8
The Effect of Exercise and Heat on Vitamin Requirements
Priscilla M. Clarkson1
INTRODUCTION
Vitamins are essential nutrients that have a wide variety of functions. The fact that many vitamins play a critical role in energy production has captured the attention of those interested in ways to optimize exercise or work performance. Moreover, increased energy production during exercise could lead to an increased vitamin requirement for those individuals who participate in rigorous physical training.
Because vitamins are essential, generally cannot be manufactured by the body, and must be ingested on a regular basis, it has been tempting to suggest that if a little is good, more is better. This reasoning was perhaps the impetus for many studies that have assessed the effects of vitamin supplementation on physical performance (Robinson and Robinson, 1954). A review of early studies suggested that vitamins were lost to a significant degree in sweat (Robinson and Robinson, 1954). For this reason, exercise—especially in hot environments—was considered to result in vitamin deficiencies. Now it is generally agreed that the vitamin loss in sweat is negligible (Brotherhood, 1984; Mitchell and Edman, 1951; Robinson and Robinson, 1954) (Table 8-1). However, some vitamins have been implicated to have beneficial effects for those individuals living and working in a hot environment.
TABLE 8-1 Concentration of Vitamins Lost in Sweat
|
Vitamin |
Concentration (µg per 100 ml) |
|
Thiamin |
0–15 |
|
Riboflavin |
0.5–12 |
|
Nicotinic acid (total) |
8–14 |
|
Pantothenic acid |
4–30 |
|
Ascorbic acid |
0–50 |
|
Pyridoxine |
7 |
|
Folic acid (plus metabolites) |
0.26 |
|
SOURCE: Mitchell and Edman (1951). Data based on ranges reported from several studies completed in the 1940s. |
|
This chapter addresses whether those individuals who expend greater amounts of energy in exercise training or work require greater amounts of vitamins and whether vitamin supplementation will enhance exercise performance. Some of this information has also been covered in a previous paper (Clarkson, 1991). This chapter also examines whether exercise in a hot environment will lead to an increased requirement for certain vitamins and whether vitamin supplements will reduce heat stress.
Vitamins are classified as either water soluble or fat soluble. Watersoluble vitamins are the B complex vitamins and vitamin C. These are stored in relatively small amounts in the body and cannot be retained for long periods. If blood levels of water-soluble vitamins exceed renal threshold, they are excreted into the urine. Most water-soluble vitamins serve major functions of either energy production or hematopoiesis. With the exception of vitamin K, fat-soluble vitamins are stored in greater amounts than the water-soluble vitamins. Fat-soluble vitamins are absorbed and transported in the body in close association with lipids and have roles that are largely independent of energy production.
WATER-SOLUBLE VITAMINS
Vitamin B complex consists of eight vitamins: vitamin B1 (thiamin), vitamin B2 (riboflavin), niacin, vitamin B6 (pyridoxine), vitamin B12 (cyanocobalamin), biotin, folic acid, and pantothenic acid. The quantity stored differs among the vitamins. For example, if an individual's diet is deficient in most of the B vitamins, clinical symptoms can sometimes occur in 3 to 7 days (Guyton, 1986). Vitamin B12 is an exception because it can be stored in the liver for a year or longer. The B vitamins, except B12 and folic acid, primarily serve as coenzymes in the metabolism of glucose and fatty
acids. Vitamin C serves many diverse functions in the body. A vitamin C-deficient diet can cause deficiency symptoms after a few weeks and can cause death from scurvy in 5 to 7 months (Guyton, 1986).
In the following discussion, these topics will be addressed for each vitamin: its function, how an individual's status is determined, changes in status by chronic exercise, effects of restriction or supplementation on performance, and relationship to heat stress.
Thiamin
The importance of thiamin ingestion was noted in the late nineteenth century when it was found that adding meat and whole grain to sailors' diets aboard ship prevented the condition known as beriberi (Brown, 1990). Thiamin is absorbed from the small intestine, and some is phosphorylated to form pyrophosphate (the coenzyme form). Pyrophosphate and free thiamin are transported via the blood to tissues, with the highest concentrations occurring in the liver, kidney, and heart. Most thiamin is stored in the pyrophosphate form.
Thiamin plays a role in carbohydrate metabolism. It functions specifically as a coenzyme in the conversion of pyruvate to acetyl coenzyme A (CoA) and alpha-ketoglutarate to succinyl CoA, as well as the transketolase reaction of the pentose phosphate pathway.
A sensitive technique for assessing thiamin status is the use of an erythrocyte enzyme stimulation test performed on blood samples. Erythrocyte transketolase activity is assessed before and after addition of thiamin pyrophosphate (TPP). If a deficiency of TPP exists, then adding TPP to the blood will increase the activity of the enzyme. The level of erythrocyte TPP is also used to determine thiamin status. Sauberlich et al. (1979) reported that urinary excretion of thiamin was a reasonably reliable indicator of thiamin nutritional status, although its use has been questioned (Gubler, 1984).
Whether physical exercise, because of the greater metabolic challenge, will increase the need for thiamin has not been fully established. The few studies that have assessed possible biochemical deficiencies of athletes have reported minimal evidence of thiamin deficiency compared with controls (Cohen et al., 1985; Guilland et al., 1989; Weight et al., 1988). Nijakowski (1966) found that blood levels of thiamin were lower in male athletes compared with a control group, however, it is possible that the lower levels were due to plasma volume expansion in athletes. Athletes were also tested after a 12-km skiing expedition, and thiamin levels showed a further decrease, which Nijakowski (1966) suggested was due to increased bodily requirements.
The National Research Council (1989) recommended that thiamin intake be proportional to caloric intake such that 0.5 mg per 1000 kcal is
required. Because of the increased energy demands of exercise, athletes ingest more food. However, many athletes are ingesting a greater proportion of carbohydrates, and it has been shown that some athletes have a high intake of refined carbohydrates with low vitamin content (Brouns and Saris 1989). Furthermore, carbohydrate loading regimens can result in low thiamin intakes. In the 1979 Tour de France, thiamin intakes were found to be too low (0.26 mg per 1000 kcal), which was attributed to the high carbohydrate meals. Van Erp-Baart et al. (1989) also pointed out that when energy intake is high, the amount of refined carbohydrate is high, and the nutrient density of thiamin drops.
Because of the role of thiamin in energy metabolism, it would seem that thiamin deficiency would lead to decrements in exercise performance. However, although thiamin-deficient diets along with deficiencies in other B complex vitamins were shown to adversely affect performance (for review see Van der Beek, 1985), there is some controversy concerning whether thiamin deficiency alone will alter performance (Williams, 1976, 1989). Wood et al. (1980) in a well-controlled study found that performance was not affected by induced thiamin deficiency. They reported no significant difference in time to exhaustion during a cycle ergometry test between subjects who ingested a low-thiamin diet (500 ºg thiamin) for 4 to 5 weeks along with a placebo (without thiamin) and subjects who had ingested the same low-thiamin diet along with a thiamin supplement (5 mg thiamin).
Few studies have assessed the effect of thiamin supplementation on exercise performance (see Keith, 1989). In two controlled studies, the effects of thiamin supplements of 5 mg per day for 1 week on an arm endurance task (Karpovich and Millman, 1942) and 0.1 mg daily for 10 to 12 weeks on grip strength and treadmill tests (aerobic and anaerobic work) (Keys et al., 1943) were examined. Both studies found that the supplement had no effect on any measure of work performance.
Mills (1941) studied the effects of heat stress on young rats and found that optimal thiamin intake for growth was increased at high temperature (91°F), although these results were not confirmed by later studies (Edison et al., 1945). Based on his own findings, however, Mills (1941) suggested that thiamin supplements should make workers in boiler or furnace rooms or in other types of heat exposure more resistant to heat effects. Other studies found that an increase in environmental temperature resulted in a decreased thiamin requirement (Edison et al., 1945), and this decrease reflected the decrease in caloric requirements at elevated temperatures. However, the animals in that study were not exercising. It has been shown that exercise in the heat is more metabolically costly perhaps because of the extra energy costs of sweating, circulation, and respiratory mechanisms (Nielsen et al., 1990). If increased caloric intake is needed for those working in a hot environment, then thiamin intake should be increased proportionally.
Thiamin loss in sweat is considered to be around 10 µg per 100 ml (Table 8-1). Working in a hot environment can produce sweat losses of up to 10 liters per day. At this value, the amount of thiamin lost would be about 1.0 mg. Although a well-balanced diet could probably satisfy this need, there should be some concern if the diet is poor or if the thiamin requirement is not increased with an increase in energy intake (to meet the demands of work).
Riboflavin
The coenzyme forms of riboflavin are flavin mononucleotide (FMN) and flavin adenine dinucleotide (FAD). These coenzymes function in cellular oxidation, specifically acting as hydrogen carriers in the mitochondrial electron transport system. Deficiencies in riboflavin are common in many Third World countries and occur invariably with deficiencies in the other water-soluble vitamins (McCormick, 1990).
Riboflavin status can be assessed reliably from blood samples. A sensitive indicator is the measurement of erythrocyte glutathione reductase (EGR) activity (Cooperman and Lopez, 1984). When riboflavin stores are low, EGR loses its saturation with FAD, and its activity drops (Cooperman and Lopez, 1984).
Whether chronic exercise alters riboflavin status is not certain. For the general U.S. population and most athlete groups studied, biochemical deficiencies of riboflavin are rare (Cohen et al., 1985; Guilland et al., 1989; Tremblay et al., 1984). However, one study found inadequate riboflavin status in 8 out of 18 athletes studied (Haralambie, 1976). It has been suggested that exercise training may increase the need for riboflavin. Belko et al. (1983) found that the need for riboflavin in healthy young women (based on an estimation of riboflavin intake required to achieve normal biochemical status) increased when they participated in jogging exercise for 20 to 50 minutes a day. Because biochemical deficiencies in athletes are rare, the increased need for riboflavin probably would be easily met by diet.
Because of the importance of riboflavin to oxidative energy production, performance could be impaired by a riboflavin deficiency. Keys et al. (1944) placed six male students on a riboflavin-restricted diet (99 mg per day or 0.31 mg per 1000 kcal) for 84 days (n = 3) and 152 days (n = 3). Subjects performed an aerobic walking test (60 minutes) and an anaerobic test (60 seconds) on the treadmill and performed grip strength tests before, every 2 weeks during, and after the restricted-diet period. The low-riboflavin diet did not adversely alter the performance measures. Van der Beek (1985) reviewed other studies on riboflavin restriction and concluded that riboflavin depletion did not alter work performance on submaximal treadmill tests.
Because studies have shown that riboflavin deficiency does not alter
exercise performance, it would seem that supplementation should not enhance performance. Belko et al. (1983) studied the effects of riboflavin supplementation in two groups of overweight women who participated in a 12-week exercise program. One group ingested a total of 0.96 mg per 1000 kcal riboflavin per day, and the other group ingested 1.16 mg per 1000 kcal per day. The improvement in aerobic capacity did not differ between groups. Also no difference in exercise performance was found when elite swimmers were supplemented with 60 mg per day of riboflavin for 16 to 20 days (Tremblay et al., 1984).
