8
Special Considerations for Reporting of Results in NHANES
This portion of the workshop considered the implications of genomic data in the context of current National Health and Nutrition Examination Survey (NHANES) policies for return of results, and whether genomic results should be viewed differently from other research results. Presenters included Jennifer Madans, National Center for Health Statistics, and Jeffrey Botkin, University of Utah. Les Biesecker, National Human Genome Research Institute, chaired the session.
STATISTICAL AGENCY CONSIDERATIONS IN RELEASING DNA RESULTS
Jennifer Madans mentioned some advantages of being part of the federal statistical system, which include an institutional commitment to long-term data collection and a commitment to maintaining the storage of specimens. While budgets will change, a project such as NHANES does not have the pressing worries that other population-based studies may have in terms of expired grants or investigator turnover. At the same time, she noted, NHANES operates under federal statistical agency regulations pertaining to confidentiality and protection, regulations that are transmitted through the informed consent process. If one thinks of a continuum of places where genetic work is done, with the clinical setting on one end, she described NHANES as being at the far “other end,” further to the extreme than the other population-based research discussed during the workshop.
Madans discussed several possible unintended consequences of or drawbacks to DNA collection that she said need to be considered (see Figure 8-1). One is the potential impact on participation in NHANES due to a perception that the government is going too far in collecting this kind of information, raising the issue of what is appropriate for the government to know. There is concern about whether the mere collection of genomic information is beyond the scope of a statistical agency, and if DNA is too sensitive a topic. If NHANES were to move to reporting genetic results, she asked, does that overstep the line of what is appropriate, and would this impact the core mission of monitoring the nation’s health?
There are consequences involved with the timing of testing and reporting of results, Madans said. In the case where NHANES results are returned very soon after the initial participant encounter, how different is that from contacting people 5 or 10 years later to say “remember us, we

FIGURE 8-1 Potential unintended consequences or drawbacks to an expanded NHANES genomic data collection.
SOURCE: Adapted from Madans (2014) presented at the Workshop on Guidelines for Returning Individual Results from Genome Research Using Population-Based Banked Specimens, February 10-11, National Research Council, Washington, DC.
may know something about you that you really might want to know?” There may be different ethical considerations in terms of agency obligations in these two scenarios, she suggested. And if genetic testing is done over time, there has to be a constant reevaluation and ongoing determination of what is reported, as is the case with other tests that NHANES does. There is likely to be increased volatility in decisions regarding genetic results and rapid changes in procedures and relatively less consensus about what should be reported, in Madans’ view.
Financial implications loom large, Madans added. NHANES does not currently have substantial in-house expertise concerning return of genetic results, and the processes of interpretation and counseling would be much more costly and complex than for nongenetic tests. These areas are outside of the existing program framework.
Madans noted potential negative impacts on agency reputation. While these impacts are difficult to anticipate, she said, agencies do have to consider whether there is an elevated risk of such impacts when dealing with highly complex and changing issues such as the ethical considerations and requirements discussed during this workshop. The thinking within the NHANES program 10 years ago was that, perhaps in 10 years, science will have coalesced and there will be more agreement on how to move forward, but this does not yet seem to be the case.
THE NOTION OF GENETIC EXCEPTIONALISM
People sometimes ask if genetic information is sufficiently different from other types of biomedical information and hence merits special rules or management, Jeffery Botkin said. He approached this question by noting that there are two angles to consider: Genetics might be treated differently than other forms of testing or information when this is not justified, which is the typical notion of exceptionalism. But the reverse might be true: Genetic information may be treated the same when, in fact, differences are justified.
Botkin said he is on record as supporting what he calls “soft genetic exceptionalism,” the idea that the aspects of a test itself or the information itself are important, not so much whether they are genetic or non-genetic (Green and Botkin, 2003). To Botkin, features of information may make it more sensitive or more problematic in certain circumstances. Genetic information may yield information relevant to the welfare of others, for example, vertical transmission wherein learning something about one’s own genetics tells one something about parental genetics. As Botkin noted, genetic information can be highly predictive of future disease; tests for BRCA1 and HNPCC have a higher predictive power than nongenetic tests for blood pressure and cholesterol. Such information
can be stigmatizing and is often more complex to analyze and interpret than that from other types of testing.1 Botkin observed that it is true that genetic tests often lack these sensitive aspects, and those are genetic tests that one need not spend a great deal of time worrying about. But when the aforementioned constellation is present, it calls for a higher level of scrutiny regardless of the platform being used.
