The efficient production of useful and beneficial goods and services has been the cornerstone of industrial development, driving economic growth for more than two centuries.11 Throughout this period, the underpinning technologies driving industrialization have evolved in response to new scientific understanding, new technological capabilities, and new market demands. Insights into the chemical nature of matter, reaction mechanisms, and the role of physical and catalytic processes transformed the industrial landscape during the 19th century. By 1882, dyes such as indigo, previously extracted from natural substances and relying on significant manual labor, could now be synthesized and made affordable. Medicines such as aspirin were similarly isolated and synthesized, making them affordable and widely available. By the early 20th century, a new understanding of chemistry transformed crude oil into a feedstock for a vast array of chemical products ranging from plastics and paints to detergents and textiles. Discoveries in physics early in the 20th century also entered the industrial landscape, leading to electronics, computers, satellites, and mobile communications, transforming economies, cultures, and the global community.
The human use and improvement of biological processes is an ancient and vital contributor to human progress, from the earliest periods of domestication of crops and animals through the agricultural revolution to the contemporary world of life sciences. Until recently, however, it has remained an essentially empirical pursuit because of the seemingly impenetrable complexity of biological systems.
Much of the underlying science for the establishment of industrial
biotechnology began to emerge in the middle of the 20th century, particularly stemming from the discovery in 1953 of the structure of DNA by Crick and Watson and the realization that DNA’s double-helix structure provided a unique mechanism for encoding information. In the decades following this discovery, significant progress has been made in understanding the relationship between these underlying biological building blocks and the functional performance of biological systems. The arrival of increasingly rapid computers and massive data handling capacity at the beginning of the 21st century facilitated the translation of data derived from high-throughput screening methods into more robust and predictive design techniques. The convergence of the life sciences with chemistry, chemical engineering, computer science, and other disciplines has increased the potential for industrialization of the biological sciences for chemical manufacturing.
The Organisation for Economic Co-operation and Development (OECD) first defined the bioeconomy as linking renewable biological resources and bioprocesses through industrial-scale biotechnologies and manufacturing to produce sustainable products, jobs, and income.12 In its 2012 National Bioeconomy Blueprint, the Obama Administration redefined the bioeconomy simply as “one based on the use of research and innovation in the biological sciences to create economic activity and public benefit.”1 It went on to observe that the U.S. bioeconomy is “all around us” with new bio-based chemicals, improved public health through improved drugs and diagnostics, and biofuels that reduce our dependency on oil.
A proactive strategy—implemented through the development of a technical roadmap similar to those that enabled sustained growth in the semiconductor industry and our explorations of space—is needed if we are to realize the widespread benefits of accelerating the industrialization of biology.
A confluence of overlapping developments has created the conditions for making this achievable: the proliferation of emerging tools, technologies, and computational models; new investment opportunities and financial instruments; exciting new insights from scientific convergence and transdisciplinary research; innovative business models and entrepreneurial enterprises (large and small); new platforms for designing biological systems for next-generation American manufacturing; and novel opportunities to enhance competitiveness and create well-paying jobs. These trends, in turn, will transform existing chemical production, create new chemical and other sectors enabled by the industrialization of biology, and open a range of new markets for bio-based products resulting from advanced chemical manufacturing.
The roadmap proposed in this report underscores the widely held view that 21st-century innovation increasingly will rely on biology and, in
particular, the convergence of biology with engineering and physical sciences such as chemistry. As a National Research Council report predicted: “Discoveries at all levels of biology will reverberate throughout science and provide the transformational insights that will lead to practical solutions in seemingly unrelated research areas.”13 A roadmap for accelerating advanced chemical manufacturing through the industrialization of biology begins to operationalize President Obama’s 2011 observation that “[t]he world is shifting to an innovation economy and nobody does innovation better than America.”14
CHARGE TO THE COMMITTEE AND INTERPRETATION OF SCOPE
At the request of the National Science Foundation and the U.S. Department of Energy, the National Research Council appointed an ad hoc committee with a broad range of expertise to identify key technical milestones for chemical manufacturing through biological routes. The committee’s task included several key components: (1) the identification of the core scientific and technical challenges; (2) the identification of and timeline for the development of tools, measurement techniques, databases, and computational techniques needed to serve as the building blocks for research and applications; (3) a discussion of how to develop, share, and diffuse common interoperable standards, languages, and measurements; and (4) when and how to integrate nontechnological insights and societal concerns into the pursuit of the technical challenges (Box 1-1).
To address this task, the committee held a 2-day workshop in Washington, DC, to gather input from a range of experts and stakeholders. Speakers provided perspectives from the chemical industry process and experiences of scaling up (or out) production; insight into challenges in biosafety and biocontainment; and technical discussions of synthesis and genome-scale engineering, measurement, computer-aided design, and advanced molecules. Insights from this workshop served as a groundwork for the committee’s deliberations, with additional data gathering occurring throughout the study process.
