5
Potential Genetic and Environmental Determinants of Food Allergy Risk and Possible Prevention Strategies
The increase in food allergy has captured the attention of the medical and research communities and the general public. Although the extent of the increase and the most affected countries are not accurately known, there is little doubt that immunoglobulin E (IgE)-mediated food allergy and anaphylaxis were rarely reported 50 years ago but are now commonly described (see Chapter 3). The prevalence of allergenic reactions to foods might differ by region of the world in part because of differences in exposures to specific foods. However, the drivers for this modern day epidemic in food allergy are poorly understood. It is not clear whether this phenomenon is part of the global rise in all allergic diseases at the end of the 20th century, or is due to a new set of unique factors, or to a combination of both.
Like other complex diseases, food allergy is thought to be caused by a combination of genetic and environmental factors. This chapter describes the state of the scientific evidence related to what are currently thought to be the most relevant genetic and environmental risk factors as well as genome-environment (GxE) interactions. The chapter starts with a discussion of the application of the developmental/ecological model (see Chapter 1) to food allergy risk factors. To that effect, a brief summary of the parallel development of the immune system of the child is included. The concept of atopic march1 is briefly introduced as potentially important when considering prevention strategies. Although other immune-related diseases, such as eczema
__________________
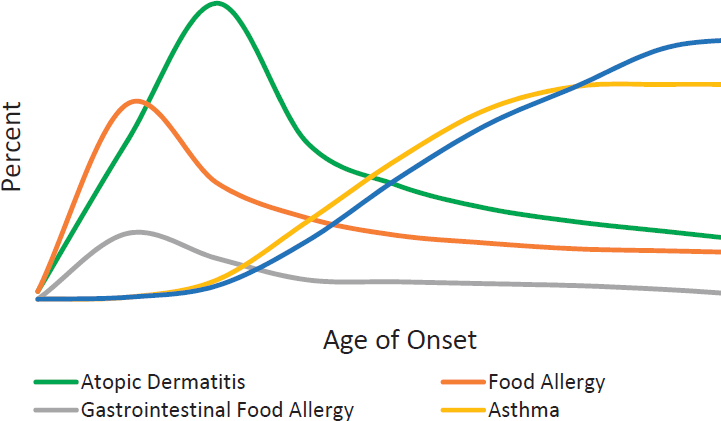
1 The atopic march refers to the idea that atopic disorders progress over time from eczema (i.e., atopic dermatitis) to asthma (see Box 5-2 and Figure 5-1).
(also known as “atopic dermatitis”), are often components of the atopic march that includes food allergy, not all people with eczema develop food allergy. Consequently, preventing eczema might not always decrease the risk of developing food allergy. Therefore, the committee decided to focus only on the relevant literature directly linked to the development of food allergy and findings associated with eczema alone are not included in this report. Also, the chapter concentrates on food allergy as an outcome except for a few risk determinants for which there are no data on food allergies. In these cases, the committee explored food sensitization2 as a potential surrogate outcome. Although food sensitization is on the causal pathway for IgE-mediated food allergy, care should be taken in interpreting these results because food sensitization may be a nonspecific marker predisposition to atopy in general, not to food allergy in particular.
To provide context for the current scientific evidence on risk determinants, the methodological limitations of studies to date are explained. The pre- and postnatal environmental risk factors that might explain the development of food allergies have been grouped into emerging hypotheses: (1) microbial hypotheses (hygiene and old friends); (2) allergen avoidance hypothesis; (3) dual allergen exposure hypothesis; (4) nutritional immunomodulation hypothesis; and (5) other hypotheses. Each section on a specific determinant factor ends with a conclusion statement about the evidence supporting the link between exposure to the considered determinant and food allergies. At the end of the chapter, the committee provides their overall conclusions, recommendations, and research needs about strategies for preventing food allergies.
FINDING PREVENTIVE MEASURES: A DEVELOPMENTAL/ECOLOGICAL APPROACH
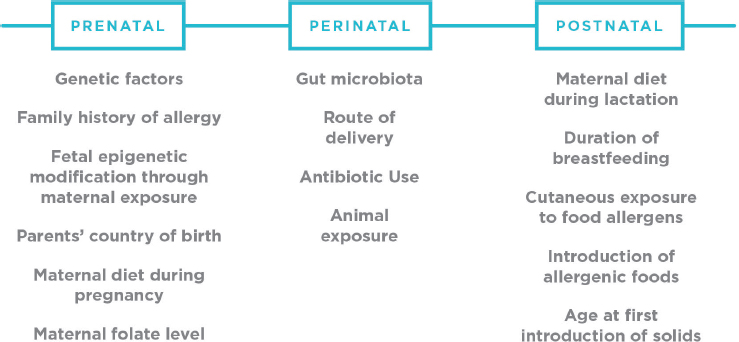
As described in Chapter 1, the committee approached its task from a developmental/ecological perspective. From the developmental perspective, the committee emphasizes the importance of developmental timing for exposures and for safety. In considering the risk determinants for developing food allergies, the committee focused on the different developmental periods—prenatal, early childhood, primary school-age, adolescence, adulthood, and elder years. In the prenatal period and first year of life, a fetus and infant’s gut goes through substantial microbiome and immune developmental changes (see Box 5-1). This key period presents a window of opportunity to modify health outcomes at a time when infants are ready to
__________________
2 Sensitization is a condition where an individual produces detectable immunoglobulin E (IgE) to a particular allergen. It can precede a food allergy reaction, but not all individuals with detectable IgE to a food allergen will experience a food allergy reaction.
begin eating solid foods. Due to the importance of this period in establishing the onset of food allergies, the scientific literature on food allergy risk factors has focused more on these early life stages and less on those changes that may occur in older children, adolescents, or adults. Therefore, while the committee’s conclusions and recommendations were crafted through a developmental lens, they are limited by the preponderance of scientific literature on these early ages.
Food Allergies and the Atopic March
Within the developmental perspective, the committee considered the concept of the atopic march (see Box 5-2) in their deliberations. The atopic march refers to the idea that atopic disorders progress over time from eczema to asthma (see Figure 5-1). In fact, in some publications, eczema is viewed as a proxy for food allergies because eczema frequently precedes the development of food allergies. In fact, eczema and food allergies are distinct conditions with different etiologies and it is not appropriate to assume that eczema is a surrogate for food allergy. Although the concept of the atopic march is generally accepted, the interplay of the various related immune conditions is still being studied and, therefore, it would be premature to adopt the general idea that strategies to prevent atopic disorders that typically occur earlier in a child’s development necessarily would also prevent the onset of food allergy. Additional prospective cohort studies with the appropriate methodologies are needed, particularly to understand the relationship between other allergic disorders and food allergy. Thus, the committee did not include other allergic disorders (i.e., wheeze, asthma, eczema, or allergic rhinitis) or their risk factors in their review of the evidence of potential determinants of food allergy.
METHODOLOGICAL LIMITATIONS
Current evidence about the risk factors associated with food allergy or sensitization is derived primarily from epidemiological (observational or ecological) studies. In addition to potential limitations in any research study—such as lack of generalizability, small number of samples, and inaccurate outcomes measurements—epidemiological studies need to be interpreted appropriately, with particular consideration to potential confounding factors and their careful adjustment. For instance, being at high risk of allergic disease could be a confounder when exploring the effects of breastfeeding in food allergies because high-risk families are more likely to follow guidelines, which might inform them about the putative protective effects of breastfeeding. If researchers do not adjust their analysis for family history of allergy (the main risk of allergy development), breast-
feeding can be misinterpreted as increasing the risk of allergic disease. This phenomenon is called “reverse causation” and is one of the reasons why randomized controlled trials (RCTs) are required to provide strong evidence that a factor is indeed causally related. Even with the best intentions, observational studies can be undermined by unmeasured confounders (i.e., residual confounding). High-quality data demonstrating causation should exist before recommendations are incorporated into public health guidelines. In most cases, this would mean RCTs. However, when evidence is not strong or trials are ethically difficult to mount (such as is the case for breastfeeding where randomization to a nonbreastfeeding arm would be unethical), clinicians need to interpret emerging or less robust evidence and provide carefully framed information to individual patients and their families to inform health decisions.
Until recently, food allergy has been less common than other allergic diseases. Therefore earlier allergy studies generally did not focus on food allergy as an outcome. It is only recently, as food allergy prevalence has increased, that attempts have been made to more precisely define and measure food allergy. Measurement methods have evolved from often inaccurate
self- or parent-reported data to better methods, such as the results of oral food challenges (OFCs). Recent literature, particularly after 2010, has more consistently reported food allergy outcomes using what is now regarded as the gold standard measurement—double-blind, placebo-controlled oral food challenge (DBPCOFC), in which the food is disguised so that neither parent nor health care professional knows whether the food or a placebo is being offered. Some experts have recommended that for children younger than 2 years, open OFC, in which foods in their natural state are offered (versus DBPCOFC) also can be included in the definition of gold standard because, in this age group, subjective symptoms do not complicate medical history and objective signs can be reliably used as endpoints.
Even DBPCOFC are limited by methodologic differences among studies (see Chapter 4). In addition, criteria for defining a positive oral challenge (i.e., a food allergic reaction) have not been formalized until recently (Koplin et al., 2012b; Sampson et al., 2012; see Chapter 4). Although most protocols state that a positive challenge is evidenced by an immediate reaction consistent with IgE-mediated food allergy, such as urticaria (hives), angioedema, or anaphylaxis, interpretation of more subjective symptoms, such as abdominal pain or nausea, or the more ubiquitous and less clearly defined sign of an eczema flare, remains difficult. Differences in criteria for defining a positive OFC across different studies and research centers hinders the ability to compare food allergy prevalence estimates among studies, to identify risk factors (because phenotypes might vary across different study cohorts), and to assess the success of different treatment strategies (including oral immunotherapy).
It should be noted, however, that performing large-scale OFCs is not always possible because of issues with compliance, risk to participants, and cost. As stated in Chapter 2, many population-based studies have relied on the detection of food-specific serum IgE (sIgE) antibodies as an indirect marker of food allergy, either alone or in conjunction with reported symptoms on ingestion of the food. These studies do provide insights into the temporal trend changes in food allergy prevalence, but should be viewed with caution when assessing risk factors for predicting food allergy owing to the high false positive rate and low specificity of this method. Self-reported measures tend to overreport food allergy due to the inability of individuals to distinguish between symptoms of food intolerance and food allergy. It is also not possible to employ reports from parents to determine allergic status to foods that have not yet been introduced into an infant’s diet (see Chapters 3 and 4).
These methodological limitations, and specifically the outcome used to define the food allergy, and their implications for the interpretation of the studies reviewed herein, are noted in conjunction with the specific studies described in this chapter.
APPROACH TO LITERATURE REVIEW
Literature Search Strategy and Study Selection
Electronic literature searches of published systematic reviews (from 2010 to September 2015) and primary studies (from 2012 to September 2015) indexed in Medline, Cochrane Database of Systematic Reviews, EMBASE, and ISI Web of Science were conducted. The complete literature search and screening strategies, study selection flow, and study eligibility criteria are described in Appendix C. The committee based its literature search strategies on the systematic reviews by Marrs et al. and de Silva et al. and on selected individual papers published after those reviews (2012 and beyond) to develop its conclusions (de Silva et al., 2014; Marrs et al., 2013). Where appropriate, other systematic reviews also were considered.
Summary tables for all systematic reviews and studies conducted after 2012 are included in Appendix C. Ongoing trials of risk determinants of food allergy for which results were not available at the time of this publication are summarized in Table 5-1. Selected public health guidelines from various countries are listed in Table 5-2.
Grading the Evidence
For each factor described, the committee made a final conclusion statement considering the preponderance of the evidence collected, as described above. The committee used the approach taken by the 2015 Dietary Guidelines Advisory Committee to grade as strong, moderate, limited, or no grade (DGAC, 2015) (see Table 5-3).
GENETIC AND EPIGENETIC RISK FACTORS
The rise in the prevalence of allergic diseases has occurred more rapidly than can be accounted for by changes in genetic sequence (Tan et al., 2012b). Therefore, similar to other complex diseases, the rising prevalence of allergic diseases is likely due to environmental factors (i.e., the exposome).3 In this way, the rise may be primarily occurring in those who are both genetically predisposed and exposed to the allergenic environment, as well as in those at risk through a heritable epigenetic mechanism from events that occurred when the parents of current children were in utero. Environmental exposures, including lifestyle and diet, interact4 with genetic
__________________
3 The exposome refers to all life course environmental exposures (including factors related to lifestyle, such as smoking or diet) from the prenatal period onward.
4 An interaction is indicated when the simultaneous influence of two or more factors on a phenotype is not additive.
predisposition to modify the risk of disease. For example, the influence of the C-159 T polymorphism on the cluster of differentiation 14 (CD14) gene may be dependent on microbial stimulation from the environment (Lau et al., 2014), with individuals who carry the TT genotype demonstrating increased protection from eczema with exposure to dogs (Myers et al., 2010).
The concept of the epigenome,5 which regulates gene expression and is largely established in utero, is relevant to early life origins of allergic disease. In contrast to deoxyribonucleic acid (DNA) sequences, which are relatively stable, the epigenome can be altered throughout the lifespan, but is particularly sensitive to environmental factors during early life periods (see Figure 5-2). Environmental factors that have often been considered in interaction with genetic risk factors include vitamin D (Koplin et al., 2016; Liu et al., 2011), smoking, air pollution, and microbial exposures (Tan et al., 2012b). Epigenetic considerations for other environmental factors, for which there is evidence of involvement in allergic diseases, have not yet been considered. It also would be useful to consider putative causative factors for food allergy, such as diet and food supplements, in relation to well-known genetic risks, such as filaggrin mutations.
A further consideration is the fact that these environmental risk factors may operate differentially based on the underlying risk category of the individual (i.e., genetic risk or family history, the more traditional form of risk stratification). As discussed below, evidence already exists of different responses to some environmental factors (e.g., vitamin D) based on a genetic risk factor (vitamin D receptor binding protein) (Koplin et al., 2016). In addition to biological variations, risk factors also may affect behavioral patterns, as has been described by Tey et al. (2014). The authors found that those with a family history of allergy were less likely to respond appropriately to guidelines revisions to introduce allergenic solids earlier in the diet of an infant. Future clinical practice guidelines and public health policy may need to take into account the way that a risk factor may differentially affect not only risk of disease, but also the behavior of the individual with a food allergy and/or their caregivers.
This section describes studies on the genetic and epigenetic factors that might affect food allergy outcomes.
__________________
5 Epigenome refers to the chemical changes to the deoxyribonucleic acid and histone proteins (e.g., methylation) of an organism that occur through life and can result in changes to the structure of chromatin and to the function of the genome. These changes can be inherited through transgenerational epigenetic inheritance.
