5
Processes to Improve Patient Care
The well-being of patients is central to the practice of medicine. This is the focal point around which the efforts of all stakeholders in health care converge, with the goals of curing disease and extending life whenever possible, while also improving or preserving quality of life. As discussed throughout this report, precision medicine, defined as targeting therapies to patients through the use of biomarker tests, is being increasingly pursued as a path toward realizing this goal, particularly in those patients for whom current treatments have proven inadequate or are nonexistent. However, challenges associated with the rapid increase in potentially therapeutically relevant biomarkers have accompanied the molecular characterization of diseases. In addition, the number of tests offered by both academic institutions and private test developers has been steadily rising (Frampton et al., 2013; Meric-Bernstam et al., 2013). The growing list of options for biomarker tests, absent guidance, can be confusing not only for academic health care providers, but especially for providers in the community where, for example, the majority of U.S. cancer patients are treated (The Moran Company, 2013).
This chapter discusses processes to improve patient care in the context of biomarker tests and molecularly targeted therapies, including equity in access to testing and relevant expertise, ensuring patient safety and adequate test performance, and the implications for generating evidence-based clinical practice guidelines. The committee’s specific recommendations are summarized in the conclusion of this chapter.
CURRENT CHALLENGES IN TEST RESULT INTERPRETATION
In the current era of precision medicine, physicians seeking to incorporate emerging tools into the management of their patients will increasingly use advanced biomarker tests to guide treatment (Evans and Khoury, 2013). Certain predictive biomarker tests, known as pharmacogenomic tests,1 evaluate genetic variations that affect pharmacokinetics (i.e., “what the body does to the drug”) and pharmacodynamics (i.e., “what the drug does to the body”), and physicians and patients use these tests to optimize treatment selection and dosage (see Box 5-1). However, in diseases with complex somatic or germline genomic etiologies, such as cancer or cystic fibrosis (CF), the molecular subsets of these diseases may
___________________
1 As defined in Chapter 1, pharmacogenomic tests are outside the scope of the committee’s charge, but their distinct role is discussed briefly here in the interest of clarity.
have unclear or emerging relationships to molecularly targeted therapies. In CF, for example, approximately 5 percent of cases are caused by the p.G551D mutation in CFTR (the gene which, when mutated, can result in the disease) and can be treated effectively by the drug ivacaftor. For the patients whose disease is linked to the far more predominant CFTR mutation p.F508del (present in 90 percent of CF patients), treatment with ivacaftor alone, or in combination with another new drug named lumacaftor, has thus far produced comparatively limited results (Davis, 2015).
Similarly, tremendous advances have been made in basic and clinical cancer research, but many challenges to widespread implementation of targeted cancer therapeutics remain (Garraway et al., 2013). Cancer is now understood to be a diverse collection of distinct acquired or inherited genomic diseases, and despite initial successes with drugs targeting specific biological processes (e.g., trastuzumab for breast cancer overexpressing HER2,2 and imatinib for chronic myelogenous leukemia with the BCR-ABL translocation), the success of new treatments depends, in part, on well-validated biomarker tests to optimally select patients for targeted therapies (Mendelsohn, 2013). Clear understanding of the biological mechanisms underlying response and resistance to molecularly targeted therapy remains incomplete; the existence of so-called exceptional responders are a testament to existing knowledge gaps (see Box 5-2). Increasingly, improving the survival of patients with advanced cancer may require targeting multiple oncogenic processes in order to overcome resistance due to tumor heterogeneity and cellular context (Vogelstein and Kinzler, 2015).
An early example of the paradigm of using a single biomarker test to select patients likely to benefit from a molecularly targeted therapy is the use of trastuzumab in breast cancer. Trastuzumab was a targeted therapy approved by the Food and Drug Administration (FDA) in 19983 for treatment of breast cancer patients. This approval was initially for patients with metastatic breast cancer that overexpressed the HER2 protein, as detected by immunohistochemistry (IHC), a technique that uses antibody-based staining to measure levels of protein expression. Subsequent studies eventually expanded approval to include use as adjuvant therapy4 in patients with early-stage breast cancer exhibiting either HER2 protein overexpression by IHC, or HER2 gene amplification (increased copies of
___________________
2 There are multiple names for the protein and gene (including ERBB2), but HER2 is the term commonly used in oncology practice.
3 See http://www.accessdata.fda.gov/drugsatfda_docs/label/2010/103792s5256lbl.pdf (accessed July 1, 2015).
4 Adjuvant therapy is given to patients who have undergone potentially curative intervention for their cancer (e.g., surgery or radiotherapy) in order to minimize the risk of local recurrence or metastasis.
a gene) as detected by a technique known as fluorescent in situ hybridization (FISH) (Hudis, 2007).
Despite the seemingly straightforward relationship between HER2 amplification/overexpression and response to the drug trastuzumab, the testing methodology continued to be refined over many years, and there is still no general agreement on optimal test methods and interpretation. As detailed in the Institute of Medicine (IOM) report Evolution of Translational Omics (IOM, 2012a), substantial discordance was reported for both IHC and FISH results performed in community laboratories compared to a central reference laboratory in the course of two clinical trials (Paik et al., 2002; Roche et al., 2002). A 2007 panel established by the American Society of Clinical Oncology/College of American Pathologists (ASCO/CAP) recommended HER2 testing for all invasive breast cancers, as well
as proposed guidelines to assist in test result interpretation and to reduce errors in the laboratory testing process (Wolff et al., 2007a,b). Additionally, the panel clarified some of the technical limitations of both IHC and FISH (Schmitt, 2009). HER2 testing has improved as a result of these guidelines and other efforts to standardize testing performance and interpretation criteria, although questions remain regarding whether some patients whose breast cancer is negative for HER2 overexpression might benefit from treatment with trastuzumab (Ithimakin et al., 2013).
A more recently FDA-approved cancer immunotherapy drug, the monoclonal antibody nivolumab, further demonstrates the ongoing difficulty in establishing well-validated biomarkers to guide treatment selection by health care providers and patients. Johnson and colleagues’ review of nivolumab for treatment of patients with melanoma described the process by which evolving evidence shapes the implications for clinical use of the drug (Johnson et al., 2015). Nivolumab was originally reported to be effective only in those melanoma patients whose tumors tested positive for overexpression of PD-L1, an immune system biomarker. Subsequent studies reported response to nivolumab in a small but significant number of patients whose tumors tested negative for PD-L1. Given the durable nature of the response and relatively limited early toxicity (patients who respond to nivolumab treatment tend to survive longer than is typical for advanced cancer patients, with fewer serious side effects), as well as the still-developing evidence around the biomarker PD-L1, FDA-approved use of nivolumab currently does not require a PD-L1 biomarker test.5
Despite this finding, similar immunotherapies are being developed, each with their own distinct PD-L1 biomarker test. FDA has recently approved the first companion diagnostic for the PD-L1 inhibitor pembrolizumab (also a monoclonal antibody), and an industry working group has been convened to harmonize the validation process across PD-1/PD-L1 biomarker tests, using different antibodies and different interpretation criteria (Averbuch et al., 2015). However, it is not yet clear whether a single test can optimally stratify patients for treatment with the various immunotherapies targeting the same pathway. Additionally, the target population for these drugs has the potential to expand rapidly. Both nivolumab and pembrolizumab have been recently approved by FDA for treatment of metastatic non-small-cell lung carcinoma, and studies have reported significant response to pembrolizumab in a separate cohort of non-melanoma patients whose tumors have abnormally high mutation rates (Le et al., 2015).
The lessons learned through the incorporation of targeted therapeu-
___________________
5 See http://www.accessdata.fda.gov/drugsatfda_docs/label/2014/125554lbl.pdf (accessed August 4, 2015).
tics into clinical practice highlight the need for standardized and well-validated biomarker tests, as well as continued evidence generation for use of both the biomarker tests and corresponding therapies. Over time, a large body of high-quality evidence could be generated from clinical experience to support optimal clinical decision making and, ultimately, improved outcomes for patients. The central importance of using high-quality evidence to guide clinical care featured prominently in the 2013 IOM report Delivering High-Quality Cancer Care, with the committee recommending adherence to evidence-based clinical practices, and further expansion of evidence gathering beyond clinical trials into real-world patient outcomes (IOM, 2013; Psek et al., 2015; Yu, 2015).
The absence of a robust evidence base, due to the rapid development and incorporation of new biomarker tests and targeted therapies, often leaves health care providers with less rigorous, unclear, and occasionally conflicting information on which to base their testing and treatment decisions (FDA, 2015; IOM, 2015). In certain rare diseases, this lack of evidence may be balanced by high unmet need for effective therapy where none currently exists. However, evidence suggests that some targeted therapies are context specific: colon cancers with BRAF mutations are largely unresponsive to BRAF inhibition despite therapeutic effectiveness in BRAF-mutant melanoma (Hyman et al., 2015; Prahallad et al., 2012). Due to the evolving nature of the evidence for the clinical use of predictive biomarker tests, careful consideration of the use of molecularly targeted therapies in general clinical practice is necessary to avoid potentially exposing patients to unnecessary risk for uncertain benefit.
The era of molecularly targeted therapy has implications for the fields of biomedical imaging and radiation therapy, which continue to be critical components of the treatment of cancer (see Box 5-3). For example, gene signatures6 may eventually be useful to identify patients for whom radiation therapy may not provide durable control of their disease, but more research is needed prior to adoption for clinical use (Ahmed et al., 2015; Torres-Roca et al., 2015).
EQUITY IN ACCESS TO TESTING AND EXPERTISE
Fair access is a key ethical concern in medicine. The bedrock ethical principle supporting fair access to health care is the principle of social justice: treat people as equals. Treating people as equals has been interpreted to mean access to opportunity (Rawls, 1971) and access to resources (Dworkin, 1981). Each interpretation entails equal access to health care,
___________________
6 Gene signatures are indexes or scores derived from testing multiple genes from one specimen.
either as an opportunity that some have and therefore all should have or as a necessary resource that should be available to all (Daniels, 2013).
In the context of precision medicine, challenges to obtaining access may confront patients of particular economic, ethnic, and cultural backgrounds and geographic locations. These challenges include the ability of patients to access and interpret complex information, to obtain coverage for biomarker testing, and to receive treatment with targeted therapies, if appropriate. In such a data-driven field, datasets skewed by the differential access to new technologies threaten the generalizability of conclusions reached through analysis of those datasets. Additionally, fair access requires that health care professionals possess the expertise to properly order tests and interpret the results to determine optimal therapy selection, in spite of the challenges posed by the rapid pace of clinical knowledge development outlined previously.
Potential Obstacles to Public Access and Understanding
Fair access to effective biomarker testing for molecularly targeted therapies may be jeopardized by a number of factors. These include (1) technical limitations of the biomarker tests and the evidence underpinning interpretation and use of the test results; (2) attitudes and behaviors relating to people from underserved communities; and (3) income inequalities (Chadwick, 2013; McClellan et al., 2013). Preventing the growth of disparities in access to biomarker tests for molecularly targeted therapies, and the resulting disparities in beneficial health outcomes, may depend on lessons learned from disparities in access to other forms of genetic testing.
Research on patterns of genetic predisposition testing reveals that more variants of unknown significance are identified in test results from minority populations than in those of European descent (Oloparde, 2004), and as a result the usefulness of genetic tests is reduced for those populations. Also, prediction models tend to be Euro-centric. Some models in particular underestimate risk for African Americans (Adams-Campbell et al., 2009) and Hispanics (Banegas et al., 2012), thereby potentially limiting access to appropriate care for those populations (Kurian, 2010). Interestingly, in a Government Accountability Office investigation, none of the testing companies that were sent specimens could provide fictitious African American and Asian clients with complete test results, though none of the companies’ advertising alerted the consumer to this testing coverage gap (GAO, 2010). The potentially harmful effect of advertising on the public’s perception of precision medicine, particularly through false or misleading advertisements, is described in Box 5-4.
A second source of unequal access to effective tests emanates from characteristics of the people in underserved communities. For example,
African Americans historically have been reluctant to use genetic services due to fears of genetic discrimination and stigmatization, and racial discordance between physicians and patients remains a persistent obstacle to shared treatment decision making (Lin and Kressin, 2015; Peterson et al., 2002). Research also suggests that minority patients are more likely to report an unmet need for discussion of genetic testing with their physician (e.g., for breast cancer risk and corresponding risk-reducing treatment); this perceived need may be influenced by cultural or language barriers, and misconceptions about genetic testing (Jagsi et al., 2015). The health care facilities available to some minority patients may be less equipped with respect to medical technology, or present other institutional barriers to participation in clinical research (Hasnain-Wynia et al., 2007; Joseph and Dohan, 2009). Nevertheless, large datasets are needed to further develop the association between genetics and disease risk or treatment response for all patient populations, and varying desire or ability among different populations to use genetic services hinders the compilation of these datasets. Moreover, accurate family histories are crucial to health assessments in the genomics era, yet both low socioeconomic status and being a member of a minority community are correlated with lower accuracy of personal and family histories (Abraham et al., 2009; Dominguez et al., 2007; Soegaard et al., 2008).
