2
The Strategic National Stockpile: Origin, Policy Foundations, and Federal Context
HISTORICAL AND CURRENT PERSPECTIVES
Burel provided historical context for the stockpile with reference to several landmark events in the history of the enterprise. In 2000, the first formulary was developed and it was configured as the initial “push-package,” that is, repacked and configured materiel assembled for rapid deployment to resupply overwhelmed local medical supplies. This was built around the understanding that in most cases, public health professionals at the state or local level would be able to break down the packages and use them rapidly.1 Push packages were loaded into specialized air cargo containers for rapid loading and air delivery, and were prepositioned in multiple secure facilities near major transportation hubs. The system was designed to be able to reach any point in the United States or its territories within 12 hours from a decision to deploy.
Burel explained that the first real deployments were during the events of September 11, 2001, and the Anthrax incident in 2001. Both deployments consisted of 12-hour push packages with appropriate equipment and medical countermeasures (MCMs); the Strategic National Stockpile (SNS) also provided support via technical assistance staff. After those events, the scope of the SNS began to widen and has continued to grow, according to Burel. The program was codified in the Public Health Service Act in 2002, which increased SNS funding to expand its capabilities. Push-package capability
___________________
1 One of the subsequent evolutions at the state level is that professional logistics firms are contracted to receive materiel and further deploy it into the state.
was expanded from 8 different types to 12 and the managed inventory of products expanded, currently sitting at more than $7 billion.2 The Division of Strategic National Stockpile’s (DSNS’s) role in the development of federal medical stations and the Cities Readiness Initiative (see Box 2-1) began in 2004, and the SNS also provided supplemental funds to stockpile pandemic influenza MCMs and personal protective equipment (PPE).
Significant Events in SNS History
Burel explained that in 2009, the H1N1 influenza pandemic triggered the largest deployment in SNS history when 12.5 million antiviral regi-
___________________
2 This includes materiel purchased by the SNS as well as materiel developed and acquired by the Biomedical Advanced Research and Development Authority (BARDA).
mens were deployed across the country (a further 300,000 were deployed internationally), as well as 19.6 million pieces of PPE, 85.1 million N95 respirators, and 2,129 regimens of Peramivir IV (the latter were deployed in conjunction with BARDA). Burel said the experience exposed the need to better integrate the operations of the SNS during an emergency with the functions of the day-to-day medical supply chain, which requires educating state and local partners about what the supply chain does and how it operates.
The 2014 outbreak of Ebola virus disease (EVD) was an emerging infectious disease (EID) event that did not fall within the original scope of the SNS, explained Burel. However, the experience revealed that the DSNS has a great reach into the commercial supply chain for many of the products required to deal with this type of threat. For instance, DSNS was asked to supply PPE for hospitals and agreed to do so, given that they would also be useful for other EIDs. They developed a tiered approach to evaluating and treating individuals for EVD, which helped hospitals to calculate needs based on the numbers of patients they would be able to treat. Even more important than acquiring materiel, Burel suggested, was the experience of supply chain and multiagency government partners working together to create a “whole government” response to a real-world incident.
Burel explained that shortages trigger what is called “allocation,” the details of which are not publicly available, but one of the determinants of how items are allocated is based on whether there is a contract with the provider, how big the contractor is, and how big the buyer is. SNS worked with supply-chain partners to find a better way to allocate scarce products during a specific event without entirely depleting the stocks for cases of immediate short-term need. Burel reflected that the expanding scope of the SNS has had a positive effect in terms of being able to work with the commercial supply chain to help in these types of allocation situations, and to provide short-term assistance as needed before the regular commercial supply chain takes back over.
Observations on the Current State of the SNS
Based on his experiences, Burel observed that none of the historic SNS responses have yet matched pre-event planning or expectations, but planning has built in the flexibility to address the unexpected and has allowed for the most effective use of available resources, material, and commercial marketplace capabilities. Coordination with other federal buyers allows agencies to work together with a coordinated government approach to obtain items that are needed without disrupting the entire supply chain.
Furthermore, Burel noted that the SNS expansion into all-hazards response (e.g., hurricanes, earthquakes, EIDs) has drained some funds that
were built around a CBRN threat. As an example, he cited federal medical stations which are an important resource, but are expensive to maintain. However, he described the SNS as a good value, with costs of less than 2 cents on the dollar for all of the products they manage, inventory accuracy of almost 100 percent, and full confidence in the safety and efficacy of products in the stockpile.
Better integration with the commercial supply chain has been fostered by working with the commercial sector to better understand its needs, noted Burel. The DSNS continues to try to better leverage the strengths of the commercial market by, for example, using commercial and third-party logistics partners for management, storage, and transportation.
SNS POLICY FOUNDATIONS
Susan E. Sherman, senior attorney, Office of the General Counsel, Department of Health and Human Services (HHS) provided a brief legal history of the SNS’s statutory foundations. The statutory origin of the SNS is in the Public Health Service Act, which authorized CDC and other components of the U.S. Public Health Service at HHS with broad authorities to assist states and localities to control communicable disease. Beginning in 1998, Congress began providing funding in annual appropriations to CDC for pharmaceutical stockpiling. Sherman explained that Congress can instruct agencies by means of enabling statutes to run a program or carry out a task, with the award of appropriations being tied to conditions. In the case of the SNS, she noted, money came before an enabling statute: CDC was running the program based on the annual appropriations that it is still receiving.
