6
Potential Supply-Chain Opportunities and Lessons from the Commercial Sector and Government Partners
STRENGTHENING THE PUBLIC HEALTH RESPONSE SUPPLY CHAIN
Brandeau co-authored a paper (Brandeau et al., 2007) with her research group that focused on how to improve the public health response supply chain with lessons from optimized commercial supply chains relevant to the public health supply chain. The paper also looked at lessons from responses to events such as natural disasters, naturally occurring outbreaks, and the 2001 anthrax attacks. She commented:
We view the public health response supply chain really as an effort that involves not only procurement and distribution of medical and pharmaceutical supplies but more broadly, we think it is about the personnel involved in response and information. We think of it more holistically . . . it’s not just material; it’s people and information.
Brandeau explained that a public health emergency necessitates rapid procurement and distribution of medical and pharmaceutical supplies, trained personnel, and information. However, publications (Bravata et al., 2004) and preparedness drills (e.g., top officials [TOPOFF] exercises) have demonstrated that gaps in preparedness exist that underscore the need to better understand the best ways to design and maintain the public health response supply chain. She noted that previous research has focused on limited aspects of the public health response supply chain, such as models for planning the facility design and personnel requirements for large-scale dispensing clinics (Hupert et al., 2002; Lee et al., 2006).
Brandeau’s group examined models (Bravata et al., 2006; Zaric et al., 2008) for evaluating local stockpiling and dispensing strategies by modeling the supply chain of materials for an anthrax attack, from the Strategic National Stockpile (SNS) to state and local stockpiles to dispensing. The limited model demonstrated that the bottleneck is likely to be dispensing capacity, not inventory.
The Public Health Response Supply Chain Versus the Commercial Supply Chain
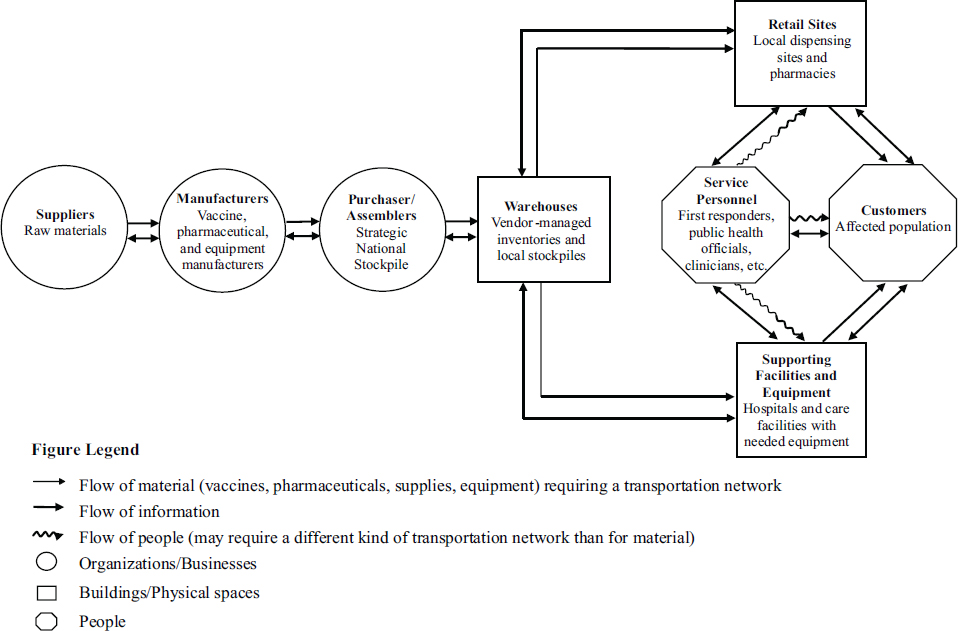
Brandeau provided a schematic of the public health response supply chain (see Figure 6-1). She emphasized that the system is more than people (suppliers, manufacturers, purchasers, assemblers, first responders, public health officials, clinicians, and the affected population), material, equipment, and sites but the flow of materials, information, and people through the system.
