3
Focus on Nanomanufacturing
Nanomanufacturing is a focus area of nanotechnology related to advanced development and commercialization that warrants special attention. It also is an area that is integrally related to other high-profile federal initiatives focused on advanced manufacturing.
Nanomanufacturing is a specialized aspect of advanced manufacturing involving nanoscale materials and processes.
The President’s Council of Advisors on Science and Technology (PCAST) has identified manufacturing as a matter of fundamental importance to the United States—economically and, in some sectors, for national security.1 In June 2011, PCAST released the report Ensuring American Leadership in Advanced Manufacturing.2 Shortly thereafter, President Obama launched the Advanced Manufacturing Partnership, a national effort bringing together industry, universities, and the federal government to invest in the emerging technologies, such as nanotechnology, that will create high-quality manufacturing jobs and enhance U.S. competitiveness. In February 2012, the National Science and Technology Council (NSTC) released A National
___________________
1 See Advanced Manufacturing National Program Office, “Advanced Manufacturing,” http://www.manufacturing.gov/advanced_manufacturing.html, accessed September 12, 2015.
2 President’s Council of Advisors on Science and Technology (PCAST), 2011, Report to the President on Ensuring American Leadership in Advanced Manufacturing, Washington, D.C., June, https://www.whitehouse.gov/administration/eop/ostp/pcast/docsreports.
Strategic Plan for Advanced Manufacturing,3 which was followed in October 2014 by the PCAST report Accelerating U.S. Advanced Manufacturing.4
Motivation for this concerted effort was the precipitous decline in U.S. manufacturing jobs, facilities, and infrastructures in many key sectors. For example, the United States lost 5 million manufacturing jobs between January 2000 and December 2014.5 Also, the United States is no longer the lead producer of manufactured goods. Today, Chinese-based businesses lead the world in total output of manufactured goods, with $2.3 trillion compared to $1.8 trillion from U.S.-based businesses.6 While the speed at which manufacture of many items moved overseas has been alarming, there are signs that reports of the demise of U.S. manufacturing may be premature. Although non-U.S. competitors are gaining, the United States still leads the world in high-tech manufacturing—for example, aircraft, spacecraft, communication products, computers, pharmaceuticals, semiconductors, and technical instruments—and U.S. manufacturing job growth is currently the highest it has been in decades.
Traditionally, manufacturing refers to making or producing something on a large scale. During the past few decades, manufacturing has evolved from the basic concept of simply “making things” into a complex value chain of global ecosystems that covers the entire life cycle of a product, from research and development and product design to manufacturing, software, applications, and disposal or recycling. A great deal of new science and engineering knowledge will be necessary to realize processes that (1) facilitate manufacturing of nano-enabled products with reliable specification tolerances, (2) have an acceptable cost, and (3) are compatible with value chain requirements. As technologies and ecosystems continue to evolve, policy makers and practitioners will need to develop standards, rules, and regulations associated with manufacturing environmental, health, and safety issues.
NANOMANUFACTURING AS A PILLAR OF THE NATIONAL NANOTECHNOLOGY INITIATIVE
Development of the capability to manufacture nanoscale materials and devices, as well as their incorporation into products, is key to realizing the potential benefits of nano-enabled technology for society. National Nanotechnology Initiative (NNI)
___________________
3 National Science and Technology Council (NSTC), 2012, A National Strategic Plan for Advanced Manufacturing, Executive Office of the President, Washington, D.C.
4 PCAST, 2014, Report to the President: Accelerating U.S. Advanced Manufacturing, Washington, D.C.
5 Office of the Press Secretary, “President Obama Launches Competition for New Textiles-Focused Manufacturing Innovation Institute; New White House Supply Chain Innovation Initiative; and Funding to Support Small Manufacturers,” Fact Sheet, release date March 18, 2015, https://www.whitehouse.gov/briefing-room/statements-and-releases.
6 R.E. Scott, 2015, Manufacturing Job Loss, Economic Policy Institute, http://www.epi.org/publications/.
leaders recognized the importance of nanomanufacturing from the outset, holding a number of NNI-sponsored workshops in the 2002 to 2004 time frame.7,8 The 2007 NNI report Manufacturing at the Nanoscale lists the following major areas as essential to expedite progress in nanomanufacturing:
- Research for hierarchical nanomanufacturing. Hierarchical integration will be used across dimensional scales, from atoms to molecules to the human length scale, to incorporate nanostructures into microscale architectures and macroscale products. Bottom-up, directed molecular or particulate assembly techniques will need to be combined with top-down, high resolution, and high-speed macroscopic fabrication techniques. Various hierarchical systems architectures will create various technology platforms for nanomanufacturing.
