7
IN VITRO CELLULAR AND SUBCELLULAR END POINTS
As in the case of the other biologic end points reviewed here, there are few reports of in vitro end points related to electromagnetic fields (EMFs) with the same combination of frequencies and waveforms as encountered in GWEN. Therefore, a general overview of results of irradiation at frequencies up to 10 GHz must be considered, but with some attention to extension of results to possible GWEN-related health effects. According to the data presented in Chapter 3 and some additional plane-wave approximations, relevant exposure and dose metrics predicted for GWEN site boundaries are listed in Table 7-1 :
TABLE 7-1 GWEN Exposure and Dose Metrics at Site Boundary
|
Exposure or Dose Metric |
Low Frequency |
Ultra-High Frequency |
|
Maximum incident power density, W/m2 |
7a |
0.01 |
|
Maximum electric field in human, V/m |
2.2 |
1a |
|
Maximum magnetic induction in human, µT |
0.07 |
0.007a |
|
Maximum specific absorption rate (SAR), W/kg |
0.001a |
0.0005 |
|
Maximum induced current density, A/m2 |
0.7 |
1a |
|
aCalculated from plane-wave approximation. |
||
A practical problem in the literature is that typically only one or a few dose metrics associated with biologic exposures are given, other measures being omitted inadvertently, for convenience, or in the belief that they are not biologically relevant. The omitted dose metric is often large; it is then difficult to compare exposure conditions associated with reported bioeffects in relation to the GWEN levels shown in Table 7-1.
Three papers by the Brighton1,2,3 group report effects of small in vitro exposures to 60-kHz continuous-wave electric fields, somewhat close to GWEN low-frequencies; these studies are described later. Otherwise, a review of the available in vitro data makes it readily apparent that almost all experiments at frequencies above GWEN
frequencies involve exposures at appreciably larger levels than shown in Table 7-1, whereas some experiments at extremely low frequency (ELF) involve exposures at or below the levels shown in Table 7-1. With very limited data at low- or ultra-high-frequencies, results with low-level ELF fields might seem applicable to the GWEN situation, but there are several pitfalls, as follows:
-
At ELF, the intracellular electric fields and current densities are usually very small, because of poor coupling with external fields. Thus, an external field of hundreds of kilowatts per meter would be required to produce the internal fields listed in Table 7-1.
-
On the basis of such endpoints as field perception threshold, which have been studied over a wide range of frequencies, biological sensitivity at GWEN frequencies would be expected to be much less (lower by a factor as large as 100) than at ELF. On the basis of biologically equivalent field levels, the numbers in Table 7-1 would be scaled down by the appropriate factor; unfortunately, this factor is unknown and speculative for many endpoints.
-
There might be qualitative differences in biological response between ELF and low-frequency (LF) or ultra-high frequency (UHF).
-
SAR is not considered to be an appropriate dose metric at ELF and is almost never reported in ELF studies.
Although population exposure to GWEN fields will produce no detectable tissue heating, most of the in vitro literature (excluding ELF exposure) deals with irradiation at SAR values higher than in the GWEN setting. That raises the question of extrapolation to low EMF levels and the specter of thermal artifacts. Some of the earlier reports of EMF biologic effects did not adequately describe temperature measurement and regulation and must be discounted. However, the temperature of cells exposed in vitro can be readily controlled by controlling the temperature of the surrounding culture medium, as reported by numerous investigators. This conclusion follows from the fact that it is impossible in aqueous media to maintain appreciable constant temperature gradients across cellular dimensions,4 and, except for extremely high-power radiation in submicrosecond pulses, transitory heating from pulsed irradiation is prevented by the brief (much less than 1µsec) thermal relaxation time for cells in aqueous media.5 Thus, reports with adequate descriptions of temperature measurements of an irradiated bulk cellular medium are acceptable on the grounds of thermometry, but extrapolation to GWEN of results obtained at higher field levels, different frequencies, and different waveforms remains open to question.
Comprehensive and recent reviews of the in vitro literature are available6,7,8,9,10,11,12 Up to a frequency of 10 GHz, most reported biological effects are in the categories shown in Table 7-2 and are considered separately in pages to follow (mutagenic effects, chromosomal aberrations, and neoplastic transformation). Analysis and numerous references through 1988 for the topics in the table are available in the cited reviews.
The amount of recent published research on EMF-induced cellular and subcellular effects appears to be small relative to the growing general concern for possible EMF hazards. Livingston et al.13 recently published the results of a battery of cytogenetic and cytotoxic tests that used human lymphocytes and Chinese hamster ovary fibroblasts exposed to 60-Hz electric current densities ranging from 0.03 to 30 A/m2 and a magnetic field of 220 µT; the results were negative. Cleary et al.14,15 reported positive results of continuous 27- and 2,450-MHz radiation in inducing cell proliferation with SAR values more than 10,000 times those of GWEN. Another pair of papers16,17 presented negative results of searches to demonstrate an ion cyclotron resonance effect. However, Ross18 reported that fibroblast proliferation changed after exposure to mixed static and sinusoidally time-varying magnetic fields in the gauss range, and interpreted these results as consistent with a calcium ion cyclotron resonance mechanism. Lyle et al.19 reported that exposure to combined static and time-varying magnetic fields changed calcium uptake in normal and leukemic lymphocytes--further support of the idea of ion cyclotron resonance. Two studies with 60-Hz, 430-V/m electric-field exposure (more than 100 times the magnitude of GWEN fields) investigated changes in growth and hydrogen ion excretion in plant-root preparations.20,21
Microwave-induced changes in channel formation in an artificial lipid bilayer system have been reported,22 but at field amplitudes and SAR values 100,000 times those relevant to GWEN. At the macromolecular level, alterations in ATPase activity after irradiation with microwaves (SAR, 20 W/kg) have recently been described.23 Effects of exposure on DNA repair were recently investigated. Treatment of cells with 5 Gy of 60Co radiation was followed by exposure to 60-Hz fields (magnetic field of 1 mT, electric field of 1 or 20 V/m, or combined fields of 0.2 V/m and 0.05 mT, 6 V/m and 0.6 mT, or 20 V/m and 1 mT) or sham exposure24 None of the exposures affected the repair of single-strand breaks. At the other extreme of amplitude, Fitzsimmons et al.25 elicited bone tissue proliferation and mitogenic activity with an ELF electric field of approximately 10-5 V/m. Finally, three reports by Cossarizza et al.26,27,28 describe increases in cell proliferation and interleukin-2 activity and decreases in DNA repair in human lymphocytes exposed to pulsed fields similar to those used clinically for low-level electric facilitation of bone repair.
