Analysis of Cancer Risks in Populations near
Nuclear Facilities:
Phase 2 Pilot Planning
The National Academy of Sciences (NAS) Committee on Analysis of Cancer Risks in Populations near Nuclear Facilities: Phase 2 Pilot Planning was charged with providing advice to NAS in performing a number of tasks related to the planning for a pilot epidemiological study of cancer risks in populations living near U.S. Nuclear Regulatory Commission (USNRC)-regulated nuclear facilities. Specifically, the Phase 2 Pilot Planning committee’s task was to advise NAS on the following activities:1
- Identify the processes for selecting qualified individuals and/or organizations to perform the epidemiological and dosimetric tasks required to carry out the pilot study.
- Initiate effluent release and meteorological data collection in preparation for estimating doses to the people who live near the pilot nuclear facilities.
- Identify state requirements for data sharing and transfer of health information.
- Obtain institutional review board (IRB) approvals for the study, as appropriate.
- Identify key stakeholders and processes for communicating with them.
Progress in carrying out these activities is described by NAS staff in Appendix B.2 The committee was also tasked with preparing a brief report that provides advice to NAS on the methods and process for carrying out the pilot study. In fact, this committee was not initially tasked with preparing a report. It decided to do so because it determined during the course of the pilot planning that it needed a written record of its advice because of the strong interest by members of the public and the sponsor in its findings. The purpose of this brief report is to serve as a public record of the committee’s advice to NAS on general methodological considerations involved in carrying out the pilot study. The NAS will consider the committee’s advice when preparing the proposal to the sponsor and other documents related to carrying out the pilot study.
This pilot study was requested by the USNRC. The purpose of the pilot study, which was a recommendation of a previous NAS report (the Phase 1 report),3 is to evaluate the technical feasibility of implementing two study
________________
1 For the full statement of task for the phase 2 pilot planning study, see Appendix A.
2 The committee is not responsible for the content of Appendix B.
3 The Phase 1 report can be accessed at http://www.nap.edu/catalog.php?record_id=13388.
designs recommended in that same report for carrying out an analysis of risks near nuclear facilities in the United States. If found to be feasible based on explicit criteria, the methods developed and tested in the pilot study could be used to conduct a nationwide study that would examine risks near all USNRC-regulated nuclear facilities. That is, the pilot study is part of a larger effort carried out by NAS at the request of the USNRC to characterize cancer risks in populations near nuclear facilities in the United States. This effort is carried out in two phases as illustrated in Figure 1.
The pilot study will be carried out under contract to the NAS by qualified individuals and/or organizations. The NAS and an NAS advisory committee will provide the overall study oversight. At the conclusion of the pilot study (i.e., at the end of the pilot execution step described in Figure 1), the NAS will prepare a consensus report with findings regarding the technical feasibility of carrying out an assessment of cancer risks at additional USNRC-regulated nuclear facilities.
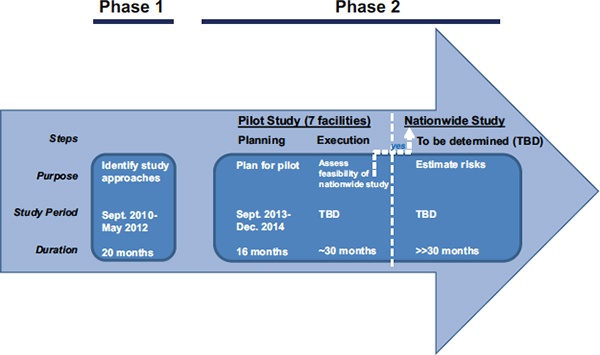
FIGURE 1 Phasing of the study on analysis of cancer risks in populations near nuclear facilities.
The USNRC asked NAS to undertake an assessment of cancer risks in populations near the nuclear facilities it regulates. NAS is performing this assessment in two phases. The Phase 1 study identified appropriate study designs to carry out an analysis of cancer risks near nuclear facilities in the United States. The Phase 1 report recommended two study designs appropriate for assessing cancer risks near nuclear facilities. It also recommended a pilot study of seven nuclear facilities to assess the technical feasibility of the recommended study designs. The Phase 2 study is the assessment of cancer risks. The pilot, which is part of the Phase 2 study, is being carried out in two steps: pilot planning and pilot execution. The pilot planning (current step) aims to plan for the pilot study. The pilot execution (next step) aims to carry out the pilot study and evaluate the technical feasibility of implementing the two study designs recommended in the Phase 1 study. If implementation of the study designs is feasible, the methods developed and tested in the pilot study could be used to conduct a nationwide study.
