7
Molybdenum-99/Technetium-99m Supply
This chapter addresses the supply component of the fourth charge of the statement of task for this study (see Sidebar 1.3 in Chapter 1), which directs the Academies to provide an assessment of
The adequacy of molybdenum-99 [Mo-99] supplies to meet future domestic medical needs, particularly in 2016 and beyond.
The demand component of this study charge was addressed in Chapter 6.
The committee interprets the term “beyond” in the study charge to mean the next 5 years (i.e., until about 2021). The committee judges that there are too many uncertainties in Mo-99 supply and demand (see Chapter 6) to look any further into the future.
This chapter is divided into three sections: Section 7.1 describes the factors that will affect future Mo-99 supplies. Section 7.2 describes the adequacy of Mo-99 supplies to meet future global demand, drawing on the careful work of the Organisation for Economic Co-operation and Development’s Nuclear Energy Agency (OECD-NEA). Section 7.3 provides the committee’s finding and recommendation on the adequacy of Mo-99 supplies to meet future domestic demand.
7.1 FUTURE AVAILABLE SUPPLY CAPACITY
Several key points important to future available supply capacity were established in Chapters 3-6:
- Current global demand for Mo-99 is estimated to be about 9,000 6-day curies (Ci) per week, about half of which is consumed in the United States. It is unlikely that domestic demand for Mo-99 will increase significantly over the next 5 years (see Chapter 6).
- The United States imports Mo-99 to meet all of its domestic needs from five global suppliers in Australia, Canada, Europe, and South Africa (see Chapter 3); there is no domestic production of Mo-99 at present (see Chapter 4).
- Canada (NRU/Nordion) will stop supplying Mo-99 after October 2016. Canada will then become a supplier of last resort until the end of March 2018 (see Chapter 3). Nordion plans to resume supplying Mo-99 produced at the University of Missouri Research Reactor Center (MURR) in 2018 (see Chapter 4).
Nordion’s exit from the Mo-99 supply chain after October 2016 will reduce available supply capacity and could affect the adequacy of Mo-99 supplies to the United States and other countries; the reduction will persist until this lost capacity is restored by Nordion or other suppliers.
Future supplies of Mo-99 will be affected by five factors:
- Current available global capacity to supply Mo-99
- Reductions in available global supply capacity after Canada stops producing Mo-99
- Planned additions to available global supply capacity by current irradiation services suppliers and/or Mo-99 suppliers
- Potential additions to available domestic supply capacity
- Other potential sources of Mo-99/Tc-99m supplies
These factors are discussed in the following subsections.
7.1.1 Current Available Global Capacity to Supply Mo-99
About 95 percent of the global supply of Mo-99 is produced by seven irradiation services suppliers and distributed by five Mo-99 global suppliers (see Chapter 3):
- Belgian Reactor-2 (BR-2) at Mol, Belgium. Mo-99 produced in this reactor is supplied to global markets by the Institut National des Radioélements (IRE) and Mallinckrodt.
- High Flux Reactor (HFR) at Petten, the Netherlands. Mo-99 produced in this reactor is supplied to global markets by IRE and Mallinckrodt.
- LVR-15 at Rez, Czech Republic. Mo-99 produced in this reactor is supplied to global markets by IRE.
- Maria at Otwock-Swierk, Poland. Mo-99 produced in this reactor is supplied to global markets by Mallinckrodt.
- National Research Universal (NRU) at the Canadian Nuclear Laboratories (CNL), Chalk River, Ontario. Mo-99 produced in this reactor is supplied to global markets by Nordion.
- Open Pool Australian Lightwater (OPAL) at Lucas Heights, Australia. Mo-99 produced in this reactor is supplied to global markets by the Australian Nuclear Science and Technology Organisation (ANSTO).
- South Africa Fundamental Atomic Research Installation 1 (SAFARI-1) at Pelindaba, South Africa. Mo-99 produced in this reactor is supplied to global markets by Nuclear Technology Products (NTP).
These seven irradiation services suppliers have a combined Mo-99 production capacity1 of about 28,000 6-day Ci per week2 (see Table 3.2 in Chapter 3). This is about three times higher than the current weekly global demand for Mo-99 of 9,000 6-day Ci. The five global Mo-99 suppliers have a combined irradiated target processing capacity of about 16,000 6-day Ci per week (see Table 3.3 in Chapter 3). This is slightly less than twice the current weekly global demand for Mo-99.
