5
Programs and Supports for Family Caregivers of Older Adults
ABSTRACT: This chapter reviews what is known about the effectiveness of interventions designed to support family caregivers of older adults, including education and skills training, environmental modifications, care management, counseling, and multicomponent models. Well-designed randomized clinical trials show that effective caregiver interventions tend to share several characteristics including, for example, assessments of caregiver risks and needs, tailored interventions that address multiple areas of risk or caregiver need and preferences, and active involvement of caregivers in skills training (rather than a didactic, prescriptive approach). Trials also suggest the potential that some caregiver interventions reduce the resource use of care recipients by delaying nursing home placement, reducing re-hospitalizations, and shortening hospital stays. Despite demonstrated effectiveness, however, promising interventions have not been disseminated and adopted in everyday settings. A variety of barriers, outlined in the chapter, have to be overcome if family caregivers are to benefit from this research.
Family members form the backbone of our health care and long-term services and supports (LTSS) systems, representing the largest single source of care for older adults. Although family caregivers assume a wide array of roles and responsibilities, as Chapter 3 described, they typically do so without sufficient education, training, or support. Caregiving can result in positive outcomes for the caregiver such as personal growth due to long-
standing expectations of mutual support (e.g., a spouse caring for a partner) or a sense of giving back to someone who has cared or provided support for them at some other time (e.g., an adult child caring for a parent). Caregiving can also result in a myriad of negative consequences for caregivers including financial strain, emotional distress, social isolation, disruption in work and other family roles, and even physical morbidities for those who are most distressed.
With the number of potential family caregivers projected to decline in the next few decades, the United States faces a looming care gap just as the population rapidly ages and many older adults have longer periods of care needs (Redfoot et al., 2013). Finding ways to support families has been and will continue to be a pressing public health focus. The past three decades have generated considerable research on the identification of interventions and supportive approaches for family caregivers and the need for advancing supportive policies will only continue. Research to date on developing, evaluating, and implementing programs for family caregivers provides invaluable insight on the challenges and consequences of caregiving and approaches for providing caregivers with needed skills for care provision, alleviating caregiver distress, and improving the quality of life for the caregiver and the older adult receiving care.
This chapter reviews the evidence on interventions directed at supporting family caregivers of older adults. Given the vast literature in this area, the committee used a framework to facilitate organization of the literature and to illustrate that, although the caregiver is the ultimate target of intervention programs, programs vary in focus, scope, funding, and the service settings and environments in which they are delivered. It also illustrates the complexity of the caregiving experience and the interactions that occur among the caregiver, the care recipient, the community, and the larger social/political environment. Because most interventions include a protocol for assessing both the caregiver (e.g., problems, needs, strengths, and resources) and the older adult, this review also includes a brief review of protocols used for caregiver assessment. A summary of the evidence for interventions is presented according to the various levels of the framework (recognizing that there are interactions among them): the individual/social level (older adult and caregiver, family, friends) organized by the specific health conditions of the older adult care recipient; the organizational level (e.g., workplace or formal health care organization); and the broader societal level.
Approach Used in the Review of the Literature
The committee defined intervention broadly to represent therapeutic strategies, care delivery models, programs, and services intended to sup-
port family caregivers of older adults. As noted, interventions may target the family caregiver or older adult (or both), organizations or the broader social context (or some combination) with the intent of modifying a particular risk factor (e.g., depression, social isolation, poor physical health, economic strain), behavioral process (e.g., communication strategies, self-care behaviors), or set of relationships (e.g., family caregiver and health and service providers; caregiver and care recipient). An intervention may include a set of social-behavioral strategies (e.g., education, skills training), psychosocial therapies (e.g., cognitive behavioral therapy), programmatic organizational strategies (e.g., workplace provisions for caregivers) or broad policy initiatives (e.g., the Family and Medical Leave Act, or FMLA). The review excluded pharmacological interventions and other interventions targeting the older adult unless caregiver outcomes were also reported.
The committee’s approach overall was to summarize the available evidence regarding intervention strategies for family caregivers of older adults with varying conditions and to draw conclusions as to what types of interventions are effective. This chapter does not present a formal systematic review of the available literature as that is beyond the scope of this report. Rather, it summarizes the characteristics of interventions, their impact on the caregiver and care recipient, and general findings regarding the extent to which diversity and issues of cost were considered. For health conditions of older adults for which the caregiver intervention literature is extensive (e.g., dementia, cancer) and recent meta-analyses or systematic reviews have been conducted, the committee summarized the results of these analyses/reviews and then examined individual articles that were not included in or published after the review was completed. For conditions such as spinal cord injury and mental health disorders in which the literature is not as extensive, the key existing intervention studies were summarized. In summary, the committee examined several important factors:
- Interventions directed at families caring for older adults with a very wide range of conditions including dementia, stroke, cancer, spinal cord injuries, and mental illnesses, were included.
- Five categories of outcomes and their measurements were considered. These included outcomes related to the psychological, physical, social/support service use, economic, and positive effects on caregiving. Encompassed in these broad outcomes is utilization of available resources by the caregiver and placement of the older adult. For example, in the National Institutes of Health (NIH)-supported Resources for Enhancing Alzheimer’s Caregiver Health II (REACH II) trial, changes in use of formal care and services by the caregiver were evaluated as the intervention included information on strategies to enhance existing use of resources.
- Consideration was given to the heterogeneity of the caregiving experience and the longitudinal trajectory of providing care, thus recognizing that different intervention approaches may be warranted for different caregivers, older adult populations, and stages in the caregiving career and stages in the life course of caregivers (e.g., young adult caregivers may require different types and levels of support than older spouses).
- Special attention was given to how interventions do (or do not) address issues of diversity given that caregivers and older adults are very heterogeneous. Diversity was defined using a broad lens to include variations in race, ethnicity/culture, geography, socioeconomic status, caregiver–older adult relationship, care arrangements, and care contexts.
- Special consideration was given to the role of technology in delivering supportive services to families. Technology can be used to provide support for the caregiver (e.g., information websites, social media); to serve as an interface with the health care system; or to foster support through the development of caregiver networks. It is also playing an increasingly important role in health care delivery, and thus caregivers often need to interact with sophisticated technologies in the delivery of care.
- In evaluating the evidence for intervention studies, deliberation was given to intervention implementation considerations such as factors that may influence access to evidence-based interventions, approaches to the design of interventions (e.g., person centeredness, tailoring to caregiver needs, training needs of health and human service providers to provide evidence-based interventions), and factors that may impede the implementation of evidence-based interventions in real-world settings, including home care, primary care, hospitals, or the aging service network.
Organizational Framework for the Interventions
To organize the available literature and understand the evidence and gaps in knowledge regarding caregiver interventions, as noted, the committee adopted a framework that recognizes that caregiving occurs within a multifaceted context that encompasses the care setting (e.g., the home or residential setting of the older adult who is impaired), the social/community networks (e.g., family members), organizations such as the workplace and health care organizations, and societal/policy environments.
Using this framework, interventions were categorized as targeting and/or delivered in various levels of the caregiver’s experience and his or her life space: the individual (older adult or caregiver), organizations (workplace,
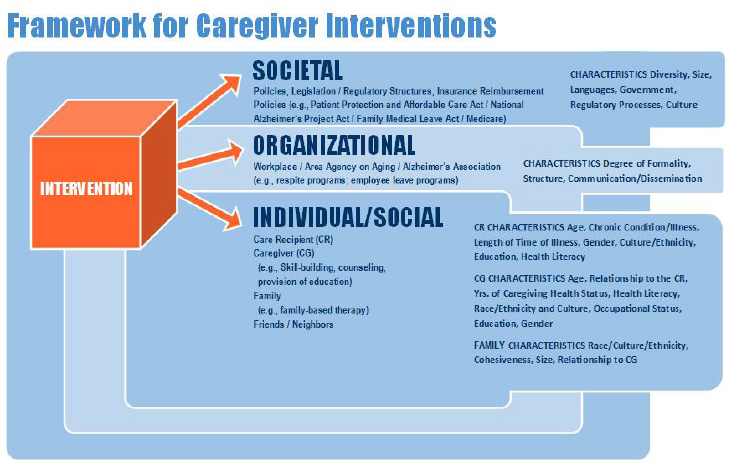
health systems, community-based agencies), or society (policy initiatives), or their combination. Each of these levels has a unique set of characteristics that influence the caregiving experience. There is also a dynamic interplay among the various levels (see Figure 5-1).
Individual Level
Interventions at this level directly target the caregiver (the relative, partner, friend, or neighbor who assists the older adult who needs help due to physical, mental, cognitive, or functional limitations), and caregiver outcomes such as their physical and emotional health, knowledge and skills, social support, coping strategies, well-being, and quality of life. Interventions at this level also include interventions that target or are delivered to entities that are proximal to a caregiver such as the family, or the immediate community in which the caregiver interacts such as the neighborhood or neighborhood organizations. Interventions targeting this level include family-based interventions (e.g., family meetings) that attempt to increase the cohesiveness or support of other family members or a support group at a neighborhood senior center, naturally occurring retirement communities, or faith-based organizations. The interventions encompass a broad range of strategies including but not limited to: education, skill building, social support groups, cognitive behavioral therapy, environmental modifications, mindfulness training, information provision, stress management, and edu-
cation. Many interventions are multicomponent and target several areas of caregiver risk. Interventions at this level may also target the dyad (both the caregiver and the older adult) or the older adult. Studies, which evaluate interventions directed at the older adult care recipient (e.g., cognitive training programs, prescription drugs) and for which caregiver outcomes are reported, are briefly summarized.
Organizational Level
The organizational level includes formal organizational structures such as health care and social service providers, the workplace, formal care settings, or community agencies (e.g., hospitals, Area Agencies on Aging). Examples of interventions that target this level include workplace benefits for caregivers, employee education and referral programs, adult day services, and in-home and outside-of-the-home respite programs.
Societal/Policy
This level includes interventions targeted at a societal and policy level, and includes insurance reimbursement policies, the National Family Caregiver Support Program (NFCSP), the National Alzheimer’s Project Act (NAPA), the Patient Protection and Affordable Care Act, FMLA, or requirements for electronic health record (EHR) technology for caregiver access to the care recipient’s health information. This chapter discusses interventions at this level briefly; more detail about them can be found in Chapters 1 and 6.
ASSESSMENT STRATEGIES
Interventions at the individual level and sometimes at the organizational level typically include an assessment of the family caregiving situation. In this report, caregiver assessment generally refers to a systematic process of gathering information about a caregiving situation, from the caregiver’s perspective, about (1) specific problems, needs, strengths, and resources; (2) the emotional and physical functioning of the caregiver and care recipient; (3) the caregiver’s ability to help meet the needs of the older adult; and (4) caregiver interactions or relationships with health care teams and/or LTSS systems. However, it may also include an assessment of the environment (e.g., clutter, safety hazards) or of the interaction between the caregiver and older adult. The specific topics of assessments vary according to the health condition of the older adults (e.g., dementia versus cancer). An assumption of caregiver assessment is that direct contact has occurred between the person performing the assessment and the family caregiver. In
other words, family caregiver assessment involves asking questions of the family caregiver about themselves, not asking the caregiver questions about the care recipient (Kelly et al., 2013). However, it frequently also involves some assessment of the care recipient such as the assessment of cognitive status. Many measures may be used to assess family caregivers and domains of assessment vary (see Box 5-1). In this regard, a comprehensive inventory of caregiver assessment measures was recently compiled and is available at the website of the Family Caregiver Alliance (2012).
In intervention research and clinical settings, a caregiver assessment is generally conducted for three purposes. First, a caregiver assessment may be motivated for the purpose of identifying caregiver eligibility for an inter-
vention trial. Second, a caregiver assessment may be incorporated in the intervention process to determine how to appropriately tailor services and skill-building strategies to best benefit family caregivers and persons receiving care (Belle et al., 2006; Fortinsky et al., 2009; Judge et al., 2011). This might involve, for example, culturally tailoring an intervention to accommodate differences in cultural values and preferences. Data from assessments may also be used in the interpretation of intervention study findings to understand how family caregiver factors relate to study outcomes.
One example of an assessment instrument that was used in a research study to tailor the intervention and that is becoming widely used in other research and clinical settings is the REACH II Risk Appraisal Measure (RAM). The RAM is a 16-item measure based on psychometric analysis of the responses of 642 caregiver dyads to the REACH II 59-item baseline assessment. It was developed as a brief, face-valid method to identify and prioritize specific areas of risk for caregivers of individuals with dementia that were amenable to intervention and relevant across diverse cultural and ethnic groups. The six RAM domains include depressive symptomatology (one item), burden (three items), self-care (two items), social support (two items), care recipient problem behaviors (two items), and safety (four items) (Czaja et al., 2009). Assessment for caregivers of individuals with other conditions such as cancer might have a strong focus on a caregiver’s ability to manage the cancer symptomatology, medication regime, and other medically oriented procedures (e.g., infusion of fluids, cleaning of feeding tubes) of the care recipient. Irrespective of the assessment instrument, the rationale for conducting a family caregiver assessment is based on the recognition that family caregivers are highly diverse and that services and supports need to be tailored to address the unique and varying needs of caregivers (Brodaty et al., 2003).