The committee identified three dimensions that will require progress to ensure the acceleration of the industrialization of biology:
- the selection of the right chemical, material, and fuel targets, based on technical and economic criteria;
- continued progress in the rapidly developing science and technology that support industrialization of biology; and
- engagement with significant societal factors impacted by the acceleration of this industry.
BOX 1-1
Statement of Task
In order to realize the full benefit of research investments intended to enable the advanced manufacturing of chemicals using biological systems, an ad hoc committee will develop a roadmap of necessary advances in basic science and engineering capabilities, including knowledge, tools, and skills. Working at the interface of synthetic chemistry, metabolic engineering, molecular biology, and synthetic biology, the committee will identify key technical goals for this next-generation chemical manufacturing, then identify the gaps in knowledge, tools, techniques, and systems required to meet those goals, and targets and timelines for achieving them. It will also consider the skills necessary to accomplish the roadmap goals, and what training opportunities are required to produce the cadre of skilled scientists and engineers needed. While focused on industrial manufacturing of chemicals, the roadmap challenges identified here will also be relevant to applications in health, energy, environment, and agriculture by advancing the tools and techniues reuired for new develoment in these areas.
Essential elements of the roadmap that the committee will consider in the study and in its report, include the following:
- identification of the core scientific and technical challenges that must be overcome;
- identification of and timeline for tools, measurement techniques, databases, and computational techniques needed to serve as the building blocks for research and applications;
- how to develop, share, and diffuse common interoperable standards, languages, and measurements; and
- when and how to integrate nontechnological insights and societal concerns into the pursuit of the technical challenges.
The report will provide guidance to both the research and research funding communities regarding key challenges, knowledge, tools, and systems needed to advance the science and engineering required for advanced manufacturing of chemicals using biological systems and to develop the workforce required to realize these advances. The report will not include recommendations related to funding, government organization, or policy issues.
A number of key terms are used throughout this report. Because many of these terms do not necessarily have universally agreed-upon definitions, we define the following for the purposes of this report:
The bioeconomy refers to the portion of the economy that is derived from biological processes and manufacturing. With reference to Figure 1-1, feedstock refers to the starting material used in the manufacturing process. This may be a form of biomass, a crude or refined petroleum hydrocarbon product, or a material that has already been chemically modified
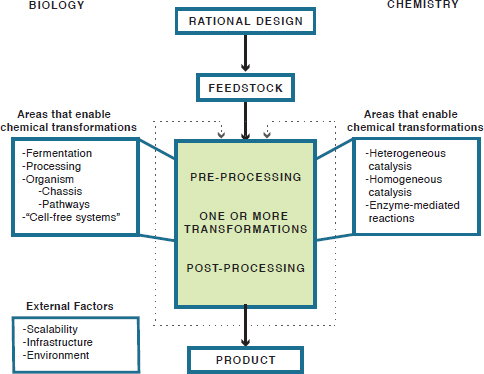
FIGURE 1-1 Chemical manufacturing flowchart showing the report’s conceptual schema of the chemical manufacturing process, from rational design to product.
in some way. Likewise, product refers to the material after it has been subject to a change in its chemical structure. Finally, transformation refers to a change in chemical structure. This could be via traditional chemical synthesis, biological routes, or both.
Biotechnology involves the “use of living cells, bacteria, etc., to make useful products.”15Genetic engineering encompasses the cutting and joining of recombinant DNA and its incorporation into an organism in order to change its characteristics,16 for example to make a new product or enhance its production. Genetic engineering is made up of a variety of technologies. Protein engineering seeks to modify the properties of an individual protein, for example to improve its stability or catalyze a new reaction. Metabolic engineering encompasses the purposeful modification of metabolic, gene regulatory, and signaling networks to achieve enhanced production of desired chemicals.
Synthetic biology is a newer discipline that seeks to deliver greater speed, cost-effectiveness, and predictability to the design of biological systems. The field applies engineering principles to reduce genetics into DNA “parts” and understand how they can be combined to build desired
functions in living cells. This has been driven in advances to build long stretches of DNA and “edit” the genomes of natural organisms. The UK Synthetic Biology Roadmap Coordination Group defined synthetic biology as “the design and engineering of biologically-based parts, novel devices and systems as well as the redesign of existing, natural biological systems.”17 Synthetic biology is a toolbox, not an end in itself. Advances in synthetic biology accelerate the industrialization of biology.
