through the potential long-term sequestration of the carbon in the soil, it is not classified in the current work as a CDR technology. Further complicating consideration of biochar as a CDR technology is the fact that pyrolysis produces less net useable energy per unit of carbon emitted to the atmosphere than does combustion of the same material (Gaunt and Lehmann, 2008). Additionally, changing the temperature and speed of the pyrolysis process can influence the mechanism by which the char forms and the stability of the resulting char (Milosavljevic et al., 1996).
Combusting waste biomass to produce energy would displace more fossil fuel and reduce net greenhouse gas emissions to a greater degree than using that material as a feedstock for biochar production. If fossil fuel use has been eliminated in the area where the biomass is produced and energy needs are not being fully met, then combusting waste material to produce bioenergy would produce lower net greenhouse gas emissions than would production of biochar. If additional energy is not needed to meet human needs, then biochar production will reduce net greenhouse gas emissions relative to allowing that waste to decompose. If the deployment of biochar requires additional mixing of the soil, the priming effect discussed previously with regards to no-till agriculture will result in increased oxidation of organic material in the soil and a concomitant increase in carbon dioxide emissions over the short to medium term. Despite not being among the CDR approaches, biochar does have benefits to agricultural practices such as improving soil structure (water and fertilizer retention), removing contaminants, and enhancing fertility in degraded soils.
Metzger and Benford (2001), by packaging and sinking land biomass into the deep ocean, especially in areas low in oxygen, the normal return of this carbon via decomposition and respiration is greatly impeded if not eliminated.
Summary of Land Management Approaches
Looking forward, there are several important future research directions that deserve consideration, together with their potential for negative ecological impacts:
- Systems analysis to develop strategies for afforestation and reforestation efforts alongside biomass and food production with minimal competition for land and maximum CDR potential;
- Development of technologies for advanced ammonia fertilizer production with lower energy requirements and related CO2 emissions (it is important to note that increased nitrogen application can result in higher rates of denitrification and N2O production, a potent greenhouse gas); and
- Engineering plant varieties that are better able to remove carbon dioxide and reliably store it for extended periods, for example, by developing plants that achieve higher photosynthetic rates than native vegetation under extreme conditions (e.g., minimal water, “nonarable” land) to limit competition with food and/or biomass production.
In summary, land management approaches—reforestation, afforestation, and changed management practices for agricultural lands—are mature technologies that are readily deployable with well-known environmental consequences. In total, they have the potential to remove significant but limited amounts of CO2 from the atmosphere (i.e., ~380 GtCO2 total out to the year 2100 at a maximum rate of between 2 and 5 GtCO2/yr for afforestation and reforestation), with a comparable potential sequestration rate from changed agricultural practices. The costs for afforestation and reforestation are generally low compared to other CDR techniques, that is, approximately $1/tCO2 to $100/tCO2 (IPCC, 2014b; Richards and Stokes, 2004; Stavins and Richards, 2005). The maximum potential for total CO2 removal from the atmosphere is on the order of the total amount that has been removed from terrestrial ecosystems by human activities—roughly 660 GtCO2, equivalent to a reduction of 40-70 ppm in atmospheric CO2 concentration by 2100 (House et al., 2002). Implementation of these techniques is unlikely to achieve anything close to this maximum potential due to the increasing demands for agricultural production and the difficulty of reaccumulating carbon on depleted landscapes. Though these techniques are clearly not a solution by themselves, they can be valuable elements of a climate change mitigation portfolio.
ACCELERATED WEATHERING METHODS AND MINERAL CARBONATION
The long-term fate for most CO2 released to the atmosphere is first to become bicarbonate ions dissolved in the ocean and later to become carbonate sediments on the sea floor (Berner et al., 1983). These transformations occur as a result of ions provided by carbonate and silicate weathering reactions that typically occur in soils or marine sediments. One class of CDR involves accelerating these carbonate and/or silicate weathering reactions so that CO2 may be stored in the ocean predominately in the
form of bicarbonate ions or stored in the ocean or on land in the form of a calcium carbonate solid (Dunsmore, 1992; Geerlings and Zevenhoven, 2013; Hartmann et al., 2013; Lackner, 2002, 2003; Olajire, 2013; Sanna et al., 2014; Stephens and Keith, 2008).3 In principal, these weathering reactions could be accelerated by bringing high CO2 concentrations in contact with appropriate naturally occurring rock formations, creating carbonate minerals in situ. Alternatively, they could be accelerated by transporting the appropriate minerals for processing in an industrial setting. Last, the appropriate minerals could be ground up, transported, and released into the ocean.
Carbon dioxide released into the atmosphere exchanges with carbon in the land biosphere and ocean on timescales ranging from seconds to millennia. However, as seawater absorbs CO2 from the atmosphere, it becomes more acidic, and this inhibits further absorption. The dissolution of calcium carbonate minerals either on land or in the ocean neutralizes some of this acidity and thus allows the seawater to absorb more CO2 (Archer et al., 2009); CO2 in addition to calcium carbonate (CaCO3) and water yields calcium ions (Ca2+) and bicarbonate ions (HCO3–) in solution:
![]()
When added to the ocean, the dissolved calcium and bicarbonate ions increase the alkalinity of seawater. It typically takes 2,000 to 8,000 years for reaction (1) to return the ocean-surface sediment carbonate system naturally to steady state following a perturbation such as the release of carbon dioxide into the atmosphere. (Equilibration with both the silicate and carbonate mineral cycles takes much longer, on the order of hundreds of thousands of years.) The long timescale associated with carbonate sediment equilibration arises in part because of slow ocean transport of dissolved carbon dioxide and because of the rates of the natural calcium carbonate cycle that involves weathering on land and deposition in marine sediments (Archer et al., 2009). Thus, one set of concepts involves strategies to accelerate the weathering reaction [Eq. (1)] (Harvey, 2008; Rau, 2011; Rau and Caldeira, 1999). The basic idea of these proposed strategies is that—if CO2 additions are going to eventually dissolve calcium carbonate minerals in the ocean and in so doing reduce both the atmospheric load of CO2 and the amount of ocean acidification caused by the CO2—it should be possible to accelerate carbonate dissolution reactions so as to achieve these perceived benefits more rapidly.
Silicate weathering reactions can also affect marine chemistry in a way similar to dissolution of carbonate minerals. However, because silicate minerals do not in general
_______________
3In the discussion here, for simplicity, the committee discusses calcium with the understanding that other divalent cations, such as magnesium, are also possible.
contain carbon, twice as much carbon can usually be stored in the ocean from weathering reactions with silicate minerals as compared with carbonate minerals per mole; that is, two moles of CO2 react with one mole of calcium silicate mineral (CaSiO3) and water, yielding calcium ions and bicarbonate ions in solution plus silica (SiO2):
![]()
In nature, it typically takes hundreds of thousands of years for reaction (2) to return the ocean-surface sediment silicate system to steady state (Caldeira and Rampino, 1990), but various strategies have been proposed to accelerate this reaction (Köhler et al., 2010, 2013; Schuiling and de Boer, 2011; Schuiling and Krijgsman, 2006).
The long-term fate for most CO2 released into the atmosphere is to become carbonate sediments in the ocean, where the cations in the carbonate minerals are derived from silicate-mineral weathering reactions. Schematically, this reaction,4 in which CO2 reacts with a silicate mineral to become a carbonate mineral plus silica, may be written as
![]()
Reaction (3) can form a solid carbonate. Note, however, that in this simplified representation, twice as much CO2 can be removed from the atmosphere if the resulting solution is allowed to be disposed of in the ocean [reaction (2)] relative to what would occur were a solid to be formed [reaction (3)] and disposed of directly as a solid.
All three of these weathering reactions, (1), (2), and (3), have been discussed as the basis for possible mechanisms for removing CO2 from the atmosphere at a large scale. It should be noted that there are no “silver bullets” in any of these accelerated weathering approaches. In reaction (1), the amount of calcium carbonate mass required is 2.3 times as large as the mass of CO2 removed.5 Similarly, for reactions (2) and (3), the silicate mineral mass must exceed the CO2 mass by a factor of 1.3 or 2.6, respectively, and for reaction (3), the mass of the resulting solids (calcium carbonate plus silica) will exceed the mass of CO2 by a factor of 3.6.6 The use of other silicate minerals, such as olivine, can potentially improve these ratios, but deployed at scale, all of these methods would involve mining of substantial masses of mineral—on the order 100 billion tons/yr to offset current CO2 emissions (~34 GtCO2/yr; see Table 2.1). For comparison, U.S. production of crushed stone or coal is about 1 billion tons/yr, and total world production of coal is about 8 billion tons/yr (USGS, 2013a). If the atmospheric CO2 is to
_______________
4These reactions should be interpreted as simplified archetypes of reactions as reactions actually used may be considerably more complicated.
5 CaCO3 is 100 g/mole and CO2 is 44 g/mole.
6 For reaction (2) 116 g of CaSiO3 would be needed for each 88 g of CO2; for reaction (3), 116 g of CaSiCO3 would be needed for each 44 g of CO2 and would produce 100 g of CaCO3 and 60 g of SiO2.
be stored in the form of a solid carbonate mineral (e.g., CaCO3), then simple examination of the elemental composition indicates that the mass of the minerals to be stored must be at least ~2.3 times the mass of the CO2. If year 2013 CO2 emissions (~36 GtCO2; Le Quéré et al., 2014) were entirely stored in the form of CaCO3, this would represent over 80 billion tons of carbonate mineral. Transport and disposal of a substantial fraction of this mass could pose formidable challenges. Furthermore, many weathering reactions are favored in relatively dilute solutions, so the volumes of water needed could in some cases be substantial (Rau and Caldeira, 1999), although not all approaches require the movement of water (Harvey, 2008; Kheshgi, 1995; Köhler et al., 2013).
Reactions similar to those listed above have been discussed in the context of carbon capture from large point sources of CO2, such as electricity generation or cement manufacturing facilities (IPCC, 2005). Examples of proposals to use accelerated mineral weathering approaches at such large point sources can be found in a wide range of sources (Béarat et al., 2006; Chizmeshya et al., 2007; Gerdemann et al., 2007; House et al., 2007; Kirchofer et al., 2012; O’Connor et al., 2004; Park and Fan, 2004; Park et al., 2003; Rau, 2011). Under the definitions used in the current work, carbon sequestration from such point sources would be considered “climate engineering” if the CDR was associated with BECCS or DACS. Such facilities at scale would require substantial amounts of mass handling. For example, Rau and Caldeira (1999) and Rau (2011) estimate that about 5,000 to 10,000 tons of water would need to be pumped for each ton of CO2 stored. Thus, these approaches favor coastally located facilities where there is ready access to seawater.
Another approach is to encourage carbonate or silicate mineral weathering reactions to occur on land (Köhler et al., 2010; Schuiling and Krijgsman, 2006) or in the ocean (Harvey, 2008; Köhler et al., 2013; Schuiling and de Boer, 2011) rather than in a centralized facility. These approaches involve crushing and distributing minerals over a broad area so that chemical weathering reactions may be accelerated by generating high amounts of reactive surface area. Such approaches involve substantial amounts of transportation and distribution of materials to have a substantial climate effect (Hangx and Spiers, 2009). An important issue is that the near-surface ocean is saturated with respect to most carbonate minerals, and the kinetics of silicate mineral dissolution are usually slow. Kheshgi (1995) suggests that by being more selective in the materials mined, or by preprocessing the mined minerals to create more soluble chemicals, compounds can be added to the near-surface ocean that would dissolve and therefore cause the ocean to take up more carbon dioxide from the atmosphere. Harvey (2008) suggests that these goals could be achieved by sinking a fine carbonate mineral powder from the surface ocean with the aim of dissolving it in undersaturated waters below. Schuiling and Krijgsman (2006) suggest silicate mineral reaction rates
could be accelerated by grinding minerals finely and then spreading them on farmlands or forests, or in the coastal ocean. A variety of electrochemical approaches to accelerating mineral weathering have also been proposed, although required electricity inputs would be substantial (House et al., 2007; Rau, 2008; Rau et al., 2013).
The approaches described above focus on bringing carbonate or silicate minerals to locations where they may react with carbon dioxide. Another strategy is to bring carbon dioxide to where it may react in situ with naturally occurring minerals. Natural uptake of CO2 by olivine has been documented in Oman (Kelemen et al., 2011; Matter and Kelemen, 2009). These studies indicate the need for improved understanding of fundamental CO2-reaction fluid-mineral interactions for mineral carbonation (Gadikota and Park, 2014; Gadikota et al., 2014a,b), which would also be relevant for understanding the fate of CO2 once it is injected into geologic formations containing silicate minerals.