Caregiver assessment is not commonly integrated in health delivery settings. At the system level, with rare exception, health care or LTSS providers have not adopted caregiver assessment into everyday practice (Feinberg and Levine, 2015). Less than one-third of states include family caregiver assessments in their Medicaid home- and community-based services (HCBS) waiver programs, with considerable variability in the scope of the assessment used (Kelly et al., 2013). The challenges of implementing caregiver assessment in practice are multifaceted. Problems not only span organizational and provider pushback but caregivers themselves may not understand the purpose of the assessment or want to be assessed (Levine et al., 2013). Importantly, caregiver assessments should also include an assessment of family structures, dynamics, and resources. In many cases, marshaling family resources can provide needed support to family members (e.g., Eisdorfer et al., 2003). Understanding the characteristics and resources of the family can also help service providers work effectively with multiple caregiver
families (or groups) and suggest strategies for sharing caregiving responsibilities. Similarly, the social/community context of the caregiver should be considered and would help to gain an understanding of interventions that are acceptable to and effective for caregivers that are population-specific and accommodate language or cultural caregiving norms.
INDIVIDUAL-LEVEL INTERVENTIONS
Interventions at the individual level employ a variety of therapeutic strategies including problem solving, skills training, information provision, support groups, counseling, and family therapy; and target various aspects of caregiver risk (e.g., symptom management, behavioral problems, lack of support and resources). They may vary in dose, intensity, and mode of delivery (e.g., face-to-face, Internet). Research evaluating individual-level interventions employs a variety of study designs such as randomized controlled trials (RCTs), case control, and pre-test/post-test designs, and includes varying outcomes related to the psychological (positive and negative), physical, social, and economic effects of caregiving. The following section summarizes the evidence regarding individual-level interventions organized according to the health condition of the older adult.
Alzheimer’s Disease/Dementia Caregiving
Alzheimer’s disease and related disorders (ADRD) are progressive, neurodegenerative conditions that result in cognitive, social, and physical functional decline, as well as behavioral and psychological symptoms. Most of the 3.6 to 5.2 million individuals with dementia in the United States live at home and are cared for by family members. In fact, families provide more than 80 percent of the LTSS that people with ADRD receive (Friedman et al., 2015; Kasper et al., 2015). As the disease progresses, the caregiving responsibilities of families increase and include advocacy, hands-on assistance with personal care and mobility tasks, emotional and social support, medical care, and surrogacy, as well as ensuring safety and quality of life and preventing and managing behavioral symptoms (Black et al., 2013; Callahan et al., 2012; Hodgson et al., 2014).
As noted in Chapter 3, compared to caregivers of older adults without cognitive impairment, caregivers of individuals with ADRD spend more time in caregiving, have more care responsibilities, and report greater objective (e.g., financial burdens, time spent in daily care routines) and subjective negative consequences (e.g., poor physical health, emotional upset and distress) (Alzheimer’s Association, 2014; Bertrand et al., 2006; Friedman et al., 2015; Kasper et al., 2015; Moon et al., 2014; Ory et al., 1999). Many interventions have been developed for this population and tested in RCTs.
Overall, an estimated 200 interventions have been tested using randomized designs (Gitlin et al., 2015; Maslow, 2012). Seven meta-analyses and 17 systematic reviews of research conducted between 1966 and 2013 have been published.
Unfortunately, there is no agreed-on categorization system for classifying caregiver interventions by their content. However, for heuristic purposes, interventions for families of persons with dementia can be categorized as follows: professional support for depression (e.g., psychotherapy); psychoeducation (e.g., education about the disease, stress reduction and support, providing information about resources); behavior management/skills training (e.g., instruction in particular approaches such as using activities, adaptive equipment, or the use of cueing to prevent and manage behaviors); situational counseling (e.g., family counseling, instruction in cognitive reframing or other positive coping techniques, mindfulness training); self-care/relaxation training (e.g., meditation, yoga); and multicomponent interventions (Gitlin and Hodgson, 2015). Multicomponent interventions tend to target caregivers of individuals at the moderate disease stage and include combinations of approaches such as dementia education, care management, environmental modification, counseling, skills training, and/or referral to community resources, all tailored to the identified unmet needs of caregivers identified via a systematic assessment.
Programs targeting family caregivers of persons with dementia have been delivered through various modalities including face-to-face (Belle et al., 2006; Gitlin et al., 2010b), group (Gallagher-Thompson et al., 2003), telephone (Bass et al., 2003; Kwok et al., 2013; Martindale-Adams et al., 2013; Tremont et al., 2015), videophone (Czaja et al., 2013), or Web-based platforms (Kajiyama et al., 2013). They range in level of dose and intensity from a brief number of sessions (e.g., four sessions delivered over 2 to 3 months) (Nichols et al., 2016) to 1 or more years of contact (Mittelman et al., 2006) and are delivered by different health and social service professionals including nurses, occupational therapists, community health workers, social workers, and care managers. Some interventions are offered in a variety of modalities. For example, the Savvy Caregiver, a psychoeducation intervention, which provides basic disease education, coping skills, and behavioral management strategies, is available in a variety of modalities (telephone, classroom, online) making it more accessible and responsive to family preferences (Hepburn et al., 2003, 2007). A few interventions have targeted both the family caregiver and the person with dementia (e.g., Whitlatch et al., 2006).
Outcome measures for dementia caregiver interventions are wide ranging and have primarily included caregiver knowledge, burden, self-efficacy, psychological morbidity (anxiety/depression), upset, confidence, skills, and desire or time to caregiver placement of the person with dementia
in assisted living or nursing homes. Many interventions, using rigorous trial designs, demonstrate effectiveness for one or more outcomes that are targeted in the trial such as reducing caregiver burden and for some interventions, reducing institutionalization and other care recipient-related outcomes such as symptomatology (Brodaty and Arasaratnam, 2012; Gitlin et al., 2006, 2008; Mittelman et al., 2006). However, the outcomes that are positively or not positively impacted vary vastly among studies (e.g., one study may report benefits for depression whereas another will focus on efficacy). An example is the Mittelman and colleagues (2006) New York University Caregiver Intervention (NYUCI), which showed reduced nursing home placement rates and increased caregiver feelings of efficacy and social support. In addition, while the intervention does not appear to change the frequency of care recipient problem behaviors overall, it does appear to help caregivers feel less distressed by these behaviors. In contrast, the REACH II intervention (see Box 5-2) resulted in improvements in a multivariate quality of life indicator that assessed caregiver burden, depressive symptoms, self-care, social support, and care recipient problem behaviors for the intervention group compared to the control group (Belle et al., 2006). For the most part, multicomponent interventions show the largest effects for most outcomes. Most changes from program participation are examined for only short duration (e.g., 3, 4, or 6 months), with few studies examining long-term benefits (i.e., more than 9 months) (Gitlin et al., 2010b; Mittelman et al., 2006). Also, most studies report positive effects on outcomes such as increased confidence in dealing with challenging situations, with very few showing no benefits at all, and no studies reporting worsening or adverse effects.
Only a few studies directly target people with dementia and also evaluate the potential benefits of the intervention for family caregivers. Of these, there are inconsistent outcomes, with some studies showing benefits for caregivers and others not. For example, Stanley and colleagues’ (2013) cognitive behavioral therapy intervention targeting anxiety in persons with dementia (Peaceful Mind) reduced caregiver distress associated with the anxiety of the person with dementia. A meta-analysis of 17 studies examining the effects on caregivers of antidementia clinical drugs administered to individuals with dementia found a small beneficial effect for caregivers including reductions in burden and time spent caregiving (Lingler et al., 2005). Gitlin and colleagues’ (2010a) activity intervention study to reduce behavioral symptoms in persons with dementia resulted in significant reductions in objective burden (e.g., time spent in providing direct care), confidence in engaging in activities (e.g., preparing light meals, grooming, and exercise), and improved mastery among caregivers, with other aspects of well-being (e.g., depressive symptoms, burden) not affected. Similarly, Tappen and colleagues’ (2014) cognitive training intervention for persons
with dementia did not result in reductions in depression or upset with behavioral symptoms for caregivers.
Although the literature is limited, interventions may also target the family or social networks of the caregiver, neighbors, neighborhoods, churches, or community-level groups such as senior centers. Family group interventions providing psychoeducation and/or counseling show positive benefits for families including reductions of caregiver negative reactions to behavioral symptoms in persons with dementia and caregiver depression (Eisdorfer et al., 2003; Ostwald et al., 1999). Home-Based Supportive Services programs that provide stipends to families to offset care expenses of individuals with disability of all ages demonstrate a wider range of benefits to family caregivers, including fewer out-of-pocket care expenses, better mental health and access to health care, and improved self-efficacy, than caregivers on a waitlist for this service (Caldwell, 2006; Heller and Caldwell, 2006; Heller et al., 1999).
The community represents a largely untapped resource for supporting families for which there are no tested interventions (see Box 5-3 for an example). Communities have a wide range of naturally occurring resources such as churches or religious places of worship, libraries, community or senior centers, or schools. Each of these could provide a range of emotional and logistical supportive services for families, including support groups, friendly visitors, chore services, and education, and serve as a supportive network for families. For example, the Naturally Occurring Retirement Communities (NORCs) and villages, which have been developing primarily in urban areas throughout the United States to provide supportive services for and to neighbors, could more purposely support family caregivers (Greenfield et al., 2013). However, the benefit of these resources for family caregivers has not been evaluated. This is a critical gap in the literature. Future research needs to be conducted concerning models for supporting family caregivers using these naturally occurring resources and other models of livable communities. There is also a lack of community-engaged interventions targeting ethnic/racial groups through outreach to ethnic media, churches, and community-based organizations that serve ethnic communities. The community can also play an important role in terms of providing support and services to “hard to reach” caregivers, such as those who live in rural locations, ethnic/racial minorities, or those who have no other source of support for the care recipient and are often unaware of or have difficulty accessing available resources and sources of caregiver support. Specifically, community programs or workers may help identify and “recruit” caregivers into educational or support programs by marketing these programs in newsletters, through service providers, or other forms of community engagement using targeted, culturally appropriate messaging. Engagement of caregivers in these programs might be facilitated by pro-
viding transportation or respite services, offering home-based programs, implementing more flexible programs with respect to scheduling, integrating them within other services, or having satellite programs in rural locations (Navaie, 2011).
Overall, although interventions vary widely in purpose, dose, intensity, and mode of delivery, effective interventions tend to share several common characteristics: adjusting dose, intensity, and specific focus of an intervention based on a caregiver’s risk or need profile (Belle et al., 2006; Czaja et al., 2009; Richards et al., 2007); active involvement of caregivers in learning a particular skill such as managing a problem behavior rather than a didactic, prescriptive approach in which information or instructions are provided (Belle et al., 2006; Chee et al., 2007; Czaja, 2009); addressing multiple areas of identified need or risk (Belle et al., 2006; Kansagara et al., 2010; Zarit and Femia, 2008); and longer interventions or episodic
(i.e., booster) support over time for the duration of caregiving (Mittelman et al., 2006).
Overall, interventions directed at the individual level (i.e., target the older adult with dementia and/or the family caregiver) appear to be feasible to implement (they can be delivered and received) and are acceptable (well received) to families. These interventions regardless of dose and intensity or place of delivery also appear to make a real and important difference in the lives of family caregivers. Box 5-4 briefly describes one such intervention—In it Together: Learning to COPE with Dementia—designed to improve the well-being of both the person with dementia and the family caregiver.
The few cost studies that have been conducted suggest that interventions directed at the individual level can be low cost and result in cost savings to the caregiver in terms of reductions in time spent in caregiving, a highly valuable resource for caregivers (Gitlin et al., 2010a; Jutkowitz et al., 2010; Nichols et al., 2008). However, few studies have examined the cost effectiveness of an intervention.
Implementation of Interventions for Caregivers of Persons with Dementia
Generally a strong body of research evidence, some cost analyses, intervention feasibility, and caregiver acceptability of an intervention are factors necessary for moving forward with the widespread translation, dissemination, and implementation of these proven interventions for delivery in service and practice settings (Gearing et al., 2011; Gitlin et al., 2015). Despite the generally positive benefit of interventions for caregivers of older adults with dementia, few studies have been translated for implementation in systems of care (see Table 5-1). One exemplar exception is the National Institutes of Health (NIH)-supported REACH II initiative. The original trial was found to improve quality of life in Hispanic and white caregivers and spousal African American caregivers (Belle et al., 2006). It has since been adapted for delivery and implemented throughout the U.S. Department of Veterans Affairs (VA) and other organizations (see Box 5-2). Initially, its 12-session structure served as a barrier to full implementation in social service settings; however, a modified four-session adaptation has shown similar outcomes as the original trial (Burgio et al., 2009; Nichols et al., 2011, 2016). Although REACH II and other evidence-based programs are currently being tested in various care settings such as Medicaid waiver programs, social services, the aging network, and home care, few caregivers of persons with dementia currently have access to such programs unless they are located in the specific regions in which the demonstration programs are being evaluated through grant-supported funds (Gitlin et al., 2015) or through VA-supported programs.
