Marine Mammal Restoration Monitoring
WHY RESTORE MARINE MAMMALS?
Marine mammals in the Gulf of Mexico are highly valued by the public, and have been adversely affected by the cumulative impacts of human activities over the past 50 years, with large die-offs receiving considerable public attention (Vollmer and Rosel, 2013). Mortality, reduced reproductive rates, and poor health of individuals have been observed over this timeframe (Litz et al., 2014). The DWH oil spill resulted in further mortality, organ damage, immunosuppression, and decreased reproduction for a number of species of marine mammals (Schwacke et al., 2014; Lane et al., 2015). The disaster also damaged habitat for prey, thus limiting food availability and exacerbating the physical and toxic effects of oil and dispersants. As marine mammals are long-lived, slow to reproduce, and are apex predators, impacts of the spill are likely to continue even as restoration activities are started. Due to the ecological diversity of the marine mammal species and stocks in the Gulf of Mexico, impacts of the spill on these stocks are varied, with the most severe injuries documented in stocks with ranges overlapping the DWH oil spill footprint and accessible to study, especially coastal bottlenose dolphin stocks from Barataria Bay and Mississippi Sound (Schwacke et al., 2014). Offshore large whales have been harder to study, but estimates of mortality reached 17% of the Bryde’s whale population and 6% of the sperm whale population (DWH MMIQT, 2015).
RESTORATION OBJECTIVES
Considering the variation in range and behavior of the many species of whales, dolphins, and manatees living in the Gulf of Mexico, restoration activities will differentially affect different marine mammal stocks. This consideration increases the logistical challenge of documenting effects of restoration for many offshore species, with the notable exception of coastal bottlenose dolphins. Restoration projects in the Gulf of Mexico will aim to recover marine mammal populations by enhancing habitat, by reducing stressors known to cause morbidity (reduced fitness and illness) and mortality, and by addressing direct human-caused threats. These projects will address the following objectives under two overarching goals:
- Restore healthy populations of marine mammals through habitat improvement and mitigation of key stressors by
- enhancing habitat for prey,
- reducing pollution of coastal waters,
- decreasing anoxic zones and harmful algal blooms,
- decreasing and/or mitigating interact-tions with commercial and recreational fishing,
- reducing illegal feeding and harassment,
- reducing noise impacts, and
- reducing vessel collisions; and
- Improve the understanding of impacts to marine mammal populations by external stressors to inform adaptive management by
- enhancing stranding network capabilities,
- enhancing stock assessments, and
- ensuring restoration activities aimed at enhancing recreational and economic resources, or restoring coastal habitat through sediment and hydrologic diversions do not adversely impact marine mammals.
Examples of common restoration objectives are provided in Table II.6, including rehabilitating healthy populations by reducing illegal feeding of dolphins, improving coastal habitat (which may affect coastal dolphins), and reducing noise from dredging activities. Table II.6 also lists a set of metrics (described below) that may help assess progress towards these objectives in many cases, and depending on the relevant monitoring purpose (note that example metrics to support monitoring for adaptive management are not included because of their inherent project/program specificity).