TABLE 5-1 Registered Randomized Controlled Clinical Trials and Observational Studies on Prevention
| Study | Study Design, Country | Population | N | Age When Outcome Is Ascertained | Food Allergy Outcome Definition | Exposure | Question to Answer |
|---|---|---|---|---|---|---|---|
| BEAT (Beating Egg Allergy) | RCT, Australia | Infants with high risk of atopy, 4-6 mo | 8 and 12 mo | Egg allergy assessment | Egg introduction versus placebo | What is the effect of early introduction of egg into the diet of infants at high risk of atopy and subsequent egg allergy? | |
| CoFAR2 | Observational, US | Children with egg and/or milk allergy, 3-15 mo | 515 | After 3 years of age | Peanut allergy | What is the development of peanut allergy in infants (3 to 15 months in age) with known milk or egg allergy? | |
| EAT (Enquiring about Tolerance) | RCT, UK | Infants 3 mo | 1,306 | 3 years | Early introduction of 6 allergenic foods together with breastfeeding versus standard introduction (6 months) | Does introducing certain foods early in a child’s diet along with continued breastfeeding stop infants from developing food allergy? | |
| STEP (Starting Time for Egg Protein) | RCT, Australia | Infants 4-6 mo without eczema but atopic mothers | 1,500 | Egg introduction versus placebo |
| HEAP | Germany | Infants 4-6 mo | 800 | 12 mo | Egg allergy | Egg introduction versus placebo | |
| PreventADALL (Preventing Atopic Dermatitis and Allergies in Children) | RCT, Norway | Infants | 6, 12, 36, and 48 mo | Early food introduction by 3-4 mo | Food allergy to any intervention allergen (cow milk, peanut, wheat, egg) | Is primary prevention of allergic diseases possible by skin care and early food introduction? | |
| PIFA (Pertussis Immunisation and Food Allergy) | Observational (case-control), Australia | Children 14-18 years | 14-18 years | History of consistent clinical symptoms following ingestion of an implicated food and evidence of sensitization to that food by laboratory testing | Whole cell versus acellular pertussis vaccine | What is the possible food allergy-preventive benefit of using whole cell pertussis vaccination compared with acelluar pertussis vaccine for whooping cough vaccination in childhood? | |
| VITALITY | RCT, Australia | Infants 6-8 weeks | 12 months | Challenge-proven food allergy in study participants with positive SPT | Vitamin D (400 IU/day) versus placebo for 10 months | Can vitamin D supplementation in infants prevent food allergy in the first year of life? |
| Study | Study Design, Country | Population | N | Age When Outcome Is Ascertained | Food Allergy Outcome Definition | Exposure | Question to Answer |
|---|---|---|---|---|---|---|---|
| Early Life Origins of the Food Allergy Epidemic | Observational, Canada | Peanut-sensitized children, 4-10 years | 5 years | DBPCOFC to peanut | Eating versus avoiding peanut | Does avoidance of peanut by children with positive SPT to peanut in the first 5 years of life increase the likelihood of developing a persistent peanut allergy by age 5 years? | |
| The Cork BASELINE Birth Cohort Study (BASELINE) | Observational, Ireland | Infants | 2 years | Incidence and prevalence of food allergy | What are the early life factors, including parental allergy, genetic susceptibility measured using fillagrin mutational status, skin barrier function, and vitamin D status and their effect on risk of eczema and food allergy in the first 2 years of life? |
| Probiotic Supplementation in Breastfed Newborn Infants | RCT, US | Infants, 1-7 days old, with intent to be exclusively breastfed for a minimum of 6 months | First 78 weeks of life | Levels of serum FABPs and glutathioneS-transferase (alpha-GST) will be measured as markers of GI permeability and potential food allergy; parental report of feeding intolerance | Probiotic supplementation versus placebo | What is the dose of a probiotic supplement (Bifidobacterium longum subsp. infantis) required to achieve predominant gut colonization in healthy newborn, breastfed infants? Does supplementation with this probiotic reduce the chance of developing eczema and food allergies in enrolled infants? | |
| PROOM-3 | RCT, Sweden | Pregnant women with at least one parent or a sibling with clinical symptoms or history of allergic disease and their newborn infants | 6 and 12 months | IgE-associated disease measured by SPT (milk, egg, wheat, peanut) | Dietary supplementation with L. reuteri and omega-3 PUFA during pregnancy and postnatally versus placebo | Can supplementation with Lactobacillus reuteri and omega-3 fatty acids during pregnancy and lactation reduce the risk of allergic disease in infancy? |
| Study | Study Design, Country | Population | N | Age When Outcome Is Ascertained | Food Allergy Outcome Definition | Exposure | Question to Answer |
|---|---|---|---|---|---|---|---|
| Mis-BAIR (Melbourne Infant Study-BCG for Allergy and Infection Reduction) | RCT, Australia | Infants, younger than 10 days old | 1 year | SPT and challenge-proven food allergy | BCG immunization for TB versus no immunization | Does BCG immunization at birth, compared to no BCG immunization, lead to a reduction in measures of allergy and infection in the first 12 months of life? | |
| Molecular Basis of Food Allergy | Observational, US | Food allergic individuals ages 4 months to 75 years | Various | What is the molecular basis of food allergy? What are the genetic factors that lead to the development of food allergy? |
NOTE: AU = Australia; DBPCOFC = double-blind, placebo-controlled oral food challenge; FABP = fatty acid binding protein; GI = gastrointestinal; IgE = immunoglobulin E; sIgE = food-specific serum IgE; SPT = skin prick test; TB = tuberculosis; UK = United Kingdom; US = United States.
Genetics
The role of genetics in food allergies was initially supported by its familial aggregation (Tsai et al., 2009) and heritability estimates derived from twin studies (Liu et al., 2009; Sicherer et al., 2000). Later, the ability to explore the genome opened the possibility to examine the involvement of specific candidate genes. More recently the potential for discovery of new loci has expanded with the use of genome-wide association studies (GWASs)6 (Hong et al., 2015). However, unlike other diseases and phenotypes, for which hundreds of loci have been identified, the number of loci that have been tentatively associated with food allergies is still rather small.
As expected, most of these candidate genes encode products influencing immune mechanisms, including antigen presentation or a shift of the immune system toward a Th2 response. The hypothesis is that genetic predispositions may result in dysregulation of the immune system and, in the context of specific environmental factors, lead to food allergy. However, the association studies performed to date that have aimed to uncover the genetic architecture of food allergies have faced similar challenges as for other complex human diseases to date. Specifically, the identified loci can explain only a very small fraction of the phenotypic variance and few of the loci examined have provided conclusive and consistent findings across populations (see Table 5-4).
Only one GWAS has been reported in relation to food allergies (peanut, milk, and egg) (Hong et al., 2015). Two single nucleotide polymorphisms (SNPs) showed an association with peanut allergy that was above the GWAS threshold for significance, both of them in the human leucocyte antigen (HLA)7 system. The first one, rs7192, is in the HLA-DR region and the second one, rs9275596, is located in the HLA-DQ region. Most interesting, both loci are also associated with differential DNA methylation. Therefore, these results support the relevance of the HLA system as well as epigenetic modifications in the predisposition to peanut allergy. In this study, though, the food allergy outcome was defined based on a convincing history of clinical allergic reaction on ingestion of a specified food and evidence of
__________________
6 Genome-wide association studies (GWASs) examine many common genetic variants in different individuals to see if any variant is associated with a trait. GWASs typically focus on associations between single-nucleotide polymorphisms (SNPs) and traits like major diseases (Gottgens, 2012).
7 The human leukocyte antigen (HLA) system is a gene complex located in chromosome 6p21 that encodes the major histocompatibility complex (MHC) proteins, which are cell proteins responsible for the regulation of the immune system. MHC class I, II, and III have different functions. MCH class I present peptides from inside the cell, MHC class II present antigens from outside of the cell to T-lymphocytes and stimulate the multiplication of T-helper cells. MHC class III are components of the complement system.
TABLE 5-2 Current Guidelines on Food Allergy Prevention
| Guideline (reference) | Year | Country | Breastfeeding |
|---|---|---|---|
| Interim Guidance Regarding Peanut Introduction from the American Academy of Pediatrics; American Academy of Allergy, Asthma & Immunology; American College of Allergy, Asthma & Immunology; and othersa (Fleischer et al., 2015) | 2015 | US, Australia, Japan, European Union (EU) |
| Early Introduction of Foods | Infant Formula | Diet of Mother | Prebiotics or Probiotics |
|---|---|---|---|
| Introduce peanut-containing products into the diets of ‘‘high-risk’’ infants early on in life (between 4 and 11 months of age) in countries where peanut allergy is prevalent. Infants with early-onset atopic disease, such as severe eczema, or egg allergy in the first 4 to 6 months of life (LEAP criteria) might benefit from evaluation by an allergist or physician to diagnose any food allergy and assist in implementing these suggestions of early peanut introduction. |
| Guideline (reference) | Year | Country | Breastfeeding |
|---|---|---|---|
| NIAID/NIH-supported Guidelines (Boyce et al., 2010) | 2010 | US | Recommends that all infants be exclusively breastfed until 4 to 6 months of age, unless breastfeeding is contraindicated for medical reasons. |
| 2016 Addendum to the NIAID/NIH-supported Guidelines (Togias et al., 2017) | 2016 | US | |
| World Health Organization and World Allergy Organization (WHO, 2003) | 2003 | Worldwide | Breastfeed exclusively until 6 months. |
| Early Introduction of Foods | Infant Formula | Diet of Mother | Prebiotics or Probiotics |
|---|---|---|---|
| Does not recommend using soy infant formula instead of cow milk infant formul as a strategy for preventing the development of food allergy or modifying its clinical course in at-risk infants. Suggests that the use of hydrolyzed infant formulas, as opposed to cow milk formula, may be considered as a strategy for preventing the development of food allergy in at-risk infants who are not exclusively breastfed. |
Does not recommend restricting maternal diet during pregnancy or lactation as a strategy for preventing the development or clinical course of food allergy. | ||
| Suggests that the introduction of solid foods should not be delayed beyond 4 to 6 months of age. Potentially allergenic foods may be introduced at this time as well. | |||
| Infants with cow milk allergy should avoid cow milk proteins; if a supplement is needed, use hypoallergenic formula, if available, and affordable to improve symptom control. | No special diet for the lactating mother. |
| Guideline (reference) | Year | Country | Breastfeeding |
|---|---|---|---|
| American Academy of Allergy, Asthma & Immunology (Fleischer et al., 2013) | 2013 | US | Exclusive breastfeeding for at least 4 and up to 6 months is endorsed. |
| European Academy of Allergy & Clinical Immunology Guidelines (Muraro et al., 2014) | 2014 | EU | Exclusive breastfeeding for at least the first 4-6 months of life is recommended. |
| Early Introduction of Foods | Infant Formula | Diet of Mother | Prebiotics or Probiotics |
|---|---|---|---|
| Complementary foods can be introduced between 4 and 6 months of age. Highly allergenic foods can be given as complementary foods once a few complementary foods have been tolerated first and should initially be given at home first rather than at day care or a restaurant. | For high-risk infants who cannot be exclusively breastfed, hydrolyzed formula appears to offer advantages to prevent allergic disease and cow milk allergy. | Avoidance diets during pregnancy and lactation are not recommended at this time, but more research is necessary for peanut. This recommendation does not apply to infants who manifest signs of allergic disease shortly after birth, because treatment may, in some cases, involve dietary interventions during lactation. |
|
| Introduction of complementary foods after the age of 4 months according to normal standard weaning practices and nutrition recommendations, for all children irrespective of atopic heredity. | For high-risk infants: If a supplement is needed during the first 4 months, a documented hypoallergenic formula is recommended. | No special diet during pregnancy or for the lactating mother. |
| Guideline (reference) | Year | Country | Breastfeeding |
|---|---|---|---|
| European Society of Pediatric Allergy and Clinical Immunology and the European Society for Pediatric Gastroenterology, Hepatology, and Nutrition Committee on Nutrition (Agostoni et al., 2008) | 2008 | Europe | Exclusive or full breastfeeding for about 6 months is a desirable goal. |
| Early Introduction of Foods | Infant Formula | Diet of Mother | Prebiotics or Probiotics |
|---|---|---|---|
| There is no convincing scientific evidence that avoidance or delayed introduction of potentially allergenic foods, such as fish and eggs, reduces allergies, either in infants considered at increased risk for the development of allergy or in those not considered to be at increased risk. Complementary foods should not be introduced before 17 weeks and foods should be added one at a time to allow detection of reactions to individual components. It is prudent to avoid both early (<4 months) and late (>7 months) introduction of gluten and to introduce gluten gradually while the infant is still breastfed because this may reduce the risk of wheat allergy. |
| Guideline (reference) | Year | Country | Breastfeeding |
|---|---|---|---|
| American Academy of Pediatrics (Greer et al., 2008) | 2008 | US | For infants at high risk of developing atopic disease, evidence suggests that exclusive breastfeeding for at least 4 months compared with feeding intact cow milk protein formula decreases the cumulative incidence of atopic dermatitis and cow milk allergy in the first 2 years of life. |
| Early Introduction of Foods | Infant Formula | Diet of Mother | Prebiotics or Probiotics |
|---|---|---|---|
| Although solid foods should not be introduced before 4 to 6 months of age, there is no current convincing evidence that delaying their introduction beyond this period has a significant protective effect on the development of atopic disease regardless of whether infants are fed cow milk protein formula or human milk. This includes delaying the introduction of foods that are considered to be highly allergic, such as fish, eggs, and foods containing peanu protein. For infants after 4 to 6 months of age, there are insufficient data to support a protective effect of any dietary intervention for the development of atopic disease. |
In studies of infants at high risk of developing atopic disease who are not breastfed exclusively for 4 to 6 months or are formula fed, there is modest evidence that atopic dermatitis may be delayed or prevented by the use of extensively or partially hydrolyzed formulas, compared with cow milk formula, in early childhood. Extensively hydrolyzed formulas may be more effective than partially hydrolyzed in the prevention of atopic disease. There is no convincing evidence for the use of soy-based infant formula for the purpose of allergy prevention. |
Current evidence does not support a major role for maternal dietary restrictions during pregnancy or lactation. |
| Guideline (reference) | Year | Country | Breastfeeding |
|---|---|---|---|
| National Health Service (NHS, 2015b) | 2015 | UK | Breast milk or first infant formula for first 6 months. |
| National Health and Medical Research Council (NHMRC, 2013) | 2012 | Australia | Exclusive breastfeeding until around 6 months of age. For infants with a family history of allergy, continue breastfeeding while introducing solid foods. |
| Early Introduction of Foods | Infant Formula | Diet of Mother | Prebiotics or Probiotics |
|---|---|---|---|
| Introduce cow milk, eggs, wheat, gluten, nuts, peanuts, peanut products, seeds, fish and shellfish one at a time and not before 6 months. | Infant formula made from cow or goat milk is the only suitable alternative to breast milk in the first 12 months. Only use soy-based infant formula if advised by health care provider. Follow-on milks are available for babies older than 6 months, but there is no need to change over to these. If child has an allergy or intolerance to milk, health care provider can advise on suitable milk alternatives. |
||
| For infants with a family history of allergy, solid foods should be introduced at about 6 months of age. | If breastfeeding is discontinued for any reason, there is no advantage in using special formulas, except under medical supervision. Soy-based formulas do not prevent or reduce the risk of developing allergies and are not a suitable alternative to cow milk–based formulas. |
Dietary elimination of potential allergens during pregnancy is not recommended for preventing childhood allergy. | The evidence on probiotics or prebiotics in infant formula to prevent atopic disease varies. |
| Guideline (reference) | Year | Country | Breastfeeding |
|---|---|---|---|
| Australasian Society of Clinical Immunology and Allergy (ASCIA, 2016a,b) | 2016 | Australia | Breastfeeding is recommended for at least 6 months.b |
| Academy of Nutrition and Dietetics (AND, 2015) | 2015 | US | Exclusive breastfeeding provides optimal nutrition and health protection for the first 6 months of life and breastfeeding with complementary foods from 6 months until at least 12 months of age is the ideal feeding pattern for infants. Breastfeeding should be supported and preserved even under adverse or challenging conditions, such as prematurity, allergies, chronic illness, and multiple births. |
NOTE: UK = United Kingdom; US = United States.
a Australasian Society of Clinical Immunology and Allergy, Canadian Society of Allergy and Clinical Immunology, European Academy of Allergy & Clinical Immunology, Israel Association of Allergy and Clinical Immunology, Japanese Society for Allergology, Society for Pediatric Dermatology, and World Allergy Organization.
b For all infants (not as a prevention for allergic diseases).
| Early Introduction of Foods | Infant Formula | Diet of Mother | Prebiotics or Probiotics |
|---|---|---|---|
| Recommends the introduction of complementary “solid” foods within the window of 4-6 months and preferably while breastfeeding, regardless of whether the food is considered to be a common food allergen. | In children with confirmed cow milk and soy allergy, appropriate formula is available on prescription. There is no consistent convincing evidence to support a protective role for partially hydrolysed formulas or extensively hydrolyzed formulas for the prevention of food allergy in infants or children. |
Exclusion of any particular foods (including foods considered to be highly allergenic) from the maternal diet during pregnancy or breastfeeding is not recommended. | Recommendations about probiotic supplements cannot currently be made. |
TABLE 5-3 Evidence-Based Review Grading System Used by the Committee to Evaluate the Association Between Potential Risk Determinants and Food Allergies
| Strong | The conclusion statement is substantiated by a large, high quality, and/or consistent body of evidence that directly addresses the question. There is a high level of certainty that the conclusion is generalizable to the population of interest, and it is unlikely to change if new evidence emerges. |
| Moderate | The conclusion statement is substantiated by sufficient evidence, but the level of certainty is restricted by limitations in the evidence, such as the amount of evidence available, inconsistencies in findings, or methodological or generalizability concerns. If new evidence emerges, there could be modifications to the conclusion statement. |
| Limited | The conclusion statement is substantiated by insufficient evidence, and the level of certainty is seriously restricted by limitations in the evidence, such as the amount of evidence available, inconsistencies in findings, or methodological or generalizability concerns. If new evidence emerges, there could likely be modifications to the conclusion statement. |
| Grade not assignable | A conclusion statement cannot be drawn due to a lack of evidence, or the availability of evidence has serious methodological concerns. |
SOURCE: DGAC, 2015.
sensitization to the same food measured by sIgE and/or a positive skin prick test (SPT) to this specified food, not by OFC.
Overall, evidence exists of genetic predisposition for food allergy based on family aggregation (Tsai et al., 2009) and heritability studies (Liu et al., 2009; Sicherer et al., 2000), the latter showing a wide range of values between 0.15 and 0.88. However, as with other complex diseases that are polygenic, challenges remain to identify what contribute to the “missing heritability.”
The committee concludes that although some evidence from various lines of investigation suggests that genetics contribute to the development of food allergies, none of the studies on the association of food allergy with specific loci examined to date has provided conclusive and consistent findings across populations.
