2
Industrial Biotechnology: Past and Present
THE BIOECONOMY AND GLOBAL CHALLENGES
The industrialization of biology offers far-reaching benefits at both the global and the national scale
- by driving the innovation economy and sustainable economic growth;
- by potentially contributing to the solutions to some of the societal grand challenges of our time, such as helping to deliver clean, affordable, and sustainable energy;
- by enabling sustainable, next-generation manufacturing; and
- by creating new skills and jobs to benefit today’s and tomorrow’s generations.
Accelerating advanced chemical manufacturing by industrializing biology can drive the rapid growth of an innovative U.S. bioeconomy. A substantial share of economic output will be increasingly related to the development and use of biological materials and bio-based processes for both chemical production and the development of new materials. The industrialization of biology creates social, environmental, and financial advantages that combine economic growth with public benefits and better lives for our citizens.
BIO-BASED MARKETS ALREADY ARE SIGNIFICANT AND THRIVING
As demonstrated throughout this report, the future economic and societal benefits from the industrialization of biology are compelling. Bio-based markets already are significant in the United States, representing more than 2.2 percent of the gross domestic product (GDP) in 2012, or more than $353 billion in economic activity in 2012.2a The European Commission estimates that the European bioeconomy (excluding health applications) already is worth more than €2 trillion annually and employs more than 21.5 million people.19
Carlson has constructed a genetically modified domestic product (GMDP) metric to compare bio-based markets and biotechnology with the economy as a whole. His current data reveal that “the U.S. economy, and in particular annual U.S. GDP growth, is becoming increasingly dependent on biotechnology.”2a The Carlson GMDP-to-GDP comparison shows that bio-based markets have grown rapidly as a percentage of American GDP and that, by 2012, they constituted 5.4 percent of annual GDP growth.2a
Bio-based chemicals also are not entirely new, nor are they a historic artifact. Current global bio-based chemical and polymer production already is estimated to be about 50 million tons each year.20 Bioprocessing techniques such as fermentation, baking, and tanning have been used throughout much of human history. In recent history, we have witnessed major advances made possible by techniques such as genetic engineering and the development of the biotechnology industry.
According to several Organisation for Economic Co-operation and Development (OECD) analyses, “[i]ndustrial biotechnology has rapidly matured, and has produced some tangible products, including a large number of bio-based chemicals and bioplastics.”21 The OECD predicted in 2009 that bio-based products would constitute at least 2.7 percent of GDP among the OECD member countries by 2030.22 The rapid advances in scientific research and technological developments in only the past 5 years have led the OECD to revise that projection significantly. In Denmark, it is estimated that about 40 percent of manufacturing already takes places in a “cell factory.”23
Other recent studies confirm the rapid growth in the thriving markets for bio-based products. Data from Agilent Technologies show that U.S. business-to-business revenues from industrial biotechnology alone reached at least $125 billion in 2012.2b Bio-based chemical applications accounted for about $66 billion of that U.S. economic activity, while biofuels added another $30 billion. Lux Research estimates that industrial chemicals made through synthetic biology currently represent a $1.5 billion market and that this likely will expand at 15 to 25 percent annual growth rates for the foreseeable future.3 A recent U.S. Department of
Agriculture (USDA) report indicates that, by 2015, bio-based chemicals will make up greater than 10 percent of the chemical market.
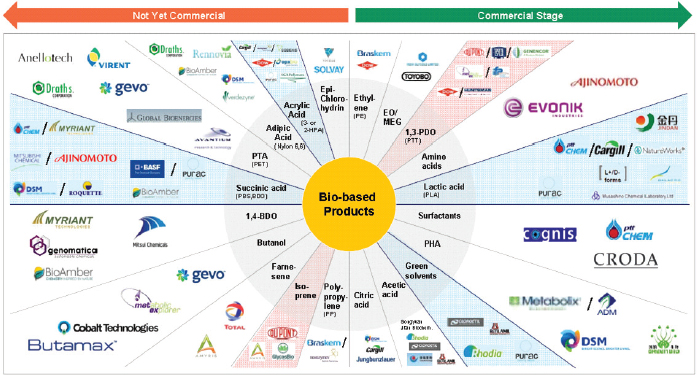
The markets for bio-based chemicals and industrial biotechnology for chemical manufacturing processes are growing roughly twice as fast as those in biomedicine or agriculture (Figure 2-1). They also reflect new structural market shifts as decentralized production processes, innovative new value chains, and collaborative ventures both compete with and complement the vertically integrated chemical manufacturing facilities that have marked the past century.
New chemicals and biochemical materials are being developed through the rapid emergence of novel bio-based technologies and processes. Biology is being used to develop innovative and resource-conserving solutions to difficult problems.
Rapid growth already is occurring in three interrelated segments of bio-based advanced chemical manufacturing: enabling technologies (tools and platforms needed for the development of advanced chemicals); core technologies (processes and inputs used to make chemical products); and enabled products (chemical products on the market). According to BCC Research, all three segments in synthetic biology are growing at more than 70 percent per year, with significant markets for enabling technologies ($653 million), core technologies ($699 million), and enabled chemical products ($2.8 billion) by 2016.24
THE INNOVATIVE POWER OF INDUSTRIAL BIOTECHNOLOGY
The biggest bioeconomy market opportunities from accelerating advanced manufacturing of chemicals through the industrialization of biology have yet to be realized. As the Obama Administration posited in its 2012 National Bioeconomy Blueprint, the rapid development of new bio-based chemicals and materials in the U.S. bioeconomy can “allow Americans to live longer, healthier lives, reduce our dependence on oil, address key environmental challenges, transform manufacturing processes, and increase the productivity and scope of the agricultural sector while growing new jobs and industries.”1
A 2013 Milken Institute report underscored the huge potential opportunities when it noted that “[n]inety-six percent of all U.S. manufactured goods use some sort of chemical product, and businesses depend[e]nt on the chemical industry account for nearly $3.6 trillion in U.S. GDP.”25 The global market for enzymes used in consumer products and industrial production processes—and a prime target for the industrialization of biology—alone is expected to reach $8 billion by 2015.
The OECD has projected that industrial biotechnology and bio-based chemical manufacturing likely will accelerate and lead the development
of a robust, global bioeconomy. The potential economic and societal benefits predicted by the OECD and others become clear when we realize that while there are more than $4 trillion of products made by chemical transformations globally, only about 5 percent of these potentially “addressable markets” have been addressed biologically. A study from BCC Research suggests that synthetic biology markets for chemicals will grow to $11 billion by 2016,24 and a broader review from McKinsey Global Institute estimates that synthetic biology and the industrialization of biology will provide a disruptive set of technologies with an economic impact of at least $100 billion by 2025.26
As a result, the broad applications of advanced chemical manufacturing for multiple uses in energy, health, advanced consumer products, agriculture and food, cosmetics, and environmental technologies are expected to produce trillions of dollars in addressable global market opportunities. Several recent studies estimate that at least 20 percent of today’s petrochemical production can be replaced by the industrialization of biology in chemical manufacturing over the next decade.25
Aside from the large size of the chemical markets that can be addressed biologically, making biology easier to engineer and developing new chemical manufacturing capabilities based on synthetic biology will have a broad range of other economic benefits. They include opening up the potential for innovative products and processes, decentralizing production process and value chains, creating incentives for new entrants (including high-growth small- and medium-sized enterprises), creating new jobs and skills, and incentivizing new business models.