- Infrastructure development. There is a need for geographically distributed nanomanufacturing research centers and user facilities with a variety of manufacturing tools to allow work on systems. These centers and shared facilities should network with existing nanoscience centers (e.g., those funded by the National Science Foundation [NSF], the Department of Energy [DOE], the National Institute of Standards and Technology [NIST], and the Department of Defense), serve as a resource for technology transfer for small and large business, and facilitate education and workforce training.
- Modeling, simulation, and design. Current molecular dynamics models are limited in time and space, such that prediction of realistic manufacturing processes is not feasible. New multiscale models need to be developed that can predict both yield and performance. Design tools using these multiscale models, equivalent to computer assisted design or finite element analysis, are needed to enable rapid product development.
- Tool development. New metrology tools and manufacturing tools are needed to measure and manipulate nanostructures and nanocomponents, with an emphasis on in-line, real-time manufacturing rate capabilities to ensure high yield and precision.
- Environmental and occupational health and safety. In order to realize the benefits of nanomanufacturing, it is necessary to better understand the ramifications for workers, users, and the environment of health, safety, and environmental issues related to nanomaterials, nanomanufacturing processes, and nanotechnology-based products and their disposal. Any potential issues or problems should be addressed proactively.
- Education and societal impact. The new nanotechnology-based processes will likely continue the manufacturing trend of decreasing physical and increasing information-processing requirements. An appropriately educated workforce, both for making the next-generation discoveries and for operating the nanomanufacturing processes, is vital to the continued economic success of the country. In addition, educating the general public about nanotechnology and nanomanufacturing is critical to achieving acceptance and realization of the promise of nanotechnology, its capabilities and risks.9
___________________
7 Chemical Industry Vision2020 Technology Partnership, 2003, Chemical Industry R&D Roadmap for Nanomaterials by Design: From Fundamentals to Function, Energetics, Inc., December, http://www.nanowerk.com/nanotechnology/reports/reportpdf/report17.pdf.
8 NSTC, 2007, Manufacturing at the Nanoscale: Report of the NNI Workshops 2002-2004, Arlington, Va., http://www.nano.gov/node/246.
9 NSTC, 2007, Manufacturing at the Nanoscale, p. vii.
The 2003 Chemical Industry R&D Roadmap for Nanomaterials by Design report had similar recommendations to Manufacturing at the Nanoscale, with one additional top priority worth noting: “develop unit operations and robust scale-up and scale-down methodologies for manufacturing.”10
The Nanoscale Science, Engineering, and Technology (NSET) agencies initiated a number of efforts to address these recommendations, including the Nanomanufacturing Program in the National Science Foundation (NSF) Engineering Directorate (established in 2002), four NSF-funded Nanoscale Science and Engineering Centers (NSECs), and the National Institute of Standards and Technology (NIST) National Nanomanufacturing and Nanometrology Facility (see Table 3.1).
In 2006, the NNI initiated a program component area (PCA) in nanomanufacturing, including all means that have the capability to reproducibly transform matter—from a bulk form and from individual atoms, molecules, and supramolecular structures—into nanoscale or nanostructured materials, devices, or systems with desired properties and performance characteristic typically in large quantities. Additionally, the PCA on nanomanufacturing included the capability to integrate such nanoscale materials and devices into systems spanning nanoscale to macroscale dimensions. The funding evolution for that PCA is shown in Table 3.2.
In 2011, the NNI complemented the Nanomanufacturing PCA with a Nanotechnology Signature Initiative (NSI), “Sustainable Nanomanufacturing—Creating the Industries of the Future,” and in 2013 dropped nanomanufacturing as a PCA separate from the NSI. The NSI is to accelerate the development of industrial-scale methods for manufacturing functional nanoscale systems. The two key thrusts of this initiative are (1) design of scalable and sustainable nanomaterials, components, devices, and processes and (2) development of nanomanufacturing measurement technologies (metrologies).
Resources and support from the NNI and its participating agencies are needed to assure continuing U.S. leadership in nanomanufacturing. But there are signs that those resources may not be forthcoming under the NNI. The 2014 update to the NNI strategic plan eliminated the objective under Goal 2 to “develop robust, scalable nanomanufacturing methods necessary to facilitate commercialization by doubling the share of the NNI investment in nanomanufacturing research over the next five years.”11 A review of the annual budget reports shows an apparent drop in nanomanufacturing funding from 2012 to 2013. “Apparent” because it appears
___________________
10 Chemical Industry Vision2020Technology Partnership, 2003, Chemical Industry R&D Roadmap for Nanomaterials By Design, p. 33.