Income inequality may also contribute to unfair access. A recent study found that among insured women, the use of a 21-gene expression assay in women with newly diagnosed breast cancer was highest in geographical areas that had the largest income inequality, whereas the use of the test was lower in areas with more equal income distribution7 (Ponce et al., 2015). Furthermore, in areas with greater income inequality, the use of the test was significantly higher among high-income women compared to lower-income women, while there was no such disparity in the areas with less income variability. Other factors that correlated positively with the use of the 21-gene expression assay included being non-Hispanic white, being a younger age, having point-of-service insurance plans rather than other types of coverage, and having a lower Charlson score8 (Ponce et al., 2015). The authors concluded that, even among the insured, high income may result in better access; this may be particularly problematic given that the United States ranks high in income inequality among developed economies (DeNavas-Walt et al., 2013). However, a similar study of Medi-
___________________
7 The Gini index is a measure of statistical spread of income across a population, commonly used to describe income inequality. In this study, a Gini index variable was computed using Census tract-level average income in three categories: less than $50,000; $50,000-$100,000; and greater than $100,000.
8 Charlson score is a measure of comorbidity, with a higher value indicating multiple co-occurring conditions and predicting higher 10-year mortality.
care recipients, albeit a different population, did not reveal any disparities in test access among groups with different incomes or racial backgrounds (Dinan et al., 2015).
These studies did not examine access to testing among the underinsured or uninsured population, but research has shown, for example, that the costs of cancer treatment for the uninsured significantly exceed public and private payers’ negotiated rates, and consequently may restrict access (Dusetzina et al., 2015). In 2006, the Department of Health and Human Services (HHS) Secretary’s Advisory Committee on Genetics, Health, and Society (SACGHS) found that significant variation in coverage of genetic testing existed among state Medicaid programs, and moreover that the ease with which this coverage, as an optional benefit, could be rolled back may exacerbate heterogeneity in access to genetic testing (SACGHS, 2006). Subsequent analysis of data from 2011 by the Office of the Inspec-
tor General (OIG) reported that all but one state Medicaid program provided some level of coverage for genetic testing, but policies remained non-specific, case-by-case, or were evaluated by a variety of factors that continued to perpetuate heterogeneity in access to testing (OIG, 2012).
The IOM has called repeatedly for policies to ensure health insurance coverage for all U.S. citizens (IOM, 2004, 2009). Health insurance coverage was discussed as a primary method to ensure access to care in Delivering High-Quality Cancer Care, though the report acknowledged that access alone would not ensure quality, and recommended the development of innovative, community-focused programs aimed at identifying and reducing disparities (IOM, 2013). Expanding U.S. health insurance coverage is a primary goal of the Patient Protection and Affordable Care Act,9
___________________
9 Patient Protection and Affordable Care Act, Public Law 111-148, 111th Congress, 2nd Sess. (March 23, 2010).
and implementation of the legislation is ongoing. However, state-level variation in implementation policies can impact enrollment and access (Sommers et al., 2015), and a recent Census Bureau report states that 10.4 percent, or approximately 33 million persons, were uninsured for the entire 2014 calendar year (Smith and Medalia, 2015). Thus, the uninsured and underinsured remain a population that is vulnerable to lack of access to health care services in general and in particular to precision medicine and molecularly targeted therapies.
Gaps in Patient Understanding
Once a patient successfully gains access to the health care system to obtain treatment, the degree to which the patient understands his or her clinical condition and recommended options can influence their health outcomes (Ancker and Kaufman, 2007; Nelson et al., 2008). Ensuring that patients understand their treatment options is challenging, particularly in diseases such as cancer, given the severity of the disease and the complicated nature of the treatments (Fallowfield and Jenkins, 1999). Many cancer patients can be overwhelmed by the complex information and terminology related to their diagnosis; Tom Brokaw, while being treated for myeloma, likened the experience to being in a foreign culture, commenting that “most patients enter a doctor’s office or hospital as if it were a Mayan temple, representing an ancient and mysterious culture with no language in common with the visitor” (Brokaw, 2015). The pervasive difficulty in understanding a cancer diagnosis and treatment options is magnified in genomic testing by patients’ low level of overall health literacy, and lack of specific genetic knowledge.
Although patients do not need to understand all of the complex concepts involved in genomic testing in order to participate in shared decision making, persistent concerns exist about the degree to which variability in health literacy and numeracy affect patient engagement and health outcomes (Paolucci and Wicklund, 2015). In Delivering High-Quality Cancer Care, the IOM described challenges to patient-centered care that included failure on the part of clinicians to understand patient needs and preferences, and cultural or language barriers that inhibit effective shared decision making for often complex cancer treatments (IOM, 2013). The IOM has also emphasized that health literacy and particularly health numeracy, defined as “the degree to which individuals have the capacity to access, process, interpret, communicate, and act on numerical, quantitative, graphical, biostatistical, and probabilistic health information needed to make effective health decisions” (Golbeck et al., 2005, p. 375), are critical for understanding biomarker-related decision making and potential outcomes (IOM, 2010a). The report noted that because numeracy does not
correlate as closely with education as literacy does (Nelson et al., 2008), further research into solutions to improve patient numeracy are needed not only for the purpose of shared health care decision making, but also to enable the public to help shape and accelerate the adoption of relevant health policy.
Another factor that contributes to the difficulty in adequately communicating information about genomic testing to patients is the high expectations that have accompanied precision medicine. Government agencies, professional organizations, and researchers often discuss the promise of precision medicine without providing realistic assessments about the current capabilities and likely timeline for meaningful breakthroughs. For example, many cancer patients believe that having their tumor genome sequenced will result in the discovery of a mutation that will enable successful treatment of their cancer with a targeted therapy. However, recent studies have found that potentially actionable targets are identified in less than half of patients specimens sequenced (an exception is an abstract presented at ASCO’s 2015 annual meeting, which reported identification of actionable targets in 77 percent of specimens), with limited data on patient outcomes (Meric-Bernstam et al., 2015; Mody et al., 2015; Nadauld et al., 2015).
Initial or highly cited scientific publications of biomarker effect sizes tend to report stronger associations with disease outcomes and risks than are demonstrated in subsequent meta-analyses (Ioannidis and Panagiotou, 2011). The media tends to disproportionately cover these publications due to their initial exciting findings, more than follow-up or conflicting studies, which can result in an unbalanced portrayal to the general public (Gonon et al., 2012). Historically, the manner in which the news media report scientific advances has been a major contributor to public understanding and expectations (Anderson et al., 2011); this can contribute to unrealistic expectations and/or fears in the context of genomic medicine (Condit, 2007; Condit et al., 1998; Lea et al., 2011). A decade ago, the Kaiser Family Foundation reported that 40 percent of the public at the time relied on news media (television, radio, newspaper) as their primary source of health and health care information (other primary sources included health professionals, friends and family, and the Internet) (Kaiser Family Foundation, 2005). More recent research shows the Internet and other new media technologies10 are increasingly pervasive, particularly for younger people (Kaiser Family Foundation, 2010). Survey data from 2013 indicated that 35 percent of U.S. adults used the Internet to obtain information about a health condition, and nearly half of
___________________
10 New media technologies include smartphones, laptops, and tablet devices, and these may serve as delivery platforms for electronic versions of television and print media.
those subsequently consulted a health care provider based on their online research, bringing their expectations into the clinic (Pew Internet, 2013).
The evolving field of precision medicine may carry with it the unintended consequence of intensifying disparities in access to advanced health care services. It is essential that during efforts to standardize the analytic and clinical validity and develop evidence of the clinical utility of biomarker tests for molecularly targeted therapies, resources be dedicated to a comprehensive investigation and assessment of disparities in access to both testing and expertise. These may be due to a variety of economic, ethnic, cultural, and geographic factors, and once identified, efforts should be focused on reducing such disparities. The Clinical and Translational Science Awards were established with many focus areas, one of which was community and health disparities research, and may serve an important role in ensuring health disparities do not increase with the widespread adoption of precision medicine (IOM, 2012b). The committee recommends that agencies that fund the development or evaluation of biomarkers should include funding to identify and overcome barriers to promote equity, access, and public understanding of precision medicine (Recommendation 8a). Existing evidence of differential understanding or access, as cited in this section of the report, is currently limited. Therefore, the committee acknowledges this research need as a first step to characterizing potential obstacles to access, which could help inhibit growth in disparities as a result of precision medicine.
Ensuring Provider Expertise
A health care provider’s recommendation for a targeted therapy depends on patient preference, clinical condition, and reliable biomarker test results, and is guided by available practice guidelines that aim to improve patient outcomes (practice guidelines are discussed further in a separate section below). The interrelated effects on clinical practice of guidelines, other published clinical data, and FDA approval and labeling are difficult to separate. Studies of physician uptake of new targeted therapies over time have demonstrated their responsiveness to both regulatory decisions and evolving guidelines (Dotan et al., 2014; Neugut et al., 2012). However, even as biomarker tests for molecularly targeted therapies have become more common and physicians have grown more accustomed to their availability, lack of confidence in the use of genetic tests has persisted in a significant proportion of providers (Cox et al., 2012b; Freedman et al., 2003; Gray et al., 2014). In 2008, SACGHS found that health care providers have difficulties keeping current with which tests to order and how to apply the results to therapy (SACGHS, 2008). In the preceding year, Guttmacher and colleagues outlined the barriers
to educating non-geneticist physicians about genetics, including medical school curriculum deficiencies, misperceptions and incorrect physician attitudes toward genetics (i.e., that genetic information is peripheral to most clinical care), lack of bridges between basic sciences and clinical care, and a need for more practical, case-based continuing education (Guttmacher et al., 2007).
Although there have been long-standing concerns that primary care physicians lack training in genomics (Scheuner et al., 2008; Suther and Goodson, 2003), recently these concerns have extended to specialty providers as well, such as oncologists and pathologists. Gray and colleagues reported that even in a comprehensive cancer center, a significant minority of physicians described themselves as having “low genomic knowledge,” and that even among those providers who were more comfortable with genomic tests, there was wide variation in the interpretation, disclosure to patients, and clinical use of genomic test results (Gray et al., 2014). Although physicians have always had to adapt to medical technology innovations, how well they can adapt to the increasing availability of omics test results remains to be seen (Vassy et al., 2015). Current variation in clinical practice points to the potential difficulties of implementing targeted therapeutics based on biomarker tests in various clinical settings.
Maintenance of Certification (MOC) is a process administered under the American Board of Medical Specialties (ABMS) that is intended to avoid limitations in training and knowledge for physicians in practice that may influence the health care of their patients (Batmangelich and Adamowski, 2004). The four components addressed by MOC are professional standing (i.e., licensure); lifelong learning and self-assessment, which is generally interpreted as a requirement for participation in continuing medical education; cognitive expertise, as assessed by a proctored examination; and performance in practice. Each of these components is adapted by the various specialty boards to meet their specific needs, though some physicians that affirm the value of recertification question the MOC process’s relevance and effectiveness as it is currently structured (Drazen and Weinstein, 2010; Goldman et al., 2010). Research into MOC examination scores has demonstrated an association between higher scores and rates of evidence-based care processes for Medicare patients (Holmboe et al., 2008) as well as for patients with diabetes (Hess et al., 2012).
However, assessments of competency related to biomarker tests and their use for directing targeted therapies are lacking outside of certain specialties (e.g., pathology). This is complicated by the fact that board certification and MOC for some types of specialists (e.g., medical oncologists) are distributed across a variety of specialties such as internal medicine or obstetrics/gynecology. This distributed framework of responsibility
ensures flexibility for assessing competence in a highly complex profession, but may also require coordinated action by both ABMS and the independent specialty boards to ensure competence in the use of crosscutting medical technologies such as biomarker tests and molecularly targeted therapies. ABMS recently released updated standards for the MOC program that direct member boards to take action to increase program quality and relevance, to continuously monitor the quality and improvement of programs, and to “incorporate ways in which diplomates may engage in specialty-relevant, performance-in-practice assessment followed by improvement activities when practice gaps are identified” (ABMS, 2014, p. 12).
The role of ensuring competence of health care providers for improving patient care has featured prominently in previous IOM reports. In Redesigning Continuing Education in the Health Professions, the IOM similarly called for collaboration to assess the impact of continuing professional development on competence, health care outcomes, and patient safety (IOM, 2010b). Delivering High-Quality Cancer Care included recommendations to ensure the competence of both oncology and nononcology providers, and particular reference was made to the use of flexible cancer core competencies to improve cancer care delivery at a number of sites (Cox et al., 2012a; IOM, 2013; Smith et al., 2009). The report also drew from a National Cancer Institute (NCI) monograph on the value of patient-centered communication and shared decision making (Epstein and Street, 2007) in outlining recommendations for high-quality cancer care. Improving Diagnosis in Health Care further emphasized the value of interpersonal and communication skills to health care providers, including communication with patients (NASEM, 2015), citing the core competencies developed by ABMS and the Accreditation Council for Graduate Medical Education (ACGME) as evidence of the role played by licensing, certification, and MOC to improve health care delivery (ABMS, 2015; ACGME, 2015; NASEM, 2015).
The previous chapter discusses the committee’s rationale and recommendations for using electronic health records (EHRs) and related tools to enhance clinician decision making, but as precision medicine continues to expand into medical practice in general, the committee recommends that licensing and specialty boards should ensure that health care professionals have and maintain competencies needed for effective use of biomarker tests for molecularly targeted therapies (Recommendation 8c). Particular attention should be given to competency in communication with patients about the therapeutic implications of their test results, and realistic expectations of resulting outcomes. In addition to the roles for ABMS and ACGME, ensuring competency will require medical education organizations, including the National Board of Medical Examiners, the
Liaison Committee on Medical Education, the Federation of State Medical Boards, and the Educational Commission for Foreign Medical Graduates, to integrate these concepts into medical school curricula.