Interaction Between Genetics and Environment: Migration Studies
As mentioned above, environmental exposures, including lifestyle and diet, interact with genetic predisposition to modify the risk of disease. The
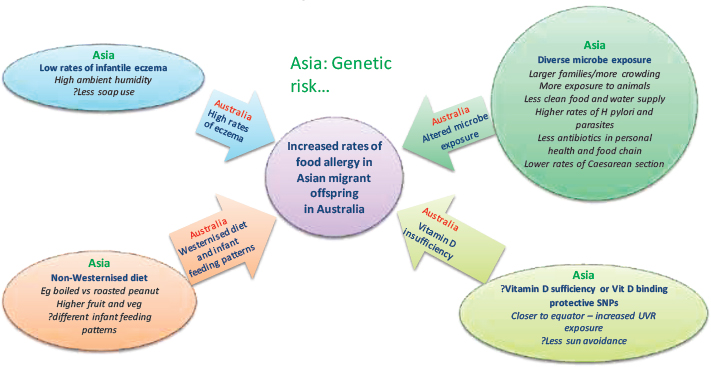
“natural experiment” of migration has provided an opportunity to postulate a possible protective effect of the Asian environment on Asian children that is removed on migration to a developed country such as the United States or Australia, where risk of allergic disease rises. In HealthNuts, challenge-confirmed peanut allergy was about three times more common in infants whose parents were born in East Asia compared to those with parents born in Australia (Koplin et al., 2014). Similar effects were seen for other food sensitizations and food allergies and for eczema. This increased risk appears to have occurred in a single generation and to be specific to infants of Asian parents. This effect was not seen among infants whose parents were born in the United Kingdom or Europe.
More recently in a large cohort of more than 65,000 children whose parents undertook a survey as their children entered primary school (age 5 years), the finding of increased rates of nut allergy in Asian children born in Australia was replicated. However the most intriguing observation in this study was the finding that children born in Asia who subsequently migrated to Australia before the age of 5 years were protected from the development of food allergy (Panjari et al., 2016).
Migration may be associated with changes to a number of factors (some of which were not measured in HealthNuts) that might be inter-related (Allen and Koplin, 2015) (see Figure 5-3). These include humidity (and its impact on skin barrier function), microbial exposure (hygiene hypothesis), and dietary changes and changes in latitude (vitamin D). For example, changes to the skin barrier function and risk of eczema as an early risk factor of food allergy may result from higher humidity in Asia than Australia
TABLE 5-4 Summary of Studies Associating Specific Genes with Food Allergy
| Author, Year | Study Design, Country | Population | N | Candidate Gene | Outcome | Summary |
|---|---|---|---|---|---|---|
| Senechal et al., 1999 | Observational, Europe | European-born white adults | 42 atopic 42 healthy |
HLA | Apple allergy | Association with the HLA-DRB1*07 allele |
| Hand et al., 2004 | Observational, UK |
Allergy clinic patients: ages 3-56 years; 81 white 3 mixed race 40 male 44 female Atopic controls: ages 16-61 years; |
84 nut-allergic patients
82 atopic non-nut-allergic patients 1,798 random blood donors |
HLA | Nut allergy (peanut, Brazil nut, hazelnut, walnut, cashew, almond, and pecan) | Increased for HLA-beta*07 and HLA-DRB1*11, HLA-DRB*13, and HLADQB1*06 alleles |
| Madore et al., 2013 | Observational, Canada |
Peanut-allergic Caucasian children, mean age 11 years
Controls: mean age 4 years |
590 cases
332 controls |
HLA | Peanut allergy | HLA-DQB1*02 and HLA-DQB1*06:03P associated with peanut allergy |
| Hong et al., 2015 | Observational, US | Participants in the Chicago Food Allergy Study | 1,315 children 1,444 parents |
HLA | Peanut allergy | HLA-DR and -DQ gene region at 6p21.32, tagged by rs7192 and rs9275596 |
| Woo et al., 2003 | Observational, US |
Food allergic patients, mean age 5.2 years, 74% male, 83% white
Non-atopic, non-asthmatic adult controls |
77 cases 61 controls |
CD14 | Food allergy | The C-159T SNP associated with food allergy |
| Campos et al., 2007 | Observational, Japan |
Food-allergic children, mean age 7.1 years
Non-food-allergic controls, mean age 9.45 years |
88 cases 101 controls |
CD14 | Food Allergy | No association with the C-159T or the C-550T |
| Torgerson et al., 2007 | Case series, France | Index case with IPEX syndrome and other family members | 11 | FOXP3 | Severe food allergy | 1300-bp deletion could cause severe food allergy |
| Siegel et al., 2013 | Observational, US |
Atopic patients: 40% female, mean age 14.8 years
Controls: 61% female, mean age 34.5 years |
65 patients with severe atopic disease
41 healthy controls |
STAT3 | Food allergies (egg, milk, or peanut) | Complex association between this locus and allergic phenotypes |
| Author, Year | Study Design, Country | Population | N | Candidate Gene | Outcome | Summary |
|---|---|---|---|---|---|---|
| Amoli et al., 2002 | Observational, UK |
Nut-allergic, Caucasian patients, mean age 10 years
Healthy atopic, non-allergic controls UK Caucasian blood donors |
71 patients 45 controls 184 blood donors |
STAT6 | Nut-allergy (peanut, cashew, Brazil nut, pecan, almond, hazelnut, or walnut) | The G allele at the G2964A SNP increased in nut-allergic patients. |
| Negoro et al., 2006 | Observational, Japan | Allergic children | 220 | STAT6 | Food allergy | No association of G2964A and severity of food allergy |
| Kusunoki et al., 2005 | Observational, Japan | Children with atopic dermatitis, >5 years of age | 118 | SPINK5 | Food allergy | The 1258AA or 1258AG carriers have higher prevalence of food allergy |
| Negoro et al., 2006 | Observational, Japan | Allergic children, mean age 7.3 years | 220 | IL10 | Food allergy | No association with the C-627A SNP |
| Campos et al., 2008 | Observational, Japan |
Food-allergic children, mean age 7.6 years, 63% male
Atopic control children without food allergy, mean age 8.2 years, 64% male |
111 cases 115 controls |
IL10 | Food allergy | No association with the C-627A SNP; but the -1082AA genotype was associated with higher risk |
| Chen et al., 2012 | Observational, Taiwan |
Food-allergic patients, age range 1-32 years; 62% male
Non-food-allergic controls, age range 1-59 years, 40% male |
37 cases 52 controls |
IL10 | Food allergy | Both the -1082A/G and the -592A/C SNPs were associated with food allergies |
| Liu et al., 2004 | Observational, Germany | German children who participated in the German Multicenter Allergy Study | 823 | IL13 | Food sensitization | C-1055T higher risk |
| Gaudieri et al., 2012 | Observational, Australia | Children recruited antenatally from healthy pregnant mothers; followed from birth to age 5 years | 35 allergic 35 non-allergic |
IL28B | Food allergy | The rs12979860 SNP associated positively with food allergy |
| Venkataraman et al., 2014 | Observational, UK | Isle of Wight birth cohort; children ages 1-18 years | 1,456 | FLG | Food allergy | FLG LOF mutations associated with food allergy. |
| Tan et al., 2012a | Observational, Australia | HealthNuts Cohort study participants; white infants, age 1 year | 700 | FLG | Food sensitization/allergy | FLG LOF mutations do not increase the risk of food allergies beyond that of food sensitization |
| Author, Year | Study Design, Country | Population | N | Candidate Gene | Outcome | Summary |
|---|---|---|---|---|---|---|
| Brough et al., 2014 | Observational, UK | Birth cohort of the Manchester Asthma and Allergy Study, children ages 1-11 years | 1,184 | FLG | Peanut allergy | Positive association with peanut allergy |
| Li et al., 2012 | Observational, China | Atopic dermatitis outpatients, mean age 3.5 years, 64.3% male | 249 | FLG | Food sensitization | Interaction of K4671X mutation and the combined mutations in FLG related to sensitization to peanut allergens in patients with atopic dermatitis |
| Oxelius et al., 2015 | Observational, Germany | Children from the German Multicenter Allergy Study, Caucasian, age 1 year or 10 years | 194 | IGHG genes | Food sensitization | The IGHG*bfn haplotype (B*bfn cells) and increased innate IgG2*n levels are predictive factors for IgE food sensitization in childhood |
NOTE: FLG = filaggrin; HLA = human leukocyte antigen; IGHG = immunoglobulin heavy locus gene; IL = interleukin; LOF = loss of function; UK = United Kingdom; US = United States.
NOTE: SNP = single nucleotide polymorphism; UVR = ultraviolet radiation.
SOURCE: Allen and Koplin, 2015. Reprinted with permission from Elsevier.
but equally may result from differences in infant washing practices (types of soap and water composition) that occur in each country and may exert an effect through the hygiene hypothesis. Microbial exposure factors that differ not only include variations in the quality of water supply (and differences in risk of waterborne gastrointestinal infections) but also differences in microbes that are a part of the food chain supply (for example, in unwashed vegetables or higher use of antibiotics in the food chain supply of meat-producing animals), number of children in a family, and issues of crowding and exposure to pets, farm animals, and stray animals (which may have higher rates of parasites), and variations in overprescribing of antibiotics in each region. Dietary differences are multiple (e.g., higher use of herbicides and pesticides that might affect the microbial load of food and increased sterilization; use of plastic in developed countries; cooking practices that may alter the allergenicity of food; different vitamin D status).
Epigenetics
The contribution of epigenetics has been more extensively studied for other allergic diseases, including asthma, eczema, and allergic rhinitis, as reviewed by Hong and Wang (2014), than for food allergies. In light of the
atopic march and common comorbidities between food allergies and these other allergic diseases, one may speculate that a link between epigenetic changes and the development of food allergies is possible, but at this time the evidence is quite limited and comes from indirect studies such as the migration studies described above.
Direct evidence to establish the relevance of epigenetic changes as a mediator of genetic susceptibility to food allergies is very limited. The most up-to-date knowledge about the role of epigenetics in food allergy has been summarized in a recent review by Neeland et al. (2015). In their epigenome-wide association study (EWAS) of food allergies,8 Martino et al. measured genome-wide DNA methylation profiles from CD4+ T-cells (see Chapter 2) on a birth cohort of 12 children with IgE-mediated food allergy diagnosed at 12 months; 12 individuals with no food allergies were controls (Martino et al., 2014). A number of statistically significant differentially methylated probes (DMPs) were identified from DNA obtained from samples taken at birth and at 12 months of age. Of interest is the finding of 96 allergy-associated non-SNP DMPs that were present at time of birth before the expression of the disease. These could be causally related to its expression, including several mitogen-activated protein kinase (MAPK) signaling molecules. Therefore, the authors concluded that “dysregulation of DNA methylation at MAPK signaling-associated genes during early CD4+ T-cell development may contribute to suboptimal T-lymphocyte responses in early childhood” that could influence the development of food allergy (Martino et al., 2014). However, this is a small study and, therefore, its findings need independent validation in larger studies and other populations.
Support for a role of epigenetics in food allergies is provided by the results from a food allergy GWAS carried out in 2,759 U.S. participants (1,315 children and 1,444 parents) from the Chicago Food Allergy Study (Hong et al., 2015). In a recent study in the Chicago cohort, Hong et al. conducted an EWAS of cow milk allergy using a two-stage approach (Hong et al., 2016). During the discovery stage, DNA methylation was measured at 485,512 genomic loci in whole blood samples from 106 Caucasian children with cow milk allergy (cases) and 76 nonallergic and nonatopic Caucasian children (controls) using the Illumina HumanMethylation450 arrays. The findings were confirmed in a small replication sample (5 cases and 20 controls). The researchers demonstrated that altered DNA methylation in genes involved in the Th1-Th2 pathways and some novel candidate genes are associated with cow milk allergy.
__________________
8 Epigenome-wide association studies (EWASs) are large-scale, systematic studies that explore the association between the epigenetic variations and diseases, equivalent to genome-wide association studies (GWASs).
The committee concludes that relative to other immune-related diseases, only a few studies have been conducted that directly support a contribution of epigenetic factors to the development of food allergies. Limited evidence from ecological studies and studies on methylation signatures of participants with food allergies suggest that gene-environment interactions and underlying epigenetic mechanisms need to be taken into account when exploring potential pre- and postnatal risk factors for food allergy.
ENVIRONMENTAL RISK FACTORS
Microbial Exposure Hypotheses
As mentioned in Box 5-1, evidence increasingly suggests that the interaction between the host microbiome and the immune system is essential to the development of immune regulation and oral tolerance (Martin et al., 2010). Exposure to microbes after birth prompts the maturation of the mucosal immune system (Kelly et al., 2007). The composition and timing of exposure to gut microbiota, and their possible role in disease development or prevention have been considered as explanations for the development of food allergy (Li et al., 2014; McLoughlin and Mills, 2011; Prince et al., 2015). The microbial hypothesis proposes that a decrease in early childhood exposure to microbes or their products may hinder the normal development of early immunoregulatory responses. This leaves the immune system more susceptible to inappropriate reactivity to innocuous antigens, resulting in the development of “allergic” diseases.
The overall microbial hypothesis encompasses two different concepts—the “Hygiene Hypothesis” and the “Old Friends Hypothesis.” The Hygiene Hypothesis, originally explained in the landmark paper by David Strachan in 1989, described a protective effect of an increasing number of siblings in a household on the risk of developing allergic rhinitis (Strachan, 1989). This was thought to potentially relate to the shared exposure to common childhood infections transmitted through direct contact with older siblings or by maternal contact with her older children prenatally. Although a protective sibling effect has been confirmed for challenge-proven food allergy outcomes (Koplin et al., 2012a) and for various food sensitization and allergy outcomes (Marrs et al., 2013), the mechanism(s) underlying this phenomenon is not clear. Although the finding is interesting and reproducible, changes to postwar houses and sanitation, and sizes of families, as well as the emergence of national immunization programs with high uptakes, also should be considered in attempting to identify the mechanisms underlying the protective effects of siblings.
Second, evidence of a protective effect of dog ownership on food allergy risk may point to the benefit of sharing of microbes or even parasites, the
latter underpinning the idea of the Old Friends hypothesis. Although this hypothesis was predicated on the assumption that IgE antibody–associated immune responses developed in part as a mechanism of host defense against parasite infestation, recent evidence indicates that, at least in mice, IgE antibody–associated immune response also can confer increased acquired resistance to the morbidity and mortality induced by arthropod and reptile venoms (Galli et al., 2016; Marichal et al., 2013; Palm et al., 2013; Starkl et al., 2016). Speaking more broadly, early evidence suggests a difference between the prevalence of food allergy in rural versus urban environments that appears to be reflected in rising rates of food allergy described in cities in China undergoing rapid urbanization (Hu et. al., 2010).
The main environmental factors contributing to the microbial exposure hypothesis include route of delivery at birth, antibiotic use, exposure to pets/animals, and immunization. Breastfeeding has been linked to infant immune development (Praveen et al., 2015) and the composition of the microbiota (Azad et al., 2016). It would therefore be plausible that a mechanism linking food allergy risk and breastfeeding could be mediated through microbiome modulation (Fooladi et al., 2013). However, no published studies to date have investigated this hypothesis, and the data linking breastfeeding and food allergy are inconclusive, potentially due to reverse causality and the inability to randomize infants for breast- versus formula-feeding. Therefore breastfeeding will not be included in this section; instead the effect of breastfeeding is included as part of the “Allergen Avoidance Hypothesis” (see p. 185). The ingestion of prebiotics and probiotics could modify the gut microbiota in a way to change immune system functionality and atopic diseases. Therefore, their use as potential risk factor for food allergies also is included.
A systematic review of the evidence linking microbial exposure and food allergy was published by Marrs et al. (Marrs et al., 2013). The authors reviewed scientific publications available in Medline between 1948 and July 2012. The key findings of this review will be briefly summarized below, but the focus will be on reviewing the findings of papers published since July 2012.
Gut Microbiota and the Use of Probiotics and Prebiotics
Data on microbial profiling and its relationship to disease are still not sufficiently detailed to consider specific microbiota modifications as a food allergy prevention strategy. However, some emerging data suggest that changes in microbiota could influence food allergies, offering further support for the microbial exposure hypothesis (West et al., 2015).
Marrs et al. included five studies in their systematic review that investigated characteristics of gut microbiota, two of which used food challenge
outcomes and three that used food sensitization parameters (Marrs et al., 2013). The two manuscripts that ranked highest in quality and measured food allergy were from the same study of Spanish infants who were diagnosed with IgE-mediated cow milk allergy by milk challenge at a tertiary referral center. Differences in microbiota were identified but unfortunately none of the results was adjusted for diet. The Marrs review also included 11 RCTs in which microbial supplementation was the intervention as a potential prevention or treatment of food allergies or sensitization. Although the quality varied, the two highest quality studies that measured food allergy by OFC to assess whether microbial supplementation may be used to prevent or treat food allergies or sensitization found no benefit.
