5
Influenza Risk Assessment
and Pandemic Preparedness
“Pandemic influenza is different from the other emergencies that we prepare for,” said Andrew Pavia, chief of the Division of Pediatric Infectious Diseases at the University of Utah. It is the one biological emergency that is certain to happen. However, given the enormous geographic diversity, the number of influenza viruses, and the rate of mutations, it is unknown how, when, and where pandemic influenza will emerge. Predictions to date have not been very accurate, and pandemics such as the 2009 H1N1 influenza confounded all preparedness models. It is also different in that it is intensely studied, and there is a commercial market, albeit small, for influenza products, which separates it from other infectious disease threats in this report. For example, all of the profits from the antiviral medication, Tamiflu, came from selling it to the SNS.
Despite how much is known about influenza, challenges in developing MCM for pandemic influenza remain. A large number of new viruses are discovered every year and consequently generate many false alarms. The inconsistent presence of pandemic influenza also creates a fluctuating market for products and the corresponding investment in research and development. When it is the “disease of the month,” Pavia said, money becomes available and efforts are made, but sustainable funding does not exist in the interepidemic periods. In this section, representatives from the government and the private sector discussed evaluating risk, developing tools and capacity, and partnering with influenza vaccine manufacturers to make a range of MCM available. (Detailed technical accounts of the MCM development process are included at the end of this chapter in the Chapter 5 Annex.)
INFLUENZA RISK ASSESSMENT TOOL
Jacqueline Katz, deputy director (acting) of the Influenza Division at CDC, gave a brief overview of the CDC Influenza Risk Assessment Tool (IRAT). IRAT is a simple, additive, multiattribute assessment tool to prioritize pandemic preparedness activities.1 Specifically, it evaluates the risk from novel influenza viruses that are circulating in animals. Using the tool, subject-matter experts from different disciplines evaluate available data and provide a quantitative risk assessment regarding the likelihood that a novel virus will emerge in humans and the likely public health impact if that does occur. Katz clarified that IRAT cannot predict the next pandemic; it is designed to compare the risk from circulating viruses. She emphasized that it will continually be updated as new information, technologies, and methods to assess data and risk become available. With new influenza A viruses constantly emerging from animal reservoirs, and the tenfold increase in the number of human infections with different novel influenza A viruses since the 1990s, Katz said, risk assessments are needed to guide decision making with respect to vaccine development, testing, and manufacturing, as well as the procurement of MCMs and other preparedness needs (e.g., diagnostics).
Putting IRAT into Action
IRAT is an objective, transparent approach that measures risk consistently with minimal bias and provides documentation to support government decisions, Katz said. The same standardized approach can be used year to year to assess influenza virus variants. The tool builds on a strong global influenza network for virus and genetic sequence sharing (WHO Global Influenza Surveillance and Response System).2 IRAT can also help to identify gaps in knowledge and data and encourages data sharing and input from both public health and animal health sectors. As noted, the tool is evaluated and reviewed regularly in an iterative process and can be rapidly updated. By evaluating many viruses including H7N9, H5N1, H9N2, and H3N2v, IRAT has informed vaccine development and procurement decisions by the government.
___________________
1See http://www.cdc.gov/flu/pandemic-resources/tools/risk-assessment.htm (accessed June 30, 2015).
2See http://www.influenzacentre.org/centre_GISRS.htm (accessed August 14, 2015).
Guiding Decision Making
Katz noted that IRAT scores are provided regularly to agencies within HHS to guide pre-pandemic risk management decisions, including which additional vaccine antigens should be produced, stockpiled, or selected for clinical trials to mitigate the potential public health impact of an emerging virus. IRAT is one component of a multifaceted decision-making process, she said. In assessing pandemic vaccine priorities, key elements are human infection, antigenic relationship, global distribution, infection in animals, and genomic variation. When considering which virus poses the greatest impact to public health, CDC considers the antigenic relatedness relative to a vaccine already available in the SNS and the antigenic relatedness to a candidate vaccine virus that may already exist but has not progressed to pilot lot production.
Risk Management
Information from IRAT is used at the HHS Pandemic and Seasonal Influenza Risk Management Meeting (also known as the Flu Risk Management Meeting, or FRMM)3 to make decisions about influenza strains for inclusion in the pre-pandemic vaccine stockpile, explained Rick Bright, acting director of the Influenza Division at BARDA. FRMM is a senior-level forum for decision makers from stakeholder agencies to identify and address risk management issues related to the development, acquisition, deployment, and use of medical and public health countermeasures for influenza. Decisions are evidence based, using a measured approach to response that ranges from monitoring novel strain emergence to a full pandemic vaccine production response, Bright said. As mentioned by Katz, IRAT is one piece of information considered in the risk management process.