In addition to substitution or displacement effects, the industrialization of biology will lead to the production of new molecules for chemical, fuel, and material applications, which are not currently possible from fossil fuel sources or traditional manufacturing. The potential for new innovation and market creation remains considerable. Major new advances in measurement tools, computer-aided design, and design-build-test-learn cycles—from the most fundamental level to engineer living material to that needed for making complex chemicals at the commercially competitive industrial scale—not only provide new tools but also create new bioeconomy markets and investment opportunities in themselves (Box 2-1).
As a result, a robust and disruptive new industrial ecosystem is emerging. Even at this early stage, the number of American synthetic biology commercial companies has grown from 54 to 131 between 2009 and 2013.27 A large number of new startups have been created, and a number of them have successfully gone public with initial public offerings. But these figures understate the full economic impact of synthetic biology because of the rapid uptake and interest by larger firms in a broad range of sectors.
BOX 2-1
Living Foundries
For example, the Defense Advanced Research Projects Agency’s Living Foundries program is working with many companies, national laboratories, and universities to develop new tools to enable rapid engineering of biology. It is tackling “impossible today” industrial projects that could become “possible” if we enable, scale, and rapidly prototype genetic designs and operating systems never before accessible for industrial production. And its most recent large-scale initiative, the 1,000 Molecules Project, seeks nothing short of a fundamental disruption of traditional chemicals and materials industries and processes by developing 1,000 new chemical building blocks for entirely new materials at the molecular scale and nanoscale in the next 3-5 years.
Established chemical companies are making significant investments internally, in partnership with startups, or both. New business models are proliferating, as are innovative collaborations, driven by advances in synthetic biology. University-industry linkages span the continuum from high-risk basic research to late-stage prototype development projects that can scale and compete at market-driven price and performance points.
Six core drivers—the “6 C’s”—are intersecting to drive the industrialization of biology and to accelerate the development of advanced manufacturing of chemicals: (1) convergence of biology and engineering; (2) challenges for society in addressing global grand challenges related to energy, climate change, the environment, agriculture and food, and health; (3) competitiveness; (4) human capital and capacity; (5) the confluence of new enabling tools, platforms, data, and processes competitiveness; and (6) the current state and readiness of both the science and industry.
The industrialization of biology in chemical manufacturing is enabled by the convergence of biology with chemistry and engineering in transformative new ways. A major part of the U.S. strategy for advanced manufacturing of chemicals is the expectation that the next industrial revolution will involve making things with greater precision, at ever
higher speeds, and at lower costs, and more sustainably by focusing on the biological processes.
Convergence includes not only transdisciplinary research and development and the integration of science but also the intersection of previously distinct industrial sectors such as chemical synthesis, industrial biotechnology and bioenergy, information technologies, and enabling tools and platforms from a number of business sectors. The growing convergence of transdisciplinary science, technology, engineering, and mathematics, along with overlapping markets and innovative business models, enables novel solutions to many previously intractable societal challenges.
Four influential reports in recent years highlight this trend and underscore the importance of convergence as a core driver. A New Biology for the Twenty-first Century was issued in 2009 and received extensive attention.13 It concluded that biology would be the key, new driver for innovation in the 21st century, much as the physical sciences had led to the information and communications technology revolution and other major breakthroughs in the 20th century. It also showed that biology increasingly would intersect with previously disparate disciplines, including chemistry. This new integration would provide the basis for not only new economic growth but also the tools and platforms for addressing many of the major global grand challenges of this century.
In 2011, a Massachusetts Institute of Technology (MIT) faculty foresight review concluded that we were entering a Third Revolution in the life sciences.28 The first had been the DNA, genetics, and molecular biology revolution that provided the basis for today’s modern biotechnology industries and approaches. The second was the genomics revolution made possible by the Human Genome Project. The Third Revolution is based on convergence and will transform next-generation manufacturing and production by merging biology and engineering in completely new ways.
The third major report was the ARISE II report from the American Academy of Arts and Sciences (AAAS), titled Advancing Research in Science and Engineering: Unleashing America’s Research & Innovation Enterprise. It noted that many of the historical distinctions between the modes of thought and organizational principles in the life sciences and the physical sciences were converging around common challenges and opportunities, but that outdated structures and approaches impeded collaborations, communication, and the translation of research into new products and services While the physical sciences have linked basic and applied research as “an interwoven continuum,” the life sciences have tended to make sharp distinctions between basic and applied sciences in its disciplines.29
The ARISE II report made two principal recommendations. First, America’s research and innovation enterprise must embrace a new
transdisciplinary organizing principle. The AAAS called for providing incentives to ensure that “tools and expertise developed within discrete disciplines are shared and combined to enable a deep conceptual and functional integration across the disciplines.”29 It also recommended creating an interdependent ecosystem for linking academic, government, and private sectors throughout the discovery and development process.
In May 2014, a follow-up National Research Council report, Convergence: Facilitating Transdisciplinary Integration of the Life Sciences, Physical Sciences, Engineering, and Beyond, sought to capture two dimensions: the convergence of the subsets of expertise necessary to address a set of research problems and the formation of the web of partnerships involved in supporting such scientific investigations. These two dimensions further enable the resulting advances to be translated into new forms of innovation and new bio-based products and services.
In this frame, convergence represents a major cultural and organizational shift for academic organizations and government science or technology ministries that have been traditionally organized around discipline-based departments. The overall ecosystem needed for convergence draws on not only academic contributors but also a much broader cross-fertilization of ideas from national laboratories, industry, citizen scientists, and funding bodies, as well as from new insights provided by economics and the social sciences. The process of convergence is applicable to basic science discovery as well as translational applications in industry. Because it is commonly focused on achieving an outcome to a challenge at the frontiers of knowledge and new markets, many convergence efforts include a major entrepreneurship component that leads to the development of new webs and ecosystems of startup companies and economic innovation.
An emerging metaphor from convergence is that of the “cell as tomorrow’s factory.” As Neri Oxman from the MIT Media Lab observed, “The biological world is displacing the machine as a general model of design.”30 In short, the industrialization of biology and synthetic biology will be as important for the next 50 years as semiconductors and related information and communication devices have been to economic growth over the past 50 years.
SOCIETAL BENEFITS IN ADDRESSING GLOBAL GRAND CHALLENGES
When compared to traditional manufacturing, advanced manufacturing of chemicals through biology might produce social benefits while requiring fewer trade-offs between growth and sustainability. In addition to the economic benefits generated through innovation, productivity
increases, and new sources of sustainable economic growth, the advanced manufacturing of chemicals through biology can address 21st-century grand challenges related to energy, climate change, sustainable and more productive agriculture, environmental sustainability, and inclusive growth.