Brandeau pointed to a set of critical differences between the commercial and public health supply chains. The commercial supply chain operates continually; demands are known with some certainty and the speed of response is important. The public health supply chain operates infrequently; demands are highly uncertain and the speed of response is essential. The latter also has more players—beyond the suppliers, manufacturers, assemblers, and retailers that constitute the former, there are agencies (national, regional, and local), local public health officials, first responders, and many others. Whereas commercial supply chains center on cost efficiency, the public health supply chain’s success metrics include costs but also health outcomes and, also importantly, social outcomes: “it’s not just that lives were saved by our response, but it was done in a way that there was minimum social disruption.”
Her group’s study suggested five strategies for public health supply-chain improvement:
- Effective supply-chain network design,
- Effective inventory management,
- Postponement of product customization and modularization of component parts,
- Coordination of supply-chain stakeholders and appropriate use of incentives, and
- Effective information management.
Commercial supply chains are very efficient in managing inventories and inventory flows, explained Brandeau. However, she advised that the public health response supply chain may well need a more robust network
SOURCE: Brandeau presentation, February 4, 2016.
design than commercial supply chains because of the unknowns: where the event will occur, what magnitude it will be, and so forth. This means that planning models and exercises are essential in thinking about what the public health response supply chain looks like and not just planning models but exercises, taking into account that there are planners at all levels of response to be organized with an overarching strategy.
The next strategy concerns effective inventory management, a management effort that strikes the right balance at the federal, regional, and local levels in the coordination of inventory management. As an example, Brandeau cited a report on the issue of prepositioning and forward deployment (IOM, 2012) and the associated trade-off between pushing inventories all the way down the supply chain versus holding them centrally, which is more cost-effective but less flexible. Inventory pooling is more cost-effective, but local inventories allow for more rapid response. A related issue is that the very lean level of inventory favored by commercial supply chains may not be as desirable in public health supply chains (which also very often have items needed for response that may need to be quickly manufactured—another key aspect of planning).
Appropriate postponement of product customization and modularization of component parts was pinpointed by Brandeau as an important point to consider. For example, a TOPOFF exercise in 2000 found that modularization, like prepackaging items for response, was extremely efficient but much less flexible. The research group maintains that a model of the public health response supply chain, including dispensing, could be used to evaluate options for modularization versus postponement of inventory customization.
Brandeau stressed that the coordination of supply-chain stakeholders and appropriate use of incentives is important because the affected public, and its expectations, are part of the supply chain that should be aligned along with all other groups involved: different branches and levels of government, private industry, and all levels of responders. Beyond the financial and cost-based incentives in the commercial supply chain, the public health supply chain also has legal and jurisdictional incentives and constraints as well as personal incentives (e.g., volunteers who are dispensing at points of dispensing [PODs] who are personally or altruistically rather than financially or legally motivated).
Finally, the research group suggested that effective management of timely and accurate information is also critical: what information the affected population needs and what the available resources for response are at all levels. Technology can play a key role here (e.g., radio-frequency identification [RFID] device tags, two-dimensional barcodes on inventory, Web-based portals for inputting and obtaining real-time information), but because of the unknown nature of events, the group recommends
building redundancies into systems for obtaining and managing response information.
Brandeau summarized the study’s conclusions: a well-designed and managed public health response supply chain can minimize costs while maximizing health outcomes and, ideally, minimizing social disruption. The public health response supply chain likely needs to be more robust than any single commercial supply chain (see Box 6-1), and it is important to coordinate and appropriately incentivize the activities of the many stakeholders. Brandeau maintained that even though the public health response supply chain is rarely activated, “we can and must learn from experience.”