As previously noted, accelerated chemical weathering approaches typically aim to dispose of (store) carbon in one of two forms, either as a solid carbonate mineral or as dissolved bicarbonate in the ocean. A seawater solution containing dissolved CO2 accompanied by added alkalinity (i.e., increasing Ca2+) stores nearly twice as much CO2 per unit of mineral dissolved; however, use of the ocean raises a range of legal and ethical issues (discussed in Chapter 4).
Scaling and Environmental Issues
Carbonate minerals, silicate minerals, and seawater are all abundant and so there are no obvious fundamental physical constraints that limit the application of these approaches at the global scale. Indeed, carbonate and silicate weathering reactions will be the way that nature slowly and eventually removes anthropogenic CO2 from Earth’s exchangeable surface reservoirs over thousands to hundreds of thousands of years. However, there are substantial real-world constraints that suggest a limited role for markedly accelerating these weathering reactions. First, as noted previously, widespread application of these approaches would require a substantial scale-up of carbonate or silicate mining, and some approaches require the use of large volumes of seawater. For many of these proposals, the large material requirements involved likely limit economically foreseeable applications to locations where appropriate minerals are coastally located.
Beyond the effects of mining and transporting so much mineral material, there are a range of environmental concerns associated with the use of the ocean. For point-source applications, there are concerns about environmental damage resulting from
the intake of large volumes of water. Depending on the ratio of CO2 to alkalinity added to the seawater, there is a potential for these approaches to increase ocean pH and carbonate mineral saturation and thereby to counter some adverse environmental effects of ocean acidification. To have substantial effects on ocean carbonate chemistry at a global scale would involve mining and crushing hundreds of cubic kilometers of carbonate and/or silicate minerals. For comparison, in 2011, worldwide coal production was equivalent to about 9 km3 (USGS, 2013a); associated mineral mass movement is likely to have been several times greater. There is also some concern about environmental consequences of adding CO2-rich alkaline fluids to the ocean. Although there is no evidence of deleterious effects of adding alkalinity to waters that have been acidified as a result of excess CO2, adding alkalinity to seawater does not remove the excess CO2 and so is not going to restore the status quo; thus, there is potential for unanticipated ecological consequences.
Looking at the entire process of possible accelerated weathering CDR strategies, a recent study carried out by Kirchofer et al. (2012) investigated the impact of alkalinity source on the life-cycle energy efficiency of mineral carbonation technologies; see Figure 3.1. The life-cycle analysis (LCA) of aqueous mineral carbonation suggests that a variety of natural and industrial byproduct-based alkalinity sources and process configurations have the potential to achieve net CO2 reductions. Natural silicate minerals (e.g., olivine and serpentine) were chosen due to their environmental abundance and widespread global availability (Krevor et al., 2009). Due to the slow kinetics of silicate dissolution in addition to the mining and grinding efforts associated with natural alkalinity sources, industrial byproducts (i.e., fly ash, cement kiln dust, and iron and steel slag) may be more reactive but are much less abundant. The LCA examined the fluxes of energy, solids, water, and CO2 for the processes of extraction (e.g., mineral mining), reactant transportation, preprocessing (e.g., grinding), chemical conversion, postprocessing, product transportation, and disposal or reuse. An example of the CO2 emissions per 1,000 tCO2 stored for mineral carbonation processes with net CO2 mitigation potential is shown in Figure 3.2. As an example, cement kiln dust (CKD) may have reasonable mitigation potential, with associated emissions of about 150 tCO2 for every 1,000 tCO2 stored. However, considerations of life-cycle CO2 emissions must be tempered with consideration of availability of reactant. A review of alkaline industrial wastes such as fly ash, CKD, steel slag, and red mud indicated that in the United States fly ash is most abundant (130 million tons/yr), followed by CKD (~18 million tons/yr), steel slag (~8-10 million tons/yr), and red mud (<5 million tons/yr) (Gadikota and Park, 2014).
Kirchofer et al. (2013) also investigated the CO2 mitigation potential of mineral carbonation with industry-based available alkaline sources in the United States. CO2
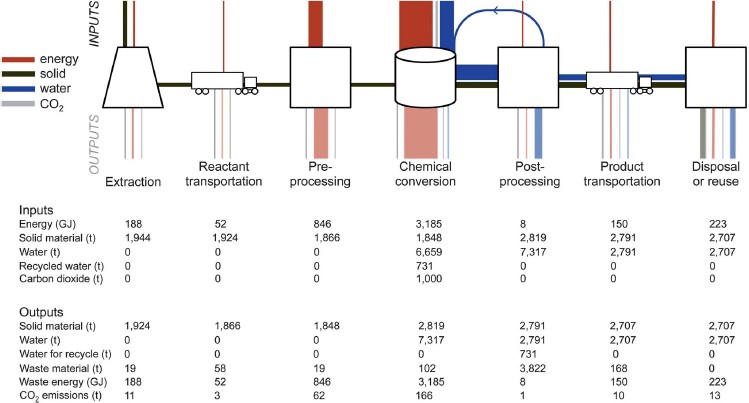
FIGURE 3.1 Life-cycle process model schematic that shows all of the steps associated with mineral carbonation of 1,000 tCO2 with mined and crushed olivine, which is a silicate mineral rich in Mg2+. This schematic shows the number of components required for mineral carbonation, including mineral extraction, transportation, preprocessing (e.g., grinding), chemical conversion, postprocessing, product transport, and disposal. The thickness of lines is scaled to the energy and mass fluxes (inputs enter from the top, outputs leave through the bottom). This process does not include the separation of CO2, and it is questionable whether atmospheric CO2 is in great enough concentration to achieve adequate conversion. It is likely that the CO2 would have to be concentrated to some extent to improve mineral carbonation conversion on timescales of interest. Taking into account the total energy (4.65 GJ/tCO2) as shown for each step results in a cost of ~$1,000/tCO2 provided coal is the electric energy source. SOURCE: Kirchofer et al., 2012.
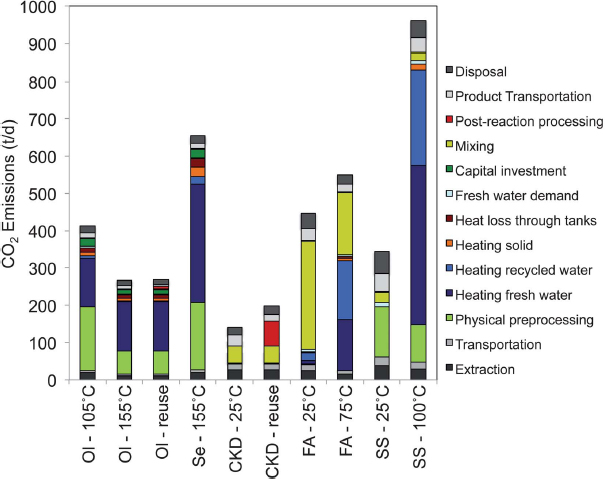
FIGURE 3.2 CO2 emissions per 1,000 tCO2/day stored for mineral carbonation processes with net CO2 mitigation potential (Kirchofer et al., 2012). Ol, olivine; Se, serpentine; CKD, cement kiln dust; FA, fly ash; SS, steel slag. Temperatures refer to reaction temperatures for chemical mixing. Emissions from using cement kiln dust for a mineral carbonation process are estimated to be the lowest, where storing 1,000 tCO2/day results in 105 tCO2/day of emissions, or a net mitigation of almost 900 tCO2/day.
point-source emissions are typically several orders of magnitude greater than the total available industrial byproduct alkalinity in locations across the country. This study found that U.S. industrial alkaline byproducts have the potential to mitigate about 7.5 MtCO2/yr, of which 7 MtCO2/yr is captured via mineral carbonation, and about 0.5 MtCO2/yr is from the avoided emissions associated with the replacement of naturally mined aggregate. Unfortunately, this is only about 0.1 percent of U.S. CO2 emissions. Including natural and industrial-sourced alkalinity yields a maximum potential of ~1.5 GtCO2/yr in the United States (Kirchofer et al., 2013); multiplied out over the rest of the century this gives a total potential of ~130 GtCO2 out to 2100.
In situ accelerated weathering, in which CO2 reacts with available alkalinity in Earth’s surface, may also provide a potentially significant means of sequestering CO2. For instance, Kelemen and Matter (2008) estimate that there is an approximate sequestration capacity of 1 trillion tons of CO2 within 3 km of the surface in the Sultanate of Oman through mineral carbonation of peridotite. Alternatively, mineral carbonation with alkalinity present below the seafloor is interesting to note, although the feasibility of such an approach may be questionable. In particular, Kelemen et al. (2011) report that approximately 1,000 trillion tons of CO2 may be mineralized in a section 10 km wide by 3 km deep along the world’s slow-spreading ridges.
The accelerated weathering concepts explored in this section are the result of theoretical explorations and limited laboratory testing. No demonstration or pilot plants exist to date. Nevertheless, the underlying geochemistry of chemical weathering and the relevant characteristics of global biogeochemical cycles are well established (Berner and Berner, 2012). Most of the engineering is straightforward—mining, crushing, and distributing minerals, or chemical engineering processes that are routinely done at laboratory scale. However, although some scaling estimates have been made (e.g., Harvey, 2008; Ilyina et al., 2013), many issues of scaling have not yet been investigated.
The large mass required if these strategies were to be deployed at a scale commensurate with the climate problem is clearly a major barrier. Proposals that rely on the ocean as a disposal site also face potential ecological and legal challenges. The legal status of such proposals under the London Convention and London Protocol is unclear (see discussion in Chapter 4). Because adding alkalinity to the ocean also helps to counteract ocean acidification, it is thought that direct biological consequences could be positive; however, no field studies have tested this hypothesis.
Because these accelerated chemical weathering approaches are relatively low-tech in their fundamental concept, it should be possible to get improved cost estimates for accelerated chemical weathering facilities and operations. These cost estimates would need to take into account geographically specific conditions; the costs of mined minerals and their transportation are likely to comprise a substantial fraction of overall cost (Figure 3.1) for ocean-based accelerated weathering, whereas land-based accelerated weathering is substantially more expensive to achieve significant impact, as previously discussed.
If such approaches are seriously contemplated, it would be important to first conduct experiments in which marine organisms or ecosystems are exposed to seawater with
the chemistry that would be expected to result from such operations.7 For proposals that involve spreading minerals on land, it would be useful to have experiments and analyses aimed at understanding what long-term application would do to these soils and the ecosystems living thereon; also, downstream impacts on streams and rivers would need to be considered.
The committee highlights several important future research directions:
- Investigations into cost-effective methods of enhancing the kinetics of carbonate and silicate mineral dissolution (or other chemical transformations) for CO2 conversion to bicarbonate or carbonate; potential approaches include mineral pretreatment, enhancement of acid-base reactivity, synergies with biotic activity, enzymes, and electrochemistry;
- Experiments and modeling to determine the environmental benefits, impacts, and fate of (bi)carbonate addition to soils, watersheds, and the ocean;
- Better determining the environmental impacts of mineral extraction and seawater pumping (where needed), especially relative to downstream environmental benefits and relative to the impacts of other CDR methods;
- Testing and modeling various approaches at meaningful scales to better determine the life-cycle economics, net cost/benefit, optimum siting, and global capacities and markets of accelerated mineral weathering in the context of CDR.
In summary, only laboratory-scale experiments of ocean-based accelerated weathering have been carried out thus far. Further research at meaningful scales could help assess concerns related to economics, global capacity, and associated environmental and sociopolitical risks. However, this technology is currently only at an intermediate level, and this approach may have significant environmental and sociopolitical risks since it concerns the ocean. This approach has the potential of cumulative CDR of ~100 GtCO2 out to the year 2100 at a rate of ~1 GtCO2/yr with estimated costs in the range of $50/tCO2 to $100/tCO2 (McLaren, 2012; Rau et al., 2013).8 Land-based mineral carbonation approaches have been investigated at limited scale as well and are likely also at an intermediate technology level, but they have minimal sociopolitical risks, except for risks associated with the mining and disposal of large masses of material. Intermediate environmental risks may exist due to the uncertainty of the effects of
_______________
7 The results of such experiments could be compared to expected effects on organisms and ecosystems from increasing pH due to ocean acidification.