DECISION-CRITICAL UNCERTAINTIES
Marine mammal species in the Gulf of Mexico vary ecologically, from coastal residents that feed on local prey, to offshore pelagic and migratory species that may feed on prey from outside the
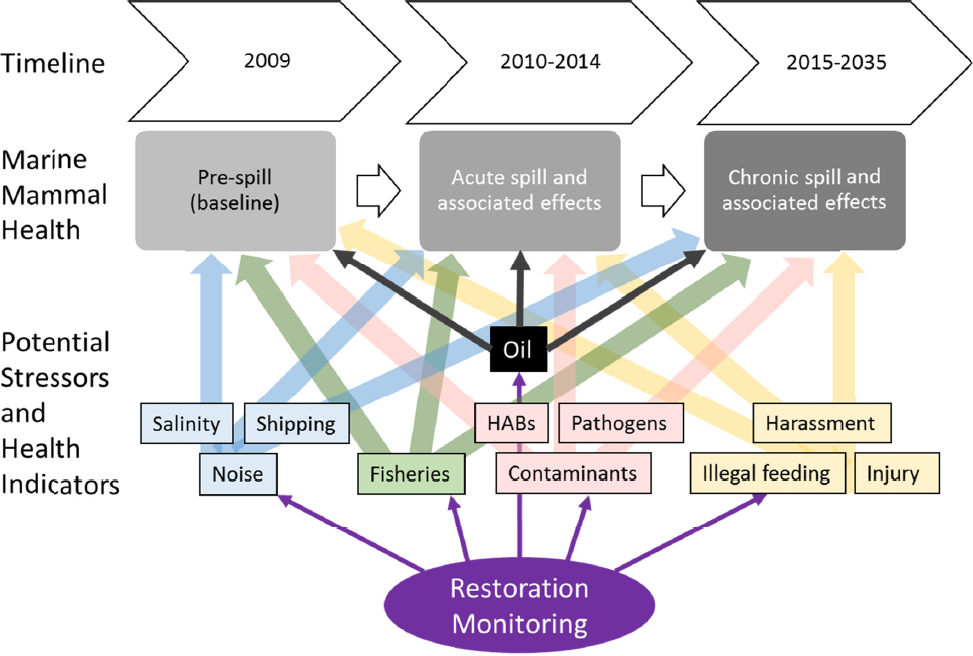
Gulf. Marine mammals are long-lived and slow to reach reproductive maturity, and as the effects of oil on health may be exerted on second generation individuals, effects of oil exposure during the DWH spill may still be occurring at the same time as effects of restoration activities a decade later. Although the release of oil during the DWH spill was mostly offshore in deep water, most restoration activities are coastal. Impacts of restoration activities thus may act on marine mammals at different temporal and spatial scales from the activity itself. In addition, many restoration activities could have cumulative impacts on marine mammals by affecting different stressors that act synergistically on marine mammal health. Understanding the effects of restoration activities relative to the continuing impacts of an oil spill requires carefully designed monitoring that may be best addressed in the context of an adaptive management program.
For some marine mammal species and stocks in the Gulf of Mexico, baseline (pre-spill) data on population size, reproductive rates, and seasonal distribution of animals are poorly known (Waring et al., 2015). For other stocks, especially coastal bottlenose dolphins, these data are available, as well as more detailed information on health and causes of death and morbidity. Unusual mortality events involving hundreds of dead marine mammals washing ashore have occurred over the last 20 years, with multiple factors including fresh water inflow, viral epidemics, harmful algal blooms (HABs), and the DWH oil spill contributing to death of these animals (Schwacke et al., 2010; Carmichael et al., 2012, Litz et al., 2014; Venn-Watson et al., 2015). The relative importance of each factor in some years is unclear, as these factors can be synergistic. HABs can cause immunosuppression, increasing susceptibility to infectious disease. Some infectious diseases predispose animals to other diseases, and changes in prey distribution and/or water temperature can alter marine mammal distribution, and thus exposure to HABS. Many of these factors can increase the likelihood to strand. Understanding the complexity of these interactions (see Figure II.6) will require extensive evaluation of health and stranding data throughout restoration activities, concurrent with the evaluation of stressors, to enable adaptive management and effective restoration of marine mammal populations.
PROJECT-LEVEL MONITORING AND ASSESSMENT PLAN CONSIDERATIONS
Information Needs Based on Monitoring Purpose and Project Objectives
As defined in Part I of this report, the three primary purposes of restoration monitoring include (1) assuring projects are built and are initially functioning as designed (construction monitoring); (2) assessing whether restoration goals and objectives have been or are being met (performance monitoring); and (3) informing restoration management, improving design of future restoration efforts, and increasing ecosystem understanding (monitoring for adaptive management). Restoration activities specifically designed to restore marine mammal populations will be monitored using a combination of construction and performance monitoring (see Table II.6). In addition, monitoring for adaptive management is needed to help reduce uncertainties and guide management decisions, given the limited experience implementing restoration for marine mammals. Furthermore, although many restoration activities that are executed with entirely different objectives will not aim to restore marine mammals, they may influence ecosystem parameters or stressors that in turn affect these species (see Box II.3).
