Recent scientific evidence points to the origins of childhood obesity as an outcome of the dynamic interplay of genetic, behavioral, and environmental factors throughout early development, with a compelling body of evidence suggesting that both maternal and paternal nutritional and other exposures affect a child’s risk of later obesity. The burgeoning field of epigenetics has led researchers to speculate that many of the observed associations between early developmental exposures and later risk of childhood obesity are mediated, at least in part, through epigenetic mechanisms (see Box 1-1). On February 26–27, 2015, the Institute of Medicine (IOM) Food and Nutrition Board and the IOM and the National Research Council Board on Children, Youth, and Families convened a workshop in Washington, DC, to explore the body of evolving science that examines the nexus of biology, environment, and developmental stage on risk of childhood obesity (see Box 1-2). The workshop focused on the prenatal period, infancy, and early childhood and addressed evidence from both animal and human studies. Workshop objectives developed by the planning committee were to (1) identify epigenetic-mediated relationships between exposure to risk factors during sensitive periods of development (gestation through age 3) and subsequent obesity-related outcomes; (2) explore the science around periods
______________
1 The planning committee’s role was limited to planning the workshop, and this summary has been prepared by the workshop rapporteur as a factual summary of what occurred at the workshop. Statements, recommendations, and opinions expressed are those of individual presenters and participants and are not necessarily endorsed or verified by the IOM, nor should they be construed as reflecting any group consensus.
BOX 1-1
What Is Epigenetics?
Shari Barkin defined epigenetics as “changes in gene expression via changes in posttranslational and posttranscriptional modifications.” Robert Waterland defined it as “the study of mitotically heritable stable alterations in gene expression potential that are not caused by changes in DNA sequence.” However, many workshop participants used “epigenetics” in its broadest sense—that is, in reference to biological phenomena that can be attributed to more than just what genes are present but also whether and how those genes actually function. Derived from the Greek prefix “epi-,” epigenetics literally means “above genetics.”
BOX 1-2
Statement of Task
An ad hoc committee will plan a 2-day public workshop exploring the body of evolving science that examines the nexus of biology, interaction between biology and environment, and developmental stage on risk for childhood obesity. The workshop will include attention to the prenatal period, infancy, and early childhood and will include evidence from animal and human studies. The committee will define the specific topics to be addressed, develop the agenda, and select and invite speakers and other participants. A summary of the workshop will subsequently be prepared by a rapporteur with the assistance of staff and then undergo the National Academies report review process prior to release.
of plasticity and potential reversibility of obesity risk in the context of early childhood development; and (3) examine the translation of epigenetic science to guide early childhood obesity prevention and intervention to reduce obesity risk (see Box 1-3). This report summarizes the information presented and discussed at the workshop and is not intended to serve as a comprehensive overview of the topic. The information and suggestions for future action included here reflect the knowledge and opinions of individual workshop participants and should not be construed as a consensus.
BOX 1-3
Workshop Objectives
The workshop objectives developed by the planning committee were to
- Identify epigenetic-mediated relationships between exposure to risk factors during sensitive periods of development (gestation through age 3) and subsequent obesity-related outcomes.
- Explore the science around periods of plasticity and potential reversibility of obesity risk in the context of early childhood development.
- Examine the translation of epigenetic science to guide early childhood obesity prevention and intervention to reduce obesity risk.
In her welcoming remarks, workshop planning committee chair Shari Barkin described epigenetics as an emerging field of study, one aimed at understanding the phenotypic changes caused not only by changes in DNA, but also by changes in gene expression. For example, when researchers transported larvae from docile European bee hives to a killer bee hive and foster-raised the normally docile bees with the killer bees, they found that not only did the European bees develop an aggressive killer bee phenotype, but their actual gene expression also changed. DNA is a code requiring context. Genes cannot change, but gene expression can. “We are built to be ‘permeable fluid beings,’ rather than ‘solitary unitary isolates,’” Barkin said, referring to science writer David Dobbs’s concept of the recursive reconstruction of the self (Dobbs, 2013). (See Dobbs, 2013, also for a description of the bee-fostering research.) “We’re constantly remaking ourselves and turning ourselves over.”