Tucker et al. (1960) studied the effects of exercise and heat stress on riboflavin excretion into the urine. In one experiment, men walked on a treadmill for 4 to 6 hours per day for six days with the temperature of the heat chamber at 49°C. The men spent a total of 10 hours per day at this temperature. Riboflavin excretion increased gradually over the course of the six days. The authors concluded that there could be a decreased requirement of riboflavin at high temperatures.
The limited data available suggest that the riboflavin requirement may be increased by exercise. However, these needs must be easily met by athletes' diets because athletes have not been shown to have a riboflavin deficiency. The one study concerning exercise and heat stress suggests that there could be a decrease in riboflavin requirement. Further study is needed to confirm this. The amount of riboflavin lost in sweat is small (Table 8-1) and should not be a problem for those working in a hot environment and profusely sweating. The recommended intake of riboflavin is linked to caloric intake (0.6 mg per 1000 kcal), and to be safe, this recommendation should be followed by people living and working in a hot environment.
Niacin
Niacin is the term used to describe nicotinic acid (niacin) and nicotinamide (niacinamide). In the body, niacin is an essential component of two coenzymes: nicotinamide adenine dinucleotide (NAD) and nicotinamide adenine dinucleotide phosphate (NADP). These coenzymes serve as electron carriers or hydrogen donors/acceptors in glycolysis, fatty acid oxidation, and the electron transport system. Severe niacin deficiency results in the condition known as pellagra (raw skin), which was common in the United States in the early 1900s but has virtually disappeared from industrialized countries (Swendseid and Swendseid, 1990).
Two available studies of niacin nutriture of athletes suggest that athletes are not deficient in niacin (Cohen et al., 1985; Weight et al., 1988). These studies used nicotinic acid or niacin levels in the blood to determine status—a questionable assessment technique because niacin and niacin metabolites in the plasma are quite low (Hankes, 1984; Swendseid and Swendseid,
1990). Erythrocyte NAD concentration or levels of 2-pyridone may be more sensitive indicators of niacin depletion (Swendseid and Swendseid, 1990).
Some evidence suggests that exercise may increase the niacin requirement (Keith, 1989). Because most adult athletes have shown no evidence of niacin deficiency, the increased requirement probably is satisfied by the athlete's diet. Chronic ingestion of niacin above the recommended dietary allowance (RDA) (National Research Council, 1989) is not recommended, because large doses are often associated with undesirable side effects, such as flushing, liver damage, increased serum uric acid levels, skin problems, and elevated plasma glucose levels (Hunt and Groff, 1990). Niacinamide in large doses is not harmful. Acute ingestion of nicotinic acid (3 to 9 g per day) has also been shown to prevent the release of fatty acids (Keith, 1989; National Research Council, 1989), which may adversely affect endurance performance.
In a double-blind placebo-controlled experiment, Hilsendager and Karpovich (1964) found that 75 mg of niacin had no effect on arm or leg endurance capacity. Bergstrom et al. (1969) compared the perception of a work load before and after subjects were given niacin, 1 g intravenously and 0.6 g perorally. After the supplementation, the subjects perceived the work load to be heavier. Niacin can decrease free fatty acid mobilization (Carlson and Oro, 1962; Williams, 1989), which may explain the negative effects of the niacin supplement. A decrease in free fatty acid mobilization would force the muscle to rely more on its muscle glycogen stores. In fact, Bergstrom et al. (1969) found that muscle glycogen content was lower in postexercise biopsy samples taken from subjects who had received the niacin supplements than with control subjects.
The only information with regard to niacin requirements in a hot environment comes from an early study that found that nicotinic acid was lost in the sweat in significant amounts (100 µg per 100 ml; Mickelsen and Keys, 1943). However, later studies did not agree with this finding (Mitchell and Edman, 1951; Robinson and Robinson, 1954). Nicotinic acid is considered to be lost in concentrations of 20 µg or less per 100 ml of sweat (Mitchell and Edman, 1951). As with thiamin and riboflavin, niacin intake should be proportional to energy intake (6.6 mg niacin per 1000 kcal). If energy intake is increased to meet the demands of exercise or work in a hot environment, then niacin should be increased as well.
Vitamin B6(Pyridoxine)
Vitamin B6 is composed of three natural compounds—pyridoxine, pyridoxamine, and pyridoxal (Merrill and Burnham, 1990)—that function in protein hemoglobin, myoglobin, and cytochromes. The coenzyme form of B6 is and amino acid metabolism; in gluconeogenesis; and in formation of
pyridoxal 5'-phosphate (PLP) and is used by over 60 enzymes. Glycogen phosphorylase, an enzyme involved in the breakdown of muscle glycogen, requires PLP as a coenzyme. Moreover, glycogen phosphorylase may serve as a reservoir for vitamin B6 storage (Merrill and Burnham, 1990) and release PLP into the circulation for use by other tissues.
Vitamin B6 status in blood samples can be assessed in several ways (Driskell, 1984). The method of choice is to assess levels of PLP, the most active form of vitamin B6 (Driskell, 1984). Chronic exercise does not appear to result in a vitamin B6 deficiency. Although biochemical deficiencies for vitamin B6 were found in 17 to 35 percent of male college athletes, similar percentages were found for the control group (Guilland et al., 1989). However, the athletes had a greater intake of vitamin B6 compared with the control subjects. Adequate vitamin B6 levels were found from assessments of blood samples of other groups of athletes (Cohen et al., 1985; Weight et al., 1988).
Although it seems that vitamin B6 status is not altered by chronic exercise, some studies have shown that acute exercise can alter the blood levels. Leklem and Shultz (1983) found that a 4500-m run substantially increased the blood levels of PLP in trained adolescent males. Hatcher et al. (1982) and Manore and Leklem (1988) reported an increase in blood levels of PLP after a 50-minute and after a 20-minute cycling exercise. PLP levels returned to baseline values after only 30 minutes rest (Manore and Leklem, 1988). It was suggested (Leklem and Shultz, 1983; Manore and Leklem, 1988) that PLP may be released from muscle glycogen phosphorylase during exercise so that PLP could be used as a cofactor for gluconeogenesis elsewhere in the body.
Holmann et al. (1991) also found that prolonged treadmill running (2 hours at 60 to 65 percent of ![]() ) resulted in significant increases in blood levels of PLP that were independent of changes in plasma volume, blood glucose, blood free fatty acid levels, and blood enzyme levels. The authors suggested that the increase in plasma PLP could be due to a release of vitamin B6 from the liver to be used in skeletal muscle to fully saturate glycogen phosphorylase or be used for other critical PLP-dependent reactions (for example, aminotransaminase reactions).
) resulted in significant increases in blood levels of PLP that were independent of changes in plasma volume, blood glucose, blood free fatty acid levels, and blood enzyme levels. The authors suggested that the increase in plasma PLP could be due to a release of vitamin B6 from the liver to be used in skeletal muscle to fully saturate glycogen phosphorylase or be used for other critical PLP-dependent reactions (for example, aminotransaminase reactions).
Another study found that 4-pyridoxic acid excretion in urine was significantly lower in trained athletes compared with controls after a vitamin B6 challenge (Dreon and Butterfield, 1986). The authors suggested that these results reflect a greater storage capacity in athletes so that 4-pyridoxic acid could be available for redistribution with increased need.
Supplementation with vitamin B6 does not appear to enhance performance. Lawrence et al. (1975a) examined swimming performance of trained swimmers who ingested 51 mg of pyridoxine hydrochloride or a placebo
daily for 6 months. No significant difference was found between the groups on 100-yard swimming times.
Because vitamin B6 is an integral part of the glycogen phosphorylase enzyme, several studies have examined the relationship between carbohydrate intake and vitamin B6. Hatcher et al. (1982) found that blood levels of PLP and vitamin B6 after exercise were lower in subjects who had consumed a low-carbohydrate diet compared with a moderate-or high-carbohydrate diet 3 days before the exercise. The authors suggested that on the low carbohydrate diet gluconeogenesis is accelerated, which increases the need for PLP as a cofactor. In another study from the same laboratory, de Vos et al. (1982) reported that vitamin B6 supplementation may cause a faster depletion of muscle glycogen stores during exercise after ingestion of a low-carbohydrate diet and may accentuate a depletion-loading manipulation used by athletes to increase glycogen stores (glycogen supercompensation). Manore and Leklem (1988) found that vitamin B6 supplementation, along with increased carbohydrate consumption, resulted in lower free fatty acids during exercise. The authors recommended that athletes who are on a highcarbohydrate diet should not supplement their diets with vitamin B6 above the RDA level.
Presently there are no data regarding vitamin B6 requirements in a hot environment. The amount of vitamin B6 lost in sweat is considered insignificant (Mitchell and Edman, 1951). However, if food intake is increased, then the amount of vitamin B6 should be increased accordingly. It is recommended that 0.016 mg per g protein of vitamin B6 be ingested (vitamin B6 and protein occur together naturally in foods) (National Research Council, 1989).
Pantothenic Acid
Pantothenic acid acts as a structural component of coenzyme A (CoA), an acyl carrier protein. Pantothenic acid is important in transport of acyl groups to the Krebs cycle and in transport of fatty acyl groups across the mitochondrial membrane (Olson, 1990). Pantothenic acid is widely distributed in nature and found in all organisms. Therefore deficiencies are rare. However, during World War II, pantothenic acid deficiency was thought to be responsible for the burning foot syndrome among prisoners in Japan and the Philippines (Fox, 1984).
It is not known whether exercise increases the requirement for pantothenic acid. Nijakowski (1966) found that athletes had higher levels of pantothenic acid in the blood compared to controls. Cycle ergometry exercise of short duration resulted in a decrease in pantothenic acid levels in the blood, but the levels were unchanged after a long-duration exercise of 4
hours. Because plasma volume was not corrected for, it is difficult to interpret these changes.
The effect of pantothenic acid supplementation on exercise performance is equivocal. Compared with the placebo group, highly trained endurance runners who ingested 2-g doses of pantothenic acid per day for 2 weeks showed decreased exercise blood lactate levels and decreased oxygen consumption during prolonged exercise at 75 percent ![]() (Litoff et al., 1985). In contrast, Nice et al. (1984), using a controlled double-blind study, examined the effect of pantothenic acid supplementation (1 g per day for 2 weeks) or a placebo on run time to exhaustion in 18 highly trained distance runners. No significant differences were found between groups in run time or any of the standard blood parameters that were assessed (that is, cortisol, glucose, creatine phosphokinase, electrolytes).
(Litoff et al., 1985). In contrast, Nice et al. (1984), using a controlled double-blind study, examined the effect of pantothenic acid supplementation (1 g per day for 2 weeks) or a placebo on run time to exhaustion in 18 highly trained distance runners. No significant differences were found between groups in run time or any of the standard blood parameters that were assessed (that is, cortisol, glucose, creatine phosphokinase, electrolytes).