UNDERSTANDING AND LEVERAGING PRACTICES OF THE COMMERCIAL SUPPLY CHAIN: LESSONS FROM PRIVATE-SECTOR THIRD-PARTY LOGISTICS PROVIDERS
Third-Party Logistics—Supporting the SNS
Thomas Mattingly, president of Metro Logics, Inc., and Frederick of FedEx Custom Critical (see Box 6-2) each spoke about their company’s working relationship with the SNS. Metro Logics, Inc., is a third-party logistics (3PL) service provider specializing in temperature-controlled distribution that has worked with the SNS for 16 years. He explained that the job of a 3PL service provider is to manage the storage and movement of inventory, in compliance with all applicable regulatory authorities and requirements. Specifically, Metro Logic’s key commitments are to ensure that its customer’s products are safe, accurately counted, continuously stored at predefined storage conditions, and ready for rapid shipment if needed.
Mattingly noted that today, the SNS actively pursues new technology opportunities to increase both speed and accuracy, whereas in the early years of the program, every function was a manual process with little or no electronic data interchange (EDI) capability. He attributed significant improvement in multiple areas of the program to the application of various types of technological advances. For instance, transactions are communicated electronically to the SNS using flat files passed through the Public Health Information Network Messaging System to ensure a secure, encrypted transfer.
Perpetual inventory files are electronically transmitted on a weekly basis to identify and report any inventory discrepancies, which are then investigated and reconciled. Internal warehouse product movement files are electronically transmitted on a daily basis to ensure the integrity of product location within the facility. Outbound orders and receipt, storage, and staging (RSS) files are processed electronically to decrease potential typing input errors and significantly reduce the amount of time necessary to process orders. Physical inventory count times have been reduced by over 80 percent by using radio-frequency technology.1 Physical inventory accuracy has also increased significantly, with the last one taken yielding a 100 percent financial accuracy rating and a 99.99 percent overall accuracy rating.
Services Provided by Third-Party Logistics
Mattingly provided an overview of the types of services provided by 3PL providers such as Metro Logics. They maintain an ongoing accounting
___________________
1 What previously took up to 42 8-hour shifts to complete now takes 5 to 7.
for all products as they are received, stored, and withdrawn, with inventory of stored projects performed to ensure that 100 percent of product is accounted for annually. All products are stored and maintained according to specified temperature requirements, and in compliance with all applicable regulations including those of the Department of Health and Human Services (HHS), the Food and Drug Administration (FDA), and the Drug
Enforcement Agency (DEA) and other such federal, state, or local laws that may apply. Metro Logics labels, tags, and otherwise prepares product for shipment as directed by the customer and ensures that the lot, dating, and quantity of product that appear on each shipment manifest reconcile with what is on the customer order identified on the pack list. To enable rapid shipment capability, facilities are designed and laid out to ensure that 100 percent of the product is available 100 percent of the time with minimum effort. They also monitor warehouse product locations to ensure that a given item is stored throughout the warehouse rather that in a concentrated area.2 Surge support is also critical; Metro Logics maintains a supplemental workforce that is utilized during an emergency. These employees receive a monthly on-call stipend and are required to work and train a minimum of 4 hours per month. This monthly training requirement also serves to help determine an employee’s level of commitment to the program.
In addition to managing the storage and movement of inventory, Mattingly explained that Metro Logics is also committed to maintaining the highest standards of quality and regulatory compliance, which is demonstrated by their highly qualified and experienced quality staff. All processes are managed through controlled procedures (e.g., document control is maintained using a validated electronic document management system). Regulatory conformance remains compliant with 21 CFR Parts 11, 210, 211, and 820 and other customer-specific requirements. Validation and calibration processes are used to ensure that all equipment and storage areas are qualified, with performance qualifications implemented annually on all validated storage areas (software systems are validated according to 21 CFR Part 11).
