A3
THE INTENSIFYING STORM: DOMESTICATION OF AEDES AEGYPTI, URBANIZATION OF ARBOVIRUSES, AND EMERGING INSECTICIDE RESISTANCE
Barry J. Beaty,1William C. Black IV,1Lars Eisen,1Adriana E. Flores,2Julián E. García-Rejón,3María Loroño-Pino,3and Karla Saavedra-Rodriguez1
Summary
It has been and continues to be the great misfortune of humankind to share time and space with the Aedes aegypti mosquito. Domestication of Ae. aegypti, urbanization of arboviruses, and globalization have created a super nidus for Ae. aegypti-transmitted diseases that spans the pantropical world. Ongoing pandemics of dengue and chikungunya are testimony to the threat posed by the super nidus. The burdens and threats of Ae. aegypti-transmitted diseases are too great to tolerate and are likely to worsen due to emerging insecticide resistance. The situation is grim; it is time to initiate a “war” on Ae. aegypti and to exploit new knowledge, tools, and approaches to control this enemy of humankind.
Introduction
As an epidemiological group, vector-borne diseases (VBDs), e.g., malaria, leishmaniasis, filariasis, onchocerciasis, trypanosomiasis, and dengue, continue to cause inestimable misery, morbidity, and mortality in humans. VBDs are major impediments to social and economic development in areas of the world that can least afford them. For the most part, there are no vaccines or therapeutics for these diseases. Thus, vector control is the principal tool to prevent and control these threats. The vectors have proven to be intractable to sustainable control, and emerging resistance to insecticides is of great concern. Notable recent successes in reducing the burden of some of these diseases, such as malaria, are now threatened by the emergence of resistance, most notably to pyrethroid insecticides, in their vectors (Strode et al., 2014; Hemingway et al., 2013). The situation is further complicated by the concomitant reduction in medical entomologists, vector biologists, and vector control personnel available to address VBD emergencies. This situation was addressed in a previous IOM publication (IOM, 2003a, 2008). Unfortunately little progress has been made in this area (IOM, 2008).
___________________
1 Colorado State University.
2 Universidad Autónoma de Nuevo León.
3 Universidad Autónoma de Yucatán.
Arthropod-borne viruses (arboviruses) continue to emerge and spread throughout the world. The introduction of West Nile virus into New York and its rapid spread throughout the Western Hemisphere is a textbook example of arbovirus epidemic potential (Petersen et al., 2013). Another great threat to humankind is the urbanization and spread of arboviruses, such as dengue virus (DENV) and chikungunya virus (CHIKV). Historically these viruses and yellow fever virus (YFV) emerged from sylvatic foci in Africa or Asia, were transported with the Aedes aegypti mosquito through much of the tropical world in sailing ships, and caused epidemics principally in port cities (Weaver, 2013; Weaver and Reisen, 2010; Nasci, 2014; Halstead, 2015). However, the ability of these viruses to emerge and to become established in tropical urban areas has increased dramatically in the past 50 years. The emergence and spread of epidemic dengue and dengue hemorrhagic fever/dengue shock syndrome (DHF/DSS) in the Americas in the 1980s and the emergence of CHIKV in the Caribbean in 2013 and its rapid spread throughout tropical America are examples of the extraordinary potential of urbanized, Ae. aegypti-transmitted arboviruses to traffic and to emerge as public health threats in pantropical urban areas.
Many reviews have addressed factors that have contributed to the emergence and resurgence and public health importance of VBDs. Clearly, lack of vaccines and therapeutics, erosion of public health infrastructure, poverty and social inequalities, population growth, unplanned urbanization, and globalization are major factors for emergence and continued importance of most VBDs. These have been discussed in detail elsewhere (Gratz, 1999; Gubler, 2005, 2011; IOM, 2003a; Weaver and Reisen, 2010). In a recent review, Gubler (2011) proposes three principal drivers that have conditioned the emergence and expansion of dengue and DHF/DSS as major threats to public health in the tropics: (1) urbanization, (2) globalization, and (3) lack of effective mosquito control. Human population growth and urbanization provide unprecedented availability of susceptible amplifying human hosts and an environment conducive to propagation of Ae. aegypti, which live in intimate association with humans. These factors in conjunction with nonsustainable mosquito control have conspired to create ideal conditions for transmission and maintenance of urbanized viruses. Globalization further fuels the flames of the epidemic potential of urbanized viruses, resulting in unprecedented trafficking of virus-infected humans and mosquitoes through the pantropical world. The combination of hyperabundant Ae. aegypti, cocirculation of multiple DENV serotypes, and increased evolution and trafficking of new virulent genotypes have led to dengue hyperendemicity and the global dengue pandemic (Gubler, 2011).
VBD specialists think in terms of the nidus of infection, in which a pathogen, susceptible hosts, and vectors intersect temporally and spatially in an environment conducive to pathogen transmission and maintenance (Weaver and Reisen, 2010). This concept has great utility, for example, in understanding the factors that maintain zoonotic virus endemic sylvatic cycles and the potential approaches
for preventing human infections. Pantropical urban areas now constitute a “super nidus” for Ae. aegypti transmitted pathogens. The large human and mosquito populations in tropical urban areas provide the ideal conditions or nidus for Ae. aegypti transmitted arbovirus maintenance, transmission, evolution, and trafficking. The large urban areas are interconnected by travel and commerce, which promote movement of viruses to new areas both within and between urban areas. Jet travel is especially efficient in moving people, viruses, and vectors around the world (Gubler, 2011).
The super nidus is certainly a key factor in the dramatic increase in importance of dengue and chikungunya in the Americas. Both DENV and CHIKV can be transmitted by other mosquito vectors, most notably Ae. albopictus, which can cause significant outbreaks of disease (Nasci, 2014). However, transmission of these viruses by Ae. aegypti in the tropical urban super nidus is an even greater threat for these diseases. Reduced efficacy of insecticides to control Ae. aegypti will likely exacerbate this situation.
In the following, we will principally focus upon entomological factors in the super nidus that conditioned emergence of epidemic dengue and DHF/DSS and chikungunya in Latin America. We will also address the implications of emerging insecticide resistance in Ae. aegypti for continued emergence, resurgence, and control of arboviruses in the super nidus. This is not a review of the literature. Rather the emphasis will be on selected examples from our long term studies of the epidemic potential of dengue and the control of Ae. aegypti in Mexico to illustrate the problems and complexity of controlling Ae. aegypti and urbanized arbovirus diseases. However, the lessons learned can certainly be extrapolated to most urban areas in the tropical world.
The Origins of the Super Nidus: Domestication of Aedes aegypti and Urbanization of Arboviruses
Domestication of Aedes aegypti
One of the great misfortunes of humankind has been the domestication and subsequent urbanization of Ae. aegypti. The ancestral form of Ae. aegypti is found in Africa; it is a sylvatic mosquito that feeds on nonhuman primates and other forest mammals and oviposits in tree holes and other natural water-containing sites (Powell and Tabachnick, 2013, Tabachnick, 2013). The mosquito is dark in color and is designated as a subspecies—Ae. aegypti formosus. A lighter colored subspecies, Ae. aegypti aegypti (hereafter called Ae. aegypti for simplicity) has adapted to feed on humans and to live and breed in and around human habitation. Domestication has likely occurred multiple times (Brown et al., 2013; Moore et al., 2013, Powell and Tabachnick, 2013). Ae. aegypti has expanded from Africa and colonized most of the pantropical world with disastrous public health consequences. Historically, the introduction of Ae. aegypti and of YFV, DENV,
and CHIKV into the New World resulted in large epidemics of the respective diseases typically in port cities (Powell and Tabachnick, 2013; Weaver, 2014). Similarly, introduction of Ae. aegypti into Asia in the mid-20th century led to spillover of DENV from its sylvatic cycles resulting in large urban epidemics of dengue (Smith, 1956).
Behavioral and genetic changes associated with domestication of Ae. aegypti dramatically impacted the vectorial capacity of the species for arbovirus transmission (Table A3-1). Ae. aegypti is anthropophilic (feeds on humans) and endophilic (lives in homes). Thus the mosquito is intimately associated with humans, thereby dramatically increasing its potential to transmit pathogens to humans. Ae. aegypti formosus is zoophilic (feeds on nonhuman hosts), exophilic (lives outdoors), and sylvatic, thereby limiting its potential to transmit pathogens to humans. However, domestication of Ae. aegypti formosus may still be occurring in West Africa, where the subspecies enters huts to feed on humans. Both Ae. aegypti and Ae. aegypti formosus occur in Senegal with the former occurring mostly in coastal urban environments (Sylla et al., 2009; Moore et al., 2009; Dickson, et al., 2014). Ae. aegypti in coastal West Africa may have resulted from a reintroduction of Ae. aegypti or may be the harbinger of a new domestication of Ae. aegypti formosus (Brown et al., 2013). Either scenario poses increased threats of arbovirus urbanization in West Africa.
Domestication of Ae. aegypti has also resulted in changes in vector competence for YFV and DENV. Vector competence is a component of the vectorial
| Anthropophily: | Feeds preferentially on humans, and when humans are available, zoophily is minimal. In addition, sugar feeding is very limited. |
|---|---|
| Endophily: | Prefers to live and feed indoors in homes and other structures. Extraordinarily close association with humans. Will oviposit in homes/structures if larval development sites are available. Readily lays eggs in cans, tires, refuse, and other manmade larval development sites surrounding homes. Endophily protects mosquitoes from outdoor insecticide-based space spraying in settings where the typical housing type is “closed” and thus prevents ingress of the spray. |
| Interrupted feeding: | May only take a partial blood meal before being disturbed and may complete feeding on other hosts. Evolved defensive behavior of the mosquito may promote mechanical and biological transmission of arboviruses. |
| Multiple feeding: | May feed multiple times during a gonadotrophic cycle, greatly promoting the potential for arbovirus transmission. |
SOURCES: Selected papers documenting the behavioral and biological factors that contribute to the vectorial capacity of Ae. aegypti: Edman et al., 1992; Harrington et al., 2001, 2014; Gubler, 2011; Garcia-Rejon et al., 2008, 2011; Hemingway et al., 2006; Reiter et al., 2003; Reiter and Gubler, 1997; Scott et al., 1993, 2000; Scott and Takken, 2012.
capacity of a mosquito population; it is more narrowly defined as the permissiveness of a mosquito for infection, replication, and subsequent transmission of a pathogen (Black et al., 2002). Genetic and environmental determinants of Flavivirus infection and transmission by Ae. aegypti and of the distribution of vector competence in natural populations have been reviewed elsewhere (Black et al., 2002; Tabachnick, 2013). Early studies revealed that Ae. aegypti formosus is less able to become infected with and to transmit YFV than Ae. aegypti (Beaty and Aitken, 1979; Tabachnick et al., 1985). Subsequent studies also revealed that Ae. aegypti formosus is also a less effective vector for DENV; however, this is dependent upon the origin of the DENV isolates used to challenge the mosquitoes (e.g., Sylla et al., 2009; Dickson et al., 2014). There also is considerable genetic variability among Ae. aegypti populations in their vector competence for DENV (Bennett et al., 2002). This will be addressed in detail below in the context of Ae. aegypti vector competence for DENV in Mexico.
Urbanization of Arboviruses
Arboviruses have the potential to spill out of their enzootic or sylvatic transmission cycles into cycles in which humans become the vertebrate amplifying hosts for the virus; such spillovers can have devastating public health consequences (Weaver and Reisen, 2010; Weaver, 2013). Spillover can take many forms; for example, humans may simply encroach upon new environments and become more frequently exposed to enzootic vectors that are willing to take human blood. Urbanization of sylvatic arboviruses is by far the greatest threat. In such scenarios, humans may become the dominant vertebrate host, eliminating the need for amplifying sylvatic hosts. YFV, DENV, and CHIKV have emerged from their respective zoonotic sylvatic cycles involving forest mosquitoes and nonhuman primates into transmission cycles involving Ae. aegypti and humans, resulting in global pandemics. The super nidus with its hyperabundant Ae. aegypti and burgeoning susceptible human populations provides unprecedented receptivity to arbovirus spillover into the urban cycle and also promotes opportunities for spillback into sylvatic cycles. Spillback is epidemiologically significant; establishment of the virus in a sylvatic cycle in a newly invaded region effectively limits opportunities for virus eradication in that region.
