Review of X-Ray Backscatter
Advanced Imaging
Technology Studies
The committee performed a comprehensive analysis of previous studies of X-ray backscatter advanced imaging technology (AIT) systems. This chapter focuses on the following reports:
- Johns Hopkins University/Applied Physics (JHU/APL), Radiation Safety Engineering Assessment Report for the Rapiscan Secure 1000 in Single Pose Configuration (2010);1
- National Institute of Standards and Technology (NIST), Assessment of the Rapiscan Secure 1000 Single Pose (ATR version) for Conformance with National Radiological Safety Standards (2012);2
- American Association of Physicists in Medicine (AAPM), Radiation Dose from Airport Scanners: Report of AAPM Task Group 217 (2013);3
_______________
1 JHU/APL, Radiation Safety Engineering Assessment Report for the Rapiscan Secure 1000 in Single Pose Configuration, NSTD-09-1085, Version 2, Laurel, Md., August 2010.
2 J.L. Glover, R. Minniti, L.T. Hudson, and N. Paulter, Assessment of the Rapiscan Secure 1000 Single Pose (ATR version) for Conformance with National Radiological Safety Standards, NIST report for the TSA, interagency agreement HSHQDC-11-X-00585, April 19, 2012; latest version available is J.L. Glover, R. Minniti, L.T. Hudson, and N. Paulter, Assessment of the Rapiscan Secure 1000 Single Pose (ATR version) for Conformance with National Radiological Safety Standards, NIST report for the TSA, interagency agreement HSHQDC-11-X-00585, September 28, 2012.
3 AAPM, Radiation Dose from Airport Scanners: Report of AAPM Task Group 217, College Park, Md., 2013.
- U.S. Army Public Health Command (USAPHC), Radiation Protection Consultation No. 26-MF-0E7K-11, Rapiscan Secure 1000 Single Pose Dosimetry Study (2012);4 and
- Food and Drug Administration (FDA), Assessment of the Rapiscan Secure 1000 Body Scanner for Conformance with Radiological Safety Standards (2006).5
The committee briefly describes how it considered radiation measurements from these previous reports, followed by an analysis of each report, including subsections on measurements, calculations, procedures. and interlocks. The chapter also includes a summary of measurements and interlock information from the reviewed studies and a discussion of other studies and their considerations—studies that usually lacked the level of measurements and calculations that the first set of studies had. The chapter concludes with findings and recommendations on exposure and dose.
COMPARING RADIATION MEASUREMENTS
In its analysis of previous reports, the committee focused on the methodologies used to measure radiation emitted by the AIT systems as well as the results. It also looked at AIT system design, such as interlocks. In interpreting and comparing the results from these reports, the committee identified two issues that may result in variations in effective dose to scanned individuals:
- The model of the AIT system evaluated, and
- The techniques used to measure kerma and half-value layer (HVL).
With respect to the model of the AIT systems, all systems evaluated in the reviewed reports were produced by the same manufacturer, Rapiscan Systems, and represent different stages in the evolution of the instruments installed in airports. As the instrument was refined, it is not unlikely that small changes were made in the physical components, operating parameters, and procedures. However, there is no evidence that major changes that would affect the radiation emitted were made during the interval covered by the reports reviewed.
Four physical quantities are characteristic of the AIT system and are the primary determinants of the effective dose to a standard subject when scanned. These
_______________
4 USAPHC, Radiation Protection Consultation No. 26-MF-0E7K-11, Rapiscan Secure 1000 Single Pose Dosimetry Study, Aberdeen Proving Ground, Md., 2012.
5 F. Cerra, Assessment of the Rapiscan Secure 1000 Body Scanner for Conformance with Radiological Safety Standards, Food and Drug Administration, Arlington, Va., July 21, 2006.
are the photon spectrum, fluence rate, scan duration, and the location of the subject in the AIT system. Additional physical quantities that describe the subject being scanned determine the effective dose for the individual, including weight, height, and shape.
The photon spectrum and fluence rate produced by a specific AIT system are controlled by the X-ray tube used, the anode voltage applied, the anode current, and the X-ray filtration inherent in the AIT system. It appears that the same tube type was used in all of the AIT systems described in the reviewed reports. Within a given tube type, the variations in output at a fixed anode voltage and current are very limited. The most significant source of variation in output is the thickness of the photon exit window of the tube. This window is a component of the photon filtration for the AIT system and thus can influence the photon spectrum. The X-ray tube used in these AIT systems has a glass window 1.5 mm thick. From 10 keV to 30 keV, the mass attenuation coefficient of borosilicate glass is 65 to 70 percent of that of aluminum; thus, this glass window contributes approximately 1 mm aluminum equivalent beam filtration. As a tube is used, a small amount of the anode material, tungsten, evaporates and deposits as a thin metallic film on the inside of the tube, including the window. Thus, the effective filtration may increase slightly with tube use.
The photon spectrum is essentially independent of the anode current, but at a fixed anode voltage, the fluence rate is directly proportional to the anode current. The standard practice for X-ray backscatter AIT systems is to control the anode current by adjusting the cathode temperature, achieved by adjusting the filament current. The filament current is provided by a regulated power supply using a sample of the anode current compared to a reference current. The anode current specified for late-model AIT systems is 3 mA, but the maximum-rated anode current for the tube is 5 mA, and the tube can probably survive a slightly higher current for short times. Consequently, it may be possible to adjust the anode current to produce a photon fluence rate and effective dose rate as much as a factor of two higher than would be obtained using the 3 mA currently specified.
The photon spectrum and the fluence rate both change with a change in anode voltage. For a tungsten target at 50 kV, a 10 percent increase in anode voltage produces approximately a 10 percent increase in the photon yield. For example, an increase in anode voltage from 50 to 55 keV results in approximately a 20 percent increase in the energy emitted as photons, the result of a 10 percent increase in the power to the anode and a 10 percent increase in the radiation yield as well as a 10 percent increase in the maximum photon energy.6 Differences of this magnitude (or larger) might exist between early prototype AIT systems and production models
_______________
6 F.H. Attix, Introduction to Radiological Physics and Radiation Dosimetry, Wiley, New York, N.Y., 1986.
but are extremely unlikely during the operation of a production unit because the regulation of high-voltage power supplies is typically better than 0.05 percent for line voltage variations from 102 to 132 VAC.7
The HVL of the photon spectrum is also influenced by any additional material in the photon path to the scanned object. It appears that the only material in addition to the glass tube window is the case of the AIT system, approximately 3 mm of plastic. Although the type and thickness of plastic may have been different on early prototype units, it is unlikely that they would differ significantly from one production AIT system to another.
When other parameters are constant, the effective dose varies directly with the scan duration. Although the scan rate, and therefore the duration, are under software control and can be adjusted by someone with the appropriate software access, it is unlikely in the usual course of operation that scan duration could be modified by more than 30 percent (i.e., increased from 3 seconds to 4 seconds) without being noticed by the operator. The fourth parameter, the location of the subject, is controlled by “footprints” painted on the floor indicating the proper pose.
With respect to the techniques used to measure kerma and HVL, one major difference in the various studies was the modification of the scanning process to increase the photon fluence available for measurement by repeating the scan at a single vertical location. The X-ray beam includes a penumbra of photons scattered by the collimator, and the contribution of these lower-energy photons to the ion chamber current may differ if the whole detector, or one line across it, is irradiated. This could lead to a small difference in the HVL, depending on the measurement conditions. This difference is expected to be very small because the collimator components appear to be made of high Z materials, and the photoelectric effect will dominate the interactions in the collimator.
