2
The State of the Science
The first of the workshop’s four panels examined the state of the science in three key areas: the prevalence of overweight and obesity in early childhood and changes over time, the epigenetics of childhood obesity, and the role of flavor in establishing lifelong food preferences. These presentations were followed by a discussion session, summarized at the end of this chapter.
PREVALENCE AND TRENDS OF OVERWEIGHT AND OBESITY IN THE EARLY CHILDHOOD YEARS
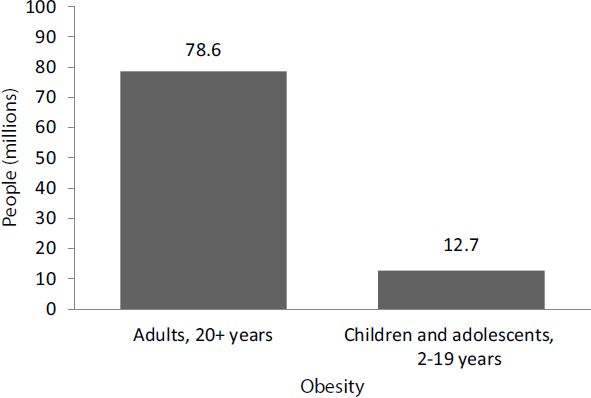
According to data from the National Health and Nutrition Examination Survey (NHANES), 16.9 percent of children and teens in the United States between the ages of 2 and 19 were obese in 2011-2012 (Ogden et al., 2014). Among adults, aged 20 and older, 34.9 percent were obese. Together, these percentages translate into more than 90 million individuals—78.6 million adults and 12.7 million youth (see Figure 2-1), observed Cynthia Ogden, an epidemiologist at the National Center for Health Statistics at the Centers for Disease Control and Prevention (CDC), in her opening presentation at the workshop. “I know you all know this, but I think it is quite striking,” she said.
The NHANES is a complex, multistage probability sample of the U.S. civilian noninstitutionalized population (Johnson et al., 2014). Because it is a sample rather than a census, all of its estimates have confidence intervals, Ogden noted. Starting in 1999, the NHANES became a continuous survey, with data being released every 2 years. About 5,000 people are surveyed each year and given standardized examinations.
SOURCES: Presented by Cynthia Ogden on October 6, 2015 (CDC, 2015a; Ogden et al., 2013).
Ogden noted that the precursor to the NHANES was the National Health Examination Survey. In 1971 nutrition was added, and the NHANES began (CDC, 2014). In that year, children aged 1 year and older were surveyed. The survey was extended to children aged 6 months and older in 1976 and to those aged 2 months and older in 1988 (CDC, 2014), and in 1999 it began encompassing all ages starting at birth. The survey includes measurements of weight and height, which are transmitted directly from the scale and stadiometer to a database to reduce the chances of recording errors. Ogden pointed out that the measured data from the NHANES are much more accurate than the proxy or self-reported data gathered in other surveys.
In children, obesity is not defined by a standard cut point as it is for adults, Ogden explained. Rather, anthropometric measures are compared with a reference population. For children less than 24 months of age, weight for recumbent body length is compared with the World Health Organization’s (WHO’s) growth standards (CDC, 2015b). Children are considered to have high weight-for-length if they are at or above the sex-specific 97.7th percentile on the WHO growth chart, which is 2 standard deviations from the mean (CDC, 2015b). Another definition sometimes used is weight-for-recumbent-length at or above the sex-specific 95th percentile on the CDC’s growth charts (CDC, 2015b).
For children aged 2 to 5, body mass index (BMI)—defined as weight divided by the square of a person’s height in metric measures—is compared with a reference population, which in the United States is generally the sex-specific 2000 CDC growth charts, said Ogden. Children are considered overweight if they have a BMI-for-age between the 85th and the 95th percentiles and obese if they are at or above the 95th percentile (CDC, 2015a). More recently, Ogden observed, some have defined severe or extreme obesity as a sex-specific BMI for age above 120 percent of the 95th percentile.
Current Prevalence of Overweight and Obesity
Ogden reported that, based on the WHO growth charts, the prevalence of high weight-for-recumbent-length (defined as greater than the 97.7th percentile) in children less than 2 years of age in the United States is 7.1 percent overall—3.5 percent for boys and 11.0 percent for girls. Using the CDC’s growth charts (which define high weight-for-recumbant-length as greater than the 95th percentile), the corresponding percentages are 8.1 percent overall, 5 percent for boys, and 11.4 percent for girls (Ogden et al., 2014).
