A
The Role of Nutrients in Coastal Waters
Urban wastewaters contain high concentrations of nutrients and as such, contribute significantly to the mass loadings of nitrogen and phosphorus to coastal waters. The various inorganic forms of nitrogen and phosphorus stimulate aquatic plant growth, and since they are relatively hydrophilic, their removal from the water column is more biologically mediated than for trace metals or hydrophobic organics. These characteristics are somewhat unique for urban wastewater contaminants. Therefore, nutrients will be considered here in detail.
Coastal waters receive large amounts of nutrients from wastewater treatment plants and nonpoint sources. In particular, estuaries receive more nutrient inputs per unit surface area than any other type of ecosystem. Many estuaries receive nutrient inputs per unit area that are more than 1,000-fold greater than those of heavily fertilized agricultural fields (Nixon et al. 1986). In moderation, nutrient inputs to estuaries and coastal seas can be considered beneficial. They result in increased production of phytoplankton (the microscopic algae floating in water), which in turn can lead to increased production of fish and shellfish (Nixon 1988, Hansson and Rudstam 1990, Rosenberg et al. 1990). However, excess nutrients can be highly damaging, leading to effects such as anoxia and hypoxia from eutrophication, nuisance algal blooms, dieback of seagrasses and corals, and reduced populations of fish and shellfish (Ryther 1954, 1989; Kirkman 1976; McComb et al. 1981; Kemp et al. 1983; Cambridge and McComb 1984; Gray and Paasche 1984; Officer et al. 1984; Larsson et al. 1985; Price et al. 1985; Rosenberg 1985; D'Elia 1987; Baden et al. 1990; Cederwall and Elmgren 1990; Hansson and
Rudstam 1990; Rosenberg et al. 1990; Parker and O'Reilly 1991; Lein and Ivanov 1992; Smayda 1992). Eutrophication also may change the plankton-based food web from one based on diatoms toward one based on flagellates or other phytoplankton, which are less desirable as food to organisms at higher trophic levels (Doering et al. 1989).
Whether or not nutrient inputs should be considered excessive depends in part upon the physics and ecological sensitivity of the receiving water body. In many parts of the world, estuaries and coastal seas clearly are receiving an excess of nutrients, and the resulting eutrophication is one of the major causes of decline of coastal waters. Nutrients should be considered a major ecological concern along with sewage disposal in many coastal urban areas and be regulated accordingly.
This section first briefly discusses the negative effects of eutrophication and nuisance algal blooms in coastal marine ecosystems. It then reviews in more detail the controls on eutrophication; discusses the issue of whether nitrogen or phosphorus is more limiting to eutrophication; presents dose-response information, which relates nitrogen to algal biomass and production; and reviews the information on controls of nuisance algal blooms.
ADVERSE CONSEQUENCES OF EUTROPHICATION AND NUISANCE ALGAE
Anoxia and Hypoxia
Anoxia is the complete removal of dissolved oxygen from the water column, an event which obviously causes widespread damage to aquatic plants and animals. Even mobile animals that can escape from anoxic waters can suffer population declines from the loss of habitat area. For example, in parts of the Baltic Sea, cod eggs laid in oxic surface waters sink into anoxic bottom waters where they die (Rosenberg et al. 1990). Oxygen concentrations in the bottom waters of the deep basins of the Baltic between 1969 and 1983 correlate negatively with codfish populations (Hansson and Rudstam 1990).
Oxygen need not be completely absent for damage to occur, and a lowering of oxygen to concentrations as low as 3 to 4.3 mg per liter can cause ecological harm in some estuaries and coastal seas (EPA 1990). Such a depletion of oxygen is termed hypoxia. Examples of ecological damage from hypoxia include lowered survival of larval fish, mortality of some species of benthic invertebrates, and loss of habitat for some mobile species of fish and shellfish that require higher concentrations of oxygen, such as lobster and codfish (Baden et al. 1990, EPA 1990). Significant mortalities of lobsters and population declines of both lobster and codfish have been
observed in some Swedish coastal waters as a result of increased incidences of hypoxia (Baden et al. 1990).
Anoxia and hypoxia are major and growing problems in many estuaries and coastal seas. Over the past few decades, the volume of anoxic bottom waters has been increasing in the Chesapeake Bay (Officer et al. 1984, D'Elia 1987), the Baltic Sea (Larsson et al. 1985), and the Black Sea (Lein and Ivanov 1992). The apex of the New York Bight (an area of some 1,250 km2) becomes hypoxic every year, and a large region of the Bight became anoxic in 1976 (Mearns et al. 1982). Hypoxic events appear to be becoming more common in waters such as the Long Island Sound (EPA 1990, Parker and O'Reilly 1991), the North Sea (Rosenberg 1985), and the Kattegat (the waters between Denmark and Sweden; Baden et al. 1990), although historical data on oxygen concentrations in coastal waters are often poor.
Anoxia and hypoxia result from oxygen consumption exceeding oxygen supply. Oxygen is consumed by the respiration of organisms, including animals, plants, and the decomposing activity of microorganisms. Oxygen is supplied to waters through the process of photosynthesis and through diffusion and surface entrapment from the atmosphere. Organic matter released in sewage effluent can contribute to anoxia and hypoxia by creating biochemical oxygen demand (BOD), the oxygen consumed during the microbial decomposition of this organic matter and chemical oxygen demand (COD), the oxygen consumed through the oxidation of ammonium and other inorganic reduced compounds. For example, altered structure, reduced diversity, and elevated biomass of benthic animal communities—related in part to high organic solids inputs—once characterized up to 95 square kilometers of the coastal shelf around major outfalls in San Diego, Orange, and Los Angeles Counties in southern California (Mearns and Word 1982). However, BOD inputs to most estuaries and coastal seas are well controlled and represent at most a localized problem. Of more concern in most estuaries and coastal marine ecosystems is the oxygen consumption that results from the decomposition of the excess phytoplankton production characteristic of eutrophication (Officer et al. 1984, Larsson et al. 1985, Jensen et al. 1990, Rydberg et al. 1990, EPA 1990, Parker and O'Reilly 1991, Lein and Ivanov 1992). Photosynthesis by phytoplankton produces oxygen, but much of the photosynthesis in eutrophic waters occurs near the surface, and oxygen readily escapes to the atmosphere. The majority of the phytoplankton material is decomposed deeper in the water column, consuming oxygen there. In contrast to the rather localized effects of BOD inputs, nutrient inputs can lead to eutrophication and anoxia or hypoxia far from the original source of the nutrient. In some cases, improved sewage treatment may aggravate this situation by resulting in more distant transport of nitrogen (Chesterikoff et al. 1992). Some evidence points to increasing hypoxia in the western basin of the Long Island Sound as being a result of
improved sewage treatment in the East River in New York City with resulting increase of flows of nitrogen from the East River into the Sound (Parker and O'Reilly 1991).
Dieback of Seagrasses, Algal Beds, and Corals
In addition to anoxia and hypoxia, eutrophication can lead to the dieback of seagrass beds, which are important habitats and nursery grounds for a variety of fish and other animals. One mechanism for such dieback is a shading out of the grasses by the abundant phytoplankton in the overlying water, a process thought to have caused the dieback of macrophytes in the upper portions of the Chesapeake Bay (Kemp et al. 1983, Twilley et al. 1985, D'Elia 1987), in the Dutch Wadden Sea (Gieson et al. 1990), and of both tropical and temperate seagrasses in Australia (Kirkman 1976, Cambridge and McComb 1984, Cambridge et al. 1986). Dieback caused by such shading usually manifests itself in a rather gradual loss of the seagrasses (Robblee et al. 1991), although the occurrence of unusual nuisance algal blooms in 1985 and 1986 greatly reduced the abundance of seagrass beds near Long Island (Dennison et al. 1989). Shading by enhanced epiphytic (Twilley et al. 1985) and macroalgal (Valiela et al. 1990) growth may be an additional cause of seagrass dieback. Nutrient enrichment may also have a direct physiological response on seagrasses, with internal nutrient imbalances appearing to lead to reduced survival (Burkholder et al. 1992a).
Beds of attached macro-algae on bottom sediments or rocks can also be adversely affected by eutrophication. Nutrient enrichment of rocky intertidal areas typically leads to a reduction in the overall diversity of both attached algae (Borowitzka 1972, Littler and Murray 1978) and associated animals (Gappa et al. 1990). These nutrient-enriched areas tend to be dominated by opportunistic algae with rapid growth rates, such as Cladophora sp. and Enteromorpha sp., which can take advantage of the elevated nutrient levels and shade out other species (Littler and Murray 1975, 1978). This phenomenon is seen clearly along the Swedish coast of the Baltic Sea, where since the mid-1970s nuisance forms of filamentous algae (Cladophora and Enteromorpha species) have become more dominant, coinciding with a decline of the former dominant bladderwrack algae, Fucus sp. (Baden et al. 1990, Rosenberg et al. 1990). The bladderwrack is used by herring in spawning, and the change to dominance by filamentous macroalgae has led to decreased hatching of herring eggs (Rosenberg et al. 1990). Subtidal forests of giant kelp (Macrocystis pyrifera) died back and failed to reproduce during the 1960s and early 1970s along the Palos Verdes Peninsula near the Los Angeles County outfalls (Wilson et al. 1980). This may have been due to light limitation, perhaps a result of eutrophication, but was more likely due directly to solids discharged in poorly treated sewage. Toxic
substances associated with these particle discharges may also have played a role. The kelp beds recovered following significant reductions in the solids emissions (Grigg 1978, Harris 1980, Wilson et al. 1980).
