ROLE OF POULTRY IN SPREAD OF NOVEL H7N9 INFLUENZA VIRUS IN CHINA32
Mary J. Pantin-Jackwood,33Patti J. Miller,33Erica Spackman,33David E. Swayne,33Leonardo Susta,33Mar Costa-Hurtado,33 and David L. Suarez33
Abstract
The recent outbreak of H7N9 influenza in China has resulted in many human cases with a high fatality rate. Poultry are the likely source of infection for humans based on sequence analysis and virus isolations from live bird markets, but it’s not clear which species of birds are most likely to be infected and shedding sufficient levels of virus to infect humans. Intranasal inoculation of chickens, Japanese quail, pigeons, Pekin ducks, Mallard ducks, Muscovy ducks, and Embden geese with 106 EID50 of the A/Anhui/1/2013 virus resulted in infection but no clinical disease signs. Virus shedding in quail and chickens was much higher and prolonged than in the other species. Quail effectively transmitted the virus to direct contacts but pigeons and Pekin ducks did not. In all species, virus was detected at much higher titers from oropharyngeal swabs than cloacal swabs. The HA gene from samples collected from selected experimentally infected birds were sequenced and three amino acid differences were commonly observed when compared to A/Anhui/1/2013: N123D, N149D, and L217Q. Leucine at position 217 is highly conserved for avian isolates and is associated with α2,6 sialic acid binding. Different amino acid combinations were observed suggesting that the inoculum had viral subpopulations that were selected after passage in birds. These experimental studies corroborate that certain poultry species are reservoirs of the H7N9 influenza virus, and that the virus is highly upper respiratorytropic so testing of bird species should preferentially be conducted with oropharyngeal swabs for best sensitivity.
IMPORTANCE: The recent outbreak of H7N9 in China has resulted in a number of human infections with a high case fatality rate. The source of the viral outbreak is suspected to be from poultry, but definitive data for
_______________
32 Reprinted with permission from the American Society for Microbiology. Originally published as Pantin-Jackwood, et al. 2014. Role of poultry in spread of novel H7N9 influenza virus in China. Journal of Virology 88(10):5381-5390.
33 Exotic and Emerging Avian Disease Research Unit, Southeast Poultry Research Laboratory, Agricultural Research Service, USDA, Athens, GA.
the source of the infection is not known. This study provides experimental data to show that quail and chickens are susceptible to infection and shed large amounts of virus and are likely important in the spread of the virus to humans. Other poultry species, including Muscovy ducks, can be infected and shed virus, but are less likely to play a role of transmitting the virus to humans. Pigeons were previously suggested as a possible source of virus because of isolation of virus from several pigeons in poultry markets in China, but experimental studies show they are generally resistant to infection and are unlikely to play a role in spread of the virus.
On 1 April 2013, the People’s Republic of China reported the first 3 human cases of a novel H7N9 subtype of influenza A virus. Over the subsequent days, the number of confirmed cases ballooned to over 82, with over 17 fatalities occurring in 6 different provinces (Li et al., 2014). Sequence analysis of the virus showed the H7 and N9 genes to be those of avian influenza viruses (AIVs) of Eurasian lineage, but at only 95% similarity to other isolates in the public sequence databases, the viruses were uniquely different from what had previously been described (Gao et al., 2013). However, the internal genes were all closely related to the well-established H9N2 lineage of avian influenza virus circulating in poultry in the region since at least 1997 (Gao et al., 2013; Guan et al., 1999). This H9N2 lineage virus has also been associated with human disease (Butt et al., 2005; Peiris et al., 1999).
The epidemiology of H7N9 virus showed that human cases were widely distributed in the affected provinces and there was no strong evidence of human-to-human transmission. Because the genome sequences of the isolates showed that they were genetically related to AIVs, Chinese veterinary officials quickly started testing poultry associated with live bird markets and commercial poultry operations and wild birds in the regions where human infections were being reported. The H7N9 viruses were detected at relatively low rates in avian species in the live bird markets, including chickens, pigeons, and ducks, and the environment (Lam et al., 2013; Shi et al., 2013a). Additional evidence of an epidemiological link of exposure to birds in markets has been found in some human cases (Chen et al., 2013b; Shi et al., 2013b). Therefore, live bird markets were suspected of being a source of human infections, and Chinese officials required temporary closure of live poultry markets in the affected provinces, resulting in an immediate reduction of human cases, providing further evidence of a role of live poultry markets in the spread of the virus and that closure of the markets is an effective control strategy (Yu et al., 2014). However, it is not clear which species of birds are most likely to be infected and are shedding levels of virus sufficient to infect humans. The lack of understanding of the virus ecology in birds has recently resulted in an additional number of human cases, demonstrating that the virus still circulates in China (Chen et al., 2013a). Based on the initial reports of this virus and previous experience with avian influenza, we evaluated the potential role of different poultry species in the epidemiology of H7N9 influenza.
Materials and Methods
Virus
The virus used in this study was A/Anhui/1/2013 (H7N9), which was kindly provided by the Centers for Disease Control and Prevention, Atlanta, GA. The virus was propagated in specific-pathogen-free (SPF) embryonating chicken eggs (ECEs) according to standard procedures (Senne, 1998). Allantoic fluid was diluted in brain heart infusion (BHI) medium (BD Bioscience, Sparks, MD) in order to prepare an inoculum with 102, 104, or 106 50% egg infective doses (EID50) per 0.1 ml per bird. All challenge doses were confirmed by back-titration in ECEs. All experiments using the H7N9 influenza virus, including work with animals, were reviewed by the institutional biosecurity committee and were performed in biosecurity level 3 enhanced (BSL-3E) and animal biosecurity level 3 enhanced (ABSL-3E) facilities at the Southeast Poultry Research Laboratory (SEPRL), Agricultural Research Service, United States Department of Agriculture (USDA), and all personnel were required to wear a powered air-purifying respirator with high-efficiency particulate air (HEPA) filtration (3M, St. Paul, MN).
Birds
Fifty-nine-week-old SPF White Leghorn chickens (Gallus gallus domesticus) (egg layer type) were obtained from SEPRL in-house flocks. Four-week-old Japanese quail (Coturnix japonica), 6- to 12-month-old rock pigeons (Columbia livia domestica), 2-week-old Pekin ducks (Anas platyrhynchos var. domestica), 2-week-old Mallard ducks (Anas platyrhynchos), 2-week-old Muscovy ducks (Cairina moschata), and 2-week-old Embden geese (Anser anser domesticus) were obtained from commercial farms. Birds were housed in self-contained isolation units that were ventilated under negative pressure with HEPA-filtered air and maintained under continuous lighting. Serum samples were collected from all birds immediately prior to challenge and found to be negative for antibodies to the H7 subtype of influenza virus by hemagglutination inhibition assay, as described below. Feed and water were provided with ad libitum access. All bird experiments were approved by and performed under the regulations of the SEPRL Institutional Animal Care and Use Committee.
Pathogenicity and Virus Transmission Studies
Eleven chickens, 11 Japanese quail, 11 pigeons, 11 Pekin ducks, 11 Mallard ducks, 7 Muscovy ducks, and 9 Embden geese were intranasally (i.n.) inoculated through the choanal cleft with an inoculum containing 106.0 EID50 of A/Anhui/1/2013 (H7N9) in 0.1 ml. At 3 days postinoculation (dpi), 2 to 3 birds from each group were euthanized and gross lesions were recorded. The following tissues were collected in 10% neutral buffered formalin solution to determine microscopic lesions and the extent of virus replication in tissues: nasal turbinates,
trachea, lung, air sac, comb, eyelid, heart, brain, esophagus, proventriculus, ventriculus, duodenum, jejunum, cecal tonsils, pancreas, liver, spleen, bursa, thymus, Harderian gland, kidney, gonads, adrenal gland, and muscle from the breast and left thigh. Lung, spleen, intestine, kidney, and thigh and breast muscle tissues, as well as allantoic fluid from eggs laid by inoculated chickens, were collected separately in BHI and kept frozen at −70°C for subsequent virus detection. The remaining birds were observed for clinical signs over an 11-day period, during which time any clinical signs were recorded.