Human health, energy, the environment, and agriculture are important domains for the application of biotechnology. Tremendous progress has been made in these areas, and these developments will be greatly accelerated by advances toward the scientific and technological milestones discussed in this report. While this committee highlights the relevance of the industrialization of biology to health, energy, the environment, and food, the focus of this report is the production of chemicals through industrialization of biology.
The arrival of this emerging capability in biological science comes at a time when new approaches are eagerly sought. As the processing of petrochemicals has become an increasingly mature industry, new global challenges have begun to emerge. To sustain the needs of an increasing global population, the provision of goods and services must in turn become more sustainable, making more efficient use of fossil feedstocks and enabling the greater use of renewable feedstocks.
As a way to frame the discussions throughout this report, Figure 1-1 provides a conceptual framework for the chemical manufacturing process, including both biological and traditional chemical routes to chemical transformations. At its most basic level, the chemical manufacturing process has four basic waypoints. After conception of the product, or properties of a product, to be made, the rational process design is considered. This includes considering the capabilities available in science and engineering, as well as beginning to consider possible chemical transformations that will lead to the product of choice. As part of the design process, a feedstock is selected. In the case of traditional chemical manufacturing, this feedstock may be crude oil. In the case of using a biological transformation, this may be a plant-derived material (e.g., switch grass, corn stover) or a crude hydrocarbon mixture. The green box represents the core components of the chemical transformation or transformations that will occur. In this case, feedstocks typically undergo some initial processing before being subject to the one or more chemical transformations that are required to generate a product. It is important to note here that either chemical or biological means may be used to enable this transformation.
Finally, some sort of post processing (e.g., separation of the product from a fermentation liquid) will yield a final product for sale, or an intermediate product that may undergo further transformations. This report addresses technical and societal challenges relevant to each aspect of this figure, including rational design, selection and development of feedstocks, preprocessing and process design, and various methods of chemical transformation. In addition, the report discusses many of the external factors that affect the entire production process, including scalability, infrastructure, the environment, and even legal and business frameworks.
This report concerns the use of biology in the production of chemicals for industrial and consumer use. As described in Figure 1-2, these materials include large-volume chemicals—the majority of which are produced through chemical routes today—and specialty chemicals, which may be uniquely suited to production via industrial biology. Large-volume (bulk) chemicals include final products such as fuels, and bulk chemical intermediates such as ethylene and butadiene, among others. Some specialty chemicals will be natural products that can be adapted to industrial biology, while other materials may be modifications of natural products, such as enzymes and polypeptides used for industrial purposes, as catalysts or additives.
Although many of the materials described in this report derive from renewable feedstocks—starch or cellulose-derived sugars—this report concerns chemicals and materials produced by the use of biological sciences, regardless of the specific starting materials used. Production processes that include both chemical transformations and biological processing are germane to this report.

FIGURE 1-2 Comparison of the technical, economic, and production differences between small-volume specialty chemical production and large-volume commodity chemical production.
The core technologies enabling the industrialization of biology are those that enable microbial biotechnology. The production of chemicals through biological processes may entail fermentation using living host organisms, “cell-free” bioprocessing, or simply enzyme-mediated syntheses. Synthetic biology is therefore at the heart of the ongoing industrialization of biology. Synthetic biology takes advantage of the science of recombinant DNA and the ability to read, write, and edit the DNA of microorganisms, allowing the design and construction of new, more efficient metabolic pathways.
These technologies generally, but not exclusively, affect our ability to perform chemical transformations through biological processes.
Several areas of science support the speed, efficiency, and cost of development of these technologies. Some important areas among these are the following:
- Advances in DNA sequencing and DNA synthesis have dramatically reduced the costs associated with synthetic biology. Proteomics and metabolomics continue to provide insights into the biochemistry of the cell. High-throughput techniques have accelerated the pace of metabolic engineering and reduced the time and expense associated with constructing metabolic pathways in host organisms.
- The tools of bioinformatics and cell profiling enable an ever more detailed understanding of gene expression and cell metabolism including the ability to collect, manipulate, interrogate, and share the large data sets associated with synthetic biology.
- Early efforts have been taken to increase the number and range of DNA “parts” that are available to engineer new functions into cells. This includes large enzyme lists that are being gleaned from the sequence databases, synthesized, and characterized for function. Also, efforts have been taken in the characterization of regulatory parts to better control expression with greater precision.18
- Modeling and visualization tools are critical to protein engineering. Predictive modeling is important at the level of proteins, the metabolic pathway, and whole-cell metabolism. Modeling is equally important at the macro level, from predicting how whole cells function as a population and interact with their environment to the design and operation of bioprocessing plants.
- The commercial-scale production of chemicals via bioprocessing requires the design and operation of large-scale facilities capable of economic production and purification of the chemical products. The science and technology needed for design and “scale-up” of new bioprocesses is important to the industrialization of biology.