TABLE 7-2 Categories of In Vitro Experiments
|
Cellular and Subcellular System |
Investigated End Points |
|
Macromolecular preparations |
Electromagnetic absorption in DNA, enzyme activity. |
|
Prokaryotes |
Growth rate, metabolism, enzyme activity. |
|
Membrane |
Cation flux, membrane fluidity, protein shedding, electrophoretic mobility. |
|
Hematopoietic and Immunological |
Cell growth, mitogenic response, lymphoblastoid transformation, T-lymphocyte activity, lymphocyte capping, protein synthesis, macrophage activity, levels of DNA and RNA. |
|
Neurons |
Firing rate, membrane potential, metabolic rate. |
|
Salivary gland cells |
RNA transcription, polypeptide synthesis. |
|
Bone cells |
Protein and DNA synthesis, Ca2+ and cyclic AMP concentrations, collagenproduction, alkaline phosphatase and mitogen activity, cell proliferation. |
|
Biochemistry |
Changes in Ca2+ efflux, ornithine decarboxylase, PKC. |
Several groups of results will be considered in more detail below, selected according to the following criteria:
-
Some of the reported dose metrics are near or below maximum GWEN values.
-
The results come from a reasonably large and consistent body of evidence.
-
The end points have perceived relevance to health effects.
BONE HEALING
In connection with in vivo experiments to stimulate bone healing in test animals, Brighton et al.1,2,3 measured in vitro responses of cultured bovine chondrocytes and rat bone cells to 60-kHz continuous sine-wave electric fields administered by capacitive coupling for various durations between 5 min and 48 h. In some conditions, modest but statistically significant field-associated changes in isotope uptake and in cyclic AMP (cAMP) concentration were observed; they depended on such factors as field amplitude and serum concentration of the culture medium. Those results are of interest in relation to GWEN, because the induced voltages and currents that yielded positive results ranged down to less than 1 V/m--lower than the highest levels of GWEN LF fields shown in Table 7-1.
As reviewed by Bassett29,30 and Tenforde,10 the use of small electromagnetic pulses to treat bone fractures and pseudoarthroses has developed into a refined clinical approach based on the delivery of well-defined electric fields to bone. That used to be accomplished via implantation of electrodes to impose the specified electric field; but pulsed external coils are now used to induce electric currents in bone by generating rapidly time-varying magnetic fields. Large magnetic fields (up to 400 µT) are also produced, but these are ignored and presumably are considered to be without biomedical significance, in light of the historical and theoretical explanation of the therapeutic action as a purely bioelectrical phenomenon. On these grounds, the relevant dose and exposure metrics are usually small, with current densities of 2-20 mA/m2 (Tenforde)10 and induced electric fields near 0.2 V/m. Those values are lower than the maximum values at GWEN site boundaries (Table 7-1), even if we apply a bioeffective scaling factor that reduces the listed LF and UHF current densities and voltages. A typical electric-field waveform used for bone-fracture treatment is shown in Figure 7-1. Similar fields have been used in in vitro investigations with bone-cell cultures,31,32 other cell types, including human lymphocytes,27,28 and tumor cells.33 Similar pulsed fields have also been found to alter RNA transcription34
MUTAGENIC EFFECTS
Variations in reported frequency, intensity, field orientation, period of exposure, and whether the field is continuous or pulsed make comparisons of data difficult. However, results of at least 20 studies of induction of gene mutations in bacteria, yeast, plants, insects, and mammalian cells in culture have been negative.35 For example, a
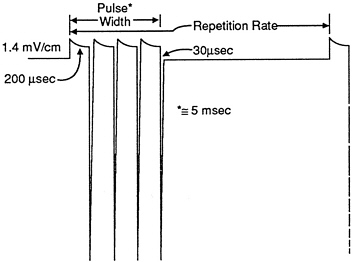
FIGURE 7-1. Electric-field waveform used in bone-healing therapy.
Source: C.A.L. Bassett, 1978, ref. 29.
recent study of forward mutations at the thymidine kinase locus in L5178 mouse leukemic cells showed no statistically significant increase in induced mutation frequency due to exposure to 2.45-GHz pulsed waves (500 or 600 W).36 Several reports of positive results have, however, appeared. A single positive study in salmonella treated with 2.45-GHz fields showed increased induction of mutations, probably because of heating.37 Studies in plants have also suggested induction of embryolethal and morphological mutants treated with 200-MHz radiowaves.38 The most reasonable conclusion to draw from the full array of studies is that direct DNA damage does not occur.
CYTOGENETIC EFFECTS
Studies of chromosomal changes in mammalian cells, although producing predominantly negative results, have sometimes produced conflicting results. Two studies have indicated chromosomal breaks after exposure to 2.45-GHz microwaves.39,40 In one study, incident power varied between 25 and 200 W;39 positive results were seen only when assays were performed without temperature control. In the other study, exposure of two kangaroo-rat cell lines maintained at 37°C with exposure to 2.45-GHz radiation demonstrated increases in chromosome aberrations in one line.40 No increase in sister chromatid exchange (SCE) was seen after treatment of Chinese hamster ovary cells with 2.45 GHz.41
Most studies of chromosomal aberrations induced in mice and hamsters with in vivo exposure, especially those at extremely low frequencies, have had negative results. For example, a recent study of 60-Hz fields with current densities of 3-3,000 µA/cm2 and a fixed magnetic field of 0.22 mT produced negative results13 However, significant increases were found in chromosomal aberrations in Ehrlich ascites tumor cells exposed in the peritoneal cavities of mice for 14 d to horizontal electric fields of 80-160 V/cm (60 Hz), compared with controls.42 Vertical electric fields produced no difference from controls, and no effect was seen with a 4-15 wk exposure.43
Female CD-1 mice treated with 2.45-GHz microwaves at 200 W/m2 did not differ from controls in SCE frequency in bone marrow cells44 Other studies in mice also failed to demonstrate significant differences in chromosomal aberrations in spermatocytes45 or bone marrow46 after treatment with 2.45-GHz microwaves; but positive results have been reported in rat bone marrow47 and Chinese hamster corneal epithelium.48
Studies on human cells exposed in vitro to low frequencies have also produced conflicting results. No increase in chromosomal breaks, SCE, or chromosomal aberrations were seen in several studies.13,49,50,51,52 For example, one study used 60-Hz fields with variable electric current densities of 3 - 3,000 µA/cm2 and a fixed magnetic field of 0.22mT.13 No changes in SCE, micronuclei, clonogenicity, or cell-cycle kinetics were observed. No increase in micronuclei after exposure to pulsed magnetic fields (50 Hz, 2.5 mT) was observed in human lymphocytes; 53 nor did the exposure influence the extent of micronuclei induction by mitomycin C. However, some studies have reported positive results. No increase in aberrations were observed in human lymphocytes following exposure of whole blood to a 50-Hz electric field, but exposure to 10-µsec long spark-discharge pulses did cause a significant increase in chromosomal breaks.54 A recent study of pulsing 50-Hz fields (1.05 mT) demonstrated a suppression of mitotic activity and higher incidence of chromosomal aberrations after 24, 48, and 72 h of treatment.55 A reduction in the cell proliferation index and an increase in SCE rate were seen only after 72 h of exposure.