A major challenge to implementation of interventions to support caregivers of older adults by the health care delivery system is the paucity of
TABLE 5-1 Outcomes and Implementation of Selected Family Caregiver Interventions
| Intervention | Description | Caregiver and Care Recipient Outcomes | Implementation Sites |
|---|---|---|---|
| BRI Care Consultation | Consumer-driven coaching program focused on finding solutions for family caregivers (CGs) and their care recipients (CRs). | Improved psychosocial outcomes for CGs and CRs (e.g., reductions in burden); reduced hospital readmissions and return emergency department visits. | 21 organizations are licensed to deliver BRI Care Consultation. Licensees have diverse missions, locations, and target populations. |
| Collaborative Care Model for Alzheimer’s Disease and Related Dementias | For CGs of persons with dementia. Primary care management by an interdisciplinary team led by an advanced practice nurse. Uses standard protocols to initiate treatment and to identify, monitor, and treat behavioral and psychological symptoms of dementia. Emphasizes nonpharmacological management. | Improved CG distress and depression; CRs had fewer behavioral and psychological symptoms of dementia. | Has been evaluated in primary care practices in two university-affiliated health care systems. |
| New York University Caregiver Intervention (NYUCI) | For CGs of persons with dementia. Task-oriented counseling and support groups to provide emotional and instrumental support to CGs and to promote communication among family members and to teach techniques for problem solving and management of difficult CR behaviors. | Improved CG competence, self-confidence, social supports, and coping with problem behaviors. Reduced CR depression and nursing home placements. | Multiple Alzheimer’s Associations and four project sites selected by the Minnesota Board on Aging and the Area Agency on Aging (AAA). |
| Intervention | Description | Caregiver and Care Recipient Outcomes | Implementation Sites |
|---|---|---|---|
| REACH II | For CGs of persons with dementia. Education, support, and skills training to improve CG’s health, safety, social support, handling of problem behaviors, and depression. | Improved CG quality of life as measured by decreased burden and depression, enhanced self-care and social support, and fewer CR fewer behavior problems. | Numerous VA sites; state aging services programs, AAAs; implemented in South Korea, Hong Kong, and Germany. |
| Savvy Caregiver | For CGs of persons with dementia. Education and skills training for CGs to help them manage stress and carry out the caregiving role effectively. | Improved CG competence, personal gain, management, positive experiences, depression, and ability to keep the CR occupied. | 4 AAA Caregiver Programs and 25 communities (including rural areas). |
| Skills2Care | For CGs of persons with dementia. Home-based intervention designed to develop a more supportive environment through modifications to the home environment and CG education and skill building. | Improved CG knowledge and skills, ability to engage persons with dementia in activities, confidence in managing behaviors, functional dependence, and care of themselves. | 20 home care agencies; AAAs and by occupational therapists in private practice; covered by Medicare. |
| New Ways Better Days: Tailored Activity Program (TAP) | Home-based occupational therapy intervention for individuals with dementia. Focuses on identifying the CR’s abilities, previous roles, habits, and interests; developing activities customized to the individual; and training their CGs in activity use and stress management. | Well-received by CGs and CRs; reduced CR behavioral symptoms; CGs gained more personal time. | Being implemented in several states through the Administration for Community Living’s Dementia-Friendly Community initiative; has been adapted in hospital settings; is undergoing testing in Baltimore and Florida; and is used in Australia, Brazil, England, and Scotland. An online adaptation is under development. |
| Care of Older Persons in the Home Environment (COPE) | For CGs of persons with dementia. Designed to support CR’s abilities by reducing environmental stressors and improving CG skills, problem-solving, and stress. | Improved CGs sense of wellbeing and confidence; improved CRs dependence and increased their engagement. | Being tested in Medicaid home and community-based programs. |
SOURCES: Belle et al., 2006; Callahan et al., 2006; Clark et al., 2004; Gitlin et al., 2001, 2003, 2010a, 2010b, 2010c; Hepburn et al., 2001; Jutkowitz et al., 2010; Mittelman, 2008; Mittelman et al., 1993, 1995, 1996, 2006; Nichols et al., 2011; Ostwald et al., 1999.
Current Procedural Terminology (CPT) or Healthcare Common Procedure Coding System (HCPCS) codes to recognize the services rendered. One exception to this model is the REACH I Skills2Care intervention, which was structured for implementation in home care delivery by occupational therapists and is reimbursed through Medicare Part A and B as long as the caregiver training is linked to the health and functional goals of the care recipient with dementia (Gitlin et al., 2015).
Table 5-2 describes the barriers to moving evidence-based interventions from the research phase to implementation in real-world, health, and social service settings. The challenges include limitations of existing evidence; funding; lack of knowledge of providers, health and social service organizations, and administrators of available evidence-based programs; and various contextual barriers. Table 5-3 outlines several strategies for addressing these barriers and facilitating the translation of evidenced-based intervention programs into clinical and community settings.
There are also notable limitations of intervention studies that point to the need for additional and new research. For example, samples are poorly characterized in terms of the disease etiology and disease stage of the person with dementia. Most interventions target the needs of families who care for persons at the moderate disease stage with a primary diagnosis of Alzheimer’s disease, with few studies targeting families caring for persons with mild cognitive impairment, early-stage dementia, severe stages of the disease, or for specific dementias such as Frontotemporal Dementia or Lewy bodies that impose unique care challenges for caregivers. Also, caregivers of older adults with dementia may also have to handle other disease challenges such as diabetes or sensory impairments such as difficulty seeing or hearing (Feil et al., 2011; Maslow, 2011). This makes it challenging for clinicians or service providers to know which families would benefit from which interventions. Also, as noted earlier, few intervention studies report long-term outcomes (i.e., more than 12 months) (Gitlin et al., 2006; Mittelman et al., 2006; Samus et al., 2014), evaluate adherence (Chee et al., 2007), or identify mechanisms by which an intervention works or why desired benefits are achieved (Roth et al., 2005). Further, interventions are, for the most part, tested outside of clinical and practice settings requiring yet additional translation and then implementation test phases (Gitlin et al., 2015; Nichols et al., 2016).
Furthermore, a psychosocial stress-process model guides most interventions and thus their focus has been on reducing stressors and caregiver burden. Consequently, the practical issues that many caregivers confront have been largely ignored such as financial and physical strain, balancing caregiving with employment responsibilities or their need for specific skills for overseeing complex medical conditions (e.g., wound care, diabetes care, vision impairments, or fall risk), or managing frustrating encounters with
| Challenge | Examples |
|---|---|
| Limitations of the existing evidence |
|
| Funding |
|
| Provider knowledge |
|
| Contextual barriers |
|
NOTES: NIA = National Institute on Aging; AoA = Administration on Aging.
SOURCES: Gitlin et al., 2015, 2016; Nichols et al., 2016; Reinhard et al., 2008.
TABLE 5-3 Selected Strategies for Addressing Barriers to Intervention Implementation
|
SOURCES: Gitlin et al., 2015; Nichols et al., 2016; Reinhard et al., 2008.
health care providers. Another limitation is that interventions target single individual caregivers even though evidence suggests that families often share care responsibilities. Finally, most interventions have been tested with white caregivers. Only a few studies have involved African Americans (Belle et al., 2006; Martindale-Adams et al., 2013), Latino (Belle et al., 2006; Gallagher-Thompson et al., 2003), and Asian (Heller and Caldwell, 2006; Reuben et al., 2013) caregivers. Other groups such as lesbian, gay, bisexual, and transgender (LGBT) caregivers, long-distance caregivers, and rural caregivers have largely been ignored. Further studies have not systematically examined caregiver health care utilization as a possible outcome of caregiving even though existing research suggests that caregiver self-care may be compromised, which has the potential of causing down-steam adverse health effects. Spouses of individuals with dementia have significantly higher monthly Medicare use than spouses of non-demented individuals, suggesting interdependence between the health and health care costs of the dyad (Dassel et al., 2015).
Impact of Pharmacological Treatments for Alzheimer’s Disease and Related Dementias on the Caregiver
The most commonly prescribed medications for older adults with ADRD are cholinesterase inhibitors (ChEIs). The majority of the research examining positive benefits of ChEIs are focused on care recipient outcomes and based on clinical trial data that support clinical effectiveness of these medications at small to modest levels for some individuals. In some cases other benefits of ChEIs have been examined such as improvements in caregiver burden,
care recipient and caregiver quality of life, and time to nursing home placement. Given the critical role that caregivers play in providing support to individuals with ADRD caregiver-specific outcome measures are gaining attention while specifying endpoints in clinical trials. Lingler and colleagues (2005) conducted a systematic review of ChEI trials that also examined indirect treatment effects, including caregiver-specific outcomes. The authors identified 17 studies involving 4,744 subjects; four trials met the inclusion for the burden analysis and six trials met the inclusion criteria for the time-use analysis. Overall the analysis revealed that providing ChEIs to care recipients had a small beneficial effect on caregiver burden and active time use among caregivers of persons with ADRD. Another systematic review by Knowles (2006) summarized major findings of effectiveness studies focusing on treatment effects of donepezil. The major findings of this review include significant improvement in cognitive function for the care recipients, delays in nursing home placement, as well as modest evidence for improvements in caregiving burden and time use. Generally, given the critical role of caregivers in providing support to older adults with ADRD, caregiver outcomes such as burden, quality of life, and time spent on caregiving should be included in any evaluation examining the effectiveness of ChEIs.
Caregivers of People Who Have Had a Stroke
Although the literature is less extensive than for persons with ADRD, interventions have also been developed for family caregivers of older adults who have had a stroke. These caregivers are typically thrust into the caregiver role with little or no warning. They typically need skills in the physical aspects of caring for the individual who had a stroke, play a large role in the person’s care coordination, and also provide emotional support to the individual, which is especially challenging if the person is confronted with long-term disabilities. They may also live with the fear that a stroke may happen again.
Overall, the evidence suggests the available programs are beneficial for both survivors of stroke and their family caregivers. The American Heart Association and American Stroke Association (AHA/ASA) (Bakas et al., 2014) recently reviewed the evidence for the efficacy of family caregiving interventions in stroke. The review examined the stroke intervention literature to determine the impact of interventions on outcomes for both stroke survivors and their family caregivers. The review included 32 interventions; 22 interventions were evaluated in RCTs. Survivor outcomes included physical functioning, anxiety, depression, social functioning, service use, and knowledge. Family caregiver outcomes included preparedness to care for survivor, burden, stress and strain, anxiety, depression, quality of life, social functioning, coping, health care utilization, and knowledge. Inter-
vention strategies are similar to those employed in interventions for other types of family caregiving. Psychoeducation elements in stroke interventions commonly include the presentation of information and warning signs of the survivor’s health. Skills training techniques include problem solving and stress management for managing the care, medication, and personal needs of the survivor, and managing emotions and behaviors. The caregiver’s emotions and health care needs are also the target of skills training techniques. Specific techniques used include problem solving, goal setting, and communication with health care professionals; hands-on training in skills such as lifting and mobility techniques and assistance with activities of daily living; and communication skills tailored to the needs of the care recipient.
Caregiver- and dyad-focused interventions have been tested. Of the 32 studies identified, 17 were caregiver focused and 15 were dyad focused. Overall, Kalra and colleagues (2004) provide strong evidence for the dyadic approach, which resulted in positive outcomes for both survivors and caregivers. However, there is also an absence of studies that target diverse groups of caregivers.
Caregivers of Older Adults with Cancer
Family members also serve as caregivers and provide critical support for older adults with cancer. For example, family caregivers can play an essential role in the delivery of medications directed at the cancer; monitoring and managing symptoms; and providing emotional support that is important to treatment and survival of the care recipient. Family caregivers of care recipients with cancer are often introduced into situations that require a working knowledge of complex medical procedures and medication regimens in the context of a life-threatening diagnosis to a member of the family. When the individual is in remission, the possibility of cancer recurrence is a concern. Interventions for family caregivers of persons with cancer have been designed to address these medical and emotional demands.
In 2010, Northouse and colleagues reported a meta-analysis of 29 RCTs of interventions for family caregivers of individuals with cancer that included an analysis of the types of interventions and their effects on various caregiver outcomes. The intervention RCTs were carried out from 1983 to 2009 and met the following criteria: (1) they had to involve family caregivers, either alone, or with the person with cancer; (2) they were psychosocially, cognitively, or behaviorally oriented; (3) the participants were randomly assigned to the intervention or control arm of the study; and (4) they were published in a peer-reviewed journal. The majority of interventions was psychoeducational and provided information regarding symptom management and other physical aspects of the individual’s care.
Attention was also directed to the emotional and psychosocial needs of care recipients, caregivers, and/or marital or family relationships. Skills training interventions that focused primarily on the development of coping, communication, and problem-solving skills with a focus on behavioral change were also included. The least frequent intervention was therapeutic counseling focused primarily on the development of therapeutic relationships to address concerns related to cancer or caregiving. Overall, although these interventions had small to medium effects on reducing caregiver burden and improving caregiver coping, they did increase caregiver self-sufficiency and improve some aspects of quality of life.
Appelbaum and Breitbart (2013) expanded on the meta-analysis conducted by Northouse and colleagues (2010) in a review article that summarized the scope and impact of 49 cancer caregiver intervention studies published between 1980 and 2011. All were classified as psychosocial and were subdivided into eight groups based on primarily therapeutic approaches, such as psychoeducational, problem solving/skill building, supportive therapy, family/couple therapy, cognitive behavioral therapy, interpersonal therapy, complementary and alternative medicine, and existential therapy. They also addressed the unique needs of family caregivers who are faced with a terminal illness of a family member. Although effect sizes were generally not reported, 65 percent of the interventions produced positive improvements in outcomes for caregivers, such as reductions in burden, anxiety, and depression and enhanced problem-solving and caregiving skills. Some interventions, such as interpersonal therapy and family/couples therapy, also resulted in better relationship quality between the caregiver and care recipient and for the care recipient’s emotional well-being.
Caregiving for Adults with Other Conditions
The empirical literature on interventions is much less robust for family caregivers of older adults with other conditions such as adults with persistent mental illness (e.g., schizophrenia, major depression) and spinal cord injury. This is an emerging area of need as many adults are living longer with these conditions and many rely on family members for support.