7.1.2 Reductions in Available Global Supply Capacity
The NRU reactor in Canada is scheduled to stop production of Mo-99 after October 2016 and permanently shut down at the end of March 2018 (see Chapter 3). During the period November 2016 through March 2018—referred to here as the contingency period—the NRU reactor could potentially resume production to support global Mo-99 supply if there is a shortage (NRCan, 2015). The associated CNL and Nordion facilities required for target processing and Mo-99 purification (see Chapter 3) will be kept in a hot standby mode during this contingency period.
The Canadian government has emphasized that the NRU reactor capacity during this contingency period should not be viewed as outage reserve capacity (ORC, see Sidebar 3.1 in Chapter 3) that the market can expect to draw upon as a matter of normal course of business. Instead, it should be viewed as a supply of last resort that will be used only in the
___________________
1 Not accounting for reactor downtime for scheduled or unscheduled maintenance.
2 All 6-day Ci estimates in this chapter are referenced to end of target processing. See Chapter 3.
event of unexpected shortages that cannot be mitigated through other means (Brady and Pruneau, 2015). The decision to resume Mo-99 production in NRU will be made by the Canadian government in consultation with other stakeholders.
The Canadian government has not publicly described the supply shortage triggers that would lead it to order a restart of Mo-99 production at NRU during the contingency period.3 The Canadian government is instead focusing on establishing communication channels with relevant stakeholders so that it can receive the information it needs to inform a restart decision. These stakeholders include the Canadian federal agencies National Resources Canada and Health Canada; international organizations with information on reactor outage schedules such as the Association of Imaging Producers & Equipment Suppliers; global Mo-99 suppliers; and other national governments, including the United States.4 The Canadian and U.S. governments, for example, have convened a bilateral working group to discuss Canada’s plans to resume Mo-99 production at NRU if significant shortages develop during the contingency period. The details of these discussions are not public.
NRU/Nordion’s ability to restart Mo-99 production and supply during the contingency period is subject to the availability of
- Highly enriched uranium (HEU) targets,
- An active operating license for NRU5 and an operating reactor, and
- Readiness at CNL and Nordion to process irradiated targets and to purify Mo-99 for commercial sale.
CNL informed the committee that it expects to have sufficient HEU targets available to respond to Mo-99 supply shortages during the contingency period. NRU should also be available to produce Mo-99 during the contingency period unless it is shut down for scheduled or unscheduled maintenance. NRU has had a lengthy unscheduled outage in 2009-2010 (see Table 3.5 in Chapter 3).
Representatives of CNL and Nordion told the committee that they plan to maintain their target processing and Mo-99 purification facilities (see Chapter 3) in standby mode during the contingency period. They also plan to retain the staff that operate these facilities and may conduct training exercises to help maintain production readiness.
___________________
3 A representative of CNL implied that the Canadian government does not intend to publicly release quantitative details of this information (Niall O’Dea, CNL, May 2016, OSTP Mo-99 Stakeholder meeting).
4 Niall O’Dea, CNL, May 2016, OSTP Mo-99 Stakeholder meeting.
5 These licenses are granted to CNL by the Canadian Nuclear Safety Commission.
The shutdown of NRU will result in loss of almost 20 percent of current global Mo-99 production capacity. It will also idle Nordion’s Mo-99 processing capacity until its new project with General Atomics and MURR comes online, probably after March 2018 (see Chapter 4).
NRU/Nordion have in the past been able to increase Mo-99 production and supply on short notice to fill supply gaps. They are a particularly important supply source for the United States because of their excess capacity production model and close geographic proximity:
- NRU has the capacity to produce Mo-99 in excess of what is needed to meet Nordion’s normal customer demand. This excess can be dispatched to customers on short notice.
- Mo-99 shipments from Nordion can arrive in the United States within about 24 hours of when they are requested. Consequently, there is relatively little Mo-99 lost to decay during shipment. Nordion ships Mo-99 to the United States using chartered aircraft, which adds flexibility and convenience.
This supply flexibility will be maintained during the contingency period, but it will be lost after March 2018.
7.1.3 Planned Additions to Available Global Supply Capacity
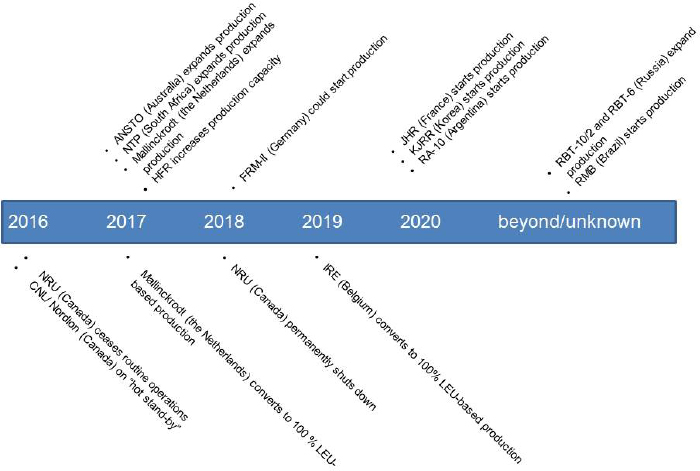
Several current global Mo-99 suppliers plan to expand their capacities to supply Mo-99 on a routine basis6 starting in 2017, and new global or regional Mo-99 suppliers plan to enter the market later this decade (see Chapter 3).
