8
Meeting the Challenge of Vision Loss in the United States: Improving Diagnosis, Rehabilitation, and Accessibility
Vision impairment can adversely affect an individual’s health, functioning, and quality of life (QOL) in numerous ways. “Low vision adversely affects many daily activities, such as writing a check, telling time, looking for daily items, using a phone, managing medications, and preparing a meal” (Liu et al., 2013, p. 280). Vision impairment may comprise a patient’s mobility, as well as his or her ability to read and drive (Brown et al., 2014; Owsley and McGwin, 2010; Swenor et al., 2015). Vision impairment in children and young adults affects development and academic performance, with a long-term, detrimental impact on QOL (Davidson and Quinn, 2011). Vision impairment is also associated with reduced mental and physical health (Crews et al., 2006, 2016b; Zhang et al., 2013a) and with increased risk of falls, injury, and mortality (Christ et al., 2014; Crews et al., 2016a; Patino et al., 2010). Vision impairment is associated with less education, lower income, and lower employment rates (Erickson, 2016). Chapter 3 discusses in further detail the adverse effects of vision impairment on QOL, independence, mobility, falls and injuries, mental health, cognition, and mortality.
A considerable portion of vision impairment can be prevented, mitigated, or reversed through public health intervention and clinical treatment. For example, estimates of uncorrected refractive error range from 8.2 million to 15.9 million (Varma et al., 2016; Wittenborn and Rein, 2016). However, even if all preventable cases of vision impairment were eliminated, millions of Americans would still live with vision impairment because of limits in the ability to prevent and manage eye disease. In 2015, 4.24 million U.S. adults ages 40 and older were affected by uncorrectable
vision impairment, including blindness; this number is projected to rise to almost 8.96 million by 2050 (Varma et al., 2016).1 Given the magnitude of this burden, meeting the health care needs of visually impaired populations is a challenge—a challenge that will only grow over time. Yet, with appropriate access to high-quality care and interventions that effectively manage vision loss, individuals with chronic vision impairment can gain significant advantages in facing this major health obstacle. Lacking these services and interventions, however, the consequences of vision impairment on one’s life can be substantial and broad ranging.
Congenital, genetic, and acquired eye diseases and conditions, such as untreated amblyopia, retinitis pigmentosa, and Stargardt’s disease can lead to chronic vision impairment in children and young adults (Bradfield, 2013; NEI, 2014, 2015). Chronic vision impairment in these populations can impose a considerable public health burden by adversely affecting health and socioeconomic outcomes across the lifespan. The greatest burden of eye disease, however, falls on older populations, who are at increased risk for age-related eye disease (CDC, 2009; Gilbert and Foster, 2001). In their advanced forms, age-related eye diseases such as glaucoma and diabetic retinopathy can result in irreversible vision loss, and the aging of the U.S. population means the prevalence of these diseases and the vision impairment they can entail is projected to increase in the coming decades (NEI, 2010).
For individuals with vision impairment that cannot be corrected by available refractive, medical, or surgical treatments, vision rehabilitation has the potential to prevent depression and emotional distress and to improve or maintain QOL, the ability to perform daily activities, and overall visual ability (Brody et al., 2005; Goldstein et al., 2015; Lamoureux et al., 2007; Walter et al., 2007).2 According to an Agency for Healthcare Research and Quality (AHRQ) report, the aim of vision rehabilitation is “to maximize the use of any residual vision that an individual might have and provide practical adaptations that reduce the disabilities associated with low vision or blindness” (AHRQ, 2004, p. 36). Desired outcomes of vision
___________________
1 In 2015, 3.22 million adults ages 40 and older had vision impairment (visual acuity worse than 20/40, but better than 20/200), and 1.02 million were blind (visual acuity of 20/200 or worse, based on visual acuity in the best-corrected, better-seeing eye). In 2050, 6.95 million adults ages 40 and older are projected to have vision impairment and 2.01 million are projected to be blind (Varma et al., 2016).
2 Vision rehabilitation and low vision rehabilitation refer to the same care processes and models. As noted in Chapter 1, this report will use the term vision impairment, rather than low vision, to describe patients with functional limitations of the eye(s) or visual system that result from vision loss. In keeping with this decision, vision rehabilitation—rather than low vision rehabilitation—will be used throughout the report. See Chapter 1 for further information on terminology.
rehabilitation include increased functional ability, independence, and QOL (AHRQ, 2004). However, achieving these outcomes is made challenging by the existence of several impediments to the provision of accessible and high-quality vision rehabilitation services.
Awareness of the benefits of vision rehabilitation may be limited among providers and patients, which creates missed opportunities for improving health outcomes (Casten et al., 2005; Lam and Leat, 2013; O’Connor et al., 2008). Limitations in the eye and vision care workforce and in the availability of some vision rehabilitation services may prevent patients from accessing needed care (Goldstein et al., 2012; Owsley et al., 2009). Minority race and ethnicity and lower socioeconomic position pose barriers to accessing primary eye and vision care and could potentially compromise access to vision rehabilitation services as well (Zhang et al., 2012, 2013b). Inadequate research on the cost, effectiveness, and cost-effectiveness of vision rehabilitation services constrains treatment options that providers may consider or confidently recommend.
This chapter explores the role of vision rehabilitation in promoting the health, functioning, and QOL of people in the United States living with chronic vision impairment. The vision rehabilitation process as well as key interventions and unique care models will be discussed. Major barriers to vision rehabilitation quality and access will be explored, as well as strategies to overcome them. The chapter will also highlight opportunities and innovative directions related to models of care that hold great promise for lessening the impacts and consequences of chronic vision impairment.
VISION REHABILITATION INTERVENTIONS AND MODELS
Overview of the Vision Rehabilitation Process
Vision rehabilitation services comprise a wide array of models of care and interventions and services provided by numerous professionals working in varied clinical settings to accomplish a diverse range of goals. According to the National Eye Health Education Program of the National Eye Institute (NEI):
Vision rehabilitation helps people adapt to vision loss and maintain their current lifestyle. A vision rehabilitation program offers a wide range of services, including training in the use of magnifiers and other adaptive devices, ways to complete daily living skills safely and independently, guidance on modifying residences, and information on where to locate resources and support. These programs typically include a team of professionals consisting of a primary eye care professional and an optometrist or ophthalmologist specializing in low vision. Occupational therapists,
orientation and mobility specialists, certified low vision therapists, counselors, and social workers may also be a part of this team. (NEHEP, n.d.)
Binns et al. (2012) states that “vision rehabilitation services conform to a variety of different models” that vary in the number and type of services they emphasize and offer (p. 36). Vision rehabilitation models of care include hospital-based vision rehabilitation services, multidisciplinary outpatient and community-based services, and self-management education programs (Ryan, 2014). Other current and proposed models offer a range of tiered, increasingly comprehensive services designed to meet the needs of patients with different needs and degrees of vision impairment (Leat, 2016; VA, 2015). In addition to optometrists and ophthalmologists, vision rehabilitation can involve opticians, occupational therapists, physical therapists, vision rehabilitation therapists, vision rehabilitation teachers, social workers, orientation and mobility specialists, psychologists, and counselors, among others (Binns et al., 2012; Markowitz, 2006b). Vision rehabilitation services can be provided in specialized vision centers, general or rehabilitation hospitals, outpatient clinics, extended care facilities, private and university-based optometry and ophthalmology practices, private homes, community-based health centers, and facilities operated by government agencies, nonprofit organizations, or community groups (Deremeik et al., 2007; Owsley et al., 2009; Ryan, 2014).
Despite this heterogeneity in the provision, organization, setting, and scope of care, vision rehabilitation services share a common, overarching model of care. This overarching model describes vision rehabilitation as a process of care that begins with examination and diagnosis, followed by development of a treatment plan, and then proceeds to management of vision impairment (Markowitz, 2006b). Reevaluation of the patient leads to adjustments as necessary to the treatment plan until the patient’s needs have been addressed or treatment options have been exhausted. Below, Figure 8-1 depicts the vision rehabilitation process in the context of the larger continuum of eye and vision care.
Clinical practice guidelines (guidelines) developed by the American Academy of Ophthalmology and American Optometric Association (AOA) recommend comprehensive eye exams as the initial step in the vision rehabilitation process (AAO, 2013; AOA, 2007). Initial care of the vision rehabilitation patient may include an initial assessment to determine the patient’s medical history, a brief examination to confirm the medical history, determination of cognitive function, and identification of priority tasks for rehabilitation, followed by comprehensive assessment of residual visual function and residual functional vision (Markowitz, 2006b). Assessment of residual visual function can include assessment of refractive error, visual acuity, visual fields, oculomotor functions, cortical
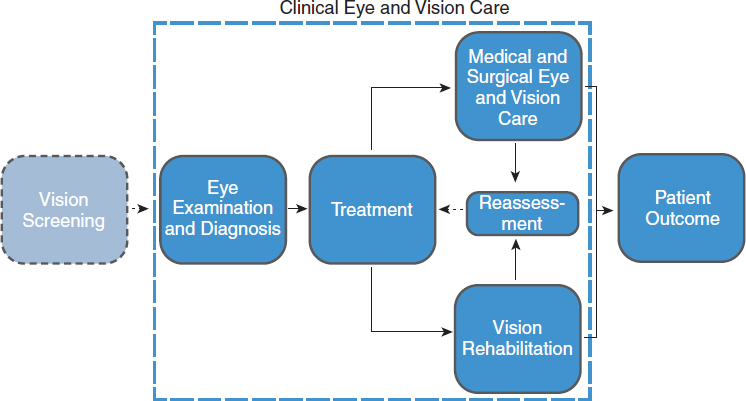
visual integration, and light characteristics affecting visual (Markowitz, 2006b). The assessment of residual functional vision is used to determine “how well a patient uses residual visual function to perform routine tasks in different places, using different items, and throughout the day,” and its results inform the development of the treatment plan (Markowitz, 2006b, p. 299).
Vision rehabilitation treatment plans are guided by the concept of patient-centered care, and therefore account for the unique preferences, goals, priorities, and relevant contextual factors (e.g., health status, social and educational supports, presence of stakeholder networks) of the individual patient when addressing the consequences of vision loss (IOM, 2001). Among U.S. adults referred to vision rehabilitation services, such consequences often include difficulty reading, driving, using assistive devices, performing daily activities inside and outside the home, walking, watching television, and performing work- and school-related activities (Brown et al., 2014).
To address these problems, vision rehabilitation treatment plans may include prescription of, and training in the use, of low vision aids and devices, orientation and mobility training, modification of home and work environments to promote functioning and safety, training in adaptive strategies and skills, and counseling, among other interventions and services (Binns et al., 2012; Markowitz, 2006b; Mogk and Goodrich, 2004; Ryan, 2014). The effectiveness and scope of vision rehabilitation models of care and services and interventions are explored below.
The Effectiveness of Vision Rehabilitation
Although evidence for the effectiveness of vision rehabilitation is still emerging, some available research does indicate that vision rehabilitation helps improve health and visual functioning for patients in numerous age groups and with different degrees and causes of vision impairment. One prospective, observational study measured the effects of vision rehabilitation on overall visual ability and on performance in four domains: reading, mobility, visual motor function, and visual information processing (Goldstein et al., 2015). Among 468 adult patients seeking vision rehabilitation services, 47 percent achieved a minimum clinically important difference in overall visual ability between baseline and reevaluation at 6 to 9 months. Over the same 6- to 9-month period, 27 percent to 44 percent of participants experienced minimum clinically important differences in their performance in a functional domain (Goldstein et al., 2015). A randomized controlled trial (RCT) compared the effects of vision rehabilitation to no treatment on reading ability, mobility, visual information processing, and visual motor skills among 126 adults with vision impairment (Stelmack et al., 2008).3 Over 4 months, participants receiving vision rehabilitation achieved significant improvements in all functional domains, while those receiving no treatment experienced declines in all functional domains. A survey of 105 patients who reported receipt of vision rehabilitation found that these services were associated with significant improvements in the ability to perform near-vision activities (e.g., reading ordinary and small print, performing near-vision work, self-grooming, ambulating down stairs during full- and low-light conditions) and distance-vision activities (e.g., reading street signs, recognizing people, watching television). Patients also reported nonsignificant improvements in perceived ability to perform vision-related social activities (Walter et al., 2007).4 Specific vision rehabilitation programs and interventions have been associated with increased social participation and overall QOL, as well as decreased emotional distress and increased vision-related functioning, especially among patients with visual impairments who were depressed prior to receiving vision rehabilitation (Berger et al., 2013; Brody et al., 2002, 2005; Lamoureux et al., 2007).
___________________
3 Vision impairment was defined as visual acuity of worse than 20/100 and better than 20/500 in the better-seeing eye. Participants had diagnosis in the better-seeing eye of macular degeneration, macular dystrophy, macular hole, or inflammation of the macula. Vision rehabilitation included examination, education on the eye disease diagnosis and prognosis, prescription of low vision devices, five weekly sessions of low vision therapy, and one home visit.
4 Vision rehabilitation included comprehensive low vision examinations and functional vision assessments, assessments of daily activities, referrals to community services, orientation and mobility evaluations, counseling, and device funding.
Although most patients in vision rehabilitation are adults, these services also appear to confer gains in visual functioning on children and adolescents. An observational study on the effects of vision rehabilitation among 183 children ages 8 to 16 with vision impairment who were living in India found that visual disability was significantly decreased 3 to 4 months after baseline for participants with mild, moderate, and severe vision impairment (Gothwal et al., 2015). Among children ages 6 to 16 with vision impairment, prescription of, and training on, low vision aids significantly improved near and distance visual acuity and self-reported ability to perform activities such as reading textbooks and copying from a blackboard (Ganesh et al., 2013).5 However, a systematic review found that vision rehabilitation in children is severely understudied, most investigations are descriptive case series with small sample sizes, and these limitations permit few conclusions to be drawn from the available literature (Chavda et al., 2014).