Underlying challenges to the effective clinical use of biomarker tests and targeted therapies include the current ambiguity surrounding standards for clinical utility (discussed in Chapter 3 of this report), as well as an often immature evidence base, further complicated by issues with research reproducibility that limit the impact on health outcomes (Bowen and Casadevall, 2015). In addition, the ability for health care providers, particularly primary care physicians, to keep pace with the speed of new clinical research and knowledge continues to decline as the number of new publications increases (Williams, 2015). As discussed previously in this chapter and throughout this report, the validation of biomarker tests and their association with response to molecularly targeted therapies in select patient populations is a process that involves accumulating large amounts of clinical data over time. The clinical implications, whether for testing methodology or treatment recommendations, would ideally evolve in parallel with the evidence and ultimately be available within clinical decision support systems (as discussed in Chapter 4).
However, in the process of developing a mature evidence base for any given intended use of a biomarker test, unclear or emerging data could lead to clinical decisions that are ineffective or put patients at unnecessary risk. Policy mechanisms to ensure that clinicians and patients have access to additional expertise, as needed, are necessary to limit the inappropriate use of biomarker tests for molecularly targeted therapies. Larger health care centers and academic medical centers are able to leverage multidisciplinary expertise to ensure evidence-based treatment in the care of patients with complex conditions. A focus on multidisciplinary care, particularly in specialties such as oncology, is recognized by accreditation entities overseeing hospitals and other large health care institutions (ACS, 2012). Multidisciplinary conferences, also referred to as multidisciplinary tumor boards, can be convened to discuss treatment options given specific pathologic features, biomarker test results, and a variety of other clinical factors specific to an individual patient. However, there is variation in the implementation of these tumor boards. While some studies have documented improvement in some outcomes or increased clinical trial enrollment, the effect of widespread adoption will require further assessment (Blayney, 2013; Keating et al., 2013; Kehl et al., 2015; Kuroki et al., 2010). Nevertheless, there is an opportunity for smaller clinical practices to access multidisciplinary, molecular expertise needed to assess biomarker tests and determine appropriate molecularly targeted therapy.
The Role of Telemedicine
Collaboration among health care teams for the purposes of evaluating treatment options for individual patients can be facilitated by the use of telemedicine, also known as telehealth. Telemedicine is defined by the American Telemedicine Association (ATA) as:
The use of medical information exchanged from one site to another via electronic communications to improve a patient’s clinical health status. Telemedicine includes a growing variety of applications and services using two-way video, e-mail, smart-phones, wireless tools and other forms of telecommunications technology. (ATA, 2015)
The Health Resources and Services Administration of HHS, which evaluates the use of telehealth through the administration of federal grants, defines telehealth in similar terms, as “the use of electronic information and telecommunications technologies to support long-distance clinical health care, patient and professional health-related education, public health and health administration” (HRSA, 2015). The terms “telemedicine” and “telehealth” today are generally used interchangeably, though historically telemedicine has been connected more closely to actual clinical services, while telehealth referred more broadly to health services including education and disease monitoring and management (ATA, 2015; CCHPCA, 2015; IOM, 2012c).
Telemedicine thus consists of multiple services, including primary care or specialist consultation, patient or provider education, and remote patient monitoring, which can be delivered through various methods. Live videoconferencing (“synchronous”), and store-and-forward systems (“asynchronous”) can be used to deliver clinical consultation with or without real-time clinician and patient interaction, for example, and electronic communication and mobile technologies facilitate remote monitoring and health education of patients and health care providers (ATA, 2015; CCHPCA, 2015). However, variation exists among individual health payer policies related to coverage and reimbursement of these services.
Until recently, the Centers for Medicare & Medicaid Services (CMS) maintained a narrow interpretation of reimbursable telemedicine services. Payment to providers previously depended on patients living in rural areas, and only certain types of clinical services were permitted (e.g., tobacco cessation, behavioral counseling for obesity), often with requirements for real-time interaction between provider and patient (e.g., video- or teleconferencing) (CMS, 2014). However, under a new rule, additional Current Procedural Technology (CPT) codes will expand CMS coverage of telemedicine beyond rural populations, and will include services such as non-real-time analysis and interpretation of clinical or physiologic data for those patients with multiple (defined as two or more) chronic
conditions, including cancer, diabetes, asthma, and heart disease (CMS, 2015). Limitations to this new policy include the multiple required chronic conditions, and a lack of payment tied to the collection of the data to be analyzed (ATA, 2014).
In Improving Diagnosis in Health Care, the National Academies of Sciences, Engineering, and Medicine acknowledged the potential for telemedicine technology to improve coordination of diagnostic management teams, as well as affordability and patient access to health care (NASEM, 2015). Relevant to molecularly targeted therapies, the report described the use of telepathology to improve diagnosis through granting immediate access to off-site subspecialty pathologists, and cited research suggesting the educational potential of having the local and consulting pathologists examine a case simultaneously. Teleoncology likewise has the potential to improve care and access. European countries have begun to use and reimburse for such care management services in an attempt to address heterogeneity in cancer outcomes. In the United States, “twinning” partnerships between larger medical centers and local centers serving rural or underserved populations may serve a similar purpose (Hazin and Qaddoumi, 2010). Studies of the influence of specific components of teleoncology, including real-time video consultations and hereditary cancer screening, suggest the ability to deliver adequate services, but conclude that further evaluation is necessary (Kitamura et al., 2010; Zilliacus et al., 2010, 2011).
To ensure judicious use of biomarker tests and molecularly targeted therapies, the committee recommends that the Secretary of HHS and CMS should conduct demonstration projects to design and assess the effectiveness of collaborative partnerships between community health care providers and larger health care centers and/or academic medical centers to be part of a rapid learning system (Recommendation 8b). These demonstration projects should examine the use of reimbursement incentives by CMS for the multidisciplinary collection and review of patient data with clinical recommendations, using distance technology or telemedicine. This would bring appropriate expertise into diagnostic management teams such as multidisciplinary tumor boards, to promote parity of access to appropriate biomarker use and clinical decision making. Individuals with molecular genomics expertise, including molecular pathologists, medical geneticists, and genetic counselors, should be included.
The continued growth of the clinical genetics workforce is integral to the shared decision making processes used to select targeted therapies. As molecularly targeted therapies continue to expand into clinical practice, clinicians will increasingly seek support from these providers (i.e., molecular pathologists, medical geneticists, and genetic counselors) and current estimates suggest a need to expand training programs to accom-
modate this (Paolucci and Wicklund, 2015; Wicklund and Trepanier, 2014). Coverage and reimbursement policies in the U.S. health care system are variable across payers, particularly those related to genetic counseling; this variability may limit access to expert guidance by non-geneticist physicians and their patients. In addition to the above recommendation to explore collaboration between larger medical centers and community centers, the committee recommends that these demonstration projects include reimbursement by CMS for genetic counseling services.
SPECIMEN ACQUISITION AND QUALITY
The clinical use of reliable biomarker tests to guide molecularly targeted therapy selection depends on many related processes. The central process is the testing itself, the oversight of which is discussed in Chapter 3. However, a number of other processes before and following clinical laboratory testing need to be considered, which can affect the accuracy and reliability of test results and patient safety. The brain-to-brain loop model11 for laboratory testing (see Figure 5-1) describes nine steps necessary to generate a laboratory test result, including test ordering, specimen collection, patient identification, specimen transportation, specimen preparation, analysis, result reporting, interpretation, and clinical action (Lundberg, 1981; Plebani et al., 2011). This model reflects the collaborative nature of clinical laboratory medicine, and provides a more granular understanding of the processes that lead to clinically useful test results. This section addresses interdisciplinary processes—including test ordering, specimen collection, processing, and handling—that are critical not only to the integrity of biomarker tests, but also to patient safety.
The acquisition of high-quality tissue specimens upon which to perform biomarker testing, as well as the development of sophisticated biobanks to store these specimens for ongoing research, is critical (Poste, 2011). High-quality specimen acquisition is particularly important for biomarker tests that use tissue-based somatic biomarkers (e.g., in cancer) because of the often discrete location of these tissues as well as specific specimen and methodological requirements of biomarker tests (Aisner and Marshall, 2012; de Gramont et al., 2015; Narmala and Boulmay, 2013). Low-quality, inadequate, or indeterminate specimens lack sufficient tissue for analysis, or lack the required tissue to be tested (e.g., obtaining normal tissue instead of tumor), and can delay a patient’s access to molecularly targeted therapies or hinder collaborative research that requires specimen collection across diverse clinical settings (Dolgin, 2016). Such errors can
___________________
11 The brain-to-brain loop model is named for the cycle of interaction among patients, physicians, and laboratory staff that can result in optimal laboratory testing and result reporting.
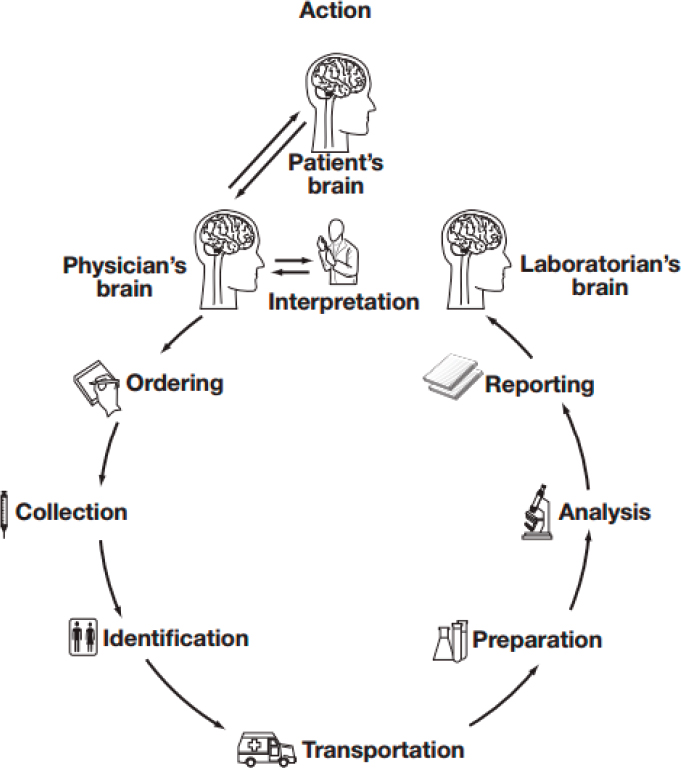
SOURCE: Plebani et al., 2011.
be due to inexperienced personnel obtaining the specimen (Choi et al., 2011), or lack of a pathologist’s involvement in the procedure to determine if diagnostic tissue has been obtained (ACR and ARRS, 2010; Gomez-Macias et al., 2009). The complexity involved in consistently obtaining adequate specimens requires communication throughout the entire health care team, including clinicians, surgeons, radiologists, and laboratory professionals, as well as consideration of the unique clinical conditions of each patient (Yamaguchi et al., 2012). The National Academies of Sciences, Engineering, and Medicine report Improving Diagnosis in Health Care also
emphasized the need for a team-based approach to reduce errors in the steps performed outside the clinical laboratory, particularly test ordering, specimen acquisition, and result interpretation (NASEM, 2015).
Across clinical practice settings, a variety of administrative, procedural, and billing issues arise when requesting biomarker tests on tissue specimens, particularly a test from a laboratory outside the institution with limited interaction with the practice. For example, typically several administrative steps are involved in establishing the relationship, including at least a brief vetting process regarding regulatory and compliance issues, or a more extensive due diligence (e.g., if the laboratory is a start-up); a formal, signed Laboratory Services Agreement12 detailing many aspects of the relationship also may be necessary. In addition, clinical questions or concerns may arise (e.g., in what specific instances patient safety might override tissue requirements for testing), which may be best discussed in a multidisciplinary group meeting, or may even warrant establishing a dedicated workgroup.
When testing involves biopsy procedures to obtain tissue, clear communication regarding the tissue requirements for biomarker tests is necessary to reduce the incidence of inadequate or insufficient specimens, and to ensure the efficiency of the entire process among all involved health care providers. Each clinical laboratory defines its own processes and procedures for the majority of tests based on the most appropriate biomarker testing technologies (e.g., next-generation sequencing platforms); cancer biomarker tests for molecularly targeted therapies, for example, may be performed on specimens with varying requirements13 (Dietel et al., 2015; IOM, 2015). Therefore, providers need to discuss the testing methodology and tissue or specimen requirements with their onsite pathologist and laboratory colleagues prior to specimen collection, and not assume they possess knowledge of the specimen requirements for a novel biomarker test. Also, special processes may be required for specimen handling (e.g., specific preservative or handling in the “gross room”14), particularly for fresh or flash-frozen tissue, which may vary with tissue type and specific biomarker test (Hatzis et al., 2011; Shabihkhani et al., 2014). Regardless of specific test requirements, requisition forms and any other required paperwork should be obtained from the specialty laboratory in advance of
___________________
12 These contractual agreements may define provided services and related standards, procedures for specimen custody, confidentiality, compensation, indemnification, and other miscellaneous provisions related to the business relationship.
13 Specimen requirements may include total tissue volume, tumor tissue volume, overall cellularity, percentage of viable cancer cells, tissue types, specimen format, fixative, and others.