To evaluate the susceptibility of quail, pigeons, and Pekin ducks, three doses of virus (102, 104, or 106 EID50) were administered i.n. to groups of five birds. At 2 dpi, three uninfected birds were placed in the same cage with the directly inoculated birds in each dose group to determine the transmission potential of the virus by contact exposure.
Oropharyngeal (OP) and cloacal (C) swab specimens were collected at 2, 4, 6, 8, and 11 dpi from directly inoculated birds and at 2, 4, 6, and 9 days after birds infected through contact exposure to determine virus shedding. Swab specimens were collected in 2 ml of BHI medium with 1× antibiotic-antimycotic. All birds remaining at the end of the experiment were euthanized by the intravenous (i.v.) administration of sodium pentobarbital (100 mg/kg of body weight).
RNA Extraction and Quantitative rRT-PCR
RNA was extracted from swab and tissue specimens using a MagMAX 96 AI/ND viral RNA isolation kit (Ambion, Inc. Austin, TX) with a KingFisher magnetic particle processor (Thermo Scientific, Waltham, MA). Quantitative real-time reverse transcription-PCR (rRT-PCR) was performed using a SmartCycler (version 2.0) apparatus and an AgPath-ID OneStep RT-PCR kit (Ambion, Inc.). The processing of the samples from the chicken, quail, and pigeon experiment was performed at the same time, and an H7 assay was used for quantitation (Slomka et al., 2009). For the duck and goose samples, the matrix gene assay was used for quantitation (Spackman et al., 2002). A standard curve for virus quantification was established with RNA extracted from dilutions of the same titrated stock of virus used to inoculate the birds. Viral titers were extrapolated from the standard curve. Data were analyzed using Prism (v.5.01) software (GraphPad Software Inc.). Two-way analysis of variance with Tukey’s posttest was used to the compare virus titers in oropharyngeal swab specimens. For statistical purposes, all oropharyngeal swab specimens from which viruses were not detected were given a numeric value of 101.0 EID50/ml. Statistical significance was set at a P value of <0.05.
Virus Replication in Tissues
Virus replication in lung, spleen, intestine, kidney, and muscle tissues from 2 to 3 birds was examined at 3 days following intranasal infection with the H7N9 virus. Titers of infectious virus were determined in ECEs or by rRT-PCR, as described above. Allantoic fluid collected from virus-inoculated laying chickens was also examined by rRT-PCR for the presence of virus.
HI Assays
Hemagglutination inhibition (HI) assays were used to evaluate antibody to H7 influenza virus prior to challenge and to confirm exposure and infection with serum collected at 11 dpi and preadsorbed with 10% chicken red blood cells (Pedersen, 2008). The HI assay was conducted in accordance with standard procedures. Briefly, 2-fold serial dilutions of 25µl of serum were made in 25µl of phosphate-buffered saline (PBS). Diluted sera were incubated for 30 min at 4°C with 4 hemagglutination units (HAU)/25µl of A/Anhui/1/2013 (H7N9) virus which had been treated with 0.1% beta-propiolactone (the pH was adjusted to 7.0 with sodium bicarbonate), and then 50µl of 0.1% chicken red blood cells was added. The test result was evaluated after 30 min of incubation at room temperature. Titers were calculated as the reciprocal of the last HI-positive serum dilution, and samples with HI titers of 8 or below were considered negative.
Histopathology and IHC
Tissues were prepared for histopathology and immunohistochemistry (IHC) as previously described (Pantin-Jackwood and Swayne, 2007). Briefly, collected tissues were fixed by submersion in 10% neutral buffered formalin and embedded in paraffin. In addition, the nasal cavity was decalcified for 2 days. Sections were made at 5µm and were stained with hematoxylin-eosin (HE). A duplicate section was immunohistochemically stained by first microwaving the sections in Citra antigen retrieval solution (BioGenex, San Ramon, CA) for antigen exposure. A 1:2,000 dilution of a mouse-derived monoclonal antibody (P13C11) (Perdue et al., 1994) specific for type A influenza virus nucleoprotein (NP) was applied, and the mixture was allowed to incubate overnight at 4°C (Perkins and Swayne, 2001). The primary antibody was then detected by the application of biotinylated goat anti-mouse IgG secondary antibody using a biotin-streptavidin detection system (supersensitive multilink immunodetection system; BioGenex). Fast Red TR (BioGenex) served as the substrate chromogen, and hematoxylin was used as a counterstain. All tissues were systematically screened for microscopic lesions and virus antigen staining.
Sequencing
Viral RNA from selected OP and cloacal swab samples collected at 2 to 11 dpi from birds of each species directly inoculated with virus and from contact-exposed birds was directly amplified by one-step reverse transcription-PCR (RT-PCR) for the HA1 gene and the region of the PB2 protein around amino acid position 627. Selected viruses isolated from the experimentally inoculated birds and propagated in ECEs were also sequenced for comparison. A commercial one-step RT-PCR kit (Qiagen Inc., Valencia, CA) and primers which matched the sequence of A/Anhui/1/2013 (H7N9) virus and which were directed to the conserved sequences at the ends of the gene segments were used. Primer sequences are available upon request. Templates were then purified by agarose gel extraction with a QIAquick gel extraction kit (Qiagen Inc., Valencia, CA) and quantified by UV spectroscopy. A BigDye Terminator kit (Applied Biosystems, Foster City, CA) was used for cycle sequencing, and the samples were subsequently run on an ABI 3730 DNA analyzer (Applied Biosystems, Foster City, CA). Bioinformatics analysis looking at the amino acid differences of the hemagglutinin (HA) gene was performed using the Influenza Research Database, which was used to look for single nucleotide polymorphisms (Squires et al., 2012).
TABLE A13-1 Susceptibility of Poultry to Influenza A (H7N9) Virus
| No. of birds virus positive/ total no. of birds sampled at the following timesa: |
||||
| 2 dpi | 4 dpi | |||
| Group | OP swabs | C swabs | OP swabs | C swabs |
| Chicken layers | 10/11 (6.0)A | 4/11 (2.1) | 8/8 (6.4)AC | 4/8 (2.5) |
| Quail | 11/11 (7.6)B | 10/11 (2.9) | 8/8 (7.5)A | 8/8 (3.2) |
| Pigeons | 7/11 (2.5)C | 5/11 (1.9) | 1/8 (1.7)B | 0/8 |
| Pekin ducks | 9/11 (5.1)C | 0/11 | 3/8 (4.5)BD | 0/8 |
| Mallard ducks | 10/11 (4.0)C | 0/11 | 2/8 (4.2)BD | 0/8 |
| Muscovy ducks | 7/7 (5.3)A | 2/7 (3.2) | 5/5 (5.9)CE | 3/5 (4.9) |
| Embden geese | 7/9 (3.4)C | 0/9 | 5/6 (4.4)DE | 0/6 |
a Results of testing for influenza A (H7N9) virus in oropharyngeal (OP) and cloacal (C) swab specimens from birds inoculated intranasally with 106 EID50 of the virus. The results are presented as the average viral shedding for each day, and samples negative by rRT-PCR were given a value 1 log unit lower than the limit of detection. Values in parentheses are the mean virus titer for positive samples determined by quantitative rRT-PCR and are reported as the log10 number of EID50/ml. The
Results
Pathogenicity of H7N9 Influenza Virus in Different Poultry Species
No clinical disease signs were observed in any of the directly inoculated or contact-exposed birds during the 11-day observation period. Results for virus shedding are presented in Table A13-1. Virus was detected in OP swabs taken at 2 dpi from all the quail and Muscovy ducks and from 10 of 11 chickens, 7 of 11 pigeons, 9 of 11 Pekin ducks, 10 of 11 Mallard ducks, and 7 of 9 Embden geese inoculated with 106.0 EID50 of the virus. At this time point, virus was detected in cloacal swabs from 4 of 11 chickens, 10 of 11 quail, 5 of 11 pigeons, 2 of 7 Muscovy ducks, and in none of the Pekin ducks, Mallard ducks, and geese. At 4 dpi, virus was detected in OP swabs of all chickens, quail, and Muscovy ducks but in smaller numbers of pigeons, Pekin ducks, Mallard ducks, and geese. Cloacal swab specimens from all quail, 3 of 5 Muscovy ducks, and 4 of 8 the chickens were virus positive, but swab specimens from the rest of the birds were negative. By 6 dpi, pigeons, Pekin ducks, and Mallard ducks had stop shedding by both the OP and C routes. Two of 6 geese had positive OP swabs at this time but were negative for both OP and C swabs on the rest of the days. Limited numbers of
| 6 dpi | 8 dpi | 11 dpi | ||||
| OP swabs | C swabs | OP swabs | C swabs | OP swabs | C swabs | Serologyb |
| 7/7 (5.5)A | 3/8 (2.1) | 6/8 (3.3)A | 0/8 | 1/8 (1.8)A | 0/8 | 5/6 (64–128) |
| 7/8 (2.6)B | 7/8 (2.6) | 6/8 (2.9)A | 4/8 (2.2) | 1/8 (2.0)A | 1/8 (1.7) | 8/8 (32–128) |
| 0/8 | 0/8 | 0/8 | 0/8 | 0/8 | 0/8 | 0/7 (5) |
| 0/8 | 0/8 | 0/8 | 0/8 | 0/8 | 0/8 | 3/8 (16) |
| 0/8 | 0/8 | 0/8 | 0/8 | 0/8 | 0/8 | 1/8 (16) |
| 5/5 (4.0)B | C 2/5 (4.1) | 3/5 (3.1)A | 3/5 (3.4) | 0/5 | 0/5 | 4/5 (16–32) |
| 2/6 (3.4)C | 0/6 | 0/6 | 0/6 | 0/6 | 0/6 | 0/6 |
estimated lower limit of sensitivity of the rRT-PCR test was 102, as determined by the limit of detection on the standard curve. For oropharyngeal virus shedding, the results for groups with different uppercase letters are significantly different (P < 0.05). dpi, days postinoculation.
