2
Definitions
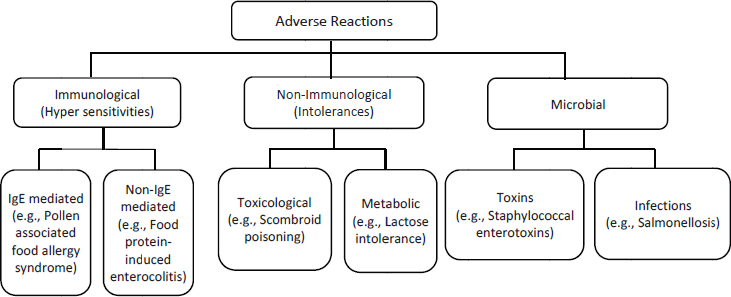
Collecting the evidence needed to develop effective diagnostic approaches, prevention strategies, therapies, and management procedures to prevent, manage, and treat food allergy requires that the physicians, biomedical and pharmaceutical scientists, policy makers, affected individuals and families, and all other stakeholders share a common understanding of what food allergy is and is not. In addition, although all proteins in foods have the potential to elicit a food allergy, some have been recognized as major allergens due to their potency in inducing a food allergy or in affecting the prevalence of allergy to those food constituents in the population. The list varies depending on the country but several ones are common globally. This introductory chapter begins by defining food allergy both by describing its signs and symptoms and by presenting our current understanding of how food allergy develops in affected individuals. The chapter also distinguishes food allergy from the many other adverse effects or conditions that could be related to foods but that have a nonimmunological origin. Considering the diversity of food adverse conditions with similar symptomatology and clinical manifestations (see Figure 2-1), it is no wonder that many misconceptions persist about food allergy. Even today, many questions are still unresolved. The most pressing research questions are detailed in Chapter 9.
FOOD ALLERGIES: DEFINITIONS
Commonly Accepted Definitions
Food allergy is “an adverse health effect arising from a specific immune response that occurs reproducibly on exposure to a given food,” according to the 2010 National Institute of Allergy and Infectious Diseases, National Institutes of Health (NIAID/NIH)-supported Guidelines for the Diagnosis and Management of Food Allergy in the United States (NIAID/NIH-supported Guidelines) (Boyce et al., 2010). “Exposure” in the food allergy context can be through ingestion, skin contact, or exposure to airborne particles. The immunologic component is central to the discussion of food allergy, including the underlying mechanisms of allergic reactions and methods of diagnosis and treatment. Adverse reactions to foods or food components that lack an identified immunologic pathophysiology are not considered food allergy, but instead are typically called food intolerances (Boyce et al., 2010). These reactions are not a focus of this report. The mechanisms behind these other conditions may include metabolic, pharmacologic, or toxic factors.
A food is defined as “any substance—whether processed, semiprocessed, or raw—that is intended for human consumption, and includes drinks, chewing gum, food additives, and dietary supplements” (Boyce et al., 2010). Food allergens are the components within foods that trigger immunologic reactions. These are most often specific glycoproteins, which can interact with the body’s immune cells in a way that initiates the development of a food allergy.
The broad terms allergy and allergic disease refer to a disease caused by immunologic dysfunction that fall under one of two key classifications: immunoglobulin E (IgE)-mediated or non-IgE-mediated (see Tables 2-1 and 2-2). This report focuses almost exclusively on IgE-mediated food allergy, which has better defined underlying cellular mechanisms and an established link to many prevalent food allergy reactions. Non-IgE-mediated food allergy reactions (e.g., food protein–induced enterocolitis) are less common and the mechanisms of the reactions are less well characterized. Celiac disease is a well-characterized, immune-mediated disease that has food as an exacerbating factor but will not be detailed in this report.
Immunoglobulin E is an antibody that, if bound to certain cells bearing receptors for IgE, can trigger intense inflammatory reactions in response to the allergen for which the cell-bound IgE has specificity. The presence and quantity of such allergen-specific IgE antibodies is a key metric in diagnosing and evaluating food allergy sensitivities. However, the quantity of IgE antibodies ranges widely, making quantification an incomplete
TABLE 2-1 Overall Differences Between IgE- and Non-IgE-Mediated Food Allergies
| Class | IgE-Mediated | Non-IgE-Mediated |
|---|---|---|
| Time to onset of reaction | Immediate <2 hoursa |
Delayed Often >4-6 hours |
| Volume usually required for reaction | Small | Sometimes larger |
| Typical symptoms | Urticaria Angioedema Vomiting Diarrhea Oral itching Anaphylaxis |
Diarrhea Food refusal Failure to thrive Gastroesophageal reflux Irritability/abdominal distension Eczema |
| Common diagnostic procedures | Above signs or symptoms by history or oral food challenge and positive IgE antibody (skin prick test or serum specific IgE) |
Sometimes can do home-based elimination and rechallenge sequence; some require rechallenge in hospital setting |
a In the case of mammalian meat, onset times for reactions related to galactose-alpha-1, 3-galactose (alpha-gal) can be longer than 2 hours. (See “Delayed Anaphylaxis Associated with Mammalian Meats” in Chapter 4.)