14 The gross room is the area where specimens from the operating room are transferred and processed for pathological review.
specimen collection and reviewed by ordering clinicians and pathologists. Any required shipping kits or labels should be obtained well in advance and made available appropriately (e.g., to the histology laboratory or to the referral laboratory staff).15
A role exists for professional societies to help ensure the integrity of biomarker testing for molecularly targeted therapies, through the development of specimen acquisition and testing guidelines for the most frequently ordered tests (Dietel et al., 2015; Schilsky, 2014). Similarly, the Improving Diagnosis in Health Care report recommended that payers provide coverage for time spent by pathologists advising health care providers on the selection, use, and interpretation of tests, which are not currently coded or covered (NASEM, 2015). To ensure the performance and accuracy of biomarker tests for molecularly targeted therapies, the committee recommends that professional organizations and accrediting entities should develop, and health care institutions and providers should implement, standards for specimen requirements, handling, and documentation (see Recommendation 6a) through an interdisciplinary effort including pathologists, interventionalists, surgeons, and other relevant experts (Recommendation 9a). Health care professionals who collect, process, and handle patient biomaterials for biomarker testing should ensure that adequate tissue is acquired to perform all necessary testing so patients are protected from unnecessary or repeated procedures, and that specimens are properly handled, with documentation in the EHR and the laboratory information system.
In making this recommendation, the committee looked to existing collaborative models of generating standards for new therapies. The Foundation for the Accreditation of Cellular Therapy (FACT) recently released common standards for cellular transplantation services, including a thorough discussion of personnel qualifications, quality controls, procedures, and processes for handling and storing cellular therapy products (FACT, 2015). The FACT standards also point out that institutions should examine their specific policies and procedures and determine what adjustments should be made or whether additional or different standards should apply. Similarly for biomarker tests for molecularly targeted therapies, the variability in specimen requirements and testing methodologies, even between tests for the same biomarker as well the number of tests needed to be performed on a given specimen, will require development of interdisciplinary best practices and workflows to ensure consistent acquisition of high-quality specimens to meet the testing needs of patients and health care providers. The NCI’s Best Practices for Biospecimen Resources could
___________________
15 Additionally, billing and reimbursement should be clarified on both sides—for the testing lab, the sending institution, and sometimes even for the ordering clinician’s office.
serve as a useful starting point for development of such interdisciplinary institutional standards (NCI, 2007). Although these general guidelines and best practices will be useful, the requirements for each specific test will need to be addressed at an institutional level.
The committee considers patient safety the ultimate goal of standardized specimen acquisition and testing. Specimens that are insufficient for all ordered tests hamper timely access to the initiation of therapy, which is critical in diseases such as cancer. Repeat procedures to acquire sufficient tissue put patients at unnecessary risk of complications. To ensure compliance with the professional standards generated in Recommendation 9a, the committee additionally recommends that the National Quality Forum (NQF) should develop quality measures that assess unnecessary repeat specimen collections (Recommendation 9b). NQF is a nonprofit organization that convenes working groups to develop and endorse quality-related measures, with the goal of improving health outcomes and the efficiency of health care delivery (NQF, 2015). NQF is particularly suited to developing the quality measure recommended by the committee, given the diverse range of stakeholders represented on the forum (including the pharmaceutical and diagnostics industries, regulatory agencies, payers, health care providers, and patients and their advocates).
CLINICAL PRACTICE GUIDELINES
The relationship between biomarkers and health outcomes can be complex. An IOM report concluded that the optimal clinical use of biomarkers depended on contextual analysis of the evidence, and further that:
It is most essential that this analysis be carried out by a panel of experts, as scientific and medical judgment is necessary to weigh the possible advantages and disadvantages of the proposed biomarker use. These evaluations should take place on a per use basis, because use depends on the context of use proposed and because knowledge and technology continually evolve. (IOM, 2010a, p. 10)
Molecularly targeted therapies represent promising treatment options for patients suffering from the conditions these drugs are approved to treat; however, the ability for biomarker test results to suggest the use of molecularly targeted therapies in other conditions (particularly in oncology) also represents a potential challenge for health care providers. For example, off-label use in oncology is relatively common and a small but growing number of oncologists choose to select treatments beyond their FDA-approved indications (i.e., “off-label”) based on emerging biomarker test results (e.g., treatment with erlotinib in patients who have exhausted standard treatment options, but whose cancer contains epidermal growth
factor receptor [EGFR] mutations) (Carlson et al., 2015; Conti et al., 2013; Krzyzanowska, 2013). However, the potential benefit of selecting treatment based on emerging biomarker tests remains uncertain, though controlled clinical trials are under way (Andre et al., 2014; Le Tourneau et al., 2015) (see also Box 4-2 in Chapter 4). Clinical practice guidelines (CPGs), generated by professional and other organizations, attempt to meet this need for guidance and distinguish clinically meaningful options that clinicians and patients can consult when making health care decisions.
This is of particular importance given the rapid rate of technological innovation in the field of precision medicine, which will continue to outpace physician knowledge of interpretation and use of biomarker tests, driving both increased reliance on CPGs (Schully et al., 2015) and increased pressure for more frequent guideline updates. The inclusion of a biomarker test and corresponding molecularly targeted therapy in a trustworthy CPG can lead to more effective use in clinical practice. Research is beginning to assess adherence rates, and the impact on survival and quality of life associated with adherence to certain guidelines in clinical conditions such as breast (Cloud et al., 2015; Henry et al., 2014) and ovarian cancers (Lee et al., 2015). However, while inclusion in a CPG may suggest a biomarker test and corresponding targeted therapy have clinical utility when used together, formal assessment of clinical utility is beyond the scope of CPG development.
The IOM report Clinical Practice Guidelines We Can Trust defines CPGs as “statements that include recommendations intended to optimize patient care that are informed by a systematic review of the evidence and an assessment of the benefits and harms of alternative care options” (IOM, 2011a, p. 4). The IOM report established standards for generating trustworthy CPGs (see Table 5-1), and a companion IOM report outlined the requirements for performing comprehensive systematic reviews of evidence to inform guideline development (IOM, 2011b). The IOM’s CPG report also detailed challenges in existing guidelines development, including weak evidentiary standards, lack of methodological transparency, concerns with conflict of interest (COI), and inconsistency among related guidelines. The report stated that guidelines that successfully surmounted these concerns would “ultimately . . . give users confidence that guidelines are based on best available evidence, largely free from bias, clear about the purpose of recommendations to individual patients, and therefore trustworthy” (IOM, 2011a, p. 77).
Methodological transparency and COI continue to be refined on the organizational level, with many expert panels outlining the criteria used in the selection of data sources, the categorization of varying levels of evidence, the rationale for resulting recommendations, and the panel’s composition with various methods for disclosure of potential COI (ACCF
TABLE 5-1 IOM Standards for Development of Trustworthy CPGs
| Focus of Standard | Application to CPG Development |
|---|---|
| Transparency | Development and funding processes should be detailed and publicly accessible. |
| Management of Conflict of Interest | Whenever possible GDG members should not have COI; relevant COI should be disclosed, with potential for divestment if such interests could be affected by CPGs. |
| Panel Composition | Panels should be balanced, multidisciplinary, and inclusive of patients and patient advocates. |
| Systematic Review | GDGs should use systematic reviews that meet IOM standards.a |
| Rating of Evidence and Strength of Recommendation |
Each recommendation should have:
|
| Articulation of Recommendation | A standardized and precise description of recommended action and applicable circumstances should be given; strong recommendations should be phrased to enable evaluation of compliance. |
| External Review | Reviewers should include a full spectrum of relevant stakeholders, and should be kept confidential unless waived; all reviewer comments should be considered; and a review draft should be made available for public comment. |
| Updating | Publication date, pertinent systematic review dates, and proposed future review dates should be documented. Literature should be monitored regularly to identify emergence of new and relevant evidence and to evaluate continued validity of the CPG. |
NOTE: COI = conflict of interest; CPG = clinical practice guideline; GDG = guideline developing group; IOM = Institute of Medicine.
SOURCE: Adapted from IOM, 2011a.
and AHA, 2010; ASCO, 2015; NCCN, 2015). However, despite the need to incorporate higher quality evidence into guideline development related to biomarker testing for molecularly targeted therapies, progress has been slow. The Centers for Disease Control and Prevention’s Evaluation of Genomic Applications in Practice and Prevention (EGAPP) Working
Group published a summary of lessons learned from the group’s publication of eight recommendation statements between 2005 and 2014. Among the testing scenarios examined, many of which addressed testing related to targeted therapies, EGAPP noted a lack of consistent demonstration of analytic and clinical validity, as well as clinical utility, resulting from the difficulty of applying traditional evidence review to rapidly advancing fields (Calonge et al., 2014). Similarly, Abernethy and colleagues examined the difficulty associated with traditional systematic review in technology assessment for off-label use of targeted therapies in cancer. Particularly problematic was the widespread heterogeneity of reviewed studies and data, and the tendency of review cut-off dates to exclude potentially practice-changing new research (Abernethy et al., 2010).
Recent CPG generation efforts reflect these difficulties. For example, a review of 16 current CPGs published jointly between the American College of Cardiology (ACC) and the American Heart Association (AHA) found that nearly half were based not on systematic review of evidence, but instead on expert opinion, case studies, or standards of care (Tricoci et al., 2009). The National Comprehensive Cancer Network’s (NCCN’s) default level of evidence category for recommendations (2A) similarly relies on lower-level evidence from “indirect comparisons among randomized trials, Phase II or non-randomized trials, or in many cases, on limited data from multiple smaller trials, retrospective studies, or clinical observations” (NCCN, 2015; Poonacha and Go, 2011) (see Table 5-2 for a summary of NCCN recommendations for biomarker testing to direct targeted therapy in common cancers). ASCO also publishes CPGs and states that “few guideline questions can be directly or completely answered only considering the evidence. Interpretation and extrapolation of evidence are often necessary” (ASCO, 2015).
Different interpretations of the relative value and harm of an intervention in the context of non-definitive evidence can result in inconsistency among related guidelines (IOM, 2011a). In contrast with NCCN guidelines, for example, a recent ASCO endorsement of a joint CAP/International Society for the Study of Lung Cancer/Association of Molecular Pathology (AMP) guideline related to testing for non-small-cell lung cancer patients prioritized EGFR and ALK testing above all other markers, but did explain that emerging tests for ROS1 and RET are under investigation (Leighl et al., 2014). Similarly, a recent draft guidance document for molecular testing in colorectal cancer (released jointly by the American Society for Clinical Pathology, CAP, ASCO, and AMP) did not consider the evidence sufficient to recommend testing for BRAF mutations for predicting response to anti-EGFR therapy in colon cancer (BRAF and KRAS mutations are considered mutually exclusive in colon cancer), which conflicts with the current NCCN recommendation (AMP, 2015).
TABLE 5-2 NCCN Guidelines for Clinical Use of Predictive Biomarker Tests in Common Cancers
| Cancer Type | Biomarker (Gene Symbol) | Quality of Evidence* |
|---|---|---|
| Non-Small Cell Lung Cancer | ALK | 1, 2A |
| EGFR | 1, 2A | |
| ROS1 | 2A | |
| KRAS | 2A | |
| BRAF | 2A | |
| ERBB2 (HER2) | 2B | |
| MET | 2A | |
| RET | 2A | |
| Breast Cancer | ESR1 | 2A |
| PGR | 2A | |
| ERBB2 (HER2) | 1 | |
| Prostate Cancer | - | - |
| Colon Cancer | NRAS/KRAS | 2A |
| BRAF | 2A | |
NOTES: * Quality of Evidence can differ between mutations within a target biomarker; Category 1: Based on high-level evidence, there is uniform NCCN consensus that the intervention is appropriate; Category 2A: Based on lower-level evidence, there is uniform NCCN consensus that the intervention is appropriate; Category 2B: Based on lower-level evidence, there is NCCN consensus that the intervention is appropriate.
NCCN = National Comprehensive Cancer Network.
SOURCE: NCCN Biomarkers Compendium, http://www.nccn.org/professionals/biomarkers/default.aspx (accessed June 2, 2015).
Compounding the variability in evidence and the inconsistency among CPG recommendations is the fact that the studies on which CPGs are based are often conducted on more restricted populations than those to which the CPGs are applied (Treweek et al., 2015), particularly in molecularly driven trials (Kim et al., 2015). This can lead CPGs to focus too narrowly on clinical conditions without considering real-world patient scenarios, where comorbidities are commonplace (Sniderman and Furberg, 2009). Efforts to reduce bias and redundancy and better direct research and clinical care have fueled calls for centralized CPG generation under the auspices of the Agency for Healthcare Research and Quality (AHRQ) or the U.S. Preventive Services Task Force (Shaneyfelt and Centor, 2009). The AHRQ’s National Guidelines Clearinghouse16 currently aggregates guidelines, enables comparison, and has a limited number of syntheses drawn from multiple guidelines in a related setting.
___________________
Given the complexity of precision medicine, there is a growing need for clinician guidance from professional organizations (Manolio et al., 2014). Clinical decision making, particularly in cancer genomics, will increasingly benefit from comparing the incremental effectiveness of therapeutic options, a process that is necessarily interdisciplinary (Goddard et al., 2012; Simonds et al., 2013). The committee endorses the existing IOM standards for trustworthy clinical practice guidelines shown in Table 5-1 (IOM, 2011a), and further recommends that guideline-developing organizations (e.g., CAP, AMP, the American College of Medical Genetics and Genomics [ACMG], ACC, NCCN, AHA, ASCO, the American College of Physicians [ACP], and others) should expand interdisciplinary collaborations to develop integrated guidelines on the appropriate use of biomarker tests for molecularly targeted therapies (Recommendation 10). Guidelines should be updated regularly and at intervals appropriate to advances in the field, widely disseminated, user-friendly, and developed with patient participation. They should conform to standards articulated by authoritative groups, including the IOM and Guidelines International Network. Additionally, guideline developers should consider the evolving clinical utility evidence, relative to the standards discussed in Recommendation 1, and from the proposed rapid learning system for biomarker tests.