Research suggests that patients with vision impairment consequent to eye diseases, including age-related macular degeneration (AMD), glaucoma, diabetic retinopathy, and amblyopia can all benefit from rehabilitation (Demers-Turco, 1999; Ganesh et al., 2013; Hooper et al., 2008; Luo et al., 2011; Nilsson, 1986). However, the extent of the benefits of vision rehabilitation may vary among patient groups, measures of visual functioning, and rehabilitation interventions and models. Wang et al. (2012) found that multidisciplinary vision rehabilitation led to significantly greater improvements in overall visual ability among visually impaired patients than in legally blind patients at 30 days and 3 months follow-up (p. 1403). Patients ages 85 and older saw significantly greater improvements in overall visual ability than patients ages 84 and younger, as did patients without glaucoma versus those with the disease (Wang et al., 2012).6 Another study found that the impact of vision rehabilitation on visual ability varies among functional domains. Specifically, a greater proportion of patients receiving vision rehabilitation achieved minimum clinically important differences in reading ability (44 percent), than in vision motor function (38 percent), vision information processing (33 percent), or mobility (27 percent) (Goldstein et al., 2015). A study comparing two methods of training adult patients
___________________
5 Vision rehabilitation included examination, educational guidance and counseling, training in use of assistive software, and prescription of, and training in, low vision aids.
6 Vision impairment replaces the term “low vision,” defined as a visual acuity of <20/60, but ≥20/200. Legally blind was defined as best-corrected visual acuity of <20/200 or constriction of visual field to within 10 degrees of fixation, irrespective of visual acuity. Intergroup differences in the impact of vision rehabilitation are based on the U.S. Department of Veterans Affairs Low-Vision Visual Functioning Questionnaire (VA LV VFQ-48) scores. Scores from the Impact of Vision Impairment Questionnaire showed no significant intergroup differences in the impact of vision rehabilitation.
with vision impairment in the use of low vision aids for reading found that extended training (i.e., five weekly 1-hour sessions) as compared to standard training (i.e., one weekly 1-hour session) was associated with significantly greater improvements in reading speed and accuracy at 5 and 12 weeks from baseline. At 12 weeks from baseline, compared to participants who received standard training, those who received extended training reported less difficulty performing common vision-related tasks and rated their eyesight higher (Scanlan and Cuddeford, 2004). As discussed below, clinical outcomes achieved by different models of care in vision rehabilitation also vary, though differences in study methodologies and patient populations make it difficult to compare the clinical effectiveness of these models.
These and other research findings suggest that generalizations about the effectiveness of vision rehabilitation services may not be applicable to a particular intervention, model of care, or treatment plan in vision rehabilitation. Similarly, the diversity in the limitations, needs, and priorities of vision rehabilitation patients mean that those interventions and models of care found to be effective for one patient or patient group may be of limited therapeutic value to another. A systematic review concluded that while research suggests that vision rehabilitation can benefit patients with vision impairment, the available evidence is insufficient to make more specific claims about the characteristics of an effective rehabilitation program, the patient populations most likely to benefit from rehabilitation, the value of rehabilitation for children, or the cost-effectiveness of vision rehabilitation (Binns et al., 2012). Future research on vision rehabilitation will need to employ a standardized methodology and prioritize investigations that identify the effectiveness of specific interventions and care models in achieving the unique treatment goals of a given population.
Vision Rehabilitation Services and Interventions
As described above, accomplishing the many individualized goals of vision rehabilitation will require an array of interventions and many providers. Optometrists and ophthalmologists may prescribe low vision optical systems that improve functionality in activities ranging from reading and writing to driving and social events (AAO, 2013; AOA, 2007). Orientation and mobility specialists can provide training in the use of white mobility canes, guide dogs, and other techniques that help preserve mobility in the home and community (ACVREP, 2014a). To improve productivity and functioning in the workplace and beyond, occupational therapists teach patients with vision impairment adaptive strategies, including the use of sensory techniques that rely on touch and hearing to perform tasks (Markowitz, 2006a). Low vision therapists and vision rehabilitation therapists may assist patients with vision impairment in adapting their work and
home environments to promote functioning and safety, or train patients in the use of low vision aids and devices, as well as techniques to maximize remaining visual function (ACVREP, 2014b). It is important to note that the individuals involved in the care of patients with vision impairment may also be informal caregivers who are not trained in vision rehabilitation. Furthermore, several types of providers, including low vision therapists and orientation and mobility specialists, are not recognized as providers by Medicare and, as a result, cannot be compensated by Medicare for the care of beneficiaries with vision impairment. The following section describes in greater detail the interventions that clinicians, therapists, and specialists provide in the course of treatment.
Optical Low Vision Aids
An impaired ability to read is a common complaint among patients seeking vision rehabilitation. Among 819 new patients at 28 vision rehabilitation centers in the United States, 66.4 percent reported difficulty reading (Brown et al., 2014). In another study, 69 of 87 participants identified improved reading ability as a goal of vision rehabilitation. Moreover, 89.9 percent of those who identified improved reading ability as a goal ranked this goal as very important (Renieri et al., 2013). Prescription and training in the use of optical low vision aids is one strategy for improving reading ability and other measures of visual functioning. Non-electronic optical low vision aids include spectacle-mounted and hand-held microscopes and telescopes for near and distance magnification, prisms for field enhancement, and selective absorption filters and occlusion for contrast and glare control (AOA, 2007; Minto and Butt, 2004). Electronic optical low vision aids include electronic video magnification (e.g., hand-held “camera,” closed circuit television [CCTV]) and computer-based magnification (AOA, 2007; Minto and Butt, 2007). A systematic review to assess the effectiveness of occupational therapy interventions for improving reading ability in older adults with vision impairment found moderately strong evidence to support the use of electronic magnifiers, while the evidence supporting the use of optical magnifiers was limited. One study reported that vision rehabilitation including training in the use of magnifying devices and vision aids resulted in significant improvements in self-estimated reading ability (Renieri et al., 2013). Another study investigated the effect of low vision aids on the reading ability of 530 adults ages 52 to 98 and diagnosed with AMD (Nguyen et al., 2009). Without low vision aids, reading speed was less than 30 words per minute (wpm) for almost all participants. After training in the use of electronic and non-electronic low vision aids, reading speed increased significantly from 20 ± 30 wpm to 72 ± 35 wpm. Improvements in reading speed were significantly greater for individuals with best-corrected distance
visual acuity of ≥20/200 or better in the better-seeing eye compared to those with best-corrected distance visual acuity of <20/200 in the better-seeing eye (Nguyen et al., 2009).
By comparison with these promising findings, several systematic reviews have found the evidence supporting the use of low vision aids to be insufficient or of low quality. For example, a systematic review examining the evidence for the impact of conventional hand-held magnification, optical telescopes, and stand-mounted or hand-held electronic magnification on reading ability in people with vision impairment, concluded that there was insufficient evidence due to the small size and moderate to low quality of available studies, and the outdated research on electronic magnification in particular (Virgili et al., 2013). Two other systematic reviews found no RCTs or quasi-RCTs comparing optical low vision aids to standard refractive correction or exploring the use of assistive technologies (e.g., CCTV, electronic vision enhancement systems, tablet computers, screen readers, screen magnifiers, optical character recognition programs) among children ages 5 to 16 with vision impairment (Barker et al., 2015; Thomas et al., 2015). The discrepancies observed among studies regarding the effect of optical low vision aids on reading ability and other measures of visual functioning may be the result of several differences in the characteristics of study cohorts, the types of optical low vision aids provided, and the duration and quality of training in the use of these aids. For example, studies suggest that difficulty using visual assistive equipment is a common concern among patients in rehabilitation programs, and that extended training in low vision aids is correlated with an improved reading ability and increased QOL (Brown et al., 2014; Scanlan and Cuddeford, 2004). As indicated by the findings of systematic reviews, more research is needed to determine how training, patient characteristics, and other factors mediate the impact of low vision aids on visual functioning.
Orientation and Mobility Training
Performance in mobility-related activities is a common concern among patients seeking vision rehabilitation services. In the Low Vision Rehabilitation Outcomes Study, 16.3 percent of participants reported difficulty with walking or with daily activities outside the home (Brown et al., 2014). A survey of older adults seeking vision rehabilitation in the United States found that 52 percent of 564 responding participants reported a fall in the previous 2 years, and that 23 percent and 16 percent of 699 responding participants reported using a straight cane or a walker/rollator, respectively (Goldstein et al., 2012). Defined as “a sequential process in which visually impaired individuals are taught to utilize their remaining senses to determine their position within their environment and to negotiate safe
movement from one place to another,” orientation and mobility is a strategy for improving the functional status of individuals with vision impairments (ACVREP, 2015, p. 5).
Techniques to improve mobility include the use of white mobility canes, guide dogs, human guides, and mental mapping. Research to assess the effectiveness of these techniques is limited. For example, a recent systematic review discovered very few studies or other documents on the development, content, feasibility, or effectiveness of white cane training programs (Ballemans et al., 2011). More generally, a 2010 systematic review was unable to reach any conclusions regarding the effectiveness of mobility and orientation training in general, and it recommended carrying out RCTs to compare the effectiveness of different training methods (Virgili, 2010). Another systematic review found insufficient evidence to support multidisciplinary vision rehabilitation, driving simulator training, or driver education programs as effective interventions for improving or maintaining community mobility (Justiss, 2013).
Zijlstra et al. (2013) report on the development of a structured and theory-based approach to white cane training that includes two in-person meetings and one telephone conference that emphasize problem-solving and addressing psychosocial issues related to the use of the cane, in addition to practical training (pp. 7, 17). An initial study comparing the effectiveness of this novel training program approach with regular training programs among 68 older adults with vision impairment found no significant differences (89 percent versus 84 percent) in the proportion of participants reporting that training was beneficial, although participant ratings of overall training, trainer performance, and participant engagement were higher for the standardized programs than for the regular program (Ballemans et al., 2012).
Environmental Modifications
Among the 819 patients recruited for the Low Vision Rehabilitation Outcomes Study, 11.7 percent reported problems with glare and lighting, 15.1 percent reported problems with performing in-home activities, and 10.3 percent reported vision-related difficulties in recognizing faces and interacting in social situations (Brown et al., 2014). Modifications to the home and work environment can help address these issues. Interventions include marking the edges of stairs with brightly colored tape, using large print reading materials, placing raised or fluorescent markings on the dials and buttons of appliances, and placing drapes and lamps to decrease glare and increase luminescence (AHRQ, 2002). Research into the impact of these interventions is limited: a recent systematic review of environmental interventions for individuals with vision impairment found no RCTs or
quasi-RCTs that compared the effect of environmental and/or behavioral interventions to other environmental and/or behavioral interventions or controls on adults ages 60 and older with vision impairment (Skelton et al., 2013). An AHRQ report observes that the paucity of controlled trials may be due to the individualized nature of these interventions, and the fact that third-party payors have not pursued or called for research to support these interventions because they “have not typically been subject to reimbursement” (AHRQ, 2002).
Nonetheless, some evidence exists to support specific modifications. For example, one study found that reading performance among study participants with vision impairment consequent to AMD was significantly improved under optimal lighting levels as compared to the lighting level found inside most homes and other buildings (Bowers et al., 2001). Lighting improvements have also been shown to improve QOL and also the performance of some activities of daily living (ADLs) among individuals with vision impairment (Brunnström et al., 2004). An RCT assessed the impact of a home safety assessment and modifications to improve safety in the home environment on falls and fall-related injuries in adults ages 75 and older with vision impairment and found that among participants who received the home safety assessment as compared to those who did not receive the assessment, the incidence rate ratio of falls and injurious falls was 0.59 and 0.81, respectively (Campbell et al., 2005). Another RCT assessed the impact of an intervention to remove home hazards on falls in older adults with and without vision impairments (Day et al., 2002). In combination with an intervention to improve vision and/or an intervention to improve strength and balance through an exercise program, reduction of home hazards significantly reduced the rate ratio of falls in comparison to participants who received no interventions (Day et al., 2002). Further research is needed on the effectiveness and cost-effectiveness of single- and multi-component environmental modifications in home, work, and academic environments for promoting safety and functioning in individuals with vision impairment.
Adaptive Strategies: Knowledge, Skills, and Tools
In the context of vision rehabilitation, adaptive strategies may include learned behaviors and skills that allow individuals with vision impairment to independently, effectively, and safely perform daily activities as well as the broader range of activities required to fully participate in social, political, economic, educational, and recreational activities. At their most basic, these strategies help individuals with vision impairment bathe, dress, and eat without assistance. For example, patients may be advised to place a finger just inside the lip of a cup while pouring liquid into it in order to avoid
spills (VisionAware, 2016b). Patients with difficulty distinguishing colors may be taught to label their clothing (e.g., “navy” or “black”) to help identify items that match (VisionAware, 2016a). More complex or particular strategies aid individuals with vision impairment in their ability to shop for food, cook meals, clean their homes, manage finances, communicate with friends and family, and move freely in the community.
Some adaptive strategies involve maximizing remaining visual function. For example, eccentric viewing is a technique in which patients learn to use portions of the retina that have not been damaged by disease. Training in eccentric viewing is included as a strategy for managing central visual field defects in the AOA guidelines for vision rehabilitation and is listed as an occupational therapy and reading rehabilitation intervention in the American Academy of Ophthalmology guidelines for vision rehabilitation (AAO, 2013; AOA, 2007). A systematic review of the effectiveness of eccentric viewing training for improving performance of daily visual activities in individuals with vision impairment consequent to AMD included five studies that had reported statistically significant improvements in reading speed, maximal reading speed, duration of reading, reading comprehension, and performance in daily activities (Hong et al., 2014). The review authors concluded that eccentric viewing training has a moderate-sized effect on the ability of the studied populations to perform daily visual activities (Hong et al., 2014). Another systematic review found that moderate quality evidence supported eccentric viewing training incorporating or in conjunction with steady eye training as a method to improve near visual acuity, reading speed, and the ability to perform ADLs in patients with central vision loss (Gaffney et al., 2014). Further research is needed to ascertain the effect of eccentric viewing training on distance visual acuity and QOL, and the comparative effectiveness and cost-effectiveness of different training programs.