Treatment of human lymphocytes with radiofrequencies has produced conflicting results, one study showing increases in chromosomal stickiness, breaks, and dicentrics,56 and another, using a magnetic resonance imaging device, showing no increase in chromosomal aberrations or SCE.57
In summary, studies of chromosomal effects of EMFs have produced conflicting results. Most studies at extremely low frequencies have been negative, although exceptions can be found. At radiofrequencies, some positive studies of chromosomal aberrations have been reported, but studies of SCE have been predominantly negative.
CELL TRANSFORMATION
Exposure to 2.45-GHz radiation at 120 pulses/sec followed by treatment with the tumor promoter 12-O-tetradecanoylphorbol-13-acetate (TPA) induced the transformation of mouse C3H/10T1/2 cells.58 TPA treatment of cells previously irradiated with microwaves and x rays led to a statistically significant 3.5- to 1.6-fold increase in transformation, compared with the transformation frequency of cells previously treated with x rays alone at 1.5 and 4.5 Gy, respectively. A later study59 showed that treatment with microwaves followed by TPA significantly increased the transformation frequency. Additional studies showed a dose-response relationship between field exposures (SAR values of 0.1, 1, and 4.4 W/kg) and transformation frequency.60 EMFs alone had no effect on cell survival or induction of transformation. Transformation by fields plus TPA was as great as that produced by 1.5 Gy of x rays. The frequency of transformation was additive when doses of x rays were given as a cocarcinogen.
In contrast with most other in vitro studies on the genotoxicity of EMFs, these results suggest that EMFs may act as an initiator. A long-standing criticism of the 10T1/2 system is that it uses cells that are immortalized, and might already have genetic changes. Hence, it is not strictly comparable to an animal model of initiation and promotion.
EFFECTS ON TRANSCRIPTION
Salivary gland chromosomes of the dipteran Sciara coprophilia have been used to study effects of weak pulsed EMFs on transcription34 Normal transcription patterns during cell differentiation are known from transcription autoradiography and can be correlated with chromosomal structure by banding and puff formation in the four polytene chromosomes. Transcription was measured in three ways: nascent RNA chains attached to specific chromosomal regions were identified with conventional autoradiography after incorporation of [3H]uridine; regions sensitive to DNase I, and therefore transcriptionally active, were examined with nick translation directly on the DNA of the cytological preparations; and RNAs of various size classes were isolated with sucrose density-gradient centrifugation. In the presence of a repetitive single pulse signal (single 380-µsec pulses used clinically to yield 0.15 V/m in bone, repeated at 72 Hz), marked and specific increases in RNA transcription in most of the bands and interbands of the chromosomes were seen at 15 and 45 min. At 30 min, transcription was approximately equal to control values. With the repetitive pulse train signal (5-msec pulse trains of 200-µsec pulses repeated at 15 Hz), increases in RNA transcription were seen only after 45 min of exposure and were lower than with the single pulse. Sucrose gradient analysis of RNA indicated a 4-fold increase in total RNA and an 11-fold increase in mRNA with the single pulse.
Those results were later extended to show that sinusoidal fields with the same frequency as the repetition frequency of the fast single-pulse signals also induce increases in transcriptional activity.61 Sine waves of 72, 222, or 4,400 Hz (measured magnetic fields of 1.15, 0.37, or 0.018 mT) were used. At higher frequencies, the same general pattern of induced transcription was seen, but it was not as dramatic as that observed at 72 Hz. Analysis of the grain-count distribution over the X chromosome of Sciara showed augmented uptake of [3H]uridine into RNA after short exposures of the cells to three signals: single repetitive pulse at 72 Hz (15 mV and 3.5 mT), repetitive pulse bursts at 15 Hz (14.5 mV and 1.9 mT), and sine waves at 72 Hz (0.8 mV and 1.1 mT).62 The quoted voltages refer to coil-probe readings with calculated electric fields in cortical bone not exceeding 0.15 V/m. The electric fields were induced by magnetic fields at the quoted strengths. Transcription was augmented in previously active loci, as well as in chromosomal regions that were not detectably active in control cells.
Polypeptide synthesis after exposure to fields of various waveforms and frequencies was studied with two-dimensional gel electrophoresis63 The polypeptide patterns were qualitatively and quantitatively different for each type of exposure and different from those of control and heat-shocked cells. Individual proteins were not identified.
The same group of investigators has demonstrated increases in selected RNA transcripts in cultured human cells (HL60).64 RNA with homology to β-actin, histone H2B, and v-myc increased 20 min after exposure to five ELF electromagnetic signals. Continuous sine waves at 60 and 72 Hz (coil-probe reading, 0.8 mV; 1.1 mT) produced the greatest increase, followed by the pulse train (burst-repetition rate, 15 Hz) and a single pulse (72 Hz, 15 mV, 3.5 mT). A followup study of sinusoidal fields at 15-150 Hz (0.5 mV, 0.2-2.3 mT) also demonstrated an increase in c-myc and histone H2B transcripts.65 The maximum increase (4-fold over control) occurred at 45 Hz.
Another group has also investigated changes in transcription by measuring [3H]-uridine incorporation into HL60 cells after exposure to a sine-wave ELF field at 60 Hz.66 Cells were exposed to a uniform magnetic field, usually at 1 mT, with different samples simultaneously exposed to induced electric fields of 0 - 4.5 mV/m. There was no effect in cells exposed to a null electric field, so the magnetic field was claimed to play no role in the transcriptional changes. A transient increase in transcription rates with a maximum of 50 or 60% enhancement was seen after 30-120 min of exposure to very small electric fields; the rate declined to basal values by 18 h.
A mathematical linear, multistep reaction model has been developed to demonstrate that a transient response to fields can lead to apparent amplitude windows.67 The model assumes that switching on the fields causes a sudden increase in an intermediate reaction rate in the synthesis of RNA and that the change in reaction rate increases with field strength.
