Possible Mechanisms for Multiple Chemical Sensitivity: The Limbic System and Others1
Claudia S. Miller and Nicholas A. Ashford
The limited data available at this time suggest that any mechanism or model that would purport to explain the syndrome of multiple chemical sensitivities would need to address the features most closely associated with this illness (Ashford and Miller, 1991):
-
Symptoms involving virtually any system in the body or several systems simultaneously, and frequently the central nervous system (particularly mood, memory and concentration difficulties).
-
Differing symptoms and severity in different individuals, even among those having the same exposure.
-
Induction by a wide range of environmental agents, including pesticides and solvents.
-
Subsequent triggering by lower levels of exposure than those involved in initial induction of the illness.
-
''Spreading'' of sensitivity to other, often chemically dissimilar substances; each substance may trigger a different, but reproducible, constellation of symptoms.
-
Concomitant food and drug intolerances, estimated to occur in a sizeable percentage of those with chemical sensitivities.
-
Adaptation (masking), that is, acclimatization or tolerance to environmental incitants, both chemical and food, with continued exposure; loss of this tolerance with removal from the incitant(s); and augmented response with reexposure after an appropriate interval (for example, 4 to 7 days).
-
An apparent threshold effect referred to by some as the patient's total load, a theoretical construct that has been invoked to help explain why an individual develops this syndrome at a particular time. Illness is said to occur when the total load of biological, chemical, physical, and psychological stressors exceeds some threshold for the patient. This concept has emerged from clinical
-
observations; no direct experiments have been done to test its validity in humans; however, animal models do exist. The concept aligns with Selye's work on the general adaptation syndrome (Selye, 1946).
Items 2 and 3 above confound epidemiological investigation of this problem.
Items 3 and 4 suggest a two step process-(1) induction or sensitization resulting from an initial "major" exposure and (2) triggering of symptoms by subsequent lower level exposures to many different incitants.
Although knowledge of the mechanism of a disease may be useful for developing better therapies, such knowledge is not a prerequisite for intervention. Preventing the development of multiple chemical sensitivities in those not yet afflicted may be possible by controlling environmental exposures that cause the initial sensitization.
The most frequently cited physiological theories to explain chemical sensitivity involve the nervous system, the immune system, or the interaction between them because these two systems most dearly link the external environment and the internal milieu.2 The rapid responsiveness of these systems also makes them attractive candidates because symptoms of food or chemical sensitivity have been reported to develop within seconds of exposure. Many chemicals, such as polybrominated biphenyls (PBBs) and trichloroethylene, affect both the nervous system and the immune system. Until 1980, the idea of a possible direct communication between the nervous and immune systems was widely debated. Subsequently, the existence of a neuroimmunoendocrine axis has been increasingly realized. Several discoveries have helped to confirm the presence of two-way communication between the nervous and immune systems (Payan et at., 1986).
Kilburn proposes that the human nervous system, because it is so highly evolved, may be most susceptible to environmental agents (Kilburn, 1989):
"Sensitivity may be its undoing. The intuitive hypothesis is advanced that the nervous system is the most liable of the body's systems to damage from environmental toxins. Appreciation of damage may be masked because subtle dysfunction is concealed by the nervous system's remarkable redundancy and substitution of functions, or it is overlooked in clinical evaluations which are usually only qualitative."
MECHANISMS INVOLVING THE LIMBIC SYSTEM
The hypothalamus (part of the limbic system) has attracted considerable attention because it is the focal point in the brain where the immune, nervous, and endocrine systems interact (Bell, 1982). Bell notes that assuming a direct cause-and-effect relationship would be premature, but that the hypothalamus could mediate food and chemical addictions in patients with multiple chemical sensitivities. The olfactory system has known links to the hypothalamus and other parts of the limbic system, which has led Bell to speculate that "the olfactory system, hypothalamus and limbic system pathways would provide the neural circuitry by which adverse food and chemical reactions could trigger certain neural, psychological and psychiatric abnormalities." Patients with chemical sensitivities have
reported food cravings, binges, violence, or hypersexual activity following chemical exposures. A model involving the hypothalamus could help to explain such behavioral changes.