Additional differences in measured HVL and kerma may result from differences in the instrumentation or techniques used for the measurements. The measurement of kerma was generally done with an ion chamber calibrated for a specific photon spectrum. If the photon spectrum produced by the AIT system does not exactly match the spectrum used for the calibration, small errors in the measurement of kerma may occur. The measurement of HVL is simpler in one way: only relative measurements are needed, but it is technically more challenging because the structure of the measuring device may be modifying the measurement. Ideally, HVL measurements would be made using a wall-less detector thin enough such that there is no variation in photon spectrum through the detector. For gas-filled detectors, the second half of this requirement is easily met, but the presence of an ion chamber wall is the equivalent of adding material to the filtering inherent
_______________
7 Glassman High Voltage, Inc., http://www.glassmanhv.com/, accessed September, 5, 2014.
in the AIT system.8 Thus, if the ion chamber used to measure HVL has a 5-mm-thick plastic wall, it will determine the HVL present at a depth of 5 mm of plastic. Differences in the structure of the ion chambers used to measure HVL may be responsible for significant differences in the measured result.
The purpose of the JHU/APL study9 was to conduct a radiation safety engineering assessment of the Rapiscan Secure 1000 in single-pose configuration.10 Measurements were made at the Rapiscan factory using two dual-pose engineering units (dated 2005 and 2007) and other components from inventory. The resulting AIT system was configured to be equivalent to a unit then being evaluated by the Transportation Security Laboratory.
Measurements
- Person being scanned
— Air kerma measurements were made using a 10X5-1800 ionization chamber manufactured by Radcal Corporation.11 This chamber was constructed with a polycarbonate wall (i.e., LexanTM) with a conductive graphite coating. The wall thickness was 3.175 mm (0.125 in.) corresponding to an arealdensity of 0.38 g/cm2. This corresponds to the aluminum equivalent thickness of 1.4 mm. The active volume was 1,800 cm3. The central collector was a cylinder centrally located in the sensitive volume of the detector with a length of 7.8 cm (3.0 in.) and diameter of 3.6 cm (1.4 in.) and 0.07 cm (0.028 in.) wall thickness.
Measurements were made using the full vertical and horizontal rasterscanning conditions at a standard location, representative of the exposure received by the individual being scanned. The detector was centered midway between the entry and exit portals and 30 cm from the surface of the wall where the beam exits. The ion chamber was orientated so that the incident beam was parallel to the long axis of the cylindrical detector. To shield the ion chamber from extraneous radiation, a piece of lead was placed on top
_______________
8 Calibration to a specified photon spectrum corrects the measured kerma for attenuation in the detector wall, but that attenuation continues to be included in a depth per dose or HVL measurement.
9 JHU/APL, Radiation Safety Engineering Assessment Report for the Rapiscan Secure 1000 in Single Pose Configuration, 2010.
10 Single-pose configuration means that the subject is scanned only a single time by two AIT systems, one for the front and one for the back.
11 Radcal Corporation, “10X5-1800,” https://www.radcal.com/10x5-1800, accessed September 5, 2014.
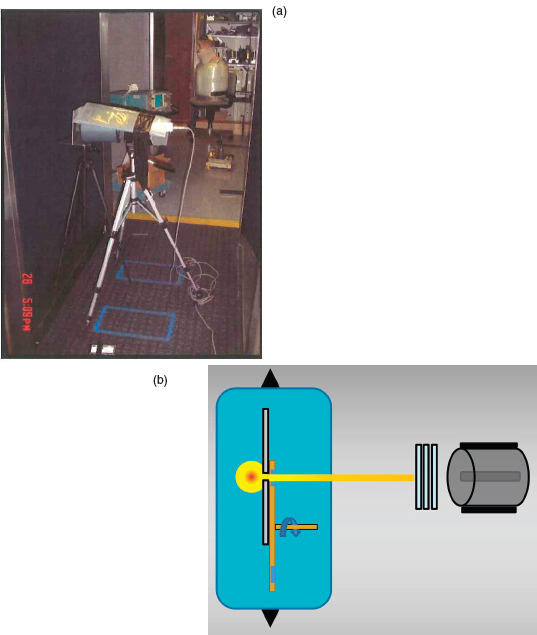
FIGURE 6.1 (a) The location of the 1800 cc ion chamber with lead shield for the JHU/APL kerma measurements. (b) Schematic of the configuration used by JHU/APL to determine half-value layer. SOURCE: (a) Department of Homeland Security, Radiation Safety Engineering Assessment Report for the Rapiscan Secure 1000 in Single Pose Configuration, August 2010, http://www.dhs.gov/advanced-imaging-technology-documents; (b) Courtesy of Tom Borak.
- of the chamber and wrapped around the sides (Figure 6.1a). The date of calibration was indicated, but details of calibration vendor and beam quality were not indicated. The report indicated that “as a conservative measure, the dose conversion coefficients were selected based on an operating potential of 50 kV.” The average air kerma was found to be 41 nGy per scan (82 nGy per screening).
— HVL measurements were made using the 1800 cc ionization chamber with the long axis oriented parallel to the beam. The Rapiscan was operated in the full scanning mode with vertical translation of the X-ray assembly, horizontal collimator, and rotating chopper wheel. Lead shielding was wrapped around the ionization chamber to shield from ambient radiation and inscatter, but the beam was not collimated before the attenuation foils (Figure 6.1b). The first HVL for the anterior unit was found to be 1.18 mm of aluminum, but the first HVL of the posterior unit was 1.63 mm of aluminum. No explanation was given for this unusually large difference in HVL for nominally identical sources. - Bystander. Geiger-Mueller and scintillator probes were used to survey the radiation outside the inspection volume. In order to simulate radiation scattered by a person being scanned, a phantom consisting of four 5-gallon containers of water supported by stacked bins was placed at the position of the person being scanned. Locations of maximum count rate were recorded and measurements were made at those locations with the 1800 cc ion chamber and with the AIT system in normal full-scanning operation. Background measurements for the same total time were made and subtracted from the readings of dose during scanning. A maximum of 10 nGy per screening was observed in locations above the inspection volume. The maximum in a location that could be occupied by the equipment operator was 8.4 nGy per screening.
Calculations
- Person being scanned. Applying the ANSI/HPS N43.17-2009 method,12 a reference effective dose of 14.6 nSv per screening was calculated.
- Bystander. Dose rate to bystanders was calculated to range from up to 1,285 nSv per hour, assuming 180 screenings per hour.
_______________
12 The ANSI/HPS N43.17-2009 standard, “Radiation Safety for Personnel Security Screening Systems Using X-Ray or Gamma Radiation,” is available at the Health Physics Society website at http://hps.org/hpssc/index.html.
Procedures and Interlocks
JHU/APL considered several potential failure mechanisms that could result in X-ray overexposure to the person being screened or the bystanders and screeners. On the engineering AIT system used for exposure dose measurements, investigators verified through measurements that the X-ray source is activated only by the “scan” button and that the X-ray source will not power on if any of the panel doors are not completely closed. They also measured the X-ray fluence as zero at the end of the scan, which indicates that the X-ray beam is off. They verified that the key used to turn the AIT system on cannot be removed unless it is in the “off” position, indicating that there can be no power to the X-ray source unless the key is present and in the “on” position. Additionally, JHU/APL reviewed the AIT system documentation and determined that the AIT system design operates the X-ray tube at its maximum voltage. Thus, it cannot produce X-rays of a higher energy or a higher fluence than the design settings, and the AIT system interlocks limit the “beam on” time to 3 seconds; there is no risk of increased exposure time.