Government Partner Supply Chain and Stockpile Strategies
Veterans Health Administration All-Hazards Emergency Cache Program
Lewis Radonovich, director, National Center for Occupational Health and Infection Control, Department of Veterans Affairs (VA), reported on the Veterans Health Administration (VHA) All-Hazards Emergency Cache Program. The VHA is the largest integrated health care system in the country, comprising 167 medical centers and 1,026 outpatient clinics.3 He out-lined several ways that the VA stockpile differs from the SNS. It is primarily intended to ensure easy accessibility and rapid access to large quantities of commonly used medical supplies and countermeasures as determined by hazards vulnerability assessments at the national, regional, and local levels;
___________________
2 This practice eliminates the “hourglass effect” that occurs when too many employees are attempting to pull product from the same aisle.
3 Approximately 823 community-based, 144 hospital-based, and 59 mobile units.
the SNS is more focused on providing access to medications and supplies that may not be commonly available. The VA system is widely geographically distributed across a national network of hospitals and clinics and is complementary in the sense that it is intended to dovetail with medical supplies and countermeasures held in local VA pharmacies and supply inventories. It is integrated and can be replenished quickly through routine supply chains, as necessary, and is scalable to meet small surge needs for routine care at the local level or larger-scale crises at the regional or national level.
Radonovich explained that the VA stockpile is designed for response to CBRN events and, as such, its content categories include antibiotics, antivirals, emergency medications, chemical countermeasures, intravenous (IV) fluids, wound care, and personal protective equipment. Full contents are available in local caches in VA medical centers with larger supplies of selected items stored in a central location. Its contents are primarily intended for the 8 million veterans and 300,000 VA staff members for the short-term preservation of VA operations in a crisis and to bridge the gap between what is available in local pharmacies and what other resources such as the SNS would provide, as well as supporting the VA facility’s involvement in the local community disaster plan. Cache operations are managed by the VA Emergency Pharmacy Service, which provides logistics support and manages product acquisition and life-cycle management, as well as VA participation in the Shelf Life Extension Program. Several executive committees oversee operations, which are reviewed on a regular basis.
Radonovich characterized the VA as both a community asset and a federal asset, which therefore deals with some competing objectives. However, he emphasized that first and foremost, it is a community medical center that is engaged in the local emergency response and at times may be called on to serve as a national resource in the event of a national or large regional emergency. One of the aims of the VA is to build the community-level understanding of its dual role as a federally governed asset and local community health care provider.
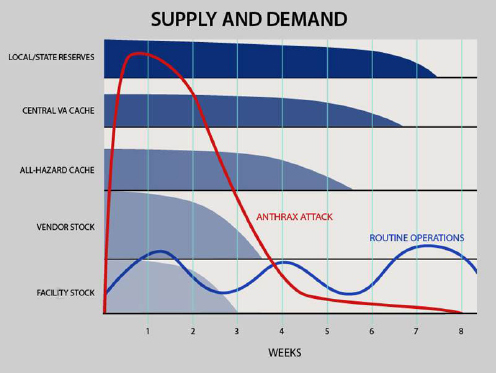
Radonovich explained the VA cache’s scalability and interoperability in Figure 6-2. He noted that routine operations use the inventory that is available in VA pharmacies, but during a national crisis, supplies that are normally available from vendors and local inventory would likely be exhausted and necessitate reaching out to the cache locally. If the local cache is insufficient, the next step is the central warehouse where additional supplies are stored. If that is exhausted, the next option is to reach out to the local and state levels (essentially, the SNS).
The VA used the Federal Emergency Management Agency National Planning Scenario to study what might happen in a large city such as Chicago in the event of an aerosolized anthrax attack. An estimated 300,000 people would be exposed to anthrax, including 20,000 veterans.
SOURCE: Radonovich presentation, February 4, 2016.
Radonovich reported that in that scenario, they have estimated that they could provide prophylactic antibiotics to all veterans and VA staff on the campus, as well as a 60-day prophylactic course after they were relocated.