b Data represent the number of positive birds/total number of birds tested (range of titers or titer by hemagglutination inhibition assay). Titers of 8 or below were considered negative.
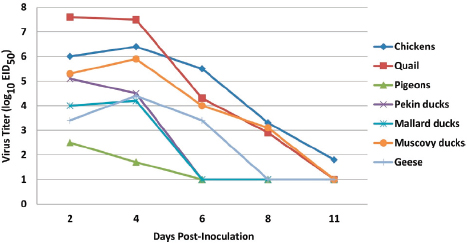
FIGURE A13-1 Comparison of oropharyngeal virus shedding after experimental challenge. Oropharyngeal shedding was detected at 2, 4, 6, 8, and 11 dpi with 106 EID50 of A/Anhui/1/2013 (H7N9) virus. The rRT-PCR values were interpolated by quantitative real-time RT-PCR using a standard curve generated with the challenge isolate. The estimated lower limit of sensitivity of the rRT-PCR test was 102, as determined by the limit of detection on the standard curve. The results are presented as the average viral shedding for each day, and samples negative by rRT-PCR were given a value 1 log unit lower than the limit of detection.
chicken and quail continued shedding virus until 11 dpi, and some Muscovy ducks were still shedding virus at 8 dpi.
High virus titers were present in OP swabs, with quail shedding an average of 107.5 EID50 on days 2 and 4 dpi and chickens shedding 106.2 EID50, on average, on the same days. Most chickens and quail continued shedding virus at 6 and 8 dpi, although the titers had decreased (Figure A13-1). The titers in cloacal swabs averaged 3 to 4 log units lower than those in OP swabs. Only 7 of 11 directly exposed pigeons in the pathogenesis experimental group shed detectable virus at 2 dpi, with most shedding virus at low titers close to the assay limit of detection. One pigeon with the highest titer at day 2 continued to shed virus on day 4, but no other pigeons had detectable viral shedding for the remainder of the study. In the waterfowl study, Muscovy ducks had the highest numbers of birds shedding virus and the highest titers, with all birds shedding virus by the oropharyngeal route on days 2, 4, and 6 and some shedding virus by the cloaca until day 8. The Pekin ducks, Mallard ducks, and Embden geese had similar patterns of infection, with most directly inoculated birds shedding virus by the oropharyngeal route on day 2 and with decreasing numbers of positive birds being detected on days 4 and 6 and no virus being detected from the cloacal swabs (Table A13-1).
Susceptibility and Transmission of H7N9 Influenza Virus in Quail, Pigeons, and Pekin Ducks
Quail, pigeons, and Pekin ducks were chosen on the basis of the differences in pathogenicity observed as described above. Results are presented in Table A13-2. All directly inoculated quail in all three dose groups eventually became infected, and virus was transmitted to all contacts. Only a single quail receiving 102 EID50 was infected at 2 dpi, but it shed enough virus to infect its cage mates. In contrast, although some pigeons in the groups inoculated with 104 and 106 EID50 were shedding virus at 2 dpi, none of the contact-exposed pigeons became infected. No virus was detected from the pigeons that received 102 EID50 of the virus. Similarly, 2 of 5 and 4 of 5 Pekin ducks in the groups receiving 104 and 106 EID50, respectively, became infected, but the virus was transmitted to only 2 contact ducks, which shed virus for only a short period of time.
Gross and Microscopic Lesions and Virus Antigen Staining in Tissues
Very mild gross lesions were observed at necropsy and mainly consisted of mild sinusitis. Microscopic lesions were consistent with low-pathogenic AIV (LPAIV) infection. Most of the lesions were confined to the upper respiratory tract. In the chickens and quail, the virus caused moderate to severe catarrhal and/or lymphocytic rhinitis and sinusitis, with mucocellular exudates containing sloughed epithelial cells, submucosal edema, and glandular hyperplasia (Figure A13-2). The trachea presented mild degenerative changes of the overlying epithelium, mild lymphocytic infiltration in the submucosa, and mild edema (Figure A13-2). In the lung, mild congestion, mild interstitial inflammation with mixed mononuclear cells, and mild catarrhal bronchitis were observed. Lesions in the gastrointestinal tract consisted of mild proliferation of gut-associated lymphoid tissues (GALTs). The remaining organs lacked significant histopathologic lesions. In ducks and geese, similar to the chickens and quail, most of the microscopic lesions were found in the upper respiratory tract (nasal turbinates, trachea); however, no lesions were observed in any other tissues, including the enteric tract. Mild to severe catarrhal rhinitis with congestion and loss of epithelial cells lining the nasal cavity was present in some ducks. In others, lymphoplasmacytic inflammation of the nasal submucosa was observed. Tracheitis with exudates and epithelial loss was common. In geese, mild to moderate lymphocytic rhinitis was the only lesion observed. No significant lesions were found in noninoculated birds.