The first enabling statute for the SNS was the Public Health Security and Bioterrorism Preparedness Act of 2002.3 It directs the Secretary of HHS to maintain a “Strategic National Stockpile,” whose statutory mission is to provide for the emergency health security of the United States, including the emergency health security of children and other vulnerable populations, in the event of a bioterrorist attack or other public health emergency. The SNS at that point was explicitly coordinated with the Department of Veterans Affairs (VA). The Act also specifically directs the SNS to procure smallpox vaccines and potassium iodide as part of the stockpile and includes provisions for stockpile management and security requirements; it also authorized annual appropriations. Sherman explained that the way the statute is written provides broad discretion to HHS and the Secretary; public health officials are the ones who decide what is needed, what constitutes a public health emergency, and how to protect the emergency health security of the
___________________
3 Strategic National Stockpile, Public Health Service (PHS) Act § 319F-2(a), 42 U.S.C. § 247d-6b(a).
United States. The language is not particularly prescriptive, but does provide guidance and a standard. Shortly thereafter, the Homeland Security Act of 2002 transferred responsibility for the SNS from HHS to the Department of Homeland Security (DHS), to be carried out in coordination with the Secretaries of HHS and VA; other requirements remained the same.
Sherman continued that under the Project BioShield Act (2004),4 responsibility for the SNS was transferred back to the Secretary of HHS to be maintained in coordination with the Secretary of DHS. The VA coordination provision was removed, and provisions for deployment authorities were added: as required by the Secretary of Homeland Security to respond to an actual or potential emergency; and at the discretion of the Secretary to respond to an actual or potential public health emergency or other situation in which deployment is necessary to protect the public health or safety. She pointed out a common misconception that the Secretary of HHS must declare a formal health emergency to deploy the stockpile; however, this is not actually required. The SNS can respond when it determines that circumstances meet the standards written in the statute. The Project BioShield Act also enacted programs related to the stockpile MCM initiative. It authorized procurement of security countermeasures against CBRN threats to be included in the SNS, and established a “Special Reserve Fund,” of $5.6 billion (available between 2004 and 2013), which was initially housed at DHS but later transferred to HHS. She noted that a process in that statute is fairly prescriptive about what determinations need to be made by DHS and HHS in order to spend the Special Reserve Fund to buy these MCMs. She explained that exceptional authorities are also included because they are considered necessary for making it easier and faster to procure products for the stockpile. The Project BioShield Act is also when the Emergency Use Authorization (EUA) was enacted for civilian purposes. Furthermore, it expands the authority for the Food and Drug Administration (FDA) to authorize emergency use5 of unapproved products or approved products for unapproved uses.
Sherman explained that the Public Readiness and Emergency Preparedness (PREP) Act of 20056 authorizes the HHS Secretary to issue a declaration to provide liability immunity (except for willful misconduct)7:
- to manufacturers, distributors, states, local governments, tribes, and others who supervise or administer countermeasures pro-
___________________
4 PHS Act § 319F-2(c), 42 U.S.C. § 247d-6b(c).
5 Emergency Use Authorization, other emergency authorities, Federal Food, Drug, and Cosmetic (FD&C) Act §§ 564 and 564A, 21 U.S.C. §§ 360bbb-3, 360bbb-3a.
6 PREP Act, PHS Act §§ 319F-3, 319F-4, 42 U.S.C. §§ 247d-6d, 247d-6e.
7 The declaration triggers emergency funds for injury compensation for serious physical injuries or deaths directly caused by administration or use of covered countermeasures.
-
grams and to licensed health professionals and others who prescribe, administer, or dispense countermeasures;
- for claims causally related to development, distribution, administration and use of FDA-approved, licensed, or authorized countermeasures against pandemic or epidemic disease, or CBRN agents as necessary to protect public health.
The PREP Act covers a very specific category of products, which are not limited to stockpile products. The PREP Act Declarations can cover any activity involving the distribution and dispensing of MCMs. Similarly, she noted that the EUA is not limited to MCMs.
Sherman next described the Pandemic and All-Hazards Preparedness Reauthorization Act (PAHPRA) of 2006, which established that the stockpile should be maintained by the Secretary of HHS in collaboration with the CDC director and added the SNS annual review requirement. This was when BARDA was established to fill the gap in advanced research and development. This Act enhances BARDA’s authority to procure CBRN countermeasures (e.g., milestone payments, warm-based surge capacity). The statute also authorizes the Assistant Secretary for Preparedness and Response (ASPR) to “exercise the responsibilities and authorities of the Secretary [of HHS] with respect to the coordination of” the stockpile and to oversee advanced research and development of MCMs carried out by BARDA. This, she noted, sets up the relationship between CDC, ASPR, and several of the other agencies from a statutory perspective.
The 2013 PAHPRA, Sherman explained, was the point at which the PHEMCE Strategy and Implementation Plan (SIP) was codified into law. It requires ASPR to submit this plan and the associated multiyear MCM budget to Congress. Another clause requires stockpile contents to be determined consistent with ASPR authority, with an explicit provision for an annual report to Congress, and another provision addressing the depletion and replacement of the current contents. It further enhances CBRN procurement and advanced research and development authorities, and reauthorizes the Special Reserve Fund. Sherman pointed to one of PAHPRA’s biggest effects, the streamlining of the EUA process, which provides new expanded authorities to FDA to authorize emergency use of approved products in emergencies and products held for emergency use, allowing for easier deployment.