Caregivers of adults with persistent mental illness have some unique challenges such as dealing with the issue of stigma, non-normative illness expectations, cyclic illness trajectory, unpredictable patterns of behavior, and often a lack of available support and resources (Biegel and Schulz, 1999). In general, the available literature suggests that psychoeducational family interventions can be effective in terms of helping families cope with and care for a relative who has a severe mental illness such as schizophrenia. Dixon and colleagues (2000) conducted a review of 15 studies on psychoeducational family interventions and found overall psychoeducational
programs for family members are efficacious in terms of family outcomes such as burden and family functioning. However, they concluded that there is incomplete knowledge on how to best design these programs. Overall, the data are quite limited regarding family caregivers of older adults with a mental illness and that which has been conducted is most often on caregivers of individuals with schizophrenia. In addition, programs targeting caregivers of individuals with mental illness are not widely available. This is clearly an area of need as a large number of people with severe and persistent mental illness live with and/or rely on their families for help and support, and literature clearly demonstrates that caring for a family member with mental illness is burdensome for the caregiver (e.g., Biegel and Schulz, 1999).
Caregivers of persons with spinal cord injury also are often confronted with some physical challenges related to lack of mobility of the care recipient and provision of care tasks related to medical complications such as the pressure sores or urinary system disorders. Similar to caregivers of individuals after a stroke, they also have to cope with being suddenly thrust into the caregiving role and the need to provide emotional support to the person with the spinal cord injury who is confronting living with disabilities.
With respect to interventions for these caregivers, the literature generally suggests that psychosocial interventions such as problem-solving therapy, family psychoeducational and dyadic multicomponent psychosocial interventions (e.g., those that combine skills training, stress management techniques) can be beneficial to family caregivers. However, the evidence is limited and some of the studies that have been conducted have involved small samples or lack of a comparison group and most of this work does not focus on older adults. However, the committee chose to include a review of some of this literature to identify strategies that might also be beneficial to older adults. For example, Elliott and Berry (2009) evaluated a brief problem-solving training for family caregivers of recent-onset spinal cord injury. They found that the intervention was beneficial in that the caregivers who received the intervention, as compared to those in the control condition, experienced a decrease in dysfunctional problem-solving styles. In another study, Elliott and colleagues (2008) evaluated a problem-solving intervention delivered via videoconferencing and found that there was a significant decline in depression for caregivers who received the intervention. They also reported an increase in social functioning. However, again the sample was relatively small. Schulz and colleagues (2009) compared a caregiver-focused multicomponent psychosocial intervention to a dual target intervention where the caregiver intervention was complemented by an intervention targeting the care recipient age 35 and older. The intervention conditions were compared to a control condition where caregivers received standard information about caregiving, spinal cord injury, and aging. One
hundred and seventy-three dyads were randomized to one of the three conditions. Overall, the results indicated that caregivers who were in the dual-target condition had improved quality of life, significantly fewer health symptoms, and were less depressed. More recently, Molazem and colleagues (2014) investigated the effectiveness of psychoeducational interventions on the life quality of the family caregivers of people with spinal cord injuries. The study, an RCT, involved 72 family caregivers in Iran who were randomized into an intervention or control group. The study results indicated that the intervention resulted in positive outcomes for caregivers who received the intervention; specifically the caregivers experienced improvement in aspects of quality of life.
Generally, the psychosocial interventions for family caregivers of older adults with chronic conditions such as persistent mental illness and spinal cord injury are similar to those targeting caregivers of adults with dementia and cancer and involve strategies such as psychoeducational programs, support groups, problem-solving training, skills training, and stress management techniques. The results are promising with respect to showing positive benefits of these interventions for caregivers and in some cases care recipients. However, the literature is rather sparse and the empirical data are limited especially for caregivers of older adults living with these conditions.
Cost and Cost-Effectiveness of Caregiver Support Programs
Although many caregiver interventions show improvement in caregiver outcomes such as health and well-being, relatively few assess economic impacts of these interventions such as health care savings associated with reduced formal health care utilization by the care recipient. These might include savings associated with delayed nursing home placement and fewer hospitalizations and emergency room visits. Most intervention studies also fail to quantify the cost of delivering the intervention such as the costs of training the interventionists and the time, travel, and monitoring costs of delivering the intervention.
Randomized trials show significant delays in nursing home placement. The New York University Caregiver Intervention, a program of enhanced supportive services for spouse and adult child caregivers of community-dwelling people with dementia (Gaugler et al., 2013; Mittelman et al., 2006) showed significantly delayed nursing home placement for care recipients in the treatment arm. In addition, caregivers who received the intervention experienced fewer depressive symptoms and less distress compared to those in the usual care control group. A simulation analysis of the widespread use of this program suggested that there would be substantial government savings through the delay in nursing home placement (Long et al., 2013).
Several trials have studied the impact of integrating caregivers into
discharge planning in a hospital or institutional setting. Compared to usual care, integrating caregivers into the discharge process and providing follow-up support to caregivers and patients resulted in significant reductions in readmissions at 90 and 180 days and overall lower costs of post-discharge care (e.g., Huang and Liang, 2005; Legrain et al., 2011; Naylor et al.. 1999). These findings taken as whole suggest that a relatively modest investment in integrating and supporting family caregivers can potentially generate significant health care cost savings. There is little information however on the long-term impact on the caregiver and the potential savings that might be accrued by preventing adverse downstream effects.
Other studies that have evaluated the cost-effectiveness of individual-level interventions suggest that these interventions can be low cost and result in cost savings to the caregiver in terms of reductions in time spent in caregiving, a highly valuable resource for caregivers (Gitlin et al., 2010a; Jutkowitz et al., 2010; Nichols et al., 2008). However, overall few studies have examined the cost-effectiveness of an intervention.
ORGANIZATIONAL LEVEL
Interventions at the organizational level include those targeting caregivers of older adults but which are embedded in or delivered through a formal organizational structure such as the workplace, primary care or other health care delivery settings, and/or community agencies such as Area Agencies on Aging or adult day services. The implementation of a caregiver program in an organizational structure typically requires adjustments to a workflow of the setting and/or connecting various service delivery and/or community agencies in a coordinated fashion to address family caregiver needs. There is a growing body of research on interventions at this level, although typically programs are in a research or evaluation phase and have not been widely implemented and sustained in organizational settings. Summarized below are some of the most promising by the type of program they represent.
Care Coordination Programs
There is a growing body of research evaluating the effects of care coordination approaches. While care coordination is defined differently across studies/programs, it typically involves an assessment phase to identify unmet needs of family caregivers and then helping families connect to and use local resources and services. The programs are intended to help caregivers and care recipients address the challenges in accessing the range of services that care recipients need and to also help support the caregiver, ameliorate problems with service fragmentation, and enhance communication with care providers. An essential feature tends to be a team approach
linking different resources and areas of expertise in a collaborative network to provide caregiver support. Additional elements may include disease education and the provision of coping or stress-reduction strategies. The role of the caregiver in these programs varies. Some programs, such as the Partners in Dementia Care (described below), actively involve the caregiver; in other programs their role is more limited/passive and care coordination primarily occurs through a nurse, case manager, or social worker (Bass et al., 2013). Programs typically last for 1 or more years, follow families over time, and seek to delay nursing home placement, reduce health care utilization, and enhance quality of life at home. However, studies are needed to establish that cost savings can be achieved for LTSS by helping the family caregiver connect to needed resources and services.
Most care coordination programs have been tested for families caring for individuals with dementia because of the lack of a systematic, coordinated care system for persons with dementia and their families and the documented high needs of this group (Black et al., 2013). For example, a survey of 307 caregivers found that only 32 percent of caregivers reported being confident in managing dementia-related problems, only 19 percent knew how to access community services to help provide care, and only 28 percent indicated that the individual’s provider helped them work through dementia care problems (Jennings et al., 2015). In response, a wide range of care management programs have been developed and evaluated.
Systematic reviews and meta-analyses of these interventions reveal that the quality of the research is highly variable with only a few being well-controlled studies (Pimouguet et al., 2010; Somme et al., 2012; Tam-Tham et al., 2013). A few studies report positive impacts on institutional delay or admission rate (e.g., nursing home placement). However, there is inconsistent evidence with regard to cost savings in health care or reductions in hospitalization.
One of the earliest care management programs to be tested was the Medicare Alzheimer’s Disease Demonstration Project, which was designed to decrease nursing home placement by improving caregiver outcomes through case management and subsidized community services. Unfortunately, although more than 5,300 dyads participated, there were no substantial benefits to families including reductions in caregiver burden and nursing home placement (Miller et al., 1999; Newcomer et al., 1999). While more recent efforts have demonstrated more positive results, the efficacy of these approaches remains inconclusive as it concerns their impact on caregiver well-being, care costs, and health care utilization.
Several more recent and well-designed trials are promising however. The Maximizing Independence at Home (MIND) study is an 18-month care coordination program that addresses dementia-related care needs for the person with dementia and his or her family caregivers through individual-
ized care planning, referral and linkage to services, provision of dementia education and skill-building strategies, and care monitoring. Delivered by non-clinical community workers from participating social agencies trained and overseen by a team of geriatric psychiatrists, the MIND program resulted in a significant delay in time to all-cause transition from home to institutional settings compared to control participants. However, there was no significant group difference in the reduction of total percentage of unmet needs from baseline to 18 months, although families receiving the MIND program did have significant reductions in the proportion of unmet needs in safety and legal/advance care domains relative to controls. Intervention participants (older adults with dementia) had significant improvement in self-reported quality of life relative to control participants but this did not extend to their family caregivers (Samus et al., 2014). Initially tested in a randomized trial with 303 dyads, a variant of this intervention is currently being replicated in a large randomized trial (MIND Plus), which integrates other evidence-based care programs (such as COPE and TAP discussed earlier) and is being tested in a Center for Medicare & Medicaid Innovation grant.
The Partners in Dementia Care (PDC) is another effective care coordination program delivered via a partnership between the U.S. Department of Veterans Affairs (VA) Medical Centers (VAMCs) and Alzheimer’s Association chapters. It targets caregivers of veterans with dementia living in the community and who are receiving primary care from the VA. In this approach, care coordinators from VAMCs and Alzheimer’s Association chapters work as a team and share computerized records to assess, reassess, develop, and implement action plans and monitor the needs of veteran families via telephone and e-mail for 12 months. A randomized trial involving 486 caregivers demonstrated positive outcomes for caregivers at 6 months and more limited improvements at 12 months in the areas of unmet needs, caregiver strain, depression, and access to support resources (Bass et al., 2013). The care coordination approach also resulted in positive outcomes for the older adult veteran at 6 months that included reduced strain in relationships, depression, and unmet needs as well as less embarrassment about memory problems. At 12 months, more impaired veterans had further reductions in unmet need and embarrassment. The extent and type of improvement appeared to vary by levels of initial need and severity of impairments among veterans, suggesting that segments of the caregiver population need different levels and types of support.
Another care management program is the Alzheimer’s and Dementia Care Program (ADC) developed by the University of California, Los Angeles and launched in 2012. The ADC involves a nurse practitioner dementia care manager who partners with primary care physicians and local community-based organizations to provide comprehensive, coordinated,
and person-centered care for individuals with dementia and their families (Reuben et al., 2013).
Several care coordination models designed to link primary care to community-based programs are also promising. Using a cluster randomized trial involving 18 primary care clinics and 408 dyads (persons with dementia and their caregivers), Vickrey and colleagues (2006) showed that a 12-month care coordination model that linked families to needed community resources as well as to health care resulted in improved adherence to treatment guidelines, care recipient quality of life and caregiver social support, mastery of caregiving, and confidence. Similarly, Callahan and colleagues (2006) tested a collaborative care intervention led by a nurse practitioner who also used standard protocols, treatment guidelines, and nonpharmacologic management to improve recognition and treatment of Alzheimer’s disease in primary care. In a controlled trial involving 153 dyads, the individuals with dementia and their caregivers receiving the collaborative care management approach were more likely to rate the care they received as good or excellent, caregivers reported less distress and depression, and individuals with dementia had significantly fewer behavioral symptoms at 12 months.
In summary, although the evidence is still inconsistent, recent tests of care coordination models targeting family caregivers demonstrate benefits for both persons with dementia and their caregivers. Most of these approaches involve an initial assessment of caregiver and older adult needs, followed by coordination and linkages to address needs. Of importance is that each program differs from the other with regard to assessment used, level of caregiver involvement, case manager level of expertise, outcome measures, and results.
Respite Programs
Respite programs occur in a wide range of settings (e.g., in home and community group settings), are provided by multiple and diverse providers, and are based on the principle that providing caregivers episodic relief from their ongoing care responsibilities benefits caregivers health and well-being and secondarily persons receiving care (Kirk and Kagan, 2015). Respite typically refers to services that provide caregivers some time away from caregiver responsibilities. There is a wide range of respite-type programs. Some provide daily medical and social services to older adults such as adult day services that in turn afford family caregivers opportunities for respite. Respite care services are available in some communities for a few hours, 1 day, or a weekend.
Although the need for respite for many high-intensity or strained caregivers of older adults is widely recognized, families are not always aware
of the availability of these services, cannot or do not want to leave their family member, or do not have access to such services (IOM, 2012). Furthermore, it has been challenging to demonstrate that such programs are effective in achieving their goal of reducing the adverse effects of caregiving. This may be due in large part to methodological challenges and the lack of consensus in the design and implementation of these programs. Information is lacking regarding how best to provide respite to maximize its benefits to both caregivers and older adults (Kirk and Kagan, 2015). Initial studies of respite programs found inconclusive results (Reifler et al., 1992). However, a second generation of studies strongly suggests that respite helps to reduce caregiver distress (Zarit et al., 2014).
One especially promising venue for respite for caregivers is adult day services (ADS), which provide out-of-home, supervised, group services with the goals of improving mood, well-being, and quality of life of the caregiver and care recipient and enabling clients to remain at home for as long as possible. ADS also provide caregivers with respite from their day-to-day care responsibilities. Most ADS offer their services during “normal business hours,” although some offer flexible hours to meet the needs of working caregivers. Although the number of ADS programs is increasing, not every community/state offers ADS thus limiting access to these programs for families.