11 For an example that includes the now removed statement, see NSTC, 2011, National Nanotechnology Initiative Strategic Plan, Washington, D.C., https://www.whitehouse.gov/sites/default/files/microsites/ostp/nni_strategic_plan_2011.pdf.
TABLE 3.1 Centers Specifically Directed Toward Nanomanufacturing
| Year | Center/Facility |
|---|---|
| 2003 | Scalable and Integrated Nanomanufacturing, University of California, Los Angeles—now University of California, Berkeley |
| Nanoscale Chemical-Electrical-Mechanical Manufacturing Systems Center, University of Illinois at Urbana-Champaign | |
| 2004 | High-Rate Nanomanufacturing, Northeastern University |
| 2005 | Hierarchical Nanomanufacturing, University of Massachusetts, Amherst |
| National Nanomanufacturing and Nanometrology Facility, National Institute of Standards and Technology |
TABLE 3.2 NNI Agency Nanomanufacturing Investment (in $millions)
| Agency | Program Component Area Year | Signature Initiative Year | 2017 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 2006 | 2007 | 2008 | 2009 | 2010 | 2011 | 2012 | 2013 | 2014 | 2015 | 2016 Est. | Prop. | |
| NSF | 20.3 | 26.6 | 20.7 | 21.9 | 21.4 | 44.8 | 44.4 | 22.7 | 30.7 | 34.1 | 26.4 | 28.4 |
| NIST | 6.9 | 12.4 | 10.8 | 10.6 | 27.2 | 14.6 | 9.2 | 3.0 | 5.6 | 6.1 | 5.4 | 4.9 |
| DOD | 3.1 | 7.5 | 7.8 | 25.3 | 26.4 | 24.3 | 42.2 | 3.7 | 2.3 | 0.9 | 0.5 | 0 |
| DOE | 0.5 | 0.5 | 6.8 | 4.9 | 6.5 | 5.0 | 0 | 0 | 0 | 0 | 0 | 0 |
| HHS/NIH | 1.7 | 0.8 | 0.7 | 0.7 | 0.8 | 1.3 | 5.7 | 0 | 1.0 | 1.0 | 1.0 | 1.0 |
| USDA/FS | 0.2 | 0.2 | 0.2 | 0.2 | 0.2 | 1.0 | 1.0 | 1 | 1.2 | 1.2 | 1.5 | 1.5 |
| USDA/NIFA | 0 | 0.1 | 0.1 | 0.2 | 0.2 | 0 | 0 | 1.2 | 0.6 | 1.0 | 1.0 | 1.0 |
| NASA | 1.0 | 0 | 0 | 0 | 2.1 | 1.0 | 2.6 | 3.2 | 5.9 | 0.6 | 0.9 | 0.6 |
| Total PCA | 33.7 | 48.1 | 47.1 | 63.8 | 84.8 | 92 | 105.1 | |||||
| Total NSI | 61 | 56 | 34.8 | 47.2 | 44.9 | 36.7 | 37.4 | |||||
NOTE: NSF, National Science Foundation; NIST, National Institute of Standards and Technology; DOD, Department of Defense; DOE, Department of Energy; HHS/NIH, Health and Human Services/National Institutes of Health; USDA/FS, U.S. Department of Agriculture—Forest Service; USDA/NIFA, United States Department of Agriculture—National Institute of Food and Agriculture; NASA, National Aeronautics and Space Administration.
SOURCE: Data from the “actual” data in the NNI Supplements to the President’s Budget, available at http://www.nano.gov/node/1071, unless otherwise designated.
the NSET funding agencies relabeled some of the efforts from manufacturing into other PCAs or signature initiatives. For instance, in the 2013 NNI supplement, the Department of Defense (DOD) reports 10 Multidisciplinary University Research Initiatives (MURIs) in 2011 as supporting nanomanufacturing (see Box 3.1). MURI efforts are on the order of $1 million to $1.5 million each and last 5 years (with rare exceptions). But the reported actual DOD support for nanomanufacturing drops from $42 million in 2012 to $3.7 million in 2013, which is not enough monies to
account for those 10 MURIs alone. Such changes in accounting call into question the credibility of the reported numbers as a means to assess the NNI investment in nanomanufacturing.
Finding 3.1: Budget figures in support of nanomanufacturing as reported in the NNI supplements to the president’s budget have been inconsistent, and progress made toward recommendations of the 2007 Manufacturing at the Nanoscale report is not clear.
Recommendation 3.1: The Nanoscale Science, Engineering, and Technology Subcommittee should prepare a report that provides a self-consistent record of the NNI nanomanufacturing program, the status relative to the recommendations of the 2007 Manufacturing at the Nanoscale report, and the NNI plans to move forward.