The NIST report13 describes radiation-exposure testing of the Rapiscan Secure 1000 Single Pose ATR (automatic target recognition) version (serial number 1041001, manufactured October 2010) in the laboratories of the Dosimetry Group at NIST.
Measurements
- Person being scanned
— Air kerma measurements were made using a 10X5-1800 ionization chamber manufactured by Radcal Corporation.14 This chamber was constructed with a polycarbonate wall (i.e., LexanTM) with a conductive graphite coating. The wall thickness was 3.175 mm (0.125 in.) corresponding to an areal density of 0.38 g/cm2. This corresponds to an aluminum equivalent thickness of 1.4 mm. The active volume was 1,800 cm3. The central collector was a cylinder centrally located in the sensitive volume of the detector with a length of 7.8 cm (3.0 in.) and diameter of 3.6 cm (1.4 in.) and 0.07 cm (0.028 in.) wall thickness.
Measurements were made using the full vertical and horizontal raster
_______________
13 Glover et al., Assessment of the Rapiscan Secure 1000 Single Pose (ATR version) for Conformance with National Radiological Safety Standards, 2012.
14 Radcal Corporation, “10X5-1800,” https://www.radcal.com/10x5-1800, accessed September 5, 2014.
- scanning conditions. Measurements were made at a standard location representative of the exposure received by an individual, 100 cm above the floor midway between the entry and exit portals and 30 cm from the surface of the wall where the beam exits to the center of the ion chamber (Figure 6.2a). The ion chamber was orientated so that the incident beam was perpendicular to the long axis of the cylindrical detector. Measurements of air kerma were also taken at the ANSI-specified Reference Location representing the point of maximum exposure, but no closer than 30 cm to the surface where the beam exits, at a height of 185 cm above the floor, midway between the entry and exit portals and 30 cm from where the beam exits the front panel. The ionization chamber was calibrated using the M50 beam quality at the NIST X-ray calibration facility. This beam had a HVL of 1.04 mm of aluminum. The measured air kerma at the standard location was 47 nGy while at the maximum location 185 cm above the floor; it was 92 nGy when averaged over the area of the ion chamber.
— HVL measurements were made using the 1800 cc ionization chamber with the long axis oriented vertically and perpendicular to the beam. The Rapiscan was operated in an engineering mode with vertical translation of the X-ray assembly disengaged. The X-ray source remained at a fixed height. The horizontal collimator and rotating chopper were in place and operated normally such that the flying spot made a repeating horizontal trace. A collimator was placed outside of the X-ray assembly upstream of the aluminum absorbers. This restricted the horizontal dimensions of the raster scanned beam incident upon the aluminum absorber. Another circular collimator was placed between the aluminum absorbers and ionization chamber to intercept scattered radiation from the aluminum and to create conditions for narrow beam geometry (Figure 6.2b-c). No significant difference was found between the first HVL for the anterior and posterior units. The HVL was found to be 1.09 mm of aluminum. - Bystander. Air kerma was measured at representative locations outside the inspection area with a RANDO phantom being scanned. Measurements were made using the 1800 cc ion chamber by averaging the charge collected over 20 scans occurring in a 5-minute interval. At many locations, the kerma was found to be less than 0.1 nGy per scan, the minimum detectable. At locations expected to have the highest kerma, based on results of a dose rate survey using a Technical Associates Neon-P8 probe, the kerma was found to range from 0.5 to 0.9 nGy per scan.
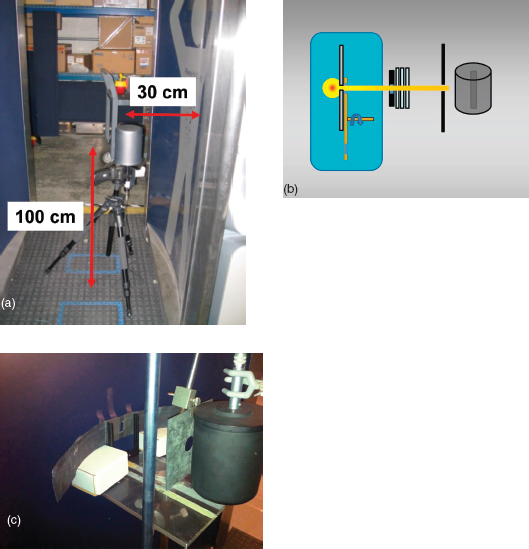
FIGURE 6.2 (a) Configuration of the 1800 cc ion chamber used by NIST to evaluate kerma. (b) Schematic and (c) photograph of the detector arrangement used by NIST to measure half-value layer. SOURCE: (a) American Association of Physicists in Medicine Task Group 217; (b) Tom Borak; (c) NIST.
Calculations
- Person being scanned
— Reference effective dose was calculated using the ANSI N43.17 relationship. The result was 12.6 ± 0.8 nSv per screening at the position that resulted in the maximum kerma, near the top of the scan range. A more realistic reference effective dose, typical of potentially occupied spaces, was 9.23 nSv per scan (18.5 nSv per screen).
- — Effective dose was calculated using the PCXMC Monte Carlo code that uses age dependent Cristy-Eckerman (geometric shape based) computational phantoms. This code uses the X-ray tube characteristics plus the “aluminum-equivalent filtration” to calculate the photon spectrum used to evaluate organ doses. The aluminum-equivalent filtration of 1.21 mm was obtained from the spectrum generator program in order to match the measured 1.085 mm of aluminum first HVL. The calculated effective dose for a properly positioned adult was 14.7 nSv. The effective dose for an adult ranged from 11.9 to 22.4 nSv, depending on positioning in the inspection area (10 cm from front panel of anterior unit to 10 cm from front panel of posterior unit). The effective dose to a standard 5-year-old child (19 kg, 109.1 cm tall, trunk thickness 15 cm) was 21 nSv when properly positioned in the scanning area.
- Bystander. Ambient dose equivalent to bystanders was calculated using conversion factor 0.85 Sv/Gy derived from ANSI N43.17, Figure C1, assuming an HVL of 1.1 mm of aluminum. Using the maximum kerma measured and assuming a workload of 180 scans per hour, the maximum ambient dose equivalent rate for bystanders was 276 nSv/hour.
Procedures and Interlocks
The NIST report did not indicate that potential failure mechanisms or verified safety interlocks were considered during the AIT system evaluation.
The aim of the AAPM report15 was to utilize the expertise of the medical physics community to make independent, detailed measurements of the Rapiscan Secure 1000 single-pose system output. The goal was to make measurements on multiple commercial AIT systems, both at the factory and in the field (i.e., in AIT systems in actual use in an airport setting).
Measurements
- Person being scanned
— Air kerma measurements were made using a 10X5-1800 ionization cham-
_______________
15 AAPM, Radiation Dose from Airport Scanners: Report of AAPM Task Group 217, College Park, Md., 2013.
- ber manufactured by Radcal Corporation.16 This chamber was constructed with a polycarbonate wall (i.e., LexanTM) with a conductive graphite coating. The wall thickness was 3.175 mm (0.125 in.), corresponding to an areal density of 0.38 g/cm2. This corresponds to an aluminum equivalent thickness of 1.4 mm. The active volume was 1,800 cm3. The central collector was a cylinder centrally located in the sensitive volume of the detector with a length of 7.8 cm (3.0 in.) and diameter of 3.6 cm (1.4 in.) and 0.07 cm (0.028 in.) wall thickness.