A recent integrative review of 19 research studies suggests that ADS benefit both the older adult client and the family caregiver although outcomes depend on the range and quality of services provided. For caregivers the treatment of the person with dementia and the availability of caregiver support services and opportunities for training in dementia care were important indicators of quality and use of the ADS. Collectively, the findings show that caregivers report feeling supported, having improved competency in care provision, and reduced burden associated with care provision (Tretteteig et al., 2016). Another recent study involving 158 family caregivers who were using ADS, demonstrated that use of ADS improved regulation of the stress hormone cortisol. Caregivers’ daytime cortisol responses on days they used ADS were compared to the days in which they did not. The study demonstrated that ADS use improved caregiver cortisol regulation, which in turn has potential to enhance long-term health outcomes (Klein et al., 2016). This important study is the first to link a social service program to biomarkers and to show physiological benefits for family caregivers.
Augmenting ADS with a systematic caregiver support program is also promising. The Adult Day Plus Program (ADS Plus) involves ADS staff who provide systematic care management, education, skills training, situational counseling, and ongoing support to family caregivers through face-to-face and telephone contact based on an initial needs assessment. The
intervention is intensive over the first 3 months and then ongoing support is provided up to 12 months. Of 106 caregivers participating in a pilot trial that used a cluster RCT design (two sites assigned to an intervention and one to a control condition), at 3 months, caregivers receiving ADS Plus reported less depression, improved confidence managing behaviors, and enhanced well-being compared to caregivers using ADS only. Long-term effects (12 months) showed that compared to the ADS only users, ADS Plus caregivers continued to report less depression and more confidence, used ADS for more days, and had fewer nursing home placements (Gitlin et al., 2006).
Thus, the benefits of respite opportunities, particularly those offered through ADS that provide a safe and secure setting for older adults, appear to be amplified by providing systematic support and education in addition to the time for respite. Research is further needed to determine the added value of respite-type services to existing evidence-based programs for family caregivers. Perhaps a dyadic focus (e.g., services for the older adult coupled with caregiver respite and other forms of support) results in better outcomes for caregivers and older adult recipients of care. Also, the long-term benefits of respite care to caregivers have not yet been demonstrated (Kansagara et al., 2010).
SOCIETAL/POLICY LEVEL
Policies and programs delivered at the societal or policy level such as Medicare payment rules and Medicaid waiver programs can also be viewed as interventions. These are discussed in the previous chapters and include those listed below. The previous chapters describe federal programs that provide direct services to caregivers of older adults (see Chapter 1), state and federal policies regarding unpaid or paid leave for family caregivers (see Chapter 4), and health care and LTSS policies that affect caregivers (see Chapter 6). These policies include
- the NFCSP;
- the VA Caregiver Support Program;
- FMLA;
- state and local paid sick leave statutes;
- various provisions in the Patient Protection and Affordable Care Act related to caregivers’ role as decision makers, caregiver assessment, quality metrics, and testing of new care models that promote person- and family-centered care;
- Medicaid’s consumer-directed options for home- and community-based services (e.g., Cash and Counseling);
- state-funded caregiver support programs;
- the Caregiver Advise, Record, Enable (CARE) Act;
- the Lifespan Respite Act;
- Medicare hospice benefits that provide interventions for both the person and the family; and
- the National Alzheimer’s Project Act (NAPA).
Medicaid policy concerning home- and community-based services for people with disabilities illustrates how state and federal policy can benefit family caregivers. Many state Medicaid programs offer consumer-directed options to Medicaid beneficiaries who are eligible for home- and community-based programs. Cash and Counseling, for example, was evaluated in the original demonstration program in three states (Arkansas, Florida, and New Jersey). While implementation varied in some ways, each demonstration provided consumers a monthly allowance to hire individuals (including family caregivers) or to help them purchase goods and services related to their care (e.g., counseling and related financial recordkeeping). In the demonstration, eligible Medicaid beneficiaries who volunteered to participate were randomly assigned to Cash and Counseling or usual Medicaid services (control group). Overall, the evaluation of the program found beneficial effects for both care recipients and their caregivers. The participants’ primary family caregivers reported significantly less physical, emotional, and financial stress than the caregivers assigned to the control group and lower rates of adverse health effects from caregiving (Brown et al., 2007; Carlson et al., 2007). Fifteen states continue to operate Cash and Counseling program and many other states have implemented similar programs.
With respect to cost, although program spending may be higher for those in the Cash and Counseling demonstration group versus usual care (e.g., agency-directed care), a new study by Coe and colleagues (2016) suggests that the program saved costs and led to improved health outcomes for participants when family caregivers were provided pay for their assistance. As anticipated, the treatment group spent significantly more Medicaid dollars on counseling, had more family involvement, and received more paid hours of care and fewer unpaid hours of care compared to the control group. However, paid family care resulted in substantial decreases for inpatient expenditures (emergency room and inpatient use), suggesting that family involvement in home care may be a substitute for hospital care. Family involvement also significantly decreased Medicaid utilization and lowered the likelihood of infections (e.g., urinary tract infections, bedsores, respiratory infections) (Coe, 2016).
THE ROLE OF TECHNOLOGY IN CAREGIVER INTERVENTIONS
In a broad sense, technology refers to the tools, equipment, machines, technical processes, or methods that are used to accomplish a task or activity. Family caregivers interact with technology to access information and support (e.g., via Internet searches, patient portals, electronic medical records, and social media), as a means of accessing or participating in intervention programs (e.g., via videoconferencing, telephone) or to provide care to an older adult. Caregivers may also use technology to monitor the functional status of a care recipient and employ telemedicine technologies to communicate with providers who can monitor the care recipient and/or the caregiver.
The Internet, videophones, videoconferencing, and other communication technologies are increasingly used to support family caregivers. They have several potential advantages including reduced cost (e.g., less travel to providers’ offices, fewer home visits by nurses); the ability to offer the intervention to greater numbers of individuals; enhanced flexibility with respect to tailoring and presentation of information; and convenience. Technology applications may be especially beneficial to long-distance caregivers in terms of enhancing access to the care recipient and other care providers. However, Internet-based technologies can also pose challenges to caregivers: not everyone has access; connectivity can be problematic; technology is constantly changing; and technology-based interventions may not be reimbursable. Nor can technology always substitute for face-to-face interactions between interventionist and caregiver (Berkowsky and Czaja, 2015; Czaja et al., 2012). Issues regarding cost and access are important areas for policy makers to consider when crafting state and federal regulations regarding payment for technology.
The range of technologies used for intervention delivery includes simple technology such as the telephone, screen-phones, videophones, touch-screen computers, videoconferencing, GPS systems, and the Internet (Web-based interventions). For example, a recent study used a videophone to deliver a modified version of the REACH II multicomponent intervention to minority family caregivers of individuals with dementia (Czaja et al., 2013). The intervention was compared to an information only/nutrition attention control group. The results indicated that it was feasible to use the videophone to deliver the intervention and that the videophone intervention was efficacious with respect to caregiver outcomes. Caregivers who received the intervention reported a decrease in burden and an increase in perceived social support and positive perceptions of the caregiving experience. Beauchamp and colleagues (2005) evaluated the efficacy of a multimedia caregiver support program delivered over the Internet as compared to a usual care
wait list control. They found caregivers who received the intervention had improvements in stress, anxiety, depression, and self-efficacy.
In a systematic review of eight psychosocial interventions (i.e., couples-based cognitive behavior therapy (CBT) via video counseling, CD-based multimedia to enhance treatment decision making, Web-based CBT, Web-based for developing a shared care-plan for managing symptoms, multimedia caregiving training program, Internet combination of CBT and education, Web-based symptom management tool kit, and Web-based supportive education, marital therapy, dyadic coping skills training), Badr and colleagues (2015) found that information technology can be a useful tool for conveying health-related information and support to caregivers. The researchers also suggest that a major research gap is the lack of integration of Web 2.0 technologies such as social media in these intervention programs given that social support and communication serve as frequent targets of interventions for caregivers and individuals with cancer. They also suggest that the use of interactive health communication technologies (IHCT) in dyadic interventions in the cancer space is still in its infancy and that more research is needed to examine impact on outcomes for individuals with cancer and caregivers (e.g., impact on relationship, communication, burden, and distress).
Chodosh and colleagues (2015) compared two modes of delivery for a caregiver management program (ACCESS)—in-person visits plus telephone and mail or telephone and mail only and found that care quality improved substantially in both arms. To date, there are no reports of mobile applications (smartphones and tablet technologies); however, it is likely that these will emerge in the future given the recent focus on mobile health applications.
Caregiver intervention research has focused primarily on caregivers of older adults with ADRD, although a few studies have included caregivers of stroke survivors (Grant et al., 2002), individuals with spinal cord injuries (e.g., Elliot et al., 2008), frail older adults (e.g., Magnusson et al., 2005), persons in hospice (Mooney et al., 2014), individuals with heart failure (Piette et al., 2015), and individuals with depression (Aikens et al., 2015). The types of interventions delivered via technology are varied and include counseling, education, skill building, links to resources and services, support groups, chat rooms, and reminiscence cues. Outcomes are wide ranging and include caregiver knowledge, burden, self-efficacy, psychological morbidity (anxiety/depression), self-efficacy, skills, service utilization, and caregiver satisfaction with the technology. Although limited, these studies suggest that caregiver interventions can be delivered via technology and can improve caregiver outcomes.
Only a few studies have examined costs. Chodosh and colleagues (2015), as noted earlier, found that the telephone only plus mail arm was
more cost-effective than the in-person plus mail and telephone arm with respect to costs associated with intervention delivery. This suggests that interventions delivered via telephone or mail may be more economical than in-person clinic-based or home-based visits by health personnel especially with recent developments in technology, which allow for video communication. However, this issue needs to be evaluated more systematically. Dang and colleagues (2008) used videophones to provide support, education, resource access, and enhanced communication to caregivers of veterans with dementia. They found that total facility utilization costs (hospitals and clinic visits) decreased for caregivers who received the intervention. However, there was no comparison group in this study—all caregivers received the intervention. In general, the findings from this study also indicate that technology-based interventions are well received by caregivers and that issues of usability are important as is caregiver training on use of the technology.
Caregivers are also increasingly going online in pursuit of information and support. A recent report by the Pew Research Center (Fox et al., 2013) indicates that 84 percent of caregivers with Internet access go online to access information about a particular treatment or disease, medications, or health insurance. A majority of those caregivers (59 percent) report that the Internet has been helpful to their ability to provide care and support to older adults with disability. There are numerous websites that offer information and support to caregivers such as the websites from the National Alliance for Caregiving, the Family Caregiver Alliance, and the Alzheimer’s Association.1 AARP also includes a section for caregivers on its website. The NIH seniorhealth.gov website provides information on resources for caregivers such as support groups and educational materials as well as tips and videos for Alzheimer’s caregivers on topics such as managing medications, safety and driving, and legal issues.
Mobile health apps are also proliferating and can also provide caregivers with support and information. For example, the VA has developed a suite of mobile health apps to support family caregivers (Frisbee, 2014). These apps include the Care4Caregiver App that includes a self-assessment tool for tracking strain, tips for coping with stress, and connections to community resources; the Summary of Care App that allows veterans and their caregivers to receive and view VA medical information; and the Journal App, which is a personal health journal that allows veterans and
___________________
1 See http://www.caregiving.org; https://www.caregiver.org; https://www.alz.org/care; http://www.aarp.org/home-family/caregiving/caregiving-tools; http://nihseniorhealth.gov/alzheimerscare/caregiversupport/01.html; and http://nihseniorhealth.gov/endoflife/supportforcaregivers/01.html (accessed August 23, 2016).
their caregivers to enter, view, and track vital signs and patient-generated data that could be shared with their VA care team as well as several others.
Emerging sensing and monitoring technologies may also prove to be beneficial to caregivers, especially those who work or live apart from their care recipient. These technologies can help caregivers track the health status and activities of the care recipient. Home monitoring systems and tracking systems are currently available and many more are emerging. There are also task management applications that can help with care coordination and medication management. However, to date these programs have not been systematically evaluated.
Very few studies involving the use of technology have examined issues of diversity in terms of differences among subgroups (e.g., race/ethnicity); however several have included diverse populations, primarily African Americans and Latinos (Chodosh et al., 2015; Czaja et al., 2013; Dang et al., 2008; Winter and Gitlin, 2006). Generally, the results of these studies suggest that using technology for intervention delivery is both feasible and acceptable (i.e., are well received by caregivers). It is important to note, however, that currently minorities generally tend to have less Internet access at home; thus, technology access may be an issue for some subgroups of caregivers. Clearly there is a need to include representatives from other ethnic/racial backgrounds, LGBT caregivers, and caregivers in rural locations in technology intervention studies. There is also a need to examine the use of technology to aid caregivers in work settings. For example, monitoring technologies might be useful to working caregivers.
The development, implementation, and evaluation of technology to support caregivers could be enhanced by innovative partnerships between researchers and the technology industry. An example is Oregon Health & Science University and Intel who are partnering in the development and testing of technology products and devices such as unobtrusive intelligent systems, which include unobtrusive sensing and monitoring systems and algorithms and assessment techniques for detecting motor and cognitive changes in older adults in these community settings (Wild et al., 2008). TigerPlace, an innovative independent living environment designed to support aging in place, is another example of an innovative partnership among the University of Missouri, the state of Missouri, and a home care agency (Fergenson, 2013; Rantz et al., 2008). Partnerships between researchers and industry can also support caregiver access to technology. For example, Cisco and AT&T provided support to the VideoCare project through the provision of equipment, technology support, and funding to help defray the Internet costs for the caregivers (Czaja et al., 2013).