Other adaptive strategies use hearing and touch to promote functioning. For example, a growing body of research has documented the ability of blind individuals to use echolocation for navigation, with studies demonstrating the ability of echolocation experts to assess with considerable accuracy the position, size, distance, and shape of objects in space, as well as the distance and location of sounds (Kolarik et al., 2014; Kondo et al., 2012; Milne et al., 2014; Schenkman and Nilsson, 2010; Schornich et al., 2012, 2013; Teng et al., 2012; Vercillo et al., 2015; Wallmeier and Wiegrebe, 2014a,b). Teng and Whitney (2011) demonstrated that, even without feedback or formal training, some normally sighted individuals can rapidly learn to determine the size and position of objects in space by means of echolocation, suggesting that echolocation training could be pursued as a strategy to promote mobility in individuals with vision impairment.
The use of low vision aids and devices, Braille typewriters and dynamic readers, white mobility canes, and environmental modifications are also examples of adaptive strategies even though these strategies use tools in addition to learned behaviors and skills to enhance functioning. Emerging technologies, such as self-driving cars, text-to-speech and speech-to-text software, electronic personal navigation systems, and a broad range of smartphone applications offering services for individuals with vision impairment hold potential for expanding opportunities for increased independence, functioning, and participation among populations with vision impairment.
Models of Vision Rehabilitation
Although the majority of existing vision rehabilitation programs share an overarching care process (i.e., examination, diagnosis and development of treatment plan, vision rehabilitation services and interventions), broad variation exists in the organization and delivery of vision rehabilitation services. For example, highly integrated vision rehabilitation programs, such the U.S. Department of Veterans Affairs’ (VA’s) Vision Impairment Services in Outpatient Rehabilitation (VISOR) Program, provide individualized care in a residential setting staffed by multidisciplinary teams that offer a range of services (VA, 2016). Other programs offer self-management education in short, weekly classes provided in a community setting.
These models of care differ in the setting, cost, duration, scope, emphasis, and organization of the services they provide. These differences can affect program effectiveness in unpredictable ways. For example, one study comparing conventional vision rehabilitation services (i.e., examination and provision of low vision aids and support services by a multidisciplinary team) to enhanced services (usual care plus an 8-week self-management program to develop problem-solving and coping skills) found no significant differences between the programs in terms of impacts on the vision-specific QOL, emotional well-being, adaption to vision loss, or self-efficacy of patients (Rees et al., 2015). Other studies confirm that more comprehensive care does not necessarily lead to better outcomes. Several systematic reviews have identified evidence indicating that multidisciplinary or multicomponent interventions are effective at promoting independent functioning in the home and community, and for maintaining or enhancing social participation (Justiss, 2013; Liu et al., 2013; Smallfield et al., 2013). Individual and group-based problem-solving—in which participants identify problems, establish goals, develop and implement solutions, and assess outcomes—was strongly supported by the evidence as an
effective method for improving social participation among older adults with vision impairment (Berger et al., 2013). The following section will describe both common and unique models of vision rehabilitation, and explore how variation in the scope, organization, and delivery of services relates to patient health outcomes.
VA Blind Rehabilitation Services
The VA Blind Rehabilitation Services provide a continuum of vision rehabilitation services to improve the functional status and QOL of veterans with any degree of vision impairment (VA, 2015). Intermediate vision impairment clinics “focus on effective use of remaining vision through the development and use of visual motor and visual perceptual skills” and include training in the use of low vision aids (VA, 2015). Advanced vision impairment clinics offer orientation and mobility training, including instruction on the use of white mobility canes, sensory training, and mental mapping. The VISOR Program provides short-term training to improve communication skills, access to technology, and the ability to perform daily activities. Inpatient Blind Rehabilitation Centers (BRCs) provide “comprehensive adjustment to blindness training,” including individual counseling and group therapy to support emotional and behavioral health. Complementing BRCs are Vision Impairment Centers to Optimize Remaining Sight (VICTORS) programs that offer interdisciplinary rehabilitation services for veterans with severe vision impairment. Together, these programs offer a range of increasingly comprehensive vision rehabilitation services (VA, 2015).
Available research suggests that several of these programs can improve the functional status and QOL of patients with vision impairment. The VA Low Vision Intervention Trial (LOVIT) assessed the effect of a 2-month vision rehabilitation program that included an initial examination, five weekly sessions to teach adaptive strategies and provide training in the use of low vision aids, as well as a home visit to assess the home environment (Stelmack et al., 2007b, 2008). Among older adults with moderate to severe vision impairment consequent to macular disease, compared to those who did not receive vision rehabilitation, those who did experienced significant improvements in mobility, reading ability, visual motor skill, visual information processing, and overall visual ability at 4 months post-baseline (Stelmack et al., 2008).7 In a follow-up study, standard low vision therapy (i.e., examination, education on eye disease, training in
___________________
7 Moderate to severe vision impairment was defined as visual acuity worse than 20/100 and better than 20/500 in the better-seeing eye.
eccentric viewing and the use of low vision aids, psychological counseling, and social work services) was offered to the original control group, and the original experimental group received no additional care (Stelmack et al., 2012a). Twelve months after baseline, the original treatment group experienced significant losses in mean scores for reading ability and visual information processing compared to performance on these outcome measures at 4 months, while the original control group experienced significant improvements in mean scores for all outcome measures compared to performance at 4 months. For both the control and treatment groups, mean scores for overall visual ability improved significantly after 12 months compared to performance at baseline, although the improvement was significantly greater for the treatment group than for the control group (Stelmack et al., 2012a).
An economic evaluation found that the average cost per patient was significantly lower for the LOVIT program ($404.70) than for comparable care in a BRC ($43,681.70). However, mobility, visual motor skills, and overall visual ability—but not reading ability or visual information processing—improved more in the BRC than in the LOVIT program (Stroupe et al., 2008). A second LOVIT trial to compare the effectiveness and cost-effectiveness of basic care (i.e., examination, provision of low vision aids) and vision rehabilitation (i.e., examination, provision of low vision aids, two to three low vision therapy sessions) is planned (Stelmack et al., 2012b). Another study investigated the effect of a BRC inpatient blind rehabilitation program on vision-specific QOL among 206 legally blind veterans (Kuyk et al., 2008). Program interventions included examination, assessment of psychological and social needs, training in the use of low vision aids, and instruction in mobility, performance of ADLs, communication, and adaptive strategies. Compared to performance at baseline, mean composite score on the National Eye Institute Visual Function Questionnaire (NEI VFQ) was significantly higher at 2 and 6 months post-rehabilitation, although the 6-month post-rehabilitation composite score was significantly lower than the 2-month post-rehabilitation composite score. Rehabilitation also significantly improved performance on measures of social function, mental health, and vision function (Kuyk et al., 2008). A prospective observational study of patients who participated in a BRC inpatient rehabilitation program found that visual ability decreased between 3 and 12 months post-rehabilitation, but remained significantly improved over performance at baseline (Stelmack et al., 2007a). Further research is needed to assess the effectiveness and cost-effectiveness of other components of the VA blind rehabilitation services, including intermediate and advanced vision impairment clinics.
Self-Management Education Programs
Self-management education programs help patients manage chronic conditions, such as uncorrectable or irreversible vision impairment. Compared to traditional patient education programs that place the health care provider in the role of educator and focus on dissemination of disease-specific technical skills and information in order to promote patient compliance with a treatment plan, self-management education programs emphasize self-efficacy by placing the patient in charge of identifying issues related to their chronic condition and teaching problem-solving skills to address those problems (Bodenheimer et al., 2002). Available evidence suggests that self-management education programs can improve functioning and emotional distress among individuals with vision impairment.
A systematic review found that self-management education programs can “improve emotional distress, functional ability[,] and self-efficacy in elderly people with AMD,” but stated that additional studies with robust methodologies were required to substantiate these findings (Lee et al., 2008, p. 174). An RCT assessed the effects of a self-management education program on 231 adults ages 60 and older with vision impairment consequent to AMD. The program involved six weekly 2-hour sessions of didactic presentations and group problem-solving, and cognitive and behavioral skills training presented to groups of 8 to 10 patients. Patients in the self-management education program experienced significant improvements in emotional distress and functioning; by comparison, changes in emotional distress and functioning were not significant for patients in the control group receiving no care (Brody et al., 2002). At 6-month follow-up, patients in the self-management education program reported significantly less emotional distress, significantly better functioning, and significantly increased self-efficacy as compared to patients in the control groups receiving either no care or 12 hours of recorded health lectures (Brody et al., 2005). In addition, among patients with depression, those randomized to the self-management education group experienced significantly greater reductions in depressive symptoms than did patients randomized to either of the control groups (Brody et al., 2006).
Studies on populations outside the United States suggest that self-management education programs can improve perceived security in the performance of several daily activities among patients with vision impairment consequent to AMD, and can increase awareness of low vision aids and practical strategies to optimize use of remaining vision among caregivers of patients with vision impairment (Dahlin Ivanoff et al., 2002; Larizza et al., 2011). Less promisingly, an RCT examining the impact of a self-management education program in addition to usual vision rehabilitation services on vision-specific QOL, emotional well-being, self-efficacy, and adaptation to
vision loss among Australian adults ages 55 and older with vision impairment found no significant difference between the outcomes of patients in the usual care group and those in the usual care plus self-management education group (Rees et al., 2015). The authors called for further research to identify the patients most likely to benefit from self-management education programs and from specific components of such programs, in order to better target these interventions.
Models of Vision Rehabilitation in Australia and the United Kingdom
Lamoureux et al. (2007) investigated the effect of a multidisciplinary vision rehabilitation service on Australian adults ages 18 and older with visual acuity <6/12 or >6/12 with restricted visual fields (p. 1477).8 Rehabilitation included an assessment by a member of a multidisciplinary team to determine the patient’s needs and goals. Subsequently, an optometrist performed an examination and offered low vision aids or rehabilitation services (e.g., occupational therapy, orientation and mobility, peer support, community services) as appropriate. A care plan, including referral pathways, is developed and considered complete when the patient “feels satisfied that the desired outcomes have been met and cannot identify any further service needs” (Lamoureux et al., 2007, p. 1477). Compared to participant scores at baseline, emotional well-being and the ability to read and access information were significantly improved 3 to 6 months after rehabilitation. Nonsignificant improvements in orientation and mobility were also observed (Lamoureux et al., 2007). Another study investigated an Australian multidisciplinary vision rehabilitation service involving an initial assessment to determine patient needs, develop a unique treatment plan, and prescribe low vision aid, followed by a review appointment within 30 days to assess the need for interventions related to orientation and mobility, independent living, recreation, adaptive technologies and devices, and counseling, among others (Wang et al., 2012). The reported effectiveness of these services depended on the assessment tool used. According to the VA Low-Vision Visual Functioning Questionnaire (VA LV VFQ-48), mean scores on measures of reading ability, visual information, visual motor, and overall visual ability had improved significantly at 30 days—but not at 3 months—compared to performance at baseline. By comparison, according to the Impact of Vision Impairment questionnaire, mean scores on measures of mobility and independence at 30 days and 3 months were significantly improved over performance at baseline, and overall mean score was significantly improved at 3 months—but not at 30 days—compared to performance at baseline. In addition, mean scores
___________________
8 A visual acuity of 6/12 (meters) converts to a visual acuity of 20/40 (feet).
on the VA LV VFQ-48 indicate that the benefits of vision rehabilitation were more pronounced for patients with best-corrected visual acuity of <20/60 and ≥20/200 compared to patients who were legally blind (i.e., best-corrected visual acuity <20/200 or constriction of visual field to within 10 degrees of fixation) for all mean scores, for patients ages 86 and older compared to those ages 85 and younger, and for patients without glaucoma compared to those with glaucoma (Wang et al., 2012). Further research is needed to determine how to adapt vision rehabilitation services to better meet the needs of all patient populations, and to develop standardized methods for assessing outcomes from vision rehabilitation.
In the United Kingdom, interdisciplinary low vision service (ILVS) programs staffed by multidisciplinary teams working out of general hospital clinics provide examination, diagnosis, and low vision aid provision and training. Home visits can involve training in mobility, independent living skills, and education on available social services. Six months after the initiation of vision rehabilitation, Hinds et al. (2003) found a significant decrease in patient anxiety regarding safety at home, coping ability, and further vision loss (p. 1393). Among 71 study participants ages 34 and older with vision impairment, 53 (75 percent) claimed to use the aid to read books, newspapers, or magazines, and 36 (51 percent) stated that they use the aids several times per day (Hinds et al., 2003).
The Welsh Low Vision Service expanded access to vision rehabilitation services in Wales by training community-based optometrists, ophthalmic medical practitioners, and dispensing opticians to provide vision rehabilitation services in a primary care setting. Based on the National Health Service Hospital Eye Service model, the Welsh Low Vision Service includes assessment of patient needs and goals, provision of low vision aids, patient education on vision rehabilitation services and interventions, referral to other services, and follow-up care (Ryan et al., 2010). Comparing access-related parameters 1 year before and 1 year after implementation of the Welsh Low Vision Service, the number of appointments for vision rehabilitation services increased by 51.7 percent, the proportion of patients waiting less than 2 months for an initial vision rehabilitation assessment increased from 11 percent to 60 percent, and 81 percent of patients traveled a shorter distance to reach their service provider than they would have if the Welsh Low Vision Service had not been implemented. Visual disability scores 3 months after rehabilitation were significantly improved over scores before rehabilitation, and nearly all patients (97.4 percent) reported that the service was useful (Ryan et al., 2010). A follow-up study found that visual disability scores 18 months after rehabilitation remained significantly higher than at baseline, and that 79 percent of study participants continued to use low vision aids (Ryan et al., 2013).
It is important to note that health care systems in Australia, the United Kingdom, and elsewhere serve different populations and are organized and funded differently than in the United States. As a result, vision rehabilitation models employed in these countries may perform differently if implemented in the United States. While these models can provide important lessons for U.S. policy makers and health care organizations, their direct applicability to vision rehabilitation services in the United States may be limited.
BARRIERS AND OPPORTUNITIES IN VISION REHABILITATION
There are several potential barriers to developing a universally high-quality and accessible vision rehabilitation system. A lack of awareness of the purpose and effectiveness of vision rehabilitation among medical providers, public health workers, and patients may unnecessarily limit the utilization of vision rehabilitation services by patients with vision impairment. Issues related to workforce education and capacity may pose barriers to accessing vision rehabilitation services. Racial, ethnic, and socioeconomic disparities in clinical eye and vision care exist and may negatively affect access and utilization of vision rehabilitation services. This section will briefly discuss barriers to achieving high-quality and accessible vision rehabilitation services in the United States and will identify strategies for eliminating these barriers.
