|
WORKSHOP IN BRIEF |
INSTITUTE OF MEDICINE Advising the nation • Improving health |
Relationships Among the Brain, the Digestive System, and Eating Behavior—Workshop in Brief
On July 9-10, 2014, the Institute of Medicine’s (IOM’s) Food Forum hosted a public workshop to explore emerging and rapidly developing research on relationships between the brain, digestive system, and eating behavior. The figure below illustrates the complex relationships between the brain, digestive system, and eating behavior and the influence of biology and the environment.
Drawing on expertise from the fields of animal and human physiology and behavior, nutrition and food science, psychology and psychiatry, and related fields, the purpose of the workshop was:
- To review current knowledge on the relationship between the brain and eating behavior:
- To explore the interaction between the brain and the digestive system.
- To explore what is known about the brain’s role in eating patterns and consumer choice.
- To evaluate the current methods used to determine the impact of food on brain activity and eating behavior.
- To identify gaps in our knowledge and discuss potential theoretical frameworks for future research.
This workshop in brief summarizes highlights of the presentations and discussion at the workshop. Importantly, the goal of the workshop was not to reach consensus on any issue or to make recommendations. The opinions expressed and suggestions summarized here are those of individual speakers or audience members and should not be construed as reflecting consensus on the part of the IOM, the Food Forum, the workshop planning committee, or any other group.
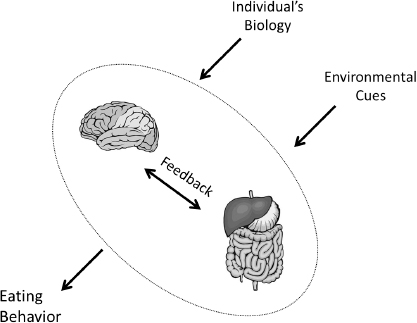
FIGURE Relationships between the brain, digestive system, and eating behavior.
NOTE: Environmental cues include commercial, physical, social, and cultural influences.
Interaction Between the Brain and Digestive System
The workshop began with an exploration of what happens in the body when food is first consumed, that is, how the presence of food in the gut triggers signals to the brain about nutrient content, character, and volume and how that information, in turn, impacts further food intake. Timothy Moran of Johns Hopkins University explained how most of the information that the brain receives about gastrointestinal contents comes from vagal afferent feedback signals,1 some of which come from the stomach and others from the intestine. Most vagal signals sent from the stomach respond to factors such as stretch and tension and are related to the volume of food, not its nutrient content. Signals from the intestine, on the other hand, respond to both load and content, with the presence of nutrients in the intestine triggering the release of gut peptides, such as cholecystokinin (CCK), which, in turn, activate vagal afferent activity. Importantly, Moran said, while vagal signals from the stomach are different than those arising in the intestine, the two intersect in the hindbrain where, together, they play a role in reducing further food intake.
According to Moran, the hindbrain is also where the brain processes yet another set of signals: sensory signals arriving from taste and taste-like receptors. Taste signals arise not just in the oral cavity, on the tongue, but deeper in the gastrointestinal tract as well. Robert Margolskee of Monell Chemical Senses Center described scientists’ discovery of taste-like receptors in the gut in the 1990s and what has been learned since then about which gut cells in particular express the receptors and how gut-expressed taste proteins contribute to the physiological response to food. For example, work in Margolskee’s laboratory has shown that expression of a molecule involved with glucose transport (SGLT1) is mediated by two gut-expressed taste proteins (gustducin and T1R3).
Robert Ritter of Washington State University elaborated on some of the information and ideas presented earlier by Moran and explored in greater mechanistic detail how vagal signals activated by gut peptides, CCK in particular, contribute to the process of satiation and reduce further food intake. Much of that activity revolves around glutamate release in the hindbrain. Ritter likened vagal afferent endings in the hindbrain to a “paintbrush that paints the feeling of satiety.” While evidence suggests that non-gut proteins, such as leptin, an adipose tissue hormone, can also contribute to the process of satiation, Ritter encouraged further research on the role non-gut proteins play in modulating vagal afferent satiety signaling.
