The committee’s charge was to “develop evidence-based clinical diagnostic criteria for ME/CFS for use by clinicians, using a consensus-building methodology.” To this end, the committee conducted a comprehensive review of the current literature pertinent to the diagnosis of ME/CFS—the results of which are reported in the preceding chapters—keeping in mind the views of clinicians, patients, and their families and caregivers, particularly as they shed light on the complex symptoms associated with this disorder. In the studies reviewed, detailed histories obtained using research questionnaires revealed that the majority of ME/CFS patients experienced post-exertional malaise (PEM) as well as intense fatigue, unrefreshing sleep, cognitive impairment, and autonomic dysfunction, regardless of the case definition or research tool used (findings that accord with those of the Centers for Disease Control and Prevention’s [CDC’s] Multi-Site Clinical Study).1 Several signs and symptoms—PEM; orthostatic intolerance; and cognitive impairment, specifically slow information processing—have distinctive findings in ME/CFS patients.
Conclusion: It is clear from the evidence compiled by the committee that ME/CFS is a serious, chronic, complex, and multisystem disease that frequently and dramatically limits the activities of affected patients.
____________________
1 Personal communication from Elizabeth Unger, 2014. Preliminary analysis of CDC Multi-Site Clinical Study.
Relying on the findings reported in the literature reviewed, as well as stakeholder input, the committee refined diagnostic criteria that focus more on the central symptoms of this disease than many other definitions (see Box 7-1). The committee anticipates that use of these criteria will make it easier for clinicians to make appropriate and timely diagnoses of ME/CFS in both children and adults and to provide appropriate treatment and management while avoiding possibly harmful interventions. The proposed criteria are quite similar to the Canadian Consensus Criteria (CCC) (Carruthers et al., 2003). The committee recognizes that some patients diagnosed by other criteria, such as the Fukuda definition (Fukuda et al., 1994), will not fulfill all of the criteria proposed here, but it emphasizes that all patients should receive appropriate care.
The committee weighed several factors in reaching consensus on these diagnostic criteria: (1) the frequency and severity with which these symptoms were experienced by patients, (2) the strength of the scientific literature, and (3) the availability of objective measures supporting the association of particular symptoms with the diagnosis. Patient reports and
BOX 7-1
Proposed Diagnostic Criteria for ME/CFS
Diagnosis requires that the patient have the following three symptoms:
- A substantial reduction or impairment in the ability to engage in pre-illness levels of occupational, educational, social, or personal activities that persists for more than 6 months and is accompanied by fatigue, which is often profound, is of new or definite onset (not lifelong), is not the result of ongoing excessive exertion, and is not substantially alleviated by rest,
- Post-exertional malaise, * and
- Unrefreshing sleep*
At least one of the two following manifestations is also required:
- Cognitive impairment* or
- Orthostatic intolerance
_____________
* Frequency and severity of symptoms should be assessed. The diagnosis of ME/CFS should be questioned if patients do not have these symptoms at least half of the time with moderate, substantial, or severe intensity.
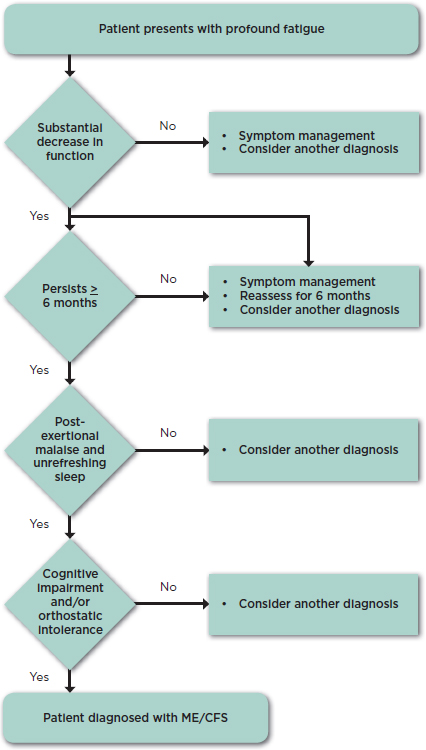
symptom surveys as well as scientific evidence consistently showed that impaired function, PEM (an exacerbation of some or all of an individual’s ME/CFS symptoms after physical or cognitive exertion, or orthostatic stress that leads to a reduction in functional ability), and unrefreshing sleep are characteristic symptoms almost universally present in ME/CFS; thus, the committee considered them to be core symptoms. The committee also found that cognitive impairment and orthostatic intolerance are frequently present in ME/CFS patients and have distinctive findings in these individuals that, particularly when viewed together with the core symptoms, distinguish ME/CFS from other fatiguing disorders. It is essential that clinicians assess the severity and duration of symptoms over an extended period of time because moderate or greater frequency and severity of symptoms are required to distinguish ME/CFS from other illnesses. Regarding the duration of the illness, the proposed criteria require 6 months to make a diagnosis in light of evidence that many other causes of similar fatigue do not last beyond 6 months (Jason et al., 2014; Nisenbaum et al., 1998). These criteria led the committee to create the diagnostic algorithm shown in Figure 7-1.