Collaboration among guideline-developing organizations has the potential to save time and resources if redundant guideline development is avoided; it also adds clarity to clinical decision making by reducing guideline inconsistency. The need for cooperative guideline development is further highlighted by the fact that no single group can cover the extensive field of genomics as it relates to clinical practice (Schully et al., 2015). The National Guideline Clearinghouse should expand its work in reviewing and rating guidelines to help ensure this collaboration is effectively improving guidelines related to biomarker tests for molecularly targeted therapies. Additionally, EHR vendors and purchasers should ensure that recommendations from high-quality guidelines are available within the EHR at the point of care (see Recommendation 6). Frequently updated guidelines should serve as input to the iterative updating of test labeling proposed in Recommendations 2 and 3.
SUMMARY AND RECOMMENDATIONS
Patients in remote or underserved areas without access to a larger health care center or academic medical center may have limited access to adequate biomarker testing technology. In addition, there is a significant lack of genetic/genomic knowledge among patients and health care providers. Rapid learning systems present a unique opportunity to
ensure evidence-based medical practice is widely available and continuously refined. Multidisciplinary conferences and specialty tumor boards have been shown to be useful vehicles for knowledge sharing and learning among health care providers. Genetic counselors have an important role to play as members of the care team, working to inform physicians about interpretation of test results and to explain biomarker test results to patients.
Goal 8: Promote equity in access to biomarker tests for molecularly targeted therapies and the expertise for effective use of the results in clinical decision making.
Recommendation 8a: Agencies that fund the development or evaluation of biomarkers should include funding to identify and overcome barriers to promote equity, access, and public understanding of precision medicine.
- Potential challenges include but are not limited to: economic factors, cultural/ethnic heterogeneity, geographic diversity, and the complexity of precision medicine.
Recommendation 8b: The Secretary of the Department of Health and Human Services and the Centers for Medicare & Medicaid Services (CMS) should conduct demonstration projects to enable and assess the effectiveness of collaboration between community health care providers and larger health care centers and/or academic medical centers to be part of a rapid learning system.
The demonstration projects should examine:
- Use of reimbursement incentives by CMS for the multidisciplinary collection and review of patient data with clinical recommendations, using distance technology or telemedicine.
- Reimbursement by CMS for genetic counseling services.
Recommendation 8c: Licensing and specialty boards should ensure that health care professionals have and maintain competencies needed for effective use of biomarker tests for molecularly targeted therapies.
- Providers should demonstrate competency in communicating with patients about biomarker tests for molecularly targeted therapies.
Inadequate tissue may be collected from a patient, requiring repeat biopsy procedures to obtain specimens sufficient for testing and exposing
the patient to unnecessary risk. Uniform standards are needed regarding specimen acquisition, handling, and subsequent documentation in the EHR and/or laboratory information system.
Recommendation 9a: Professional organizations and accrediting entities should develop, and health care institutions and providers should implement, standards for specimen requirements, handling, and documentation (see Recommendation 6a) through an interdisciplinary effort, including pathologists, interventionalists, surgeons, and other relevant experts.
- Health care professionals who collect, process, and handle (label and ship) patient biomaterials for biomarker testing should ensure that adequate tissue is acquired to perform all necessary testing; that patients are protected from unnecessary/repeated procedures; and that samples are properly handled, with documentation in the electronic health record and/or the laboratory information system.
Recommendation 9b: The National Quality Forum should develop quality measures that assess unnecessary repeat specimen collections.
CPGs exist for some biomarker tests for molecularly targeted therapies, but may not be frequently updated, may not be sufficiently user-friendly or clinically relevant, and often conflict with related guidelines. Increasingly, a broader base of interdisciplinary expertise is needed to generate trustworthy guidelines related to biomarker tests for molecularly targeted therapies. Consistent with the committee’s vision of a learning health care system, CPGs serve an important dual purpose, both for clinical decision making and for data input for decisions regarding test and drug labeling.
Goal 10: Improve the processes for developing and updating clinical practice guidelines for the effective use of biomarker tests for molecularly targeted therapies.
Recommendation 10: Guideline-developing organizations (e.g., the College of American Pathologists, Association for Molecular Pathology, American College of Medical Genetics and Genomics, American College of Cardiology, National Comprehensive Cancer Network, American Heart Association, American Society of Clinical Oncology, American College of Physicians, and others) should expand interdisciplinary collaborations to develop integrated
guidelines on the appropriate use of biomarker tests for molecularly targeted therapies.
- Guidelines should be updated regularly and at intervals appropriate to advances in the field, widely disseminated, user-friendly, and developed with patient participation. They should conform to standards articulated by authoritative groups, including the Institute of Medicine and Guidelines International Network.
- Guideline developers should consider the evolving clinical utility evidence, relative to the standards discussed in Recommendation 1, and from the proposed rapid learning system for biomarker tests.
- The National Guideline Clearinghouse should expand its work in reviewing and rating guidelines.
- Electronic health record (EHR) vendors/EHR purchasers should ensure that recommendations from high-quality guidelines are available within the EHR at the point of care (see Recommendation 6).
- Frequently updated guidelines should serve as input to the iterative updating of test and drug labeling by the integrated federal review process (see Recommendation 2).
REFERENCES
Abel, G. A., H. J. Burstein, N. D. Hevelone, and J. C. Weeks. 2009. Cancer-related direct-to-consumer advertising: Awareness, perceptions, and reported impact among patients undergoing active cancer treatment. Journal of Clinical Oncology 27(25):4182-4187.
Abernethy, A., R. R. Coeytaux, K. Carson, D. McCrory, S. Y. Barbour, M. Gradison, R. J. Irvine, and J. L. Wheeler. 2010. Report on the evidence regarding off-label indications for targeted therapies used in cancer treatment. Rockville, MD: Agency for Healthcare Research and Quality.
ABMS (American Board of Medical Specialties). 2014. Standards for the ABMS program for Maintenance of Certification (MOC). http://www.abms.org/media/1109/standards-for-the-abms-program-for-moc-final.pdf (accessed January 15, 2016).
ABMS. 2015. Based on core competencies. http://www.abms.org/board-certification/a-trusted-credential/based-on-core-competencies (accessed January 15, 2016).
Abraham, L., B. M. Geller, B. C. Yankaskas, E. J. Bowles, L. S. Karliner, S. H. Taplin, and D. L. Miglioretti. 2009. Accuracy of self-reported breast cancer among women undergoing mammography. Breast Cancer Research and Treatment 118(3):583-592.
ACCF (American College of Cardiology Foundation) and AHA (American Heart Association). 2010. Methodology manual and policies from the ACCF/AHA Task Force on Practice Guidelines. Journal of the American College of Cardiology 62(16):e147-e239.
ACGME (American Council for Graduate Medical Education). 2015. Accreditation Council for Graduate Medical Education. www.acgme.org/acgmeweb (accessed January 15, 2016).
ACR (American College of Radiology) and ARRS (American Roentgen Ray Society). 2010. On-site pathology improves the inadequacy rate of ultrasound-guided thyroid biopsies. www.sciencedaily.com/releases/2010/05/100505091628.htm (accessed July 28, 2015).
ACS (American College of Surgeons). 2012. Cancer program standards 2012: Ensuring patient-centered care. https://www.facs.org/~/media/files/quality%20programs/cancer/coc/programstandards2012.ashx (accessed October 8, 2015).
Adams-Campbell, L. L., K. H. Makambi, W. A. Frederick, M. Gaskins, R. L. Dewitty, and W. McCaskill-Stevens. 2009. Breast cancer risk assessments comparing Gail and CARE models in African-American women. The Breast Journal 15(Suppl 1):S72-S75.
Ahmed, K. A., W. J. Fulp, A. E. Berglund, S. E. Hoffe, T. J. Dilling, S. A. Eschrich, R. Shridhar, and J. F. Torres-Roca. 2015. Differences between colon cancer primaries and metastases using a molecular assay for tumor radiation sensitivity suggest implications for potential oligometastatic SBRT patient selection. International Journal of Radiation Oncology*Biology*Physics 92(4):837-842.
Aisner, D. L., and C. B. Marshall. 2012. Molecular pathology of non-small cell lung cancer: A practical guide. American Journal of Clinical Pathology 138(3):332-346.
AMP (Association for Molecular Pathology). 2015. ASCP/CAP/AMP/ASCO evaluation of molecular markers for colorectal cancer draft recommendations. http://www.amp.org/committees/clinical_practice/CRCOpenComment.cfm (accessed October 4, 2015).
Ancker, J. S., and D. Kaufman. 2007. Rethinking health numeracy: A multidisciplinary literature review. Journal of the American Medical Informatics Association 14(6):713-721.
Anderson, A. A., D. A. Scheufele, D. Brossard, and E. A. Corley. 2011. The role of media and deference to scientific authority in cultivating trust in sources of information about emerging technologies. International Journal of Public Opinion Research 24(2):225-237.
Andre, F., T. Bachelot, F. Commo, M. Campone, M. Arnedos, V. Dieras, M. Lacroix-Triki, L. Lacroix, P. Cohen, D. Gentien, J. Adelaide, F. Dalenc, A. Goncalves, C. Levy, J. M. Ferrero, J. Bonneterre, C. Lefeuvre, M. Jimenez, T. Filleron, and H. Bonnefoi. 2014. Comparative genomic hybridisation array and DNA sequencing to direct treatment of metastatic breast cancer: A multicentre, prospective trial (SAFIR01/UNICANCER). Lancet Oncology 15(3):267-274.
ASCO (American Society of Clinical Oncology). 2015. Guideline development process. http://www.instituteforquality.org/guideline-development-process (accessed July 20, 2015).
ATA (American Telemedicine Association). 2014. Update on CMS payment decisions—two steps forward, one back. http://www.americantelemed.org/news-landing/2014/11/07/update-on-cms-payment-decisions---two-steps-forward-one-back#.Vhf4Yd9VhHx (accessed October 5, 2015).
ATA. 2015. What is telemedicine? http://www.americantelemed.org/about-telemedicine/what-is-telemedicine#.Vhf6bd9VhHw (accessed October 5, 2015).
Averbuch, S., K. Emancipator, I. McCaffery, A. McElhinny, D. Stanforth, J. Walker, and D. Ward. 2015. A blueprint proposal for companion diagnostic comparability. Silver Spring, MD: Food and Drug Administration.
Banegas, M. P., M. H. Gail, A. LaCroix, B. Thompson, M. E. Martinez, J. Wactawski-Wende, E. M. John, F. A. Hubbell, S. Yasmeen, and H. A. Katki. 2012. Evaluating breast cancer risk projections for Hispanic women. Breast Cancer Research and Treatment 132(1):347-353.
Batmangelich, S., and S. Adamowski. 2004. Maintenance of certification in the United States: A progress report. Journal of Continuing Education in the Health Professions 24(3):134-138.
Blayney, D. W. 2013. Tumor boards (team huddles) aren’t enough to reach the goal. Journal of the National Cancer Institute 105(2):82-84.
Bowen, A., and A. Casadevall. 2015. Increasing disparities between resource inputs and outcomes, as measured by certain health deliverables, in biomedical research. Proceedings of the National Academy of Sciences of the United States of America 112(36):11335-11340.
Brokaw, T. 2015. A lucky life interrupted: A memoir of hope. New York: Random House Publishing Group.
Calonge, N., R. D. Klein, A. O. Berg, J. S. Berg, K. Armstrong, J. Botkin, D. Campos-Outcalt, B. Djulbegovic, N. L. Fisher, T. G. Ganiats, J. E. Haddow, M. Hayes, A. Janssens, C. Kaye, D. O. Lyman, K. Offit, S. G. Pauker, K. A. Phillips, M. Piper, C. S. Richards, J. A. Scott, O. L. Strickland, S. Teutsch, S. R. Tunis, D. L. Veenstra, M. S. Williams, D. T. Zallen, and the EGAPP Working Group. 2014. The EGAPP initiative: Lessons learned. Genetics in Medicine 16(3):217-224.
Carlson, J. J., B. Ely, R. N. Hansen, and G. H. Lyman. 2015. Precision oncology: Molecular testing and targeted treatment beyond indication. Journal of Clinical Oncology 33(15).
Caulfield, T., and A. L. McGuire. 2012. Direct-to-consumer genetic testing: Perceptions, problems, and policy responses. Annual Review of Medicine 63:23-33.
CCHPCA (Center for Connected Health Policy). 2015. What is telehealth? http://cchpca.org/ what-is-telehealth (accessed October 5, 2015).
Chadwick, R. 2013. Ethical issues in personalized medicine. Drug Discovery Today: Therapeutic Strategies 10(4): e171-e174.
Choi, S. H., K. H. Han, J. H. Yoon, H. J. Moon, E. J. Son, J. H. Youk, E. K. Kim, and J. Y. Kwak. 2011. Factors affecting inadequate sampling of ultrasound-guided fine-needle aspiration biopsy of thyroid nodules. Clinical Endocrinology 74(6):776-782.
Cloud, A. J., A. Thai, Y. Liao, and M. B. Terry. 2015. The impact of cancer prevention guideline adherence on overall mortality in a high-risk cohort of women from the New York site of the Breast Cancer Family Registry. Breast Cancer Research and Treatment 149(2):537-546.
