Summary
Scientists have studied gene drives for more than 50 years. The development of a powerful genome editing tool in 2012, CRISPR/Cas9,1 led to recent breakthroughs in gene drive research that built on that half century’s worth of knowledge, and stimulated new discussion of the potential applications and implications of gene drive technologies. Just prior to the beginning of this study and since the committee was first convened, scientists published four proofs of concept—one in yeast, one in fruit flies, and two in different species of mosquitoes—that demonstrate the successful development of gene drives in the laboratory, at least in these organisms. Proposed applications for gene-drive modified organisms for basic research, conservation, agriculture, public health and other purposes will likely continue to expand as gene editing tools become more refined. Gene-drive modified organisms are on the horizon.
The fast moving nature of this field is both encouraging and concerning. While gene-drive modified organisms hold promise for addressing difficult to solve, persistent challenges, such as the eradication of vector-borne diseases and the conservation of threatened and endangered species, these proposed applications are based on limited proof-of-concept studies. The presumed efficiency of gene-drive modified organisms may lead to calls for their release in perceived crisis situations, before there is adequate knowledge of their ecological effects, and before mitigation plans for unintended harmful consequences are in place.
Responding to this fast moving field, the National Institutes of Health (NIH) and the Foundation for the National Institutes of Health (FNIH)2 asked the National Academies of Sciences, Engineering, and Medicine to convene a committee with a broad range of expertise to summarize the scientific discoveries related to gene drives and considerations for their responsible use.
Proof-of-concept in a few laboratory studies is not sufficient in and of itself to support a decision to release gene-drive modified organisms into the environment. Laboratory and field research is needed to refine CRISPR/Cas9-based gene drives and other gene drive mechanisms, and to understand how gene drives might work under different environmental conditions and in a wide variety of organisms. The considerable gaps in knowledge about potential off-target (within the organism) and non-target (in other species or the environment) effects necessitate a collaborative, multidisciplinary approach to research, ecological risk assessment, development of public policy, and decision making for each proposed application of a gene drive technology. General principles to guide responsible practices for gene drives from the laboratory setting through to field release and monitoring are embedded as recommendations throughout the report.
STATE OF THE SCIENCE OF GENE DRIVES
Gene drives are systems of biased inheritance in which the ability of a genetic element to pass from a parent to its offspring through sexual reproduction is enhanced (see Figure S-1).
Thus, the result of a gene drive is the preferential increase of a specific genotype, the genetic makeup of an organism that determines a specific phenotype (trait), from one generation to the next, and potentially throughout the population.
___________________
1CRISPR (Clustered regularly-interspaced short palindromic repeats) are segments of bacterial DNA that, when paired with a specific guide protein, such as Cas9 (CRISPR associated protein 9), can be used to make targeted cuts in an organism’s genome.
2This study was sponsored by the National Institutes of Health and the Foundation for the National Institutes of Health, and the National Academy of Sciences Biology and Biotechnology Fund. The Defense Advanced Research Projects Agency and The Bill & Melinda Gates Foundation provided support to the NIH and the FNIH, respectively for this study.
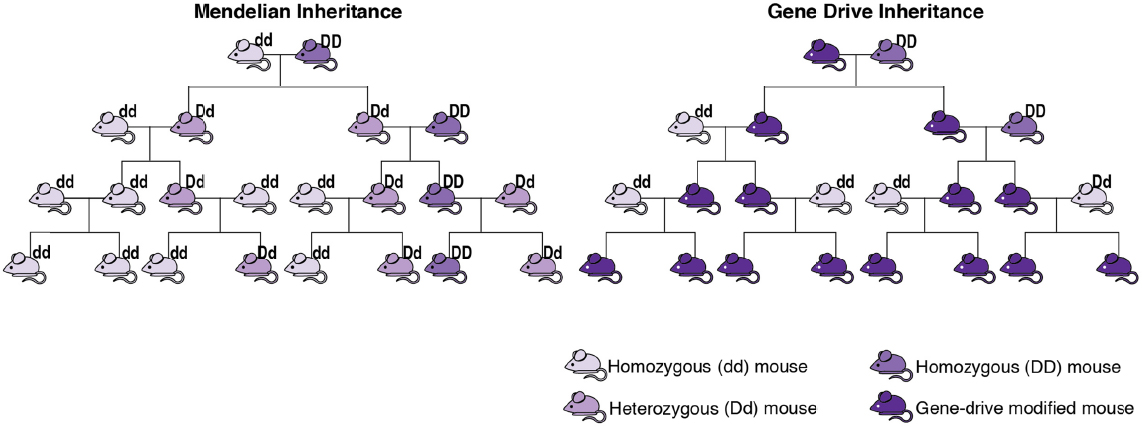
A wide variety of gene drives occur in nature that can cause genetic elements to spread throughout populations to varying degrees. Researchers are studying how to harness such natural mechanisms (e.g., transposable elements, homing endonucleases, and meiotic drive) to develop gene-drive modified organisms. Preliminary evidence suggests that gene drives developed in the laboratory with CRISPR/Cas9 could spread a targeted gene through nearly 100% of a given population of yeast, fruit flies, or mosquitoes.