Some authors have alleged that psychological conditioning to odors is responsible for patients' reactions to chemicals. Of course, odor conditioning may occur in selected cases. However, physiological mechanisms involving the limbic system may also occur. A direct pathway from the oropharynx to the brain and hypothalamic and limbc region has been demonstrated in rats (Kate, 1968; Mailer et al., 1967). Substances placed in the oropharynx migrated to the brain in minutes via a pathway other than the blood stream and in higher concentrations than if administered via the gastrointestinal tract, suggesting a direct route from mouth (or nose) to brain. Similarly, Shipley showed that inhaled substances that contact the nasal epithelium may cross into the brain and be distributed widely via transneuronal transport (Shipley, 1985). Thus, molecules that are inhaled and contact the olfactory apparatus could influence functions in other parts of the brain.
Ryan and associates studied 17 workers who attributed changes in thought processes, particularly memory and concentration difficulties, or changes in mood to their exposure to solvents (Ryan et al., 1988). Those workers with "cacosmia" (a heightened sensitivity to odors resembling that reported by chemically sensitive individuals) performed most poorly on neurobehavioral tests requiring verbal learning or visual memory. The authors felt their findings supported a hypothesis that chronic solvent exposure may affect the "rhinencephalic structures" (the primitive "smell'' brain), the evolutionary precursor of the limbic system.
This phylogenetically ancient part of the brain is present in all mammals. It influences the organism's interaction with its environment in many subtle ways essential for preservation of the individual, its offspring, and the species. Limbus (Latin for "margin" or "rim") refers to its appearing like a rim around the edge of the cerebral hemispheres. Figure I shows its component parts. Note the dose anatomical relationship to the olfactory bulb. The olfactory nerves are the brain's most direct contact with the external environment. Each nerve consists of bipolar neurons, which have one end in the upper part of the nose and the other in the brain (olfactory bulb). Strong odors and even milder ones cause electrical activity in the amygdala and hippocampal areas of the limbic system (Monroe, 1986). Subsensory exposure to chemicals can cause protracted, if not permanent, alterations in the electrical activity of the limbic region, beginning first with the most sensitive structures, particularly that portion of the amygdala that analyzes odors (Bokina, 1976). All parts of the limbic system are intimately interconnected. Interestingly, ablation of the olfactory bulb in laboratory animals serves as a model for depression which investigators have used for testing the efficacy of various antidepressants (Jesberger and Richardson, 1988).
The amygdala is involved in feelings and activities related to self-preservation, such as searching for food, feeding, fighting and self-protection (MacLean, 1986). The cingulate gyrus appears to influence maternal care and nursing, separation cries between mother and offspring, and playful behavior, including wit and humor (MacLean, 1986). The septum involves feeling and expression relating to procreation. Lesions in the septal area may cause hyperresponsiveness to physical stimuli (such as touching, sounds, or temperature changes), hyperemotionality, loss of motivation, excessive sugar and water intake, and fear of unfamiliar situations (Isaacson, 1982), phenomena reported by some chemically sensitive individuals.
The hippocampus appears important for laying down new memories and thus is essential for learning (Gilman, 1982). Hippocampal lesions may cause difficulty in retaining
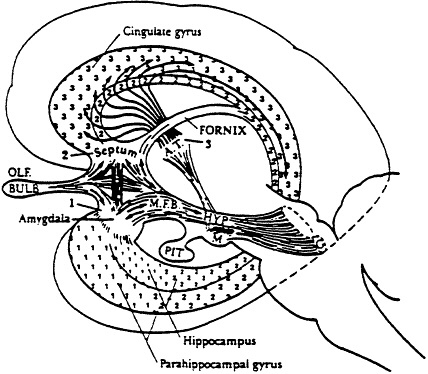
Figure 1 Three major subdivisions of the limbic system. The small numerals 1, 2, and 3 overlie, respectively, the amygdaloid, septal, and thalamocingulate divisions. The corresponding large numerals identify connecting nuclei in the amygdala, septum, and anterior thalamus. Abbreviations: AT, anterior thalamic nuclei; G, tegmental nuclei of Gudden; HYP, hypothalamus; M, mammillary bodies; MFB, reed/an forebrain bundle; PIT, pituitary, OLF, olfactory. Source: MacLean, P.D., "A Triune Concept of the Brain and Behavior," in Boag, T., and Campbell, D., The Hincks Memorial Lectures (1973), University of Toronto Press, Toronto, Ontario, p. 15.