CMS (Centers for Medicare & Medicaid Services). 2014. Telehealth services: Rural health fact sheet series. https://www.cms.gov/Outreach-and-Education/Medicare-Learning-Network-MLN/MLNProducts/downloads/RuralHlthClinfctsht.pdf (accessed October 4, 2015).
CMS. 2015. Chronic care management services. https://www.cms.gov/Outreach-andEducation/Medicare-Learning-Network-MLN/MLNProducts/Downloads/ChronicCareManagement.pdf (accessed October 5, 2015).
Coleman, C. N., T. S. Lawrence, and D. G. Kirsch. 2014. Enhancing the efficacy of radiation therapy: Premises, promises, and practicality. Journal of Clinical Oncology 32(26):2832-2835.
Condit, C. 2007. How culture and science make race “genetic”: Motives and strategies for discrete categorization of the continuous and heterogeneous. Literature and Medicine 26(1):240-268.
Condit, C. M., N. Ofulue, and K. M. Sheedy. 1998. Determinism and mass-media portrayals of genetics. The American Journal of Human Genetics 62(4):979-984.
Conti, R. M., A. C. Bernstein, V. M. Villaflor, R. L. Schilsky, M. B. Rosenthal, and P. B. Bach. 2013. Prevalence of off-label use and spending in 2010 among patent-protected chemotherapies in a population-based cohort of medical oncologists. Journal of Clinical Oncology 31(9):1134-1139.
Cox, K. A., A. P. Smith, and M. Lichtveld. 2012a. A competency-based approach to expanding the cancer care workforce part III—improving cancer pain and palliative care competency. Journal of Cancer Education 27(3):507-514.
Cox, S. L., A. I. Zlot, K. Silvey, D. Elliott, T. Horn, A. Johnson, and R. F. Leman. 2012b. Patterns of cancer genetic testing: A randomized survey of Oregon clinicians. Journal of Cancer Epidemiology 2012:294730.
Crews, K. R., J. K. Hicks, C. H. Pui, M. V. Relling, and W. E. Evans. 2012. Pharmacogenomics and individualized medicine: Translating science into practice. Clinical Pharmacology & Therapeutics 92(4):467-475.
Daniels, N. 2013. Justice and access to health care. In The Stanford Encyclopedia of Philosophy (Spring 2013 Edition), Edward N. Zalta (ed.). Stanford, CA: Stanford University.
Davis, P. B. 2015. Another beginning for cystic fibrosis therapy. New England Journal of Medicine 373(3):274-276.
de Gramont, A., S. Watson, L. M. Ellis, J. Rodon, J. Tabernero, A. de Gramont, and S. R. Hamilton. 2015. Pragmatic issues in biomarker evaluation for targeted therapies in cancer. Nature Reviews Clinical Oncology 12(4):197-212.
DeNavas-Walt, C., B. D. Proctor, and J. C. Smith. 2013. Income, poverty, and health insurance coverage in the United States, Current Population Reports P60-245. Washington, DC: U.S. Government Printing Office.
Dietel, M., K. Johrens, M. V. Laffert, M. Hummel, H. Blaker, B. M. Pfitzner, A. Lehmann, C. Denkert, S. Darb-Esfahani, D. Lenze, F. L. Heppner, A. Koch, C. Sers, F. Klauschen, and I. Anagnostopoulos. 2015. A 2015 update on predictive molecular pathology and its role in targeted cancer therapy: A review focussing on clinical relevance. Cancer Gene Therapy 22(9):417-430.
Dinan, M. A., X. Mi, S. D. Reed, B. R. Hirsch, G. H. Lyman, and L. H. Curtis. 2015. Initial trends in the use of the 21-gene recurrence score assay for patients with breast cancer in the Medicare population, 2005-2009. Journal of the American Medical Association Oncology 1(2):158-166.
Dolgin, E. 2016. Shoddy biopsies deny cancer patients a shot at personalized treatment. http://www.statnews.com/2016/01/22/precision-medicine-cancer-biopsies (accessed January 21, 2016).
Dominguez, F. J., C. Lawrence, E. F. Halpern, B. Drohan, G. Grinstein, D. M. Black, B. L. Smith, M. A. Gadd, M. Specht, D. B. Kopans, R. H. Moore, S. S. Hughes, C. A. Roche, and K. S. Hughes. 2007. Accuracy of self-reported personal history of cancer in an outpatient breast center. Journal of Genetic Counseling 16(3):341-345.
Dotan, E., T. Li, M. J. Hall, N. J. Meropol, J. R. Beck, and Y. N. Wong. 2014. Oncologists’ response to new data regarding the use of epidermal growth factor receptor inhibitors in colorectal cancer. Journal of Oncology Practice 10(5):308-314.
Drazen, J. M., and D. F. Weinstein. 2010. Considering recertification. New England Journal of Medicine 362(10):946-947.
Dusetzina, S. B., E. Basch, and N. L. Keating. 2015. For uninsured cancer patients, outpatient charges can be costly, putting treatments out of reach. Health Affairs 34(4):584-591.
Dworkin, R. 1981. What is equality? Part 2: Equality of resources. Philosophy and Public Affairs 10:185-246.
Epstein, R. M., and R. L. Street. 2007. Patient-centered communication in cancer care: Promoting healing and reducing suffering. Bethesda, MD: National Cancer Institute.
Evans, J. P., and M. J. Khoury. 2013. The arrival of genomic medicine to the clinic is only the beginning of the journey. Genetics in Medicine 15(4):268-269.
FACT (Foundation for the Accreditation of Cellular Therapy). 2015. Common standards for cellular therapies. Omaha, NE: University of Nebraska Medical Center.
Fallowfield, L., and V. Jenkins. 1999. Effective communication skills are the key to good cancer care. European Journal of Cancer 35(11):1592-1597.
FDA (Food and Drug Administration). 2015. The public health evidence for FDA oversight of laboratory developed tests: 20 case studies. Silver Spring, MD: Office of Public Health Strategy and Analysis.
Frampton, G. M., A. Fichtenholtz, G. A. Otto, K. Wang, S. R. Downing, J. He, M. Schnall-Levin, J. White, E. M. Sanford, P. An, J. Sun, F. Juhn, K. Brennan, K. Iwanik, A. Maillet, J. Buell, E. White, M. Zhao, S. Balasubramanian, S. Terzic, T. Richards, V. Banning, L. Garcia, K. Mahoney, Z. Zwirko, A. Donahue, H. Beltran, J. M. Mosquera, M. A. Rubin, S. Dogan, C. V. Hedvat, M. F. Berger, L. Pusztai, M. Lechner, C. Boshoff, M. Jarosz, C. Vietz, A. Parker, V. A. Miller, J. S. Ross, J. Curran, M. T. Cronin, P. J. Stephens, D. Lipson, and R. Yelensky. 2013. Development and validation of a clinical cancer genomic profiling test based on massively parallel DNA sequencing. Nature Biotechnology 31(11):1023-1031.
Freedman, A. N., L. Wideroff, L. Olson, W. Davis, C. Klabunde, K. P. Srinath, B. B. Reeve, R. T. Croyle, and R. Ballard-Barbash. 2003. US physicians’ attitudes toward genetic testing for cancer susceptibility. American Journal of Medical Genetics Part A 120A(1):63-71.
FTC (Federal Trade Commission). 2014. Consumer information: Direct-to-consumer genetic tests. https://www.consumer.ftc.gov/articles/0166-direct-consumer-genetic-tests (accessed September 5, 2015).
GAO (Government Accountability Office). 2006. Nutrigenetic testing: Tests purchased from four web sites mislead consumers. http://www.gao.gov/new.items/d06977t.pdf (accessed June 12, 2015).
GAO. 2010. Direct-to-consumer genetic tests: Misleading test results are further complicated by deceptive marketing and other questionable practices. http://www.gao.gov/new.items/d10847t.pdf (accessed June 12, 2015).
Garraway, L. A., J. Verweij, and K. V. Ballman. 2013. Precision oncology: An overview. Journal of Clinical Oncology 31(15):1803-1805.
Goddard, K. A., W. A. Knaus, E. Whitlock, G. H. Lyman, H. S. Feigelson, S. D. Schully, S. Ramsey, S. Tunis, A. N. Freedman, M. J. Khoury, and D. L. Veenstra. 2012. Building the evidence base for decision making in cancer genomic medicine using comparative effectiveness research. Genetics in Medicine 14(7):633-642.
Golbeck, A. L., C. R. Ahlers-Schmidt, A. M. Paschal, and S. E. Dismuke. 2005. A definition and operational framework for health numeracy. American Journal of Preventive Medicine 29(4):375-376.
Goldman, L., A. Goroll, and B. Kessler. 2010. Do not enroll in the current MOC program. New England Journal of Medicine 362:950-952.
Gomez-Macias, G. S., R. Garza-Guajardo, J. Segura-Luna, and O. Barboza-Quintana. 2009. Inadequate fine needle aspiration biopsy samples: Pathologists versus other specialists. Cytojournal 6:9.
Gonon, F., J. P. Konsman, D. Cohen, and T. Boraud. 2012. Why most biomedical findings echoed by newspapers turn out to be false: The case of attention deficit hyperactivity disorder. PLoS ONE 7(9):e44275.
Gray, S. W., K. Hicks-Courant, A. Cronin, B. J. Rollins, and J. C. Weeks. 2014. Physicians’ attitudes about multiplex tumor genomic testing. Journal of Clinical Oncology 32(13):1317-1323.
Gray, S. W., A. Cronin, E. Bair, N. Lindeman, V. Viswanath, and K. A. Janeway. 2015. Marketing of personalized cancer care on the Web: An analysis of Internet websites. Journal of the National Cancer Institute 107(5).
Guttmacher, A. E., M. E. Porteous, and J. D. McInerney. 2007. Educating health-care professionals about genetics and genomics. Nature Reviews Genetics 8(2):151-157.
Hasnain-Wynia, R., D. W. Baker, D. Nerenz, J. Feinglass, A. C. Beal, M. B. Landrum, R. Behal, and J. S. Weissman. 2007. Disparities in health care are driven by where minority patients seek care: Examination of the hospital quality alliance measures. Archives of Internal Medicine 167(12):1233-1239.
Hatzis, C., H. Sun, H. Yao, R. E. Hubbard, F. Meric-Bernstam, G. V. Babiera, Y. Wu, L. Pusztai, and W. F. Symmans. 2011. Effects of tissue handling on RNA integrity and microarray measurements from resected breast cancers. Journal of the National Cancer Institute 103(24):1871-1883.
Hazin, R., and I. Qaddoumi. 2010. Teleoncology: Current and future applications for improving cancer care globally. Lancet Oncology 11(2):204-210.
Henry, N. L., D. F. Hayes, S. D. Ramsey, G. N. Hortobagyi, W. E. Barlow, and J. R. Gralow. 2014. Promoting quality and evidence-based care in early-stage breast cancer followup. Journal of the National Cancer Institute 106(4):dju034.
Hess, B. J., W. Weng, E. S. Holmboe, and R. S. Lipner. 2012. The association between physicians’ cognitive skills and quality of diabetes care. Academic Medicine 87(2):157-163.
Higgins, G. S., S. M. O’Cathail, R. J. Muschel, and W. G. McKenna. 2015. Drug radiotherapy combinations: Review of previous failures and reasons for future optimism. Cancer Treatment Reviews 41(2):105-113.
Holmboe, E. S., Y. Wang, T. P. Meehan, J. P. Tate, S. Y. Ho, K. S. Starkey, and R. S. Lipner. 2008. Association between maintenance of certification examination scores and quality of care for Medicare beneficiaries. Archives of Internal Medicine 168(13):1396-1403.
HRSA (Health Resources and Services Administration). 2015. Telehealth. http://www.hrsa.gov/ruralhealth/about/telehealth/telehealth.html (accessed October 5, 2015).
Hudis, C. A. 2007. Trastuzumab—mechanism of action and use in clinical practice. New England Journal of Medicine 357(1):39-51.
Hyman, D. M., I. Puzanov, V. Subbiah, J. E. Faris, I. Chau, J. Y. Blay, J. Wolf, N. S. Raje, E. L. Diamond, A. Hollebecque, R. Gervais, M. E. Elez-Fernandez, A. Italiano, R. D. Hofheinz, M. Hidalgo, E. Chan, M. Schuler, S. F. Lasserre, M. Makrutzki, F. Sirzen, M. L. Veronese, J. Tabernero, and J. Baselga. 2015. Vemurafenib in multiple nonmelanoma cancers with BRAF V600 mutations. New England Journal of Medicine 373(8):726-736.
Ioannidis, J. P., and O. A. Panagiotou. 2011. Comparison of effect sizes associated with biomarkers reported in highly cited individual articles and in subsequent meta-analyses. Journal of the American Medical Association 305(21):2200-2210.
IOM (Institute of Medicine). 2004. Insuring America’s health: Principles and recommendations. Washington, DC: The National Academies Press.
IOM. 2007. Cancer biomarkers: The promises and challenges of improving detection and treatment. Washington, DC: The National Academies Press.
IOM. 2009. America’s uninsured crisis: Consequences for health and health care. Washington, DC: The National Academies Press.
IOM. 2010a. Evaluation of biomarkers and surrogate endpoints in chronic disease. Washington, DC: The National Academies Press.
IOM. 2010b. Redesigning continuing education in the health professions. Washington, DC: The National Academies Press.