Other clinical features that may be seen in patients with this disorder are a history of certain infections known to act as triggers for ME/CFS that preceded the onset of symptoms and many types of pain, including headaches, arthralgia, and myalgia. Other complaints, such as gastrointestinal and genitourinary problems, sore throat, tender axillary/cervical lymph nodes, and sensitivity to external stimuli, are reported less frequently (Buchwald and Garrity, 1994; Jason et al., 2013; McGregor et al., 1996). These features, when present, can support the diagnosis of ME/CFS.
These criteria and the algorithm in Figure 7-1 can be used for pediatric patients as well. Although they experience the same symptoms as adult patients, current evidence suggests that children and adolescents with ME/CFS are more likely to have onset following infection with Epstein-Barr virus (EBV) and to have difficulty with orthostatic intolerance and autonomic dysfunction. Profound fatigue, unrefreshing sleep, PEM, and cognitive impairment under stress are common in pediatric patients.
The current evidence base is insufficient to determine whether the illness defined by existing ME/CFS criteria or by the new criteria proposed here has subtypes or is actually a collection of potentially distinguishable disorders. Fukuda and colleagues (1994, p. 956) urged that individuals with ME/CFS “be sub-grouped before analysis or stratified after analysis” by researcher-determined clinical characteristics (e.g., length of illness, infectious onset) or objective test results (e.g., treadmill testing, immunologic markers). Today, many clinicians and researchers agree that ME/CFS is a heterogeneous disease that may be composed of subgroups (Bassi et al., 2008; Carruthers et al., 2011; Jason et al., 2005; May et al., 2010). Yet in practice, large studies that include individuals with diverse symptoms and
yield diverse findings are needed to identify subgroups reliably, and few such studies have been conducted. Nor is there adequate evidence to enable comment on the manifestations of ME/CFS across the life course, except to the extent that orthostatic intolerance and postural orthostatic tachycardia syndrome (POTS) appear to be more prominent in children than in adults with the disorder (see the sections on orthostatic intolerance and autonomic dysfunction in adults and children with ME/CFS in Chapters 4 and 6, respectively). Notably, clinical impression suggests that a larger percentage of children and adolescents relative to adults recover from the disease, although this impression has not been confirmed by the few longitudinal studies conducted to date (Bell et al., 2001; Brown et al., 2012; Cairns and Hotopf, 2005). The committee notes, however, that many of these questions are under active investigation.
Conclusion: One of the committee’s most important conclusions is that a thorough history, physical examination, and targeted workup are necessary and often sufficient for diagnosis of ME/CFS.
Table 7-1 provides examples of (1) terms ME/CFS patients commonly use to describe their symptoms; (2) potential questions that can alert clinicians to the diagnosis; (3) in-office questionnaires, tools, or clinical observations that may aid diagnosis or indicate the need for further assessment or referral to a specialist (see also Appendix D); and (4) the objective tests that reveal distinctive findings in these patients. The information provided in this table can be used to operationalize the committee’s proposed diagnostic criteria in the steps described below. Table 7-2 provides similar information for other symptoms/manifestations of ME/CFS that can support diagnosis.