Based on 2012 data (the most recent data available), the percentages of 2- to 5-year-olds who are overweight are 14.4 percent for boys, 14.5 percent for girls, and 14.5 percent overall, while the corresponding percentages for obesity are 9.5 percent, 7.2 percent, and 8.4 percent (Ogden et al., 2014).
Among children less than 2 years of age, Ogden reported, there are no statistically significant differences in the prevalence of high weight-for-recumbent length by race/Hispanic origin (Ogden et al., 2014). By contrast, obesity prevalence differs significantly among non-Hispanic white, non-Hispanic black, non-Hispanic Asian, and Hispanic 2- to 5-year-olds (see Figure 2-2). For example, the prevalence of obesity among African Americans and Hispanics is significantly higher than that among non-Hispanic whites or non-Hispanic Asians in this age group. The reason for these differing patterns in the two age groups is unclear, Ogden said, but she suggested that it might be partially explained by differences in both the definition and measurement of excess weight in the two groups. The prevalence of obesity among young children also varies substantially by income in the United States. As of 2011, according to the Pediatric Nutrition Surveillance System, obesity among low-income, preschool-aged children ranged from less than 9.2 percent to 17.9 percent in different states, compared with an overall obesity rate in the general U.S. population of 8.4 percent (see Figure 2-2).
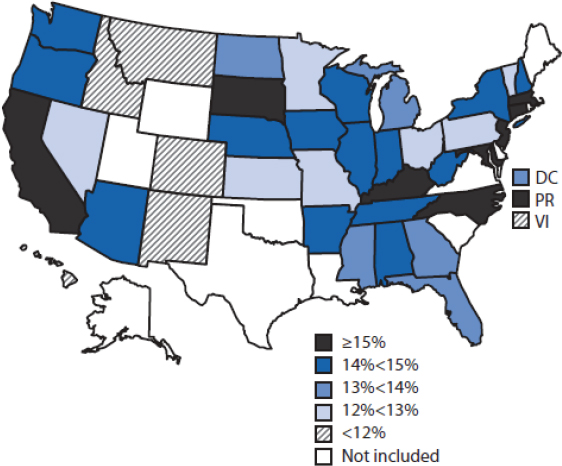
NOTE: DC = District of Columbia; PR = Puerto Rico; VI = U.S. Virgin Islands.
SOURCES: Presented by Cynthia Ogden, October 6, 2015 (May et al., 2013).
Trends Over Time
Ogden reported that among 2- to 19-year-olds in the United States, the prevalence of obesity has more than tripled—from approximately 5 percent in the 1970s to 17 percent in the most recently available data, for 2011-2012 (Ogden et al., 2014) (see Figure 2-3). She noted that if children and adolescents aged 2-19 years are divided into three groups—ages 2-5, 6-11, and 12-19—one sees that obesity has risen more among the latter two groups than among the former (Ogden et al., 2014).
Still, the average weight of 2- to 5-year-olds has risen since the 1970s, Ogden stated. Average weight for 5-year-old girls was 46.6 pounds in 2007-2010, compared with 43.2 pounds in 1976-1980 (Fryar et al., 2012; Najjar and Rowland, 1987). For children less than 2 years of age, the prevalence of excess weight has not changed significantly since data on this age group were first collected in 1999-2000. The difference in trends between these youngest children and 2- to 5-year-olds is particularly interesting, said Ogden. More longitudinal data could help in understanding the difference, she suggested.
Childhood obesity trends were essentially flat over the decade before the most recently available data (Ogden et al., 2014), meaning that the increases occurred largely in the 1980s, the 1990s, and the first few years of the 2000s, said Ogden. Furthermore, for 2- to 5-year-olds, the prevalence of obesity decreased from 13.9 percent to 8.4 percent over the period 2003-
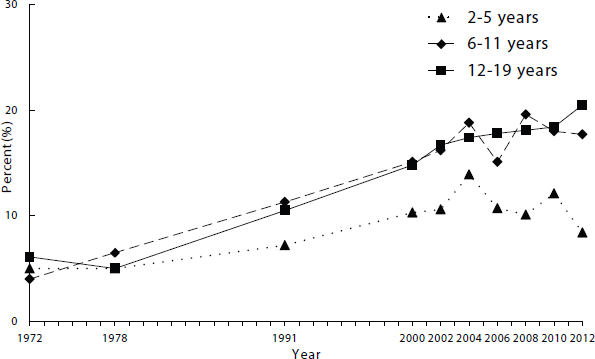
SOURCES: Presented by Cynthia Ogden on October 6, 2015 (Fryar et al., 2014).