The industrialization of biology can serve to enhance the United States’ energy independence. Advanced chemical manufacturing based on biological sources such as plants, algae, bacteria, yeast, filamentous fungi, and other organisms can replace many chemicals now derived from petroleum or other fossil fuels. If properly designed, bio-based production processes, including new bio-based inputs, can improve energy efficiency and, in some cases, reduce energy costs.
Over time, a growing part of the demand for chemical products and processes likely will come from the increasing economic activity of emerging and developing countries. Given the burgeoning demand for oil and other scarce natural resources in many emerging markets, sustainable sources of new and advanced bio-based chemicals may be the only viable way to meet the needs of their populations.
A driver for the transition to the bioeconomy and novel advanced chemical manufacturing is the anticipation by some energy experts, such as the International Energy Agency, that oil, gas, and coal “will reach peak production in the not too distant future and that prices will climb.” The OECD recently demonstrated that the scope and platforms for the bio-based production of chemicals and fuels increased significantly in 2013. Its analysis concludes that these developments “may open the door to greater replacement of the oil barrel.”31
Climate Change and Environmental Sustainability
The advanced manufacturing of bio-based chemicals could provide numerous environmental benefits. Many producers of consumer products are now committed to “green growth,” which will require new enzymes and other chemical inputs, together with more sustainable production processes. Bio-based production, properly designed and managed, has the potential for generating fewer toxic by-products and less waste than traditional chemical manufacturing.
New approaches to the advanced manufacturing of chemicals are well aligned with American efforts to mitigate the adverse effects of greenhouse gases and to enable the United States to meet its global climate change commitments. By using biomass as a feedstock and through advanced manufacturing techniques that industrialize biology, the manu-
facturing of advanced chemicals can achieve significant savings in greenhouse gas emissions compared to production from oil or other fossil fuels.
The development of advanced manufacturing for bio-based chemicals also means increasing the number of products that are carbon neutral in terms of not producing any net increase in carbon dioxide or other greenhouse gases over their entire life cycle: from design and production through disposal. At the same time, significant waste reduction may be achieved through bio-based production processes and the resulting products’ life cycle, including manufacturing the advanced chemicals used to produce them. The milder bioprocess conditions—such as generally lower temperature and pressure—used for bio-based manufacturing compared to fossil fuels also contribute to sustainability. Additional environmental benefits will be related to using synthetic biology and related techniques for bio-remediation that can bring contaminated soil back into productive use. The OECD notes that the world’s soil is being lost 18-30 times faster than it is formed, and that new methods such as synthetic biology are important for limiting soil destruction and for growing crops more efficiently.
A roadmap focused on manufacturing at commercially competitive scale will create new opportunities for American agriculture and provide new value chains that do not require costly trade-offs with land. As a Milken Institute study concluded, “[b]io-based chemicals offer the prospect of new cash crops like switchgrass, new demand for the cellulosic fiber in traditional crops, and new jobs in bio-chemical production and process.”25
The increased use of biomass as a feedstock for the production both of high-value, low-volume, bio-based chemicals and bioplastics and of low-value, high-volume, bulk biofuels and commodity chemicals provides new opportunities for innovation in sustainable agriculture. Integrated production facilities that offer the ability to produce not only biofuels but also bio-based chemicals and bioplastics are becoming increasingly technologically feasible and economically viable. Advanced feedstocks will also permit farmers to produce larger yields on smaller amounts of land to feed a growing population.
Competitiveness and Innovation
The promise and importance of the industrialization of biology has not gone unnoticed around the world. China is investing huge amounts in synthetic biology, and it has made this set of technologies a priority in its current 15-year Science and Technology Plan.32 The United Kingdom
developed a list of “The Eight Great Technologies” for the future of Britain, and synthetic biology is considered number two of the top technologies for the United Kingdom’s future. A number of countries now are developing national strategies or plans related to synthetic biology, the industrialization of biology, and the future bioeconomy, including many emerging markets such as South Africa, Brazil, and Mexico.
THE TIME IS RIGHT: CURRENT STATE AND ADVANCES IN SCIENCE AND INDUSTRY
Opportunities Arising from DNA Technologies, Systems Biology, Metagenomics, and Synthetic Biology
Biology has the potential to build intricate material and chemical structures with atomic precision. Biotechnology has only begun to harness this capability, and leading-edge products in development have simple structures, such as butanediol, isobutanol, farnesene, and lactic acid. Biology excels at producing more complex molecules and mixtures of molecules. Access to this chemical complexity via biotechnology has been limited by the investment needed to engineer multistep biological transformations.
The past decade has seen an explosion in the technologies to read, write, compose, and debug DNA. This is rapidly increasing the scale and sophistication of genetic engineering projects. In the near term, this will lead to more complex chemical structures and composite nanomaterials, which require precise control over dozens of genes. Examples of this include mining drugs from the human microbiome, obtaining pesticides from environmental samples, and producing metal nanoparticles for electronics and medical devices. In the longer term, one can imagine organisms designed from the ground up for consolidated bioprocessing and automatic assembly of a product that requires multiple steps.
The genetics underlying the natural world are rapidly being illuminated by DNA sequencing, the cost of which is declining faster than Moore’s law. The first human genome (3.2 billion base pairs [bp]) was sequenced in 2001 at a cost of $2.7 billion. Nine years later 1,000 human genomes (3.2 trillion bp) were sequenced, and in 2014, Illumina released the HiSeq X, promising a $1,000 genome. Beyond human genetics, this technology has been applied to sequence communities of organisms (metagenomics) populating niches in the environment or associated with hosts, such as the human gut. Databases of sequences have been rapidly grown with information; as of 2013, there were 160 million sequences from 240,000 organisms. This has built an enormous potential catalogue of natural parts, from which high-value chemical pathways can be discovered or created.
Accessing these chemicals requires more than the sequence information. Historically, collaboration required the physical transfer of DNA materials, such as genes, between labs. The rise of DNA synthesis has moved biology toward an information science where the DNA can be reconstructed from the sequence information alone, thus eliminating the need for physical transfer and enabling the direct access to biological functions encoded in the sequence databases.33 DNA synthesis has been applied to build entire 1 MB bacterial genomes and yeast chromosomes.34 Synthesis provides the genetic designer with full operational control over the identity of each bp of a large design, as opposed to previously, where DNA was stitched together from existing pieces. There is still significant room for improvement; while a large DNA sequencing center can sequence >4 trillion bp/day, the top industrial synthesis companies only produce ~300,000 bp/day.