IOM. 2011a. Clinical practice guidelines we can trust. Washington, DC: The National Academies Press.
IOM. 2011b. Finding what works in health care: Standards for systematic reviews. Washington, DC: The National Academies Press.
IOM. 2012a. Evolution of translational omics: Lessons learned and the path forward. Washington, DC: The National Academies Press.
IOM. 2012b. Genome-based therapeutics: Targeted drug discovery and development: Workshop summary. Washington, DC: The National Academies Press.
IOM. 2012c. The role of telehealth in an evolving health care environment: Workshop summary. Washington, DC: The National Academies Press.
IOM. 2013. Delivering high-quality cancer care: Charting a new course for a system in crisis. Washington, DC: The National Academies Press.
IOM. 2015. Policy issues in the development and adoption of biomarkers for molecularly targeted cancer therapies: Workshop summary. Washington, DC: The National Academies Press.
Ithimakin, S., K. C. Day, F. Malik, Q. Zen, S. J. Dawsey, T. F. Bersano-Begey, A. A. Quraishi, K. W. Ignatoski, S. Daignault, A. Davis, C. L. Hall, N. Palanisamy, A. N. Heath, N. Tawakkol, T. K. Luther, S. G. Clouthier, W. A. Chadwick, M. L. Day, C. G. Kleer, D. G. Thomas, D. F. Hayes, H. Korkaya, and M. S. Wicha. 2013. HER2 drives luminal breast cancer stem cells in the absence of HER2 amplification: Implications for efficacy of adjuvant trastuzumab. Cancer Research 73(5):1635-1646.
Jagsi, R., K. A. Griffith, A. W. Kurian, M. Morrow, A. S. Hamilton, J. J. Graff, S. J. Katz, and S. T. Hawley. 2015. Concerns about cancer risk and experiences with genetic testing in a diverse population of patients with breast cancer. Journal of Clinical Oncology 33(14):1584-1591.
Johnson, D. B., C. Peng, and J. A. Sosman. 2015. Nivolumab in melanoma: Latest evidence and clinical potential. Therapeutic Advances in Medical Oncology 7(2):97-106.
Joseph, G., and D. Dohan. 2009. Recruiting minorities where they receive care: Institutional barriers to cancer clinical trials recruitment in a safety-net hospital. Contemporary Clinical Trials 30(6):552-559.
Kaiser Family Foundation. 2005. Public opinion snapshot on health information sources. http://www.kff.org/kaiserpolls/pomr071805oth.cfm (accessed June 24, 2015).
Kaiser Family Foundation. 2010. Generation M: Media in the lives of 8- to 18-year-olds. Menlo Park, CA: Kaiser Family Foundation.
Keating, N. L., M. B. Landrum, E. B. Lamont, S. R. Bozeman, L. N. Shulman, and B. J. McNeil. 2013. Tumor boards and the quality of cancer care. Journal of the National Cancer Institute 105(2):113-121.
Kehl, K. L., M. B. Landrum, K. L. Kahn, S. W. Gray, A. B. Chen, and N. L. Keating. 2015. Tumor board participation among physicians caring for patients with lung or colorectal cancer. Journal of Oncology Practice 11(3):e267-e278.
Kim, E. S., D. Bernstein, S. G. Hilsenbeck, C. H. Chung, A. P. Dicker, J. L. Ersek, S. Stein, F. R. Khuri, E. Burgess, K. Hunt, P. Ivy, S. S. Bruinooge, N. Meropol, and R. L. Schilsky. 2015. Modernizing eligibility criteria for molecularly driven trials. Journal of Clinical Oncology 33(25):2815-2820.
Kitamura, C., L. Zurawel-Balaura, and R. K. Wong. 2010. How effective is video consultation in clinical oncology? A systematic review. Current Oncology 17(3):17-27.
Krzyzanowska, M. K. 2013. Off-label use of cancer drugs: A benchmark is established. Journal of Clinical Oncology 31(9):1125-1127.
Kurian, A. W. 2010. BRCA1 and BRCA2 mutations across race and ethnicity: Distribution and clinical implications. Current Opinion in Obstetrics and Gynecology 22(1):72-78.
Kuroki, L., A. Stuckey, P. Hirway, C. A. Raker, C. A. Bandera, P. A. DiSilvestro, C. O. Granai, R. D. Legare, B. J. Sakr, and D. S. Dizon. 2010. Addressing clinical trials: Can the multidisciplinary tumor board improve participation? A study from an academic women’s cancer program. Gynecologic Oncology 116(3):295-300.
Lachance, C. R., L. A. Erby, B. M. Ford, V. C. Allen, Jr., and K. A. Kaphingst. 2010. Informational content, literacy demands, and usability of websites offering health-related genetic tests directly to consumers. Genetics in Medicine 12(5):304-312.
Le, D. T., J. N. Uram, H. Wang, B. R. Bartlett, H. Kemberling, A. D. Eyring, A. D. Skora, B. S. Luber, N. S. Azad, D. Laheru, B. Biedrzycki, R. C. Donehower, A. Zaheer, G. A. Fisher, T. S. Crocenzi, J. J. Lee, S. M. Duffy, R. M. Goldberg, A. de la Chapelle, M. Koshiji, F. Bhaijee, T. Huebner, R. H. Hruban, L. D. Wood, N. Cuka, D. M. Pardoll, N. Papadopoulos, K. W. Kinzler, S. Zhou, T. C. Cornish, J. M. Taube, R. A. Anders, J. R. Eshleman, B. Vogelstein, and L. A. Diaz, Jr. 2015. PD-1 blockade in tumors with mismatch-repair deficiency. New England Journal of Medicine 372(26):2509-2520.
Le Tourneau, C., J. P. Delord, A. Goncalves, C. Gavoille, C. Dubot, N. Isambert, M. Campone, O. Tredan, M. A. Massiani, C. Mauborgne, S. Armanet, N. Servant, I. Bieche, V. Bernard, D. Gentien, P. Jezequel, V. Attignon, S. Boyault, A. Vincent-Salomon, V. Servois, M. P. Sablin, M. Kamal, X. Paoletti, and S. investigators. 2015. Molecularly targeted therapy based on tumour molecular profiling versus conventional therapy for advanced cancer (SHIVA): A multicentre, open-label, proof-of-concept, randomised, controlled phase 2 trial. Lancet Oncology 16(13):1324-1334.
Lea, D. H., K. A. Kaphingst, D. Bowen, I. Lipkus, and D. W. Hadley. 2011. Communicating genetic and genomic information: Health literacy and numeracy considerations. Public Health Genomics 14(4-5):279-289.
Lee, J. Y., T. H. Kim, D. H. Suh, J. W. Kim, H. S. Kim, H. H. Chung, N. H. Park, Y. S. Song, and S. B. Kang. 2015. Impact of guideline adherence on patient outcomes in early-stage epithelial ovarian cancer. European Journal of Surgical Oncology 41(4):585-591.
Leighl, N. B., N. Rekhtman, W. A. Biermann, J. Huang, M. Mino-Kenudson, S. S. Ramalingam, H. West, S. Whitlock, and M. R. Somerfield. 2014. Molecular testing for selection of patients with lung cancer for epidermal growth factor receptor and anaplastic lymphoma kinase tyrosine kinase inhibitors: American Society of Clinical Oncology endorsement of the College of American Pathologists/International Association for the Study of Lung Cancer/Association for Molecular Pathology guideline. Journal of Clinical Oncology 32(32):3673-3679.
Lin, M. Y., and N. R. Kressin. 2015. Race/ethnicity and Americans’ experiences with treatment decision making. Patient Education and Counseling 98(12):1636-1642.
Lundberg, G. D. 1981. Acting on significant laboratory results. Journal of the American Medical Association 245(17):1762-1763.
Manolio, T. A., M. F. Murray, and Inter-Society Coordinating Committee for Practitioner Education in Genomics. 2014. The growing role of professional societies in educating clinicians in genomics. Genetics in Medicine 16(8):571-572.
Martin, M. A., T. E. Klein, B. J. Dong, M. Pirmohamed, D. W. Haas, D. L. Kroetz, and Clinical Pharmacogenetics Implementation Consortium. 2012. Clinical Pharmacogenetics Implementation Consortium guidelines for HLA-B genotype and abacavir dosing. Clinical Pharmacology & Therapeutics 91(4):734-738.
McClellan, K. A., D. Avard, J. Simard, and B. M. Knoppers. 2013. Personalized medicine and access to health care: Potential for inequitable access? European Journal of Human Genetics 21(2):143-147.
Mega, J. L., S. L. Close, S. D. Wiviott, L. Shen, R. D. Hockett, J. T. Brandt, J. R. Walker, E. M. Antman, W. Macias, E. Braunwald, and M. S. Sabatine. 2009. Cytochrome P-450 polymorphisms and response to clopidogrel. New England Journal of Medicine 360(4):354-362.
Mega, J. L., T. Simon, J. P. Collet, J. L. Anderson, E. M. Antman, K. Bliden, C. P. Cannon, N. Danchin, B. Giusti, P. Gurbel, B. D. Horne, J. S. Hulot, A. Kastrati, G. Montalescot, F. J. Neumann, L. Shen, D. Sibbing, P. G. Steg, D. Trenk, S. D. Wiviott, and M. S. Sabatine. 2010. Reduced-function CYP2C19 genotype and risk of adverse clinical outcomes among patients treated with clopidogrel predominantly for PCI: A meta-analysis. Journal of the American Medical Association 304(16):1821-1830.
Mendelsohn, J. 2013. Personalizing oncology: Perspectives and prospects. Journal of Clinical Oncology 31(15):1904-1911.
Meric-Bernstam, F., C. Farhangfar, J. Mendelsohn, and G. B. Mills. 2013. Building a personalized medicine infrastructure at a major cancer center. Journal of Clinical Oncology 31(15):1849-1857.
Meric-Bernstam, F., L. Brusco, K. Shaw, C. Horombe, S. Kopetz, M. A. Davies, M. Routbort, S. A. Piha-Paul, F. Janku, N. Ueno, D. Hong, J. De Groot, V. Ravi, Y. Li, R. Luthra, K. Patel, R. Broaddus, J. Mendelsohn, and G. B. Mills. 2015. Feasibility of large-scale genomic testing to facilitate enrollment onto genomically matched clinical trials. Journal of Clinical Oncology 33(25):2753-2762.
Mirghani, H., F. Amen, Y. Tao, E. Deutsch, and A. Levy. 2015. Increased radiosensitivity of HPV-positive head and neck cancers: Molecular basis and therapeutic perspectives. Cancer Treatment Reviews 41(10):844-852.
Mody, R. J., Y. M. Wu, R. J. Lonigro, X. Cao, S. Roychowdhury, P. Vats, K. M. Frank, J. R. Prensner, I. Asangani, N. Palanisamy, J. R. Dillman, R. M. Rabah, L. P. Kunju, J. Everett, V. M. Raymond, Y. Ning, F. Su, R. Wang, E. M. Stoffel, J. W. Innis, J. S. Roberts, P. L. Robertson, G. Yanik, A. Chamdin, J. A. Connelly, S. Choi, A. C. Harris, C. Kitko, R. J. Rao, J. E. Levine, V. P. Castle, R. J. Hutchinson, M. Talpaz, D. R. Robinson, and A. M. Chinnaiyan. 2015. Integrative clinical sequencing in the management of refractory or relapsed cancer in youth. Journal of the American Medical Association 314(9):913-925.
The Moran Company. 2013. Results of analyses for chemotherapy administration utilization and chemotherapy drug utilization, 2005-2011 for Medicare fee-for-service beneficiaries. Arlington, VA: The Moran Group.
Morris, Z. S., and P. M. Harari. 2014. Interaction of radiation therapy with molecular targeted agents. Journal of Clinical Oncology 32(26):2886-2893.
Mouchawar, J., S. Hensley-Alford, S. Laurion, J. Ellis, A. Kulchak-Rahm, M. L. Finucane, R. Meenan, L. Axell, R. Pollack, and D. Ritzwoller. 2005a. Impact of direct-to-consumer advertising for hereditary breast cancer testing on genetic services at a managed care organization: A naturally-occurring experiment. Genetics in Medicine 7(3):191-197.
Mouchawar, J., S. Laurion, D. P. Ritzwoller, J. Ellis, A. Kulchak-Rahm, and S. Hensley-Alford. 2005b. Assessing controversial direct-to-consumer advertising for hereditary breast cancer testing: Reactions from women and their physicians in a managed care organization. American Journal of Managed Care 11(10):601-608.
Nadauld, L., D. S. Haslem, G. Stone, P. J. Mishra, S. Raghunath, J. L. Gillman, D. L. Loughmiller, and J. M. Ford. 2015. Implementation of a precision cancer program in an integrated health care system. Journal of Clinical Oncology 33(15) (suppl; abstr e17647).
Narmala, S. K., and B. C. Boulmay. 2013. Adequacy of lung cancer tissue samples for ancillary molecular testing in conventionally obtained biopsy material: A 3-year experience of the Louisiana State University (LSU) public hospital in New Orleans. Journal of Clinical Oncology 31(31).
NASEM (The National Academies of Sciences, Engineering, and Medicine). 2015. Improving diagnosis in health care. Washington, DC: The National Academies Press.
NCCN (National Comprehensive Cancer Network). 2015. Development and update of the NCCN guidelines. http://www.nccn.org/professionals/physician_gls/guidelines-development.asp (accessed July 20, 2015).