In humans, good data exist to show that rapid infant weight gain in the first 6 months of life is strongly associated with obesity, insulin resistance, and metabolic dysfunction in early adulthood (Leunissen et al., 2009). The infant weight gain observed in that study represented an increase of just 1 centile, Barkin noted. As a clinician, she regularly observes infants crossing many centiles. She emphasized the dynamic nature of the multiple external and internal factors that interact during this early period and contribute to later childhood obesity, and encouraged workshop participants to consider how scientists’ growing knowledge of this dynamic, including the role of epigenetics, can shed light on where to target childhood obesity prevention efforts (see Figure 1-1).
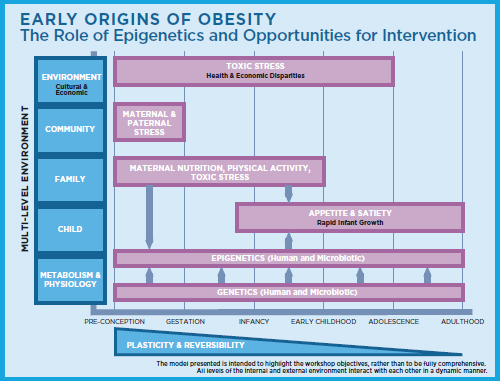
FIGURE 1-1 Early origins of obesity: Multiple external and internal factors interact to cause childhood obesity.
SOURCE: Distributed by the workshop planning committee on February 26–27, 2015.
David Klurfeld of the U.S. Department of Agriculture’s Agricultural Research Service briefly touched on the history of the origins of epigenetics in his opening remarks. Although C. H. Waddington is credited with coining the term “epigenetics” in 1942 to describe how genes interact with their surroundings to produce a phenotype, Klurfeld referred workshop participants to an article in the March–April 2015 issue of American Scientist on Ernest E. Just, an embryologist on the faculty of Howard University in Washington, DC (Byrnes, 2014). While Thomas Hunt Morgan of Columbia University proposed that genes control all cellular events throughout development, Just proposed what he called the theory of genetic restriction, that is, that cytoplasmic factors interact with nuclear elements to induce cell differentiation. The truth, Klurfeld stated, appears to be somewhere between these two extremes. Although the concept, or field, of epigenetics can be traced back at least as far as Just, for Klurfeld epigenetics is new. He described it as a “gee whiz” field, with a lot of fascinating numbers associ-
ated with it, such as the fact that DNA in a single cell is 2 meters long but folds into a nucleus 6 microns across.
For Sandra Hassink, on the other hand, epigenetics has been on the American Academy of Pediatrics’s strategic plan for the past 5 years, representing what she described as a deep desire among clinicians to understand the underpinnings of child health. Hassink showed graphs from the Centers for Disease Control and Prevention of the prevalence of obesity across the United States and stated that two things always strike her when she looks at the graphs. The first is the geographic variation, which leads her to ask what is happening in those areas where prevalence is so high, and the second is that the country is still dealing with massive health disparities. Hassink started her obesity practice in 1988, when, she said, obesity was not on people’s radar screens. Initially, as she watched obesity move through the adult community, she and others thought that children would somehow be spared. But they are not spared. They are suffering from obesity in high numbers at very early ages and with a dramatic impact on their health. Today, according to Hassink, 17 percent of children are obese and 30 percent are overweight or obese. These children, she observed, look in many ways like middle-aged adults in terms of their health profiles. She remarked that while she did not enter pediatrics expecting to see this, in fact that is what pediatricians are seeing and dealing with every day. Moreover, they are operating in what Hassink described as a “very complex socioecological milieu,” and they have only “very blunt instruments,” namely, food and exercise.
Clinicians know that they need to focus on very young children in particular because, Hassink said, once a child develops obesity or an adolescent develops morbid obesity, “there’s no going back,” even with extraordinary effort. But they don’t have ways to intervene in any practical sense. Additionally, clinicians know that what happens to the mother happens to the fetus and infant, with environmental influences having profound effects on the physiology and behavior of energy intake and expenditure. But again, she said, “We don’t know exactly where to break this cycle or how to break this cycle.” Yet, there are many opportunities during early childhood to intervene. Not only is it an important period of time when lifelong nutrition and activity behaviors are being established, but, moreover, the first 20 of a family’s (on average) 31 well-child visits are concentrated in the first 5 years of life, creating a window of opportunity for clinicians to partner with families and to teach parenting skills. The American Academy of Pediatrics is ready, Hassink said, to serve as an engine for translating pediatric obesity research findings into action.