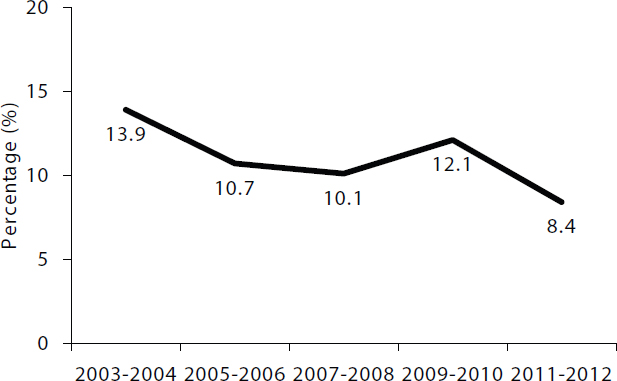
NOTE: 5.5 percentage point decrease (95 percent confidence interval of 1.4 to 9.6 percentage point decrease).
SOURCES: Presented by Cynthia Ogden on October 6, 2015 (Fryar et al., 2014).
2004 to 2011-2012 (see Figure 2-4). Ogden called attention to the need for more data to understand this drop. For example, were the 2011-2012 data an anomaly? Also, the numbers vary from each 2-year survey to the next, although averaging over a longer time period also shows a decline in obesity. “There may be a decrease in these young children,” said Ogden. “We need to see the 2013-2014 [NHANES] data to confirm that.”
Other data sources also show a drop in obesity among preschoolers, Ogden stated. For example, the Pediatric Nutrition Surveillance System found a slight but statistically significant decline in 18 states from 2008 to 2011 among low-income 2- to 4-year-olds participating in federal nutrition programs (May et al., 2013). Similarly, data from Southern California gathered by Kaiser Permanente researchers showed a decline in obesity among 2- to 5-year-olds—from 13.0 to 11.6 percent—between 2008 and 2013, although the change was insignificant (Koebnick et al., 2015).
Persistence of Obesity
A number of studies have found an association between childhood obesity and adult obesity, Ogden noted. For example, the Bogalusa Heart
Study found a moderate association (correlation of 0.3-0.4) between obesity in children when they were 2 to 5 years of age and obesity in those same individuals when they were adults. Compared with children with BMIs at or below the 50th percentile, children who were overweight were four times as likely to be obese as adults (Freedman et al., 2005).
Likewise, Ogden noted, data from the Fels Longitudinal study showed that children who were obese at 3-4 years old had a 15-25 percent chance of being obese in adulthood (Guo et al., 2002). The Early Childhood Longitudinal Study of kindergarteners in 1998-1999 found that children had a 47 percent probability of being obese in 8th grade if they had been obese in kindergarten (Cunningham et al., 2014). A study of data from a Washington State health maintenance organization found that obesity at ages 1 to 2 years was not a significant predictor of obesity during young adulthood, but that obesity between the ages of 3 and 5 years increased the odds of obesity in young adulthood to 4.1 (Whitaker et al., 1997). This study also found that the risk depended on whether the child had a parent with obesity. Finally, the longitudinal Cardiovascular Risk in Young Finns Study found a moderate association of 0.29 for girls and 0.36 for boys between obesity at age 3 and in adulthood (Juhola et al., 2011).
These studies still leave many questions unanswered about the persistence of obesity, noted Ogden, such as whether cohort differences are important. But, she said, they show that persistence occurs and is increasingly likely with older children.
In summary, said Ogden, 7-8 percent of children less than 5 years of age have excess weight, although the percentage depends on the age group in question and the measures used to quantify obesity. Finally, she noted, there are disparities in the prevalence of early childhood obesity along racial, ethnic, and economic lines.