More recent data originate from the Canadian Synergy in Microbiota (SyMBIOTA) study, part of a larger Canadian research effort on the microbiota. This large 6-year longitudinal study is using metadata and samples from the Canadian Healthy Infant Longitudinal cohort to discern relationships between infant fecal microbiota and each of a group of factors, including antibiotic use, pets, and food sensitization (Kozyrskyj, 2015). Their data suggest that lower species richness in microbiota of infants (N=166, ages 3 and 12 months) might be a predictor of food (i.e., for egg, milk, and peanut) sensitization (SPT at age 12 months), even when adjusting for birth delivery mode, antibiotic use, or breastfeeding (Azad et al., 2015). Their research also revealed that sensitization occurred after the changes in microbiota diversity and richness, two commonly used indexes. Therefore, this ratio could potentially be used as a predictor of food sensitization, a potential surrogate for food allergies. Each quartile increase in richness at 3 months was associated with a 55 percent reduction in risk for food sensitization by 1 year (adjusted odds ratio [aOR] 0.45; 95% confidence interval [CI]: 0.23-0.87).
One meta-analysis of 10 RCTs (Kong et al., 2014) reported no significant difference in the incidence of food allergies comparing prenatal and postnatal probiotics supplementation with placebo or control. However, the food allergy assessments were not described in the meta-analysis. The World Allergy Organization (WAO) has recently conducted a systematic review on the relationship between supplementing the diet of pregnant or lactating women or infants with probiotics and allergy diseases. Six trials explored the relationship with food allergies but none of them made the direct comparison of probiotics versus no probiotics in pregnant women or in breastfeeding women for prevention of allergy in their children. None of the trials found differences in food allergy with probiotic supplementation (Cuello-Garcia et al., 2015). Two additional observational studies found during the committees’ evidence-based search did not find an association between the addition of probiotics to infants’ diets (Loo et al., 2014; West et al., 2013). The most recent work on the effect of prebiotics in
food allergy, also conducted by the WAO (Cuello-Garcia et al., 2016), is a guideline that seems to be based on a systematic review. The methods of systematic review, however, were not fully reported and no other source or citation to the systematic review was found. The guideline is based on studies investigating the relationship between prebiotics consumption by women during pregnancy or lactation and by healthy infants for preventing various allergic symptoms, including food allergy. Only one intervention study assessed the risk of developing food allergy in infants consuming an infant formula containing oligosacharides (Ivakhnenko and Nyankovskyy, 2013). That study (N=240) found that infants who had been fed with breast milk or oligosaccharide-supplemented infant formula had significantly fewer allergic reactions to food products compared to the infants fed the standard formula (3.92 percent and 4.84 percent versus 16.98 percent, respectively; P<0.05).
The committee concludes that, at this time, only a few studies have been conducted on the relationship between changes in the microbiota and food sensitization and, therefore, the evidence supporting this relationship is limited. RCTs on probiotic and prebiotics supplementation are few and have methodological limitations. Therefore, the committee concludes that the evidence is limited and does not yet support a decrease in food allergy risk from the use of probiotics or prebiotics by pregnant and lactating women or by infants. Additional research would be needed before recommending the use of prebiotics or probiotics to prevent the onset of food allergies.
Route of Delivery
The composition of the gut microbiota is influenced by route of delivery. Vaginally-delivered infants harbor bacterial communities resembling their mother’s vaginal microbiota. In contrast, infants delivered by cesarean section have bacterial communities similar to those found on the skin surface (Dominguez-Bello et al., 2010). In light of the fact that the gut microbiome plays a central role in the development of immune regulation and oral tolerance, it is not surprising that investigators have examined the question of whether caesarean delivery increased the risk of food allergy.
In their systematic review, Marrs et al. identified 13 publications. Of these, five identified food allergy through OFCs. All 13 publications, except for the study of lowest quality, reported an increased risk of developing food allergy or food sensitization in children delivered by cesarean section (Marrs et al., 2013). Six of these associations were significant. However, only two included clinical food allergy diagnoses. Of the studies included for review, these two studies yielded the highest quality data. The studies used 2,803 consecutive mother-infant pairs from a Norwegian birth
cohort surveyed at 12, 18, and 24 months. When children were challenged with food orally using open or double-blind protocols, cesarean section was associated with a significantly higher risk for cow milk allergy. This occurred only in the subgroup of children with atopic mothers, however (aOR: 9.6 [95% CI: 1.8-52.4]) (Eggesbo et al., 2005). They also observed a nonsignificant 60 percent increase in egg allergy risk up to age 2 years (Eggesbo et al., 2003).
The Marrs review also included a prospective nested case-control study of 16,237 infants in Finland, ages 0 to 2 years (Metsala et al., 2010). Infants whose parents had received a reimbursement for the cost of specialized formula based on diagnosis of cow milk allergy were recruited, and the allergy was certified by a pediatrician using clinical exam, symptoms, elimination diet, SPT, and elevated sIgE or open challenge test (Metsala et al., 2010). Controls were randomly selected infants who were matched for age, sex, and delivery hospital. A significant relationship between cesarean delivery and cow milk allergy was observed (aOR: 1.18; 95% CI: 1.10-1.27).
Lodge et al. conducted a more recent review of systematic reviews and found two systematic reviews that included six original studies (Lodge et al., 2013). An association between cesarean section delivery and increase in food allergy is seen in only the three smallest studies. Two of these studies used specific IgE to food allergens as the outcome measurement. No conclusion was reached by the authors due to methodological flaws (i.e., small size studies or inaccurate food allergy measurement).
Since the Marrs’ systematic review, six prospective cohort studies investigating associations between cesarean delivery and allergy risk have been published. They include studies conducted in Australia (Peters et al., 2014), France (Pele et al., 2013), the United Kingdom (Grimshaw et al., 2014), the United States (Luccioli et al., 2014; McGowan et al., 2015), and a five-country study (Depner et al., 2013) totaling 25,688 cases and controls. Overall, these studies found no significant associations between cesarean delivery and a variety of food allergies. The age of the children in the studies ranged from 0 to 5 years, and most included physician-diagnosed food allergy. Minimum criteria for diagnosis were sIgE to food allergen or a positive SPT. However, Luccioli et al. used physician diagnosis based on parental report (Luccioli et al., 2014). The largest study was the Australian HealthNuts Study (Peters et al., 2014), which recruited 5,276 infants at immunization clinics. These infants (2,848 of the total recruited) were investigated for open challenge-proven egg, peanut, and sesame allergy. However, no significant association was demonstrated with mode of delivery (Peters et al., 2014). Two retrospective case-control studies from Finland (N=3,181) (Pyrhonen et al., 2013) and the United States (N=291) (Dowhower Karpa et al., 2012) also did not show an association between cesarean delivery and food allergy.
The variation in association between mode of delivery and risk of food allergy may be partly explained by the fact that some studies have been unable to distinguish between whether cesarean delivery had been done on an elective or emergency basis (e.g., Koplin et al., 2012a; Peters et al., 2014). Emergency cesarean delivery is generally associated with rupture of membranes. As a result, the baby has some exposure to vaginal commensal bacteria during labor. However, the exposure is not usually to the same extent as vaginal delivery. However, because the proportion of emergency cesarean deliveries is usually relatively small compared to elective cesarean deliveries, we would still expect to see some association between mode of delivery and food allergy. This would be true even in those studies that could not differentiate emergency from elective cesarean deliveries, particularly in the larger and better powered studies. It also should be noted that the association between cesarean delivery and allergic risk could be misinterpreted due to the potential for reverse causation similar to breastfeeding.
Only a few observational studies have been conducted on the relationship between food sensitization or food allergy and cesarean delivery. The studies have methodological limitations. Therefore, the committee concludes that, at this time, evidence to support an increased risk for food sensitization or food allergy due to giving birth by cesarean delivery is limited. Strong evidence is unlikely to be forthcoming because of the ethical inability to randomize a population to deliver a baby by cesarean section. However, additional prospective research studies are needed.
Antibiotic Use
Antibiotics are known to cause short-term and, in some cases, lasting alterations in the microbiota (Faa et al., 2013). Infants can be exposed to antibiotics pre-, peri-, or postnatally as individual exposures or multiple exposures across this time, when the microbiome is not well established and is more susceptible to perturbations. The Marrs et al. systematic review reported no relationship between antenatal or postnatal antibiotic exposure and increased risk of food allergy (Marrs et al., 2013).
Since 2012, two prospective cohort studies of food allergic children have been published that were not included in the Marrs systematic review (Marrs et al., 2013). Studies in Finland (Metsala et al., 2013) and the United Kingdom (Grimshaw et al., 2014) and one retrospective case control study from the United States (Dowhower-Karpa et al., 2012) investigated associations between antibiotic exposure and food allergy risk. In those infants whose mother used antibiotics before or during pregnancy, respectively, the Finnish prospective, nested case-control study (N=16,237) reported a statistically significant 26 percent (aOR: 1.26; 95% CI: 1.20-1.33) and 21
percent (aOR: 1.21; 95% CI: 1.14-1.28) increased risk for cow milk allergy (determined by OFC) (Metsala et al., 2013). An even greater risk of cow milk allergy (aOR: 1.71; 95% CI: 1.59-1.84) was reported in infants who were treated with antibiotics between birth and 1 month of age (Metsala et al., 2013).
However, two other studies described below showed no statistically significant association. Cases (N=41) and controls (N=82) in the UK study were drawn from the Prevalence of Infant Food Allergy (PIFA) study (Grimshaw et al., 2014). Children in this study were part of the larger EuroPrevall birth cohort. Food allergy was diagnosed using SPT, physical exam, clinical history, sIgE, and DBPCOFC. Maternal antibiotic use during or after pregnancy or during breastfeeding was not associated with increased risk of food allergy in the infant. However, administration of the antibiotic to the infant was not assessed (Grimshaw et al., 2014). In a retrospective case (N=99) control (N=192) design, Dowhower Karpa et al. found no association between peripartum or neonatal antibiotic exposure and food allergy, diagnosed by positive sIgE or SPT (Dowhower Karpa et al., 2012).
Thus, taking together the results of the Marrs systematic review (Marrs et al., 2013) and the three studies published since, only one study (Metsala et al., 2013) has reported a link between antibiotic use and food allergy. The strengths of that study is the large sample size (more than 16,000 children) and the prospective design. However, additional studies are needed to conclusively demonstrate a link between antibiotic use in early life and food allergy risk.
Only a few studies have explored the relationship between food allergies and antibiotic use. The committee concludes that evidence from observational studies suggesting a link between antibiotic use in early life and food allergies is limited. Additional studies with information on the type and dose of antibiotic, the timing of exposure along the perinatal continuum, and whether the infant is repeatedly exposed are needed to conclusively demonstrate a link with food allergies.
Animal Exposure
As noted above, the premise of the “Hygiene” and “Old Friends” hypotheses is based on the concept that the lack of early childhood exposure to infectious agents, symbiotic microorganisms, and/or parasites increases susceptibility to allergic diseases and asthma by suppressing the natural development of the immune system (Strachan, 1989).
The Marrs review reported on four studies investigating associations between farm and animal exposure and food allergy (Marrs et al., 2013). In their review, only the HealthNuts Study supported the microbial hypothesis. The study reported data on risk of pets and siblings for the develop-
ment of challenge-proven egg allergy (Koplin et al., 2012a). It also assessed the role of these factors on any food allergy using latent class analysis, a sophisticated analytical epidemiological method (Peters et al., 2015). Marrs et al. also reported findings from the European Protection against Allergy Study in Rural Environments (PASTURE), which described a cohort of families living in proximity to farm animals in rural settings (Marrs et al., 2013). This study showed significantly less food sensitization in the cord blood of mothers who consumed raw cow milk (versus boiled milk) in the perinatal period. However, the authors applied a lower cutoff for sIgE concentration than is conventionally used (>0.2 versus 0.35 IU/ml), which may have overestimated the incidence of food sensitization (Ege et al., 2008).
Since 2012, several prospective cohort studies have investigated whether exposure to farm animals (Depner et al., 2013; Pele et al., 2013) or pets (Goldberg al., 2013; Grimshaw et al., 2014; Martin et al., 2015; Peters et al., 2015; Stelmach et al., 2014) influenced the risk of food allergy or food sensitization. Depner et al. performed an additional analysis of data from 686 children in the rural European PASTURE cohort (Depner et al., 2013). Again using sIgE as their diagnostic criterion for food sensitization, they explored the more traditionally used sIgE cutoff of 0.35 IU/ml compared to 0.2 IU/ml in their previous study by Ege et al. (2008). They found that allergen-specific IgE levels rarely exceeded 0.35 IU/mL (<3% of all children) at age 1 year and the 95th percentiles at 1 year were consistently less than 0.7 IU/mL (RAST class 2) for any IgE. The only exception was cat (1.3 IU/mL) (Depner et al., 2013). They also found that early life exposure to farm animals, such as sheep, goats, and rabbits, did not confer protection against food allergen sensitization. However, exposure to farming increased (P=0.0015) the risk of food allergen sensitization (aOR: 2.11; 95% CI: 1.33-3.34). A total of 793 (378 farm and 415 nonfarm) children were included in the analyses. Pele et al. also reported no effect of farm animal contact on food allergy incidence in more than 1,400 children participating in the PELAGIE mother–child cohort. However, mold or dampness in the home increased (P≤0.001) the incidence of food allergy (23.9% versus 8.8%, yes versus no) in this cohort, as measured by parent report (Pele et al., 2013).
All other prospective cohort studies published since 2012 investigated exposure to pets. Two studies with a total of 350 children reported no association between pets in the home (Israel) and food sensitization (measured by specific IgE to cow milk) (Goldberg et al., 2013) nor an association of pet ownership (United Kingdom) with food allergy risk (measured by DBPCOFC or convincing history of anaphylaxis) (Grimshaw et al., 2014). In contrast, Stelmach et al. reported an increased risk of food allergy based on diagnosis by a doctor following international guidelines (aOR: 1.48; 95% CI: 1.02-2.16) associated with pets in the home during pregnancy in
a cohort of 501 children from the Polish Mother and Child Cohort Study (REPRO_PL cohort) (Stelmach et al., 2014).
Two studies from the HealthNuts cohort, a prospective, population-based cohort of 5,276 infants age 12 months in Melbourne, Australia, investigated whether direct exposure to pets (Koplin et al., 2012a; Peters et al., 2015) or the co-incidence of eczema (Martin et al., 2015) moderated the effect of pets on food allergy risk. Koplin et al. examined the relationship between environmental and demographic factors and egg allergy, the most common food allergy in infants and young children (Koplin et al., 2012a). Using SPT to egg white and oral food challenge at 12 months revealed that children with a pet dog at home (dog ownership ascertained by questionnaire) were less likely to develop egg allergy than those without a pet dog at home (aOR: 0.72; 95% CI: 0.52-0.99). Peters et al. observed that, compared to not having a dog in the home, having a dog significantly reduced the risk of multiple food allergies (including peanut) by 60 percent (aOR: 0.4; 95% CI: 0.21-0.73), whereas having a dog that was kept outside only (versus no dog) provided no protection. In this latter scenario, a significantly increased risk in egg allergy was actually observed (aOR: 1.56; 95% CI: 1.1-2.21) (Peters et al., 2015). Within the same cohort, Martin et al. compared the effect of dog or cat exposure on infants with (N=2,795) or without (N=1,903) eczema (Martin et al., 2015). Having a dog reduced the risk of food allergy in infants with eczema (aOR: 0.7; 95% CI: 0.5-0.9), but not in infants without eczema. A similar effect on food allergies was observed for infants with (aOR: 0.6; 95% CI: 0.4-0.9) or without eczema in homes with cats (Martin et al., 2015).
Results from studies exploring the relationship between animal exposures and food allergies are inconsistent. The few observational studies related to living on a farm found that exposure to farm animals offers no protection against food allergies. Also, from observational studies, the committee concludes that evidence is limited regarding the potential for a close interaction with a pet being more protective against a food allergy than pet ownership in general or having a pet who is restricted to outside the home. Further studies should be conducted on the nature of the association between exposure to farm animals or pet ownership and food allergies.
Allergen Avoidance Hypothesis
As mentioned in the introduction of this chapter, in considering the risk determinants for developing food allergies, the committee focused on the prenatal and early childhood developmental periods. In that vein, this section focuses on allergen exposure beginning at conception. The allergen avoidance hypothesis was predicated on the basis of the concept that
avoiding common food allergens early in life when the immune system is developing would prevent the onset of food allergies.