The ability to compose DNA has lagged behind our ability to read and write it. The most valuable functions require many genes and complex regulatory control over how much, when, and where they are turned on. Synthetic biology offers some tools to tackle this challenge, including genetic circuits, precision regulatory parts, and computer-aided design to systematically recode multigene systems.35 While it is possible to synthesize entire genomes, we are far from being able to write them from scratch from the bottom up. The current state of the art is the top-down “editing” of existing genomes using technologies such as MAGE9 and CRISPR/Cas910 to introduce incremental changes in an otherwise natural genome.9, 36 Similarly, genome-scale design tools have emerged to control flux through metabolic pathways (e.g., COBRA and Optknock), but the output of these are predictions of the impact of the top-down knockout of enzymes in a defined host.37
Genome-scale engineering, where designs are composed of thousands of genes assembled from the bottom up, will become the norm. This will require computational tools that merge the simulation tools from systems biology with biophysical methods that can convert a desired feature into a specific DNA sequence. New design paradigms are also needed in order to manage a large project and integrate across different cellular systems.38 The ability to build synthetic regulation (sensors and circuits) needs to be combined with metabolic engineering and the ability to control cellular functions (e.g., protein secretion) and stress responses.39 As synthetic systems become larger, it will be more important to be quantitative in understanding the distribution of resources and their load on cellular growth and maintenance. Increasingly, it will be important to develop methods to insulate a synthetic system from the background processes of the host.40
Genome-scale design will require genome-scale debugging. It is currently impossible to get a snapshot of how a change in the genetic design
impacts all of the processes in a cell. Advances in -omics technologies make it possible to characterize the mRNA, proteins, and small molecules in a cell. However, each of these requires specialized expertise and instrumentation and is cost prohibitive to perform on failed designs. The form of the data is nonstandard, and integrating information across transcriptomics, proteomics, and metabolomics is difficult. Furthermore, it is difficult to convert the results into actionable design changes to optimize a system.
Integrated national-level infrastructure can help accelerate the transition to genome-scale designs. Strain databanks and sequencing centers provide surveillance of the natural world and could populate parts libraries with billions of natural enzymes and pathways. BioFABs provide the substrate for large designs by providing high-quality genetic parts through large-scale engineering and characterization.41 Foundries work to systematically produce products by pushing the scale of genetic designs and integrating DNA manufacturing with cellular analytics.38 Finally, metrology institutes (e.g., run by the National Institute of Standards and Technology) provide the standards for characterizing genetic parts, reporting construction precision, and software integrating -omics data.42
New High-Value Chemical Products Unobtainable by Traditional Chemical Synthesis
Organic synthesis is a mature discipline where nearly any target molecule can be made through a logical combination of reaction steps. A similar capability has not been realized in biotechnology, the products of which have been somewhat limited to chemicals naturally made by cells. Only small numbers of enzymes have been characterized relative to the potential of chemical space, and there is a lack of standards in reporting enzyme activity and specificity. However, we are at an inflection point in enzymology, where sequence databases have been populated with tens of millions of enzymes and access to this resource en masse is enabled by DNA synthesis. This will lead to a revolution in pathway design, where obtaining a non-natural target molecule by combining enzymes will be intellectually analogous to the logic of organic chemistry.
In the early 2000s, access to even a single enzyme required the physical transfer of DNA materials between labs. The decline in DNA synthesis has made it routine to build all of the genes from a sequence database predicted to have a desired function. Such “part mining” typically involves constructing the genes for hundreds of enzymes and has been very successful at identifying variants with desired properties. This often results in the combination of genes from diverse organisms in building a pathway.
This mining will soon grow from individual enzymes to entire pathways.33a Potentially high-value products are encoded by 10- to 150-kb
gene clusters that encode multienzyme cellular factories. Many natural products from these pathways are produced industrially by the native organisms; for example, rapamycin is a pharmaceutical that may have an effect in treating cancer and spinosyn is a biological pesticide. While synthetic routes to complex natural products can be found, there are few labs capable of this and the routes are often low yield and not viable industrially. It is clear that only the surface has been scratched in identifying potential products. Just recently, bioinformatics algorithms have been developed that enable the enumeration of clusters in genome databases. To date, 40,000 have been found in the National Center for Biotechnology Information, and metagenomic samples, including the human microbiome, yield tens of thousands of new clusters.43 The Agency for Science, Technology and Research of Singapore’s collection consisting of 120,000 strains is estimated to contain several million novel pathways. Fully accessing their chemical products will require a further reduction in synthesis costs and the application of synthetic biology to control multigene systems and functionally transfer activity to new organisms.
Collectively, the mining efforts are yielding a deluge of new enzyme data across entire families that encompass activity and specificity information. In addition to the mapping of natural diversity, advances in engineering enzymes have yielded transformations that are chemically difficult, for example, the use of cytochrome P450s for C-C and C-N chemistry. The expansion of the enzyme toolbox will feed new computational methods whose input is a desired chemical structure and whose output is a combination of enzymes predicted to build the molecule. Examples of this include the Biochemical Network Integrated Computational Explorer (BNICE)44 and the ACT ontology, which has been used to build a synthetic pathway to N-acetyl-p-aminophenol (Tylenol).45
Implementing Computation in Cells
Living cells have an incredible capacity to sense and interpret their surroundings. They achieve this using gene regulation that functions as sensors to respond to environmental stimuli and circuits that process this information and commit cells to a response.39, 46 To date, this has not been utilized as part of chemical production in biotechnology. However, the potential impact is enormous. Even simple operations would be valuable, such as turning on different pathways at various times during fermentation and implementing feedback regulation to avoid the accumulation of toxic pathway intermediates.47 Sophisticated circuitry would enable new options in cellular design. For example, entire process control algorithms could be implemented that optimize the uptake of feedstock and control of flux through metabolic pathways. Consolidated bioprocessing could
be implemented, where the degradation of biomass and construction of a complex chemical is performed via a preprogrammed order of events. Finally, the composite materials that are possible via biology require precise timing and spatial location of gene expression.48
The use of biological organisms to transform precursor molecules into targeted molecular end points dates back to the earliest days of recorded human history in the form of fermentation to produce beer, cheese, and bread. Jokichi Takamine’s work on the “koji” process—resulting in an 1894 patent on a microbial enzyme in the United States—marked a turn toward the industrialization of biological processes.49 The modern era of industrial biotechnology began more recently, with its birth typically tied to large-scale fermentation of penicillin. The antibiotic, first isolated by Sir Alexander Fleming in 1928, was not advanced to large-scale production until World War II, driven by the need for an alternative to sulfa drugs to treat bacterial infections.50 Although initial titers in 1939 were estimated to be on the order of 0.001 g/L, by the end of the war U.S. capacity for penicillin production was sufficient for ~100,000 patients per year. Penicillin was not the only major fermentation process that attained industrial prominence during this time. The “ABE” process, in which Clostridium acetobutylicum ferments sugars to a mixture of acetone, butanol, and ethanol, was first developed in the early 20th century and was a primary source of acetone during World War I. During World War II, the ABE process was the primary source of butanol, and fermentation remained popular as a source for this commodity until lower-cost petroleum-derived products emerged in the 1960s.