In order to determine sites of virus replication, immunohistochemical staining for AIV antigen with an antibody to NP was conducted. Common viral staining was present in the epithelial cells and macrophages of the nasal cavity and adjacent glands in all of the quail and chickens examined (Figure A13-2). Similar viral antigen staining was present in the nasal epithelium of 2 of 3 Pekin ducks, 3 of 3 Mallards, and 1 of 2 Muscovy ducks but not in the geese or pigeons. Viral staining was also present in epithelial cells, macro phages, and desquamated
TABLE A13-2 Transmission of Influenza A (H7N9) Virus in Quail, Pigeons, and Ducks
| Group/virus dose | No. of birds virus positive/ total no. of birds sampled at the following timesa: |
|||
| 2 dpi | 4 dpi | |||
| OP swabs | C swabs | OP swabs | C swabs | |
| Quail/102 EID50 | 1/5 (2.4) | 0/5 | 2/5 (3.0) | 2/5 (1.9) |
| Quail/102 EID50 contacts | 1/3 (1.7) | 0/3 | ||
| Quail/104 EID50 | 5/5 (7.5) | 5/5 (3.0) | 5/5 (7.4) | 4/5 (2.7) |
| Quail/104 EID50 contacts | 3/3 (7.6) | 2/3 (2.3) | ||
| Quail/106 EID50 | 5/5 (7.6) | 5/5 (2.9) | 5/5 (7.5) | 5/5 (3.2) |
| Quail/106 EID50 contacts | 3/3 (7.7) | 3/3 (3.0) | ||
| Pigeon/102 EID50 | 0/5 | 0/5 | 0/5 | 0/5 |
| Pigeon/102 EID50 contacts | 0/3 | 0/3 | ||
| Pigeon/104 EID50 | 2/5 (2.2) | 1/5 | 0/5 | 0/5 |
| Pigeon/104 EID50 contacts | 0/3 | 0/3 | ||
| Pigeon/106 EID50 | 2/5 (2.5) | 2/5 (1.9) | 0/5 | 0/5 |
| Pigeon/106 EID50 contacts | 0/3 | 0/3 | ||
| Pekin duck/102 EID50 | 0/5 | 0/5 | 0/5 | 0/5 |
| Pekin duck/102 EID50 contacts | 0/3 | 0/3 | ||
| Pekin duck/104 EID50 | 2/5 (3.7) | 0/5 | 1/5 (4.4) | 0/5 |
| Pekin duck/104 EID50 contacts | 0/3 | 0/3 | ||
| Pekin duck/106 EID50 | 4/5 (4.0) | 0/5 | 1/5 (2.7) | 0/5 |
| Pekin duck/106 EID50 contacts | 0/3 | 0/3 | ||
a Results of testing for influenza A (H7N9) virus in oropharyngeal (OP) and cloacal (C) swabs specimens from inoculated birds and birds exposed through contact. Values in parentheses are the mean virus titer for positive samples determined by quantitative rRT-PCR and are reported as the log10 number of EID50/ml. The estimated lower limit of sensitivity of the rRT-PCR test was 102, as
cells of the trachea of one quail and one Muscovy duck and in enterocytes and submucosal macrophages in the intestine of one quail and two Muscovy ducks.
Virus Detection in Tissue Samples
Virus isolation and/or virus detection by rRT-PCR from tissues collected at 3 dpi from birds infected with 106 EID50 of the H7N9 virus was attempted. Low
| 6 dpi | 8 dpi | 11 dpi | ||||
| OP swabs | C swabs | OP swabs | C swabs | OP swabs | C swabs | Serologyb |
| 5/5 (5.8) | 3/5 (2.1) | 5/5 (6.1) | 4/5 (4.8) | 5/5 | 4/5 | 3/5 (8–64) |
| 3/3 (4.0) | 0/3 | 3/3 (6.1) | 0/3 | 3/3 (7.0) | 2/3 (2.1) | 1/3 (8) |
| 5/5 (5.8) | 4/5 (2.7) | 4/5 (2.8) | 4/5 (2.7) | 0/5 | 0/5 | 5/5 (16–256) |
| 3/3 (6.1) | 2/3 (2.6) | 3/3 (4.5) | 1/3 (2.2) | 1/3 (2.3) | 2/3 (2.3) | 3/3 (32) |
| 5/5 (4.3) | 3/5 (2.6) | 3/5 (2.9) | 3/5 (2.2) | 2/5 | 1/5 | 5/5 (74) |
| 3/3 (7.9) | 2/3 (2.7) | 3/3 (4.9) | 3/3 (3.2) | 3/3 (2.2) | 3/3 (2.2) | 3/3 (8–128) |
| 0/5 | 0/5 | 0/5 | 0/5 | 0/5 | 0/5 | 1/5 (16) |
| 0/3 | 0/3 | 0/3 | 0/3 | 0/3 | 0/3 | 0/3 |
| 0/5 | 0/5 | 0/5 | 0/5 | 0/5 | 0/5 | 0/5 |
| 0/3 | 0/3 | 0/3 | 0/3 | 0/3 | 0/3 | 0/3 |
| 0/5 | 0/5 | 0/5 | 0/5 | 0/5 | 0/5 | 0/5 |
| 0/3 | 0/3 | 0/3 | 0/3 | 0/3 | 0/3 | 0/3 |
| 0/5 | 1/5 | 0/5 | 0/5 | 0/5 | 0/5 | 0/5 |
| 0/3 | 0/3 | 0/3 | 0/3 | 0/3 | 0/3 | 0/3 |
| 0/5 | 0/5 | 0/5 | 0/5 | 0/5 | 0/5 | 1/5 (16) |
| 1/3 (3.8) | 0/3 | 2/3 (4.5) | 0/3 | 0/3 | 0/3 | 0/3 |
| 0/5 | 0/5 | 0/5 | 0/5 | 0/5 | 0/5 | 1/5 (32) |
| 0/3 | 0/3 | 0/3 | 0/3 | 0/3 | 0/3 | 0/3 |
determined by the limit of detection on the standard curve. The results are presented as the average viral shedding for each day, and samples negative by rRT-PCR were given a cycle threshold value 1 log unit lower than the limit of detection. dpi, days postinoculation.
b Data represent the number of positive birds/total number of birds tested (range of titers or titer by hemagglutination inhibition assay). Titers of 8 or below were considered negative.
virus titers (100.97 to 101.23 EID50) were detected in the intestine of two Muscovy ducks, one quail, and one chicken; in the spleen of three quail, one chicken, and one Mallard duck; and in the kidney of one Pekin duck, two Mallard ducks, one Muscovy duck, and two geese. However, the lungs and muscle tissues and the contents of eggs laid by infected chickens were virus negative.
Serology
When examined at 11 dpi, all quail, most chickens and Muscovy ducks, and some Pekin and Mallard ducks infected with 106 EID50 of the H7N9 virus had detectable titers of antibodies against the virus, but no pigeons or geese seroconverted (Table A13-1). All quail given 104 to 106 EID50 of the virus and 3 of 5 quail given 102 EID50 also seroconverted (Table A13-2), and antibodies were also detected in contact-exposed quail. However, most pigeons and Pekin ducks had undetectable antibody titers regardless of the virus dose given.
Sequencing
The HA1 gene and part of the PB2 gene were RT-PCR amplified and sequenced from selected virus-positive OP swabs collected at 2 to 11 dpi from inoculated chickens (n = 3), quail (n = 2), a pigeon (n = 1), a Pekin duck (n = 1), and a Muscovy duck (n = 1) and from contact control birds chickens (n = 3) and quail (n = 6). Similar numbers of sequences were obtained from virus isolated in embryonating chicken eggs from OP swabs. All samples had lysine (K) at position 627 of the PB2 gene, which is the same as the sequence of the parent human isolate. Lysine at this position is associated with virulence in mammals (Hatta, 2001). However, three amino acid differences were commonly observed in the HA1 gene sequence compared to the A/Anhui/1/2013 sequence: N123D, N149D, and L217Q (H7 numbering) (Table A13-3). Both the virus that was originally received from China and the virus that was used as the inoculum for these experiments and that had been passaged once in embryonating chicken eggs were sequenced. While the virus received from China had asparagine (N) residues at both positions 123 and 149, the egg-passaged inoculum virus had aspartic acid
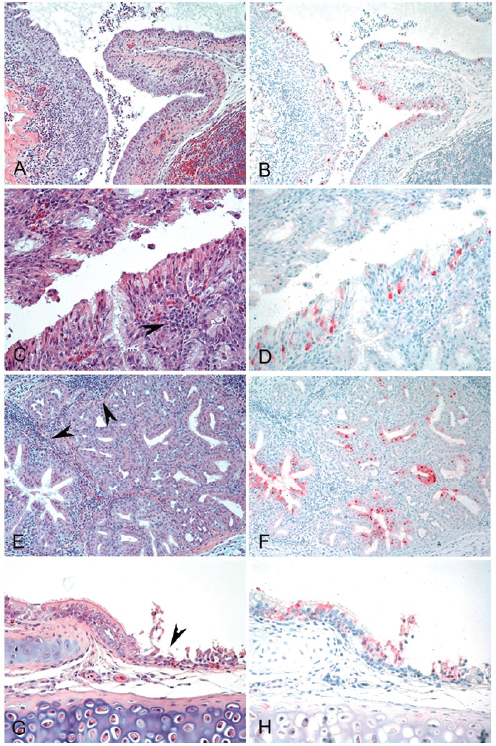
FIGURE A13-2 Histopathology and immunohistochemical staining for avian influenza virus antigen in tissues of quail intranasally infected with the A/Anhui/1/2013 (H7N9) virus 3 dpi. Virus is stained in red. (A and B) Nasal epithelium. Severe necrotizing rhinitis with submucosal congestion and edema, glandular hyperplasia, and lymphoplasmacytic infiltration (A) and viral antigen in epithelial cells (B) are shown (magnification, ×200). (C and D) Nasal epithelium. Necrosis of epithelial cells and lymphocytic infiltration (arrowhead) (C) and viral antigen in epithelial cells (D) are shown (magnification, ×400). (E and F) Nasal gland. Lymphocytic infiltration in submucosa (arrowheads) (E) and viral antigen in epithelial cells (F) are shown (magnification, ×200). (G and H) Trachea. Necrosis of epithelial cells (arrowhead) (G) and viral antigen staining in epithelial cells (H) are shown (magnification, ×400).
