Integrating Clinical Research and Practice: Examples
KEY SPEAKER POINTS
- The keys to a successful research partnership, according to Susan Huang, include a trial design and operational plan that cater to the strengths of each partner, choosing a research question that aligns well with a major health system priority, leveraging existing infrastructure in order to smoothly integrate research into clinical care, and using routinely collected data that already exist in EHRs.
- Uma Kotagal stressed the importance of using data that are collected systematically for research in the EHR in order to place minimal data collection burdens on care providers.
- David Grossman reiterated the importance of selecting research projects that improve care and outcomes as well as supporting the economic health of the system. He noted that a major challenge for researchers is to ensure that disruptive innovation does not negatively affect the stability of the health care system.
- Another important reason for being part of a network dedicated to improving care while reducing costs, even when it uses scarce resources, is that it enables staff to “do the right thing,” Edward Havranek said. “I think we forget that one of the most important aspects of quality, value, diffusing research, new
-
knowledge, and doing things better is that it appeals to that very uniquely moral and idealistic motivation for doing what we do.”
- Havranek also noted that there are times when the reimbursement system acts as an impediment to bringing research findings into the clinic, an issue that Kotagal said arises from the fact that revenue streams and incentives are too often dissociated. One solution to this problem, she said, would be for those who pay for health care to start paying for continuous innovation.
To provide the workshop with a vision of what is possible when care and research are integrated effectively, four speakers highlighted examples of organizations that are on the leading edge of these efforts. These four presenters described the effects that integration can have on efficiency, value, and health outcomes and discussed how the resulting value proposition can help organizations embrace the integration of care and research. Susan Huang of the University of California, Irvine, described the multi-partner REDUCE MRSA trial; Uma Kotagal of Cincinnati Children’s Hospital Medical Center provided details on the Improve Care Now Network; David Grossman of Group Health and the Group Health Research Institute discussed Group Health’s approach to integrating care delivery and research; and Edward Havranek of Denver Health spoke about the High Value Healthcare Collaborative. An open discussion, moderated by Harold Luft of the Palo Alto Medical Foundation, followed the presentations.
The REDUCE MRSA trial is an example of a successful partnership between a health system and an academic research group, Susan Huang said in her opening remarks. The rationale for conducting this trial was that health care–associated infections, many of which are caused by MRSA, or methicillin-resistant Staphylococcus aureus, are 1 of the top 10 causes of death in the United States and are likely to be the most preventable (National Center for Health Statistics, 2002). Moreover, there has been considerable debate as to whether the best approach for preventing these infections is to target the bacteria directly or to target at-risk populations, such as those in intensive care units (ICUs). This debate, said Huang, has been going on for over a decade. “The need for a definitive trial was quite prominent,” she said.
The trial was conducted as a partnership between the Hospital Corporation of America (HCA), the largest private health system in the United States and one that accounts for about 5 percent of all in-patient hospital stays, and a group of academic researchers who were well-versed in health care–associated infection research and experienced at conducting clinical trials. The trial was done using cluster randomization, which in this case meant randomizing hospitals and assigning all adult ICUs in each hospital to the same MRSA prevention strategy (Huang et al., 2013). The three strategies tested were:
- Routine care: This strategy involved screening patients for MRSA when they were admitted to the ICU. Health care providers used gloves and gowns when caring for patients who tested positive for MRSA.
- Bathing and treating patients who tested positive for MRSA: Health care providers used gloves and gowns when caring for patients who tested positive for MRSA, the patients were bathed daily with a 2 percent chlorhexidine-containing cloth, and for 5 days they had mupirocin antibiotic ointment applied twice daily inside their noses, the noses being the body site most commonly colonized with MRSA.
- Universal decolonization: Patients entering the ICU were not screened for MRSA colonization. Instead, all patients were bathed daily with a 2 percent chlorhexidine-containing cloth and received mupirocin ointment twice daily inside their noses for 5 days. Gloves and gowns continued to be used when treating patients who tested positive for MRSA.