Eating is impacted by much more than what happens when food enters the body and triggers feedback signals to the brain. Laurette Dubé of McGill University considered the broader context within which brain–digestive interactions operate and impact eating behavior. She described experiments showing how food is framed—for example, whether people are told that the food they are about to eat is “healthy” versus “tasty” and how that impacts their perception of fullness. Dubé also described studies showing that birth weight is linked with eating behavior. Yet other evidence suggests that parental or familial context—for example, whether a child learns to associate food with reward—can also influence eating behavior. Finally, Dubé described studies showing that the broader social environment matters; for example, decreased income has been associated with increased consumption of carbonated soft drinks. Dubé urged a greater focus on these broader contexts when studying physiological responses to food and suggested a systems-level approach. She said, “We need to study [the broader context] scientifically as much as we study the body scientifically.”
Assessing the Science Behind Methodologies Being Used to Characterize Food as Addictive
Scientists increasingly rely on data from neuroimaging studies and questionnaires, specifically the Yale Food Addiction Scale (YFAS) self-report questionnaire, to answer questions about the potential addictive nature of foods and eating behaviors. One session of the workshop explored current methods used to determine the impact of food on brain activity and eating behavior.
___________________
1 Vagal afferent signals are signals transmitted toward the central nervous system—in this case from the gastrointestinal tract—via the vagus nerve.
In the opinion of Dana Small, Yale University, neuroimaging will be critical to understanding how the food environment impacts the brain and interacts with neural circuits in a way that promotes obesity. Small described neuroimaging evidence indicating that food cues in the environment can trigger eating even in the absence of hunger. Heightened food cue responses in the brain (e.g., responses to food pictures), as measured by fMRI,2 are not only associated with higher body mass index (or BMI) but also predict success in weight loss trials. At least for carbohydrates, Small described data demonstrating that the underlying physiological mechanism appears to be a post-ingestive glucose metabolic effect as opposed to something sensory, with greater metabolic effect causing a greater anticipatory response to food cues. She observed that the effects are different for liquids versus solids.
While Small did not directly address the food addiction implications of the evidence she described, Hisham Ziauddeen of the University of Cambridge stated that whether imaging data reveal anything about eating behaviors is open to debate. He highlighted the many assumptions underlying neuroimaging studies on eating behaviors and urged caution when interpreting results. For example, neuroimaging studies on “food addiction” are largely guided by what is known about drug addiction and changes in the dopamine pathways associated with drug addiction. However, data have shown that similar responses to different substances in the same region of the brain do not necessitate that the same neuronal circuits are being engaged. In Ziauddeen’s opinion, there is little direct neuroimaging evidence of food addiction.
Ashley Gearhardt of the University of Michigan reviewed several YFAS studies demonstrating that addictive mechanisms may be at play in problematic eating but emphasized that existing findings need to be replicated. She also urged more longitudinal YFAS studies and a greater understanding of which foods in particular trigger addictive-like responses.
The YFAS is based on DSM-IV criteria for drug addiction, which themselves are based on opioid addiction studies extending back to the 1930s. Charles O’Brien of the University of Pennsylvania questioned whether concepts and terminology from the opioid addiction field are applicable to food. If there is a good reason for adopting those criteria, then they likely/potentially should be adopted. But is there? “That is one of the questions that we have to try to answer,” he said. He and his colleagues who worked on the more recent DSM-V considered including food addiction as a diagnosis but found existing neuroimaging and other evidence inconclusive.
Future Directions: Is the Addiction Model for Drugs and Alcohol Appropriate for Food?
Throughout the workshop, individual participants expressed varying opinions about how to interpret existing evidence and whether it is appropriate to characterize food as addictive. In the latter half of the workshop, an entire session was organized around counterpoint presentations on whether the drug and alcohol addiction model is appropriate for food. Nicole Avena of Columbia University argued for the use of the addiction model as appropriate for food, while Peter Rogers of University of Bristol used some of the same evidence to argue against the use of the addiction model for food.
Avena’s argument for the appropriateness of the addiction model was based, in part, on research with rats. Studies from her lab have demonstrated that sugar acts on the brain in a way similar to that of drugs on the brain. Rats that binge on sugar not only release dopamine in their brains in a drug-like way, but also show brain chemistry signs of withdrawal when sugar is removed from their diet. Avena urged a rethinking of the concept of addiction. Rather than the extreme loss of control typically associated with addiction, she observed that the typical pathway to obesity involves a gradual loss of control with repeated consumption of large portions and failure to resist energy-rich foods.