The use of the term “genetic exceptionalism” implies a set of rules that people agree on and are making exceptions for with regard to genetics, Botkin said. It does not appear that today’s debate is sufficiently ripe to say that those rules exist, he posited. Speaking about exceptionalism is premature simply because nothing is an exception yet from a standard set of acceptable rules. In NHANES at this point in time, results for physical examinations and tests that are routinely conducted in clinical care as part of the original survey assessment are provided back to participants. But, he noted, there is no return-of-research results generated later, whether those results are genetic or not. So in that sense, Botkin said, there is no genetic exceptionalism at the present time.
Botkin presented a simple schematic (see Figure 8-2) of broad differences between different forms of testing, which he grouped into three categories: (1) physiologic tests, tests of current biological function, blood counts, blood gases, electrolytes, renal function tests, etc.; (2) imaging and other types of physical or anatomic testing; and (3) genetic or genomic testing. In physiologic testing, he explained, there is clinical validity and clinical utility for the sorts of things tested, and results often suggest a reasonably urgent response. Results are plainly evident and easy to interpret (one does not need a sophisticated analytic protocol to determine if many of these results are problematic) and for the most part are not context specific.
With imaging, clinical validity is variable depending on the finding. Utility is often present, Botkin said, as when one observes an anatomic abnormality and considers appropriate interventions. Similarly with urgency: A brain tumor on a scan is something that needs a response. Results are often plainly evident, but are often not context-specific; a brain tumor is a brain tumor whether one is 10 years old or 50 years old.
Genomic testing has variable clinical validity and variable clinical utility, depending on what is targeted. There usually is no urgency of action because for the most part genetic results do not need to be responded to
__________________
1Concern about stigma resulting from genetic testing may have increased in response to increases in genetic research (see, e.g., Sankar et al., 2006, and World Health Organization (http://www.who.int/genomics/gender/en/index3.html [June 2014]). This workshop did not directly consider the risks of stigmatization following the return of genetic (and other) information.
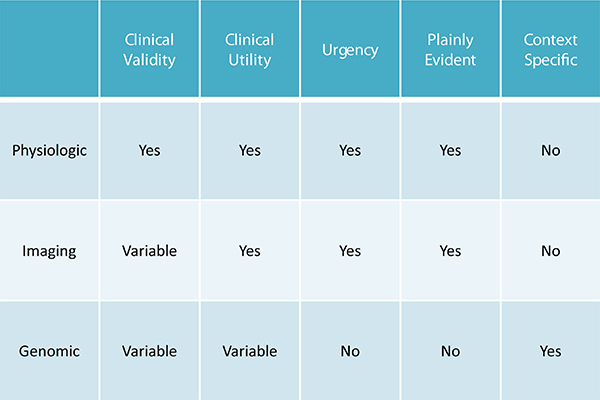
FIGURE 8-2 Characteristics of three forms of testing.
SOURCE: Botkin (2014) presented at the Workshop on Guidelines for Returning Individual Results from Genome Research Using Population-Based Banked Specimens, February 10-11, National Research Council, Washington, DC.
today or this week. They are not plainly evident, as it takes a fair amount of sophisticated analysis to figure out what is occurring, Botkin added. And they often are context-specific, an example being someone who has already lived through much of their risk; hence the risk is contingent upon the person’s age.
According to Botkin, these differences among test characteristics tend to favor return of results for physiologic and imaging tests above genetics, which may be an argument in favor of genetic exceptionalism. The big differences are the urgency, the plainly evident nature of results, and the fact that results often are not context-specific. Botkin said these differences make it easier to say “these are results that will not be returned,” as opposed to saying “maybe these are returnable depending on the specific situation of the person.”
Further discussion at the end of the workshop session about incidental findings and return of results highlighted the fact that discrete analyzers have now replaced most automated laboratory equipment that used to routinely perform a wide range of tests on a panel, regardless of the test or tests ordered. A participant pointed out that laboratories now try to focus analyses to include only what has been ordered, and clinicians
and laboratorians are comfortable with “gating” machines to produce only ordered results. The widespread use of discrete analyzers makes it unlikely that a laboratory would conduct tests other than those that are specifically requested. Further, the participant elaborated, CLIA requires laboratories to perform tests only at the written or electronic request of an authorized person, thereby further decreasing the likelihood that a laboratory would conduct a test without receiving a specific order from an authorized person to do so. In light of these developments and in view of ethical analyses suggesting that investigators do not have an obligation to search for incidental findings, it can be argued that any new standards saying that additional genomic analyses should routinely be done to identify actionable results would constitute genetic exceptionalism. Another participant noted that the NHANES practice of screening DNA bank proposals based on the potential for finding reportable results might be in the same category as gating genetic/genomic analyses to avoid known pathologic variants that are not relevant to specified research.