Yellow fever virus YFV is the archetypical virus in terms of spillover and urbanization. YFV originated in Africa and was maintained in cycles involving principally canopy dwelling mosquitoes and nonhuman primates (Mutebi and Barrett, 2002; Beck et al., 2013). Spillover of YFV into the urban transmission cycle involving humans and Ae. aegypti resulted in yellow fever epidemics that decimated cities, especially port cities, in the Americas and Africa (Weaver and Reisen, 2010). YFV is also the archetypical example of arbovirus spillback into a sylvatic cycle involving nonhuman primates and forest mosquitoes. YFV spilled
back into sylvatic cycles in the Americas, posing an ongoing threat of urbanization of YFV from these sylvatic foci. The lack of reurbanization of YFV from sylvatic cycles in South America is a mystery (Barrett and Higgs, 2007). Another great mystery has been the lack of emergence of YFV in Asia, which would be a public health catastrophe. Early studies revealed that the vector competence of Asian Ae. aegypti mosquitoes was lower than that of Caribbean populations of the vector, which may in part condition the lack of emergence in Asia (Tabachnick et al., 1985). However, dengue hyperendemicity first in Asia and now in the New World could provide cross-protective herd immunity in humans and ironically thereby restrict urbanization of YFV in both regions.
Dengue virus DENV originated in Southeast Asia where the four DENV serotypes (DENV1–4) diverged and are maintained in cycles involving canopy-dwelling mosquitoes and nonhuman primates (Hanley et al., 2013; Messina et al., 2014). Spillover of all four DENV serotypes from sylvatic cycles has occurred and continues to occur into human cycles with anthropophilic vectors, including Ae. aegypti and Ae. albopictus. There is apparently no need for adaptation for human transmission and virulence (Vasilakis et al., 2007, 2010; Weaver, 2013). Clearly the expanding super nidus is even more receptive to emergence and urbanization of DENV from sylvatic cycles. Spillback of DENV-2 into a sylvatic nonhuman primate and forest mosquito cycle has occurred in West Africa (Weaver and Reisen, 2010; Weaver, 2013), but thus far, spillback of DENV into sylvatic cycles in Latin America has not been detected. Epidemic dengue and DHF/DSS emerged in Southeast Asia following the introduction of Ae. aegypti and the rapid urbanization following World War II, highlighting the importance of the urban transmission cycle for dengue hyperendemicity (Smith, 1956; Gubler, 2011).
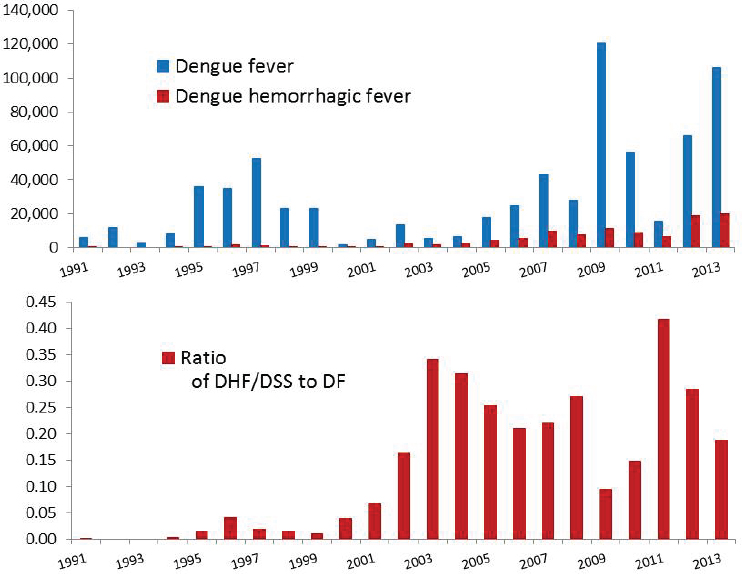
Dengue is the most important arthropod-borne viral disease of humans with more than 3 billion people living in dengue endemic areas (Guzman and Harris, 2014). Worldwide, more than 390 million infections, 100 million DF cases, and 500,000 cases of the more severe DHF occur each year (Bhatt et al., 2013). Infection with one of the four antigenically related DENV serotypes confers long-term protection to that serotype but no or very short-lived cross-protection to the other serotypes. Dengue disease severity ranges from asymptomatic to fatal (Srikiatkhachorn et al., 2011). Most symptomatic cases are classified as dengue fever (DF), an acute and self-limited condition characterized by fever, generalized pains, rash, lymphadenopathy, and minor hemorrhages. Even the asymptomatic infections are likely to be epidemiologically significant; these silent infections can prime patients for the more serious forms of the disease. People who experience secondary infections with a heterologous serotype of the virus are primed for the more serious forms of the disease—DHF/DSS, which is characterized by hemostatic disorders, hepatic involvement, and plasma leakage resulting from increased vascular permeability. DSS is potentially fatal. In Mexico and much of Latin America, almost 30 percent of patients are now progressing to severe
dengue disease (see Figure A3-1). The surge of patients experiencing severe dengue disease has overwhelmed the public health infrastructure in many cities and countries. The emergence of epidemic dengue and DHF/DSS in the pantropical world has been a public health disaster.
Chikungunya virus CHIKV has emerged from its sylvatic nidus in Africa multiple times, causing major epidemics in Africa, Asia, and Latin America. Historically, CHIKV emerged from its enzootic cycle involving forest mosquitoes and nonhuman primates and was transported around the world in sailing ships with Ae. aegypti, as with YFV and DENV (Weaver and Reisen, 2010; Carey, 1971). In the 1950s, epidemics of chikungunya were reported in India and Southeast Asia, but the virus disappeared in India. The situation changed dramatically in recent years. Major epidemics caused by different strains of CHIKV have occurred with millions of cases reported (Nasci, 2014, Weaver, 2014). The recent reintroduction of CHIKV into the Caribbean (Halstead, 2015) and its explosive spread throughout Latin America are the latest manifestations of urbanization and spread of the virus. Considering the size of the super nidus in Latin America, it is likely that CHIKV will become endemic in the tropical Americas as it has in Asia following its emergence there (Nasci, 2014; Weaver, 2014). It will be of great interest to see if CHIKV spills back into a sylvatic cycle in the Americas. In Southeast Asia, the virus has been endemic for many years in the Ae. aegypti–human cycle, yet there is no evidence of spillback into a sylvatic cycle.
Virus infection can cause a febrile disease with severe, debilitating arthritis, which can last for several weeks, and in some cases may become chronic. Historically, chikungunya was thought to be a serious but self-limiting disease. However, mortality rates in the range of 1 per 1,000 cases have been reported in epidemics in India and Asia, likely due to comorbidities (Renault et al., 2008; Tandale et al., 2009). CHIKV exerts a major socioeconomic burden during epidemics (Soumahoro et al., 2011). Following CHIKV introduction into India in 2006, millions of cases occurred and transmission continues (Mohan et al., 2010). The explosive nature of the epidemic of chikungunya in the New World has been amazing. More than 850,000 cases have been reported since its introduction early in 2013 (http://www.cdc.gov/chikungunya), and it is likely that all of pantropical America will soon experience the burden of chikungunya.
Other arbovirus threats Arboviruses maintained in cycles involving nonhuman primates clearly pose a great threat for urbanization. Mayaro virus, a relative of CHIKV, is maintained in a forest mosquito–nonhuman primate cycle in the Americas and Zika virus, which is presumably maintained in enzootic cycles involving nonhuman primates and forest mosquitoes in Africa and Asia recently caused an outbreak of febrile disease in a Pacific Island in which humans were the amplifying hosts, are clear candidates for urbanization (Weaver and Reisen, 2010; Weaver, 2013). Other non-Ae. aegypti vectored viruses have the potential
to adapt to Ae. aegypti and to become urbanized. The dramatic expansion of the super nidus and its increasing encroachment on sylvatic cycles of other important arboviruses provides unprecedented opportunities for arbovirus urbanization. For example, Ae. aegypti has been demonstrated to be a competent vector of Venezuelan equine encephalitis virus (VEEV) in laboratory studies, and humans develop a significant viremia (Weaver and Reisen, 2010). The expanding super nidus in South America ensures that VEEV and Ae. aegypti will overlap; urbanization of VEEV would be a public health catastrophe.
Comment We are entering new territory in terms of urbanization of arboviruses; the pantropical super nidus and globalization combine to provide (1) unprecedented opportunities for spillover of arboviruses from sylvatic cycles, (2) extraordinary potential for viruses and vectors to be transported rapidly throughout the world, and (3) major threats of ongoing epidemics in the super nidus, with enormous public health and economic consequences. Some of the causes, threats, and needs for control of the super nidus will be addressed in the context of our studies of epidemic dengue and DHF/DSS and control of Ae. aegypti in Mexico.
Epidemic Dengue and Dengue Hemorrhagic Fever in Mexico
Epidemic DHF/DSS emerged in the Americas in the 1980s (Gubler, 2005; Guzman and Harris, 2014). Since then dengue has emerged as a major public health problem and is considered to be hyperendemic in most of Latin America. In Mexico, Ae. aegypti has resurged and is hyperabundant in most urban areas in the country (with the exception of those at higher altitudes in the central plateau), all four serotypes of DENV now cocirculate in endemic areas, more virulent viruses and genotypes have been introduced, and the number of secondary infections and severe disease manifestations are greatly increased (see Figure A3-1). Indeed, dengue epidemics are annual occurrences and can overwhelm public health capacity in the affected areas. As mentioned above, CHIKV has exploded throughout much of Latin America since its introduction in 2013. The Ministry of Health of Mexico reported the first autochthonous (locally contracted) case of chikungunya on November 7, 2014, in Chiapas State (http://www.cenaprece.salud.gob.mx/programas/interior/emergencias/descargas/pdf/Declaratoria_Emergencia_Chiapas_Chikungunya.pdf). There is great concern about a major epidemic of chikungunya in Mexico in 2015. In the following, we will focus upon selected factors that have resulted in Ae. aegypti hyperabundance in Mexico and contributed to the dengue hyperendemicity.
Collaborative Studies of Dengue Hyperendemicity and Control in Mexico
In the early 1990s, before the emergence of DHF/DSS, we embarked upon collaborative studies with Mexican scientists that focused upon viral, vector,
Top: Laboratory-confirmed dengue fever cases in Mexico.
Bottom: Ratio of DHF/DSS (dengue hemorrhagic fever/dengue shock syndrome) to DF (dengue fever) laboratory-confirmed cases.
SOURCE: Beaty et al. Data from Mexican Ministry of Health (http://www.epidemiologia.salud.gob.mx/dgae/infoepid/inicio_anuarios.html).
and epidemiological determinants of the lack of DHF/DSS in Mexico, which was occurring elsewhere in the Americas. The collaborations then evolved to develop innovative approaches to control Ae. aegypti and DENV transmission to humans in homes. Mérida City in Yucatán State became a major field site and focus of many of our studies. Some of the studies and lessons learned will be briefly reviewed here.
Viral Determinants of Dengue Hyperendemicity
Molecular epidemiological studies were conducted to investigate the viral determinants of the emergence of epidemic dengue and DHF/DSS as major public health problems in Mexico (Diaz et al., 2006). Phylogenetic analyses were
conducted to determine the origin, persistence, and geographical dispersion of the four serotypes of DENV isolated in Mexico between 1980 and 2002. Changes in the incidence and severity of dengue were temporally associated with the introduction and circulation of different serotypes and more virulent genotypes of DENV into the Yucatán State of Mexico in 2002 (Loroño-Pino et al., 2004). This was associated with increased incidence of DHF/DSS. Nucleotide sequencing and phylogenetic analyses identified isolates from patients with severe disease as DENV-2 viruses of the American-Asian genotype, which was the first report of this genotype in Yucatán State. Ominously, 31 percent of the patients met the World Health Organization criteria for DHF. The majority (77 percent) of the patients experienced secondary infections in this epidemic. The new virus genotypes supplanted the DENV-2 American genotype viruses in Mexico. The reasons for these genetic sweeps remain to be determined, and this is an important area of research. The introduction of new virulent virus genotypes resulted in a dramatic increase in severe dengue cases (see Figure A3-1). Increased surveillance for such introductions is critical to allow public health authorities to intervene in impending epidemics.
Vector Determinants of Dengue Hyperendemicity
Many entomological factors have contributed to dengue hyperendemicity and Ae. aegypti resurgence in Mexico:
- The collapse of sustainable vector control
- Introduction and trafficking of vectors
- Presence and distribution of highly competent vectors
- The extreme endophily of the vector in concrete housing and buildings typical of Latin America
- The emergence of the throw-away society providing inexhaustible breeding sites for the vector
- Behavioral changes permitting Ae. aegypti to more effectively exploit the urban environment
Selected investigations and lessons learned concerning these entomological factors follow to illustrate the threats and complexities of controlling Ae. aegypti-transmitted arboviral diseases in the super nidus.