Although the VA stockpile was not necessarily intended to be used for pharmaceuticals shortages, Radonovich explained that it is in fact tapped into somewhat regularly for that purpose. Each week they might receive five pages’ worth of pharmaceutical shortages, some of which trigger usage of the supplies in the cache. Noting that the VA stockpile is used regularly to relieve drug shortages, Carlin asked about the decision-making process and how the potential negative consequences are balanced with the benefits. Radonovich explained that it is needs based, not a formula. If they run out of supply that cannot be immediately acquired from the vendor, then the most logical option is the stockpile.
Radonovich described how future efforts are aimed at being able to dispense MCMs to large numbers of people efficiently (e.g., through host demonstrations optimizing VHA dispensing capabilities) as well as refining the contents of the VHA emergency cache based on vulnerability assess-
ments and clarifying its role during pharmaceutical shortages. Finally, they seek to optimize federal and local community partnerships by enhancing the integration of each VA medical facility within each local community and working closely with the Centers for Disease Control and Prevention (CDC), the Department of Defense (DoD), and PHEMCE.
DoD Support to Contingency Operations
Colonel Alex Zotomayor, director of the Medical Supply Chain for the Defense Logistics Agency (DLA), DoD, described DLA’s role as the logistics provider for DoD and U.S. military forces. The medical supply chain is situated within DLA Troop Support, which manages a range of supply chains, contracts, and programs. The DLA medical support strategy has a dual focus: both institutional and operational. Its everyday focus is the former, which requires speed, reliability, responsiveness, and affordability for military treatment facilities worldwide. During a crisis, the focus shifts to operational and being ready to respond immediately to surge requirements to support contingency operations spanning the range of natural disasters up to wars on multiple fronts.
Zotomayor explained that prior to the 1990s, medical troop support was a stockpile-heavy organization, but during Operations Desert Shield and Desert Storm, the support provided was not optimal due to both the lack of responsiveness to time lines and the fact that the items being pushed out were not actually needed or used by deployed forces. That forced DLA to reorganize, realizing the warehousing and depot strategy was not optimal. They pared down the inventory of depot stock (from tens of thousands of lines to 1,100 current lines of stock) and adopted a commercial-type “just-in-time” model, drawing on e-commerce solutions such as Prime Vendor and Electronic Catalog. These two programs meet 91 percent of their customers’ requirements, with a delivery time frame of 1 day within the United States and 5 to 7 days for overseas customers. Materiel not covered by the e-commerce programs is purchased through direct vendor delivery (e.g., for seasonal flu vaccines). The DLA distribution depot does maintain a small stockpile for military unique materiel and MCM supplies.
Forward Presence Inventory: Delivery Agents
Zotomayor described the Army Medical Materiel Agreement (AMMA), a formalized partnership between DLA and the Army Medical Command which grants designated army activities access to and the use of DLA’s working capital fund. This provides the Army with a reliable source of funds for ordering and maintaining supplies for particular activities, including the support of surge requirements. There are currently four Army hos-
pitals designated as AMMA sites, including three overseas; the agreement is that the Army manages the stock for DLA, but it remains DLA property until sold by the Army. The advantage for DLA is that they have these AMMA sites as mini distribution depots.
The majority of distribution is handled by commercial vendors, according to Zotomayor, who deliver directly to customers using their own robust distribution networks, with DLA providing movement via Transportation Command’s commercial carrier contracts for the transport of hazardous material, controlled substances, and cold-chain management. As a DoD entity, DLA also has access to military assets and distribution hubs that are set up during contingency operations.