Measurements were made on three AIT systems (1.5 SP systems) at the Rapiscan factory and three AIT systems at Los Angeles International Airport. Measurements were made at a standard location representative of the exposure received by the individual being screened. This was similar to the NIST study reviewed above, but the height is 91 cm above the floor, midway between the entry and exit portals, and 30 cm from the surface of the wall where the beam exits. The ion chamber was orientated so that the incident beam was perpendicular to the long axis of the cylindrical detector. The ionization chamber was calibrated using the M50 beam,17 which has a HVL of 1.04 mm of aluminum. The average air kerma at the reference point for nine AIT systems was 46 nGy with a standard deviation of 3 nGy and a range of 40 to 52 Gy.
— HVL measurements were made both at the factory and at the airport. Measurements at the factory were made with a 6 cc thimble ion chamber suitable for mammography beams with the scanning mechanisms completely disassembled. This created a stationary beam defined only by the horizontal slit attached to the X-ray tube housing. Measurements were made with beam collimation before and after the aluminum absorber (Figure 6.3). Measurements at the airport were made with the 1800 cc ion chamber described above. For the factory bench top configuration, which did not include the AIT system faceplate, the HVL was 0.81 mm of aluminum. For the measurements at the airport, the HVL was 0.93 mm of aluminum. - Bystander. An attempt was made to determine the dose to bystanders using both the 1800 cc chamber and a Fluke 451P survey meter. Measurements were made at the airport, with an anthropomorphic torso phantom in the AIT system. Locations expected to have maximum scattered radiation were measured by integrating the reading for 10 screenings, but no exposure above background was detected.
_______________
16 Radcal Corporation, “10X5-1800,” https://www.radcal.com/10x5-1800, accessed September 5, 2014.
17 For a definition of M50 beam, see Table 3 in NIST Handbook 150-2D, Calibration Laboratories: Technical Guide for Ionizing Radiation Measurements, 2004.

FIGURE 6.3 (a) Schematic and (b) photograph of the system used by AAPM to measure half-value layer. SOURCE: (a) Tom Borak; (b) American Association of Physicists in Medicine Task Group 217.
Calculations
- Person being scanned
— Reference effective dose was calculated using the ANSI N43.17 formula. The result was 11 nSv per screening.
— Effective dose and average absorbed dose for three organs were calculated for the standard man, a 10-year-old child (height 1.398 m, mass 32.4 kg), a Centers for Disease Control and Prevention (CDC) average U.S. adult, and a CDC 90th percentile adult using the PCXMC Monte Carlo code. The input data for beam quality was 50 kV, 20 degree anode angle, and 1 mm of aluminum total filtration to give the measured 0.93 mm of aluminum HVL. The results are summarized in Table 6.1. - Bystander. No calculations were made for the bystander.
Procedures and Interlocks
The AAPM report includes potential failure mechanisms that could result in overexposure to the person being screened or the bystanders or screeners, including
TABLE 6.1 Effective Dose and Three Organ Doses Calculated Using PCXMC and Measured Kerma
| Phantom | Effective Dose (nSv) | Skin Dose (nGy) | Breast Dose (nGy) | Thyroid Dose (nGy) |
| Standard man | 11.1 | 40.4 | 20.0 | 21.0 |
| 10–year-old child | 14.8 | 42.4 | 32.8 | 22.4 |
| CDC average adult | 10.4 | 41.0 | 19.2 | 20.0 |
| CDC 90th percentile | 9.4 | 40.9 | 18.7 | 18.2 |
NOTE: CDC, Centers for Disease Control and Prevention.
overvoltage of the X-ray tube and the vertical scan stopping during the screening and increasing the exposure dose to one area of the passenger. They did not verify design or interlocks that would prevent these scenarios but indicated that the power supply to the X-ray tube is already operating at its maximum voltage and that the image would be affected in a noticeable manner if the vertical or horizontal beam motion stopped.
The primary focus of the USAPHC report was to estimate the effective dose per screening to individuals being scanned and to operators and others in the area of the AIT system using passive dosimeters. Dosimeters have response characteristics that are essentially independent of dose rate. The use of these passive dosimeters avoids the possible complications of high dose rate in the beam, which has been mentioned as a concern when the effective dose, E, is determined based on measurements of kerma, K, using a large ion chamber. The most sensitive passive dosimeters available for this type of measurement are aluminum oxide crystals, with the absorbed dose determined by optically stimulated luminescence (OSL).
A commercial dosimetry service was used for these measurements. The minimum dose reported by the vendor for OSL dosimeters used in this experiment was 10,000 nSv, and the resolution for higher doses was also 10,000 nSv. In order to produce data with nanosievert-per-screening resolution, the dosimeters were subjected to 93,105 screenings in a Rapiscan Secure 1000 single-pose system before being returned to the dosimetry service for reading. For each dosimeter, utilizing a system of physical filters over the OSL chip, the dosimetry service provided estimates of the deep dose equivalent (DDE), lens dose equivalent, and shallow dose equivalent. They also interpreted the readings behind the separate filters slightly differently, assuming that the ambient radiation field is a mixture of photon and
electron radiation, unless they were informed that it was a pure photon field. In this report, it was assumed that the true dose at each depth is between the values obtained assuming a pure photon field and a mixture of photons and electrons.
Measurements
- Person being scanned. —In order to evaluate the radiation exposure produced by the Rapiscan Secure 1000 single-pose system, a physical phantom consisting of plastic containers of water was assembled on a wooden framework in the inspection area. OSL dosimeters were placed on the surface of the plastic containers facing the anterior AIT system unit at nine different locations representing specific locations on the body. These dosimeters do not provide a value for air kerma. In addition, measurements of HVL and kerma were made using a Radcal Model 10X5-1800 ion chamber. The results were as follows: a HVL of 1.646 (anterior unit) and 1.281 (posterior unit); kerma of 63 (anterior) and 55 (posterior) nGy.
- Bystander. Dosimeters were also placed at the outer edges of the shielding wings of the AIT systems and on a plastic framework just outside the inspection area in order to determine the dose to AIT system operators.
Calculations
- Person being scanned. The use of dosimeters on only the surface of the phantom did not provide the data needed to evaluate the average dose to each organ, as is required for evaluation of effective dose. Instead, it was assumed that the DDE, which estimates the dose at 1 cm depth, would overestimate E because most of the organs used to evaluate E are deeper than 1 cm and therefore receive less dose when exposed to the soft X rays used for scanning. Thus, the DDE was assumed to be an acceptable estimate of E.
The average DDE to the nine locations on the water phantom was 49 nSv per screening using the photon-only algorithm to evaluate the OSL reading and 38 nSv per screening using the mixed-radiation algorithm. These values are somewhat higher than the values of E obtained in other studies, as expected, but confirm that the effective dose is substantially less than the 250 nSv limit. These results also indicate that measurements of kerma made with a large-volume ion chamber do not have serious errors due to the dose rate in the beam and possible recombination issues.
The reference effective dose was calculated using the ANSI N43.17 formula and ion chamber data, resulting in EREF equal to 33 nSv.
- Bystander. DDE measured outside the inspection area ranged from below the detection limit to 140,000 nSv with an average (counting below limit readings as 10,000 nSv) of 11,300 nSv for the 93,105 screenings. Per screening, this is a maximum of 1.5 nSv and an average of 0.12 nSv.