A total of 74,256 patients in 74 ICUs at 43 HCA hospitals across 16 states were enrolled in the trial. This scope and depth of participation, which was achieved over a mere 18 months, would have been impossible without the collaborative partnership at the heart of this trial, Huang said. For baseline data, the study used 12 months of retrospective EHR data available in the HCA data warehouse. The results of the study were unequivocal: Universal decolonization resulted in a significantly greater reduction in the rate of all bloodstream infections than either targeted decolonization or screening and isolation.
Huang credited the success of this partnership to several factors. The most important reason, she said, was that the research question aligned well with a major health system priority and with HCA’s “strong dedication to quality.” As an aside, Huang noted that HCA’s administration emphasized the need to conduct the trial rapidly in order to not impede the health system’s quality improvement goals, because the trial would prohibit the
implementation of other interventions that could affect the study outcomes. Another factor contributing to the partnership’s success was that the trial design and operational plan catered to the strengths of each partner. “It is not conceivable that each entity, whether it is an academic center or the health system, would excel in every part of what it takes to conduct a clinical trial,” Huang said. As examples of how the trial design highlighted each partner’s strengths, she cited HCA’s centralized data systems and its ability to command a large number of hospitals, the experience of the academic partners at running large clinical trials and at conducting the highly specialized tests for bacterial isolates, and the longstanding experience with quality improvement studies of the Harvard Pilgrim Health Care IRB and that board’s willingness to serve as a central IRB for 38 of the 43 hospitals. This last factor played a major role in securing all IRB approvals within 6 weeks.
Huang noted that one interesting feature of this trial was that its cost was approximately $40 per patient, compared to $10,000 or more per patient for previously reported intervention trials. Furthermore, the intervention itself is low cost because it involves swapping out bathing soap and adding a nasal ointment while eliminating the cost and effort of screening patients for MRSA colonization. Huang also noted that 42 of the 43 hospitals that participated in the study were community hospitals and these were distributed evenly across the nation, making it likely that the results of this trial should be generalizable across hospital settings.
Huang explained that there were no onsite investigators for this trial. “We did not parachute in people who had special expertise in research,” she said. ‘We leveraged the existing quality improvement infrastructure and gave [the hospitals] the protocols and computer-based training modules that [the hospitals] needed, and we had coaching calls twice monthly.” In addition, the trial used pragmatic outcomes rather than forcing people to do extensive chart reviews. “We used routinely collected data that existed in the electronic health record to determine the outcomes of this trial,” Huang said. Also simplifying the trial was the decision for all data to remain in the hands of the HCA hospitals. “We reached behind their firewall, did all of the analyses, and then only moved summary level data into our hands that HCA approved to maximize protection and privacy of patient data,” Huang said.
The Improve Care Now Network was created to bring patients, families, and providers together to produce a system that would transform care for children with inflammatory bowel disease, Uma Kotagal told the workshop audience. The primary requirement for this learning health system was that research and health care delivery should continuously inform each
other in order to standardize care and achieve the best possible practice while reducing variation in outcomes. The framework for the network (see Figure 4-1) started with patients and clinicians creating a learning engine at the point of care, which is where data are collected and recorded in EHRs. The data are used to generate reports that support and highlight variations in practice.
A fundamental principle of the Improve Care Now Network, Kotagal said, is that data are collected systematically and at the point of care using an EHR. This minimizes the burden imposed on providers to collect data and allows it to be used for both quality improvement and research. Both patient-reported outcomes and clinical outcomes are recorded, collected, and analyzed, with the results being fed back to clinicians and patients for use in daily care. Outcomes data are analyzed for each center and displayed in graphical form, enabling each center to work individually but also to learn from each other in a collaborative fashion. In practice, this feedback-based system has increased remission rates to over 80 percent among more than 20,000 patients—representing some 35 percent of all children in the United States with inflammatory bowel disease—at 63 care centers.