BASIC RESEARCH IN SUPPORT OF VIABLE NANOMANUFACTURING
Several government agencies sponsor programs in fundamental research on nanomanufacturing methods and techniques, including research on methods for retaining nanoscale properties when scaling up for use in larger products and
components. Examples include the NSF’s core Sustainable Nanomanufacturing Program; the NSF nanoscale science and engineering centers, which includes the National Nanomanufacturing Network (NNN); and the Defense Advanced Research Projects Agency’s (DARPA’s) Atoms to Product (A2P) Program.
Scalable Nanomanufacturing Program (NSF)
Initiated in 2002, the NSF nanomanufacturing program supports fundamental research in novel methods and techniques for batch and continuous processes and top-down (addition/subtraction) and bottom-up (directed self-assembly) processes leading to the formation of complex heterogeneous nanosystems. In 2011, this program was followed by the Scalable Nanomanufacturing Program,12 which supports basic research in nanostructure and process design principles, integration across length-scales, and system-level integration. The program leverages advances in the understanding of nanoscale phenomena and processes (physical, chemical, electrical, thermal, mechanical, and biological), nanomaterials discovery, novel nanostructured architectures, and new nanodevice and nanosystem concepts. It seeks to address quality, efficiency, scalability, reliability, safety, and affordability issues that are relevant to manufacturing. To address these issues, the program encourages research on processes and production systems based on computation; modeling and simulation; use of process metrology; sensing, monitoring, and control; and assessment of product (nanomaterial, nanostructure, nanodevice, or nanosystem) quality and performance. The program also supports education of the next generation of researchers and encourages building a workforce trained in nanomanufacturing systems. It is also interested in understanding long-term environmental, health, and safety (EHS) implications of large-scale production and use of nanoscale materials, devices, and systems.
Nanoscale Science and Engineering Centers/National Nanomanufacturing Network
Nanomanufacturing collaborations have been the focus of the NNN. The NNN was started in 2006 when NSF funded the Center for Hierarchical Manufacturing (CHM). The four NSECs (see Table 3.1) became the core NNN, which today includes the DOE Center for Integrated Nanotechnologies at Sandia National Laboratory and the NIST Center for Nanoscale Science and Technology.
According to the NSF nanomanufacturing program director, the work funded through the NNN NSECs is one step closer (up to technology readiness level [TRL] 3)
___________________
12 National Science Foundation, “Scalable Nanomanufacturing (SNM) Program Solicitation,” https://www.nsf.gov/publications/pub_summ.jsp?ods_key=nsf16513, accessed September 1, 2016.
to commercialization than basic science (TRL1-2).13 For example, the NNN led the effort to create the International Organization for Standardization (ISO) Technical Committee 229 Nanomanufacturing Terminology Standard, a project involving participants from 31 countries. This document, OSI/TS 80004-8,14 lists 156 terms and definitions focusing on various types of nanomanufacturing processes. Standard terminology is key to any industry, and having this new ISO standard enables everyone to speak the same nanomanufacturing language. Processing tools were developed, such as the cluster tool for directed assembly and transfer developed at the Center for High-Rate Nanomanufacturing. The CHM provides tool sets that its partners can use to demonstrate process feasibility and scalability.15,16 The NNN also sponsored a series of workshops on nanoinformatics, an enabling technology for process control, streamlined product and manufacturing design, and experimental design and analysis.
NNN research results, along with other nanomanufacturing related information, have been curated on the website InterNano.org. The database of processes for materials, devices, and structures is particularly informative. However, the future of InterNano.org and access to the information it contains is uncertain because funding for the last NSEC of the NNN is scheduled to end in 2016.
Atoms to Product Program (DARPA)
In late 2015 the DARPA Defense Science Office launched its A2P program, with the goal of developing technologies and processes to assemble nanometer-scale pieces—whose dimensions are near the size of atoms—into systems, components, or materials that are at least millimeter-scale in size. At the heart of that goal was a frustrating reality: Many common materials, when fabricated at nanometer-scale, exhibit unique and attractive “atomic-scale” behaviors—including quantized current-voltage behavior, dramatically lower melting points, and significantly higher specific heats—but they tend to lose these potentially beneficial traits when they are manufactured at larger “product-scale” dimensions, typically on the order of a few centimeters, for integration into devices and systems. This effort directly
___________________
13 K. Cooper, “NSF Nanomanufacturing Program Activities,” presentation to the committee on September 9, 2015.
14 International Organization for Standardization, 2013, “Nanotechnologies: Vocabulary: Part 8: Nanomanufacturing Processes,” ISO/TS 80004-8:2013, http://www.iso.org/iso/iso_catalogue/catalogue_tc/catalogue_detail.htm?csnumber=52937.