Procedures and Interlocks
The USAPHC report includes potential failure mechanisms that could result in overexposure to the person being screened or the bystanders or screeners, including overvoltage of the X-ray tube and the vertical scan stopping during the screening and increasing the exposure dose to one area of the passenger. They did not verify design or interlocks that would prevent these scenarios but indicate that the operator cannot adjust the power to the X-ray tube or the scan speed, so these failure modes cannot be caused by operators.
In 2006 a Rapiscan Secure 1000 dual-pose AIT system (serial number S701201213, dated May 2001) was evaluated at the FDA’s Center of Devices and Radiological Health.18 The AIT system was evaluated against ANSI N43.17-2002, which was the current standard at the time. Tests confirmed that the tube anode potential was 50 kV, the same as the units later installed at airports.
Measurements
- Person being scanned
— Exposure (proportional to kerma) was mapped using the solid-state detector and measured using a 10X5-1800 ion chamber manufactured by Radcal Corporation19 mounted with its axis vertical and 30 cm from the face of the AIT system. Ion collection efficiency at the operating voltage of 300 V was evaluated by decreasing it to 270 V. This decrease resulted in only a 2.3 percent decrease in charge per scan, indicating that very little recombination was occurring at 300 V. The measured exposure per scan was 9,600 nrem, equivalent to kerma of 84 nGy at 30 cm from the face of the AIT system and 100 cm above the floor (the height of maximum kerma in the range of 0 to 180 cm). The kerma decreased to approximately 60 percent of this value at
_______________
18 Cerra, Assessment of the Rapiscan Secure 1000 Body Scanner for Conformance with Radiological Safety Standards, 2006.
19 Radcal Corporation, “10X5-1800,” https://www.radcal.com/10x5-1800, accessed September 5, 2014.
- heights of 0 cm and 180 cm, but a peak value of 2.5 times the value at 100 cm was found at 197 cm for alternate scans.
— HVL was determined using a R100B solid-state detector placed at 50 cm from the AIT system face and 90 cm above the floor. HVL was measured during normal scan operation. To ensure good (narrow beam) geometry, a 6.25 mm thick lead shield with a 25 mm diameter aperture was placed 35 cm from the face of the AIT system, in line between the X-ray tube and the detector. High-purity aluminum absorbers were placed on the detector side of the shield. The first HVL was found to be 1.1 mm of aluminum, but a second HVL was not determined. - Bystander. The AIT system was monitored for leakage radiation using an array of eight Geiger-Muller detectors 5 cm in diameter. No detectable leakage was found over the majority of the AIT system surface, but leakage was found along a line up both sides and across the top of the unit, approximately 29 cm from the front edge of the unit. Using the 1800 cc ion chamber, the maximum leakage rate was found to be 75 nR (0.65 nGy) per scan. Radiation scattered from a phantom was measured with the 1800 cc ion chamber (calibrated for the M50 spectrum) at two locations, on the mid-plane of the phantom and on the plane of the AIT system back plate (100 cm above the floor and 30 cm outside the plane of the AIT system side surface in both cases). The scattered radiation was found to be 200 nR (1.8 nGy) per scan and 110 nR (0.96 nGy) per scan, respectively.
Calculations
- Person being scanned
— Based on fitting the kerma as a function of distance (r) from the AIT system face to a 1/r2 relationship, it was concluded that the effective position of the X-ray source was 91 cm behind the face of the AIT system.
— The first HVL was compared with data for HVL as a function of total filtration for a 50 kV spectrum, and it was determined that the effective filtration was 1.4 mm of aluminum, which was supplied to the PCXMC program for determining an effective dose of 23.6 nSv per frontal scan. - Bystander. No results were provided.
Procedures and Interlocks
Not applicable.
SUMMARY OF MEASUREMENTS FROM REVIEWED STUDIES
The characteristics of the detectors used and the AIT system operation mode utilized to make the measurements reported in the reports reviewed in the previous sections are summarized in Table 6.2. The results are summarized in Tables 6.3 and 6.4.
The studies described above do not define how they reached margins of error for their estimates of average exposure. At a minimum, all of the studies deviate from standard statistical methods for describing the uncertainty in key parameters. A more detailed discussion on the statistics for some of the available studies is available in Appendix D.
TABLE 6.2 Characteristics of the Rapiscan Secure 1000 Measurements in the Reviewed Reports
| Report | AIT System Type (Pose) | Kerma Detector | Kerma Scan | Kerma Calibration Spectrum | HVL Detector | HVL Scan | Leakage Detector |
| USAPHC | Single | 1800 cc | Full | Unknown | 1800 cc | Full | OSL |
| FDA | Dual | 1800 cc | Full | Solid state | Full | 1800 cc GM survey | |
| JHU/APL | Single | 1800 cc | Full | Unknown | 1800 cc | Full | 1800 cc GM survey |
| AAPM | Single | 1800 cc | Full | M50 | 6 cc thimble | None | 1800 cc |
| 1800 cc | full | ||||||
| NIST | Single | 1800 cc | Full | M50 | 1800 cc | Line | 1800 cc GM survey |
NOTE: GM, Geiger-Muller tube; HVL, half-value layer; N/A, not applicable; OSL, optically stimulated luminescence.
TABLE 6.3 Summary of Rapiscan Secure 1000 Measurement Results in the Reviewed Reports
| KStd (nGy) per Scan |
Kmax (nGy) per Scan |
HVL1 (mm of aluminum) |
|
| USAPHC | 63 | N/A | 1.646 (anterior) |
| 1.281 (posterior) | |||
| FDA | 84 | 212 | 1.1 |
| JHU/APL | 41 ±1 | N/A | 1.18 (anterior) |
| 1.63 (posterior) | |||
| AAPM | 46 ±3 | N/A | 0.93 |
| NIST | 68 ±1 | 95 ±6 | 1.09 |
NOTE: HVL, half-value layer; N/A, not applicable.
TABLE 6.4 Effective Dose per Rapiscan Secure 1000 Screening Results in the Reviewed Reports
| Person Screened | ||
| EREF (nSv) | E (nSv) | |
| USAPHC | 38-49a | NA |
| FDA | 29 | 23.6b |
| JHU/APL | 14.7 | NA |
| AAPM | 10.7 | 11.1 |
| NIST | 18.5 | 14.7 |
a Deep dose equivalent.
b Per anterior scan.
SUMMARY OF THE INTERLOCK TOPIC FROM REVIEWED STUDIES
Three of the reviewed studies considered the potential for accidental or deliberate malfunction of the Rapiscan Secure 1000 that could result in X-ray overexposure to the person being scanned, bystanders, or the operators. The USAPHC, JHU/APL, and AAPM reports indicate that increasing the voltage or current to the X-ray tube could result in increasing maximum X-ray energy or increasing the X-ray photon flux, either of which would result in an increased X-ray dose to the person being screened. All three studies concluded that the X-ray tube is operated at its maximum voltage and that the operator does not have access to increase the voltage or current to the X-ray tube. In addition, the JHU/APL report refers to an interlock documented in the AIT system manual that includes a photodiode measurement that reflects the X-ray photon flux and is interlocked to turn off the voltage to the X-ray tube if the flux increases from nominal by more than 17 percent or is more than 23 percent below nominal.
The USAPHC, JHU/APL, and AAPM reports also consider a malfunction where the vertical scan slows or stops, resulting in increased X-ray exposure to the person being scanned. Both the USAPHC and AAPM reports note that the operator cannot adjust scan speed and that if the beam stops, the image would be affected and noticed by the screener reviewing the images. The JHU/APL report review of the AIT system manual also notes that an interlock prevents the “beam on” time from exceeding 3 seconds, which would not result in overexposure to the person being screened. The JHU/APL report also notes that the Secure 1000 meets the ANSI N43.17-2002 standard requirement 6.2.2, “Subject exposure during a malfunction.”