| Virus or virus source | No. of viruses with identical sequences | Source | Sequence in the following proteinb: | |||
| H7 position | H7 Position | H7 position | PB2 position | |||
| 123 (132) | 149 (158) | 217 (226) | 627 | |||
| A/Anhui/1/2013 (egg passage 2) | 1 | Original | N | N | L | K |
| A/Anhui/1/2013 (egg passage 3) | 1 | Inoculum | D | D | L | K |
| Chicken | 1 | Challenged | D | D | Q | K |
| Chicken | 2 | Challenged | N | N | Q | K |
| Chicken, egg isolation | 1 | D | D | Q | K | |
| Chicken, egg isolation | 2 | N | N | Q | K | |
| Quail | 1 | Challenged | D | D | Q | K |
| Quail | 1 | Challenged | D | D | L | K |
| Quail | 5 | Contact | D | D | Q | K |
| Quail | 1 | Contact | D | D | L | K |
| Quail, egg isolation | 8 | D | D | Q | K | |
| Pigeon, egg isolation | 1 | D | D | Q | K | |
| Pekin duck | 1 | Challenged | D | D | Q | K |
| Mallard duck | 1 | Challenged | N | N | L | K |
| Muscovy duck | 1 | Challenged | N | N | L | K |
a Viruses were obtained directly from swabs or after egg passage.
b Numbers in parentheses are the analogous positions in H3.
(D) at both positions. Both isolates maintained the leucine (L) at position 217. Four different combinations of amino acids were observed in the infected birds. Most birds in this study continued to have aspartic acid at positions 123 and 149, but some isolates from chickens and Mallard and Pekin ducks had asparagine at both positions. The most consistent difference observed was the change at position 217 from leucine (L) to glutamine (Q). Position 217 correlates to position 226 in H3 human influenza viruses, and position 226 is critical for determining specificity to either α2,6 human-like sialic acid receptors or α2,3 avian-like sialic acid receptors (Connor et al., 1994). Most of the Chinese H7N9 sequences have leucine at this position, but passage in poultry seems to provide selection pressure for glutamine. Leucine was still found in a minority of isolates from both challenged and contact-infected birds. All the viruses that were passaged in eggs had glutamine at position 217. Examination of GenBank for sequence variation of Eurasian H7 influenza viruses at position 217 showed that over 99% of AIVs have glutamine at this position, and none are reported to have leucine. At position 123, asparagine is found in over 97% of viruses and aspartic acid is found in less than 3% of viruses, and at position 149, asparagine is found in 99% of isolates, with no viruses having aspartic acid.
Discussion
Quail are experimentally susceptible to many subtypes of both mammalian and avian influenza viruses (Bonfante et al., 2013; Cilloni et al., 2010; Makarova et al., 2003; Perez et al., 2003) and have been proposed to be a bridging species or disease amplifiers between wild waterfowl and domestic gallinaceous poultry (Cilloni et al., 2010; Hossain et al., 2008; Sorrell and Perez, 2007; Thontiravong et al., 2012; Yamada et al., 2012). In this study, we demonstrate that quail are susceptible to even a low-dose challenge of the Chinese H7N9 virus. The virus replicated to high titers in the upper respiratory tract for at least a week, and the virus transmitted easily by direct contact to cage mates. Although quail are proportionally a minor poultry species, they have the potential to be a major reservoir of the H7N9 virus for transmission to other poultry and to humans. The adult chickens in this study also shed high levels of virus, indicating that chickens are also an important source of virus which could be infecting humans either through direct contact or by aerosolization of virus, which occurs in particular during the slaughter process in live bird markets (Belser, 2010).
Our data support the suggestion that pigeons are generally resistant to AIV infection, with only 1 of 26 pigeons shedding moderate titers of virus. Historically, isolation of AIV from pigeons is rare, with less than 40 sequences appearing in GenBank, including the sequences of 8 H9N2 viruses and no H7 viruses (Squires et al., 2012). Experimentally, pigeons have generally been resistant to H5N1 highly pathogenic AIV (HPAIV) infection (Boon et al., 2007; Liu et al., 2007; Perkins and Swayne, 2001; Yamamoto et al., 2012); however, inoculation
of pigeons with high virus doses or with specific strains resulted in infrequent morbidity and mortality (Jia et al., 2008; Klopfleisch et al., 2006; Werner et al., 2007). Experimental data suggest that pigeons are unlikely to play a major role in the maintenance and transmission of the Chinese H7N9 virus. Exposure to high levels of virus from chickens or other species in live bird markets could explain the reported H7N9 isolations from pigeons (Shi et al., 2013a).
In this study, we also examined the pathogenesis of the H7N9 influenza virus in three different types of ducks and one type of goose. Pekin and Muscovy ducks and Embden geese are domestic waterfowl frequently present in live bird markets in China. Although closely related to Pekin ducks, we chose to also include Mallard ducks to address the possibility that wild birds could become infected with this virus by contact with domestic poultry and possibly spread the virus to other areas. Wild Mallard ducks also have one of the highest isolation rates for AIV and are a primary reservoir in the wild (Stallknecht et al., 2008). All four species could be infected by high-dose challenge with the virus, but the birds did not show any clinically observable disease and most species shed relatively small amounts of virus for shorter periods of time than quail and chickens. There was some evidence of contact transmission in Pekin ducks, but the ducks infected through contact shed little virus and shed virus for only short periods of time. Of the four species, Muscovy ducks shed the most virus. This is not surprising, since Muscovy ducks have been shown to be more susceptible to infection with highly pathogenic H5N1 AIV strains, show more severe disease, and shed larger amounts of virus than other domestic duck species (Cagle et al., 2011, 2012; Pantin-Jackwood et al., 2013). Muscovy ducks, it must be remembered, are a different species (Cairina moschata) than Pekin and Mallard ducks (Anas platyrhynchos) and should not be expected to have a similar response to infection (Brown et al., 2006). These differences in response to AIV infection in different waterfowl species should be taken into account when determining which species are involved in the transmission of emerging viruses. In this study, Muscovy ducks appeared to play a more important role as a possible biological vector of H7N9 AIV than Pekin ducks, Mallard ducks, and Embden geese.
AIV is maintained in wild birds, but occasionally, the virus can spread from its natural reservoir to poultry. Wild-bird AIVs are generally poorly adapted to domestic galliformes (chickens, quail, partridge), but as conditions permit, the virus can be transmitted and adapt to the new host. Wild aquatic birds do not typically show clinical signs of infection with AIVs, and although AIVs can replicate in cells of both the respiratory and intestinal tracts, in ducks they are reported to favor the intestinal tract (Swayne and Slemons, 2008; Webster et al., 1978). The results of these studies are consistent with those of previous studies indicating that chicken-adapted AIVs replicate better in chickens than in ducks (Pillai et al., 2010; Spackman et al., 2010). The underlining mechanism is not clear, but a shorter neuraminidase protein due to a deletion in the stalk region may be linked to this feature (Banks et al., 2001; Matrosovich et al., 1999; Mundt et al., 2009).