Diagnostic Tools
First and foremost, listening to patients and taking a careful history are key diagnostic tools. Patients’ symptoms and the words they frequently use in describing them should lead clinicians to consider the diagnosis of ME/CFS. Patients often use a variety of evocative words to describe particular symptoms. Histories of many of these symptoms are frequently and consistently elicited when any of an assortment of research questionnaires are used. Clinicians should ask a series of questions to elicit history consistent or inconsistent with ME/CFS. It is essential that clinicians assess the severity and duration of symptoms over the past month or more. Chronic, frequent, and moderate or severe symptoms are required to distinguish ME/CFS from other illnesses. Questionnaires and visual analog scales in
| Major Symptom | Patient Descriptions | Potential Questions for Eliciting the Medical History (all questions should explore frequency and severity of symptoms) | Potential In-Office Questionnaires, Tools, or Clinical Observations | Abnormal Findings on Reliable Objective Tests That, When Considered in the Context of Other Symptoms, Support the Diagnosis of ME/CFS (these tests are not routinely required, nor do negative results preclude the diagnosis) |
| Fatigue |
|
|
History of this symptom can consistently be elicited using research questionnaires but without specificity. For information on tools or questionnaires for assessing fatigue in adults and children, see Appendix D. Observe for progressive fatigue, need for help or need to lie down during a prolonged exam. |
|
| Substantial decrease or impairment in function |
|
Think back to what you were able to do before you became sick. How much has this illness affected:
What are you able to do now? How does it compare with what you were able to do before? Are you able to attend school regularly? Are you able to participate in any extracurricular clubs or sports activities? |
For information on other tools or questionnaires for assessing decreased or impaired function in adults and children, see Appendix D. |
| Major Symptom | Patient Descriptions | Potential Questions for Eliciting the Medical History (all questions should explore frequency and severity of symptoms) | Potential In-Office Questionnaires, Tools, or Clinical Observations | Abnormal Findings on Reliable Objective Tests That, When Considered in the Context of Other Symptoms, Support the Diagnosis of ME/CFS (these tests are not routinely required, nor do negative results preclude the diagnosis) |
| Post-exertional malaise (prolonged exacerbation of a patient’s baseline symptoms after physical/cognitive/orthostatic stress; may be delayed relative to the trigger) |
|
(Consider asking patients to keep a diary for a week or two, documenting activities and symptoms.) |
History of this symptom can consistently be elicited using research questionnaires in adults and children (see Appendix D). | Two cardiopulmonary exercise tests (CPETs) separated by 24 hours that demonstrate marked inability to reproduce maximal or anaerobic threshold measures on the second day; values on the second CPET must be much lower than those on the first CPET (note that this test may induce severe exacerbation of symptoms in these patients); 2-day CPETs are not required for diagnosis, although they may be needed for other reasons, such as obtaining Social Security Disability. |
| Feeling unrefreshed despite sleeping many hours and other sleep disturbances |
|
|
History of this symptom can be consistently elicited using research questionnaires (see Appendix D). | No evidence that currently available sleep studies contribute to the diagnosis of this disorder. |
| Cognitive impairments (problems with thinking exacerbated by exertion, effort, or stress or time pressure) |
|
|
While research questionnaires frequently elicit cognitive impairments (see Appendix D), they are less successful at eliciting issues with slow processing. Observe for difficulties with thinking during the clinic visit—unusual trouble remembering medications, relating details of history or understanding questions/recommendations, expressing self. |
Formal neuropsychology testing is not required for diagnosis, although it may be needed for other reasons, such as obtaining Social Security Disability. |
| Major Symptom | Patient Descriptions | Potential Questions for Eliciting the Medical History (all questions should explore frequency and severity of symptoms) | Potential In-Office Questionnaires, Tools, or Clinical Observations | Abnormal Findings on Reliable Objective Tests That, When Considered in the Context of Other Symptoms, Support the Diagnosis of ME/CFS (these tests are not routinely required, nor do negative results preclude the diagnosis) |
| Orthostatic intolerance (Symptoms worsen upon assuming and maintaining upright posture and are ameliorated, though not necessarily abolished, by recumbency.) |
|
|
This is an area that is not currently explored in depth in research questionnaires, but some suggestions are included in Appendix D.
|
Standing test or tilt test to evaluate for postural tachycardia syndrome, neurally mediated hypotension, and orthostatic hypotension (See the section on orthostatic intolerance and autonomic dysfunction in Chapter 4.) Tilt testing is not required for diagnosis, although it may be needed for other reasons, such as obtaining Social Security Disability |
|
|
||
NOTES: The patient descriptions included in this table were obtained from the Food and Drug Administration’s Voice of the Patient report (FDA, 2013), the public comments submitted for the study, and the ME/CFS experts on the committee.