CONGRESS AND THE SNS
Frank Gottron, specialist in science and technology policy at the Congressional Research Service, examined the relationship between Congress and the SNS. It began in 1998 with the Consolidated Appropriations
Act,8 which, in response to an emergency budget supplement request by President Clinton, provided $51 million for pharmaceutical and vaccine stockpiling activities at CDC. To provide more direction to the statute, in 2002 Congress passed the Public Health Security and Bioterrorism Preparedness and Response Act9 to emphasize consultation with a working group to coordinate stakeholders and establish the HHS Secretary’s role in ensuring appropriate inventory, accounting, and security of the stockpile. It was further aimed at ensuring consultation with federal, state, and local official partners with respect to special events and special needs, as well as periodic review and revision of the contents of the stockpile.
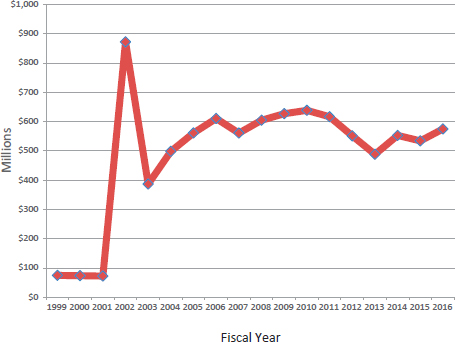
Congress has been broadly supportive of the program, according to Gottron, roughly appropriating the amounts the Executive Branch has requested over the years and funding the SNS with between $500 million and $625 million per year since 2004 (see Figure 2-1).
Gottron explained how Congress’s significant investment in the program has given rise to significant policy concerns since its inception. In a Senate Appropriations Report from 1999, they asked the department to articulate a clear and coherent biosecurity strategy to the public and to Congress that is rooted in both national security needs and scientific opportunities, as well as requesting a long-term strategic plan (including 5 years of funding requirements) for the National Pharmaceutical Stockpile based on the results of a documented national-level threat and risk assessment. This required estimating the cost and time needed to acquire and establish proposed MCMs and identifying the long-term costs and benefits of establishing and sustaining the production and inventory infrastructure for the stockpile and management of its inventory (including stock replacement and rotation).
The same questions raised by Congress in 1999 still persist, noted Gottron. Progress has been made to address them, but they are still ongoing concerns. Many of those issues were incorporated into the 2013 PAHPRA.
Transparency in the planning and operation of the SNS is a priority for Congress, according to Gottron. PAHPRA addressed certain transparency requirements relating to the annual revision of the stockpile. Additional reporting requirements written into the statute to improve transparency include integration with annual PHEMCE SIPs as well as the 5-year coordinated biodefense budget. More recently, Congress requested additional reports on certain supplies within the stockpile, and for future budgets to include additional information about planning for replacement and acquisition of new MCMs, for example. PAHPRA also includes additional in-statute requirements regarding coordination with other federal agencies
___________________
8 Omnibus Consolidated and Emergency Appropriations Act, 1999 (P.L. No. 105-277).
9 See https://www.gpo.gov/fdsys/pkg/BILLS-107hr3448enr/pdf/BILLS-107hr3448enr.pdf (accessed June 6, 2016).
SOURCE: Gottron presentation, February 4, 2016.
and nonfederal partners, although there are ongoing concerns about the process and whether the outreach has been sufficient. In 2015, Gottron reported that Congress called for CDC to update guidance to state and local public health officials regarding new acquisitions to the SNS and how they would be used.
Gottron noted that Congress, on certain occasions, has also been interested in the inclusion of specific items in the stockpile; for example, the originating legislation specifically calls out the inclusion of potassium iodide and the smallpox vaccine. In the intervening years there have been strong suggestions to consider including psychotropic medications, various antiviral medications, and specific medical equipment such as face masks. The protection of pediatric populations is also an ongoing concern being taking into consideration.
Also ongoing are concerns about item expiration and replacement costs; Gottron commented that Congress is particularly interested in the repurposing of soon-to-expire medications (e.g., efforts to allow DoD to use the expiring anthrax vaccine in their vaccination program; proposals to allow first-responders access to the anthrax vaccine on a voluntary basis).
SNS AND THE FEDERAL MEDICAL COUNTERMEASURES ENTERPRISE: FEDERAL PROGRAM AND AGENCY PARTNERSHIPS
Sherman explained that the HHS Secretary maintains the stockpile in collaboration with CDC, in coordination with DHS, and consistent with ASPR authorities. He or she deploys the stockpile to respond to an actual or potential public health emergency or other situations in which deployment is necessary to protect the public health or safety, and issues declarations needed for liability protections and emergency use of countermeasures. The ASPR exercises the authorities of the HHS Secretary to coordinate the stockpile, manages the PHEMCE and multiyear budget, and oversees Project BioShield procurements and BARDA advanced research and development. CDC manages and operates the stockpile and deploys products for specific individual conditions. It is also responsible for testing and evaluation of government programs, sharing with other federal agencies, prepositioning, and other preparedness activities. FDA conducts regulatory oversight of products and authorizes emergency use.
The Public Health Emergency Medical Countermeasures Enterprise
Phillips provided an overview of PHEMCE. Established in 2006 and led by the Office of the ASPR, it provides direction and jointly partners with the SNS. It is tasked with strategic planning surrounding MCM prioritization and support for developing, procuring, and planning for the effective use of MCMs against CBRN threats and EIDs, including influenza. This effort involves a wide range of partnerships (BARDA, CDC, FDA, National Institutes of Health [NIH], DoD, DHS, VA, and the Department of Agriculture; see Figure 2-2).