Kotagal said that this process makes possible several types of research. Research on improvement includes studies on the factors that motivate clinicians, researchers, and patients and families to participate, contribute,
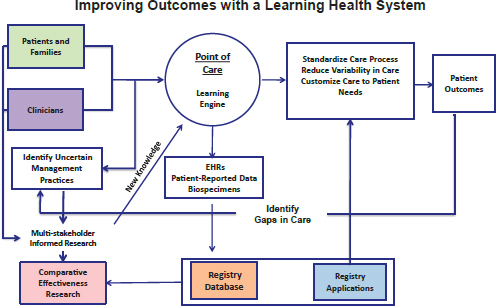
FIGURE 4-1 A schematic representation of the Improve Care Now Network.
SOURCE: Reprinted with permission from Christopher B. Forrest.
and work to improve their health care system. It also addresses questions regarding who uses the system and their goals and needs. Additionally, the network is used as a high-throughput system for comparative effectiveness research to identify which treatments work best for which patients and to extend the results of studies in adults to those involving children, she said.
An important feature of this system, explained Kotagal, is that it enables the research team to try to discern what is happening with the 20 percent of children with inflammatory bowel disease who are not doing well. Children are taught how to keep track of their own health and to understand the relationship between dietary changes and symptoms in a way that encourages learning. Ongoing studies using both patient-reported data and clinical outcomes data are looking at the potential for telehealth to improve self-adherence among children and to look for biomarkers that would predict remission rates. Kotagal noted in closing that networks such as this are critically important in the pediatric world because childhood diseases are rare. “It is difficult for us to learn at a single site what needs to happen,” Kotagal said, adding that “for those health systems that have continued to participate in these networks there is a strategic commitment from their boards, their CEOs, and their clinical chiefs and an understanding that these networks offer us the best chance to rapidly bring new knowledge to the bedside to improve the outcomes for care.”
EMBEDDED RESEARCH AT GROUP HEALTH
The process of embedding research into clinical care at Group Health takes a great deal of hard work, said David Grossman, speaking as both a researcher and the operational medical director of Group Health, a nonprofit health system with about 600,000 members in Washington State and $3.8 billion in annual revenue. Grossman said that two-thirds of Group Health’s members receive care at 25 Group Health Medical Centers, which have used EHRs since 2005. The Group Health Research Institute (GHRI), established in 1983, has 75 resident and affiliated investigators and an annual revenue stream of about $50 million, more than 95 percent of it from grants and contracts. Core funding for the Institute comes to about 40 cents per member per month. The Institute is a member of several multisite networks, including PCORnet, and it publishes its research in the public literature. “This is a nonproprietary research center,” Grossman said. “Institute researchers understand that they must thoroughly consider both the patients’ and the business enterprise’s interests in a research project before presenting it to the IRB to improve community health and health systems at large.”
An important aspect of the research GHRI conducts is that it largely addresses practical problems facing health systems. “Group Health has a
long history of doing practical research,” Grossman said, “and all research projects have to be important and salient to Group Health’s members.”
As an illustration of embedded research, Grossman described a project that originated with the Group Health CEO, who wanted to explore the potential impact of offering Group Health employees a new health plan with value-based benefits. “The questions he asked,” Grossman said, “were, why do we have this standard approach to cost sharing that ignores variation in the value of individual services that are delivered to patients? Does it really make sense, for example, to charge the same co-pay for every drug when we know that some drugs have much greater value than others?” To test the impact of value-based benefits, Group Health is offering health plan incentives that encourage employees to engage in health-promoting activities such as exercise. Group Health also reduced co-pays for certain services such as the provision of highly effective medications to control hypertension, heart failure, asthma, and depression, with a goal of improving drug adherence and preventing complications from chronic disease. GHRI is evaluating the impact of the changes on employee health status and workplace productivity as primary outcomes, and on care quality, utilization, and total cost of care as secondary outcomes.