THE EPIGENETICS OF CHILDHOOD OBESITY
The risk of obesity is impacted by the dynamic relationship among genetics, environment, and early childhood development, observed Shari Barkin, William K. Warren Foundation endowed chair and professor of pediatrics at the Vanderbilt University School of Medicine. Study of this relationship is still in its early stages, she noted. But enough is now known, she said, to “examine potential ideas for what might be modifiable or able to be mitigated as we examine innovative solutions for pediatric obesity prevention.”
Human beings are 99.9 percent identical at the genetic level, noted Barkin, yet still have many differences (NHGRI, 2014). Part of the reason for these differences is epigenetics. Barkin defined the science of epigenetics as the study of changes in the expression of genes via post-translational and
post-transcriptional modifications. “Think of it this way,” she said. “You can’t change your genes, but you can change the way your genes behave.”
Barkin explained that in all living species, the environment affects gene expression. Organisms receive cues from the environment that can change which genes are turned on and off (Lobo, 2008), a process that has been described as the social life of genes (Dobbs, 2013). For example, when the larvae of docile European bees are placed in the hives of the killer bee, the normally placid European bees acquire a phenotype identical to that of killer bees (Dobbs, 2013). “The genes didn’t change, but the expression of the genes did,” said Barkin.
Barkin noted that in February 2015, the Institute of Medicine held a workshop on the role of epigenetics in the origins of obesity in fetal development and early life and opportunities for intervention (IOM, 2015). The workshop examined gene expression across the lifetime and at multiple levels of environmental interaction (see Figure 2-5). From the many topics
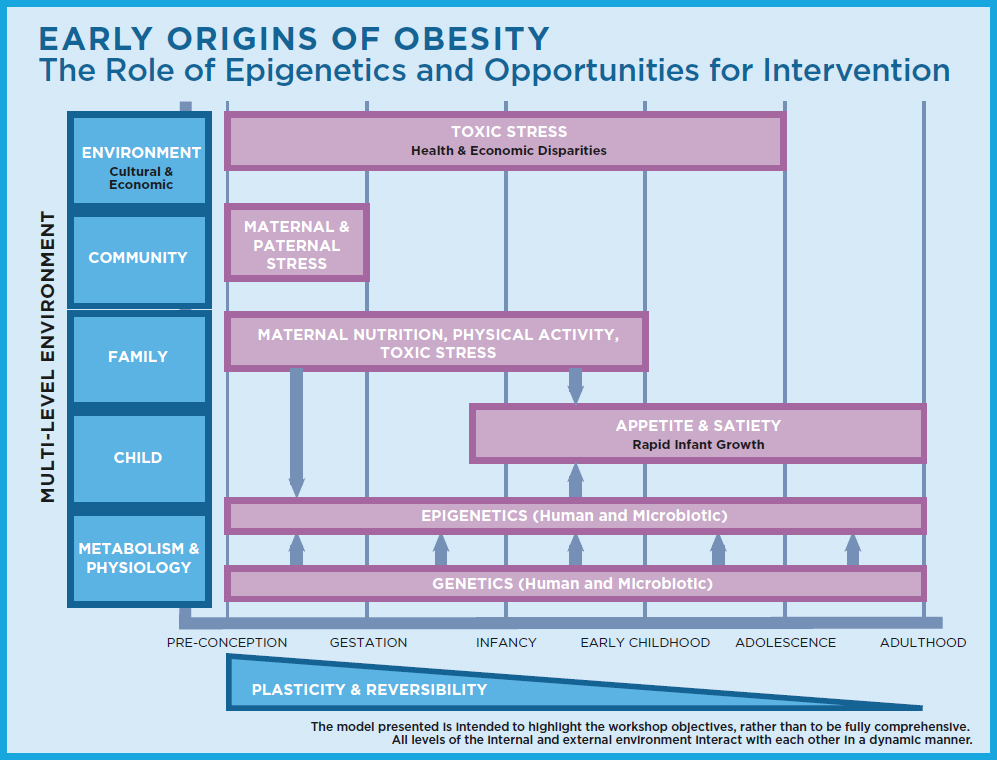
SOURCES: Presented by Shari Barkin on October 6, 2015. From Examining a Developmental Approach to Childhood Obesity: The Fetal and Early Childhood Years (IOM, 2015).
discussed at that workshop, Barkin focused specifically on maternal and paternal nutrition.