Control of H7N9 influenza is complicated by the lack of disease signs in poultry. Detection of LPAIVs is more difficult than detection of HPAIVs, like H5N1, because testing cannot target sick or dead birds like syndromic surveillance can. Critically, the data from both quail and chickens show high levels of viral replication in the upper respiratory tract and the shedding of much less virus in cloacal swabs, findings which are not unexpected, because poultry-adapted AIVs are typically shed at much higher levels in the respiratory tract in gallinaceous poultry (Claes et al., 2013; Gonzales et al., 2012; Marché et al., 2012; Pillai et al., 2010; Spackman et al., 2010). The disease pathogenesis of the H7N9 virus was unusual, in that virus replication was primarily restricted to the upper respiratory tract for all the species examined and the virus did not replicate well in lungs. Testing of gallinaceous and waterfowl bird species should preferentially be conducted with OP swabs for the best sensitivity.
One of the unusual features of this H7N9 virus was the presence of leucine at position 217 in the HA1 protein, a sequence which many have speculated increased the ability of the virus to infect humans. This position, analogous to position 226 of H3 viruses, forms part of the receptor binding site. The presence of leucine at this position is associated with binding to α2,6-sialic acid, which is the primary type of sialic acid found in the human upper respiratory tract. The presence of glutamine at this position is associated with binding to α2,3-sialic acid, the most common type of sialic acid found in avian species (Naeve et al., 1984; Vines et al., 1998). Receptor binding studies have confirmed that the human H7N9 virus with leucine at position 226 binds more strongly to α2,6-sialic acid than an avian H7 strain, but the human virus also had strong binding to α2,3-sialic acid (Xiong et al., 2013). Examining published Eurasian H7 sequences, glutamine is almost exclusively found at position 217 in avian influenza viruses, so the presence of leucine in the human isolate appears to support a change for adaptation to humans. The change of leucine back to glutamine in most of the challenged birds shows positive selection for glutamine in poultry and because of the mixed results suggests that the challenge virus was a mixed population. The change of L217Q in egg-passaged virus is also not surprising, as egg adaptation of human viruses is a common occurrence (Rocha et al., 1993). Experimental studies examining a European H7 virus showed that when the double mutation of Q226L and G228S was introduced into the hemagglutinin protein, the tropism of the hemagglutinin changed from α2,3-sialic acid to α2,6-sialic acid (Spackman et al., 2002), further supporting our observations. The single mutation Q226L was not examined, but because most Chinese H7N9 viruses already have leucine, it suggests that a single additional change could result in a major shift in viral tropism. Two other amino acid changes at positions 123 and 149 were also commonly observed. The amino acid at position 123 is part of the 120 loop that is projected to form part of the receptor binding site, but it is not clear how this amino acid might affect viral binding (Yang et al., 2012). The role of aspartic acid at position 149 also remains unclear. The data from this study and the sequence data for H7 viruses
in general suggest a conundrum about the H7N9 reservoir, because although the H7N9 virus with leucine at position 226 was transmitted in poultry, the majority of transmitted virus in this study had glutamine. Are there other animal species in the Chinese wet markets that may be a bridge species that supports leucine at position 226, or does the Q226L mutation occur after the virus jumps the species barrier to humans?
The epidemiology of the H7N9 virus outbreak clearly has an important poultry component, on the basis of the sequence analysis and the epidemiology of the virus, and these experimental studies have shown that chickens and quail are likely important reservoirs of the virus and pigeons are not. The role of domestic waterfowl is less clear, with relatively high levels of virus being shed by Muscovy ducks and smaller amounts being shed by Pekin ducks and Embden geese. Efforts in China have targeted live bird markets in an attempt to control the virus, and understanding the contribution of poultry farms, wholesale markets, and the markets themselves in the maintenance of the virus is critical. In the live bird market system in the United States, poultry farms are generally free of infected birds, but with H7N2 LPAIV, wholesale markets seemed to be the major sources of the virus (Bulaga et al., 2003).
The H7 virus has infected humans on numerous occasions, although the clinical disease has usually been mild. The Chinese H7N9 virus is unusual in the manifestation of severe disease in humans with a high case fatality rate. The viral sequence suggests that the virus is poised to mutate to a form that has 2,6-sialic acid receptor specificity, which is likely a prerequisite for human-to-human transmission, which could lead to a highly virulent pandemic. An understanding of viral pathogenesis and key reservoir species can potentially allow interventions in animals to stop a future human pandemic.
Acknowledgments
We thank Tim Olivier, Suzanne Deblois, Diane Smith, Kira Moresco, Mark Freeman, Michelle Edenfield, Scott Lee, and Raul Otalora for technical assistance and Ruben Donis and Todd Davis at the Centers for Disease Control and Prevention for providing the challenge virus.
This research was supported by U.S. Department of Agriculture CRIS Project 6612-32000-063-00D and federal funds from the National Institute of Allergy and Infectious Diseases, National Institutes of Health (NIH), under IAA no. AAI12004-001-00001.
The contents of this article are solely the responsibility of the authors and do not necessarily represent the official views of the NIH.
References
Banks, J., E. S. Speidel, E. Moore, L. Plowright, A. Piccirillo, I. Capua, P. Cordioli, A. Fioretti, and D. J. Alexander. 2001. Changes in the haemagglutinin and the neuraminidase genes prior to the emergence of highly pathogenic H7N1 avian influenza viruses in Italy. Archives of Virology 146(5):963-973.
Belser, J. A. T., T. M. Tumpey, J. M. Katz, and D. E. Swayne. 2010. Possible transmission modes for avian influenza viruses to people: Studies in experimental models (Abstract). Influenza and Other Respiratory Viruses 4(Suppl 1):38.
Bonfante, F., L. V. Patrono, R. Aiello, M. S. Beato, C. Terregino, and I. Capua. 2013. Susceptibility and intra-species transmission of the H9N2 G1 prototype lineage virus in Japanese quail and turkeys. Veterinary Microbiology 165(1–2):177-183.
Boon, A. C., M. R. Sandbulte, P. Seiler, R. J. Webby, T. Songserm, Y. Guan, and R. G. Webster. 2007. Role of terrestrial wild birds in ecology of influenza A virus (H5N1). Emerging Infectious Diseases 13(11):1720-1724.
Brown, J. D., D. E. Stallknecht, J. R. Beck, D. L. Suarez, and D. E. Swayne. 2006. Susceptibility of North American ducks and gulls to H5N1 highly pathogenic avian influenza viruses. Emerging Infectious Diseases 12(11):1663-1670.
Bulaga, L. L., L. Garber, D. A. Senne, T. J. Myers, R. Good, S. Wainwright, S. Trock, and D. L. Suarez. 2003. Epidemiologic and surveillance studies on avian influenza in live-bird markets in New York and New Jersey, 2001. Avian Diseases 47(s3):996-1001.
Butt, K., G. J. Smith, H. Chen, L. Zhang, Y. C. Leung, K. Xu, W. Lim, R. G. Webster, K. Yuen, and J. M. Peiris. 2005. Human infection with an avian H9N2 influenza A virus in Hong Kong in 2003. Journal of Clinical Microbiology 43(11):5760-5767.
Cagle, C., T. L. To, T. Nguyen, J. Wasilenko, S. C. Adams, C. J. Cardona, E. Spackman, D. L. Suarez, and M. J. Pantin-Jackwood. 2011. Pekin and Muscovy ducks respond differently to vaccination with a H5N1 highly pathogenic avian influenza (HPAI) commercial inactivated vaccine. Vaccine 29(38):6549-6557.
Cagle, C., J. Wasilenko, S. C. Adams, C. J. Cardona, T. L. To, T. Nguyen, E. Spackman, D. L. Suarez, D. Smith, E. Shepherd, J. Roth, and M. J. Pantin-Jackwood. 2012. Differences in pathogenicity, response to vaccination, and innate immune responses in different types of ducks infected with a virulent H5N1 highly pathogenic avian influenza virus from Vietnam. Avian Diseases 56(3):479-487.
Chen, E., Y. Chen, L. Fu, Z. Chen, Z. Gong, H. Mao, D. Wang, M. Ni, P. Wu, and Z. Yu. 2013a. Human infection with avian influenza A (H7N9) virus re-emerges in China in winter 2013. Euro Surveillance 18(43).
