The relationships between buildings and microbes discussed in this report suggest that human microbial exposures may be modulated and controlled through interventions related to building design, construction, and operation. Such interventions are intended to improve human health and have two broad goals: (1) reducing human exposure to harmful microbes and (2) encouraging human exposure to beneficial microbes. This chapter describes existing and potential interventions for modifying human microbial exposures to improve health. The discussion identifies potential trade-offs associated with such interventions, such as increased energy consumption or building costs. The focus is on control of air- and surface-borne microbes in buildings.1 Where relevant, selected results on the effectiveness of common interventions for the built environment with respect to the microbiome and human health are presented.
There are a wide variety of biological particles and chemicals of both biological and nonbiological origin inside buildings, multiple sources of these agents, and varied exposure routes, leading to myriad intervention approaches that merit consideration. Both physical- and chemical-based interventions exist or have been proposed for controlling or reducing microbial exposures in buildings. These types of interventions include changes to building design and operation, such as control of ventilation rates or the use of air filtration systems, and the use of disinfectants to inactivate viable microorganisms. Other types of interventions focus on the promotion of exposures to potentially beneficial microorganisms, such as by enhancing building and human connections with outdoor microbial diversity. Assessment of the appropriateness of these interventions will need to go beyond their effectiveness at changing building microbial exposures to include broader cost-benefit analyses that consider positive outcomes; weigh potential negative outcomes; and include economic factors, environmental factors, and effects on human health. The chapter includes a discussion of approaches to better understanding and optimizing interventions and their potential trade-offs in the framework of building engineering controls.
PHYSICAL INTERVENTIONS TO REDUCE EXPOSURE TO HAZARDOUS MICROBES
Physical interventions are most commonly practiced through engineering controls in the built environment. These typically include local or systemic changes to temperature, ventilation, moisture, and light (including ultraviolet [UV]).
___________________
1 Most traditional control measures for microorganisms in water supplies, such as filtration and inclusion of disinfectants at water treatment facilities, are conducted outside of the building.
Changes to Ventilation Practice
Increasing outdoor air ventilation rates is a common strategic intervention intended to reduce occupants’ microbial exposure in buildings, either through dilution of indoor air with outdoor air of different or lower microbial loads, or by supporting indoor environmental conditions (i.e., changes to relative humidity) that are less conducive to microbial growth. All buildings experience some degree of indoor–outdoor air exchange, some of which is purposeful (ventilation) and some unintended (infiltration). Mechanical ventilation approaches, whether natural or hybrid, are preferred to infiltration because they provide better control of the ventilation, the ability to treat outdoor contaminants and dehumidify outdoor air, and potentially the ability to reduce energy impacts by recovering or discharging heat from outgoing air.
A simplified material balance equation for a single-zone space and steady-state contaminant concentration with no internal loss terms due to deposition or engineered control systems is shown below:

where Cin (mass or number per volume) is the steady-state indoor contaminant concentration, Cout (mass or number per volume) is the outdoor concentration, P (unitless) is the penetration factor for outdoor contaminants, G is the indoor contaminant generation rate (mass or number per time), and Qo (volume per time) is the outdoor airflow rate into the building being considered.
The impacts of ventilation can be understood by considering the terms in Equation 5.1. In this case, airborne microorganisms are considered the “contaminant” in the equation. Assuming for purposes of discussion that all terms in the equation are constant, increasing the outdoor ventilation rate (Qo) will decrease the term G/Qo on the right side of the equation, decreasing the level of the indoor contaminant. However, if the outdoor concentration of this contaminant is greater than zero, the indoor concentration can never be lower than PCout (the amount of the contaminant that penetrates indoors from outside). Increasing the rate of outdoor air ventilation is effective as an intervention only when the air contaminant concentration outdoors is lower than that indoors. A possible practical effect, for example, could be to decrease the concentration of bacteria with an indoor source (such as shedding from humans) while increasing the concentration of fungi introduced from outdoor air.
This same conclusion is valid when considering water vapor as a contaminant. In that case, the material balance is more complicated than
reflected in Equation 5.1, as it needs to consider temperature effects; moisture removal by the heating, ventilation, and air conditioning (HVAC) system; and moisture storage in building materials. Because microorganisms require moisture to grow, increased ventilation with humid outdoor air is not likely to be an effective intervention without careful consideration of the impact on indoor moisture levels or the addition of dehumidification (possibly in association with air conditioning).
The paradigm of using ventilation for treatment of indoor air has been applied mainly to removal of chemical contaminants with exclusive or primarily indoor sources. This model can be extended to many infectious viruses and other microorganisms, where humans are the sources and the microbial concentrations outdoors are considered to be very low. However, a number of hazardous microbes, such as plant-derived and fungal allergens, commonly have an outdoor source that is more important than any indoor sources. Thus, increasing ventilation would be an effective strategy for such species only if the outdoor air were subjected to filtration or other treatment.
In considering the impact of increased outdoor air ventilation on levels of indoor airborne contaminants, it is also important to consider the loss mechanisms that could be added to Equation 5.1 and the generation mechanisms embodied in the term G. Loss mechanisms include removal through particle filtration in the ventilation system or in-room filtration devices, as well as the decay of microbial infectivity and particle and gas deposition onto surfaces. It is important to recognize here that hypersensitivity potential (e.g., allergenicity) is often not related to the decay of infectious potential. Generation mechanisms include human microbial shedding, resuspension of microorganisms from surfaces, and desorption of volatile organic compounds (VOCs) from surfaces to air. Semivolatile organic compounds (SVOCs) adsorbed by building materials, including SVOCs produced by microbes, are very slow to release or partition into the air, making ventilation less effective in their removal than removal of more volatile compounds. The relative magnitude of these mechanisms compared with dilution through ventilation will determine the overall effectiveness of increased ventilation as an intervention.
Analyses of material and mass balance need to take particle size into account because deposition rates, resuspension rates, filtration efficiencies, and building penetration rates depend strongly on this parameter (Nazaroff, 2004). The average geometric mean diameters for bioaerosol particles that contain bacteria and fungi (which may be in the form of aggregated cells and/or spores attached to particulate matter) have been reported to be 5.5 and 5.9 μm, respectively (Hospodsky et al., 2015). Particles in this size range penetrate building envelopes inefficiently and can be removed effectively by most ventilation system filters. For viruses, a study of day
care centers and airplanes found that 64 percent of influenza genomes in air were associated with particles smaller than 2.5 μm in diameter (Yang et al., 2011), corresponding to lower deposition rates and effective removal by ventilation. However, resuspension of microbes in densely occupied settings, such as classrooms, can dramatically increase indoor air concentrations of bacteria and fungi, even at high outdoor air ventilation rates (Hospodsky et al., 2015). To understand the more complex systems that represent real buildings and built environment microbiomes, it would be necessary to develop separate mass balance equations that appropriately treat generation and loss rates for each microbe or contaminant of interest. Given the diversity of microorganisms that form built environment microbiomes, developing and integrating individual mass balance equations for each can be challenging, although the creation of models based on this concept that are more precise than the simplified Equation 5.1 will be useful.
Natural Ventilation and Envelope Tightness
Employing natural ventilation (i.e., ventilation without use of mechanical systems) instead of or in addition to mechanical ventilation has been advocated as an intervention for its potential benefits for occupants’ health and comfort. Studies have shown that the prevalence of symptoms of sick building syndrome is lower in naturally ventilated than in mechanically ventilated buildings, but the mechanism for this finding has not been established (Seppänen and Fisk, 2002). Natural ventilation often is proposed to increase outdoor ventilation rates, which will decrease the levels of internally generated airborne microbes. However, outdoor air entering buildings through natural ventilation may not be (cost) effectively captured for filtration and conditioning. As a result, such practices can introduce outdoor contaminants, including allergens and moisture, at undesirable levels, and therefore may not necessarily provide net improvements with regard to indoor environmental conditions. Moreover, the rates of outdoor air entry and the distribution of this air within a building must be carefully considered for appropriate building analyses in this context. More advanced systems that provide better control of ventilation rates and air distribution than those often found in the United States (CIBSE, 2005; Schulze and Eicker, 2013) may warrant further study for domestic implementation.
Increasing the tightness of the building envelope is another way to affect ventilation and exposures in buildings. In both mechanically and naturally ventilated buildings, the entry of unconditioned, unfiltered, and infiltrated air will interfere with the performance of the ventilation system. Reducing infiltration results in better indoor temperature and humidity control, lower likelihood of indoor moisture accumulation, and lower entry rates of outdoor microbes, all of which can reduce the likelihood of elevated
indoor microbe levels. In buildings ventilated predominantly by infiltration through spurious air leakage, which includes most U.S. residences, envelope tightening provides for better airflow control. However, tighter building envelopes need to be accompanied by reliable ventilation, mechanical or natural, that meets the outdoor air exchange and humidity control requirements of a building.