It is possible that even if feasible, the nationwide study will have low statistical power to detect any excess cancer risks in populations near nuclear facilities, if they exist. In that case the recommendation to proceed with the nationwide study will require weighing the potential for false positive associations together with the value of communicating with the public that the best information available, even if limited, is being used to answer its questions about cancer risks near nuclear facilities.
The Phase 1 report recommended that seven nuclear facilities be selected for the pilot study of cancer risks in populations near nuclear facilities. These are:
- Dresden Nuclear Power Station, Morris, Illinois
- Millstone Power Station, Waterford, Connecticut
- Oyster Creek Nuclear Generating Station, Forked River, New Jersey
- Haddam Neck Plant, Haddam Neck, Connecticut
- Big Rock Point Nuclear Plant, Charlevoix, Michigan
- San Onofre Nuclear Generating Station, San Clemente, California; and
- Nuclear Fuel Services, Erwin, Tennessee.
The selected nuclear facilities include nuclear power plants that feature different reactor designs, years of operation, and current operational statuses. They also include a nuclear fuel cycle facility regulated by the USNRC, the Nuclear Fuel Services, which is a uranium fuel fabrication facility. A pilot study of these nuclear facilities will likely reveal the logistical difficulties and potential problems with accessing the required information to perform a nationwide study of cancer risks near USNRC-regulated nuclear facilities.
The study designs to be examined for their feasibility during the pilot study are a population-level (or ecologic) study and a linkage-based case-control study.
The population-level, or ecologic, study would describe cancer incidence and mortality in populations living in census tracts within approximately 50 kilometers (30 miles) of the nuclear facilities. This study would examine all radiogenic and relatively common cancer types at all ages in populations that are potentially exposed to radiation from nuclear facility operations during their operational histories to the extent feasible with available data. Organ doses to the population residing within a census tract will be assigned on the basis of radiation exposure calculated at the geographic centroid of the census tract.
The ecologic study is intended to answer the following questions:
- Do cancer incidence and mortality vary by proximity to nuclear facilities?
- Does cancer incidence or mortality reflect patterns of radiation exposure associated with the nuclear facility?
A linkage-based case-control study (hereafter referred to as the case-control study) would assess whether children younger than 15 years of age whose mothers lived close to the nuclear facilities at the time of their birth are at higher risk of developing cancer compared with those whose mothers lived farther away but within a 50-kilometer (30-mile) radius from the facilities. This study would attempt to provide a more focused assessment of the association of pediatric cancers in relation to in utero and early-life exposure to radiation to the extent feasible with available data. Organ doses to the fetus and child will be assigned on the basis of address where the mothers lived at the time of delivery of the children, as reported in birth certificates.
The case-control study is intended to answer the following questions:
- Is a mother’s residential proximity to a nuclear facility at time of delivery associated with cancer in her children?
- Is estimated radiation exposure during pregnancy, early infancy, or childhood associated with childhood cancer occurrence?
The Phase 1 report recommended these ecologic and case-control study designs based on scientific merit, a preliminary analysis of their technical feasibility, and their ability to address public concerns about cancer risks near nuclear facilities.
To conduct the pilot study, NAS and its contractors will make use of existing health information from state cancer registries and vital statistics offices and data from the facilities, the state, and other entities on radioactive effluent releases. There is no intention to conduct interviews with people who live near the pilot nuclear facilities or to make new measurements of radioactive effluent releases from the facilities or in the environment. Still, the committee judges that the pilot study will likely require substantial effort and resources. The monetary cost for the retrieval of the data, construction of databases, and subsequent analyses will also be substantial.
This report comprises the committee’s advice to NAS about general methodological considerations in carrying out the pilot study of cancer risks near nuclear facilities. It is not intended to be a comprehensive workplan of how to conduct the pilot study—this is outside the committee’s study charge. The committee has also provided advice on study requirements and specifications that are described in a request for information that was issued to the public on October 7, 2014.4 These study requirements and specifications will be further developed at the next step of the pilot, the pilot execution step.