There are no data to suggest that the need for pantothenic acid would be increased by living and working in a hot environment. Pantothenic acid is not lost to a significant degree in sweat (Mitchell and Edman, 1951).
Vitamin B12(Cyanocobalamin)
Vitamin B12 plays a role in the formation and function of red blood cells (Ellenbogen, 1984) and may also function in protein, fat, and carbohydrate metabolism (Van der Beek, 1985). The condition of pernicious anemia was first described in 1924, and in 1929 a factor in liver was found to act as an antipernicious factor. It was not until 1948 that vitamin B12 was isolated and used to treat pernicious anemia (Ellenbogen, 1984).
No information is available on vitamin B12 status in athletes. However, it should be noted that athletes who are complete vegetarians may acquire a vitamin B12 deficiency because vitamin B12 is found mainly in animal products. Red cell vitamin B12 levels can indicate vitamin B12 status; however, low levels may also indicate a folate deficiency (Herbert, 1990). Several other tests are available to discern the two deficiencies; these are detailed elsewhere (Herbert, 1990).
Existing evidence suggests that vitamin B12 supplementation has no effect on performance (Williams, 1976). Montoye et al. (1955), in a doubleblind study, placed 51 adolescent boys (ages 12 to 17) into either an experimental group that consumed 50 µg of vitamin B12 daily, a placebo group, or a control group. No significant difference was found after 7 weeks between the supplemented group or the placebo group in the time to run 0.5 mile or in the Harvard step-test score (Montoye et al., 1955). Tin-May-Than et al. (1978) studied performance capacity in 36 healthy male subjects before and after injection of 1 mg cyanocobalamin given 3 times a week for 6 weeks. They found no significant improvement in ![]() , grip strength, pull-ups, leg lifts, or standing broad jump performance.
, grip strength, pull-ups, leg lifts, or standing broad jump performance.
There is no information concerning the effects of heat stress on vitamin B12 status. Recent studies have shown that megadoses of vitamin C (500 mg) may detrimentally affect the availability of vitamin B12 from food (Herbert, 1990). Doses of vitamin C of 3 g per day may even result in vitamin B12 deficiency disease. How this occurs is still unclear, but Herbert (1990) states that nutritionists should advise persons taking megadoses of vitamin C to have their blood checked regularly for vitamin B12 status. These findings should be taken into account with regard to the use of vitamin C to reduce heat stress (see section on vitamin C).
Folic Acid (Folate) and Biotin
Folic acid (pteroylglutamic acid) and folate (pteroylglutamate) are involved with DNA synthesis and nucleotide and amino acid metabolism, and they are especially important in tissues undergoing rapid turnover, such as red blood cells. Folic acid deficiency has been suggested to be the most common vitamin deficiency in humans and can result in anemia (Keith, 1989). No studies have assessed the relationship of folic acid status and exercise performance or the effect of folic acid supplementation on performance.
Biotin acts as a coenzyme for several carboxylase enzymes that are important in supplying intermediates for the Krebs cycle and for amino acid metabolism. It is also important in fatty acid and glycogen synthesis. Biotin deficiencies are rare in individuals consuming a nutritionally sound diet. One study found no difference in blood biotin levels in athletes compared with controls (Nijakowski, 1966). No studies have examined the effect of biotin supplementation on performance.
B Complex Vitamins
Many studies have shown that a deficiency of more than one of the B complex vitamins could lead to a decrease in physical performance capacity (for detailed reviews, see Van der Beek, 1985; Williams, 1989). Deficiency of a combination of several B vitamins produced subjective symptoms of fatigue, loss of ambition, irritability, and pain and loss of efficiency during normal work (see Van der Beek, 1985). Most of the studies that evaluated the effects of depletion of several B vitamins were done in the 1940s. More recently, Van der Beek et al. (1988) placed 12 men on a thiamin-, riboflavin-, vitamin C-, and vitamin B6-poor diet for 8 weeks. After 8 weeks, this diet caused borderline or moderately deficient blood levels of the four vitamins. These deficiencies were associated with a 9.8 percent decrease in ![]() and a 19.6 percent decrease in anaerobic threshold. Thus, a restricted diet of 21.3 to 32.5 percent of the Dutch RDA of these B vitamins
and a 19.6 percent decrease in anaerobic threshold. Thus, a restricted diet of 21.3 to 32.5 percent of the Dutch RDA of these B vitamins
and vitamin C led to decreased endurance capacity within a few weeks. This decrease was most probably due to the deficiency in the B vitamins rather than vitamin C (see section on vitamin C).
Because deficiencies of several B vitamins will lead to performance decrements, it is reasonable to assume that supplementation with a combination of B vitamins would enhance performance. Several studies have evaluated the effects of vitamin B complex supplementation (an excellent and detailed review can be found in Williams, 1989). Using a controlled, crossover design, Keys and Henschel (1941) examined the effect of supplementation with 100 mg nicotinic acid amide, 5 mg thiamin chloride, and 100 mg ascorbic acid daily for 4 weeks. Subjects were eight infantry men, and the exercise test was a 15-minute submaximal treadmill test (marching) where the subjects carried a pack and rifle. Compared with the placebo, the supplementation did not result in improved physiological parameters during exercise. In a follow-up study, Keys and Henschel (1942) examined the effects of a supplement containing 5 to 17 mg thiamin, 10 mg riboflavin, 100 mg nicotinic acid, 10 to 100 mg vitamin B6, 20 mg calcium pantothenate, and 100 to 200 mg ascorbic acid for 4 to 6 weeks. Subjects were 26 soldiers, and the exercise test was a strenuous treadmill run. Like their first study, Keys and Henschel found no beneficial effects on performance, so that endurance and resistance to fatigue were unaltered.
The effect of B complex supplementation on endurance capacity during a treadmill test was examined in physically active male college students (Read and McGuffin, 1983). The supplement contained 5 mg thiamin, 5 mg riboflavin, 25 mg niacin, 2 mg pyridoxine, 0.5 µg vitamin B12, and 12.5 mg pantothenic acid. After 6 weeks of supplementation, there was no significant improvement in endurance capacity.
Early and Carlson (1969) suggested that vitamin B complex supplementation could enhance exercise in the heat because these water-soluble vitamins may be lost via sweating. They studied the effect of one dose of a vitamin B supplement, which contained 100 mg thiamin, 8 mg riboflavin, 100 mg niacinamide, 5 mg pyridoxine, 25 mg cobalamin, and 30 mg pantothenic acid. High school males were given either the supplement or a placebo 30 minutes before running 10 50-yard dashes during hot weather. The running times were recorded for each trial. The group that received the supplement showed less fatigue (drop-off in running time) over the trials. These authors suggested that the amount of supplement and the combination of ingredients may be important for a supplement to be effective. They stated that the lower dosages of vitamins used in previous studies may not have been adequate to fulfill the additional vitamin requirement because of sweat loss and heightened metabolic activity with exercise in the heat.
Henschel et al. (1944a) examined the effects of a supplement containing 200 mg ascorbic acid or 0.5 mg thiamin, 10 mg riboflavin, and 100 mg
nicotinamide ingested for 3 days prior to exposure to heat for 2 to 4 days. During the heat exposure, the temperature was 110° to 120°F in the day and 85° to 90°F at night. The vitamin supplementation had no effect on sweat composition, water balance, strength tests, or exercise performance and recovery. Thus, the vitamin supplementation did not affect the rate and degree of acclimatization, the incidence of heat exhaustion, and the ability to perform work in the hot environment (Mayer and Bullen, 1960).
Although studies are equivocal with regard to whether supplementation of several of the B complex vitamins will enhance performance in a hot environment, these studies mainly assessed the effects of a short period of supplementation and a short exposure to exercise in the heat. Because exercise in the heat may increase energy expenditure (Consolazio, 1963), it is possible that a deficiency in B complex vitamins could occur if the dietary intake is not increased accordingly. This is especially true because of the loss of several of the B complex vitamins in sweat. Although the loss is small, if the intake of these vitamins is also compromised, a deficiency could occur. If caloric intake should be increased by work in a hot environment, then the intake of these vitamins would increase accordingly. Thus, for adults, 0.5 mg thiamin per 1000-kcal diet, 0.6 mg riboflavin per 1000-kcal diet, 0.016 mg vitamin B6 per g protein, and 6.6 mg niacin per 1000-kcal diet are recommended (National Research Council, 1989).
Vitamin C (Ascorbic Acid)
Scurvy was identified as far back as the ancient Greeks and Romans. This condition proved to be a scourge to armies, navies, and explorers until the early 1900s when Albert Szent-Gyorgyi first identified a substance that was later named vitamin C and used to prevent scurvy (Sauberlich, 1990). Vitamin C has numerous functions, including the biosynthesis of collagen, catecholamines, serotonin, and carnitine. It also plays a role as an antioxidant and is needed for nonheme iron absorption, transport, and storage (Keith, 1989).
Vitamin C is probably one of the most studied vitamins and one of the most controversial. The popularly believed benefits of vitamin C supplementation range from curing or preventing the common cold to reducing fatigue, wound healing, preventing injury, and enhancing performance capacity (Jaffe, 1984; Keith, 1989; National Research Council, 1989; Pike and Brown, 1984). Vitamin C is widely distributed throughout the body with highest concentrations in the pituitary, adrenals, and leukocytes. Major concentrations also are found in skeletal muscle, brain, and liver.
Ascorbic acid can be measured in the serum or plasma, leukocytes, and urine; however, levels in the plasma or serum are most commonly used (Sauberlich, 1990). Of several groups of athletes studied, most had adequate
or above adequate blood levels of vitamin C (for review see Clarkson, 1991). These data provide no evidence to suggest that chronic exercise creates a vitamin C deficiency.
Acute exercise appears to increase blood levels of ascorbic acid. Plasma and lymphocyte ascorbic acid levels increased in nine men who completed a 21-km race (Gleeson et al., 1987). This study also found that the increase in plasma ascorbic acid levels correlated significantly with an increase in plasma cortisol. The authors suggested that exercise may cause ascorbic acid to be released from the adrenal glands into the circulation along with the release of cortisol. Normally, vitamin C inhibits adrenocorticotropic hormone synthesis (Strydom et al., 1976). If chronic stress increases the release of vitamin C from the adrenals, an abnormal release of adrenocorticotropic hormones could occur followed by a ''fatigue'' of the adrenal glands. At this point the adrenals could not function adequately in another stress situation (Strydom et al., 1976).
Van der Beek et al. (1990) assessed the effect of vitamin C restriction on physical performance in 12 healthy men. The subjects ingested a diet providing only 10 mg per day of vitamin C for 3 weeks and 25 mg per day for 4 weeks. During this time, vitamin C levels in the blood decreased significantly. However, no effect of the vitamin C restriction was found on ![]() or the onset of blood lactate accumulation. The marginal vitamin C deficiency did not alter exercise performance.
or the onset of blood lactate accumulation. The marginal vitamin C deficiency did not alter exercise performance.
