3
Environmental Processes, Behavior,
and Toxicity of Diluted Bitumen
This chapter is concerned with what happens to crude oil after it is released from a pipeline, and with the potential environmental and ecological consequences of that release. It thus considers the chemical and physical processes affecting the oil and its residues, the resulting behaviors that manifest across various environmental settings, and the toxicity of the spilled and eventual residual oil. The discussion further distinguishes ways in which the behavior of diluted bitumen is similar to or distinct from that of the light and medium crude oils that are commonly transported in U.S. pipelines.
As mentioned in the previous chapter, crude oil released to the environment experiences a host of chemical and physical changes—processes collectively referred to as “weathering.” For the purposes of this chapter, weathering processes are divided into three categories based on the nature of the chemical and physical effects on the oil. Chemical processes include photooxidation and biodegradation and cause alteration to molecular structures through the cleavage and formation of covalent bonds, the linkages between atoms. Physical-chemical partitioning processes include evaporation and dissolution. These act without changing molecule structures and partition material, for example, between the atmosphere and a liquid phase. Physical processes include spreading, dispersion, emul-
sification, adhesion, and sedimentation. All of these change the physical properties and behavior of the oil but do not always partition it between phases or change its molecular structure.
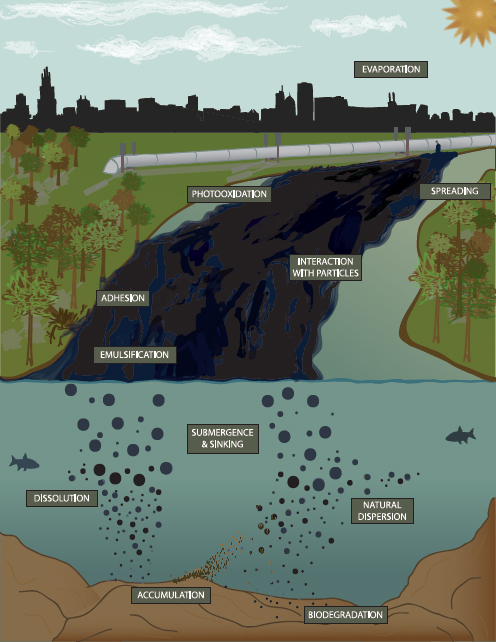
Processes occurring for a crude oil spill on water are outlined in Figure 3-1. The details of each process are discussed in this section. Given the statement of task, the focus is on the processes most relevant to oil spilled in subaerial and aquatic continental and coastal environments, including groundwater, lakes, estuaries, and streams of all sizes. By tracing the flow of the oil, where the origin of the spill represents day 0 of the spill, a general sequence of processes that occur can be summarized.
Although many processes act on spilled oil, few processes lead to chemical decomposition. The two processes of greatest relevance to oil spills in the environment are photochemical oxidation and biodegradation. These processes tend to occur slowly over a period of weeks to years and represent the breakdown of oil at the molecular level.
Photooxidation
Photochemical processes result from exposure of spilled oil to sunlight, leading to cleavage and formation of covalent bonds. Oxygen is typically incorporated into the products and thus the term photooxidation is commonly used. These oxidized products include both carbon dioxide and other oxygenated compounds.32 Typically, aromatic hydrocarbons are transformed more rapidly than alkanes,33 thereby increasing the relative abundance of resins and asphaltenes in the residual oil.32,33c In one set of laboratory experiments, the photooxidation of crude oil in fresh water under direct ultraviolet irradiation showed oxidation of 5% of the branched alkanes, 9% of the linear alkanes, and 37% of the aromatic hydrocarbons.34
Although photooxidation can be important when spills receive intense solar exposure, the photooxidation of diluted bitumen in the environment has hardly been studied and its role in the weathering of spilled diluted bitumen is not well understood. Notably, one potential outcome of photooxidation of crude oils is the production of persistent molecules in the environment32 containing carboxylic acids and alcohols, which may be soluble in water. Thus, these photoproducts may be transported in surface waters or groundwater.35
Photochemical enhancement of toxicity has been demonstrated for some polycyclic aromatic hydrocarbons (PAHs) via photomodification and photosensitization. In one study,36 organisms exposed to PAHs from
spilled oil experienced up to a 48-fold greater toxicity when illuminated with natural sunlight instead of standard laboratory light. This mechanism of toxicity is important for early-life-stage and translucent organisms that often accumulate PAHs in their tissues and inhabit surface waters.
Biodegradation
Biodegradation is a process by which living organisms, mainly bacteria, degrade hydrocarbons.37 Biodegradation can occur either aerobically or anaerobically, with aerobic processes typically occurring more rapidly and extensively. Biodegradation is accelerated in the presence of abundant oxygen and nutrients,38 moderate temperature and salinity, and reasonable oil-water interfacial surface area.39 For diluted bitumen, the extent of biodegradation depends on a combination of environmental factors, the proportions of bitumen and diluent, the nature of the diluent, and how fast the diluent is lost to evaporation. Since the deposits from which bitumen is extracted are themselves residues remaining after extensive anaerobic biodegradation, a spill of diluted bitumen may be less susceptible to biodegradation than a comparable spill of light or medium crude oil. However, biodegradation of diluted bitumen in environments containing aerobic bacteria and nutrients warrants further study.40
The main classes of crude oil components highlighted in Chapter 2 (see Figure 2-1)—saturates, aromatics, resins, and asphaltenes—provide a useful framework for understanding the relevance of biodegradation to the environmental fate of diluted bitumen. There are no quantitative field studies on the biodegradation of spills of diluted bitumen, but saturates and aromatics are expected to biodegrade within weeks to years.
Conversely, resins and asphaltenes in the bitumen are expected to remain recalcitrant for a longer time.
The U.S. Environmental Protection Agency (USEPA) studied short-term biodegradation in the laboratory using residual oil in sediment from the Kalamazoo River that was collected 19-20 months after a diluted bitumen spill.41 Over 28 days of aerobic incubation of sediment slurries with inorganic nutrients added, about 25% of the total petroleum hydrocarbons degraded, mostly in the first 14 days. The decreasing rate of biodegradation over the 28-day period suggested that the majority of the spilled oil would not degrade over time scales of at least a few months in spite of the experiment’s favorable conditions for bacterial activity.
Physical-Chemical Partitioning Processes
Once crude oil is spilled from a transmission pipeline, its composition can be affected by evaporation of volatile compounds and aqueous disso-
lution of water-soluble compounds. These processes tend to occur rapidly and strongly impact the composition and behavior of residual spilled oil.
Evaporation
Following a spill that brings oil into contact with the atmosphere, light components will evaporate at relative rates that depend on their volatility. As a result, compounds with greater volatility tend to evaporate from oil more rapidly than those with lesser volatility. This relative relationship tends to hold across various environments but absolute rates vary substantially based on concentrations of the volatile compounds in the oil and on ambient conditions including exposed surface area and volume; temperature of the oil, water, and air; and velocities of the wind current.42 Fingas43 has argued that the importance of the wind speed is moderated by the fact that the supply of hydrocarbon molecules to the oil-atmosphere interface is often a limiting step for evaporation.
For diluted bitumen, as with other crude oils, evaporation of light components can occur readily. The relatively light natural-gas condensates often used as the diluent in diluted bitumen are particularly volatile. The loss of volatiles thus leads to a residue strongly resembling the original bitumen, and this is a key behavior that distinguishes diluted bitumen from other commonly transported crude oils. As noted in Chapter 2, such processes increase both density and adhesion of the residual oil. The relationship between density and submergence is considered further here, whereas adhesion is considered in the context of physical processes.
The increase in density that occurs with evaporative loss of the diluent increases the likelihood that the residual oil will submerge beneath the water surface and potentially sink to the bottom.8,9b The rate at which density increases will depend on the composition of the diluted bitumen and especially on the nature of the diluent, but significant density increases have been observed to occur over in the first 1-2 weeks of a spill. Diluted bitumen with relatively high proportions of light and heavy hydrocarbons and a paucity of compounds in the C15-C25 range, such as Access Western Blend (AWB), Christina Lake, and Borealis Heavy Blend, are expected to achieve a higher density more rapidly with evaporation.
As evidence, King et al.9b conducted oil weathering studies in an experimental tank placed outside in Dartmouth, Nova Scotia, Canada (Figure 3-2). As the curves fit to the experimental data in Figure 3-2 show, within 13 days, the density of residues derived from Access Western Blend exceeded that of fresh water, and the oil sank until reaching an underlying layer of saline water. The maximum density of AWB (1.008 g/cm3) is greater than that of the density of weathered residues of Cold Lake Blend (1.0014 g/cm3) and it takes less time for AWB to approach its final density
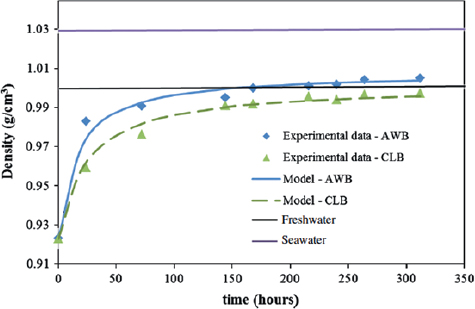
FIGURE 3-2 Observed increases in density for two diluted bitumen (AWB, Access Western Blend; CLB, Cold Lake Blend) added to a flume tank containing seawater, compared to the typical densities of fresh water and seawater. Curved lines show model fits to the observations.