An integrated and coordinated approach to monitoring marine mammals in the Gulf is needed to determine how they are affected by restoration activities operating under a variety of objectives, rather than only those activities targeting marine mammals specifically, in an ecosystem that is impacted continually by multiple stressors. This effort will depend upon a synthetic data management system that integrates monitoring data from different sources and projects (see Chapter 4). Collaborative, consistent, comparable, and standardized data collection and analysis is important, due to the long timescale of the data series and the multiple collectors. Investment in marine mammal data management beyond existing levels will enable these considerations, so that synthesis of the effects of restoration projects on marine mammals can be effective (see Chapter 5), and performance of projects can be refined to ensure enhancement of marine mammal populations (see Chapter 6).
Construction Monitoring
Construction monitoring is needed to determine whether a particular project was implemented as designed, such as monitoring enforcement, fisheries bycatch observer effort, and compliance with state and federal laws protecting marine mammals. Ready access to data on the status of other restoration projects not aimed specifically at marine mammal restoration, but likely to affect marine mammals (e.g., through salinity changes, dredging activity, noise), is also needed.
Performance Monitoring
Performance of restoration activities for marine mammals will be monitored by quantifying parameters such as abundance, distribution, and health, as well as the key stressors that impact these factors (e.g., salinity, prey abundance, shipping activity, noise, HABs, direct human actions such as illegal feeding, sport killing, and fisheries bycatch) (Vollmer and Rosel, 2013). The extent to which each of these factors can be measured will vary among marine mammal stocks, due to differences in accessibility and logistical challenges. It is good practice to determine impacts on population abundance for all species and stocks broadly using coordinated ship-based and aerial surveys and passive acoustic monitoring. Ideally, each stock abundance would be determined at least every
two years, and opportunities to coordinate with sea turtle surveys would be investigated.
Monitoring for Adaptive Management
Data obtained from the marine mammal stranding network on marine mammal health and causes of morbidity and mortality can be synthesized to aid adaptive management (see Table 4.1). For example, if changes in dolphin skin health resembling those typically caused by changes in salinity were detected in association with restoration projects that are known to affect the salinity of dolphin habitat, a likely link could be determined. NOAA Fisheries Service’s Marine Mammal Health and Stranding Response Program manages responses to stranded cetaceans and evaluates their health and health trends; correlates health and trend data with biological, physical, and chemical environmental parameters; and coordinates responses to unusual mortality events. The Manatee Salvage and Necropsy Program at the Florida Fish and Wildlife Research Institute supports efforts to salvage and necropsy Florida manatees throughout their range, including animals that strand outside the state of Florida, and monitors trends in manatee mortality. The U.S. Fish and Wildlife Service also responds to calls about injured and distressed manatees throughout the southeastern United States. Archiving these diverse data in an integrated database will allow sharing and synthesis from multiple sources so that data can be synthesized to produce population abundance, distribution, and health estimates. Such a database will require considerable investment and management, yet would ensure maximal benefit to marine mammal population restoration from executed projects.
Choose Suitable Metrics
Abundance
Abundance of marine mammal stocks (21 species and 56 stocks of marine mammals occur within the Gulf of Mexico) is mostly determined through ship-based and aerial surveys, which in the Gulf are performed and coordinated by NOAA Fisheries Service through the Stock Assessment Reports1 (Waring et al., 2015). The Fisheries Service has validated different survey techniques (aerial and ship-based line transects, mark-recapture techniques using photographs and biopsies, passive acoustic monitoring) and modeling methodologies, as the lead agency in coordinating abundance estimates of marine mammals in the Gulf (Fulling et al., 2003; Hubard et al., 2004). However, current federal resources to support abundance estimates are inconsistent and would benefit from augmentation to ensure more extensive and consistent spatial and temporal coverage of abundance estimates. As small-scale restoration projects will not likely have the ability to perform marine mammal abundance surveys, a program to which small projects or project coordinating agencies can contribute resources, would be beneficial. Data from federal, state, private, and academic institutions that can augment abundance estimates (e.g., photo-identification data, acoustic monitoring, movements) would benefit from integration by NOAA Fisheries Service with investments in data management and synthesis. Good practice is to continue monitoring abundance for at least two marine mammal generation times, which for some large whales is 70 years (Thomas et al., 2015).