While evidence presented by Avena and others showed an overlap between brain mechanisms and behaviors associated with foods and those associated with drugs, Rogers cautioned that such an overlap is not evidence for addiction. He proposed an alternative explanation to changes in the brain that occur when rats (or people) overeat. Rather than an addiction-like vicious cycle where rats have to eat more food over time to get the
___________________
2 fMRI is functional magnetic resonance imaging, a technology that measures brain activity by detecting changes in blood flow.
same reward, he suggested that the brain chemistry changes being observed may be a negative feedback mechanism for weight-gaining rats to stop overeating. Rogers cautioned that not only does the evidence not support the case for addiction, except possibly in the case of binge eating, but the addiction model also could be counterproductive. By attributing overeating to addiction, people may be less motivated to make healthier choices.
Future Directions: Integrating the Evidence
As highlighted thus far in this workshop in brief, much of the workshop discussion revolved around how the brain processes two kinds of food-related signals: satiety signals sent from the digestive system indicating fullness, and sensory signals triggered not only by food in the gut but also food cues in the environment. In his concluding presentation, Edmund Rolls of the Oxford Center for Computational Neuroscience hypothesized that an imbalance between these two systems may contribute to obesity, with sensory signals overriding satiety signals and over-stimulating the reward system in the brain. Revisiting Dubé’s argument that eating behavior is influenced by the broader context in which the brain–digestive system operates, Rolls suggested that whether the systems are imbalanced depends, in part, on “top-down” cognitive processes in the brain that influence how people actually perceive and respond to food rewards. A better understanding of individual differences in sensitivity to food rewards and whether a greater sensitivity might contribute to obesity is one of many topics Rolls suggested for future research.
Panel Discussions with the Audience: Strategies for Future Research
Research on relationships between the brain, digestive system, and eating behavior is developing rapidly, with several new methodologies being employed and new avenues of study being explored. Still, many unanswered questions remain. During the panel discussions with the audience, workshop participants explored potential research strategies for addressing some of those questions. Dubé called for a systems-level approach to understanding how body weight is managed and how brain–digestive system interactions are influenced by “higher-level” cognition and the broader social environment. At the same time, she and others stressed the need for continued research aimed at understanding the pieces by themselves. Amid multiple queries about whether addictive-like overeating is triggered by a substance in food, a predisposition in overeating individuals, or both, Ziauddeen stressed the importance of having a clearly identifiable addictive substance as a starting point for studying any addiction. Finally, a number of participants urged a better understanding of the purpose and consequences of applying an addiction model to food. ![]()
___________________
DISCLAIMER: This workshop in brief has been prepared by Leslie Pray, rapporteur, as a factual summary of what occurred at the meeting. The statements made are those of the authors or individual meeting participants and do not necessarily represent the views of all meeting participants, the planning committee, or the National Academies.
REVIEW: To ensure that it meets institutional standards for quality and objectivity, this workshop in brief was reviewed by Sylvia B. Rowe, SR Strategy, and Joseph Herskovic, Joseph E. Herskovic Consulting; and coordinated by Chelsea Frakes, Institute of Medicine.
SPONSORS: This workshop was partially supported by the University of Massachusetts—Amherst; in addition to Abbott Laboratories; Agriculture Research Service—U.S. Department of Agriculture; Center for Nutrition Policy and Promotion—U.S. Department of Agriculture; The Coca-Cola Company; ConAgra Foods; Division of Nutrition Research Coordination—National Institutes of Health; Food and Nutrition Service—U.S. Department of Agriculture; Food Safety and Inspection Service—U.S. Department of Agriculture; General Mills Inc.; Kellogg Company; Kraft Foods; Mars, Inc.; McDonald’s; Monsanto; National Cancer Institute—National Institutes of Health; Nestlé Nutrition; Office of Dietary Supplements—National Institutes of Health; Office of Disease Prevention and Health Promotion—Department of Health and Human Services; PepsiCo; and Tate & Lyle.
For additional information regarding the meeting, visit www.iom.edu/FoodForum.