Over the past 10 years, BARDA has made decisions early on to buy bulk lots of vaccine candidates for H5N1 influenza and store those bulk lots in long-term storage. Although BARDA has gained valuable knowledge in doing this, it is an expensive approach, Bright noted. Producing and storing a full-sized bulk lot of vaccine for each threat that might emerge is not economically feasible. Recalling Robinson’s previous comments on the need for prioritization and assessment, Bright said BARDA works with CDC to implement IRAT and other factors that
___________________
3See http://report.nih.gov/crs/View.aspx?Id=2641 (accessed August 14, 2015).
inform decisions on how, where, and to what level a response is initiated. This facilitates better preparedness for a wider variety of threats instead of investing all or a significant portion of funds into only one or two threats. Importantly, everything along this continuum involves the manufacturer, he added. BARDA has a flexible contracting mechanism so it can respond in a variety of ways, to a variety of threats, with different manufacturers who may or may not be able to respond at the time needed.
BARDA Decision Making for Pandemics
Bright described several examples where the risk-based, measured approach was used. The 2005 H5N1 influenza outbreak in Southeast Asia, for example, was determined to be a significant threat. In response, BARDA established a stockpile, met stockpiling goals, and implemented an innovative mix-and-match program with NIH to begin evaluating different antigen-adjuvant combinations that might be needed in a pandemic response. In 2009, the H1N1 influenza pandemic “took us by surprise,” Bright said, and BARDA went into full response mode. A total of 186 million doses of H1N1 vaccine were produced and filled by the manufacturers, along with production of 120 million doses of bulk adjuvants as a contingency. One of the first times IRAT was used was for the H3N2v influenza outbreak in 2012. In this case, clinical lots of candidate vaccines were made, and clinical trials were conducted to determine the dosage needed, but no further development was done. Currently, the 2013 emergence of H7N9 influenza is the highest threat, according to the IRAT, and the decision was made to produce clinical lots, to conduct clinical studies, and to stockpile bulk antigen. Bright emphasized that the stockpile program and measured response approach have enabled BARDA to gain valuable knowledge about the physical properties of antigens and adjuvants, storage conditions, potency over time, and other information that is needed to manage risk and inform development of the next generation of influenza vaccines.
INDUSTRY PARTNERS IN PANDEMIC
PREPAREDNESS AND RAPID RESPONSE
The national vaccine goals for pandemic influenza preparedness call for pre-pandemic vaccine stockpiles to protect 20 million people as well
as for manufacturing infrastructure to support rapid production of 600 million doses, said Monique Mansoura of Novartis. She emphasized the dynamic nature of the influenza threat and questioned the match of vaccine stockpiles that were purchased 10 years ago against today’s circulating strains. The H5N1 influenza emerging in Egypt in 2015 is not necessarily the H5N1 strain that emerged in Vietnam in 2004. She concurred with O’Toole of In-Q-Tel and others that the public health sector is largely underestimating and under-requesting what is needed to protect Americans from pandemic influenza and other threats. Although focus has been largely on the cost of purchasing pharmaceuticals for the nation’s SNS, she highlighted that little attention is given to the cost of inaction. There are countless other dynamic pandemic threats, and Mansoura noted that creating a dynamic and strategic stockpile strategy should be considered.
We are focused on the cost of buying a stockpile, but we are not necessarily focused on the cost of inaction.
— Monique Mansoura, Head, Medical
Countermeasures & Government
Affairs, Americas, Novartis
Influenza Vaccines
Following the 2009 H1N1 influenza outbreak, during which the vaccine supply lagged behind the demand, the President’s Council of Advisors on Science and Technology (PCAST) released a report on reengineering the nation’s influenza vaccine enterprise to enable a more rapid vaccine response to pandemic influenza (PCAST, 2010). The recommendations from the White House focused on new or enhanced science and technological advances, including investment in new vaccine manufacturing platforms. The PCAST report also called for studies of the use of adjuvants and development of the FDA guidance on the use of adjuvants with seasonal influenza vaccines to improve vaccine efficacy.