Surge Requirements and Readiness Contracts
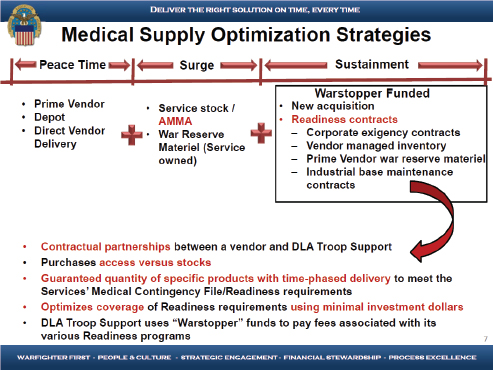
During a surge requirement, Zotomayor explained that the military forces maintain a limited amount of stock in their own units and are supported with AMMA sites. Beyond that, DLA relies primarily on readiness contracts with the health care industry through which they purchase guaranteed access to materiel or production capability with time-phased deliveries that minimize DLA’s requirement to stockpile and maintain material at our distribution depots (see Figure 6-3). Currently, DLA has invested approximately $27 million per year in readiness contracts providing access to approximately $780 million of material in stock (a $28.10 return on investment). Zotomayor outlined the four types of readiness contracts. Corporate exigency contracts are long-term partnerships with manufacturers providing guaranteed product availability, in order to provide sustainment material to meet the surge requirements. The benefit from this type of contract is large quantities due to access to manufacturers and to their production capabilities. Vendor-managed inventory contracts focus on long-term partnerships with distributors to provide guaranteed product availability, with the benefit of timely response within a 24-hour delivery period (currently the only vendor-managed inventory contracts that they have are for pharmaceutical items). Prime vendor war readiness contracts are adjunct to prime vendor contracts that allow prenegotiated access to stocks in support of surge requirements. Industrial base maintenance contracts are long-term partnerships with select manufacturers to maintain a warm production base for particular items of military significance; these are items with production time lines and capabilities such that DLA needs to ensure that the industry maintains a consistent flow of production. DLA Troop Support uses “Warstopper” funds to pay fees associated with its various readiness programs.
DLA has strategic relationships that are formalized in the Defense Medical Logistics Enterprise; Zotomayor emphasized that DLA cannot be
NOTE: AMMA = Army Medical Material Agreement; DLA = Defense Logistics Agency.
SOURCE: Zotomayor presentation, February 4, 2016.
stove piped and look at things only from a DoD perspective. Looking at things from a broader government perspective and incorporating other government partners regarding procurement of medical supplies is important because of competition in sources of supply. DLA is currently conducting industrial preparedness planning assessments, a collaborative effort among DLA Medical Troop Support, the Armed Services, and the health care industry to assess industrial capabilities of manufacturers and distributors to provide materiel to meet the time-phased mobilization and sustainment requirements of the Services. Once the assessment is completed, decisions will be made about purchasing and maintaining stock, negotiating a contingency contract, and/or accepting risks regarding the ability of future production time lines to meet requirements in light of current inventory.
In the context of the large number of products in the SNS inventory and the associated tensions of sustainability, Inglesby asked how DoD and the VA determine the boundaries for their stockpiles and whether their stockpiles are subject to the same types of tensions as the SNS. Decision making
is simple and streamlined for DoD, according to Zotomayor, because they are told what the requirements are by Health Affairs, Defense Health Agency, and the Services. Radonovich explained that the VA uses two parallel processes. At the national stockpile level, they back into numbers by estimating the number of veterans in a community and the number of expected exposures in that group, then extrapolating that to estimate the number of MCMs needed for the entire country. At the local level, they use standard hazard vulnerability assessment levels.
Sosin asked about DoD’s use of multiple intermediary holders of material, as one of the challenges for CDC is ensuring that the quality of the management and control of that material by intermediaries. Zotomayor explained that there is a standard across DoD that military medical logistics forces have to maintain in the management of items, which is clearly laid out in formalized agreements. They also perform follow-up inspections, follow-up inventories, and so forth to ensure that there are good management practices and established quality control.