15 M. Tuominen, “The National Nanomanufacturing Network,” presentation to the committee on September 9, 2015.
16 University of Massachusetts, Amherst, Center for Hierarchiacal Manufacturing, “About CHM,” http://chm.pse.umass.edu/, accessed September 18, 2015.
addresses one of the high-priority recommendations of the Chemical Industry R&D Roadmap for Nanomaterials by Design report.17
Finding 3.2: Basic research programs focused on nanomanufacturing have been a strength of the NNI. NSF centers focused on nanomanufacturing have more adequate budgets for facilities and education than do single investigators who have smaller awards. Ending support for nanomanufacturing centers will lead to a decrease in coordinated education and facility efforts.
Recommendation 3.2: The National Science Foundation should find ways to continue some nanomanufacturing center-scale efforts. Such centers might be explicitly tasked to pursue early-stage research in support of advanced manufacturing programs, such as the Manufacturing Innovation Institutes.
APPLIED RESEARCH AND DEVELOPMENT IN SUPPORT OF NANOMANUFACTURING
Applied research and development is by definition aimed at creating new systems to address a targeted need. Such efforts generally are agnostic as to the technical solution, nanotechnology, or otherwise. However, nanotechnology can be part of the solution to many system-level challenges. Therefore, more applied federal programs can benefit from focused investment by the NNI. There are several examples of such programs. The semiconductor industry is actively partnering with federal agencies to fund research that will lead to future nano-enabled information technologies.18 DOD and NASA are invested in the manufacturing of nanostructures for structural materials and coatings; the U.S. Department of Agriculture Forest Service is investing in the manufacture of innovative nanocellulose products; and DOE is pursuing the manufacture of nanostructures for renewable energy conversion and storage. The National Institutes of Health is investing in biologic nanoparticles and nanoparticle-based drugs to fight cancer and other diseases.
To regain manufacturing momentum and grow jobs for the United States, in 2012 the U.S. government initiated a national manufacturing initiative and established a National Advanced Manufacturing National Program Office at NIST. Two key components of the initiative are (1) fostering public-private partnership supported Manufacturing Innovation Institutes (MIIs) and (2) the creation of the NIST Advanced Manufacturing Consortia Program (AMTech). The MIIs are an
___________________
17 Chemical Industry Vision 2020 Technology Partnership, 2003, Chemical Industry R&D Roadmap for Nanomaterials By Design.
18 See the Nanoelectronics Research Initiative and STARnet research programs, which are collaborations with federal agencies and the Semiconductor Research Corporation.
example where “pull” from a higher TRL program can provide focus for the NNI. Each MII has a unique focus (see Box 3.2), with a common goal to create, showcase, and deploy new capabilities and new manufacturing processes. The institutes seek to bridge the funding gap (manufacturing readiness level [MRL] 4 to 7) for applied research and development. These institutes are coordinated by the Advanced Manufacturing National Program Office located at NIST and serve as a point of private-public collaboration for suppliers, schools, colleges, and other organizations to develop and scale particular manufacturing technologies and processes.
The creation of an MII focused solely on nanomanufacturing was considered but not pursued; however, nearly all of the existing MIIs will benefit from—or even depend on—nanotechnology or nanomanufacturing advances. NNI sponsored research is a source of TRL 1-3 concepts for these institutes.
Advanced Manufacturing Technology Program
AMTech, launched in 2013, aims to establish new, or strengthen existing, industry-driven consortia that address high-priority research challenges imped-
ing the growth of advanced manufacturing in the United States. The AMTech program funds broad participation across the value chain including companies of all sizes, universities, and government agencies. After two rounds of funding, there are 35 planning awards, totaling ~$17 million, to identify critical gaps in advanced manufacturing technology infrastructure and create industry-driven technology roadmaps for addressing those gaps. Each award supports an industry-driven consortium to develop research plans and chart collaborative actions to solve high-priority technology challenges and to accelerate the growth of advanced manufacturing in the United States. As with the MIIs, many of the AMTech-funded projects address topics in which nano-enabled technology will be beneficial, if not critical.