One study in bacteria demonstrated effects on the lac operon in E. coli.68 The β-galactosidase gene is under control of the lac operon and normally is repressed. Changes in repressor protein lead to changes in the rate of synthesis of β-galactosidase. Bacteria exposed to a 50-Hz square-wave magnetic field of 0 - 0.7 mT displayed a complex response with increasing field strength. A decrease was seen at fields up to 0.3 mT, then an increase up to 0.54 mT, then another decrease at field levels greater than 0.56 mT. Results also depended on cell concentration. Strengths of induced electric fields were not reported.
Those studies demonstrate biological effects, but not health consequences. Changes in transcription can result from normal stimulation of cell growth. In most of the studies, the electric-field strength was lower than the corresponding GWEN values (Table 7-1), but the magnetic fields were much larger.
TUMOR PROMOTION
The preponderance of negative over positive results with respect to direct genotoxic or clastogenic effect of EMFs has led many investigators to believe that these fields can be carcinogenic only in the sense of tumor promotion. The idea of two-stage carcinogenesis and tumor promotion was developed originally in animal models, and it assumes that a subthreshold exposure to a carcinogen (initiation) is followed by continuous application of a nongenotoxic irritant (promotion). Although tumor promoters are not carcinogenic themselves and are not genotoxic agents, they have a proliferative effect that allows the expansion and selection of initiated (genetically altered) cells69,70 It is now accepted that many promoters act at the level of membrane signal transduction, altering the normal homeostatic mechanisms of the cell. It is also accepted that, to be called a tumor promoter, a substance must induce proliferation in the target cell population71
Three lines of evidence have been used to support a putative promotional effect of EMFs on tumor formation: field-induced changes in Ca2+, induction of ornithine decarboxylase, and alterations in cellular kinases. Because the reported evidence involves relatively small fields with respect to GWEN and has generated concern for hazards, a critique of methods and interpretations is appropriate.
Calcium Homeostasis
It is generally accepted that Ca2+ plays a major role in signal-transduction mechanisms in the cell membrane. Increases in free Ca2+ in the cytoplasm have been shown after exposure of cell cultures to epidermal growth factor (EGF),72 and it is known that calcium-dependent processes, such as the generation of cAMP, alter the binding of some hormones to their receptors.73 From the point of view of promotion,
the most relevant aspect of calcium-dependent processes is the phospholipase C/protein kinase C (PKC) cascade.74 The activation of phospholipase C results in an increase in cytoplasmic free Ca2+ as a result of the release of inositol phosphate from the cell membrane. PKC is activated by diacylglycerol (also released by the action of phospholipase C) and calcium. We know that TPA, the classical skin-tumor promoter, binds and activates PKC, and it is believed that tumor promotion is mediated through PKC activation. It has been speculated that modulation of intracellular Ca2+ by EMF has an effect on signal transduction mechanisms; therefore, this modulation could be a mechanism of tumor promotion.
Since the early work of Bawin et al.,75,76 at least two laboratories have published reports showing that EMFs affect the efflux of Ca2+ from brain tissue. The basic assay used in most of the studies76,77 is carried out on entire forebrain removed from the animal and pre-incubated in a physiologic solution containing radioactive calcium. After being washed in a nonradioactive solution to eliminate loosely associated radioactive calcium, the tissues are exposed to the EMF, and the radioactive calcium released by the tissue is determined. Bawin et al.75 showed a modestly increased efflux of calcium after exposure to 147-MHz RF fields modulated between 6 and 25 Hz, but not the unmodulated carrier wave (147 MHz) alone. The incident power density was 10-20 W/m2. The resulting tissue electric field of 1-10 V/m is too low to directly influence Ca2+ binding by a thermal mechanism.78 The maximum effect occurred at a modulation frequency of 16 Hz. The same laboratory reported an apparently contradictory study in which a decrease in Ca2+ efflux, rather than an increase, occurred when electric fields without an RF carrier were used; this study also contradicted the study of Blackman et al.,77 which showed that fields without an RF carrier produce an increase in Ca2+ efflux.
The work of Bawin et al.75,76 and the data of Blackman et al.77,79 indicate that the effect of EMFs on Ca2+ efflux occurs only at specific amplitudes and frequencies. Unlike most chemical- or radiation-induced biological effects in which a dose-response relation is normally shown (i.e., increasing dose results in an increase in effect until the effect eventually reaches a plateau), EMFs produced a maximum effect at specific windows (e.g., 5-10 V/m in air and 30-60 V/m in air), with insignificant trends at lower or higher intensities. Frequency windows were also described. RF waves appear not to affect Ca2+ efflux, except when they are modulated between 6 and 25 Hz. Therefore, the essential component seems to be an ELF field centered around 16 Hz. (Effects have also been reported at 45, 60, 75, 90, and 105 Hz, and it has been suggested that the results are related to the fact that these frequencies are multiples or harmonics, of 15 Hz.) The electric field in cell-culture media or tissue was much less than 1 V/m in the ELF experiments.
The effect of Ca2+ efflux seems to be largely restricted to neural tissue. In the Bawin and Adey study,76 no effect was observed in muscle subjected to the same experimental conditions as the brain.73,79 However, a positive effect was obtained by
Lin-Liu and Adey80 in isolated synaptosomes, and this further suggests the tissue specificity of the phenomenon. In contrast, the positive results with efflux in bone tissue exposed in vitro to small electromagnetic pulses must be recalled.31,32
More recent studies showed similar results in tissue-culture systems. Dutta et al.81 reported enhanced efflux of ions in cultured neuroblastoma cells exposed to 915 or 147 MHz,82 both modulated at 16 Hz. SAR values in those experiments were as low as 0.0005 W/Kg; positive results were reported at 0.05 and 0.005 W/Kg.
Two major subjects warrant discussion regarding these studies: the experimental procedure itself and the possible relevance of the findings to tumor promotion. The studies have been criticized for their experimental model.83 Almost all have been performed with the experimental approach described by Bawin and Adey in 1976,76 which uses isolated whole-brain hemispheres. It has been shown that electrical activity disappears within minutes of tissue removal from the animal and that the amount of available O2 is drastically reduced in the first 10 min. Although Blackman et al.77,79 have argued that the tissue is metabolically active, it is difficult to assess the extent to which residual metabolic activity is comparable with physiological cell metabolism and the extent to which normal cell structures are preserved. It is well known that Ca2+ movements take place and are part of the physiopathology of necrosis caused by anoxia. As to the relevance of the studies for tumor promotion, although they might explain some of the behavioral or neurophysiological effects of EMFs, the implications for tumor promotion are speculative. As discussed earlier, it is accepted that tumor promoters induce proliferation in the target tissue. However, in the experiments on modulation of Ca2+ efflux, it has not been shown that the effects are related to perturbation in any homeostatic mechanism. Furthermore, no experimental evidence links the changes in Ca2+ movement with cell proliferation. In summary, although a link between EMFs and signal transduction by alteration of Ca2+ levels cannot be ruled out, it is premature to infer that such mechanisms are involved in tumor promotion.