Excellent and comprehensive reviews of studies concerning the effects of ascorbic acid supplementation on performance can be found elsewhere (Keith, 1989; Williams, 1989). Keith (1989) cited 19 studies, many from outside the United States, that have shown a positive effect, and 18 that have shown no effect, of vitamin C supplementation on performance. Although several studies have shown that vitamin C supplementation will enhance performance (for example, Howald et al., 1975), these studies are flawed by poor designs, or the subjects may have been deficient in vitamin C. There are equally as many studies, and often better controlled ones, to demonstrate that vitamin C supplementation has no effect (for example, Keith and Merrill, 1983; Keren and Epstein, 1980).
Smokers have been shown to have a greater requirement for vitamin C, and the RDA for smokers is set at a minimum of 100 mg of vitamin C per day (compared with 60 mg per day for nonsmokers) (National Research Council, 1989). Keith and Driskell (1982) examined whether vitamin C supplementation of 300 mg per day for 3 weeks improved measures of lung function, resting and exercise heart rate, resting and exercise blood pressure, and the amount of work performed during a treadmill test in chronic smokers and nonsmokers. They concluded that vitamin C supplementation had little effect on lung function and exercise performance in either smokers or nonsmokers.
One study examined the effect of vitamin C supplementation on injury rate as well as on performance. Gey et al. (1970) placed 286 U.S. Air Force officers into two groups: officers in one group received 1000 mg vitamin C and officers in the other received a placebo daily for 12 weeks during moderate training. After 12 weeks, the groups showed no differences in improvement of performance on the Cooper 12-minute walk-run test (Gey et al., 1970). Also, the group taking vitamin C supplements had no reduction in injury rate compared with the group without supplementation.
Vitamin C also acts as an antioxidant to protect cells from free radical damage (see vitamin E section) (Machlin and Bendich, 1987). Because muscle soreness after exercise may result from muscle tissue damage (Ebbeling and Clarkson, 1989), it could be hypothesized that vitamin C supplementation may affect the development of soreness. Staton (1952) examined whether vitamin supplementation of 100 mg per day for 30 days would affect the performance of sit-ups on the second day of performance of the sit-ups (assuming that subjects were sore from the first day of sit-ups). The number of fewer repetitions the subjects were able to perform on the second day was taken to indicate the amount of soreness experienced. Vitamin C did not affect the number of sit-ups that could be performed, and Staton (1952) concluded that vitamin C had no effect on soreness. Whether the criterion score reflected an individual's soreness is uncertain. Also, because the exercise used in this study may not have produced significant muscle damage, especially with regard to the generation of free radicals, further study of the relationship of vitamin C and exercise-induced muscle damage is warranted.
In 1942, Holmes reviewed the use of vitamin C during World War II. He stated that "under certain severe conditions soldiers may need dietary supplements of certain vitamins. This is especially true of vitamin C, ascorbic acid, of which the United States used 17 tons in 1940 and may soon reach an annual output (synthetic) of 100 tons." Although Holmes provides citations to support the loss of vitamin C in appreciable quantities in sweat, this claim has not been substantiated by other studies. For example, one study found that at a sweat secretion of 700 ml per hour or more, the loss in vitamin C would not exceed 3 mg per day (Mitchell and Edman, 1951). However, Holmes stated that "the function of the vitamin C may go beyond mere replacement of the amount lost. It may combat heat shock." He also suggested that vitamin C may play a role in the healing of fractures and other wounds.
An interesting letter (Poda, 1979) regarding vitamin C intake and heat stroke appeared in a medical journal in 1979 and is excerpted below:
In 1951, a salesman with an Indiana-Illinois district sustained "heat stroke." Thereafter, if the temperature rose to more that 29.5°C he got "sick," very weak, and shocklike. He thus missed most of his summer saleswork, since air conditioned cars were not common then. On a hunch, from an old army
tale from World War II, I had him take 100 mg of vitamin C (ascorbic acid) three times daily during the summer months. Even though temperatures stayed at 32°C and higher (up to 40.6°C), he was able to drive his non-air-conditioned car and function. If he forgot to take his vitamin C, he got "sick."
In an early study, Henschel et al. (1944b) studied men who (a) were on a rigidly controlled vitamin C-restricted diet or on a vitamin C-supplemented (500 mg per day) diet for 4 to 7 days and who (b) were exercising in the heat for 2 hours per day for 4 days. The criterion measures were pulse rate at rest, pulse rate during exercise, and rectal temperature. No differences were found between the restricted and the supplemented conditions. Heat exhaustion occurred with equal frequency in each condition.
Strydom et al. (1976) reevaluated the Henschel et al. (1944b) study and noted that the authors had acknowledged that the vitamin C-supplemented condition showed a slight advantage with regard to rectal temperature. Furthermore, Henschel et al. (1944b) had studied only 4 days of heat stress, which may not have been sufficient to determine the effects of vitamin C supplementation. Therefore, Strydom et al. (1976) decided to further investigate whether vitamin C ingestion would affect the rate and degree of acclimatization to heat stress. In a study done in South Africa, they placed 60 mining recruits into three groups and administered a placebo, vitamin C (250 mg per day), or vitamin C (500 mg per day) for 10 days. During the supplementation period, the subjects were exposed to temperatures of 33.9°C for 4 hours per day while working at an intensity of 35 watts. Measurement of rectal temperature showed that vitamin C supplementation enhanced the rate and degree of acclimatization, with no difference between the two levels of supplementation. No effect of the supplement was found on sweat rates or heart rate response to the work. The initial blood levels of vitamin C for the three groups were 0.48 mg per 100 ml (placebo group), 0.60 mg per 100 ml (250-mg ascorbic acid group), and 0.43 mg per 100 ml (500-mg ascorbic acid group). These levels are considered adequate (Hunt and Groff, 1990), but at the low end of normal (Strydom et al., 1976).
In a subsequent study from the same laboratory (Kotze et al., 1977), a similar experiment was performed, but blood ascorbic acid levels were also assessed daily during the 10 days of heat stress. Resting blood ascorbic acid levels increased by the same amount in subjects receiving either the 250-mg or the 500-mg vitamin C supplement, and blood levels reached the saturation point between the third and the fifth day. The increase in blood ascorbic acid levels was associated with a reduction in rectal temperature and a reduction in total sweat output (which was independent of the reduction in rectal temperature). The maximum beneficial effect of the supplements in reducing rectal temperature occurred over the first 3 days of heat stress.
Thus, the supplements were more effective when heat stress was relatively high and blood levels of vitamin C had not reached the saturation point.
Although vitamin C status was adequate for the subjects in both of the studies cited above, the status was probably lower than in the normal healthy population, and the vitamin C supplementation raised blood levels of vitamin C to that found in healthy, well-fed individuals. However, blood levels of 0.43 to 0.60 mg per 100 ml may not be uncharacteristic of many lower socioeconomic groups, especially those who have poor diets, do not take supplements, and are smokers. In fact, Woteki et al. (1986) reported that in a 1976–1980 survey, about 5 percent of young adult American males were found to have blood vitamin C levels below 0.25 mg per 100 ml. Thus it could be expected that a sizable proportion of those individuals rapidly mobilized into military service may have suboptimal vitamin C status (between 0.43 and 0.60 mg per 100 ml).
Some data show that vitamin C status may be compromised by living and working in a hot environment for an extended period of time (see Scott, 1975). Visagie et al. (1974) found among mine workers in South Africa a high incidence of vitamin C deficiency during the first 3 months of employment. This deficiency occurred despite diets adequate in vitamin C.
Hindson (1970) examined vitamin C levels in the white blood cells of apparently healthy Europeans living in the tropics, a subject population consisting of British forces and their families living in Singapore. Anyone taking vitamin C supplements was excluded. Results showed a significant drop in vitamin C levels for the men but only a modest fall for the women. Although vitamin C is not lost in sweat to a significant degree in acclimatized individuals, vitamin C is needed in increased quantities for the process of sweating. Hindson (1970) concluded that vitamin C supplements should be taken by men who are working in the tropics. Also, vitamin C has been shown to be beneficial in treating prickly heat, a common disease of sweat glands for those living in the tropics (Hindson, 1970).
Recently, Chen et al. (1990) developed a sports drink especially for athletes training in hot environments. Made from Actinidia sinensis Planch (ASP; also known as kiwifruit), the drink contained several minerals and 48 mg per 100 ml vitamin C (Chen et al., 1990). During the summer of 1982, elite Chinese soccer and track athletes were tested at their training site. Environmental temperatures were 26.6° to 31.5°C. Athletes drank 500 to 1200 ml of ASP 10 minutes prior to a 1.5-to 2.7-hour normal training session and again halfway through the training session. On a separate occasion (training session), subjects drank an equivalent volume of a placebo drink. Vitamin C content in the athletes' urine averaged 132 mg per day when ingesting the ASP drink and 44 mg per day when ingesting the placebo. The authors concluded that vitamin C status of athletes ingesting the
ASP drink was improved. However, when the body's pool of vitamin C is greater than 1500 mg, the efficacy of kidney reabsorption decreases, and vitamin C is excreted into the urine (Hunt and Groff, 1990). Thus, increased vitamin C in the urine may simply indicate that the athletes had high levels of vitamin C already, and the excess "spilled over" into the urine.
FAT-SOLUBLE VITAMINS
Vitamins A, D, E, and K are fat-soluble vitamins, and these can be stored in appreciable amounts in the fat stores of the body. Because vitamin D is involved in calcium metabolism and vitamins A and E can function as antioxidants, these supplements may be important to exercise or work performance. Because no studies have been uncovered that examined the relationship of vitamin K (a vitamin necessary for blood clotting) with exercise performance or heat stress, this vitamin will not be discussed in the following sections.
Vitamin A
Night blindness was recognized by the ancient Egyptians and was treated by adding liver to the diet or by topically applying liver extract to the eyes (J. A. Olson, 1990). In 1914 the compound now known as vitamin A was found to prevent night blindness. Interestingly, the early Egyptian remedies had been lost over the years so that in the nineteenth century, night blindness plagued armies throughout the world (J. A. Olson, 1990).
Vitamin A designates a group of compounds including retinol, retinaldehyde, and retinoic acid. The body's need for vitamin A can be met by intake of preformed retinoids with vitamin A activity, which are generally found in animal products (National Research Council, 1989). Also, the need can be met by ingesting carotenoid precursors of vitamin A (beta-carotene, alphacarotene, and cryptoxanthin) commonly found in plants (National Research Council, 1989). The primary function of vitamin A is for maintenance of vision. Vitamin A is also involved in the growth process and the body's immune response. Beta-carotene, the major carotenoid precursor of vitamin A, plays a role as an antioxidant.
Blood levels of vitamin A (retinol) provide a relatively good index of total body stores. When the liver stores of vitamin A are low, the plasma levels fall (Olson, 1984). The few studies that have examined vitamin A status of athletes found no deficiencies (Guilland et al., 1989; Weight et al., 1988). The absence of deficiencies is most probably due to the body's relatively large storage capacity for vitamin A.