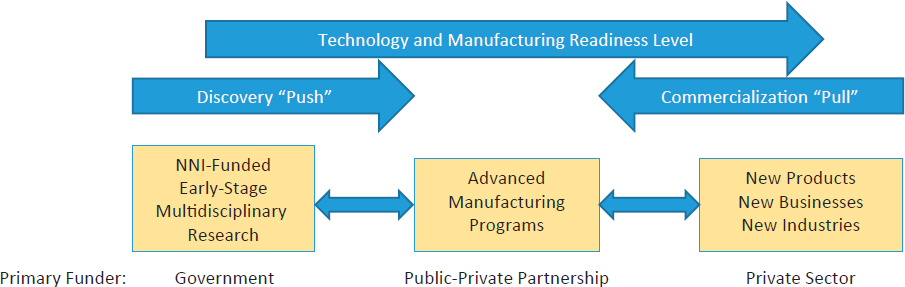
The relationship between NNI-funded basic research and the more applied research and development programs described above is illustrated in Figure 3.1. As discussed in Chapter 2, the push of new ideas as well as the pull of product-based needs both contribute to innovation. Connections between the NNI and advanced manufacturing programs such as the MII program and AMTech can accelerate progress toward the goals of those programs.
Finding 3.3: In many cases, progress or success in the MIIs and in implementation of the roadmaps developed under the AMTech program will require advances in nanomanufacturing.
Recommendation 3.3a: NNI-participating agencies should explicitly support the early-stage (technology readiness level 1-3) nanomanufacturing research needed to enable the roadmaps and goals of current advanced manufacturing programs, in particular the existing Manufacturing Innovation Institutes.
Recommendation 3.3b: The Nanoscale Science, Engineering and Technology Subcommittee should form a nanomanufacturing working group to identify nanoscale research needs of advanced manufacturing, coordinate efforts between the NNI and the federal programs focused on advanced manufacture, and foster greater investment by those programs in nano-enabled technologies.
The roles of the nanomanufacturing working group could include the following.
- Engage with the NSTC Subcommittee on Advanced Manufacturing to inform the subcommittee about implications of nanotechnology in various areas of manufacturing.
- Work with the MII and AMTech consortia to understand their TRL 4-7 roadmaps and identify and support relevant underlying TRL 1-3 nanoscale research needs.
- Work with the various mission agency leaders and managers responsible for the applied research and advanced development programs to identify opportunities for, and encourage investments in, nanotechnology-enabled technology solutions.
- Identify and report as part of the NNI annual report activities under way within the NNI-participating agencies in support of MIIs.
Other existing federal programs that target manufacturing could be—and in some cases are being—used to advance nanomanufacturing technology. As noted in Chapter 2, several agencies have called for Small Business Innovation Research/Small Business Technology Transfer proposals related to nanotechnology. More specific to nanomanufacturing, the DOD Defense Production Act Title III, a DOD-wide program under the Deputy Assistant Secretary of Defense, Manufacturing and Industrial Base Policy, has been used to initiate several nanomanufacturing facilities (see Table 3.3 and Box 3.3).
Other programs that could support nanomanufacturing research and development are the DOD MANTECH programs,19 the Defense-wide Manufacturing Science and Technology Program, the Air Force Research Laboratory Manufacturing and Industrial Technologies program, the DOE Office of Energy Efficiency and Renewable Energy Advanced Manufacturing Program, the DOE Technology Commercialization Fund,20 the Oak Ridge National Laboratory Manufacturing Demonstration Facility,21 and the NASA Space Technology Mission Directorate.
___________________
19 Department of Defense, ManTech, https://www.dodmantech.com/, accessed August 16, 2016.
20 Department of Energy, “Technology Commercialization Fund,” http://energy.gov/technologytransitions/technology-commercialization-fund, accessed August 16, 2016.
21 Oak Ridge National Laboratory, “Manufacturing Demonstration Facility,” http://web.ornl.gov/sci/manufacturing/mdf/, accessed August 16, 2016.
TABLE 3.3 Defense Production Act Title III Projects
| Facility | Year | Company | Government Funds ($million) | Company Cost Share ($million) |
|---|---|---|---|---|
| Advanced Carbon Nanotube Volume Production | 2011 | Nanocomp Technologies | 22 | 9.2 |
| Conductive Composites (Nickel-based nanomaterials) | 2011 | Conductive Composites | 10.2 | 2.8 |
| Atomic Layer Deposition Hermetic Coatings | 2007 | Raytheon RF Component | 5.4 | 0.5 |
| Hybrid Plastics and POSS Nanotechnology | 2005 | Hybrid Plastics | 21.2 | 2.2 |
NOTE: Last column is the company contribution.
SOURCE: See the “Defense Production Act Title III” website, maintained by Northrop Grumman Technology Services, Beavercreek, Ohio, http://www.dpatitle3.com/dpa_db/index.php, accessed September 12, 2015.