Needs for Future Stockpiles and Capabilities
Mansoura called for a “rebranding” of how stockpiles and response in the pre-pandemic phase are typically conceived. The H7N9 influenza response was an unprecedented response to a pre-pandemic threat, she said. Within 8 months of identifying the first case, HHS had a vaccine stockpile. Much was learned along the way about integrating new technologies, synthetic seeds, adjuvants, clinical trials, antigens, measured response, and stockpiling. Mansoura raised a concern that it took five separate contracting actions to secure an HHS decision to
purchase the stockpile once created, and there seemed to be an expectation that manufacturers would do this type of work when needed at their own risk. This, she said, is a very challenging way to do business. While the development of innovative platforms may someday enable a sufficiently rapid response after a new threat emerges, Mansoura contended that current assumptions and existing capabilities must be tested to provide verifiable assessments of the nation’s state of preparedness before an event occurs (e.g., developing and testing a vaccine, determining the dose, stockpiling, understanding the stability profile of the pharmaceutical over time).
Mansoura went on to note that the vaccine industry is among the first responders to pandemic influenza threats, and as a result, manufacturers need to have reliable pandemic plans that need to be exercised to ensure effective performance under pandemic conditions. Vaccine development is challenging, and the ability to produce seasonal influenza vaccine is not the only preparedness activity for a pandemic response. Mansoura highlighted the importance of testing and evaluating innovative technologies, strategies, and policies (e.g., biosafety assessment, permitting, contracting) for continued improvement. With each practice round, she said, we understand more about the nature of this dynamic threat. She advocated for the need to expand the strain-specific knowledge base and develop regulatory science to integrate new technologies.
Pre-Pandemic Versus Pandemic Development
David Vaughn, head of External Research and Development at GSK Vaccines described his company’s experience with the H5N1 and H1N1 influenza outbreaks. The GSK AS03 adjuvanted H5N1 vaccine was licensed in the United States in November 2013, but the usual economic incentives that typically encourage companies to develop influenza vaccines did not apply for a vaccine to address the virulent strain of H5N1, Vaughn said. In this case, GSK received most of its external funding from BARDA, with additional funding from the Canadian and Japanese governments. Due to concerns about an impending H5N1 pandemic, GSK got off to a very fast start. The contract with BARDA was signed in January 2007, and the pre-biologics license application (pre-BLA) meeting with the FDA took place in September 2008. However, when H1N1 influenza emerged in 2009, focusing on an H5N1 vaccine became difficult because all attention had shifted to the crisis at hand. Instead of submitting the BLA in 2009 as planned, it was not
submitted until 2012. Because of the pre-pandemic development of the H5N1 vaccine, Vaughn noted there was “the luxury” of extending the timelines, as there was no current pandemic demanding the final product. In this case the BLA was ultimately submitted, but he cautioned that, in some cases, a project is never seen to completion without the immediate demand. Vaughn also stressed the importance of considering pediatric development from the onset.
Increasing Capabilities Between Pandemics
Looking beyond H5N1 and H1N1 influenza, Vaughn said there is a need to improve the overall response to pandemics and ways to address interpandemic drift. In this regard, GSK is striving to increase its capabilities in research, development, and manufacturing in order to respond to a number of biosecurity threats. GSK has entered into unique public–private partnerships with the Defense Threat Reduction Agency and BARDA for antibacterial product development. Through recent acquisitions, GSK has also gained new platform technologies that may facilitate a faster response, both in terms of development and production. Vaughn noted that any plug-and-play rapid technology needs to be repeatedly successful with a variety of disease threats so that regulatory authorities and governments can make decisions in urgent situations to deploy vaccines under an EUA with little or no clinical data.
To enhance national preparedness, GSK proposes to embed MCM activities within the pharmaceutical industry’s research and development capabilities, Vaughn said. One of the best ways to ensure that technologies are scalable is to start the development process within the final manufacturer from beginning to end. Funding rapid response technologies will depend on multiyear federal appropriations to allow for continued pharmaceutical development and prevent the delay or termination of viable projects once they have been started. Vaughn concurred with others that current contracting approaches can be complex and time consuming, especially when programmatic responsibilities fall within one company and financial responsibilities another. Contracting hurdles have been overcome quickly in exceptional circumstances, he acknowledged, but for these efforts to be sustainable, streamlined contracting and integrating MCM activities into a company’s portfolio should be the norm, not an exception.