Phillips commented that these partnerships have been fruitful in establishing new capabilities10 as well as delivering 12 new MCMs to the stockpile, building a more robust interagency governance structure, and conducting 10 major portfolio reviews of MCM development by threat. Two major studies by the Government Accountability Office have ensured the integrity of the process from beginning to end. She highlighted the SNS’s integration as a core part of PHEMCE. ASPR and CDC have coordinated and integrated with respect to state and local grantees, as well as public
___________________
10 FDA Regulatory Science Initiatives, the BARDA Centers for Innovation in Advanced Development & Manufacturing, National Institute of Allergy and Infectious Diseases’ Concept Acceleration Program; the HHS Five Year Budget Overlook for the Medical Countermeasures Program; the annual review of the National Stockpile, and the National and International Portfolio Tracking Tool for CBRN and MCM products.
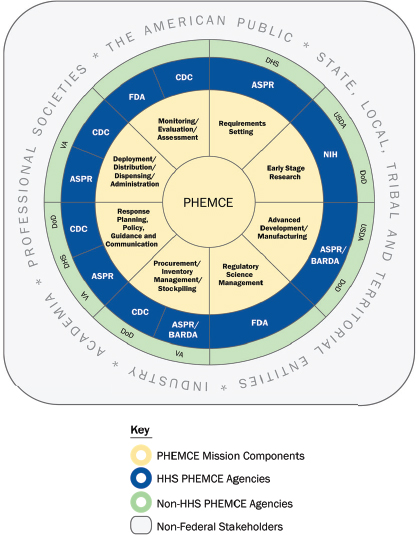
NOTE: ASPR = Office of the Assistant Secretary for Preparedness and Response; BARDA = Biomedical Advanced Research and Development Authority; CDC = Centers for Disease Control and Prevention; DHS = Department of Homeland Security; DoD = Department of Defense; FDA = Food and Drug Administration; HHS = Department of Health and Human Services; NIH = National Institutes of Health; PHEMCE = Public Health Emergency Medical Countermeasures Enterprise; USDA = U.S. Department of Agriculture; VA = Department of Veterans Affairs.
SOURCE: Disbrow presentation, February 4, 2016.
health emergency preparedness programs and health care communities (through the grant programs).
Phillips explained that the 2012 PHEMCE SIP examined response planning as a strategic goal for PHEMCE. There are ongoing joint efforts to engage with state and local agencies (as well as other stakeholders) and to evaluate the risk posed by emerging threats to public health, with PHEMCE providing structure and institutional support through a working group co-led by DHS and CDC. For those EIDs determined to require MCM intervention, resources from across PHEMCE partner agencies are leveraged for requirement setting, research and development, procurement, and/or possible utilization levels. Partnerships are focusing on the SNS’s role in developing enhanced diagnostics. The network of partnerships also allows interagency partners to come together on major emerging issues when convened for other purposes.
Philips outlined some of the challenges faced by PHEMCE, including dealing with competing demands as to what the SNS is and what it should be, as well as the constantly evolving issues that are intrinsic to the entire MCM enterprise (pandemics, new EIDs, CBRN threats, and its BioShield underpinnings). Addressing these challenges, she noted, requires a balancing act between the original intent of the stockpile and these new concerns. Doing so requires multiple levels of assessment and prioritizing to inform investments and critical decisions. She further maintained that the PHEMCE model provides an opportunity to broadcast a continuous, loud, and clear message about what is being done, the progress being made, and strategies being implemented moving forward. Box 2-2 explains the public expectations around the SNS.
PHEMCE and the Strategic National Stockpile Annual Review
An overview of PHEMCE’s role in the SNS annual review, and how the review process fits into the requirements process and informs preparedness, was provided by Richard Jaffe, director, Division of Medical Countermeasures Strategy and Requirements, Office of Policy and Planning, ASPR, HHS. The SNS annual review is mandated by the Public Health Services Act (as amended by the PAHPRA in 2013)11 and the Homeland Security Presidential Directive 21.12 This is important from an interagency perspective for enabling agreed-upon formulary policy recommendations and a defensible budget, with responsibility spread over multiple agencies.
Jaffe noted that the review also provides for both top-down and bottom-up analyses of the makeup of threat areas, and allows for the use of integrated program teams composed of subject-matter experts to set requirements and analyze threats (with senior-level review) to make recommendations on increasing preparedness across the SNS formulary.
Jaffe explained that the review is developed via partnership between the ASPR and CDC to ensure that policies are not disconnected from the available assets at present. The ASPR leads and coordinates departmental policy development and implementation on SNS-related policy, ensures documentation of plans and procedures that include SNS assets, and oversees the Enterprise Senior Council (ESC), with participation of the CDC director, to advise the HHS Secretary regarding strategic priorities for the SNS. He clarified that the ASPR generally leaves policy planning to the oversight of the ESC (the senior level of the PHEMCE), where they make major policy decisions and recommendations for the SNS assets. CDC participates in ASPR-led policy and planning activities, coordinates the end-to-end management of the SNS,13 and coordinates with state, local, tribal, and territorial (SLTT) public health officials on plans and exercises for SNS-provided assets.
Jaffe described how the ASPR and CDC co-lead the SNS annual review process, which involves five phases14 to prioritize formulary gaps for the SNS
___________________
11 “Secretary shall conduct an annual review (taking into account at-risk individuals) of the contents of the stockpile, including nonpharmaceutical supplies, and make necessary additions or modifications to the contents based on such review and shall submit such review annually to the appropriate congressional committees of jurisdiction to the extent that disclosure of such information does not compromise national security.”
12 The SNS annual review is to be delivered to the director of the Office of Management and Budget (OMB) and assistant to the President for Homeland Security and Counterterrorism at the time of HHS’s budget submission.
13 Budget proposals, procurements as directed by the ESC and the HHS Secretary, management of SNS facilities, and disposal of expired products.