THE PIPELINE FOR CAREGIVER INTERVENTIONS
Although a wide range of programs have been tested in randomized clinical trials and have demonstrated small to moderate treatment effects on important outcomes, few caregivers have access to these programs. Unlike the drug discovery pipeline, there is not a similar trajectory for developing, evaluating, and then implementing interventions for families (Gitlin and Czaja, 2016). As most interventions are tested external to service delivery settings and payment mechanisms and/or for specific populations, they need to undergo a translational phase in which the program is adapted, shortened, or modified in some way to fit within the daily routines of an agency or clinic, the current structure of a payment mechanism, and/or the unique situations of diverse caregivers. Interventions that require extensive training of interventionists for their delivery also face challenges of scalability and delivery to reach all family caregivers in need. To move proven interventions for delivery into different health and human service settings, several actions may be required.
First, most individually targeted caregiver interventions have been developed and evaluated in efficacy trials with community-based populations. With few exceptions such as the ADS Plus intervention that was tested for delivery within ADS (Gitlin et al., 2006), the collaborative care model tested for delivery in primary care (Callahan et al., 2006), and the Guided Care program (Wolff et al., 2010), interventions have not been evaluated within a particular delivery context. Thus, most interventions require what has been referred to as a translational phase in which manuals, procedures, and protocols are revised in order to better fit a particular health and human service setting (Burgio et al., 2009; Gitlin et al., 2015; Nichols et al., 2016).
Proven interventions may also need to be adapted to meet the needs of different targeted populations or cultural groups. Adaptations may include changing format, setting, language, and health literacy levels of materials as well as evaluating cultural appropriateness (Nápoles et al., 2010). Even for those interventions tested for efficacy within a delivery setting, pilot testing may be necessary to identify effective implementation processes and strategies (e.g., marketing and referral procedures, workflow, staff training) prior to widespread dissemination and implementation. No studies to date have evaluated the feasibility of sustaining interventions over time and the business plans and associated costs. Remarkably few studies have taken into account the involvement of multiple caregivers and how best to tailor existing interventions and assess outcomes for such circumstances.
More research is needed to understand the best strategies for widespread dissemination and implementation of proven programs. A primary barrier has been the lack of adequate funding for this effort. Notably, only
two federal agencies have funded dissemination efforts. The Administration of Community Living (ACL) has funded dissemination efforts, but funding levels for ACL also remain low. The VA’s Caregiver Support Program funds dissemination of the REACH individual intervention and telephone support groups through training of VA staff, in addition to funding the online Building Better Caregivers classes. As implementation science provides the theoretical foundation and the evidentiary base for the strategies most effective in moving proven interventions into care settings, there is an opportunity to more rapidly overcome the research-to-practice gap in this area.
Other methodological challenges relate to understanding how interventions that primarily target older adults, such as care coordination or supportive services, impact family caregivers. Such programs are typically developed using embedded designs in which delivery is integrated and examined within mainstream care delivery and often include older adults with variable underlying diseases or conditions. The design and evaluation of such interventions present unique challenges relating to determining eligibility criteria for family caregivers, determining adequate sample to observe desired effects, and outcomes of care in light of the widely varied needs and circumstances of older adults (Wolff et al., 2010; Zarit et al., 2016). For example, caregivers who are not experiencing caregiving-related negative emotions, strain, or stress may nevertheless benefit from education and skills to increase their knowledge, confidence, and skills to provide care, or additional services to better support the older adult they assist that reduce time spent caregiving and caregiver-related work productivity loss. For such caregivers, appropriate outcomes may relate to the ability to participate in valued activities, confidence for the caregiving role, or the perceived quality of care delivered to the person they assist. Few interventions of older adults have developed programs for family caregivers or examined outcomes for family caregivers.
In addition to more federal funding for these efforts, private–public partnerships could be considered to advance the implementation of proven interventions. Exploration of how such interventions may fit within existing and new funding streams, health care organizations such as Accountable Care Organizations, and/or existing programs, such as the NFCSP, would be important to pursue and should be a high priority for helping families and health and human service professionals gain access to evidence-based programs (Lynn and Montgomery, 2015).
CONCLUSIONS
The committee’s key findings and conclusions are described in detail in Box 5-5. In summary, the committee concludes that there is a rich body of research assessing a wide range of caregiver interventions designed to help
alleviate the negative consequences of caregiving, especially for caregivers of older adults with dementia. Well-designed trials, conducted in a variety of settings, have demonstrated that caregiver education and skills training, environmental modifications of care recipients’ homes, care coordination and management, counseling, self-care and relaxation training, respite programs, and other approaches can improve quality of life for both caregivers and care recipients, increase caregivers’ abilities and confidence, and delay care recipients’ institutionalization.
Trials have also demonstrated that interventions that involve caregivers may reduce the resource use of care recipients by delaying nursing home placement, reducing rehospitalizations, and shortening hospital stays.
Effective caregiver interventions tend to share several characteristics. They incorporate an assessment of caregivers’ needs, tailor the intervention accordingly, and consider the caregivers’ preferences. Training programs that actively involve caregivers in learning a particular skill result in better outcomes compared to didactic, prescriptive approaches such as handing out information sheets.
Yet, few of the nation’s millions of family caregivers of older adults have access to evidence-based interventions. Numerous barriers have stymied translation of research successes into everyday settings. Many interventions have not been evaluated in or integrated into real-world settings where third-party reimbursement rules and financial and other organizational constraints prevail.
Wider dissemination of effective caregiver interventions will also require attention to the limitations of the available evidence. So far, trials have only rarely included sufficient numbers of diverse caregivers and care recipients to allow an assessment of their effectiveness for Hispanic, African American, and other ethnic and racial groups; long-distance caregivers; LGBT caregivers; and others. Future trials should assess whether existing models are effective across diverse populations to determine if further modifications or cultural adaptations are needed. Additional work is also needed to identify optimal strategies to disseminate and sustain effective models in diverse communities (Nápoles et al., 2013). Most caregiving research has focused primarily on improving outcomes for family caregivers of persons with Alzheimer’s disease and often on a single caregiver rather than on situations where multiple family caregivers are involved. Although the lessons learned from these studies likely apply to a variety of caregiving contexts, additional research on caregivers of older adults with other impairments is needed.
REFERENCES
Aikens, J. E., R. Trivedi, A. Heapy, P. N. Pfeiffer, and J. D. Piette. 2015. Potential impact of incorporating a patient-selected support person into mHealth for depression. Journal of General Internal Medicine 30(6):797-803.
Alzheimer’s Association. 2014. 2014 Alzheimer’s disease fact and figures. https://www.alz.org/downloads/facts_figures_2014.pdf (accessed December 14, 2015).
Appelbaum, A., and W. Breitbart. 2013. Care for the cancer caregiver: A systematic review. Palliative and Supportive Care 11(3):231-252.
Badr, H., C. L. Carmack, and M. A. Diefenbach. 2015. Psychosocial interventions for patients and caregivers in the age of new communication technologies: Opportunities and challenges in cancer care. Journal of Health Communications 20(3):328-342.
Bakas, T., P. C. Clark, M. Kelly-Hayes, R. B. King, B. J. Lutz, and E. L. Miller. 2014. Evidence for stroke family caregiver and dyad interventions: A statement for healthcare professionals from the American Heart Association and American Stroke Association. Stroke 45(9):2836-2852.
Bass, D. M., P. A. Clark, W. J. Looman, C. A. McCarthy, and S. Eckert. 2003. The Cleveland Alzheimer’s managed care demonstration: Outcomes after 12 months of implementation. The Gerontologist 43(1):73-85.
Bass, D. M., K. S. Judge, A. Lynn Snow, N. L. Wilson, R. Morgan, W. J. Looman, C. A. McCarthy, K. Maslow, J. A. Moye, R. Randazzo, M. Garcia-Maldonado, R. Elbein, G. Odenheimer, and M. E. Kunik. 2013. Caregiver outcomes of Partners in Dementia Care: Effect of a care coordination program for veterans with dementia and their family members and friends. Journal of the American Geriatrics Society 61(8):1377-1386.
Beauchamp, N., A. B. Irvine, J. Seeley, and B. Johnson. 2005. Work-site-based Internet multimedia program for family caregivers of persons with dementia. The Gerontologist 45(6):793-801.
Belle, S., L. Burgio, R. Burns, D. Coon, S. Czaja, D. Gallagher-Thompson, L. Gitlin, J. Klinger, K. Koepke, C. Lee, J. Martindale-Adams, L. Nichols, R. Schulz, S. Stahl, A. Stevens, L. Winter, and S. Zhang. 2006. Enhancing the quality of life of dementia caregivers from different ethnic or racial groups: A randomized, controlled trial. Annals of Internal Medicine 145(10):727-738.
Berkowsky, R. W., and S. J. Czaja. 2015. The use of technology in behavioral intervention research: Advantages and challenges. In Behavioral intervention research: Designing, evaluating, and implementing, edited by L. Gitlin and S. J. Czaja. New York: Springer. Pp. 119-136.
Bertrand, R. M., L. Fredman, and J. Saczynski. 2006. Are all caregivers created equal? Stress in caregivers to adults with and without dementia. Journal of Aging and Health 18(4):534-551.
Biegel, D. E., and R. Schulz. 1999. Caregiving and caregiver interventions in aging and mental illness. Family Relations 48(4):345-354.
Black, B. S., D. Johnston, P. V. Rabins, A. Morrison, C. Lyketsos, and Q. M. Samus. 2013. Unmet needs of community-residing persons with dementia and their informal caregivers: Findings from the Maximizing Independence at Home Study. Journal of the American Geriatrics Society 61(12):2087-2095.
Brodaty, H., and C. Arasaratnam. 2012. Meta-analysis of nonpharmacological interventions for neuropsychiatric symptoms of dementia. American Journal of Psychiatry 169(9): 946-953.
Brodaty, H., A. Green, and A. Koschera. 2003. Meta-analysis of psychosocial interventions for caregivers of people with dementia. Journal of the American Geriatric Society 51(5):657-664.
Brown, R., B. L. Carlson, S. Dale, L. Foster, B. Phillips, and J. Schore. 2007. Cash and Counseling improving the lives of Medicaid beneficiaries who need personal care or home and community-based services. Princeton, NJ: Mathematica Policy Research.
Burgio, L. D., I. B. Collins, B. Schmid, T. Wharton, D. McCallum, and J. DeCoster. 2009. Translating the REACH caregiver intervention for use by area agency on aging personnel: The REACH OUT program. The Gerontologist 49(1):103-116.
Caldwell, J. 2006. Consumer-directed supports: Economic, health, and social outcomes for families. Mental Retardation 44(6):405-417.
Callahan, C. M., M. A. Boustani, F. W. Unverzagt, M. G. Austrom, T. M. Damush, A. J. Perkins, B. A. Fultz, S. L. Hui, S. R. Counsell, and H. C. Hendrie. 2006. Effectiveness of collaborative care for older adults with Alzheimer disease in primary care: A randomized controlled trial. Journal of the American Medical Association 295(18):2148-2157.
Callahan, C. M., G. Arling, W. Tu, M. B. Rosenman, S. R. Counsell, T. E. Stump, and H. C. Hendrie. 2012. Transitions in care for older adults with and without dementia. Journal of the American Geriatrics Society 60(5):813-820.
Carlson, B. L., L. Foster, S. B. Dale, and R. Brown. 2007. Effects of Cash and Counseling on personal care and well-being. Health Services Research 42(1p2):467-487.
Chee, Y. K., L. N. Gitlin, M. P. Dennis, and W. W. Hauck. 2007. Predictors of adherence to a skill-building intervention in dementia caregivers. Journal of Gerontology Medical Sciences 62(6):673-678.
Cheung, K. S., B. H. Lau, P. W. Wong, A. Y. Leung, V. W. Lou, G. M. Chan, and R. Schulz. 2015. Multicomponent intervention on enhancing dementia caregiver well-being and reducing behavioral problems among Hong Kong Chinese: A translational study based on REACH II. International Journal of Geriatric Psychiatry 30(5):460-469.
Chodosh, J., B. A. Colaiaco, K. I. Connor, D. W. Cope, H. Liu, D. A. Ganz, M. J. Richman, D. L. Cherry, J. M. Blank, R. P. Carbone, S. M. Wolf, and B. G. Vickrey. 2015. Dementia care management in an underserved community: The comparative effectiveness of two different approaches. Journal of Aging and Health 27(5):864-893.
Clark, P. A., D. M. Bass, W. J. Looman, C. A. McCarthy, and S. Eckert. 2004. Outcomes for patients with dementia from the Cleveland Alzheimer’s managed care demonstration. Aging & Mental Health 8(1):40-51.
Coe, N. B., J. Guo, R. T. Konetzka, and C. H. Van Houtven. 2016. What is the marginal benefit of payment-induced family care? National Bureau of Economic Research Working Paper No. 22249. http://www.nber.org/papers/w22249 (access June 27, 2016).
Czaja, S. J., L. N. Gitlin, R. Schulz, S. Zhang, L. D. Burgio, A. B. Stevens, N. O. Nichols, and D. Gallagher-Thompson. 2009. Development of the Risk Appraisal Measure: A brief screen to identify risk areas and guide interventions for dementia caregivers. Journal of the American Geriatrics Society 57(6):1064-1072.
Czaja, S. J., C. C. Lee, and R. Schulz. 2012. Quality of life technologies in supporting family caregivers. In Quality of life technology handbook, edited by R. Schulz. Boca Raton, FL: CRC Press. Pp. 245-262.