POTENTIAL OPPORTUNITY FOR BETTER ENGAGEMENT WITH THE COMMERCIAL AND PHARMACEUTICAL INDUSTRIES
Fri pointed out the “gray area” of overlap between products in the stockpile and those in the commercial supply chain (e.g., antivirals). He suggested finding ways to collaborate in overlap spots in the supply chains to ensure that medicines are reaching the right people in the right place at the right time. He elaborated that unique medicines in the stockpile are governed by very specific contracts, but many products in the commercial supply chain are made in China, Europe, India, and Israel. Much of that responsibility is external to the United States, even though the United States still is by far the largest pharmaceutical marketplace in the world. Furthermore, at least two of the large U.S. distributors are now international entities negotiating contracts on a global basis.
Mansoura suggested that relationships with industry need more attention, as the notion of developing products with an indefinite shelf life, for example, would likely be detrimental from the perspective of the manufacturer: “things that are upside and cost-efficient on the government side are potentially the opposite on the other side of the partnership.” She noted that vaccines, for example, are a particularly fragile system; expanding production will require looking at some of the broader, strategic, and system-level issues in the vaccine industry.
Khan suggested the use of reverse inventory contracts such that SNS materials can be returned to industry partners if requested for distribution through their routine supply chains in order to address nationwide need, and then replenished with new materials. But he cautioned that the SNS has
previously assessed scenarios where this actually would not be helpful, and products in the SNS have never been used to support select shortages in the commercial supply chain. Either the supply chain has recovered quickly so that the federal assistance has not been needed, he explained, or products to support the commercial inventory gaps did not exist in sufficient amounts in the SNS to make a substantial difference.
SNS Role in Fiscally Sustaining the MCM Industry
Referring to economic tension between the Division of Strategic National Stockpile’s (DSNS’s) desire for lower-cost countermeasures and the limited market viability of some of the products it stockpiles, Sosin maintained that the SNS’s role in fiscally sustaining the MCM production is a legitimate one, but pointed to the question of whether that optimizes the use of resources for the spectrum of risk: “Does it buy down the most risk? Can we effectively do both or do we have to choose?” He contended that Khan’s suggestion of moving toward the development of products with indefinite shelf lives would not alleviate the problem, because manufacturing those product lines would not make financial sense from the industry’s perspective. Khan clarified that these products were not necessarily purchased with SNS funds and that SNS dollars are relatively stable if adjusted for inflation—a process is needed then to buy down the biggest risk given the resources that are available.
Price Negotiation
Lewis Grossman, professor of law, American University Washington College of Law, noted that the pharmaceutical market is an unusual one (e.g., regulatory barriers to entry) and that the products stocked by the SNS are also particularly atypical from a market perspective. From a procurement and price negotiation perspective, he questioned whether the SNS faces any challenges and whether its statutory underpinnings provide any special provisions regarding procurement. Sherman explained that the Project BioShield Act put forth the authority to procure security countermeasures, with quite a few special contracting authorities for exceptional circumstances that have been adjusted over time. In terms of price negotiation and conditions, Burel reported that historically, the SNS has been very effective in obtaining the best possible prices for products given the circumstances, but he conceded that for commercial products, they are far off the retail prices. For sole-provider products, there are enough benchmark products to make reasonable estimates (e.g., vaccines can be compared to other vaccines). Khan noted that while the SNS is already working on the ability to bargain for materials that come into the SNS based on actual cost, the reality is that most of these products
have no other commercial market, and so the SNS should be doing more. Burel explained that MCMs have benchmarks for the early and advanced development stages that BARDA can use to suggest the financial model that is appropriate to achieve a fair and reasonable price for that product. Those data, coupled with other procurement-sensitive data, are used to further negotiate new pricing on products as they come for purchase.