TABLE 2-2 Types of Food Allergies
| GI Food Allergies | |
|---|---|
| IgE-mediated | |
| Immediate gastrointestinal (GI) hypersensitivity | Upper GI symptoms may occur within minutes; lower GI symptoms may occur either immediately or with a delay of up to several hours. Immediate vomiting is the most common reaction and the one most clearly mediated by IgE. |
| Pollen-associated food allergy syndrome (PFAS) | PFAS is an IgE-mediated allergy, often to raw fruits or vegetables, with symptoms including itching or swelling of the lips, mouth, and throat. |
| Non-IgE-mediated | |
| Eosinophilic gastroenteritis (EG) | EG is thought to be non-IgE-mediated although IgE-mediated is possible. EG symptoms vary depending on the portion of the GI tract involved and the localized or widespread infiltration of the GI tract by eosinophils. |
| Eosinophilic esophagitis (EoE) | EoE symptoms vary depending on the age of the person, from reflux-like symptoms and vomiting in school-age children, to refusal to eat and impaction in teenagers and adults. |
| Food protein–induced enteropathy | Vomiting, diarrhea, and sometimes protein-losing enteropathy occur in this condition. |
| Food protein–induced allergic proctocolitis (AP) | AP typically presents as specks or streaks of blood mixed with mucus in the stool of otherwise healthy infants. Food-specific IgE is generally absent. The suspected role of food allergy is based on history of exposure to allergens, not diagnostic tests. |
| Food protein–induced enterocolitis syndrome (FPIES) | Non-IgE-mediated FPIES usually occurs in infants and presents as chronic emesis, diarrhea, and failure to thrive. Milk and soy protein are the leading, but not exclusive, causes. The reaction is delayed, occurring approximately 2 hours or later after ingesting the food. |
| Cutaneous | |
| IgE-mediated | |
| Acute urticaria | Round and irregular pruritic (itchy) lesions appear quickly after ingesting an allergenic food. Although IgE-mediated food allergy often causes urticaria, it is not the leading cause. |
| Angioedema | Likely IgE-mediated when caused by food and involves “non-pitting, non-pruritic, well-defined edematous swelling that involves subcutaneous tissues, abdominal organs, or the upper airway.” Upper airway involvement signifies a likely medical emergency. |
| Contact urticaria | Can be induced by either an IgE-mediated food allergy or a nonimmunologic histamine reaction. |
| Non-IgE-mediated | |
| Atopic dermatitis (AD) | AD involves complex interactions between skin barrier dysfunction and environmental factors, linked in some individuals to mutations in the protein filaggrin. The role of food allergy, from sensitization to subsequent skin reaction, remains a topic of debate. |
| Allergic contact dermatitis (ACD) | ACD is a form of eczema caused by reactions to chemical haptens in foods; it is associated with marked pruritus, erythema (redness of the skin), papules, vesicles, and edema. |
SOURCE: Boyce et al., 2010.
indicator of function, allergen sensitivity, or reaction severity. Moreover, some individuals with measurable IgE specific for particular food allergens do not exhibit clinical signs and symptoms of food allergy when they ingest such allergens. This supports the conclusion that allergen-specific IgE is (by definition) required for a person to exhibit clinical food allergy to that allergen, but the presence of such allergen-specific IgE is not sufficient for a person to exhibit a food allergy to that allergen. IgE is typically measured in serum or determined through allergen skin prick tests (Berin, 2015). The IgE-mediated reactions observed in patients with food allergy are often grouped into immediate onset reactions and immediate plus late-phase reactions and can include life-threatening anaphylaxis, gastrointestinal hypersensitivity, urticaria, and pollen-associated food allergy syndrome (Berin, 2015). Allergen-specific IgE may be detectable in atopic dermatitis (AD) and eosinophilic esophagitis (EoE) and these may be food-responsive disorders, but a direct correlation of the pathology in these disorders with IgE and specific food allergen triggers is less clear (see Box 2-1 for these and other basic definitions).