14 Phase I: DSNS Review of SNS Contents; Phase II: IPT Gap Analysis and Threat Specific Recommendations; Phase III: Enterprise Executive Committee (EEC) Cross-Threat Prioritization; Phase IV: ESC Deliberation and Finalization; Phase V: Report Development and Approval.
and to ensure that the best portfolio of stockpiled products are included, given budget constraints, to optimize the HHS’s capability. The process relies heavily on input from threat-specific subject-matter experts and senior leaders to inform program prioritization and budget formulation. This is coordinated with a PHEMCE multiyear budget that is aligned with the SIP. In the first phase, the SNS performs a content review and a business process review to find ways to improve the process. The second phase is a deep dive into the threat-specific areas to see if there are any “game changers” in threats or technologies. The third phase is cross-threat prioritization, looking within and across the Integrated Program Teams (IPTs)15 to see how MCMs could be leveraged (i.e., if there are different ways MCMs can be used for one threat versus another) to obviate the need for threat-specific countermeasures. The fourth phase incorporates senior-level input; Jaffe noted that this requires clear communication in explaining these complex issues to senior leadership outside of the domain. The final phase is writing the report to Congress, obtaining executive clearance, and delivering the report to the Office of Management and Budget (OMB) and Congress.
Jaffe explained that the goal of the assessments themselves is to continue to improve estimates of what is needed in the stockpile and what can be effectively used, driving toward achieving a process that effectively balances the current status and the preparedness goals. To that end, he outlined five parameters for inclusion of MCMs in the SNS as a high-level overview for workshop participants:
- Can we develop it (research and development)?
- Can we make it (manufacturing)?
- Can we stockpile it and procure it (purchasing)?
- Can we plan on how we are going to use it (response planning) and how we use the operational capacity?
- Can we use everything that we say we are going to use?
Turning to the requirements process, Jaffe explained that there is a three-tiered process to identify what the threat is, how to mitigate that threat, and how much it needs to be mitigated. He clarified that the aims are not to stock more MCMs than are needed to mitigate threats and determining what the product looks like in a stockpiling goal. At that point,
___________________
15 “The IPTs provide an end-to-end vision of MCMs against a particular threat type (e.g., anthrax, smallpox) or capability (e.g., diagnostics) that ranges from requirements setting (i.e., stockpiling targets and product characteristics) through to stockpiling, delivery and dispensing, and monitoring and evaluating MCM effectiveness. The IPTs develop strategies for addressing key cross-cutting issues, in consideration of available programmatic resources at the federal and SLTT levels. IPTs serve as subject-matter expert communities of practice for interagency vetting and input on issues within their purview. They report to the EEC” (http://www.phe.gov/Preparedness/mcm/phemce/Pages/governance.aspx [accessed April 15, 2016]).
PHEMCE members work together with the partners at BARDA to develop acquisition strategies and other downstream components.
Jaffe commented that the preparedness goals to “develop, make, stockpile, plan, and use” track very well with how much is needed, how much is being used, and what the product would look like in a stockpiling goal for requirements. All of these goals are informed by the SNS, tracked, and aligned to create the best stockpile with the best product to mitigate threats.
The Biomedical Advanced Research and Development Authority
Gary Disbrow, director, CBRN Division, ASPR/BARDA, explained that the mission of BARDA is to support advanced development and availability of countermeasures for CBRN threats, pandemic influenza, and EIDs through product development, innovation, acquisition, stockpiling, building manufacturing infrastructure, and core service assistance.
Disbrow explained that BARDA’s role within PHEMCE is, in essence, to act as a virtual pharmaceutical company. In addition to providing contractual support for the development of MCMs, BARDA also is responsible for the initial acquisition and purchasing of products, stockpiling them, and working in collaboration with the DSNS to place them in the SNS or other venues. BARDA also builds manufacturing infrastructure and provides core service assistance to certain companies.
Disbrow highlighted two methods for procurement and stockpiling. The first method is for the DSNS to use its fund to purchase licensed products that were not developed by PHEMCE partners. The second is for BARDA to support16 successful products (those that meet scientific milestones for safety, efficacy, and control of manufacturing) through late-stage development, procurement, and maintenance when products are under potential EUA. Products are also supported through approval/licensure under Project BioShield (which is a different funding stream). Once a product is approved or licensed, BARDA works with them to fulfill postmarketing commitments and requirements.17
In addition to contractual support, BARDA has established a National Countermeasure Response Infrastructure, which was presented by Disbrow (see Figure 2-3).
___________________
16 BARDA’s advanced research and development contracts do not permit them to actually buy products, only to support them.
17 Products supported by Project BioShield have included Anthrax Immune Globulin (AIG), Anthrax Vaccine Adsorbed general use prophylaxis (AVA GUP), heptavalent Botulinum Antitoxin Therapeutic (hBAT), Neulasta, Neupogen, and Raxibacumab. Approved/licensed products supported under PI (pandemic influenza) funding include Peramivir, 5N1 (one) and H1N1 (six) vaccine candidates, cell-based vaccine and recombinant vaccine, and diagnostics (five).
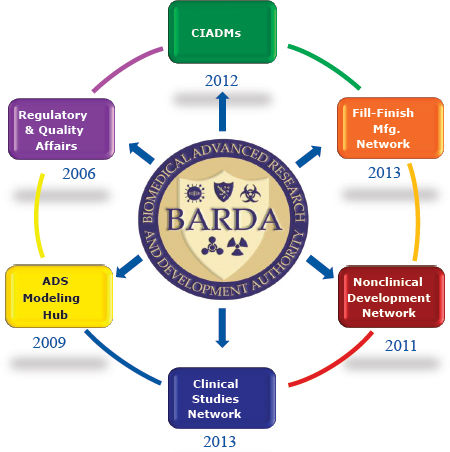
SOURCE: Disbrow presentation, February 4, 2016.