Only one study has examined the effect of vitamin A supplementation on exercise performance. Five men were placed on a vitamin A-deficient
diet (100 IU per day) for about 6 months followed by vitamin A supplementation (25,000 to 75,000 IU per day) for 6 weeks (Wald et al., 1942). No significant difference in run to exhaustion on a treadmill was found between the deficient and supplemented condition. Because the subjects had supplemented their diets with 75,000 IU for 30 days prior to the depletion phase of the experiment, the 6-month period of vitamin deficiency may not have been long enough to deplete the body's stores of vitamin A. However, it seems that vitamin A stores are generally adequate to meet the demands of exercise.
An antioxidant supplement containing 10 mg beta-carotene, 1000 mg vitamin C, and 800 IU of vitamin E was given to subjects before a downhill running exercise on a treadmill (Viguie et al., 1989). Although the details of the study are not available because this was a published abstract, it seems that the subjects performed the same exercise twice, the first time without the supplement and the second time with the supplement. Results showed that the supplement enhanced glutathione status (antioxidant status) and reduced indicators of exercise-induced muscle damage. However, other studies have shown that when the same damage-inducing exercise was repeated, the indices of damage were always reduced on the second bout regardless of any treatment (Clarkson and Tremblay, 1988). Further studies of the effects of beta-carotene as an antioxidant to reduce muscle damage from strenuous exercise are warranted. Presently there is no information on the effects of heat stress on vitamin A requirements.
Vitamin D
In the seventeenth century, rickets was scientifically described as resulting from a dietary deficiency (Norman, 1990). Later vitamin D was found to be synthesized by the body when skin was exposed to sunlight. The major function of vitamin D is its action as a hormone in the mineralization of bones and teeth (Keith, 1989). When the skin is exposed to ultraviolet radiation of the sun, a stero1 (7-dehydrocholesterol) is converted into vitamin D (cholecalciferol). Eventually vitamin D is converted to its hormone forms, 25-hydroxycholecalciferol (25(OH)D3) and 1, 25-dihydroxycholecalciferol (1,25(OH)2D3), by the liver and kidney, respectively. Also, vitamin D is obtainable from a few food sources including fortified milk and milk products.
Biochemical status of vitamin D is generally assessed from measurement of 25(OH)D3 in the blood; however, blood levels do not fully reflect the extent of storage. Although few studies have examined biochemical status of vitamin D in athletes (Adams et al., 1982; Cohen et al., 1985), vitamin D deficiencies generally are believed to be rare for those individuals with adequate milk consumption and exposure to sunlight.
Because vitamin D is involved with calcium metabolism, it could be thought to be related to exercise performance. However, existing evidence suggests that vitamin D supplementation does not affect work performance (Keith, 1989). Unique findings have been reported by Bell et al. (1988) who showed that blood levels of Gla-protein, an indicator of bone formation, and vitamin D were higher in subjects involved in muscle building training compared to controls. The authors suggested that the muscle building exercises stimulated (a) osteoblastic bone formation and (b) the production of vitamin D, possibly to provide calcium for newly forming muscle tissue. Whether these data indicate a greater vitamin D requirement for strenuous work where large loads are carried or moved is not known, and the question warrants further investigation.
Exposure to sunlight in a hot environment should be sufficient to prevent a vitamin D deficiency. In fact one study found no cases of vitamin D deficiency rickets in a survey of 224 African infants (Kendall, 1972). Because the mothers spent time in sunlight, and breast feeding is universal in the African population studied, babies get sufficient vitamin D. Also, it has been suggested that tropical vegetable foods contain appreciable amounts of vitamin D (Kendall, 1972).
Vitamin E
The major symptom of vitamin E deficiency in animals, which was identified in 1922, is a damping of the reproductive ability. However, muscle wasting or dystrophic muscles have also been noted in vitamin E-deficient animals (Bieri, 1990). It was not until the 1950s that vitamin E was shown to be important for humans as well as other animals.
Vitamin E comprises at least four compounds known as tocopherols. The most active and well known of these is alpha-tocopherol. Vitamin E has been shown to function as an antioxidant of polyunsaturated fatty acids in cellular membranes (Machlin and Bendich, 1987). In this role, vitamin E serves as a free radical scavenger to protect cell membranes from lipid peroxidation. Free radicals are chemical species with one or more unpaired electrons in their outer orbit, which makes them highly reactive. Because strenuous exercise can increase lipid peroxidation (Kanter et al., 1988; Maughan et al., 1989), vitamin E may have important implications for exercise or work capacity.
Plasma or serum tocopherol levels can provide a relatively good index of vitamin E status (Machlin, 1984). Although few studies have assessed vitamin E status of athletes (Cohen et al., 1985; Guilland et al., 1989; Weight et al., 1988), vitamin E deficiencies are considered rare (Kagen et al., 1989). Vitamin E intake among athletes is considered to be more than sufficient (Buskirk, 1981; Clarkson, 1991). High vitamin E intakes were
routinely used by athletes in the Mexico City and Munich Olympic Games (Buskirk, 1981).
Acute exercise has been shown to affect blood levels of tocopherol. Pincemail et al. (1988) found that plasma tocopherol levels were significantly increased in nine men during intense cycle ergometer exercise. The authors suggested that tocopherol was mobilized from adipose tissue into the blood and distributed to exercising muscles. At the muscle level, tocopherol could act to prevent lipid peroxidation induced by the exercise. However, because this study did not correct for hemoconcentration, and the small increase in plasma tocopherol was back to baseline after 10 minutes of rest, the results may simply be due to plasma volume changes induced by exercise.
To study the effects of vitamin E deficiency, Bunnell et al. (1975) fed subjects who were employed in jobs of hard physical labor a low vitamin E diet for 13 months. Although vitamin E levels dropped significantly during the study, subjects did not perceive any muscle weakness, pain, or cramps. Work capacity was not assessed.
Results from several well-controlled experiments have shown that vitamin E supplementation had no effect on the following:
-
The performance of standard exercise tests, including bench step tests, 1-mile run, 400-m swim, and motor fitness tests in adolescent male swimmers given 400 mg of alpha-tocopherol daily for 6 weeks (Sharman et al., 1971).
-
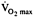 or muscle strength in college swimmers given 1200 IU daily for 85 days (Shephard et al., 1974).
or muscle strength in college swimmers given 1200 IU daily for 85 days (Shephard et al., 1974). -
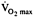 in ice hockey players given 1200 IU daily for 50 days (Watt et al., 1974).
in ice hockey players given 1200 IU daily for 50 days (Watt et al., 1974). -
A swimming endurance test and blood lactate in competitive swimmers given 900 IU daily for 6 months (Lawrence et al., 1975a,b).
-
Motor fitness tests, cardiorespiratory efficiency during cycle ergometry exercise and bench stepping, and 400-m swim times in male and female trained swimmers given 400 mg daily for 6 weeks (Sharman et al., 1976).
-
100-or 400-m swim performance in swimmers given 1600 IU daily for 5 weeks (Talbot and Jamieson, 1977).
Because of the role of vitamin E as an antioxidant, two studies examined the effect of vitamin E supplementation on performance at high altitudes, where oxygen availability may be compromised. Nagawa et al. (1968) reported that supplementation of 300 mg per day for at least 4 to 5 weeks had a moderate effect on several exercise tests, including cycle ergometry exercise and running sprints, performed at altitudes of 2700 and 2900 m. Using a better controlled design, Kobayashi (1974) examined the effect of vitamin E supplementation of 1200 IU daily for 6 weeks on submaximal
cycle ergometry exercise. Testing was done at altitudes of 1525 m (5000 feet) and 4570 m (15,000 feet). Submaximal oxygen intake, oxygen debt, and blood lactate levels were significantly lower in the vitamin E-supplemented group compared with the placebo group. At the higher altitudes, the decreased availability of oxygen may increase lipid peroxidation of the red blood cell and muscle cell membranes and thereby enhance their destruction. Williams (1989) suggested that increased levels of vitamin E could counteract this effect. A recent study by Simon-Schnass and Pabst (1988) showed that lipid peroxidation was lower in a group of mountain climbers supplemented with vitamin E.
Vitamin E may play an antioxidant role in reducing muscle damage from strenuous exercise (Ebbeling and Clarkson, 1989). Exhaustive exercise that produces muscle damage also results in an increase in free radical activity (Kanter et al., 1988; Maughan et al., 1989). However, results are equivocal on whether muscle damage is reduced by vitamin E supplementation. Helgheim et al. (1979) found that vitamin E (447 IU per day) supplementation for 6 weeks did not reduce the leakage of muscle enzymes into the blood following strenuous exercise. Also, muscle soreness, a general indicator of muscle damage, was not reduced in subjects taking vitamin E supplements (600 IU per day) for 2 days before performing a strenuous exercise (Francis and Hoobler, 1986). Although Sumida et al. (1989) found that 4 weeks of vitamin E supplementation (447 IU per day) resulted in a reduced serum enzyme response to exercise, a balanced design was not used. Rather, subjects performed the same exercise before supplementation and then again after supplementation. It has been well documented that serum enzyme response is substantially reduced the second time an exercise regimen is performed (Clarkson and Tremblay, 1988; Ebbeling and Clarkson, 1989) However, Goldfarb et al. (1989) examined the effect of 800 IU of vitamin E per day for 4 weeks on lipid peroxidation in blood samples taken after a run at 80 percent ![]() . Compared to the placebo group, the vitamin E-supplemented group showed reduced levels of lipid peroxidation at rest and after running
. Compared to the placebo group, the vitamin E-supplemented group showed reduced levels of lipid peroxidation at rest and after running
There is currently no information concerning vitamin E supplementation for exercise in the heat. Vitamins A, C, and E are all antioxidants and may have significant roles in reducing muscle damage (via lipid peroxidation) induced by strenuous exercise. It has been suggested that work in the heat could create a hypoxic condition in the muscle due to the redistribution of blood from the muscle to the skin, although there is some question whether this occurs (Young, 1990)· While no studies have examined lipid peroxidation during exercise in the heat, it is possible that hypoxia, dehydration, or other changes induced by heat stress could exacerbate lipid peroxidation in exercising muscle. If so, the antioxidant vitamins may be useful in the reduction of heat stress. Further research in this area seems warranted.
CONCLUSIONS AND RECOMMENDATIONS
The requirement for B vitamins does not seem to be increased by living and working in a hot environment. Although loss of these vitamins in sweat is minimal, a deficiency could occur over time from profuse sweating coupled with an insufficient dietary intake. Because thiamin, riboflavin, niacin, and vitamin B6 are important to energy metabolism, the level of vitamin intake should be related to the amount of food consumed. Thus, for adults, 0.5 mg of thiamin per 1000-kcal diet, 0.6 mg of riboflavin per 1000-kcal diet, 0.016 mg of vitamin B6 per g protein, and 6.6 mg of niacin per 1000-kcal diet are recommended (National Research Council, 1989). If calorie intake is not sufficient to meet the demands of exercise in the heat, then the vitamin intake will be compromised as well.
Folic acid and vitamin B12 are not linked to energy production, and their intake should be that of the 1989 RDA. There is no information to suggest that exposure to a hot environment would increase their need above levels recommended by the National Research Council.