The development of CRISPR/Cas9 as a genome editing tool has spurred biologists to propose a range of applications for gene drives to solve various public health, agricultural, conservation, and other challenges where solutions are limited or entirely lacking (see Table S-1). Most research to date is focused on controlling or altering organisms that transmit infectious diseases to humans, such as mosquito vectors of dengue, malaria, Zika, and chikungunya.
A gene drive that alters the female mosquito’s ability to become infected with the malaria parasite, or prevents parasite development within the mosquito, could block malarial transmission without affecting mosquito populations. In November 2015, researchers demonstrated that CRISPR/Cas9 can be used to create a gene drive to spread anti-Plasmodium genes in populations of a malaria-carrying mosquito, Anopheles stephensi (Gantz et al., 2015). However, the system transmits the drive construct at Mendelian frequencies in some instances, suggesting that this valuable proof-of-concept needs further modification and research before field release (Gantz et al, 2015). Alternatively, a gene drive that alters the fitness of the female mosquito could result in reducing vector populations over time. In December 2015, researchers demonstrated that CRISPR/Cas9 can be used to create a gene drive that causes sterility in female Anopheles gambiae mosquitoes (Hammond et al., 2016).
It is important to note that, until proven otherwise, cell types and species are expected to differ in their capacity to carry a gene drive, and therefore the effects and efficacy of gene drives will be largely species-dependent.
ECOLOGICAL AND ENVIRONMENTAL CONSIDERATIONS
Research on the molecular biology of gene drives has outpaced research on population genetics and ecosystem dynamics, two fields of study whose perspectives are essential to determining the efficacy of gene drives and their biological and ecological outcomes. There are considerable gaps in knowledge regarding a gene drive’s effectiveness, both on the target organism and the environment, over time and across diverse genetic backgrounds. It is also essential to consider how gene drives will propagate throughout a population and affect not only the target species, but its entire ecological community. Key factors that influence the propagation of gene drives include the following:
- The evolutionary “fitness” of individuals carrying the gene drive—that is, their ability to produce fertile offspring—as compared to individuals not carrying the gene drive.
- The “conversion rate,” which describes how the gene drive is passed to subsequent generations when one parent carries the gene drive and the other does not.
- “Gene flow,” which describes how the gene drive moves between different populations of the target species.
- “Horizontal gene transfer,” or the potential for gene drives to move from the target species into entirely different species.
These four factors interact in complex ways. Improved modeling capabilities and more empirical evidence would enhance our ability to understand and predict how gene drives might propagate through populations. It is also vital to consider how changes in a species’ population size or distribution that are caused by a gene drive might reverberate through the ecosystem as a whole. Pertinent community dynamics and ecological factors to consider include the following:
- What is the species’ role in its community?
TABLE S-1 Potential Applications for Gene Drive Research
| Public Health | |
 Aedes aegypti Image Source: US Centers for Disease Control and Prevention |
|
| Ecosystem Conservation | |
 Hemignathus munroi (‘Akiapōlā’au honeycreeper) Image Source: US Fish and Wildlife Service |
|
| Agriculture | |
 Fruit damage from spotted wing drosophila infestation Image Source: US Department of Agriculture |
|
| Basic Research | |
 DNA Double Helix Image Source: National Institutes of Health |
|
- Are there other species that would fill a similar ecological niche in the community if the target species were to disappear?
- Is there a tipping point at which the community may change rapidly from one configuration to another, and could the gene drive lead to such a tipping point?
- How might a dramatic change in the population of the target species affect other species with which it has coevolved?
- Could the target species develop mechanisms to neutralize the gene drive (e.g., evolve resistance), and how might that dynamic affect others in the ecological community?