Reduced light levels from excessive phytoplankton and macroalgal growth in eutrophic environments can cause coral dieback. Shading affects coral growth by decreasing the productivity of the zooxanthallae, symbiotic algae in the coral tissue, which provides much of the coral's nutrition (Smith 1981). Excess nutrients can also cause a shift in the composition of the coral community as other species outcompete corals for space. Filter-feeding species such as sponges take advantage of the high phytoplankton productivity and become dominant, displacing the corals (Pastorak and Bilyard 1985). Sedimentation of decomposing phytoplankton may have an additional detrimental impact on coral growth (Smith 1981).
Nuisance Algal Blooms
Blooms of nuisance algae are characterized by very high abundances of one overwhelmingly dominant species in the phytoplankton. These blooms often result in noticeable color and are popularly named by this color: red tides, green tides, and brown tides. As with eutrophication generally, these blooms can result in anoxic or hypoxic conditions. In addition, many nuisance blooms produce substances toxic to aquatic organisms or humans (Cosper 1991). Green tides during the 1950s heavily damaged oyster populations on Long Island (Ryther 1954, 1989), and brown tides in 1985 and 1986 greatly reduced populations of bay scallops on Long Island (Cosper et al. 1987, Bricelj and Kuenstner 1989) and of blue mussels in Narragansett Bay (Tracey et al. 1989). These shellfish starved to death since they were unable to graze on the brown-tide algae. Blooms of some dinoflagellates (red tides) can result in the accumulation of toxins in shellfish, which, when eaten by humans, cause paralytic or diarrhetic shellfish poisoning (Smayda 1989). Frequent blooms of a gold-brown dinoflagellate in Northern Europe have caused extensive fish mortality since the mid-1960s (Smayda 1989). In 1991, toxins produced by a diatom bloom concentrated in anchovy and caused the death of pelicans that fed on these fish (Work et al. undated, as cited in Smayda 1992). Recently, Burkholder et al. (1992b) discovered a new toxic dinoflagellate that releases toxins only in the presence of fish and appears to be responsible for several fish kills in estuaries in North Carolina.
Nuisance-bloom tides have been known since biblical times (Cosper 1991), but blooms of many species appear to be occurring with greater frequency throughout the world (Hallegraeff et al. 1988; Anderson 1989; Smayda 1989, 1992; Robineau et al. 1991). Red-tide blooms of toxic dinoflagellates appear to be more frequent in many parts of the world (Ander-
son 1989, Smayda 1989, Wells et al. 1991), and blooms of cyanobacteria have become more prevalent in the less saline portions of the Chesapeake Bay (D'Elia 1987) and in the Baltic Sea and related waters over the past 10 to 20 years (Smayda 1989 and references therein). Many of the new toxic phytoplankton blooms are subpopulations of previously non-toxic species, which now occur at previously unseen abundances (Smayda 1989, 1992). Brown-tide blooms of Alureococcus anophagefferens were unknown before 1985 (Sieburth et al. 1988). As discussed below, the cause(s) of increased nuisance blooms is not known, but evidence points toward the importance of increased nutrient inputs (nitrogen, phosphorus, iron) to estuaries and coastal seas.
CONTROLS ON EUTROPHICATION AND NUISANCE BLOOMS IN COASTAL WATERS
Nutrient Limitation
Nutrients are elements essential for plant growth, such as nitrogen, phosphorus, silica, and sulfur. Phytoplankton production in most coastal marine ecosystems and estuaries is nutrient limited, and increased nutrient inputs lead to higher production and eutrophication (Ryther and Dunstan 1971; Graneli 1978, 1981, 1984; McComb et al. 1981; Boynton et al. 1982; Nixon and Pilson 1983; Smith 1984; Valiela 1984; D'Elia et al. 1986; Nixon et al. 1986; D'Elia 1987; Howarth 1988; Andersen et al. 1991). Unfortunately, the discussion of nutrient limitation in coastal marine waters has been surrounded by some confusion, in part because the term can have many different meanings and is often used quite loosely (Howarth 1988) and in part because of potential methodological problems in determining nutrient limitation (Hecky and Kilham 1988, Howarth 1988, Banse 1990). If one is concerned with eutrophication, then the appropriate definition of nutrient limitation is the regulation of the potential rate of net primary production by phytoplankton (Howarth 1988). Net primary production is defined as the total amount of photosynthesis minus the amount of plant respiration occurring in a given area (or volume) of water in a given amount of time. If an addition of nutrients would increase the rate of net primary production, even if this means a complete change in the species composition of the phytoplankton, then production is considered to be nutrient limited (Howarth 1988, Vitousek and Howarth 1991).
Factors other than nutrient input can also influence or partially control primary production. For instance, phytoplankton production in some estuaries (such as the Hudson River) is limited by light availability. This tends to occur in extremely turbid estuaries or in estuaries where moderate turbidity coexists with deep mixing of the water. The turbidity can result both
from suspension of inorganic particles and from high phytoplankton biomass, and so light limitation often is a result of self-shading by the phytoplankton, as is the case in many lakes (Wetzel 1983). In estuaries where nutrient inputs are high and production is limited by light, the nutrients are simply transported further away from the source before being first assimilated by phytoplankton, as is seen in the transport of nutrients from the Hudson River and New York Harbor into the New York Bight (Malone 1982). This further transport may or may not provide sufficient dilution to avoid excessive eutrophication, which may just occur further afield from the nutrient source.
Through their grazing on phytoplankton, zooplankton and other animals can also influence the rate of primary production and the biomass of phytoplankton. This has received extensive study and discussion in freshwater ecosystems (Carpenter et al. 1985, Morin et al. 1991) and in offshore ocean ecosystems (Steele 1974, Banse 1990) although it is virtually unstudied in estuaries and coastal seas. Nonetheless, changes in grazing in estuaries may have serious effects on water quality. For instance, some researchers believe that at one time, oyster populations in the Chesapeake Bay were sufficiently high to filter the bay's entire water volume on average once every week; the currently declined oyster populations probably filter the water of the bay only once per year on average (Newell 1988). This lower grazing pressure on phytoplankton populations may be contributing to eutrophication of the Chesapeake. However, reductions in grazing pressure can result in algal blooms only where nutrient availabilities are high. Thus, nutrient supply should be viewed as the cause of eutrophication with grazing pressures being a secondary regulator.
Nitrogen Versus Phosphorus Limitation
Nitrogen is the element usually limiting to primary production by phytoplankton in most estuaries and coastal seas of the temperate zone (Ryther and Dunstan 1971; Vince and Valiela 1973; Smayda 1974; Norin 1977; Graneli 1978, 1981, 1984; Boynton et al. 1982; Nixon and Pilson 1983; Valiela 1984; D'Elia et al. 1986; Nixon et al. 1986; Frithsen et al. 1988; Howarth 1988; Rydberg et al. 1990; Vitousek and Howarth 1991; Nixon 1992), although some temperate estuaries such as the Apalachicola in the Gulf of Mexico may be phosphorus limited (Myers and Iverson 1981; Howarth 1988) and others such as parts of the Chesapeake Bay and the Baltic Sea may switch seasonally between nitrogen and phosphorus limitation (McComb et al. 1981, D'Elia et al. 1986, Graneli et al. 1990, Andersen et al. 1991). Many tropical estuarine lagoons may be phosphorus limited as well (Smith 1984, Smith and Atkinson 1984, Howarth 1988, Vitousek and Howarth 1991). Sewage often accounts for 50 percent or more of the nitrogen inputs to
TABLE A.1 Total Annual Inputs of Dissolved Inorganic Nitrogen to Various Estuaries and the Percent of this Input which comes from Sewage Treatment Plants (Source: Nixon and Pilson 1983. Reprinted, by permission, from Academic Press, 1983.)
|
|
DIN input (mmole m-2 year-1) |
Sewage (%) |
|
Kaneohe Bay |
230 |
78 |
|
Long Island Sound |
400 |
67 |
|
Chesapeake Bay |
510 |
33 |
|
Apalachicola Bay |
560 |
2 |
|
Barataria Bay |
570 |
<1 |
|
Patuxent Estuary |
600 |
48 |
|
Potomac Estuary |
810 |
48 |
|
Pamlico Estuary |
860 |
<1 |
|
Narragansett Bay |
950 |
41 |
|
Mobile Bay |
1,280 |
7 |
|
Delaware Bay |
1,300 |
50 |
|
Raritan Bay |
1,460 |
86 |
|
South San Francisco Bay |
1,600 |
˜100 |
|
North San Francisco Bay |
2,010 |
45 |
|
New York Bay |
31,900 |
82 |
various estuaries and may be a more controllable input than those from agriculture and other nonpoint sources (Table A.1, Nixon and Pilson 1983).