Studies on Building Ventilation and Microbial Exposures
Associations between increased outdoor ventilation rates (typically >10–15 liters/second per person) and improved human health are well established (Bornehag et al., 2005; Menzies and Bourbeau, 1997). Common symptoms noted in ventilation and health studies include allergies and other hypersensitivity responses, respiratory infections, and neurological and other symptoms. As a result, it is plausible that microbes are an important mediator between ventilation and health (Sundell et al., 2011). In cases in which hazardous microbial agents have exclusive indoor sources (e.g., influenza virus and the bacterium Mycobacterium tuberculosis), the importance of outdoor air ventilation is predictable. In one study, for example, installation and operation of heat recovery ventilators (ventilation systems that bring outdoor air into buildings and transfer heat from the outgoing airstream to the incoming airstream) in Inuit homes was found to result in reductions in reported wheeze and rhinitis (Kovesi et al., 2009).
More broadly, studies in naturally and mechanically ventilated commercial buildings (Meadow et al., 2014) have demonstrated that the rate and method of ventilation may influence the composition of bacterial communities in indoor air. These relationships are affected by such factors as occupancy level, ventilation design and operation, and outdoor microbe concentrations and ecology. In a hospital study, bacteria closely related to human pathogens were relatively more abundant in rooms with lower ventilation rates, while rooms with natural ventilation had a more diverse bacterial community compared with mechanically ventilated rooms (Kembel et al., 2012). In a recent school study aimed at disentangling the impacts of ventilation and occupancy, indoor air concentrations of bacteria and fungi were dominated by indoor sources associated with human occupancy (resuspension and shedding) rather than outdoor air ventilation. This was true for all but the extreme cases of high ventilation rate and high outdoor air microbial concentration (Hospodsky et al., 2015).
Moisture Control
Decreasing Relative Humidity
Evidence indicates that decreasing relative humidity (RH), as modified by ventilation, can impact occupant microbial exposures. Increased ventilation in Swedish homes, for example, is indirectly associated with lower concentrations of airborne dust mite allergens because dust mite activity and growth are associated with high RH. In the cold, dry Scandinavian climates, increased outdoor air ventilation results in lower indoor RH levels (Sundell et al., 1995). Increased outdoor ventilation to reduce RH, especially in cold climates, is a commonly recommended approach for controlling mold growth in buildings. Sustained building RH levels greater than 70 percent are associated with fungal growth on building materials (Arundel et al., 1986), while building RH levels below 50 percent are expected to contribute to transmission of influenza (Yang et al., 2012). Although evidence suggests that greater biodiversity surrounding people’s homes influences the classes of bacteria on their skin and is associated with reduced incidence of allergic disposition (atopy) (Hanski et al., 2012), there is little evidence that increased outdoor air ventilation has a benefit for immune system development.
The literature indicates general consensus that ventilation, either direct or through the control of RH, influences the concentration and ecology of microbes in buildings. In most cases, and especially for potentially beneficial microbes, this information is not sufficiently developed to support design decisions or well-founded recommendations on how to design, control, and operate ventilation systems specifically for control of microbial communities and exposures.
Remediating in Damp Buildings
Dampness and visible mold have been reported in approximately 50 percent of U.S. homes (Mudarri and Fisk, 2007; Spengler et al., 1994). Water damage and visible mold inside buildings are consistently associated with respiratory and allergic health effects in infants, children, and adults (Mendell et al., 2011). Prior research and practice have led to the development of some operative approaches for reducing associated health symptoms. Intense remediation and environmental intervention for homes with moisture sources and visible mold have been shown to reduce some microbial exposures and can reduce allergy symptoms and asthma morbidity. In research previously sponsored by the National Institutes of Health (NIH), the U.S. Department of Housing and Urban Development, and the U.S. Environmental Protection Agency (EPA), remediation that included
removal of water-damaged building materials and alteration of HVAC systems resulted in a large decrease in asthma symptom days and asthma exacerbation versus a no-remediation control group (Kercsmar et al., 2006). A 2004 report of the Institute of Medicine reviews building dampness, associated mold growth, and health impacts and recommends the development of national guidelines for preventing indoor dampness, as well as economic or other incentives to spur adherence to moisture prevention practice by those that construct and manage buildings (IOM, 2004).
An additional concern regarding interventions for damp buildings is the lack of critical guidance on when to initiate costly remediation and whether remediation efforts are successful. The Institute for Inspection, Cleaning and Restoration Certification (IICRC) has established widely used guidance documents in the mold and building restoration industry, such as American National Standards Institute (ANSI)/IICRC S520 2015, Standard and Reference Guide for Professional Mold Remediation. The purpose of remediation is to restore the property to an acceptable state similar to that prior to the occurrence of the indoor mold contamination, designated by S520 as “Condition 1.” Condition 1 is defined as “(normal fungal ecology): an indoor environment that may have settled spores, fungal fragments or traces of actual growth whose identity, location, and quantity are reflective of a normal fungal ecology for a similar indoor environment” (ANSI/IICRC, 2015, p. 16).
The major uncertainty in the S520 document and all other mold remediation guidance is that there are no accepted methods for defining and quantifying the “normal fungal ecology” reference point. The types, abundances, and concentrations of microbial taxa that constitute the normal ecology of a building are not clearly defined, and they likely differ based on such environmental variables as climate and land use (Amend et al., 2010; Kembel et al., 2014; Qian et al., 2012; Reponen et al., 2011). Such variability could potentially be addressed by the detailed ecological information leveraged by modern DNA sequencing technologies and bioinformatics analyses now used in building microbiome studies. Gaps in knowledge about what represents “normal” microbial ecology and how to interpret particular microbial findings are one reason that building microbial sampling often is not recommended. The issue of microbial sampling for building microbial assessment and remediation strategies is beyond the scope of this report.2
___________________
2 The American Industrial Hygiene Association, for example, has a position statement on Mold and Dampness in the Built Environment (AIHA, 2013) that provides guidance on remediation efforts.
Particle Filtration
It is common practice to treat the air in building HVAC systems using particle filtration, which could be an effective intervention for removing airborne microbes. Particle filtration has been used in buildings for many decades, primarily to reduce fouling of heat transfer surfaces by particulate matter in outdoor air and airborne organic substances in the outdoor airstream. More recently, the benefits of reducing indoor fine and ultrafine particle concentrations for occupant health have been considered, leading to requirements for and use of higher levels of filter efficiency in buildings. Conventional air filter performance is a strong function of particle diameter. Smaller particles (on the order of 0.1 μm in diameter and lower) are removed primarily via diffusion, in which random motion of the particle leads to contact with a fiber in the filter and subsequent capture. Larger particles (diameters of about 1 μm and larger) are carried along by the airstream and collide with fibers as a result of impaction. Both diffusion and impaction are less effective at particle removal between roughly 0.1 μm and 1 μm. Thus, removal efficiencies are lower in this size range, which encompasses the known aerodynamic diameter ranges of some viruses, such as influenza (Lindsley et al., 2010), but this range is smaller than most indoor bacteria and fungi (Hospodsky et al., 2015).
Particle filtration efficiencies are rated based on testing using American Society of Heating, Refrigerating and Air-Conditioning Engineers (ASHRAE) Standard 52.2 (ASHRAE, 2017), which leads to filters being classified using the so-called Minimum Efficiency Reporting Value (MERV) scale.3 MERV ratings range from 1 to 20, with higher values corresponding to more effective particle removal. ASHRAE Standard 62.1 requires MERV 8 filters upstream of wetted surfaces in HVAC systems to reduce the accumulation of organic matter on these surfaces and the subsequent likelihood of microbial growth (ASHRAE, 2010). The standard also requires MERV 6 filters in the outdoor air intake when outdoor levels of particulate matter (PM)10 exceed ambient air quality standards and MERV 11 filters when PM2.5 exceeds ambient standards. It is important to note that filtration will treat only air passing through the filters, making filter installation and sealing important to achieving the intended level of particle removal. Filtration is an additional loss mechanism that could be incorporated into Equation 5.1, the rate of which will be affected by removal efficiency and amount of airflow through the filter.
Low-rise residential buildings that are ventilated by infiltration and open windows do not generally provide for systemic filtration of outdoor
___________________
3 MERV is a scale used to indicate the effectiveness of air filters. Fewer particles will pass through filters with a higher MERV rating.
air, although some loss of particles is expected to result from deposition as outdoor air penetrates the building envelope, with particles below 0.1 and greater than 1 μm in diameter penetrating less efficiently (Nazaroff, 2004). Thus, filtration is achieved by the circulation of indoor air through a residential HVAC system. Among U.S. residences with HVAC systems, surveys indicate that 25 percent use HVAC filters of MERV 5 or less, 60 percent use MERV 6–8, 10 percent use MERV 12, and 5 percent use MERV 16 (Stephens and Siegel, 2012). Portable air filtration systems—that is, standalone units typically intended to provide filtration in a single room—are employed in some applications. These units are rated using the so-called clean air delivery rate (CADR) based on an Association of Home Appliance Manufacturers (AHAM) test method that converts the particle removal rate to an equivalent volumetric airflow (dilution) rate.