Exposure to Antigen Through Maternal Diet During Pregnancy or Lactation
Maternal diet during pregnancy and lactation has been of great interest in understanding the etiology of food allergies in offspring. The fetal programming hypothesis supports the idea that the maternal diet has long-term influence on children’s health (Barker, 1990; Langley-Evans, 1997). Its application to food allergies would suggest that consuming specific allergenic foods during this critical period might be associated with the development of the immune system in utero that may later manifest itself as food allergies over the life course, given specific childhood exposures. Results from two prospective cohort studies (Bunyavanich et al., 2014; Frazier et al., 2014) (total N=9,482 mother–child pairs) show that a higher consumption of allergenic foods before or during pregnancy (e.g., peanut), as measured by a food frequency questionnaire, was associated with a reduced risk of having a child with food allergies. This finding supports the fetal programming hypothesis. The HealthNuts Study also assessed the role of allergen avoidance in pregnancy and lactation and the risk of challenge-proven egg allergy and found no association (Koplin et al., 2010). Another recent prospective cohort study (Pele et al., 2013) reported an association between maternal pre-pregnancy consumption of shellfish and food allergy (1.62; 95% CI: 1.11-2.37). However, this study assessed food allergy by parental report. Randomized studies on this subject have involved the elimination of certain allergenic foods as opposed to increasing their consumption among primarily high-risk families. Kramer and Kakuma conducted a high-quality systematic review that included three RCTs of foods avoided during pregnancy and/or lactation and the outcomes of egg and milk sensitization (but not food allergy itself) among women at high risk of having an atopic offspring (Kramer and Kakuma, 2012). In two of the RCTs (Falth-Mangnusson and Kjellman, 1987; Lilja et al., 1988) (total N=334), women either avoided or decreased their intake of cow milk and eggs beginning in the third trimester of pregnancy and this was associated with a nonsignificant reduction of egg sensitization in their infants at 6 months, but not at 18 months. Sensitization for cow milk allergy was not reduced at either time point. The remaining RCT (Appelt et al., 2004) (total N=497) had women totally avoid peanuts, nuts, and fish as well as decrease their intake of cow milk and eggs beginning in the third trimester through 1 year postpartum. This study found no significant associations with milk or peanut sensitization in offspring at age 1, 2, or 7 years. However, for egg sensitization, an increased risk was seen at age 2 years only (1.91; 95% CI:
1.03-3.5). This trial is published in abstract form only, with no details on the randomization being available.
Another recent systematic review by de Silva et al. found seven high-quality studies on maternal diets and also concluded that “overall, the evidence is not strong enough to recommend changing the diet or supplements of pregnant or breastfeeding women” to prevent food allergies in infants at normal or high risk of food allergies (de Silva et al., 2014).
The committee concludes that, to date, study findings provide limited evidence to support or discourage eliminating allergenic foods from the diet of pregnant or lactating women at high risk of having a child with allergies. Because the evidence about the benefits of consuming or eliminating allergenic foods during pregnancy and lactation is not clear, additional RCTs are warranted before providing advice in this regard. Studies exploring the effect on the development of food allergies in children of intake of allergens by the mother are in progress.
Breastfeeding
Breastfeeding is an important early life factor that determines an individual’s gut microbiota and likely indirectly modulates immune responses. In addition, breastfeeding transfers bioactive compounds from the mother to the child that can also influence immune responses. However, the evidence assessing any potential link between breastfeeding and food allergies risk is not clear. Systematic analysis of observational studies on the protective effect of breastfeeding have shown conflicting results, and many of the studies included were conducted decades ago when food allergy was uncommon and methods of assessment were limited (Grimshaw et al., 2009). Most systematic reviews have failed to find a specific beneficial effect of breastfeeding on food allergy or food sensitization (de Silva et al., 2014; Kramer and Kakuma, 2012). Moreover, two cohort studies reviewed in de Silva et al. (2014) suggested that exclusive breastfeeding for 8 weeks did not reduce the risk of cow milk allergy (measured by parents report followed by SPT and oral food challenge) (Saarinen et al., 1999) and breastfeeding for 5 months or more may increase the likelihood of sensitization to egg in infants at high risk of atopy, although food allergy was not assessed (Wetzig et al., 2000). Importantly, the apparent negative effects of extensive breastfeeding may relate to the delayed introduction of first complementary foods rather than the effects of breast milk per se (see the section “Dual Allergen Exposure Hypotheses” in this chapter). Alternatively, these recent findings of increased risk of breastfeeding may simply be a misinterpretation of the data related to the reverse causation phenomenon (see the section “Methodological Limitations” in this chapter). One study found that the effects
of breastfeeding on food sensitization can be modified by genetic variants relevant to allergic diseases (Hong et al., 2011).
Lodge et al. undertook a systematic review to assess the role of breastfeeding in food allergy (Lodge et al., 2015). The review included nine cohort and four cross-sectional studies. The numbers of participants ranged from 163 to 21,766 (cohort studies) and from 1,278 to 13,110 (cross-sectional studies). No association with food allergy was found for more versus less9 breastfeeding in the pooled estimate (6 cohort and 6 cross-sectional), although study heterogeneity was high. Various sub-analyses failed to find any protective association of breastfeeding for food allergy. The primary issue concerning the quality of these studies was the poor accuracy of outcome assessment. Only two studies used OFCs, the recognized gold standard for food allergy diagnosis; most studies relied on parental report of symptoms or on physician diagnosis.
The committee’s review of the evidence found eight studies (seven cohort and one cross-sectional) that explored breastfeeding as a food allergy risk determinant. Although Ivakhnenko and Nyankovskyy showed that infants (N=240) who were breastfed had significant risk of developing an allergy to cow milk protein and had gastrointestinal symptoms of food allergy by age 18 months compared with those who were fed standard infant formula, the risk of bias of this trial was high due to unclear definitions and diagnoses of food allergy outcomes and high dropout rates (Ivakhnenko and Nyankovskyy, 2013). Two studies performed only unadjusted analyses so the results (mostly no significant associations) are likely to be confounded (Grimshaw et al., 2014; McGowan et al., 2015). The other four cohort studies showed associations between longer duration of breastfeeding (any or exclusive) and a lower risk of developing cow milk sensitization (Liao et al., 2014; N=258), food allergy (Stelmach et al., 2014; N=501; aOR: 0.88; 95% CI: 0.82-0.95), or multiple food allergy (predominantly egg) (Peters et al., 2015; N=5276; aOR: 1.17; 95% CI: 1.09-1.24) after adjusting for potential confounders. The single cross-sectional study did not find a significant association between exclusive breastfeeding (poorly defined) and food allergies among children (N=386) ages 0 to 18 years with atopic dermatitis. Luccioli et al. collected data from prospective cohort of children (N=1,363) who participated in the Infant Feeding Practices Study (IFPS) II and also found no significant relationship between breastfeeding for various periods and food allergies (Luccioli et al., 2014). Only some studies used OFC as an outcome measure (Grimshaw et al., 2014; Peters et al., 2015). The single
__________________
9 More or Less: The authors included all studies. When multiple odds ratios were available for a single study, the authors preferentially selected estimates for exclusive breastfeeding, then longest duration versus shortest. When multiple ages of outcome were available, the authors chose the oldest up to 18 years.
cross-sectional study did not find a significant association between exclusive breastfeeding (poorly defined) and food allergies among children, ages 0 to 18 years, with atopic dermatitis (Mailhol et al., 2014).
As mentioned above, investigation of the role of breastfeeding in allergic disease is particularly prone to confounder bias because families who are at high risk of allergy are more likely to breastfeed, as recommended by some guidelines. In addition, the composition of human milk changes from colostrum to late lactation and throughout the day, and differs from mother to mother (Ballard and Morrow, 2013) and could therefore affect health outcomes of the child. Compounding the difficulties in this area is the inability to randomize to a nonbreastfeeding arm, as this would be unethical given the many well-established benefits of breastfeeding, such as protection against some chronic diseases, obesity, and infections.
The committee concludes that due to inconsistencies in results from prospective studies, the evidence that breastfeeding is protective against food allergies is limited. Strong evidence is unlikely to be forthcoming because of the ethical inability to randomize a population to breastfeeding alternatives. However, additional well-designed prospective research studies in infants at low and high risk for food allergy are needed.
Types of Infant Formula
Significant interest has been expressed in the use of modified infant formulas—especially partially hydrolyzed formulas (PHF), which include longer cow milk peptides, and extensively hydrolyzed cow’s milk formulas (EHF), which include di- and tri-peptides derived from cow milk protein—as a way to avoid allergen exposure and prevent early childhood allergic disease. As a result of demand from families with a history of allergy seeking readily available primary prevention interventions, industry has responded with the development of a variety of “allergy prevention” formulae, and expert bodies have provided recommendations regarding their use for preventing allergies. Some infant feeding guidelines have recommended that hydrolyzed formula can be considered as primary prevention therapy for some allergic diseases. In the United States, a policy statement from the American Academy of Pediatrics indicated that in studies of infants at high risk of atopy, modest evidence supports the delay or prevention of onset atopic dermatitis by the use of hydrolyzed, and particularly extensively hydrolyzed, formulas (Greer et al., 2008). In Australia, the Australasian Society of Clinical Immunology and Allergy Guidelines: Infant Feeding and Allergy Prevention no longer recommends hydrolyzed formulas as primary prevention therapy for allergic diseases. The guidelines now state, “Based on a recently published review of studies (Boyle et al., 2016), no consistent convincing evidence supports a protective role for partially hydrolyzed for-
mulas (usually labelled ‘HA’ or Hypoallergenic) or extensively hydrolyzed formulas for the prevention of eczema, food allergy, asthma or allergic rhinitis in infants or children” (ASCIA, 2016b).
A Cochrane review supports the use of hydrolyzed formula to prevent allergy in high-risk infants who are unable to be completely breastfed but not for those infants who can breastfed (Osborn and Sinn, 2006, 2009). Critics of this Cochrane review have pointed out that it suffers from small-study publication bias (i.e., scarcity of small negative studies) (Lowe et al., 2013) and thus the beneficial effect of PHF was likely overestimated. Due to the methodological concerns and inconsistency of the findings of the studies included in the review, the authors themselves recommend that further larger trials be conducted. Subsequently, new evidence from a large intervention trial of 620 high-risk infants (the Melbourne Atopic Cohort Study) has emerged. Findings from this trial challenge the effectiveness of PHF (Lowe et al., 2011).
The German Infant Nutritional Intervention (GINI) study was a trial aimed at exploring the effect of hydrolyzed formulas (compared to cow milk formula) in preventing allergic diseases in infants at high risk of atopy. Infants (N=2,252) were randomly assigned at birth to receive partially or extensively hydrolyzed whey formula, extensively hydrolyzed casein formula, or cow milk formula as milk substitute for the first 4 months when breastfeeding was insufficient. In a follow up until the children were age 6 years, hydrolyzed infant formulas prevented eczema and allergic manifestation (atopic dermatitis, food allergy, allergic urticaria, asthma, and hay fever/allergic rhinitis) (von Berg et. al., 2008). However, subsequent results showed little evidence of an ongoing preventive effect between the ages of 7 and 10 years (von Berg et al., 2013a). These more recent findings have not yet been incorporated into the Cochrane review. Likewise, the European Academy of Allergy & Clinical Immunology (EAACI) systematic review included both the Cochrane review and the Melbourne Atopic Cohort Study as well as the preliminary GINI results but did not include the latest results from the GINI study. Therefore, their conclusion supported the protective effect for PHF. Interestingly, the most recent findings from the GINI study suggest that casein-predominant EHF might be expected to have a greater biological effect than PHF because the formula is more extensively modified (von Berg et al., 2013a,b). However, most infant feeding guideline recommendations are based on the reality that PHF is both cheaper and more palatable than EHF and therefore should be considered instead of EHF. Additionally, in some countries EHF is only available with a prescription, which significantly increases costs to the health care system.
Most recently, Boyle et al. conducted a systematic review and meta-analysis of studies to determine whether feeding infants with hydrolyzed formulas reduces their risk of allergic disease (Boyle et al., 2016). Their
search yielded 37 intervention trials of more than 19,000 participants, although few studies included in the meta-analysis were published in the past 10 years. For the majority of studies, infants were considered to be at high risk of allergy because a first degree relative had a history of allergic disease. Overall, the pooled data showed no significant reduction in risk of any food allergy in infants ages 0 to 4 years when they were fed EHF or PHF compared to standard cow milk formula. On concluding the review, the authors found that previous studies suffered from unclear or high risk of bias. The review also showed evidence of conflict of interest and had inadequate methods of randomization and treatment allocation (selection bias). The authors recommended that international infant guidelines should be revised to remove the recommendation that hydrolyzed formula protects against allergic disease. In addition, a review of systematic reviews also stated that evidence is insufficient to conclude that the use of hydrolyzed formulas may reduce food allergy or sensitization when compared with standard formula in children with high atopy risk, and no evidence supports hydrolyzed formulas over breast milk for prevention of food sensitization or food allergy (Lodge et al., 2013).
The committee concludes that the studies on the effects of PHF or EHF for preventing food allergies have methodological flaws and their findings are inconsistent. Therefore, evidence on the effect of PHF or EHF for the prevention of food allergies is limited. If this area were to be investigated, high-quality RCT studies on the effects of PHF and EHF to determine whether hydrolyzed infant formulas influence the onset of food allergies would be needed before the use of these formulas could be recommended for prevention.
Dual Allergen Exposure Hypothesis
The “Dual Allergen Exposure” hypothesis proposes that allergic sensitization to foods may occur through exposure to low doses of allergen through the skin due to food allergens in the environment being absorbed through a damaged skin barrier (such as in eczema or presence of filaggrin loss-of-function mutations). This hypothesis also proposes that oral exposure to these allergens through consumption of allergenic foods early in infancy, before skin sensitization, leads to lasting oral tolerance and prevents the development of sensitization and allergy even with subsequent skin exposure (Du Toit et al., 2016; Lack, 2012; Lack et al., 2003) (see Figure 5-4).
Mechanistic evidence supporting this hypothesis comes from mouse models (Strid et al., 2005). Recent studies suggest that the activation of innate immune pathways in the skin through thymic stromal lymphopoietin, an interleukin (IL)-7-like cytokine associated with atopic dermatitis
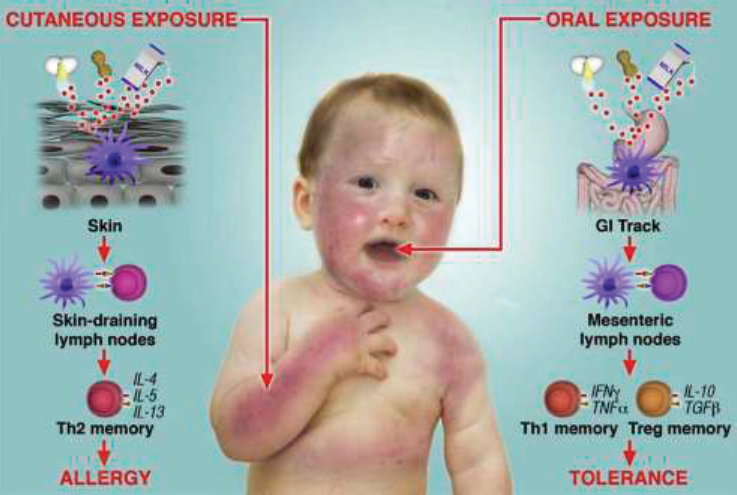
NOTE: GI = gastrointestinal, Treg = T regulatory.
SOURCE: Lack, 2012. Reprinted with permission from Elsevier.
and asthma, and basophil activation may play a key role in development of food allergy secondary to cutaneous sensitization in animal models (Muto et al., 2014; Noti et al., 2014).
Studies of human populations to date have primarily focused on peanut allergy. One human study demonstrated that peanut allergens can be found in the household environment and that higher exposure to environmental peanut antigens appears to increase the risk of peanut allergy in children with either filaggrin loss-of-function mutations or atopic dermatitis (Brough et al., 2014).
Other contextual factors support this hypothesis. Weaning practices in developed countries, coupled with high eczema rates may contribute to the high prevalence of food allergy. In the Western world, eczema prevalence is as high as 25 percent by age 1 year (Martin et al., 2013). (As noted previously, eczema frequently co-associates with food allergy, with 50 percent of those with early onset, moderately severe eczema developing food allergy by age 1 year [Martin et al., 2015].) This, coupled with recommendations
in the late 1990s and early 2000s to delay allergenic solids (e.g., egg until age 24 months and peanut until age 3 years) provide the correct temporal framework for this practice to have had a potential effect on the epidemic (Koplin and Allen, 2013). The following section presents the evidence on the main factors related to this hypothesis, namely skin barrier function and timing of introduction of foods. The section also includes the results from recent studies on introduction of diet diversity in early life.