When genetically identical mice are exposed to different maternal diets in utero, said Barkin, they can have very different phenotypes, including different weights and different appearances (Waterland and Jirtle, 2003). In particular, mice that would normally be obese instead have a lean phenotype if their mothers are exposed to a diet supplemented with choline, folic acid, and vitamin B12, which affects the methylation of DNA at a genetic locus known as agouti (Waterland and Jirtle, 2003). “Clearly, maternal nutrition is important,” said Barkin.
In animal models, high-fat diets during pregnancy similarly are associated with differential gene expression related to lipid metabolism, glucose metabolism, and appetite, Barkin reported. This altered gene expression can affect carbohydrate and lipid metabolism in offspring, in turn affecting the phenotypes of offspring (Lillycrop and Burdge, 2011). Some of these effects can be mitigated if postnatal nutrition has normal fat composition, Barkin added.
Barkin stated that maternal nutrition also affects the microbiome of mothers and their children—the community of single-celled organisms that live in close association with the body (Peterson et al., 2009). In a study of Japanese macaques, for example, mothers on a Western diet high in fats experienced a shift in microbiome species that affected lipid metabolism and inflammatory response (Friedman, 2015). Exposure to a maternal Western diet appears to pattern the microbiome and effect a pro-inflammatory state in infants (Friedman, 2015; Kumar et al., 2014). Although studies in the past have focused on correlations, many studies currently under way are looking at causation, Barkin reported.
Paternal nutrition also matters, said Barkin. Epigenetic modifications appear to act through the sperm that fertilize eggs (Lane et al., 2015). In rats, for example, a high-fat paternal diet increases beta cell dysfunction in female offspring (Ng et al., 2010). Paternal obesity also appears to affect the metabolic and reproductive health of the offspring for multiple generations (Lane et al., 2015). Barkin suggested that some of these effects could be addressed with improved paternal nutrition.
These findings have important implications for better understanding childhood obesity, Barkin pointed out. For example, Godfrey and colleagues (2011) examined longitudinal cohorts of children and epigenetic signatures of their umbilical cord tissue and then looked at fat distribution in the same children at age 9. Methylation patterns at several genes were associated with increased adiposity in an independent sample of children, although no causal mechanism was identified. The authors concluded that perinatal epigenetics could provide an opportunity to assess individual vulnerability to childhood obesity.
Barkin also described a 7-year randomized controlled trial of family-based community-centered interventions to prevent obesity in preschool-aged children that she and her colleagues are conducting. Using saliva to probe epigenetic signatures, they have found methylation patterns in children at risk of obesity that are associated with the BMI of their mothers.
To summarize this intriguing body of research, Barkin highlighted the field’s potential “to identify at-risk [for obesity] children as well as understand mechanisms . . . [and] shed light on factors that could be potentially mitigated.”
THE FLAVOR WORLD OF CHILDHOOD: BASIC BIOLOGY AND HEALTH IMPLICATIONS
Many chronic illnesses that plague society derive in large part from poor food choices that are dictated by people’s taste preferences, stated Julie Mennella, a member at the Monell Chemical Senses Center in Philadelphia. “We don’t have a problem of children over-consuming broccoli,” she said. “They are over-consuming added sugars and salt, [with] too few fruits and vegetables. This pattern of food choice that is evident in the youngest members of our society mirrors that of our nation’s adults.”
What can account for patterns of food choice that appear to be antithetical to health and for the difficulty of changing those patterns? Mennella pointed to two factors that can predispose some children to consume obesogenic diets: inborn, evolutionarily driven taste preferences, and the detrimental consequences of not being exposed to the flavors of healthful foods early in life (Mennella and Ventura, 2011; Trabulsi and Mennella, 2012).
Evolution has shaped the taste of foods children initially prefer or reject, said Mennella. In an environment with limited nutrients and abundant poisonous plants, sensory systems evolved to detect and prefer the once rare energy (carbohydrate)-rich and sodium-rich foods that taste sweet and salty and to reject toxic foods that taste bitter (Forestell and Mennella, 2015).
Mennella went on to explain that the chemical senses that underlie the flavors of foods and beverages are functioning by at least the second trimester of pregnancy (Forestell and Mennella, 2015). Taste receptors are found not only in the oral cavity but also in the nasal cavity (Lee and Cohen, 2015) and in the gut (Egan and Margolskee, 2008), and activation of these receptors in the mouth can be a source of extreme pleasure or pain. While some responses are inborn (e.g., the liking of sweets), Mennella said, they can be shaped by learning.