Ornithine decarboxylase
Ornithine decarboxylase (ODC) is the rate-limiting enzyme in the polyamine biosynthetic pathway. Although the function of polyamines has not been completely elucidated, it is known that they play a role in cell proliferation. Induction of proliferation in quiescent cell populations by hormones, growth factors, or tumor promoters is normally preceded by ODC induction and increases in cellular polyamines84 The induction of ODC by most tumor promoters has been considered a necessary, although not sufficient, step in tumor promotion, so ODC has been used as an early indicator of tumor promotional effects69,70 Some studies have suggested that EMFs can induce ODC in tissue-culture cells. Byus et al.85 studied the effect of small 60-Hz electric fields on ODC activities of three established cell lines (human CEM lymphoma cells, mouse P3 myeloma cells, and Reuber H35 hepatoma cells).
Cells were exposed for 1 h to an electric field of 1 V/m. The EMF produced a transient increase in ODC activity in all cell lines, from about 40% in Reuber cells to about 400% in CEM cells. The peak of activity was different in the three cell types, but all the cells returned to normal values 3 h after exposure. In the same study, Reuber H35 cells were exposed to fields of varied strength (0.01-1 V/m), and an effect on ODC was seen only immediately after the termination of exposure and only at the highest and lowest exposures. Exposures for longer periods either had no effect or else decreased ODC activity, depending on the duration of exposure.
In a second study,86 the same group exposed three cell lines (Reuber H35, CHO, and 294T melanoma) to a 16-Hz-modulated microwave field (450 MHz, 10 W/m2). The cultures were measured during the logarithmic phase growth, as opposed to the quiescent phase in the previous study. ODC activity increased significantly (by up to 50%) in all three lines. The effect of different modulation frequencies on ODC activity was also investigated in Reuber H35 hepatoma cells. The maximum effect was produced with modulation at 16 Hz; frequencies of 60 and 100 Hz failed to produce any effect. Interaction of EMFs and TPA was another subject of the study. The experiments suggested a synergistic effect of TPA and EMFs on ODC induction.
Although those authors have shown that ELF electric fields and 16-Hz amplitudemodulated RF fields have some biological effect on ODC activity, they have not shown ODC induction by these fields. Enzymatic activity can be regulated at transcriptional, translational, and post-translational levels. The modest reported changes in enzymatic activity (compared with those seen with growth factors and tumor promoters) and the kinetics of the effect of EMFs (very different from the kinetics of induction by tumor promoters) argue against the possibility that these changes represent true enzymatic induction. No known studies have attempted to show at what degree of cellular metabolic regulation the fields interact. All these studies were performed in transformed cell lines, and there are no reports of similar EMF effects on animals or even on nontransformed cells in culture. Finally, in all cases where tumor promoters have been shown to induce ODC, the induction of the enzyme has been coupled with induction of cell proliferation; this has not been the case in the studies on EMF effects.
Protein Kinase C
In a single study published in 1984, Byus et al.87 studied the effect of 450-MHz microwaves modulated at 3-100 Hz on protein kinases in cultures of human tonsil lymphocytes. The incident power density was 10 W/m2. The RF carrier field did not produce any effect on cAMP-dependent kinase activity, but a decrease of about 50% in the cAMP-independent kinase activity, PKC, was reported for fields modulated at 16 Hz. The decrease in enzymatic activity was transient, and activity returned to normal within 1 h, even in the presence of continued field exposure. Other modulation
frequencies produced less marked results, and unmodulated 450-MHz fields did not produce any detectable changes in PKC.
That study has been considered of special interest, because PKC has been shown to mediate many of the actions of TPA and other tumor promoters (e.g., teleocidine). The putative modulation (down-regulation) of a protein kinase by EMFs has been used to support a putative tumor-promotional activity of EMFs. However, although the study was reported 8 yr ago, there has been no confirmation that the protein kinase involved was actually PKC. It is surprising that, in spite of the fact that PKC modulation by EMFs would constitute a strong argument in favor of tumor promotion, no attempt has been made to use more novel approaches to confirm the results independently.
CONCLUSIONS
There are no data on cellular and subcellular end points after exposure to EMFs matching those of GWEN in frequency, waveform, or duty cycle. We are forced to state, in the absence of reports on cellular response to fields even remotely resembling GWEN fields, that firm conclusions cannot be drawn. In a search for a match of experimental exposures with maximum exposures expected at a GWEN station site boundary, we found some experiments that yielded positive results. However, the exposures used are much higher than expected population exposures associated with GWEN fields, and the experimental end points involved have no demonstrated connection with health risk. Furthermore, the extremely low duty cycle of GWEN must be acknowledged, and the precautions regarding interfrequency comparisons must be applied.
An interesting perspective for risk assessment is provided by the observation of beneficial results of low-level magnetic pulses in bone healing. Those pulses are regarded by practitioners and patients as therapeutic, and the possible adverse effects on exposed tissues are not considered to be important in clinical practice.
REFERENCES
1. Brighton, C. T., A. S. Unger, and J. L. Stambough. 1984. In vivo growth of bovine articular cartilage chondrocytes in various capacitively coupled electrical fields. J. of Ortho. Res. 2 : 15-22.
2. Brighton, C. T., and P. F. Townsend. 1988. Increased cAMP production after short-term capacitively coupled stimulation in bovine growth plate chondrocytes. J. of Ortho. Res. 6 : 552-558.
3. Brighton, C. T., and W. P. McCluskey. 1988. Response of cultured bone cells to a capacitively coupled electric field: inhibition of cAMP response to parathyroid hormone. J. of Othro. Res. 6 : 567-571.
4. Love, L. A., and F. W. Kremkau. 1980. Intracellular temperature distribution produced by ultrasound. J. Acoust. Soc. Am. 67 : 1045-1051.
5. Friend, A. W., Jr., S. L. Gartner, K. R. Foster, and H. Howe, Jr. 1981. The effects of high power microwave pulses on red blood cells and the relationship to transmembrane thermal gradients. IEEE Trans. Microwave Theory and Techniques MTT 29 : 1271-1276.