Grossman said that an important factor in the success of implementing this project was close collaboration between researchers and Group Health’s human resources and health plan operating divisions to design the evaluation. In addition, early planning enabled the project to secure bridge funding for primary data collection while a grant application to AHRQ was pending. “This seed support from the Institute was absolutely critical for the timely start of the study,” Grossman said. Other key factors were the strong collaboration with Kaiser Permanente as partners in the research and their willingness to serve as a control site and use of the existing HMO Research Network virtual data warehouse.
One of the biggest challenges facing the study, Grossman said, is managing leadership expectations, particularly about the timing and speed at which the study will produce meaningful results. Another challenge for researchers is taking into account how the organization might react if study results show that value-based benefits lead to improved health and care quality but at higher cost. “It is important to have frank discussions with organizational leaders about these considerations at the beginning of the process,” Grossman said. For embedded research institutions like GHRI, ongoing major challenges include conducting research quickly enough for delivery system expectations and ensuring that research that involves disruptive innovation does not negatively impact the overall stability of the health care system. “We researchers can have fantastic ideas that are really cutting edge, but you can throw a huge wrench in operations by implementing too many changes too quickly,” Grossman said. In closing, Grossman
noted that he and his colleagues have come to understand the value of engaging stakeholders, including patients and leaders, early in the design process and sharing interim findings even when they are tentative.
RESEARCH AND PRACTICE IN A SAFETY NET SYSTEM
Denver Health is typical of “safety net” health systems, Edward Havranek said, in that the patients that it serves are mostly poor, with fewer than half having commercial insurance, and as a result it has a very thin operating margin. Denver Health is atypical, though, in three respects: It has gone 22 years without an operating deficit, it has a strong academic affiliation with the University of Colorado that goes back to 1947, and it has an inpatient risk-adjusted mortality for each of the past 5 years that puts it among the top 5 percent of all health systems as well as outpatient blood pressure control rates that rank it in the top quartile. “This finding is robust across subsets within the hospital and over time,” Havranek said, adding that the health system is not satisfied with being in the top quartile on measures of blood pressure control and that new data suggest how value-based propositions could improve that metric.
Two questions that a safety net health system faces are whether it should put resources into providing care of higher value and whether it should become a learning health system. “Let me argue here that the answer to both of these is yes when the measure of success is easily monetized and when the time to return is short,” Havranek said. “When you don’t have much operating margin to play with, you really have to be careful about what you invest in. We really want to invest first and foremost in things where we can carefully and explicitly prove to ourselves that we are not wasting money, and we need to know that quickly. We cannot be 2 or 3 or 4 years into a project before we discover that we are losing money.”
An example of a quality improvement project that met these criteria involved testing an evidence-based approach for venous thromboembolic prophylaxis. After the project was initiated in 2008, there was a clear decrease in the amount of money spent on venous thromboembolic prophylaxis, while at the same time the incidence of postoperative deep vein thrombosis and pulmonary embolism went down. “In other words,” Havranek said, “saving money improved outcomes by using the best available evidence, organizing and motivating people to work on this together, and giving it a high priority in a system such as ours. You really can do this kind of work.”
In this final remarks, Havranek discussed why Denver Health joined the High Value Healthcare Collaborative, a voluntary, largely self-funded network of some 19 health care systems that for the past 4 years has been working on ways of improving the value of health care (see Figure 4-2). “One reason is that safety net providers are isolated,” he said. “If you look
FIGURE 4-2 The High Value Healthcare Collaborative.
SOURCE: Reprinted with permission from the High Value Healthcare Collaborative.
around our communities, we don’t have competitors—nobody wants our patients. That lack of competition and local collaboration are both things that we would predict would diminish uptake of innovation. Being able to compare ourselves to the other members of the network creates a context for our efforts that is important.”
Another important reason for being part of a network dedicated to improving care while reducing costs, even when it uses scarce resources, is that it enables staff to “do the right thing,” Havranek said. “I think we forget that one of the most important aspects of quality, value, diffusing research, new knowledge, and doing things better is that it appeals to that very uniquely moral and idealistic motivation for doing what we do.”