Acceleration of the industrialization of biology will require the convergence of several societal factors, including a properly trained workforce; appropriate legal frameworks; and physical infrastructure and standard operating procedures for safely containing, working with, and disposing of organisms used in bioprocessing. Public acceptance and endorsement of bioprocessed chemicals will be an important consideration in their commercial viability. In addition, international harmonization of policies would make the economic and governance environment more conducive to advances in industrial biology. The challenge for policy makers is to find the right mix of governance tools to promote innovation while also respecting a diversity of values and supporting effective oversight.
The workforce required for industrial biology to reach its potential will need a multidisciplinary education, with expertise in the biological sciences, chemistry, engineering, and computing. Expertise in environmental science will also be crucial for some industrial applications. Robust development of industrial biology will require a workforce with the expertise to create and safely operate complex organisms.
Industrial biotechnology will need a governance framework that balances important social goals and manifests important values (Figure 1-3). Governance involves deployment of a variety of policy tools by which an industry’s behavior can be shaped, including education of industry actors, industry self-governance through standard setting, accreditation, government standard setting and regulation, public engagement and public scrutiny, tort liability, and other mechanisms for developing safety standards and controls. For governing the industrialization of biology, key goals pertain to safety (risk identification and mitigation) and sustainability. For industrial biotechnology to deliver widespread benefits it must have low environmental impacts, use biological feedstocks sustainably, and operate according to high safety standards with respect to humans, animals, and the environment.
Ultimately, the right to operate bioprocesses is conferred by society. Regulatory frameworks to promote safety are necessary but not sufficient. The public must have sufficient understanding and acceptance of the science and technology involved to ensure comfort with and acceptance of goods produced through new techniques.
The remainder of this report addresses the elements of the statement of task outlined above.
Chapter 2 examines the unique opportunity for the acceleration of the industrialization of biology presented by the convergence of biology,
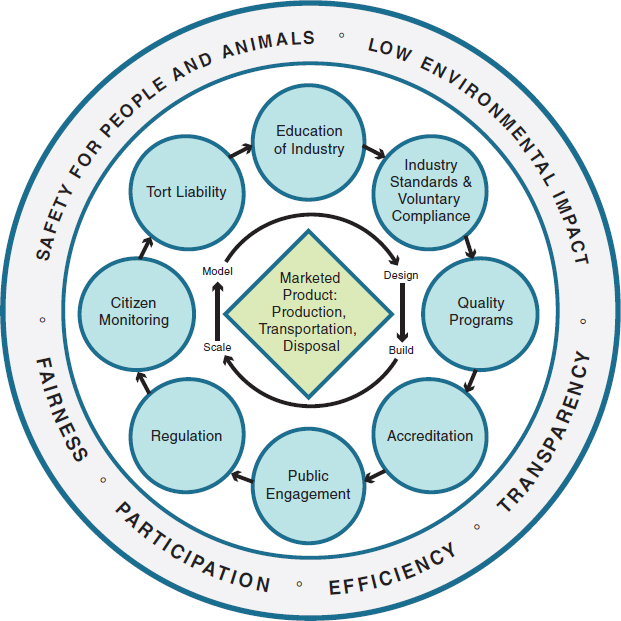
FIGURE 1-3 Tools of governance. This figure indicates the relationships between a specific production process (represented by the central quadrilateral figure), the various tools of governance (represented by the blue circles), and the concepts that a governance structure for chemical manufacturing should value (the outer circle).
chemistry, chemical engineering, and other critical fields; the development of new tools and methods; and the current economic success of chemicals produced through biological routes. The chapter discusses the core drivers of the industrialization of biology and identifies the societal challenges that the industrialization of biology is poised to address.
Chapter 3 develops a vision of a future in which the industrialization of biology is ubiquitous. It explores what materials might plausibly be produced through biological routes, what kind of economic infrastructure might be developed, and how these changes might affect society. This
chapter also identifies those societal questions that should be addressed as chemical manufacturing through biological routes matures and discusses potential governance mechanisms that might be used to address those questions.
Chapter 4 presents the committee’s technical roadmap for the industrialization of biology, including specific roadmap goals and timelines for feedstock utilization and development, chemical transformations, and deeper understanding of organisms. This chapter discusses critical aspects of each of these major technical areas and provides specific recommendations for the rapid achievement of the roadmap goals and the necessity of viewing the roadmap and the process that generated it as an evergreen process.
Chapter 5 distills the committee’s analysis and assessment of both technical and nontechnical issues into a set of specific recommendations to stakeholders involved in the industrialization of biology.