SOURCE: King, et al.9b
in comparison with CLB. The time difference could be important when responding to an oil spill, where a difference of 12 hours could result in submergence of the oil.
Dissolution
If the spilled oil is in contact with water, components that are at least slightly soluble in water will be lost by dissolution. For diluted bitumen, the most soluble components originate from the diluent and, because of their volatility, also tend to be readily lost by evaporation. Therefore, unless the oil propagated or spread under water for an extended time, such as in the Deepwater Horizon oil spill,44 evaporative losses are expected to be larger than dissolution losses. However, evaporative losses may be slower than dissolution if a spill spread under the ice in an ice-covered lake or river. Also, dissolution may become important for diluted bitumen that has percolated to or been released beneath the water table.
Oil released to the environment may experience changes due to a host of physical processes including spreading, dispersion, emulsification, adhesion, and sedimentation. While these processes do not affect the molecular composition of oil directly, they do result in complex, environment-dependent interactions with the chemical and physical-chemical partitioning processes.
Spreading
On land, spreading of spilled oil is often limited, but when oil reaches a water surface it starts to spread immediately.45 Unless constrained, the oil will continue to spread out into a thin film, or slick, due mostly to interfacial tension.45 Spreading rapidly increases the footprint of oil in the environment and can make recovery efforts more difficult, but it also makes the oil more exposed to photooxidation and evaporation.
Small amounts of crude oil can spread into a very thin layer, or “sheen,” that is readily visible. Such sheening represents an impairment of water quality that can determine the degree of oil recovery that is required, as happened in the case of the spill in Marshall, Michigan (Box 3-1, Figure 3-3). Submerged or sunken oil residues, mostly out of sight, can still serve as a source of a sheen. The effect can continue for long periods, either spontaneously in response to temperature and water-level changes, or as a result of disturbance of the sediments by animals, boats, etc.
Dispersion
In the context of oil spills, dispersion refers to the entrainment of oil droplets in the water column. The extent of oil dispersion depends on the interfacial tension between oil and water, oil viscosity, and the mixing energy that may be driven by wind, currents, or tides.46 The interfacial tension between the oil and water does not vary widely among oil types. The range is typically less than twofold. When applied, chemical dispersants can decrease interfacial tension by 10- to 200-fold,47 allowing a greater proportion of oil to disperse into the water column. The mixing energy varies across environments as well as over space and time in a particular environment (e.g., rivers passing through dams or cataracts, and windy versus calm weather on lakes and coastal marine waters).
The droplet size distribution (DSD) of oil dispersed in water plays an important role in the behavior of oil in the aquatic environment. Larger droplets are more buoyant than smaller droplets and thus rise to the water surface, regardless of whether they were released underwater or released
BOX 3-1
Marshall, Michigan: Enbridge
The largest release of diluted bitumen into the environment occurred in July 2010 when the Line 6B pipeline operated by Enbridge Energy Partners LLC ruptured and released an estimated 843,000 gallons of oil into a tributary of the Kalamazoo River near Marshall, Michigan.12, 52 This spill is recognized as one of the largest, and most costly, inland spills in North America, with estimated costs exceeding $1 billion.
Oil flowed a couple of miles down the tributary and entered the Kalamazoo River, ultimately affecting about 40 miles of stream and river channels. The release occurred when the river was at flood stage and had temporarily inundated its floodplain; shortly thereafter, falling water levels left oil stranded on vegetation and soils up to about a meter above the normal summer river level. The river carries a lower suspended sediment load (i.e., less turbid) than most U.S. rivers and is not particularly turbulent because it has a low elevational gradient.
The U.S. Environmental Protection Agency (USEPA) quickly assumed control of the emergency response under the National Contingency Plan (NCP) and worked with Enbridge and a host of federal, state, and local government agencies.12 The USEPA remained in this function through 2014, an exceptionally protracted response period. The Michigan Department of Environmental Quality is still engaged in remediation as of 2015.
Initial response focused on capturing and collecting floating oil using conventional techniques such as conventional and sorbent boom, and the majority of the oil was recovered as floating oil or deposits on land, including from the wetland at the source of the release. A major wildlife rehabilitation effort commenced, ultimately cleaning and releasing over 3,000 turtles as well as some mammals and birds. Visible oil on floodplain and riparian vegetation was removed, mostly manually.
Within a few weeks it became apparent that significant amounts of the oil had sunk to the bottom of the river. Recovery of sunken oil increasingly became the focus of response efforts after the initial autumn, although some heavily oiled islands and floodplain areas still required excavation. Oil accumulated on the bot-
on the water surface and entrained into the water column by turbulence. In the Deepwater Horizon spill in the Gulf of Mexico, where the oil was released at depth, large droplets (> 1.0 mm) rose almost vertically and reached the surface within hours,44c,48 while small droplets rose more slowly or became permanently entrained.49 Studies of surface oil slicks50 predicted rapid transport of large oil droplets and slow transport of smaller droplets, causing the formation of a comet-shaped oil slick on the water surface. The DSD affects not only the transport but also the fate and toxicity of spilled oil. Increasing the proportion of small droplets results in an increase in the surface area per unit mass of oil, which enhances the
tom wherever the river flow slowed down, but particularly behind three areas with manmade dams, and in some oxbows (abandoned river channels).
Detection and quantification of the sunken oil was challenging.9a Laboratory measurements of total petroleum hydrocarbons suffered from interferences by natural organic matter. Sunken oil was mapped out using a method called poling, in which the sediments were disturbed with a disk on the end of a pole and the amount of sheen and floating globules that appeared on the surface was estimated. Poling showed that the oil tended to accumulate in areas of slow or no flow and fine, often highly organic sediment. Chemical fingerprinting, requiring an expensive set of measurements, was employed selectively to establish that the sunken oil and the sheen it produced were in fact from the pipeline release and not a legacy from other pollution sources.
With the exception of dredging, proven techniques for recovery of this type of sunken oil in a riverine setting were lacking. Sediment agitation and collection of the resultant sheen was employed extensively in an attempt to recover the sunken oil without sediment removal, but eventually that approach was shown to be inadequate and dredging was conducted in the most extensive contaminated areas. In the later stages of the cleanup, a net environmental benefits analysis was conducted to examine the distribution of sunken oil, as indicated by poling, and to determine whether further recovery attempts were justified against the environmental disturbance of recovery operations.12 Many small areas of sheen-producing sediments were left alone but continue to be monitored. The USEPA estimates that as much as 80,000 gallons of oil may remain in the sediments, but major recovery efforts ended in 2014.
The toxicity of the spilled oil to fish and wildlife received some study. A histopathological study of fishes conducted shortly after the spill showed evidence for toxic effects, whereas short-term bioassays with invertebrates conducted in later years were less conclusive. Sampling of fishes and benthic invertebrates indicated that communities returned to normal after the first year, although the apparent decline in the first year was clearest for invertebrates, and it is difficult to distinguish the relative roles of direct toxicity of the spilled oil from the effects of cleanup activities (particularly sediment disturbance).
dissolution of hydrocarbons in the water column44b and their potential for bioaccumulation via transport across biological membranes.
The time for a slick to break up and reach a stable droplet size distribution (the equilibrium DSD) is also an important factor because the delay enables other processes to act on the oil in a differential way. For example, turbulence could change during this time, leading toward another equilibrium DSD, or particles of a certain size might preferentially interact with mineral or organic particles, thus forming oil-particle aggregates. Figure 3-4 shows how the median droplet size varies with time for oils with various viscosities subjected to agitation at an energy dissipation

FIGURE 3-3 Sheening in the Kalamazoo River, the site of a diluted bitumen spill (see Box 3-1). Left: Sheen emanating from an island after a modest rise in river level (the spill occurred at a higher river level). Right: Sheen generated from sunken oil upon disturbance of the sediments. Photo credit: USEPA.
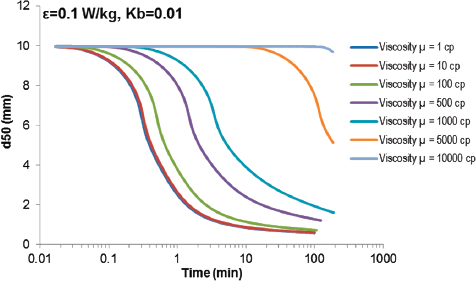
FIGURE 3-4 Evolution of the median droplet diameter (d50) with time as a function of the oil viscosity, based on model simulations.53 High-viscosity oils would require hours to disperse into an equilibrium droplet size distribution.
rate (ε, W/kg) representative of spilling breaking waves. Even under these conditions, which can be expected to be rare in inland waters, but more likely in a coastal environment, oils with viscosities >10,000 cP require many hours to approach equilibrium, leaving ample opportunity for other processes to act.
Emulsification
Emulsification is the process by which one liquid is dispersed into another one in the form of small droplets.51 To be called an emulsion, the product must have some physical stability; otherwise the process is called water uptake rather than emulsification. Water droplets can remain in an emulsified oil layer in a stable form and the resulting material, sometimes called mousse or chocolate mousse because of its appearance, has properties differing strongly from those of the parent oil.
