7
Role of Research Funders in Multimodal Therapy Development
As demonstrated in the previous chapter and mentioned by FDA commissioner Robert Califf and others, governmental and charitable funding agencies play an important role in filling the gaps that may exist with respect to industry investment in developing novel multimodal interventions. For example, Karl Kieburtz suggested that third party investments might help mitigate the risk of partnerships among competitors, and Erich Mohr commented that peer-reviewed funding adds essential credibil-
ity to risky projects, such as the GDNF−CED project discussed in the previous chapter.
Califf suggested another example of how a neutral third party can help facilitate progress: by providing funding for a study that industry partners have no commercial incentive to pursue. Califf described a head-to-head trial of two interventions—one a drug and the other a device—for congestive heart failure (Bardy et al., 2005). Neither of the manufacturers was motivated to do the trial, but the field was driven to make it happen, so NIH put up foundational money to help the industry participate. The answer was clear when the trial was completed: The device was superior. This led not only to an update of the label by the FDA, but also the reimbursement policy of CMS, said Califf.
Partnerships among potential competitors may be easier to achieve when the partners have overlapping goals or synergistic components to offer, noted Christopher Shaffer. Brian Fiske agreed, noting that MJFF routinely brings competitors together in precompetitive space for a variety of purposes related to PD therapy development, including fundamental research to understand the biology of a target, and building infrastructure, including patient registries, to increase the efficiency of clinical trials. Another area where funding agencies—both public and private—may facilitate progress is by bringing industry partners together to contribute compounds for repurposing, which have passed through safety endpoints in Phase II, but failed for the intended indication, said Stevin Zorn.
ROLE OF RESEARCH AGENCIES IN DE-RISKING MULTIMODAL THERAPY DEVELOPMENT
The NIH Blueprint for Neuroscience Research is a cooperative effort across 15 NIH centers and institutes to support discovery and development of therapeutics to combat nervous system disorders and conditions through grants, contract resources, and expert consultants. The Blueprint Neurotherapeutics Network is specifically designed to help investigators translate knowledge into products that could benefit patients, according to Amir Tamiz. For example, the Blueprint network is among several funding streams that enable NINDS to focus funding at the early stages of drug discovery and development with strong scientific rational, rigorously designed studies, and transparent reporting in order to move projects to a stage where they can be handed off to industry and nonprofits,
who may have the resources to conduct clinical trials and bring products to commercialization.
Tamiz provided two examples of projects that NINDS is funding that combine therapies as a way of achieving greater effects. In the first, Karen Aboody, professor of developmental and stem cell biology at the City of Hope, is using neural stem cells (NSCs) as a delivery vehicle to target tumor cells in the brain with anticancer agents. She has engineered the NSCs to express an enzyme that converts the nontoxic prodrug 5-fluorocytosine to the active chemotherapeutic 5-fluorouracil (Aboody et al., 2013). Her preclinical work has led to approval of a first-in-human study of NSC-mediated enzyme/prodrug targeted cancer therapy in patients with recurrent glioblastoma.