TABLE 7-2 Assessing Other Symptoms/Manifestations of ME/CFS That May Support Diagnosis
| Other Symptoms/Manifestations | Patient Descriptions | Potential Questions for Eliciting the Medical History (all questions should explore frequency and severity of symptoms) | Potential In-Office Questionnaires, Tools, or Clinical Observations | Abnormal Findings on Reliable Objective Tests That, When Considered in the Context of Other Symptoms, Support the Diagnosis of ME/CFS (these tests are not routinely required, nor do negative results preclude the diagnosis) |
| Pain |
|
|
For information on tools or questionnaires for assessing decreased or impaired function, see Appendix D. | |
| History of prior triggering infection from which patient never fully recovered | Patients report coming down with a viral-like, respiratory, or gastrointestinal infection from which they never recovered entirely. |
|
Laboratory evidence of prior infection is not required to diagnose this disorder, although it may be needed for other reasons, such as obtaining Social Security Disability; the presence of virus-specific immunoglobulin M (IgM), if documented near the onset of illness, may support diagnosis. | |
| Abnormal immune function | Patients may report that they are more or less susceptible to infections. |
|
Decreased function of natural killer cells in those with severe disease (Note that this is not unique to this disorder and that commercially available tests may not give accurate results.) | |
NOTES: The patient descriptions included in this table were obtained from the Food and Drug Administration’s Voice of the Patient report (FDA, 2013), the public comments submitted for the study, and the ME/CFS experts on the committee.
which patients rate the frequency and severity of symptoms may be helpful in making these assessments (see Appendix D).
Second, there are several in-office tests and clinical observations that may aid in making the diagnosis or indicate a need for further evaluation or referral. The clinical utility of these tests and observations, however, needs more thorough evaluation. There are objective measures of function that can be helpful in making or confirming the diagnosis. The committee notes that these tests are not required in every case; indeed, they should be performed for purposes of diagnosis only when the initial assessment is not definitive. The tests are neither pathognomonic nor diagnostic of ME/CFS on their own, nor does every patient have to have abnormal results on any particular test. Indeed, some tests should be undertaken with caution as they may seriously exacerbate the patient’s symptoms. Rather, the results of these tests, when performed in cases of uncertainty, should be considered in the context of the patient’s overall clinical picture. The committee recognizes that objective tests also may be useful for patient management but notes that evaluating such uses is beyond its charge.
Conclusion: The central point is that ME/CFS is a diagnosis to be made.
The clinician who suspects this disorder should obtain a detailed history, perform a comprehensive physical examination, and consider conducting an appropriate symptom-focused diagnostic work-up to determine a differential diagnosis and, where clinically indicated, to exclude other disorders that could cause the patient’s symptoms as well as to identify any comorbid conditions. As noted previously, the objective functional tests listed in Table 7-1 and Appendix D frequently are not required to make the diagnosis of ME/CFS. If these tests are performed, however, abnormal results can support the diagnosis.
Evidence shows that some tests are not helpful in making the diagnosis of ME/CFS. The committee found tests of neuroendocrine function to be an example, warranted only if disorders in those systems are suspected on other clinical grounds. Similarly, currently available sleep studies are not useful for identifying the presence or absence of unrefreshing sleep.
Recommendation 1: Physicians should diagnose myalgic encephalomyelitis/chronic fatigue syndrome if diagnostic criteria are met following an appropriate history, physical examination, and medical work-up. A new code should be assigned to this disorder in the International Classification of Diseases, Tenth Revision (ICD-10), that is not linked to “chronic fatigue” or “neurasthenia.”
Even if patients do not meet the criteria for this disorder, clinicians should address their symptoms and concerns. Patients who have not yet been symptomatic for 6 months should be followed over time to see whether they meet the criteria for ME/CFS at a later time.
Recommendation 2: The Department of Health and Human Services should develop a toolkit appropriate for screening and diagnosing patients with myalgic encephalomyelitis/chronic fatigue syndrome in a wide array of clinical settings that commonly encounter these patients, including primary care practices, emergency departments, mental/behavioral health clinics, physical/occupational therapy units, and medical subspecialty services (e.g., rheumatology, infectious diseases, neurology).