In addition to abundance, data to inform stock structure are needed (e.g., genetics, movements) to improve characterization of stocks beyond current knowledge of coastal bottlenose dolphin stock structure (Sellas et al., 2005). These can be obtained through biopsy, photo-identification, tagging, and passive acoustic monitoring (Wells and Scott 1990). A unique lineage of Bryde’s whale, for example, was only recently characterized (Rosel and Wilcox, 2014). As mortality of this population following DWH was estimated at 17%, and these animals are affected by human activities such as noise, ship strikes, and fisheries interactions, their designation as a discrete stock can affect these activities if mortality exceeds permissible unintentional human-induced mortality for the stock.2
Distribution
Distribution data informs spatial planning tools, such as habitat use mapping, so spatial adaptive management approaches can be used during restoration activities. Data on marine mammal distribution can be obtained through direct observation from vessels and airplanes;
___________________
1 NOAA Stock Assessment Reports: http://www.nmfs.noaa.gov/pr/sars/species.htm.
2 See NOAA Fisheries Stock Assessment reports and Potential Biological Removal process: http://www.nmfs.noaa.gov/pr/sars.
acoustic monitoring of produced sounds by acoustic buoys, bottom-mounted acoustic units, towed arrays, and glider mounted devices; and tagging of animals with radio and satellite-linked telemetry devices (Hansen et al,. 1996; Garrison et al., 2002; MMC, 2011; Hildebrand et al., 2015). Different approaches are needed for different species, due to variations in accessibility, movement, and availability of baseline data. Frequency of monitoring will depend upon the monitoring tool(s) used, and the species to be assessed, but need to obtain data on species distribution at least biennially (see Chapter 3 for general good practices on monitoring scale and frequency). Tagging of representative offshore species, such as sperm whales at sites with differing degrees of anthropogenic activity, would be a useful approach to monitoring as some baseline data exist for this species (Jochens et al., 2008; McConnell et al., 2010). Passive acoustic monitoring is useful for gathering data on recovery of deep diving offshore species (Hildebrand et al., 2015). Integrating data into a Gulf-wide database including other environmental parameters and stressors that influence marine mammal distribution, such as the Gulf of Mexico Coastal Ocean Observing System (GCOOS), would be beneficial. Data on distribution can be combined with habitat use and stressor information to generate fine scale habitat use models that can guide management and restoration activities.
Health
Monitoring health of marine mammals focuses on monitoring representative populations at index sites for which baseline data exist and logistically feasible techniques have been developed. Animals that are most likely to be affected by perturbations resulting from restoration activities, as well as other stressors, are logical subjects for health monitoring. Coastal bottlenose dolphins, especially the Barataria Bay and Mississippi Sound populations, meet these criteria and were used by NOAA to assess the damage to natural resources from the DWH spill. Representative populations’ health is monitored through photography, remote biopsy, and live capture-sampling-release programs that include clinical evaluations, life history assessments, and sample collection. Samples can be evaluated for specific health parameters and measures of organ function, and individual animals given health scores using predetermined suites of parameters (Schwacke et al., 2014). Good practice is to monitor dolphins that include a coastal population at a restoration site (e.g., Barataria Bay, Mississippi Sound) and a population at a site within the Gulf unaffected by the spill or restoration projects (e.g., Sarasota Bay). Sampling for health monitoring ought to be performed annually at reference sites.