Since World War I, vitamin C has received popular attention as a nutrient that can reduce heat stress. More recent studies have generally confirmed the anecdotal studies. Increased vitamin intake of 250 mg seems to have a positive effect on reducing heat stress during acclimatization in those individuals with adequate but low vitamin C levels. Some data have shown that vitamin C status may be compromised by long-term exposure to a hot environment. Thus, vitamin C supplements may be useful for those individuals who live and work in a hot environment. However, intakes of greater than 250 mg per day are not recommended because high doses of vitamin C can adversely affect the absorption of vitamin B12.
The one study (Bell et al., 1988) suggesting that vitamin D may be related to muscle building induced by strenuous resistance exercise is interesting and warrants further attention. However, at this time, there is no reason to recommend vitamin D supplements for people who work in the heat. Exposure to sunlight probably is sufficient for adequate vitamin D status.
Vitamins A, C, and E are antioxidants and may be useful in the reduction of lipid peroxidation induced by exercise stress. However, there have been no studies to examine whether lipid peroxidation is exacerbated by exercise in a hot environment. Further studies on whether these vitamins will be important as antioxidants for people living and working in a hot environment are warranted.
The following recommendations are made:
-
Thiamin, riboflavin, vitamin B6, and niacin should be linked to changes in food consumption as recommended by the RDAs (National Research Council, 1989). Insufficient data exist to recommend otherwise.
-
The RDA of folic acid and vitamin B12 should be sufficient to meet the body's requirement. Insufficient data exist to recommend otherwise.
-
Further studies should be done to determine the effects on vitamin status of long-term exposure to living and working in the heat.
-
During acclimatization, vitamin C intake should be about 250 mg per day to reduce heat stress and enhance acclimatization in those people who have adequate but low vitamin C status. Further study is warranted to confirm this recommendation and to determine whether supplementation may be effective in reducing heat stress in people with optimal vitamin C status.
-
Existing data show that vitamin C supplements may be needed for extended periods of living and working in hot environments. Further studies are needed to confirm this finding and to determine the amount of vitamin C needed to prevent a decrease in status.
-
There is no need to supplement vitamin D in hot environments.
-
Vitamin A (beta-carotene), vitamin C, and vitamin E function as antioxidants and may be useful as supplements in a hot environment. Further research is needed for confirmation.
REFERENCES
Adams, M.M., L.P. Porcello, and V.M. Vivian 1982 Effect of a supplement on dietary intakes of female collegiate swimmers. Physician Sportsmed. 10:122–134.
Belko, A.Z., E. Obarzanek, H.J. Kalwarf, M.A. Rotter, S. Bogusz, D. Miller, J.D. Haas, and D.A. Roe 1983 Effects of exercise on riboflavin requirements of young women. Am. J. Clin. Nutr. 37:509–517.
Bell, N.H., R.N. Godsen, D.P. Henry, J. Shary, and S. Epstein 1988 The effects of muscle-building exercise on vitamin D and mineral metabolism. J. Bone Miner. Res. 3:369–373.
Bergstrom, J., E. Hultman, L. Jorfeldt, B. Pernow, and J. Wahren 1969 Effect of nicotinic acid on physical working capacity and on metabolism of muscle glycogen in man. J. Appl. Physiol. 26:170–176.
Bieri, J.G. 1990 Vitamin E. Pp. 117–121 in Present Knowledge in Nutrition, M.L. Brown, ed. Washington, D.C.: International Life Sciences Institute.
Brotherhood, J.R. 1984 Nutrition and sports performance. Sports Med. 1:350–389.
Brouns, F., and W. Saris 1989 How vitamins affect performance. J. Sports Med. Phys. Fitness 29:400–404.
Brown, M.L. 1990 Thiamin. Pp. 142–144 in Present Knowledge in Nutrition, M.L. Brown, ed. Washington, D.C.: International Life Sciences Institute.
Bunnell, R.H., E. DeRitter, and S.H. Rubin 1975 Effect of feeding polyunsaturated fatty acids with a low vitamin E diet on blood levels of tocopherol in men performing hard physical labor. Am. J. Clin. Nutr. 28:706–711.
Buskirk, E.R. 1981 Some nutritional considerations in the conditioning of athletes. Ann. Rev. Nutr. 1:319–350.
Carlson, L.A., and L. Oro 1962 The effect of nicotinic acid on the plasma free fatty acids. Acta Med. Scand. 172:641–645.
Chen, J.D., Z.Y. Yang, S.H. Ma, and Y.C. Zhen 1990 The effects of Actinidia sinensis Planch (kiwi) drink supplementation on athletes training in hot environments. J. Sports Med. Phys. Fitness 30:181–184.
Clarkson, P.M. 1991 Vitamins and trace minerals. Pp. 2142 in Perspectives in Exercise Science and Sports Medicine, vol. 4: Ergogenics: Enhancement of Performance in Exercise and Sport, D.R. Lamb and M.H. Williams, eds. Dubuque, Iowa: W. C.B. Brown and Benchmark.
Clarkson, P.M., and I. Tremblay 1988 Rapid adaptation to exercise induced muscle damage. J. Appl. Physiol. 65:1–6.
Cohen, J.L., L. Potosnak, O. Frank, and H. Baker 1985 A nutritional and hematological assessment of elite ballet dancers. Physician Sportsmed. 13:43–54.
Consolazio, C.F. 1963 The energy requirements of men living under extreme environmental conditions. 3. Extremely hot environments. Pp. 65–77 in World Review of Nutrition and Dietetics, vol. 27, G.H. Bourne, ed. New York: Hafner Publishing.
Cooperman, J.M., and R. Lopez 1984 Ribofiavin. Pp. 299–327 in Handbook of Vitamins. Nutritional, Biochemical and Clinical Aspects, L.J. Machlin, ed. New York: Marcel Dekker.
deVos, A.M., J.E. Leklem, and D.E. Campbell 1982 Carbohydrate loading, vitamin B6 supplementation, and fuel metabolism during exercise in man. Med. Sci. Sports Exerc. 14:137 (abstract).
Dreon, D.M., and G.E. Butterfield 1986 Vitamin B6 utilization in active and inactive young men. Am. J. Clin. Nutr. 43:816–824.
Driskell, J.A. 1984 Vitamin B6. Pp. 379–401 in Handbook of Vitamins. Nutritional, Biochemical and Clinical Aspects, L. J. Machlin, ed. New York: Marcel Dekker.
Early, R.G., and B.R. Carlson 1969 Water-soluble vitamin therapy in the delay of fatigue from physical activity in hot climatic conditions. Int. Z. angew. Physiol. 27:43–50.
Ebbeling, C.B., and P.M. Clarkson 1989 Exercise-induced muscle damage and adaptation. Sports Med. 7:207–234.
Edison, A.O., R.H. Silber, and D.M. Tennent 1945 The effect of varied thiamine intake on the growth of rats in tropical environment. Am. J. Physiol. 144:643–651.
Ellenbogen, L. 1984 Vitamin B12. Pp. 497–547 in Handbook of Vitamins. Nutritional, Biochemical and Clinical Aspects, L.J. Machlin, ed. New York: Marcel Dekker.
Fox, H.M. 1984 Pantothenic acid. Pp. 437–457 in Handbook of Vitamins. Nutritional, Biochemical and Clinical Aspects, L.J. Machlin, ed. New York: Marcel Dekker.
Francis, K.T., and T. Hoobler 1986 Failure of vitamin E and delayed muscle soreness. J. Med. Assoc. Alabama 55:15–18.
Gey, G.O., K.H. Cooper, and R.A. Bottenberg 1970 Effect of ascorbic acid on endurance performance and athletic injury. J. Am. Med. Assoc. 211:105.
Gleeson, M., J.D. Robertson, and R.J. Maughan 1987 Influence of exercise on ascorbic acid status in man. Clin. Sci. 73:501–505.
Goldfarb, A.H., M.K. Todd, B.T. Boyer, H.M. Alessio, and R.G. Cutler 1989 Effect of vitamin E on lipid peroxidation at 80 percent max. Med. Sci. Sports Exerc. 21:S16 (abstract).
Gubler, C.J. 1984 Thiamin. Pp. 245–297 in Handbook of Vitamins. Nutritional, Biochemical and Clinical Aspects, L.J. Machlin, ed. New York: Marcel Dekker.
Guilland, J., T. Penaranda, C. Gallet, V. Boggio, F. Fuchs, and J. Klepping 1989 Vitamin status of young athletes including the effects of supplementation. Med. Sci. Sports Exerc. 21:441–449.
Guyton, A.C. 1986 Textbook of Medical Physiology, 7th ed. Philadelphia: W.B. Saunders.
Hankes, L.V. 1984 Nicotinic acid and Nicotinamide. Pp. 329–377 in Handbook of Vitamins. Nutritional, Biochemical and Clinical Aspects, L.J. Machlin, ed. New York: Marcel Dekker.
Haralambie, G. 1976 Vitamin B2 status in athletes and the influence of riboflavin administration on neuromuscular irritability . Nutr. Metab. 20:1–8.
Hatcher, L.F., J.E. Leklem, and D.E. Campbell 1982 Altered vitamin B6 metabolism during exercise in man: Effect of carbohydrate modified diets and vitamin B6 supplements. Med. Sci. Sports Exerc. 14:112 (abstract).
Helgheim, I., O. Hetland, S. Nilsson, F. Ingjer, and S.B. Stromme 1979 The effects of vitamin E on serum enzyme levels following heavy exercise. Eur. J. Appl. Physiol. 40:283–289.
Henschel, A., H.L. Taylor, O. Mickelsen, J.M. Brozek, and A. Keys 1944a The effect of high vitamin C and B intake on the ability of man to work in hot environments. Fed. Proc. 3:18.
Henschel, A., H.L. Taylor, J. Brozek, O. Mickelsen, and A. Keys 1944b Vitamin C and ability to work in hot environments. Am. J. Trop. Med. Hyg. 24: 259–264.
Herbert, V. 1990 Vitamin B-12. Pp. 170–178 in Present Knowledge in Nutrition, M. L. Brown, ed. Washington, D.C.: International Life Sciences Institute.
Hilsendager, D., and P. Karpovich 1964 Ergogenic effect of glycine and niacin separately and in combination. Res. Q. 35:389–392.
Hindson, T.C. 1970 Ascorbic acid status of Europeans resident in the tropics. Br. J. Nutr. 24:801–802.
Hofmann, A., R.D. Reynolds, B.L. Smoak, V.G. Villanueva, and P.A. Duester 1991 Plasma pyridoxal and pyridoxal 5'-phosphate concentrations in response to ingestion of water or glucose polymer during a 2-h run. Am. J. Clin. Nutr. 53:84–89.
Holmes, H.N. 1942 Vitamin C in the war. Science 96:384–386.
Howaid, H., B. Segesser, and W.F. Korner 1975 Ascorbic acid and athletic performance. Ann. N.Y. Acad. Sci. 258:458–463.