Common Food Allergy Signs and Symptoms
Food allergy can manifest through a wide range of signs and symptoms with varying severity, which makes diagnosis challenging, particularly if a history of allergic reactions has not already been established (see Chapter 4). The most common signs and symptoms typically manifest on the skin (i.e., cutaneous), in the gastrointestinal (GI) tract, in the respiratory system, or in all of these areas. These signs and symptoms include development of uticaria (hives), angioedema (tissue swelling), circulatory collapse, dizziness, coughing, vomiting, stomach cramps, nausea, and others (ACAAI, 2015).
Food allergy is rarely the principal cause of respiratory conditions, but IgE-mediated respiratory symptoms can be a key finding in diagnosing anaphylaxis (James, 2003). In addition, occupational asthma caused by exposure to food occurs more frequently among individuals in certain professions such as bakers, millers, or grain elevator workers. A number of specific cutaneous and GI food allergy conditions, as defined by the NIAID, are listed in Table 2-2. Food-induced anaphylaxis—which may accompany or encompass other conditions—is an acute, potentially life-threatening syndrome with multisystemic manifestations due to the rapid release of inflammatory mediators (Boyce et al., 2010). It can occur within minutes to a couple of hours of ingesting the food (or longer for alpha-gal reactions related to mammalian meat). The reaction usually develops and, if appropriately treated, resolves completely within hours, but in rare
instances symptoms can occur hours later. For some individuals, exercise after ingesting an allergenic food may increase the likelihood of anaphylaxis and severity of clinical symptoms (Romano et al., 2001). In those with severe and potentially life-threatening anaphylaxis, findings include airway compromise (swelling of tissues in the throat and upper airways), impaired breathing (e.g., nasal congestion and rhinorrhea and narrowing of airways in the lungs), and/or circulatory problems (e.g., changes in heart rate, shock). Skin and mucosal changes usually, but not always, also occur (e.g., flushing, pruritus, hives in the skin; swelling of the tissues of the lips, mouth, and GI system). For a more detailed description of anaphylaxis and its diagnosis and management, see Chapter 6.
Common Allergenic Foods and Food Allergens
Although, in theory, any protein molecule could be allergenic and a large number of foods have been noted to cause IgE-mediated food allergy, a small number of foods cause most of the clinical reactions (Boyce et al., 2010). Foods that are categorized as allergenic differ by country because the prevalence of food allergy caused by various foods differs by region of the world and according to the eating habits within a population. In the United States, the foods listed below are currently considered allergenic for regulatory purposes. However, the committee did not restrict its findings, conclusions, and recommendations to this list, and has included foods that are viewed as allergenic by other countries (e.g., sesame and some fruits and vegetables). The lists have regulatory implications for managing allergens (e.g., food product labeling) that affect many stakeholders. Importantly, the foods that are or are not on these “official” lists of allergens affect consumers who need to avoid specific allergens, both in their country and when they travel internationally. Chapter 7 includes a description of the criteria that different countries follow in order to categorize a food as allergenic and a list of foods that are commonly considered allergenic in various countries. Chapter 7 also includes the committee’s recommendations to update the list of allergens in the United States.
In each of these allergenic foods, specific glycoproteins trigger the reaction and production of IgE antibodies that are reactive with those glycoproteins (during the period when the patient is becoming sensitized to those allergens). Clinical reactions are then triggered upon re-exposure to such foods after sensitization has occurred. Identifying and tracking these allergenic proteins and how they are affected by factors such as variation in food preparation is crucial to understanding mechanisms of food allergy reactions and potential avenues of prevention or treatment. For example, it will be important to understand how and why certain processes of food preparation can neutralize or diminish the ability of allergens either to induce sensitization or elicit clinical reactions (see also Chapter 7). Each type of allergenic food can contain a major or several allergenic proteins, as illustrated by the following list:
- Peanuts: Ara h 1, Ara h 2, Ara h 3, Ara h 8, Ara h 9
- Milk: aS1-casein, aS2-casein, β-casein, κ-casein, β-lactoglobulin, α-lactalbumin
- Eggs: ovomucoid, ovalbumin, ovotransferrin, lysozyme
- Fish: parvalbumin
- Shellfish: tropomyosin
- Wheat: Tri a 12, Tri a 14, Tri a 19, Tri a 21, Tri a 26
- Soy: Gly m 1, Gly m 4, Gly m 5, Gly m 6, Gly m 8
Food Allergy Misconceptions
Perceptions of food allergy conditions, patterns, and treatments can have a profound impact on both patient safety and cultural/societal accommodations for those with food allergy. Misconceptions persist among doctors, patients with allergies, and the general public—some of which could be potentially dangerous. Misconceptions are particularly significant among parents or guardians, as food allergy often manifests first in children.