DEVELOPING AND SUSTAINING NEW
VACCINE PLATFORMS
The market for seasonal influenza vaccine is a large-volume, low-margin market, Pavia of the University of Utah said, which does not particularly encourage innovation. Larger companies that already have a substantial share of the seasonal influenza vaccine market face challenges in moving to new platforms. Mansoura said the ability to leverage a licensed manufacturing platform in a licensed facility provides an enormous enhancement to current capabilities in responding to threats. For example, synthetic vaccine seed technology allows for rapid progress from viral sequence to vaccine manufacturing within days. While the science and technology have advanced, key rate-limiting steps in the process include permitting, funding, and intellectual property issues. The application and review process for the funding of new programs can take 1 year. In addition, program focus and availability of government funding can shift from year to year. If establishing rapid response capabilities to make vaccines quickly is a government priority, then it should be stated and funded as such, Mansoura argued. But there are no signals from government to industry leadership of such a commitment. The opportunity costs for large companies are significant, and for smaller companies, the investor community will not be supportive of these endeavors if government is the only partner, making any sustained progress difficult to achieve.
Manon Cox of Protein Sciences Corporation said that BARDA invested significantly in her company’s recombinant influenza vaccine platform because it could potentially shave 6 to 12 weeks off the delivery timeline for an influenza vaccine. However, she noted that one of the challenges after achieving product licensure is successful commerciallization. Existing players aggressively defend the sizable seasonal influenza market, creating a barrier for smaller newcomers to establish themselves in the market—even with innovative platforms. Cox encouraged government funders, such as BARDA, to consider how they can then help sustain the technology over time. Mansoura agreed that the seasonal influenza vaccine market is oversupplied and hypercompetitive compared with the pandemic influenza vaccine market that comes and goes. A combined market between seasonal and pandemic influenza vaccine is needed, she said. Government officials are limited in what they can do to incentivize industry without a market. At current funding
levels, BARDA cannot mount rapid, nimble responses for new pandemic threats under existing contracts, she said.
BARDA’s Platform Investments
Bright noted his encouragement by the amount of progress that has been made by BARDA’s partnerships with industry. BARDA had set out to implement the national pandemic influenza strategy in a stage-wise approach to build better vaccines that are available sooner. This was achieved by investing heavily in public–private partnerships to build the domestic manufacturing infrastructure and increase the capacity for egg-based vaccine production, which was the routine process at the time. BARDA then invested heavily in cell-based manufacturing, which does not rely on the availability of eggs and the idiosyncrasies of egg-based vaccine production. Next, BARDA invested in three different recombinant-based technologies, which are even faster because they do not rely on growing the virus itself. This process further clarified that the use of adjuvants offered an antigen-sparing approach to make more doses of vaccine available sooner. All of these platform technologies can be incorporated to enable a more rapid and effective response. A side benefit of the increased pandemic capacity has been the application of the new technologies to the seasonal influenza vaccine. Domestic manufacturers have been able to provide quadrivalent flu vaccines, high-dose vaccines, intradermal vaccines, and egg-free vaccines.
Although vaccine capacity has increased, there is still room for improvement in vaccine efficacy, which remains at about 50 percent. Bright explained that a new BARDA initiative will invest in the development of more effective influenza vaccines, with a goal of greater than 50 percent efficacy in all populations and broader immunity to overcome vaccine strain mismatch challenges. This process also has the potential to create a baseline readiness level for response to pandemic strains. It may also be possible to develop vaccines that afford longer duration of immunity. BARDA has demonstrated that capacity can be built, next-generation technologies developed and implemented, and end-to-end time reduced through interagency and industry partnerships. Bright hoped this would serve as a strong signal to industry and academic stakeholders to engage in another public–private partnership approach to improving PHEMCE.
FUNDING A BETTER INFLUENZA VACCINE
Bright pointed out that 2015 was the first year that BARDA received appropriated dollars from Congress. BARDA did receive significant supplemental funding in 2006 and 2009 to respond to the influenza pandemics, which has been supporting all of its programs until this year. He added, though, that the 2015 appropriation was much lower than what BARDA considers to be necessary to accomplish all of its goals. Developing a more effective influenza vaccine is going to cost nearly $1 billion, Bright said, and will likely entail having four to six different technologies in the portfolio. As previously discussed, a product typically takes 10 to 20 years to move from discovery to reaching FDA approval. This will require a very prudent, prioritized approach in partnership with industry. Cost-sharing approaches will be critical, and government agencies will need long-term support from Congress and others, he added. Mansoura said that because BARDA has shifted from multibillion dollar, multiyear supplemental budgets to an annual appropriation, it looks like an increase (i.e., the annual budget has gone from nothing to something). However, there is a lack of transparency regarding the financing needs, and it is not clear if the pandemic preparedness plan from a decade ago is the basis for the current appropriation. What is needed, Mansoura said, is a dialogue about what pandemic preparedness should look like in 2015. She added that there has to be an aggressive research and development program that will improve vaccines, but not at the expense of testing and sustaining the systems and infrastructure that need to be in place to produce these vaccines. BARDA budgets over the past 3 years have been 90 percent dedicated to a research and development program, and lack a lifecycle management plan.