Changes in health may also be detected through surveillance of stranded animals. The stranding network is also vital to monitoring efficacy of restoration projects to reduce marine mammal bycatch and interaction rates with fishing gear (Byrd et al., 2008, 2014). Enhancing the stranding network throughout the Gulf of Mexico (potentially in coordination with sea turtle stranding response operations) would result in standardized detection, monitoring, sampling, and reporting systems, as well as data integration through GCOOS with information such as water quality and harmful algal blooms. This would allow data on marine mammal health and abundance to inform managers of effects of restoration activities (positive or negative) on marine mammal populations, and thus guide future restoration project planning to optimize marine mammal restoration (see Table II.6). Data collection by the stranding network ought to be continual, with real time reporting to managers.
Table II.6 Metrics Considered Good Practice to Monitor Marine Mammal Restoration Activities for Construction, Performance Toward Project Objectives, and Program-Level Or Large-Scale Assessments
| Monitoring Purpose | |||
|---|---|---|---|
| Construction | Performance | Program-level | |
| Potential Monitoring Metrics | Examples | Examples | Suggested |
| Habitat | |||
|
Map of animal distributions (transect observations, biologging tag coordinates, GPS photo identification, passive acoustic monitoring, stranded animal location) |
#1, #2, #3 | #1, #2, #3 | X |
|
Location and timing of extractive activities |
X | ||
|
Location and timing of recreational activities |
#1 | #1 | X |
|
Location and timing of construction activities |
#3 | #3 | X |
|
Date and location of possibly interacting management activities |
#2, #3 | X | |
| Oceanographic Conditions | |||
|
Salinity |
#2 | #2 | X |
|
Calibrated noise measurements with GPS coordinates of recorders and times |
#3 | #3 | |
|
Weather (fog, glare, visibility) |
|||
|
Sea state |
|||
|
Tidal state |
|||
|
Water depth |
|||
|
Biotoxin concentration |
|||
|
Number of boats illegally feeding animals |
#1 | #1 | |
| Fauna | |||
|
Abundance and distribution (counted or estimated) |
#2 | X | |
|
Photographs of coastal dolphins |
#2 | ||
|
Species composition |
|||
|
Group size |
|||
|
Age/size/gender categories |
|||
|
Biopsies of coastal dolphins |
#2 | ||
|
Health assessment of live-capture-released coastal dolphins or manatees |
#2 | #2 | X |
|
Number of stranded dolphins with fresh water skin lesions |
#2 | #2 | |
|
Number of injured dolphins, whales, or manatees (photo-IDs) |
#1 | ||
|
Number of stranded dolphins, whales, or manatees |
#2 | #1 | X |
|
Number of harassed individuals |
|||
|
Number of individuals taken/killed |
|||
|
Behavior patterns (possibly extrapolated from distribution information) |
|||
| Ecosystem Services | |||
|
Existence value |
|||
|
Wildlife watching |
|||
SOURCES: Wells and Scott, 1990; Hansen et al., 1996; Garrison et al., 2002; Mullin and Fullin, 2004; Wells et al., 2004; Torres et al., 2005; ION, 2012; Department of the Navy, 2012.
NOTES: Examples are provided to illustrate linkages between restoration situations/objectives and appropriate metrics. Example #1 (linkages shown in the table by “#1”) is to reduce illegal feeding of dolphins; Example #2 is to improve coastal habitat; and Example #3 is to control noise from dredging. The “X” symbol indicates metrics that are suggested by the committee as appropriate to sample across multiple projects at a program, region, or Gulf-wide scale.
REFERENCES
Barros, N.B., and D.K. Odell. 1990. Food habits of bottlenose dolphins in the southeastern United States. Pp. 309-328 in The Bottlenose Dolphin. S. Leatherwood and R. Reeves, eds. San Diego, CA: Academic Press.