Disbrow explained that there are three Centers for Innovation in Advanced Development & Manufacturing (CIADMs) in the United States that can help developers to manufacture their products and provide other core services. The Fill-Finish Manufacturing Network has four centers located throughout the United States that can assist companies that have manufacturing capacity but not fill-finish capacity. The Nonclinical Development Network can develop animal models that can stand up to regulatory rigor; most of these products are licensed or approved under the
Animal Rule.18 This network can also evaluate products on the commercial market for suitability for another purpose in treating threats that should be addressed. The Clinical Studies Network (four sites located throughout the United States) can perform clinical studies for products. This infrastructure also includes a modeling hub as well as regulatory and quality affairs.
Disbrow explained that these infrastructure capabilities were actually activated during the EVD response. For the EVD therapeutic, a regulatory and quality affairs group helped the company write some sections of their investigational new drug (IND) application so they could submit it more quickly. The Fill-Finish and Manufacturing Network actually took over and was eventually responsible for shipping the finished product to West Africa. The Nonclinical Development Network was used to evaluate other EVD therapeutics; the CIADMs were actually leveraged to manufacture monoclonal antibodies that could address the EVD outbreak through a partnership with Genentech Inc.
Disbrow noted that as the value of the SNS formulary has increased over the past several years, the range of products supported under Project BioShield that have been (or will soon be) added to the SNS include products for smallpox, botulism, anthrax,19 chemical injury, and radiation and burn injury from nuclear detonation. During the discussion, Skip Skivington, vice president of operations, Kaiser Permanente Finance Operations, asked Burel about the cause of the rapid increase in inventory cost from $6 billion to more than $7 billion. Burel attributed the valuation increase to the inclusion of additional products in the SNS, due in large part to BARDA’s advanced development efforts. But he commented that while the products delivered by BARDA are of high quality, many have very high costs associated with them. Thus, acquiring even a few of those products drives the valuation very high. He noted also that those products will have to be continually procured and replaced by the DSNS or BARDA’s great work and great investment will have been squandered. Burel commended BARDA for its role in developing new products and expanding capabilities for the SNS, but warned that plans will need to be in place to maintain that level of contribution going forward.
According to Disbrow, BARDA provides a range of stockpiling options for the SNS. They can deliver CBRN countermeasures and pandemic influenza products to the SNS if there is not a commercial market. Vendor-managed inventory can be used when there is a commercial indication and
___________________
18 See http://www.fda.gov/downloads/drugs/guidancecomplianceregulatoryinformation/guidances/ucm399217.pdf (accessed June 3, 2016).
19 The anthrax vaccine has completely transitioned to the SNS. Disbrow commented that he is aware that costs are being transferred to the SNS (personnel, inventory storage, etc.) even without purchasing or production costs.
market that can be leveraged to decrease life-cycle management costs.20 In such cases the product is stored at the vendor in a cage that is owned by the U.S. government. As it nears expiration, it rotates into the commercial market and then new product comes into that cage. This means that the product is available to the U.S. government at any time; however, this method for managing and storing pharmaceuticals is not possible for all MCMs.
BARDA also manages pre-pandemic stockpiles of bulk vaccine, adjuvant, plasma, or bulk intermediates for CBRN threats, according to Disbrow. Although bulk intermediates have a longer shelf life, a several-month-long lead time may be necessary to actually take that bulk and manufacture it into final drug product. Furthermore, if the product is fill-finished but does not meet release specifications, then the product is unusable. BARDA also makes investments in next-generation products to potentially decrease life-cycle management costs, for example, products in lyophilized forms to extend the shelf life from 3 to 10 years. Disbrow explained that such investments always require a return on investment (ROI) analysis: there is very clear justification to invest going from 3 to 10 years, but less so from 5 to 6 years, or going from an IV administration to a pill. He estimated that such investments might require several hundred million dollars over a much longer period of time. During the discussion, Sheldon Jacobson, professor and director, Simulation and Optimization Laboratory, Department of Computer Science, University of Illinois at Urbana-Champaign, asked for clarification about how ROI is measured for MCMs that are never actually used. Disbrow explained that this is determined by how quickly the investment will be recouped to develop the second-generation MCM (life-cycle cost includes the development and then the out-year cost for replenishment); such investments require savings over time.
Disbrow outlined numerous ways that BARDA interacts with CDC and DSNS, arguing that they are not in fact siloed (as Skivington had suggested). For instance, Flu Risk Management Meetings involve high-level PHEMCE partners. BARDA must brief both the Enterprise Executive Committee and the Enterprise Senior Council before making acquisitions for Project BioShield, meaning that all PHEMCE partners are informed. Integrated program teams are PHEMCE-wide teams of subject-matter experts that make decisions on requirements for different types of products. DSNS and CDC personnel participate in BARDA-established technical evaluation panels for proposals and in-process reviews, where decisions are made about moving forward with a contractor on the basis of scientific merit. Project coordination teams manage the day-to-day oversight of individual
___________________
20 Neupogen and Leukine are products that were previously approved to treat cancer patients undergoing myelosuppressive therapy. Neupogen has now been approved for the acute radiation syndrome indication. These products are out in the commercial market.
contracts. The multiyear budget allows NIH to inform BARDA about potential product transfers and transitions over a 5-year period; BARDA also informs DSNS about potential product transfers to the stockpile over the same period, according to Disbrow. The SNS annual review informs the review committee about when potential products will be ready for procurement and stockpiling, and BARDA and the SNS work together to support submission of pre-EUA packages, among numerous other venues of collaboration.