Czaja, S. J., D. Loewenstein, R. Schulz, S. N. Nair, and D. Perdomo. 2013. A videophone psychosocial intervention for dementia caregivers. American Journal of Geriatric Psychiatry 21(11):1071-1081.
Dang, S., N. Remons, J. Harris, J. Malphurs, L. Sandals, A. Lozada Cabrera, and N. Nedd. 2008. Care coordination assisted by technology for multiethnic caregivers of persons with dementia: A pilot clinical demonstration project on caregiver burden and depression. Journal of Telemedicine and Telecare 14(8):443-447.
Dassel, K. B., D. C. Carr, and P. Vitaliano. 2015. Does caring for a spouse with dementia accelerate cognitive decline? Findings from the Health and Retirement Study. The Gerontologist: Advance Access. http://gerontologist.oxfordjournals.org/content/early/2015/11/18/geront.gnv148.abstract (accessed April 6, 2015).
Dixon, L., C. Adams, and A. Lucksted. 2000. Update on family psychoeducation for schizophrenia. Schizophrenia Bulletin 26(1):5-20.
Eisdorfer, C., S. J. Czaja, D. Loewenstein, M. P. Rubert, S. Argüelles, V. B. Mitrani, and J. Szapocznik. 2003. The effect of a family therapy and technology-based intervention on caregiver depression. The Gerontologist 43(4):521-531.
Elliott, T. R., and J. W. Berry. 2009. Brief problem-solving training for family caregivers of persons with recent-onset spinal cord injuries: A randomized controlled trial. Journal of Clinical Psychology 65(4):406-422.
Elliott, T. R., D. Brossart, J. W. Berry, and P. R. Fine. 2008. Problem-solving training via videoconferencing for family caregivers of persons with spinal cord injuries: A randomized controlled trial. Behaviour Research and Therapy 46(11):1220-1229.
Family Caregiver Alliance. 2006. Caregiver assessment: Principles, guidelines and strategies for change, Vol. I. San Francisco, CA.
Family Caregiver Alliance. 2012. Selected caregiver assessment measures: A resource inventory for practitioners, 2nd ed. https://www.caregiver.org/sites/caregiver.org/files/pdfs/SelCGAssmtMeas_ResInv_FINAL_12.10.12.pdf (accessed April 6, 2016).
Feil, D. G., R. Lukman, B. Simon, A. Walston, and B. Vickrey. 2011. Impact of dementia on caring for patients’ diabetes. Aging & Mental Health 15(7):894-903.
Feinberg, L. F., and C. Levine. 2015. Family caregiving: Looking to the future. Generations 39(4):11-20.
Fergenson, M., 2013. TigerPlace: An innovative “Aging in Place” community. The American Journal of Nursing 113(1):68-69.
Fortinsky, R. H., M. Kulldorff, A. Kleppinger, and L. Kenyon-Pesce. 2009. Dementia care consultation for family caregivers: Collaborative model linking an Alzheimer’s Association chapter with primary care physicians. Aging & Mental Health 13(2):162-170.
Fox, S., M. Duggan, and K. Purcell. 2013. Family caregivers are wired for health. Washington, DC: Pew Research Center’s Internet & American Life Project. http://www.pewinternet.org/2013/06/20/family-caregivers-are-wired-for-health (accessed March 23, 2016).
Friedman, E. M., R. A. Shih, K. M. Langa, and M. D. Hurd. 2015. U.S. prevalence and predictors of informal caregiving for dementia. Health Affairs 34(10):1637-1641.
Frisbee, K. 2014. Assessing the impact of mobile health apps on family caregiver burden levels and the factors predicting mobile health app use. International Journal of Integrated Care 14(8):e035.
Gallagher-Thompson, D., W. Haley, D. Guy, M. Rupert, T. Argüelles, L. M. Zeiss, C. Long, S. Tennstedt, and M. Ory. 2003. Tailoring psychological interventions for ethnically diverse dementia caregivers. Clinical Psychology: Science and Practice 10(4):423-438.
Gaugler, J. E., M. Reese, and M. S. Mittelman. 2013. Effects of the NYU Caregiver Intervention-Adult Child on residential care placement. The Gerontologist 53(6):985-997.
Gearing, R. E., N. El-Bassel, A. Ghesquiere, S. Baldwin, J. Gillies, and E. Ngeow. 2011. Major ingredients of fidelity: A review and scientific guide to improving quality of intervention research implementation. Clinical Psychology Review 31(1):79-88.
Gitlin, L. N., and Czaja, S. J. 2016. Behavioral intervention research: Designing, evaluating and implementing. New York: Springer.
Gitlin, L., and N. Hodgson. 2015. Caregivers as therapeutic agents in dementia care: The evidence-base for interventions supporting their role. In Family caregiving in the new normal, edited by J. E. Gaugler and R. L. Kane. London, UK: Academic Press. Pp. 305-353.
Gitlin, L. N., M. Corcoran, L. Winter, A. Boyce, and W. W. Hauck. 2001. A randomized, controlled trial of a home environmental intervention effect on efficacy and upset in caregivers and on daily function of persons with dementia. The Gerontologist 41(1):4-14.
Gitlin, L. N., L. Winter, M. Corcoran, M. P. Dennis, S. Schinfeld, and W. W. Hauck. 2003. Effects of the home environmental skill-building program on the caregiver-care recipient dyad: 6-month outcome from the Philadelphia REACH initiative. The Gerontologist 43(4):532-546.
Gitlin, L. N., K. Reever, M. P. Dennis, E. Mathieu, and W. W. Hauck. 2006. Enhancing quality of life of families who use adult day services: Short- and long-term effects of the Adult Day Services Plus program. The Gerontologist 46(5):630-639.
Gitlin, L. N., L. Winter, J. Burke, N. Chernett, M. P. Dennis, and W. W. Hauck. 2008. Tailored activities to manage neuropsychiatric behaviors in persons with dementia and reduce caregiver burden: A randomized pilot study. The American Journal of Geriatric Psychiatry 16(3):229-239.
Gitlin, L. N., N. Hodgson, E. Jutkowitz, and L. Pizzi. 2010a. The cost-effectiveness of a nonpharmacologic intervention for individuals with dementia and family caregivers: The Tailored Activity Program. The American Journal of Geriatric Psychiatry 18(6):510-519.
Gitlin, L. N., L. Winter, M. P. Dennis, N. Hodgson, and W. W. Hauck. 2010b. Targeting and managing behavioral symptoms in individuals with dementia: A randomized trial of a nonpharmacological intervention. Journal of the American Geriatrics Society 58(8):1465-1474.
Gitlin, L. N., L. Winter, M. P. Dennis, N. Hodgson, and W. W. Hauck. 2010c. A biobehavioral home-based intervention and the well-being of patients with dementia and their caregivers: The COPE randomized trial. Journal of the American Medical Association 304(9):983-991.
Gitlin, L. N., K. Marx, I. H. Stanley, and N. Hodgson. 2015. Translating evidence-based dementia caregiving interventions into practice: State-of-the-science and next steps. The Gerontologist 55(2):210-226.
Gitlin, L. N., N. A. Hodgson, and S. S. W. Choi. 2016. Home-based interventions targeting persons with dementia: What is the evidence and where do we go from here? In Dementia care, edited by M. Boltz and J. Galvin. New York: Springer. Pp. 167-188.
Grant, J. S., T. R. Elliott, M. Weaver, A. A. Bartolucci, and J. N. Giger. 2002. Telephone intervention with family caregivers of stroke survivors after rehabilitation. Stroke 33:2060-2065.
Greenfield, E. A., A. E. Scharlach, A. J. Lehning, J. K. Davitt, and C. L. Graham. 2013. A tale of two community initiatives for promoting aging in place: Similarities and differences in the national implementation of NORC programs and villages. The Gerontologist 53(6):928-938.
Heller, T., and J. Caldwell. 2006. Supporting aging caregivers and adults with developmental disabilities in future planning. Mental Retardation 44(3):189-202.
Heller, T., A. B. Miller, and K. Hsieh. 1999. Impact of a consumer-directed family support program on adults with developmental disabilities and their family caregivers. Family Relations 48(4):419-427.
Hepburn, K. W., J. Tornatore, B. Center, and S. W. Ostwald. 2001. Dementia family caregiver training: Affecting beliefs about caregiving and caregiver outcomes. Journal of the American Geriatrics Society 49(4):450-457.
Hepburn, K. W., M. Lewis, C. W. Sherman, and J. Tornatore. 2003. The Savvy Caregiver Program: Developing and testing a transportable dementia family caregiver training program. The Gerontologist 43(6):908-915.
Hepburn, K., M. Lewis, J. Tornatore, C. W. Sherman, and K. L. Bremer. 2007. The Savvy Caregiver Program: The demonstrated effectiveness of a transportable dementia caregiver psychoeducation program. Journal of Gerontological Nursing 33(3):30-36.
HHS (U.S. Department of Health and Human Services). 2014. National plan to address Alzheimer’s disease: 2014 update.https://aspe.hhs.gov/national-plan-address-alzheimer%E2%80%99s-disease-2014-update (accessed February 12, 2016).
Hodgson, N., L. N. Gitlin, L. Winter, and W. W. Hauck. 2014. Caregiver’s perceptions of the relationship of pain to behavioral and psychiatric symptoms in older community residing adults with dementia. The Clinical Journal of Pain 30(5):421-427.
Huang, T., and S. Liang. 2005. A randomized clinical trial of the effectiveness of a discharge planning intervention in hospitalized elders with hip fracture due to falling. Journal of Clinical Nursing 14(10):1193-1201.
IOM (Institute of Medicine). 2012. Living well with chronic illness: A call for public health action. Washington, DC: The National Academies Press.
Jennings, L. A., D. B. Reuben, L. C. Evertson, K. S. Serrano, L. Ercoli, J. Grill, J. Chodosh, Z. Tan, and N. S. Wenger. 2015. Unmet needs of caregivers of individuals referred to a dementia care program. Journal of the American Geriatrics Society 63(2):282-289.
Judge, K. S., D. M. Bass, A. L. Snow, N. L. Wilson, R. Morgan, W. J. Looman, C. McCarthy, and M. E. Kunik. 2011. Partners in dementia care: A care coordination intervention for individuals with dementia and their family caregivers. The Gerontologist 51(2):261-272.
Jutkowitz, E., L. N. Gitlin, and L. T. Pizzi. 2010. Evaluating willingness-to-pay thresholds for dementia caregiving interventions: Application to the Tailored Activity Program. Value in Health 13(6):720-725.
Kajiyama, B., L. W. Thompson, T. Eto-Iwase, M. Yamashita, J. Di Mario, Y. Marian Tzuang, and D. Gallagher-Thompson. 2013. Exploring the effectiveness of an Internet-based program for reducing caregiver distress using the iCare Stress Management e-Training Program. Aging & Mental Health 17(5):544-554.
Kalra, L., A. Evans, I. Perez, A. Melbourn, A. Patel, M. Knapp, and N. Donaldson. 2004. Training carers of stroke patients: Randomised controlled trial. BMJ 328(7448):1099-1104.
Kansagara, D., E. Goy, and M. Freeman. 2010. A systematic evidence review of interventions for non-professional caregivers of individuals with dementia. Washington, DC: U.S. Department of Veterans Affairs.
Kasper, J. D., V. A. Freedman, B. C. Spillman, and J. L. Wolff. 2015. The disproportionate impact of dementia on family and unpaid caregiving to older adults. Health Affairs 34(10):1642-1649.
Kelly, K., N. Wolfe, M. Gibson, and L. F. Feinberg. 2013. Listening to family caregivers: The need to include caregiver assessment in Medicaid home and community-based service waiver programs. Washington, DC: AARP Public Policy Institute.
Kirk, R. S., and J. Kagan. 2015. A research agenda for respite care: Deliberations of an expert panel of researchers, advocates and funders.http://archrespite.org/images/docs/2015_Reports/ARCH_Respite_Research_Report_web.pdf (accessed March 22, 2016).
Klein, L. C., K. Kim, D. M. Almeida, E. E. Femia, M. J. Rovine, and S. H. Zarit. 2016. Anticipating an easier day: Effects of adult day services on daily cortisol and stress. The Gerontologist 56(2):303-312.
Knowles, J. 2006. Donepezil in Alzheimer’s disease: An evidence-based review of its impact on clinical and economic outcomes. Core Evidence 1(3):195-219.
Kwok, T., I. I. Bel Wong, K. Chui, D. Young, and F. Ho. 2013. Telephone-delivered psychoeducational intervention for Hong Kong Chinese dementia caregivers: A single-blinded randomized controlled trial. Clinical Interventions in Aging 8:1191-1197.
Legrain, S., F. Tubach, D. Bonnet-Zamponi, A. Lemaire, J. P. Aquino, E. Paillaud, E. Taillandier-Heriche, C. Thomas, M. Verny, B. Pasquet, A. L. Moutet, D. Lieberherr, and S. Lacaille. 2011. A new multimodal geriatric discharge-planning intervention to prevent emergency visits and rehospitalizations of older adults: The optimization of medication in AGEd multicenter randomized controlled trial. Journal of the American Geriatrics Society 59(11):2017-2028.
Levine, C., D. E. Halper, J. L. Rutberg, and D. A. Gould. 2013. Engaging family caregivers as partners in transitions: TC-QuIC: A quality improvement collaborative. New York: United Hospital Fund.
Lingler, J. H., L. M. Martire, and R. Schulz. 2005. Caregiver-specific outcomes in antidementia clinical drug trials: A systematic review and meta-analysis. Journal of the American Geriatrics Society 53(6):983-990.
Long, K. H., J. P. Moriarty, M.S. Mittelman, and S. S. Foldes. 2013. Estimating the potential cost savings from The New York University Caregiver Intervention in Minnesota. Health Affairs 33(4):596-604.