Lack of Awareness and Knowledge of Vision Rehabilitation Services
Patients, medical providers, and public health actors often lack an awareness of the goals, content, effectiveness, and availability of vision rehabilitation. Patients may simply be unaware of vision rehabilitation services, or they may misunderstand their purpose. The stigma surrounding blindness or individuals’ failure to acknowledge deficits in visual functioning may also contribute to less-than-optimal use of vision rehabilitation by populations with vision impairment. A lack of knowledge and inadequate communication between clinical care providers and public health officials prevents the larger population health workforce from addressing the issue of limited patient awareness. This section will discuss the gaps in knowledge of vision rehabilitation among patients and providers, and will suggest corrective strategies.
Knowledge Gaps Among Patients
The limited available evidence suggests that the knowledge and awareness of vision rehabilitation among the U.S. population is poor. The 2005 Survey of Public Knowledge, Attitudes, and Practices Related to Eye Health
and Disease found that among 3,180 U.S. adults ages 18 and older, 16 percent reported having heard the term “low vision” (NEI/LCIF, 2008, p. v).9 Of the 2 percent of surveyed adults who reported that an eye care provider had diagnosed them with vision impairment, only 31 percent had been recommended to a low vision specialist (NEI/LCIF, 2008).10 One study assessed the knowledge of, experience with, and interest in vision rehabilitation services and devices among U.S. adults ages 65 and older who were diagnosed with AMD (Casten et al., 2005). More participants were interested in, than were aware of, vision rehabilitation (83 percent versus 24 percent), and for the majority of vision rehabilitation services or devices, the proportion of participants who were aware of a particular service or device was lower than the proportion of participants who were interested in that service or device. For example, participants were more likely to be interested in than aware of support groups (50 percent versus 31 percent), home modifications (54 percent versus 15 percent), and mobility training (49 percent versus 11 percent) (Casten et al., 2005).
Studies on the awareness of vision rehabilitation among populations in Canada and Australia suggest that the lack of awareness can vary among groups and that numerous gaps in patient knowledge may exist. Two studies of adult Canadians with vision impairment found that individuals of African descent were significantly less likely to be aware of vision rehabilitation services than individuals of other surveyed ancestries (Mwilambwe et al., 2009; Overbury and Wittich, 2011).11 Another study reported that among 749 Canadian adults with vision impairment, 32.3 percent lacked information about vision rehabilitation services (Fraser et al., 2015). Compared to study participants who attended vision rehabilitation services, or who did not attend but were aware of such services, a greater proportion of those who lacked information about vision rehabilitation services reported great difficulty or inability to read traffic, street, or store signs, as well as being unable to perform fine handiwork (e.g., sewing, knitting). Males were also less likely than females to be aware of vision rehabilitation services (Fraser et al., 2015). Other studies have shown that patients with vision impairment may not attend vision rehabilitation services because they may not believe that vision rehabilitation will be helpful or may not identify themselves as having an impairment, or may believe that vision rehabilitation is only for
___________________
9 The survey defined low vision as a visual impairment that is not corrected by standard eyeglasses, contact lenses, medication, or surgery and that interferes with the ability to perform everyday activities.
10 “Vision impairment” replaces the term “low vision” used in the survey.
11Overbury and Wittich (2011) define vision impairment as a best-corrected visual acuity in the better-seeing eye of <20/60 [6/18] or a visual field of <60 degrees in either the horizontal or vertical meridian. Mwilambwe et al. (2009) define vision impairment as best-corrected visual acuity worse than 20/70 in the better-seeing eye.
blind patients (Matti et al., 2011; Pollard et al., 2003). Although these findings do not necessarily apply to populations in the United States, the implications they have for developing targeted health education activities strongly suggest the need for similar research in the United States.
Improving public awareness of vision rehabilitation begins with ensuring that health care providers are knowledgeable about vision rehabilitation and that they consistently share this knowledge with visually impaired patients. Health education campaigns implemented by stakeholders within vision rehabilitation and the clinical vision care system represent another strategy for promoting knowledge of vision rehabilitation among patients. To the extent that vision rehabilitation can mitigate the high societal and health care costs associated with vision impairment and blindness, all groups that directly or indirectly bear these costs (e.g., governments, nonprofit organizations, insurance providers, and health care organizations) also have a stake in promoting awareness of vision rehabilitation.
Knowledge Gaps Among Health Care Providers
Providers must be aware of vision rehabilitation services in order to refer patients to, and educate them about, these services. Studies from Australia and Canada support the claim that patient counseling on vision rehabilitation is essential for promoting patient awareness of these services and that failure to provide such counseling is a prominent barrier to access and utilization of vision rehabilitation services. For example, one study found that among Australian patients with vision impairment who followed up on a referral to vision rehabilitation services, 85.4 percent cited receiving a referral to and/or information about vision rehabilitation services as a facilitating factor in the decision to attend vision rehabilitation (O’Connor et al., 2008). In another study, peer workers and patients at vision rehabilitation centers in Australia reported that patients arriving at vision rehabilitation centers may have no understanding of why they are there or the nature of the service, that referral for vision rehabilitation often came late in the care process, and that patients with limited vision impairments are sometimes less likely than patients with total blindness to receive information on vision rehabilitation services (Pollard et al., 2003). In Australia, health professionals working in eye and vision care have identified a lack of knowledge of vision rehabilitation services among health professionals and poor communication between patients and providers as barriers to use of vision rehabilitation services (O’Connor et al., 2008). In the United States, patients with vision impairment have identified the quality of communication between patients and providers as the most important factor affecting the quality of care and have reported difficulties accessing
health-related information (O’Day et al., 2004). As mentioned above, caution must be exercised when assessing studies performed in Canada and Australia, because these findings may not necessarily apply to populations in the United States.
Potential strategies exist for promoting the consistency and quality of patient–provider communications concerning vision rehabilitation, as well as to ensure that all health care professionals are knowledgeable about these services. For example, modules on vision rehabilitation services could be incorporated into the formal education and post-professional training of optometrists, ophthalmologists, primary care physicians, and other health care professionals who interact with visually impaired patient populations. In one study, an online continuing education program significantly improved knowledge of assessment and the treatment of vision impairment among generalist occupational therapists. After completing the program, an increased number of occupational therapists reported that they frequently screened for vision impairment, frequently provided environmental modifications to enhance visual functioning, and felt comfortable providing interventions for patients with vision impairment (Nipp et al., 2014). The need for these kinds of professional training programs has been demonstrated. For example, in a survey of 100 occupational therapists that did not specialize in vision rehabilitation, researchers found that only 52 percent believed that they had received adequate training in occupational therapy school to address the unique rehabilitation needs of patients with vision impairment (Winner et al., 2014).
Medical professional groups could also collaboratively pursue the development of guidelines describing best practices for interprofessional referral to assist care providers in the identification and appropriate referral of patients with vision impairment. Finally, health care organizations, professional groups, policy makers, and other stakeholders could promote the integration of vision rehabilitation service providers into clinical settings to better develop interprofessional trust and lines of communication and to encourage the sharing of knowledge and best practices.
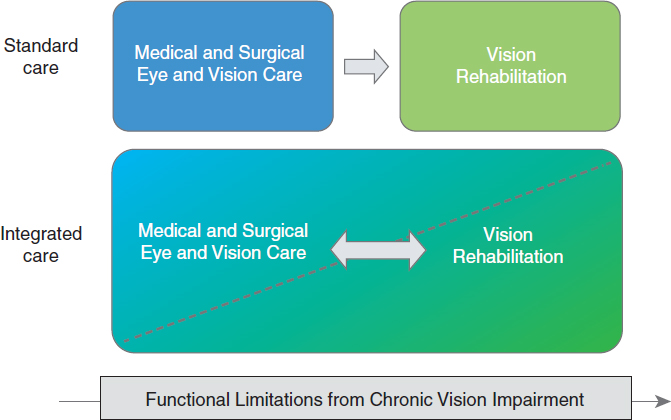
These actions could be pursued as part of a larger effort to promote collaborative care and the adoption of integrated models of care in which medical, surgical, and rehabilitation interventions and services together improve the quality and accessibility of eye and vision care. Figure 8-2 depicts how the relationship between vision rehabilitation services and the surgical and medical services described in Chapter 7 could evolve toward an integrated model in which vision rehabilitation gradually assumes priority over medical and surgical care as functional limitations associated with chronic vision impairment increase.
Knowledge Gaps Among Population Health Actors
In addition to patients and providers, members of the population health workforce also need to be knowledgeable about vision rehabilitation services. This workforce includes public health officials; epidemiologists; health policy analysts; nonprofit groups working in health care, vision care, or patient advocacy; insurance providers; administrators of health care organizations; research organizations; and health economists. The potential benefit derived from imparting knowledge about vision rehabilitation to the population health workforce is that members of each class of actor could use this information in accordance with their own interests, competencies, and professional obligations. Insurance providers could provide coverage for vision rehabilitation care and treatment under major medical insurance plans; epidemiologists could assess the impact of discrete components of vision rehabilitation on patient health outcomes; nonprofit organizations could improve care access and continuity by strengthening relationships between providers of vision rehabilitation and other community health services; health economists could assess a range of rehabilitation models to determine which provide the most cost-effective care; and health policy analysts could investigate which social policies support vision rehabilitation.
If the value of educating the population health workforce about vision rehabilitation is clear, the most appropriate means of doing so is not. Developing and running individual health education campaigns targeted to each of these groups would incur considerable costs. On the other hand, general
awareness campaigns would likely have diminished penetration in at least some target groups. One strategy would be to focus health education efforts on public health departments and to rely on their unique role as conveners and coordinators to ensure that, through their dissemination activities, the population health workforce as a whole is made aware of the effectiveness of vision rehabilitation and its role in the clinical vision services system.
Workforce Education and Capacity
In considering ways to improve the value and increase the uptake of vision rehabilitation services for an increasing number of Americans with chronic vision impairment, the potential barriers related to the development of a competent multidisciplinary workforce should be addressed. The relatively limited number of optometric or ophthalmic residency or fellowship programs focused on vision rehabilitation may be one such barrier. As of academic year 2014–2015, there were no ophthalmology subspecialty residency programs in vision rehabilitation accredited by the Accreditation Council for Graduate Medical Education (ACGME), and the American Board of Ophthalmology does not currently offer specialty or subspecialty certification in vision rehabilitation (ABMS, 2016; ACGME, 2015). However, ACGME does require that physician faculty members of ophthalmology residency programs have expertise in a broad range of ophthalmic disciplines, including vision rehabilitation, and that residents must demonstrate competence in their knowledge of vision rehabilitation (ACGME, 2012). According to the American Council on Optometric Education, there were 34 accredited optometric residency programs with a focus or partial focus on vision rehabilitation in 2016 (ACOE, 2016), and the Association of Schools and Colleges of Optometry lists 20 residency programs affiliated with 12 colleges of optometry that offered focused training in vision rehabilitation in academic year 2015–2016 (ASCO, 2016).12
Occupational therapists have a number of opportunities for developing knowledge of or competency in vision rehabilitation. Doctoral programs are available in occupational therapy with specialty tracks or areas of concentration in vision rehabilitation and a graduate certificate in vision rehabilitation
___________________
12 The 14 programs included those with a focus on low vision/ocular disease, primary care and vision rehabilitation, ocular disease and low vision rehabilitation, geriatrics and low vision, low vision rehabilitation, vision therapy and rehabilitation, vision therapy/low vision rehabilitation, low vision/primary care, vision therapy/low vision rehabilitation, geriatric and low vision rehabilitative optometry, ocular disease/low vision rehabilitation, and primary care/low vision rehabilitation. Three additional programs offer training in vision rehabilitation (neuro-optometric rehabilitation), brain injury vision rehabilitation, and brain injury vision rehabilitation and ocular disease. An additional program in primary care optometry included training in low vision (ASCO, 2016).
(AOTA, 2016c,d). American Occupational Therapy Association (AOTA) offers a specialty certification in low vision, as well as a continuing education course on vision rehabilitation, and several AOTA-approved continuing education providers offer courses in vision rehabilitation (AOTA, 2015, 2016b). There are currently no AOTA-approved occupational therapy residency programs focused on vision rehabilitation (AOTA, 2016a). Research is needed to determine whether expanding the vision rehabilitation workforce through the development of additional training and education opportunities for optometrists, ophthalmologists, and occupational therapists can improve access to vision rehabilitation services.
Another potential barrier to access may be the distribution of the burden of care across providers and the limited availability of specific rehabilitative services. A census to determine the characteristics of vision rehabilitation patients, providers, and services in the United States found that although government agencies and independent services for visually impaired persons accounted for just 7.5 percent and 11.2 percent of all service entities, respectively, they were responsible for the provision of 28.4 percent and 22.7 percent of all vision rehabilitation services (Owsley et al., 2009). By comparison, private optometry and ophthalmology practices together comprised 60.1 percent of service entities, but provided only 22.6 percent of vision rehabilitation services. General hospitals, rehabilitation hospitals, and outpatient rehabilitation centers were more likely than private optometry and ophthalmology practices to offer several types of vision rehabilitation services (e.g., psychological services, support groups, social work services, driving rehabilitation, home visits, orientation and mobility training, eccentric viewing training, advanced/intensive training in device use), but they comprised only 2.6 percent, 1.5 percent, and 2.6 percent of service entities, respectively (Owsley et al., 2009). Research is needed to determine how the scope of practice of service entities and the distribution of the burden of care across service entities affect the quality of and access to care.