8 Rau et al.’s estimate includes an ocean-land requirement of <7 × 105 km2/GtCO2 captured per year and assumes wind as an energy resource; the total of 85 GtCO2 assumes approximately 1 GtCO2/yr for 85 years until 2100.
mining large masses of minerals, in the case of ex situ mineralization, and injection of large amounts of alkalinity and CO2, in the case of in situ mineral carbonation. Land-based approaches using silicate minerals have been estimated to have a potential capacity of roughly 4 GtCO2/yr with an estimated cost of $23/tCO2 to $66/tCO2 (IPCC, 2014b; Rau and Caldeira, 1999; Rau et al., 2007). In considering ex situ mineral carbonation, these low cost estimates do not consider all steps of preparation and utilization of CO2 and the minerals as outlined in Figure 3.1. Taking into account the total energy (4.65 GJ/tCO2) as shown for each step results in a cost of ~$1,000/tCO2 provided coal is the electric energy source (Kirchofer et al., 2012).
A natural biological pump exists in the sea: planktonic algae and other microscopic plants take up CO2 at the ocean surface and convert it to particulate organic matter. Some of this organic matter settles into the deep ocean and serves as food for animals, bacteria, and other microorganisms that respire and reverse the reaction, converting organic carbon back to CO2, which is re-released at depth. The net result of the biological pump is to sequester inorganic carbon in the deep ocean and thus maintain a lower preindustrial atmospheric CO2. Numerical modeling studies suggest that variations in the magnitude and geographic patterns of the biological pump could drive changes in atmospheric CO2 of a few tens to perhaps more than 100 ppm over timescales of several decades to centuries (Marinov et al., 2008; Sarmiento and Gruber, 2006). To a first-order approximation, the present-day biological pump is thought to be in steady state and does not materially influence the concentration of anthropogenic CO2 in the atmosphere, and the current rate of ocean uptake of anthropogenic CO2 is governed by physical-chemical processes and ocean circulation (Sabine and Tanhua, 2010).
The strength of the marine biological pump and resulting ocean carbon sequestration depends, among other factors, on the quantity of the phytoplanktonic nutrients nitrogen and phosphorus in the global ocean and the completeness with which the supply of these nutrients to the surface ocean are utilized by phytoplankton. There are several mechanisms by which a natural or deliberate human perturbation of the biological pump could potentially enhance the net uptake and ocean sequestration of CO2 from the atmosphere. First, if a limiting nutrient like nitrate or phosphate is added to the ocean from an external source, the utilization of that nutrient by primary producers would increase the net formation of organic matter. That additional organic material would ultimately be exported to the ocean interior and respired as CO2, thus increasing deep-ocean CO2 sequestration. Second, there are regions in the ocean where some
of the nutrients brought from depth to the surface are not consumed before they are returned to depth by ocean circulation. If the efficiency of nutrient utilization in those regions, primarily in the Southern Ocean, were to be somehow enhanced, more carbon would be stored in the intermediate and deep ocean. Third, if the elemental ratio of carbon to nutrients in organic matter were to increase from the average value at present, then the net new flux of carbon to depth would also increase. Fourth, a reduction in the biological formation of particulate inorganic carbon in the surface ocean would increase surface alkalinity and enhance ocean carbon sequestration. Finally, most of the organic matter produced by plankton is respired in the upper few hundred meters of the water column, with only a small fraction reaching the mid-depth to deep ocean where the respired CO2 is isolated from the atmosphere for many decades to centuries because of the relatively slow overturning circulation of the ocean. In model simulations, increasing the depth where sinking particles are respired back to CO2 results in increased ocean carbon sequestration (Kwon et al., 2009). These scenarios are not mutually exclusive and could arise because of changes in ocean circulation, external nutrient and trace-metal inputs, and plankton food-web dynamics. One perturbation will be climate change forced by the combustion of fossil fuels.
In a future warmer world, climate change will almost certainly alter ocean circulation and stratification, which in turn may also affect the aforementioned biological processes that are critical to the biological pump (Sarmiento et al., 1998). Model simulations suggest that the changes in ocean physics and biology may be sufficient to reduce by a small degree the ocean’s ability to remove anthropogenic CO2 and store inorganic carbon (Arora et al., 2013). Some studies have suggested that climate change is already reducing ocean carbon uptake at least regionally (e.g., Le Quéré et al., 2009), but this relatively small long-term climate effect is difficult to discern robustly from the limited available historical and present-day observations (McKinley et al., 2011).
Approaches have been proposed to increase the strength of the biological pump (through increasing either the size of nutrient reservoirs or the degree to which they are used) by deliberately adding nutrients to fertilize ocean plankton. The large quantities of nitrogen and phosphorus that must be added to the ocean to significantly affect atmospheric CO2 render this approach far less practical than iron fertilization, reflecting the fact that the organic matter formed by plankton has a relatively low ratio of carbon to either nitrogen or phosphorus (for example, the carbon-to-nitrogen ratio is only about a factor of 5 to 8). Instead, the focus has been on more modest additions of the essential micronutrient iron because of the large ratios of carbon to iron in planktonic organic matter (1,000 to more than 100,000 on a mole/mole basis; Boyd et al., 2007).
The basic principal behind ocean iron fertilization (OIF) is that by adding iron to surface waters in some specific regions of the ocean, one could stimulate increased growth by phytoplankton, which would increase the completeness with which the natural supplies of nitrogen and phosphorus are used in those waters, increasing the flux of organic carbon into the deep ocean. Under an appropriate set of conditions, the enhancement of the biological pump would result in CDR from the upper ocean and atmosphere and sequestration in the subsurface ocean (Martin, 1990). A primary focus is on the high-latitude surface waters of the Southern Hemisphere that typically have abundant macronutrients (e.g., nitrogen and phosphorus) but low chlorophyll and phytoplankton growth—particularly of large cells that lead to carbon export—relative to other nutrient-abundant regions, because of limitation by low surface iron levels (Martin and Fitzwater, 1988). This discovery resulted in proposals to influence the biological pump’s effect on ocean anthropogenic CO2 uptake through the deliberate addition of iron to the ocean surface (Box 3.2). The Southern Ocean contains the largest area of iron-limited conditions and is the focus of many discussions on ocean iron fertilization approaches; other iron-limited regions, including the subpolar North Pacific and eastern Equatorial Pacific, have been the sites of scientific field experiments on iron addition and are often included in numerical simulations of ocean iron fertilization methods.
Other related ocean biological CDR approaches have been proposed but have been studied in less detail than ocean iron fertilization (Williamson et al., 2012). Fertilization with surface addition of macronutrients, such as bioavailable nitrogen in the form of urea as well as phosphate (Lampitt et al., 2008), has the advantage that it can be applied in low-latitude, nutrient-poor surface waters and has possible co-benefits because of enhanced biological productivity. However, as already noted, there are drawbacks relative to micronutrient fertilization because of the much larger mass requirements associated with the plankton biological needs of nitrogen and phosphorus relative to carbon. Another proposed alternative would be to artificially enhance ocean upwelling of subsurface nutrients with some form of active pumping method using, for example, wave-driven pipes (e.g., Lovelock and Rapley, 2007). Artificial upwelling has also been suggested as a carbon sequestration method for some specific ocean regions where the supply of excess phosphorus could stimulate nitrogen fixation (Karl and Letelier, 2008). Beyond issues of the technical feasibility of ocean pipes and the resulting cooling of the ocean surface, the major drawback from a CDR perspective is that any upwelled subsurface water with enriched nutrients would also have elevated CO2 levels that would effectively cancel most, if not all, of the benefit of biological carbon drawdown (Oschlies et al., 2010; Yool et al., 2009).
BOX 3.2 HISTORICAL CONTEXT OF OCEAN IRON FERTILIZATION
“Give me half a tanker of iron, and I’ll give you an ice age,” biogeochemist John Martin reportedly quipped in a Dr. Strangelove accent at a conference at Woods Hole in 1988 (Fleming, 2010). Martin and his colleagues at Moss Landing Marine Laboratories proposed that iron was a limiting nutrient in certain ocean waters and that adding it stimulated explosive and widespread phytoplankton growth. They tested their iron deficiency, or “Geritol,” hypothesis in bottles of ocean water, and subsequently experimenters added iron to the ocean in a dozen or so shipborne “patch” experiments extending over hundreds of square miles (see text for discussion). OIF was shown to be effective at inducing phytoplankton growth, and the question became this: Was it possible that the blooming and die-off of phytoplankton, fertilized by the iron in natural dust, was the key factor in regulating atmospheric carbon dioxide concentrations during glacial-interglacial cycles? Dust bands in ancient ice cores encouraged this idea, as did the detection of natural plankton blooms by satellites.
This realization led to further questions. Could OIF speed up the biological carbon pump to sequester carbon dioxide? And could it be a solution to climate change? Because of this possibility, Martin’s hypothesis received widespread public attention. What if entrepreneurs or governments could turn patches of ocean green and claim that the carbonaceous carcasses of the dead plankton sinking below the waves constituted biological “sequestration” of undesired atmospheric carbon? Several companies—Climos,1 Planktos (now out of the business), GreenSea Ventures, and the Ocean Nourishment Corporation2—have proposed entering the carbon-trading market by dumping either iron or urea into the ocean to stimulate both plankton blooms and ocean fishing (Climos, 2007; Freestone and Rayfuse, 2008; Powell, 2008; Rickels et al., 2012; Schiermeier, 2003).
OIF projects could be undertaken unilaterally and without coordination by an actor out to make a point; in fact, one such incident took place off the coast of Canada in 2012 (Tollefson, 2012). However, as this section describes, there are still unresolved questions with respect to the effectiveness and potential unintended consequences of large-scale ocean iron fertilization.
_______________
An extensive series of small-scale iron release experiments have shown that artificially adding iron to high-nitrate, low-chlorophyll regions in the Equatorial Pacific and Southern Ocean does cause increased phytoplankton growth rates and the development of phytoplankton blooms (Boyd et al., 2007; de Baar et al., 2005). Mesoscale iron fertilization experiments also have demonstrated that a shift toward larger phytoplankton species, in particular diatoms, occurs and that the short-term ocean drawdown of atmospheric carbon dioxide increases to varying degree (Coale et al., 1996; Pollard et al., 2009). Collecting evidence of increased sinking of particulate carbon has proved more elusive, in part because of limitations on the duration and scope of field
experiments to date (Buesseler and Boyd, 2003). Few studies have measured well the changes in particle fluxes and respiration rates in the subsurface ocean below a bloom because experiments ended before the bloom terminated or because the patch of fertilized water had expanded to cover a much broader area, making it more difficult to observe changes using sediment traps. Thus, the effect on long-term CO2 drawdown and increase in ocean carbon sequestration in the interior of the ocean is not well documented and appears to vary substantially across experiments and ocean regions, with examples of both minimal and large sinking particle flux events associated with specific experiments (Martin et al., 2013; Smetacek et al., 2012).
An iron-fertilized increase in sinking organic matter will not necessarily translate directly into a comparable increase in the rate of long-term ocean inorganic carbon sequestration. Much of the sinking organic matter flux due to an iron fertilization–induced bloom will be respired back to CO2, nutrients, and dissolved iron by bacteria and zooplankton in the upper few hundred meters of the water column, and ocean circulation will carry the resulting excess CO2 back to the ocean surface, where it can be released back to the atmosphere on relatively short timescales of a few years to decades, unless there is sufficient iron available to support biological transformation of the excess CO2 back into organic matter (Robinson et al., 2014). Therefore, an important factor is the degree to which the iron released at depth during organic matter respiration remains in the water column or is removed to the sediments through scavenging and particle export. Rapid iron scavenging would imply that ocean fertilization would need to be continued essentially indefinitely to result in permanent carbon disposal from the atmosphere. Alternatively, if a substantial amount of the added iron that sinks with and is released from respired organic particles is not scavenged from subsurface waters, it could limit the escape of the excess CO2 to the atmosphere when the subsurface water returns to the ocean surface and could extend the duration of enhanced ocean carbon sequestration due to iron fertilization. Enhanced long-term carbon sequestration, typically defined as a duration of more than 100 years, would also occur from the small fraction of sinking particles that reach intermediate or deep waters (greater than 1,000 m).
Because of the large natural background levels and variability of subsurface dissolved inorganic carbon, the direct measurement of small changes in ocean carbon sequestration at depth from ocean iron fertilization experiments is challenging. Furthermore, it is not possible in the field to track the subsequent fate of water parcels for sufficiently long time to quantify the rate of return to the surface ocean. Therefore, estimates of the efficiency of iron fertilization on ocean carbon sequestration are restricted so far to numerical model studies that require a number of assumptions about biological dynamics and iron biogeochemistry. With these caveats in mind, modeling
studies indicate that the potential upper limit for a sustained ocean iron fertilization CO2 sink is relatively modest at 1.0 to 3.7 GtCO2/yr9 and that the total ocean sequestration capacity until the end of the century is 85 to 315 GtCO2, assuming continuous iron fertilization of the entire iron-limited Southern Ocean, Equatorial Pacific, and subpolar North Pacific (Aumont and Bopp, 2006; Zahariev et al., 2008).