That nitrogen limits primary production in most temperate-zone estuaries and coastal seas was much debated throughout the 1980s (D'Elia 1987, Howarth 1988, Nixon 1992). One argument against nitrogen limitation was that phosphorus is generally limiting in temperate-zone lakes (Edmondson 1970; Vollenwieder 1976, 1979; Schindler 1977, 1978; Wetzel 1983) and, until recently, there was little evidence that the biogeochemical processes regulating nutrient limitation were fundamentally different between freshwater and marine ecosystems (Schindler 1981, Smith 1984). Another argument was that much of the evidence for nitrogen limitation in marine ecosystems came from extremely short-term (generally a few days), small-scale enrichment experiments in flasks or bottles. It may not be possible to extrapolate the results of such short-term enrichment experiments to the whole ecosystem, in part because they only measure the physiological response of phytoplankton species that are present in the water and can respond in a significant way during the time of the experiment (Smith 1984, Hecky and Kilham 1988, Howarth 1988, Banse 1990, Marino et al. 1990).
In recent years, increasing evidence has accumulated both that nitrogen is limiting in many coastal marine ecosystems and that the biogeochemical
processes regulating nutrient limitation do in fact vary between marine and freshwater ecosystems. The evidence for nitrogen limitation, in addition to the short-term enrichment experiments, consists of generally low concentrations of dissolved nitrogen compared to dissolved phosphorus (Boynton et al. 1982, Graneli 1984, Valiela 1984) and longer, larger scale enrichment experiments (D'Elia et al. 1986), including one mesocosm experiment of many months duration in the Marine Ecosystem Research Laboratory (MERL) facility (Frithsen et al. 1988, and unpublished data). While any one such piece of evidence may not be entirely convincing, the agreement among the several lines of evidence convincingly demonstrates nitrogen limitation (Howarth 1988, Vitousek and Howarth 1991).
What differences in biogeochemical cycles lead toward nitrogen limitation in temperate coastal marine ecosystems and toward phosphorus limitation in temperate lakes? At least three factors appear important (Figure A.1): the ratio of nitrogen to phosphorus in nutrient inputs to estuaries is

FIGURE A.1 Summary of biogeochemical processes controlling nutrient limitation in aquatic ecosystems. The ratio of nitrogen to phosphorus from terrestrial inputs varies greatly, generally being lower for ecosystems receiving more sewage inputs. Nitrogen and phosphorus are assimilated by phytoplankton in an approximate molar ratio of 16:1. The N:P ratio of nutrients released in the water column during zooplankton feeding and during decomposition frequently approximates 16:1 but can be lower. Conversely, the N:P ratio of sedimenting material also frequently approximated 16:1 but can be as high as 30:1 in oligotrophic waters. Nutrients released from marine and estuarine sediments back to the water column tend to have low N:P ratios, but such releases in lack sediments often have N:P ratios near 16:1 or higher. The extent of nitrogen fixation varies but tends to be much greater in lakes than in marine ecosystems. (Source: Howarth 1988. Reproduced, with permission, from the Annual Review of Ecology and Systematics, Vol. 19, C 1988 by Annual Reviews Inc.)
frequently less than for lakes, the sediments are often a more important sink of phosphorus in lakes than in marine ecosystems, and nitrogen fixation is a more prevalent process in the plankton of lakes (Howarth 1988). Each of these is discussed briefly below.
In both freshwater and marine ecosystems, the relative requirements of phytoplankton for nitrogen and phosphorus are fairly constant, with the two elements being assimilated in the approximate molar ratio of 16:1, the Redfield ratio (Redfield 1958). If there were no biogeochemical processes acting within a water body, the ratio of nitrogen to phosphorus in the nutrient inputs to the ecosystem would determine whether the system were nitrogen or phosphorus limited. Ratios below 16:1 would lead to nitrogen limitation and higher ratios would lead to phosphorus limitation (Howarth 1988). In fact, the N:P ratio in nutrient loadings to many (but by no means all) estuaries and coastal seas are below this ratio, while nutrient inputs to temperate lakes tend to have higher N:P ratios (Jaworski 1981, Kelly and Levin 1986, NOAA/EPA 1988). This difference in ratios probably reflects the relative importance of sewage, which tends to have a low N:P ratio, as a nutrient source to coastal waters.
Biogeochemical processes within sediments act to alter the relative abundance of nitrogen and phosphorus in an ecosystem. Denitrification, the bacterial reduction of nitrate to molecular nitrogen, removes nitrogen and tends to make coastal marine ecosystems more nitrogen limited (Nixon et al. 1980, Nixon and Pilson 1983). However, this process appears to be even more important in lakes than in estuaries and coastal seas; a higher percentage of the nitrogen mineralized during decomposition is denitrified in lake sediments than in estuarine sediments (Seitzinger 1988, Gardner et al. 1991, Seitzinger et al. 1991). Of more importance in explaining a tendency for nitrogen limitation in coastal marine ecosystems of the temperate zone, therefore, is the relatively high phosphorus flux from sediments; nutrient fluxes from these sediments have fairly low N:P ratios (Rowe et al. 1975, Boynton et al. 1980, Nixon et al. 1980). In many lakes, phosphorus is bound in the sediments (Schindler et al. 1977), although in others, phosphorus fluxes are comparable to marine sediments (Khalid et al. 1977). Nutrient fluxes from lake sediments can be either enriched or depleted in nitrogen relative to phosphorus (Kamp-Nielsen 1974). Caraco et al. (1989, 1990) have suggested that the abundance of sulfate in an ecosystem partially regulates the sediment flux of phosphorus. Phosphorus binding in sediments is greatest where sulfate concentrations are lowest, which is consistent with variable fluxes in lakes and higher fluxes in coastal marine ecosystems.
When the relative abundance of nitrogen to phosphorus is low in the water column of lakes, nitrogen-fixing species of cyanobacteria are favored since they can convert molecular nitrogen to ammonium or organic nitrogen. Under such nitrogen-depleted conditions in lakes, these cyanobacteria
often are the dominant phytoplankton species and fix appreciable quantities of nitrogen. As a result, nitrogen deficits (relative to phosphorus) can be alleviated, and primary production in the lake is phosphorus limited (Schindler 1977, Flett et al. 1980, Howarth 1988, Howarth et al. 1988a). In contrast, nitrogen-fixing cyanobacteria are rare or absent from the plankton of most estuaries and coastal seas, which is a condition helping to maintain nitrogen limitation in these ecosystems (Howarth 1988, Howarth et al. 1988a). Exceptions are found in the Baltic Sea (Lindahl and Wallstrom 1985) and in the Harvey-Peel estuary in Australia (McComb et al. 1981) but are unknown in the waters of the United States. The explanation for the rarity of planktonic, nitrogen-fixing cyanobacteria in coastal marine waters is still subject to debate (Paerl et al. 1987, Howarth et al. 1988b, Paerl and Carlton 1988, Carpenter et al. 1990, Marino et al. 1990, Vitousek and Howarth 1991). Possible reasons include one or more of the following: a lower availability of iron and molybdenum, trace metals required for nitrogen fixation, in saline water (Howarth and Cole 1985, Howarth et al. 1988b, Marino et al. 1990); greater turbulence in coastal marine systems, allowing oxygen to poison the nitrogenase enzyme responsible for nitrogen fixation (Paerl et al. 1987, Paerl and Carlton 1988); greater grazing pressure on cyanobacteria in marine systems (Vitousek and Howarth 1991); and a lower light availability in estuaries and coastal waters due to higher turbidity and/or deeper mixed layers (Howarth and Marino 1990, Vitousek and Howarth 1991).
As noted above, many tropical estuaries and coastal systems may be phosphorus limited (Smith 1984, Smith and Atkinson 1984). Although the evidence for limitation of production by phytoplankton is not entirely clear in tropical systems (Howarth 1988), and production by seagrasses and attached macroalgae is sometimes nitrogen limited in tropical systems (Lapointe et al. 1987, McGlathery et al. 1992), primary production by seagrasses in many tropical areas is clearly limited by phosphorus (Short et al. 1985, 1990; Littler et al. 1988; Powell et al. 1989). Phosphorus limitation in these systems is probably the result both of a high degree of phosphorus adsorption in the calcium-carbonate sediments that dominate such tropical systems (Morse et al. 1985) and the high rates of nitrogen fixation associated with benthic algal mats and with symbionts of seagrasses in clear, relatively oligotrophic lagoons (Howarth 1988, Howarth et al. 1988a).
This report does not explicitly consider nutrient limitation in open ocean systems away from the coast because there is no reason to believe that such ecosystems can be significantly affected by wastewater inputs.