The particle removal efficiency of MERV filters can be estimated as a function of particle aerodynamic diameter (Azimi et al., 2014; EPA, 2008), and the particle size distributions of several relevant viruses, bacteria, and fungi have been described (Hospodsky et al., 2015; Lindsley et al., 2010; Pastuszka et al., 2000; Yamamoto et al., 2014). Studies on human health improvements due to filtration have been reviewed for residential and commercial buildings. Results suggest that filtration does result in reductions in exposure to biologic particles, including cat, dog, and dust mite allergens, and offers modest improvements in allergy or asthma severity (Fisk, 2013). Although the fundamental information exists for testing and predicting the effects of filtration on reducing microbial exposure and associated disease, this research is not well developed.
A suite of less common air treatment technologies for particle removal, including electrostatically enhanced filtration and electrostatic precipitation, has previously been reviewed (EPA, 2008).
UV Germicidal Irradiation
Microbes can be transported to and from indoor surfaces via aerosol routes, and it is recognized by the engineering and medical communities that airborne microbes deposit onto fomites and vice versa in response to human activity and common environmental perturbations (Prussin and Marr, 2015; see also Chapter 3). Thus, disinfection practice needs to include both air and surfaces. UV germicidal irradiation (UVGI) is one common physical approach for disinfection of air and surfaces.
Exposure to light between wavelengths of 100 and 400 nanometers (nm) (UV-A, -B, -C) can damage the DNA of living organisms and result in an inability to replicate, thus rendering a cell noninfectious. UVGI, typically from low-pressure mercury vapor lamps with spectral power distribution focused at 254 nm, has been applied to disinfect indoor air and surfaces for
more than 50 years (Reed, 2010). UV systems that emit wavelengths below 242 nm can generate ozone and are not appropriate for air disinfection in occupied buildings. While UVGI can be effective at microbial inactivation, its effect against the allergenic or toxigenic properties of microbes is not well described. UV disinfection has commonly been adapted for use within the upper levels of rooms for effective air disinfection, placed on cooling coils to reduce microbial growth on and fouling of coils, placed in air-supply ducts, and more recently applied in surface disinfection. Guidelines are available for use by practitioners in applying this relatively mature technology to a variety of indoor settings (ASHRAE, 2016). Although UV equipment for residential systems is readily available from HVAC vendors in the domestic consumer market, UV air treatment generally has been dominated by health care applications and specialty commercial sectors (ASHRAE, 2016).
UV Air Disinfection
Upper-level UVGI typically is applied in health care settings for the purpose of interrupting transmission of Mycobacterium tuberculosis. The technology entails irradiation of the upper ~20 percent of a room with UV-C lamps (reviewed by Brickner et al., 2003). This technology leverages rapid vertical mixing of air in a room rather than removal of air from a room and subsequent treatment in a ventilation duct. (It should be noted that UV sources present acute radiation exposure risks to occupants, and UV lamps must be specially louvered for in-room applications [Sliney, 2013].) Full-room experiments have demonstrated the utility of upper-level UVGI for reducing infectious bacterial exposures to occupants (Xu et al., 2003), and the effects of environmental conditions, especially RH, on UV inactivation efficiency have been documented for bacterial pathogens and surrogates (Peccia et al., 2001). There also exists epidemiologic evidence of the effectiveness of upper-level UVGI. A significantly lower incidence of influenza in a veterans hospital tuberculosis (TB) ward equipped with upper-level UVGI suggests the efficacy of this approach in decreasing airborne transmission of influenza virus (McLean, 1961). Similarly, a clinical trial in an HIV–TB ward in Lima, Peru, demonstrated a more than double reduction in TB infection rate in wards equipped with UVGI versus those without (Escombe et al., 2009).
UV Surface Disinfection
Surface disinfection by UVGI has recently focused on the use of mobile UV units in hospitals to disinfect surfaces, as well as irradiation of cooling coils in large building HVAC systems. The mobile technology is new, and
the operation and workflow of these units have not been optimized. However, recent randomized trials have found a significantly reduced relative risk of infection for methicillin-resistant Staphylococcus aureus (MRSA) and vancomycin-resistant enterococci (VRE) when terminal UV disinfection was added to the standard cleaning regimen in hospital rooms (Anderson et al., 2017; Weber et al., 2016). In that study, large and significant reductions in the culturable concentrations of MRSA, VRE, and multidrug-resistant Acinetobacter were also observed on hospital room surfaces. And multiple studies suggest reductions in Clostridium difficile hospital infections when UV-C technology is used to treat hospital room surfaces (Levin et al., 2013; Miller et al., 2015; Nagaraja et al., 2015).
The application of UV on HVAC cooling coils has been recognized for its potential to inhibit microbial fouling of heat transfer equipment, thereby reducing pressure drops across the system and improving heat transfer (Luongo et al., 2017; Wang et al., 2016). An additional benefit of irradiating coils is the previously observed reductions in respiratory, mucosal, and musculoskeletal symptoms in workers in a building when UVGI was installed in the HVAC system. Reductions of symptoms in that study were coincident with UV operation and a large reduction in microbial and endotoxin concentration on irradiated surfaces within the ventilation system (Menzies et al., 2003).
CHEMICAL INTERVENTIONS TO REDUCE EXPOSURE TO HAZARDOUS MICROBES
Chemical interventions in the built environment focus on the inactivation of surface-bound microbes through the use of chemical disinfectants and, to a lesser extent, on the introduction of antimicrobial materials. These types of interventions include the treatment of air and surfaces through chemical disinfection and the design and use of antimicrobial materials or coatings.
Chemical Disinfection
Surface-associated microorganisms are a central component of the indoor microbiome. Important sources of microbes on surfaces include tracked-in dust, microbes shed from humans, settled airborne microbial aerosols and droplets, and microbial growth (Adams et al., 2013; Grant et al., 1989; Roberts et al., 1999; see also Chapter 3). In addition to contact- and fomite-based exposure, resuspension of microbes from flooring and elevated surfaces often is a significant source of airborne bacteria and inhalation exposure to bacteria and fungi (Bhangar et al., 2014; Hospodsky et al., 2014; Qian et al., 2012). Thus, chemical disinfection of surfaces is
an important type of intervention for modifying exposure to infectious microorganisms and viruses. A significant literature describes studies testing the efficacy of disinfection in schools, health care facilities, and industrial facilities (e.g., food preparation) where elevated surfaces and flooring are considered reservoirs of infectious bacteria and viruses (Donskey, 2013).
Regarding the building microbiome, uncertainty exists in chemical and other surface disinfection practices in two important areas. First, the rate at which the redevelopment of surface microbial communities occurs is poorly understood, resulting in uncertainty in determining optimal cleaning practices. This rate is affected by building parameters that include occupancy, ventilation, building materials, and moisture. Second, growing evidence suggests that early-life exposure to house dust containing increased fungal and bacterial diversity (Dannemiller et al., 2014; Ege et al., 2011) and elevated content of some specific bacteria (Lynch et al., 2014) may be protective concerning the development of asthma and recurrent wheeze in children (see Chapter 2). The impact of chemical cleaning interventions on the exposure of children to the chemicals and chemical by-products, as well as to these microorganisms, and on health outcomes is not known.
It is also important to note that tremendous differences exist among indoor building materials, all of which have different porosities, as well as chemical compositions that affect their ability to host dirt, microbes, skin cells, hair, and other human effluents in and on which microbes survive. Specialty practices exist for introducing aerosols containing oxidants and surfactants inside built environments for the express purpose of indoor disinfection. These practices most often include, but are not limited to, the introduction of ozone, vaporized hydrogen peroxide, chlorine derivatives (vaporized hypochlorous acid and chlorine dioxide), and peracetic acid microdroplets (Boyce, 2016). Like UV irradiation, however, aerosolized chemical disinfection practices present acute exposure risks to indoor occupants that need to be carefully managed prior to utilization. Oxidizing aerosol applications in the residential sector remain limited to the remediation of large-scale water damage and use in some health care settings. These strong chemical oxidants are known to react with indoor building materials (Hubbard et al., 2009), and in the case of ozone, carbonyls are released as ozonation by-products (Poppendieck et al., 2007). Because of costs and liabilities, the health care and government building sectors will likely remain the largest users of oxidizing aerosols.