The JHU/APL group verified several other interlocks against potential malfunctions on a Secure 1000 engineering AIT system built for their measurements at the Rapiscan factory. They verified that the X-ray beam cannot be activated
without the power key present and in the “on” position, and the X-ray source does not remain on after the scan is complete. Additionally, the panel doors are interlocked, and the group verified that the X-ray source cannot be activated if the panel doors have been removed.
It is worth mentioning that Mowery et al.20 investigated potential cyber physical threats and discusses software and hardware issues that could compromise the safety of the device. The paper (reviewed in the section below) makes the following observations: the PC controlling the AIT system could potentially be infected with malware; there is a bypass signal that disables the hardware interlocks; all safety interlocks can be bypassed by the software; and corrupt firmware could potentially deliver the whole radiation dose from a scan to a single random point on the body. The authors tried to cause the code on the PC to malfunction but failed.
In summary, none of the reports identified a potential AIT system malfunction that would result in overexposure to the person being screened or to the operator. For an overview of all the information this far, see Tables 6.5 and 6.6.
OTHER STUDIES AND CONSIDERATIONS
There have been additional studies and reports that examined the dose received during X-ray backscatter AIT screening. Three are described here in some detail. These studies are based on previous measurements and estimates of AIT system parameters.
The Dose from Compton Backscatter Screening
Retz et al.21 report an effort to determine the effective dose per screening by estimating the number of scattered photons per pixel that must be detected to produce a usable image. They then estimate the number of photons that must be incident on the surface of the scanned individual in order to produce the needed number of scattered photons. Finally, they estimate the effective dose from the number of incident photons per pixel. In order to make these calculations, a great deal of information is required about the scanning AIT system, including the beam spot size, the photon spectrum, the beam scanning rate, the location and efficiency of the scattered photon detectors, and much more. However, most of this information was not available to the authors because it was considered proprietary by the
_______________
20 K. Mowery, E. Wustrow, T. Wypych, C. Singleton, C. Comfort, E. Rescorla, S. Checkoway, J.A. Halderman, and H. Shacham, Security analysis of a full-body scanner, Proceedings of the 23rd USENIX Security Symposium, 2014, https://www.usenix.org/conference/usenixsecurity14/technical-sessions/presentation/mowery.
21 P. Retz, R.L. Metzger, and K.L. Mossman, The dose from Compton backscatter screening, Radiation Protection Dosimetry 145:75-81, 2010.
TABLE 6.5 Summary of Rapiscan Secure 1000 Malfunction Investigations from the Reviewed Reports
| Report | Potential Malfunction | Page | Conclusion | How Verified |
| JHU/APL | X-ray source activated by “on” switch without pressing “scan” button | 36 | X-ray source activated only by “scan” button | Verified by measurement on engineering AIT system |
| X-ray source can be activated with power key removed | 36 | Key can be removed with power switch only in “off” position | Verified on engineering AIT system | |
| X-ray source can be activated for a scan with panel doors removed | 37 | Panel doors are interlocked, and AIT system will not scan if interlock is tripped | Verified on engineering AIT system | |
| X-ray beam fails to turn off at the end of a scan | 41 | No X-rays are emitted after a scan is complete | Verified by measurement on engineering AIT system | |
| X-ray tube can be operated with higher voltage resulting in higher X-ray photon flux and higher X-ray photon energies | 38 | X-ray tube is operated at the maximum voltage. Photodiode measures X-ray photon intensity and is interlocked if the flux varies from nominal by <−23% or +17% | Confirmed in AIT system documentation | |
| Vertical scan stops and X-ray exposure is focused on one area of the body for enough time to overexpose the passenger | 40 | AIT system interlock limits “beam-on” time to 3 seconds, which would not result in overexposure. Meets ANSI requirement N43.17.2002 6.2.2 “Subject exposure during a malfunction” | Confirmed in AIT system documentation | |
| AAPM | X-ray tube can be operated with higher voltage resulting in higher X-ray photon flux and higher X-ray photon energies | 13 | Power supply cannot provide voltage higher than tube specification | No evaluation |
| Vertical scan stops and X-ray exposure is focused on one area of the body for enough time to overexpose the passenger | 13 | If beam stops, the image would be affected and the operator would notice “immediately” | No evaluation | |
| Report | Potential Malfunction | Page | Conclusion | How Verified |
| USAPHC | X-ray tube can be operated with higher voltage or current resulting in higher X-ray photon flux or higher X-ray photon energies | 2 | Operator cannot adjust X-ray tube voltage or current | No evaluation |
| Vertical scan speed is lower than set value and increases X-ray exposure, resulting in overexposure to the passenger | 2 | Operator cannot adjust scan speed | No evaluation | |
TABLE 6.6 Summary of Rapiscan Secure 1000 Malfunction Investigations from the Committee’s Investigation of the Unit at NIST
| Report | Potential Malfunction | Conclusion | How Verified |
| Committee | X-ray source stays on after emergency-off (EMO) button is pressed | X-ray source turns off when EMO button is pushed; vertical scan bar completes full travel to top or bottom | Verified by EMO activation on engineering AIT system |
| X-ray source activated by “on” switch without pressing “scan” button | X-ray source only activated by “scan” button | Verified by measurement on engineering AIT system | |
| X-ray source can be activated with power key removed | Key can be removed only with power switch in “off” position | Verified on engineering AIT system | |
| X-ray source can be activated for a scan with panel doors removed | Panel doors are interlocked, and AIT system will not scan if interlock is tripped | Verified on engineering AIT system | |
| X-ray beam fails to turn off at the end of a scan | No X-rays are emitted after a scan is complete | Verified by measurement on engineering AIT system | |
| Vertical scan can be locked into a single position | Cannot initiate scan if vertical scan motor is turned off; can activate X-ray source with vertical scan locked only in engineering mode, which requires a separate password | Verified on engineering AIT system | |
AIT system manufacturers. In order to proceed with the calculations, the authors made estimates of the needed information based on a simple analysis of published images made by commercial scanning equipment (for pixel size and gray scale resolution) and on scanning AIT system size and shape (for detector size and location).
Assuming a 50 kV X-ray source, 2 mm square pixel size, a scan duration of 8 seconds, and a maximum of 3,200 photons detected per pixel, they estimated the effective dose of 0.880 nSv per screening, on the order of 20 times the value reported for measurements.22 The authors point out that if the number of scattered photons per pixel is held constant, the effective dose is proportional to 1/(pixel area); similarly, if the pixel area is constant, the effective dose is proportional to 1/(scattered photon number per pixel). Thus, if they had assumed a pixel size of 4 mm square and a maximum of 640 photons, they would have predicted an effective dose of 44 nSv. Although they express the opinion that the difference between their calculation and measured results is likely due to recombination in the ion chambers used to measure air kerma, considering the tenuous nature of their estimates of pixel size and number of scattered photons it is perhaps more likely that the difference is due to differences between their estimated input values and those actually used in AIT systems. The authors also assert that if there is a failure of the scanning AIT system, the beam would have to be turned off within ~15 milliseconds to avoid exceeding an effective dose of 250,000 nSv. This conclusion is apparently based on a misinterpretation of effective dose. Effective dose is the sum of the average dose to each organ times that tissue’s weighting factor. The average dose to an organ (e.g., the skin) is the same if 1,000 pixels receive a dose of 100 nGy or if 1 pixel in a population of 1,000 receives a dose of 100,000 nGy. Thus, the effective dose is relatively insensitive to scanning failures if the total beam time is unchanged.23
Letter to the Presidential Science and Technology Advisor
Four professors at the University of California, San Francisco, questioned the safety of the X-ray backscatter AITs in a letter to the presidential science and tech-
_______________
22 They also estimate that an X-ray tube operating at 125 kV would require an anode current of 3.5 mA. An anode current of over 17 mA would be required to produce the same photon energy fluence for a tube operating at 50 kV. This is at least three times the anode current actually used, indicating that they have also overestimated the number of photons required.