Early cost estimates for ocean iron fertilization were quite low (<$10/tCO2), reflecting the large leverage of the amount of iron added per organic carbon fixed via photosynthesis (e.g., Ritschard, 1992). However, more recent studies factor in new information, suggesting lower biological efficiency leading to carbon export and sequestration and leakage of CO2 back to the atmosphere (Markels et al., 2011). For example, one estimate of the cost of ocean iron fertilization is approximately $450/tCO2 (Harrison, 2013). Improved cost estimates would also require information on technological issues (e.g., iron spreading and approaches to limit scavenging), the efficiency of atmospheric CO2 uptake, and verification and monitoring requirements.
Studies have identified a number of possible drawbacks to iron fertilization as a CDR method (Buesseler et al., 2008; Strong et al., 2009; Williamson et al., 2012). In particular, the ecological impacts on the marine food web and fisheries due to continuous, extensive iron fertilization may be substantial but are poorly characterized. It is also likely that iron fertilization will have downstream effects on nutrient supply, and thus productivity and food web dynamics, in other ocean regions. An intended consequence of ocean iron fertilization involves shifting plankton community composition toward larger cells that will lead to enhanced downward-sinking flux; the long-term impact of this shift on higher trophic levels, including fish, seabirds, and marine mammals, is not well known but may be addressable in part by studying analogous regions with substantial natural iron fertilization. Iron addition often stimulates the growth of Pseudonitzschia diatom species, some of which are associated with toxin-producing harmful algal blooms (Moore et al., 2008). In the case of a specific iron addition experiment in the subpolar North Pacific Ocean, the iron-stimulated Pseudonitzschia diatoms were shown to produce domoic acid, a neurotoxin that has the potential to harm fish, marine mammals, and humans (Trick et al., 2010).
A number of scientific studies have raised concerns about how ocean iron fertilization may potentially also alter ocean biogeochemistry. Changes in the air-sea fluxes of climate-active trace gases such as dimethylsulfide, methane, and nitrous oxide (N2O) could in principle either partially cancel out or amplify the benefits from enhanced ocean CO2 uptake (Diaz and Rosenberg, 2008). A substantial component of ocean N2O
_______________
9 Only two significant figures reported here.
production is thought to arise from microbially driven nitrification of ammonia and organic nitrogen released from sinking particles in the upper ocean. Nitrification is expected to increase due to iron fertilization, and because N2O is a much more powerful greenhouse gas than CO2, the effect could be to greatly diminish the climate impact of iron fertilization (Barker et al., 2007; Jin and Gruber, 2003). There is also the potential for the release of methyl halides to the atmosphere that might lead to possible depletion of stratospheric ozone (Wright, 2003). Increased export of organic carbon to the subsurface ocean would also likely reduce local subsurface dissolved oxygen levels, exacerbating the declines in subsurface oxygen already expected under a warmer climate. A resulting expansion of low-oxygen, hypoxic regions of the coastal or open ocean would potentially have significant biological ramifications (Keeling et al., 2010). Iron fertilization on a large scale could potentially also have downstream effects by reducing the nutrient supply to low-latitude ecosystems. Although ocean iron fertilization would act to remove CO2 from the surface ocean and transport it to depth, the effects on partially mitigating ocean acidification in surface waters due to rising atmospheric CO2 levels would be minimal at best and would somewhat increase the rate of acidification of subsurface waters (Cao and Caldeira, 2010). In addition to these concerns over the effectiveness and environmental impacts of OIF projects, there are significant ethical and legal concerns as well. These are discussed further in Chapter 4.
Looking forward, the committee highlights several important future research directions:
- Understanding the effectiveness of iron inputs on stimulating biological organic carbon production and increasing carbon export;
- Determining the fate of the sinking organic carbon and iron in the subsurface ocean as a result of deliberate ocean iron fertilization;
- Assessing potential downstream effects that may limit biological productivity or change other aspects of biogeochemistry in other regions;
- Detection and accounting of net changes in subsurface ocean carbon sequestration and the effective lifetime of the carbon sequestration; and
- Understanding the ecological and biogeochemical consequences of extended and large-scale iron fertilization.
In summary, current limitations of ocean iron fertilization as a viable CDR method include the limited knowledge regarding the method’s effectiveness in regard to carbon capture, concerns regarding the environmental impacts and cost of large-scale and sustained OIF, and the associated ethical and legal issues. Although about a dozen ocean iron fertilization field experiments have been conducted, their purpose was fundamental scientific research primarily related to the basic controls on ocean biology and biogeochemistry. Many unresolved issues remain regarding scalability, ef-
ficacy, verification, and environmental impacts. Given these limitations and unknowns, the committee concludes that the risks and costs currently outweigh the benefits. The committee considers this an immature CDR technology with high technical and environmental risk.
BIOENERGY WITH CARBON CAPTURE AND SEQUESTRATION AND DIRECT AIR CAPTURE AND SEQUESTRATION
Bioenergy with Carbon Capture and Sequestration
BECCS is a process in which biomass is converted to heat, electricity, or liquid or gas fuels, followed by CO2 capture and sequestration. The BECCS cycle (Figure 2.1) begins with plants assimilating CO2 from the atmosphere via photosynthesis with sufficient sunlight, water, and nutrients (e.g., bioavailable nitrogen and phosphorus or fertilizers) as additional inputs. The biomass is then used in either an energy generation (electricity or process heat) or chemical process plant, thereby creating CO2 and water vapor. Biomass also can be used to produce liquid fuels such as ethanol or methanol, gas fuels such as hydrogen, or engineered algal systems designed to directly produce hydrocarbons. The CO2 is captured in a similar manner to how it would be captured from point-source emitters firing coal or natural gas.10 To form liquid fuels, the synthesis gas would be catalytically reacted through a Fischer-Tropsch process.11 The formation of alcohols, polymers, and various carbon-based chemicals is also possible through this catalytic process. Formation of liquid fuels does not cause a net sequestration of carbon; it involves chemical conversion for use as an energy source and emission to the atmosphere.
Current estimates show that if BECCS were deployed to its theoretical maximum feasible amount, it could account for a significant portion of the world’s energy supply. Literature estimates for bioenergy potential range from 50 to 675 EJ/yr (Berndes et al., 2003). Many integrated assessment models (Azar et al., 2010; Rao et al., 2008; Riahi et al., 2011; Thomson et al., 2011) assume large-scale bioenergy usage by the end of the century, in the range of 150 to 400 EJ/yr.
Both the availability of land for biomass cultivation and the need to transport bulky biomass to processing facilities severely limit the feasible use of bioenergy. The higher
_______________
10 Capture technologies from point-source emitters (e.g., coal- and natural gas–fired power plants) include absorption via amine scrubbing (or other chemical solvent), adsorption, and membrane technologies for pre- and postcombustion applications (Wilcox, 2012).
11 A Fischer-Tropsch process is a series of chemical reactions that converts gas-phase carbon monoxide and hydrogen into liquid hydrocarbons.
reported estimates of energy from bioenergy, 200 to 400 EJ/yr (Azar et al., 2010),12 assume that diets change dramatically in response to increasing carbon prices, because these costs become embedded into land rents and food prices, leading to a shift from products with high land requirements, such as beef, to products with lower land requirements, such as grains (Wise et al., 2009). It is assumed that these effects are not undercut by the dramatically increasing growing global population or by increased global affluence. Edmonds et al. (2013) report that reduced herd sizes have the potential to free up 4.5 million km2 of pastureland and 1.2 million km2 of cropland,13 allowing for the expansion of bioenergy production. To put this into perspective, 200 EJ/yr (Azar et al., 2010) is roughly equal to current world oil consumption (190 EJ/yr) and represents ~40 percent of today’s global energy production (550 EJ/yr).14 In these scenarios, about 80 to 100 EJ/yr is derived from byproducts of agriculture and forest industries (Azar et al., 2010), with the remaining 180 to 300 EJ/yr coming from dedicated energy crops that require land, water, and nutrients. Biomass growth at this scale requires extensive land area. More specifically, 100 EJ/yr may require up to 500 million hectares of land, assuming an average biomass yield of 10 tons of dry biomass per hectare annually. For comparison, about 1,600 million hectares are currently planted with agricultural crops, and an additional 3,400 million hectares are used for pasture (FAO, 2010). Global food demands are projected to nearly double over the next 50 years (Tilman et al., 2001), which will, in the absence of dramatic yield increases or diet changes, put energy crops in direct competition with food crops for arable land. There is no empirical evidence that the globe is inclined to move away from animal agriculture; rather, demand for meat is increasing globally (Foley et al., 2011).
Large-scale deployment of BECCS would have risks and complications; it is not materially relevant until such time as fossil fuel use is limited and linking CCS with bioenergy use has a net benefit to the climate. Prior to that point, there is no difference in net carbon emissions to the atmosphere whether the CCS is tied to bioenergy or fossil fuel use. Large-scale expansion of biomass plantations may displace forests that have significant biodiversity that the new growth would lack. Primary forests tend to have greater biodiversity than secondary ones (Barlow et al., 2007; Lindenmayer and Hobbs, 2004; Zurita et al., 2006), and restored grasslands and forests are known to have reduced biodiversity compared to neighboring native ecosystems (Camill et al., 2004). In addition, large old-growth forests and undisturbed grasslands have significant
_______________
12 For reference, Azar et al., (2010) report that 100 EJ/yr from bioenergy, if used in conjunction with carbon capture and sequestration (CCS), would remove 2.5 GtC (9.2 GtCO2).
13 For reference, the state of Alaska has a total area of 1.7 million km2.
14http://www.eia.gov/cfapps/ipdbproject/IEDIndex3.cfm?tid=44&pid=44&aid=2; http://www.eia.gov/cfapps/ipdbproject/iedindex3.cfm?tid=5&pid=5&aid=2&cid=regions&syid=2008&eyid=2012&unit=QBTU.
amounts of carbon sequestered, and conversion to other land uses usually leads to large greenhouse gas emissions such that it would take decades or more to provide a net reduction in the atmospheric carbon dioxide stock as a result of bioenergy on these lands (Creutzig et al., 2012; IPCC, 2011c; Mitchell et al., 2012; Tilman et al., 2009).
Smith and Torn (2013) focused on using switchgrass specifically as the biomass feedstock for BECCS and report 200 million hectares of land (20 times the area currently used for U.S. bioethanol production), 20 Tg/yr of nitrogen (20 percent of global fertilizer production), and 4,000 km3/yr of water (equal to current global water withdrawals for irrigation and 4 percent of total renewable water resources) would be required to remove 1 PgC/yr (3.7 GtCO2/yr). Hence, adoption of bioenergy reliance at this scale will be constrained by available land and resources and the secondary impacts on greenhouse gas emissions (e.g., N2O). One area of research is to identify energy crops with lower water, nutrient, and energy requirements and the capacity to grow on marginal agricultural lands (Heaton et al., 2008; McLaughlin and Walsh, 1998; Msangi et al., 2007).
According to Kriegler et al. (2013), the costs associated with BECCS are lower than the most optimistic DACS15 case (Lackner, 2010) up to a removal of 12 GtCO2/yr, and then the costs increase abruptly due to biomass supply limitations. To put this number into context, CO2 emissions from fossil fuel combustion were 31.6 Gt in 2011 (IEA, 2011). They argue that when BECCS approaches levels of between 13 and 14 GtCO2/yr, it will be outcompeted by DACS in terms of cost. Their model (ReMIND) also assumes a sequestration potential of 3,670 GtCO2 with an injection rate of 0.5 percent per year, which results in an upper bound of 18 GtCO2/yr. Although this capacity of CO2 sequestration is consistent with Dooley’s (2013) “practical” capacity estimates (3,900 GtCO2), it is important to keep in mind that today with the existing five CCS projects in place (see below section, Geological Sequestration of Carbon Dioxide), sequestration is only taking place on the order of MtCO2/yr. In addition, the IEA 2013 CCS Roadmap (IEA, 2013b) estimates that an increase to ~7 GtCO2/yr through 2050 is required in order to prevent a 2°C increase in warming, among other strategies including nuclear power, efficiency and fuel switching, and renewables. Hence, it is uncertain whether the injection and sequestration of 18 GtCO2/yr is a reasonable estimate. Furthermore, these studies are misleading since none of the cost estimates include compression or sequestration, but only capture. In addition, it is important to keep in mind that there are many challenges associated with accurately determining sequestration potential and that geological sequestration technologies are still in their infancy (see Benson et al. [2012] and section Geological Sequestration of Carbon Dioxide, below). The seques-
_______________
15 See next section, Direct Air Capture and Storage.
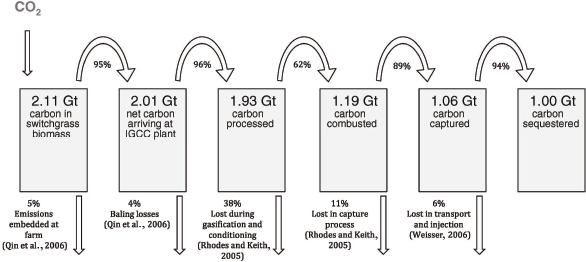
FIGURE 3.3 BECCS carbon flow. Carbon losses upon processing switchgrass to a useful fuel. SOURCE: Smith and Torn, 2013.
tration potential of CO2 may ultimately dictate the viability and impact of BECCS and DACS as CDR approaches.