recent memories (Isaacson, 1982). The hippocampus, at the intersection of numerous neural pathways and in a critical position to affect the transfer of information from one brain region to another, acts as an information switching center. Comment: Learning and memory decrements are a frequent consequence of exposure to toxic substances, and some researchers view the hippocampus as a prime target for such toxins (Office of Technology Assessment, 1990; Walsh, 1988). Damage to the hippocampus itself, or to nerves leading to or from it, may adversely affect the synthesis, storage, release, or inactivation of the excitatory and inhibitory amino acids that serve as neurotransmitters in this region of the brain. Toxins may disrupt the delicate balance of these amino adds, perhaps leading to the release of a flood of excitatory neurotransmitters that damage neighboring cells, a phenomenon that has been called "excitotoxicity" (Office of Technology Assessment, 1990). Remarkably small perturbations of hippocampal function may have large and long-lasting effects upon behavior and cognition (Walsh, 1988).
The most vital component of the limbic system, the hypothalamus, governs: (1) body
temperature via vasoconstriction, shivering, vasodilation, sweating, fever, and behaviors such as moving to a cooler or warmer environment or putting on or taking off clothing; (2) reproductive physiology and behavior; (3) feeding, drinking, digestive, and metabolic activities, including water balance, addictive eating leading to obesity, and complete refusal of food and water leading to death; (4) aggressive behavior, including such physical manifestations of emotion such as increased heart rate, elevated blood pressure, dry mouth, and gastrointestinal responses (Gilman, 1982).
The hypothalamus is also the locus at which sympathetic and parasympathetic nervous systems converge. Many symptoms experienced by patients with food and chemical sensitivities relate to the autonomic (sympathetic and parasympathetic) nervous systems; for example, altered smooth muscle tone produces Raynaud's phenomenon, diarrhea, constipation, and other symptoms reported by these individuals.
The hypothalamus also appears to influence anaphylaxis and other aspects of immunity (Stein, 1981). Conversely, antigens may affect electrical activity in the hypothalamus (Besedovsky, 1977).
It is important to recognize that thoughts arising in the cerebral cortex that have strong emotional overtones also can trigger hypothalamic responses and recreate the physical effects associated with intense anger, fear, and other feelings. To implement its effects, the hypothalamus not only has a direct electrical output to the nervous system but also produces its own hormones, many of which stimulate or inhibit the pituitary's production of hormones (Gilman, 1982). Of interest in this regard, is that a disproportionate number of chemically sensitive individuals seem to have been treated for thyroid hormone deficiency at some time in their lives.
Most of the neural input to the hypothalamus comes from the nearby limbic and olfactory areas (Isaacson, 1982). Lesions in the limbic region may be associated with irrational fears, feelings of strangeness or unreality, wishing to be alone, and sadness (MacLean, 1962). A feeling of being out of touch with or out of control of one's feelings and thoughts, not unlike that described by many patients with chemical sensitivity, may be perceived. Some report feeling "spacey" or that "the camera isn't on" unless they make an enormous effort to focus their attention.
Doane describes potential difficulties for patients with limbic dysfunction (Doane, 1986):
Activity controlled by the limbic system may seem largely irrational and often is not perceived within one's self in ways that are easily understood or communicated in verbal language.
The dynamic involvement of the hypothalamus and limbic system in virtually every aspect of human physiology and behavior makes injury to these structures an intriguing hypothesis to explain the development of chemical sensitivity with its diverse manifestations. Rich neural connections lie between the olfactory system and the limbic and temporal regions of the brain. Surgical or epileptic patients with damage to the limbic or medial temporal portions of the brain may experience persistent alterations in odor perception (for example, an unusual smell that characteristically precedes seizure activity) as well as learning and memory difficulties (Ryan, 1988).