23 The effective dose will depend on which organs and tissues are in the beam when scanning fails. This is because different tissues have different weighting factors and occur at different depths in the body. However, the absorbed dose to the skin is much higher than that to deeper organs and, therefore, dominates the effective dose. Although other organs may or may not be in the beam if scanning fails, the skin will be, and thus the effective dose will be relatively insensitive to the beam location.
nology advisor.24 Many of the concerns expressed in the letter deal with specific aspects of radiation biology or segments of the population with unusual sensitivity to radiation and are outside the scope of this report. However, some of the points raised relate to the physics of energy deposition by ionizing radiation and the definitions of quantities used to describe radiation exposure. These concerns bear, indirectly, on the accuracy and relevancy of measurements of exposure from AIT systems that have been reported previously and would also apply to the measurements made specifically for this report.
The letter asserts that because backscatter screening utilizes a small beam of relatively low-energy photons, it produces unique forms of damage. While it is true that these photons scatter from electrons involved in maintaining molecular structures, and thus break molecular bonds, the vast majority of the bonds broken by any dose of ionizing radiation are broken by the secondary electrons liberated by Compton scattering, or the photoelectric effect. A 28 keV photon will typically interact twice: once by Compton scattering transferring some of its energy to an electron, and then by photoelectric effect, transferring the rest of its energy to an electron and disappearing in the process. These two electrons produce on the order of 1,000 ionizations and many more excitations in the molecules they interact with. Similar ionizations and excitations are produced by the secondary electrons released by photons of any energy above a few hundred electronvolts. Although differences in the energy spectra of electrons produced by photons of different energy produce differences in the average distance between ionizations along the path of an electron, and this may produce small differences in the biological effectiveness of the radiation,25 the interactions produced by the X rays used for backscatter screening are essentially the same as those produced by X rays used in many medical procedures.
The letter also asserts that because the energy delivered by an AIT system is mostly absorbed in the skin, it may produce an excessive risk of skin cancer. Most radiation exposures, including those delivered by inspection systems, do not deliver the same energy per mass to all parts of the body. However, the exposures received by the survivors of the nuclear bombs used in Japan, the source of most of the data used to set radiation protection limits, were nearly uniform. Radiation protection standards in the United States and internationally are based on maintaining the risk of lethal effects at an acceptable level based on an assumption of linear, no threshold, response. Assuming the linear response, the risk of a lethal effect in
_______________
24 J.W. Sedat, Ph.D., Marc Shuman, M.D., David Agard, Ph.D., and Robert Stroud, Ph.D., Letter to Dr. John P. Holdren, Assistant to the President for Science and Technology, April 6, 2010, available at http://www.whitehouse.gov/sites/default/files/microsites/ostp/ucsf-jph-letter.pdf, accessed February 2014.
25 A committee of the National Council on Radiation Protection and Measurements is currently attempting to determine the magnitude of such differences.
a nonuniformly irradiated individual can be evaluated by summing the risks of lethal events in each of the tissues of the body. The bomb survivor data provide the radiation sensitivity information needed to relate the average dose in an organ to the risk in that organ. Following this logic, the International Commission on Radiation Protection developed the quantity “effective dose,” which is the sum of the average dose to each organ times the weighting factor for that organ, a version of the radiation sensitivity as described above. For example, the weighting factor for the skin is 0.01 and the sum of the weighting factors for all other organs is 0.99. It is thus possible to state:
An absorbed dose of 1,000 nGy to the skin only, or 10 nGy to each of the organs of the body (including the skin), results in the same effective dose, 10 nSv.
In addition, a dose of 1,000 nGy to the skin only, or 10 nGy to each of the organs of the body, is expected to result in the same risk of a lethal event. If the irradiation is uniform, that lethal event could occur in any organ, but if the dose is limited to the skin, the lethal event has to occur in the skin. Thus, if the dose is limited to the skin, the risk of skin cancer is no more than the total risk of all cancers that would have occurred if the same effective dose was distributed uniformly.
Furthermore, the letter asserts that the correct quantity for measuring the exposure from an AIT system would be the photon flux. For some types of irradiation, there are significant advantages to the use of charged particle flux—or the equivalent, fluence rate—to characterize the exposure.26 However, for photons over a wide range of energies, there is a simple relationship between fluence and kerma. If the photon spectrum and atomic composition of the target are known, the fluence can be calculated from the kerma, and the kerma can be calculated from the fluence. In the case of the X-ray backscatter AIT systems currently under consideration, the fluence is relatively low because it is limited by the X-ray tube voltage, the anode current, and anode composition, all of which are comparable to that used for a conventional dental X ray. However, the fluence (photons per area) is lower than in a dental X ray because an area is exposed to the beam for only a few thousandths of a second. One way to put the fluence or kerma into prospective is to estimate the number of radiation events in a cell of the skin. If one assumes that the photon spectrum produces an average linear energy of 5 keV/µm and that cells are 10-µm diameter spheres, the average dose to a cell hit by the radiation is 0.01 Gy, and at a kerma of 100 nGy, more than the kerma per screening produced by most AIT systems, the chance of a cell being hit is 10–5—that is, about 1 in 100,000 of the cells in the skin are hit.
_______________
26 International Commission on Radiation Units and Measurements, Report 85: Fundamental Quantities and Units for Ionizing Radiation, Journal of the ICRU 11(1):1-31, 2011.
Estimation of Organ and Effective Dose Due to Compton Backscatter Security Scans
Hoppe and Schmidt27 calculated the organ and effective doses for four voxelized phantoms using published data for incident kerma and X-ray filtration. They did the calculations for a 34-year-old male, a 26-year-old female, an 11-year-old female, and a 6-year-old male, all using 2 mm cubic voxels. They used the air kerma values provided by the JHU/APL report cited above, 41 nGy, to determine the photon fluence. They estimated the photon spectrum using the SPEC78 software assuming the X-ray anode voltage of 50 kV, tube description, and filtration of 1.0 mm of aluminum, the minimum consistent with the ANSI/HPS N43.17-2009 reference effective dose method. They used the estimated photon spectrum to calculate energy deposited in the 1800 cc ion chamber used by JHU/APL in order to determine the kerma to fluence conversion, but they did not do additional calculations to test the accuracy of the photon spectrum. This process has been contested because the ion chamber was simulated as a water cylinder instead of air, which results in an overestimate by a factor of about 3 in the source fluence.28 They found the effective dose for a person at 30 cm from the beam exit surface to range from 50 nSv for an adult male to 70 nSv for the 6-year-old male child. The other two phantoms, female adult and child, both showed 60 nSv per screening. They found the highest organ doses in the adipose tissue, ranging from 37.3 nGy in the adult male to 63.3 nGy in the female child, per scan. They present numbers for the skin, but the 2 mm pixel size averages the skin dose with that of adjacent tissue and introduces significant errors due to partial pixels in the field, so these numbers are not reliable.