Ineffective or nonsustainable Ae. aegypti control Ae. aegypti control in the super nidus is a major challenge to public health. It is noteworthy that in the mid-20th century, countries in the Western Hemisphere including Mexico as well as the Pan American Health Organization (PAHO) waged an effective campaign to control Ae. aegypti. In the early 1900s, Ae. aegypti and dengue were widely distributed, but a successful hemispheric campaign against yellow fever led by
Fred Soper of PAHO that was initiated in 1947 and continued to the early 1970s resulted in Ae. aegypti and DENV being eliminated from most of Central and South America. This campaign was based on spraying of larval development sites and indoor environments with DDT. The campaign was quite effective, and Ae. aegypti presence and/or abundance was dramatically reduced in the Americas (Gubler, 2005; Gratz, 1999). Ironically, the success of the program led to its demise. The resources devoted to vector control, which is quite expensive, were diverted to other programs. This resulted in an astounding reemergence of Ae. aegypti and also of DENV with dengue outbreaks and the emergence of DHF/DSS across the Americas in the following decades (Gubler, 2005, 2011). The measures used in Soper’s campaign would likely not be as effective today (IOM, 2008). The urban super nidus vastly increases the difficulty of controlling Ae. aegypti, and current vector control programs have not stemmed the rising tide of the dengue pandemic.
These control programs typically include activities to control both immature and adult stages of Ae. aegypti. Chemical or biological larviciding and physical source reduction are widely used to control immatures and to try to maintain mosquito populations below threshold levels thought to interrupt DENV transmission (Gubler, 2005; Eisen et al., 2009). This overall strategy has not proven to be sustainable. Programs were often poorly funded or did not receive long-term support by government agencies. Indeed, source reduction may no longer be a practically sustainable control strategy because of the emergence of the “throw-away society” where breeding sites for Ae. aegypti accumulate rapidly and are almost ubiquitous. Chemical or biological larviciding can be effective, but it is tremendously labor intensive and costly, and locating breeding sites can be difficult as described below. The extreme endophily of female Ae. aegypti complicates efforts to control adults (Edman et al., 1992; Harrington et al., 2001; Bonds 2012). Outdoor spraying of insecticides during dengue outbreaks is likely to be ineffective in most situations because of poor penetration of the insecticide into cement housing, but it is still routinely used in many control programs (Gubler, 2005). Ironically, exposure of mosquitoes to sublethal doses of insecticides during outdoor spraying may increase evolution of resistance. Indoor space spraying can be an effective dengue outbreak intervention strategy; recent studies in Iquito, Peru, have demonstrated that indoor space spraying three times reduces Ae. aegypti populations and the number of dengue cases (T. Scott, personal communication). Although effective, the approach is laborious, expensive, and needs to be targeted to be cost-effective.
Thus despite the tremendous efforts and resources expended by public health organizations in dengue-endemic countries, it has proven difficult to achieve sustainable control of Ae. aegypti and to prevent or disrupt dengue outbreaks (Eisen et al., 2009). Ae. aegypti control in the super nidus is very difficult and complex as illustrated by some of the following examples.
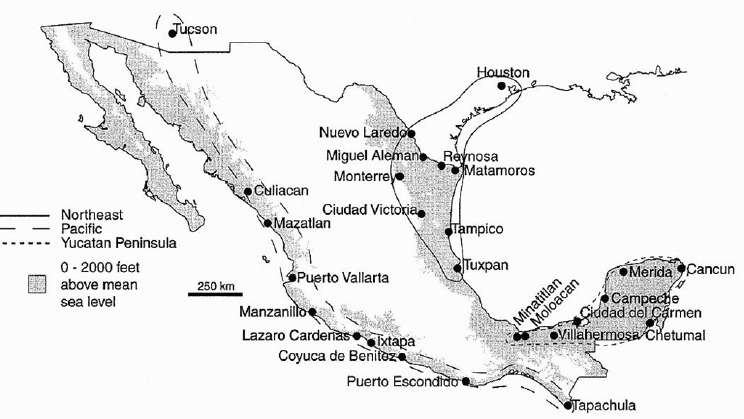
Trafficking of vectors Globalization is predicated upon commerce and rapid and efficient transport of goods and people, and it provides great threats of trafficking of Ae. aegypti between and within super nidus regions (IOM, 2003a; Gubler, 2011). The mosquito can be transported as eggs in tires and other containers into new regions and hatch upon exposure to water, or as adults in vehicles and even in airplanes (Lounibus, 2002; Whelan et al., 2012). Such trafficking potential threatens sustainability of vector control. Understanding the breeding structure and trafficking potential of Ae. aegypti in a country is critical for developing effective vector surveillance, monitoring, and control strategies and for understanding spatial heterogeneities in DENV transmission. To characterize the breeding structure of Ae. aegypti in Mexico, we conducted a population genetic analysis of 38 collections of Ae. aegypti from throughout coastal regions of Mexico (Gorrochotegui-Escalante et al., 2002). Single-strand conformation polymorphism analysis was used to screen for variation in a 387-bp region of the ND4 mitochondrial gene, and 25 haplotypes were detected. Northeastern Mexico collections were genetically differentiated from and had lower genetic diversity than southern Yucatán and western coastal Pacific collections (see Figure A3-2). Yucatán and Pacific collections were genetically homogeneous. Regression analysis of geographic and genetic distances indicated that collections were genetically isolated by distance in the Pacific and the Yucatán, but not among collections in the northeast. Free gene flow occurred among all collections within 130 km of one another in the northeast and within 180 km in the Yucatán. F(ST) values were never large among Pacific collections, suggesting extensive gene flow along the Pacific coast (see Figure A3-2).
SOURCE: Gorrochotegui-Escalante et al., 2002, with permission from American Journal of Tropical Medicine and Hygiene.
The extraordinary gene flow in Ae. aegypti populations along the western coast of Mexico was presumed to be attributable to trafficking of Ae. aegypti in the extensive commerce and tourism trade routes located there. The reasons for limited gene flow between Ae. aegypti populations in the northeast of Mexico and the Yucatán in the south were investigated (Lozano-Fuentes et al., 2009). Targeted population genetic studies revealed that the intersection of the neovolcanic axis (NVA) with the Gulf of Mexico coast in the state of Veracruz acts as a discrete barrier to gene flow among Ae. aegypti populations north and south of the NVA, presumably because of the lack of commerce and tourism routes through this region of eastern Mexico. The breeding structure of Ae. aegypti in Mexico is complex and dynamic. Vector (and vector gene) trafficking into previously controlled areas will clearly complicate sustainable control efforts by public health officials.
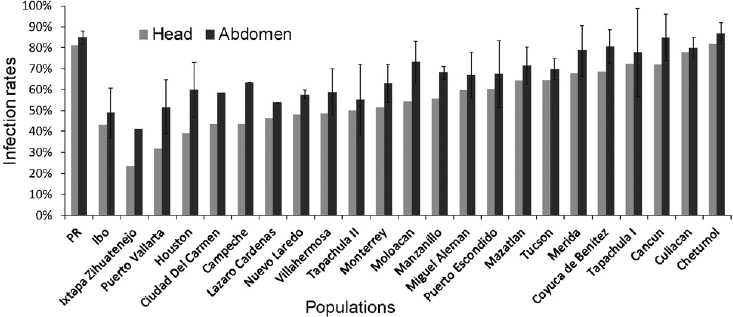
Competence of vectors for virus transmission Vector competence is a critical component of the transmission potential of an arbovirus and may contribute significantly to the heterogeneities seen in DENV transmission. Concomitantly with our breeding structure studies, we characterized heterogeneities in vector competence of Mexican populations of Ae. aegypti from 24 collections in Mexico and the United States (see Figure A3-3). Mosquitoes were challenged orally with DENV-2 JAM1409, an American-Asian genotype isolated during one of the initial epidemics of DHF/DSS in the New World (Bennett et al., 2002). The presence or absence of a midgut infection barrier (MIB) and a midgut escape barrier (MEB) was determined for mosquitoes in each population. The percentage of mosquitoes exhibiting a MIB ranged from 14 percent to 59 percent, and those exhibiting a MEB ranged from 4 percent to 43 percent in the collections. Midgut infection rates were dose dependent. Thus new, more virulent genotypes
SOURCE: Adapted from Bennett et al., 2002.
of DENV that cause higher titered viremias in humans would likely be more infectious for mosquitoes. The vector competence rate of examined collections (i.e., the number of mosquitoes with disseminated DENV infections/number of mosquitoes orally challenged with the virus) ranged from 24 percent to 83 percent (see Figure A3-3). Considerable genetic variability in vector competence for DENV occurs in Ae. aegypti collections in Mexico, with mosquitoes from the Yucatán Peninsula being highly competent vectors (Bennett et al., 2002).
The mosquito populations north and south of the NVA also differed in their vector competence for DENV-2 (Lozano-Fuentes et al., 2009). The average vector competence rate for Ae. aegypti from populations north of the NVA was 55 percent, as compared with 20 percent south of the NVA. Most of this variation was attributable to midgut infection and escape barriers. In Ae. aegypti north of the NVA, 22 percent failed to develop midgut infections and 30 percent of those with an infected midgut failed to develop a disseminated infection. In contrast, 45 percent of the mosquitoes from south of the NVA failed to develop a midgut infection, and 63 percent of those with an infected midgut failed to develop a disseminated infection.
Mexican vector and virus interactions We also conducted studies to understand heterogeneities in dengue prevalence and mechanisms conditioning the genetic sweeps of virus genotypes. For example, Ae. aegypti from Chetumal were orally challenged with American and American-Asian genotype viruses isolated from severe dengue patients in Mérida City (Loroño-Pino et al., 2004). The American-Asian DENV-2 isolates were much more fit in their ability to be transmitted by Ae. aegypti (see Figure A3-4). The American genotype virus was dramatically less efficient than the American-Asian genotypes in escaping the midgut to infect
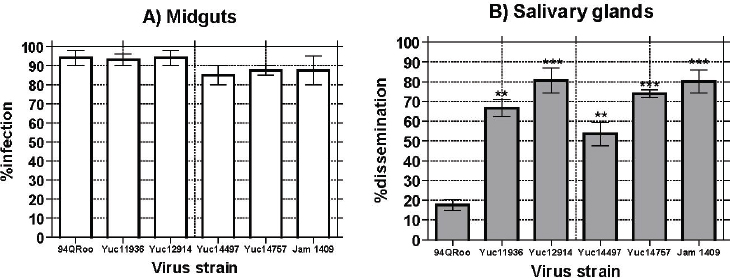
NOTES: American-Asian genotype viruses—Yuc11936, Yuc12914, Yuc14497, Yuc14757; American genotype virus—94QRoo.
SOURCE: Salazar et al., 2010. Reproduced with permission from Revista Biomédica.
salivary glands. The American-Asian genotype DENVs could be detected in salivary glands of the Chetumal strain of Ae. aegypti as early as 4 days post oral challenge (Salazar-Sanchez et al., 2007), a dramatically shorter incubation than previously reported. The infection and replication efficiency of the American-Asian viruses was attributable in part to mutations in the 3′UTR of the virus (see Figure A3-5; Salazar et al., 2010). The 3′UTR contains motifs that are critical for translation and RNA synthesis, and secondary structure of the 3′UTR is a determinant of virus replication efficiency. Efficient and rapid productive infection of vectors could be major determinants of the genetic sweep of the newly introduced genotypes. Identification of genetic markers for vector competence in mosquitoes could be exploited as a marker for risk assessment and surveillance programs for improved and targeted vector control.
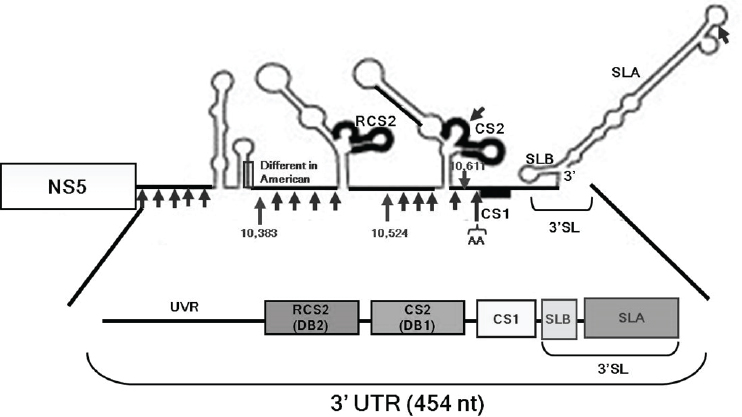
NOTES: The secondary structure of the 3′UTR and the different functional domains are shown. Arrows point out the sites where the principal secondary structures were affected by mutations. The 3′UTR consists of: (1) a variable region (VR) adjacent to the stop codon of the viral polyprotein that encloses 2 hairpin structures (HP), (2) a core region containing two predicted secondary structures, the DB1 (containing CS2) and DB2 (containing RCS2), and (3) a 3′-terminal region enclosing the CS1 and the 3′SL (formed by SLA and SLB).