Antimicrobial Materials
The indoor built environment has a plethora of textile and nontextile surfaces constructed of a wide range of natural and synthetic materials. A significant fraction of indoor surfaces, including textiles, have incorporated
antibiotics and metal nanoparticles—often silver—for the express purpose of imparting antimicrobial properties to their facade or other structural base (Chen and Schluesener, 2008). Recognizing that the association of microbes with furnishings and structural surfaces can negatively affect those in close contact, as well as the surface itself, the manufacturing of specialty surfaces incorporating broad-spectrum biocides has received increased attention for built environment design, especially in the health care sector but also in homes. Comprehensive reviews address antimicrobial finishing practices, qualitative and quantitative evaluations of antimicrobial efficacy, and methods for applying antimicrobial agents. Some of the most recent developments in antimicrobial treatment of surfaces and textiles include using various active agents, such as metal nanoparticles, quaternary ammonium salts, polyhexamethylene biguanide, triclosan, chitosan, dyes, and regenerable halamine compounds (Gao and Cranston, 2008; Hasan et al., 2013). Activating antimicrobial surfaces with such metals as copper or silver and applying liquid compounds, including biocidal paints, that confer on surfaces persistent antimicrobial activity are additional strategies that require validation and further investigation for built environment application.4
INTERVENTIONS TO ENCOURAGE EXPOSURE TO BENEFICIAL MICROBES
Although the focus of a variety of microbially motivated building interventions has been on reducing exposure to harmful microbes, a more recent emphasis is on encouraging exposure to potentially beneficial microbes (an idea also known as “environmental probiotics”). The nascent arena concerning beneficial microbe exposures within buildings is emerging into the following major perspectives. First, environmental probiotics may protect against colonization by and expansion of opportunistic pathogens in the environment and thus reduce human exposure to an infectious or otherwise harmful agent. This concept has initially been explored in the context of pathogen control in plumbing systems (Wang et al., 2013) and the proposed
___________________
4 An antimicrobial pesticide must be registered by the U.S. Environmental Protection Agency under the Federal Insecticide, Fungicide, and Rodenticide Act (FIFRA), 7 U.S.C. § 136 et seq., prior to sale or distribution within the United States. An antimicrobial pesticide is defined as any product intended to disinfect, sanitize, reduce, or mitigate the growth or development of microbiological organisms or protect inanimate objects, industrial processes or systems, surfaces, water, or other chemical substances from contamination, fouling, or deterioration caused by bacteria, viruses, fungi, protozoa, algae, or slime. Wood preservatives and antifoulants are also classified as antimicrobial pesticides if the products have antimicrobial claims. These products are handled by EPA’s Office of Pesticide Programs Antimicrobials Division. See https://www.epa.gov/pesticide-registration/antimicrobial-pesticide-registration (accessed July 27, 2017) (Communication, EPA, July 27, 2017).
use of environmental probiotic cleaning agents in the hospital environment (Caselli et al., 2016). Second, the presence of beneficial microbes in buildings may act as a source and modulator of the human microbiome, and the changes in the human microbiome thus introduced may result in the prevention or reduction of disease, although these connections remain to be fully explored (see Chapter 2).5 The concept of environmental probiotics has gained consumer market attention, and several products are available for residential use that emit microbes claimed (by the vendor) to be beneficial. Yet, despite these promising hypotheses, the efficacy and potential drawbacks of any specific built environment probiotic have not been rigorously investigated in the peer-reviewed literature.6 Below, two of the most compelling links between microbial exposures in buildings and protection against disease are discussed. Early work in this area suggests there may be benefits to designing, operating, and maintaining buildings to encourage exposure to microbiota from other humans, animals, plants, and biodiverse natural environments.
Indirectly Adding Microbes to Buildings: Animals in or Near Homes
A significant body of scientific literature supports the hypothesis that some microbial exposures are protective against the development of asthma (von Mutius, 2016; see also Chapter 2). In nonfarm environments, opportunities for beneficial microbial exposures may come through dog ownership. Evidence from cross-sectional epidemiology studies demonstrates that ownership of pets (especially dogs) is associated with reduced risk of allergic sensitization (Ownby et al., 2002). Fujimura and colleagues (2014) exposed mice to dust collected from homes with and without dogs. They found that exposure to dog-associated house dust resulted in a gut microbiome that was enriched in Lactobacillus, which in turn protected the mice against airway allergen challenge and virus infection. The presence of dogs is known to exert a strong influence on the microbiome of homes and to result in increased bacterial and fungal diversity (Dannemiller et al., 2016b; Dunn et al., 2013).
___________________
5 As discussed in Chapter 2, many open questions remain. For example, accumulating evidence suggests that perinatal exposures to microorganisms are important in establishing the human microbiome in early life, but the effects of microbial exposures in adulthood are much less well understood.
6 Environmental probiotics would require registration under FIFRA if claims of pesticidal effect were made; however, if there were no claims of pesticidal effect, the product might not need to be registered. Probiotic pesticide products would be handled by EPA’s Office of Pesticide Programs Biopesticide and Pollution Prevention Division (Communication, EPA, July 27, 2017).
Increasing Outdoor Biodiversity Around Occupied Buildings
It has been estimated that 66 percent of humanity will live in cities by 2050 (UN, 2015), and current trends of increasing urban density are resulting in the loss and increased fragmentation of green spaces. Evidence suggests that human well-being in urban areas is linked to neighborhood greenness, and recent research, discussed below, suggests that this link may be driven in part by indoor exposure to the diverse microbial communities associated with plants. Questions remain, however, as to how outdoor landscape features and the presence of indoor plants influence indoor microbiome quality.
The increase in some illnesses in higher-income, urbanized societies may be associated with a trend of failing immunoregulation and poorly regulated inflammatory responses in humans. It has been hypothesized that these immune system failures are due to a lack of exposure to organisms (also sometimes called “old friends”) from humankind’s evolutionary past that needed to be tolerated and therefore evolved roles in driving immunoregulatory mechanisms (Rook, 2013).
Through modern living and reduced exposure to outdoor spaces, plants, and animals, humans may lose contact with the commensal microorganisms transmitted by their mother, other people, animals, and the environment (Rook et al., 2014). Hanski and colleagues (2012) explored the connections among land use, the human microbiome, and allergy risk. They used DNA sequencing technology to compare the skin microbiome and allergic disposition of adolescents living with more or less forest and agricultural land within a 3 km radius from their homes. The authors observed that in healthy individuals, greater green (vegetative) space around the home was associated with higher concentrations of skin Proteobacteria. They also found that healthy individuals had a greater diversity of Gammaproteobacteria on their skin relative to individuals with allergic sensitization. Finally, they showed that individuals’ greater amounts of bacteria from the genus Acinetobacter (which belongs to the class Gammaproteobacteria) on the skin produced greater amounts of interleukin-10 (an anti-inflammatory cytokine that is known to increase immune tolerance). Together, these findings suggest that land use and the environmental context surrounding a building influence the human microbiome and health (see Ruokolainen et al., 2015, for further analysis of this hypothesis and dataset). Hanski (2014) interprets his research in a way that builds on the “old friends” concept, as framed by the “biodiversity hypothesis.” According to that hypothesis, reduced contact of people with nature (and in particular with plants and their associated microbial communities) may adversely affect the human microbiome and immune function. (See Stamper et al., 2016, for a recent review of the topic.)
Potential interventions to increase indoor exposure to vegetation have been suggested—for example, the use of plant “biowalls” in build-
ings, which harbor microbial communities and have been claimed to play potential roles in filtering volatile organic gases. Such walls have aesthetic value, and investigations continue into their roles in affecting air quality (Darlington et al., 2000; Russell et al., 2014). However, the health effects of human exposure to vegetation-associated microorganisms in such walls remain unknown, and the walls also require careful design to manage the moisture they introduce into the indoor environment.
A FRAMEWORK FOR ASSESSING BUILT ENVIRONMENT INTERVENTIONS
Approaches to dealing with the myriad factors involved in the control of microbial exposures in buildings include tools for design and assessment and the development of new building technologies. There is growing evidence of benefits from interventions that reduce exposures to hazardous microbes or encourage exposures to certain beneficial ones. However, real or proposed interventions may conflict with economic, indoor air quality, energy, and other human health–related building goals or constraints. Recognized and potential negative health, energy, and economic trade-offs associated with ventilation- and cleaning-based interventions are presented below, along with holistic strategies for designing interventions that can reduce or eliminate these trade-offs.
Material Balance Modeling
Central to assessment of built environment interventions is understanding the fate and transport of and human exposure to microbes in indoor air. All buildings are unique, but the dynamics of microbes and microbial communities within the built environment are controlled by a narrower regime of mostly physical processes (reviewed by Nazaroff [2016]). With the general exception of moisture damage, which allows for bacterial and fungal growth on building materials and in floor dust (Dannemiller et al., 2016a; Mudarri and Fisk, 2007; Spengler et al., 1994), microbes in air and on surfaces of buildings likely contain relatively low levels of metabolic activity, if at all. Thus, the dynamics and assemblages of microbial communities are largely a response to physical rather than chemical gradients. These physical processes link air with surfaces and sources, and an understanding of these processes is essential for making quantitative assessments to track the effectiveness of interventions and the liabilities of the trade-offs involved. Models of these processes often result in the ability to predict indoor concentrations and human exposures. If dose-response information is available, microbial risk analysis can be added to reflect health as an endpoint more directly (Fabian et al., 2014).
While not commonly applied to microbes in buildings, dispersion modeling approaches have significant potential for use as an assessment tool. The basis of a dispersion model is a mass (or material) balance. The role of a mass balance in understanding systems was introduced in Chapter 1 and represented in Equation 5.1. It is reintroduced here as an aid to modeling microbial concentrations and movements in a built environment and providing a priori exposure estimates for impacts of different interventions. Figure 5-1 depicts a number of physical variables that are important to incorporate into dispersion models. This simplified aerosol balance at steady state shows that the amount of biomass entering from outside through ventilation plus the amount entering from outside through infiltration and the contributions from indoor “generation” sources (growth, resuspension, and shedding) is equivalent to the amount of indoor biomass that is flowing or leaking out of the building plus other indoor “losses” (deposition, decay, and removal by filtration).