It is notable that these achievements preceded the elucidation of the structure of DNA by nearly a decade. The status of penicillin, acetone, and butanol as natural products derived from (reasonably) productive host organisms enabled the use of methods such as classical mutagenesis that required neither complete understanding of the underlying biochemical pathways nor knowledge of the genes encoding the constituent enzymes in order to enhance productivity. In the three decades that followed, fermentation processes were developed for large-scale commercial production of several additional products including citric acid, vitamin B12, glutamic acid, and lysine. Even in the absence of detailed genetics, understanding of pathway regulation facilitated the use of classical mutagenesis to select improved strains. For example, the observation that lysine inhibits upstream enzymes in the pathway for its synthesis led to the use of the lysine analog S-2-aminoethyl cysteine as a selective inhibitor to identify feedback-resistant mutants. Such methods could be routinely used to
improve natural product fermentations, but the utility of biology for chemical manufacturing remained limited to a small subset of molecules.
In 1973, Herbert Boyer of the University of California, San Francisco, and Stanley Cohen of Stanford University and colleagues published a manuscript titled Construction of Biologically Functional Bacterial Plasmids In Vitro.51 This publication described what is widely considered to be the first genetic engineering experiment and signaled the start of the Biotechnology Age. However, despite the decades-old history of the industrialization of biology for small-molecule production, “biotechnology” became nearly synonymous with “biopharmaceuticals” in the aftermath of this breakthrough. Less than 10 years later, recombinant human insulin was approved by the Food and Drug Administration and numerous protein drugs followed.
Why did biopharmaceuticals eclipse biochemicals as the first and most obvious beneficiaries of recombinant DNA technology? This is perhaps best answered in consideration of the complexity of the final products and the reactions that encode them.
Therapeutic proteins are structurally more complex than small molecules, yet their synthesis is directly related to the heterologous DNA chosen for expression. Simple overexpression in the right host of as little as a single gene produces the product of interest.
Conversely, biochemicals are the result of a series of enzyme-catalyzed reactions, with each enzyme encoded by at least one gene. This complexity of pathways over products creates a systems-level challenge that requires more systems-oriented solutions. This challenge and the discipline that emerged to address it was first codified in Toward a Science of Metabolic Engineering by James E. Bailey.52 Metabolic engineering sought to take advantage of the tools of recombinant DNA technology while applying systems and network analyses to the challenge of engineering more productive strains.53 These principles were successfully applied to generate highly efficient and productive fermentation processes for a number of products, including, for example, 1,3-propanediol and lysine from an engineered strain of E. coli with extraordinary maximum production rates of 8 g/L/hr.
In addition, unlike large-volume chemicals produced to replace or supplement an already existing commodity (e.g., ethanol), novel biopharmaceuticals are highly specialized products for which no other production route is possible or economically realistic. As Figure 2-2 indicates, biopharmaceuticals and other specialized products (such as industrial enzymes) operate at a much higher gross margin than products in large-volume commodity markets. The biology and chemistry of potential products helps to explain why biopharmaceuticals were some of the first
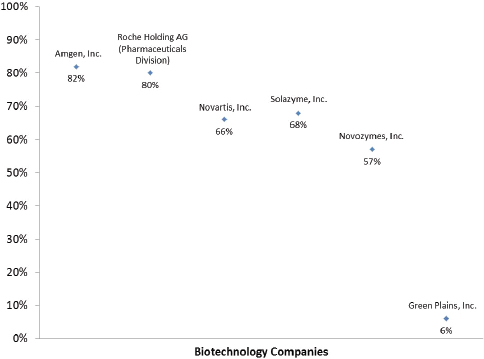
FIGURE 2-2 This figure displays the gross margin of several representative companies producing biopharmaceuticals, industrial enzymes, high-value oils, and ethanol
SOURCES: Amgen, Inc. (2014, February 24) 10-K. Retrieved from http://www.sec.gov; Solazyme, Inc. (2014, March 14) 10-K. Retrieved from http://www.sec.gov; Novartis, Inc. (2014) Novartis Annual Report 2014. Retrieved from http://www.novartis.com; Roche Holding AG (2014) Roche Financial Review 2014. Retrieved from http://www.roche.com [note: gross margin is exclusive to Roche pharmaceuticals division]; Novozymes, Inc. (2014) 2014 Annual Report. Retrieved from http://www.novozymes.com; Green Plains, Inc. (2014, February 10) 10-K. Retrieved from http://www.sec.gov
products of the industrialization of biology, but the economic factors are also critical.
While bio-based chemical production from whole-cell organisms was being advanced, the tools of biotechnology were also being applied in other arenas. Of note is the use of genetic engineering in agriculture. Engineered crops have led to such favorable properties as increased yields, decreased pesticide use, and reduced greenhouse gas emissions in field operations. Additionally, the ability to clone and heterologously express recombinant proteins has greatly facilitated the use of biology to perform single-step reactions in vitro. Purified enzymes have been used to produce
a number of products, especially for pharmaceutical synthesis, but the power to extend this lies in the advanced tools of biotechnology. Protein engineering, especially through directed evolution, has generated enzyme variants with activities on previously naïve substrates and enabled the synthesis of numerous small molecules, particularly chiral ones, of pharmaceutical significance.54 The use of biocatalysis for chemical synthesis can not only access structures with higher atom economy but also significantly reduce the environmental footprint of the associated process. In the case of sitagliptin, a molecule produced through collaboration between Merck and Codexis, the optimized use of a biocatalytic step to replace an analogous chemical one in the process resulted in reduction in the ratio of total mass of materials used to mass of isolated product from 37 to 6.55
The key drivers for a company using bio-based methods for chemical production can vary greatly based on the products being manufactured. Under ideal circumstances, an analysis of the margin on a series of products would allow us to understand the economic drivers that are most relevant to their production processes. Margin, however, is not typically released by most corporations. In the absence of specific margin, gross margin can be calculated based on annual statements of public companies. An analysis of gross margin for 6 companies that have products limited to an individual sector can give some general insight into the considerations of margin that they will face. As shown in Figure 2-2, Amgen, Roche (Pharmaceuticals), and Novartis have relatively high gross margins ranging from 66 percent to 82 percent. This is not unexpected for a pharmaceutical manufacturer, because they typically develop low-volume, high-cost chemicals. By contrast, Green Plains, Inc., a biofuels manufacturer, is selling a true commodity chemical and is able to make only a small margin (6 percent). Each of these entities will have different economic factors to consider, for example, feedstock costs will be the dominant driver for a biofuels manufacturer with a small margin. Feedstocks are likely to be less of a concern for the others shown in Figure2-2.
Today, the use of biology in industry is both strong and increasing. Applications range from traditional areas such as food to newer markets such as renewable energy. In all cases, the potential for increased use of biology will be enhanced by advances in the scale at which biological systems can be engineered.