Additionally, an important aspect of BECCS to consider is the CO2 emissions associated with the energy required to process the biomass for gasification or combustion. In the absence of CO2 sequestration, bioenergy from biomass is not inherently a carbon-neutral process. Figure 3.3 shows an example of the process of gasifying switchgrass. Initially the switchgrass has 2.1 GtC (7.7 GtCO2), but after drying, processing, and gasifying, there is a resulting 1.06 GtC (3.9 GtCO2) separated from the synthesis gas and finally 1.00 GtC (3.67 GtCO2) that will ultimately become stored. Hence, storing 1 GtC (3.67 GtCO2) requires fixing 2.1 GtC (7.7 GtCO2) considering the carbon losses along the life cycle of the process. The high carbon-to-energy ratio of bioenergy feedstocks (roughly equal to that of coal and half that of natural gas for dry biomass) and the decrease in net energy resulting from the combustion of bioenergy feedstocks with a high moisture content mean that, in the most common situation, there is lower net reduction in GHG emissions relative to using the same CCS capacity with fossil fuel–generated energy, particularly natural gas–generated energy. If the amount of fossil fuel and bioenergy burned is held constant there is no net reduction in atmospheric CO2 stocks if CCS is deployed to sequester bioenergy (BECCS) versus fossil fuel–generated carbon dioxide. BECCS is important as a CDR approach once fossil use is limited, and CCS capacity can be used effectively to drive energy emissions net negative.
Looking forward, the committee highlights several important future research directions:
- Small-scale boiler redesign for co-firing natural gas with biomass and
- Advanced technologies for drying biomass at the recovery site to minimize water transport costs and heating inefficiencies.
In summary, the technological readiness of BECCS is similar to that of CCS. The use of biomass as an energy feedstock as practiced in the United States is currently dominated by its use for process heat (EIA, 2013a). Further advances in boiler and gasification technologies will assist in the potential for increased bioenergy use. Similarly, the costs are on the order of conventional CCS at ~$60/tCO2 to $250/tCO2 unless less expensive alternative CO2 capture and sequestration methods can be used. The cumulative CDR theoretical potential of BECCS is large at >1,000 GtCO2 removed and stored by 2100 (Kriegler et al., 2013; Lenton and Vaughan, 2009) at a rate of 15 to 18 GtCO2/yr (Azar et al., 2010; Kriegler et al., 2013). (Note that the rate in the near term, i.e., out to 2050, may only be up to 3 to 10 GtCO2/yr [IPCC, 2014b].) However, that potential is likely to be significantly constrained for some time, if not indefinitely, by the need for most arable land to be used to meet global food demand and the competing demand to use global CCS capacity to sequester fossil fuel emissions.
Direct Air Capture and Sequestration
Direct air capture (DAC) refers to chemical scrubbing processes for capturing CO2 directly from the atmosphere via absorption or adsorption separation processes. Although other abiotic (and biotic) processes can also directly remove CO2 from air, DAC is distinguished by producing concentrated CO2 as its end product. Following CO2 capture, the material used to carry out the separation (e.g., amine- or hydroxide-based sorbents) must then be regenerated; this leads to the production of a near-pure stream of CO2, which can be used (e.g., enhanced oil recovery, chemical production, or other uses) or sequestered.16 The separation technologies for DAC are similar, but not necessarily identical, to those used in conventional CCS, in which CO2 is captured from point sources where CO2 concentrations are much higher, such as coal-fired power plants or chemical plants producing ammonia or ethylene oxide. Although chemical absorption-based separation employing amines for point-source capture of CO2 is well established (the first patent was filed in 193017), it is not clear that this technol-
_______________
16 The process of DAC with sequestration is referred to as DACS.
17 Chemical scrubbing using amine-based absorption is often referred to as the current state-of-the-art technology for point-source capture of CO2 and is a technology that has been ongoing since the first patent filed by Bottoms in 1930.
ogy will be the primary solution for the required scale of significant CO2 reductions due to its negative environmental impacts, water requirements, and moderately high cost. Solvent-based approaches to chemically scrubbing CO2 out of the atmosphere are considered here without focus on solid sorbents due to the infancy in adsorption-based processes compared to solvent-based processes for CO2 separation. There has yet to be a study carried out that involves a detailed cost analysis of an adsorption process from capture to regeneration of CO2 from the atmosphere.
More specifically, the primary difference between DACS and CCS is that the CO2 concentration in air is 100 to 300 times lower than in the flue gas of a gas- or coal-fired power plant, respectively. The more dilute a system is, the more energy intensive the capture or separation process is. As shown in Figure 3.4, the minimum amount of energy required to capture CO2 from air is 2 to 10 times the amount required to capture CO2 from point sources. For this and related reasons, the cost of capturing CO2 from air will be higher than from point sources, and DACS is likely to become attractive only after CCS has been widely implemented.
There are other important differences between DAC and point-source capture. The design of an absorbing unit for DAC is likely to be large in terms of its cross-sectional area, but very shallow due to pressure-drop limitation requirements (Figure 3.5), whereas a similar unit for point-source capture is likely to be tall and potentially thin by comparison (Figure 3.6). For example, a 500-MW coal-fired power plant with a plant size of about 15 ha18 emits on average 11,000 tons of CO2 per day. Using current state-of-the-art technology based on amine scrubbing, capturing 90 percent of the CO2 (i.e., 10,000 tons) requires 2 ha, or 13 percent of the footprint of the power plant. Alternatively, capturing 10,000 tons of CO2 per day directly from the air, assuming an air flow rate of 2 m/s, requires about 15 ha, equal to the land area of a 500-MW power plant (EPRI, 2010). Capital costs generally scale with land area; that is, more units will be required to capture the same amount of CO2 and will generally require more land area. The energy required (shown in Figure 3.4) generally relates to operating and maintenance costs, where overall costs are the sum of capital costs plus operating and maintenance costs.
Costs of DAC vary in the literature significantly due to the different underlying assumptions factored into the costs (APS, 2011; Holmes and Keith, 2012; House et al., 2011; Mazzotti et al., 2013). In particular, the studies of the American Physical Society (APS), Holmes and Keith, House et al., and Mazzotti et al. are the only ones considered in this cost comparison since they represent the few works that explicitly outline
_______________
18 Roughly the size of 15 football fields.
FIGURE 3.4 Comparison of minimum work for CO2 capture for various capture percentages and purity percentages for applications spanning the extremely dilute atmosphere to the concentrated fuel gas of coal gasification. The more dilute a system is, the greater the energy required for separation. For instance, direct air capture is more energetic than separation from natural gas combustion, which is more energy intensive than separation from coal combustion flue gas (Wilcox et al., 2014).
whether capture and/or regeneration are included in their cost estimates. Although adsorption-based approaches have also been carried out for DAC applications (Choi et al., 2011; Wang et al., 2011), these approaches are not considered explicitly in this report since they have yet to be presented at the demonstration scale in detail in the peer-reviewed literature in a detailed enough fashion. Once CO2 is captured, the sorbent or solvent used must be regenerated for reuse, producing a near-pure stream of CO2 for pipeline compression. Table 3.1 highlights several studies from the literature with the underlying assumptions considered in the cost estimates. For instance, House et al.’s $1,000/ton estimate is based on the first and second laws of thermodynamics, assuming 90 percent capture and 95 percent purity combined with a Sherwood analysis based on the dilution of CO2 in the atmosphere. In addition, this cost assumes the energy source is CO2 free since using natural gas or coal would result in greater

FIGURE 3.5 Carbon Engineering’s slab air-contactor design is shown as an example of the design of a DAC plant. The surface area is optimized to achieve maximum air contact for reasonable CO2 capture, and the width of the column is shallow to minimize pressure drop and subsequent energy requirements. Comparing with Figure 3.6, it is clear that the design and footprint of a separation system is dependent on the starting CO2 concentration. SOURCE: Holmes and Keith, 2012 (top); Carbon Engineering, Ltd. (bottom).

FIGURE 3.6 Conventional amine solvent plant for CO2 separation at the National Carbon Capture Center in Wilsonville, Alabama. This is a demonstration plant that bridges technologies from the bench scale to the pilot scale, and the absorber unit that is currently in place captures 3,650 tCO2/yr. In contrast to the air contactor used for extremely dilute air capture, this flue-gas contactor (absorption column) can be tall for increased CO2 separation due to the inherent driving force of the flue-gas stream. This image provides an example for dimensional comparison to the DAC plant in Figure 3.5, not a comparison of scale as the annual removal rate by the National Carbon Capture Center is small since it is for demonstration purposes only. SOURCE: Courtesy of Frank Morton, Business Development Manager of the National Carbon Capture Center, Southern Company.
TABLE 3.1 Comparison of Assumptions and Costs of DAC in the Literature
| Cost [$/tCO2 captured] | ||||
| Capture | Regeneration | Total | Assumptions | Reference |
| Yes | Yes | 1,000 |
Calculation based on minimum work. |
House et al., 2011 |
|
Capture and regeneration included. |
||||
| Yes | Yes | 376-600 |
Optimization case study. Counterflow contactor. Considered air velocity, liquid velocity, and recovery as decision variables. Capture and regeneration included. |
APS, 2011; Mazzotti et al., 2013 |
| Yes | No | 60 |
Optimization case study. Cross-flow contactor. Air velocity and mass-transfer coefficient as decision variables. Only capture included. |
Holmes and Keith, 2012 |
CO2 emissions than the CO2 captured. Because this estimate is based on the minimum work required to separate CO2 from a gas mixture, capture and regeneration are both inherently included. In the case of the APS report and the more recent work of Mazzotti et al., both capture and regeneration are included in their estimates, which range from $400/tCO2 to $600/tCO2 captured. The study by Mazzotti et al. was an optimization based on a case study that assumed capture would take place in a conventional absorption process (tall and thin tower as shown in Figure 3.6) with the flue gas and solvent contacting in a counterflow configuration. The decision variables considered in their optimization were air and liquid velocities and percent capture. In the recent work of Holmes and Keith, only the cost of capture was considered ($60/tCO2 captured) with a cross-flow air contactor (high cross-sectional area and thin unit as shown in Figure 3.5) and air velocity and the mass-transfer coefficient being the decision variables in their optimization procedure. Again, given these differences in assumptions and the decision to focus on just half of the story in some cases, it is difficult to directly compare these estimates. By comparison, the cost of CO2 capture from a coal-fired power plant is about $100/tCO2 (Al-Juaied and Whitmore, 2009; Deutch and Moniz, 2007; DOE, 2010; IPCC, 2005).
None of the cost estimates above include compression or sequestration, which is required for DAC to be a CDR technology. In addition, since there has not been a DAC plant built to capture CO2 to date, costing such a design is a difficult task; such a system may look quite different from that used to capture CO2 from more concentrated sources.
Similar to BECCS, in order for DACS to be a viable component for reducing global warming, the sequestration capabilities have to be well defined. Reservoir quality, proximity to capture plant, and injection rates will all dictate the feasibility, capacity, and rate associated with the CDR from a DACS approach. In addition, safety, public perception, and sequestration reliability will all be primary factors (further discussion of geological sequestration is in the next section). Also, alternative uses of the concentrated CO2 need to be considered, for example, its conversion via accelerated mineral weathering to solid carbonate or dissolved bicarbonate for stable ocean sequestration (see Accelerated Weathering Methods and Mineral Carbonation). One advantage of DACS over CCS and BECCS is that capture equipment can be sited close to sequestration or utilization19 sites (if the CO2 is to be utilized) without regard to considerations that influence power plant siting (e.g., fuel supply and electricity transmission).