Bechdel, S.E., M.S. Mazzoil, M.E. Murdoch, E.M. Howells, J.S. Reif, and S.D. McCulloch. 2009. Prevalence and impacts of motorized vessels on bottlenose dolphins (Tursiops truncatus) in the Indian River Lagoon, Florida. Aquatic Mammals 35(3):367-377.
Byrd, B.L., A.A. Hohn, G.N. Lovewell, K.M. Altman, S.G. Barco, A. Friedlaender, C.A. Harms, W.A. McLellan, K.T. Moore, P.E. Rosel, and V.G. Thayer. 2014. Strandings as indicators of marine mammal biodiversity and human interactions off the coast of North Carolina. Fishery Bulletin 112(1):1-23.
Byrd, B.L., A.A. Hohn, F.H. Munden, G.N. Lovewell, and R.E. LoPiccolo. 2008. Effects of commercial fishing regulations on strandings rates of bottlenose dolphins (Tursiops truncatus). Fishery Bulletin 106(1):72-81.
Carmichael, R.H., W.M. Graham, A. Allen, G. Worthy, and S. Howden. 2012. Were multiple stressors a ‘perfect storm’ for northern Gulf of Mexico bottlenose dolphins (Tursiops truncates) in 2011? PLoS One 7:e41155. doi: 10.1371/journal.pone.0041 155.
Department of the Navy. 2012. Final Marine Mammal Monitoring Plan. http://www.nmfs.noaa.gov/pr/pdfs/permits/navy_kitsap_ehw2_marinemammalmonitoring.pdf.
DWH MMIQT (Deepwater Horizon Marine Mammal Injury Quantification Team). 2015. Models and analyses for the quantification of injury to Gulf of Mexico cetaceans from the Deepwater Horizon oil spill. DWH Marine Mammal NRDA Technical Working Group Report. Washington, DC: U.S. Department of the Interior. https://www.doi.gov/deepwaterhorizon/adminrecord.
Garrison, L.P., S.L. Swartz, A. Martinez, C. Burks, and J. Stamate. 2002. A Marine Mammal Assessment Survey of the Southeast U.S. Continental Shelf: February-April 2002. NOAA Technical Memorandum NOAA Fisheries-SEFSC-492. Silver Spring, MD: NOAA.
Fulling, G.L., K.D. Mullin, and C.W. Hubbard. 2003. Abundance and distribution of cetaceans in outer continental shelf waters of the U.S. Gulf of Mexico. Fishery Bulletin 101:923-932.
Hansen, L.J., K.D. Mullin, T.A. Jefferson, and G.P. Scott. 1996. Visual surveys aboard ships and aircraft. Pp. 55-132 in Distribution and Abundance of Marine Mammals in the North-Central and Western Gulf of Mexico: Final report. Volume II: Technical report. OCS Study MMS 96-0027. R.W. Davis and G.S. Fargion, eds. New Orleans, LA: Minerals Management Service, Gulf of Mexico OCS Region.
Hildebrand, J.A., S. Baumann-Pickering, K.E. Frasier, J.S. Trickey, K.P. Merkens, S.M. Wiggins, M.A. McDonald, L.P. Garrison, D. Harris, T.A. Marques, and L. Thomas. 2015. Passive acoustic monitoring of beaked whale densities in the Gulf of Mexico. Scientific Reports 5. doi: 10.1038/srep16343.
Hubard, C.W., K.Maze-Foley, K.D. Mullin, and W.W. Schroeder. 2004. Seasonal abundance and site fidelity of bottlenose dolphins (Tursiops truncatus) in Mississippi Sound. Aquatic Mammals 30(2):299-310.
ION Geophysical. 2012. Marine mammal monitoring and mitigation plan for marine seismic surveys in the Arctic Ocean. Houston, TX: ION Geophysical. http://www.nmfs.noaa.gov/pr/pdfs/permits/ion_2012_monitoring_plan.pdf.