Dose-response Relationship: Nitrogen and Eutrophication
The relationship between phosphorus loadings, phosphorus concentrations, chlorophyll (a measure of phytoplankton biomass), and primary pro-

FIGURE A.2a Regression plot relating nitrogen loadings to annual phytoplankton production in a variety of estuarine ecosystems: 1) Chesapeake Bay, 2) Patuxent River, 3) Pamlico River, 4) Byfjord, 5) Apalachicola Bay, 6) Narragansett Bay, 7) San Francisco Bay, 8) St. Margarets Bay, 9) Long Island Sound, 10) Kungsbacka Fjord, 11) Loch Etive, 12) St. Lawrence River, 13) Baltic Sea, and 14) Kaneohe Bay. (Source: Boynton et al. 1982. Reprinted, by permission, from Academic Press, 1982.)
duction has been extensively studied through statistical analysis in freshwater lakes using large, multi-lake data sets (Vollenweider 1976, 1979; Schindler 1978; Smith 1979; Wetzel 1983; Molot and Dillon 1991; and references therein). Generally, a positive relationship is found between either phosphorus loadings or concentrations of phosphorus in the water column and either chlorophyll or primary production. These relationships have been used successfully to manage eutrophication in lakes (Wetzel 1983).
Although the available data sets are smaller, similar approaches have been applied to study nitrogen and eutrophication in estuaries and coastal marine ecosystems. Data compiled from a variety of estuaries show a positive relationship between loadings of either total nitrogen per unit area (Boynton et al. 1982) or dissolved inorganic nitrogen per unit volume (Nixon and Pilson 1983, Nixon 1992) to the estuary and rates of primary production (Figures A.2a and A.3). Interestingly, nitrogen loadings to estuaries are a better predictor of primary production than are phosphorus loadings (Figures A.2a and A.2b)-further evidence that a majority of these ecosystems are probably nitrogen limited (Boynton et al. 1982). Whether a particular
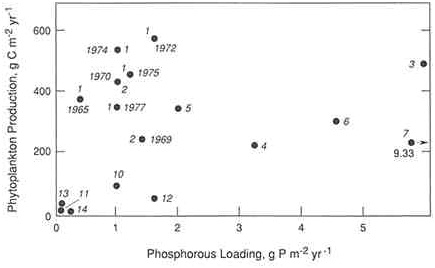
FIGURE A.2b Regression plot relating phosphorus loadings to annual phytoplankton production in a variety of estuarine ecosystems: 1) Chesapeake Bay, 2) Patuxent River, 3) Pamlico River, 4) Byfjord, 5) Apalachicola Bay, 6) Narragansett Bay, 7) San Francisco Bay, 8) St. Margarets Bay, 9) Long Island Sound, 10) Kungsbacka Fjord, 11) Loch Etive, 12) St. Lawrence River, 13) Baltic Sea, and 14) Kaneohe Bay. (Source: Boynton et al. 1982. Reprinted, by permission, from Academic Press, 1982.)
level of production should be considered excessive would depend upon the circulation of the water body and other factors affecting the supply of oxygen to the ecosystem.
Chlorophyll concentrations (phytoplankton biomass) are also correlated with inputs of dissolved inorganic nitrogen to various estuarine and coastal marine ecosystems (Nixon and Pilson 1983). Figure A.4a shows the relationship between annual inputs of dissolved inorganic nitrogen per unit volume and the average annual abundance of chlorophyll for a variety of estuaries. Data from an enrichment experiment at MERL are also included. Figure A.4b is similar but relates nitrogen inputs to average chlorophyll abundances during the summer season instead of giving annual values. The results from the MERL experiment compare favorably with the data from natural ecosystems, particularly at lower rates of nitrogen input. At the higher nutrient inputs, compared with natural ecosystems, the MERL mesocosms tend to have lower chlorophyll values during the summer and higher annual average chlorophyll values (Figures A.4a and A.4b).
No guidelines exist by which to determine whether coastal marine eco-

FIGURE A.3 Annual measurements of primary production as a function of the estimated annual input of dissolved inorganic nitrogen (DIN) in various estuaries. 1) Narragansett Bay, Rhode Island; 2) Long Island Sound; 3) Lower New York Bay; 4) Lower Delaware Bay; 5) Chesapeake Bay; 6) Patuxent estuary, Maryland; 7) Pamlico estuary, North Carolina; 8) Apalachicola Bay, Florida; 9) Barataria Bay, Louisiana; 10) North and South San Francisco Bay, California; 11) Kaneohe Bay, Hawaii. (Source: Nixon and Pilson 1983. Reprinted, by permission, from Academic Press, 1983.)
systems are in fact eutrophic. In lakes, however, over the past few decades, collective expert opinion has tended toward the following relationship of annual chlorophyll to trophic status (Wetzel 1983): oligotrophic lakes average 1.7 mg m-3 chlorophyll a (range = 0.3 to 4.5), mesotrophic lakes average 4.7 mg m-3 (range = 3 to 11), and eutrophic lakes average 14.3 mg m-3 (range = 3 to 78). There is no reason why these criteria should not be equally applicable to marine ecosystems, and the averages and ranges are drawn onto Figures A.4a and A.4b for reference. Using these delineations, few estuaries are oligotrophic, many are mesotrophic, and many are extremely eutrophic. Note that based on this interpretation of trophic status, Kaneohe Bay (Honolulu, Hawaii) is considered oligotrophic (average annual chlorophyll of 2 and 6 mg m-3; Nixon and Pilson 1983), and Long Island Sound is considered mesotrophic (average annual chlorophyll of 6 mg m-3; Nixon and Pilson 1983). Yet nutrient enrichment has severely modified the coral community of Kaneohe Bay (Smith 1981, Pastorak and Bilyard 1985), and eutrophication in Long Island Sound appears to be leading to increasingly frequent hypoxic events (EPA 1990). Thus, the applica-
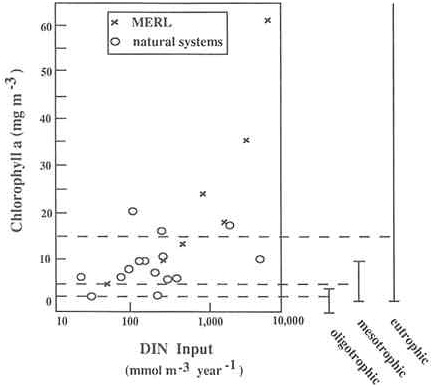
FIGURE A.4a Annual average concentration of chlorophyll compared to inorganic nitrogen inputs in a variety of natural estuaries and in experimental mesocosms at the Marine Ecosystem Research Laboratory, University of Rhode Island. The mesocosms were fertilized with nutrients. Data are from Nixon and Pilson (1983). Chlorophyll is a measure of phytoplankton abundance. Bars to the right indicate ranges of chlorophyll believed to characterize lakes as being oligotrophic, mesotrophic, or eutrophic. Dashed horizontal lines represent the mean chlorophyll values for each classification (Wetzel 1983). See text for a discussion of the applicability of this classification to estuaries.
tion of this lake-based approach to the trophic status of estuaries may not provide sufficient protection. This is the case, in part, because such trophic guidelines are based on phytoplankton response to nutrients yet problems such as those experienced in Kaneohe Bay are the result of benthic algae overgrowing the coral.
Jaworski (1981) proposed ''permissible" nutrient inputs to protect shallow (4-meters to 9-meters deep) temperate-zone estuaries from eutrophica-

FIGURE A.4b Summer mean concentration of chlorophyll. The comparison shown here is the same as for Figure A.4a, except chlorophyll values represent the mean for the summer season rather than annual means.
tion. For phosphorus-limited estuaries, he suggested that inputs of phosphorus should be kept below 24 mmol P m-2 year-1. For nitrogen-limited estuaries, Jaworski proposed that inputs be kept below 380 mmol N m-2 year-1. For these shallow estuaries, this corresponds to a nitrogen loading of 40 to 95 mmol m-3 year-1. From Figures A.4a and A.4b, it would appear that Jaworski's proposed nitrogen loadings would indeed keep most estuaries from becoming eutrophic. Note that numerical models of the Chesapeake Bay suggest that current nitrogen loadings need to be reduced by 40 percent to eliminate anoxia there (Butt 1992); this reduction corresponds to a decrease in nitrogen inputs from 510 mmol m-2 year-1 (Nixon and Pilson 1983, and Table A.1) to some 300 mmol m-2 year-1 or within the range suggested by Jaworski (1981) for nitrogen-limited estuaries.
Causes of Nuisance Algal Blooms
Smayda (1989) has compiled extensive evidence in support of the hypothesis that the worldwide increase in nuisance algal blooms is related to increased nutrient availability. For instance, a 2.5-fold increase in nutrient loadings accompanied an 8-fold increase in the annual number of red-tide blooms in a harbor in Hong Kong between 1976 and 1986. Increased nutrient concentrations in the North Sea, the Baltic Sea, and in waters between Denmark and Sweden (the Skagerrak and Kattegat) have co-occurred with increased primary production and increased incidence of blooms in these waters (Smayda 1989). The green-tides which occurred in the Great South Bay of Long Island in the 1950s were also clearly associated with nitrogen loading from duck farms there (Ryther 1954), and the reduction of nutrient loadings and the opening of a channel to increase water exchange between the bay and ocean have greatly reduced these blooms (Ryther 1989). Also, nuisance algal blooms are much more likely to occur in nutrient-rich estuarine waters than in more coastal or shelf waters (Cosper 1991, Prego 1992). The new dinoflagellate discovered by Burkholder et al. (1992b), which produces toxins only in the presence of fish, seems to be stimulated by phosphorus additions.