Some current global supplies are planning to increase their available supply capacities:
- ANSTO plans to increase available supply capacity from 1,100 to 3,500 6-day Ci per week by mid-2017.
- Mallinckrodt plans to increase Mo-99 supply capacity from 3,500 to 5,000 6-day Ci per week in 2017.
- NTP plans to increase available supply capacity from 3,000 to 3,500 6-day Ci per week in 2017.
One current irradiation services supplier plans to expand its Mo-99 production capacity:
___________________
6 This routine supply is different than ORC, which is intended to meet temporary supply shortages during planned or unplanned shutdowns of other facilities. ORC-based production is not intended to be sustained indefinitely.
- HFR plans to increase its Mo-99 production capacity from 5,400 to 6,200 6-day Ci per week starting in 2017.
Additionally, BR-2 is considering a gradual expansion of its Mo-99 production capacity from 7,800 6-day Ci per week to 10,530 6-day Ci per week (this expansion is not included in Figure 7.1 because it has not been decided).
Two additional European research reactors may become irradiation services suppliers:
- FRM-II, Germany, could start producing Mo-99 in 2018.
- Jules Horowitz Reactor (JHR), France, is expected to start operation in 2020 and could produce Mo-99 thereafter.
And several countries may expand Mo-99 supplies to global or regional markets:
- Russia plans to capture about a 20 percent share of the world Mo-99 market. There is no published schedule for Russia’s expansion plan.7
- South Korea plans to build the Kijang Research Reactor and start producing Mo-99 in 2020 for domestic and eventual regional consumption.
- Argentina plans to build a new reactor with a weekly Mo-99 production capacity of 2,500 6-day Ci to cover domestic and regional supply needs. Argentina plans to have this reactor operational by 2020.
- Brazil plans to build a reactor with a weekly Mo-99 production capacity of 1,000 6-day Ci. This reactor, if built, would start producing Mo-99 after 2021.
Figure 7.1 shows the timeline for these projects. The schedule estimates shown in the figure were provided by suppliers and were not independently verified by the committee. Some of the timelines are likely optimistic. Reactor and radiochemical facility construction and commissioning projects are complex undertakings. These projects often experience unanticipated technical upsets and schedule delays.
The available supply capacity by current Mo-99 suppliers is estimated to be about 15,880 6-day Ci per week during the first three quarters of 2016 (see Table 3.3 in Chapter 3). Nordion’s Mo-99 processing capacity,
___________________
7 It is also unclear at present whether Russia will use HEU or low enriched uranium (LEU) targets to produce Mo-99. See Chapter 5.
currently about 4,680 6-day Ci per week, will drop to zero after October 2016. The scheduled expansions in available supply capacity by ANSTO, Mallinckrodt, and NTP will add about 4,400 6-day Ci per week in 2017, about 300 6-day Ci per week less than the capacity loss from Nordion.
7.1.4 Potential Additions to Available Domestic Supply Capacity
Several private-sector companies in the United States are planning to produce Mo-99 for medical use. These efforts are described in Chapter 4. Three companies have signed cooperative agreements with the Department of Energy’s National Nuclear Security Administration (DOE-NNSA) and are actively working8 to develop domestic Mo-99 supply capabilities:
- General Atomics, in collaboration with Nordion and MURR
- NorthStar Medical Radioisotopes
- SHINE Medical Technologies
___________________
8 Two other companies, Babcock & Wilcox and General Electric-Hitachi, also signed cooperative agreements with NNSA but subsequently suspended their projects. See Chapter 4.
At least five other private-sector companies (also described in Chapter 4) are actively planning to develop domestic Mo-99 supply capabilities:
- Coquí RadioPharmaceuticals
- Eden Radioisotopes
- Flibe Energy
- Niowave
- Northwest Medical Isotopes
The NNSA-supported Mo-99 production projects are further along the development path than are production projects initiated by the private sector. As discussed in Chapter 4, none of these projects will be supplying Mo-99 to domestic markets by October 2016 when the NRU reactor ceases routine production. One project (NorthStar) may be supplying Mo-99 to domestic markets in 2018. It is unclear whether any of the five other private-sector projects will ever be completed (see Chapter 4).