Commercialization of the SNS Supply Chain
Larry Glasscock, senior vice president, Global Accounts, MNX Global Logistics, contended that moving to the commercial arena as much as possible is important for the SNS, because a very high percentage of material and ingredients that go into the more sophisticated and complex countermeasures are imported. He questioned how the SNS can address this additional layer of complexity (e.g., shortages in the commercial supply chain and changes in the marketplace) in the move toward commercialization. Burel responded that the SNS distribution process tries to act like the commercial market to the extent possible because it is known to work, but that they will have to plug into systems that are designed to dispense in a different way than they normally operate. Challenges further upstream in the supply chain, back to the raw material for particular holdings, becomes even more complex. Burel noted that although nothing prevents the SNS or BARDA (or a joint effort) from buying and stocking raw materials from a procurement perspective, it adds a layer of procurement responsibility—and a layer of cost—for which resources are not currently available. Pharmaceutical supply-chain security will pose an issue going forward, but again, he noted that the SNS does not have the financial capability to move into that space except for specifically targeted concerns that must be addressed.
POTENTIAL OPPORTUNITY FOR IMPROVED ELECTRONIC DATA INTERCHANGE
Mattingly highlighted areas for improvement with respect to EDI issues. In the early stages, the SNS was not able to preload the RSSs, requiring extensive time spent manually keying in RSS files. This problem was rectified by creating a program to match files submitted by the SNS (flat file to flat file), but there is room for improvement. His organization would benefit from being able to communicate with vendors and manufacturers directly by importing their information and scanning their native labels, which allows more capacity to be devoted to logistical operations. He also suggested using available technology for advanced shipping notices to allow visibility of trucks at all times.
Fri noted that there are specific standards in the pharmaceutical supply chain for transmitting information electronically and for labeling case- and pallet-level barcodes; thus any company in the commercial supply chain that works with large wholesalers must be doing EDI and be compliant with these barcode guidelines. Given that the SNS labels differently, she asked about the potential to use some of those industry standards. Burel explained that the SNS can accept any type of commercial barcode and has not specified a different standard, because it already exists. However, the SNS’s own inventory system does have compliance issues around barcoding.
Mattingly clarified that the flat-file-to-flat-file system is effective, but not in the standard EDI format used widely; it would be beneficial to move toward the point of being able to communicate in real time:
I’ll move a pallet in my warehouse. I’m scanning. It updates now. Their files don’t update until the end of the day. It’s an automatic. We send it automatically to PHINMS [Public Health Information Network Messaging System], they pick it up, and they execute it. But there are still a couple hours our locations could be off, but we’re working on that. We’re not the standard format that’s recognized in the industry, but we are trading information.
Burel continued that the use of flat files is related to specific security issues around how data can be exchanged between CDC’s firewall systems and external firewall systems—at this point, exchanging that flat file is the best option that does not involve prohibitive cost, and the delay is not an issue for the SNS.
Poole described a common problem from the state perspective: they receive the product from CDC and the bill of lading, but they or their private vendors cannot import that into the RSS. Burel explained that because they are in a unique position of interfacing with every state, and possibly every locality, further facilitating EDI would require dealing with a wide range of different systems. However, for localities that have contracted with private-sector vendors who are accustomed to receiving electronic data, it would be relatively easy for DSNS to provide them with EDI information in addition to the bill of lading.
Petersen related that during H1N1, his department received the product from CDC but did not get the expected electronic files; instead they received many pages of physical bills of lading that they had to input manually into the inventory management system. This was time intensive, especially during the initial phase when the severity of the pandemic and how or whether the product would be distributed were unknowns. He commented that improving that EDI side for managed inventory needs further progress. Burel agreed, noting that the SNS does not mandate an inventory system
to be used in every state and every locality; that said, having 62 separate Public Health Emergency Preparedness Cooperative Agreement holders gives rise to the potential for 62 different formats. However, he stated that the SNS would continue to work to address this issue.
Judith Robinson, supervisory public health analyst, District of Columbia (DC) Department of Health, asked if in addition to the bill of lading, it would be possible to also send a flat file that could be imported into the inventory management system. Burel replied that there is the potential to provide files for import into inventory management systems easily if the locality is using the system that the SNS offers. Otherwise, the file would need to be formatted in such a way that it would match up with the location-specific inventory management system. Every project area could potentially use a different file format, which might change over time and have to be continually updated.


