Adequate Early Life Skin Barrier Function
It is important to note that mutations leading to filaggrin loss-of-function appear to be equally common among individuals with asymptomatic food sensitization and those with true food allergy (Tan et al., 2012a), suggesting that filaggrin confers a risk for food sensitization—the first step to food allergy—but not for food allergy itself. Previous studies reporting an association with food allergy were not designed to untangle any differential effect between sensitized tolerant and sensitized allergic individuals (Brown et al., 2011). Recent data from the Isle of Wight birth cohort used path analysis to demonstrate that the effect of filaggrin loss-of-function mutations on food allergy at age 10 years occurred indirectly through an effect on eczema and food sensitization in early childhood (Venkataraman et al., 2014). Together, these findings suggest that skin barrier function plays a role in sensitization status but not in food allergy or tolerance.
Two recent RCTs have investigated the application of daily moisturizer from birth in an attempt to reduce infantile eczema. Although the studies are small in size, the results support the idea that the integrity of the skin barrier is related to preventing food allergy. One RCT in the United States and the United Kingdom (N=124) examined the effects of an intervention that consisted of the use of an emollient at least once per day on neonates at risk of atopic dermatitis (Simpson et al., 2014). Atopic dermatitis was measured at 6 months. The results demonstrated a significant protective effect against atopic dermatitis (relative risk [RR]: 0.50; 95% CI: 0.28-0.9; P=0.017). The second trial examined the effect of using a moisturizer from the first week of life on eczema as a primary outcome and egg sensitization (but not allergy) as a secondary outcome in a group of 118 neonates at high risk of atopic dermatitis (Horimukai et al., 2014). At 32 weeks postnatal age, application of moisturizer to neonates was effective at preventing atopic dermatitis after 32 weeks, but unfortunately the trial showed no evidence of a reduction in sensitization to egg white in this relatively small study of 118 infants. However, a higher proportion of infants with atopic dermatitis showed egg sensitization compared with infants without atopic dermatitis.
The committee concludes that limited but consistent evidence on mutations on the filaggrin gene and on preventing eczema at early age suggests that impairment of skin barrier function plays a role in sensitization status as the first step on the path to food allergy.
Timing of Introduction of Solids and Infant Feeding
The dual antigen exposure hypothesis states that the second factor in the two steps to food allergy is the delay in oral allergen exposure. Until recently, delayed introduction of solids and particularly allergenic solids into the infant’s diet was a strategy adopted in many countries with the aim of reducing or preventing food allergies. Although exclusive breastfeeding for the first 6 months of life has been universally recommended in all countries to promote its health benefits (WHO, 2016), as described above, no evidence indicates that exclusive breastfeeding prevents the development of food allergies.
In 2008 and 2009, specific dietary advice to avoid peanuts in the United Kingdom and the United States, respectively, was rescinded (Greer et al., 2008; NHS, 2015a) based largely on the premise that evidence was insufficient to promote avoidance as a strategy to prevent food allergies. More recent advice does not state whether infants should actively receive allergenic foods, and if so at what age. Indeed, a recent nationwide UK dietary survey showed that only 8 percent of children younger than age 1 year had consumed any foods containing peanut (McAndrew et al., 2012).
The EAACI systematic review includes three cohort studies that found that the concept of delaying solid foods or cow milk consumption until 4 months of age does not appear to confer any benefit in terms of food allergies (de Silva et al., 2014). Most recently, evidence has been accumulating about the benefits of introducing allergens early. This section will focus on the most recent RCTs that evaluate the benefits of introducing allergens early in life.
In 2008, Du Toit et al. found that the level of peanut allergy in Jewish children in the United Kingdom was 10-fold higher than that of Jewish children in Israel and that median consumption of peanut protein was 0 g per month in the United Kingdom versus 7.1 g per month in Israel10 (Du Toit et al., 2008). Based on these results, Du Toit et al. conducted a large RCT to formally assess whether early introduction of peanut prevented the development of peanut allergy at age 5 years (Du Toit et al., 2015). The LEAP (Learning Early about Peanut Allergy) study randomized 640 highly
__________________
10 At the time, it was common practice in Israel to introduce a peanut snack (Bamba) as a weaning food into the diet of infants around the age of 4 to 6 months. In contrast, UK guidelines at the time recommended that children avoid peanut until after age 3 years.
atopic children with severe eczema and/or egg allergy to either consumption or avoidance of peanut at ages 4 to 11 months and the intervention continued until the children were age 5 years. The results showed that early consumption of peanut reduced the prevalence of peanut allergy (diagnosed by DBPCOFC) at age 5 years by more than 80 percent. The reduction was effective in children who were either SPT negative or SPT positive to peanut (wheals of 1, 2, 3, or 4 mm). As 17 percent of the LEAP cohort had peanut-specific IgE ≥0.35 at entry into the study and 27 percent had detectable IgE (≥0.1 kUA/L), prevention of peanut allergy was occurring for the majority of children after IgE sensitization had occurred; this represents secondary prevention (Du Toit et al., 2013). However, for infants in the group who were SPT negative at enrollment and who had no detectable IgE, early consumption of peanut also reduced the prevalence of peanut allergy (6 percent and 1 percent in the peanut avoidance group and in the peanut consuming group, respectively). This primary prevention strategy also was effective in a secondary analysis in children of different races.
As reviewed in Chapter 4, at the moment, we do not have definitive biomarkers to define tolerance. It is of interest that during the LEAP study, an early rise in peanut-specific IgG4 and peanut-specific IgG4/IgE ratio occurred in the peanut-consuming group (Du Toit et al., 2015). A high peanut-specific IgG4/IgE ratio was associated with protection against peanut allergy. Although the peanut-specific IgG4/IgE ratio decreased in the original peanut-consuming group during the period of peanut avoidance in the follow-up LEAP-On Study (Du Toit et al., 2016), it remained significantly higher than in the original peanut avoidance group. Interestingly in LEAP, peanut-specific IgE was not significantly different between the original peanut consuming and peanut avoidance groups throughout the study. However, peanut-specific IgE to Ara h 2 started to decline in the original peanut consuming group after 2.5 years of consumption, and continued to decline despite 1 year of peanut avoidance in that group between ages 5 and 6 years (Du Toit et al., 2016). This suggests potentially that high production of allergen-specific IgG4 may be important in the initiation of tolerance and that inhibition of IgE synthesis may be important in long-lived tolerance.
In order to determine whether early introduction of peanut was effective at preventing peanut allergy in the absence of ongoing peanut consumption, the LEAP-On Study was designed (Du Toit et al., 2016). Children (N=566) from the original LEAP cohort, irrespective of whether they were in the original peanut consumer or avoidant group, were asked to completely avoid peanut consumption for 1 year and then their peanut allergy status was determined by OFC, SPT, and specific IgE. Despite high adherence to this protocol of avoidance, the protective effects of early consumption remained and the original peanut consuming group had a 74 percent reduction in peanut allergy at age 6 years compared to the original peanut avoid-
ant group. Another follow-up study, the LEAP Adlib Study, is currently being designed. In this trial the original LEAP participants will continue to be followed up for a 4-year period of ad libitum consumption of peanut to determine whether the effects of early introduction remain protective.
In regard to other foods, observational studies suggest that delayed introduction of egg (Koplin et al., 2010), cow milk (Katz et al., 2010), and wheat (Poole et al., 2006) are associated with an increased risk of those respective food allergies. Various trials are in progress to confirm or refute these observations (see Table 5-1). Early evidence from Koplin and Allen (2013, p. 830) “suggests that if a window of opportunity for promoting tolerance exists, it may be different for each food” (Koplin and Allen, 2013, p. 830). However, further investigation is required. In the large HealthNuts study, where egg allergy was determined by challenge (among other food allergens), it was found that early introduction (age 4 to 6 months) of hen egg in the infant’s diet protected against the development of egg allergy, but introduction after 6 months of age was associated with significantly increased risk of developing egg allergy and even more so if introduced after age 9 months (Koplin et al., 2010). The results from the LEAP study (Du Toit et al., 2015) also are supported by data from the Solids Timing for Allergy Research (STAR) trial, which randomized 86 infants with eczema to egg avoidance or early regular egg consumption from age 4 months. The study found a lower, nonsignificant prevalence of egg allergy by 12 months in the intervention group (33% versus 51%; P=0.11) (Palmer et al., 2013). In a large birth cohort study conducted in Israel, IgE-mediated cow milk allergy did not occur in infants (N=13,019) who had received cow milk–based formula regularly in the first 2 weeks of life. In contrast, children who had formula milk introduced at age 3 to 4 months had the highest rate of cow milk allergy (Katz et al., 2010).
The EAT (Enquiring about Tolerance) intervention trial, which has recently been published, also examined the effects of early introduction of common allergenic foods. Unfortunately, compliance with intervention in this trial was low and the intention to treat analysis did not reveal a protective effect from early introduction of solids. In contrast, the per protocol analysis did suggest that early introduction of other common allergenic foods into the diet of infants may protect against the development of food allergies in general (Perkin et al., 2016). In the EAT study, exclusively breastfed infants (N=1,303) were recruited in the general population and randomly assigned at age 3 months to either introduction of six allergenic foods (cooked egg, peanut, cow milk, sesame, white fish, and wheat) (Early Introduction Group) or to the current recommended practice of exclusive breastfeeding until approximately 6 months of age (Standard Introduction Group). The primary outcome was determined to be food allergy between 1 and 3 years of age determined in nearly all participants by DBPCOFC.
The study showed a modest and nonsignificant 20 percent overall decrease in the rate of food allergies in the Early Introduction Group (5.6 percent compared to 7.1 percent in the Standard Introduction Group). However, in a per protocol analysis, the prevalence of any food allergy was significantly lower in the Early Introduction Group compared to the Standard Introduction Group (2.4 percent versus 7.3 percent; P=0.01) representing a 66 percent reduction in the prevalence of overall food allergy. The effects were most apparent for peanut allergy in the per protocol analysis (0 percent in the Early Introduction Group versus 2.5 percent in the Standard Introduction Group; P=0.003) and for egg allergy (1.4 percent versus 5.5 percent; P=0.009). These changes also were accompanied by decreases in SPT to the foods in the Early Introduction Group. A dose–response analysis revealed that 2 g of peanut protein or egg white protein per week appeared to be most protective against these food allergies. Interestingly 2 g of peanut protein per week is the dose that was observed in the Du Toit et al. study in Israel where children appeared to be protected against peanut allergy (Du Toit et al., 2008).
The EAT study shows that early introduction of foods was safe, as the intervention group did not experience an increased number of reactions compared to the controls. However, it is difficult to make any certain conclusions from the EAT study about the efficacy of early introduction of foods, given that efficacy was seen only in the per protocol group. Although careful analysis did not show any evidence of bias that could account for these results, it is not possible to completely exclude unmeasured bias.
A number of factors appeared to be associated with nonadherence to early introduction of foods relating to atopic predisposition. These include ethnicity, family life, readiness to eat solid foods, and parental perception of possible food allergic reactions (IgE- or non-IgE-mediated). The EAT study therefore suggests that if early introduction of allergenic foods from 3 months of age is to be adopted as a prevention strategy, numerous potential obstacles must be overcome with respect to implementation of adherence. Importantly, early introduction of allergens in the LEAP study or the EAT study did not reduce duration of breastfeeding (Feeney et al., 2016). It is noteworthy, however, that the participants in the EAT study are from the general population rather than a high-risk population and therefore any effect size may be less pronounced compared to the LEAP study. Furthermore, the intervention was more complex because it involved six foods, not one.
Diet diversity Two studies have examined the role of diversity of early life food exposures, which may be one factor that coincides temporally with the rise in food allergy, in the development of food sensitization and food allergy. A prospective birth cohort study of 856 children found that
increased diversity of complementary foods introduced in the first year of life was associated with a reduced risk of food allergy (Roduit et al., 2014). In another prospective longitudinal study of 123 participants, the authors found that dietary patterns in the first year of life consisting of more fresh fruit and vegetables and home-prepared meals were associated with less challenge-proven food allergy by the age of 2 years (Grimshaw et al., 2014).
The committee concludes that results of the LEAP trial provide strong evidence that early introduction of peanut (between 4 and 11 months) is protective against peanut allergy in infants who are at high risk (as defined by early onset eczema or coexistent egg allergy). Limited evidence from observational studies also suggests that delaying the introduction of egg, cow milk, and wheat to decrease risk of those food allergies has no benefits. Results from one RCT show a not significant decrease in food allergy if allergenic foods (i.e., cooked egg, peanut, cow milk, sesame, white fish, and wheat) are introduced starting at 3 months of age. More studies are necessary to assess whether early introduction of other allergenic foods, in addition to peanut, affect food allergy.
Nutritional Immunomodulation Hypothesis
Proper functioning of the immune system is crucial to health, and diet is a major and common exogenous factor modulating immunocompetence. Thus, nutrition research has focused on the role of foods or specific food components in enhancing immune system responsiveness to challenges and thus improving health and reducing disease risks (Albers et al., 2005). Along these lines, evidence supports the notion that sensitization or expression of food allergies does not depend exclusively on the food allergens per se, but on the exposure to other immunomodulatory exposures, such as other dietary factors, during specific critical periods. This section provides an overview of the immunomodulatory capacities of selected food components, including vitamin D, selected fatty acids, and folate. Better knowledge of these interactions should provide additional avenues for preventing and/or ameliorating food allergies.
Vitamin D
Vitamin D has become increasingly recognized as an important regulator of immune response (Adams and Hewison, 2008). 1,25(OH)2D can be converted from 25(OH)D locally based on widespread expression of vitamin D activating enzyme CP27B in a broad spectrum of cells involved in immune response, such as macrophages, B cells, and T cells. This active form of vitamin D exerts its function through interaction with the vitamin D receptor (VDR), which is also present in the above immune cells. Vitamin
D has been demonstrated to inhibit the differentiation of B lymphocytes to plasma cells and suppress immunoglobulin production (Chen et al., 2007). However, the effects of vitamin D on T lymphocytes are more complicated. Vitamin D has been shown to inhibit T cell proliferation and production of Th1 cytokines, which induces a shift in the balance between Th1 and Th2-type cytokines toward Th2 dominance (Cantorna et al., 2004; Iho et al., 1985; Reichel et al., 1987). In contrast, in CD4+ and CD8+ T-cells from human cord blood, vitamin D inhibits IL-12-generated interferon (IFN)-γ production and IL-4 production, as well as IL-4-induced expression of IL-13.
It has been hypothesized that in the presence of vitamin D, T regulatory cells function normally to suppress inappropriate Th1 and Th2 responses to environmental exposures leading to disease (Litonjua and Weiss, 2007). Research suggests that vitamin D deficiency might impair epithelial barrier integrity, which would in turn result in increased and inappropriate mucosal exposure to food antigens and also a pro-sensitization immune imbalance that compromises immunological tolerance (Roider et al., 2013).
Two opposing hypotheses have been proposed regarding the connection between vitamin D and allergic disease in general. In 1999, Wjst postulated that excess vitamin D might be associated with an increased risk of allergic disease based on its effects on the shift in the T-cell phenotype from a balance on Th1/Th2 to aTh2 dominance, and parallel patterns of increased oral vitamin D supplementation with a “Western lifestyle” (Wjst, 2008; Wjst and Dold, 1999). In contrast, Litonjua and Weiss raised an opposite hypothesis, suggesting that vitamin D might protect against asthma and allergies (Litonjua and Weiss, 2007, 2008). They believed that the immune effects of vitamin D are probably found on dendritic cells and Treg cells, and that these effects may differ depending on the stage of human development.
Two lines of ecological enquiry support the more recent hypothesis that low vitamin D may increase the risk of food allergy. First, countries further from the Equator (and thus receiving lower ambient ultraviolet radiation) have recorded more pediatric admissions to the hospital for food allergy–related events, and more prescriptions of hypoallergenic formulas for the treatment of cow milk allergy and adrenaline auto injectors for the treatment of anaphylaxis in children, compared to countries closer to the Equator (Camargo et al., 2007; Mullins et al., 2009, 2010; Rudders et al., 2010). These findings appear to be independent of longitude, socioeconomic status, or physician density. Second, children receiving care at a large medical center in Boston for food-related acute allergic reactions were more likely to be born in autumn/winter than in spring/summer (Vassallo et al., 2010). Similar relationships of food allergy to birth seasonality have been reported in the Southern hemisphere (Mullins
et al., 2011). Furthermore, children residing in Australia’s southern states have twice the odds (95% CI: 1.2-5.0) of peanut allergy at age 4 to 5 years and three times (95% CI: 1.0-9.0) the odds of egg allergy than children in the northern states (Osborne et al., 2012). A recent study from Australia described that infants with vitamin D insufficiency were three times more likely to have egg allergy than those who had adequate stores of the vitamin, with the odds increasing to 10-fold among those with two or more food allergies. Furthermore, among food-sensitized infants, those with vitamin D insufficiency were six times more likely to be food allergic than tolerant (Allen et al., 2013). These effects were observed among infants with Australian-born parents but not those with parents born outside Australia. Genetic polymorphisms contribute to variation in vitamin D binding protein levels, explaining almost 80 percent of variation in levels (Koplin et al., 2016). Binding protein levels in turn alter the biological availability of serum vitamin D, with lower levels increasing the availability of serum vitamin D (25OHD3). It was recently described that polymorphisms resulting in lower VDR levels appeared to compensate for adverse effects of low serum vitamin D on food allergy risk (Koplin et al., 2016), presumably by increasing the ability to use available vitamin D. These findings suggest that references ranges for optimal levels of serum vitamin D may need to take into account differences in VDR level.