On the other hand, there is little if any evidence to show a direct connection between either nitrogen or phosphorus concentrations and blooms of most brown-tide or red-tide organisms (Cosper 1991, Wells et al. 1991). Red-tide blooms in Florida are not correlated with concentrations of any measured form of nitrogen or phosphorus (Rounsefell and Dragovich 1966). Similarly, the brown-tide blooms of the mid-1980s along the northeastern coast of the United States did not appear to be correlated with higher levels of nitrogen or phosphorus (Cosper et al. 1989, Cosper 1991). However, it is important to note that the concentration of a nutrient at any given point of time may not be correlated with its availability to phytoplankton (Howarth 1988), and phytoplankton can grow for long periods of time off of internally stored pools of nutrients (Andersen et al. 1991).
Perhaps more importantly, it may not be the availability of nitrogen or phosphorus alone that matters in controlling nuisance algal blooms but rather the relative availability of these nutrients in comparison to silicon (Officer and Ryther 1980, Smayda 1989). When Si:N and Si:P ratios are relatively high, silicon is relatively available, favoring the growth of diatoms, which have a high requirement for silicon. However, as the Si:N and Si:P ratios decrease, competition begins to favor other algae with no silicon requirement, such as the red-tide, green-tide, and brown-tide organisms. Most silicon comes from natural sources, and as inputs of nutrients from sewage increase, the Si:N and Si:P ratios decrease (Officer and Ryther 1980). Eutrophication itself can decrease the abundance of silicon by increasing sedimenta-
tion of phytoplankton, as has been demonstrated in the Baltic Sea (Wulff et al. 1990). Where long-term nutrient data are available, the increased occurrence of nuisance algal blooms has always been found to be correlated with a decrease in Si:N and Si:P ratios (Smayda 1989 and references therein). Net primary production probably remains controlled by nitrogen or phosphorus availability throughout the range of silicon availabilities (Howarth 1988), but the relative availability of silicon may well control the abundance of diatoms versus other phytoplankton species, thereby setting the stage for nuisance blooms (Smayda 1989).
Trace-metal availability may be another factor in the initiation of many nuisance algal blooms, with high iron availability favoring the bloom-forming species. A variety of physiological studies with pure cultures and short-term enrichment studies with natural populations have shown that the requirement for iron is high for red-tide dinoflagellates (Wilson 1966, Martin and Martin 1973, Graneli et al. 1986, Doucette and Harrison 1990), for the brown-tide algae (Cosper et al. 1990), and for cyanobacteria (Wurtsbaugh and Horne 1983, Howarth et al. 1988b). Red-tide outbreaks over a 25-year period in Florida have been correlated with iron inputs from rivers (Kim and Martin 1974) although inputs of a number of other substances are undoubtedly correlated with the iron inputs (Wells et al. 1991). However, most iron is not directly available to phytoplankton, and the available fraction of iron is not correlated with the outbreak of dinoflagellate blooms in Maine (Wells et al. 1991). Proof of a critical role for iron in initiating blooms must await further study.
CONCLUSIONS
Many estuaries and coastal marine ecosystems receive excessive inputs of nutrients, and sewage effluent is often a major component of this. These excessive inputs cause eutrophication, leading to anoxic and hypoxic conditions, loss of seagrass and algal beds, and damage to coral reefs. In many temperate estuaries and coastal seas, nitrogen is the primary nutrient of concern leading to increased eutrophication. Phosphorus controls eutrophication in some coastal marine ecosystems, at least during some seasons, and may be the primarily controlling element in tropical seas. To control coastal eutrophication, both nitrogen and phosphorus need to be controlled (D'Elia et al. 1986, Howarth 1988, Graneli et al. 1990). For phosphorus, the approach used in lakes—correlational models relating phosphorus to phytoplankton in lakes—should provide an adequate strategy for determining acceptable inputs to most temperate estuaries and coastal marine ecosystems as well. Acceptable inputs for nitrogen to marine ecosystems can be developed from similar models relating nitrogen inputs to phytoplankton production or biomass. However, such approaches may not adequately protect tropical and
semi-tropical marine ecosystems that contain nutrient-sensitive corals or other ecosystems with sensitive benthic communities in shallow water.
No strong consensus exists on the extent to which excess nutrients contribute to the increasingly frequent occurrence of toxic algal blooms in many parts of the world's coastal oceans. However, to minimize problems from such blooms, it seems prudent to maintain availabilities of nitrogen and phosphorus at levels low compared to silicon availability, thereby favoring diatoms. Since increased iron availabilities may also contribute to the formation of toxic blooms, increases in the availability of iron in coastal waters should also be avoided. Factors that increase iron availability include erosion from land and increased reducing conditions in sediments from eutrophication.
REFERENCES
Andersen, T., A.K.L. Schartau, and E. Paasche. 1991. Quantifying external and internal nitrogen and phosphorus pools, as well as nitrogen and phosphorus supplied through remineralization, in coastal marine plankton by means of a dilution technique. Mar. Ecol. Progr. Ser. 69:67-80.
Anderson, D.M. 1989. Toxic algal blooms and red tides: A global perspective. In Red Tides: Biology, Environmental Science and Toxicology, T. Okaichi, D.M. Anderson, and T. Nemoto, eds. New York: Elsevier.
Baden, S.P., L.O. Loo, L. Pihl, and R. Rosenberg. 1990. Effects of eutrophication on benthic communities including fish: Swedish west coast. Ambio 19:113-122.
Banse, K. 1990. Does iron really limit phytoplankton production in the offshore subarctic Pacific? Limnol. Oceanogr. 35:772-775.
Borowitzka, M.A. 1972. Intertidal algal species diversity and the effect of pollution. Australian Journal of Marine and Freshwater Science 23:73-84.
Boynton, W.R., W.M. Kemp., and C.G. Osborne. 1980. Nutrient fluxes across the sediment-water interface in the turbid zone of a coastal plain estuary. In Estuarine Perspectives, V.S. Kennedy, ed. New York: Academic Press.
Boynton, W.R., W.M. Kemp, and C.W. Keefe. 1982. A comparative analysis of nutrients and other factors influencing estuarine phytoplankton production. In Estuarine Comparisons, V.S. Kennedy, ed. New York: Academic Press.
Bricelj, M., and S. Kuenstner. 1989. The feeding physiology and growth of bay scallops and mussels. In Novel Phytoplankton Blooms: Causes and Impacts of Recurrent Brown Tides and Other Unusual Blooms. Lecture Notes on Coastal and Estuarine Studies, E.M. Cosper, E.J. Carpenter, and V.M. Bricelj, eds. Berlin: Springer-Verlag.
Burkholder, J.M., K.M. Mason, and H.B. Glasgow. 1992a. Water-column nitrate enrichment promotes decline of eelgrass Zostera marina: Evidence from seasonal mesocosm experiments. Mar. Ecol. Prog. Ser. 81:163-178.
Burkholder, J.M., E.J. Noga, C.H. Hobbs, and H.B. Glasgow. 1992b. Now "phantom" dinoflagellate is the causative agent of major estuarine fish kills. Nature 358:407-410.
Butt, A.J. 1992. Numerical models and nutrient reduction strategies in Virginia. Coastal Management 20:25-36.
Cambridge, M.L., and A.J. McComb. 1984. The loss of seagrasses in Cockburn Sound, Western Australia. I. The time course and magnitude of seagrass decline in relation to industrial development. Aquatic Botany 20:229-242.
Cambridge, M.C., A.W. Chaffings, C. Brittan, L. Moore, and A.J. McComb. 1986. The loss of seagrass in Cockburn Sound, Western Australia. II. Possible causes of seagrass decline. Aquatic Botany 24:269-285.
Caraco, N., J.J. Cole, and G. E. Likens. 1989. Evidence for sulfate-controlled phosphorus release from sediments of aquatic systems. Nature 341:316-318.
Caraco, N., J.J. Cole, and G.E. Likens. 1990. A comparison of phosphorus immobilization in sediments of freshwater and coastal marine systems. Biogeochemistry 9:277-290.
Carpenter, E.J., J. Chang, M. Cottrell, J. Schubauer, H.W. Paerl, B.M. Bebout, and D.G. Capone. 1990. Re-evaluation of nitrogenase oxygen-protective mechanisms in the planktonic marine cyanobacterium Trichodesmium. Mar. Ecol. Prog. Ser. 65:151-158.
Carpenter, S.R., J.F. Kitchell, and J.R. Hodgson. 1985. Cascading trophic interactions and lake productivity. BioScience 35:634-639.