Hunt, S.M., and J.L. Groff 1990 Advanced Nutrition and Human Metabolism. St. Paul, Minn.: West Publishing.
Jaffe, G.M. 1984 Vitamin C. Pp. 199–244 in Handbook of Vitamins. Nutritional, Biochemical and Clinical Aspects, L. J. Machlin, ed. New York: Marcel Dekker.
Kagen, V.E., V.B. Spirichev, and A.N. Erin 1989 Vitamin E, physical exercise, and sport. Pp. 255–278 in Nutrition in Exercise and Sport, J.E. Hickson and I. Wolinsky, eds. Boca Raton, Florida: CRC Press.
Kanter, M.M., G.R. Lesmes, L.A. Kaminsky, J. La Ham-Saeger, and N.D. Nequin 1988 Serum creatine kinase and lactate dehydrogenase changes following an eighty kilometer race. Eur. J. Appl. Physiol. 57:60–63.
Karpovich, P.V., and N. Millman 1942 Vitamin B1 and endurance. N. Engl. J. Med. 226:881–882.
Keith, R.E. 1989 Vitamins in sport and exercise. Pp. 233–253 in Nutrition in Exercise and Sport, J.E. Hickson and I. Wolinsky, eds. Boca Raton, Florida: CRC Press.
Keith, R.E., and J.A. Driskell 1982 Lung function and treadmill performance of smoking and nonsmoking males receiving ascorbic acid supplements. Am. J. Clin. Nutr. 36:840–845.
Keith, R.E., and E. Merrill 1983 The effects of vitamin C on maximal grip strength and muscular endurance. J. Sports Med. 23:253–256.
Kendall, A.C. 1972 Rickets in the tropics and sub-tropics. Cent. Afr. J. Med. 18:47–49.
Keren, G., and Y. Epstein 1980 The effect of high dosage vitamin C intake on aerobic and anaerobic capacity. J. Sports Med. 20:145–148.
Keys, A., and A.F. Henschel 1941 High vitamin supplementation (B1, nicotinic acid and C) and the response to intensive exercise in U.S. Army infantrymen. Am. J. Physiol. 133:350–351.
1942 Vitamin supplementation of U.S. Army rations in relation to fatigue and the ability to do muscular work. J. Nutr. 23:259–269.
Keys, A., A.F. Henschel, O. Mickelsen, and J. M. Brozek 1943 The performance of normal young men on controlled thiamine intaes. J. Nutr. 26:399–415.
Keys, A., A.F. Henschel, O. Mickelsen, J.M. Brozek, and J.H. Crawford 1944 Physiological and biochemical functions in normal young men on a diet restricted in riboflavin. J. Nutr. 27:165–178.
Kobayashi, Y. 1974 Effect of vitamin E on aerobic work performance in man during acute exposure to hypoxic hypoxia. Ph.D. dissertation. University of New Mexico, Albuquerque.
Kotze, H.F., W.H. van der Wait, G.G. Rogers, and N.B. Strydom 1977 Effects of plasma ascorbic acid levels on heat acclimatization in man. J. Appl. Physiol. 42:711–716.
Lawrence, J.D., J.L. Smith, R.C. Bower, and W.P. Riehl 1975a The effect of alpha-tocopherol (vitamin E) and pyridoxine HCI (vitamin B6) on the swimming endurance of trained swimmers. J. Am. Coil. Health Assoc. 23:219–222.
Lawrence, J.D., R.C. Bower, W.P. Riehl, and J.L. Smith 1975b Effects of alpha-tocopherol acetate on the swimming endurance of trained swimmers. Am. J. Clin. Nutr. 28: 205–208.
Leklem, J.E., and T.D. Shultz 1983 Increased plasma pyridoxal 5'-phosphate and vitamin B6 in male adolescents after 4500-meter run. Am. J. Clin. Nutr. 38:541–548.
Litoff, D., H. Scherzer, and J. Harrison 1985 Effects of pantothenic acid supplementation on human exercise. Med. Sci. Sports Exerc. 17:287 (abstract). Machlin, L.J.
1984 Vitamin E. Pp. 99–145 in Handbook of Vitamins. Nutritional, Biochemical and Clinical Aspects, L.J. Machlin, ed. New York: Marcel Dekker.
Machlin, L.J., and A. Bendich 1987 Free radical tissue damage: Protective role of antioxidant nutrients. Fed. Am. Soc. Exp. Biol. J. 1:441–445.
Manore, M.M., and J.E. Leklem 1988 Effect of carbohydrate and vitamin B6 on fuel substrates during exercise in women. Med. Sci. Sports Exerc. 20: 233–241.
Maughan, R.J., A.E. Donnelly, M. Gleeson, P.H. Whiting, and K.A. Walker 1989 Delayed-onset muscle damage and lipid peroxidation in man after a downhill run. Muscle Nerve 12:332–336.
Mayer, J., and B. Bullen 1960 Nutrition and athletic performance. Physiol. Rev. 40:369–397.
McCormick, D.B. 1990 Riboflavin. Pp. 146–154 in Present Knowledge in Nutrition, M.L. Brown, ed. Washington, D.C.: International Life Sciences Institute.
Merrill, Jr., A.H., and F.S. Burnham 1990 Vitamin B-6. Pp. 155–162 in Present Knowledge in Nutrition, M. L. Brown, ed. Washington, D.C.: International Life Sciences Institute.
Mickelsen, O., and A. Keys 1943 The composition of sweat with special reference to the vitamins. J. Biol. Chem. 149: 479–490.
Mills, C.A. 1941 Environmental temperatures and thiamine requirements. Am. J. Physiol. 133:525–532.
Mitchell, H.H., and M. Edman 1951 Nutrition and Climatic Stress. Springfield, Ill.: Charles C. Thomas.
Montoye, H.J., P.J. Spata, V. Pinckney, and L. Barron 1955 Effects of vitamin B12 supplementation on physical fitness and growth of young boys. J. Appl. Physiol. 7:589–592.
Nagawa, T., H. Kita, J. Aoki, T. Maeshima, and K. Shiozawa 1968 The effect of vitamin E on endurance. Asian Med. J. 11:619–633.
National Research Council 1989 Recommended Dietary Allowances, 10th ed. Washington, D.C.: National Academy Press.
Nice, C., A.G. Reeves, T. Brinck-Johnsen, and W. Noll 1984 The effects of pantothenic acid on human exercise capacity. J. Sports Med. 24:26–29.
Nielsen, B., G. Savard, E.A. Richter, M. Hargreaves, and B. Saltin 1990 Muscle blood flow and muscle metabolism during exercise and heat stress. J. Appl. Physiol. 69:1040–1046.
Nijakowski, F. 1966 Assays of some vitamins of the B complex group in human blood in relation to muscular effort. Acta Physiol. Pol. 17:397–404.
Norman, A.W. 1990 Vitamin D. Pp. 108–116 in Present Knowledge in Nutrition, M. L. Brown, ed. Washington, D.C.: International Life Sciences Institute.
Olson, J.A. 1984 Vitamin A. Pp. 1–43 in Handbook of Vitamins. Nutritional, Biochemical and Clinical Aspects, L. J. Machlin, ed. New York: Marcel Dekker.
1990 Vitamin A. Pp. 96–107 in Present Knowledge in Nutrition, M. L. Brown, ed. Washington, D.C.: International Life Sciences Institute.
Olson, R.E. 1990 Pantothenic acid. Pp. 208–211 in Present Knowledge in Nutrition, M. L. Brown, ed. Washington, D.C.: International Life Sciences Institute.
Pike, R.L., and M.L. Brown 1984 Nutrition, An Integrated Approach, 3rd ed. New York: Macmillan.
Pincemail, J., C. Deby, G. Camus, F. Pirnay, R. Bouchez, L. Massaux, and R. Goutier 1988 Tocopherol mobilization during intensive exercise. Eur. J. Appl. Physiol. 57:189–191.
Poda, G.A. 1979 Vitamin C for heat symptoms? Ann. Intern. Med. 91(4):657.
Read, M.H., and S.L. McGuffin 1983 The effect of B-complex supplementation on endurance performance. J. Sports Med. 23:178–184.
Robinson, S., and A.H. Robinson 1954 Chemical composition of sweat. Physiol. Rev. 34:202–220.
Sauberlich, H.E. 1990 Ascorbic acid. Pp. 132–141 in Present Knowledge in Nutrition, M.L. Brown, ed. Washington, D.C.: International Life Sciences Institute.
Sauberlich, H.E., Y.F. Herman, C.O. Stevens, and R.H. Herman 1979 Thiamin requirement of the adult human. Am. J. Clin. Nutr. 32:2237–2248. Scott, M.L.
1975 Environmental influences on ascorbic acid requirements in animals. Ann. N.Y. Acad. Sci. 258:151–155.
Sharman, I.M., M.G. Down, and R.N. Sen 1971 The effects of vitamin E and training on physiological function and athletic performance in adolescent swimmers. Br. J. Nutr. 26:265–276.
Sharman, I.M., M.G. Down, and N.G. Norgan 1976 The effects of vitamin E on physiological function and athletic performance of trained swimmers. J. Sports Med. 16:215–225.
Shephard, R.J., R. Campbell, P. Pimm, D. Stuart, and G.R. Wright 1974 Vitamin E, exercise, and the recovery from physical activity. Eur. J. Appl. Physiol. 33:119–126.
Simon-Schnass, I., and H. Pabst 1988 Influence of vitamin E on physical performance. Internat. J. Vit. Nutr. Res. 58:49–54.
Staton, W.M. 1952 The influence of ascorbic acid in minimizing post-exercise muscle soreness in young men. Res. Q. 23:356–360.
Strydom, N.B., H.F. Kotze, W.H. Van der Wait, and G.G. Rogers 1976 Effect of ascorbic acid on rate of heat acclimatization. J. Appl. Physiol. 41:202–205.
Sumida, S., K. Tanaka, H. Kitao, and F. Nakadomo 1989 Exercise-induced lipid peroxidation and leakage of enzymes before and after vitamin E supplementation. Int. J. Biochem. 21:835–838.
Swendseid, J., and M.E. Swendseid 1990 Niacin. Pp. 163–169 in Present Knowledge in Nutrition, M. L. Brown, ed. Washington, D.C.: International Life Sciences Institute.
Talbot, D., and J. Jamieson 1977 An examination of the effect of vitamin E on the performance of highly trained swimmers. Can. J. Appl. Sport Sci. 2:67–69.
Tin-May-Than, Ma-Win-May, Khin-Sann-Aung, and M. Mya-Tu 1978 The effect of vitamin B12 on physical performance capacity. Br. J. Nutr. 40:269–273.
Tremblay, A., B. Boilard, M.F. Breton, H. Bessette, and A.G. Roberge 1984 The effects of riboflavin supplementation on the nutritional status and performance of elite swimmers. Nutr. Res. 4:201–208.
Tucker, R.G., O. Mickelsen, and A. Keys 1960 The influence of sleep, work, diuresis, heat, acute starvation, thiamine intake and bed rest on human riboflavin excretion. J. Nutr. 72:251–261.