A study to assess the baseline traits of 764 patients seeking vision rehabilitation at 28 outpatient clinical centers found that 42 percent, 23 percent, and 22 percent of patients described their current emotional state as “frustrated,” “anxious,” and “depressed,” respectively, while 85 percent reported that they limited their driving in some way (Goldstein et al., 2012, p. 1031). Another study found that 44.9 percent of vision rehabilitation patients had problems with emotional or psychological adjustments related to vision impairment and that 67.7 percent had problems or difficulties with driving (Owsley et al., 2009). Yet only a minority of service entities had a psychologist on staff (4.9 percent) or offered psychological services (21.1 percent), and only 11.4 percent offered driving rehabilitation (Owsley et al., 2009). Research is needed to ascertain whether this apparent misalignment between the needs of patients
seeking vision rehabilitation and the types of interventions offered by vision rehabilitation service entities affects patient outcomes.
Finally, Medicare policies that do not recognize some members of the vision rehabilitation team, such as orientation and mobility specialists and low vision therapists, may create a barrier to care access, by failing to incentivize the growth of these professional groups or their involvement in vision rehabilitation. To address these barriers to access, policy makers could support intelligently designed vision rehabilitation demonstration projects that evaluate the efficacy and cost-effectiveness of models of care and payment and that promote and appropriately incentivize team-based vision rehabilitation services. Professional education programs could be expanded to include metrics that ensure an adequate, well-trained workforce and that address misperceptions related to vision rehabilitation. Policy makers could consider loan forgiveness and other incentives for those interested in pursuing careers in vision rehabilitation.
Racial, Ethnic, Socioeconomic, and Geographic Barriers to Accessing Vision Rehabilitation
Members of minority groups and populations living in lower socioeconomic groups access and utilize clinical eye and vision care less frequently than whites and populations in higher socioeconomic groups. Studies have found an association between lower income or less education and reduced rates of clinical eye and vision care (Zhang et al., 2012, 2013b). Other studies and reviews have reported disparities among racial and ethnic groups in access to and utilization of clinical eye and vision care (Chou et al., 2012, 2014; Elam and Lee, 2013). Because eye exams are the entry point into the vision rehabilitation process of care, it is reasonable to infer that groups that are less likely to use those services are also less likely to use vision rehabilitation services. However, there is limited direct evidence to support this inference.
There are few peer-reviewed studies on racial, ethnic, or socioeconomic barriers to vision rehabilitation access and utilization in the United States. However, studies on vision rehabilitation in Canada and Australia show that these barriers do exist in these other countries’ health care systems. Among 702 Canadian adults ages 26 to 100 with vision impairment, those individuals with more education were more likely to be aware of and use vision rehabilitation services (Overbury and Wittich, 2011).13 Lack of awareness of vision rehabilitation—which can pose a barrier to utilization—varies among racial and socioeconomic groups in Canada and is discussed above (Mwilambwe et al., 2009; Overbury and Wittich, 2011). Lack of
___________________
13 Vision impairment was defined as best-corrected visual acuity in the better-seeing eye of <20/60 [6/18], or a visual field of <60 degrees in either the horizontal or vertical meridian.
transportation, which is often associated with lower socioeconomic status, is also cited as a barrier to accessing vision rehabilitation. For example, one study found that among 98 Australian patients with vision impairment who were referred to vision rehabilitation services, only 49 percent attended the service. Of those who declined or did not comply with the referral, respectively 33.3 percent and 50.0 percent cited transportation difficulties or the lack of an accompanying person as a reason for non-attendance (O’Connor et al., 2008).14 It is important to note that differences among health care systems and patient populations mean that access barriers to vision rehabilitation in Australia and Canada may differ from those in the United States. However, the evidence of the existence of racial, ethnic, and socioeconomic barriers to accessing vision rehabilitation in other countries can be used to spur and guide efforts to determine whether such barriers also exist in the United States, and to eliminate them if they do.
In the United States, geographic variation in vision rehabilitation entities may affect the accessibility of vision rehabilitation services. One study found that the density of vision rehabilitation service entities varied among states, with higher density in some Northeastern states, and in Montana, Nebraska, North Dakota, South Dakota, and Wyoming, while many states across the southern part of the country (e.g., Alabama, Arizona, California, Georgia, Mississippi, Oklahoma, Texas) had lower densities of service entities (Owsley et al., 2009). Another study found that Alabama, Florida, Mississippi, and other states in the Southeast were among the states with the highest per capita prevalence of blindness in 2015, while several states in the Southeast, Midwest, and Northeast are among the states projected to have the highest per capita prevalence of blindness in 2050 (Varma et al., 2016). Research is needed to determine whether and how regional variations in the prevalence of vision impairment and blindness, and in the per capita density of vision rehabilitation service entities, affect, or are projected to affect, access to vision rehabilitation and demands on services.
NOVEL TECHNOLOGIES, NEW THERAPIES, AND PROMISING RESEARCH
Between technological advances that increase the effectiveness of low vision aids, an urgent push to integrate counseling and other QOL interventions into models of vision rehabilitation, and research efforts to improve the quality of care, vision rehabilitation is undergoing dramatic and beneficial changes. This section will explore how technological advance, innovative clinical care and care models, and promising research are currently
___________________
14 All patients with worse than 6/12 visual acuity, or who reported functional or emotional difficulties because of their vision, were eligible to attend vision rehabilitation services.
improving health outcomes among people with vision impairment. Challenges to optimizing the impact of these powerful new tools, methods, and scientific findings will be discussed, as will strategies for overcoming those challenges.
Novel Technologies and Advances
Products and technologies that enhance the functioning and mobility of populations with vision impairment are a key component of effective vision rehabilitation services. These include products that are designed to meet the specific needs of individuals with vision impairment (e.g., dynamic Braille readers, large-print books), as well as products for use by the general public (e.g., smartphones, text-to-speech software) that also meet—or can be adapted to meet—a need common to individuals with vision impairment. This section explores the current and emerging technologies that promote QOL among populations with vision impairment and discusses the role of intentional design in supporting the development of such technologies.
Current and Emerging Technologies
As discussed above, compromised reading ability is a common complaint of patients referred to vision rehabilitation services (Brown et al., 2014; Renieri et al., 2013). Technologies to improve reading ability or otherwise provide access to written materials are numerous. Refreshable Braille displays can convert text on a digital display into Braille, though currently the available models can be prohibitively expensive (Russomanno et al., 2015). Electronic book readers with refreshable Braille displays and other devices with dynamic tactile displays are under development (Moore, 2015; Motto Ros et al., 2014). Smartphones and tablet computers have enhanced the convenience and accessibility of audiobooks, can magnify text and other images, and are increasingly equipped with speech-activated functions and text-to-speech software. A survey of 132 individuals with vision impairment found that among the 81 percent of respondents that used smartphones, 34 percent used the device to listen to audiobooks, 51 percent used the integrated camera to see an image more clearly, and 59 percent found the speech functions useful (Crossland et al., 2014). Other products and tools are designed to aid individuals with vision impairment in the performance of common activities such as cooking, exercising, and cleaning (AFB, 2014; NFB, 2016). There is a need for sustained research to continue the development of products and technologies that are accessible and that expand options for social participation, employment, and education among populations with vision impairment.
Several emerging technologies hold promise for enhancing the mobility of populations with vision impairment. For example, technology companies and car manufacturers are developing and testing self-driving cars that may create new transportation options for individuals who are otherwise unable to drive due to vision impairment (Stenquist, 2014). Difficulty with driving is a common complaint of individuals referred for vision rehabilitation (Brown et al., 2014), and individuals with some types of vision impairment (e.g., reduced contrast sensitivity) may restrict their driving habits (Fraser et al., 2013; Sandlin et al., 2014). Moreover, Owsley and McGwin (2010) note that available evidence points to an association between impaired driving performance and reduced visual acuity, visual field, or contrast sensitivity (Owsley and McGwin, 2010). These findings suggest that self-driving cars could address a documented need. Navigation systems that use smartphones and other mobile devices to provide users with real-time information about their surroundings hold potential for improving the safety and mobility of individuals with vision impairment and are being developed for use in public transportation systems, indoor settings, and other areas (Basulto, 2015; Legge et al., 2013; Pym, 2015). Mobility devices that use lasers, infrared sensors, and/or echolocation to convey information about the environment to the user via vibrational and auditory cues have also been developed and have outperformed white mobility canes in tests of hazard avoidance (Bhatlawande et al., 2014; Maidenbaum et al., 2014). Ongoing research is needed to support the development of new products and technologies that safely and effectively enhance the functioning and mobility of populations with vision impairment.
Intentional Technological Advancement and Research Needs and Opportunities
It is important to note that both existing and emerging technologies are in themselves value neutral regarding the well-being of people with vision loss. Technology can be a tool to promote or a barrier to prevent health and independent functioning. Once a luxury item, now a necessary tool, the endless adaptability of smartphones allows developers to constantly expand their uses and capabilities, and their ubiquity encourages employers, social groups, and merchants to assume every employee, acquaintance, and consumer has access to one. If accessibility remains a concern for developers of smartphones and their applications, individuals with vision impairment will have a highly portable tool with flexibility to support independence. However, if developers do not ensure their products are accessible to populations with vision impairment, these groups will have limited ability to utilize an increasingly large and crucial set of services and opportunities.
It is therefore of signal importance that entrepreneurs, developers, and innovators develop their products with the vision impairment population in mind. Moreover, the embrace of accessibility as a design principle should not be construed as a regulation that threatens business prospects and hampers creative impulse, but as an opportunity to offer goods and services to a broader set of consumers and as an engineering problem that encourages the development of technological solutions valuable in their own right.
Ensuring the universal accessibility of current and emerging technologies is one method of leveraging technological advance for the benefit of those with vision impairment. A more direct method is to invest in research and technologies that cater exclusively to this population. As part of its ongoing strategic planning activities, the NEI has previously identified the need to “[d]evelop assistive devices, environmental modifications, and rehabilitation strategies to minimize the impact of visual impairment in everyday life and reduce disability and societal limitations in visually impaired persons” (NEI, 2004, p. 28). More recently and specifically, the NEI identified the need for research to inform development of visual prostheses; tools that improve access to the internet, print materials, and navigation aids; and technologies that effectively translate key aspects of visual information into tactile and auditory information. The NEI also noted that developers of assistive technologies need to account for the unique needs and limitations of visually impaired populations that are also “elderly, cognitively impaired, or technologically naive” (NEI, 2012, p. 59). Addressing these research gaps and opportunities could accelerate development of new assistive technologies that improve independence, functioning, and QOL among individuals with vision impairment.
New Therapies and Models of Care
New Therapies for Comorbid Depression
As discussed above and in Chapter 3, several studies have found an association between vision impairment and depression (Evans et al., 2007; Horowitz et al., 2005b; Kempen et al., 2012; Nollett et al., 2016). Vision rehabilitation has been shown to be effective at mitigating depression in populations with vision impairment. An RCT compared the effects of vision rehabilitation combined with either behavior activation or supportive therapy on the prevention of depressive symptoms among patients older than 65 with bilateral AMD, functional deficits consequent to visual acuity of <20/70 in the better-seeing eye, and subthreshold depression or depressed
mood.15 After 4 months, 12.6 percent and 23.4 percent of patients in the behavioral activation and supportive therapy groups, respectively, had developed a depressive disorder. Patients in the supportive therapy group were significantly more likely to have developed a depressive disorder (Rovner et al., 2014). Another study evaluated the effects of vision rehabilitation interventions on patients ages 65 and older with vision impairment. Two years into the study, the prevalence of significant depressive symptoms had declined from 33.7 percent to 25.3 percent, and the receipt of vision rehabilitation services and skills training was associated with significantly fewer depressive symptoms. In total, vision rehabilitation interventions accounted for 10 percent of the variance in the prevalence of depression after controlling for baseline depression and other variables (Horowitz et al., 2005a).
These findings support the American Academy of Ophthalmology and AOA guidelines on vision rehabilitation, which recommend assessing the cognitive/psychological status of patients with vision impairment (AAO, 2013; AOA, 2007). The U.S. Preventive Services Task Force (USPSTF) has recommended that primary care providers screen adult patients ages 18 and older—including older adults and pregnant and postpartum women—for depression, and states that primary care providers should consider patient “risk factors, comorbid conditions, and life events to determine if additional screening of high-risk patients is warranted” (USPSTF, 2016).
Provider adherence to these guidelines and recommendations may promote early identification, and subsequent diagnosis and treatment, of depression in populations with vision impairment. Research findings suggest that novel vision rehabilitation treatment plans and training programs for providers working in vision rehabilitation may also improve clinical outcomes and care quality for patients with vision impairment and comorbid depression (Rees et al., 2012; van der Aa et al., 2015). Further research is needed to develop interventions that improve the mental health of populations with vision impairment.
New Therapies for Comorbid Cognitive Impairment
Compared to younger adults with vision impairment, cognitive impairment is more prevalent and progresses more rapidly in older adults with vision impairment (Lin et al., 2004; Reyes-Ortiz et al., 2005; Rogers and
___________________
15 Behavioral activation “emphasized the link between action, mood, and mastery, and promoted self-efficacy and social connection as ways to improve mood and function and counter self-defeating behaviors” (Rovner et al., 2014, p. 3). Supportive therapy “facilitated personal expression about vision loss and disability and, in this trial, controlled for the nonspecific effects of attention” (Rovner et al., 2014, p. 4).
Langa, 2010; Tay et al., 2006; Whitson et al., 2007). Both cognitive impairment and vision impairment are disabling in their own right, but their co-occurrence has been associated with staggeringly high rates of disability and low self-rated health (Whitson et al., 2007, 2012a). Cognitive deficits were detected in about 40 percent of older adults with macular disease who were referred to an outpatient vision rehabilitation clinic (Whitson et al., 2010). Considering that rehabilitation typically entails education and training on new techniques and devices, it is easy to imagine that deficits in short-term memory, language processing, or executive function could limit a patient’s progress in the program. Indeed, among patients receiving customary vision rehabilitation services, the patient and caregiver experience as well as functional outcomes were all found to be negatively affected by comorbid cognitive impairment (Lawrence et al., 2009; Whitson et al., 2012b). However, efforts are under way to evaluate and implement programs specifically designed to detect and accommodate comorbid cognitive deficits. In a pilot study of one such program, Memory or Reasoning Enhanced Low Vision Rehabilitation, participants experienced subjective and objective improvements in several measures of visual function after participation in the 6-week program (Whitson et al., 2013). Additional work is needed to evaluate the sustainability of these effects and determine whether the new models are superior to “usual care” services for individuals with co-existing vision and cognitive impairment.