Misconceptions fall into two major types: those related to basic concepts or management of a food allergy. This section addresses the former. The timing of the clinical symptoms after food ingestion, how long symptoms of food allergy actually last after ingestion, and the foods more or less likely to cause severe symptoms are often misunderstood. Bock (1987) reported that 28 percent of parents thought that their children had adverse reactions to foods but only 8 percent of the children actually did when challenged with the food. The Chicago Food Allergy Research Survey for Parents of Children with Food Allergy, a study conducted in 2008, solicited answers from 2,945 parents from across the United States (Gupta et al., 2010a); significant misconceptions or absences in knowledge were revealed. Some 52 percent of parents answered that anaphylaxis was more likely to be fatal in children than in adolescents, while the opposite is true. Almost half of participants believed that there is a cure for food allergy and more than two-thirds believed that a medicine could be taken as prevention. Furthermore, 40 percent of respondents reported “experiencing hostility from other parents when trying to accommodate their child’s food allergy.” That perceived hostility might point to a lack of awareness among the general public, which can fail to recognize legitimate food allergy dangers. However, another study (Gupta et al., 2009) concluded that most members of the public recognize the real risk of food allergy–related deaths and can even identify key symptoms. Other misconceptions among surveyed parents include a belief that food additive allergies are common (actually rare, despite the prevalence of additives in processed foods) and a lack of awareness about the rates at which children outgrow certain food allergy sensitivities (Gupta et al., 2010a).
These inaccurate beliefs were less common among parents who visited allergists rather than primary care physicians, which points to the potential lack of knowledge outside of specialists. Medical practitioners, and especially allergists, have an ongoing responsibility to educate the public, patients, and their fellow physicians (see Chapter 6). However, numerous studies suggest deficits in understanding these basic concepts among many different stakeholders, including physicians (Desjardins et al., 2013; Morawetz et al., 2014). Various surveys indicate misunderstandings among medical professionals in recognizing risk factors for food allergy reactions,
including anaphylaxis (Clark et al., 2004; Gupta et al., 2010b; Turner et al., 2016; Wang et al., 2014).
Knowledge deficits regarding food allergy also have been noted among school nurses (Carlisle et al., 2010), child care providers (Greiwe et al., 2015), emergency response providers (Jacobsen et al., 2012), restaurant personnel (Ahuja and Sicherer, 2007; Bailey et al., 2011), and teachers (Ercan et al., 2012; Polloni et al., 2013). Overall, stakeholders and the general public are currently insufficiently educated (see also Chapters 4, 5, and 6).
MECHANISMS
Mechanisms of Disease
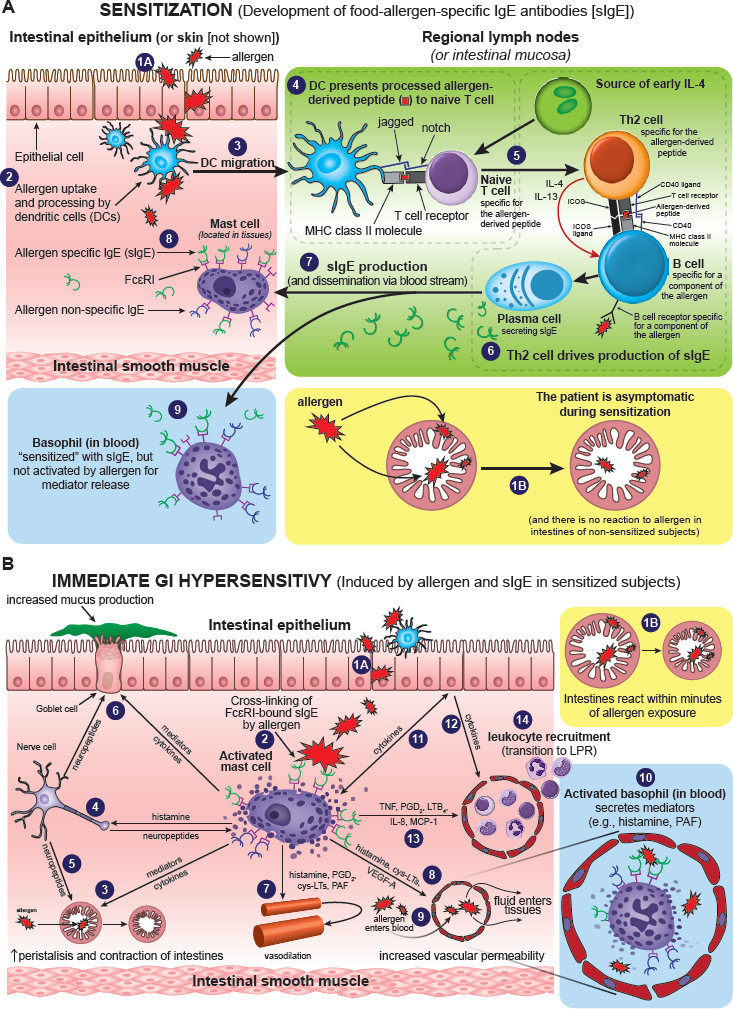
This report is not meant to delve deeply into the basic mechanisms underlying food allergy, but mainly to address more practical aspects, such as diagnosis and management. Still, unraveling the pathological processes of food allergy is critical for understanding how to diagnose and clinically evaluate food allergy and for developing short- and long-term mitigation strategies. The intricate biological systems involved and the wide range of clinical manifestations of food allergy make this a long-term process characterized by incremental, albeit ultimately important, progress. For purposes of this report, this section briefly explores two principal aspects of clinical food allergy: the mechanism of the reaction and the mechanism of immunological tolerance. Figure 2-2 represents the mechanistic interactions and complexities of food allergy, which are not fully described in this section. For a more detailed description of the processes readers are referred to other publications (e.g., Berin, 2015; Chinthrajah et al., 2016).