7.1.5 Other Potential Sources of Mo-99/Tc-99m Supply
As discussed in Chapter 3, at least two of the four Canadian government-funded projects for Tc-99m production (Canadian Isotope Innovations and Advanced Cyclotron Systems) intend to supply Tc-99m to the U.S. market. However, these suppliers must obtain Food and Drug Administration approval to sell cyclotron-produced Tc-99m in the United States, and they will have to demonstrate the ability to produce this isotope reliably and cost-effectively. Even if successful, these suppliers are unlikely to provide more than a few hundred 6-day Ci per week to the U.S. market during the contingency period.
7.2 OECD-NEA ANALYSIS OF Mo-99 SUPPLY AND DEMAND
OECD-NEA has published several reports on Mo-99/Tc-99m market demand and projections (OECD-NEA, 2011b, 2012, 2014b, 2015, 2016). These reports are intended to inform policy makers, Mo-99 supply chain participants, and the medical community about high-risk periods for Mo-99 supply shortages. High-risk periods are defined in the latest reports as the periods when supply is near or below demand plus a 35 percent ORC. The OECD-NEA-recommended 35 percent ORC is based on the (n–1) criterion established by the High-level Group on the Security of Supply of Medical Radioisotopes (HLG-MR) (see Chapter 3):
In the HLG-MR principles, it was proposed that a processor should hold sufficient paid reserve capacity to replace the largest supplier of irradiated
targets in their supply chain and likewise participants further down the supply chain should hold similar levels of ORC. This is the so-called (n-1) criterion. (OECD-NEA, 2015, p. 7)
The 2016 OECD-NEA report presents three scenarios for Mo-99 capacity and supply for the period 2016-2021. These scenarios are described in Sidebar 7.1. OECD-NEA concluded that
the current irradiator and processor supply chain should be sufficient, and if well maintained, planned, and scheduled, be able to manage an unplanned outage of a reactor or a processor [in the 2016-2021 period]. (OECD-NEA, 2016, p. 21)
OECD-NEA also concluded that, starting in 2017, the capability to manage an unplanned outage will be reduced because of the planned exit of NRU/Nordion from the market. The organization highlighted the need to add irradiation and processing capacity by 2017 to offset this reduced capacity.
7.3 FINDING AND RECOMMENDATION
The committee analyzed the adequacy of Mo-99 supplies to meet domestic demand until about 2021 using the information provided in Chapters 3-6. The results of this analysis are presented below in one finding and one recommendation:
FINDING 4B: Global supplies of molybdenum-99 are adequate at present to meet U.S. domestic needs. However, available supply capacity will be reduced substantially after October 2016 when the Canadian supplier shuts down, and supply capacity could be reduced further in 2017-2018 when European suppliers convert to low enriched uranium targets and the Australian supplier starts up a new target processing facility, especially if these suppliers encounter conversion and/or start-up delays. The committee judges that there is a substantial (>50 percent) likelihood of severe molybdenum-99/technetium-99m supply shortages after October 2016, lasting at least until current global suppliers complete their planned capacity expansions.
RECOMMENDATION 4B: The U.S. government should continue to work with the Canadian government to ensure that there is an executable and well-communicated plan in place to restart Canadian supply of molybdenum-99 after October 2016.
Current available supply capacity, about 15,880 6-day Ci of Mo-99 per week, is more than adequate at present to meet current global (~9,000 6-day Ci per week) and U.S. (~50 percent of global) demand. However, this supply capacity will be reduced to about 11,200 6-day Ci per week when NRU stops producing Mo-99 after October 2016. This represents about a 25 percent ORC, well below the 35 percent ORC recommended by OECD-NEA. Planned expansions by current suppliers, if realized, would restore most of the available supply capacity lost after October 2016 (see Figure 3.3 in Chapter 3).
A number of efforts are under way by current global Mo-99 suppliers and potential new domestic Mo-99 suppliers to fill the expected supply gap (see Section 7.1.3 in this chapter). The organizations responsible for these efforts have put forward what the committee considers to be best-case schedules for bringing new Mo-99 supplies to market. Several of these schedules have already slipped during the course of this study; additional slippage would be entirely unsurprising.