CDC’s CFS Toolkit (CDC, 2014) and the International Association for CFS/ME’s ME/CFS: Primer for Clinical Practitioners (IACFS/ME, 2014) may be potential places to start, but both need updating in a number of areas in light of the findings presented in this report. The development of clinical questionnaire or history tools that are valid across populations of patients should be an urgent priority. The DePaul Symptom Questionnaire, which has been used extensively in research (DePaul Research Team, 2010), as well as the CDC Symptom Inventory (Wagner et al., 2005), may provide a solid base from which to begin developing questionnaires and interview guides that can be validated for clinical use. Also critical is the development of brief in-office tests for detecting PEM and orthostatic intolerance, for which there are known distinctive findings on more complex clinical tests. For example, evaluation of the extent to which results of the 10-minute lean test correlate with abnormalities on tilt table testing would be useful. It would also be helpful to develop a brief set of neuropsychology tests targeting the information processing problems that affect patients with ME/CFS. This is a particular need as current neuropsychology tests require many hours to complete, frequently precipitating symptoms that patients may find intolerable, and often are impractical for patients because they are quite expensive and not covered by many insurance plans (Lange, 2010). Identification of a set of distinctive biomarkers for this disorder should also be a priority. Finally, all of the above tools should be evaluated to determine how well they distinguish ME/CFS from other complex, multisystem, and fatiguing disorders.
Comorbidities
Over the years, case definitions of ME/CFS have differed significantly in their classification of exclusionary conditions and comorbidities. As a re-
sult, a number of disorders, such as morbid obesity and an array of psychiatric disorders, are listed as exclusionary in one definition and as comorbid in another, despite the lack of scientific evidence that being affected by such disorders precludes having ME/CFS. Indeed, it has become increasingly clear that many patients with ME/CFS have other disorders as well, some of which—including fibromyalgia, irritable bowel syndrome, metabolic syndrome, sleep disorders, and depression—may have symptoms that overlap with those of ME/CFS (Buchwald and Garrity, 1994; Johnson et al., 1996; Maloney et al., 2010). Some of these other disorders may develop as part of the spectrum of ME/CFS or in response to the burdens of this disorder.
The committee decided against developing a comprehensive list of potential comorbid conditions, but it points to conditions that clinicians may wish to consider that have been identified by the International Consensus Criteria for ME (ME-ICC) and CCC, including fibromyalgia, myofascial pain syndrome, temporomandibular joint syndrome, irritable bowel syndrome, interstitial cystitis, irritable bladder syndrome, Raynaud’s phenomenon, prolapsed mitral valve, depression, migraine, allergies, multiple chemical sensitivities, Sicca syndrome, obstructive or central sleep apnea, and reactive depression or anxiety.
In the section on pain in Chapter 5, the committee discusses the presence of fibromyalgia in patients with ME/CFS. The lack of rigorous studies assessing how ME/CFS patients with fibromyalgia differ from those without fibromyalgia and from patients with fibromyalgia who do not meet criteria for ME/CFS prevents a complete understanding of how these two entities differ. However, the available literature shows that differences between them exist. In addition to the evidence presented in Chapter 5, Cook and colleagues (2012) found that patients with ME/CFS + fibromyalgia showed cardiopulmonary changes during submaximal exercise that differentiated them from controls; the ME/CFS-only group did not show these effects. Also, an analysis of sleep records showed very different results for the two study groups: those with ME/CFS alone had a significantly higher probability of transitioning from rapid eye movement (REM) sleep to wakefulness relative to the ME/CFS + fibromyalgia group; the latter group, on the other hand, showed both sleep pressure and sleep disruption—increased transitions from wakefulness to light sleep and increased transitions from deep to lighter sleep or wakefulness, respectively (Kishi et al., 2011). More research in this area is clearly needed.
The committee recognizes that diagnosis and treatment of comorbid conditions are necessary when caring for patients. For example, a patient with ME/CFS with a prominent history of snoring and sleep apnea may have polysomnography diagnostic of sleep apnea. Treatment with continuous positive airway pressure could improve the patient’s overall condition but not resolve all the symptoms of ME/CFS, signifying that in this indi-
vidual, obstructive sleep apnea is a comorbid condition rather than the cause of the patient’s ME/CFS symptoms.
Areas Deserving Further Study
While there was sufficient evidence with which to carry out the first steps of its task, the committee was struck by the relative paucity of research on ME/CFS conducted to date in many areas related to this disorder. Remarkably little research funding has been made available to study the etiology, pathophysiology, and effective treatment of this disease, especially given the number of people afflicted. Thus, the committee was unable to define subgroups of patients or even to clearly define the natural history of the disease. More research is essential.
Future diagnostic research will be most instructive when protocols include patients identified using the committee’s proposed diagnostic criteria for ME/CFS as well as patients with other complex fatiguing disorders. Almost all of the studies conducted to date have compared patients with ME/CFS with healthy controls rather than with patients with these other fatiguing disorders. As a result, there is a paucity of data to guide clinicians in distinguishing among these disorders, a gap that urgently needs to be filled.