Specific food allergies likely are a result of complex interactions among genes and the environment (including not only factors in the “external environment,” such as pollen, pollution, and pathogenic microbes, but also effects of the microbes that normally reside in us—the “internal environment” of the microbiome) (see Chapter 5 for a detailed description of current knowledge on food allergy determinant factors).
With IgE-mediated food allergy—the classification under review here—allergic sensitization must precede manifestation of the full reaction. Sensitization is defined as the process by which an individual produces detectable IgE to a particular allergen (allergen-specific IgE [sIgE]). This can be called, operationally, the “offending allergen.” (See also Figure 2-2 for an explanation of this process and Box 2-2 for definitions of key cellular components in food allergy reactions.) However, it is important to recognize that sensitization alone does not constitute clinical food allergy. In fact, sensitization can persist without the patient manifesting any clinical signs of food allergy.
This finding is an important part of understanding the diagnostic workup in food allergy. Having sIgE against a food allergen means the person has been sensitized to that allergen and therefore might exhibit a clinical food allergy reaction to that allergen, but a more specific test (the double-blind, placebo-controlled oral food challenge) would be required to diagnosis an allergy to that food in such a sensitized person.
Some allergens produce organ-specific reactions, but the mechanisms that could explain such clinical variation are not well understood. For example, peanut and egg are the most common triggers of GI symptoms, and peanut causes more respiratory reactions than other allergens (Berin, 2015).
It has been increasingly recognized that skin exposure can be a powerful driving factor in food sensitization. One leading hypothesis about how sensitization occurs is that humans naturally become tolerant to food encountered orally in the diet, but sensitization is favored, at least in “susceptible” individuals, through skin exposure (see Chapter 5). The interplay between reactions occurring in the skin and within the GI tract is thought to be an important element of sensitization. For example, loss-of-function variants in the gene encoding filaggrin, a key protein in the regulation of epidermal barrier function and health, have an established link to eczema, but research also links variants in filaggrin to food allergy (Lack, 2012). Lack of normal skin barrier integrity facilitates the development of food allergy. Peanut sensitization in particular is linked with skin exposure, but studies also show that childhood use of lotions containing oat led to much higher rates of oat allergies (Boussault et al., 2007). The molecular underpinnings of this hypothesis, however, are not fully understood.
Mechanisms of Tolerance and Desensitization
Two major terms that are used for defining a situation that is commonly known as “growing out” of a food allergy are desensitization and tolerance (see Figure 2-1). In some instances, natural tolerance (as opposed to the tolerance induced by specific therapeutic interventions) to some foods that once induced food allergy in that individual will develop over time. Accounts of spontaneous resolution of IgE-mediated food allergy vary according to food, age, and geography, but estimates indicate that 65 to 80 percent of individuals will develop such natural tolerance for cow milk, wheat, soy, and egg, and only 10 to 20 percent for peanut and tree nuts (Campbell et al., 2015).
For those who have not acquired tolerance naturally, a cure for food allergy does not exist yet. Strategies of management and treatment include avoidance of allergens, immediate treatment of symptoms, and the induction of tolerance. Multiple mechanisms play a role in regulating food allergy, many of which are extrathymic, resulting in a range of clinical
recommendations and conflicting data (Campbell et al., 2015). Hallmarks of tolerance include a reduction in sIgE production, decreased allergen-IgE-induced basophil activation, increased allergen-specific IgG4, and induction of T regulatory (Treg) cells (Berin, 2015; Campbell et al., 2015; Chinthrajah et al., 2016).