Challenges in Government Funding
Phyllis Arthur, senior director for BIO, pointed out that funding for BARDA and preparedness has bipartisan support and has been a priority for the president. Yet, Pavia said that even with a highly effective economic analysis of the net present value of investment in new influenza vaccines, creating the political will to spend the tax dollars could be difficult. With a multilayered budgeting process, agencies can ask for money in the departmental budget, but that budget is edited by the Office of Management and Budget, and then by Congress. Those
who are best positioned by their subject-matter expertise to estimate need are not in control of the financial decision making to impact the final product. Pavia said the alternative is to use government as the facilitator or the convener of the process. Osterholm of CIDRAP noted that now is the time to enlist the support of the business community in pandemic preparedness, and not just government and pharmaceutical industry partners, as the private sector understands how pandemics can impact both their supply chains and the company’s workforce.
In the push to increase flu vaccination among the general public, public health and the media have created an environment where everyone thinks the current influenza vaccine sufficiently protects the public and does not need improvement, according to Osterholm. He shared that he received significant negative feedback on a published meta-analysis showing that current influenza vaccines were only moderately protective (Osterholm et al., 2012). He also suggested that there was systematic bias in the 2010 and 2011 Advisory Committee on Immunization Practices statements that favored the current influenza vaccines. He emphasized the need to take a step back and assess what is known about the current vaccine and explain why a better vaccine is needed—because with the current mindset that it is very effective and does not need improvement, investors and companies will not step forward to develop a new season flu vaccine. However, he did stress that the current vaccine method and annual international decision process are the best available, and even 10 percent protection is better than zero. Though, understanding the limitations and assessing what is truly needed will be an important step in moving forward.
A Manhattan-Like Project for a Better Influenza Vaccine
Osterholm referred to an increasing number of studies showing that annual influenza vaccination is leading to waning immunity. Those who are protected could have about 90 to 100 days of protection, and vaccination is now commonly started as early as August or September, meaning that during the winter months—peak times for flu season in many parts of the country—even those who are vaccinated will have limited levels of immunity. Participants discussed the progress on development of a universal influenza vaccine. One participant suggested that the knowledge has sufficiently advanced and the technology exists, but there has not been investment in taking a product through to clinical development. An additional advantage to a universal flu vaccine, as Cox
pointed out, would be an opportunity to evaluate the platform’s use for a range of other diseases instead of creating new and untested additional platforms. No resources have been allocated yet to evaluate the usefulness of platform technologies for other emerging viruses. Several participants likened the level of organization and commitment (including funding) needed for such an endeavor to that of the Manhattan Project.4 Katz of CDC said the key is to bring together different manufacturers, different intellectual property, and different platforms. The task is twofold: develop a broader-immunity vaccine for the long term that will provide longer-lasting, broader protection (perhaps also against a potentially pandemic subtype) and simultaneously address the limitations of current vaccines for the short term by developing new platforms for seasonal vaccines. These new platforms could then be sustained for broader MCM applications if they are sufficiently resourced.
This annex contains technical details from the
speakers’ presentations related to the development of
products targeting pandemic influenza.
Jacqueline Katz
Deputy Director (acting) of the Influenza Division at CDC
To implement the tool, she explained, subject-matter experts score influenza viruses on 10 elements (Trock et al., 2012). The elements are grouped into properties of the virus, attributes of the population, and the ecology and epidemiology of the virus (see Figure 5-1). The scores for each of the 10 criteria are then weighted by the significance of each to virus emergence and public health impact. Each reviewer also provides a confidence score to address uncertainty. Composite scores are then used to rank and compare viruses in terms of potential pandemic risk.
___________________
4The Manhattan Project is the unofficial designation for the former U.S. War Department’s secret program, organized in 1942, to explore the isolation of radioactive isotopes and the production of an atomic bomb. Initial research was conducted at Columbia University in New York City, New York.