Generally speaking, a gene drive would likely be most harmful to an ecosystem if it is released in a native keystone species, but potentially less impactful if deployed in an invasive species for which there is a native ecological equivalent, thus containing the impacts to a relatively small part of the food web. Unintended consequences should be considered, especially in regard to the risk of horizontal gene transfer. In order to address knowledge gaps, gene drive research will require the convergence of multiple fields of study including molecular biology, genome editing, population genetics, evolutionary biology, and ecology.
CHARTING HUMAN VALUES
Questions about gene drives rest on values at every step, from whether, why, and how research should be conducted to whether and where a gene-drive modified organism should be released into the environment. Three broad categories of concern were identified and explored:
- the potential benefits and harms of gene drive research for people,
- the potential impact of gene-drive modified organisms on the environment, and
- the use of gene drives and who will make decisions about them.
The potential benefits and harms of gene-drive modified organisms will be central in deciding whether to allow field testing or open environmental release. Some of the fundamental reasons to conduct gene drive research include widely shared commitments to fighting human disease, promoting human welfare, and protecting and restoring the natural environment. A hypothetical example is the potential development of a gene drive that prevents mosquitoes from transmitting dengue, a virus that occurs predominately in urban environments throughout the tropics, could save many lives.
On the other hand, some gene-drive modified organisms might pose harm to humans. One hypothetical example is a mosquito modified so that it could not host the dengue virus that becomes more susceptible as a host to another virus. Deciding whether to go forward with environmental release of a gene-drive modified organism will require a reasonable level of assurance that the possible harms have been identified and studied and that they are outweighed by the potential benefits.
A potential environmental benefit is that a gene drive may be less harmful than alternative solutions to a problem. For example, a gene drive to suppress non-native rodent populations on remote islands could reduce the need for alternative forms of control such as the use of rodenticides. The cost of administering rodenticides is estimated to be in the millions of dollars and rodenticides may also harm non-target species.
Nonetheless, because gene-drive modified organisms are intended to spread in the environment, there is a widespread sense among researchers and commentators that they may have harmful effects for other species or ecosystems. For example, using a gene drive to suppress a non-native weed population may lead to unexpected consequences, such as the loss of habitat for native species or even the establishment of a second, more resilient invasive species. Assessments of the environmental harms of a proposed release will require careful, case-by-case analysis
Values related to human welfare and environmental harms will be weighed in developing public policy guidelines, some of which may constrain research on gene drives or the release of gene-drive modified organisms into the environment. Such guidelines will require integrating precautionary measures into the research process and the assessment of potential benefits and harms. Precautionary measures can provide opportunities to gather further information and revisit decisions about how to proceed with a gene drive technology, but, at the same time, not hinder research progress.
Perspectives on the place of human beings in ecosystems and their larger relationship to nature—and their impact on and manipulation of ecosystems—have an important role in the emerging debate about gene drives. The increased power for human beings to alter wild species and perhaps to eliminate them, thereby altering the shared environment—will be intrinsically objectionable to some people. Proposals to use gene drives in ways that might lead to the extinction of species or significantly alter the environment will require especially careful review.
When selecting sites for field trials or environmental releases of gene-drive modified organisms, it is important to consider the values of researchers and the affected publics, and their understanding of the balance of potential benefits and harms. Approaches to ensure that communities participate meaningfully in decision making about the use of gene-drive modified organisms will be essential, particularly in low- and middle-income countries where power differentials may preclude such participation.
PHASED TESTING AND SCIENTIFIC APPROACHES TO REDUCE GENE DRIVES’ POTENTIAL HARMS
Before field testing or environmental release of gene-drive modified organisms, it is crucial to establish a rich understanding of the target organism, its relationship with its environment, and potential unintended consequences. A phased testing pathway, such as the one outlined by the World Health Organization (WHO) for testing genetically modified mosquitoes, can facilitate a precautionary, step-by-step approach to research on gene drives (WHO, 2014). Each step in such a pathway promotes careful study and evaluation, includes checkpoints to determine whether and when research should move to the next phase, and provides vital data to inform and enhance the effectiveness of other phases.
In contrast with other genetic modification techniques, which are typically designed to minimize inheritance or transmission of altered genetic elements, the goal of a gene drive is to rapidly spread genetic information throughout a population. This makes it especially important to minimize the potential for unintended consequences. Reducing the potential for unintended consequences will require a combination of confinement and containment strategies.