Bell hypothesizes that chemically sensitive patients may have olfactory-limbic-temporal pathways that are more easily "kindled" (Bell, 1990). In other words, a small signal or insult would more readily trigger firing of nerve cells in brain regions where kindling was present.
Kindling might be enhanced by genetic endowment, prior environmental exposures, psychological stress, hormonal variations, or other factors. Unlike surgical ablation, which destroys a brain area, kindling is a kind of stimulatory lesion (Girgis, 1986).
Kindling has been described previously in the context of seizures. The amygdala, for example, which is particularly susceptible to electrical discharge following either electrical (Girgis, 1986) or chemical provocation (Bokina, 1976), is subject to long-lasting alteration when strong or repetitive stimuli are administered. Very potent or repeated stimuli, whether electrical or chemical, may permanently augment the tendency for neurons to fire in the presence of future stimuli even when challenged with much lower levels than those originally involved.
Girgis reports a decrease in acetylcholinesterase (AChE), an enzyme that breaks down the neurotransmitter acetylcholine in junctions between nerve cells, that parallels the increase in supersensitivity to stimuli (Girgis, 1986). The limbic system is especially rich in AChE, which is strongly bound to the nerve cell membranes and very stable. The AChE may play a protective role by enzymatically maintaining acetylcholine concentrations at nerve junctions within safe bounds and protecting susceptible cells in the limbic system from developing "bizarre sensitivity" (Girgis, 1986). Interestingly, physicians who treat patients with multiple chemical sensitivities have noted some of the most severe and debilitating exposures for these patients have involved organophosphate pesticides, which inhibit AChE.
Bokina found impaired speed of execution and coordination of execution and complex motor processes in humans repeatedly exposed to carbon disulfide for 10- to 15-minute intervals at subsensory levels (Bokina, 1976). Animals primed by high or chronic lower concentrations of various chemicals, such as formaldehyde and ozone, and subsequently reexposed to even lower concentrations of the same chemicals showed an increased tendency toward paroxysmal electrical discharge in the amygdala (Bokina, 1976). Bokina observed that although the chemicals he used to sensitize the animals were different in terms of their structure and physical and chemical properties, their effects upon the limbic system were remarkably similar.
Kindling could help to explain the apparent loss of adaptive capacity and spreading of sensitivities to chemically unrelated substances reported in multiple chemical sensitivity. Formerly well-tolerated low-level exposures to, for example, tobacco smoke or perfume might trigger symptoms in individuals whose limbic areas have been kindled by a prior pesticide or solvent exposure.
One intriguing aspect of the limbic system as a mechanism for multiple chemical sensitivities is the system's responsiveness to both chemical and cortical stimuli. Therefore, conscious thought processes and emotional states influence limbic activity just as chemical or physical stimuli can. The former may be under more or less conscious control of the individual, whereas the latter arc almost entirely unconscious and automatic However, conscious efforts that play into the delicate circuitry of the limbic system may be able to alter or suppress concurrent electrical activity evoked by environmental agents. Some patients with chemical sensitivities report being able to "will" their way out of a mild reaction to a food or chemical and attempt to control their symptoms in this manner. Most say such efforts do not work for their most problematic incitants. In fact, the ability to exercise any conscious effort, even that of simply getting away from the exposure, may be lost during a reaction. Monroe reported the case of a man for whom exposure to the odor of stale beer caused greatly increased electrical activity in the limbic system (amygdala and hippocampal areas). Various memories, some associated with beer, also increased electrical activity in the same region. However, simple arithmetic computations would immediately
stop such activity. Therefore, conscious thought processes could alter some electrical activity in the limbic system (Monroe, 1986).