Mennella focused first on sweet taste, which she contended is one of the most powerful taste signals for a baby and humans’ oldest reward (Mennella, 2008). Babies are born attracted to sweet taste, the predominant taste quality of mother’s milk. Within hours of birth, infants ingest more
of a sweetened solution than water and can differentiate varying degrees of sweetness (Desor et al., 1973). They suck more strongly on a nipple sweetened with sucrose than on an unsweetened nipple (Maone et al., 1990), and this potentiating of sucking is evident even in premature infants (Maone et al., 1990). Putting a drop of sugar in solution in a baby’s oral cavity produces a smile, a calming effect, and a sucking effect (Steiner, 1979), and it blunts expressions of pain (Pepino and Mennella, 2005; Stevens et al., 2010). “Sugar is very powerful,” said Mennella. “It is not just a preferred taste.”
The preference for sweet taste—the taste signal for energy (e.g., fruits)—remains heightened during childhood and periods of maximal growth, Mennella continued (Coldwell et al., 2009; Mennella et al., 2014). It is evident among children around the world (Mennella, 2008). Relative to adults, children as a group prefer a more intense sweet sensation, Mennella noted, and the adult pattern of sweet taste preferences does not emerge until mid-adolescence (Mennella et al., 2011). “Even your teenager is living in a different world when it comes to sweet,” she said.
“As much as children like the taste of sweet, they dislike the taste of bitter,” Mennella continued. This dislike serves an important role, by “protecting the animal from consuming that which is toxic,” she reiterated. Like sweet preferences, sensitivity to some bitter tastes also changes with age, attaining the adult pattern during mid-adolescence (Mennella et al., 2010, 2011).
In the current food environment, the elegant biology that attracts children to mother’s milk and to sources of energy and protects them from poison makes them vulnerable to obesity, contended Mennella. “The biology hasn’t changed,” she said. “The environment has changed. [It is] rich in added sugars or salt.” While the biological drive to avoid bitter and prefer salty and sweet foods may have served children well in a feast-or-famine setting, this biology explains why children are vulnerable to the current food environment abundant in highly processed and palatable foods and rich in added sugars, non-nutritive sweeteners, and salt. Understanding this vulnerability is key to developing evidence-based strategies for obesity prevention in children, said Mennella.
Mennella acknowledged that the ingrained biology of avoiding bitter tastes and liking sweet ones will not be changed easily. However, good news comes from research on how sensory experience beginning early in life can modify flavor and food preferences. “Our biology is not our destiny,” said Mennella. “These senses can learn. We can get children off to a healthy start if they are provided with those experiences, and the earlier the better.”
Mennella and her colleagues have focused on fruits and vegetables. A diet insufficient in fruit ranks as the leading dietary risk factor for the global burden of disease, with a diet low in vegetables ranking somewhat lower
(Ezzati and Riboli, 2013). The earliest information about these important sources of nutrients comes from the mother, both before birth and after (Neville et al., 2012). A variety of flavors are transmitted from the mother’s diet to amniotic fluid (Mennella et al., 1995) and, if she breastfeeds, to breast milk (Mennella, 2007; Mennella and Beauchamp, 1991a,b, 1996, 1999). Infants detect these flavors, Mennella noted, and experience modifies their acceptance of similarly flavored foods. She and her colleagues found, for example, that the babies of mothers who ate carrots during pregnancy or lactation ate more carrots and made less negative faces while doing so than the babies of mothers who avoided carrots (Mennella et al., 2001). Similarly, the babies of mothers who had more fruits in their diet were more accepting of fruits upon first tasting them (Forestell and Mennella, 2007). The same phenomenon has been found in experimental results from other animals (Aigueperse et al., 2013; Mennella, 2007), Mennella noted. “It is a fundamental feature of how animals first learn about what foods are safe,” she said. “The young learn from their mothers what foods she prefers, what foods she has access to, and what foods define their culture.”