NANOMEDICINE MANUFACTURING: AN EMERGING OPPORTUNITY
Many biological structures and processes are inherently nanoscale, and nanotechnology research related to medicine and health applications is starting to bear fruit. For example, some drugs, including cancer-fighting drugs such as Docetaxel, are more soluble and move through cell walls and membranes to disease sites
more easily if they are nanoscale.22 Nanomedical breakthroughs include increasing success in gene therapy and development of new antiviral vaccines (hepatitis C, pneumonia). Most of these novel disease-fighting solutions are still in early stages; in 2015, there were more than 250 nanomedicine projects in clinical trials.23
At the same time, three relatively new federal initiatives address biologically inspired challenges—the BRAIN Initiative, Precision Medicine, and the “moonshot” program to find a cure for cancer announced in the 2016 State of the Union address. Tools and techniques for nanoscale synthesis and characterization will be important for research under these initiatives and nanotechnology-based diagnostic and therapeutic products are likely outputs.
The 2014 PCAST assessment of the NNI24 includes an appendix on “Manufacturing Nanomedicine” that states the following:
A significant emphasis must be placed early in the commercialization pathway on refining or replacing laboratory fabrication procedures with reliable, consistent, and economically viable manufacturing methods that can be scaled up for clinical development and, ultimately, to reliably generate commercial drug supply. Start-ups frequently must focus considerable time and capital on developing these methods. The need to scale up is likely to occur early on the product development timeline since animal testing in the relevant disease models and understanding of how these nanotherapies are distributed in the body are required for making the decision to proceed toward clinical development.
Another important consideration in manufacturing nanomedicines is the need to conduct preclinical toxicity studies and any subsequent clinical trials using drug supply generated under Good Manufacturing Practice (GMP) conditions in an approved facility. Thus this investment must be made prior to knowing whether the nanomedicine will be effective in humans for its intended indication. Depending on the novelty and complexity of the manufacturing process, there may be few options to source the manufacturing to outside parties. In the absence of established nanomanufacturing facilities in which to explore methods, for preclinical studies, complete method validation, or deploy a GMP-based manufacturing scale-up protocol, the start-up has no alternative but do this internally. Clearly, access to manufacturing facilities for scaling up nanomedicines in the amounts necessary for animal testing and preclinical development would accelerate the transition of these novel therapies to proof-of-concept human testing in clinical development.25
With more than 250 nanomedicines in clinical trials, now is the time to expand the study of nanomedicine manufacturing. Although many scale up and manufac-
___________________
22 J.S. Murday, R.W. Siegel, J. Stein, and J.F. Wright, 2009, Translational nanomedicine: Status assessment and opportunities, Nanomedicine: Nanotechnology, Biology, and Medicine 5:251-273.
23 M. Tomczyk, 2015, Nanoinnovation: What Every Manager Needs to Know, Wiley-VCH Verlag GMBH, Weinheim, Germany.
24 PCAST, 2014, Report to the President and Congress on the Fifth Assessment of the National Nanotechnology Initiative, Executive Office of the President, https://www.whitehouse.gov/sites/default/files/microsites/ostp/PCAST/pcast_fifth_nni_review_oct2014_final.pdf.
25 Ibid, p. 67.
turing hurdles for a new nanomedicine are similar to those of any new therapeutic, manufacturing nanomedicines poses special issues. Perhaps the most prevalent is the need for nanoscale characterization at all stages of discovery, development, and commercialization. In addition, nanomaterials that meet medical-grade requirements for purity and reproducibility can be difficult to obtain. It is not uncommon for expensive nanomedicines, produced under Food and Drug Administration (FDA) current Good Manufacturing Practices (cGMPs), to not meet specifications or give poor efficacy reproducibility. Safety extends not only to dosage and clinical results, but also to manufacturing and the need to avoid contamination and toxicological factors.
The United States currently leads the world in nanomedicine. In 2012, the United States accounted for 53 percent of nanomedicine patent applications, followed by Europe (25 percent) and Asia (12 percent). Drug delivery represented the largest segment in nanomedicine, accounting for 76 percent of publications and 59 percent of nanomedicine patents. The second-largest segment was in vitro diagnostics (11 percent of publications and 14 percent of patent filings).26 In addition to the obvious health benefits, nanomedicine is a sizable “industry” that is projected to grow to over $130 billion in 2016, compared to $63.8 billion in 2010.
To retain U.S. leadership in nanomedicine, a sustainable medical nanomanufacturing infrastructure is needed to move innovative medical research, including in cell and gene therapies, into commercial use. For instance, there are few contract manufacturing organizations with capabilities for cGMP27 manufacturing of nanomedicines, quality assurance/quality control testing protocols are inadequate, and there are few services with the ability to incorporate small molecule drugs in FDA-approved, bio-compatible nanoparticle-based formulations.