Finally, Disbrow explained that as products become approved and licensed, the DSNS will be responsible for their replenishment and maintenance. BARDA/PHEMCE partners have achieved approval for 19 products, with additional approvals expected in the coming fiscal years. However, Disbrow cautioned that the SNS budget will need to increase as the role of maintenance transitions to the SNS.
The Food and Drug Administration and the Federal Medical Countermeasures Enterprise
The role of FDA in the federal MCM enterprise was described by Brad Leissa, deputy director and emergency coordinator, Counter-Terrorism and Emergency Coordination Staff, Center for Drug Evaluation and Research. He explained that FDA’s overarching MCM objective is to facilitate the development of and access to safe and effective MCMs (i.e., drugs, biologics, and devices, including diagnostics and PPE to counter high-priority CBRN and EID threats).
Leissa explained that FDA has numerous MCM roles throughout products’ life cycles.21 FDA engages with product sponsors throughout the MCM development process and collaborates with government partners (e.g., PHEMCE, state and/or local governments) as needed. It is involved in approving, licensing, clearing, and regulating MCMs with the Animal Rule as the authority for when safety still needs to be assessed in both animals and humans.22 FDA has legal mechanisms to prepare for and facilitate the emergency use of MCMs (e.g., EUA, IND application or investigational device exemption [IDE], and other emergency-use authorities) and associated import/export laws and liability protections (PREP Act). FDA supports MCM product development via scientific research. The Medical Countermeasures Initiatives (MCMi) can support anticipating, monitoring for, and managing potential MCM shortages.
___________________
21 MCMi Annual Report: http://www.fda.gov/EmergencyPreparedness/Counterterrorism/MedicalCountermeasures/AboutMCMi/ucm270744.htm (accessed April 15, 2016).
22 The principal efficacy determination is made through animals with the extrapolation of pharmacokinetics and pharmacodynamic endpoints to humans.
FDA works closely with the DSNS regarding potential MCM shortages, according to Leissa. The DSNS will occasionally contact FDA to seek advice about whether the size and magnitude of a procurement being considered will affect general use of the product in the community. If supported by science, FDA has the ability to extend drug expiration dates through the shelf-life extension program. FDA monitors MCM use for adverse events (e.g., through MedWatch and the Vaccine Adverse Event Reporting System) and ensures consumer protection against fraudulent claims, enforcing against misbranded and adulterated products.23 FDA addresses evolving challenges with respect to emerging threats during an event through intraevent MCM surveillance that evaluates both the safety and the operational effectiveness of the product.
Leissa explained that use of investigational new drugs is relatively straightforward for approved products, unless they have an indication for which they are not approved. When a product is in the SNS it can be used under IND application.
Emergency Use Authorization and Legal Mechanisms
EUA opened the door for mass preparedness planning, noted Leissa. Pre-EUA is a mechanism by which FDA communicates with drug developers or with other agencies in the government about a product that is not yet at the stage of EUA and comes to an agreement about what is needed. An example of an EUA from 2009 is Peramivir IV; CDC then was on the operational side of distributing Peramivir to requesting physicians who agreed to the conditions of the EUA. A summary of EUAs issued between 2005 and 2015 is listed in Table 2-1.
Emergency-use legal mechanisms allow expanded access to investigational drugs and devices; Leissa explained that in certain circumstances, clinical trials under an IND/IDE may be the most ethical and fairest means to provide access, given limited supplies and need to assess products.24 EUA25 was established by the Project BioShield Act (2004) and amended by PAHPRA in 2013.26
Leissa described how under EUA authority,27 FDA can authorize for use in CBRN emergencies the unapproved MCMs (despite lacking the amount of data necessary for approval) and the unapproved use of approved MCMs
___________________
23 Six Warning Letters were issued in response to Ebola claims, including three in September 2014. See http://www.cnn.com/2014/09/26/health/ebola-fake-drugs (accessed June 6, 2016).