Overall, looking forward, the committee highlights several important future research directions in direct air capture:
- System optimization that couples material properties for CO2 separation to the process properties;
- In terms of technological advancements, determining if overlap in separation technologies exists between dilute versus concentrated CO2 sources;
- Alternative CO2 conversion, sequestration, or use options other than underground injection of concentrated CO2; and
- Systems analysis between DAC plant design coupled to noncarbonized energy resources such as solar and wind.
In addition, a possible alternative to DAC for which further research could provide benefits is the internal consumption of or the extraction of CO2 from seawater (Box 3.3).
In summary, DAC is an immature technology with only laboratory-scale experiments carried out to date and demonstration-scale projects in progress, with limited public results (see, for example, Choi et al., 2011; Holmes and Keith, 2012; Lackner, 2009; it is too early for peer-reviewed studies to have documented the performance of
_______________
19 Utilization includes enhanced oil recovery (EOR), exhaust gas recirculation, and chemical production.
BOX 3.3 SEAWATER CAPTURE
As shown in Figure 2.1, the ocean contains 50 times more carbon than the atmosphere, and about 28 percent of the CO2 emitted by humans is dissolved in the ocean (see Table 2.1). The concentration of carbon in seawater is more than 100 times the concentration of carbon in air per unit volume (100 mg/L versus 0.8 mg/L in air), but 6 times less per unit mass (100 mg/kg versus 600 mg/kg). For natural seawater (pH ~ 8), most of the dissolved carbon is in the form of bicarbonate ion (HCO3−). Consuming CO2 in seawater via in situ biological means (see Ocean Iron Fertilization) or by chemical or geochemical reactions (see Accelerated Mineral Weathering with Land-Ocean Sequestration) causes the partial pressure of CO2 (pCO2) in that seawater to decline. As ocean pCO2 drops below the pCO2 of air, CO2 will passively diffuse from air into the ocean and will mostly equilibrate into other forms, principally HCO3−, thus removing and sequestering air CO2. Alternatively, CO2 can be extracted from seawater by heating, placing under a vacuum, purging or bubbling with a non-CO2 gas, or acidification via means other than CO2 addition. In this vein, Eisaman et al. (2012) demonstrated the extraction of nearly 60 percent of the dissolved carbon in seawater in the form of CO2 using bipolar membrane electrodialysis. Following removal of CO2 from seawater, the pCO2 of the remaining seawater would be reduced and hence would become a sink for atmospheric CO2. The electrochemical energy consumption for this nonoptimized process was experimentally observed to be about 240 kJ/mol. Additional energy would be required to pump seawater through the plant. Although this is more than 10 times that given in Figure 3.4 for DAC (~20 kJ/mol), this is a measured value rather than a theoretical minimum. Such energy requirements are substantially lower than the 400 to 1,000 kJ/mol estimated for DAC systems (APS, 2011; House et al., 2011; and references therein). Other marine electrochemical approaches have been proposed (House et al., 2007; Rau, 2008; Rau et al., 2013), with estimated energy expenditures at scale of <300 kJ/mol CO2. In general seawater capture is much less technologically mature than air capture, so research in this area could yield potential benefits.
some other systems). An additional limitation is the energy-intensive nature of this approach, making it cost prohibitive compared to point-source CO2 capture. Cost estimates including both CO2 capture and sorbent regeneration range between $400/tCO2 and $1,000/tCO2 (House et al., 2011; Mazzotti et al., 2013).20 Point-source CO2 capture costs are currently lower, but costs for reducing emissions from distributed sources (e.g., replacing large numbers of cars with electric vehicles) may be considerably higher. Benefits of DAC are the flexibility associated with plant placement in addition to its minor environmental impacts. As with BECCS and conventional CCS, DAC needs to be coupled with sequestration in order for negative emissions to take place (Figure 2.2). In addition, the energy source for DAC needs to be free of CO2
_______________
20 The wide range of estimates stems from including both capture and sorbent regeneration steps.
emissions for this approach to be optimally CO2-emissions-negative. The annual and cumulative CDR potential up to 2100 was considered for the United States only. Assuming that solar energy is used to fuel the DAC process and that ~100,000,000 acres of Bureau of Land Management (BLM) land are available in the southwestern United States, this could lead to a removal of ~13 GtCO2/yr and a cumulative removal of ~1,100 GtCO2 up to 2100 (see Table 2.2 as well).
Geological Sequestration of Carbon Dioxide
The sequestration of CO2 is directly connected with BECCS, DACS, and CCS technologies, as previously discussed. Once CO2 is captured, it must be sequestered to prevent its return to the atmosphere. Depleted hydrocarbon reservoirs and saline aquifers are the primary options for geological sequestration of CO2. EOR and coal-bed methane recovery are utilization techniques that inadvertently store some CO2, but for the most part the CO2 used in these processes is recovered for subsequent reuse (see Utilization of Carbon Dioxide and Available Markets). Research is ongoing as to whether CO2 might be used as an enhancement fluid for gas recovery from shale (Firouzi et al., 2014; Heller and Zoback, 2014). To give a sense of scale, cumulative emissions are on the order of 2,000 GtCO2, which corresponds to a volume of approximately 2,300 km3 (equivalent to Lakes Erie and Ontario combined21).
Total capacity estimates show that geological sequestration has the potential to sequester large amounts of CO2. In Global Energy Assessment: Toward a Sustainable Future, Benson et al. (2012) estimate that global sequestration capacities for depleted oil and gas reservoirs are ~1,000 GtCO2 for coal beds up to 200 GtCO2 and sequestration in saline aquifers is highly variable between 4,000 and 23,000 GtCO2 (Benson et al., 2012). A recent study by Dooley (2013) provides updated geologic sequestration capacities, with a global “theoretical” capacity of 35,300 GtCO2, an “effective” capacity of 13,500 GtCO2, and a “practical” capacity of 3,900 GtCO2. The IPCC (2005, 2011a) estimates a minimum sequestration capacity in geologic formations of 550 GtC (~2000 GtCO2), with the potential to be significantly larger (i.e., thousands of gigatonnes of carbon), due to the uncertainty associated with saline aquifers. In 2012, the U.S. Geological Survey (USGS) identified technically accessible sequestration resources totaling 3,000 GtCO2 in 36 geological formations in the United States (USGS Geologic Carbon Dioxide Storage Resources Assessment Team, 2013). Figure 3.7 shows the estimated CO2 sequestration potential of saline aquifers, depleted oil and gas, and coal-bed reservoirs
_______________
21 The volume of 2,000 GtCO2 is approximately 2,300 km3 assuming a CO2 condensed-phase density of 0.02 mol/cm3 (Liu and Wilcox, 2012); the volume of the U.S. Great Lakes can be found here: USEPA (2011).
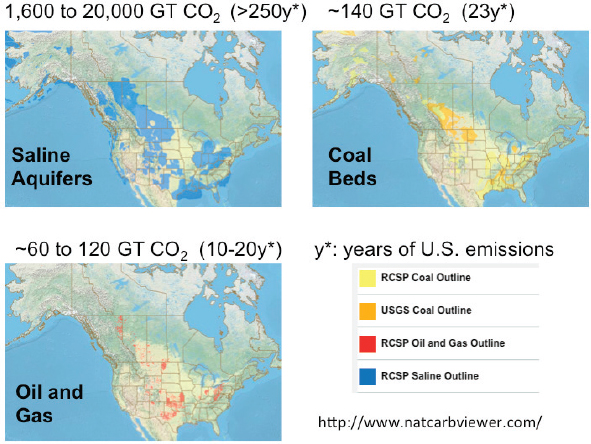
FIGURE 3.7 U.S. CO2 sequestration capacity estimates for various geological reservoirs. Saline aquifers have the highest potential for CO2 sequestration, followed by depleted coal beds and oil and gas fields. SOURCE: Created using NATCARB, http://www.natcarbviewer.com/.
in North America. The Benson et al. (2012) review emphasizes the need for research, geological assessments, and—even more crucial to the viability of sequestration—commercial-scale demonstration projects for improvement of confidence in capacity estimates.
Current annual rates of CO2 sequestration from existing projects are small compared to the amount required to make a significant change to atmospheric CO2 concentrations. The current scale of CCS is on the order of millions of tons of CO2 per year, with four large-scale CCS projects in place totaling ~50 MtCO2 sequestered and demonstrated monitoring sufficient to ensure efficacy of the injected CO2. These projects
have been operating from a few years to almost two decades, thereby demonstrating the effective and safe deployment of CCS (Benson et al., 2012).22
However, it is important to recognize that the scale required for adequate CO2 sequestration to significantly reduce negative climate change impacts is much greater. Nine additional projects are under construction and are expected to be operational by 2016, with an estimated 13 MtCO2 stored per year. The IEA CCS Roadmap (IEA, 2013b) reports that in order for CCS to make up 17 percent of the CO2 mitigation portfolio through 2050, the scale of CCS needs to increase from the order of millions of tons of CO2 per year to ~7 GtCO2/yr (Global CCS Institute, 2013; IEA, 2013b).
Important considerations include the long-term integrity of the cap rock and other qualities of the reservoir that minimize leakage, and, due to the large net volumes of injected fluids needed, the risk of inducing seismic events through overpressurizing the reservoir (NRC, 2013a). With nearly 40 years of experience from EOR, a great deal of information has been gained associated with the safe subsurface injection of CO2 (Benson et al., 2012). A recent study by Gan and Frohlich (2013) suggests that supercritical CO2 injection since 2004 in the Cogdell oil field north of Snyder, Texas, may be a contributing factor to seismic activity taking place between 2006 to 2011, with a total of 18 earthquakes having magnitudes of 3 or greater. Zoback and Gorelick (2012) state that the injection of large volumes of CO2 into the brittle rocks commonly found in continental interiors will likely trigger seismic events, which could subsequently affect seal integrity, thereby increasing the potential for CO2 leakage. These recent studies highlight the importance of ongoing research in the field of CO2 sequestration in geological reservoirs if CDR methods such as BECCS and DACS are to move forward and contribute significantly to reducing negative impacts of climate change.
As shown in Figure 3.8, the leakage probability generally decreases over time from secondary trapping mechanisms, such as solubility trapping due to dissolution of supercritical CO2 into saltwater (brine) already present in the porous rock. In addition, given the higher density of the saltwater containing dissolved CO2 compared to the surrounding fluids, the mixture will sink to the bottom of the formation over time, further trapping CO2. Over even longer timescales, mineral trapping may take place due to the formation of carbonic acid in the reservoir. Over time, this weak acid can react
_______________
22 It should also be noted that the U.S. Department of Energy already has in place a number of key carbon capture and sequestration research programs and initiatives in place under the Office of Fossil Energy (OFE). More specifically, the OFE allocated $1.52 billion of the $3.4 billion it received from the Recovery Act in 2009 for a competitive solicitation of industrial carbon capture and energy efficiency improvement projects, including for innovative concepts for beneficial reuse of CO2. Three projects chosen are aimed at testing large-scale industrial CCS, expected to capture 6.5 MtCO2 per year by the end of September 2015. Further information can be found at http://energy.gov/fe/office-fossil-energy.
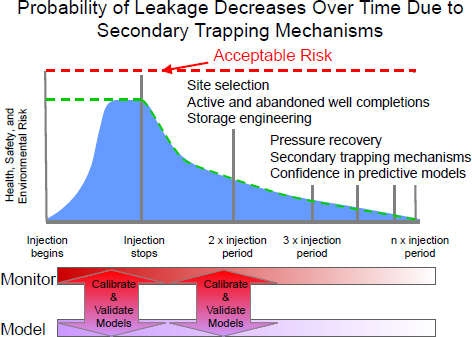
FIGURE 3.8 Evolution of health, safety, and environmental risk over time. Leakage generally decreases over the lifetime of the CO2 sequestering activity. SOURCE: GEA, 2012.
with minerals in the rock, leading to the formation of carbonate minerals in which the CO2 is chemically transformed and, hence, more permanently trapped.
A study by Hepple and Benson (2005) was carried out that discusses the performance requirements and implications of surface seepage. Figure 3.9 (left) from this study compares allowable emissions for stabilization of carbon dioxide concentrations at 550 parts per million by volume (ppmv) and expected emissions for different leakage rates as a function of time. This assumes that carbon sequestration is the only mitigation measure used to reduce CO2 emissions below a particular reference scenario (IPCC SRES scenario A1B), which results in the sequestration of about 10,000 GtCO2 over a period of 300 years. In this case, leakage rates below 0.1 percent of sequestered CO2 lead to emissions less than those that are allowable for stabilization. However, from Figure 3.9 (right), an annual leakage rate of 0.1 percent of stored CO2 leads to only ~40 percent of the CO2 stored after 1,000 years, while a leakage rate of 0.01 percent leads to ~90 percent stored over 1,000 years.