SOURCE: Salazar et al., 2010. Reproduced with permission from Revista Biomédica.
Extreme Endophily and Hyperabundance of Ae. aegypti in Human Structures
The domicile Domestication of Ae. aegypti and its consequent close association with humans in their domiciles is one of the major factors promoting efficient DENV transmission. A classic epidemiological investigation of dengue in Laredo, Texas, and Nuevo Laredo, Mexico, revealed the seminal importance of protecting the home to reduce DENV transmission (Reiter et al., 2003). In the U.S. city there were minimal DENV infections, but Ae. aegypti breeding sites were common. In contrast, in the Mexican City, there were many DENV infections, but mosquito breeding site control was more effective than in the U.S. city. The reduction in dengue in the United States was attributed to screens and air conditioning protecting the home from Ae. aegypti and thus dramatically reducing indoor DENV transmission.
Following publication of these results and in anticipation of initiating mosquito control efforts, we investigated the abundance of Ae. aegypti and presence of DENV in females collected from within and around homes of laboratory-confirmed dengue patients over a 12-month period in Mérida City (Garcia-Rejón et al., 2008). Backpack aspiration from 880 homes produced 1,836 females indoors (predominantly from bedrooms) and 102 females (< 5 percent of the females collected) from patios or backyards. The mean weekly indoor catch rate per home peaked at 8 females in late August. In some homes, up to 40 Ae. aegypti females were collected in one visit (see Table A3-2). Clearly these highly infested homes are threats to both the occupants and visitors for transmission of DENV and CHIKV. Other important outcomes of these investigations are provided in Table A3-3. DENV-infected Ae. aegypti females were recovered from 34 homes, and up to 7 DENV-infected females were collected in a home. DENV-infected females were collected from homes of dengue patients up to 27 days after the onset of symptoms. The epidemiological significance of the long-term persistence of DENV-infected mosquitoes in homes cannot be overstated. Ae. aegypti feeds multiple times during a gonadotrophic cycle, is an efficient interrupted feeder, and can survive for more than a month in the protective confines of the home. Obviously, all the members of the household, visitors, and nearby neighbors are
TABLE A3-2 Ae. aegypti and Culex quinquefasciatus Females in Dengue Patient Homes
| Species | No. Collected | % of Total | Range for Individual Homes | No. (%) of Homes with Females |
|---|---|---|---|---|
| Aedes aegypti | 1,836 | 41 | 0–40 | 332 (37.7) |
| Culex quinquefasciatus | 2,641 | 59 | 0–59 | 312 (35.5) |
| Total | 4,477 | 100 |
SOURCE: Adapted from Garcia-Rejon et al., 2008.
TABLE A3-3 The Critical Epidemiological Need to Control Aedes aegypti in the Indoor Environment
|
SOURCE: Adapted from Garcia-Rejon et al., 2008.
at great risk for being infected by these mosquitoes. Clearly the domicile is one of the most epidemiologically significant points of contact between infected vectors and humans, making it a key target for interventions. For example, indoor insecticide application in homes of suspected dengue patients could be used to reduce intradomiciliary transmission of DENV and prevent their homes from becoming sources for dispersal of DENV by persons visiting and being bitten by infected mosquitoes.
The school Following the results of the domicile studies, we decided to determine the abundance of Ae. aegypti mosquitoes and presence of DENV in females collected from schools in Mérida City (Garcia-Rejón et al., 2011b). Backpack aspiration from 24 schools produced 468 Ae. aegypti females and 1,676 Culex quinquefasciatus females, another human biter (see Figure A3-6). Ae. aegypti females were collected most commonly from classrooms, followed by offices and bathrooms. DENV RNA was detected in 19 of 118 pools (16 percent) of Ae. aegypti females (total of 415 females). The overall rate of DENV infection per 100 Ae. aegypti females was 4.8. DENV-infected pools were detected from 11 of 24 schools (46 percent) and came from different room types, including classrooms, offices, and bathrooms (see Figure A3-6). Clearly, schools in Mérida City and elsewhere in tropical areas are a risky environment for students, teachers, and other personnel to be exposed to DENV-infected Ae. aegypti females. Children infected at school could in turn introduce the virus into their respective domiciles, infect Ae. aegypti, and initiate an intradomiciliary cycle. Schools are clearly an important target for dengue vector control.
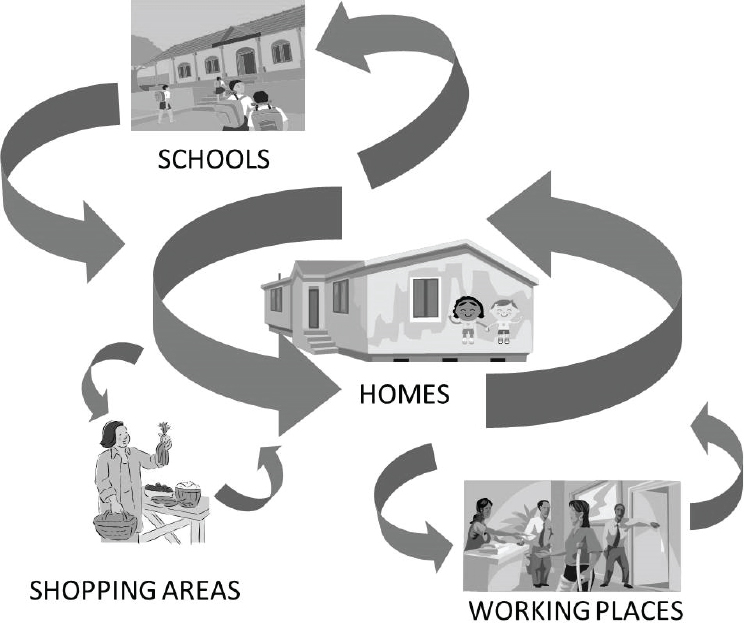
Based upon the results of the home and school studies, we have proposed a new model of dengue epidemiology in urban areas in Mexico (see Figure A3-7). There is an intradomiciliary cycle in which DENV is transmitted in the home or peridomestic environment. There is also an extradomiciliary cycle in which
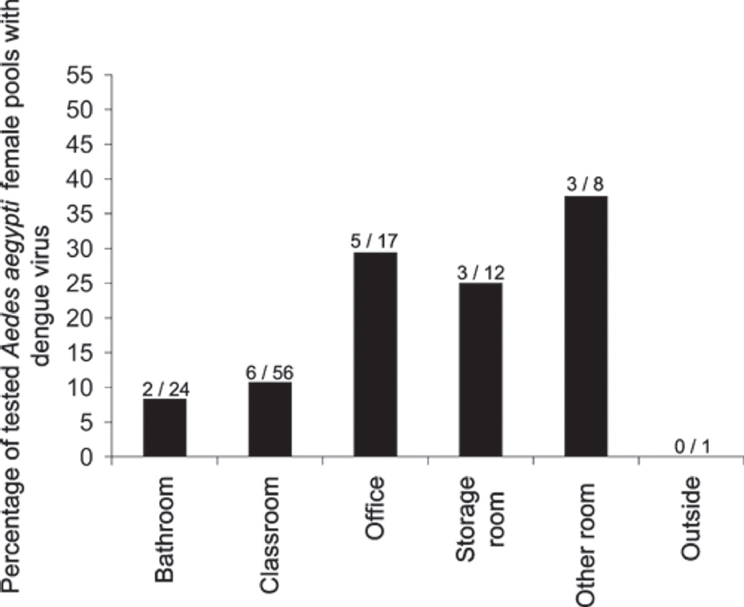
NOTE: Numbers above the bars indicate numbers of positive pools per total tested pools.
SOURCE: Garcia-Rejon et al., 2011b. Reproduced with permission from American Journal of Tropical Medicine and Hygiene.
individuals can be infected outside of the home, such as in schools and work places. DENV can be introduced into and amplified in either cycle by transmission to susceptible individuals and noninfected mosquitoes. Effective dengue control will require reducing Ae. aegypti abundance in both cycles.
Because the home is such a critical environment for transmission of DENV between humans and mosquito vectors and because community-wide distribution of insecticide-treated curtains (ITCs) showed promise in reducing DENV infections (Kroeger, et al., 2006), we conducted a Casa Segura study in Mérida City to determine the potential to reduce intradomiciliary DENV transmission through ITC use in individual homes (Loroño-Pino et al., 2013). Windows of homes were covered with ITCs, and humans and mosquitoes were monitored for DENV infections. DENV infections in mosquitoes and in humans were reduced in homes with ITCs in one of two study subareas. Overall, ITCs reduced intradomiciliary DENV transmission. DENV-infected Ae. aegypti females were
reduced within the ITC homes where the treated curtains were used most appropriately (see Table A3-4). Indeed, no infected Ae. aegypti were detected in homes where the curtains were used most appropriately (4th quartile use index). In these homes, curtains were present, covered the window, and not tied up or covered by a privacy curtain (nontreated), all of which would aid mosquitos in entering or exiting homes (Loroño-Pino et al., 2013). ITC homes were also significantly less likely to experience multiple DENV infections in humans than control homes (see Table A3-5), indicating interruption of the intradomiciliary transmission cycle. Some homes yielded up to nine infected Ae. aegypti females, emphasizing again the potential importance of highly infested homes in DENV transmission (Loroño-Pino et al., 2013). The studies were promising and revealing of best practices for protecting the homes from intradomiciliary transmission. However,
| Curtain Use Index (CUI) | Mean DENV-Infected Ae. aegypti Females per Home | |
|---|---|---|
| CUI quartile | Nontreated Curtain Homes | Insecticide-Treated Curtain Homes |
| 1st to 3rd quartile (limited, low and medium use) | 0.13 (34)* | 0.09 (16) |
| 4th quartile (high use) | 0.23 (16) | 0.00 (0) |
*Total number of infected females collected.
SOURCE: Adapted from Loroño-Pino et al., 2013.
| Dengue Infections in Humans by Individual Home | Percentage of Homes with Single or Multiple DENV Infections in Humans | |
|---|---|---|
| Nontreated Curtain Homes | Insecticide-Treated Curtain Homes | |
| Single infection | 51 (40/78) | 49 (38/78) |
| Multiple infection | 71 (24/34) | 29 (10/34) |
SOURCE: Adapted from Loroño-Pino et al., 2013.
the potential for ITCs for DENV vector control remain to be determined, especially in the face of emerging insecticide resistance, which could reduce ITC efficacy (see Table A3-4).
Breeding site identification and source reduction The emergence of our throw-away society and rapid urbanization have greatly complicated vector control, especially in urban areas. Larval development sites, such as tires, cans, bottles, and other water-holding containers are now ubiquitous breeding sites for Ae. aegypti. In addition, the urban environment provides multiple other breeding sites that Ae. aegypti may exploit and that sometimes are difficult to locate and to control. Detection of Ae. aegypti breeding in sewer systems (Barrera et al., 2008) and demonstration that the mosquitoes obtained from these cryptic breeding sites were genetically the same as those collected from conventional breeding sites, is clear evidence of the plasticity of Ae. aegypti behavior in the super nidus (Somers et al., 2011).
In our Casa Segura studies, breeding sites around and near premises in Mérida City were identified, using the classification scheme of Servicios de Salud de Yucatán, and characterized for productivity (Garcia-Rejón et al., 2011a). The
most productive breeding sites for Ae. aegypti immatures included small and larger discarded water-holding containers, tires, and so on. The importance of different container types varied between dry and wet periods. Such information is important for targeting productive containers in source reduction campaigns. Entomological investigations also revealed the presence of two categories of extremely productive but uncontrolled breeding sites in the city: storm water drains and vacant lots.
Storm water drains Storm water drains near some of the homes contained large numbers of mosquito immatures. This prompted a survey of storm water drains and catch basins in Mérida City for production of Ae. aegypti and Cx. quinquefasciatus (Arana-Guardia et al., 2014). We examined 1,761 storm water drains located in 45 different neighborhoods spread across the city over dry and wet seasons; 262 (14.9 percent) held water and 123 yielded mosquito immatures. In total, we collected 64,560 immatures representing nine species, including 39,269 Cx. quinquefasciatus and 23,313 Ae. aegypti. Clearly storm water drains produce massive numbers of potential vector mosquitoes across Mérida City, both in the wet and dry seasons, and are nonresidential development sites that should be included in mosquito surveillance and control programs.