Recent studies have begun to investigate specific treatments to induce food allergy desensitization or tolerance. It is important to understand that the term desensitization is used here to mean that, while continuing on a specific course of treatment with the offending allergen, the individual will tolerate more of the food on food challenge, even in some cases to the point of not reacting to “serving sized” amounts of the food. Desensitization, however, does not guarantee true tolerance (defined here as a long-term loss of clinical reactivity to the allergen under conditions of no further exposure to the offending allergen). A more recent term, “sustained unresponsiveness” was coined to describe what happens when the treatment for food allergy is stopped. In some such individuals, unresponsiveness to the offending allergen lasts weeks to months, while in others, desensitization is lost more quickly. The mechanisms that may explain desensitization versus sustained unresponsiveness versus true tolerance are being intensely investigated, as are approaches of immune system monitoring that might help classify individuals into one of these three groups with respect to the outcome of treatment. Some of the mechanisms by which treatments for food allergy may be associated with changes in the clinical symptoms include the occurrence during the treatment of natural tolerance noted above (this is one reason why clinical studies of new treatments would include a placebo group), reduction in production of allergen-specific IgE, decreased antigen- and food-specific IgE-dependent basophil activation, increased allergen-specific IgG4 (one effect of which may be to bind allergen before it can be encountered by sIgE and the surface of basophils and mast cells), and the induction of Treg cells or anergic T cells (Campbell et al., 2015). However, these possibilities, and others, are still under investigation). The major unknown about the mechanism of oral treatment–induced desensitization or tolerance is whether ongoing exposure to the protein in the food is necessary to sustain long-term beneficial effects of the treatment.
OVERALL CONCLUSIONS
Food allergy is defined as an adverse health effect arising from a specific immune response that occurs reproducibly on exposure to a given food. Various types of food allergies, such as immediate gastrointestinal hypersensitivity or eosinophilic gastroenteritis, occur and they can be classified as IgE-mediated and non-IgE-mediated food allergies. Many of them present common respiratory, gastrointestinal, and cutaneous signs and symptoms.
The definition and diagnosis of an allergy is rendered even more complicated because other gastrointestinal conditions, such as a food intolerance, can easily be misinterpreted as a food allergy. Given this diversity in signs and symptoms and underlying mechanisms, many misconceptions exist among the general public about what a food allergy is and how to identify one. More importantly, these misconceptions also are common among physicians, emergency care personnel, nurses, and others who are recognized as public health professionals. These misconceptions have tremendous implications for the public at large and specifically for allergic individuals and their families. For example, a diagnostic error can affect health outcomes, including psychological distress, or can lead to unnecessary management strategies.
Many fundamental mechanisms are now understood regarding how IgE-mediated food allergies develop and what is responsible for the signs and symptoms induced during allergic reactions to food. For example, it is well known that upon re-exposure following sensitization to an antigen, the antigen-induced aggregation of antigen-specific IgE binds to receptors on specialized cells (including mast cells in tissues and basophils in the blood). Such aggregation activates those specialized cells, releasing a variety of potent biological mediators that in turn result in the typical food allergy signs and symptoms. However, many questions are still being explored. A better understanding of the mechanistic processes underlying food allergy, and of the mechanisms that contribute to the various potential host responses to different forms of therapy for food allergy, will be invaluable in advancing the development of better prevention strategies, diagnostic methods, and treatments of food allergy.
RESEARCH NEEDS
Conducting research related to the mechanistic processes underlying food allergy is essential in making significant advances to develop better methods to prevent disease or reduce its severity; predict, diagnose, and monitor disease; and optimally manage and treat, and ultimately to cure, food allergy. These mechanistic processes include disease predispositions, origins and onset, normal and disordered oral tolerance to foods, factors that contribute to disease severity, and variation in individual responses to different forms of therapy.