24 IND application (21 CFR §§ 312.300-312.320), IDE (21 CFR Part 812).
25 FD&C Act § 564.
26 Other emergency-use authorities include FD&C Act §§ 564A, 505-1, and 564B; established by PAHPRA in 2013.
27 FD&C Act § 564.
TABLE 2-1 Summary of EUAs Issued (2005-2015)
| EUAs Issued by FDA | |||
|---|---|---|---|
| Year | MCM | Requester | Status |
| Anthrax (Bacillus anthracis) | |||
| 2005 | Anthrax Vaccine Adsorbed (AVA) | DoD | Terminated |
| 2008 (reissued in 2009, 2010, and 2011) | Doxycycline hyclate 100 mg oral tablets (in National Postal Model home/workplace kits) | HHS (ASPR/ BARDA) | Current (2011 version) |
| 2011 | All oral formulations of doxycycline (mass dispensing) | HHS (CDC) | Current |
| 2009 H1N1 Influenza Pandemic | |||
| 2009-2010 | Antivirals (3) | HHS (CDC) | Terminated (all H1N1 EUAs) |
| IVDs (18) | Various | ||
| Disposable N95 respirators | HHS (CDC) | ||
| Novel Influenza A (H7N9) Virus | |||
| 2013 | CDC Human Influenza Virus Real-Time RT-PCR Diagnostic Panel-Infleunza A/H7 (Eurasian Lineage) Assay | HHS (CDC) | Current |
| 2014 | Lyra Influenza A Subtype H7N9 Assay | Quidel Corporation | Current |
| 2014 | A/H7N9 Influenza Rapid Test | Arbor Vita Corporation | Current |
| Middle East Respiratory Syndrome Coronavirus (MERS-CoV) | |||
| 2013 (reissued in 2014) | CDC Novel Coronavirus 2012 Real-time RT-PCR Assay | HHS (CDC) | Current |
| 2015 | RealStar MERS-CoV RT-PCR Kit U.S. | Altona Diagnostics GmbH | Current |
| Enterovirus D68 (EV-D68) | |||
| 2015 | CDC EV-D68 2014 rRT-PCR Assay | HHS (CDC) | Current |
| Ebola EUAs Issued by FDA | |||
| Year | MCM | Requester | Status |
| 2014 (reissued in 2014) | DoD EZ1 Real-time RT-PCR Assay | DoD | Current |
| 2014 (reissued in 2015) | CDC Ebola VP40 rRT-PCR Assay | HHS (CDC) | Current |
| 2014 (reissued in 2015) | CDC Ebola NP rRT-PCR Assay | HHS (CDC) | Current |
| EUAs Issued by FDA | |||
| Year | MCM | Requester | Status |
| 2014 (reissued in 2015) | FilmArray NGDS BT-E Assay | BioFire Defense, LLC | Current |
| 2014 | FilmArray Biothreat-E test | BioFire Defense, LLC | Current |
| 2014 (reissued in 2014) | RealStar Ebolavuris RT-PCR Kit 1.0 | Altona Diagnostics, GmbH | Current |
| 2014 | LightMix Ebola Zaire rRT-PCR Test | Roche Molecular Systems, Inc. | Current |
| 2015 (reissued in 2015) | ReEBOV Antigen Rapid Test | Corgenix, Inc. | Current |
| 2015 | Xpert Ebola Assay | Cepheid | Current |
| 2015 | OraQuick Ebola Rapid Antigen Test | OraSure Technologies, Inc. | Current |
NOTE: ASPR = Office of the Assistant Secretary of Preparedness and Response; BARDA = Biomedical Advanced Research and Development Authority; CDC = Centers for Disease Control and Preparedness; DoD = Department of Defense; EUA = Emergency Use Authorization; HHS = Department of Health and Human Services; IVD = in vitro diagnostics; PCR = polymerase chain reaction; rRT-PCR = real-time reverse transcription polymerase chain reaction; RT-PCR = reverse transcription polymerase chain reaction.
SOURCE: Leissa presentation, February 4, 2016.
(e.g., for a new indication). When scientific evidence is available to support MCM use in a CBRN emergency, issuing an EUA enables response stakeholders to use, or prepare to use, an MCM without violating the FD&C Act of 1938, as amended. The DHS, DoD, or HHS Secretary makes a specific type of determination regarding requirements for EUA issuance. The HHS Secretary issues a declaration that circumstances exist to justify EUA issuance based on one of the four determinations, and FDA ensures that EUA criteria for issuance are met (e.g., based on totality of scientific evidence, the known/potential benefits outweigh known/potential risks; no adequate, approved, available alternative). Conditions of authorization are put in place as safeguards for use of the product under EUA.28
Leissa explained that there are also other emergency-use authorities available in certain circumstances. PAHPRA established emergency-use
___________________
28 For example, information on emergency use, fact sheets for recipients and health care professionals, dispensing/screening procedures, monitoring of adverse events, clarification of roles.
authorities for eligible FDA-approved MCMs intended for use in CBRN emergencies to facilitate stakeholder preparedness and response without an EUA.29 Emergency dispensing orders (FDA authority) allow mass dispensing at PODs without an individual prescription or all required labeling. Emergency-use instructions (CDC authority) are streamlined fact sheets on MCM use for recipients and health care providers. Expiration dating extensions are allowed when FDA determines, through either the Shelf Life Extension Program (SLEP) or other announcements it makes about extensions, that these are legally covered as well under these emergency-use authorities.
Current Good Manufacturing Practice (CGMP) waivers (FDA authority) are used when there may be a limited period of time during an emergency in which a product might need to be transported or stored outside of its labeled storage conditions. Risk Evaluation and Mitigation Strategy waivers (FDA authority) are also in place for certain products. Prepositioning of approved or unapproved MCMs30 facilitates rapid deployment during an actual emergency; it allows prepositioning of MCMs by or on behalf of government entities (federal, state, or local) in anticipation of FDA approval, clearance, or licensure or EUA issuance. These authorities preserve otherwise applicable liability protections (e.g., PREP Act) for MCM planning, preparedness, and response stakeholders.
During the discussion, Boris Lushniak, professor and chair, Department of Preventive Medicine and Biostatistics, F. Edward Hébert School of Medicine, Uniformed Services University, asked whether there have been legislative or regulatory approaches that have been discussed at FDA or other agencies to improve the process, given existing constraints. Leissa replied that on the emergency preparedness side with respect to state and local capabilities, there are preexisting authorities that can be used. Emergency-use guidance for these authorities will be issued soon, so that local-level officials will know that they exist and to resolve any misunderstanding about when EUAs are required or not in various circumstances. In terms of MCM development, he explained that FDA is somewhat limited in its authorities for improving the process (e.g., the Animal Rule does require animal data, not in vitro data, in quality animal models). Various avenues are being explored to assist in the development of MCMs, according to Leissa, such as an established qualified animal model that could be used by multiple developers, not just the government.
___________________
29 FD&C Act § 564A.
30 FD&C Act § 564B.






