This learning about foods continues during complementary feeding, when solid foods are introduced into the infant’s diet, Mennella continued. She pointed out that the presence of a food in a child’s environment does not ensure that the child will learn to eat that food. Rather, the child must taste the food to learn to like it (Birch et al., 1987). With respect to fruits and vegetables, infants, whether breastfed or formula fed, continue to learn to like their flavor through repeated (8-10 days) exposure to a particular fruit or vegetable or to a variety of these healthy foods (Mennella et al., 2001; Sullivan and Birch, 1994). This learning is reinforced if the foods are part of the maternal diet (Sullivan and Birch, 1994), and is more efficient during infancy than in later childhood (Birch and Marlin, 1982). But the reality is that many children are deprived of learning to like the taste of fruits and vegetables, Mennella said. From the age of 2 years, children are more likely to consume a manufactured sweet than a fruit or vegetable on any given day (Saavedra et al., 2013). By age 4, 92 percent of children consume less than the minimum recommended amount of vegetables, while 99 percent of children exceed the recommended intake of added sugars (Krebs-Smith et al., 2010).
These dietary patterns, which are set before the age of 2 years (Saavedra et al., 2013), cannot be underestimated, Mennella stressed. Infrequent intake of fruits and vegetables during late infancy is associated with infrequent intake at 6 years (Grimm et al., 2014). The evidence suggests that the diets of children often mirror those of their parents, and consequently many are not having the experiences they need to learn to like fruits and vegetables, Mennella said.
Ending on a hopeful note, Mennella pointed to periods of life in which
old routines fall away and habits are suddenly in flux (Duhigg, 2012). One such period is around the birth of a child. For example, Mennella noted, many smoking cessation plans have been successful with pregnant women (Lumley et al., 2009). “Being pregnant and then a parent is probably one of the strongest motivators to change or modify behaviors,” she said. For some women, simply knowing that their babies are learning about what they eat could make a difference, she suggested. She also recommended focusing on older adolescents as their taste and flavor preferences begin to change (Mennella et al., 2010, 2011). “They are the future parents of the next generation,” she said. “It may be another opportune time for individuals who may be motivated to change to establish healthy eating habits that will be passed on to their children.”
In conclusion, Mennella emphasized that basic research in both humans and animal models is critical to continued advances and applications in this area. Food is much more than taste or a source of calories, she said. “It gives us pleasure. It identifies who we are as a people, as a family, as a culture. It symbolizes our relationship to our environment.”
DISCUSSION SESSION: OPPORTUNITIES FOR INTERVENTIONS
During the discussion session, the presenters turned their attention more directly to some of the kinds of interventions that could be supported by the science they had described. For example, Barkin emphasized the potential for further research to identify critical periods or populations in which interventions could be especially effective. “When we supplement appropriately, it might have greater benefits for certain ages or genders,” she said. “We also have to manage the unintended consequences of who it might not be good for.” Improved diets before and during pregnancy likely have an effect beyond any nutritional supplement. “There is no magical pill that seems to be associated with better outcomes; rather pre-pregnancy BMI and gestational weight gain seem to be potential points for intervention.”
Mennella emphasized the importance of focusing on the family environment in investigations of how to improve diets. “You cannot feed babies separately than you feed a family,” she said. “The baby is nourished by what the mother eats and that learning continues after birth, especially if the baby is breastfed.” She noted that, while children often eat specially prepared foods during early complementary feeding, the goal is to have the child eat the foods of the family. For that reason, she suggested thinking about the diet of the family rather than the diet of only the child, a perspective that could help make healthy food habits more sustainable. Children can learn to like healthy foods, she reiterated, and the earlier they are exposed to them in the positive context of their families, the more successful the outcomes will be.
Barkin recommended looking at the framing of food and beverage consumption. If the default option with a fast food meal is fruit and milk rather than french fries and a soda, more people will eat these foods, she said. She also observed that discussion of healthy eating inevitably involves public policies, which again highlights the importance of both the evidence and family dynamics. “If we can consider how policies address families together as a unit, we could have the opportunity to shift normative behaviors,” she suggested. For example, policies could help spread knowledge about what kinds of foods and what kinds of eating habits are best, such as not grazing on foods and beverages throughout the day. “Grazing overrides our homeostatic mechanism of saying, ‘Hey, I’m full and I don’t need to eat right now,’” she noted.
Finally, Barkin suggested that evidence needs to be presented to policy makers and the public in a way that tells a story. “Evidence, unfortunately, is not enough to change policy,” she said. “It is the way in which you tell the story of the evidence.”