New Therapies for General Medical Comorbidity
Because most of the diseases that cause chronic vision impairment are age-related, many individuals who need vision rehabilitation have a host of other medical problems. An analysis of participants in the Medicare Expenditure Panel Survey found that “eye disorders” rarely occurred in isolation: more than 80 percent of beneficiaries with eye disorders also had at least one of the four other chronic conditions assessed (heart disease, diabetes, arthritis, or hypertension) (Anderson and Horvath, 2004). Vision rehabilitation services can be highly beneficial for individuals with comorbid medical conditions, by enabling them to overcome vision-related barriers to chronic disease management. Many important activities related to self-care for medical disease (e.g., medication administration, foot checks for diabetics, blood pressure monitoring, daily weight checks for heart failure) rely on vision. However, medical comorbidities (aside from depression and cognitive deficits) can interfere with vision rehabilitation. One study analyzed more than 600 interviews with patients and their companions and identified five broad themes related to how the patients’ experience in vision rehabilitation was affected by comorbid conditions (Whitson et al., 2011). For example, patients with multiple chronic conditions
frequently experienced “good days and bad days,” with fluctuations in symptoms such as pain or shortness of breath. When the appointments fell on “bad days,” these medically complex patients were less able to focus and subsequently perceived less benefit. While the study identified many such challenges related to comorbidity, it also identified potential solutions for each challenge. For example, the issue of “good days, bad days” might be addressed through flexible scheduling options that allow patients to stack appointments on “good days” or through Web-based or take-home materials that can supplement learning that may have suffered on a “bad day.”
CONCLUSION
Vision rehabilitation is essential to maximizing the independence, functioning, participation, safety, and overall QOL of people with chronic vision impairment. Yet there are numerous barriers to high-quality and universally accessible vision rehabilitation services. A lack of awareness among patients and providers of the purpose, methods, and effectiveness of vision rehabilitation; the lack of interprofessional guidelines on the clinical findings that should trigger referral of patients to vision rehabilitation; limited research, and limited resources for research, on the cost, effectiveness, and cost-effectiveness of interventions and models of care in vision rehabilitation; and barriers to access based on socioeconomic position and race all detrimentally impact care quality, access, and health outcomes. Improving the state of vision rehabilitation services and population vision health will require correcting each of these issues through a collaborative development of interprofessional guidelines, review and revision of the Centers for Medicare & Medicaid Services policy regarding coverage of vision services, design and funding of a health care services research agenda, and implementation of other strategies discussed above. In addition, stakeholders should continue to support and foster successes in vision rehabilitation related to emerging technologies and advancing medical knowledge and treatments.
This chapter identifies numerous information gaps. Box 8-1 lists these research gaps. Although this list is not comprehensive, it represents a starting point for developing a concrete set of prioritized research goals that may be fruitfully and collaboratively pursued by academic researchers, government centers, nonprofit research groups, and other stakeholders in eye and vision health.
Too often, the need for high-quality, equitable, and accessible vision rehabilitation services is lost in the focus on early detection and curative treatment of eye disease through surveillance and clinical care. Rather than treating vision rehabilitation as a last resort to be pursued only when “nothing else can be done,” optometrists and ophthalmologists should integrate
discussions of vision rehabilitation services into the earliest stages of clinical care, where they can improve QOL and visual functioning throughout the care process. This shift will require both the concrete measures described above and the conceptual and cultural work of rethinking the purposes and priorities of eye and vision care.
REFERENCES
AAO (American Academy of Optometry). 2013. Vision rehabilitation—Preferred Practice Pattern® guideline. http://www.aao.org/preferred-practice-pattern/vision-rehabilitation-ppp--2013 (accessed April 5, 2016).
ABMS (American Board of Medical Specialties). 2016. Specialty and subspecialty certificates. http://www.abms.org/member-boards/specialty-subspecialty-certificates (accessed May 27, 2016).
ACGME (Accreditation Council for Graduate Medical Education). 2012. ACGME program requirements for graduate medical education in ophthalmology. http://www.acgme.org/portals/0/pfassets/programrequirements/240_ophthalmology_2016.pdf (accessed June 6, 2016).
———. 2015. ACGME data resource book academic year 2014–2015. http://www.acgme.org/Portals/0/PFAssets/PublicationsBooks/2014-2015_ACGME_DATABOOK_DOCUMENT.pdf (accessed June 6, 2016).
ACOE (American Council on Optometric Education). 2016. Accredited optometric residency programs. http://www.aoa.org/Documents/students/residency_Directory_3_18_2016.pdf (accessed June 6, 2016).
ACVREP (Academy for Certification of Vision Rehabilitation and Education Professionals). 2014a. Orientation and mobility specialist certification handbook. https://www.acvrep.org/certifications/coms (accessed June 6, 2016).
———. 2014b. Vision rehabilitation therapist certification handbook. https://www.acvrep.org/certifications/cvrt (accessed June 6, 2016).
———. 2015. Certified orientation and mobility specialist. https://www.acvrep.org/ascerteon/control/certifications/coms (accessed March 31, 2016).
AFB (American Foundation for the Blind). 2014. Products for people who are blind or visually impaired. http://www.afb.org/prodmain.asp (accessed April 2, 2016).
AHRQ (Agency for Healthcare Research and Quality). 2002. Vision rehabilitation: Care and benefit plan models: Literature review. Applying the framework to vision rehabilitation. http://www.ahrq.gov/professionals/clinicians-providers/resources/vision/vision2.html (accessed June 6, 2016).
———. 2004. Vision rehabilitation for elderly individuals with low vision or blindness. https://www.cms.gov/Medicare/Coverage/InfoExchange/downloads/rtcvisionrehab.pdf (accessed June 6, 2016).
Anderson, G., and J. Horvath. 2004. The growing burden of chronic disease in America. Public Health Reports 119(3):263–270.
AOA (American Optometric Association). 2007. Care of the patient with visual impairment (low vision rehabilitation).http://www.aoa.org/documents/optometrists/CPG-14.pdf (accessed June 6, 2016).
AOTA (American Occupational Therapy Association). 2015. AOTA specialty certification in low vision occupational therapist candidate application. https://www.aota.org//media/Corporate/Files/EducationCareers/BSC/initial/SCLV-A-OTA-APPLICATION.pdf (accessed June 6, 2016).
———. 2016a. Approved & candidate residency sites. http://www.aota.org/Education-Careers/Advance-Career/Residency/ResidencySites.aspx (accessed May 27, 2016).
———. 2016b. Online course: Low vision in older adults: Foundations for rehabilitation, 2nd edition. https://myaota.aota.org/shop_aota/prodview.aspx?TYPE=D&PID=165106646&SKU=OL37?sc_camp=6970EB610A00413DAD32FA9102E6A366 (accessed May 27, 2016).
———. 2016c. Postprofessional programs in OT—Doctoral level programs. http://www.aota.org/Education-Careers/Find-School/Postprofessional/PostprofessionalOT-D.aspx (accessed May 27, 2016).
———. 2016d. Postprofessional programs in OT—Masters degree programs. http://www.aota.org/Education-Careers/Find-School/Postprofessional/PostprofessionalOTM.aspx (accessed May 27, 2016).
ASCO (Association of Schools and Colleges of Optometry). 2016. Optometry residency programs in AY 2015-2016. http://www.opted.org/wp-content/uploads/2016/05/OptometryResidency-Programs-2015-16-updated-5-23.pdf (accessed May 27, 2016).
Ballemans, J., G. I. J. M. Kempen, and G. A. R. Zijlstra. 2011. Orientation and mobility training for partially-sighted older adults using an identification cane: A systematic review. Clinical Rehabilitation 25(10):880–891.
Ballemans, J., G. R. Zijlstra, G. H. van Rens, J. S. Schouten, and G. I. Kempen. 2012. Usefulness and acceptability of a standardised orientation and mobility training for partially-sighted older adults using an identification cane. BMC Health Services Research 12(1):1–14.
Barker, L., R. Thomas, G. Rubin, and A. Dahlmann-Noor. 2015. Optical reading aids for children and young people with low vision. Cochrane Database of Systematic Reviews 3:CD010987.
Basulto, D. 2015. The new app that serves as eyes for the blind. Washington Post. October 22. https://www.washingtonpost.com/news/innovations/wp/2015/10/22/the-new-app-that-serves-as-eyes-for-the-blind (accessed September 1, 2016).
Berger, S., J. McAteer, K. Schreier, and J. Kaldenberg. 2013. Occupational therapy interventions to improve leisure and social participation for older adults with low vision: A systematic review. American Journal of Occupational Therapy 67(3):303–311.
Bhatlawande, S., M. Mahadevappa, J. Mukherjee, M. Biswas, D. Das, and S. Gupta. 2014. Design, development, and clinical evaluation of the electronic mobility cane for vision rehabilitation. IEEE Transactions on Neural Systems and Rehabilitation Engineering 22(6):1148–1159.
Binns, A. M., C. Bunce, C. Dickinson, R. Harper, R. Tudor-Edwards, M. Woodhouse, P. Linck, A. Suttie, J. Jackson, J. Lindsay, J. Wolffsohn, L. Hughes, and T. H. Margrain. 2012. How effective is low vision service provision? A systematic review. Survey of Ophthalmology 57(1):34–65.
Bodenheimer, T., K. Lorig, H. Holman, and K. Grumbach. 2002. Patient self-management of chronic disease in primary care. JAMA 288(19):2469–2475.
Bowers, A. R., C. Meek, and N. Stewart. 2001. Illumination and reading performance in age-related macular degeneration. Clinical and Experimental Optometry 84(3):139–147.
Bradfield, Y. S. 2013. Identification and treatment of amblyopia. American Family Physician 87(5):348–352.
Brody, B. L., A. C. Roch-Levecq, A. C. Gamst, K. Maclean, R. M. Kaplan, and S. I. Brown. 2002. Self-management of age-related macular degeneration and quality of life: A randomized controlled trial. Archives of Ophthalmology 120(11):1477–1483.
Brody, B. L., A. C. Roch-Levecq, R. G. Thomas, R. M. Kaplan, and S. I. Brown. 2005. Self-management of age-related macular degeneration at the 6-month follow-up: A randomized controlled trial. Archives of Ophthalmology 123(1):46–53.
Brody, B. L., A. C. Roch-Levecq, R. M. Kaplan, C. Y. Moutier, and S. I. Brown. 2006. Age-related macular degeneration: Self-management and reduction of depressive symptoms in a randomized, controlled study. Journal of the American Geriatrics Society 54(10):1557–1562.
Brown, J. C., J. E. Goldstein, T. L. Chan, R. Massof, and P. Ramulu. 2014. Characterizing functional complaints in patients seeking outpatient low-vision services in the United States. Ophthalmology 121(8):1655–1662, e1651.
Brunnström, G., S. Sörensen, K. Alsterstad, and J. Sjöstrand. 2004. Quality of light and quality of life—The effect of lighting adaptation among people with low vision. Ophthalmic and Physiological Optics 24(4):274–280.
Campbell, A. J., M. C. Robertson, S. J. La Grow, N. M. Kerse, G. F. Sanderson, R. J. Jacobs, D. M. Sharp, and L. A. Hale. 2005. Randomised controlled trial of prevention of falls in people aged ≥75 with severe visual impairment: The VIP trial. British Medical Journal 331(7520):817.
Casten, R. J., E. K. Maloney, and B. W. Rovner. 2005. Knowledge and use of low vision services among persons with age-related macular degeneration. Journal of Visual Impairment & Blindness 99(11):720–724.
CDC (Centers for Disease Control and Prevention). 2009. The burden of vision loss. http://www.cdc.gov/visionhealth/basic_information/vision_loss_burden.htm (accessed March 24, 2016).
Chavda, S., W. Hodge, F. Si, and K. Diab. 2014. Low-vision rehabilitation methods in children: A systematic review. Canadian Journal of Ophthalmology 49(3):e71–e73.
Chou, C. F., L. E. Barker, J. E. Crews, S. A. Primo, X. Zhang, A. F. Elliott, K. McKeever Bullard, L. S. Geiss, and J. B. Saaddine. 2012. Disparities in eye care utilization among the United States adults with visual impairment: Findings from the Behavioral Risk Factor Surveillance System 2006–2009. American Journal of Ophthalmology 154(6 Suppl):S45–S52, e41.
Chou, C. F., C. E. Sherrod, X. Zhang, L. E. Barker, K. M. Bullard, J. E. Crews, and J. B. Saaddine. 2014. Barriers to eye care among people aged 40 years and older with diagnosed diabetes, 2006–2010. Diabetes Care 37(1):180–188.
Christ, S. L., D. D. Zheng, B. K. Swenor, B. L. Lam, S. K. West, S. L. Tannenbaum, B. E. Muñoz, and D. J. Lee. 2014. Longitudinal relationships among visual acuity, daily functional status, and mortality: The Salisbury Eye Evaluation Study. JAMA Ophthalmology 132(12):1400–1406.
Crews, J. E., G. C. Jones, and J. H. Kim. 2006. Double jeopardy: The effects of comorbid conditions among older people with vision loss. Journal of Visual Impairment & Blindness 100:824.
Crews, J. E., C. F. Chou, J. A. Stevens, and J. B. Saaddine. 2016a. Falls among persons aged ≥65 years with and without severe vision impairment—United States, 2014. Morbidity and Mortality Weekly Report 65(17):433–437.
Crews, J. E., C. F. Chou, M. M. Zack, X. Zhang, K. M. Bullard, A. R. Morse, and J. B. Saaddine. 2016b. The association of health-related quality of life with severity of visual impairment among people aged 40-64 years: Findings from the 2006–2010 Behavioral Risk Factor Surveillance System. Ophthalmic Epidemiology 23(3):145–153.
Crossland, M. D., R. S. Silva, and A. F. Macedo. 2014. Smartphone, tablet computer and e-reader use by people with vision impairment. Ophthalmic and Physiological Optics 34(5):552–557.
Dahlin Ivanoff, S., U. Sonn, and E. Svensson. 2002. A health education program for elderly persons with visual impairments and perceived security in the performance of daily occupations: A randomized study. American Journal of Occupational Therapy 56(3):322–330.