Jochens, A., D. Biggs, K. Benoit-Bird, D. Engelhaupt, J. Gordon, C. Hu, N. Jaquet, M. Johnson, R. Leben, B. Mate, P. Miller, J. Ortega-Ortiz, A. Thode, P. Tyack, and B. Würsig. 2008. Sperm whale seismic study in the Gulf of Mexico: Synthesis report. OCS Study MMS 2008-006. New Orleans, LA: Minerals Management Service, Gulf of Mexico OCS Region.
Laist, D., A. Knowlton, J.G. Mead, A.S. Collett, and M. Podesta. 2001. Collisions between whales and ships. Marine Mammal Science 17(1):35-75
Lane, S.M., C.R. Smith, J. Mitchell, B.C. Balmer, K.P. Barry, T. McDonald, C.S. Mori, P.E.
Rosel, T.K. Rowles, T.R. Speakman, and F.I. Townsend. 2015. Reproductive outcome and survival of common bottlenose dolphins sampled in Barataria Bay, Louisiana, USA, following the Deepwater Horizon oil spill. Proceedings of the Royal Society B 282(1818). doi: 10.1098/rspb.2015.1944.
Litz, J.A., M.A. Baran, S.R. Bowen-Stevens, R.H. Carmichael, K.M. Colegrove, L.P. Garrison, S.E. Fire, E.M. Fougeres, R. Hardy, S. Holmes, W. Jones, B.E. Mase-Guthrie, D.K. Odell, P.E. Rosel, J.T. Saliki, D.K. Shannon, S.F. Shippee, S.M. Smith, E.M. Stratton, M.C. Tumlin, H.R. Whitehead, G.A.J. Worthy, and T.K. Rowles. 2014. Review of historical unusual mortality events (UMEs) in the Gulf of Mexico (1990−2009): Providing context for the multi-year northern Gulf of Mexico cetacean UME declared in 2010. Diseases of Aquatic Organisms 112:161-175. doi: 10.3354/dao 02807.
Martins, M., P.M. Costa, J. Raimundo, C. Vale, A.M. Ferreira, and M.H. Costa. 2012. Impact of remobilized contaminants in Mytilus edulis during dredging operations in a harbour area: Bioaccumulation and biomarker responses. Ecotoxicology and Environmental Safety 85:96-103.
McConnell, B., M. Fedak, S. Hooker, and T. Patterson. 2010. Telemetry. Pp. 222-242 in Marine Mammal Ecology and Conservation: A Handbook of Techniques. I.L. Boyd, W.D. Bowen, and S.J. Iverson, eds. New York: Oxford University Press.
MMC (Marine Mammal Commission). 2011. Mariculture and harbor seals in Drakes Estero, California. Bethesda, MD: MMC. https://www.mmc.gov/wp-content/uploads/drakes_estero_report.pdf.
Mullin, K.D., and G.L. Fullin. 2004. Abundance of cetaceans in the oceanic northern Gulf of Mexico, 1996-2001. Marine Mammal Science 20(4):787-807.
Nowacek, S.M., R.S. Wells, and A.R. Solow. 2001. Short-term effects of boat traffic on bottlenose dolphins, Tursiops truncatus, in Sarasota Bay, Florida. Marine Mammal Science 17(4):673-688.
Pirotta, E., B.E. Laesser, A. Hardaker, N. Riddoch, M. Marcoux, and D. Lusseau. 2013. Dredging displaces bottlenose dolphins from an urbanised foraging patch. Marine Pollution Bulletin 74:396-402.
Rosel, P.E., and L.A. Wilcox. 2014. Genetic evidence reveals a unique lineage of Bryde’s whales in the northern Gulf of Mexico. Endangered Species Research 25:19-34.
Schwacke, L.H., C.R. Smith, F.I. Townsend, R.S. Wells, L.B. Hart, B.C. Balmer, T.K. Collier, S. De Guise, M.M. Fry, L.J. Guillette, Jr., S.V. Lamb, S.M. Lane, W.E. McFee, N.J. Place, M.C. Tumlin, G.M. Ylitalo, E.S. Zolman, and T.K. Rowles. 2014. Health of common bottlenose dolphins (Tursiops truncatus) in Barataria Bay, Louisiana, following the Deepwater Horizon oil spill. Environmental Science and Technology 48:93-103.