Vacant lots We also assessed the potential for vacant lots and other nonresidential settings to serve as source environments for Ae. aegypti (Baak-Baak et al., 2014). Mosquito immatures were collected from residential premises (n = 156 site visits) and nonresidential settings represented by vacant lots, parking lots, and streets or sidewalks. Collections totaled 46,025 mosquito immatures of 13 species. Ae. aegypti was the most commonly encountered species accounting for 81.0 percent of total immatures, followed by Cx. quinquefasciatus (12 percent). Site visits to vacant lots (74 percent) were more likely to result in collection of Ae. aegypti immatures than residential premises (36 percent). Tires accounted for 75.5 percent of Ae. aegypti immatures collected from vacant lots. Vacant lots should be included in mosquito surveillance and control efforts; they often are located near homes and frequently harbor numerous small and large discarded water-holding containers that serve as development sites for immature mosquitoes.
Comment Vacant lots, storm water drains and sewer systems, and other cryptic breeding sites clearly must be included in the efforts to control Ae. aegypti in source reduction and larviciding control programs. The plethora and rapid accumulation of breeding sites in the throw-away society certainly complicate vector control. Even if homeowners clear their own patios and surroundings of breeding sites (for example as in the Patio Limpio program in Mexico and/or in the Recicla por tu Bienestar program in the state of Yucatán; Mendoza-Mezquita et al., 2014), their homes can be inundated by mosquitoes from sites on uncontrolled storm drains, vacant lots, or neighboring patios and yards.
Human mobility complicates Ae. aegypti and dengue control studies Human mobility in the urban environment can confound dengue control studies targeting the home or other indoor environments. Human movement is extensive, thus complicating identification of where individuals become infected with DENV (Stoddard et al., 2013; Vazquez-Prokopec et al., 2013). In the Casa Segura study (Loroño-Pino et al., 2013) and other studies involving protection of the domicile, it is difficult to ascertain whether or not the individual was infected in the home or outside of the home during movement around the city. Thus, if epidemiological outcomes are being measured (e.g., human infections or seroconversions), it is difficult to determine if that infection occurred in the home or outside the home. Consequently, we feel that monitoring homes for DENV-infected Ae. aegypti, which are more restricted to the specific home environment than humans, may be a better measure of the protective effect of the intervention than human infection, because the latter may well have occurred elsewhere.
The Threat of Emerging Insecticide Resistance for Ae. Aegypti Control
Insecticides are critical now and for the foreseeable future to control Ae. aegypti and the pathogens it transmits. Pyrethroids and temephos for adult and larval control, respectively, are the cornerstones of Ae. aegypti control in Mexico and much of the pantropical world. Numerous studies have now documented resistance in Ae. aegypti to these commonly used pesticides, most notably to pyrethroids (e.g., Flores et al., 2013). The increases in insecticide resistance are of great concern. Although the operational significance of these forms of resistance need to be determined, there is the frightening possibility that these tools may be removed from the armamentarium used by mosquito control officials to control dengue. Here the main focus will be upon one form of resistance, “knockdown resistance” (kdr), which has exploded in Ae. aegypti in Mexico and the Yucatán, a pattern that is being documented throughout the pantropical super nidus.
Knock Down Resistance
In mosquitos, kdr is caused by mutations in the voltage-gated sodium channel transmembrane protein (para) that reduce pyrethroid binding (Kasai et al., 2014). Insect sodium channels contain four homologous repeats (domains I–IV) each with six transmembrane segments (S1–S6); interestingly, the majority of pyrethroid-resistance associated mutations occur in the IIS5, IIS6, and IIIS6 segments (see Figure A3-8). In Ae. aegypti, many point mutations associated with pyrethroid resistance had been identified in different geographical mosquito populations (see Figure A3-8). We screened the IIS6 segment in para in 1,318 mosquitoes in 30 strains from throughout Latin America (Saavedra-Rodriguez et al., 2007) and identified two alternate nonsynonymous mutations in codon Ile1,011 in exon 20, and one nonsynonymous mutation at codon Val1,016 in
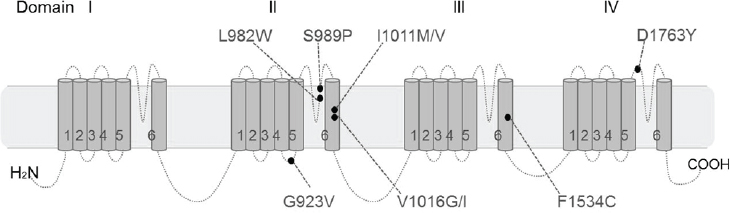
NOTES: Pyrethroid resistance-associated mutations identified in the voltage-gated sodium channel of Aedes aegypti. Point mutations are designated based on the house fly sodium channel gene (GenBank accession number X96668). Replacements G923V, L982W, I1011M and V1016G were first identified by Brengues et al., 2003; S989P was first described by Srisawat et al., 2010; Replacements I1011V and V10161 are described in Saavedra-Rodriguez et al., 2007; F1534 was identified by Yanola et al., 2011; and D1763Y mutation was identified in Chang et al., 2009.
exon 21 of Ae. aegypti. From these point mutations, a transition in the first position of codon 1,016 encoding an Ile replacement (Ile1,016) rapidly increased in frequency in two separate selection experiments, one with deltamethrin on a field strain from Santiago de Cuba and another with permethrin on a strain from Isla Mujeres in Mexico.
The frequency of the kdr-conferring allele, Ile1,016, was then determined in Mexico (Garcia et al., 2009; see Figure A3-9). A total of 81 field collections containing 3,951 Ae. aegypti were made throughout Mexico from 1996 to 2009. These mosquitoes were analyzed for the frequency of the 1,016 mutation using a melting-curve PCR assay. Dramatic increases in frequencies of Ile1,016 were recorded from the late 1990s to 2006–2009 in several states including Nuevo León in the north, Veracruz on the central Atlantic coast, and Yucatán, Quintana Roo, and Chiapas in the south. From 1996 to 2000, the overall frequency of Ile1,016 was 0.04 percent. The earliest detection of Ile1,016 was in Nuevo Laredo on the U.S. border in 1997. By 2003–2004 the overall frequency of Ile1,016 had increased approximately 100-fold to 3 percent. When checked again in 2006, the frequency had increased slightly to 4 percent. This was followed in 2007–2009 by a sudden jump in Ile1,016 frequency to 33 percent. There was spatial heterogeneity in Ile1,016 frequencies among 2007–2008 collections, which ranged from 46 percent in the state of Veracruz to 51 percent in the Yucatán Peninsula and 15 percent in and around Tapachula in the state of Chiapas. Spatial heterogeneity was also evident at smaller geographic scales. For example, within the city of Chetumal, Quintana Roo, Ile1,016 frequencies varied from 38 to 88 percent. This dramatic and rapid increase in kdr frequencies has also been documented in Ae. aegypti populations from throughout the world. This may be related to heavy
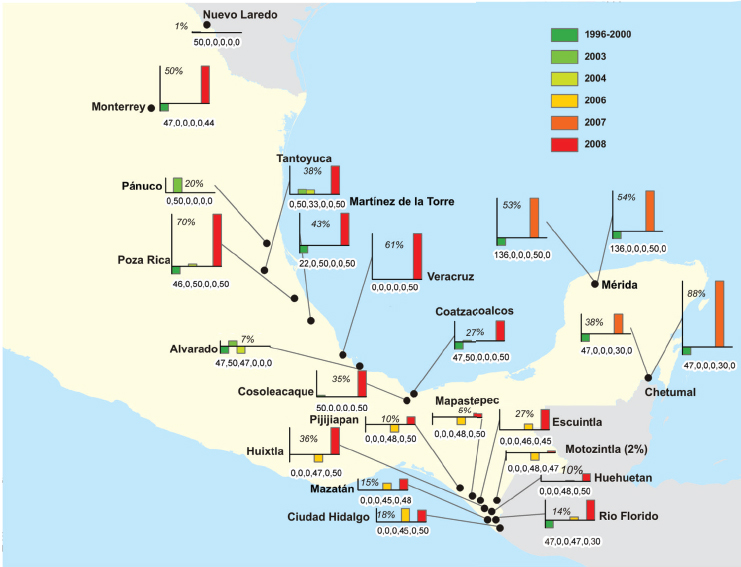
SOURCE: Garcia et al., 2009. Available from PLoS Neglected Tropical Disease under Creative Commons license.
use of permethrin-based insecticides in mosquito control programs. A simple model of positive directional selection predicted rapid fixation of Ile1,016 unless there is negative fitness associated with Ile1,016 in the absence of permethrin. If so, then spatial refugia of susceptible Ae. aegypti or rotational schedules of different classes of adulticides could be established to slow or prevent fixation of Ile1,016 (Garcia et al., 2009).
There was also a dramatic increase in kdr-conferring allele frequencies in Mérida City during the last decade. Previous analyses (Saavedra-Rodriquez et al., 2007) revealed that the kdr allele was absent in Ae. aegypti in Mérida City in 1999. By 2010 the kdr allele was approaching fixation in mosquitoes in the urban center of Mérida City (see Table A3-6). Our model had predicted this rapid fixation in the absence of refugia (Garcia et al., 2009). The operational significance of fixation of the kdr allele remains to be determined. In An. gambiae, kdr alleles have become fixed and have had limited impact upon vector control; however, there continues to be evolution of metabolic resistance mechanisms in the mosquito, which may increase resistance by 1,000 fold (Hemingway, 2014). The same may be true for metabolic resistance in Ae. aegypti (Donnelly et al., 2009).
TABLE A3-6 Temporal Increase in kdr in Aedes aegypti in Mérida City
| Year | Generation | Ile/Ile AA | Ile/Val AG | Val/Val GG | Ile1,016 n | Ile1,016 Allele Frequency |
|---|---|---|---|---|---|---|
| 1999 | F1 | 0 | 0 | 0 | 272 | 0.000 |
| 2007 | F2 | 26 | 55 | 19 | 100 | 0.535 |
| 2010 | F1 | 147 | 77 | 7 | 231 | 0.803 |
| 2011 | F1 | 309 | 135 | 10 | 454 | 0.829 |
| 2013 | F1 | 91 | 22 | 1 | 114 | 0.895 |
Metabolic Resistance
We have also characterized metabolic insecticide resistance mechanisms in Ae. aegypti from the Yucatán using biochemical assays (Flores et al., 2006). The activities of alpha and beta esterases, mixed-function oxidases (MFO), glutathione-S-transferase (GST), acetylcholinesterase (AChE), and insensitive acetylcholinesterase (iAChE) were assayed in microplate assays. Elevation of alpha and beta esterases in some of the populations suggested potential insecticide-resistance mechanisms against organophosphate, carbamate, and pyrethroid insecticides.
Metabolic resistance gene expression before, during, and after five generations of permethrin laboratory selection were monitored in Ae. aegypti from the Yucatán Peninsula (Saavedra-Rodriguez et al., 2012). Changes in expression of 290 metabolic detoxification genes were measured using the Aedes Detox microarray. Selection simultaneously increased the LC(50), KC(50), and Ile1,016 frequency. Very few of the same genes were differentially transcribed among field strains, but 10 cytochrome P(450) genes were upregulated in more than one strain. Identification of one or a few metabolic genes that are predictably associated with permethrin adaptation may very be difficult, but such information would be invaluable for incorporation into mosquito surveillance and control programs.
Potential Factors Conditioning the Increase in Knockdown Resistance
Public health insecticide usage The selective pressures that produced the observed dramatic increase in Ile1,016 frequencies in Mexico remain a subject of discussion. Clearly major insecticidal control efforts have been expended to control Ae. aegypti in Mexico, especially with the emergence of DHF/DSS. Pyrethroids are applied in and around dengue case households and intensive space spraying of areas with dengue cases may promote evolution of resistance. We demonstrated local adaptation to pyrethroids by comparing patterns of variation among 27 Ae. aegypti collections at 13 single nucleotide polymorphisms
(SNPs): Ile1,016 and Cys1,534 in the voltage-gated sodium channel gene (see Figure A3-8), three in detoxification genes previously associated with resistance, and eight in putatively neutral loci. The SNPs in para varied greatly in frequency among collections, whereas SNPs at the remaining 11 loci showed little variation supporting previous evidence for extensive local gene flow. Thus, local adaptation to pyrethroids appears to offset the homogenizing effects of gene flow (Saavedra-Rodriguez et al., 2014). Such control efforts are much less extensive in rural areas and villages, and the importance of vector control in selection for resistance is likely reflected in the fact that kdr frequencies are significantly less in rural areas surrounding Mérida City, where public health vector control is much less intensive than in Mérida City. This is also evidence for negative fitness associated with the Ile1,016 and Cys1,534 alleles in the absence of selection in rural areas.