An intriguing example of the competing effects of exposure and psychological state has been reported (Sanderson, 1989). Carbon dioxide levels greater than 5 percent in the air have been shown to induce panic attacks ("fight or Right" responses depend upon limbic activity) in patients suffering from panic disorder. The fact that patients in this study who believed that they had control over the carbon dioxide level to which they were exposed had fewer and less intense panic disorder symptoms suggests that psychological factors (that is, the illusion of control), could mitigate the biological response to an environmental stressor.
Thus, experimental evidence suggests a delicate interplay occurring in the limbic region. Conceivably, chemicals contacting olfactory nerve projections in the nose could either be transported into or relay electrical signals to the limbic region, leading to a vast array of symptoms. Likewise, thought processes and mood states trigger limbic activity or may, in some cases, interrupt preexisting limbic activity. At present, however, there is no evidence to suggest that limbic activity triggered by environmental exposures can be entirely overcome by psychological interventions.
One important ramification of a limbic hypothesis, if true, is that there may be no convenient biological marker for multiple chemical sensitivity.
Detection of chemical stimuli in the nose is not limited to the olfactory nerve, but involves the trigeminal nerve and its afferents which may also play a role in this condition. Trigeminal free nerve endings in the nose and mouth detect noxious chemicals and reflexively initiate protective responses including cessation of breathing, constriction or dilatation of the airways, reduction in heart rate and cardiac output, constriction of most blood vessels (except capillaries in the head), increased epinephrine release, changes in blood pressure and efforts to withdraw (Silver and Maruniak, 1981). This is a powerful reflex which serves an obvious protective role. Most familiar is the trigeminal reflex response to smelling salts. Apparently trigeminal responses eau occur with non-irritating stimuli. Thus far, no odor has been found that stimulates the trigeminal nerve alone or the olfactory nerve alone (Silver and Maruniak 1981) making study of either system in isolation difficult. Potentially both play a role in chemical sensitivity.
BIOCHEMICAL MECHANISMS
Rea and other ecologists who see chemically sensitive patients have noted vitamin and mineral abnormalities in many of them. Others argue that these patients are often sick, debilitated, and malnourished, and therefore, such findings are not surprising.
Individuals who have defective enzyme detoxification systems could be more susceptible to low level exposures. Conceivably, chemically sensitive individuals could have defective detoxification pathways, and be more affected while others in the same environment tolerate the same exposures without symptoms (Rogers, 1990). Rea has noted that many of his chemically sensitive patients have decreased levels of detoxifying enzymes, such as glutathione peroxidase. This possibility is particularly intriguing because such enzyme systems are inducible (that is, can be stimulated) and thus might conform to an adaptation hypothesis. Scadding and associates noted poor sulfoxidation ability in 58 of 74 patients with well-defined reactions to foods versus 67 of 200 normal controls p <0.005) (Scadding et al., 1988). Similarly, Reidenberg reported the case of a laboratory technician who developed a lupuslike disease in response to hydrazine (Reidenberg et al., 1988). She
was genetically a slow acetylator, which may might have predisposed her to developing a lupuslike disorder after sufficient exposure to the chemical A deficiency of one or more particular enzymes could help to explain why some patients are more susceptible to foods and chemicals than others. Further, damage by a toxin might compromise detoxification pathways so that other substances formerly metabolized by this pathway could not be degraded properly and thus might provoke symptoms at low exposure levels, a hypothetical basis for the spreading phenomenon.
Levine has proposed that environmental sensitivities are the result of toxic chemicals reacting with cell constituents to create free radicals (which are formed when a molecule loses an electron) (Levine, 1983). He hypothesizes that if an antioxidant molecule (such as vitamin A, C, E, or selenium) is not present nearby to supply the missing electron, then an electron may be removed from an unsaturated lipid in a cell membrane, leading to membrane damage, release of prostaglandins and other inflammatory mediators, and formation of antibodies to chemically altered tissue macromolecules.