When developing confinement and containment strategies, consideration should be given to their benefits, costs, and weaknesses. For example, adding a visible marker to help identify gene-drive modified organisms in some cases could have negative consequences for the organism, which should be weighed against the benefits of this strategy. It is particularly imperative to use caution when considering the development of a “reversal drive”—a gene drive designed to mitigate the unintended consequences of another gene drive—as it may be impossible to employ this strategy effectively without off-target effects or to redress fully ecological and environmental effects from the original gene drive.
After release into the environment, a gene drive knows no political boundaries. Thus, it is desirable to expand the intellectual capital of governing bodies and research capacity of relevant institutions around the world to facilitate appropriate engagement in governance, research, and collaboration pertaining to gene drives. In particular, this includes building long-term relationships with scientists in low- and middle-income countries where field research on gene-drive modified organisms is most likely to occur.
ASSESSING RISKS OF GENE-DRIVE MODIFIED ORGANISMS
The potential for gene drives to spread throughout a population, to persist in the environment, and to cause irreversible effects on organisms and ecosystems calls for a robust method to assess risks. Environmental assessments and environmental impact statements required by the National Environmental Protection Act, though widely acknowledged as valuable in other contexts, are inappropriate tools to characterize the risks of gene-drive modified organisms. Instead, ecological risk assessment would be beneficial in the context of gene drive research, because this method can be used to estimate the probability of immediate and long-term environmental and public health harms and benefits.
Ecological risk assessment allows comparisons among alternative strategies, incorporates the concerns of relevant publics, and can be used to identify sources of uncertainty, making it well-suited to inform research directions and support public policy decisions about emerging gene drive technologies. Two key features of ecological risk assessments are the ability to trace cause-and-effect pathways and the ability to quantify the probability of specific outcomes. This approach could also potentially be built into a structured, adaptive process to oversee the release and management of gene-drive modified organisms in the environment. As of May 2016, no ecological risk assessment has yet been conducted for a gene-drive modified organism.
Some amount of uncertainty is unavoidable. There is currently sufficient knowledge to begin constructing ecological risk assessments for some potential gene-drive modified organisms, including mosquitoes and mice. In some other cases it may be possible to extrapolate from research and risk analyses focused on other genetically modified organisms and non-indigenous species. However, laboratory studies and confined field tests (or studies that mimic confined field tests such as large cage trials and greenhouse studies) represent the best approaches to reduce uncertainty in an ecological risk assessment, and are likely to be of greatest use to risk assessors.
In the United States, the primary source of federal guidance on ecological risk assessment comes from the US Environmental Protection Agency’s 1998 Guidelines for Ecological Risk Assessment. Since 1998 EPA has also published documents that update the approaches to specific technical features and incorporate ecosystem services into ecological risk assessment. The 1998 guidelines and subsequent documents focus predominantly on evaluating the risks to ecosystems posed by toxic chemicals, and do not yet adequately address the assessment of multiple stressors and endpoints. Consequently, these documents are not yet sufficient on their own, to guide ecological risk assessment of gene drive technology. The lack of guidance from the US federal government applicable to ecological risk assessment for gene drive research is a critical gap.
PUBLIC ENGAGEMENT
There is broad agreement on the importance of engaging affected communities, stakeholders, and broader publics in decision making about activities involving gene drives. Public engagement can help to frame and define the risks of gene-drive modified organisms and provide input into practical decision making and policy. The outcomes of engagement may be as crucial as the scientific outcomes to decisions about whether to release a gene-drive modified organism into the environment. Thus, engagement cannot be an afterthought; it requires effort, attention, resources, and advanced planning.
Mechanisms for public engagement and deliberation already exist within some authorized US agencies that oversee biotechnology, but there is generally little clarity on how public engagement should feed into governance and a lack of consensus about best practices in this regard. This is due to at least two factors: first, because regulatory authority remains unclear, the availability of particular formal and customary mechanisms for public engagement also remain unclear; second, although the National Environmental Protection Act will in some cases require public input and
afford opportunity for public comment, these mechanisms are an inadequate platform for the more robust forms of engagement discussed in this report.
GOVERNANCE OF GENE DRIVES
The nature of gene drives—which are intended to spread select genetic elements into populations of living organisms—raises many ethical questions and presents a challenge for existing governance paradigms to identify and assess environmental and public health risks. The governance of research begins with the personal responsibility of the investigator, is formalized in professional guidelines, and often extends to legally binding policies and enforceable regulations. In the United States, it is clear that gene drive activities will trigger a variety of governance mechanisms. However, some of these mechanisms may be inadequate for identifying immediate and long-term potential environmental and public health implications of individual gene drive applications because they lack clarity in their jurisdiction, they are challenged by the distinguishing characteristics of gene drives, or they provide insufficient structures for public engagement.