Water can be present in oil in five distinct ways. First, some oils contain about 1% water as soluble water. This water does not significantly change the physical or chemical properties of the oil. Second, oil can contain water droplets without forming a stable emulsion. They are formed when water droplets are incorporated into oil by wave action and the mixture is not viscous enough to prevent droplets from separating from the oil.
Mesostable emulsions represent the third way that water can be present in oil and are formed when the small droplets of water are stabilized by a combination of the viscosity of the oil and the interfacial action of asphaltenes and resins. The viscosities of mesostable emulsions are 20-80 times higher than that of the starting oil. These emulsions generally break down within a few days into oil and water or sometimes into water, oil, and emulsion remnants.51 Mesostable emulsions are viscous liquids that are reddish brown in color.
The fourth way that water exists in oil is in the form of stable emulsions, which form in a way similar to mesostable emulsions and persist because the concentration of asphaltenes and resins is high enough to stabilize the oil-water interface. The viscosity of stable emulsions is 800-1,000 times higher than that of the starting oil. This emulsion will remain stable for weeks and even months after formation. Stable emulsions are reddish brown in color and appear to be nearly solid. Because of their high viscosity and near solidity, these emulsions do not spread and tend to remain in lumps or mats on water or shore.
The fifth way that oil can contain water is by viscosity entrainment. If the viscosity of the oil is such that droplets can penetrate, but will only slowly migrate downward, the oil can contain about 30% to 40% water as long as it is in a turbulent water body. Once the water calms or the oil is removed, the entrained water slowly drains. Typically most of the water will be gone before about 2 days.
The formation of emulsions is an important event in an oil spill. First, and most importantly, emulsification substantially increases the actual volume of the spill. Emulsions of all types initially contain about 50% to 70% water and thus, when emulsions are formed, the volume of the spill can be more than tripled. Even more significantly, the viscosity of the oil
increases by as much as 1,000 times, depending on the type of emulsion formed. For example, oil that has the viscosity of a motor oil can triple in volume and become almost solid through the process of emulsification. These increases in volume and viscosity make cleanup operations more difficult. Oil in stable emulsions is difficult or impossible to disperse, to recover with skimmers, or to burn. Mesostable emulsions are relatively easy to break down, whereas stable emulsions may take months or years to break down naturally. Emulsion formation also changes the fate of the oil. When oil forms stable or mesostable emulsions, evaporation slows considerably. Biodegradation also slows. The dissolution of soluble components from oil may also cease once emulsification has occurred.
Adhesion
Adhesion was considered in Chapter 2 as a physical property of oil, and diluted bitumen tends to be more adhesive than other commonly transported crude oils. In this section, adhesion is further considered in the context of weathering, as an environmental process that changes the physical properties and behavior of oil.
When diluted bitumen is spilled and the diluent evaporates, the residual oil will increasingly adhere to surfaces. Many inland and coastal waters contain submerged, floating, and emergent aquatic vegetation and debris that would provide surfaces for adhesion of bitumen. In addition, various animals may also become coated with oil, including turtles, amphibians, insects, and mammalian species (see Figure 3-5). Residual bitumen can be difficult to remove from tree trunks and other biota, rocks, concrete, and manufactured surfaces (Figure 3-5). Strongly adhesive behavior is not unique to bitumen, as some heavy oils with high contents of resins and asphaltenes can also become highly adhesive following weathering. However, diluted bitumen is unique in terms of the rate at which its physical properties can begin to revert back to those of bitumen, due to the evaporative loss of volatile hydrocarbons that comprise the diluent. The emergence of strong adhesion following the evaporative loss of volatile components can impede recovery efforts and, as discussed further below, is expected to increase the tendency of the residue to adhere to particulate matter and sink.
Sedimentation
In the context of this report, sedimentation refers to oil that sinks and comes to rest on the underlying bed in an aquatic setting. Sedimentation may occur by several routes, including an increase in density of the oil through physical-chemical partitioning or chemical processes, the adhe-

FIGURE 3-5 Biota coated in adhesive oil: (a) Oil that coated the shoreline after the Marshall spill into the Kalamazoo River in Michigan (Box 3-1) as the river level fell in August 2010. Photo credit: S. Hamilton. (b) Residual bitumen oil on a tree along the Kalamazoo River in September 2015, over 5 years after the spill. Photo credit: S. Hamilton. (c) Oiled map turtle from the Kalamazoo River, collected shortly after the spill. Photo credit: Michigan Department of Natural Resources. (d) Ladybird beetle collected at a natural hydrocarbon seep off Coal Oil Point, Santa Barbara, CA. Like bitumen, the seep oil is highly adhesive because of its high resin and asphaltene content. Photo credit: D. Valentine and R. Harwood.
sion of entrained (dispersed) droplets of oil to the bed, and formation of oil-particle aggregates (OPAs) of sufficient density to submerge. This section focuses on OPAs, although the high density of weathered diluted bitumen is expected to increase sedimentation by all three routes, relative to commonly transported crude oils.
Aggregation of oil with natural particulate matter can cause submergence of an initially floating oil.9a There are two major types of OPAs: oil droplets coated by small particles54 and oil trapped within or adhering to large particles (Figure 3-6). The first type is more common and has
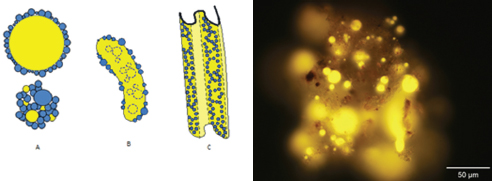
FIGURE 3-6 Left: Types of oil-particle aggregates (OPAs): A, single and multiple oil droplets (yellow) aggregated with natural particles (blue); B, solid aggregate of a large, usually elongated mass of oil with interior particles (dashed blue circles); and C, flake aggregate of thin layers of clay that incorporate oil and fold up (c.f. Stoffyn-Egli and Lee55 and Fitzpatrick, et al56). Right: Kalamazoo River sediment spiked with weathered source oil from the Marhsall, MI spill after 48 hours, under ultraviolet epifluorescence microscopy at 320× magnification.57 Images from Fitzpatrick et al.9a
been studied in some detail. The particles on the oil droplet surfaces prevent coalescence with other droplets, thus stabilizing the suspension. The OPAs tend to separate from the oil droplets from which they form, and then they may sink because the aggregate of residual oil (density near, or slightly less than, that of water) with inorganic particles (density at least twice that of water) yields an aggregate particle density that is neutrally or negatively buoyant in water.
The formation of aggregates depends on the viscosity of the oil droplet, the surface areas and mineralogy of the particles, and the salinity of the water.58 Salinity enhances the formation of OPAs, but the onset of aggregation is very steep, becoming important at salinities as low as 1/200 that of seawater.54b, 58b The extent of adsorption also depends on the surface properties of the sediment particles and other particles such as algal cells. The relative quantity of residual oil produced by weathering is from 5- to 50-fold greater than the quantity produced by weathering of equal volumes of light and medium crude oils.
In quiescent water, the density of the OPA determines whether it remains at the surface or sinks to the bottom. However, when turbulence is present, the suspension of the OPA in the water is due to the balance between hydrodynamic forces that tend to lift the OPA and keep it in suspension versus the gravitational force on the OPA. This means that density alone does not determine the location of the OPA; its size and shape
are also important. A large OPA would be less prone to displacement by turbulence than a smaller one and, thus, tends to settle faster than a smaller OPA. Because viscous oils tend to break into large droplets,59 the OPAs resulting from these oils would tend to be large, and if their density is greater than that of the receiving water, they would settle to the bottom. This formation of large OPAs is a major distinction between diluted bitumen, which rapidly weathers to a viscous residue, and commonly transported crude oils.
Two recent laboratory studies dealt specifically with the formation and sedimentation of oil-particle aggregates in fresh water. The results of the first of these studies60 are fully consistent with the second, far more detailed report, by Waterman et al.,61 which focused specifically on the mechanisms underlying the submergence and deposition of diluted bitumen residues in the bed of the Kalamazoo River. Cold Lake Blend, a diluted bitumen, was added to pure water and agitated by bubbling air at 21ºC. The study found that it lost at most 17.4% of its mass and reached a density of 0.993 g/cm3, thus remaining buoyant.61 However, when the diluted bitumen was added to a mixture of Kalamazoo River water and sediment collected upstream from the spill site, the results were very different. Abundant OPAs formed quickly, on time scales of minutes to an hour, and included both inorganic and organic particles. Measured settling velocities ranged from 1 to 11 mm/s, with most being around 2 mm/s. This observation—that aggregates formed readily in a natural, fresh water—is not in conflict with laboratory studies showing that aggregates form slowly, if at all, in nonsaline water. The natural water is definitely “fresh,” but its content of ions derived from clays and soils is high enough to allow the formation of aggregates. Specifically, the electrical conductance of the water from the Kalamazoo River was 640 µS/cm, typical of North American river waters and indicating a salinity about 1/100 that of seawater.