Chen, Y., W. Liang, S. Yang, N. Wu, H. Gao, J. Sheng, H. Yao, J. Wo, Q. Fang, D. Cui, Y. Li, X. Yao, Y. Zhang, H. Wu, S. Zheng, H. Diao, S. Xia, Y. Zhang, K.-H. Chan, H.-W. Tsoi, J. L.-L. Teng, W. Song, P. Wang, S.-Y. Lau, M. Zheng, J. F.-W. Chan, K. K.-W. To, H. Chen, L. Li, and K.-Y. Yuen. 2013b. Human infections with the emerging avian influenza A H7N9 virus from wet market poultry: Clinical analysis and characterisation of viral genome. Lancet 381(9881):1916-1925.
Cilloni, F., A. Toffan, S. Giannecchini, V. Clausi, A. Azzi, I. Capua, and C. Terregino. 2010. Increased pathogenicity and shedding in chickens of a wild bird–origin low pathogenicity avian influenza virus of the H7N3 subtype following multiple in vivo passages in quail and turkey. Avian Diseases 54(s1):555-557.
Claes, G., S. Welby, T. Van Den Berg, Y. Van Der Stede, J. Dewulf, B. Lambrecht, and S. Marché. 2013. The impact of viral tropism and housing conditions on the transmission of three H5/H7 low pathogenic avian influenza viruses in chickens. Epidemiology and Infection 141(11):2428-2443.
Connor, R. J., Y. Kawaoka, R. G. Webster, and J. C. Paulson. 1994. Receptor specificity in human, avian, and equine H2 and H3 influenza virus isolates. Virology 205(1):17-23.
Gao, R., B. Cao, Y. Hu, Z. Feng, D. Wang, W. Hu, J. Chen, Z. Jie, H. Qiu, K. Xu, X. Xu, H. Lu, W. Zhu, Z. Gao, N. Xiang, Y. Shen, Z. He, Y. Gu, Z. Zhang, Y. Yang, X. Zhao, L. Zhou, X. Li, S. Zou, Y. Zhang, X. Li, L. Yang, J. Guo, J. Dong, Q. Li, L. Dong, Y. Zhu, T. Bai, S. Wang, P. Hao, W. Yang, Y. Zhang, J. Han, H. Yu, D. Li, G. F. Gao, G. Wu, Y. Wang, Z. Yuan, and Y. Shu. 2013. Human infection with a novel avian-origin influenza A (H7N9) Virus. New England Journal of Medicine 368(20):1888-1897.
Gonzales, J. L., A. R. W. Elbers, A. Bouma, G. Koch, J. J. de Wit, and J. A. Stegeman. 2012. Transmission characteristics of low pathogenic avian influenza virus of H7N7 and H5N7 subtypes in layer chickens. Veterinary Microbiology 155(2–4):207-213.
Guan, Y., K. F. Shortridge, S. Krauss, and R. G. Webster. 1999. Molecular characterization of H9N2 influenza viruses: Were they the donors of the “internal” genes of H5N1 viruses in Hong Kong? Proceedings of the National Academy of Sciences 96(16):9363-9367.
Hatta, M. N., G.Neumann, and Y. Kawaoka. 2001. Reverse genetics approach towards understanding pathogenesis of H5N1 Hong Kong influenza A virus infection. Philosophical Transactions of the Royal Society of London. Series B: Biological Sciences 356(1416):1841-1843.
Hossain, M. J., D. Hickman, and D. R. Perez. 2008. Evidence of expanded host range and mammalian-associated genetic changes in a duck H9N2 influenza virus following adaptation in quail and chickens. PloS One 3(9):e3170.
Jia, B., J. Shi, Y. Li, K. Shinya, Y. Muramoto, X. Zeng, G. Tian, Y. Kawaoka, and H. Chen. 2008. Pathogenicity of Chinese H5N1 highly pathogenic avian influenza viruses in pigeons. Archives of Virology 153(10):1821-1826.
Klopfleisch, R., O. Werner, E. Mundt, T. Harder, and J. P. Teifke. 2006. Neurotropism of highly pathogenic avian influenza virus A/chicken/Indonesia/2003 (H5N1) in experimentally infected pigeons (Columbia livia f. domestica). Veterinary Pathology Online 43(4):463-470.
Lam, T. T.-Y., J. Wang, Y. Shen, B. Zhou, L. Duan, C.-L. Cheung, C. Ma, S. J. Lycett, C. Y.-H. Leung, X. Chen, L. Li, W. Hong, Y. Chai, L. Zhou, H. Liang, Z. Ou, Y. Liu, A. Farooqui, D. J. Kelvin, L. L. M. Poon, D. K. Smith, O. G. Pybus, G. M. Leung, Y. Shu, R. G. Webster, R. J. Webby, J. S. M. Peiris, A. Rambaut, H. Zhu, and Y. Guan. 2013. The genesis and source of the H7N9 influenza viruses causing human infections in China. Nature 502(7470):241-244.
Li, Q., L. Zhou, M. Zhou, Z. Chen, F. Li, H. Wu, N. Xiang, E. Chen, F. Tang, D. Wang, L. Meng, Z. Hong, W. Tu, Y. Cao, L. Li, F. Ding, B. Liu, M. Wang, R. Xie, R. Gao, X. Li, T. Bai, S. Zou, J. He, J. Hu, Y. Xu, C. Chai, S. Wang, Y. Gao, L. Jin, Y. Zhang, H. Luo, H. Yu, J. He, Q. Li, X. Wang, L. Gao, X. Pang, G. Liu, Y. Yan, H. Yuan, Y. Shu, W. Yang, Y. Wang, F. Wu, T. M. Uyeki, and Z. Feng. 2014. Epidemiology of human infections with avian influenza A(H7N9) virus in China. New England Journal of Medicine 370(6):520-532.
Liu, Y., J. Zhou, H. Yang, W. Yao, W. Bu, B. Yang, W. Song, Y. Meng, J. Lin, C. Han, J. Zhu, Z. Ma, J. Zhao, and X. Wang. 2007. Susceptibility and transmissibility of pigeons to Asian lineage highly pathogenic avian influenza virus subtype H5N1. Avian Pathology 36(6):461-465.
Makarova, N. V., H. Ozaki, H. Kida, R. G. Webster, and D. R. Perez. 2003. Replication and transmission of influenza viruses in Japanese quail. Virology 310(1):8-15.
Marché, S., G. Claes, S. Van Borm, D. Vangeluwe, T. van den Berg, and B. Lambrecht. 2012. Different replication profiles in specific-pathogen-free chickens of two H7 low pathogenic avian influenza viruses isolated from wild birds. Avian Diseases 56(4s1):959-965.
Matrosovich, M., N. Zhou, Y. Kawaoka, and R. Webster. 1999. The surface glycoproteins of H5 influenza viruses isolated from humans, chickens, and wild aquatic birds have distinguishable properties. Journal of Virology 73(2):1146-1155.
Mundt, E., L. Gay, L. Jones, G. Saavedra, S. M. Tompkins, and R. Tripp. 2009. Replication and pathogenesis associated with H5N1, H5N2, and H5N3 low-pathogenic avian influenza virus infection in chickens and ducks. Archives of Virology 154(8):1241-1248.
Naeve, C., V. Hinshaw, and R. Webster. 1984. Mutations in the hemagglutinin receptor-binding site can change the biological properties of an influenza virus. Journal of Virology 51(2):567-569.
Pantin-Jackwood, M. J., and D. E. Swayne. 2007. Pathobiology of Asian highly pathogenic avian influenza H5N1 virus infections in ducks. Avian Diseases 51(s1):250-259.
Pantin-Jackwood, M., D. E. Swayne, D. Smith, and E. Shepherd. 2013. Effect of species, breed and route of virus inoculation on the pathogenicity of H5N1 highly pathogenic influenza (HPAI) viruses in domestic ducks. Veterinary Research 44:62.