6. Cleary, S. F. 1989. Biological effects of radiofrequency radiation: an overview. Pp. 59-79 in Electromagnetic Biointeraction. Mechanisms, Safety Standards, Protection Guides, G. Franceshetti, O. P. Gandhi, and M. Grandolfo, eds. New York and London : Plenum Press.
7. Cleary, S. F. 1990. Cellular effects of radiofrequency electromagnetic fields. Pp. 339-366 in Biological Effects Medical Applications of Electromagnetic Energy , O. P. Gandhi, ed. Englewood Cliffs, NJ : Prentice Hall.
8. Anderson, L. E. 1990. Biological effects of extremely low frequency and 60 Hz fields. Chapter 9 in Biological Effects and Medical Appliances of Electromagnetic Energy, O.P. Gandhi, ed. Englewood Cliffs, NJ : Prentice Hall.
9. Miller, M. W. 1986. Extremely low frequency (ELF) electrical fields: experimental work on biological effects. Pp. 139-168 in CRC Handbook of Biological Effects of Electromagnetic Fields, C. Polk and E. Postow, eds. Boca Raton, FL : CRC Press.
10. Tenforde, T. S. 1986. Interaction of ELF magnetic fields with living matter. Pp. 197-225 in CRC Handbook of Biological Effects of Electromagnetic Fields, C. Polk and E. Postow, eds. Boca Raton, FL : CRC Press.
11. Michaelson, S. M. 1986. Interaction of nonmodulated fields with living matter. Pp. 339-423 in CRC Handbook of Biological Effects of Electromagnetic Fields, C. Polk and E. Postow, eds. Boca Raton, FL : CRC Press.
12. Postow, E., and M. L. Swicord. Modulated fields and “window” effects. 1986. Pp. 425-460 in CRC Handbook of Biological Effects of Electromagnetic Fields, C. Polk and E. Postow, eds. Boca Raton, FL : CRC Press.
13. Livingston, G. K., K. L. Witt, O. P. Gandhi, I. Chatterjee, and J. L. Roti Roti. 1991. Reproductive integrity of mammalian cells exposed to power frequency electromagnetic fields. Environ. Mol. Mutag. 17 : 49-58.
14. Cleary, S. F., L.-M. Liu, and R. E. Merchant. 1990. In vitro lymphocyte proliferation induced by radio-frequency electromagnetic radiation under isothermal conditions. Bioelectromagnetics 11 : 47-56.
15. Cleary, S. F., L.-M. Liu, and R. E. Merchant. 1990. Glioma proliferation modulated in vitro by isothermal radiofrequency radiation exposure. Radiat. Res. 121 : 38-45.
16. Liboff, A. R., and W. C. Parkinson. 1991. Search for ion-cyclotron resonance in an Na+-transport system. Bioelectromagnetics 12 : 77-84.
17. Parkinson, W. C., and C. T. Hanks. 1989. Search for cyclotron resonance in cells in vitro. Bioelectromagnetics 10 : 129-146.
18. Ross, S. M. 1990. Combined DC and ELF magnetic fields can alter cell proliferation. Bioelectromagnetics 11 : 27-36.
19. Lyle, D.B., X. Wang, R. D. Ayotte, A. R. Sheppard, and W. R. Adey. 1991. Calcium uptake by normal and leukemic T-lymphocytes exposed to lowfrequency magnetic fields. Bioelectromagnetics 12 : 145-156.
20. Azadniv, M., M. W. Miller, A. A. Brayman, and C. Cox. 1990. Repetitive pulsed-train “off” duration mitigates reductions in root growth rates of Pisum sativim L. induced by 60-Hz electric field. Radiat. Res. 124 : 62-65.
21. Brayman, A. A., and M. W. Miller. 1990. 60-Hz electric field exposure inhibits net apparent H+-ion excretion from excised roots of Zea mays L. Radiat. Res. 123 : 22-31.
22. Sandblom, J., and S. Theander. 1991. The effect of microwave radiation on the stability and formation of gramicidin-A channels in lipid bilayer membranes. Bioelectromagnetics 12 : 9-20.
23. Brown, H. D., and S. K. Chattopadhyay. 1991. Ouabain inhibition of kidney ATPase by 9.14 GHz radiation. Bioelectromagnetics 12 : 137-144.
24. Frazier, M. E., J. A. Reese, J. E. Morris, R. F. Jostes, and D. L. Miller. 1990. Exposure of mammalian cells to 60-Hz magnetic or electric fields: analysis of DNA repair of induced, single-strand breaks. Bioelectromagnetics 11(3) : 229-234.
25. Fitzsimmons, R. J., J. R. Farley, W. R. Adey, and D. J. Baylink. 1989. Frequency dependence of increased cell proliferation in vitro in exposures to a low-amplitude, low-frequency electric field: evidence for dependence on increased mitogen activity released into culture medium. J. Cellular Physiology 139 : 586-591.
26. Cossarizza, A., D. Monti, F. Bersani, R. Paganelli, G. Montagnani, R. Cadossi, M. Cantini, and C. Franceschi. 1989. Extremely low frequency pulsed electromagnetic fields increase interleukin-2 (IL-2) utilization and IL-2 receptor expression in mitogen-stimulated human lymphocytes from old subjects. FEBS 248 : 141-144.
27. Cossarizza, A., D. Monti, F. Bersani, R. Cadossi, M. Cantini, and C. Franceschi. 1989. Extremely low frequency pulsed electromagnetic fields increase cell proliferation in lymphocytes from young and aged persons. Biochem. Biophys. Res. Comm. 160 : 692-698.
28. Cossarizza, A., D. Monti, P. Sola, G. Moschini, R. Cadossi, F. Bersani, and C. Franceschi. 1989. DNA repair after gamma irradiation in lymphocytes exposed to low-frequency pulsed electromagnetic fields. Radiat. Res. 118 : 161-168.
29. Bassett, C. A. L. 1978. Pulsing electromagnetic fields: a new approach to surgical problems . Pp. 255-306 in Metabolic Surgery, H. Buchwald and R. L. Varco, eds. New York : Grune and Stratton.
30. Bassett, C. A. L., S. N. Mitchell, and S. R. Gaston. 1982. Pulsing electromagnetic field treatment in ununited fractures and failed arthroses. JAMA 247 : 623-626.
31. Colacicco, G., and A. A. Pilla. 1983. Electromagnetic modulation of biological processes: bicarbonate effect and mechanistic considerations in the
Ca2+ uptake by embryonal chick tibia in vitro. Z. Naturforsch. [C] 38 : 465-467.