Finding the cause of and cure for ME/CFS may require research that enlists a homogeneous sample of patients from which important subsets can be identified in terms of disease symptomatology, responses to physical and cognitive stressors, brain imaging, the microbiome, virology, immune function, and gene expression. Integrative approaches using systems biology may be useful in unraveling illness triggers. Studies aimed at assessing the natural history of the disease and its temporal characteristics (onset, duration, severity, recovery, and functional deficits) are essential for a better understanding of ME/CFS and also are important to further refine the diagnostic criteria proposed in this report.
DISSEMINATING THE DIAGNOSTIC CRITERIA
The criteria proposed here will not improve the diagnosis and care of patients unless health care providers use them. Accordingly, the committee developed an outreach strategy for disseminating the clinical diagnostic criteria resulting from this study nationwide to health care professionals so patients will receive this diagnosis in an accurate and timely manner (see Chapter 8). The committee believes that focusing dissemination efforts on reaching primary care and other providers who encounter these patients will increase awareness of and familiarity with the new criteria in a manner that will be most beneficial to patients with ME/CFS.
UPDATING THE DIAGNOSTIC CRITERIA
The committee recognizes that new and accumulating evidence will likely enable refinement of the diagnostic criteria proposed in this report and possibly define subtypes of the disease or even distinct entities. Ideally, diagnostic criteria should be updated when evidence supports a change in order to improve the identification and care of affected individuals (IOM, 2011). The need for reexamination could arise for a number of reasons. Evidence providing greater understanding of the etiology and pathophysiology of ME/CFS might facilitate more discriminating, efficient, or precise diagnostic approaches. Evidence validating either screening or diagnostic tools that are practical for widespread use by generalists might lead to improved identification of affected individuals and so warrant inclusion. And a demonstration of subtypes or even discrete disease entities within the current case definition might enable better prognostication and help target treatment approaches.
Given the current pace of scientific research, the committee expects that the criteria proposed here may warrant reassessment in the not too distant future. Therefore, the committee recommends continuing surveillance of the evidence and revisiting of the criteria in no more than 5 years. Efforts to update the criteria should comply with the process recommendations set forth in the Institute of Medicine (IOM) report Clinical Practice Guidelines We Can Trust, which were developed for clinical practice guidelines but are also pertinent to the development of trustworthy diagnostic criteria (IOM, 2011). Regardless of who convenes the update effort, the process for updating should be transparent and open to the views of all stakeholders to ensure that all relevant perspectives are considered. An evidence review that is guided by well-formulated questions informed by stakeholders’ views and is conducted by individuals with methodological expertise should underpin any update effort. Ideally, experienced individuals without significant conflicts of interest should conduct a systematic literature review to address the key questions. A multidisciplinary group that includes individuals with clinical expertise should identify those questions and rely heavily on the findings derived from the literature review when making recommendations about modifications of the diagnostic criteria. Members of this group should clearly disclose their potential conflicts of interest, and the conveners of the group should try to limit the number of members with significant conflicts, who should in no case represent a majority of the group’s membership. It is critical that support for these activities come from nonconflicted sources such as the Agency for Healthcare Research and Quality (AHRQ) or foundations.
Recommendation 3: A multidisciplinary group should reexamine the diagnostic criteria set forth in this report when firm evidence supports modification to improve the identification or care of affected individuals. Such a group should consider, in no more than 5 years, whether modification of the criteria is necessary. Funding for this update effort should be provided by nonconflicted sources, such as the Agency for Healthcare Research and Quality through its Evidence-based Practice Centers process, and foundations.
As noted in Chapter 3, many concerns have been raised about the term “chronic fatigue syndrome” by clinicians and particularly by patients, their families, and their caregivers.
Conclusion: The committee agrees that the term “chronic fatigue syndrome” can result in stigmatization and trivialization and should no longer be used as the name of this illness.
In response to its directive to “recommend whether new terminology for ME/CFS should be adopted” (see Box 1-1 in Chapter 1), the committee considered the variety of case definitions and diagnostic criteria that have been proposed for ME/CFS as well as the public comments submitted for this study (see Box 3-2 in Chapter 3). In considering what name would be most appropriate, the committee turned first to “myalgic encephalomyelitis” or “encephalopathy” (ME), which was the name most commonly supported in the public comments. The committee, however, was concerned that the term “encephalomyelitis” is not well supported by the evidence and that there is substantial controversy surrounding the two versions of this name. Neither version conveys the full complexity of this disorder. The committee notes that many of the other names that have been proposed focus on particular organ systems. Others suggest particular etiologies, such as immune or infectious, for this disorder that are not yet proven.