The cost of geological sequestration includes the site characterization, capital costs (i.e., well surface equipment, drilling costs, and additional CO2 compression in some in-
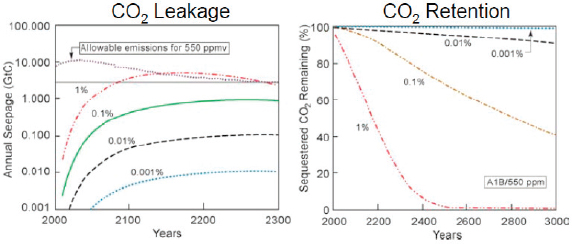
FIGURE 3.9 Comparison between “allowable” and expected emissions (left) and percent stored CO2 remaining (right) for different leakage rates as a function of time. SOURCE: Hepple and Benson, 2005.
stances), operating and maintenance, monitoring, and verification. Benson et al. (2012) estimate that CCS has the potential of increasing the cost of electricity generation by 50 percent to 100 percent, with capital costs and parasitic energy requirements of 15 percent to 30 percent being the substantial cost drivers. The reservoir characterization costs are less expensive in the case of oil and gas formations compared to saline aquifers or deep coal seams because oil and gas sites were previously characterized from exploration efforts. The cost of characterization will also depend on the aerial extent of the potential CO2 migration plume, which may be determined by regulations (Rubin et al., 2007). The costs associated with drilling primarily depend on the number of wells, including those for produced water as required, field injectivity, and the allowed overpressure. Operating and maintenance costs are expected to be comparable to the costs of water injection for secondary oil recovery processes (Bock et al., 2003; Rubin et al., 2007). Monitoring costs are expected to be a small fraction of the overall costs (Benson et al., 2004). According to the International Energy Agency (IEA, 2009), the potential global investment required solely for CO2 sequestration for stabilization of atmospheric CO2 to 450 ppmv ranges between $0.8 to $6 billion in 2020 and $90 to $600 billion in 2050.23 More specifically, the costs associated with existing CO2 sequestration projects are $11/tCO2 to $17/tCO2 for the project at Sleipner (North Sea), $20/tCO2 for the one at Weyburn (Saskatchewan), and $6/tCO2 for the one at In Salah (Algeria) (ITFCCS, 2010). If CO2 is used for EOR, the revenues from the additional gas and
_______________
232009 U.S. dollars.
oil produced will reduce the cost of CO2 disposal, but in these cases the majority of the CO2 is recovered and reused. The purchase price of CO2 is about $40/tCO2 to $50/tCO2 for EOR operations (Benson et al., 2012).
Benson et al. (2012) emphasize that environmental risks of geological sequestration appear manageable, but regulations will be required to govern site selection, operating guidelines, and the monitoring and closure of a sequestration facility. Public perception of the safety and effectiveness of geological sequestration will likely be a challenge until more projects are under way with an established safety record.
In addition to geological sequestration, CO2 can also be injected into the mid-depth ocean (i.e., 1,000 to 3,000 m deep; see Figure 3.10). Within this approach, the CO2 is stored on the order of hundreds to thousands of years before it returns to the atmosphere through ocean circulation. Alternatively, there is deep-injection ocean disposal,
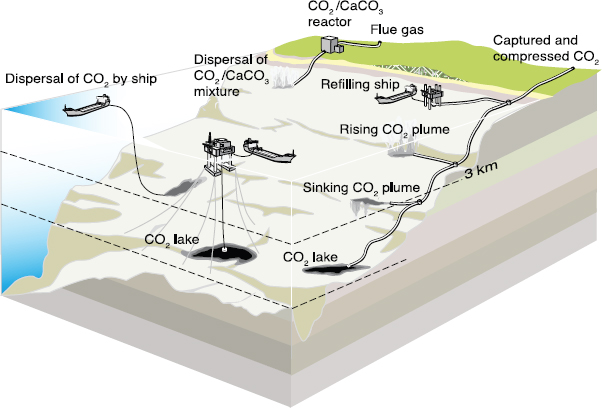
FIGURE 3.10 Ocean disposal strategies for inorganic processes (i.e., not ocean fertilization). CO2 could potentially be placed in the ocean either as a highly compressed gas (CO2), or dissolved in alkalinity-enriched seawater (CO2/CaCO3). Highly compressed CO2 could be placed on the seafloor or dispersed in plumes. Pipes or ships could be used to transport the CO2. CO2 and alkalinity-enriched seawater would need to be dispersed in the ocean. SOURCE: IPCC, 2005, Chapter 6 on Ocean Storage.
in which stationary pools of CO2 are created near the bottom of the ocean, with a potential sequestration capacity on the order of 1,000 GtCO2 (IPCC, 2005). Due to the potential biological impacts, high cost, sequestration reversibility, and public acceptance concerns, little research is being conducted on ocean sequestration of CO2 today.
CO2 could potentially be stored in the ocean in a form where the CO2 acidity is neutralized in solution by the addition of alkalinity derived by carbonate (IPCC, 2005; Rau, 2011; Rau and Caldeira, 1999) or silicate minerals (Kirchofer et al., 2012). It has been proposed that such solutions could be added to the ocean (Figures 2.1 and 3.10). These alkaline and CO2-enriched waters would bring ocean pH and the carbonate mineral saturation state back closer to preindustrial values, offsetting at least some of the ocean acidification caused by excess CO2, and thus might be expected to have a positive effect on marine calcifiers (NRC, 2010b; Rau et al., 2012). However, these approaches require a substantial mining infrastructure and large volumes of inflowing water, so there is potential for substantial local adverse environmental consequence. Economic considerations indicate that application of these approaches, if they can be cost competitive, would largely be limited to coastal environments with co-located availability of concentrated CO2 streams, carbonate or silicate minerals, and ocean water (Rau and Caldeira, 1999).
A hybrid sequestration scheme has been proposed (Schrag, 2009) in which CO2 is injected under a thin layer of sediments at the ocean’s floor to combine aspects of geological sequestration with ocean sequestration. A related hybrid scheme is to inject CO2 into deep-sea basalt reservoirs, such as the Juan de Fuca plate (Goldberg et al., 2008; Marieni et al., 2013). Another concept involves the displacement of methane from methane hydrate structure with CO2, which could potentially enhance methane production with the co-benefit of CO2 sequestration (Ohgaki et al., 1996). The hybrid and methane displacement in hydrates approaches are still at the very early research stages.
Looking forward, the committee identified a couple of important future research directions for CO2 sequestration:
- Rapid expansion and scale-up of CO2 sequestration demonstration projects with monitoring to gain experience, improve procedures, and increase public understanding of the safety of the process; and
- Increased research focus on reservoir quality and capacity to ensure safety and efficacy; all sequestration reservoirs are unique and require dedicated characterization.
The technical readiness of CO2 sequestration is at the intermediate stage since prototypes already exist, but not at the required scale for significant CO2 sequestration, i.e., on the order of tens of gigatonnes CO2 per year. Although efforts have been made to monitor the CO2 leakage for the operations currently in practice at the scale of millions of tons of CO2 per year, it is still uncertain whether the CO2 will be stored on the order of millennia. Additional monitoring and leakage studies need to be carried out. The environmental impacts of CO2 sequestration may be considered medium given the potential of induced seismicity and Earth’s unknown response to long-term CO2 mineralization sequestration. Although studies by Benson et al. (2012) and Dooley (2013) show significantly high capacity estimates for geologic sequestration of CO2, this does not indicate the timescale of allowable injection. Based on the required projections to limit 2°C warming from the IEA Roadmap (IEA, 2013b), the required annual progress in CO2 sequestration is significant, with 1 GtCO2/yr up to 2025, ~8 GtCO2/yr to 2050, and up to ~20 GtCO2/yr through 2100. These projects result in ~800 GtCO2 cumulatively sequestered up to 2100. Costs of CO2 sequestration range from $10/tCO2 to $20/tCO2 (ITFCCS, 2010).
Utilization of Carbon Dioxide and Available Markets
The primary market for CO2 today is EOR. Although the food beverage industries and chemical markets exist for CO2, they do so to a lesser extent than EOR. In the United States, ~54 MtCO2/yr is used for EOR and most of the CO2 is sourced naturally rather than anthropogenically. In particular, 80 percent of the CO2 is sourced from natural reservoirs, while the remaining is from anthropogenic source (Kuuskraa et al., 2013). According to Advanced Resources International (2011), state-of-the-art and “next-generation” EOR in the United States have a long-term total capacity of 10 and 20 GtCO2, respectively. In addition to EOR, ~80 to 120 MtCO2/yr is sold commercially for various applications, primarily including chemical solvent production, coffee decaffeination, fertilizer production, and carbonated beverages. The CO2 demand for refrigerants and solvents is less than 1 MtCO2/yr, while the beverage industry is on the order of ~8 MtCO2/yr. Although EOR has the potential to involve some degree of permanent CO2 sequestration, it is important to note that most utilization methods ultimately return CO2 into the atmosphere (Global CCS Institute, 2013; IPCC, 2005).
In the section Accelerated Weathering and Mineral Carbonation, the transformation of CO2 with alkalinity to form stable or dissolved carbonates was reviewed. These options could potentially store CO2 in useful or marketable forms. A limitation of solid carbonate sequestration is the relatively small size of available markets for solid carbonates, which primarily include road building and concrete. Consideration of the current ag-
gregate market provides a reasonable estimate on the potential impact of this utilization option, which is small (on the order of less than 1 percent of emissions) and was discussed in greater detail previously. A study by Sridhar and Hill (2011) estimated that replacing 10 percent of building materials with carbonate minerals has the potential to reduce CO2 emissions by 1.6 Gt/yr. If there is at some point in the future a market for substances that help reduce ocean acidification (NRC, 2010a), there could conceivably be a market for the high-alkalinity, CO2-rich solutions that could be generated by accelerated weathering processes.
Looking forward, the committee identified several important future research directions for utilization of CO2:
- Catalyst design for CO2 conversion processes with reduced energy; and
- Advanced uses of CO2 that can expand capacity and verify permanent sequestration of CO2 without re-release into the atmosphere, for example, monitored EOR, monitored enhanced natural gas recovery, geothermal heat recovery, waterless fracking, carbonate formation, or use of high-alkalinity high-CO2 solutions to counter ocean acidification.
Some of the methods listed in Table 2.2 are both affordable and benign, while some may be benign but costly in the near term. For instance, land management methods including reforestation and afforestation, water management, low- or no-till agriculture, and cover crop agriculture have the potential to store ~2 to 5 GtCO2/yr at a cost of ~$1/tCO2 to $100/tCO2 (see Table 2.2). Today, the upper range of these estimates equates to just over 10 percent of global CO2 emissions. Specifically, tropical afforestation accounts for over half of this potential sequestration (Smith and Torn, 2013) and is based on land and resource availability. It is important to consider the potential scale of methods, even if they are affordable and benign, to determine whether they can reasonably contribute to a portfolio of responses to the CO2 problem.
Another strategy of significant impact with reasonable costs is the concept of bioenergy. In this process, biomass may be directly combusted or co-fired with coal or natural gas to produce process heat or electricity. The generation of a variety of outputs (i.e., polygeneration) such as electricity, process heat, fuel, and chemicals is also possible through gasification of the biomass, which results in the production of synthetic gas (i.e., CO + H2), allowing for product synthesis flexibility dependent on market potential. However, without separation of CO2 from the flue (combustion) or fuel (gasification) gas, this process is not a negative-emissions strategy. Therefore, CO2 separation
and subsequent sequestration are required for BECCS to be capable of CDR. From the perspective of reducing the growth of atmospheric CO2 levels, employing BECCS has the same impact as the comparable amount of CCS and bioenergy being deployed until such time as fossil fuel emissions are minimal, which is unlikely to occur on any large scale for decades. Very similar to conventional CCS, the CO2 separation costs are in line with CCS at ~$100/tCO2. BECCS has the theoretical potential to remove up to 18 GtCO2/yr; however, this removal rate would require ~1,000 million acres of arable land for biomass cultivation, which represents nearly three-quarters of the planet’s available arable land, and thus is not realistic under any plausible scenario. Therefore, depending on world population and competition for land for food production and urban expansion, the level of CDR impact from BECCS is likely to be dramatically lower than the theoretical potential might suggest.