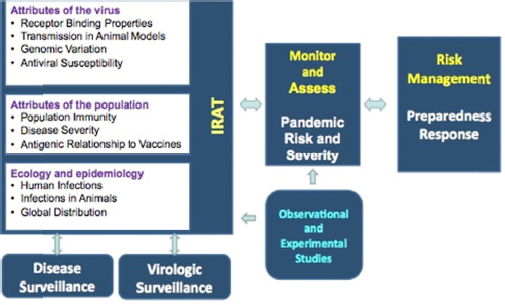
FIGURE 5-1 Influenza risk assessment and management using the CDC Influenza Risk Assessment Tool (IRAT).
SOURCE: Katz presentation, March 26, 2015.
Rick Bright
Acting Director of the Influenza Division at BARDA
Applying IRAT to Pandemic Scenarios
To illustrate a measured response, Bright described two pandemic scenarios involving a high pathogenicity avian influenza and a high pathogenicity influenza in special populations. The former would have a rapid, high peak and then ebb, and the latter would have a slower, gradual, lower peak, and then ebb (see Figure 5-2). It takes 24 to 26 weeks to make and release vaccines using current influenza vaccine manufacturing technologies and production processes. With no preparedness, therefore, the vaccine would become available only as the pandemic ebbs in both scenarios. If, by using IRAT and other available tools, an emerging virus is perceived to pose some risk, a decision could be made to
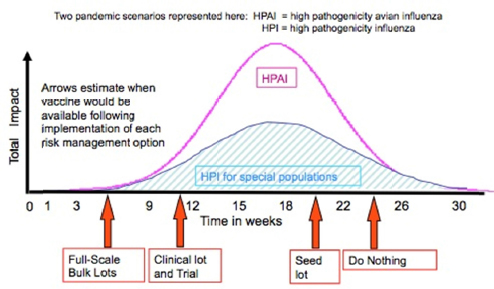
FIGURE 5-2 Pre-pandemic influenza vaccine availability by risk management option.
SOURCE: Bright presentation, March 26, 2015.
have CDC or manufacturers produce a seed lot of vaccine, thereby shaving 2 to 3 weeks off of a response time. If the risk assessment deems the threat to be higher, a decision could be made to produce a clinical lot of vaccine and conduct a clinical study, collecting valuable data (e.g., dosage, adjuvant) that could shave 12 to 14 weeks off the time to respond to a pandemic. If the virus is assessed to be very high risk, a full-scale bulk lot could be manufactured and stored just short of finishing and filling the vaccine, and available vaccine would be ready in several weeks.
David Vaughn
Head of External Research and Development at GSK Vaccines
Vaughn explained that the H1N1 vaccine development experience was very different from the H5N1 experience, due to the extent of the outbreak and immediate need for MCMs. While the H5N1 development process had the “luxury” of extending timelines, H1N1 vaccine development needed to be done in a timely manner. Importantly though, there was a global market for an H1N1 vaccine, giving some prediction of a positive return on investment. In the summer and fall of 2009, GSK initiated 28 clinical trials with funding from BARDA for the H1N1 vaccine. Three of those trials evaluated the use of an adjuvant in case it was needed in the United States to respond to that pandemic. The GSK
H5N1 vaccine requires an adjuvant, but for the H1N1 vaccine, the use of an adjuvant is optional. GSK took a global approach, by prioritizing the importance of vaccine coverage, and the use of adjuvants such as ASO3 or MF59 with the H1N1 vaccine allowed for the vaccination of more people in less time. Vaughn noted that GSK conducted an efficacy trial of AS03 in 6,000 children, and the data showed that the use of an adjuvant paired with the antigen increased the protective efficacy of the vaccine by 77 percent over two doses of antigen alone (Nolan et al., 2014). Because of some adverse events reported following the H1N1 adjuvant vaccination of Pandemrix in Europe in 2009, and a link to increased incidence of narcolepsy in children, public concern surrounding the use of adjuvants in flu vaccines also exists.5 Largely because of this, it would not have been possible to license an adjuvanted H1N1 vaccine in the United States in fall 2009, Vaughn said, as no approved flu vaccines in the United States then or now contain an adjuvant. However, as of November 2013, an H5N1 vaccine has been licensed for use in the SNS, and he suggested that, in the future, there may be greater public acceptance of adjuvants if they are needed for an influenza pandemic—recognizing their advantages in treating more people in a shorter time.
___________________
5For more on the adverse events linked to the Pandemrix vaccination, see http://www.cdc.gov/vaccinesafety/Concerns/h1n1_narcolepsy_pandemrix.html (accessed August 14, 2015).
