Security Analysis of a Full-Body Scanner
Mowery et al. 29 performed a radiation dose assessment of the Rapiscan Secure 1000 by measuring the dose from a total of 4,033 scans totaling 6 hours and
_______________
27 M.E. Hoppe and T.G. Schmidt, Estimation of organ and effective dose due to Compton backscatter security scans, Medical Physics 39:3396-3403, 2012.
28 A revised estimate of effective dose to passengers was published in Medical Physics, based on comments from NIST (see J.L. Glover and L.T. Hudson, Comment on “Estimation of organ and effective dose due to Compton backscatter security scans” [Med. Phys. 39, 3396 (2012)], Medical Physics 39(9):5782-5787). However, the committee did not have access to the revised material while working on the report and have, therefore, not been able to comment on it.
29 Mowery et al., Security analysis of a full-body scanner, 2014.
23 minutes using 21 InLight30 dosimeters placed on a RANDO31 chest phantom (neck-to-floor distance of 144 cm and source-to-detector distance of 66 cm). As the authors point out, the detectors are sensitive to the angular placement in the radiation field as well as the time from exposure to signal readout (fading). Following the manufacturer’s suggested calibration procedures and using the dose equation algorithm,32 the authors deduce an average DDE per scan to be 73.8 nSv, the average shallow dose equivalent (SDE) to be 70.6 nSv, and the eye lens dose to be 77.9 nSv with a DDE standard deviation of 7.5 and a coefficient of variation of 0.10. The authors do note the higher deep dose than shallow dose and suggest further investigations to account for this unexpected behavior. The authors conclude the paper by stating that the dose equivalent33 measured is below the recommendation of 250 nSv per screening established by the applicable ANSI/HPS standard and that a person would have to undergo approximately 3,200 scans per year to exceed the standard’s annual exposure limit of 250,000 nSv/year. It is also worth mentioning that DDE and SDE are operational quantities used in radiation protection for external radiation. DDE is defined as the dose equivalent at a tissue depth of 1 cm and is not an assessment of the dose equivalent to organs and tissues distributed throughout the entire body; therefore, it should not be used as a surrogate for effective dose received by airline passengers from bilateral scans during a backscatter screen.
FINDINGS AND RECOMMENDATIONS ON EXPOSURE AND DOSE
Key Finding: Previous radiation dose studies employed different methodologies and instrumentation to estimate the dose delivered by Rapiscan Secure 1000 X-ray backscatter AIT systems. The reports reviewed by the committee generally agreed that the effective dose per screening to an average passenger
_______________
30 See Landauer, Inc., “Dosimeters,” http://www.landauer.com/Industry/Products/Dosimeters/Dosimeters.aspx, accessed October 9, 2014. These are aluminum oxide (Al2O3:C) crystals functioning through optically stimulated luminescence (OSL).
31 The Phantom Laboratory, “RANDO® Phantoms,” http://www.phantomlab.com/products/rando.php, accessed October 9, 2014.
32 The shallow dose equivalent (SDE) and deep dose equivalent (DDE) are obtained from a proprietary algorithm using a combination of ratios of the OSL dosimeters behind each of the filters.
33 The authors did not measure HVL in aluminum or air kerma and thus could not determine the ANSI reference effective dose for the scanning unit. The exact definition of DDE and the extent to which it represents effective dose to the passenger is not clear. Estimating DDE for soft X rays emitted from a tube operating at 50 kV may be problematic because the X rays are not mono-energetic, and most of the incident photons are less than 35 keV.
is about a factor of 10 below the limit of 250 nSV per screening, the dose set by ANSI/HPS N43.17-2009.
Finding: In most cases, kerma was measured with a large cylindrical ion chamber with a relatively thick wall that was calibrated for the M50 NIST spectrum. This chamber averages the kerma over a relatively large area, and the distance between the face of the AIT system and the effective center of the ion chamber is not known exactly. This may have resulted in a small error relative to the kerma at a specified point, the quantity needed to calibrate effective dose calculations.
Recommendation: During kerma measurements, one should use a parallel-plate ion chamber with precisely known effective center and a relatively small diameter.
Finding: In most cases, the HVL was measured using a large cylindrical ion chamber with relatively thick walls (3 mm air equivalent plastic) and varying degrees of control of scattered radiation. In other cases, detectors with unknown cover thicknesses were used. These measurements may misrepresent the first HVL because of attenuation of the lowest energy photons by the wall of the detector or detector cover. None of the measurements included the second HVL.
Recommendation: One should measure first and second half-value layers (HVLs) using a thin-window, parallel-plate ion chamber with effective shielding to ensure narrow beam geometry. The increments of aluminum thickness should be small compared to the HVL in order to maximize the information for comparison with Monte Carlo simulations of attenuation using different photon spectra.
Depth Dose Distribution Measurements
Finding: Depth dose distribution with a resolution of approximately 25 µm in tissue is needed to ensure that the dose to critical cells of the skin in scanned individuals is not being underreported and to provide a test of the photon spectrum used to calculate the effective dose. None of the reports provided the depth dose distribution in tissue in the broad beam geometry.
Recommendation: One should measure depth dose distributions in tissue using a thin-window, parallel-plate ion chamber. Embed the ion chamber in a tissue-equivalent phantom large enough to simulate broad beam geometry, and use tissue-equivalent absorbers with 25 µm depth increments.
Dose Outside Inspection Area Potentially Affecting Bystanders
Outside the inspection area, the dose rate and the photon energy are very low, so using the correct detector is important. Detectors are typically calibrated for a specific photon spectrum—for example, M50—which is expected to approximate the spectrum of the radiation source being measured. However, the average energy of the photon spectrum scattered from a person being scanned will be less than the average energy of the incident spectrum. As a result, an ion chamber calibrated to detect radiation from the incident spectrum may not accurately detect radiation scattered from a person being screened.
Key Finding: Measurements of radiation outside the inspection area have generally been made with detectors that are calibrated for X-ray energies higher than those scattered from a passenger being scanned by the Rapiscan Secure 1000 AIT system. As a result, the detectors may indicate a lower dose than is actually present. The detectors have sometimes failed to distinguish a signal from the background radiation.
Finding: Dose measurements outside the inspection area were often made with radiation survey instruments using pressurized ion chambers or other detectors with unknown, but likely very low, response for photons below 30 keV. Because most of the dose outside the inspection area is probably due to scattered photons, and because the peak of the scattered photon spectrum is well below 30 keV, such detectors may indicate a significantly lower dose than is present.
Recommendation: Dose per screening outside the inspection area should be measured with a thin-window ion chamber or other detector with relatively uniform response to photons down to approximately 10 keV.
Based on all the detailed recommendations from this chapter, it is possible to state a key recommendation.
Key Recommendation: To estimate X-ray radiation exposure outside the inspection area, measurements should be made with detectors calibrated for X-ray energies below the maximum for the advanced imaging technol-
ogy (AIT) system’s X-ray tube and for the radiation levels expected. Use of detectors that are appropriate for other applications but not ideal for measuring dose in a X-ray backscatter AIT system may result in inaccurate measurements.
As discussed in Chapter 7, for both AIT systems the committee studied, the radiation measured outside the inspection area by the National Research Council subcontractor was found to be so low as to be statistically indistinguishable from zero (7.3 ±8.0 nGy) when using appropriate detectors calibrated for the correct energies.






