Water temperature and salinity are important determinants of the propensity of residual bitumen to submerge. Short62 reviewed existing experimental studies of the submergence of weathered bitumen and concluded that the studies had not sufficiently considered the differential effects of temperature on the densities of bitumen products versus that of freshwater or seawater. The density of bitumen increases faster with decreasing temperature than does the density of water, and therefore a weathered bitumen may sink in cooler water while floating in warmer water, yet the studies did not investigate a range of temperatures. Furthermore, Short62 noted that where there is salinity stratification with fresh or brackish water overlying seawater, as is particularly common at freshwater inlets to coastal marine zones, submerged oil may accumulate at density interfaces beneath the surface. Such interfaces can be dynamic
under the influence of winds, tides, and changing freshwater discharge, presenting a particularly challenging situation for crude oil detection and recovery.
In sum, in a diluted bitumen spill subject to weathering, there is much more residue and its density is much closer to that of water, a combination that is likely to translate to enhanced OPA formation and oil submergence relative to commonly transported crude oils.
This section addresses how oil spills behave in and potentially impact specific environments, with particular attention to how spills of diluted bitumen may be similar to or distinct from those of other commonly transported crude oils. The toxicity of diluted bitumen is reviewed in a following section.
Water bodies merit particular attention. Pipelines traverse innumerable inland water bodies ranging from headwater streams to wetlands to larger rivers and lakes. Spills can enter water directly or indirectly via overland flow or transport in groundwater. These inland waters are immensely diverse. Once an oil spill contacts water bodies, it may be more difficult to recover, it can potentially be transported quickly from the site of release, it presents a hazard to aquatic life (see toxicity discussion below), and it may impair human uses of the water body.
The land-water interface often presents special challenges for oil spill cleanup. Water levels in lakes, rivers, and coastal zones vary under the influence of precipitation and runoff, dam operations, and/or tides. Accordingly, oil floating on the water can subsequently become stranded in intermittently flooded areas, from which it could be remobilized later. The land-water interface is often disproportionately important to its area, serving as habitat for fish and wildlife and providing recreational and aesthetic values for people.
Spills on Land and into Groundwater
When diluted bitumen or other crude oil is released onto land, or into soils beneath the ground surface, its movement in the ground depends on the soil type, oil viscosity, and the depth to the water table. Oil percolates deeper in coarse sediments, such as gravel, than in fine sediments, such as fine sand, silt, and clay. Extensive studies of a spill of crude oil at Bemidji, Minnesota (Box 3-2), have provided an in-depth understanding of the behavior of crude oil in a subsurface setting. If crude oil is sufficiently fluid to move in the subsurface, it would migrate downward until reaching the water table, where it will sit above the water and could
BOX 3-2
Groundwater Contamination in Bemidji, Minnesota
One of the most comprehensively studied sites for crude oil transport and fate in groundwater has been at Bemidji, Minnesota, where 10,700 barrels of crude oil, characterized as light to medium,67 spilled from a failed weld on a pipeline in August 1979. About 2,500 barrels seeped into the subsurface and could not be recovered. Remediation of this site remains a challenge today. In 2011, reviewing three decades of remediation and investigation, scientists from the U.S. Geological Survey wrote, “Considerable volumes of NAPL oil [non-aqueous-phase liquid oil] still remain in the subsurface despite 30 years of volatilization, dissolution, and biodegradation, and 5 years of pump-and-skim remediation.”68 The current approach for the contaminated Bemidji aquifer is to rely on natural attenuation, including both aerobic and anaerobic biodegradation by microorganisms.64
Some of the liquid oil reached the groundwater table, where it has floated on top of the water, while some was retained by sediments in the unsaturated zone. As depicted in the cross-section figure below, various processes have affected the amount and composition of the subsurface oil, including differential sorption to the soils and underlying glacial deposits, evaporation of volatile components into the soil air space, dissolution of the more soluble components into groundwater, and biodegradation. Natural attenuation by biodegradation has substantially reduced the amount of oil and slowed its movement relative to the underlying groundwater, even though rates are limited by the availability of oxygen and other oxidants.
The oil has gradually migrated in the direction of groundwater flow, yielding a plume of liquid oil, an associated vapor plume in the overlying unsaturated zone, and a plume of dissolved constituents that moves with the groundwater. The different zones in the figure below have distinct concentrations of petroleum contaminants, dissolved oxygen, and the products of microbial reactions that degrade the petroleum compounds. Research at the Bemidji site has provided a wealth of information on the rates and limitations of the microbial processes that biodegrade oil, and this information has been integrated into a mathematical model that informs decisions about remediation of subsurface oil spills.
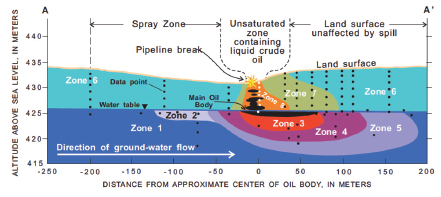
SOURCE: Delin et al.69
propagate horizontally along the groundwater flow gradient.63 In most instances, the underground migration of the free phase oil (or diluted bitumen) is limited, and many small crude oil spills onto land have been remediated by excavating the contaminated soils. In the case of diluted bitumen, if spilled oil is exposed to the atmosphere, the evaporation of the diluent and the light fractions increases the oil viscosity and thus limits oil migration in the ground. However, if not lost to evaporation, the light components of crude oil, especially those that are more soluble in water such as benzene, toluene, ethyl benzene, xylenes (BTEX), and naphthalenes, could migrate relatively easily in the aquifer, especially if the aquifer material contains only small amounts of organic matter. Examples of such materials include gravel and sand aquifers, typical of glacial outwash. For organic-rich soils, water-soluble oil components could be adsorbed onto sediments, depending on their water-to-octanol (KOW) partition coefficients.63 A concern with crude oil spills underground is the mobilization of trace metals such as arsenic, as has been observed at the Bemidji site in Minnesota.64
If oil were spilled into karst aquifers, where transmission of groundwater can be exceptionally rapid, its behavior might be expected to be comparable to that in surface waters. Rapid migration of the oil could pose a threat to water supplies for drinking or irrigation. However, to date there is no experience with diluted bitumen spills into karst aquifers.
The surface soils at the site of the release of diluted bitumen from a pipeline near Marshall, MI, as well as the downstream floodplains of the Kalamazoo River, were significantly contaminated (Box 3-1). At the release site, diluted bitumen saturated soils in a small wetland before overflowing into a creek. Large amounts of residual oil were deposited on downstream floodplains as river levels fell. Much adhered to vegetation and other surfaces as the diluent evaporated. Residual oil did not move long distances in groundwater, presumably because the groundwater flows to the river from the surroundings in almost all of the affected reaches.65 Had the flows been from the river to the alluvium, a common situation, groundwater contamination may have been more pervasive.
The ecological impacts of the oil deposited on land at the Marshall, MI spill were not studied in depth. Because remobilization of oil by subsequent floods was a major concern, oil-coated vegetation and soils were largely removed within months wherever oil deposits were visible. Little immediate mortality of wildlife was reported. Removal of oily vegetation and soils severely disturbed the riparian and floodplain ecosystems, even though this was done manually in all but the most heavily oiled locations, where excavation was necessary.
In terrestrial ecosystems, spills of diluted bitumen and of light or medium crude oils present similar challenges for cleanup. Contaminated
soils, vegetation, and man-made surfaces must be removed or cleaned. The most distinctive feature of diluted bitumen in this regard is its tendency to be strongly adhesive and to coat surfaces, and the difficulty of washing it off. Contact with the sticky bitumen by wildlife and people would increase exposure although toxicity implications are not clear given limited data (see Toxicity section, Chapter 3). The bitumen would be expected to be highly persistent due to its abundance and resistance to dissolution or biodegradation.
Inland lakes and reservoirs vary greatly in depth, exposure to wind and currents, suspended matter, and submerged and emergent aquatic vegetation, all of which potentially affect the behavior and impacts of oil spilled into these environments. Spilled oil may enter a lake or reservoir along a shoreline, or from tributaries. Submerged oil carried in suspension (including as OPAs) by streams and rivers may accumulate in depositional areas once the water enters a lake or reservoir, and this is particularly pertinent to spills of diluted bitumen because of their greater propensity to form OPAs, as discussed above. Lakes and reservoirs are often used as water supplies and for recreation, thus raising the possibility of risks to the public, and they provide important habitat for fish and wildlife.
The 2010 spill in Marshall, MI (Box 3-1) reached a reservoir known as Morrow Lake, and that location provides our only experience to date with a diluted bitumen spill in a reservoir. The extensive delta at the upper end of the reservoir, which begins 60 km downstream of the pipeline release, accumulated submerged oil. Sheening was observed during warm months for 3 years after the spill. A hydrodynamic model indicated the potential for resuspension of the sediment containing oil and its movement further into the reservoir.66 The most heavily oiled sediments had to be dredged in 2014.
The Great Lakes system of the U.S. and Canada has distinct characteristics that would affect the behavior and impacts of an oil spill. Transmission pipelines capable of transporting diluted bitumen products cross the Great Lakes system at two points: the Straits of Mackinac between Lake Michigan and Lake Huron,70 and the St. Clair River upstream of Detroit and Lake Erie. A release at either the Mackinac Straits or the St. Clair River would lead to movement of oil into the lakes. Additionally, pipelines cross many streams and rivers that flow short distances to either the southwestern shores of Lake Superior or the southern shores of Lake Michigan.