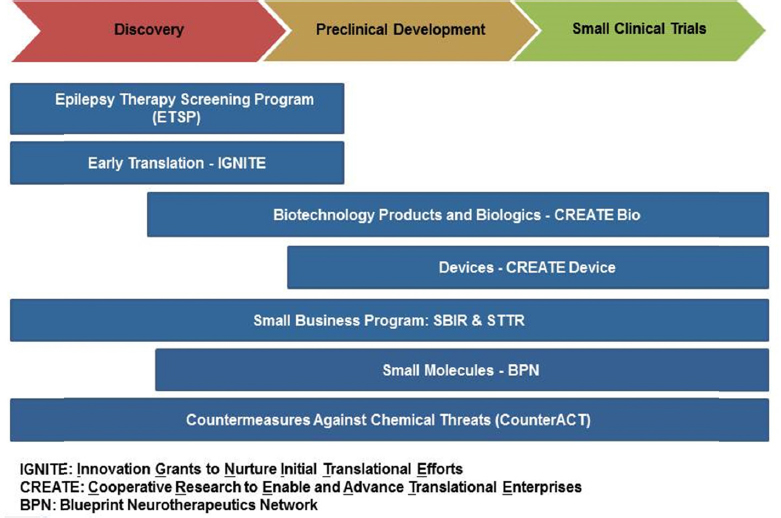
The second example combines a catheter-based intervention for brain aneurysm—one of two currently approved treatment options for aneurysms—with a bioactive compound that will stabilize the clot to reduce the rate of recurrence. Tamiz said this project benefitted from a Small Business Innovation Research grant. This is just one of several translational funding mechanisms available through NIH (see Figure 7-1). Of particular interest for early discovery efforts are the IGNITE (Innovation Grants to Nurture Initial Translational Efforts), CREATE (Cooperative Research to Enable and Advance Translational Enterprises), and Blueprint Neurotherapeutics (BPN) programs. CREATE is further split into two programs, one for therapeutic devices (CREATE Device) and the other for biotechnology products and biologics (CREATE Bio). Under the auspices of BPN, NIH has also established robust contracts within the United States to provide investigators with medicinal chemistry, pharmacokinetic and toxicology, data management, manufacturing and formulation, and clinical trials resources, added Tamiz.
Multimodal treatment research is also funded and conducted by other government agencies, such as the Department of Defense (DoD) and the Veterans Health Administration (VHA). Indeed, said Stuart Hoffman, senior scientific advisor for brain injury in the VHA Office of Research and Development, multimodal approaches are often necessary because a condition such as TBI has multiple consequences, including cognitive disability, psychological and behavioral disorders, sensory dysfunction, and pain; such a condition is typically accompanied by other visceral and orthopedic injuries. Since 2000, nearly 350,000 service members from the recent wars in Iraq and Afghanistan have been diagnosed with TBI (DVBIC, 2016). Most of these cases are mild, but in some people may result in persistent postconcussional symptoms often accompanied by
NOTE: SBIR = small business innovation research; STTR = small business technology transfer.
SOURCE: Presentation by Tamiz, June 15, 2016.
comorbidities, such as posttraumatic stress disorder, and chronic pain, said Hoffman (see Figure 2-2). In addition, he noted that recent studies have identified long-term chronic degeneration and inflammatory responses in the brains of service members and veterans exposed to blast. Sarah Lisanby commented that collaboration across disciplines, including neuropsychiatry, will be needed to study the full spectrum of manifestations of brain injury. Recognizing this, in 2012 President Obama issued an Executive Order calling for improved access to mental health services for veterans, service members, and military families and the establishment of a National Research Action Plan coordinated by DoD, VA, and Departments of Health and Human Services and Education.
Stuart Hoffman, senior scientific advisor for brain injury at the VA, co-chairs the steering committee for a large VA and DoD clinical study on the chronic effects of TBI. The study is made possible because of the integration of research and care within the VA health system, and access
to data housed in the Federal Interagency TBI Research informatics system (FITBIR). The VA also created the Million Veteran Program (MVP) in 2012 to advance genomic research and bring precision medicine to veterans. Now partnered with the federal government’s Precision Medicine Initiative, MVP already has nearly 500,000 veterans. Other partnerships, including with industry, are also planned through the VA Center for Innovation, said Hoffman.
ROLE OF DISEASE-SPECIFIC FUNDING IN MULTIMODAL THERAPY DEVELOPMENT
The Alzheimer’s Association and MJFF exemplify how disease-specific organizations can advance the development of multimodal therapies through targeted research funding. According to James Hendrix, the Alzheimer’s Association currently funds more than $80 million for 300 active projects in 20 countries worldwide. MJFF is the largest nonprofit funder of Parkinson’s research, last year providing more than $87 million globally to research programs, said Brian Fiske.