Consumer product insecticide usage Extensive usage of insecticide consumer products for indoor mosquito control has emerged as a potential additional source of kdr in Ae. aegypti. We conducted a study to evaluate the household use of insecticide consumer products to kill mosquitoes and the expenditures for using these products in Mérida City (Loroño-Pino et al., 2014). A questionnaire was administered to 441 households; 382 (86.6 percent of) surveyed households took action to kill insect pests with consumer products. The most commonly used product types were insecticide aerosol spray cans (74 percent), electric plug-in insecticide emitters (37 percent), and mosquito coils (28 percent) (see Table A3-7). Mosquitoes were targeted by 90 percent of households using insecticide aerosol spray cans, and more than 99 percent of households used electric plug-in insecticide emitters or mosquito coils. Products were used daily or every 2 days in most of the households. For all products used to kill insect pests, the median annual estimated expenditure per household that took action was approximately US$31. These numbers are suggestive of an annual market in excess of US$5.7 million
TABLE A3-7 Consumer Usage of Mosquito Control Products in Homes
| Methods Used to Control Insect Pests* | All Households Number and (percent) | Urban Area Number and (percent) | Rural Area Number and (percent) |
|---|---|---|---|
| Interviewed households | 382 | 300 (79) | 82 (21) |
| Insecticide aerosol spray can | 281 (74) | 238 (79) | 43 (52) |
| Electric plug-in insecticide emitter | 143 (37) | 99 (33) | 44 (54) |
| Mosquito coil | 108 (28) | 82 (27) | 26 (32) |
| Smoke | 19 (5) | 15 (5) | 4 (5) |
| Electric insect racquet | 11 (3) | 10 (3) | 1 (1) |
SOURCE: Adapted from Loroño-Pino et al., 2014.
for Mérida City alone. Homeowners spent substantial amounts of money on insecticide consumer products. Clearly, there is a large market and incentive for companies to provide effective consumer products for vector control.
The constant exposure to pyrethroid-based insecticides resulting from public health control efforts and the use of consumer products could promote insecticide resistance in the mosquito populations.
The Global Threat of Pyrethroid Resistance for Control of VBDs
Pyrethroid resistance also may affect control of other globally important VBDs by insecticides. Pyrethroids are by far the most commonly used mosquito adulticides, and evolution of resistance to these compounds is a major threat to public health. Pyrethroid resistance, both kdr and metabolic resistance, has now been documented in vectors of most globally important pathogens (Hemingway et al., 2006). This resistance threatens some significant advances that have been made in control of VBDs in the last decade. The situation is potentially most grave for control of the Anopheles spp. vectors of malaria in Africa. The use of long-lasting insecticide-treated bed nets (LLIN) and indoor residual spraying (IRS) has reduced malaria deaths by a third (Hemingway, 2014). All LLINs and most IRS have pyrethroid active ingredients. Pyrethroids are relatively safe for use around humans, are easy to formulate, and cheap to produce. Other classes of insecticides do not share these attributes, making pyrethroids the current insecticides of choice (Hemingway, 2014). Some insecticides also have been lost to the armamentarium for vector control. The discovery of DDT and its use to control VBDs was a landmark achievement in public health. However indiscriminant usage of DDT to control insect pests led to detrimental effects on nontarget organisms, and DDT was banned even for public health use in IRS programs. The widespread termination of DDT usage coincided with a resurgence in malaria, leishmaniasis, dengue, and other diseases that are transmitted principally indoors in the Americas and elsewhere (Attaran et al., 2000). Indoor use of DDT disrupted the close association between the human host and important anthropophilic and endophilic vectors, such as Ae. aegypti and An. gambiae, thereby reducing transmission and disease. Pyrethroids have largely taken the place of DDT in vector control, especially in IRS and LLINs. But now this important tool for vector control is in jeopardy.
Although kdr has now been demonstrated in most of the major vectors of important human pathogens, its operational significance remains to be determined. Pyrethroid resistance in An. gambiae in Africa has occurred in waves. Pyrethroid resistance was relatively rare in these species until the start-up of massive vector control programs using LLINs and IRS. Initial kdr resistance was not associated with obvious operational impact, but subsequent waves of resistance have involved relatively new metabolic mechanisms, such as P450-based metabolic enzymes. Metabolic resistance can result in mosquitoes that are 1,000 fold more
resistant to pyrethroids than kdr mosquitoes (Hemingway, 2014). If a similar scenario occurs with Ae. aegypti, pyrethroids may also be lost to the armamentarium for dengue and chikungunya control. The same can be said for control programs for vectors of Chagas, leishmaniasis, filariasis, and other VBDs. The emerging resistance to pyrethroids is a potential public catastrophe on the order of emerging resistance to antibiotics in bacteria. These subjects were addressed in an IOM workshop that for the first time gathered experts in resistance in vectors, bacteria, parasites, and viruses to discuss common mechanisms, threats, and opportunities for mitigating resistance in their respective systems (IOM, 2003b).
Clearly, there is a public health imperative to develop new, environmentally sensitive insecticides and formulations with the efficacy of DDT or pyrethroids to augment existing and future control programs. The development of new insecticides is not trivial; no new chemical insecticides have been brought to market for decades (Hemingway et al., 2006). To help address this public health threat, the Innovative Vector Control Consortium (IVCC) was formed in 2005 to facilitate the development and applications of new insecticides for vector control. The IVCC partners with industry to develop new insecticides of different classes with different modes of action to replace or complement pyrethroids. The new insecticides will provide the potential for rotational or mosaic applications to minimize development of insecticide resistance and thereby provide improved stewardship of existing and new pesticides and sustainable vector control.
Needs and Opportunities
The pantropical urban super nidus for Ae. aegypti-transmitted diseases is a major threat to public health and is proving to be intractable to classical vector control measures. Unprecedented population growth, unplanned urbanization, and the throw-away society are major factors contributing to the creation of the super nidus and a dramatic global increase in Ae. aegypti-transmitted diseases. The situation is exacerbated by globalization and the extraordinary movement of humans, vectors, and viruses throughout the pantropical world. Control measures that proved successful in the past are unlikely to be as successful in the super nidus. The situation is dire. Conventional vector control measures, even when applied most efficiently, as in Singapore (Ooi et al., 2006), are not sufficient to control dengue hyperendemicity. New approaches are sorely needed to augment insecticidal control of Ae. aegypti (Morrison et al., 2008).
Despite the gravity of the situation, new and innovative tools evolving from the digital, genomic, and molecular revolutions are available to augment and enhance control of Ae. aegypti and dengue; some are being developed and tested for efficacy in Ae. aegypti control currently. A major breakthrough occurred with the transfer of Wolbachia, an insect endosymbiont, into Ae. aegypti. Wolbachia infection causes cytoplasmic incompatibility in Ae. aegypti, and thus the bacteria traffics efficiently in laboratory and wild populations, rapidly infecting most of
the target mosquitoes (Hoffman et al., 2011). Most remarkably the Wolbachia-infected mosquitoes are resistant to DENV (and a variety of other pathogens transmitted by mosquitoes), thereby reducing DENV transmission to humans (Moreira et al., 2009). Wolbachia control trials are ongoing in many parts of the world. RIDL (release of insects carrying a dominant lethal) technology is another innovative approach being tested for Ae. aegypti control in field trials. This is more akin to but differs from sterile insect technology (Wise de Valdez et al., 2011; Black et al., 2011). Transgenic males propagated in the presence of tetracycline are innundatively released to mate with wild females (Carvalho et al., 2014). Progeny of these mosquitoes die, thus suppressing the Ae. aegypti population. Field trials are ongoing or planned in other parts of the world. A variety of other genetic approaches are being explored for vector control (Franz et al., 2014). Exciting new chemical interventions for Ae. aegypti control are also being tested. For example, pyriproxyfen (a synthetic juvenile hormone) is being tested for mosquito control. The active ingredient can be used to coat nets and other surfaces. When female mosquitoes contact these nets, they transfer the chemical back to oviposition sites, thereby stopping larval development. Treating LLINs with pyrethroids and pyriproxyfen potentially provides a negative cross-selection resistance-blocking mechanism for vector control (Devine et al., 2009). These strategies are environmentally sensitive and particularly suited to dengue vector control in the super nidus, as in using mosquitoes to find cryptic breeding sites and deliver the control agent. New and effective traps also are being designed that offer exciting new potential to control vector populations (Barrera et al., 2014; Hiscox et al., 2014).
Vector control programs would benefit by not only having new insecticides and innovative control interventions in their armamentarium but also from application of modern management tools. Failure of some vector programs can be attributable to inconsistent implementation of vector control strategies (Hemingway et al., 2006). The digital revolution has provided unprecedented computational power for modeling of control interventions and for developing surveillance and decision support systems, which could enhance the efficacy of control programs (IOM, 2008). Decision support systems can provide improved vector control through rapid and efficient monitoring of entomological and epidemiological parameters related to pathogen transmission; provide more effective vector control through prompt, timely, and focused application of the appropriate insecticides, which can mitigate insecticide resistance; and provide efficient and effective use of resources (Eisen et al., 2011). Many tools with potential to improve vector control are now freely available on the web (e.g., Lozano-Fuentes et al., 2008). It is likely that all of these new technologies and innovative approaches will be needed to control Ae. aegypti in the super nidus. Finally, although not the subject of this contribution, significant advances are being made in the development of a new generation of vaccines for DENV and CHIKV (e.g., da Costa et al., 2014; Powers, 2014).
Conclusion
The burden of Ae. aegypti-transmitted diseases is too great to bear, and new approaches and technologies are critical. It is clearly time to declare “war” on Ae. aegypti and to control this enemy of humankind!
Acknowledgment: This work was supported in part by NIH grant AI088647.
References
Arana-Guardia, R., C. M. Baak-Baak, M. A. Loroño-Pino, C. Machain-Williams, B. J. Beaty, L. Eisen, and J. E. García-Rejón. 2014. Stormwater drains and catch basins as sources for production of Aedes aegypti and Culex quinquefasciatus. Acta Tropica 134:33-42.
Attaran, A., D. R Roberts, C. F. Curtis, and W. L. Kilama. 2000. Balancing risks on the backs of the poor. Nature Medicine 6(7):729-731.
Baak-Baak, C. M., R. Arana-Guardia, N. Cigarroa-Toledo, M. A. Loroño-Pino, G. Reyes-Solis, C. Machain-Williams, B. J. Beaty, L. Eisen, and J. E. García-Rejón. 2014. Vacant lots: Productive sites for Aedes (Stegomyia) aegypti (Diptera: Culicidae) in Mérida City, Mexico. Journal of Medical Entomology 51(2):475-483.
Barrera, R., M. Amador, V. Acevedo, R.R. Hemme, and G. Félix. 2014. Sustained, area-wide control of Aedes aegypti using CDC autocidal gravid ovitraps. American Journal of Tropical Medicine and Hygiene 15. pii: 14-0426.
Barrera, R., M. Amador, A. Diaz, J. Smith, J. L. Munoz-Jordan, and Y. Rosario. 2008. Unusual productivity of Aedes aegypti in septic tanks and its implications for dengue control. Medical Veterinary Entomology 22(1):62-69.
Barrett, A. D., and S. Higgs. 2007. Yellow fever: A disease that has yet to be conquered. Annual Review of Entomology 52:209-229.
Beck, A., H. Guzman, L. Li, B. Ellis, R. B. Tesh, and A. D. Barrett. 2013. Phylogeographic reconstruction of African yellow fever virus isolates indicates recent simultaneous dispersal into east and west Africa. PLoS Neglected Tropical Diseases 7(3):e1910.
Beaty, B. J., and T. H. G. Aitken. 1979. In vitro transmission of yellow fever virus by geographic strains of Aedes aegypti. Mosquito News 39:232238.
Bennett, K. E., K. E. Olson, L. Muñoz, I. Fernandez-Salas, J. A. Farfán-Alé, S. Higgs, W. C. Black 4th, and B. J. Beaty. 2002. Variation in vector competence for dengue 2 virus among 24 collections of Aedes aegypti from Mexico and the United States. American Journal of Tropical Medicine and Hygiene 67(1):85-92.
Bhatt, S., P. W. Gething, O. J. Brady, J. P. Messina, A. W. Farlow, C. L. Moyes, J. M. Drake, J. S. Brownstein, A. G. Hoen, O. Sankoh, M. F. Myers, D. B. George, T. Jaenisch, G. R. Wint, C. P. Simmons, T. W. Scott, J. J. Farrar, and S. I. Hay. 2013. The global distribution and burden of dengue. Nature 496(7446):504-507.
Black, W. C., 4th, K. E. Bennett, N. Gorrochótegui-Escalante, C. V. Barillas-Mury, I. Fernández-Salas, M. de Lourdes Muñoz, J. A. Farfán-Alé, K. E. Olson, and B. J. Beaty. 2002. Flavivirus susceptibility in Aedes aegypti. Archives of Medical Research 33(4):379-388.
Black, W. C., 4th, L. Alphey, and A. A. James. 2011. Why RIDL is not SIT. Trends in Parasitology 27(8):362-370.