Currents can be complex in the Great Lakes, with currents in the Straits of Mackinac depending on relative water levels of Lakes Michigan
and Huron as well as on wind speed and direction. It could be very difficult to anticipate the movement of the spilled oil and to recover the oil, even at the surface, due to the expansive area and potential for strong wave action. Ice cover during winter could impede detection and recovery of spilled oil.
Pipelines often traverse wetlands, which are common landscapes in the U.S. in all but the arid West. Wetlands are highly variable ecosystems in terms of their hydrology (water sources, flow, and connectivity with water bodies) and ecology (vegetation and animal life). An oil release into a wetland may affect a limited area if the system is hydrologically isolated or has only seasonal flow. However, wetlands commonly occur along land-water interfaces of lakes, rivers, and coastal marine waters, and spilled oil could spread from wetlands into adjacent waters, or vice versa, if they are connected via surface water flow.
Wetlands are valued and protected because they provide important ecosystem services and can harbor rare or threatened plants and animals. Oil spills into wetlands present challenges because of the sensitivity of these ecosystems;71 for example, in Marshall, MI (Box 3-1), oil trapped in floodplain swamps and marshes had to be recovered manually by cutting vegetation and scraping soils wherever possible, and care had to be taken to not alter the natural hydrology (e.g., by building access roads).
Unless the water is deep enough that submergence of oil poses an issue, diluted bitumen and commonly transported crude oils pose many similar challenges following release in wetlands, but three factors specific to diluted bitumen can be mentioned. First, the amount of residual oil will be large compared to those produced by spills of conventional crude oils. Second, the increased level of adhesion of bitumen may complicate operations in a wetland environment. And third, diluted bitumen residues may persist longer in wetlands, as in other environments, because of their resistance to biodegradation.
Streams and rivers vary in several ways that are important for the transport and fate of oil released into them. Key factors include gradients and velocities of flow; the type and concentration of suspended particulate matter; the types and abundance of underwater algae and plants; the extents and types of zones in which oil may be deposited, such as sedimentation in impoundments and side channels; and varia-
tions in flow and water levels. Submerged oil carried by streams and rivers can continue to move downstream until reaching depositional zones in water bodies or floodplains, where it can settle in relatively extensive areas.
The transport of oil in rivers could be also affected by whether the reach is gaining or losing water by exchange with groundwater. Rivers may transport sediment (and associated crude oil) by wash load, suspended load, and bed load.72 Wash load consists of very fine particles that are relatively evenly distributed through the water column; examples include suspended clays and organic matter. The wash load may represent those particles that would interact with spilled diluted bitumen and influence its initial behavior. Knowledge of bed material composition does not allow one to predict wash load transport. Suspended load is the load that is suspended in the water column but still interacts with the bed. The interaction usually occurs at riffles causing the shear stress to increase and sediments to be suspended from the lee side of the ripples. Bed load has continuous contact with the bed and has a direct relation to the turbulence along the bottom in rivers. While both suspended and bed loads are commonly predicted using sediment transport models, the wash load is not predicted by these models, limiting the prediction of OPA formation in rivers. Furthermore, prediction requires a better understanding of which types of sediment are most likely aggregate with oil to form OPAs, and how those OPAs are likely to be transported in streams and rivers. The answers to these questions are likely to be different for diluted bitumen compared to other commonly transported crude oils.
Pipeline spills can readily reach coastal zones if they occur in tributaries or along estuaries, or at coastal processing facilities. Coastal zones in the U.S. where diluted bitumen spills could occur in the future as diluted bitumen pipelines and infrastructure expand include Anacortes in the Pacific Northwest, the Gulf Coast, and Portland, Maine. Each of these regions has distinct physical and ecological characteristics that would influence the fate and ecological effects of spilled diluted bitumen. Many oil spills have occurred in coastal areas29 and have included heavy refined oil products, though until recently the only one in North America that involved diluted bitumen occurred in Burnaby, British Columbia. The recent Refugio oil spill in California also entered a coastal area and all active cleanup for this spill ended August 31, 2015 (Box 3-3).
Estuaries occur at the interface of terrestrial and marine systems. Estuaries are characterized by periodic reversals of flow due to tides and
BOX 3-3
Santa Barbara, California: Refugio Pipeline Spill
On May 19, 2015, a pipeline rupture occurred near Santa Barbara, California, on Line 901, operated by Plains All American Pipeline L.P. The heavy crude oil was discharged from a heated transmission pipeline and comprised a blend of oils from four nearby platforms. Despite the chemical and physical differences between the heavy crude oil spilled at Refugio and diluted bitumen, several aspects of the event provide for a valuable point of comparison for this study, which led to a site visit from members of the study committee.
Unanticipated Complexities
The May 19, 2015, rupture to Line 901 occurred on the inland side of a four-lane highway during daylight hours, though an earthen berm paralleling the highway prevented motorists from seeing the oil. The discharged oil pooled in a local depression and then overflowed to a drainage culvert that ran under the berm. The oil flowed unseen to a second drainage culvert that ran underneath the freeway, to a drainage that led under a railway line, down a gulley to a cliff, and then to a sand and gravel beach from which it entered the ocean. The total discharge was estimated at 101,000 gallons, with 20,000 gallons estimated to have reached the ocean.
The partitioning of the discharge between land and ocean dictated response actions from both USEPA and the U.S. Coast Guard, and a joint command structure was established in which two federal On-Scene Coordinators, one from each agency, shared the lead responsibilities for the response. The bifurcation of discharge and the resulting structure of the Unified Command underscore the importance of response planning for diluted bitumen spills in a terrestrial setting that might subsequently affect lacustrine, riverine, and/or coastal marine environments. This incident further underscores the need for coordinated planning among federal agencies for response to diluted bitumen spills.
by the presence of freshwater-saltwater mixing zones. Large estuaries can be strongly affected by wind and waves. The interaction of fresh and saline waters in estuaries is important not only for physical transport of spilled crude oil but also because mixing of these waters often results in the flocculation and settling of clay particles. Where crude oil is associated with these particles, salinity gradients can be important in controlling the sinking rate.
Coastal zones are characterized by three-dimensional flows due to waves, longshore drift, and rip currents. Although there are numerous regional ocean models, their highest spatial resolution tends to be around
Coastal Zone Transport and Submerged Oil
The product spilled from Line 901 was a heavy crude oil and much of the mass floated at the sea surface. The heavy crude oil that reached the ocean was initially transported along the coast to the east, driven by longshore drift, and opposite to the direction of the prevailing offshore current. This transport gave way to heavy oiling of beaches extending several miles to the east of the rupture. Transport offshore by the prevailing current and then to the southeast caused oiling events and beach closures in Ventura and Los Angeles Counties, up to 100 miles away. Scientists identified submerged and sunken oil in near-shore reefs and kelp beds, near the most heavily oiled beaches. The submerged oil was presumably ballasted by mineral and organic material to which it had adhered while on the beach and in the surf zone. The documentation of submerged oil by scientists triggered response officials to conduct a formal assessment of submerged oil, the result of which was a finding that quantities were too low to warrant removal efforts.

Photo credit: D. Valentine
1.0 km, a scale that is too large to quantify the processes occurring in the coastal zone. While waves in coastal areas tend to bring the crude oil to the beach, there are situations where regional, longshore, and rip currents transport crude oil offshore and parallel to the shoreline and deposit it miles away from where it was released (Box 3-3).
One distinct characteristic of coastal waters (including estuaries) that affects the potential for crude oil to submerge is salinity, which ranges from fresh to oceanic levels and can fluctuate seasonally or daily due to tides and river discharge. Salinity affects the density of the water, and as noted earlier, OPAs that may sink in brackish water can remain in suspen-
sion in more saline water. Water flow in coastal zones is often complex and this extends to spatial and temporal patterns of salinity. Tides cause bidirectional flow of water in channelized areas and coastal currents are also commonly induced by waves. Wave action can be produced by local winds and also ocean swells. Freshwater inputs may lie above saltwater. Suspended particulate matter, composed of mixtures of inorganic sediment, detrital organic particles, and algae, also is spatially and temporally variable in coastal waters but often occurs at high concentrations compared to offshore waters. All of these features complicate the prediction of the behavior of an oil spill in these environments, including its propensity to form OPAs and become submerged.
There has been only one significant release of diluted bitumen into a coastal marine zone—the Burnaby Inlet in British Columbia (Box 3-4)—and in that case most of the spilled oil was recovered quickly. However, there is a great deal of experience with crude oil spills in coastal zones because of the high tanker, barge, pipeline, and refinery activities in close proximity. These have included spills where the crude oil became submerged or sank mostly as a result of oil-particle interactions.46a
Crude oil penetration and persistence in beaches (or river banks and bars) is affected by both crude oil properties and beach hydrodynamics. The mobility of crude oil in porous media such as sand is inversely proportional to its viscosity and, thus, high-viscosity oils (such as weathered diluted bitumen) tend to penetrate very little within the sediment. High adhesion of heavy crude and weathered diluted bitumen oils further reduces their mobility in porous media, making them as amenable to recovery as other crude oils.