32. Colacicco, G., and A. A. Pilla. 1984. Electromagnetic modulation of biological processes: influence of culture media and significance of methodology in the Ca-uptake by embryonal chick tibia in vitro. Calcified Tissue Int. 36 : 167-174.
33. Colacicco, G., and A. A. Pilla. 1983. Electromagnetic modulation of biological processes: ATPase function and DNA production by Raji cancer cells in vitro. Z. Naturforsch. [C] 38 : 468-470.
34. Goodman, R., C. A. Bassett, and A. S. Henderson. 1983. Pulsing electromagnetic fields induce cellular transcription. Science 220 : 1283-1285.
35. Brown, H. D., and S. K. Chattopadhyay. 1988. Electromagnetic-field exposure and cancer. Cancer Biochem. Biophys. 9 : 295-342.
36. Meltz, M. L., P. Eagan, and D. N. Erwin. 1990. Proflavin and microwave radiation: Absence of a mutagenic interaction . Bioelectromagnetics 11 : 149-158.
37. Blevins, R. D., R. C. Crenshaw, A. E. Hougland, and C. E. Clark. 1980. The effects of microwave radiation and heat on specific mutants of Salmonella typhimurium LT2. Radiat. Res. 82 : 511-517.
38. Harte, C. 1973. Mutations in Oenothera hookeri after prolonged influence of radiowaves during one vegetation period. Theor. Appl. Genet. 43 : 6-12.
39. Alam, M. T., N. Barthakur, N. G. Lambert, and S. S. Kasatiya. 1978. Cytological effects of microwave radiation in Chinese hamster cells in vitro. Can. J. Genet. Cytol. 20 : 23-30.
40. Yao, K. T. S., and M. M. Jiles. 1970. Effects of 2450 MHz microwave radiation on cultivated kangaroo-rat cells. Pp. 123-133 in Biological Effects and Microwave Radiation: Symposium Proceedings , S.F. Cleary, ed. Public Health Service Publication No. BRH/DBE 70-2. Government Printing Office : Washington, D.C.
41. Ciaravino, M., M. L. Meltz, and D. N. Erwin. 1987. Effects of radiofrequency radiation and simultaneous exposure with mitomycin C on the frequency of sister chromatid exchanges in Chinese hamster ovary cells. Environ. Mutagen. 9 : 393-399.
42. Mitchell, J. A., A. A. Marino, T. J. Berger, and R. O. Becker. 1978. Effect of electrostatic fields on the chromosomes of Ehrlich ascites tumor cells exposed in vivo. Physiol. Chem. Phys. 10 : 79-85.
43. Marino, A. A., T. J. Berger, J. T. Mitchell, B. A. Duhacek, and R. O. Becker. 1974. Electric field effects in selected biologic systems. Ann. N. Y. Acad. Sci. 238 : 436-443.
44. McRee, D. I., G. MacNichols, and G. K. Livingston. 1981. Incidence of sister chromatid exchange in bone marrow cells of the mouse following microwave exposure. Radiat. Res. 85 : 340-348.
45. Beechey, C. V., D. Brooker, C. I. Kowalczuk, R. D. Saunders, and A. G. Searle. 1986. Cytogenetic effects of microwave irradiation on male germ cells of the mouse. Int. J. Radiat. Biol. 50 : 909-918.
46. Ramaiya, L. K., M. D. Pomerantseva, G. A. Vilkina, and V. S. Tikhonchuk. 1980. Study of the action of UHF microwaves on mammalian germ and somatic cells. Cytol. Genet. 14 : 1-5.
47. Kapustin, A. A., M. I. Rudnev, G. I. Leonskaya, and G. K. Konobeeva. 1976. Cytogenetic effects of a varying UHF electromagnetic field. Cytol. Genet. 10 : 16-18.
48. Yao, K. T. S. 1982. Cytogenetic consequences of microwave irradiation on mammalian cells incubated in vitro. J. Hered. 73 : 133-138.
49. Cohen, M. M., A. Jybsjam, J. A. Asterborski, D. McCulloch, and D. A. Paskewitz. 1986. Effect of low-level 60-Hz electromagnetic fields on human lymphoid cells: I. Mitotic lymphocytes. Bioelectromagnetics 7 : 415-424.
50. Cohen, M. M., A. Kunska, J. A. Astemborski, and D. McCulloch. 1986. The effect of low-level 60-Hz electromagnetic fields on human lymphoid cells: II.Sister chromatid exchanges in peripheral lymphocytes and lymphoblastoid cell lines. Mutat. Res. 172 : 177-184.
51. Rosenthal, M., and G. Obe. 1989. Effects of 50-Hz electromagnetic fields on proliferation and on chromosomal alterations in human peripheral lymphocytes untreated or pretreated with chemical mutagens. Mutat. Res. 210 : 329-335.
52. Garcia-Sagredo, J. M., L. A. Parada, and J. L. Monteagudo. 1990. Effect on SCE in human chromosomes in vitro of low-level pulsed magnetic field. Environ. and Mol. Mutag. 16 : 185-188.
53. Scarfi, M. R., F. Bersane, A. Cossarizza, D. Monti, G. Castellani, R. Cadossi, G. Franceschetti, and C. Franceschi. 1991. Spontaneous and mitomycin C-induced micronuclei in human lymphocytes exposed to extremely low frequency pulsed fields. Biochem. Biophys. Res. Commun. 176 : 194-200.
54. Nordenson, I., K. Hansson-Mild, S. Nordstrom, A. Sweins, and E. Birke. 1984. Clastogenic effects in human lymphocytes of power frequency electric fields: In vivo and in vitro studies. Radiat. Environ. Biophys. 23 : 191-201.
55. Khalil, A. M., and W. Qassem. 1991. Cytogenetic effects of pulsing electromagnetic fields on human lymphocytes in vitro: chromosome aberration, sister chromatid exchanges and cell kinetics. Mutat. Res. 274 : 141-146.
56. Stodolnik-Baranska, W. 1984. The effects of microwaves on human lymphocyte cultures. Biologic effects and health hazards of microwave radiation. Proc. Int. Symp. Warsaw, 1973. Pol. Med. Publishers. Pp. 189-195.
57. Wolff, S., T. L. James, G. B. Young, A. R. Margulis, J. Bodycote, and V. Afzal. 1985. Magnetic resonance imaging: Absence of in vitro cytogenetic damage . Radiology 155 : 163-165.
58. Balcer-Kubiczek, E. K., and G. H. Harrison. 1985. Evidence for microwave carcinogenesis in vitro. Carc. 6(6) : 859-864.