A few studies have been published on the effect of maternal vitamin D status during pregnancy and the development of food allergy in offspring. A follow-up study from an RCT (N=164) reported that Vitamin D supplementation of the mothers during lactation may increase the risk of later food allergy up to 2 years of age (unadjusted analysis), although the authors reported high loss in subjects in the follow-up (Norizoe et al., 2014). However, results from cross-sectional studies (Allen et al., 2013) suggest that vitamin D sufficiency in infants age 1 year may be an important protective factor for food allergy at that age. Another cross-sectional study that followed a German birth cohort for 10 years reported that specific IgE for food allergens (OR: 1.07; 95% CI: 1.02-1.11) at age 10, as well as lifetime prevalence were significantly related to the vitamin D status (Wawro et al., 2014). Conversely, a study in Korea (N=226) showed that vitamin D deficiency increased the risk of sensitization to food allergens (Baek et al., 2014). In a longitudinal study (N=231), Jones et al. showed that maternal intake of supplemental vitamin D was significantly correlated with cord blood 25(OH)D3 concentration (Jones et al., 2012). However, the associations between cord blood 25(OH)D3 concentration and allergen sensitization, IgE-mediated food allergy, or eczema severity were not significant. Another prospective birth cohort study (N=378) in Germany reported that maternal and cord blood 25(OH)D3 was positively associated with
children’s risk for food allergy within the first 2 years of life (Weisse et al., 2013).
Liu et al. reported that the combination of persistently low vitamin D status at birth and in early childhood (ages 1 to 3 years) increased the risk of food sensitization (defined as specific IgE ≥0.35 kUA/L to any common food allergen, that is, egg white, milk, peanut, walnut, soy, shrimp, cod fish, and wheat) (aOR: 2.03; 95% CI: 1.02-4.04); the risk was particularly higher among children carrying the C allele of rs2243250 (aOR: 3.23; 95% CI: 1.37-7.60) (N=460) (Liu et al., 2013).
Multiple genes are known to be involved in 25(OH)D3 metabolism and regulatory pathways: genes encoding the molecules to convert 25(OH) D3 into its bioactive form 1,25(OH)2D (i.e., CYP27B1) and then a water-soluble metabolite (i.e., calcitroic acid; CYP24A1), as well as the receptor complex of vitamin D (i.e., VDR, RXRA, RXRB) and vitamin D binding protein (i.e., GC). Liu et al. evaluated children in the Boston Birth Cohort (N=649) and did not find an association between vitamin D levels in cord blood and sensitization to food allergens in early childhood (Liu et al., 2011). However, when examined with candidate gene single nucleotide polymorphisms, a significant interaction was identified for an IL-4 gene polymorphism and three other genes, indicating a risk for sensitization. In an Australian study, Koplin et al. investigated whether polymorphisms in a VDR-binding protein gene (low, the GT/TT genotype; high, the GG genotype) could modify the relationship between serum vitamin D and food allergy (Koplin et al., 2016). The study (N=5,276) found that low serum 25(OH)D3 levels (≤50 nM/L) at age 1 year had a modest association with food allergy, particularly among infants with the GG genotype (aOR: 6.0; 95% CI: 0.9-38.9) but the CI was wide. There was no association with food allergy in children with those with low serum 25(OH)D3 levels and GT/TT genotypes (aOR: 0.7; 95% CI: 0.2-2.0; P interaction=0.014).
The committee concludes that the quantity of evidence on the role of vitamin D in the development of food allergy during critical developmental windows (in utero, infancy, and early childhood) is limited. Further research is needed to confirm or refute this relationship.
Lipids/Omega-3 Fatty Acids
Dietary fat consumption has been hypothesized to influence atopy development by modulation of IgE production (Black and Sharpe, 1997). Among the different dietary fats, the ones that have been studied most extensively are the omega-3 fatty acids. Omega-3 fatty acids are known to have anti-inflammatory and immune modulator properties (Wall et al., 2010). Current evidence suggests that the intake of omega-3 fatty acids has decreased from ancestral times, whereas the consumption of omega-6 has
probably increased. Consequently, the dietary ratios of omega-6 to omega-3 fatty acids have changed over time from approximately 1:1 to almost 17:1 in certain industrialized societies (Simopoulos, 2002). The parallel increases in this ratio and in the prevalence of allergic disease, as well as information from experimental models, have elicited the hypothesis that dietary omega-3 fatty acids in early life may influence immune system development and immune cell function (Calder, 2013; Shek et al., 2012).
This hypothesis has been tested using a variety of experimental models, and the results of individual studies have been the focus of several reviews and meta-analyses that reveal the uncertainties that currently afflict this area of knowledge. Contributing to the current controversies are (1) the different experimental designs (observational versus RCTs), (2) the times of intervention and follow up, (3) the usually small size of the populations studied, (4) the different approaches to supplying the omega-3 fatty acids and the doses used, (5) the different periods investigated (fetal life, infancy, childhood), (6) the different outcomes examined, and (7) the potential confounder introduced by the wide-ranging presence of pro-allergenic pollutants and contaminants in fish, the major source of dietary omega-3.
The systematic review of Klemens et al., which reviewed the literature from 1950-2010, is considered to be of medium quality (Klemens et al., 2011). The review included three RCTs (Dunstan et al., 2003; Furuhjelm et al., 2009; Lauritzen et al., 2005; total N=264) of omega-3 fatty acids supplementation compared to olive or soy oil during pregnancy and/or lactation in a high-risk population for outcomes of food allergy, as defined by SPT and clinical diagnosis. When supplementation started during pregnancy egg sensitization decreased at 12 months of age (OR: 0.33; 95% CI: 0.16-0.70). Receiving the supplementation during pregnancy and/or lactation and food allergy at age 12 months were not significantly associated.
A recent Cochrane review, which included manuscripts published until August 2014, assessed the effect of omega-3 supplementation in pregnant and/or breastfeeding women on allergy outcomes (food allergy, atopic dermatitis, allergic rhinitis, and asthma/wheeze) in their children (Gunaratne et al., 2015). Overall, the results showed little reduction of allergic disease in the children resulting from maternal omega-3 supplementation during pregnancy and/or breastfeeding. Five trials reported food allergy outcomes (Dunstan et al., 2003; Furuhjelm et al., 2009; Lauritzen et al., 2005; Makrides et al., 2009, 2010). There was only one study where omega-3 supplementation reduced the incidence of IgE-mediated food allergies in children up to 12 months of age (Furuhjelm et al., 2009) (N=117; RR: 0.13; 95% CI: 0.02-0.95). Similarly, another recent review identified three RCTs (Dunstan et al., 2003; Furuhjelm et al., 2009; Palmer et al., 2012) and two follow-up studies (Furuhjelm et al., 2011; Palmer et al., 2013) with pregnant women whose infants were at high risk of atopy. After adjusting for
potential confounders or after long-term follow-up only one study showed an association between maternal omega-3 fatty acid supplementation and lower risk of food sensitization (Newberry et al., 2016).
The committee concludes that the current evidence does not support a link between increased maternal omega-3 intake and a protective effect on childhood food allergy.
Folate
Emerging interest in the role of folate in immune development and allergic disease has been driven by the recent understanding that folate, a dietary methyl donor, can affect immune function and alter gene expression through epigenetic mechanisms (Brown et al., 2014). Concerns have been raised about whether folic acid supplementation during pregnancy and/or early childhood is a potential risk factor for the development of atopic diseases in children. As animal models have demonstrated, maternal supplementation with dietary methyl donors during pregnancy induces hypermethylation of key regulatory genes in lung tissue, resulting in subsequent allergic airway disease in offspring (Hollingsworth et al., 2008). Exposure to folate in utero can affect DNA methylation during fetal development in humans (Amarasekera et al., 2014), which can influence transcriptional activity. For example, hypermethylation can silence the expression of genes. During polarization of naive T helper cells to Th2 cells, methylation of the promoter region of the IFN-γ gene blocks transcription factor binding and thus expression of the IFN-γ gene (Jones and Chen, 2006). Consequently, increased folic acid intake could influence the expression of genes that may be involved in T-cell differentiation during gestation. In turn, this may influence the allergic predisposition in the neonate.
To date, most human studies on this topic have focused on asthma, with very limited number of studies specific to food allergy or food sensitization. An Australian study (N=484) assessed maternal folic acid intake and serum folate levels during the third trimester, and cord blood folate status at birth (N=285), and allergic outcomes at age 12 months, including IgE-mediated food allergy, eczema, and asthma, in offspring (Dunstan et al., 2012, p. 51). In their study, food allergy was defined as “a history of immediate symptoms following contact and/or ingestion and a positive SPT to the implicated food.”
However, maternal serum folate status and allergic outcomes were not associated (Dunstan et al., 2012). In a study of 2,834 Dutch children, maternal folic acid supplement intake across the whole pregnancy, and intracellular folate status (measured in the third trimester of pregnancy in 837 [29.5%] participants) was not significantly associated with specific
IgE against hen egg, cow milk, peanut, and aeroallergens at age 2 years or eczema until age 6 to 7 years (Magdelijns et al., 2011).
A recent study that measured serum folate (at ages 2, 4, 6, and 8 years) in 138 U.S. children found that increased serum folate levels at or before age 6 years were significantly associated with increased incidence of sensitization to both food and aeroallergens, but not with serum total IgE, asthma, or wheezing at ages 6 or 9 years (Okupa et al., 2013). In the National Health and Nutrition Examination Survey (NHANES) (which covers ages 2 to 85 years), a cross-sectional study, serum folate levels were inversely associated with atopy, wheeze, and elevated total IgE levels (Matsui and Matsui, 2009).
Of note, the inconsistent results of previous studies are likely due to many reasons, including differences in sample size, participants’ ages, clinical characteristics, allergic outcomes, methods used for measurement of folate status, and statistical methods used in the analysis.
The committee concludes that evidence to assess the causal association between folate and the development or prevention of food allergy is lacking. Further research to study this potential association is needed.
Other Nutrients
A prospective cohort study assessed the relationship between maternal dietary antioxidant intake (B carotene, vitamins C and E, copper, and zinc) during pregnancy and food allergy of the child at age 12 months among families at high risk (West et al., 2012). This study of 300 mother-infant dyads found a protective effect of vitamin C intake on food allergy, with higher intakes that were limited to one quartile of vitamin C intake. For copper, intake in the highest quartile also showed a protective effect. However, as previously noted, observational studies suffer from inherent methodological flaws. Thus, proper RCTs are required to determine the causal effect of the maternal diet on the etiology of food allergies in offspring.
The committee concludes that evidence to assess the causal association between other nutrients and the development or prevention of food allergy is lacking.
Other Hypotheses
Do the Obesity and Diabetes Epidemics Have a Role in the Rise of Food Allergy?
The parallel increase in the prevalence of obesity and type 2 diabetes and allergic diseases raises the question of whether these conditions may be linked. Obesity is known to induce systemic inflammation, which
might adversely influence the immature immune system and atopic outcomes. Increased adipose tissue also could lead to reduced adiponectin levels, which in turn down-regulates the secretion of IL-10 and decreases regulatory T cells (Hersoug and Linneberg, 2007). Although the precise mechanism underlying the link between obesity and allergic disease including food allergies remains to be elucidated, the hypothesis is biologically plausible.
Very limited data are available on the association between having overweight or obesity and food allergy. Observational studies have shown that obesity is associated with a higher risk of atopy (elevated specific IgE to allergen) (Ouyang et al., 2009; Visness et al., 2009; Xu et al., 2000). For example, data from the 2005-2006 NHANES demonstrated that children with overweight or obesity had a higher geometric mean of total IgE levels and were at a higher risk of atopy than children with normal weight. This association was driven largely by allergic sensitization to food allergens, and systemic inflammation (measured as serum c-reactive protein) in children with obesity may play a role in the development of allergy (Visness et al., 2009). In contrast, ample studies show the association between overweight and obesity and asthma in both children and adults (Baumann and Lorentz, 2013; Granell et al., 2014).
The role of maternal overweight and obesity and diabetes on the developing fetus and the subsequent risk of allergic diseases has not been well studied but deserve attention. In the prospective Boston Birth cohort, Kumar et al. reported that in term births, gestational diabetes was significantly associated with allergen sensitization in the child, and such association was also driven by food sensitization (Kumar et al., 2009). In contrast, others reported no associations between obesity measures and atopy (Jarvis et al., 2002; Ma et al., 2010), or inverse associations (Van Gysel et al., 2009).
Other Unsubstantiated Hypothesis for the Rise in Food Allergy
Media interest in food allergies has become significant and sustained as food allergies have become more common. As such, public conjectures about potential causes for the rise are widespread. In particular, awareness about unfortunate cases of food-induced anaphylaxis is high. Added to that is the increased awareness by various community or commercial organizations (such as schools, restaurants, airlines, and sporting clubs) of their need to be careful about how they provide foods for food allergic individuals. As a result, communities are greatly interested in why the prevalence of food allergy appears to be rising.
One of the most widely held theories, among the many that abound, as to why food allergy is on the rise holds that it is due to the increasing
consumption of processed foods and food additives. Unfortunately, to date no significant research has been conducted on this issue. Websites and blogs tout the dangers of processed foods and food additives, and evidence from clinical observation suggests that some parents believe that food additives aggravate a range of clinical symptoms and signs, from difficult behavior and autism to gastrointestinal reactions. Clinically, the best way to understand whether a food is aggravating symptoms is to eliminate that food and later challenge with it—provided the risk of anaphylaxis has been excluded. However, the role of additives and preservatives in the development of food allergy in the first place has never been examined at the ecological or epidemiological level. In addition to understanding whether preservatives or additives have a direct toxicological effect on the developing immune system, it would be valuable to assess whether these substances actually influence the composition of the gastrointestinal microbiome.
Concerns over genetically modified crops (Nordlee et al., 1996) has resulted in consideration of the role that such foods may play in aggravating food allergy and in a requirement to assess the potential allergenicity of genetically modified crops (CAC, 2009; FAO/WHO, 2001). Although an online tool recently has been developed to help assess the role a novel protein may play in cross-reactivity (Goodman et al., 2016) based on criteria from the Codex Alimentarius Commission (CAC, 2009), current methodologies are considered inadequate to predict de novo allergenicity. Little or no research exists on whether the increased use of genetically modified crops could be linked to the rise in food allergy.
Numerous lay books and review articles argue that the increased consumption of fast food in the Westernized diet may have a significant impact on immunity (Myles, 2014). Although emerging indirect evidence suggests that fresh fruit and vegetables and food diversity might be important for an optimal and healthy start to life, to date little work has been done on their role specifically in preventing food allergy. Some of the first emerging evidence of diet diversity and its impact on food allergy development has been generated by the EuroPrevall study (Grimshaw et al., 2014). In a nested case-control within-cohort study of 41 infants using gold standard food challenge outcomes and 82 age-matched controls, the authors found that an infant diet with high levels of fruits, vegetables, and home-prepared foods is associated with less food allergy by 2 years of age. As an observational study, these results are subject to confounding but they generate a hypothesis worth testing in systematic trials.
The committee concludes that speculation abounds regarding why food allergy is on the rise. Although some ideas are based in appropriate theoretical frameworks, the absence of RCTs prevents firm conclusions to be drawn on their validity.
OVERALL CONCLUSIONS
The development of food allergies, like other complex diseases, might be regulated by the epigenome and in that way be caused by a genetic predisposition interacting with environmental exposures. The epigenome can be altered throughout the lifespan, but is particularly sensitive to environmental factors during early life periods. There appears to be a window of opportunity in the perinatal and early childhood period that may modulate the functionality of the immune system and related health conditions, specifically food allergies.
Many factors have been postulated to contribute to the onset of sensitization and to food allergies. A few of them have been extensively researched and sufficient evidence exists to support guidelines or to continue research to gain more insights (e.g., about the optimal timing and dosing of early introduction of foods). For other factors, either evidence is lacking about their association with food allergy but the association is biologically plausible (e.g., folate) or limited evidence exists about their association (e.g., vitamin D or fatty acids). For these, a recommendation based on their association with food allergy development cannot be made at this time and more research is needed. For other factors, direct or indirect evidence is lacking, but myths continue to prevail among the public (e.g., food additives).