Two distinguishing characteristics of gene drives, intentional spread of a genetic trait through a population and the potential for their effects on ecosystems to be irreversible, present increased uncertainties, making robust assessment of their risk more critical, but also more difficult. Because of the existing uncertainties associated with gene drives, regulation will be needed that facilitates fundamental, applied, and translational research so that the potential harms and benefits of gene drives can be responsibly explored in laboratory and field studies.
It is important to note that a one-size-fits-all approach to governance is not likely to be appropriate. Each phase of research activity—from developing a research plan to post-release surveillance—raises different levels of concern depending on the organism being modified and the type of gene drive being developed. Governance and regulation of gene drive research will need to be proportionate to the hazards posed by the specific activity. In addition, governance will need to be responsive to changes in scientific best practices and ethical considerations as gene drive technologies develop.
Investigators’ Responsibility and Professional Guidelines
Currently, institutions, funders, and professional societies work in concert to encourage professional best practices in research. Such cooperation will be instrumental to maintaining high standards in gene drive research. Appropriate resources for education (conceptual) and training (practical) in the responsible conduct of research, as well as public acknowledgement of researchers for their standards of practice, will be important for reinforcing responsible practices in gene drive research.
Federal Guidelines
Laboratory-based research conducted at an institution that receives funding from the National Institutes of Health (NIH) is subject to NIH’s guidelines on biosafety and oversight by Institutional Biosafety Committees (IBCs). These guidelines, although international in nature, are adapted to specific institutional contexts and are complemented by good laboratory practices. Moreover, the NIH guidelines clearly stipulate that all research at NIH-funded institutions may be regulated by laws established at the local, state, and federal levels, even in the absence of NIH funding for a specific project (e.g., other federal agencies, private foundations). IBCs have provided a robust system of health and environmental protection for laboratory research over the last few decades.
Nonetheless, due to the novel characteristics of gene drives, capacity issues, and an absence of clearly defined guidelines for gene drive research, current IBCs may not have the expertise or resources to evaluate the biosafety of gene drives effectively. IBCs are also not equipped
to examine biosecurity or willful misuse issues. However, there is potential to learn from institutional biosafety committees at institutions where gene drive research has been ongoing.
Federal Regulations
In the United States, regulation of the gene-drive modified organisms will most likely fall under the Coordinated Framework for the Regulation of Biotechnology. However, the US Food and Drug Administration (FDA), the US Department of Agriculture (USDA), and the US Environmental Protection Agency (EPA), the federal agencies included in the current Coordinated Framework, do not have clear lines of authority over the potential applications of gene drive research. The diversity of potential gene-drive modified organisms and contexts in which they might be used reveal a number of regulatory overlaps and gaps. For some potential applications of gene drive technologies, regulatory jurisdiction may overlap, which suggests the need for a process to quickly determine which agency should coordinate governance of that technology.
Potential Dual Use Issues
Gene drive research raises concerns about biosafety, biosecurity, and potential dual use of the technology. The scientific community, including individual researchers, institutions, and funders, have an obligation to engage in conversations with policy makers about best practices to safeguard against unintentional or intentional misuse of gene-drive modified organisms. Safeguards will be aided by rigorous attention to confinement and containment protocols in laboratory and field tests; active awareness about the potential for misuse; and participation in education and training programs about the dual use potential of gene drive research. Governance mechanisms need to be in place to address questions about the biosecurity implications of gene drive research and consider developing mitigation strategies that are not dependent on the underlying technology.
Need for International Coordination
Research on gene drives is global. Responsible governance will need to be international and inclusive, with clearly defined global regulatory frameworks, policies, and best practice standards for implementation. Low- and middle-income countries where gene-drive modified organisms may be employed will certainly need to be involved in governance, recognizing that many countries lack the capacity to develop a comprehensive regulatory scheme for gene drives from scratch.
The United Nations Convention on Biological Diversity is the main international regulatory instrument governing the development and use of genetically modified organisms, as implemented through the Cartagena and Nagoya Protocols. Many countries are now developing regulatory systems in response to the Cartagena Protocol. Many such systems are predicated on a strong precautionary, nearly preventive approach, which may restrict further gene drive research out of a precautionary concern about gene drives’ intrinsic ability to spread and persist in the environment. Given that the United States is not a Party to the Cartagena Protocol, it is a major gap in international governance that the United States does not have a clear policy for collaborating with other countries with divergent systems of governance, especially when such countries may, in fact, lack the capacity to assess the safety of gene drive research, undertake public engagement and societal dialogue, and maintain regulatory institutions.