The committee was convinced of the value of creating a name that conveys the central elements of this disease, a practice for which there is much precedent in medicine for disorders whose etiology or pathophysiology is not yet well understood. After extensive consideration, and being mindful of the concerns expressed by patients and their advocates, the committee recommends that the disorder described in this report be named “systemic exertion intolerance disease” (SEID). “Systemic exertion intolerance” captures the fact that exertion of any sort—physical, cognitive, emotional—can adversely affect these patients in many organ systems and in many aspects
of their lives. The committee intends for this name to convey the complexity and severity of this disorder.
Recommendation 4: The committee recommends that this disorder be renamed “systemic exertion intolerance disease” (SEID). SEID should replace myalgic encephalomyelitis/chronic fatigue syndrome for patients who meet the criteria set forth in this report.
The committee’s careful review of the literature, together with the input of patients and clinicians, revealed that ME/CFS is a complex disorder characterized by a prolonged, significant decrease in function; fatigue; postexertional malaise; unrefreshing sleep; difficulties with information processing, especially under time pressure; and orthostatic intolerance. ME/CFS frequently is accompanied by pain and a variety of other symptoms. It often is triggered by specific infections. Although research using complex investigative testing is yielding some unique findings, there is an urgent need to develop a series of questionnaires and objective tests that can easily be used to diagnose these patients in a variety of clinical settings. Current evidence does provide a path forward now, which is outlined in this report, but it is the committee’s hope that new developments in research, particularly in the creation of clinical diagnostic tools, will warrant revisiting these guidelines in the near future. To that end, the committee has proposed a process that should be used in updating its proposed diagnostic criteria.
In what will likely be its most controversial recommendation, the committee has proposed that the name “chronic fatigue syndrome” no longer be used to describe this disorder. The committee repeatedly heard from patients that this term was stigmatizing and too often precluded their receiving appropriate care. The committee instead determined that the name “systemic exertion intolerance disease” (SEID) better characterizes the disorder for which its proposed diagnostic criteria were developed.
Bassi, N., D. Amital, H. Amital, A. Doria, and Y. Shoenfeld. 2008. Chronic fatigue syndrome: Characteristics and possible causes for its pathogenesis. Israel Medical Association Journal 10(1):79-82.
Bell, D. S., K. Jordan, and M. Robinson. 2001. Thirteen-year follow-up of children and adolescents with chronic fatigue syndrome. Pediatrics 107(5):994-998.
Brown, M. M., D. S. Bell, L. A. Jason, C. Christos, and D. E. Bell. 2012. Understanding long-term outcomes of chronic fatigue syndrome. Journal of Clinical Psychology 68(9):1028-1035.
Buchwald, D., and D. Garrity. 1994. Comparison of patients with chronic fatigue syndrome, fibromyalgia, and multiple chemical sensitivities. Archives of Internal Medicine 154(18):2049-2053.
Cairns, R., and M. Hotopf. 2005. A systematic review describing the prognosis of chronic fatigue syndrome. Occupational Medicine (Oxford) 55(1):20-31.
Carruthers, B. M., A. K. Jain, K. L. De Meirleir, D. L. Peterson, N. G. Klimas, A. M. Lemer, A. C. Bested, P. Flor-Henry, P. Joshi, A. C. P. Powles, J. A. Sherkey, and M. I. van de Sande. 2003. Myalgic encephalomyelitis/chronic fatigue syndrome: Clinical working case definition, diagnostic and treatment protocols (Canadian case definition). Journal of Chronic Fatigue Syndrome 11(1):7-115.
Carruthers, B. M., M. I. van de Sande, K. L. De Meirleir, N. G. Klimas, G. Broderick, T. Mitchell, D. Staines, A. C. P. Powles, N. Speight, R. Vallings, L. Bateman, B. Baumgarten-Austrheim, D. S. Bell, N. Carlo-Stella, J. Chia, A. Darragh, D. Jo, D. Lewis, A. R. Light, S. Marshall-Gradisbik, I. Mena, J. A. Mikovits, K. Miwa, M. Murovska, M. L. Pall, and S. Stevens. 2011. Myalgic encephalomyelitis: International consensus criteria. Journal of Internal Medicine 270(4):327-338.