Malaria is a global health problem that threatens 300 to 500 million people and kills more than 1 million people annually.56 Disease control is
hampered by the occurrence of multi-drug-resistant strains of the malaria parasite Plasmodium falciparum.57 Artemisinin, a sesquiterpene lactone endoperoxide extracted from Artemisia annua L (family Asteraceae; commonly known as sweet wormwood), is highly effective against multidrug-resistant Plasmodium spp. but is in short supply and unaffordable to most malaria sufferers.58 Although total synthesis of artemisinin is difficult and costly,59 the semisynthesis of artemisinin from E. coli–sourced artemisinic acid, its immediate precursor, could be a cost-effective, environmentally friendly, high-quality, and reliable source of artemisinin.60
The production of semisynthetic artemisinin is one of the first success stories for the combined use of metabolic engineering and synthetic biology in the production of a pharmaceutical at industrial scale (see Figure 2-3). As semisynthetic artemisinin is functionally equivalent to the plant-derived drug,61 it has now been approved by the WHO (World Heath Organization) for the preparation of approved artemisinin deriva-
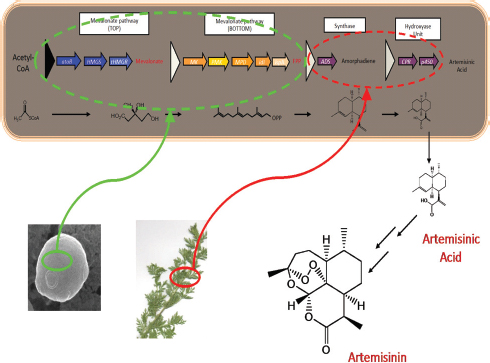
FIGURE 2-3 Overall scheme for engineering artemisinic acid production into Escherichia coli. Genes from Saccharomyces cerevisiae and Artemisia annua were expressed in E. coli to transform acetyl-CoA, energy, and reducing equivalents into artemisinic acid, which was then chemically transformed into artemisinin. SOURCE: Keasling, J. (May, 2012) Synthetic biology and the development of tools for metabolic engineering. Metabolic Engineering 14(3), 189-195.
tives (such as artesunate) for incorporation into artemisinin-combination therapies.62 Despite concerns over the amount of semisynthetic artemisinin made available to people in countries affected by malaria,63 semisynthetic artemisinin is a major success of industrial biotechnology.
Biofuels: Moving to Commercial
Realization of the sustainable benefit of the innovations described above requires commercialization on a massive scale. Advanced biofuels must be economically competitive with existing products, overcoming the primary economic drivers of feedstock price and overall process productivity and yield. While many biofuel candidates have desirable performance characteristics, the potential yield for each product is limited by the theoretical yield of the production pathway,64 and this limit sets a bottom price that a product can achieve for a given feedstock. Commercialization requires advancing lab-scale processes to yields and productivities that approach the maximum theoretical output of that process (generally >85 percent) and scaling them to reactors that will be >600 cubic meters in volume (~106-fold scale-up from most lab-scale fermentations). The engineering of catalysts to reach the yields and productivities to meet economic targets and the scaling of these processes without losing performance are the greatest challenges to commercialization and represent an expensive purgatory for many of the advanced biofuels in the pipeline. To date, only a few promising technologies for advanced biofuels have reached the later phases of commercialization, and these are for the higher alcohols, butanol and isobutanol. Both are anaerobic processes that can leverage existing ethanol facilities, and whose products are either natural or engineered pathways linked to microbial fermentative growth,65 While tremendous investments have gone into isoprenoid and fatty acid–derived products, commercial success is yet to be demonstrated. Interestingly, as these technologies become more defined they appear to differentiate in regard to industrial host, process and product trade-offs, and feedstock choice. Indeed, as processes move to commercialization, choice of host becomes a critical decision.
Yeast is an attractive host organism because of its robustness, extensive fermentation knowledge, availability of genetic tools, tolerance to industrial conditions and solvents (butanol tolerance >20 g/L), low media pH, and lack of susceptibility to bacteriophage.20 Yeasts’ main limitations are an inability to digest C5 sugars, such as xylose and arabinose, which are present in lignocellulosic biomass; a natural ability to produce ethanol, which may hinder metabolic engineering efforts to produce advanced biofuels; limited synthetic biology tools for pathway optimization; and
reduced protein expression levels when compared to E. coli, which may limit the flux through biofuel-producing pathways.
Yeast is currently the preferred organism for the production of butanol and isoprenoid-based biofuels.66 According to publicly available information, at least three large enterprises are pursuing higher alcohol production in yeast using slightly different strategies. Gevo’s strategy has been to link the production of isobutanol to anaerobic growth and to select for strains that approach theoretical yields.65a This in combination with a stripping of the isobutanol from the broth through continuous flash evaporation has led to a process with over 90 percent theoretical yield and for which a commercial-scale plant is now in place. One strategy being pursued by Butamax is to construct many different metabolic pathways leading to butanoyl.67 Finally, Butalco, which has strains that metabolize C5 sugars, proposes to use only endogenous genes to improve isobutanol production.68
Technologies exploiting fatty acid metabolism are pursuing a variety of host organisms. E. coli is being exploited for the production of fatty acid–derived compounds, such as fatty acid methyl esters, fatty alcohols, alkanes, and olefins, directly from carbohydrate in a single-step fermentation. E. coli is believed to be preferred for this application because of its exceptionally high rate of fatty acid biosynthesis (3g/L/hr per gram of dry cell weight, based on 30-minute doubling time and 9.7 percent lipid content of cell mass), its natural ability to secrete these products, its natural ability to consume both C5 and C6 sugars, its extensive industrial precedent in the commercial production of metabolically engineered small molecules (1,3-propanediol, lysine, phenylalanine, etc.), and its ease of engineering. E. coli does have limitations, such as preference for neutral pH and susceptibility to bacteriophage. Cyanobacteria are also being pursued, but for the production of these compounds from CO2 in photobioreactors. Technologies for the production of triacyl glyceride (TAG), which is the precursor for biodiesel and renewable diesel (hydrodeoxygenated TAG),69 have gravitated to oleaginous algae, Generally Recognized As Safe (GRAS) organisms that naturally produce high levels of intracellular oil in both photobioreactors and heterotrophic fermentations.70 Until any of the promising technologies described above begin producing fuels at prices competitive with existing products, the jury shall remain out as to how effective they can be to meet the goal of widespread adoption.
Genomatica is a San Diego–based startup company that has been active for 15 years. Its focus has been the biological production of chemicals. Its initial focus has been polymer intermediates—specialty monomers
that are used as ingredients for plastics and fibers. Genomatica71 has faced all the challenges that will be faced by others entering this field. Bringing new-to-world technology to a mature industry is difficult. It is important to start with a thoroughly validated value proposition. Product targets must be chosen carefully. Process economics are always a challenge. Feedstock costs and feedstock flexibility are critically important. Conventional sugars are subject to price variability and represent a viable feedstock only in Brazil, India, the United States, and Thailand. Concerns about the use of sugar for fuel (and feedstocks) versus food are also an issue. At this stage biomass-based sugars are challenged to meet the cost and quality needed to produce polymer intermediates. C1 feedstocks look attractive, but today face major challenges for engineering production strains and for production process technology.