Carpenter, S.R., J.R. Kitchell, and J.R. Hodgson. 1987. Regulation of lake primary production by food web structure. Ecology 68:1863-1876.
Cederwall, H., and R. Elmgren. 1990. Biological effects of eutrophication in the Baltic Sea, particularly the coastal zone. Ambio 19(3):109-112.
Chesterikoff, A., B. Garban, G. Billen, and M. Poulin. 1992. Inorganic nitrogen dynamics in the River Seine downstream from Paris (France) . Biogeochemistry 17(3):147-164.
Cosper, E.M. 1991. Recent and Historical Novel Algal Blooms. Monospecific Blooms Occurred Along Northeast Coast in 1980s. Waste Management Research Report (SUNY Buffalo, SUNY Stony Brook, and Cornell Univ.) 3(2):3-6.
Cosper, E.M., C. Lee, and E.J. Carpenter. 1990. Novel "brown tide" bloom in Long Island embayments: A search for the causes. In Toxic Marine Phytoplankton, E. Graneli, B. Sundsttrom, L. Edler, and D.M. Anderson, eds. New York: Elsevier.
Cosper, E.M., W.C. Dennison, E.J. Carpenter, V.M. Bricelj, J.G. Mitchell, S.H. Kuenstner, D.C. Colflesh, and M. Dewey. 1987. Recurrent and persistent "brown tide" blooms perturb coastal marine ecosystem. Estuaries 10:284-290.
Cosper, E.M., W. Dennison, A. Milligan, E.J. Carpenter, C. Lee, J. Holzapfel, and L. Milanese. 1989. An examination of the environmental factors important to initiating and sustaining "brown tide" blooms. In Novel Phytoplankton Blooms: Causes and Impacts of Recurrent Brown Tides and Other Unusual Blooms. Lecture Notes on Coastal and Estuarine Studies , E.M. Cosper, E.J. Carpenter, and V.M. Bricelj, ed. Berlin: Springer-Verlag.
D'Elia, C.F. 1987. Nutrient enrichment of the Chesapeake Bay—Too much of a good thing. Environment 29:6-33.
D'Elia, C.F., J.G. Sanders, and W.R. Boynton. 1986. Nutrient enrichment studies in a coastal plain estuary: Phytoplankton growth in large-scale, continuous cultures. Can. J. Fish. Aquat. Sci. 43:397-406.
Dennison, W.C., G.J. Marshall, and C. Wigand. 1989. Effect of "brown tide" shading on eelgrass (Zostera marina L.) distributions. In Novel Phytoplankton Blooms: Causes and Impacts of Recurrent Brown Tides and Other Unusual Blooms. Lecture Notes on Coastal and Estuarine Studies, E.M. Cosper, E.J. Carpenter, and V.M. Bricelj, eds. Berlin: Springer-Verlag.
Doering, P.H., C.A. Oviatt, L.L. Beatty, V.F. Banzon, R. Rice, S.P. Kelly, B.K. Sullivan, and J.B. Frithsen. 1989. Structure and function in a model coastal ecosystem: Silicon, the benthos and eutrophication. Mar. Ecol. Prog. Ser. 52:287-299.
Doucette, G.J., and P.J. Harrison. 1990. Some effects of iron and nitrogen stress on the red tide dinoflagellate Gymnodinium sanguineum. Mar. Ecol. Prog. Ser. 62:293-306.
Edmondson, W.T. 1970. Phosphorus, nitrogen, and algae in Lake Washington after diversion of sewage. Science 169:690-691.
EPA (U.S. Environmental Protection Agency). 1990. Long Island Sound Study, Status Report and Interim Actions for Hypoxia Management. New York: U.S. Environmental Protection Agency.
Flett, R.J., D.W. Schindler, R.D. Hamilton, and N.E.R. Campbell. 1980. Nitrogen fixation in Canadian precambrian shield lakes. Can. J. Fish. Aquat. Sci. 37:494-505.
Frithsen, J.B., C.A. Oviatt, M.E.Q. Pilson, R.W. Howarth, and J.J. Cole. 1988. A comparison of nitrogen vs. phosphorus limitation of production in coastal marine ecosystems. EOS 69(44):1100.
Gappa, J., J. Lopez, A. Tablado, and N.H. Magaldi. 1990. Influence of sewage pollution on a rocky intertidal community dominated by the mytilid Brachidontes rodriguezi. Mar. Ecol. Prog. Ser. 63:163-175.
Gardner, W.S., S.P. Seitzinger, and J.M. Malczyk. 1991. The effects of sea salts on the forms of nitrogen released from estuarine and freshwater sediments: Does ion pairing affect ammonium flux? Estuaries 14:157-166.
Gieson, W.B.J.T., M.M. van Katwijk, and C. den Hartog. 1990. Eelgrass condition and turbidity in the Dutch Wadden Sea. Aquatic Botany 37:71-85.
Graneli, E. 1978. Algal assay of limiting nutrients for phytoplankton production in the Oresund. Vatten 2:117-128.
Graneli, E. 1981. Bioassay experiments in the Falsterbo Channel—nutrients added daily. Kieler Meeresforsch. Sonderh. 5:82-90.
Graneli, E. 1984. Algal growth potential and limiting nutrients for phytoplankton production in Oresund water of Baltic and Kattegat origin. Limnologica (Berlin) 15:563-569.
Graneli, E., H. Persson, and L. Edler. 1986. Connection between trace metals, chelators, and red tide blooms in the Laholm Bay, SE Kattegat—An experimental approach. Mar. Env. Res. 18:61-78.
Graneli, E., K. Wallstrom, U. Larsson, W. Graneli, and R. Elmgren. 1990. Nutrient limitation of primary production in the Baltic Sea area. Ambio 19:142-151.
Gray, J.S., and E. Paasche. 1984. On marine eutrophication. Mar. Pollut. Bull. 15:349-350.
Grigg, R.W. 1978. Long-term changes in rocky bottom communities of Palos Verdes. Pp. 157-184 in Coastal Water Research Project Annual Report, 1978, W. Bascom, ed. El Segundo, California: Southern California Coastal Water Research Project.
Hallegraeff, G.M., D.A. Steffensen, and R. Wetherbee. 1988. Three estuarine dinoflagellates that can produce paralytic shellfish toxins. Journal of Plankton Research 10:533-541.
Hansson, S., and L.G. Rudstam. 1990. Eutrophication and Baltic fish communities. Ambio 19:123-125.
Harris, L. 1980. Changes in intertidal algae at Palos Verdes. Pp. 35-73 in Coastal Water Research Project Biennial Report 1979-1980, W. Bascom, ed. Long Beach, California: Southern California Coastal Water Research Project.
Hecky, P.E., and P. Kilham. 1988. Nutrient limitation of phytoplankton in freshwater and marine environments: A review of recent evidence on the effects of enrichment. Limnol. Oceanogr. 33:796-822.
Howarth, R.W. 1988. Nutrient limitation of net primary production in marine ecosystems. Annual Review of Ecology & Systematics 19:89-110.
Howarth, R.W., and J.J. Cole. 1985. Molybdenum availability, nitrogen limitation, and phytoplankton growth in natural waters. Science 229:653-655.
Howarth, R.W., and R. Marino. 1990. Nitrogen-fixing cyanobacteria in the plankton of lakes and estuaries: A reply to the comment by Smith. Limnol. Oceanogr. 35:1859-1863.
Howarth, R.W., R. Marino, J. Lane, and J.J. Cole. 1988a. Nitrogen fixation in freshwater, estuarine, and marine ecosystems. 1. Rates and importance. Limnol. Oceanogr. 33:669-687.
Howarth, R.W., R. Marino and J.J. Cole. 1988b. Nitrogen fixation in freshwater, estuarine, and marine ecosystems. 2. Biogeochemical controls. Limnol. Oceanogr. 33:688-701.
Jaworski, N.B. 1981. Sources of nutrients and the scale of eutrophication problems in estuaries. In Estuaries and Nutrients, B.J. Neilson and L.E. Cronin, eds. New York: Humana.
Jensen, L.M., K. Sand-Jensen, S. Marcher, and M. Hansen. 1990. Plankton community respiration along a nutrient gradient in a shallow Danish estuary. Mar. Ecol. Prog. Ser. 61:75-85.
Kamp-Nielsen, L. 1974. Mud-water exchange of phosphorus and other ions in undisturbed sediment cores and factors affecting the exchange rate. Arch. Hydrobiol. 13:218-237.
Khalid, R.A., W.H. Patrick, and R.D. DeLaune. 1977. Phosphorus sorption characteristics of flooded soils. Soil Sci. Soc. Am. J. 41:305.
Kelly, J., and S. Levin. 1986. A comparison of aquatic and terrestrial nutrient cycling and production processes in natural ecosystems, with reference to ecological concepts of relevance to some waste disposal issues. In The Role of Oceans as a Waste Disposal Option, G. Kullenber, ed. Amsterdam: Reidel.