VASCULAR MECHANISMS
Rea hypothesizes that blood vessel constriction, inflammation, or leakage in multiple organ systems could explain the bizarre combinations of symptoms in these patients. In his view, particular complaints could mirror the site and size of affected blood vessels. Spasms in large-caliber arteries, either acutely or chronically, could reduce blood supply to an organ or limb and result in dysfunction, pain, or even necrosis (Rea, 1975). Chemical injury to the fragile wails of smaller vessels, however, would be more likely to cause hemorrhage (resulting in petechiae and bruises) or edema (Rea, 1979). The walls of blood vessels contain smooth muscle. Rea notes that other tissues containing smooth muscle such as the respiratory, gastrointestinal, and genitourinary systems are frequently involved in these patients (Rea, 1977). Impaired blood vessel or altered smooth muscle function might explain the diverse and seemingly unrelated symptoms occurring in patients with multiple chemical sensitivities. In the case of either blood vessel or smooth muscle dysfunction, dearly, neurological and immune alterations or changes in membrane permability or receptors (for example, denervation-supersensitivity) could play primary roles. A vascular hypothesis might also explain why some patients experience increased pain or other symptoms at the site of an earlier injury or surgery, where blood flow may be relatively compromised.
Perhaps the mechanism for multiple chemical sensitivities is not identifiable; that is, after all avenues of biochemical and immunological inquiry have been exhausted, no single explanation for this disorder is forthcoming. The theory of substance-specific adaptation is based upon observations of the responses of patients in a deadapted state who are worked up in an environmental unit. Adaptation is only an observation at this time, not a mechanism. However, biological limits might regulate how much an organism can adapt, limits that could be highly individual and vary by orders of magnitude. Certainly adaptation occurs at all levels of biological systems, from enzyme systems to cells, tissues, organs, and even behavior (Fregly, 1969). Theoretically, a major insult or the accumulation of lower-level injuries within these systems could lead to a kind of "overload' or "saturation" effect with respect to adaptive capacity that would cause an individual to have environmental responses, which, instead of being flexible and fluid, are now fragile and overly responsive. Many patients report that even after years and in some cases decades following the onset of
REFERENCES
Ashford, N. A. and Miller, C. S. 1991. Chemical Exposures: Low Levels and High Stakes. New York: Van Nostrand Reinhold.
Bell, I. R., Clinical Ecology. 1982. Common Knowledge Press: Bolinas, California.
Bell, I. R. 1990. The biopersonality of allergies and environmental illness. Paper presented at the Eighth Annual International Symposium on Man and His Environment in Health and Disease. Dallas, Texas.
Besedovsky, H., Sorkin, E., Felix, D. and Haas, H. 1977. Hypothalamic changes during the immune response. Eur. J. Immunol. 7:323-325.
Bokina, A. I. Eksler, N. D., Semenenko, A. D., and Merkur'yeva, R. V. 1976. Investigation of the mechanism of action of atmospheric pollutants on the central nervous system and comparative evaluation of methods of study. Environ. Health. Perspect. 13:3742.
Doane, B. K. and Livingston, K. E., editors. 1986. Clinical psychiatry and the physiodynamics of the limbic system. Pp. 285-315 in The Limbic System: Functional Organization and Clinical Disorders. New York: Raven Press.
Fregly, M.J. 1969. Comments on cross-adaptation. Environ. Res. 2:435-441.
Gilman, S., and Winans, S. W. 1982. Manter and Gatz's Essentials of Clinical Neuroanatomy and Neurophysiology. Philadelphia: F. A. Davis Company.
Girgis, M. 1986. Biochemical patterns in limbic system circuitry: biochemical-electrophysiological interactions displayed by chemitrode techniques. Pp. 55-65 in Limbic System: Functional Organization and Clinical Disorders. New York: Raven Press.
Isaacson, R.L. 1982. The Limbic System. New York: Plenum Press.
Jesberger, J. A. and Richardson, J.S. 1988. Brain output dysregulation induced by olfactory bulbectomy: an approximation for the rat of major depressive disorders in humans. Intern. J. Neurosci. 38:241-265.
Kare, M. 1986. Direct pathways to the brain. Science. 163:952-953.
Kilburn, K. H. 1989. Is the human nervous system most sensitive to environmental toxins? Arch. Environ. Health. 44(6)-343-344.