This brief report differs from typical reports of the National Research Council of the National Academy of Sciences and National Academy of Engineering that provide full supporting rationale and context for study findings and recommendations. The committee’s advice to NAS was informed by the independent expert views of its members (see Appendix C for committee membership and expertise) and by information gathered during this study (see Appendix B). The committee held meetings to obtain information from the Electric Power Research Institute, the USNRC, subject-matter experts, several nonprofit organizations, and a variety of other stakeholder groups and individuals. It also received a number of written comments. Additional information was received when a subgroup of the committee hosted a public meeting near the Oyster Creek Nuclear Generating Station.
The committee’s advice is presented in the form of 14 considerations related to the pilot study procedures (1-5), general methodologies (6-10) and specific methodologies for conducting the ecologic (11-13) and case-control studies (14). This brief report was subject to the National Research Council’s report review process prior to its release. The names and affiliations of the reviewers are presented in Appendix D.
Procedural Considerations for Conducting the Pilot Study
1. The pilot study should be guided by the principles of independence, objectivity, ethics, openness, and excellence. In addition, transparency should be a central aim throughout the pilot study. This involves providing to interested parties information on the study process and regular updates on the study progress and making information accessible to the public (except where health data privacy rules or other ethical/legal considerations preclude disclosure).
2. Stakeholders should be given the opportunity to provide input on issues relevant to the study throughout the course of the pilot study. Such input could be obtained by following different and complementary models including forming a stakeholder advisory board and inviting stakeholders to speak at meetings. Ongoing communication with stakeholders during the study is essential.
3. All tasks of the pilot study should have a procedure manual that describes the specific approach to carrying
________________
4 NAS issued a request for information (RFI) regarding the provision of research support and the associated costs for executing the pilot study. Attachment A to the RFI is the first draft of the pilot study requirements and specifications. See http://nas-sites.org/cancerriskstudy/.
out the task in sufficient detail to support replication by others. It is important to demonstrate transparency by making these procedure manuals available upon request.
4. The recommended study designs have several inherent limitations as well as limitations due to the unavailability of information to accurately reconstruct doses to the study populations. For example, in the absence of information on residential history, the recommended study designs by necessity will make assumptions about relevant exposures based on information about location of residence at one time point in the lifetime of the study populations, such as place of residence at time of diagnosis or death or place of residence of the mother at the time of delivery of the child. The pilot study should provide a comprehensive discussion of the assumptions and resulting uncertainties and their potential impacts on the risk assessments.
5. As noted in the Phase 1 report, the pilot study is intended to be a feasibility5 study, not a small-scale study of analysis of risks around the pilot nuclear facilities, and is also intended to identify potential modifications needed in the design of the nationwide study. These objectives of the pilot study should be made clear to the stakeholders.
Committee members have different views about whether the pilot study should test specific hypotheses and about the appropriateness and utility of presenting risk estimates from the pilot study. However, there was full agreement among committee members about the following three points:
i. Any data collected during the pilot study will have limited use for estimating cancer risks in populations near each of the nuclear facilities or for the seven nuclear facilities combined because of the imprecision inherent in estimates from small samples.
ii. Interpretation and communication of risk estimates from the pilot study, if reported, should be done with great caution.
iii. The decision to proceed with the nationwide study should be based solely on conclusions related to feasibility and not on risk estimates.
General Methodological Considerations for Health and Dosimetry Data Processing
6. State cancer registries and vital statistics offices that hold the health information needed to conduct the pilot study should, to the extent possible, use the same protocol for preparing the data for release to the contractors who will carry out the pilot study. This includes protocols for linking cancer registration and birth records, assigning census tracts, and other procedures related to data acquisition. Having a common protocol makes it easier to compare and integrate data from different sources. The successful development and use of a common protocol would require regular communication among the state entities that hold the information, the contractors who will carry out the pilot study, and the NAS committee and staff that will provide study oversight.
7. The methodology used for the assessment of the annual doses from atmospheric and aquatic effluent releases from the individual nuclear power plants will be similar across the plants because the radionuclides released and the pathways of human exposure (inhalation, ingestion, and direct radiation) are the same. The specific model parameters used for the assessment of radionuclide concentrations in air and water, however, may differ from plant to plant depending on the complexity of the geography, weather patterns, nearby water bodies, and other factors. Independent of the models used, particular attention should be paid to the releases of carbon-14, which is believed to account for a substantial fraction of the doses in recent years.