Jamie Bussel of the Robert Wood Johnson Foundation began her welcome remarks by noting that although epigenetics is not on the foundation’s
strategic plan, early childhood is. More than 8 years ago, the foundation announced its $500 million commitment to preventing childhood obesity, which Bussel said was the largest prospective commitment that the foundation had ever made in a single health issue. They set what she described as a “pretty audacious” goal to decrease the prevalence of childhood obesity by 2015. The evidence of progress is “strong and absolutely exciting,” she said. However, while the foundation believes that it has turned a corner on childhood obesity and is on the right track, it has not gone far enough. Despite the gains, progress has been shared unequally, Bussel stated, with white children and children in higher income areas showing greater decreases in obesity rates. Today, more than 25 million children remain at risk for cardiovascular disease and type 2 diabetes. Unless this is addressed, Bussel predicted that this generation of children will be the first to live sicker and die younger than their parents.
The Robert Wood Johnson Foundation announced in February 2015 that it would be contributing an additional $500 million over the next decade to continue and expand its work in reducing childhood obesity. An important theme that will be driving the next decade of effort, not just in terms of building the evidence base, but also supporting action and advocacy, will be an intensified focus on reducing disparities. The foundation’s vision is for all children in the United States to be at a healthy weight no matter who they are or where they live. This is part of a broader effort focused on the healthy development of children—a critical element of the foundation’s mission to build a Culture of Health in the nation.
Bussel emphasized, as Hassink had and as many other participants would over the course of the workshop, the importance of starting early. According to Hassink, evidence indicates that children who enter school at a healthy weight are more likely to maintain a healthy weight through adolescence and adulthood. The first 1,000 days following conception is a crucial period for development and for the prevention of obesity and its consequences. Of the foundation’s five “big bets,” that is, five key strategies where it plans to concentrate its obesity prevention efforts, Bussel identified as the most relevant to this IOM workshop the effort to ensure that all children enter kindergarten at a healthy weight. She stressed the importance of working with parents even before their infants are born, linking families with the support and resources they need beyond the clinic walls to help their children achieve and maintain healthy weights, and the importance of gaining a better understanding of how to change what needs to be changed not just at the individual level but at the family and systems levels as well.
Much of the workshop discussion revolved around the emerging nature of the evidence for epigenetics as a key component of the “Early Origins of Obesity” model (featured in the workshop infographic) and whether experimental findings indicate causal versus correlational or confounding associations. Session 1, moderated by Matthew Gillman of the Harvard School of Public Health, set the conceptual stage for this discussion. Chapter 2 summarizes the Session 1 presentations and discussion.
Following a conceptual overview by Robert Waterland of the Baylor College of Medicine and Andrea Baccarelli of the Harvard School of Public Health, workshop participants in Session 2, moderated by Karen Lillycrop of Southampton University, considered how the risk of childhood obesity can be affected by (1) maternal and paternal nutrition and other exposures before conception, (2) maternal and placental nutrition and health during pregnancy, and (3) postnatal maternal and infant nutrition and health. Chapter 3 summarizes the Session 2 presentations and discussion.
Moderated by Leann Birch of the University of Georgia, Session 3 speakers discussed potential opportunities for intervention and prevention based on rapidly advancing knowledge of the role of epigenetics and other factors in the early origins of obesity. Chapter 4 summarizes the Session 3 presentations and discussion.
Given the dynamism, complexity, and context-dependency of childhood obesity etiology, it is difficult to translate research results, especially findings from animal experiments, into real-world application. Moderated by Debra Haire-Joshu of Washington University in St. Louis, the session explored in detail some of the challenges as well as opportunities for real-world application. Chapter 5 summarizes the Session 4 presentations and discussion.
To conclude the workshop, Esa Davis of the University of Pittsburgh Medical Center moderated a discussion with workshop speakers on possible future research directions, with an emphasis on expanding what scientists know about epigenetic-mediated associations between early developmental exposures and subsequent obesity-related health outcomes. Judith Hall of the University of British Columbia facilitated a second discussion open to all workshop participants that focused on opportunities and challenges in epigenetic research. Chapter 6 summarizes both discussions together. The chapter ends with a summary of the workshop’s major overarching themes as described by Shari Barkin.
This page intentionally left blank.