Dispersion models are commonly applied to assess indoor air concentrations and exposures for particles or chemicals. Computer programs for simulating air movement and transport of airborne particle and chemical contaminants in buildings have existed since the 1980s, and they continue to be developed and applied to a range of building performance issues (e.g., CONTAM [Dols and Polidoro, 2015; see also Chapter 4]). These programs fall into two broad categories—micro and macro. Micro models can be used to predict detailed airflow patterns and airborne contaminant concentrations in a building space at scales of centimeters or less using computational fluid dynamics. Macro models simulate whole buildings, typically representing each space (e.g., room, hallway, vertical shaft) as a single node at a uniform concentration and a single pressure. Both types of models can be used to model many processes relevant to microbes, including filtration and deposition on surfaces, and they could be modified to include a broader suite of processes relevant to microbial exposure.
The application of models specific to microbes is limited largely by the lack of microbial information with which to run dispersion models for assessing exposure, limited dose-response information for microbial agents, and poor understanding of the variety of responses to microbes within a human population. Because particle size drives many of the important physical processes detailed in Figure 5-1, more information on the size distributions of indoor air microbes of importance is necessary. These limitations are most extreme for potentially beneficial microbes, as large uncertainty exists with respect to the beneficial agents, the necessary doses, and the relevant sectors of the human population.
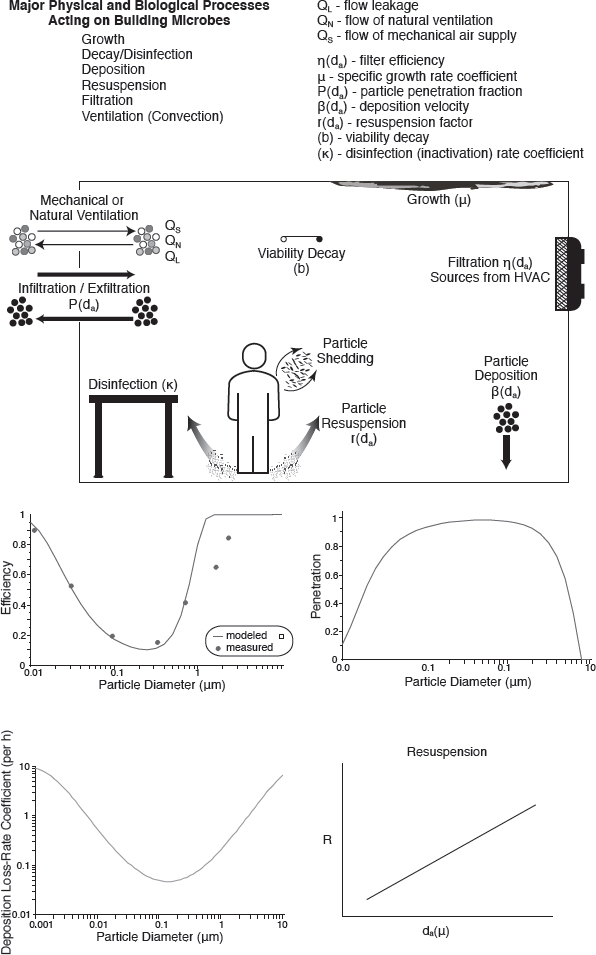
SOURCES: Filtration efficiency, penetration efficiency, and deposition plots modified from Nazaroff (2004); data for resuspension rates and house figure from Thatcher and Layton (1995).
Balancing Ventilation and Energy Usage
Through dilution and humidity control, increased outdoor air ventilation with filtration is a common approach for removing hazardous microbes, allergens, and toxins from indoor air. A potential negative tradeoff for this increased flow of outdoor air is an increase in energy consumption. When outdoor and indoor temperature and humidity conditions differ, energy in proportion to the rate at which outdoor air enters a building is required to raise or lower the temperature and adjust the water content necessary to condition the air (Persily, 2016), in accordance with
![]()
where qs (energy/time) is the energy consumption associated with changing the outdoor ventilation air temperature and water content to the desired indoor conditions. The first term on the right-hand side of this equation is the sensible energy consumption, where Q (volume/time) is the rate at which outdoor air enters the building, ρ (mass/volume) is the outdoor air density, Cp (energy/mass per temperature difference) is the specific heat of air, and ΔT is the indoor–outdoor temperature difference. The second term on the right-hand side is the latent energy consumption, where hw (energy/mass) is the latent heat of vaporization of water, and ΔW is the indoor–outdoor humidity ratio difference (ASHRAE, 2013). In addition, fan energy is required to provide ventilation air in mechanically ventilated buildings and to overcome pressure drops across filters. In mechanically ventilated buildings, the relationship between ventilation and energy consumption is complex and depends on the type of HVAC system and the associated control and operating strategies, and it can be evaluated only through detailed simulation or measurement efforts. In U.S. Mid-Atlantic states, for example, elevated energy consumption for ventilation is associated with heating requirements in the winter and dehumidification and cooling in the summer, while spring and fall operations can make use of economizers to introduce outdoor air that requires less conditioning during these seasons. Depending on outdoor climate and conditions and the use of strategies that include economizers, increased building envelope tightness, heat recovery ventilation, and demand-controlled ventilation, increasing outdoor ventilation rates to a level that improves indoor air quality in general and reduces exposure to indoor-generated microbial contaminants does not necessarily come at significantly increased energy or capital costs (Persily and Emmerich, 2012). The economic and energy costs of ventilation and the balance between these costs and benefits have not been rigorously studied.
Balancing Ventilation and Outdoor Air Quality
Growing evidence from the biodiversity hypothesis indicates a potential health benefit of human exposure to a diverse cohort of microorganisms in outdoor air. Such exposures may be facilitated through increased unfiltered outdoor air ventilation. An unintentional consequence of the increased use of mechanical ventilation with particle filtration is a shift in the indoor air microbiome away from the outdoor microbiome (Kembel et al., 2012; Meadow et al., 2014). Some limitations may be inherent in ventilation as a result of its nonselective nature. Depending on the locations and sensitivities of occupants, outdoor air may introduce into occupied spaces ambient or localized air quality hazards in both particulate matter and gas phase, as well as allergenic fungi and pollen. In areas with poor outdoor air quality, increased ventilation introduces biologic particles (associated with crustal materials), chemicals, and particulate matter into the built environment. Filtration of incoming outdoor air may reduce the influence of hazardous outdoor particles, but it will not remove gases and may also eliminate potentially beneficial outdoor microbes associated with an outdoor biodiverse environment. Enhanced filtration of outdoor air, especially to reduce PM2.5 exposure, requires the use of filters with higher MERV ratings and can also incur significant capital and operational costs. However, lower-pressure-drop, high-efficiency filtration technology is being developed that reduces the associated operating costs. Recognizing the inherent link between outdoor and indoor air, further efforts to produce good indoor air quality will be influenced by efforts and legislation aimed at improving outdoor ambient air quality.
Balancing Microbial Removal with Exposure to UV Radiation and Cleaning Products
As noted earlier, several trade-offs are associated with efforts to kill or remove hazardous microbes in the built environment. A primary concern has been exposure to cleaning chemicals, which are common sources of VOCs in indoor air. Both so-called green and conventional cleaning products emit primary chemical aerosols and may also result in secondary aerosol formation (Nazaroff and Weschler, 2004). A second concern is the presence of antibiotics or antimicrobial chemicals in many consumer cleaning products and the risk of producing resistant bacterial strains (Aiello and Larson, 2003). Moreover, as noted previously in the discussion of UV germicidal air and surface treatments, these technologies present radiation exposure or ozone generation hazards, respectively, for occupants, and systems must be designed and operated to manage these risks.
An emerging concern with respect to cleaning and disinfection practices is the unintentional removal of beneficial bacteria and other microbes. The tools used for these purposes are imprecise and provide no opportunity to remove pathogens selectively without also removing microbial “old friends” that may be present or reducing diversity (Rook et al., 2014).
Expanding Models to Include Health Outcomes, Economics, and Energy
The output of the dispersion models described earlier in this chapter includes indoor air concentrations and human exposures. However, approaches to designing sustainable built environment interventions will not be derived through standard engineering practices focused on the optimization of a single variable. To truly capture the complex nature of interventions and associated trade-offs, these models for microbes need to be expanded to consider health, energy, and economics. The strength of this broader approach is the potential to link building characteristics quantitatively to health outcomes and compare costs of health care and interventions. Recently, for example, indoor airflow and contaminant dispersion models (CONTAM) have been coupled with energy evaluation models (EnergyPlus) to capture interdependencies between airflow and heat transfer and thereby directly link indoor air quality and energy analyses (Dols et al., 2016).