NCI (National Cancer Institute). 2007. National Cancer Institute best practices for biospecimen resources. http://biospecimens.cancer.gov/bestpractices/2011-NCIbestpractices.pdf (accessed October 12, 2015).
NCI. 2016. Imaging and biomarkers for early detection of aggressive cancer (U01). http://grants.nih.gov/grants/guide/pa-files/PAR-16-089.html (accessed January 26, 2016).
Nelson, W., V. F. Reyna, A. Fagerlin, I. Lipkus, and E. Peters. 2008. Clinical implications of numeracy: Theory and practice. Annals of Behavioral Medicine 35(3):261-274.
Neugut, A. I., D. J. Becker, B. J. Insel, and D. L. Hershman. 2012. Uptake of oxaliplatin and bevacizumab for treatment of node-positive and metastatic colon cancer. Journal of Oncology Practice 8(3):156-163.
NQF (National Quality Forum). 2015. National quality forum—mission and vision. http://www.qualityforum.org/About_NQF/Mission_and_Vision.aspx (accessed October 20, 2015).
OIG (Office of the Inspector General). 2012. Memorandum report: Coverage and payment for genetic laboratory tests. Washington, DC: Department of Health and Human Services.
Oloparde, O. I. 2004. Genetics in clinical cancer care: A promise unfulfilled among minority populations. Cancer Epidemiology Biomarkers & Prevention 13(11):1683-1686.
Paik, S., J. Bryant, E. Tan-Chiu, E. Romond, W. Hiller, K. Park, A. Brown, G. Yothers, S. Anderson, R. Smith, D. L. Wickerham, and N. Wolmark. 2002. Real-world performance of HER2 testing—national surgical adjuvant breast and bowel project experience. Journal of the National Cancer Institute 94(11):852-854.
Pantaleo, M. A., M. Nannini, S. Fanti, S. Boschi, P. L. Lollini, and G. Biasco. 2009. Molecular imaging of EGFR: It’s time to go beyond receptor expression. Journal of Nuclear Medicine 50(7):1195-1196; author reply 1196, 1197.
Paolucci, S. A., and C. Wicklund. 2015. Perspective: Genomic literacy and shared decision making. Written input to the Committee on Policy Issues in the Clinical Development and Use of Biomarkers for Molecularly Targeted Therapies, June 15, 2015, Washington, DC.
Peterson, E. A., K. J. Milliron, K. E. Lewis, S. D. Goold, and S. D. Merajver. 2002. Health insurance and discrimination concerns and BRCA1/2 testing in a clinic population. Cancer Epidemiology, Biomarkers & Prevention 11(1):79-87.
Pew Internet. 2013. Health online 2013. Washington, DC: Pew Research Center.
Plebani, M., M. Laposata, and G. D. Lundberg. 2011. The brain-to-brain loop concept for laboratory testing 40 years after its introduction. American Journal of Clinical Pathology 136(6):829-833.
Ponce, N. A., M. Ko, S. Y. Liang, J. Armstrong, M. Toscano, C. Chanfreau-Coffinier, and J. S. Haas. 2015. Early diffusion of gene expression profiling in breast cancer patients associated with areas of high income inequality. Health Affairs 34(4):609-615.
Poonacha, T. K., and R. S. Go. 2011. Level of scientific evidence underlying recommendations arising from the National Comprehensive Cancer Network clinical practice guidelines. Journal of Clinical Oncology 29(2):186-191.
Poste, G. 2011. Bring on the biomarkers. Nature 469(7329):156-157.
Prahallad, A., C. Sun, S. Huang, F. Di Nicolantonio, R. Salazar, D. Zecchin, R. L. Beijersbergen, A. Bardelli, and R. Bernards. 2012. Unresponsiveness of colon cancer to BRAF(V600E) inhibition through feedback activation of EGFR. Nature 483(7387):100-103.
Psek, W. A., R. A. Stametz, L. D. Bailey-Davis, D. Davis, J. Darer, W. A. Faucett, D. L. Henninger, D. C. Sellers, and G. Gerrity. 2015. Operationalizing the learning health care system in an integrated delivery system. EGEMS (Wash DC) 3(1):1122.
Rawls, J. 1971. A theory of justice. Cambridge, MA: Harvard Univeristy Press.
Roche, P. C., V. J. Suman, R. B. Jenkins, N. E. Davidson, S. Martino, P. A. Kaufman, F. K. Addo, B. Murphy, J. N. Ingle, and E. A. Perez. 2002. Concordance between local and central laboratory HER2 testing in the breast intergroup trial N9831. Journal of the National Cancer Institute 94(11):855-857.
SACGHS (Secretary’s Advisory Committee on Genetics, Health, and Society). 2006. Coverage and reimbursement of genetic tests report. Washington, DC: Department of Health and Human Services.
SACGHS. 2008. U.S. system of oversight of genetic testing: A response to the charge of the Secretary of Health and Human Services. Washington, DC: Department of Health and Human Services.
Scheuner, M. T., P. Sieverding, and P. G. Shekelle. 2008. Delivery of genomic medicine for common chronic adult diseases: A systematic review. Journal of the American Medical Association 299(11):1320-1334.
Schilsky, R. L. 2014. Implementing personalized cancer care. Nature Reviews Clinical Oncology 11(7):432-438.
Schmitt, F. 2009. HER2+ breast cancer: How to evaluate? Advances in Therapy 26(Suppl 1):S1-S8.
Schully, S. D., T. K. Lam, W. D. Dotson, C. Q. Chang, N. Aronson, M. L. Birkeland, S. J. Brewster, S. Boccia, A. H. Buchanan, N. Calonge, K. Calzone, B. Djulbegovic, K. A. Goddard, R. D. Klein, T. E. Klein, J. Lau, R. Long, G. H. Lyman, R. L. Morgan, C. G. Palmer, M. V. Relling, W. S. Rubinstein, J. J. Swen, S. F. Terry, M. S. Williams, and M. J. Khoury. 2015. Evidence synthesis and guideline development in genomic medicine: Current status and future prospects. Genetics in Medicine 17(1):63-67.
Shabihkhani, M., G. M. Lucey, B. Wei, S. Mareninov, J. J. Lou, H. V. Vinters, E. J. Singer, T. F. Cloughesy, and W. H. Yong. 2014. The procurement, storage, and quality assurance of frozen blood and tissue biospecimens in pathology, biorepository, and biobank settings. Clinical Biochemistry 47(4-5):258-266.
Shaneyfelt, T. M., and R. M. Centor. 2009. Reassessment of clinical practice guidelines: Go gently into that good night. Journal of the American Medical Association 301(8):868-869.
Simonds, N. I., M. J. Khoury, S. D. Schully, K. Armstrong, W. F. Cohn, D. A. Fenstermacher, G. S. Ginsburg, K. A. Goddard, W. A. Knaus, G. H. Lyman, S. D. Ramsey, J. Xu, and A. N. Freedman. 2013. Comparative effectiveness research in cancer genomics and precision medicine: Current landscape and future prospects. Journal of the National Cancer Institute 105(13):929-936.
Smith, A. P., M. Y. Lichtveld, K. R. Miner, S. L. Tyus, and L. N. Gase. 2009. A competency-based approach to expanding the cancer care workforce: Proof of concept. MEDSURG Nursing Journal 18(1):38-49; quiz 50.
Smith, J., C. Medalia. 2015. Health insurance coverage in the United States: 2014. Current Population Reports, P60-253. Washington, DC: U.S. Government Printing Office.
Sniderman, A. D., and C. D. Furberg. 2009. Why guideline-making requires reform. Journal of the American Medical Association 301(4):429-431.
Soegaard, M., A. Jensen, K. Frederiksen, E. Hogdall, C. Hogdall, J. Blaakaer, and S. K. Kjaer. 2008. Accuracy of self-reported family history of cancer in a large case-control study of ovarian cancer. Cancer Causes Control 19(5):469-479.
Sommers, B. D., B. Maylone, K. H. Nguyen, R. J. Blendon, and A. M. Epstein. 2015. The impact of state policies on ACA applications and enrollment among low-income adults in Arkansas, Kentucky, and Texas. Health Affairs 34(6):1010-1018.
Suther, S., and P. Goodson. 2003. Barriers to the provision of genetic services by primary care physicians: A systematic review of the literature. Genetics in Medicine 5(2):70-76.
Torres-Roca, J. F., W. J. Fulp, J. J. Caudell, N. Servant, M. A. Bollet, M. van de Vijver, A. O. Naghavi, E. E. Harris, and S. A. Eschrich. 2015. Integration of a radiosensitivity molecular signature into the assessment of local recurrence risk in breast cancer. International Journal of Radiation Oncology*Biology*Physics 93(3):631-638.
Treweek, S., R. Dryden, C. McCowan, A. Harrow, and A. M. Thompson. 2015. Do participants in adjuvant breast cancer trials reflect the breast cancer patient population? European Journal of Cancer 51(8):907-914.
Tricoci, P., J. M. Allen, J. M. Kramer, R. M. Califf, and S. C. Smith, Jr. 2009. Scientific evidence underlying the ACC/AHA clinical practice guidelines. Journal of the American Medical Association 301(8):831-841.
van Kruchten, M., A. W. Glaudemans, E. F. de Vries, R. G. Beets-Tan, C. P. Schroder, R. A. Dierckx, E. G. de Vries, and G. A. Hospers. 2012. PET imaging of estrogen receptors as a diagnostic tool for breast cancer patients presenting with a clinical dilemma. Journal of Nuclear Medicine 53(2):182-190.
Vassy, J. L., B. R. Korf, and R. C. Green. 2015. How to know when physicians are ready for genomic medicine. Science Translational Medicine 7(287):287fs219.
Vogelstein, B., and K. W. Kinzler. 2015. The path to cancer—three strikes and you’re out. New England Journal of Medicine 373(20):1895-1898.
Wicklund, C., and A. Trepanier. 2014. Adapting genetic counseling training to the genomic era: More an evolution than a revolution. Journal of Genetic Counseling 23(4):452-454.
Williams, M. S. 2015. Clinical practice issues. Presentation to the Committee on Policy Issues in the Clinical Development and Use of Biomarkers for Molecularly Targeted Therapies, April 1, 2015, Washington, DC.
Wolff, A. C., M. E. H. Hammond, J. N. Schwartz, K. L. Hagerty, D. C. Alfred, R. J. Cote, M. Dowsett, P. L. Fitzgibbons, W. M. Hanna, A. Langer, L. M. McShane, S. Paik, M. D. Pegram, E. A. Perez, M. F. Press, A. Rhodes, C. Sturgeon, S. E. Taube, R. Tubbs, G. H. Vance, M. Van De Vijver, T. M. Wheeler, and D. F. Hayes. 2007a. American Society of Clinical Oncology/College of American Pathologists guideline recommendations for human epidermal growth factor receptor 2 testing in breast cancer. Archives of Pathology & Laboratory Medicine 131(1):18-43.
Wolff, A. C., M. E. H. Hammond, J. N. Schwartz, K. L. Hagerty, D. C. Allred, R. J. Cote, M. Dowsett, P. L. Fitzgibbons, W. M. Hanna, A. Langer, L. M. McShane, S. Paik, M. D. Pegram, E. A. Perez, M. F. Press, A. Rhodes, C. Sturgeon, S. E. Taube, R. Tubbs, G. H. Vance, M. Van De Vijver, T. M. Wheeler, and D. F. Hayes. 2007b. American Society of Clinical Oncology/College of American Pathologists guideline recommendations for human epidermal growth factor receptor 2 testing in breast cancer. Journal of Clinical Oncology 25(1):118-145.
Yamaguchi, R., S. Tsuchiya, T. Koshikawa, T. Yokoyama, K. Mibuchi, Y. Nonaka, S. Ito, H. Higuchi, M. Nagao, K. Higaki, J. Watanabe, M. Kage, and H. Yano. 2012. Evaluation of inadequate, indeterminate, false-negative and false-positive cases in cytological examination for breast cancer according to histological type. Diagnostic Pathology 7:53.
Yard, B., E. K. Chie, D. J. Adams, C. Peacock, and M. E. Abazeed. 2015. Radiotherapy in the era of precision medicine. Seminars in Radiation Oncology 4(4):227-236.
Yu, P. P. 2015. Knowledge bases, clinical decision support systems, and rapid learning in oncology. Journal of Oncology Practice 11(2):e206-e211.
Zabalza, M., I. Subirana, J. Sala, C. Lluis-Ganella, G. Lucas, M. Tomas, R. Masia, J. Marrugat, R. Brugada, and R. Elosua. 2012. Meta-analyses of the association between cytochrome CYP2C19 loss- and gain-of-function polymorphisms and cardiovascular outcomes in patients with coronary artery disease treated with clopidogrel. Heart 98(2):100-108.
Zilliacus, E. M., B. Meiser, E. A. Lobb, J. Kirk, L. Warwick, and K. Tucker. 2010. Women’s experience of telehealth cancer genetic counseling. Journal of Genetic Counseling 19(5):463-472.
Zilliacus, E. M., B. Meiser, E. A. Lobb, P. J. Kelly, K. Barlow-Stewart, J. A. Kirk, A. D. Spigelman, L. J. Warwick, and K. M. Tucker. 2011. Are videoconferenced consultations as effective as face-to-face consultations for hereditary breast and ovarian cancer genetic counseling? Genetics in Medicine 13(11):933-941.
This page intentionally left blank.












