Levine, S. A. and Reinhardt, J.H. 1983. Biochemical pathology initiated by free radicals, oxidant chemicals, and therapeutic drugs in the etiology of chemical hypersensitivity disease. J Orthomolecular Psychiatry. 12:166-183.
MacLean, P. D. 1967. The brain in relation to empathy and medical education. J Nerv
Ment Dis. 144(5):374-382.
Maclean, P. D., Boag, T. and Campbell, D. 1973. A Triune Concept of the Brain and Behavior: The Hincks Memorial Lectures. Pp. 6-66. Toronto, Ontario. University of Toronto Press.
Maclean, P.D. 1986. Culminating developments in the evolution of the limbic system: the thalamocingulate division. In Doane, B. K. and Livingston, K. E., editors. Pp. 1-28. Organization and Clinical Disorders. New York: Raven Press.
Mallet, O., Kate, M. R., Welt, M. and Behrman, H. 1967. Movement of glucose and sodium chloride from the oropharyngeal cavity to the brain. Nature 213(2):713-714.
Monroe, R. R. 1986. Episodic behavioral disorders and limbic ictus. Pp. 251-266 in The Limbic System: Functional Organization and Clinical Disorders. Doane, B. K., and Livingston, K. E., editors. New York: Raven Press.
Office of Technology Assessment, Congress of the United States. 1990. Identifying and Controlling Poisons of the Nervous System. Washington, D.C.: United States Government Printing Office.
Payan, D. G., McGillis, J. P. and Goetzl, E.J. 1986. Neuroimmunology. Adv. Immunology. 39:299-323.
Rea, W. J. 1977. Environmentally triggered small vessel vasculitis. Ann. Allergy. 38:245-51.
Rea, W. J. 1979. The environmental aspects of ear, nose and throat disease. Part I. Oto-Rhino-Laryngology & Allergy Digest 41(7):41-56.
Rea, W. J., Bell, I. R. and Smiley, R.E. 1975. Environmentally triggered large-vessel vasculitis. Pp. 185-198 in Allergy and Medical Treatment. Johnson, J. and Spencer, J. T., editors. Chicago: Symposia Specialists.
Reidenberg, M. 1983. Lupus erythematous-like disease due to hydrazine. Am. J. Med. 75:363-370.
Rogers, S. A. 1990. A practical approach to the person with suspected indoor air quality problems. Indoor Air. Fifth International Conference on Indoor Air Quality and Climate, Toronto, Ontario 5:345-349.
Ryan, C. M., Morrow, L. A. and Hodgson, M. 1988. Cacosmia and neurobehavioral dysfunction associated with occupational exposure to mixtures of organic solvents. Am. J. Psychiatry 145(11):1442-1445.
Sanderson, W. 1989. The influence of an illusion of control on panic attacks induced via inhalation of 55 percent carbon dioxide-enriched air. Arch. Gen. Psychiatry 46:157-162.
Scadding L. 1988. Poor sulphoxidation ability in patients with food sensitivity . Br. Meal. J. 297(6641):105-107.
Selye, H. 1946. The general adaptation syndrome and the diseases of adaptation. J. of Allergy. 17:231-247, 289-323, 358-398.
Shipley, M., 1985. Transport of molecules from nose to brain: transneuronal antegrade and retrograde labeling in the rat olfactory system by wheat germ agglutinnin-horseradish peroxidase applied to nasal epithelium. Brain Res. Bulletin. 15:129-142.
Silver, W. L. and Maruniak, J.A. 1981. Trigeminal chemoreception in the nasal and oral cavities. Chemical Senses 6(4):295-305.
Stein, M., Keller, S. and Schleifer, S. 1981. The hypothalamus and the immune response. Pp. 45-65. In Weiner, H., Hofer, M. A. and Stunkard, A. J., editors. Brain. Behavior and Bodily Disease. New York: Raven Press 45-65.
Walsh, T. J., and Emerich, D .F. 1988. The hippocampus as a common target of neurotoxic agents. Toxicology. 49:137-140.