The number of irradiation services suppliers will be reduced from seven to six after NRU stops producing Mo-99. Four of the remaining suppliers use reactors that are over 50 years old (BR-2, HFR, LVR-15, and SAFARI-1), and one supplier also uses a reactor that is over 40 years old (Maria) (see Table 3.2 in Chapter 3). Irradiation services suppliers put great emphasis on maintenance so that their reactors can continue to operate safely and reliably. However, the potential for unplanned reactor maintenance outages increases as reactors age. Indeed, several of the reactors used to produce Mo-99 have already had unplanned and extended outages for major repairs (see Table 3.6 in Chapter 3). Such outages may well occur in the future.
Unplanned outages of Mo-99 suppliers could also reduce available supply capacity below current global demand. The number of global Mo-99 suppliers will be reduced from five to four after Canada stops supplying Mo-99. Three of these suppliers (IRE, Mallinckrodt, and NTP) are capable of supplying one-third or more of current global demand for Mo-99 (see Table 3.2 in Chapter 3). The loss of any one of these suppliers after October 2016 could result in severe global Mo-99 supply shortages.
Three global Mo-99 suppliers are currently making substantial modifications to their target processing facilities:
- IRE and Mallinckrodt are converting their facilities to process LEU targets and will be running two parallel processing lines (one for LEU targets and one for HEU targets) in 2017 and possibly 2018 (Chapter 5).
- ANSTO is constructing a new target processing facility (completion is planned for mid-2017) and will be implementing a target dis
solution and Mo-99 recovery process in that facility that contains elements of ANSTO’s and NTP’s current processes (see Chapter 3).
The potential for unexpected supply disruptions increases any time a supplier moves to a new facility or implements a new process. For example, ANSTO encountered a several-month delay in starting up its current target processing facility. IRE and Mallinckrodt have encountered unexpected delays in converting to low enriched uranium targets.9 Such delays are a normal part of the start-up process for complex facilities but are difficult to anticipate or schedule.
IRE, Mallinckrodt, and NTP expect to lose about 20 percent10 of their available supply capacity after conversion to LEU because of reduced uranium-235 (U-235) loadings and increased neutron capture (see Section 5.2 in Chapter 5). They are planning to increase target throughputs and make other changes to restore this lost capacity. Delays in these efforts could further reduce supply capacities.
NTP and ANSTO rely on one reactor each (SAFARI-1 and OPAL, respectively) for all of their target irradiations. They have no backup irradiation services suppliers. Consequently, their entire available supply capacity will be lost whenever these reactors shut down, whether for planned or unplanned maintenance. The reactor operators coordinate the planned outages for these reactors to minimize potential supply disruptions. However, unplanned outages of one of both of these reactors could result in severe supply shortages, especially if the outages extend over multiple weeks.
The committee agrees in principle with OECD-NEA that
the current irradiator and processor supply chain should be sufficient, and if well maintained, planned, and scheduled, be able to manage an unplanned outage of a reactor or a processor [in the 2016-2021 period]. (OECD-NEA, 2016, p. 21)
However, the committee judges that it will be difficult to achieve a “well-maintained, planned, and scheduled supply chain” in light of the factors discussed above. The committee therefore finds (Finding 4B above) that there is a substantial (>50 percent) likelihood of severe molybdenum-99/technetium-99m supply shortages after October 2016, lasting at least until the planned production expansions described in Section 7.1.3 of this chapter are completed. In particular, unplanned outages at IRE, Mallinckrodt, or NTP would likely cause severe Mo-99 supply shortages, and delays by
___________________
9 NTP, which already produces a portion of its supply with LEU targets, also encountered unexpected delays.
10OECD-NEA (2016) currently estimates production capacity losses to be 10 percent because some steps have already been taken by suppliers to overcome them.
ANSTO in expanding its available supply capacity will extend the period of global supply vulnerability. This period of vulnerability could last into 2018 and possibly beyond if there are substantial delays in completing these production expansions.
The committee recommends (see Recommendation 4B above) that the U.S. government should continue to work with the Canadian government to ensure that there is an executable and well-communicated plan in place to restart emergency production of molybdenum-99 in Canada if there are extended unplanned facility outages. The committee is particularly concerned about the potential for unplanned outages in global Mo-99 supplier facilities and the impacts of those outages on global Mo-99 supplies.
If such unplanned outages occur, the Canadian government might have only 1 week or at most 2 weeks to restart Mo-99 production in NRU to avoid severe global Mo-99 supply shortages. The decision and consultation processes will have to be well established and practiced to execute on this schedule. Of course, once a restart decision was made, NRU/Nordion will have to be operationally ready to resume Mo-99 production and supply. This will require that both organizations maintain an adequately sized and trained staff to operate the target irradiation and processing facilities during the contingency period.