Davidson, S., and G. E. Quinn. 2011. The impact of pediatric vision disorders in adulthood. Pediatrics 127(2):334–339.
Day, L., B. Fildes, I. Gordon, M. Fitzharris, H. Flamer, and S. Lord. 2002. Randomised factorial trial of falls prevention among older people living in their own homes. British Medical Journal 325(7356):128.
Demers-Turco, P. 1999. Providing timely and ongoing vision rehabilitation services for the diabetic patient with irreversible vision loss from diabetic retinopathy. Journal of the American Optometric Association 70(1):49–62.
Deremeik, J., A. T. Broman, D. Friedman, S. K. West, R. Massof, W. Park, K. Bandeen-Roche, K. Frick, and B. Muñoz. 2007. Low vision rehabilitation in a nursing home population: The seeing study. Journal of Visual Impairment & Blindness 101(11):701–714.
Elam, A. R., and P. P. Lee. 2013. High-risk populations for vision loss and eye care underutilization: A review of the literature and ideas on moving forward. Survey of Ophthalmology 58(4):348–358.
Erickson, W., C. Lee, S. von Schrader. 2016. Disability statistics from the 2014 American Community Survey (ACS). Ithaca, NY: Cornell University Yang Tan Institute (YTI).
Evans, J. R., A. E. Fletcher, and R. P. Wormald. 2007. Depression and anxiety in visually impaired older people. Ophthalmology 114(2):283–288.
Fraser, M. L., L. B. Meuleners, A. H. Lee, J. Q. Ng, and N. Morlet. 2013. Which visual measures affect change in driving difficulty after first eye cataract surgery? Accident Analysis and Prevention 58:10–14.
Fraser, S. A., A. P. Johnson, W. Wittich, and O. Overbury. 2015. Critical success factors in awareness of and choice towards low vision rehabilitation. Ophthalmic Physiological Optics 35(1):81–89.
Gaffney, A. J., T. H. Margrain, C. V. Bunce, and A. M. Binns. 2014. How effective is eccentric viewing training? A systematic literature review. Ophthalmic Physiological Optics 34(4):427–437.
Ganesh, S., S. Sethi, S. Srivastav, A. Chaudhary, and P. Arora. 2013. Impact of low vision rehabilitation on functional vision performance of children with visual impairment. Oman Journal of Ophthalmology 6(3):170–174.
Gilbert, C., and A. Foster. 2001. Childhood blindness in the context of vision 2020: The right to sight. Bulletin of the World Health Organization 79(3):227–232.
Goldstein, J. E., R. W. Massof, J. T. Deremeik, S. Braudway, M. L. Jackson, K. B. Kehler, S. A. Primo, J. S. Sunness, and the Low Vision Research Network Study Group. 2012. Baseline traits of low vision patients served by private outpatient clinical centers in the United States. Archives of Ophthalmology 130(8):1028–1037.
Goldstein, J. E., M. Jackson, S. M. Fox, J. T. Deremeik, and R. W. Massof, for the Low Vision Research Network Study Group. 2015. Clinically meaningful rehabilitation outcomes of low vision patients served by outpatient clinical centers. JAMA Ophthalmology 133(7):762–769.
Gothwal, V. K., R. Sumalini, and S. Bharani. 2015. Assessing the effectiveness of low vision rehabilitation in children: An observational study. Investigative Ophthalmology & Visual Science 56(5):3355–3360.
Hinds, A., A. Sinclair, J. Park, A. Suttie, H. Paterson, and M. Macdonald. 2003. Impact of an interdisciplinary low vision service on the quality of life of low vision patients. British Journal of Ophthalmology 87(11):1391–1396.
Hong, S. P., H. Park, J. S. Kwon, and E. Yoo. 2014. Effectiveness of eccentric viewing training for daily visual activities for individuals with age-related macular degeneration: A systematic review and meta-analysis. NeuroRehabilitation 34(3):587–595.
Hooper, P., J. W. Jutai, G. Strong, and E. Russell-Minda. 2008. Age-related macular degeneration and low-vision rehabilitation: A systematic review. Canadian Journal of Ophthalmology 43(2):180–187.
Horowitz, A., J. P. Reinhardt, and K. Boerner. 2005a. The effect of rehabilitation on depression among visually disabled older adults. Aging and Mental Health 9(6):563–570.
Horowitz, A., J. P. Reinhardt, and G. J. Kennedy. 2005b. Major and subthreshold depression among older adults seeking vision rehabilitation services. American Journal of Geriatric Psychiatry 13(3):180–187.
IOM (Institute of Medicine). 2001. Crossing the quality chasm: A new health system for the 21st century. Washington, DC: National Academy Press.
Justiss, M. D. 2013. Occupational therapy interventions to promote driving and community mobility for older adults with low vision: A systematic review. American Journal of Occupational Therapy 67(3):296–302.
Kempen, G. I., J. Ballemans, A. V. Ranchor, G. H. van Rens, and G. A. Zijlstra. 2012. The impact of low vision on activities of daily living, symptoms of depression, feelings of anxiety and social support in community-living older adults seeking vision rehabilitation services. Quality of Life Research 21(8):1405–1411.
Kolarik, A. J., S. Cirstea, S. Pardhan, and B. C. Moore. 2014. A summary of research investigating echolocation abilities of blind and sighted humans. Hearing Research 310:60–68.
Kondo, H. M., D. Pressnitzer, I. Toshima, and M. Kashino. 2012. Effects of self-motion on auditory scene analysis. Proceedings of the National Academy of Sciences of the United States of America 109(17):6775–6780.
Kuyk, T., L. Liu, J. L. Elliott, H. E. Grubbs, C. Owsley, G. McGwin, R. L. Griffin, and P. S. Fuhr. 2008. Health-related quality of life following blind rehabilitation. Quality of Life Research 17(4):497–507.
Lam, N., and S. J. Leat. 2013. Barriers to accessing low-vision care: The patient’s perspective. Canadian Journal of Ophthalmology 48(6):458–462.
Lamoureux, E. L., J. F. Pallant, K. Pesudovs, G. Rees, J. B. Hassell, and J. E. Keeffe. 2007. The effectiveness of low-vision rehabilitation on participation in daily living and quality of life. Investigative Ophthalmology & Visual Science 48(4):1476–1482.
Larizza, M. F., J. Xie, E. Fenwick, E. L. Lamoureux, J. E. Keeffe, and G. Rees. 2011. Impact of a low-vision self-management program on informal caregivers. Optometry & Visual Science 88(12):1486–1495.
Lawrence, V., J. Murray, D. Ffytche, and S. Banerjee. 2009. “Out of sight, out of mind”: A qualitative study of visual impairment and dementia from three perspectives. International Psychogeriatrics 21(3):511–518.
Leat, S. J. 2016. A proposed model for integrated low-vision rehabilitation services in Canada. Optometry & Visual Science 93(1):77–84.
Lee, L., T. L. Packer, S. H. Tang, and S. Girdler. 2008. Self-management education programs for age-related macular degeneration: A systematic review. Australasian Journal on Ageing 27(4):170–176.
Legge, G. E., P. J. Beckmann, B. S. Tjan, G. Havey, K. Kramer, D. Rolkosky, R. Gage, M. Chen, S. Puchakayala, and A. Rangarajan. 2013. Indoor navigation by people with visual impairment using a digital sign system. PLoS ONE 8(10):e76783.
Lin, M. Y., P. R. Gutierrez, K. L. Stone, K. Yaffe, K. E. Ensrud, H. A. Fink, C. A. Sarkisian, A. L. Coleman, and C. M. Mangione. 2004. Vision impairment and combined vision and hearing impairment predict cognitive and functional decline in older women. Journal of the American Geriatrics Society 52(12):1996–2002.
Liu, C.-J., M. A. Brost, V. E. Horton, S. B. Kenyon, and K. E. Mears. 2013. Occupational therapy interventions to improve performance of daily activities at home for older adults with low vision: A systematic review. American Journal of Occupational Therapy 67(3):279–287.
Luo, R.-J., S.-R. Liu, Z. Tian, W.-H. Zhu, Y.-H. Zhuo, and R.-D. Liao. 2011. Rehabilitation of vision disorder and improved quality of life in patients with primary open angle glaucoma. Chinese Medical Journal 124(17):2687–2691.
Maidenbaum, S., S. Hanassy, S. Abboud, G. Buchs, D. R. Chebat, S. Levy-Tzedek, and A. Amedi. 2014. The “eyecane,” a new electronic travel aid for the blind: Technology, behavior & swift learning. Restorative Neurology and Neuroscience 32(6): 813–824.
Markowitz, M. 2006a. Occupational therapy interventions in low vision rehabilitation. Canadian Journal of Ophthalmology 41(3):340–347.
Markowitz, S. N. 2006b. Principles of modern low vision rehabilitation. Canadian Journal of Ophthalmology 41(3):289–312.
Matti, A. I., K. Pesudovs, A. Daly, M. Brown, and C. S. Chen. 2011. Access to low-vision rehabilitation services: Barriers and enablers. Clinical and Experimental Optometry 94(2):181–186.
Milne, J. L., M. A. Goodale, and L. Thaler. 2014. The role of head movements in the discrimination of 2-D shape by blind echolocation experts. Attention, Perception & Psychophysics 76(6):1828–1837.
Minto, H., and Butt, I. A. 2004. Low vision devices and training. Community Eye Health 17(49):67.
Mogk, L., and G. Goodrich. 2004. The history and future of low vision services in the United States. Journal of Visual Impairment & Blindness 98(10):585–600.
Moore, N. C. 2015. Bringing Braille back with better display technology. http://www.engin.umich.edu/college/about/news/stories/2015/december/bringing-braille-back-with-a-betterdisplay-technology (accessed June 30, 2016).
Motto Ros, P., V. Dante, L. Mesin, E. Petetti, P. Del Giudice, and E. Pasero. 2014. A new dynamic tactile display for reconfigurable Braille: Implementation and tests. Frontiers in Neuroengineering 7:6.
Mwilambwe, A., W. Wittich, and E. E. Freeman. 2009. Disparities in awareness and use of low-vision rehabilitation. Canadian Journal of Ophthalmology 44(6):686–691.
NEHEP (National Eye Health Education Program). n.d. Vision rehabilitation: Helping people with low vision. https://nei.nih.gov/sites/default/files/nehep-pdfs/Low_Vision_Fact_Sheet_v3.pdf (accessed June 5, 2016).
NEI (National Eye Institute). 2004. National plan for eye and vision research. https://nei.nih.gov/sites/default/files/nei-pdfs/nationalplan1.pdf (accessed June 30, 2016).
———. 2010. Vision impairment tables. https://nei.nih.gov/eyedata/vision_impaired/tables (accessed March 25, 2016).
———. 2012. Vision research needs, gaps, and opportunities. https://nei.nih.gov/sites/default/files/nei-pdfs/VisionResearch2012.pdf (accessed February 22, 2016).
———. 2014. Facts about retinitis pigmentosa. https://nei.nih.gov/health/pigmentosa/pigmentosa_facts (accessed June 15, 2016).
———. 2015. Facts about Stargardt disease. https://nei.nih.gov/health/stargardt/star_facts (accessed June 15, 2016).
NEI/LCIF (National Eye Institute/Lions Club International Foundation). 2008. 2005 survey of public knowledge, attitudes, and practices related to eye health and disease. https://nei.nih.gov/kap (accessed June 6, 2016).
NFB (National Federation of the Blind). 2016. Technology resource list. https://nfb.org/technology-resource-list (accessed April 2, 2016).
Nguyen, N. X., M. Weismann, and S. Trauzettel-Klosinski. 2009. Improvement of reading speed after providing of low vision aids in patients with age-related macular degeneration. Acta Ophthalmologica 87(8):849–853.
Nilsson, U. L. 1986. Visual rehabilitation of patients with advanced diabetic retinopathy. A follow-up study at the low vision clinic, Department of Ophthalmology, University of Linkoping. Documenta Ophthalmologica 62(4):369–382.
Nipp, C. M., L. K. Vogtle, and M. Warren. 2014. Clinical application of low vision rehabilitation strategies after completion of a computer-based training module. Occupational Therapy in Health Care 28(3):296–305.
Nollett, C. L., N. Bray, C. Bunce, R. J. Casten, R. T. Edwards, M. T. Hegel, S. Janikoun, S. E. Jumbe, B. Ryan, J. Shearn, D. J. Smith, M. Stanford, W. Xing, and T. H. Margrain. 2016. High prevalence of untreated depression in patients accessing low-vision services. Ophthalmology 123(2):440-441.
O’Connor, P. M., L. C. Mu, and J. E. Keeffe. 2008. Access and utilization of a new low-vision rehabilitation service. Clinical and Experimental Ophthalmology 36(6):547–552.
O’Day, B. L., M. Killeen, and L. I. Iezzoni. 2004. Improving health care experiences of persons who are blind or have low vision: Suggestions from focus groups. American Journal of Medical Quality 19(5):193–200.
Overbury, O., and W. Wittich. 2011. Barriers to low vision rehabilitation: The Montreal Barriers Study. Investigative Ophthalmology & Visual Science 52(12):8933–8938.
Owsley, C., and G. McGwin. 2010. Vision and driving. Vision Research 50(23):2348–2361.
Owsley, C., G. McGwin, Jr., P. P. Lee, N. Wasserman, and K. Searcey. 2009. Characteristics of low-vision rehabilitation services in the United States. Archives of Ophthalmology 127(5):681–689.
Patino, C. M., R. McKean-Cowdin, S. P. Azen, J. C. Allison, F. Choudhury, and R. Varma. 2010. Central and peripheral visual impairment and the risk of falls and falls with injury. Ophthalmology 117(2):199–206, e191.
Pollard, T. L., J. A. Simpson, E. L. Lamoureux, and J. E. Keeffe. 2003. Barriers to accessing low vision services. Ophthalmic Physiological Optics 23(4):321–327.
Pym, H. 2015. Technology helps visually impaired navigate the tube. BBC. March 5. http://www.bbc.com/news/health-31754365 (accessed June 30, 2016).