One of the multimodal intervention programs funded by the Alzheimer’s Association—the Finnish Geriatric Intervention Study to Prevent Cognitive Impairment and Disability (FINGER)—investigated whether a combination of lifestyle interventions, including exercise, diet, social engagement, and cognitive training, could slow cognitive decline among non-demented older individuals. Results from this 2-year randomized controlled trial study published in 2015 suggested that the multidomain intervention improved or maintained cognitive functioning in at-risk elderly (Ngandu et al., 2015). The Alzheimer’s Association followed this by publishing a summary of evidence collected in many prevention studies, concluding that strong evidence indicates that physical exercise and management of cardiovascular risk factors reduce the risk of cognitive decline and may reduce the risk of dementia, and that healthy diet and lifelong learning or cognitive training may also reduce the risk of cognitive decline. However, the study also noted many unanswered questions and called for more research in the area of prevention (Baumgart et al., 2015).
Other combinatorial approaches that target multiple symptoms are being investigated for the treatment of PD, said Fiske. This is essential, he said, because there is much more to PD than motor symptoms, including the possibility of cognitive impairment, autonomic dysfunction, sleep
dysregulation, and pain. Indeed, sleep disturbances are common to many neurological and psychiatric diseases, such as depression and TBI, said Lisanby and Hoffman.
Potentially more promising for both AD and PD would be combinations that target multiple disease mechanisms. For example, protein misfolding and dysfunction in how proteins are trafficked and cleared, neuroinflammation, mitochondrial dysfunction, oxidative stress, and other aging pathways have all been implicated in both diseases and may need to be targeted in combination. Fiske noted, however, that many questions remain about the role of these pathways in PD; for example, do they represent distinct forms of the disease with a similar clinical phenotype or are all these pathways converging on the same process? Should combinations target multiple mechanisms or multiple points along one mechanistic pathway? Kieburtz asked another question: Do these phenomena coexist with the disease or are they core features? Answers to these questions will be needed to build a rationale or argument for any type of combination therapy, said Fiske.
In addition to funding studies, another way disease-focused organizations can help advance the development of multimodal therapies is by convening experts to push things forward. For example, as described in Chapter 3, in 2015 the Alzheimer’s Association convened a workshop with experts from academia and industry to recommend a path forward for drug−drug combination therapies to treat AD (Hendrix et al., 2016). They followed this with a new grant mechanism, Alzheimer’s Combination Therapy Opportunities (ACTO), which will provide up to $2 million for a clinical trial testing repurposed drug combinations that target multiple biological mechanisms through Phase II proof of concept. Hendrix said another recent meeting of the Alzheimer’s Association’s Research Roundtable focused on neuropsychiatric symptoms associated with AD, in recognition that AD is more than a disease of cognition and that treatments are needed for these other disabling symptoms. Hendrix noted that one of the strengths of the Research Roundtable is that it brings companies together in precompetitive space to advance drug development.
Disease-focused foundations can also expedite drug development by building trial infrastructure such as platform and adaptive trials and registries that provide access to patients, said Fiske. He said MJFF supports cohort development that enables the collection of clinical and biological
data from large numbers of patients with PD and that can also be used to validate biomarkers1 and other outcome measures.
The Alzheimer’s Association and MJFF have also led the field in promoting data sharing among academic and industry researchers. For example, Hendrix said that a workgroup convened after a Research Roundtable on Amyloid-Related Imaging Abnormalities (ARIA)—a rare side effect of treatment with antiamyloid antibodies—convinced five or six companies to share their data. The workgroup subsequently issued recommendations for monitoring ARIA in clinical trials (Sperling et al., 2011). Meanwhile, MJFF has brought a number of companies together to address potential safety issues associated with inhibitors of the LRRK2 gene. They have been remarkably willing to contribute information and data, said Fiske. The key is to find the right common ground.
___________________
1 Additional information on biomarkers and their importance for therapeutic development for specific disorders can be found at: https://www.michaeljfox.org/research/priority-area-detail.php?biomarkers (accessed October 24, 2016); and http://www.alz.org/research/funding/global_biomarker_consortium.asp (accessed October 24, 2016).
This page intentionally left blank.