Important considerations include the socioeconomic status of home/ building occupants and unique environments. Buildings are commonly private, and there are no regulations concerning biological exposures to drive research and practice. While prevention of infection from drinking water and food is regulated and expected by the general public, few expectations are focused on transmission of infectious diseases or exacerbation of allergenic disease in buildings. The enthusiasm and organization of efforts to develop rational interventions are thereby limited. In addition, residents who rent or are of low socioeconomic status will have a limited ability to pay for interventions or innovative technologies. Such environments as schools, health care facilities, and heavily water-damaged buildings may require specialized approaches and trade-offs, which likely will be different for different building types. For example, ventilation with efficient filtration in hospitals is necessary to reduce the incidence of hospital-associated infection. In hospitals, but perhaps not homes with young children, these needs may override concerns about encouraging biodiverse microbial exposures.
SUMMARY OBSERVATIONS AND KNOWLEDGE GAPS
Summary Observations
A number of potential physical and chemical interventions that can significantly affect indoor environmental quality can be undertaken in the built environment. The discussion in this chapter has focused particularly on examples of interventions that affect indoor air.
Buildings with tight envelopes and well-designed mechanical or engineered natural ventilation provide more potential to control and modulate microbial exposures relative to buildings ventilated by unintentional air leakage. The design of effective ventilation-based interventions will require a greater understanding of the potential (and limitations) of these ventilation schemes in modulating indoor microbial exposures and risks.
Understanding and improving the environment outside of buildings should be considered part of intervention design. The onus for improved indoor environmental quality cannot reside solely with building operations, particularly because air and water from outside sources are important inputs to indoor environments. Instead of reliance on the building envelope to control all exposures, researchers will need to consider the broader perspectives for meeting the diverse goals of improving microbial indoor air quality, recognizing which goals cannot be achieved absent good outdoor air quality.
Critical guidance is lacking on when to initiate interventions for damp buildings and on how to gauge the success of these interventions. Built environment interventions are most commonly proposed or undertaken with a goal of reducing exposure to microorganisms that may have negative health effects. As discussed in prior chapters, the presence of dampness, water damage, and visible mold in buildings is associated with negative respiratory health effects, and as a result, remediation aimed at drying and removing building materials affected by these conditions from indoor environments is often considered. Significant questions remain about what constitutes normal microbial ecology in different building types and under different conditions. This information will be needed to delineate impacted versus normal microbial ecologies to enable understanding and assessing the impact of interventions.
Interventions for promoting human exposure to beneficial microbes are in a nascent stage. Research continues to explore whether microbes with beneficial effects can be identified; how or whether exposures to these microorganisms in the built environment are associated with various types of health impacts; and the building conditions that promote, hinder, or alter these exposures.
Quantitative frameworks provide valuable insights to support the design of interventions and to understand the trade-offs among potentially competing priorities. Such frameworks can be used to better understand the anticipated effects of interventions and to aid in the design of intervention approaches. To design and deploy interventions that promote human and environmental health, such frameworks as building airflow and contaminant transport models, risk analyses, and building energy models will need to be linked systematically and holistically to infrastructure design and occupant health data.
Knowledge Gaps
On the basis of the above summary observations and the information developed in this chapter on interventions in the built environment, the committee identified the following goals for research to address knowledge gaps and advance the field:
- Improve understanding of “normal” microbial ecology in buildings of different types and under different conditions. This information provides important input into assessment of and decision making about potential interventions. In the case of damp building remediation to alter mold exposures, for example, if remediation goals are to fit within a “normal fungal ecology” standard, research will be required to define the normal fungal (and other microbial) ecologies for buildings within specific geographic areas and climates. DNA sequencing and the associated bioinformatics may be well suited to assessing the natural microbial ecology of buildings.
- Further explore the concept of interventions that promote exposure to beneficial microorganisms, and whether and under what circumstances these might promote good health. Provided that microbes with beneficial health effects are identified, additional research will be needed both to determine whether these microbes, while in the built environment, can be transmitted to humans and impact the human microbiome and to design and test interventions that encourage and control exposure to these microbes in buildings.
- Obtain additional data necessary to support the use of a variety of quantitative frameworks for understanding and assessing built environment interventions. A variety of information will need to be incorporated into models that link building and microbial information systematically and holistically to additional design, energy, environmental, and health data. To apply dispersion and risk models effectively for microbes, more research will be required to understand the size distributions of health-relevant microbiota; more de-
tailed information will be needed on the emission rates of microbes in buildings; and clear dose-response information will be needed for health-relevant microorganisms. To understand the economic implications of interventions, more research will be required to determine such parameters as health care costs of microbe-associated disease (or prevention of disease via beneficial microbes) and the energy, greenhouse gas, and other implications of interventions, such as the energy trade-offs associated with increasing outdoor air ventilation to control infectious disease.
REFERENCES
Adams, R. I., M. Miletto, J. W. Taylor, and T. D. Bruns. 2013. Dispersal in microbes: Fungi in indoor air are dominated by outdoor air and show dispersal limitation at short distances. The ISME Journal 7(7):1751-7362.
Aiello, A. E., and E. Larson. 2003. Antibacterial cleaning and hygiene products as an emerging risk factor for antibiotic resistance in the community. The Lancet Infectious Diseases 3(8):501-506.
AIHA (American Industrial Hygiene Association). 2013. Position statement on mold and dampness in the built environment. https://www.aiha.org/government-affairs/PositionStatements/P-Mold-03-26-13.pdf (accessed July 18, 2017).
Amend, A. S., K. A. Seifert, R. Samson, and T. D. Bruns. 2010. Indoor fungal composition is geographically patterned and more diverse in temperate zones than in the tropics. Proceedings of the National Academy of Sciences of the United States of America 107(31):13748-13753.
Anderson, D. J., L. F. Chen, D. J. Weber, R. W. Moehring, S. S. Lewis, P. F. Triplett, M. Blocker, P. Becherer, J. C. Schwab, L. P. Knelson, Y. Lokhnygina, W. A. Rutala, H. Kanamori, M. F. Gergen, and D. J. Sexton. 2017. Enhanced terminal room disinfection and acquisition and infection caused by multidrug-resistant organisms and Clostridium difficile (the Benefits of Enhanced Terminal Room Disinfection study): A cluster-randomised, multicentre, crossover study. The Lancet 389(10071):805-814.
ANSI/IICRC (American National Standards Institute/Institute of Inspection, Cleaning and Restoration Certification). 2015. S520: 2015 Reference guide for professional mold remediation. Las Vegas, NV: IICRC.
Arundel, A. V., E. M. Sterling, J. H. Biggin, and T. D. Sterling. 1986. Indirect health effects of relative humidity in indoor environments. Environmental Health Perspectives 65:351-361.
ASHRAE (American Society of Heating, Refrigerating and Air-Conditioning Engineers). 2010. ANSI/ASHRAE Standard 62.1-2010. Ventilation for acceptable indoor air quality. Atlanta, GA: ASHRAE.
ASHRAE. 2013. Fundamentals handbook. Atlanta, GA: ASHRAE.
ASHRAE. 2016. Ultraviolet air and surface treatment proceedings. In TC 2.9. https://tc0209.ashraetcs.org (accessed May 1, 2017).
ASHRAE. 2017. Standard 52.2: Method of Testing General Ventilation Air-Cleaning Devices for Removal Efficiency by Particle Size (ANSI Approved). Atlanta, GA: ASHRAE.
Azimi, P., D. Zhao, and B. Stephens. 2014. Estimates of HVAC filtration efficiency for fine and ultrafine particles of outdoor origin. Atmospheric Environment 98:337-346.
Bhangar, S., J. A. Huffman, and W. W. Nazaroff. 2014. Size-resolved fluorescent biological aerosol particle concentrations and occupant emissions in a university classroom. Indoor Air 24(6):604-617.
Bornehag, C. G., J. Sundell, L. Hägerhed-Engman, and T. Sigsgaard. 2005. Association between ventilation rates in 390 Swedish homes and allergic symptoms in children. Indoor Air 15(4):275-280.
Boyce, J. M. 2016. Modern technologies for improving cleaning and disinfection of environmental surfaces in hospitals. Antimicrobial Resistance & Infection Control 5(1):10.
Brickner, P. W., R. L. Vincent, M. First, E. Nardell, M. Murray, and W. Kaufman. 2003. The application of ultraviolet germicidal irradiation to control transmission of airborne disease: Bioterrorism countermeasure. Public Health Reports 118(2):99-114.
Caselli, E., M. D’Accolti, A. Vandini, L. Lanzoni, M. T. Camerada, M. Coccagna, A. Branchini, P. Antonioli, P. G. Balboni, D. Di Luca, and S. Mazzacane. 2016. Impact of a probiotic-based cleaning intervention on the microbiota ecosystem of the hospital surfaces: Focus on the resistome remodulation. PLOS ONE 11(2):e0148857.
Chen, X., and H. J. Schluesener. 2008. Nanosilver: A nanoproduct in medical application. Toxicology Letters 176(1):1-12.
CIBSE (Chartered Institution of Building Services Engineers). 2005. Natural ventilation in non-domestic buildings. CIBSE application manual AM10. London, UK: CIBSE.