Pedersen, J. C. 2008. Neuraminidase-Inhibition Assay for the Identification of Influenza A Virus Neuraminidase Subtype or Neuraminidase Antibody Specificity. In Avian Influenza Virus. Vol. 436, Methods in Molecular Biology, edited by E. Spackman. New York: Humana Press. Pp. 67-75.
Peiris, M., K. Y. Yuen, C. W. Leung, K. H. Chan, P. L. S. Ip, R. W. M. Lai, W. K. Orr, and K. F. Shortridge. 1999. Human infection with influenza H9N2. Lancet 354(9182):916-917.
Perdue, M. L., J. Latimer, C. Greene, and P. Holt. 1994. Consistent occurrence of hemagglutinin variants among avian influenza virus isolates of the H7 subtype. Virus Research 34(1):15-29.
Perez, D. R., R. J. Webby, E. Hoffmann, and R. G. Webster. 2003. Land-based birds as potential disseminators of avian/mammalian reassortant influenza A viruses. Avian Diseases 47(s3): 1114-1117.
Perkins, L. E. L., and D. E. Swayne. 2001. Pathobiology of A/chicken/Hong Kong/220/97 (H5N1) avian influenza virus in seven gallinaceous species. Veterinary Pathology Online 38(2):149-164.
Pillai, S. P. S., M. Pantin-Jackwood, D. L. Suarez, Y. M. Saif, and C. W. Lee. 2010. Pathobiological characterization of low-pathogenicity H5 avian influenza viruses of diverse origins in chickens, ducks and turkeys. Archives of Virology 155(9):1439-1451.
Rocha, E. P., X. Xu, H. E. Hall, J. R. Allen, H. L. Regnery, and N. J. Cox. 1993. Comparison of 10 influenza A (H1N1 and H3N2) haemagglutinin sequences obtained directly from clinical specimens to those of MDCK cell-and egg-grown viruses. Journal of General Virology 74:2513-2513.
Senne, D. 1998. Virus propagation in embryonating eggs. A laboratory manual for the isolation and identification of avian pathogens. Kennet Square, PA: American Association of Avian Pathologists. Pp. 235-240.
Shi, J., G. Deng, P. Liu, J. Zhou, L. Guan, W. Li, X. Li, J. Guo, G. Wang, J. Fan, J. Wang, Y. Li, Y. Jiang, L. Liu, G. Tian, C. Li, and H. Chen. 2013a. Isolation and characterization of H7N9 viruses from live poultry markets — Implication of the source of current H7N9 infection in humans. Chinese Science Bulletin 58(16):1857-1863.
Shi, J., J. Xie, Z. He, Y. Hu, Y. He, Q. Huang, B. Leng, W. He, Y. Sheng, F. Li, Y. Song, C. Bai, Y. Gu, and Z. Jie. 2013b. A detailed epidemiological and clinical description of 6 human cases of avian-origin influenza A (H7N9) virus infection in Shanghai. PloS One 8(10):e77651.
Slomka, M. J., T. Pavlidis, V. J. Coward, J. Voermans, G. Koch, A. Hanna, J. Banks, and I. H. Brown. 2009. Validated real-time reverse transcriptase PCR methods for the diagnosis and pathotyping of Eurasian H7 avian influenza viruses. Influenza and Other Respiratory Viruses 3(4):151-164.
Sorrell, E. M., and D. R. Perez. 2007. Adaptation of influenza A/Mallard/Potsdam/178-4/83 H2N2 virus in Japanese quail leads to infection and transmission in chickens. Avian Diseases 51(s1): 264-268.
Spackman, E., D. A. Senne, T. Myers, L. L. Bulaga, L. P. Garber, M. L. Perdue, K. Lohman, L. T. Daum, and D. L. Suarez. 2002. Development of a real-time reverse transcriptase PCR assay for type A influenza virus and the avian H5 and H7 hemagglutinin subtypes. Journal of Clinical Microbiology 40(9):3256-3260.
Spackman, E., J. Gelb, L. A. Preskenis, B. S. Ladman, C. R. Pope, M. J. Pantin-Jackwood, and E. T. Mckinley. 2010. The pathogenesis of low pathogenicity H7 avian influenza viruses in chickens, ducks and turkeys. Virology Journal 7(331):974-977.
Squires, R. B., J. Noronha, V. Hunt, A. García-Sastre, C. Macken, N. Baumgarth, D. Suarez, B. E. Pickett, Y. Zhang, C. N. Larsen, A. Ramsey, L. Zhou, S. Zaremba, S. Kumar, J. Deitrich, E. Klem, and R. H. Scheuermann. 2012. Influenza Research Database: An integrated bioinformatics resource for influenza research and surveillance. Influenza and Other Respiratory Viruses 6(6):404-416.
Stallknecht, D. E., J. D. Brown, and D. Swayne. 2008. Ecology of avian influenza in wild birds. In Avian Influenza. Ames, IA: Blackwell Publishing. Pp.43-58.
Swayne, D. E., and R. D. Slemons. 2008. Using mean infectious dose of high- and low-pathogenicity avian influenza viruses originating from wild duck and poultry as one measure of infectivity and adaptation to poultry. Avian Diseases 52(3):455-460.
Thontiravong, A., P. Kitikoon, S. Wannaratana, R. Tantilertcharoen, R. Tuanudom, S. Pakpinyo, J. Sasipreeyajan, K. Oraveerakul, and A. Amonsin. 2012. Quail as a potential mixing vessel for the generation of new reassortant influenza A viruses. Veterinary Microbiology 160(3–4):305-313.
Vines, A., K. Wells, M. Matrosovich, M. R. Castrucci, T. Ito, and Y. Kawaoka. 1998. The role of influenza A virus hemagglutinin residues 226 and 228 in receptor specificity and host range restriction. Journal of Virology 72(9):7626-7631.
Webster, R. G., M. Yakhno, V. S. Hinshaw, W. J. Bean, and K. Copal Murti. 1978. Intestinal influenza: Replication and characterization of influenza viruses in ducks. Virology 84(2):268-278.
Werner, O., E. Starick, J. Teifke, R. Klopfleisch, T. Y. Prajitno, M. Beer, B. Hoffmann, and T. C. Harder. 2007. Minute excretion of highly pathogenic avian influenza virus A/chicken/Indonesia/2003 (H5N1) from experimentally infected domestic pigeons (Columbia livia) and lack of transmission to sentinel chickens. Journal of General Virology 88(11):3089-3093.
Xiong, X., S. R. Martin, L. F. Haire, S. A. Wharton, R. S. Daniels, M. S. Bennett, J. W. McCauley, P. J. Collins, P. A. Walker, J. J. Skehel, and S. J. Gamblin. 2013. Receptor binding by an H7N9 influenza virus from humans. Nature 499(7459):496-499.
Yamada, S., K. Shinya, A. Takada, T. Ito, T. Suzuki, Y. Suzuki, Q. M. Le, M. Ebina, N. Kasai, H. Kida, T. Horimoto, P. Rivailler, L. M. Chen, R. O. Donis, and Y. Kawaoka. 2012. Adaptation of a duck influenza A virus in quail. Journal of Virology 86(3):1411-1420.
Yamamoto, Y., K. Nakamura, M. Yamada, and M. Mase. 2012. Limited susceptibility of pigeons experimentally inoculated with H5N1 highly pathogenic avian influenza viruses. Journal of Veterinary Medical Science 74(2):205-208.
Yang, H., P. J. Carney, R. O. Donis, and J. Stevens. 2012. Structure and receptor complexes of the hemagglutinin from a highly pathogenic H7N7 influenza virus. Journal of Virology 86(16): 8645-8652.
Yu, H., J. T. Wu, B. J. Cowling, Q. Liao, V. J. Fang, S. Zhou, P. Wu, H. Zhou, E. H. Y. Lau, D. Guo, M. Y. Ni, Z. Peng, L. Feng, H. Jiang, H. Luo, Q. Li, Z. Feng, Y. Wang, W. Yang, and G. M. Leung. 2014. Effect of closure of live poultry markets on poultry-to-person transmission of avian influenza A H7N9 virus: An ecological study. Lancet 383(9916):541-548.






