Returning to what Havranek considers a failure—the inability to improve its hypertension control outcomes beyond being in the top quartile—he described a study that Denver Health, Kaiser Permanente, and the U.S. Department of Veterans Affairs (VA) conducted, which looked at the effect of home-based blood pressure monitoring. The protocol called for every patient in the study to receive a blood pressure cuff and instructions on how to use it and transmit the data to the health system. Once per week, a pharmacist reviewed the data and called patients to tell them what to do with their medications. The result of this intervention was to reduce systolic blood pressure by an average of 7 millimeters compared to controls. “That was more effective than any single drug that we could give,” Havranek said.
The failure came when he went to Denver Health’s administration and was told that the cost of this intervention, which Havranek said would raise its quality metric into the top 10 percent, was too high. “So we have
a problem,” he said. “We have a research-based solution that we demonstrated within our system and we cannot deploy it. Our partners at Kaiser and the VA have the financial structures where they can justify doing that sort of intervention, and we cannot.” The lesson here, he noted in closing, is that “our reimbursement system is fighting against us as we try to bring research findings into the clinical realm.”
Harold Luft started the discussion by noting that this last example highlights the tension between researchers and the organizations in which they work. “From the researcher’s side,” he said, “being in an organization makes the research more valuable. But what we are hearing is that speed and the ability to answer important questions quickly is a challenge for the research side, yet it is also the way to provide more value to the organization.” James Rohack from Baylor Scott & White Health agreed with this idea and asked if there can be a national solution to the tension between research and operations or if everything has to be done locally in individual communities. Havranek replied that he thinks this tension is not real—that most researchers in health systems understand the need to balance knowledge generation with operational and financial efficiency. Grossman said that if there is a tension it is positive, as it motivates the different constituencies in a health system to work and communicate effectively together, creating a stronger system overall.
Kotagal disagreed, saying there is a tension that has to do with time-lines and producing pragmatic results versus developing ideal solutions. She said that another type of tension arises when trying to generalize these small-scale examples. “I do not think there are good designs for how to do end-to-end research,” she said. “There are designs, but they have not been replicated to say we know how they work and to scale them. That is where we need to go.” Huang also thought that this tension between research and operations exists but that, as the examples presented show, those tensions can be overcome when thought leaders and academics work together with a health system that desires to make quality improvement a priority.
Huang commented on the importance of involving networks of health systems in research because of the large number of patients that such networks can bring into a study. She said that such collaborations provide the speed that health system administrators want as well as the ability to disseminate results more quickly.
In response to a question from Robert Jesse from the VHA about the boundaries between operational and clinical research with regard to the Common Rule, Havranek said that IRB issues are an impediment to doing operational research. “Depending on where you are, it is anywhere from
a minor to a major annoyance,” he said. “I think there is some ethical imperative in improving the system and to the extent that we impede the improvement in the system we are doing something that is probably unethical. I think systems need to recognize that when there needs to be a very quick expedited process for demonstrating that things are of no or little risk to individual patients and that the risk in not doing it is substantial, we need to move ahead. There needs to be a real lifting of this cloud over us that is imposed by the IRBs.”
Jonathan Tobin from the Clinical Directors Network and Rockefeller University asked if there was a way to look at some of the organizational variables that may facilitate or inhibit uptake by health systems. Huang said that this is where networks of health systems, such as the one that conducted the REDUCE MRSA trial, can play an important role by providing many settings in which to conduct research. She also noted that it is important to ask questions about dissemination and implementation at the outset of a project rather than as an afterthought.
Patrick Conway from CMS continued this line of questioning by asking if the panelists had any ideas on how to accelerate the pace of change beyond these individual great examples of success. Kotagal replied that those that pay for health care have to start paying for innovation. “Currently, our streams of revenue and incentives are dissociated,” she said, adding that this is not the case in most other industries. “It would be important for people paying for health care to require continuous innovation,” she added. Grossman said that researchers need to be more explicit in laying out the business case along with the clinical case when they design a study and present results to administrators.
This page intentionally left blank.