Dannemiller, K. C., M. J. Mendell, J. M. Macher, K. Kumagai, A. Bradman, N. Holland, K. Harley, B. Eskenazi, and J. Peccia. 2014. Next-generation DNA sequencing reveals that low fungal diversity in house dust is associated with childhood asthma development. Indoor Air 24(3):236-247.
Dannemiller, K. C., C. J. Weschler, and J. Peccia. 2016a. Fungal and bacterial growth in floor dust at elevated relative humidity levels. Indoor Air 27(2):354-363.
Dannemiller, K. C., J. F. Gent, B. P. Leaderer, and J. Peccia. 2016b. Influence of housing characteristics on bacterial and fungal communities in homes of asthmatic children. Indoor Air 26(2):179-192.
Darlington A., M. Chan, D. Malloch, C. Pilger, and M. A. Dixon. 2000. The biofiltration of indoor air: Implications for air quality. Indoor Air 10(1):39-46.
Dols, W. S., and B. J. Polidoro. 2015. CONTAM user guide and program documentation. NIST Technical Note 1887. Gaithersburg, MD: National Institute of Standards and Technology.
Dols, W. S., S. J. Emmerich, and B. J. Polidoro. 2016. Coupling the multizone airflow and contaminant transport software CONTAM with EnergyPlus using co-simulation. Building Simulation 9(4):469-479.
Donskey, C. J. 2013. Does improving surface cleaning and disinfection reduce health care-associated infections? American Journal of Infection Control 41(5, Suppl.):S12-S19.
Dunn, R. R., N. Fierer, J. B. Henley, J. W. Leff, and H. L. Menninger. 2013. Home life: Factors structuring the bacterial diversity found within and between homes. PLOS ONE 8(5):e64133.
Ege, M. J., M. Mayer, A.-C. Normand, J. Genuneit, W. O. C. M. Cookson, C. Braun-Fahrländer, D. Heederik, R. Piarroux, and E. von Mutius. 2011. Exposure to environmental microorganisms and childhood asthma. New England Journal of Medicine 364(8):701-709.
EPA (U.S. Environmental Protection Agency). 2008. Critical assessment of building air cleaner technologies. EPA/600/R-08/053. Washington, DC: EPA.
Escombe, A. Roderick, D. A. J. Moore, R. H. Gilman, M. Navincopa, E. Ticona, B. Mitchell, C. Noakes, C. Martínez, P. Sheen, R. Ramirez, W. Quino, A. Gonzalez, J. S. Friedland, and C. A. Evans. 2009. Upper-room ultraviolet light and negative air ionization to prevent tuberculosis transmission. PLOS Medicine 6(3):e1000043.
Fabian, M. P., G. Adamkiewicz, N. K. Stout, M. Sandel, and J. I. Levy. 2014. A simulation model of building intervention impacts on indoor environmental quality, pediatric asthma, and costs. Journal of Allergy and Clinical Immunology 133(1):77-84.
Fisk, W. J. 2013. Health benefits of particle filtration. Indoor Air 23(5):357-368.
Fujimura, K. E., T. Demoor, M. Rauch, A. A. Faruqi, S. Jang, C. C. Johnson, H. A. Boushey, E. Zoratti, D. Ownby, N. W. Lukacs, and S. V. Lynch. 2014. House dust exposure mediates gut microbiome Lactobacillus enrichment and airway immune defense against allergens and virus infection. Proceedings of the National Academy of Sciences of the United States of America 111(2):805-810.
Gao, Y., and R. Cranston. 2008. Recent advances in antimicrobial treatments of textiles. Textile Research Journal 78(1):60-72.
Grant, C., C. A. Hunter, B. Flannigan, and A. F. Bravery. 1989. The moisture requirements of moulds isolated from domestic dwellings. International Biodeterioration 2(4):259-284.
Hanski, I. 2014. Biodiversity, microbes and human well-being. Ethics in Science and Environmental Politics 14(1):19-25.
Hanski, I., L. von Hertzen, N. Fyhrquist, K. Koskinen, K. Torppa, T. Laatikainen, P. Karisola, P. Auvinen, L. Paulin, M. J. Mäkelä, E. Vartiainen, T. U. Kosunen, H. Alenius, and T. Haahtela. 2012. Environmental biodiversity, human microbiota, and allergy are interrelated. Proceedings of the National Academy of Sciences of the United States of America 109(21):8334-8339.
Hasan, J., R. J. Crawford, and E. P. Ivanova. 2013. Antibacterial surfaces: The quest for a new generation of biomaterials. Trends in Biotechnology 31(5):295-304.
Hospodsky, D., H. Yamamoto, W. W. Nazaroff, and J. Peccia. 2014 (unpublished). Size-resolved, baseline characterization data for indoor emission rates of bacteria, fungi, and particulate matter.
Hospodsky, D., N. Yamamoto, W. W. Nazaroff, D. Miller, S. Gorthala, and J. Peccia. 2015. Characterizing airborne fungal and bacterial concentrations and emission rates in six occupied children’s classrooms. Indoor Air 2(6):641-652.
Hubbard, H., D. Poppendieck, and R. L. Corsi. 2009. Chlorine dioxide reactions with indoor materials during building disinfection: Surface uptake. Environmental Science & Technology 43(5):1329-1335.
IOM (Institute of Medicine). 2004. Damp indoor spaces and health. Washington, DC: The National Academies Press.
Kembel, S. W., E. Jones, J. Kline, D. Northcutt, J. Stenson, A. M. Womack, B. J. M. Bohannan, G. Z. Brown, and J. L Green. 2012. Architectural design influences the diversity and structure of the build environment. The ISME Journal 6(8):1469-1479.
Kembel, S. W., J. F. Meadow, T. K. O’Connor, G. Mhuireach, D. Northcutt, J. Kline, M. Moriyama, G. Z. Brown, B. J. M. Bohannan, and J. L. Green. 2014. Architectural design drives the biogeography of indoor bacterial communities. PLOS ONE 9(1):e87093.
Kercsmar, C. M., D. G. Dearborn, M. Schluchter, L. Xue, H. L. Kirchner, J. Sobolewski, S. J. Greenberg, S. J. Vesper, and T. Allan. 2006. Reduction in asthma morbidity in children as a result of home remediation aimed at moisture sources. Environmental Health Perspectives 114(10):1574-1580.
Kovesi, T., C. Zaloum, C. Stocco, D. Fugler, R. E. Dales, A. Ni, N. Barrowman, N. L. Gilbert, and J. D. Miller. 2009. Heat recovery ventilators prevent respiratory disorders in Inuit children. Indoor Air 19(6):489-499.
Levin, J., L. S. Riley, C. Parrish, D. English, and S. Ahn. 2013. The effect of portable pulsed xenon ultraviolet light after terminal cleaning on hospital-associated Clostridium difficile infection in a community hospital. American Journal of Infection Control 41(8):746-748.
Lindsley, W. G., F. M. Blachere, R. E. Thewlis, A. Vishnu, K. A. Davis, G. Cao, J. E. Palmer, K. E. Clark, M. A. Fisher, R. Khakoo, and D. H. Beezhold. 2010. Measurements of airborne influenza virus in aerosol particles from human coughs. PLOS ONE 5(11):e15100.
Luongo, J. C., J. Brownstein, and S. L. Miller. 2017. Ultraviolet germicidal coil cleaning: Impact on heat transfer effectiveness and static pressure drop. Building and Environment 112:159-165.
Lynch, S. V., R. A. Wood, H. Boushey, L. B. Bacharier, G. R. Bloomberg, M. Kattan, G. T. O’Connor, M. T. Sandel, A. Calatroni, E. Matsui, C. C. Johnson, H. Lynn, C. M. Visness, K. F. Jaffee, P. J. Gergen, D. R. Gold, R. J. Wright, K. Fujimura, M. Rauch, W. W. Busse, and J. E. Gern. 2014. Effects of early-life exposure to allergens and bacteria on recurrent wheeze and atopy in urban children. Journal of Allergy and Clinical Immunology 134(3):593-601. doi:10.1016/j.jaci.2014.04.018.
McLean, R. L. 1961. The mechanism of spread of Asian influenza: General discussion. American Review of Respiratory Disease 83(2P2):36-38.
Meadow, J. F., A. E. Altrichter, S. W. Kembel, J. Kline, G. Mhuireach, M. Moriyama, D. Northcutt, T. K. O’Connor, A. M. Womack, G. Z. Brown, J. L Green, and B. J. M. Bohannan. 2014. Indoor airborne bacterial communities are influenced by ventilation, occupancy, and outdoor air source. Indoor Air 24(1):41-48.
Mendell, M. J., A. G. Mirer, K. Cheung, M. Tong, and J. Douwes. 2011. Respiratory and allergic health effects of dampness, mold, and dampness-related agents: A review of the epidemiologic evidence. Environmental Health Perspectives 119(6):748-756.
Menzies, D., and J. Bourbeau. 1997. Building-related illnesses. New England Journal of Medicine 337(21):1524-1531.
Menzies, D., J. Popa, J. A. Hanley, T. Rand, and D. K. Milton. 2003. Effect of ultraviolet germicidal lights installed in office ventilation systems on workers’ health and wellbeing: Double-blind multiple crossover trial. The Lancet 362(9398):1785-1791.