CDC (Centers for Disease Control and Prevention). 2014. CDC CFS toolkit. http://www.cdc.gov/cfs/toolkit (accessed July 22, 2014).
Cook, D. B., A. J. Stegner, P. R. Nagelkirk, J. D. Meyer, F. Togo, and B. H. Natelson. 2012. Responses to exercise differ for chronic fatigue syndrome patients with fibromyalgia. Medicine & Science in Sports & Exercise 44(6):1186-1193.
DePaul Research Team. 2010. DePaul symptom questionnaire. http://condor.depaul.edu/ljason/cfs/measures.html (accessed August 20, 2014).
FDA (Food and Drug Administration). 2013. The voice of the patient: Chronic fatigue syndrome and myalgic encephalomyelitis. Bethesda, MD: Center for Drug Evaluation and Research (CDER), FDA.
Fukuda, K., S. E. Straus, I. Hickie, M. C. Sharpe, J. G. Dobbins, and A. Komaroff. 1994. The chronic fatigue syndrome: A comprehensive approach to its definition and study. Annals of Internal Medicine 121(12):953-959.
IACFS/ME (International Association for Chronic Fatigue Syndrome/Myalgic Encephalomyelitis). 2014. ME/CFS: Primer for clinical practitioners. Chicago, IL: IACFS/ME.
IOM (Institute of Medicine). 2011. Clinical practice guidelines we can trust. Washington, DC: The National Academies Press.
Jason, L. A., K. Corradi, S. Torres-Harding, R. R. Taylor, and C. King. 2005. Chronic fatigue syndrome: The need for subtypes. Neuropsychology Review 15(1):29-58.
Jason, L. A., M. Sunnquist, A. Brown, M. Evans, S. Vernon, J. Furst, and V. Simonis. 2013. Examining case definition criteria for chronic fatigue syndrome and myalgic encephalomyelitis. Fatigue: Biomedicine, Health & Behavior Fatigue: Biomedicine, Health & Behavior 2(1).
Jason, L. A., B. Z. Katz, Y. Shiraishi, C. Mears, Y. Im, and R. R. Taylor. 2014. Predictors of post-infectious chronic fatigue syndrome in adolescents. Health Psychology & Behavioural Medicine 2(1):41-51.
Johnson, S. K., J. DeLuca, and B. H. Natelson. 1996. Depression in fatiguing illness: Comparing patients with chronic fatigue syndrome, multiple sclerosis and depression. Journal of Affective Disorders 39(1):21-30.
Kishi, A., B. H. Natelson, F. Togo, Z. R. Struzik, D. M. Rapoport, and Y. Yamamoto. 2011. Sleep-stage dynamics in patients with chronic fatigue syndrome with or without fibromyalgia. Sleep 34(11):1551-1560.
Lange, G. 2010. Cognitive function in CFS: A measure of disability? http://www.hhs.gov/advcomcfs/meetings/presentations/presentation_10132010_gudrunlannge.pdf (accessed August 5, 2014).
Maloney, E. M., R. S. Boneva, J. M. Lin, and W. C. Reeves. 2010. Chronic fatigue syndrome is associated with metabolic syndrome: Results from a case-control study in Georgia. Metabolism: Clinical & Experimental 59(9):1351-1357.
May, M., A. Emond, and E. Crawley. 2010. Phenotypes of chronic fatigue syndrome in children and young people. Archives of Disease in Childhood 95(4):245-249.
McGregor, N. R., R. H. Dunstan, M. Zerbes, H. L. Butt, T. K. Roberts, and I. J. Klineberg. 1996. Preliminary determination of the association between symptom expression and urinary metabolites in subjects with chronic fatigue syndrome. Biochemical and Molecular Medicine 58(1):85-92.
Nisenbaum, R., M. Reyes, A. C. Mawle, and W. C. Reeves. 1998. Factor analysis of unexplained severe fatigue and interrelated symptoms: Overlap with criteria for chronic fatigue syndrome. American Journal of Epidemiology 148(1):72-77.
Wagner, D., R. Nisenbaum, C. Heim, J. F. Jones, E. R. Unger, and W. C. Reeves. 2005. Psychometric properties of the CDC symptom inventory for assessment of chronic fatigue syndrome. Population Health Metrics 3(8).