Within the NNI, the National Cancer Institute (NCI) supports the Alliance for Nanotechnology in Cancer, designed in part to expedite movement of discoveries of cures and treatments from laboratory bench to patient bed (see Box 3.4). A component of the alliance that is especially vital to the translation of research toward application is the Nanotechnology Characterization Laboratory (NCL). By developing and performing a standard set of appropriate tests for nanomaterials proposed for cancer diagnosis or treatment, NCL has greatly expedited the development, trial, and regulatory review process. As highlighted by NCL Director Scott McNeil in his presentation to the committee,28 the obvious strengths of
___________________
26 V. Morigi, A. Tocchio, C.B. Pellegrini, J.H. Sakamoto, M. Arnone, and E. Tasciotti, 2012, Nanotechnology in medicine: From inception to market domination, Journal of Drug Delivery 2012: 389485.
27 Refers to the Current Good Manufacturing Practice regulations enforced by the U.S. Food and Drug Administration.
28 Scott McNeil, Nanotechnology Characterization Laboratory Director, “Critical Infrastructure Gap in Nanomedicine: Scale Up and cGMP Manufacturing,” presentation to the committee on July 29, 2015.
NCL notwithstanding, there is a serious “opportunity gap” with respect to the ability of developers to move beyond sub-gram quantities of material that are readily produced in an individual laboratory, to the kilogram quantities required for preclinical safety assessments and Phase I clinical trials. Any advantage held by the United States is being challenged, for example, by the European Union’s Framework for Research and Innovation Programme, known as “Horizon 2020,” which clearly articulates a set of goals that addresses the challenge of scale-up for the generation of nanomedicines.29
The updated NIH/NCI Nanotechnology Cancer Plan released in 2015 includes a section on Commercialization of Nano-Products for Cancer and Manufacturing Challenges of Nano-Products.30 The plan focuses only on the manufacture of nanoparticles, not nanoscale devices or other medical applications of nanotechnology. It notes that “perhaps the most frequent shortcoming manufacturers encounter in the advancement of therapeutic nanoparticles is a lack of thorough characterization of the product and the identification, to the extent possible, of
___________________
29 See the European Commission website “HORIZON 2020” at http://ec.europa.eu/programmes/horizon2020/, accessed August 19, 2016.
30 National Cancer Institute, 2015, Cancer Nanotechnology Plan 2015, http://nano.cancer.gov/about/plan/.
the critical quality attributes. This requires, among other things, an early emphasis on the appropriate analytical methods, which is something that is frequently neglected.”31 While the plan does a good job of outlining the challenges related to manufacturing nanoparticles for medical use, the alliance (and NIH in general) is not set up to support manufacturing research.
In addition to the Alliance for Nanotechnology in Cancer, the Translation of Nanotechnology in Cancer (TONIC) Consortium was established in 2011 to bring together Alliance-funded research centers, pharmaceutical and biotechnology companies, and patient advocacy groups to promote collaboration between academia and industry and share knowledge about best practices in translating nanotechnology from the laboratory to the marketplace. The consortium has formed a working group on nanodrugs to develop clinical protocols for testing nanoparticle drugs in patients, while in the process addressing limitations and gaps specific to nanoparticle therapeutics.
Another program that is related to manufacturing for nanomedicine and nanohealth is the Nano-Bio Manufacturing Consortium (NBMC) funded by the Air Force Research Laboratory. The mission of NBMC is to mature an integrated suite of nano-bio manufacturing technologies and transition it to industry. The program envisions the convergence of nanotechnology, biotechnology, advanced (additive) manufacturing, and flexible electronics enabling real-time, remote physiological and health/medical monitoring. Early research is focused on developing a technology platform for human performance monitors for military and civilian personnel in high stress situations such as pilots, special operations personnel, firefighters, and trauma care providers.
Finding 3.4: Nanomedicine manufacturing is an essential step in realizing the benefits of the considerable investment in nanomedicine research under the NNI. Nanomedicine manufacturing poses a number of specific challenges that are not being met by other NNI manufacturing efforts. Two reports—the NCI Cancer Nanotechnology Plan 2015 and the PCAST Report to the President and Congress on the Fifth Assessment of the National Nanotechnology Initiative (Appendix II, Manufacturing Nanomedicine)—provide a sound basis for NNI focus on this topic.
Recommendation 3.4: The National Institutes of Health should lead the development of a roadmap, in collaboration with the nanomedicine industry, to identify technical barriers to scaling up the manufacture of nanomedicines, as well as areas in which research is needed to overcome those barriers.
__________________
31 National Cancer Institute, 2015, Cancer Nanotechnology Plan 2015, p. 9.

