In tidally influenced beaches, crude oil deposition is usually highest in the upper intertidal zone and decreases moving toward the lower intertidal zone. However, crude oil buried in the upper intertidal zone tends to weather faster than that in the lower intertidal zone due to high water exchange resulting from tidal action and often from local groundwater inputs that replenish oxygen and nutrients.73 For example, groundwater movement was found to be a major factor in the persistence of the Exxon Valdez oil in Alaska beaches.38b
BOX 3-4
Burnaby, British Columbia: Kinder Morgan
On July 24, 2007, approximately 1,400 barrels (58,800 gallons) of Albian heavy synthetic crude oil leaked from the Westridge Transfer Line in Burnaby, British Columbia. The spill resulted from an excavator bucket striking the pipeline during excavation for a new storm sewer line. The pipeline was operated by Kinder Morgan Canada and owned by Trans Mountain Pipeline L.P. The pipeline linked the Burnaby terminal to the Westridge Dock, where oil could be loaded to tankers.37b After the oil was spilled, it flowed in Burnaby’s storm sewer systems until it reached Burrard Inlet.37b In total, 11 houses were sprayed by the rupture, 50 properties were affected, 250 residents voluntarily left, and the Burrard Inlet’s marine environment and 1,200 m of shoreline were affected by the spill.37b
Cleanup took months and cost roughly $15 million and resulted in the recovery of approximately 1,321 barrels of oil.75 Responders used three distinct methods to recover the oil, based on the oil’s location. In residential areas, peat moss was used successfully to absorb oil on land. In storm sewers, oil in the storm sewers was vacuumed up. Much of the oil was collected in the pump station. And finally, in Burrard Inlet, booming was used to contain oil around the release points, skimmers and absorbent pads were used to remove oil, and tarballs, debris, and oiled macroalgae were manually removed.
To treat the oil that had adhered to the shoreline, responders successfully used the chemical shoreline cleaner Corexit 9580.76 An estimated 35 barrels of oil were not recovered and were considered to be released to the marine environment.
The recovery effort during the Burnaby Harbor spill was relatively successful. Because the synthetic crude traveled on a predictable path through the storm sewer system, the responders were able to set up booms in a quick and efficient manner. The effects of the spill were limited due to favorable conditions for recovery:77
- There was sunny weather with little stormwater flow (slowed the movement of oil in storm drains and facilitated evaporation of oil).
- The spill occurred on slack tide (incoming tide helped keep the oil near shore while booms were placed, and helped limit the movement of oil in the Inlet).
- It occurred outside the primary migration and overwintering period for birds, and after the breeding bird season.
- It occurred prior to the main period of salmon migration to creeks and rivers for spawning.
There were no reports of the oil sinking or becoming submerged in the water column.
Toxicity of Commonly Transported Crude Oils
As described in Chapter 2 and displayed in Table 2-1, crude oils contain a variety of chemical constituents whose percentage compositions differ widely depending upon the specific product or blend, and thus generalizations about toxicity are difficult. However, the toxicity of typical constituents of crude oil has been demonstrated by many studies. Specific toxic compounds include the monoaromatic hydrocarbons comprising BTEX. The acute toxicity, respiratory issues, and potential carcinogenic properties of BTEX are of concern regarding the health of humans and of wildlife. Various diluted bitumen products show lower BTEX levels (percentage volume)74 compared to light and medium crudes, with values similar to those of heavy crudes (Table 3-1).
Other crude oil components of interest include the low-molecular-weight linear alkanes, other monoaromatics, and 2-ring PAHs that are often of acute aquatic toxicity concern, mainly due to acute narcosis-based mechanisms of toxicity. Also of concern are the 3- to 5-ring (unsubstituted
TABLE 3-1 Five-Year Average Concentrations and Ranges of BTEX in Various Crude Oils in Percent Volumeii
| Light Crude | Medium Crude | Heavy Crude | Diluted Bitumen | |
| Benzene | 0.22 (<0.01-0.38) | 0.42 (0.14-0.77) | 0.12 (0.02-0.21) | 0.16 (0.06-0.28) |
| Ethylbenzene | 0.26 (0.24-0.27) | 0.35 (0.19-0.60) | 0.11 (0.05-0.18) | 0.07 (0.04-0.11) |
| Toluene | 0.67 (0.03-1.14) | 0.87 (0.29-1.34) | 0.26 (0.12-0.45) | 0.32 (0.18-0.47) |
| Xylenes | 0.99 (0.18-1.46) | 0.77 (0.43-1.09) | 0.36 (0.23-0.49) | 0.33 ± 0.05 (0.27-0.43) |
| TOTAL BTEX | 2.10 (0.24-3.26) | 2.34 (0.97-3.11) | 0.84 (0.56-1.26) | 0.89 (0.64-1.16) |
iiData are from the Crudemonitor database accessed May 2015. Values in parentheses list the range of concentrations across oils in each category. All crude oils with data spanning at least two years were included for comparison. The crude oils include five light crude oils (BC light, Boundary Lake, Koch Alberta, Pembina light sour, and Scotian light); four medium crude oils (Medium Gibson sour, Midale, Peace Pipe Sour, and West Texas Intermediate); eight heavy crude oils (Lloyd blend, Wabasca Heavy, Western Canadian Blend, Bow River North, Bow River South, Fosterton, Seal Heavy, and Sockeye 2000) and eight diluted bitumen (Access Western Blend, Cold Lake Crude, Western Canadian Select, Christina dilbit blend, Borealis Heavy Blend, Kearl Lake).
and alkylated) PAHs, which have been demonstrated to cause both acute and sublethal toxicity via numerous mechanisms of action that are important in delayed responses and in long-term residual and chronic effects, such as carcinogenesis, reproductive failures, developmental deformities, and immune suppression. A summary of PAH concentrations in some commonly transported crude oils and diluted bitumen is presented in the previous chapter (Table 2-2).
There is a wealth of toxicological literature describing the sublethal effects of commonly transported crude oil components.29 The individual-level acute and sublethal effects include early-life-stage developmental defects, reduced growth and reproductive capacity, behavioral impairment, altered bioenergetics, genetic damage, impaired immune function and hence disease resistance, and enzymatic and hormonal changes including endocrine and hypothalamic-pituitary-adrenal axis implications, which can lead to population- and ecosystem-level impacts (e.g., changes to the base of food webs that affect higher-level consumers).
In addition to direct, chemically based mechanisms, crude oils can also result in acute and sublethal effects via physical mechanisms. The physical coating of biological surfaces impedes an organism’s movement and can alter behavior and/or hamper respiration. An example of this would be the coating of gills and permeable skin surfaces of fish.
As noted in Chapter 2, many of the chemical compounds in diluted bitumen are found in other crude oils, and thus toxic properties are expected to be similar in many respects, although the relative proportions differ. The bitumen is a concentrate of the high-molecular-weight components of a conventional crude, while the diluent is a concentrate of the low-molecular-weight components. In contrast to other crude oils, very limited data exist on the toxicity of diluted bitumen, although much is known about the diluent components as they are commonly found in other crude oils. The potential for diluted bitumen to significantly weather, adhere to particles, submerge, and possibly sink in quiescent areas, coupled with its high content of recalcitrant resins and asphaltenes, can result in unrecoverable sunken oil and thus prolonged chronic exposure of benthic organisms.
Without a more detailed chemical characterization of this initial chemical pool, coupled with characterization of weathering or biodegradation products the comparison of diluted bitumen toxicity to other commonly transported crudes cannot be completed. As discussed in Chapter 2, a large fraction of diluted bitumen consists of an array of currently uncharacterized chemicals. This situation is not unique to diluted bitumen and
applies to other crude oils; however, diluted bitumen has a larger number of unknown polar compounds. Furthermore, the adhesion of residual bitumen oil to biological surfaces may lead to physical coating of organisms (Figure 3-5), impacting movement, behavior, feeding, thermoregulation, and respiration.
There are very few published laboratory experiments specifically investigating the toxicity of diluted bitumen, despite its use and transport in North America for over 40 years. Currently, there is only one laboratory study investigating the toxicity of a diluted bitumen, working with larval fish (Japanese medaka, Oryzias latipes).78 Sublethal concentrations of soluble components of the oil or “water-accumulated fraction” derived from Access Western Blend caused an increased prevalence of blue sac disease, impaired development, and abnormal swim bladders upon hatching. In addition, exposures resulted in various genetic markers for physiological stress that are commonly observed with exposure to other crude oils. It has been well established that exposure to conventional crude oils can cause embryo toxicity in fish primarily due to the 3- to 5-ring alkylated PAHs. Concentrations of 3- and 4-ring unsubstituted parent and alkyl PAHs are similar or higher in Cold Lake Blend in comparison to other commonly transported crudes as listed in Table 2-2 in Chapter 2, although that blend contains higher levels of total phenanthrenes, fluorenes and chrysenes compared with light and medium crude oils. Therefore, based on the limited available research, it is expected that exposure to diluted bitumen would cause similar or higher chronic toxicity to fish embryos, although further chemical characterization of diluted bitumen along with more toxicity studies in other fish species would be required to confirm this. Delayed effects from acute or chronic exposure or chronic toxicity studies of the residual bitumen component have also not been investigated. Other additional mechanisms of action and sub-lethal effects to other species and life stages have also not been studied for diluted bitumen.