One of the most prominent hypotheses for how food allergy develops—the dual-allergen hypothesis—proposes that environmental exposure to food allergens through the skin early in life can lead to allergy, while consumption of these foods during a developmentally appropriate period early in life results in tolerance. Under this hypothesis, children who avoid allergens in their diet but are still exposed to them in the environment might
be more likely to develop an allergy than those not exposed. Supporting this hypothesis are data suggesting that early dietary introduction of peanut products may confer protection against peanut allergy as well as data suggesting that loss of function of filaggrin, a protein important for epithelial structure, confers a risk for food sensitization. However, many questions remain about the mechanisms by which sensitization and tolerance occur and about which elements of the immune system represent the most important contributors to the severity of food allergy or the establishment of tolerance. For example, studies have shown that biochemical indicators of tolerance include a reduction in allergen-specific IgE production, decreased allergen-IgE-induced basophil activation, increased allergen-specific IgG4, and induction of Treg cells or anergic T cells. However, some of the data are conflicting and more studies are needed to better understand the role of these factors in food allergy
Another prevalent hypothesis is the microbial hypothesis, which states that the decrease in early childhood exposure to microbes may alter the development of early immunoregulatory responses, leading to the development of allergic disorders. For example, exposure to microbes during the perinatal period, may influence interactions between the developing microbiota and the immune system at the cellular and molecular levels and in turn affect health outcomes. Although the potential relationships between exposure to microbes early in life and the onset of food allergies have been explored, specific changes in the microbial profile of individuals, their particular interactions with the immune system, and how these interactions might be associated with food allergy have not been studied in depth.
To fill gaps in knowledge in this area, studies should be conducted to accomplish the following objectives:
- Elucidate the molecular and cellular mechanisms that account for the differences between innate tolerance versus food sensitization and between food sensitization versus food allergy.
- Identify the mechanisms, in patients with food allergies, for acquiring tolerance to the offending food allergen, without therapeutic intervention, as well as for responding to therapeutic interventions by developing transient desensitization versus sustained unresponsiveness versus true tolerance to the offending food allergens.
- Define how particular products and functions of mast cells, basophils, and other effector cells can contribute to the signs and symptoms of food allergic reactions, including anaphylaxis, and identify factors that may contribute to individual variation in the pathophysiological responses to such products.
- Study the role of immunoglobulins other than IgE, such as IgG4 or IgA, and of effector cells in addition to mast cells and basophils, in modulating (i.e., enhancing or reducing) food allergic responses.
- Identify and describe the roles of the skin and intestinal barriers in protecting individuals from developing food sensitization or a food allergy, and identify ways in which protective aspects of barrier function can be enhanced and factors that diminish barrier function be reduced.
- Examine the interactions between the microbiota and the host immune system that may favor or protect against the development of a food allergy, and define the extent to which the microbiota or its products can be manipulated to enhance resistance to the development of food allergy.
REFERENCES
ACAAI (American College of Allergy, Asthma & Immunology). 2015. Types of allergies: Food allergy. http://acaai.org/allergies/types/food-allergies (accessed September 15, 2015).
Ahuja, R., and S. H. Sicherer. 2007. Food-allergy management from the perspective of restaurant and food establishment personnel. Ann Allergy Asthma Immunol 98(4):344-348.
Bailey, S., R. Albardiaz, A. J. Frew, and H. Smith. 2011. Restaurant staff’s knowledge of anaphylaxis and dietary care of people with allergies. Clin Exp Allergy 41(5):713-717.
Berin, M. C. 2015. Pathogenesis of IgE-mediated food allergy. Clin Exp Allergy 45(10): 1483-1496.
Bock, S. A. 1987. Prospective appraisal of complaints of adverse reactions to foods in children during the first 3 years of life. Pediatrics 79(5):683-688.
Boussault, P., C. Leaute-Labreze, E. Saubusse, S. Maurice-Tison, M. Perromat, S. Roul, A. Sarrat, A. Taieb, and F. Boralevi. 2007. Oat sensitization in children with atopic dermatitis: Prevalence, risks and associated factors. Allergy 62(11):1251-1256.
Boyce, J. A., A. Assa’ad, A. W. Burks, S. M. Jones, H. A. Sampson, R. A. Wood, M. Plaut, S. F. Cooper, M. J. Fenton, S. H. Arshad, S. L. Bahna, L. A. Beck, C. Byrd-Bredbenner, C. A. Camargo, Jr., L. Eichenfield, G. T. Furuta, J. M. Hanifin, C. Jones, M. Kraft, B. D. Levy, P. Lieberman, S. Luccioli, K. M. McCall, L. C. Schneider, R. A. Simon, F. E. Simons, S. J. Teach, B. P. Yawn, and J. M. Schwaninger. 2010. Guidelines for the diagnosis and management of food allergy in the United States: Report of the NIAID-sponsored expert panel. J Allergy Clin Immunol 126(6 Suppl):S1-S58.
Campbell, D. E., R. J. Boyle, C. A. Thornton, and S. L. Prescott. 2015. Mechanisms of allergic disease—Environmental and genetic determinants for the development of allergy. Clin Exp Allergy 45(5):844-858.