In practice, a significant amount of field research on genetically modified mosquitos operates under guidelines established by international organizations, such as the WHO, and by the research community itself. These should provide a useful foundation for the establishment of guidelines for gene-drive modified organisms. However, these existing guidelines have important gaps and may not address all of the distinctive aspects of gene drives or the range of potential organisms to be used. For example, guidelines may need to be adapted to align to local
contexts in order to be implemented. Moreover, most guidelines are not themselves tied explicitly to public oversight and implementation.
GENE DRIVES ON THE HORIZON
There is insufficient evidence available at this time to support the release of gene-drive modified organisms into the environment. However, the potential benefits of gene drives for basic and applied research are significant and justify proceeding with laboratory research and highly controlled field trials.
A phased testing pathway and robust ecological risk assessments are essential for navigating uncertainty and informing decisions around the development and application of gene-drive modified organisms.
A comprehensive approach to the development and governance of gene-drive modified organisms will need to go beyond considerations for public health and the environment, and must also consider the benefits of technological innovation, the implications of intellectual property arrangements, public engagement, and economics, among other valued societal commitments.
Specific recommendations related to these overarching conclusions, presented in Chapter 9 of this report, include:
Recommendation 9-1: Funders of gene drive research should coordinate, and if feasible collaborate, to reduce gaps in knowledge not only about the molecular biology of gene drives, but also in other areas of fundamental and applied research that will be crucial to the responsible development and application of gene drive technology, including population genetics, evolutionary biology, ecosystem dynamics, modeling, ecological risk assessment, and public engagement.
Recommendation 9-2: Funders of gene drive research should establish open access, online repositories of data on gene drives as well as standard operating procedures for gene drive research to share knowledge, improve frameworks for ecological risk assessment, and guide research design and monitoring standards around the world.
Recommendation 9-3: The distinguishing characteristics of gene drives—including their intentional spread and the potential irreversibility of their environmental effects—should be used to frame the societal appraisal of the technology, and they should be considered in ecological risk assessment, public engagement, regulatory reform, and decision making.
Recommendation 9-4: Proposed field tests or environmental releases of gene-drive modified organisms should be subject to ecological risk assessment and structured decision making processes. These processes should include modeling of off-target and non-target effects from the genome level through ecosystem level. When possible, empirical estimates of such variables as gene flow, population change, trophic interactions, and community dynamics should be developed as part of the models.
Recommendation 9-5: Governing authorities, including research institutions, funders, and regulators, should develop and maintain clear policies and mechanisms for how public engagement will factor into research, ecological risk assessments, and public policy decisions on gene drives. Defined mechanisms and avenues for such engagement should be built into the risk assessment and decision-making processes from the beginning.
Recommendation 9-6: In selecting sites for field testing and environmental releases, researchers and funders should be guided by their professional judgment, the feasibility of
risk assessment and community engagement, and the community’s values and understanding of the balance of benefits and harms. In site selection, preference should be given to locations in countries with the existing scientific capacity and governance frameworks to conduct and oversee the safe investigation of gene drives and development of gene-drive modified organisms.
REFERENCES
Gantz, V.M., N. Jasinskiene, O. Tatarenkova, A. Fazekas, V.M. Macias, E. Bier, and A.A. James. 2015. Highly efficient Cas9-mediated gene drive for population modification of the malaria vector mosquito Anopheles stephensi. Proc. Natl. Acad. Sci. U.S.A. 112:E6736-E6743.
Hammond, A., R. Galizi, K. Kyrou, A. Simoni, C. Siniscalchi, D. Katsanos, M. Gribble, D. Baker, E. Marois, S. Russell, A. Burt, N. Windbichler, A. Crisanti, and T. Nolan. 2016. A CRISPR-Cas9 gene drive system targeting female reproduction in the malaria mosquito vector Anopheles gambiae. Nat. Biotechnol. 34(1):78-83.
WHO (World Health Organization). 2014. The Guidance Framework for Testing Genetically Modified Mosquitoes. World Health Organization, Programme for Research and Training in Tropical Diseases [online]. Available at http://apps.who.int/iris/bitstream/10665/127889/1/9789241507486_eng.pdf?ua=1 [accessed April 19, 2016].