Kemp, W.M., R.R. Twilley, J.C. Stevenson, W.R. Boynton, and J.C. Means . 1983. The decline of submerged vascular plants in upper Chesapeake Bay: Summary of results concerning possible causes. J. Mar. Technol. Soc. 17:78-85.
Kim, Y.S., and D.F. Martin. 1974. Interrelationship of Peace River parameters as a basis of the iron index: A predictive guide to the Florida red tide. Wat. Res. 8:607-616.
Kirkman, R.H. 1976. A Review of the Literature on Seagrass Related to its Decline in Moreton Bay, Qld. CSIRO Report no. 64. CSIRO.
Lapointe, B.E., M.M. Littler, and D.S. Littler. 1987. A comparison of nutrient-limited productivity in macroalgae from a Caribbean barrier reef and from a mangrove ecosystem. Aquatic Botany 28:243-255.
Larsson, U.R., R. Elmgren, and F. Wulff. 1985. Eutrophication and the Baltic Sea: Causes and consequences. Ambio 14:10-14.
Lein, A.Y., and M.V. Ivanov. 1992. Interaction of carbon, sulphur, and oxygen cycles in continental and marginal seas. In Sulphur Cycling on the Continents: Wetlands, Terrestrial Ecosystems, and Associated Water Bodies, R.W. Howarth, J.W.B. Stewart, and M.V. Ivanov, eds. Chichester, U.K.: Wiley & Sons.
Lindahl, G., and K. Wallstrom. 1985. Nitrogen fixation (acetylene reduction) in planktonic cyanobacteria in Oregrundsgrepen, SW Bothnian Sea. Arch. Hydrobiol. 104:193-204.
Littler, M.M., and S.N. Murray. 1975. Impact of sewage on the distribution, abundance and community structure of rocky intertidal macro-organisms. Mar. Biol. 30:277-291.
Littler, M.M., and S.N. Murray. 1978. Influence of domestic wastes on energetic pathways in rocky intertidal communities. J. Appl. Ecol. 15:583-596.
Littler, M.M., D.S. Littler, and B.E. Lapointe. 1988. A comparison of nutrient- and lightlimited photosynthesis in psarnmophyfic versus epilithic forms of Halimeda (caulerpales, halimedaceae) from the Bahamas. Coral Reefs 6:219-225.
Malone, T.C. 1982. Factors influencing the fate of sewage-derived nutrients in the lower Hudson estuary and New York bight. In Ecological Stress and the New York Bight: Science and Management, G.F. Mayer, ed. Columbia, South Carolina: Estuarine Research Federation.
Marino, R., R.W. Howarth, J. Shamess, and E.E. Prepas. 1990. Molybdenum and sulfate as controls on the abundance of nitrogen-fixing cyanobacteria in saline lakes in Alberta. Liminol. Oceanogr. 35:245-259.
Martin, D.F., and B.B. Martin. 1973. Implications of metal organic interactions in red tide outbreaks. In Trace Metals and Metal-organic Interactions in Natural Waters, P.C. Singer, ed. Ann Arbor, Michigan: Ann Arbor Science.
McComb, A.J., R.P. Atkins, P.B. Birch, D.M. Gordon, and R.J. Luketelich. 1981. Eutrophication in the Peel-Harvey Estuarine System, Western Australia. In Estuaries and Nutrients, B.J. Nielson and L.E. Cronin, eds. New York: Humana.
McGlathery, K.J., R.W. Howarth, and R. Marino. 1992. Nutrient limitation of the macroalga,
Penicillus capitatus, associated with subtropical seagrass meadows in Bermuda. Estuaries 15:18-25
.Mearns, A.J., and J.Q. Word. 1982. Forecasting the effects of sewage solids on marine benthic communities. Pp. 495 in Ecological Stress in the New York Bight: Science and Management, G.F. Mayer, ed. Columbia, South Carolina: Estuarine Research Federation.
Mearns, A.J., E. Haines, G.S. Klepple, R.A. McGrath, J.J.A. McLaughlin, D.A. Segar, J.H. Sharp, J.J. Walsh, J.Q. Word, D.K. Young, and M.W. Young. 1982. Effects of nutrients and carbon loadings on communities and ecosystems. In Ecological Stress and the New York Bight: Science and Management, G.F. Mayer, ed. Columbia, South Carolina: Estuarine Research Federation.
Molot, L.A., and P.J. Dillon. 1991. Nitrogen/phosphorus ratios and the prediction of chlorophyll in phosphorus-limited lakes in central Ontario. Can. J. Fish. Aquat. Sci. 48:140-145.
Morin. A., K.D. Hambright, N.G. Hairston, D.M. Sherman, and R.W. Howarth. 1991. Consumer control of gross primary production in replicate freshwater ponds. International Vereinigung fuer Theoretische und Angewandte Limnologie. Verhandlungen IVTLAP 24(3):1512-1516.
Morse, J.W., J.J. Zullig, L.D. Bernstein, F.J. Millero, P. Milne, A. Mucci, and G.R. Choppin. 1985. Chemistry of calcium carbonate-rich shallow water sediments in the Bahamas. Am. J. Sci. 285:147-185.
Myers, V.B., and R.I. Iverson. 1981. Phosphorus and nitrogen limited phytoplankton productivity in northeastern Gulf of Mexico coastal estuaries . In Estuaries and Nutrients, B.J. Nielson, and L.E. Cronin, eds. New York: Humana.
Newell, R.I.E. 1988. Ecological changes in Chesapeake Bay: Are they the result of over harvesting the American oyster, Crassotrea virginica? Pp. 29-31 in proceedings of Understanding the Estuary: Advances in Chesapeake Bay Research, March 1988. Publication 129. Baltimore, Maryland: Chesapeake Research Consortium.
Nixon, S.W. 1988. Physical energy inputs and the comparative ecology of lake and marine ecosystems. Limnol. Oceanogr. 33:1005-1025.
Nixon, S.W. 1992. Quantifying the relationship between nitrogen input and the productivity of marine ecosystems. Adv. Mar. Techn. Conf. 5:57-83.
Nixon, S.W., and M.E.Q. Pilson. 1983. Nitrogen in estuarine and coastal marine ecosystems. In Nitrogen in the Marine Environment, E.J. Carpenter and D.G. Capone, eds. New York: Academic Press.
Nixon, S.W., J.R. Kelly, B.N. Fumas, C.A. Oviatt, and S.S. Hale. 1980. Phosphorus regeneration and the metabolism of coastal marine bottom communities. In Marine Benthic Dynamics, K.R. Tenore and B.C. Coull, eds. Columbia, South Carolina: University of South Carolina Press.
Nixon, S.W., C. Oviatt, J. Frithsen, and B. Sullivan. 1986. Nutrients and productivity of estuaries and coastal marine ecosystems. J. Limnol. Soc. S. Afr. 12:43-71.
National Oceanographic and Atmospheric Administration and the U.S. Environmental Protection Agency (NOAA/EPA). 1988. Strategic Assessment of Near Coastal Waters: Northeast Case Study. Susceptibility and Status of Northeast Estuaries to Nutrient Discharges. Rockville, Maryland: National Oceanic and Atmospheric Administration.
Norin, L.L. 1977. 14C-bioassays with the natural phytoplankton in the Stockholm archipelago. Ambio Spec. Rep. 5:15-21.
Officer, C.B., and J.H. Ryther. 1980. The possible importance of silicon in marine eutrophication. Mar. Ecol. Prog. Ser. 3:83-91.
Officer, C.B., R.B. Biggs, J. Taft, L.E. Cronin, M.A. Tyler, and W.R. Boynton. 1984. Chesapeake Bay anoxia: Origin, development, and significance. Science 223:22-27.
Paerl, H.W., and R.C. Carlton. 1988. Control of nitrogen fixation by oxygen depletion in surface-associated microzones. Nature 332:260-262.
Paerl, H.W., K.M. Crocker, and L.E. Prufert. 1987. Limitation of N2 fixation in coastal marine waters: Relative importance of molybdenum, iron, phosphorous, and organic matter availability. Limnol. Oceanogr. 32:525-536.
Parker, C.A., and J.E. O'Reilly. 1991. Oxygen depletion in Long Island Sound: A historical perspective. Estuaries 14:248-264.
Pastorak, R.A., and G.R. Bilyard. 1985. Effects of sewage pollution on coral-reef communities. Mar. Ecol. Prog. Ser. 21:175-189.
Prego, R. 1992. Flows and budgets of nutrient salts and organic carbon in relation to a red tide in the Ria of Vigo (NW Spain). Marine Ecology Progress Series 79:289-302.
Price, K.S., D.A. Flemer, J.L. Taft, and G.B. Mackierrnan. 1985. Nutrient enrichment of Chesapeake Bay and its impact on the habitat of striped bass: A speculative hypothesis. Trans. Am. Fish. Soc. 114:97-106.
Powell, G.V.N., W.J. Kenworthy, and J.F. Fourqurean. 1989. Experimental evidence for nutrient limitation of seagrass growth in a tropical estuary with restricted circulation . Bull. Mar. Sci. 44:324-340.
Redfield, A.C. 1958. The biological control of chemical factors in the environment. Am. Sci. 46:205-221.