Van der Beek, E.J. 1985 Vitamins and endurance training: Food for running or faddish claims? Sports Med. 2:175–197.
Van der Beek, E.J., W. van Dokkum, J. Schrijver, M. Wedel, A.W.K. Gaillard, A. Wesstra, H. van de Weerd, and R.J.J. Hermus 1988 Thiamin, riboflavin, and vitamins B-6 and C: Impact of combined restricted intake on functional performance in man . Am. J. Clin. Nutr. 48:1451–1462.
Van der Beek, E.J., W. van Dokkum, J. Schrijver, A. Wesstra, C. Kistemaker and R.J.J. Hermus 1990 Controlled vitamin C restriction and physical performance in volunteers. J. Am. Coil. Nutr. 9:332–339.
Van Erp-Baart, A.M.J., W.M.H. Saris, R.A. Binkhorst, J.A. Vos, and J.W.H. Elvers 1989 Nationwide survey on nutritional habits in elite athletes. Part 2. Mineral and vitamin intake. Int. J. Sports Med. 10:S11–16.
Viguie, C.A., L. Packer, and G.A. Brooks 1989 Antioxidant supplementation affects indices of muscle trauma and oxidant stress in human blood during exercise. Med. Sci. Sports Exerc. 21:S16 (abstract).
Visagie, M.E., J.P. Du Plessis, G. Groothof, A. Alberts, and N.F. Laubscher 1974 Changes in vitamin A and C levels in Black mine workers. S. African Med. J. 48:2502–2506.
Wald, G., L. Brouha, and R.E. Johnson 1942 Experimental human vitamin A deficiency and the ability to perform muscular exercise . Am. J. Physiol. 137:551–556.
Watt, T., T. T. Romet, I. McFarlane, D. McGuey, C. Allen, and R.C. Goode 1974 Vitamin E and oxygen consumption. Lancet 2:354–355 (abstract).
Weight, L.M., T.D. Noakes, D. Labadarios, J. Graves, P. Jacobs, and P.A. Berman 1988 Vitamin and mineral status of trained athletes including the effects of supplementation. Am. J. Clin. Nutr. 47:186–191.
Williams, M. H. 1976 Nutritional Aspects of Human Physical and Athletic Performance. Springfield, Ill.: Charles C. Thomas.
1989 Vitamin supplementation and athletic performance, an overview. Int. J. Vitam. Nutr. Res. 30:161–191.
Wood, B., A. Gijsbers, A. Goode, S. Davis, J. Mulholland, and K. Breen 1980 A study of partial thiamin restriction in human volunteers. Am. J. Clin. Nutr. 33:848–861.
Woteki, C., C. Johnson, and R. Murphy 1986 Nutritional status of the U.S. population: Iron, vitamin C, and zinc. Pp. 21–39 in What is America Eating? Proceedings of a Symposium, Food and Nutrition Board, National Research Council. Washington, D.C.: National Academy Press.
Young, A.J. 1990 Energy substrate utilization during exercise in extreme environments. Pp. 65–117 in Exercise and Sport Sciences Reviews, vol. 18, K. E. Pandolf, ed. Baltimore, Md.: Williams & Wilkins.
DISCUSSION
DR. NESHEIM: Thank you, Dr. Clarkson. We have a few minutes for questions or comments.
DR. EVANS: Well, we have one paper that we published in January and two more that are about to be published in which we have looked at the effects of vitamin E supplementations on skeletal muscle damage, circulating and skeletal muscle cytokine (CK) levels, and neutrophil generation.
And it appears that vitamin E has a profound effect in subjects that are over 60 in terms of altering all of their responses so that they look like young people in terms of CK release and neutrophil generation and monocyte function.
But it has very little effect in young people in all of those things and it may well be that membrane function is very different in old people as compared to young people.
But the other thing that vitamin E does is that it causes almost a total suppression of interleukin-1 (IL-1) production which may also have some very interesting effects. If IL-1 is necessary for adaptation to increased use, vitamin E may have some not such great effects.
PARTICIPANT: Dr. Clarkson, I was particularly interested in how you arrived at the quantitative figure of 250 milligrams (mg) for vitamin C.
DR. CLARKSON: That is what Strydom (Strydom et al., 1976) actually used in his paper. He used 250 milligrams as a supplement as well as 500 mg.
PARTICIPANT: Did he titrate the dose or was that just something that he chose?
DR. CLARKSON: I believe he based it on the Henshel et al. earlier study, (Henschel et al., 1944b) and it was no different. That graph depicting the 250 mg and the 500 mg dose showed no difference between the two doses. So 250 mg seems to be sufficient.
PARTICIPANT: You know it seems to be striking that all of the potential
effects on vitamin supplementation have been measured just using a gross measure in a ![]() . Maybe you can comment on that.
. Maybe you can comment on that.
It seems to me that there are so many other potentially more sensitive measurements that we can make of metabolic responses to exercise that have been ignored for the most part because ![]() is easy to measure.
is easy to measure.
DR. CLARKSON: I agree. Many studies that we find have used ![]() but there are also several studies that used submaximal exercise and studies that used strength.
but there are also several studies that used submaximal exercise and studies that used strength.
These are easy to measure. I think that is why they are used. Also, except for vitamin C where I only showed you three representative studies, mostly all the studies that are available were presented here. So it is not that there are a hundred other studies out there.
I think that more people should be involved in looking at the effects of vitamins on performance. I think one of the problems why people aren't involved in looking at vitamins is that it is hard to measure in the blood and therefore difficult to determine initial status.
PARTICIPANT: My question is specifically in terms of looking at measurements as opposed to plasma or sweat loss. What about some other measure—urine or something else.
DR. CLARKSON: Well, urine levels are hard to interpret because what happens is, as soon as you reach a threshold level the nutrient spills over, so you are not quite sure what urinary secretion means.
Does increased excretion mean you need less? Perhaps for a nonexercising person this is true. I am not ready to really believe that for an exercising person. In this case when you get an increased excretion it is not clear what this really means.
If I gave a sedentary individual large doses of a particular vitamin and it increases in the urine, then we would say, yes, the status is adequate and the person does not need a supplement.
However, when you add stressors like heat and exercise, I am not really sure what an increase in urinary levels of vitamins means.
PARTICIPANT: I just wanted to follow up one comment you had made on niacin. There are two papers—certainly submitted—in those studies Evelyn Stephasson(?) had administered niacin supplementation to individuals and had them exposed to heat and attempted exercise.
She found a very profound dilation and increased incidence of syncopy. So niacin supplementation in heat could actually reduce performance.
DR. CLARKSON: Yes, I mentioned the flushing.
PARTICIPANT: In the Strydom (Strydom et al., 1976) paper, do you hap-
pen to recall whether he assessed what the vitamin C status was before he did the supplementation?
DR. CLARKSON: No, on the second paper they did check vitamin C levels.
PARTICIPANT: And they were adequate before this?
DR. CLARKSON: Yes, but low.
PARTICIPANT: Based on plasma concentrations?
DR. CLARKSON: Yes.
PARTICIPANT: I just want to make a comment. I don't know if anyone noticed, about a week or two ago in Science magazine, there was just a short note on Dr. Linus Pauling who is still at age 90 consuming 18 grams per day of vitamin C. I don't know what type of effects that would have on absorption and interference that you talk about.
And the other thing, I was also interested in the work in the South Africans on vitamin C apparently accelerating the acquisition of acclimation. Do you know of any other papers that have followed that up? That was mid-1970s; correct?
DR. CLARKSON: Yes, and that is it. I found that one.
PARTICIPANT: I could offer a technical comment. My thesis was on vitamin B12 chemistry and I did study some of the interactions of vitamin C and B12 and so has Victor Herbert (RDA, ninth edition). And a lot of these effects are an artifact of the analytical techniques. I don't take that too seriously.
It turns out that vitamin C plus certain forms of B12 will be generating singlet oxygen and will destroy the chromophore in the test tube. So if you don't prevent this artifact—it is a pro-oxidant when you add it to iron usually.
So a lot of the studies are flawed because they didn't prevent this. You have high C levels carried over in your serum when you are doing analysis in B12.
DR. CLARKSON: In his (Herbert, 1990) recent review of literature on vitamin B12, Herbert suggests that vitamin C does have an effect on absorption of vitamin B12.
PARTICIPANT: Just a comment on vitamin B12. I would think it would be rather unlikely that you would see a B12 deficiency if you were to put adults on a low intake for a period of time. It is going to take a long long time to get a deficiency.
Actually, I did my thesis work on B12 requirements in baby pigs and the only way we could ever produce a B12 requirement in those pigs was to put the dams on a low or almost no B12 intake and then take the pigs away from the dam almost immediately after birth and put them on a vitamin B12-free diet and then we could produce a deficiency and, as a matter of fact, we produced it very quickly.
But if we let them have the colostrum milk for even four or five days, it just went a long time to ever produce a B12 deficiency.
PARTICIPANT: I would like to comment that there is some data that I think has appeared in the literature now by Doris Calloway and colleagues who were involved in a three-country study—Mexico, Kenya, and Egypt—and were looking at growth and other performance parameters in children.
They appeared to be finding an impact of animal protein intake per day in terms of the growth and development of these young children and they are looking very hard at trying to get data on the actual B12 content of these diets.
It is a possibility, since these populations tend to be very much on a vegetarian type of program—very little meat in these poorer populations—that you are seeing some of it (vitamin B12 deficiency) in the military.
But then again, I think it is highly unlikely that we would see a B12 deficiency as it relates to that.
PARTICIPANT: Just maybe one other comment. Haven't there been some reported vitamin D deficiencies in Middle Eastern countries in which women, in fact, have very little skin exposure to the sun?
I mean, it is a complicating factor. In a desert environment, many people have kind of an ironic effect of D deficiency because their skin doesn't see the sun.
PARTICIPANT: I seem to recall reading some comments to that but I don't know of any specific literature.
DR. EVANS: We are in the process of conducting some studies in vitamin D deficiencies in older people but vitamin D deficiency is very present. They don't drink milk and they don't see the sun very much and it may be associated with a profound muscle weakness due to a calcium metabolism problem.
DR. CLARKSON: There might also be vitamin D deficiencies in some athlete groups like dancers who don't drink milk, because quite a few of them do have a low consumption of milk and they do not spend much time in the sunlight.
PARTICIPANT: I was going to ask a question, and this relates to the microorganisms in the GI tract and the vitamin C. I wonder, has anyone
done any studies and looked at the types of microbes that are in the GI tract, the possibility of infection (subclinical infections) that occur in long-distance runners? Has anyone ever done that type of work?
PARTICIPANT: If anything, there was one paper that suggested that too much vitamin C might contribute to some of the lesions that have been observed in the GI tract in athletes.
PARTICIPANT: You would have to take in a large amount, wouldn't you?
DR. EVANS: With some athletes, apparently they do take in quite a bit.
DR. NESHEIM: Thank you very much for your interesting comments.




