Schwacke, L.H., M.J. Twiner, S. De Guise, B.C. Balmer, R.S. Wells, F.I. Townsend, D.C. Rotstein, R.A. Varela, L.J. Hansen, E.S. Zolman, T.R. Spradlin, M. Levin, H. Leibrecht, Z. Wang, and T.K. Rowles. 2010. Eosinophilia and biotoxin exposure in bottlenose dolphins (Tursiops truncatus) from a coastal area impacted by repeated mortality events. Environmental Research 110:548-555.
Sellas, A.B., R.S. Wells, and P.E. Rosel. 2005. Mitochondrial and nuclear DNA analyses reveal fine scale geographic structure in bottlenose dolphins (Tursiops truncatus) in the Gulf of Mexico. Conservation Genetics 6:715-728.
Thomas, P.O., R.R. Reeves, and R.L. Brownell, Jr. 2015. Status of the world’s baleen whales. Publications, Agencies and Staff of the U.S. Department of Commerce 544. http://digitalcommons.unl.edu/usdeptcommercepub/544.
Torres, L.G., W.A. McLellan, E.Meagher, and D.A. Pabst. 2005. Seasonal distribution and relative abundance of bottlenose dolphins, Tursiops truncatus, along the U.S. mid-Atlantic coast. Journal of Cetacean Research and Management 7(2):153-161.
Van Dolah, F.M. 2000. Marine algal toxins: Origins, health effects, and their increased occurrence. Environmental Health Perspectives 108(Supplement 1):133-141.
Venn-Watson, S., L. Garrison, J. Litz, E. Fougeres, B. Mase, G. Rappucci, E. Stratton, R. Carmichael, D. Odell, D. Shannon, S. Shippee, S. Smith, L. Staggs, M. Tumlin, H. Whitehead, and T. Rowles. 2015. Demographic clusters identified within the northern Gulf of Mexico common bottlenose dolphin (Tursiops truncates) Unusual Mortality Event January 2010-June 2013. PLoS One 10(2):e0117248. doi: 10.1371/journal.pone.0117248.
Vollmer, N.L., and P.E. Rosel. 2013. A review of common bottlenose dolphins (Tursiops truncatus) in the northern Gulf of Mexico: Population biology, potential threats, and management. Southeastern Naturalist 12(monograph 6):1-43
Waring, G.T., E. Josephson, K. Maze-Foley, and P.E. Rosel. 2015. U.S. Atlantic and Gulf of Mexico marine mammal stock assessments - 2014. Woods Hole, MA: U.S. Department of Commerce, National Oceanic and Atmospheric Administration, National Marine Fisheries Service, Northeast Fisheries Science Center. http://www.nmfs.noaa.gov/pr/pdfs/sars/ao2011.pdf.
Wells, R.S., and M.D. Scott. 1990. Estimating bottlenose dolphin population parameters from individual identification and capture-release techniques. Report of the International Whaling Commission, Special Issue 12:407-415.
Wells, R.S., H.L. Rhinehart, L.J. Hansen, J.C. Sweeney, F.L. Townsend, R. Stone, D. Casper, M. Scott, A. Hohn, and T.K. Rowles. 2004. Bottlenose dolphins as ecosystem sentinels: Developing a health monitoring system. EcoHealth 1:246-254.
Wilson, B., H. Arnold, G. Bearzi, C.M. Fortuna, R. Gaspar, S. Ingram, C. Liret, S. Pribanic, A.J. Read, V. Ridoux, K. Schneider, K.W. Urian, R.S. Wells, C. Wood, P.M. Thompson, and P.S. Hammond. 1999. Epidermal disease in bottlenose dolphins: Impacts of natural and anthropogenic factors. Proceedings of the Royal Society of London B 256:1077-1083.
This page intentionally left blank.