8. A modified methodology for the assessment of the annual doses from atmospheric and aquatic effluent
________________
5 Feasibility refers to the ability to
• Obtain sufficient nuclear facility airborne and waterborne effluent release and meteorology data,
• Obtain cancer incidence and mortality data at the census-tract level, and
• Link birth registration and cancer incidence data to identify eligible cases of pediatric cancers and matched controls.
Explicit criteria for feasibility will be defined during the pilot execution step.
releases to that used for the nuclear power plants will need to be used for the Nuclear Fuel Services (NFS) facility. NFS is a uranium fuel fabrication facility that releases primarily alpha emitters, unlike nuclear power plants which release primarily beta and gamma emitters. Thus, for NFS, internal irradiation via inhalation or ingestion is the most important pathway of exposure.
9. The pilot study should independently validate a sample of effluent release data reported by each of the pilot nuclear facilities. Validation of the releases and dose estimates can include demonstrating that the doses could not have exceeded specific levels based on detection limits of environmental monitoring by independent entities such as states and research organizations.
10. There should be an investigation of available data on variations in natural background radiation levels with distance and direction from the pilot nuclear facilities. Such an investigation would determine whether sufficient information exists for consideration of natural background radiation as a potential confounder in the epidemiological analysis.
11. Risk estimates from the ecologic study should be interpreted with caution because of the inherent limitations of the study design; these include aggregation of information and the difficulty of characterizing within-area (census tract) variability in exposures and potential confounders. The pilot ecologic study could provide indications of the heterogeneity of populations across census tracts and possible confounding by characterizing demographic differences of the populations in relation to proximity to the nuclear facilities.
12. Investigation of available data conducted by NAS staff during the current study highlighted two limitations of information collected by the state vital statistics offices that may compromise the feasibility of an ecologic study of cancer mortality:
i. The year at which address at time of death from cancer6 is first recorded electronically ranges from 1949 to 2008. In fact, five out of the seven pilot states for which information is available started collecting address at time of death from cancer electronically in 2000 or later.
ii. Information on the specific cancer site as underlying cause of death may be missing from the death certificate in some states. Also, in some cases, if a cancer metastasizes, the metastatic site may be listed as the underlying cause of death instead of the primary cancer site.
13. The abovementioned limitations in carrying out and interpreting findings from the ecologic study and the uncertainties in estimating exposure for a population where data for individuals are not known outweigh the benefits from performing a detailed dose assessment. Still, the ecologic study should include a facility-specific dose assessment in addition to a distance-based analysis of risks.7 This dose assessment can be based on simplifying assumptions (e.g., that there have been no changes with time on water use, land use and food consumption and origin) and using annual average concentrations of radionuclides released and commensurate averaging of meteorological data. These simplifications would reduce the amount of effort and resources required; however, they would still be substantial because doses need to be calculated starting from the first year that the nuclear facility of interest started operation in order for the study to
________________
6 Information on address at time of death from cancer is central to estimating cancer mortality risks for the census tracts within 50 kilometers from the pilot nuclear facilities.
7 This is because distance alone is not a good indicator of the doses received by the populations of the various census tracts. There are several
reasons why the dose pattern around a nuclear facility may be complex and cannot be predicted using distance alone. Two are discussed here:
i The distribution of wind speed and direction is not isotropic around a nuclear facility and therefore airborne releases are not transported uniformly in all directions.
ii The population of a given census tract would typically consume water and foodstuffs that, at least in part, originate from another census tract.
assess cancer risks later in life. For some nuclear facilities this would require estimating releases and calculating doses associated with about 55 years of facility operation.
Specific Considerations for the Pilot Case-Control Study of Childhood Cancers
14. The dose assessment will start as early as 16 years before a case is diagnosed.8 Many assumptions will have to be made to estimate risks because residential history and dietary and lifestyle habits of the children examined will not be available. The dose assessment should use more detailed information than that for the ecologic study to determine concentrations of radionuclides released and time-specific parameters such as water use, land use, and amounts and origins of consumed foodstuffs.
________________
8 This applies to a case diagnosed at age 15. Since the case-control study will attempt to answer the question of whether estimated radiation exposure during pregnancy is associated with childhood cancer occurrence, doses will be calculated also for the 9 months (for convenience rounded to 1 year) of pregnancy.
This page intentionally left blank.