Bonds, J. A. S. 2012. Ultra-low-volume space sprays in mosquito control: A critical review. Medical Veterinary Entomology 26:121-130.
Brengues, C., N. J. Hawkes, F. Chandre, L. McCarroll, S. Duchon, P. Guillet, S. Manguin, J. C. Morgan, and J. Hemingway. 2003. Pyrethroid and DDT cross-resistance in Aedes aegypti is correlated with novel mutations in the voltage-gated sodium channel gene. Medical Veterinary Entomology 17(1):87-94.
Brown, J. E., C. S. McBride, P. Johnson, S. Ritchie, C. Paupy, H. Bossin, J. Lutomiah, I. Fernandez-Salas, A. Ponlawat, A. J. Cornel, W. C. Black 4th, N. Gorrochotegui-Escalante, L. Urdaneta-Marquez, M. Sylla, M. Slotman, K.O. Murray, C. Walker, and J. R. Powell. 2011. Worldwide patterns of genetic differentiation imply multiple domestications’ of Aedes aegypti, a major vector of human diseases. Proceedings of the Royal Society of London. Series B, Biological Sciences 278(1717):2446-2454.
Carey, D.E. 1971. Chikungunya and dengue: A case of mistaken identity? Journal of the History of Medicine and Allied Sciences 26:243-262.
Carvalho, D. O., D. Nimmo, N. Naish, A. R. McKemey, P. Gray, A. B. Wilke, M. T. Marrelli, J. F. Virginio, L. Alphey, and M. L. Capurro. 2014. Mass production of genetically modified Aedes aegypti for field releases in Brazil. Journal of Visualized Experiments 4(83):e3579.
Chang, C., W. K. Shen, T. T. Wang, Y. H. Lin, E. L. Hsu, and S. M. Dai. 2009. A novel amino acid substitution in a voltage-gated sodium channel is associated with knockdown resistance to permethrin in Aedes aegypti. Insect Biochemistry and Molecular Biology 39(4):272-278.
da Costa, V. G., A. C. Marques-Silva, V. G. Floriano, and M. L. Moreli. 2014. Safety, immunogenicity and efficacy of a recombinant tetravalent dengue vaccine: A meta-analysis of randomized trials. Vaccine 32(39):4885-4892.
Díaz, F. J., W. C. Black 4th, J. A. Farfán-Alé, M. A. Loroño-Pino, K. E. Olson, and B. J. Beaty. 2006. Dengue virus circulation and evolution in Mexico: A phylogenetic perspective. Archives of Medical Research 37(6):760-773.
Devine, G. J., E. Z. Perea, G. F. Killeen, J. D. Stancil, S. J. Clark, and A. C. Morrison. 2009. Using adult mosquitoes to transfer insecticides to Aedes aegypti larval habitats. Proceedings of the National Academy of Sciences 106(28):11530-11534.
Dickson, L. B., I. Sanchez-Vargas, M. Sylla, K. Fleming, and W. C. Black 4th. 2014. Vector competence in West African Aedes aegypti is flavivirus species and genotype dependent. PLoS Neglected Tropical Diseases 8(10):e3153.
Donnelly, M. J., V. Corbel, D. Weetman, C. S. Wilding, M. S. Williamson, and W. C. Black 4th. 2009. Does kdr genotype predict insecticide-resistance phenotype in mosquitoes? Trends in Parasitology 25(5):213-219.
Edman, J. D., D. Strickman, P. Kittayapong, and T. W. Scott. 1992. Female Aedes aegypti (Diptera: Culicidae) in Thailand rarely feed on sugar. Journal of Medical Parasitology 29(6):1035-1038.
Eisen, L., B. J. Beaty, A. C. Morrison, and T. W. Scott. 2009. Proactive vector control strategies and improved monitoring and evaluation practices for dengue preventional strategies and improved monitoring and evaluation practices for dengue prevention. Journal of Medical Entomology 46(6):1245-1255.
Eisen, L., M. Coleman, S. Lozano-Fuentes, N. McEachen, M. Orlans, and M. Coleman. 2011. Multidisease data management system platform for vector-borne diseases. PLoS Neglected Tropical Diseases 5:e1016.
Franz, A. W., R. J. Clem, and A. L. Passarelli. 2014. Novel genetic and molecular tools for the investigation and control of dengue virus transmission by mosquitoes. Current Tropical Medicine Reports 1(1):21-31.
Flores, A. E., J. S. Grajales, I. F. Salas, G. P. Garcia, M. H. Becerra, S. Lozano, W. G. Brogdon, W. C. Black 4th, and Beaty, B. 2006. Mechanisms of insecticide resistance in field populations of Aedes aegypti (L.) from Quintana Roo, Southern Mexico. Journal of the American Mosquito Control Association 22(4):672-677.
Flores, A. E., G. Ponce, B. Silva, S. Gutierrez, C. Bobadilla, B. Lopez, R. Mercado, and I. W. C. Black. 2013. Wide spread cross resistance to pyrethroids in Aedes aegypti (Diptera: Culicidae) from Veracruz state Mexico. Journal of Economic Entomology 106(2013):959-969.
Garcia, G. P., A. E. Flores, I. Fernández-Salas, K. Saavedra-Rodríguez, G. Reyes-Solis, S. Lozano-Fuentes, J. Guillermo Bond, M. Casas-Martínez, J. M. Ramsey, J. García-Rejón, M. Domínguez-Galera, H. Ranson, J. Hemingway, L. Eisen, and W. C. Black IV. 2009. Recent rapid rise of a permethrin knock down resistance allele in Aedes aegypti in Mexico. PLoS Neglected Tropical Disease 3(10):e531.
Garcia-Rejón, J., M. A. Loroño-Pino, J. A. Farfán-Alé, L. Flores-Flores, E. Del Pilar Rosado-Paredes, N. Rivero-Cardenas, R. Najera-Vazquez, S. Gomez-Carro, V. Lira-Zumbardo, P. Gonzalez-Martinez, S. Lozano-Fuentes, D. Elizondo-Quiroga, B. J. Beaty, and L. Eisen. 2008. Dengue virus-infected Aedes aegypti in the home environment. American Journal of Tropical Medicine and Hygiene 79(6):940-950.
García-Rejón, J. E., M. P. López-Uribe, M. A. Loroño-Pino, J. A. Farfán-Alé, M. R. Del Najera-Vazquez, S. Lozano-Fuentes, B. J. Beaty, and L. Eisen. 2011a. Productive container types for Aedes aegypti immatures in Mérida, Mexico. Journal of Medical Entomology 48(3):644-650.
García-Rejón, J. E., M. A. Loroño-Pino, J. A. Farfán-Alé, L. F. Flores-Flores, M. P. López-Uribe, R. Najera-Vazquez, G. Nuñez-Ayala, B. J. Beaty, and L. Eisen. 2011b. Mosquito infestation and dengue virus infection in Aedes aegypti females in schools in Merida, Mexico. American Journal of Tropical Medicine and Hygiene 84(3):489-496.
Gorrochotegui-Escalante, N., C. Gomez-Machorro, S. Lozano-Fuentes, L. Fernandez-Salas, M. De Lourdes Munoz, J. A. Farfán-Alé, J. Garcia-Rejón, B. J. Beaty, and W. C. Black 4th. 2002. Breeding structure of Aedes aegypti populations in Mexico varies by region. American Journal of Tropical Medicine and Hygiene 66(2):213-22.
Gratz, N. G. 1999. Emerging and resurging vector-borne diseases. Annual Review of Entomology 44:51-75.
Gubler, D. 2005. The emergence of epidemic dengue fever and dengue hemorrhagic fever in the Americas: A case of failed public health policy. Revista Panamericana de Salud Pública 17(4):221-224.
Gubler, D. J. 2011. Dengue, urbanization, and globalization: The unholy trinity of the 21(st) century. Tropical Medicine and Health 39:3-11.
Guzman, M. G., and E. Harris. 2015. Dengue. Lancet 85(9966):453-465.
Halstead, S.B. 2015. Reappearance of chikungunya, formerly called dengue, in the Americas. Emerging Infectious Diseases 21:557-561.
Hanley, K. A., T. P. Monath, S. C. Weaver, S. L. Rossi, R. L. Richman, and N. Vasilakis. 2013. Fever versus fever: The role of host and vector susceptibility and interspecific competition in shaping the current and future distributions of the sylvatic cycles of dengue virus and yellow fever virus. Infectious, Genetics, and Evolution 19:292-311.
Harrington, L. C., J. D. Edman, and T. W. Scott. 2001. Why do female Aedes aegypti (Diptera: Culicidae) feed preferentially and frequently on human blood? Journal of Medical Entomology 38(3):411-422.
Harrington, L. C., A. Fleisher, D. Ruiz-Moreno, F. Vermeylen, C. V. Wa, R. L. Poulson, J. D. Edman, J. M. Clark, J. W. Jones, S. Kitthawee, and T. W. Scott. 2014. Heterogeneous feeding patterns of the dengue vector, Aedes aegypti, on individual human hosts in rural Thailand. PLoS Neglected Tropical Diseases 8(8):e3048. doi: 10.1371/journal.pntd.0003048.
Hemingway, J. 2014. The role of vector control in stopping the transmission of malaria: Threats and opportunities. Philosophical Transactions of the Royal Society of London. Series B: Biological Sciences 369(1645):20130431.
Hemingway, J., B. J. Beaty, M. Rowland, T. W. Scott, and B. L. Sharp. 2006. The Innovative Vector Control Consortium: Improved control of mosquito-borne diseases. Trends in Parasitology 22(7):308-312.
Hemingway, J., J. Vontas, R. Poupardin, J. Raman, J. Lines, C. Schwabe, A. Matias, and I. Kleinschmidt. 2013. Country-level operational implementation of the global plan for insecticide resistance management. Proceedings of the National Academy of Sciences of the United States of America 110(23):9397-9402.
Hiscox, A., B. Otieno, A. Kibet, C. K. Mweresa, P. Omusula, M. Geier, A. Rose, W. R. Mukabana, and W. Takken. 2014. Development and optimization of the Suna trap as a tool for mosquito monitoring and control. Malaria Journal 13:257.
Hoffmann, A. A., B. L. Montgomery, J. Popovici, I. Iturbe-Ormaetxe, P. H. Johnson, F. Muzzi, M. Greenfield, M. Durkan, Y. S. Leong, Y. Dong, H. Cook, J. Axford, A. G. Callahan, N. Kenny, C. Omodei, E. A. McGraw, P. A. Ryan, S. A. Ritchie, M. Turelli, and S. L. O’Neill. 2011. Successful establishment of Wolbachia in Aedes populations to suppress dengue transmission. Nature 476(7361):454-457.
Kasai, S., O. Komagata, K. Itokawa, T. Shono, L. C. Ng, M. Kobayashi, and T. Tomita. 2014. Mechanisms of pyrethroid resistance in the dengue mosquito vector, Aedes aegypti: Target site insensitivity, penetration, and metabolism. PLoS Neglected Tropical Diseases 8(6):e2948.
IOM (Institute of Medicine). 2003a. Microbial threats to health. Washington, DC: The National Academies Press.
IOM. 2003b. The resistance phenomenon in microbes and infectious disease vectors. Implications for human health and strategies for containment. Washington, DC: The National Academies Press.
IOM. 2008. Vector-borne diseases: Understanding the environmental, human health, and ecological connections. Washington, DC: The National Academies Press.
Kroeger, A., A. Lenhart, M. Ochoa, E. Villegas, M. Levy, N. Alexander, and P. J. McCall. 2006. Effective control of dengue vectors with curtains and water container covers treated with insecticide in Mexico and Venezuela: Cluster randomised trials. British Medical Journal 332(7552): 1247-1252.
Loroño-Pino, M. A., J. A. Farfán-Alé, A. L. Zapata-Peraza, E. P. Rosado-Paredes, L. F. Flores-Flores, J. E. García-Rejón, F .J. Díaz, B. J. Blitvich, M. Andrade-Narváez, E. Jiménez-Ríos, C. D. Blair, K. E. Olson, W. Black 4th, and B. J. Beaty. 2004. Introduction of the American/Asian genotype of dengue 2 virus into the Yucatan State of Mexico. American Journal of Tropical Medicine and Hygiene 71(4):485-492.
Loroño-Pino, M. A., J. E. García-Rejón, C. Machain-Williams, S. Gomez-Carro, G. Nuñez-Ayala, R. Nájera-Vázquez, A. Losoya, L. Aguilar, K. Saavedra-Rodriguez, S. Lozano-Fuentes, M. K. Beaty, W. C. Black 4th, T. J. Keefe, L. Eisen, and B. J. Beaty. 2013. Towards a Casa Segura: A consumer product study of the effect of insecticide-treated curtains on Aedes aegypti and dengue virus infections in the home. American Journal of Tropical Medicine and Hygiene 89(2):385-397.