Lynn, J., and A. Montgomery. 2015. Creating a comprehensive care system for frail elders in “age boom” America. The Gerontologist 55(2):278-285.
Magnusson, L., E. Hanson, and M. Nolan. 2005. The impact of information and communication technology on family carers of older people and professionals in Sweden. Ageing and Society 25(5):693-713.
Martindale-Adams, J., L. O. Nichols, R. Burns, M. J. Graney, and J. Zuber. 2013. A trial of dementia caregiver telephone support. Canadian Journal of Nursing Research 45(4):30-48.
Maslow, K. 2011. Dementia, diabetes and family caregiving. Aging & Mental Health 15(8): 933-935.
Maslow, K. 2012. Translating innovation to impact: Evidence-based interventions to support people with Alzheimer’s disease and their caregivers at home and in the community. Washington, DC: Alliance for Aging Research, Administration on Aging, and MetLife Foundation.
Miller, R., R. Newcomer., and P. Fox. 1999. Effects of the Medicare Alzheimer’s Disease Demonstration on nursing home entry. Health Services Research 34(3):691-714.
Mittelman, M. S. 2008. Psychosocial intervention research: Challenges, strategies and measurement issues. Aging & Mental Health 12(1):1-4.
Mittelman, M. S., S. H. Ferris, G. Steinberg, E. Shulman, J. A. Mackell, A. Ambinder, and J. Cohen. 1993. An intervention that delays institutionalization of Alzheimer’s disease patients: Treatment of spouse-caregivers. The Gerontologist 33(6):730-740.
Mittelman, M. S., S. H. Ferris, E. Shulman, G. Steinberg, A. Ambinder, J. A. Mackell, and J. Cohen. 1995. A comprehensive support program: Effect on depression in spouse-caregivers of AD patients. The Gerontologist 35(6):792-802.
Mittelman, M. S., S. H. Ferris, E. Shulman, G. Steinberg, and B. Levin. 1996. A family intervention to delay nursing home placement of patients with Alzheimer disease: A randomized controlled trial. Journal of the American Medical Association 276(21):1725-1731.
Mittelman, M. S., W. E. Haley, O. J. Clay, and D. L. Roth. 2006. Improving caregiver wellbeing delays nursing home placement of patients with Alzheimer disease. Neurology 67(9):1592-1599.
Molazem, Z., T. Falahati, I. Jahanbin, P. Jafari, and S. Ghadakpour. 2014. The effect of psycho-educational interventions on the quality of life of the family caregivers of the patients with spinal cord injury: A randomized controlled trial. International Journal of Community Based Nursing and Midwifery 2(1):31-39.
Moon, H., C. J. Whitlatch, M. H. Kim, and P. Dilworth-Anderson. 2014. Predictors of discrepancy between care recipients with early-stage dementia and their caregivers on perceptions of the care recipients’ quality of life. Paper presented at Society for Social Work and Research 18th Annual Conference: Research for Social Change: Addressing Local and Global Challenges, January 17. San Antonio, TX.
Mooney, K., P. Berry, B. Wong, and G. Donaldson. 2014. Helping cancer-family caregivers with end-of-life home symptom management: Initial evaluation of an automated symptom monitoring and coaching system. Paper presented at ASCO Annual Meeting, October 24. Chicago, IL.
Nápoles, A. M., L. Chadia, R. Eveersley, and G. Moreno-John. 2010. Developing culturally sensitive caregiver interventions: Are we there yet? American Journal of Alzheimer’s Disorders and Other Dementias 25(5):389-406.
Nápoles, A. M., J. Santoyo-Olsson, and A. L. Stewart. 2013. Methods for translating evidence-based behavioral interventions for health-disparity communities. Preventing Chronic Disease 10:E193.
Navaie, M. 2011. Accessibility of caregiver education and support programs: Reaching hard-to-reach caregivers. Education and Support Programs for Caregivers: Research, Practice, Policy 13-28.
Naylor, M. D., D. Brooten, R. Campbell, B. S. Jacobsen, M. D. Mezey, M. V. Pauly, and J. S. Schwartz. 1999. Comprehensive discharge planning and home follow-up of hospitalized elders—A randomized clinical trial. JAMA 281(7):613-620.
Newcomer, R., C. Yordi, R. DuNah, P. Fox, and A. Wilkinson. 1999. Effects of the Medicare Alzheimer’s Disease Demonstration on caregiver burden and depression. Health Services Research 34(3):669-689.
Nichols, L. O., C. Chang, A. Lummus, R. Burns, J. Martindale-Adams, M. J. Graney, D. W. Coon, and S. Czaja. 2008. The cost-effectiveness of a behavior intervention with caregivers of patients with Alzheimer’s disease. Journal of the American Geriatrics Society 56(3):413-420.
Nichols, L. O., J. Martindale-Adams, R. Burns, M. J. Graney, and J. Zuber. 2011. Translation of a dementia caregiver support program in a health care system—REACH VA. Archives of Internal Medicine 171(4):353-359.
Nichols, L. O., J. Martindale-Adams, R. Burns, J. Zuber, and M. J. Graney. 2016. REACH VA: Moving from translation to system implementation. The Gerontologist 56(1):135-144.
Northouse, L. L., M. Katapodi, L. Song, L. Zhang, and D. W. Mood. 2010. Interventions with family caregivers of cancer patients: Meta-analysis of randomized trials. CA: A Cancer Journal for Clinicians 60(5):317-339.
Ory, M. G., R. R. Hoffman, J. L. Yee, S. Tennstedt, and R. Schulz. 1999. Prevalence and impact of caregiving: A detailed comparison between dementia and nondementia caregivers. The Gerontologist 39(2):177-185.
Ostwald, S. K., K. W. Hepburn, W. Caron, T. Burns, and R. Mantell. 1999. Reducing caregiver burden: A randomized psychoeducational intervention for caregivers of persons with dementia. The Gerontologist 39(3):299-309.
Piette, J. D., D. Striplin, N. Marinec, J. Chen, and J. E. Aikens. 2015. A randomized trial of mobile health support for heart failure patients and their informal caregivers: Impacts on caregiver-reported outcomes. Medical Care 53(8):692-699.
Pimouguet, C., T. Lavaud, J. Dartigues, and C. Helmer. 2010. Dementia case management effectiveness on health care costs and resource utilization: A systematic review of randomized controlled trials. The Journal of Nutrition, Health and Aging 14(8):669-676.
Rantz, M., M. Aud, G. L. Alexander, D. P. Oliver, D. Minner, M. Skubic, J. M. Keller, Z. He, M. Popescu, G. Demiris and S. J. Miller. 2008. TigerPlace: An innovative educational and research environment. In AAAI Fall Symposium: AI in eldercare: New solutions to old problems Pp. 84-91.
Redfoot, D., L. Feinberg, and A. Houser. 2013. The aging of the baby boom and the growing care gap: A look at future declines in the availability of family caregivers. Washington, DC: AARP Public Policy Institute.
Reifler, B. V., R. S. Henry, K. A. Sherrill, and C. H. Asbury. 1992. A national demonstration program on dementia day centers and respite services: An interim report. Behavior, Health, & Aging 2(3):199-206.
Reinhard, S. C., R. Montgomery, and M. J. Gibson. 2008. Informal caregivers: Sustaining the core of long-term services and supports. Paper presented at Building Bridges: Making a Difference in Long-Term Care Fifth Annual Long-Term Care Colloquium, Washington, DC.
Reuben, D. B., L. C. Evertson, N. S. Wenger, K. Serrano, J. Chodosh, L. Ercoli, and Z. S. Tan. 2013. The University of California at Los Angeles Alzheimer’s and Dementia Care program for comprehensive, coordinated, patient-centered care: Preliminary data. Journal of American Geriatrics Society 61(12):2214-2218.
Richards, K. C., C. A. Enderlin, C. Beck, J. C. McSweeney, T. C. Jones, and P. K. Roberson. 2007. Tailored biobehavioral interventions: A literature review and synthesis. Research and Theory for Nursing Practice 21(4):271-285.
Rosalynn Carter Institute. 2012. REACH II intervention (Schulz and colleagues). http://www.rosalynncarter.org/caregiver_intervention_database/dimentia/reach_ii_intervention (accessed February 17, 2016).
Roth, D. L., M. S. Mittelman, O. J. Clay, A. Madan, and W. E. Haley. 2005. Changes in social support as mediators of the impact of a psychosocial intervention for spouse caregivers of persons with Alzheimer’s disease. Psychology and Aging 20(4):634-644.
Samus, Q. M., D. Johnston, B. S. Black, E. Hess, C. Lyman, A. Vavilikolanu, J. Pollutra, J. M. Leoutsakos, L. N. Gitlin, P. V. Rabins, and C. G. Lyketsos. 2014. A multidimensional home-based care coordination intervention for elders with memory disorders: The Maximizing Independence at Home (MIND) pilot randomized trial. American Journal of Geriatric Psychiatry 22(4):398-414.
Schulz, R., S. J. Czaja, A. Lustig, B. Zdaniuk, L. M. Martire, and D. Perdomo. 2009. Improving the quality of life of caregivers of persons with spinal cord injury: A randomized controlled trial. Rehabilitation Psychology 54(1):1-15.
Somme, D., H. Trouve, M. Dramé, D. Gagnon, Y. Couturier, and O. Saint-Jean. 2012. Analysis of case management programs for patients with dementia: A systematic review. Alzheimer’s & Dementia 8(5):426-436.
Stanley, M. A., J. Calleo, A. L. Bush, N. Wilson, A. L. Snow, C. Kraus-Schuman, A. L. Paukert, N. J. Petersen, G. A. Brenes, and P. E. Schulz. 2013. The Peaceful Mind program: A pilot test of a cognitive–behavioral therapy–based intervention for anxious patients with dementia. The American Journal of Geriatric Psychiatry 21(7):696-708.
Stevens, A. B., E. R. Smith, L. R. Trickett, and R. McGhee. 2012. Implementing an evidence-based caregiver intervention within an integrated healthcare system. Translational Behavioral Medicine 2(2):218-227.
Stevens, A., J. Cho, J. L. Thorud, and M. Ory. 2016 (unpublished). Live well our health initiative year 6 6 month report. Fort Worth, TX: United Way of Tarrant County, Scott & White Healthcare, and Texas A&M Health Science Center.
Tam-Tham, H., M. Cepoiu-Martin, P. E. Ronksley, C. J. Maxwell, and B. R. Hemmelgarn. 2013. Dementia case management and risk of long-term care placement: A systematic review and meta-analysis. International Journal of Geriatric Psychiatry 28(9):889-902.
Tappen, R. M., and D. Hain. 2014. The effect of in-home cognitive training on functional performance of individuals with mild cognitive impairment and early-stage Alzheimer’s disease. Research in Gerontological Nursing 7(1):14-24.
Tremont, G., J. D. Davis, G. D. Papandonatos, B. R. Ott, R. H. Fortinsky, P. Gozalo, M. S. Yue, K. Bryant, C. Grover, and D. S. Bishop. 2015. Psychosocial telephone intervention for dementia caregivers: A randomized, controlled trial. Alzheimer’s & Dementia 11(5):541-548.
Tretteteig, S., S. Vatne, and A. M. M. Rokstad. 2016. The influence of day care centres for people with dementia on family caregivers: An integrative review of the literature. Aging & Mental Health 20(5):450-462.
Vickrey, B. G., B. S. Mittman, K. I. Connor, M. L. Pearson, R. D. Della Penna, T. G. Ganiats, J. R. W. DeMonte, J. Chodosh, X. Cui, S. Vassar, N. Duan, and M. Lee. 2006. The effect of a disease management intervention on quality and outcomes of dementia care: A randomized, controlled trial. Annals of Internal Medicine 145(10):713-726.
Whitlatch, C. J., K. Judge, S. H. Zarit, and E. Femia. 2006. Dyadic intervention for family caregivers and care receivers in early-stage dementia. The Gerontologist 46(5):688-694.
Wild, K., L. Boise, J. Lundell, and A. Foucek. 2008. Unobtrusive in-home monitoring of cognitive and physical health: Reactions and perceptions of older adults. Journal of Applied Gerontology 27(2):181-200.
Winter, L., and L. N. Gitlin. 2006. Evaluation of a telephone-based support group intervention for female caregivers of community-dwelling individuals with dementia. American Journal of Alzheimer’s Disease and Other Dementias 21(6):391-397.
Wolff, J. L., E. R. Giovannetti, C. M. Boyd, L. Reider, S. Palmer, D. Scharfstein, J. Marsteller, S. T. Wegener, K. Frey, B. Leff, K. D. Frick, and C. Boult. 2010. Effects of guided care on family caregivers. The Gerontologist 50(4):459-470.
Zarit, S., and E. Femia. 2008. Behavioral and psychosocial interventions for family caregivers. American Journal of Nursing 108(9 Suppl):47-53.
Zarit, S. H., K. Kim, E. E. Femia, D. M. Almeida, and L. C. Klein. 2014. The effects of adult day services on family caregivers’ daily stress, affect, and health: Outcomes from the Daily Stress and Health (DaSH) study. The Gerontologist 54(4):570-579.
Zarit, S. H., L. R. Bangerter, Y. Liu, and M. J. Rovine. 2016. Exploring the benefits of respite services to family caregivers: Mmethodological issues and current findings. Aging & Mental Health (E-publication ahead of print). http://www.tandfonline.com/doi/abs/10.1080/13607863.2015.1128881 (accessed April 8, 2016).
This page intentionally left blank.




















