Although both capture and sequestration are inherent within those approaches that increase terrestrial carbon stocks, this is not the case for bioenergy. Hence, application of BECCS on an annual basis may also be limited by the sequestration potential. Geologic sequestration is currently practiced on the order of millions of tons of CO2 per year. (Other concepts, such as accelerated weathering approaches, have not yet progressed beyond benchtop scale.) IEA and Word Energy Outlook roadmaps indicate that through 2050 this scale needs to be on the order of gigatonnes of CO2 per year if warming beyond 2°C is to be avoided. However, this requires a thousandfold increase in the current sequestration activity and the construction and operation of hundreds to thousands of individual sources and injection sites. Although theoretically this large number of sources and injection sites is possible (IEA, 2005), to be technically feasible at this scale will require additional demonstration and pilot plants to be brought online very soon. In addition to the existing four projects globally, another nine projects are under construction today, with the potential to capture and store 13 MtCO2/yr, and should be operational by 2016 (IEA, 2013b). Again, this scale will have to increase by at least an order of magnitude to achieve any significant impact on net carbon emissions.
Although the scales of utilization are limited, it is important to consider their potential. Due to the immense scale of CO2 to be captured, some types of CO2 utilization will undoubtedly make up part of the portfolio of responses for preventing rerelease of CO2 into the atmosphere in addition to geologic sequestration. For instance, another option may be to produce carbonate minerals by reacting CO2 with available alkalinity. The carbonate may be used as “synthetic” aggregate for available construction markets. In addition, these carbonate minerals could potentially be left in dissolved form where they could be released into the ocean, thereby countering acidification caused by passive uptake of excess CO2 from the atmosphere.
Overall, all of these options have trade-offs that are described in greater detail in the previous sections of this chapter. Land management approaches and BECCS are generally characterized by lower risk and lower costs, whereas ocean iron fertilization is generally characterized as higher risk and DACS as currently higher cost.
Tables 3.2 and 3.3 provide a quick summary overview of the committee’s judgments on aspects such as effectiveness, technical readiness, ramp-up time, duration of effects, cost, ability to detect and monitor, and various risks of the CDR strategies presented in this chapter; aspects of capture and sequestration systems are discussed in the two tables, respectively. In each category, the committee has provided an estimate of not only the magnitude of the effect (e.g., high, medium, low, and what those categories mean for that table entry), but also the committee’s confidence in that categorization. The entries in the tables are the product of committee deliberation based on an understanding of the available literature. Although capture from point-source emitters coupled to sequestration (i.e., CCS) is not considered a CDR technology, it is included in Table 3.2 for comparison with the CDR technologies considered in this report.
CDR Summary Table 3.2 CO2 Capture Approaches
| Committee Confidence: |
||||||
| NOTES: | Point-Source Capture | Direct Air Capture | Biological Land Based | Biological Ocean Based | Accelerated Weathering Land Based | Accelerated Weathering Ocean Based |
| Fuel/fuel gas | Afforestation, soil, land management | Ocean iron fertilization | ||||
| Technological readiness, speed to deployment, technical risk | ||||||
| Mature technology (ready to deploy quickly, low technical risk): technology exists at scale | ||||||
|
Intermediate maturity technology: prototypes exist, not to scale |
|
|
||||
|
Immature technology (not ready to deploy quickly, high technical risk): needs prototyping…¬…… |
|
|
|
|||
| Time required to scale to maximum deployment with major effort, achieving significant capture rate (~1 GtCO2/yr) | ||||||
| Fast: years (i.e., < 10 yr) | ||||||
|
Medium: decades (i.e., 10 < x < 30 yr) |
|
|
|
|
||
|
Slow: many decades (i.e., >30 yr) |
|
|
||||
| Effect per unit cost for pilot scale with currently available technology | ||||||
| High: dollars per ton CO2 (i.e., <$10/tCO2) | ||||||
|
Medium: tens of dollars per ton CO2 (i.e., $10/tCO2 < x < $100/tCO2) |
|
|
|
|||
|
Low: hundreds of dollars per ton CO2 (i.e., >$100/tCO2] |
|
|
|
|||
| Maximum feasible deployment capture rate | ||||||
| High: >10 GtCO2/yr (i.e., >30 percent of current emission rate; order of magnitude of current emission rate) | ||||||
|
Medium: 1 < x < 10 GtCO2/yr (i.e., order 10 percent of current emission rate) |
|
|
|
|
|
|
|
Low: < 1 GtCO2/yr (i.e., order 1 percent of current emission rate) |
|
|||||
| Verifiability: Ability to confirm that capture has happened and quantify how much CO2 has been captured | ||||||
| Easily verifiable: existing and planned observation systems can verify without retasking | ||||||
|
Moderately easy to verify: existing observation systems would need retasking or known technology would need to be deployed |
|
|
||||
|
Difficult to verify: new technology or methods would need to be developed or deployed |
|
|||||
| NOTES: | Point-Source Capture | Direct Air Capture | Biological Land Based | Biological Ocean Based | Accelerated Weathering Land Based | Accelerated Weathering Ocean Based |
|
Fuel/fuel gas |
Afforestation, soil, land management |
Ocean iron fertilization |
||||
| Negative environmental consequences | ||||||
| Minor: mostly local impacts; can be mitigated consistent with current national environmental protection standards | ||||||
|
Medium: potentially serious impacts that may be difficult to mitigate to current environmental protection standards |
|
|
|
|||
|
Major: severe national or global impacts incompatible with current environmental protection standards; impacts may exceed environmental benefits of climate change mitigation |
|
|||||
| Environmental co-benefits | ||||||
| High: numerous and/or very likely co-benefits, such as protection of watersheds from erosion, wildlife habitat and diversity, recreational opportunities, or reduction in ocean acidification | ||||||
|
Medium: modest or uncertain co-benefits |
|
|||||
|
Low: very few or no co-benefits |
|
|
|
|
||
| Sociopolitical risks (include national security) | ||||||
| Minor: limited and mostly local economic and social impacts | ||||||
|
Medium: potential for serious national or regional economic, social, political, or security impacts that may be difficult for governments to manage |
|
|
|
|
||
|
Major: potential for severe national and regional economic hardship, social dislocation, political instability, and civil or military conflict |
||||||
| NOTES: | Point-Source Capture | Direct Air Capture | Biological Land Based | Biological Ocean Based | Accelerated Weathering Land Based | Accelerated Weathering Ocean Based |
|
Fuel/fuel gas |
Afforestation, soil, land management |
Ocean iron fertilization |
||||
| Governance challenges for deployment at scale | ||||||
|
No novel governance challenges |
|
|
|
|||
|
Governance challenges likely to be primarily territorial, but with some legitimate interest by other states |
|
|||||
|
Potential for substantial adverse effects across international borders or to an international commons |
|
|
||||
| Risk of detrimental deployment from unilateral and uncoordinated actors | ||||||
| Low risks: few actors (individuals, groups, nations) have large enough resources to deploy technique and motivation to do so | ||||||
|
Medium risks |
|
|||||
|
High risks: many actors with resources and motivation |
||||||
CDR Summary Table 3.3 CO2 Sequestration Approaches
| Committee Confidence: |
|||||
| NOTES: | Reduced Carbon (e.g., land plants, biochar) | Oxidized Carbon Sequestered as Molecular CO2 in Solid Earth (geologic) | Oxidized Carbon Sequestered as Molecular CO2 in Ocean | Oxidized Carbon Sequestered in Other Compound (e.g., CaCO3), Solid Form | Oxidized Carbon Sequestered in Ocean |
| Land management | For example, as pa of CCS, BECCS, or DACS | rt Ocean iron fertilization | Accelerated weathering on land | Accelerated weathering in or near ocean | |
| Technological readiness, speed to deployment, and technical risk | |||||
| Mature technology (ready to deploy quickly, low technical risk): technology exists at scale | |||||
|
Intermediate maturity technology: prototypes exist, not to scale |
|
|
|||
|
Immature technology (not ready to deploy quickly, high technical risk): needs prototyping |
|
|
|||
| Time required to scale to maximum deployment with major effort | |||||
| Fast: years (i.e., <10 yrs) | |||||
|
Medium: decades (i.e., 10 < x < 30 yr) |
|
|
|
|
|
|
Slow: many decades (i.e., >30 yr) |
|||||
| NOTES: | Reduced Carbon (e.g., land plants, biochar) | Oxidized Carbon Sequestered as Molecular CO2 in Solid Earth (geologic) | Oxidized Carbon Sequestered as Molecular CO2 in Ocean | Oxidized Carbon Sequestered in Other Compound (e.g., CaCO3), Solid Form | Oxidized Carbon Sequestered in Ocean |
| Land management | For example, as part of CCS, BECCS, or DACS | Ocean iron fertilization | Accelerated weathering on land | Accelerated weathering in or near ocean | |
| Persistence (sequestration lifetime) | |||||
| Millenia | |||||
|
Centuries |
|
|
|||
|
Decades |
|||||
| Maximum sequestration amount | |||||
| High: >10,000 GtCO2 (i.e., >30 percent of fossil fuel resource, order 100 percent of fossil fuel resource) | |||||
|
Medium: 1,000 < x < 10,000 GtCO2 (i.e., order 10 percent of fossil fuel resources) |
|||||
|
Low: order <1,000 GtCO2 (i.e., order 1 percent of fossil fuel resources) |
|
|
|||
| Verifiability: Ability to detect and quantify the rate at which CO2 was captured and added to the sequestration reservoir | |||||
| Easily verifiable: existing and planned observation systems can verify without retasking | |||||
|
Moderately easy to verify: existing observation systems would need retasking or known technology would need to be deployed |
|
|
|
||
|
Difficult to verify: new technology or methods would need to be developed or deployed |
|||||
| Verifiability: Ability to detect and quantify the rate at which CO2 is leaking out of the reservoir | |||||
| Easily verifiable: existing and planned observation systems can verify without retasking | |||||
|
Moderately easy to verify: existing observation systems would need retasking or known technology would need to be deployed |
|
|
|
|
|
|
Difficult to verify: new technology or methods would need to be developed or deployed |
|
||||
| NOTES: | Reduced Carbon (e.g., land plants, biochar) | Oxidized Carbon Sequestered as Molecular CO2 in Solid Earth (geologic) | Oxidized Carbon Sequestered as Molecular CO2 in Ocean | Oxidized Carbon Sequestered in Other Compound (e.g., CaCO3), Solid Form | Oxidized Carbon Sequestered in Ocean |
| Land management | For example, as part of CCS, BECCS, or DACS | Ocean iron fertilization | Accelerated weathering on land | Accelerated weathering in or near ocean | |
| Verifiability: Ability to quantify increase in carbon stocks of the sequestration reservoir (i.e., verification of change in carbon mass stored) | |||||
| Easily verifiable: existing and planned observation systems can verify without retasking | |||||
|
Moderately easy to verify: existing observation systems would need retasking or known technology would need to be deployed |
|
|
|
||
|
Difficult to verify: new technology or methods would need to be developed or deployed |
|||||
| Negative environmental consequences | |||||
| Minor: mostly local impacts; can be mitigated consistent with current national environmental protection standards | |||||
|
Medium: potentially serious impacts that may be difficult to mitigate to current environmental protection standards |
|
|
|
|
|
|
Major: severe national or global impacts incompatible with current environmental protection standards; impacts may exceed environmental benefits of climate change mitigation |
|||||
| Sociopolitical risks (include national security) | |||||
| Minor: limited and mostly local economic and social impacts | |||||
|
Medium: potential for serious national or regional economic, social, political, or security impacts that may be difficult for governments to manage |
|
|
|
|
|
|
Major: potential for severe national and regional economic hardship, social dislocation, political instability, and civil or military conflict |
|||||
| NOTES: | Reduced Carbon (e.g., land plants, biochar) | Oxidized Carbon Sequestered as Molecular CO2 in Solid Earth (geologic) | Oxidized Carbon Sequestered as Molecular CO2 in Ocean | Oxidized Carbon Sequestered in Other Compound (e.g., CaCO3), Solid Form | Oxidized Carbon Sequestered in Ocean |
| Land management | For example, as part of CCS, BECCS, or DACS | Ocean iron fertilization | Accelerated weathering on land | Accelerated weathering in or near ocean | |
| Governance challenges for deployment at scale | |||||
| No novel governance challenges | |||||
|
Governance challenges likely to be primarily territorial, but with some legitimate interest by other states |
|
|
|||
|
Potential for substantial adverse effects across international borders or to an international commons |
|
||||


























