As a startup company, Genomatica has faced significant challenges that derive from the development timelines (5-8 years) needed to bring a product to market. The long development time is a contributing factor to the elevated costs to develop a new product platform, on the order of $100 million. Genomatica is focused on reducing both the timeline and costs associated with future product platform developments. Many elements in the product development are beyond the capabilities of any single company. Genomatica has relied on a network of key partnerships to deliver its initial products. Partners have assisted with feedstocks, scale-up, engineering, commercial-scale production, and market-related activities.
In 2008, Genomatica announced its novel bioprocess for the production of 1,4-butanediol (renewable BDO), an important intermediate used in the production of poly(butylene terephthalate) and polyethers. Petrochemical-based BDO is a high-volume chemical, produced in large scale and largely depreciated assets. Current estimates put worldwide production of fossil-based BDO at > 106 T/year. With an installed competitive base, economics for the bioprocess were a paramount consideration.
By 2012, this process had been demonstrated at commercial scale, and in 2012/2013 both Novamont and BASF entered into a license of this GENO BDO technology from Genomatica. Both licensees have begun customer sampling and have communicated their intent to build commercial-scale facilities to produce renewable BDO.
Genomatica constructed the BDO pathway in an industrial E. coli production host.72 Making use of the naturally occurring sugar metabolism via glycolysis and the TCA cycle, Genomatica researchers added the genes necessary to convert Succinyl-CoA to 4-hydroxy-butyrate and on to 1,4-butanediol. As is typical for bioproduction of chemicals, once the basic metabolic pathway is constructed, Genomatica tuned the pathway to enhance production rate and titer, and to improve yield by eliminat-
ing metabolites produced via competing pathways. These improvements were critical to achieving the market-competitive process economics.
Following the development of the production host, process engineering, both fermentation and a multistep separation and purification process was needed to produce polymer-grade BDO.
The industrial enzyme industry rapidly expanded in the 1960s. Most of the early products were produced through fermentation processes, using wild-type hosts, either bacterial or fungal. These hosts are still predominant in today’s industry.
Starting from the 1970s, the use of recombinant DNA techniques, combined with protein engineering, had a profound impact on the industry, allowing for more efficient enzyme production, reducing enzyme production costs, and leading to growth of markets and applications. The development of deep-tank, fed-batch aerobic fermentation, replacing the earlier Koji process of culturing of microorganisms on semisolid media, enabled improved efficiency and reduced costs.
Today, the market for industrial enzymes exceeds $5 billion, worldwide.73 Enzymes enable industries ranging from high-fructose corn syrup and citric acid as food ingredients, to fuel ethanol, to enzyme-based “stone washing” of blue jeans, to more efficient stain removal by detergents (Box 2-2).
Industrial biotechnology will need a governance frameworki that balances important social goals and manifests important values. Governance involves deployment of a variety of policy tools by which an industry’s behavior can be shaped, including education of industry actors, industry self-governance through standard setting, accreditation, government standard setting and regulation, public engagement and public scrutiny, tort liability, and other mechanisms for developing safety standards and controls. For governing the industrialization of biology, key goals pertain to safety (risk identification and mitigation) and sustainability. For industrial biotechnology to deliver widespread benefits, it must have low
_______________________
i Governance framework refers to the process of governing the industrial biotechnology, including industry norms, government regulations, and trade associations, among other methods. A more detailed background of U.S. government regulations can be found in Appendix C and at Carter S. R., Rodemeyer M., Garfinkel M.S., and Friedman R.M. Synthetic Biology and the U.S. Biotechnology Regulatory System: Challenges and Options, J. Craig Venter Institute: Rockville, MD, 2014.
BOX 2-2 Cold-Water Protease Enzyme
Each day, Americans do 123 million loads of laundry. Most of us choose to set our dials to warm or hot water to ensure that stains are removed. We now have a new choice—one that is better for our clothes, lowers our costs, and protects the environment. A new cold-water protease enzyme makes this possible.
Enzymes have been used to improve the cleaning efficiency of detergents for decades. Detergent enzymes account for about 30 percent of industrial enzyme production and are one of the most successful applications of industrial bioscience. Proteases, in particular, are the most widely used enzymes in detergents and facilitate the removal of proteinaceous deposits and stains. Subtilisins are the prototypical group of bacterial alkaline serine proteases that are extensively used in detergents. Historically, they were not particularly stable, or active, in the high-surfactant, alkaline washing processes
Recently, new protein engineering technology has enabled the tailoring of new protease enzymes that meet the trend toward cold-water washing with uncompromised cleaning performance. This breakthrough came from the use of synthetic genes and massively parallel predictive screens to map key properties such as enzyme activity and stability. In parallel, computation and modeling of physicochemical and structural constraints were used to design and to inform the hi h-throu hput screenin
The new protease delivers the cleaning power of warm-water wash at temperatures below 20°C. The mutations introduced near the enzyme’s active site serve to broaden its specificity toward more protein substrates and to cleave them faster. Other mutations serve to increase the affinity of structural calcium ligands, thus conferring stability in harsh detergent environments. The new enzyme demonstrated superior cleaning performance for a broad panel of protein stains, such as blood, grass, and dairy.
Current energy consumption for U.S. residential clothes washing is substantial, amounting to 54,000 GWh, annually. This represents CO2 emissions of 40 million annual tons. About 80 percent of this energy is used to heat the wash water. Universal adoption of cold-water washing could save as much as 45,000 GWh each year, reducing CO2 emissions by the equivalent of more than 6 million cars.
environmental impacts, use biological feedstocks sustainably, and operate according to high safety standards with respect to humans, animals, and the environment.
The governance framework should aim to achieve other goals as well, such as trustworthiness. For the industry to be trustworthy, its actors should adhere to high safety and environmental standards, and the public or its governmental representatives must have access to information that allows them to assess industry adherence to such standards.
For the governance framework to have legitimacy in the eyes of the public and the industry, it should be perceived as fair, transparent, effi-
cient, and inclusive of diverse viewpoints. These values sometimes come into tension, and all governance systems must make trade-offs between or among them. For instance, efficiency may be hindered by the need for public participation (inclusion of diverse viewpoints) and fair processes. Governance values can also interact synergistically. For instance, public participation and citizen oversight are promoted by transparency, which permits the public to acquire information about industry activities, contemplated regulations, and other relevant issues on which individuals from outside the industry might want to provide feedback. For the governance framework to achieve a reasonable balance among the relevant goals and important values, the framework must be carefully designed. Furthermore, governance can be more or less adaptive, insofar as it explicitly builds on opportunities for learning and adjustment as technologies evolve along with the social, political, and environmental contexts. Adaptive governance poses information problems, however, because somebody must be collecting data on the functioning of the governance processes and on changes in the technology and in the surrounding context for policy makers to know how and when the governance should change.
Regulation is one component of governance—one mechanism by which governments, representing and balancing a broad array of interests, shape behavior in the industry by setting and enforcing standards. Regulation can be very prescriptive, so-called command-and-control style, or it can be more flexible, such as market-based frameworks (e.g., carbon trading), negotiated project-specific licensing, multiparty collaborative planning, and other alternative or second-generation approaches.