While there have been few experimental dose-response investigations of diluted bitumen toxicity, the toxicity of diluted bitumen spilled into the environment has been inferred from post-spill field observations and investigations as well as laboratory studies using field samples from recent diluted bitumen spills. In addition, organisms collected near the oil sands deposits have been examined.79 Natural bitumen deposits are exposed in the banks of rivers in the Athabasca oil sands region, and studies of wild fish from these locations have found sublethal biochemical and hormonal responses, including the classic response to exposures of PAHs, namely increased levels of ethoxyresorufin-O-deethylase activity and a reduction in steroid production in comparison to fish from reference areas.79d For early life stages of fish, these biochemical responses can
be related to the observed deformities in embryos and larvae following exposure to waters affected by oil sands.79c
The significance of the field observations was confirmed by laboratory-based studies of sediment toxicity in water bodies of the oil sands region.79a,79b Compared to controls, fish eggs and embryos of fathead minnow (Pimephales promelas) and white sucker (Catostomus commersonii) showed increased mortality, reduced hatching success, delayed timing of hatching, abnormal development of embryos, and deformations and reduced size of larvae. The same effects have been observed during exposures to weathered conventional crude oils.
Bitumen contains several metals that are potentially toxic and are discussed in further detail in the human toxicity section below. However, potential bioavailability and toxicity of metals in the diluted bitumen transported by pipelines remains little studied. Studies in the vicinity of the oil sands mining sites in Alberta have documented increased concentrations of cadmium, copper, lead, mercury, nickel, silver, and zinc in snowmelt (reflecting atmospheric emissions and redeposition) and river waters, reaching levels of concern for the protection of aquatic life.7b Commonly measured metals, such as vanadium and nickel are found at higher levels in bitumen (and diluted bitumen) compared to other crude oils but these metals are predominantly bound in organic-metal porphyrin structures that are less bioavailable. Few data address this issue, however, and weathering and biodegradation processes have the potential to release these metals. Overall the toxicity (chronic and acute) of fresh and weathered diluted bitumen and its residues to freshwater, estuarine, and marine species at various life stages is at this time very understudied compared with other commonly transported light and medium crude oils. Furthermore, bioaccumulation and impacts to the food web and trophic transfer issues have not received attention for diluted bitumen in contrast to that of commonly transported crudes.
In the case of the spill of diluted bitumen into the Kalamazoo River (Box 3-1), toxicological effects on fishes were studied by a team from the U.S. Geological Survey within the framework of the Natural Resource Damage Assessment (NRDA).80 Fish were sampled within a few weeks of the spill to obtain a gross pathological assessment of general health. When fish in oiled reaches were compared to fish in an upstream reference reach, significant adverse changes were evident. These differences were not observed in subsequent years.
The likelihood of submergence and sinking of weathered diluted bitumen, often as OPAs, merits particular attention because it presents distinct routes of exposure to the biota. In addition, the sunken oil may not be recoverable, thus resulting in protracted periods of exposure. Many aquatic animals also consume particles directly or indirectly from the bot-
tom sediment, underwater plant or macroalgal surfaces, or the water column, which may include oil droplets and OPAs. Contact of droplets with respiratory surfaces (e.g., fish gills) or with permeable dermal surfaces can interfere with respiration. Organisms feeding on oil-contaminated material can in turn be consumed by predators, which can pass contaminants up the food web.
The major ways in which humans may be exposed to chemicals following an oil spill are via (i) inhalation of volatile organics, (ii) dermal (skin) exposure through direct physical contact with the oil, (iii) ingestion of contaminated drinking water, and (iv) ingestion of contaminated food. This section aims to compare potentially hazardous characteristics of diluted bitumen with those of other commonly transported crude oils and identify whether diluted bitumen may pose similar or distinct human health risks.
Inhalation and dermal exposure are typically immediate concerns for the first responders and the public in the vicinity of the spill. Dermal exposure can be addressed by use of appropriate personal protective equipment, and so the focus here is on inhalation hazards and contamination of drinking water and food sources. While the issue of drinking water contamination can also be immediate, such concerns may persist for some time and may occur well beyond the initial site of the spill, depending on the fate and transport of the spilled oil. Similarly, contamination of agricultural produce (e.g., by irrigation water) and fisheries may be a concern in the near term. However, there may also be longer-term concern for food safety. During a spill, water bodies for human recreational use (swimming, boating, and fishing) are closed until deemed safe. For these reasons, the immediate human health issues are considered first and then the potential longer-term human health concerns are considered separately.
Initial human health concerns
During the initial days of spill response, the major components of concern to human health in crude oils include the volatile compounds—benzene, toluene, ethylbenzene, and xylenes (collectively called BTEX) and hydrogen sulfide (H2S)—that can result in acute and sublethal effects via inhalation exposure. Benzene is also a well-known human carcinogen. Benzene is typically present in crude oils and is frequently monitored to assess both inhalation and drinking water supplies. Health and safety
concerns regarding exposure to volatile organic compounds have also been addressed in the context of response (see Chapter 4). Oil spills can occur in populated areas and result in immediate human exposure; the 2013 Mayflower, Arkansas, oil spill in a residential area exemplifies the importance of an immediate, effective response to mitigate human health hazards (Box 3-5).
Table 3-1 shows the 5-year average BTEX concentrations and ranges for light, medium, heavy and diluted bitumen crude including the four representative crude oils described in Chapter 2.74 The data indicate that the BTEX constituents in diluted bitumen (reported in % by volume) are not distinctly different from other crude oils. The average BTEX in diluted bitumen at 0.89 % vol was similar to the heavy crude oils at 0.84 % vol, whereas light and medium crude oils were 2.56 and 2.80 % vol respectively. The light crude oils category represented the highest variability. By comparison, the 5-year average values reported for all condensates listed in the crude monitor database averaged 4.22% BTEX by volume.74
Exposure to hydrogen sulfide (H2S) gas is also of concern as it can immediately damage the central nervous system and act as a chemical asphyxiant.81 Given that hydrogen sulfide is corrosive, its levels were discussed in the previous report.2 Diluted bitumen typically contains similar or lower levels of hydrogen sulfide compared to the other crude oils.
Another immediate (and potentially longer-term) concern relative to human health is the contamination of drinking water in the vicinity of or downstream of the spill. The more soluble BTEX components, especially benzene, are of most concern for drinking water and are also discussed in Chapter 4. In water, the solubilities of BTEX compounds range from approximately 150 to 1,800 mg/L82 making them significantly more soluble than most other hydrocarbons. The USEPA has established water quality standards for BTEX compounds that are regulated by the Safe Drinking Water Act (SDWA). USEPA’s National Primary Drinking Water Regulations set maximum contaminant level (MCLs). For example, MCLs for benzene are 0.005 mg/L (5 parts per billion or ppb). In a 2015 pipeline spill of Bakken crude oil into the Yellowstone River in Montana, oil related volatile organic contaminants (e.g., benzene) were detected at levels of concern at a drinking water treatment plant forcing the closure of the water intakes. Given the similar concentrations of BTEX in diluted bitumen and other commonly transported crude oils in the U.S. pipeline system, it is not expected that the BTEX components in diluted bitumen will pose a higher immediate risk to human health during the initial spill response phase.
Of additional concern with respect to immediate human (and any exposed organism) toxicity is the presence of elevated concentrations of
BOX 3-5
Mayflower, Arkansas: ExxonMobil
On March 29, 2013, the 20-in. Pegasus oil pipeline, constructed in 1947-1948 and operated by ExxonMobil Pipeline Corporation, ruptured in Mayflower, Arkansas, releasing 3,190 barrels of Wabasca Heavy crude oil in a residential area. The Pegasus pipeline is buried 24 in. underground at the release site. The oil flowed down the street and into a drainage ditch and tributary that led to Dawson Cove, an arm of Lake Conway. The upper part of Dawson Cove was heavily forested and flooded at the time of the spill. The lower part of Dawson Cove is separated from the lake by a road with open culverts. Response crews were on site within 30 min of detection of the release, including ExxonMobil Pipeline employees and federal, state, and local responders. Staff from the Arkansas Game and Fish Commission quickly constructed earthen berms at the head of Dawson Cove, which allowed the oil to be contained within the cove, with no oil documented as entering Lake Conway.
Wabasca Heavy crude oil is a blend typically composed of ~80% bitumen obtained by polymer injection and water flooding from the Athabasca region and 20% diluent; this blend typically has a 19-20 API gravity, ~1% BTEX, and 4.15% sulfur.74
Twenty-two homes were evacuated. Air quality monitoring in residential areas in the first week or so after the release reported benzene below detection (0.05 ppm) but volatile organic compounds (VOCs) of up to 29 parts per million (ppm) on the day of the spill and 3 ppm by the third day. Higher levels of both benzene and VOCs were measured in work areas, and workers wore respiratory protection as specified in the health and safety plan.