Miller, R., S. Simmons, C. Dale, J. Stachowiak, and M. Stibich. 2015. Utilization and impact of a pulsed-xenon ultraviolet room disinfection system and multidisciplinary care team on Clostridium difficile in a long-term acute care facility. American Journal of Infection Control 43(12):1350-1353.
Mudarri, D., and W. J. Fisk. 2007. Public health and economic impact of dampness and mold. Indoor Air 17(3):226-235.
Nagaraja, A., P. Visintainer, J. P. Haas, and J. Menz, G. P. Wormser, and M. A. Montecalvo. 2015. Clostridium difficile infections before and during use of ultraviolet disinfection. American Journal of Infection Control 43(9):940-945.
Nazaroff, W. W. 2004. Indoor particle dynamics. Indoor Air 14(S7):175-183.
Nazaroff, W. W. 2016. Indoor bioaerosol dynamics. Indoor Air 26(1):61-78.
Nazaroff, W. W., and C. J. Weschler. 2004. Cleaning products and air fresheners: Exposure to primary and secondary air pollutants. Atmospheric Environment 38(18):2841-2865.
Ownby, D. R., C. Johnson, and E. L. Peterson. 2002. Exposure to dogs and cats in the first year of life and risk of allergic sensitization at 6 to 7 years of age. Journal of the American Medical Association 288(8):963-972.
Pastuszka, J. S., U. Kyaw Tha Paw, D. O. Lis, A. Wlazło, and K. Ulfig. 2000. Bacterial and fungal aerosol in indoor environment in Upper Silesia, Poland. Atmospheric Environment 34(22):3833-3842.
Peccia, J., H. M. Werth, S. Miller, and M. Hernandez. 2001. Effects of relative humidity on the ultraviolet induced inactivation of airborne bacteria. Aerosol Science and Technology 35(3):728-740.
Persily, A. K. 2016. Field measurement of ventilation rates. Indoor Air 26(1):97-111.
Persily, A. K., and S. J. Emmerich. 2012. Indoor air quality in sustainable, energy efficient buildings. HVAC&R Research 18(1-2):4-20.
Poppendieck, D. G., H. F. Hubbard, C. J. Weschler, and R. L. Corsi. 2007. Formation and emissions of carbonyls during and following gas-phase ozonation of indoor materials. Atmospheric Environment 41(35):7614-7626.
Prussin, A. J., and L. C. Marr. 2015. Sources of airborne microorganisms in the built environment. Microbiome 3(1):78.
Qian, J., D. Hospodsky, N. Yamamoto, W. W. Nazaroff, and J. Peccia. 2012. Size-resolved emission rates of airborne bacteria and fungi in an occupied classroom. Indoor Air 22(4):339-351.
Reed, N. G. 2010. The history of ultraviolet germicidal irradiation for air disinfection. Public Health Reports 125(1):15-27.
Reponen, T., S. Vesper, L. Levin, E. Johansson, P. Ryan, J. Burkle, S. A. Grinshpun, S. Zheng, D. I. Bernstein, J. Lockey, M. Villareal, G. K. Hershey, and G. LeMasters. 2011. High environmental relative moldiness index during infancy as a predictor of asthma at 7 years of age. Annals of Allergy, Asthma & Immunology 107(2):120-126.
Roberts, J. W., W. S. Clifford, G. Glass, and P. G. Hummer. 1999. Reducing dust, lead, dust mites, bacteria, and fungi in carpets by vacuuming. Archives of Environmental Contamination and Toxicology 36(4):477-484.
Rook, G. A. W. 2013. Regulation of the immune system by biodiversity from the natural environment: An ecosystem service essential to health. Proceedings of the National Academy of Sciences of the United States of America 110(46):18360-18367.
Rook, G. A. W., C. L. Raison, and C. A. Lowry. 2014. Microbial “old friends,” immunoregulation and socioeconomic status. Clinical & Experimental Immunology 177(1):1-12.
Ruokolainen, L., L. von Hertzen, N. Fyhrquist, T. Laatikainen, J. Lehtomäki, P. Auvinen, A. M. Karvonen, A. Hyvärinen, V. Tillmann, O. Niemelä, M. Knip, T. Haahtela, J. Pekkanen, and I. Hanski. 2015. Green areas around homes reduce atopic sensitization in children. Allergy 70(2):195-202.
Russell, J. A., Y. Hu, L. Chau, M. Pauliushchyk, I. Anastopoulos, S. Anandan, and M. S. Waring. 2014. Indoor-biofilter growth and exposure to airborne chemicals drive similar changes in plant root bacterial communities. Applied and Environmental Microbiology 80(16):4805-4813.
Schulze, T., and U. Eicker. 2013. Controlled natural ventilation for energy efficient buildings. Energy and Buildings 56:221-232.
Seppänen, O., and W. J. Fisk. 2002. Association of ventilation system type with SBS symptoms in office workers. Indoor Air 12(2):98-112.
Sliney, D. 2013. Balancing the risk of eye irritation from UV-C with infection from bioaerosols. Photochemistry and Photobiology 89(4):770-776.
Spengler, J., L. Neas, S. Nakai, D. Dockery, F. Speizer, J. Ware, and M. Raizenne. 1994. Respiratory symptoms and housing characteristics. Indoor Air 4(2):72-82.
Stamper, C. E., A. J. Hoisington, O. M. Gomez, A. L. Halweg-Edwards, D. G. Smith, K. L. Bates, K. A. Kinney, T. T. Postolache, L. A. Brenner, G. A. W. Rook, and C. A. Lowry. 2016. Chapter 14. The microbiome of the built environment and human behavior: Implications for emotional health and well-being in postmodern western societies. In International review of neurobiology, edited by J. F. Cryan and G. Clarke. Cambridge, MA: Academic Press. Pp. 289-323.
Stephens, B., and J. A. Siegel. 2012. Penetration of ambient submicron particles into single-family residences and associations with building characteristics. Indoor Air 22(6):501-513.
Sundell, J., M. Wickman, G. Pershagen, and S. L. Nordvall. 1995. Ventilation in homes infested by house-dust mites. Allergy 50(2):106-112.
Sundell, J., H. Levin, W. W. Nazaroff, W. S. Cain, W. J. Fisk, D. T. Grimsrud, F. Gyntelberg, Y. Li, A. K. Persily, A. C. Pickering, J. M. Samet, J. D. Spengler, S. T. Taylor, and C. J. Weschler. 2011. Ventilation rates and health: Multidisciplinary review of the scientific literature. Indoor Air 21(3):191-204.
Thatcher, T. L., and D. W. Layton. 1995. Deposition, resuspension, and penetration of particles within a residence. Atmospheric Environment 29(13):1487-1497.
UN (United Nations). 2015. World urbanization prospects: The 2014 revision. ST/ESA/ SER.A/366. New York: UN, Department of Economic and Social Affairs, Population Division. https://esa.un.org/unpd/wup/Publications/Files/WUP2014-Report.pdf (accessed July 20, 2017).
von Mutius, E. 2016. The microbial environment and its influence on asthma prevention in early life. Journal of Allergy and Clinical Immunology 137(3):680-689.
Wang, H., M. A. Edwards, J. O. Falkinham, and A. Pruden. 2013. Probiotic approach to pathogen control in premise plumbing systems? A review. Environmental Science & Technology 47(18):10117-10128.
Wang, Y.., C. Sekhar, W. P. Bahnfleth, K. W. Cheong, and J. Firrantello. 2016. Effectiveness of an ultraviolet germicidal irradiation system in enhancing cooling coil energy performance in a hot and humid climate. Energy and Buildings 130:321-329.
Weber, D. J., H. Kanamori, and W. A. Rutala. 2016. “No touch” technologies for environmental decontamination: Focus on ultraviolet devices and hydrogen peroxide systems. Current Opinion in Infectious Diseases 29(4):424-431.
Xu, P., J. Peccia, P. Fabian, J. W. Martyny, K. P. Fennelly, M. Hernandez, and S. L. Miller. 2003. Efficacy of ultraviolet germicidal irradiation of upper-room air in inactivating airborne bacterial spores and mycobacteria in full-scale studies. Atmospheric Environment 37(3):405-419.
Yamamoto, N., W. W. Nazaroff, and J. Peccia. 2014. Assessing the aerodynamic diameters of taxon-specific fungal bioaerosols by quantitative PCR and next-generation DNA sequencing. Journal of Aerosol Science 78:1-10.
Yang, W., S. Elankumaran, and L. C. Marr. 2011. Concentrations and size distributions of airborne influenza A viruses measured indoors at a health centre, a day-care centre and on aeroplanes. Journal of The Royal Society Interface 8(61):1176-1184.
Yang, W., S. Elankumaran, and L. C. Marr. 2012. Relationship between humidity and influenza A viability in droplets and implications for influenza’s seasonality. PLOS ONE 7(10):e46789.