59. Balcer-Kubiczek, E. K. and G. H. Harrison. 1989. Induction of Neoplastic transformation in C3H/10T1/2 cells by 2.45 GHz microwaves and phorbol ester. Radiat. Res. 117 : 351-537.
60. Balcer-Kubiczek, E. K., and G. H. Harrison. 1991. Neoplastic transformation of C3H/10T1/2 cells following exposure to 120-Hz modulated 2.5 GHz microwaves and phorbol ester tumor promoter . Radiat. Res. 126 : 65-72.
61. Goodman, R., and A. S. Henderson. 1986. Sine waves enhance cellular transcription. Bioelectromagnetics 7 : 23-29.
62. Goodman, R., J. Abbott, and A. S. Henderson. 1987. Transcriptional patterns in the X chromosome of Sciara coprophilia following exposure to magnetic fields. Bioelectromagnetics 8 : 1-7.
63. Goodman, R., and A. S. Henderson. 1988. Exposure of salivary gland cells to low-frequency electromagnetic fields alters polypeptide synthesis. Proc. Natl. Acad. Sci. (USA) 85 : 3928-3932.
64. Goodman, R., L. X. Wei, J. C. Xu, and A. Henderson. 1989. Exposure of human cells to low-frequency electromagnetic fields results in quantitative changes in transcripts. Biochem. Biophys. Acta 1009 : 216-220.
65. Wei, L. X., R. Goodman, and A. Henderson. 1990. Changes in levels of c-myc and histone H2B following exposure of cells to low-frequency sinusoidal electromagnetic fields: evidence for a window effect. Bioelectromagnetics 11 : 269-272.
66. Greene, J. J., W. J. Skowronski, J. M. Mullins and R. M. Nardone. 1991. Delineation of electric and magnetic field effects of extremely low frequency electromagnetic radiation on transcription. Biochem. Biophys. Res. Commun. 174 : 742-749.
67. Litovitz, T. A., C. J. Montrose, R. Goodman, and E. C. Elson. Amplitude windows and transiently augmented transcription from exposure to electromagnetic fields. Bioelectromagnetics 11 : 297-312.
68. Aarholt, E., E. A. Flinn, and C. W. Smith. 1982. Magnetic fields affect the Lac operon system. Phys. Med. Biol. 27(4) : 606-610.
69. Yuspa, S. H. 1986. Cutaneous chemical carcinogenesis. J. Am. Acad. Dermatol. 15 : 1031-1044.
70. Slaga, T. J. 1989. Cellular and molecular mechanisms involved in multistage skin carcinogenesis . Pp. 1-18 in Skin Tumor: Experimental and Clinical Aspects, C. J. Conti, T. J. Slaga, and A. J. P. Klein-Szanto, eds. New York : Raven Press.
71. Weinstein, I. B. 1988. The origins of human cancer: Molecular mechanisms of carcinogenesis and their implications for cancer prevention and treatment. Twenty-seventh G. H. A. Clowes Award Lecture. Cancer Res. 48 : 4135-4143.
72. Grynkiewicz, G., M. Poenie, and R. Y. Tsien. 1985. A new generation of Ca2+ indicators with greatly improved fluorescence properties. J. Biol Chem. 260 : 3440-3450.
73. Adey, W. R. 1990. Electromagnetic fields, cell membrane amplification, and cancer promotion . Pp. 211-249 in Extremely Low Frequency Electromagnetic Fields: The Question of Cancer , B. W. Wilson, R. G. Stevens, and L. E. Anderson, eds. Columbus, OH : Battelle Press.
74. Nishizuka, Y. 1984. The role of protein kinase C in cell surface signal transduction and tumor promotion. Nature 308 : 693-698.
75. Bawin, S. M., L. K. Kaczmarek, and W. R. Adey. 1975. Effects of modulated VHF fields on the central nervous system. Ann. N.Y. Acad. Sci. 274 : 74-81.
76. Bawin, S. M., and W. R. Adey. 1976. Sensitivity of calcium binding in cerebral tissue to weak environmental electric fields oscillating at low frequency. Proc. Natl. Acad. Sci. (USA) 73 : 1999-2003.
77. Blackman, C. F., S. G. Genane, L. S. Kinney, W. T. Jones, and D. E. House. 1982. Effects of ELF fields on calcium-ion efflux from brain tissue in vitro. Radiat. Res. 92 : 510-520.
78. Tenforde, T. S. 1980. Thermal aspects of electromagnetic field interactions with bound calcium ions at the nerve cell surface. J. Theoretical Biol. 83 : 517-521.
79. Blackman, C. F. 1990. ELF effects on calcium homeostasis. Pp. 187-208 in Extremely Low Frequency Magnetic Fields: The Question of Cancer, B. W. Wilson, R. G. Stevens, and L. E. Anderson, eds. Columbus, OH : Battelle Press.
80. Lin-Liu, S., and W. R. Adey. 1982. Low frequency amplitude modulated microwave fields change calcium efflux rates from synaptosomes. Bioelectromagnetics 3 : 309-322.
81. Dutta, S. K., A. Subramoniam, B. Ghosh, and R. Parshad. 1984. Microwave radiation-induced calcium ion efflux from human neuroblastoma cells in culture. Bioelectromagnetics 5 : 71-78.
82. Dutta, S. K., B. Ghosh, and C. F. Blackman. 1989. Radiofrequency radiation-induced calcium ion-efflux enhancement from human and other neuroblastoma cells in culture. Bioelectromagnetics 19(2) : 197-202.
83. Myers, R. D., and D. H. Ross. 1981, Radiation and brain calcium. A review and critique. Neurosci. Biobehav. Rev. 5 : 503-543.
84. Pegg, A. E., and H. G. Williams-Ashman. 1981. Pp. 3-42 in Polyamines in Biology and Medicine. D. A. Morris and L. J. Marton, eds. New York : Marcel Dekker.
85. Byus, C. V., S. E. Pieper, and W. R. Adey. 1987. The effects of low-energy 60 Hz environmental electromagnetic fields upon the growth-related enzyme ornithine decarboxylase. Carcinogenesis 8 : 1385-1389.
86. Byus, C. V., K. Kartun, S. Pieper, and W. R. Adey. 1988. Increased ornithine decarboxylase activity in cultured cells exposed to low energy modulated microwave fields and phorbol ester tumor promoters. Cancer Res. 48 : 4222-4226.
87. Byus, C. V., R. L. Lundak, R. M. Fletcher, and W. R. Adey. 1984. Alterations in protein kinase activity following exposure of cultured human lymphocytes to modulated microwave fields. Bioelectromagnetics 5 : 341-351.
