There were no reports of oil sinking in the quiet waters of Dawson Cove. However, the dense vegetation had to be removed to access the oil, and intensive mechanical methods were used to remove trees, shrubs, rootballs, and other or-
unknown volatile and/or water-soluble compounds. This concern is not unique to diluted bitumen and is of concern for all crude oils.21 However, the concentrations of polar compounds such as those containing nitrogen are higher in diluted bitumen. Therefore, there may be chemicals of toxicological concern unique to diluted bitumen that have not yet been characterized. This consideration is an important caveat for an assessment of relative human health risk for diluted bitumen spills compared to spills of commonly transported crude oils: an assessment based only on known BTEX concentrations must be considered incomplete and therefore tentative.
ganic debris from about 14.5 acres of forested wetland. The intensive mechanical operations mixed oil into the soils in the cove area. The post-cleanup assessment found low levels of PAHs in the soils that were mostly below sediment toxicity concerns. Nonetheless, because of chronic sheening in the cove, in August 2014 ExxonMobil was required to (i) excavate 800 yd3 of sediment from the 1,300-ft-long tributary upstream of the cove, and backfill as needed with clean sediment; (ii) place an organoclay soil amendment on 2 acres of sediment surface within the remaining vegetated area; and (iii) place a reactive cap on about 4.5 acres of open water.
This spill shows that quick response can be very effective. Diluted bitumen products will initially float with the lack of turbulence in fresh water, and at that time they can be cleaned to levels of low toxicity, but are persistent and can cause chronic sheening that can trigger the need for extensive treatment, particularly in inland areas where natural removal processes are slow.

Photo Credit: U.S. Environmental Protection Agency
Longer-term human health concerns
In addressing longer term human health concerns, protection of water supplies is a focus of spill response activities. For reasons described earlier in the report, weathered diluted bitumen has a greater potential to submerge or sink, presenting a greater potential for chronic contamination of a water supply that may result in a long closure time for drinking water sources. Another serious outcome in the case of incomplete removal of sunken weathered bitumen could be a longer lasting impairment of a surface-water source of drinking water.
In the United States, groundwater supplies about 32% of drinking water83 and there have been studies of crude oil contamination of groundwater, such as the site in Bemidji, MN (Box 3-2). Given the potential for longer travel time of crude oil in groundwater systems, the impacts may be detected later than for surface waters and may be inherently more difficult to remediate than for surface water bodies. The environmental behavior of a diluted bitumen spill with respect to groundwater contamination is discussed above.
There are various delineations of groundwater zones in which limits are set for both residential and industrial zones84 for acceptable levels of Total Petroleum Hydrocarbons (TPH) and BTEX components. In the recent diluted bitumen spill in Marshall, MI, over 150 drinking water wells were monitored for a variety of inorganic and organic oil (and non-oil) related chemical contaminants. The contaminants monitored included nickel, vanadium, and organic compounds, including BTEX. TPH in the gasoline range (GRO, ranging in carbon atoms from C6-C10) and the diesel range (DRO, >C10-C28) were measured. The public health assessment report released in 201385 stated that there were no detections of crude oil related chemicals in any of the sampled wells, other than iron and nickel. These elevated levels of iron and nickel were only found in a few of the over 150 wells sampled and were deemed to be a natural occurrence for the wells in Calhoun and Kalamazoo Counties. The absence of groundwater contamination along the river was not unexpected given that groundwater flows toward the river and its floodplain in almost all of the affected reach.86
When there is deemed to be a risk of human exposure via food consumption, the collection of fish and other food items (e.g., shellfish) may be prohibited over space and time based on sheening and/or the presence of targeted compounds in the tissues of food items. The main class of compounds that are measured for food safety is the polycyclic aromatic hydrocarbons (PAHs), although others, including metals may be monitored. As Table 2-2 in Chapter 2 shows, there are a number of higher molecular weight PAHs that the USEPA has listed as probable human carcinogens for a representative set of light, medium, and heavy crudes, in addition to diluted bitumen. Out of the 16 EPA priority PAHs listed, 11 have slightly higher to over 3 times higher levels of these carcinogenic compounds in Cold Lake diluted bitumen compared to the four other crude oils. For example, benzo[a]pyrene levels in Cold Lake diluted bitumen are 3.01 µg/g compared to the light and medium crudes ranging from 0.25–0.74 µg/g. As mentioned above there also are many unknown compounds in crude oils for which we do not know the environmental fate, bioavailability and potential impact to organisms. Further chemical composition details are required, together with assessments on bioavail-
ability and toxicity to organisms. Furthermore, details on how and to what the chemical constituents are biodegraded when diluted bitumen is spilled into the environment or metabolized to by organisms that bioaccumulate oil constituents are also required for a full understanding of the toxicity of diluted bitumen.
Accumulation of oil-derived metals into the food chain and ultimately into food for human consumption is also possible. However, levels of metals are usually very low in crude oils and only a few are measured (typically, nickel, vanadium, copper, cadmium and lead). Levels of vanadium and nickel in the various crude oils are reported as these are usually the metals of highest concentration compared to other trace metals. Using the 5 year average levels of vanadium and nickel in the same oils as detailed for the BTEX comparison shows that diluted bitumen concentrations are over 7 and 2.5 times higher than in the example light and medium crude oils respectively. For example, five year average levels of nickel and vanadium in diluted bitumen are 60 and 152 mg/L respectively compared to 8 and 20 mg/L in light and 23 and 60 mg/L in medium crudes. The bioavailability and toxicity of metals can also be very dependent upon the specific form (speciation) of the metal, which is dependent upon a variety of environmental parameters, including redox status (related to the presence or absence of oxygen) and pH content, both of which can be modified due to an oil spill. However, these measurements do not take the bioavailability of these metals into consideration and they are likely to be less bioavailable given that they are commonly found in tightly bound organic structures (porphyrins) in diluted bitumen. Although evidence of food web contamination from a diluted bitumen spill is lacking, risks for benthic organisms and their consumers cannot be ruled out given the limited data available regarding chemical composition and how biodegradation and other weathering processes might change the chemical composition of the residual oil. A more complete understanding of the chemical constituents of all crude oils and their weathering products is necessary to support a more thorough toxicological assessment. Based on the limited available evidence for diluted bitumen, however, it appears that it would pose a lower or similar hazard to human health in the short-term for the chemicals currently monitored and assessed. However, the potential toxicological risks to humans and animals, particularly longer-term exposures, are currently unknown compared to commonly transported crude oils.
In some respects the environmental effects of diluted bitumen spills resemble those of spills of other commonly transported crude oils, as long
as the diluent and bitumen remain mixed and in their original proportions. The movement of the oil on land and on the water surface, and its toxicity to wildlife and people, are similar at the outset. Once a spill occurs, however, exposure of diluted bitumen to the atmosphere allows the lighter diluent fraction to evaporate, resulting in residual bitumen that has several distinctive characteristics, being particularly dense, viscous, and with a strong tendency to adhere to surfaces and to submerge beneath the water surface and potentially sink to the sediments.
In light of these characteristics, diluted bitumen spills in the environment pose particular challenges when they reach water bodies. Progressive evaporative loss of the diluent leaves behind the relatively dense and viscous bitumen, which can then become submerged, perhaps first by adhering to particles, and ultimately sink to the sediments. The density of the residual oil need not exceed that of the water to submerge if conditions are conducive to the formation of oil-particle aggregates with densities greater than water, and this may be a common situation in inland and coastal waters where suspended particulate matter abounds. The loss of the lighter fraction and resultant potential for submergence of residual oil manifests more quickly and will involve a greater fraction of the spilled oil than in the case of light and medium crude oils. Toxicity of the residual bitumen has received little study, although toxic effects of both organic substances and associated metals have been observed in the vicinity of oil sands deposits in western Canada. The difficulty of recovering sunken oil and the recalcitrant nature of bitumen mean that aquatic biota may be exposed to the material for longer periods than in the case of lighter oils that sometimes sink to the bottom but are relatively biodegradable.
Impacts of diluted bitumen spills are expected to vary across the great diversity of inland and coastal water settings, which present varying scenarios for transport and submergence of the oil, for the feasibility of recovering oil that has sunk, for the nature of the aquatic biota that would be exposed, and for human uses of the water and water bodies. Flow and mixing patterns, turbulence, and the nature and abundance of natural particulate matter are among the most important considerations.
The toxicity of diluted bitumen has scarcely been studied using a direct, experimental dose-response approach in the laboratory, although it has been inferred from studies in surface waters draining the oil sands deposits, as well by post-spill sampling and bioassays. Evidence so far suggests that the BTEX in the diluent—as well as certain PAHs in the bitumen—can have toxic effects, but these same compounds also occur in other commonly transported crude oils. Many of the PAHs known to cause chronic and sublethal effects, such as alkyl PAHs and EPA priority PAHs, are at similar or higher levels than those of commonly transported crude oils. Diluted bitumen or its weathered residues may contain
other, uncharacterized compounds with toxic properties, but this awaits further investigation. There may also be various metabolites produced from diluted bitumen components that are also currently unknown and uncharacterized for their toxicological consequences. Until there is more toxicological research specifically targeting diluted bitumen, the acute, chronic, sub-lethal and longer-term toxicities of diluted bitumen relative to conventional crude oils will be poorly known.