Loroño-Pino, M. A., Y. N. Chan-Dzul, R. Zapata-Gil, C. Carrillo-Solís, A. Uitz-Mena, J. E. García-Rejón, T. J. Keefe, B. J. Beaty, and L. Eisen. 2014. Household use of insecticide consumer products in a dengue-endemic area in Mexico. Tropical Medicine & International Health 19(10):1267-1275.
Lounibos, L. P. 2002. Invasions by insect vectors of human disease. Annual Review of Entomology 47:233-266.
Lozano-Fuentes, S., D. Elizondo-Quiroga, J. A. Farfán-Alé, M. A. Loroño-Pino, J. Garcia-Rejón, S. Gomez-Carro, V. Lira-Zumbardo, R. Najera-Vazquez, I. Fernandez-Salas, J. Calderon-Martinez, M. Dominguez-Galera, P. Mis-Avila, N. Morris, M. Coleman, C. G. Moore, B. J. Beaty, and L. Eisen. 2008. Use of Google Earth to strengthen public health capacity and facilitate management of vector-borne diseases in resource-poor environments. Bulletin of the World Health Organization 86(9):718-725.
Lozano-Fuentes, S., Fernandez-Salas, I., M. de Lourdes Munoz, J. Garcia-Rejón, K. E. Olson, B. J. Beaty, and W. C. Black 4th. 2009. The neovolcanic axis is a barrier to gene flow among Aedes aegypti populations in Mexico that differ in vector competence for dengue 2 virus. PLoS Neglected Tropical Diseases 3(6):e468.
Mendoza-Mezquita, J. E., N. Torres-Arcila, M. M. Tec-Kumul, W. A. Páez-Cantón, J. L. Fraga Moreno, J. R. Torres-Castro, S. M. Vázquez-Narváez, and M. R. Nájera-Vázquez. 2014. Recicla por tu bienestar para prevenir el dengue. Poster session presented at: LXVIII Reunión Anual de Salud Pública en Mexico. November 19-22, Mérida, Yucatán, Mexico.
Messina, J. P., O. J. Brady, T. W. Scott, C. Zou, D. M. Pigott, K. A. Duda, S. Bhatt, L. Katzelnick, R. E. Howes, K. E. Battle, C. P. Simmons, and S. I. Hay. 2014. Global spread of dengue virus types: Mapping the 70 year history. Trends in Microbiology 22(3):138-146.
Mohan, A., D. Kiran, I. Manohar, and Kuman D. 2010. Epidemiology, clinical manifestations, and diagnosis of chikungunya fever: Lessons learned from the re-emerging epidemic. Indian Journal of Dermatology 55(1):54-63.
Moore, M., M. Sylla, L. Goss, M. W. Burugu, R. Sang, L. W. Kamau, E. U. Kenya, C. Bosio, L. Munoz, M. Sharakova, and W. C. Black. 2013. Dual African origins of global Aedes aegypti s.l. populations revealed by mitochondrial DNA. PLoS Neglected Tropical Diseases 7(4):e2175.
Moreira, L. A., I. Iturbe-Ormaetxe, J. A. Jeffery, G. Lu, A. T. Pyke, L. M. Hedges, B. C. Rocha, S. Hall-Mendelin, A. Day, M. Riegler, L. E. Hugo, K. N. Johnson, B. H. Kay, E. A. McGraw, A. F. van den Hurk, P. A. Ryan, and S. L. O’Neill. 2009. A Wolbachia symbiont in Aedes aegypti limits infection with dengue, chikungunya, and Plasmodium. Cell 139(7):1268-1278.
Morrison, A. C., E. Zielinski-Gutierrez, T. W. Scott, and R. Rosenberg. 2008. Defining challenges and proposing solutions for control of the virus vector Aedes aegypti. PLoS Medicine 5(3):e68. doi: 10.1371/journal.pmed.0050068.
Mutebi, J. P., and A. D. Barrett. 2002. The epidemiology of yellow fever in Africa. Microbes and Infection 4(14):1459-1468.
Nasci, R. 2014. Movement of chikungunya virus into the Western Hemisphere. Emerging Infectious Diseases 20(8):1394-1395.
Ooi, E. E., K. T. Goh, and D. J. Gubler. 2006. Dengue prevention and 35 years of vector control in Singapore. Emerging Infectious Diseases 12(6):887-893.
Petersen, L. R., A. C. Brault, and R. S. Nasci. 2013. West Nile virus: Review of the literature. JAMA 310(3):308-315.
Powell, J. R., and W. J. Tabachnick. 2013. History of domestication and spread of Aedes aegypti—A review. Memórias do Instituto Oswaldo Cruz 108(Suppl 1):11-17.
Powers, A. M. 2014. Chikungunya virus control: Is a vaccine on the horizon? Lancet S0140-6736(14)61290-61293.
Reiter P., and D. J. Gubler. 1997. Surveillance and control of urban dengue vectors. In: D. J. Gubler and G. Kuno, editors. Dengue and dengue hemorrhagic fever. New York: CAB International. Pp. 425-462.
Reiter, P., S. Lathrop, M. Bunning, B. Biggerstaff, D. Singer, T. Tiwari, L. Baber, M. Amador, J. Thirion, J. Hayes, C. Seca, J. Mendez, B. Ramirez, J. Robinson, J. Rawlings, V. Vorndam, S. Waterman, D. Gubler, G. Clark, and E. Hayes. 2003. Texas lifestyle limits transmission of dengue virus. Emerging Infectious Diseases 9(1):86-89.
Renault, P., L. Josseran, and V. Pierre. 2008. Chikungunya-related fatality rates, Mauritius, India, and Reunion Island. Emerging Infectious Diseases 14(8):1327.
Saavedra-Rodriguez, K., L. Urdaneta-Marquez, S. Rajatileka, M. Moulton, A. E. Flores, I. Fernandez-Salas, J. Bisset, M. Rodriguez, P. J. McCall, M. J. Donnell, H. Ranson, J. Hemingway, and W. C. Black 4th. 2007. A mutation in the voltage-gated sodium channel gene associated with pyrethroid resistance in Latin American Aedes aegypti. Insect Molecular Biology 16(6):785-798.
Saavedra-Rodriguez, K., A. F. Suarez, I. F. Salas, C. Strode, H. Ranson, J. Hemingway, and W. C. Black 4th. 2012. Transcription of detoxification genes after permethrin selection in the mosquito Aedes aegypti. Insect Molecular Biology 21(1):61-77.
Saavedra-Rodriguez, K., M. Beaty, S. Lozano-Fuentes, S. Denham, J. Garcia-Rejón, G. Reyes-Solis, C. Machain-Williams, M. A. Loroño-Pino, A. Flores-Suarez, G. Ponce-Garcia, B. Beaty, L. Eisen, and W. C. Black 4th. 2014. Local evolution of pyrethroid resistance offsets gene flow among Aedes aegypti collections in Yucatan State, Mexico. American Journal of Tropical Medicine and Hygiene 92(1):201-209.
Salazar, M. I., M. A. Loroño Pino, J. A. Farfán Alé, K. E. Olson, and B. J. Beaty. 2010. American and American/Asian genotypes of dengue virus differ in mosquito infection efficiency: Candidate molecular determinants of productive vector infection. Revista Biomédica 21(3):121-135.
Salazar-Sanchez, I., I. Sanchez-Vargas, J. H. Richardson, K. E. Olson, and B. J. Beaty. 2007. Dengue virus type 2: Virogenesis and tropisms in orally infected Aedes aegypti mosquitoes. BMC Microbiology 7:9.
Scott, T. W., G. G. Clark, L. H. Lorenz, P. H. Amerasinghe, P. Reiter, and J. D. Edman. 1993. Detection of multiple blood feeding in Aedes aegypti (Diptera: Culicidae) during a single gonotrophic cycle using a histologic technique. Journal of Medical Entomology 30(1):94-99.
Scott, T. W., P. H. Amerasinghe, A. C. Morrison, L. H. Lorenz, G. G. Clark, D. Strickman, P. Kittayapong, and J. D. Edman. 2000. Longitudinal studies of Aedes aegypti (Diptera: Culicidae) in Thailand and Puerto Rico: Blood feeding frequency. Journal of Medical Entomology 37(1):89-101.
Scott, T. W., and W. Takken. 2012. Feeding strategies of anthropophilic mosquitoes result in increased risk of pathogen transmission. Trends in Parasitology 28(3):114-121. doi: 10.1016/j. pt.2012.01.001.
Smith, C. 1956. The history of dengue in tropical Asia and its probable relationship to the mosquito Aedes aegypti. Journal of Tropical Medicine and Hygiene 59:3-11.
Somers, G., J. E. Brown, R. Barrera, and J. R. Powell. 2011. Genetics and morphology of Aedes aegypti (Diptera: Culicidae) in septic tanks in Puerto Rico. Journal of Medical Entomology 48(6):1095-1102.
Soumahoro, M., P. Boelle, B. Gaüzere, K. Atsou, C. Pelat, B. Lambert, G. Ruche, M. Gastellu-Etchegorry, P. Renault, M. Sarazin, Y. Yazdanpanah, A. Flahault, D. Malvy, and T. Hanslik. 2011. The chikungunya epidemic on La Réunion Island in 2005–2006: A cost-of-illness study. PLoS Neglected Tropical Diseases 5(6):e1197.
Srikiatkhachorn, A., A. L. Rothman, R. V. Gibbons, N. Sittisombut, P. Malasit, F. A. Ennis, S. Nimmannitya, and S. Kalayanarooj. 2011. Dengue—How best to classify it. Clinical Infectious Diseases 53(6):563-567.
Srisawat, R., N. Komalamisra, C. Apiwathnasorn, P. Paeporn, S. Roytrakul, Y. Rongsriyam, and Y. Eshita. 2012. Field-collected permethrin-resistant Aedes aegypti from central Thailand contain point mutations in the domain IIS6 of the sodium channel gene (KDR). Southeast Asian Journal of Tropical Medicine and Public Health 43(6):1380-1386.
Stoddard, S. T., B. M. Forshey, A. C. Morrison, V. A. Paz-Soldan, G. M. Vazquez-Prokopec, H. Astete, R. C. Reiner Jr., S. Vilcarromero, J. P. Elder, E. S. Halsey, T. J. Kochel, U. Kitron, and T. W. Scott. 2013. House-to-house human movement drives dengue virus transmission. Proceedings of the National Academy of Sciences of the United States of America 110(3):994-999.
Strode, C., S. Donegan, P. Garner, A. A. Enayati, and J. Hemingway. 2014. The impact of pyrethroid resistance on the efficacy of insecticide-treated bed nets against African anopheline mosquitoes: Systematic review and meta-analysis. PLoS Medicine 11(3):e1001619.
Sylla, M., C. Bosio, L. Urdaneta-Marquez, M. Ndiaye, and W. C. Black 4th. 2009. Gene flow, subspecies composition, and dengue virus-2 susceptibility among Aedes aegypti collections in Senegal. PLoS Neglected Tropical Diseases 3(4):e408.
Tabachnick, W. 2013. Nature, nurture, and evolution of intra-species variation in mosquito arbovirus transmission competence. International Journal of Environmental Research and Public Health 10:249-277.
Tabachnick, W. J., G. P. Wallis, T. H. G. Aitken, B. R. Miller, G. D. Amato, L. Lorenz, J. R. Powell, and B. J. Beaty. 1985. Oral infection of Aedes aegypti with yellow fever virus: Geographic variation and genetic considerations. American Journal of Tropical Medicine and Hygiene 34:12191224.
Tandale, B. V., P. S. Sathe, V. A. Arankalle, R. S. Wadia, R. Kulkarni, S. V. Shah, S. K. Shah, J. K. Sheth, A. B. Sudeep, A. S. Tripathy, and A. C. Mishra. 2009. Systemic involvements and fatalities during chikungunya epidemic in India, 2006. Journal of Clinical Virology 46(2):145-149.
Vasilakis, N., E. J. Shell, E. B. Fokam, P. W. Mason, K. A. Hanley, D. M. Estes, and S. C. Weaver. 2007. Potential of ancestral sylvatic dengue-2 viruses to re-emerge. Virology 358(2):402-412.
Vasilakis, N., J. Cardosa, M. Diallo, A. A. Sall, E. C. Holmes, K. A. Hanley, S. C. Weaver, J. Mota, and R. Rico-Hesse. 2010. Sylvatic dengue viruses share the pathogenic potential of urban/endemic dengue viruses. Journal of Virology 84(7):3726-3728.



