Rees, G., E. E. Holloway, G. Craig, N. Hepi, S. Coad, J. E. Keeffe, and E. L. Lamoureux. 2012. Screening for depression: Integrating training into the professional development programme for low vision rehabilitation staff. Clinical and Experimental Ophthalmology 40(9):840–848.
Rees, G., J. Xie, P. P. Chiang, M. F. Larizza, M. Marella, J. B. Hassell, J. E. Keeffe, and E. L. Lamoureux. 2015. A randomised controlled trial of a self-management programme for low vision implemented in low vision rehabilitation services. Patient Education and Counseling 98(2):174–181.
Renieri, G., S. Pitz, N. Pfeiffer, M. E. Beutel, and R. Zwerenz. 2013. Changes in quality of life in visually impaired patients after low-vision rehabilitation. International Journal of Rehabilitation Research 36(1):48–55.
Reyes-Ortiz, C. A., Y. F. Kuo, A. R. DiNuzzo, L. A. Ray, M. A. Raji, and K. S. Markides. 2005. Near vision impairment predicts cognitive decline: Data from the Hispanic Established Populations for Epidemiologic Studies of the Elderly. Journal of the American Geriatrics Society 53(4):681–686.
Rogers, M. A. M., and K. M. Langa. 2010. Untreated poor vision: A contributing factor to late-life dementia. American Journal of Epidemiology 171(6):728–735.
Rovner, B. W., R. J. Casten, M. T. Hegel, R. W. Massof, B. E. Leiby, A. C. Ho, and W. S. Tasman. 2014. Low Vision Depression Prevention Trial in Age-Related Macular Degeneration: A randomized clinical trial. Ophthalmology 121(11):2204–2211.
Russomanno, A., S. O’Modhrain, R. B. Gillespie, and M. W. Rodger. 2015. Refreshing refreshable Braille displays. IEEE Transactions on Haptics 8(3):287–297.
Ryan, B. 2014. Models of low vision care: Past, present and future. Clinical and Experimental Optometry 97(3):209–213.
Ryan, B., S. White, J. Wild, H. Court, and T. H. Margrain. 2010. The newly established primary care based Welsh Low Vision Service is effective and has improved access to low vision services in Wales. Ophthalmic Physiological Optics 30(4):358–364.
Ryan, B., J. Khadka, C. Bunce, and H. Court. 2013. Effectiveness of the community-based low vision service Wales: A long-term outcome study. British Journal of Ophthalmology 97(4):487–491.
Sandlin, D., G. McGwin, Jr., and C. Owsley. 2014. Association between vision impairment and driving exposure in older adults aged 70 years and over: A population-based examination. Acta Ophthalmologica 92(3):e207–e212.
Scanlan, J., and J. E. Cuddeford. 2004. Low vision rehabilitation: A comparison of traditional and extended teaching programs. Journal of Visual Impairment & Blindness 98(10):601–610.
Schenkman, B. N., and M. E. Nilsson. 2010. Human echolocation: Blind and sighted persons’ ability to detect sounds recorded in the presence of a reflecting object. Perception 39(4):483–501.
Schornich, S., A. Nagy, and L. Wiegrebe. 2012. Discovering your inner bat: Echo-acoustic target ranging in humans. Journal of the Association for Research in Otolaryngology 13(5):673–682.
Schornich, S., L. Wallmeier, N. Gessele, A. Nagy, M. Schranner, D. Kish, and L. Wiegrebe. 2013. Psychophysics of human echolocation. Advances in Experimental Medicine and Biology 787:311–319.
Skelton, D. A., T. E. Howe, C. Ballinger, F. Neil, S. Palmer, and L. Gray. 2013. Environmental and behavioural interventions for reducing physical activity limitation in community-dwelling visually impaired older people. Cochrane Database of Systematic Reviews 6:CD009233.
Smallfield, S., K. Clem, and A. Myers. 2013. Occupational therapy interventions to improve the reading ability of older adults with low vision: A systematic review. American Journal of Occupational Therapy 67(3):288–295.
Stelmack, J. A., D. Moran, D. Dean, and R. W. Massof. 2007a. Short- and long-term effects of an intensive inpatient vision rehabilitation program. Archives of Physical Medicine and Rehabilitation 88(6):691–695.
Stelmack, J. A., X. C. Tang, D. J. Reda, D. Moran, S. Rinne, R. M. Mancil, R. Cummings, G. Mancil, K. Stroupe, N. Ellis, and R. W. Massof. 2007b. The Veterans Affairs Low Vision Intervention Trial (LOVIT): Design and methodology. Clinical Trials 4(6):650–660.
Stelmack, J. A., X. C. Tang, D. J. Reda, S. Rinee, R. M. Mancil, R. W. Massof, for the LOVIT Study Group. 2008. Outcomes of the Veterans Affairs Low Vision Intervention Trial (LOVIT). Archives of Ophthalmology 126(5):608–617.
Stelmack, J. A., X. C. Tang, Y. Wei, and R. W. Massof, for the Low-Vision Intervention Trial Study Group. 2012a. The effectiveness of low-vision rehabilitation in 2 cohorts derived from the Veterans Affairs Low Vision Intervention Trial. Archives of Ophthalmology 130(9):1162–1168.
Stelmack, J. A., X. C. Tang, D. J. Reda, K. T. Stroupe, S. Rinne, and R. W. Massof. 2012b. VA LOVIT II: A protocol to compare low vision rehabilitation and basic low vision. Ophthalmic Physiological Optics 32(6):461–471.
Stenquist, P. 2014. In self-driving cars, a potential lifeline for the disabled. New York Times. November 7. http://www.nytimes.com/2014/11/09/automobiles/in-self-driving-cars-apotential-lifeline-for-the-disabled.html?_r=0 (accessed June 30, 2016).
Stroupe, K. T., J. A. Stelmack, X. C. Tang, D. J. Reda, D. Moran, S. Rinne, R. Mancil, Y. Wei, R. Cummings, G. Mancil, N. Ellis, and R. W. Massof. 2008. Economic evaluation of blind rehabilitation for veterans with macular diseases in the Department of Veterans Affairs. Ophthalmic Epidemiology 15(2):84–91.
Swenor, B. K., E. M. Simonsick, L. Ferrucci, A. B. Newman, S. Rubin, and V. Wilson, for the the Health, Aging, and Body Composition Study. 2015. Visual impairment and incident mobility limitations: The Health ABC Study. Journal of the American Geriatrics Society 63(1):46–54.
Tay, T., J. J. Wang, A. Kifley, R. Lindley, P. Newall, and P. Mitchell. 2006. Sensory and cognitive association in older persons: Findings from an older Australian population. Gerontology 52(6):386–394.
Teng, S., and D. Whitney. 2011. The acuity of echolocation: Spatial resolution in the sighted compared to expert performance. Journal of Visual Impairment & Blindness 105(1):20–32.
Teng, S., A. Puri, and D. Whitney. 2012. Ultrafine spatial acuity of blind expert human echolocators. Experimental Brain Research 216(4):483–488.
Thomas, R., L. Barker, G. Rubin, and A. Dahlmann-Noor. 2015. Assistive technology for children and young people with low vision. Cochrane Database of Systematic Reviews 6:CD011350.
USPSTF (U.S. Preventive Services Task Force). 2016. Screening for depression in adults: U.S. Preventive Services Task Force recommendation statement. JAMA 315(4): 380–387.
VA (U.S. Department of Veterans Affairs). 2015. Rehabilitation and prosthetic services: About blind rehabilitation services. http://www.prosthetics.va.gov/blindrehab/BRS_Coordinated_Care.asp (accessed March 31, 2016).
———. 2016. Kansas City VA Medical Center. http://www.kansascity.va.gov/services/Vision_Impairment_Rehabilitation.asp (accessed May 27, 2016).
van der Aa, H. P. A., G. H. van Rens, H. C. Comijs, T. H. Margrain, F. Gallindo-Garre, J. W. Twisk, and R. M. van Nispen. 2015. Stepped care for depression and anxiety in visually impaired older adults: Multicentre randomised controlled trial. British Medical Journal 351:h6127.
Varma, R., T. S. Vajaranant, B. Burkemper, S. Wu, M. Torres, C. Hsu, F. Choudhury, and R. McKean-Cowdin. 2016. Visual impairment and blindness in adults in the United States: Demographic and geographic variations from 2015 to 2050. JAMA Ophthalmology 134(7):802–809.
Vercillo, T., J. L. Milne, M. Gori, and M. A. Goodale. 2015. Enhanced auditory spatial localization in blind echolocators. Neuropsychologia 67:35–40.
Virgili, G., and G. Rubin, 2010. Orientation and mobility training for people with low vision. Cochrane Database of Systematic Reviews 5:CD003925.
Virgili, G., R. Acosta, L. L. Grover, S. A. Bentley, and G. Giacomelli. 2013. Reading aids for adults with low vision. Cochrane Database of Systematic Reviews 10:CD003303.
VisionAware. 2016a. Organizing and labeling clothing when you are blind or have low vision. http://www.visionaware.org/section.aspx?FolderID=8&SectionID=115&TopicID=485&DocumentID=5814&rewrite=0 (accessed June 17, 2016).
———. 2016b. Safe cooking techniques for cooks who are blind or have low vision. http://www.visionaware.org/info/everyday-living/cooking/safe-cooking-techniques/135 (accessed June 17, 2016).
Wallmeier, L., and L. Wiegrebe. 2014a. Ranging in human sonar: Effects of additional early reflections and exploratory head movements. PLoS ONE 9(12):e115363.
———. 2014b. Self-motion facilitates echo-acoustic orientation in humans. Royal Society Open Science 1(3):140185.
Walter, C., R. Althouse, H. Humble, W. Smith, and J. V. Odom. 2007. Vision rehabilitation: Recipients’ perceived efficacy of rehabilitation. Ophthalmic Epidemiology 14(3):103–111.
Wang, B. Z., K. Pesudovs, M. C. Keane, A. Daly, and C. S. Chen. 2012. Evaluating the effectiveness of multidisciplinary low-vision rehabilitation. Optometry and Visual Science 89(9):1399–1408.
Whitson, H. E., S. W. Cousins, B. M. Burchett, C. F. Hybels, C. F. Pieper, and H. J. Cohen. 2007. The combined effect of visual impairment and cognitive impairment on disability in older people. Journal of the American Geriatrics Society 55(6):885–891.
Whitson, H. E., D. Ansah, D. Whitaker, G. Potter, S. W. Cousins, H. MacDonald, C. F. Pieper, L. Landerman, D. C. Steffens, and H. J. Cohen. 2010. Prevalence and patterns of comorbid cognitive impairment in low vision rehabilitation for macular disease. Archives of Gerontolology and Geriatrics 50(2):209–212.
Whitson, H. E., K. Steinhauser, N. Ammarell, D. Whitaker, S. W. Cousins, D. Ansah, L. L. Sanders, and H. J. Cohen. 2011. Categorizing the effect of comorbidity: A qualitative study of individuals’ experiences in a low-vision rehabilitation program. Journal of the American Geriatric Society 59(10):1802–1809.
Whitson, H. E., R. Malhotra, A. Chan, D. B. Matchar, and T. Ostbye. 2012a. Comorbid visual and cognitive impairment: Relationship with disability status and self-rated health among older Singaporeans. Asia Pacific Journal of Public Health 26(3):310–319.
Whitson, H. E., D. Whitaker, L. L. Sanders, G. G. Potter, S. W. Cousins, D. Ansah, E. McConnell, C. F. Pieper, L. Landerman, D. C. Steffens, and H. J. Cohen. 2012b. Memory deficit associated with worse functional trajectories in older adults in low-vision rehabilitation for macular disease. Journal of the American Geriatric Society 60(11):2087–2092.
Whitson, H. E., D. Whitaker, G. Potter, E. McConnell, F. Tripp, L. L. Sanders, K. W. Muir, H. J. Cohen, and S. W. Cousins. 2013. A low-vision rehabilitation program for patients with mild cognitive deficits. JAMA Ophthalmology 131(7):912–919.
Winner, S., H. K. Yuen, L. K. Vogtle, and M. Warren. 2014. Factors associated with comfort level of occupational therapy practitioners in providing low vision services. American Journal of Occupational Therapy 68(1):96–101.
Wittenborn, J., and D. Rein. 2016 (unpublished). The potential costs and benefits of treatment for undiagnosed eye disorders. Paper prepared for the Committee on Public Health Approaches to Reduce Vision Impairment and Promote Eye Health. http://www.nationalacademies.org/hmd/~/media/Files/Report%20Files/2016/UndiagnosedEyeDisordersCommissionedPaper.pdf (accessed September 15, 2016).
Zhang, X., M. F. Cotch, A. Ryskulova, S. A. Primo, P. Nair, C.-F. Chou, L. S. Geiss, L. Barker, A. F. Elliott, J. E. Crews, and J. B. Saaddine. 2012. Vision health disparities in the United States by race/ethnicity, education, and economic status: Findings from two nationally representative surveys. American Journal of Ophthalmology 154(6 Suppl):S53–S62, e51.
Zhang, X., K. M. Bullard, M. F. Cotch, M. R. Wilson, B. W. Rovner, G. McGwin, C. Owsley, L. Barker, J. E. Crews, and J. B. Saaddine. 2013a. Association between depression and functional vision loss in persons 20 years of age or older in the United States, NHANES 2005–2008. JAMA Ophthalmology 131(5):573–581.
Zhang, X., G. L. Beckles, C. F. Chou, J. B. Saaddine, M. R. Wilson, P. P. Lee, N. Parvathy, A. Ryskulova, and L. S. Geiss. 2013b. Socioeconomic disparity in use of eye care services among U.S. adults with age-related eye diseases: National Health Interview Survey, 2002 and 2008. JAMA Ophthalmology 131(9):1198–1206.
Zijlstra, G. A. R., J. Ballemans, and G. I. J. M. Kempen. 2013. Orientation and mobility training for adults with low vision: A new standardized approach. Clinical Rehabilitation 27(1):3–18.
This page intentionally left blank.














