Carlisle, S. K., P. A. Vargas, S. Noone, P. Steele, S. H. Sicherer, A. W. Burks, and S. M. Jones. 2010. Food allergy education for school nurses: A needs assessment survey by the Consortium of Food Allergy Research. J Sch Nurs 26(5):360-367.
Chinthrajah, R. S., J. D. Hernandez, S. D. Boyd, S. J. Galli, and K. C. Nadeau. 2016. Molecular and cellular mechanisms of food allergy and food tolerance. J Allergy Clin Immunol 137(4):984-997.
Clark, S., S. A. Bock, T. J. Gaeta, B. E. Brenner, R. K. Cydulka, and C. A. Camargo. 2004. Multicenter study of emergency department visits for food allergies. J Allergy Clin Immunol 113(2):347-352.
Desjardins, M., A. Clarke, R. Alizadehfar, D. Grenier, H. Eisman, S. Carr, T. K. Vander Leek, L. Teperman, N. Higgins, L. Joseph, G. Shand, and M. Ben-Shoshan. 2013. Canadian allergists’ and nonallergists’ perception of epinephrine use and vaccination of persons with egg allergy. J Allergy Clin Immunol Pract 1(3):289-294.
Ercan, H., A. Ozen, H. Karatepe, M. Berber, and R. Cengizlier. 2012. Primary school teachers’ knowledge about and attitudes toward anaphylaxis. Pediatr Allergy Immunol 23(5):428-432.
Greiwe, J. C., F. Pazheri, and B. Schroer. 2015. Nannies’ knowledge, attitude, and management of food allergies of children: An online survey. J Allergy Clin Immunol Pract 3(1):63-67.
Gupta, R. S., J. S. Kim, E. E. Springston, B. Smith, J. A. Pongracic, X. Wang, and J. Holl. 2009. Food allergy knowledge, attitudes, and beliefs in the United States. Ann Allergy Asthma Immunol 103(1):43-50.
Gupta, R. S., E. E. Springston, B. Smith, J. S. Kim, J. A. Pongracic, X. Wang, and J. Holl. 2010a. Food allergy knowledge, attitudes, and beliefs of parents with food-allergic children in the United States. Pediatr Allergy Immunol 21(6):927-934.
Gupta, R. S., E. E. Springston, J. S. Kim, B. Smith, J. A. Pongracic, X. Wang, and J. Holl. 2010b. Food allergy knowledge, attitudes, and beliefs of primary care physicians. Pediatrics 125(1):126-132.
Jacobsen, R. C., S. Toy, A. J. Bonham, J. A. Salomone, 3rd, J. Ruthstrom, and M. Gratton. 2012. Anaphylaxis knowledge among paramedics: Results of a national survey. Prehosp Emerg Care 16(4):527-534.
James, J. M. 2003. Respiratory manifestations of food allergy. Pediatrics 111(6 Pt 3):1625-1630.
Lack, G. 2012. Update on risk factors for food allergy. J Allergy Clin Immunol 129(5): 1187-1197.
Morawetz, D. Y., H. Hiscock, K. J. Allen, S. Davies, and M. H. Danchin. 2014. Management of food allergy: A survey of Australian paediatricians. J Paediatr Child Health 50(6):432-437.
Polloni, L., F. Lazzarotto, A. Toniolo, G. Ducolin, and A. Muraro. 2013. What do school personnel know, think and feel about food allergies? Clin Transl Allergy 3(1):39.
Romano, A., M. Di Fonso, F. Giuffreda, G. Papa, M. C. Artesani, M. Viola, A. Venuti, V. Palmieri, and P. Zeppilli. 2001. Food-dependent exercise-induced anaphylaxis: Clinical and laboratory findings in 54 subjects. Int Arch Allergy Immunol 125(3):264-272.
Turner, P. J., J. L. Baumert, K. Beyer, R. J. Boyle, C. H. Chan, A. T. Clark, R. W. Crevel, A. DunnGalvin, M. Fernandez-Rivas, M. H. Gowland, L. Grabenhenrich, S. Hardy, G. F. Houben, J. O’B Hourihane, A. Muraro, L. K. Poulsen, K. Pyrz, B. C. Remington, S. Schnadt, R. van Ree, C. Venter, M. Worm, E. N. Mills, G. Roberts, and B. K. Ballmer-Weber. 2016. Can we identify patients at risk of life-threatening allergic reactions to food? Allergy 71(9):1241-1255.
Wang, J., M. C. Young, and A. Nowak-Wegrzyn. 2014. International survey of knowledge of food-induced anaphylaxis. Pediatr Allergy Immunol 25(7):644-650.