Robblee, M.B., T.R. Barber, P.R. Carlson, M.J. Durako, J.W. Fourqurean, L.K.Muehlstein, D. Porter, L.A. Yarbro, R.T. Zieman, and J.C. Zieman. 1991. Mass mortality of the tropical seagrass Thalassia testudinum in Florida Bay (USA). Mar. Ecol. Prog. Ser. 71:297-299.
Robineau, B., J.A. Gagne, L. Fortier, and A.D. Cembella. 1991. Potential impact of a toxic dinoflagellate (Alexandrium excovatum) bloom on survival of fish and crustacean larvae. Marine Biology 108:293-301.
Rosenberg, R. 1985. Eutrophication-The future marine coastal nuisance? Mar. Poll. Bull. 16:227-231.
Rosenberg, R., R. Elmgren, S. Fleischer, P. Jonsson, G. Persson, and H. Dahlin. 1990. Marine eutrophication case studies in Sweden. Ambio 19:102-108.
Rounsefell, G.A., and A. Dragovich. 1966. Correlation between oceanographic Rfactors and abundance of the Florida redtide (Gymnodiniwn breve Davis), 1954-1961. Bull. Mar. Sci. 16:402.
Rowe, G.T., C.H. Cliffer, K.L. Smith, and P.L. Hamilton. 1975. Benthic nutrient regeneration and its coupling to primary productivity in coastal waters. Nature 225:215-217.
Rydberg, L., L. Edler, S. Floderus, and W. Graneli. 1990. Interaction between supply of nutrients, primary production, sedimentation and oxygen consumption in SE Kattegat. Ambio 19:134-141.
Ryther, J.H. 1954. The ecology of phytoplankton blooms in Moriches Bay and Great South Bay, Long Island, New York. Biol. Bull. 106:198-209.
Ryther, J.H. 1989. Historical perspective of phytoplankton blooms on Long Island and the green tides of the 1950's. In Novel Phytoplankton Blooms: Causes and Impacts of Recurrent Brown Tides and Other Unusual Blooms. Lecture Notes on Coastal and Estuarine Studies, E.M. Cosper, E.J. Carpenter, and V.M. Bricelj, eds. Berlin: Springer-Verlag.
Ryther, J.H., and W.M. Dunstan. 1971. Nitrogen, phosphorus and eutrophication in the coastal marine environment. Science 171:1008-1012.
Schindler, D.W. 1977. Evolution of phosphorus limitation in lakes. Science 195: 260-262.
Schindler, D.W. 1978. Factors regulating phytoplankton production and standing crop in the world's freshwaters. Limnol. Oceanogr. 23:478-486.
Schindler, D.W. 1981. Studies of eutrophication in lakes and their relevance to the estuarine environment. In Estuaries and Nutrients, B.J. Neilson and L.E. Cronin, eds. New York: Humana.
Schindler, D.W., R. Hesslein, and G. Kipphut. 1977. Interactions between sediments and overlying waters in an experimentally eutrophied precambrian shield lake. In Interactions Between Sediments and Fresh Water, H.L. Golttemian, ed. Junk, The Hague.
Seitzinger, S.P. 1988. Denitrification in freshwater and marine ecosystems: Ecological and geochemical significance. Limnol. Oceanog. 33:702-724.
Seitzinger, S.P., W.S. Gardner, and A.K. Spratt. 1991. The effect of salinity on ammonium sorption in aquatic sediments: Implications for benthic nutrient cycling . Estuaries 14:167-174.
Short, F.T., W.C. Dennison, and D.G. Cappone. 1990. Phosphorus-limited growth of the tropical seagrass Syringodiumfiliforme in carbonate sediments. Mar. Ecol. Prog. Ser. 62:169-174.
Short, F.T., M. W. Davis, R.A. Gibson, and C.F. Zimmerman. 1985. Evidence for phosphorus limitation in carbonate sediments of the seagrass Syringodiumfiliforme. Estuarine, Coastal and Shelf Science. 20:419-430.
Sieburth, J., P.W. Johnson, and P.E. Hargraves. 1988. Ultrastructure and ecology of Aureococcus anophagefferens gen. et sp. nov. (Chrysophyceae); the dominant picoplankter during a bloom in Narragansett Bay, Rhode Island, summer 1985. J. Phycol. 24:416-425.
Smayda, T.J. 1974. Bioassay of the growth potential of the surface water of lower Narragansett Bay over an annual cycle using the diatom Thalassiosira pseudonana (oceanic clone, 131). Limnol. Oceanogr. 19:889-901.
Smayda, T.J. 1989. Primary production and the global epidemic of phytoplankton blooms in the sea: A linkage? Pp. 449-483 in Novel Phytoplankton Blooms: Causes and Impacts of Recurrent Brown Tides and Other Unusual Blooms. Lecture Notes on Coastal and Estuarine Studies, E.M. Cosper, E.J. Carpenter, and V.M. Bricelj, ed. Berlin: Springer-Verlag.
Smayda, T.J. 1992. A phantom of the ocean. Nature 358:374-375.
Smith, V.H. 1979. Nutrient dependence of primary productivity in lakes. Limnol. Oceanogr. 24:1051-1064.
Smith, S.V. 1981. Responses of Kaneohe Bay, Hawaii, to relaxation of sewage stress. In Estuaries and Nutrients, B.J. Neilson and L.E. Cronin, eds. New York: Humana.
Smith, S.V. 1984. Phosphorus vs. nitrogen limitation in the marine environment. Limnol. Oceanogr. 29:1149-1160.
Smith, S.V., and M.J. Atkinson. 1984. Phosphorus limitation of net production in a confined aquatic ecosystem. Nature 207:626-627.
Steele, J.H. 1974. The Structure of Marine Ecosystems. Cambridge, Massachusetts:Harvard University Press.
Tracey, G.A., R.L. Steele, J. Gatzke, D.K. Phelps, R. Nuzzi, M. Waters, and D.M. Anderson. 1989. Testing and application of biomonitoring methods for assessing environmental effects of noxious algal blooms. In Novel Phytoplankton Blooms: Causes and Impacts of Recurrent Brown Tides and Other Unusual Blooms. Lecture Notes on Coastal and Estuarine Studies, E.M. Cosper, E.J. Carpenter, and V.M. Bricelj, eds. Berlin: Springer-Verlag.
Twilley, R.R., W.M. Kemp, K.W. Staver, J.C. Stevenson, and W.R. Boynton. 1985. Nutrient enrichment of estuarine submerged vascular plant communities. 1. Algal growth and effects on production of plants and associated communities. Mar. Ecol. Prog. Ser. 23:179-191.
Valiela, I. 1984. Marine Ecological Processes. New York: Springer-Verlag.
Valiela, I., Costa, J., Foreman, K., Teal, J.M., Howes, B., and Aubrey, B. 1990. Transport of groundwater-borne nutrients from watersheds and their effects on coastal waters. Biogeochemistry 10:177-197.
Vince, S., and I. Valiela. 1973. The effects of ammonium and phosphate enrichment on
chlorophyll a, a pigment ratio and species composition of phytoplankton of Vineyard Sound. Mar. Biol. 19:69-73
.Vitousek, P.M., and R.W. Howarth. 1991. Nitrogen limitation on land and in the sea: How can it occur'? Biogeochemistry 13:87-115.
Vollenweider, R.A. 1976. Advances in defining critical loading levels for phosphorus in lake eutrophication. Mem. Ist. Ital. Idrobiol. 33:53-83.
Vollenweider, R.A. 1979. Das Nahrstoffbelastungskonzept als Grundlage fur den externen Eingriff in den Eutrophierungsprozess stehender Gewasser und Talsperren. Z. Wasser-u. Abwasser-Forschung 12:46-56.
Wells, M.L., L.M. Mayer, and R.R.L. Guillard. 1991. Evaluation of iron as a triggering factor for red tide blooms. Mar. Ecol. Prog. Ser. 69:93-102.
Wetzel, R.G. 1983. Limnology. Philadelphia, Pennsylvania: Saunders.
Wilson, W.B. 1966. The Suitability of Seawater for the Survival and Growth of Gymnodinium breve Davis, and Some Effects of Phosphorus and Nitrogen on its Growth. Professional Paper Series, Marine Laboratory Florida 7:1-42.
Wilson, K., A.J. Mearns, and J.J. Grant. 1980. Changes in kelp forests at Palos Verdes. Pp. 77-92 in Coastal Water Research Project Biennieal Report 1979-1980, W. Bascom, ed. Long Beach. CA: Southern California Coastal Water Research Project.
Wulff, F., A. Stigebrandt, and L. Rahm. 1990. Nutrient dynamics of the Baltic Sea. Ambio 19:126-133.
Wurtsbaugh, W.A., and A.J. Horne. 1983. Iron in eutrophic Clear Lake, California: Its importance for algal nitrogen fixation and growth. Can. J. Fish. Aquat. Sci. 40:1419-1429.


