The current regulatory environment for approval and control of organisms used in bioprocessing is complex and still developing. Biologically produced chemicals can be regulated by the Environmental Protection Agency (EPA) under the Toxic Substances Control Act (TSCA) or the Federal Insecticide, Fungicide, and Rodenticide Act (FIFRA); the USDA’s Animal and Plant Health Inspection Service (USDA-APHIS) under the Plant Protection Act; and the FDA under the Food, Drug, and Cosmetics Act. The applicable U.S. legal regime will depend on a product’s intended use rather than on the method by which it was made. The Occupational Safety and Health Administration’s (OSHA’s) general regulations also address the health and safety of people who work in industries where biotechnology is used. OSHA does not have regulations specific to work with engineered organisms, but it does require that employers create a workplace free from serious, recognized hazards, and it lays out principles and precautions for working with hazardous chemicals. The overlap of legal regimes, and the uncertainty over how and whether regimes will apply to complex engineered organisms, can lead to uncertainty that may hinder
technology investment, development of new products, or more efficient production processes (Box 2-3).
In 1986, the United States developed the Coordinated Framework for the Regulation of Biotechnology, a formal policy for coordinating the activities of the agencies that regulate biotechnology products and research. Where regulatory oversight involves more than one agency, the policy specifies that one agency will take the lead in consolidating and coordinating regulatory reviews. The Coordinated Framework provides a good basis for reconciling the overlapping jurisdiction of various agencies in situations where uncertainty might arise. However, despite the multiple statutory authorities under whom agencies can regulate industrial biology, the existing legal regimes may fail to adequately address some foreseeable risks. Neither the EPA nor USDA-APHIS regulates production processes, and both focus most of their biotechnology-specific regulation on the “premarket” phase of the product life cycle.ii It is, therefore, unclear whether these agencies have adequate authority or expertise to ensure that proper containment and disposal procedures will be used at commercial manufacturing facilities once a manufacturer engages in legal commercial production of a biologically produced chemical. It is also unclear whether either agency has authority to sanction a firm that creates a public health or environmental hazard by inappropriately disposing of host microorganisms, waste biomass, or co-products.
It is not clear whether any agency has adequate authority to oversee worker, environmental, or public safety when research to design and test industrial microorganisms is privately funded and conducted by commercial firms. Working with engineered microorganisms requires taking precautions commensurate with the level of risk. The design of manufacturing facilities should include appropriate containment features, and firms should include biosafety considerations in their standard practices. Firms that have long been involved in the production of chemicals by ordinary chemical synthesis will have a safety culture and safety engineering in their facilities, but their expectations and practices may have to evolve to accommodate biological processes. There currently are no unified federal standards for commercial production of chemicals through biological routes; there is nothing for industry that is comparable to the National Institutes of Health (NIH) Guidelines.
Similarly, the Centers for Disease Control and Prevention and NIH have created the Biosafety in Microbiological and Biomedical Labora-
_______________________
ii It may be possible that some waste streams could fall under the Resource Conservation and Recovery Act (RCRA); however, the EPA implementation under this act provides two categories of regulated waste, “Listed Wastes” and “Characteristic Wastes,” each with very specific lists of chemicals and their concentrations that may be regulated. See 40 CFR §261.
BOX 2-3 Biosafety Design Considerations
The U.S. Department of Health and Human Services has put forward screening framework guidance for providers of double-stranded DNA. These guidelines have been implemented by the International Gene Synthesis Consortium’s (IGSC’s) harmonized screening protocol, which consists of both gene sequence and customer screening approaches to promote biosecurity. Biosafety and biosecurity gene sequence screening approaches can readily be incorporated into the integrated design toolchain. Although IGSC’s protocol does not enable companies to identify and predict problematic biosafety and biosecurity properties that emerge when multiple components come together in an organism or bioprocess, it does when an individual component is of concern. A similar problem is the development of algorithms to assign or discern design attribution for legal or law enforcement purposes. Methods and tools that address emergent biosafety and biosecurity concerns, including tools that identify the metabolic dependencies and physical containment properties of designed organisms, should also be included in the integrated design toolchain. Furthermore, methods and tools for identifying hazardous chemical properties of feedstocks, intermediates, and products (from material safety data sheets or other information sources) will be very important for assessing the biosafety aspects of an overall bioprocess.
tories (BMBL) manual, which complements and expands on the NIH Guidelines. Now in its fifth edition, the BMBL sets forth principles for risk assessment and containment of hazardous biomaterials and has become the code of practice for biosafety. No agency, however, has specified the precautions to be taken in privately funded research and product development, or mandated adoption of the BMBL or similar principles.
“In modern democracies . . . the public plays a central role in determining how science is funded, used, and regulated.”74 The governance regime for industrializing biology should enhance opportunities for the public to engage with regulators and industry. By doing so, the regime can manifest the values of trustworthiness, transparency, and participation and can help ensure that science serves the greater public good. Such engagement enables members of the public to learn about technologies and oversight mechanisms, and to play a role in governance processes. There are numerous methods of public engagement, and the optimal approach will vary with the particular social context, technology, and applicable regulatory regime(s). This report does not recommend particular approaches for public engagement but does emphasize its importance.
Another set of social factors that will influence the pace and direction of industrial biology is the balance between open innovation and informa-
tion sharing, on the one hand, and proprietary product development, on the other hand. In the context of synthetic biology, a great deal has been written about the virtues of open versus proprietary research as pathways for advancing the field. This report does not extend that previous work. Here, we merely note two important points. First, “open science” does not necessarily require that findings be put in the public domain, although that is one approach. Open science can also be promoted by licensing intellectual property to create a science commons, or otherwise to protect broad access to a discovery or product. Second, patents are often viewed as one component in the proprietary and private property–driven approach to innovation. To date, patents have played a significant role in attracting and protecting investments in the biotechnology and chemical industries; however, the patent law has recently changed in ways that constrain opportunities to patent some biotechnology inventions and processes. The committee did not attempt to predict the degree to which recent legal changes will affect patenting of this report’s subject matter—complex engineered microorganisms and industrial production processes using those microorganisms. Patent protection will still be available for some important products and processes in industrial biology, but recent legal changes could affect the number and nature of patents, which may in turn influence the ways businesses organize and collaborate. To promote the industrialization of biology, academic and industry scientists in synthetic biology and related fields will need to determine an acceptable balance between open and proprietary approaches to innovation.
Related to balancing open and proprietary science is the increasingly prevalent practice of data sharing. In other contexts (e.g., biomedical science), sharing of precompetitive, detailed data has proven beneficial for a broad array of stakeholders, from academia to industry. Such sharing, which is being encouraged or mandated75 by many public funders of science and across the U.S. government, requires that researchers make available types of data beyond the summarized, aggregated, highly analyzed data typically found in publications. Stakeholders in industrial biotechnology might also identify opportunities to advance the field through appropriate data sharing.