For some factors (e.g., breastfeeding or vaginal delivery), although the evidence is inconsistent, it would be unethical to pursue RCTs; therefore, the evidence about their contribution to food allergies is derived solely from epidemiological studies. The review of the evidence by the committee neither confirmed nor rebutted current hypotheses related to any association between these factors and the increase in the prevalence of food allergies. The most recent research on the effects of allergen exposure at early age, however, strongly supports the dual allergen exposure hypothesis. The strongest data on potential prevention practices derives from a large RCT supporting the hypothesis that delaying the introduction of peanuts, coupled with high eczema rates, may have contributed to the high prevalence of peanut allergy in the Western world. Similar trials are being conducted for other allergenic foods and some of them are still being analyzed and interpreted (see Table 5-1). The LEAP study found that within a very narrow time range (ages 4 to 11 months), early introduction of peanut is protective against peanut allergy in infants who are at high risk (as defined by early onset eczema or coexistent egg allergy). Other studies have found that delaying introduction of other allergenic foods (cooked egg, cow milk, and wheat) has no benefits.
The lack of strong evidence for a link between most of the potential risk determinants and food allergy has created inconsistencies in public health
advice among different guidelines (see Table 5-2) and corresponding confusion among physicians, patients, and their families. Consensus of infant feeding guidelines to prevent food allergy across different public health authorities is needed for health care providers to counsel patients and their caregivers with consistent recommendations. Moreover, future clinical practice guidelines and public health policy should take into account the way in which a risk factor may differentially affect the risk of disease as well as the behavior of individuals with food allergy or their caregivers.
RECOMMENDATIONS
The committee recommends that public health authorities and clinical practice guidelines include consistent, clear, and evidence-based advice for families and health care providers, including dietitians, about the potential benefits of introducing allergenic foods (e.g., peanut products, egg, dairy, and wheat) in the first year of life to infants, when an infant is developmentally ready (around 6 months of age), but not before 4 months of age, particularly to those at high risk of allergy. Guidelines also should include information about the circumstances in which health care providers should advise their patients about the safest way to introduce in their diet peanut products (and/or other foods, as determined by the results of ongoing research).
In addition, as mentioned in Chapter 6, the committee recommends that public health authorities regularly update food allergy guidelines on diagnosis, prevention, and management based on strong scientific evidence. For example, current evidence is insufficient to associate any of the following behaviors with prevention of food allergy: food allergen avoidance diets for pregnant or lactating women, prolonged allergen avoidance in infancy, vaginal delivery, breastfeeding, infant formulas containing extensively or partially hydrolyzed protein, and supplementation with specific nutrients (e.g., vitamin D, folate, fatty acids) in children or adults.
RESEARCH NEEDS
Considerations for Study Designs
Studies on the etiological factors associated with food allergies frequently present methodological flaws due to various reasons, including lack of accounting for confounding factors (e.g., breastfeeding), use of inaccurate food allergy measures (e.g., self-reporting), or disregard for the
fact that different populations (e.g., those at high risk of developing a food allergy) might respond differently to the various risk factors. For example, due to a variety of differential gene-environment factors (e.g., genetics, epigenetics, microbiomes, and other pre- and postnatal environmental factors), populations will respond differently to interventions. Also, the etiology and early life onset of food allergy seems to be multifactorial, and collecting specimen for future analyses would be advantageous. Future research design on etiological determinants should consider the following:
- Conduct longitudinal birth cohort studies that explore the effects of environmental factors during critical developmental windows (in utero, infancy, and early childhood) on food allergy.
- Couple relevant prenatal, perinatal, and early childhood epidemiological and clinical data with appropriate biospecimen collections (e.g., serum, cord blood, breast milk) for current and future bio-marker analyses.
- Design studies so that the responses to various exposures of individuals and populations at high risk and low risk of developing food allergy can be differentiated.
- Use the currently accepted gold standard—double-blind, placebo-controlled oral food challenges (employing standard dosing protocols and scoring systems, so that the results of various studies can better be compared)—as the food allergy outcome in research intervention studies until a simpler reliable method to measure food allergy is identified and validated.
- Account for the potential influence of confounding factors, in addition to age, sex, and geography, such as breastfeeding, composition of breast milk, dietary intake, other allergic disorders in the patient or family history (particularly atopic dermatitis), genetic susceptibility, presence of dogs or cats in the household, number of siblings, history of antibiotic usage, and exposure to agents or practices that might impair skin barrier function.
- Engage patients or groups representing patients so that research designs may take into consideration potential socio-psychological, cultural, and behavioral considerations.
Overall Research Needs
Many genetic and environmental factors could contribute to the onset of sensitization and to food allergy. For the majority of factors reviewed by the committee, some, but largely insufficient or inconsistent, evidence exists at this time about their association with sensitization or food allergy. Nev-
ertheless, health care providers, patients, and their caregivers still need clear prevention approaches and authoritative and clear public health guidelines. Therefore, research needs to continue to support or refute the contribution of these factors to food sensitization or food allergy. The committee recognizes, though, that for other factors direct or indirect evidence is lacking and research is not currently warranted (e.g., food additives). Although some public health guidelines have been developed to guide practices of health care providers and individuals, efforts have not been undertaken to assess the impact of such public health guidelines on practices related to food allergy and on prevalence of food allergy. Prospective studies and behavioral research should be conducted to accomplish the following objectives:
- Examine risk factors for food allergies in all populations (ages, sex, ethnicities, comorbidities, socioeconomic strata), especially in those populations that might have been underrepresented in past research.
- Gain insights about the behaviors of those with (or at risk of) food allergy and their caregivers as well as about the impact of public health guidelines on health care providers and individuals’ practices.
- Examine the etiology of the rising prevalence of food allergy within the past two decades, which could identify new targets for allergy prevention and treatment. For example, what changes have occurred in food preparation and consumption behavior in communities and what is their potential relationship to the increase in food allergies? What changes may have occurred in the use of agents (such as detergents) or practices (such as in personal hygiene) that might contribute to impaired skin barrier function?
- Elucidate, through prospective studies, the role of environmental factors and gene-environment interactions in the atopic march and the development of food allergy. For example, do specific factors increase the risk of an individual progressing from eczema to food allergy?
- Explore potentially unidentified risk factors that may influence food allergy. For example, although the data available to date have not shown evidence of a relationship, it is plausible that maternal and early childhood adiposity and metabolic disorders could be risk factors for food allergy development.
- Using prospective birth cohort studies, evaluate the effects of multiple early life factors (individually and in combination) and of possible gene-environmental interactions in the development and
-
prevention of food allergy in order to inform the design of specific RCTs.
- Identify best practices to engage patients and their families in the planning stages of research studies so that patients’ and families’ concerns are considered, and assess the value of using these approaches.
Specific Research Needs
In addition, high-quality prospective studies and RCTs are needed on specific risk determinants for which some evidence exists about their effect on food allergy related to the most plausible hypotheses to make meaningful conclusions. These studies should be conducted to accomplish the following objectives:
The Microbial Hypothesis
- Determine, using well-designed prospective studies, the role of mode of birth delivery (vaginal, emergency versus elective cesarean section) and early life microbiome composition on the development of food allergy.
- Assess, through well-designed prospective studies, potential links between food allergy and antibiotic exposure in children (studies should include information on the type, dose, and frequency of antibiotic exposure).
- Determine whether pet ownership is related to food allergy by using well-designed prospective studies.
- Assess, with RCTs, the potential benefits of prebiotics and probiotics to prevent the onset of food allergy.
Allergen Avoidance and Exposure
- Elucidate the relationship, if any, between breastfeeding and the onset of food allergy (may also influence through microbiome modulation) with well-designed prospective studies and take into account the potential effect of differences in breast milk composition.
- Determine, with RCTs, whether consuming or eliminating or avoiding specific allergenic foods during pregnancy and lactation has any benefits.
- Conduct RCTs, similar to the Learning Early About Peanut study, to determine whether early introduction of peanut products has
-
benefit in individuals other than high-risk infants, who were studied in the original trial.
- Examine early introduction of allergenic foods in addition to peanut to determine whether this approach is beneficial in preventing the development of food allergy.
Nutrition Immunomodulation Hypothesis
- Assess, with RCTs, the potential role of specific nutrients, such as vitamin D, folate, or fatty acids, in preventing food allergy.
REFERENCES
Adams, J. S., and M. Hewison. 2008. Unexpected actions of vitamin D: New perspectives on the regulation of innate and adaptive immunity. Nat Clin Pract Endocrinol Metab 4(2):80-90.
Adlerberth, I., and A. E. Wold. 2009. Establishment of the gut microbiota in Western infants. Acta Paediatr 98(2):229-238.
Agostoni, C., T. Decsi, M. Fewtrell, O. Goulet, S. Kolacek, B. Koletzko, K. F. Michaelsen, L. Moreno, J. Puntis, J. Rigo, R. Shamir, H. Szajewska, D. Turck, and J. van Goudoever. 2008. Complementary feeding: A commentary by the ESPGHAN Committee on Nutrition. J Pediatr Gastroenterol Nutr 46(1):99-110.
Albers, R., J. M. Antoine, R. Bourdet-Sicard, P. C. Calder, M. Gleeson, B. Lesourd, S. Samartin, I. R. Sanderson, J. Van Loo, F. W. Vas Dias, and B. Watzl. 2005. Markers to measure immunomodulation in human nutrition intervention studies. Br J Nutr 94(3):452-481.
Alduraywish, S. A., C. J. Lodge, B. Campbell, K. J. Allen, B. Erbas, A. J. Lowe, and S. C. Dharmage. 2016. The march from early life food sensitization to allergic disease: A systematic review and meta-analyses of birth cohort studies. Allergy 71(1):77-89.
Allen, K. J., and J. J. Koplin. 2015. Why does Australia appear to have the highest rates of food allergy? Pediatr Clin North Am 62(6):1441-1451.
Allen, K. J., J. J. Koplin, A. L. Ponsonby, L. C. Gurrin, M. Wake, P. Vuillermin, P. Martin, M. Matheson, A. Lowe, M. Robinson, D. Tey, N. J. Osborne, T. Dang, H. T. Tina Tan, L. Thiele, D. Anderson, H. Czech, J. Sanjeevan, G. Zurzolo, T. Dwyer, M. L. Tang, D. Hill, and S. C. Dharmage. 2013. Vitamin D insufficiency is associated with challenge-proven food allergy in infants. J Allergy Clin Immunol 131(4):1109-1116.
Amarasekera, M., D. Martino, S. Ashley, H. Harb, D. Kesper, D. Strickland, R. Saffery, and S. L. Prescott. 2014. Genome-wide DNA methylation profiling identifies a folate-sensitive region of differential methylation upstream of ZFP57-imprinting regulator in humans. Faseb J 28(9):4068-4076.
Amoli, M. M., S. Hand, A. H. Hajeer, K. P. Jones, S. Rolf, C. Sting, B. H. Davies, and W. E. Ollier. 2002. Polymorphism in the STAT6 gene encodes risk for nut allergy. Genes Immun 3(4):220-224.
AND (Academy of Nutrition and Dietetics). 2015. Practice paper of the Academy of Nutrition and Dietetics: Promoting and supporting breastfeeding. http://www.eatrightpro.org/resource/practice/position-and-practice-papers/practice-papers/practice-paper-promoting-and-supporting-breastfeeding (accessed July 16, 2016).
Appelt, G. K., M. Chan-Yeung, W. T. A. Watson, H. Dimich-Ward, A. Ferguson, J. Manfreda, and A. B. Becker. 2004. Breastfeeding and food avoidance are ineffective in preventing sensitization in high risk children. J Allergy Clin Immunol 113(2):S99.
ASCIA (Australasian Society of Clinical Immunology and Allergy). 2016a. Food allergy FAQs. http://www.allergy.org.au/patients/food-allergy/faqs (accessed July 16, 2016).
ASCIA. 2016b. Guidelines: Infant feeding and allergy prevention. http://www.allergy.org.au/images/pcc/ASCIA_guidelines_infant_feeding_and_allergy_prevention.pdf (accessed May 25, 2016).
Azad, M. B., T. Konya, D. S. Guttman, C. J. Field, M. R. Sears, K. T. HayGlass, P. J. Mandhane, S. E. Turvey, P. Subbarao, A. B. Becker, J. A. Scott, and A. L. Kozyrskyj. 2015. Infant gut microbiota and food sensitization: Associations in the first year of life. Clin Exp Allergy 45(3):632-643.
Azad, M. B., T. Konya, R. R. Persaud, D. S. Guttman, R. S. Chari, C. J. Field, M. R. Sears, P. J. Mandhane, S. E. Turvey, P. Subbarao, A. B. Becker, J. A. Scott, and A. L. Kozyrskyj. 2016. Impact of maternal intrapartum antibiotics, method of birth and breastfeeding on gut microbiota during the first year of life: A prospective cohort study. BJOG 123(6):983-993.
Baek, J. H., Y. H. Shin, I. H. Chung, H. J. Kim, E. G. Yoo, J. W. Yoon, H. M. Jee, Y. E. Chang, and M. Y. Han. 2014. The link between serum vitamin D level, sensitization to food allergens, and the severity of atopic dermatitis in infancy. J Pediatr 165(4):849-854.
Ballard, O., and A. L. Morrow. 2013. Human milk composition: Nutrients and bioactive factors. Pediatr Clin North Am 60(1):49-74.
Barker, D. J. 1990. The fetal and infant origins of adult disease. BMJ 301(6761):1111.
Baumann, S., and A. Lorentz. 2013. Obesity—a promoter of allergy? Int Arch Allergy Immunol 162(3):205-213.
Black, P. N., and S. Sharpe. 1997. Dietary fat and asthma: Is there a connection? Eur Respir J 10(1):6-12.
Boyce, J. A., A. Assa’ad, A. W. Burks, S. M. Jones, H. A. Sampson, R. A. Wood, M. Plaut, S. F. Cooper, M. J. Fenton, S. H. Arshad, S. L. Bahna, L. A. Beck, C. Byrd-Bredbenner, C. A. Camargo, Jr., L. Eichenfield, G. T. Furuta, J. M. Hanifin, C. Jones, M. Kraft, B. D. Levy, P. Lieberman, S. Luccioli, K. M. McCall, L. C. Schneider, R. A. Simon, F. E. Simons, S. J. Teach, B. P. Yawn, and J. M. Schwaninger. 2010. Guidelines for the diagnosis and management of food allergy in the United States: Report of the NIAID-sponsored expert panel. J Allergy Clin Immunol 126(6 Suppl):S1-S58.
Boyle, R. J., D. Ierodiakonou, T. Khan, J. Chivinge, Z. Robinson, N. Geoghegan, K. Jarrold, T. Afxentiou, T. Reeves, S. Cunha, M. Trivella, V. Garcia-Larsen, and J. Leonardi-Bee. 2016. Hydrolyzed formula and risk of allergic or autoimmune disease: Systematic review and meta-analysis. BMJ 352:i974.
Brough, H. A., A. Simpson, K. Makinson, J. Hankinson, S. Brown, A. Douiri, D. C. Belgrave, M. Penagos, A. C. Stephens, W. H. McLean, V. Turcanu, N. Nicolaou, A. Custovic, and G. Lack. 2014. Peanut allergy: Effect of environmental peanut exposure in children with filaggrin loss-of-function mutations. J Allergy Clin Immunol 134(4):867-875.
Brown, S. B., K. W. Reeves, and E. R. Bertone-Johnson. 2014. Maternal folate exposure in pregnancy and childhood asthma and allergy: A systematic review. Nutr Rev 72(1):55-64.
Brown, S. J., Y. Asai, H. J. Cordell, L. E. Campbell, Y. Zhao, H. Liao, K. Northstone, J. Henderson, R. Alizadehfar, M. Ben-Shoshan, K. Morgan, G. Roberts, L. J. Masthoff, S. G. Pasmans, P. C. van den Akker, C. Wijmenga, J. O. Hourihane, C. N. Palmer, G. Lack, A. Clarke, P. R. Hull, A. D. Irvine, and W. H. McLean. 2011. Loss-of-function variants in the filaggrin gene are a significant risk factor for peanut allergy. J Allergy Clin Immunol 127(3):661-667.
Bunyavanich, S., S. L. Rifas-Shiman, T. A. Platts-Mills, L. Workman, J. E. Sordillo, C. A. Camargo, Jr., M. W. Gillman, D. R. Gold, and A. A. Litonjua. 2014. Peanut, milk, and wheat intake during pregnancy is associated with reduced allergy and asthma in children. J Allergy Clin Immunol 133(5):1373-1382.
CAC (Codex Alimentarius Commission). 2009. Foods derived from modern biotechnology. Rome: World Health Organization and Food and Agriculture Organization.
Calder, P. C. 2013. n-3 Fatty acids, inflammation and immunity: New mechanisms to explain old actions. Proc Nutr Soc 72(3):326-336.
Camargo, C. A., Jr., S. Clark, M. S. Kaplan, P. Lieberman, and R. A. Wood. 2007. Regional differences in EpiPen prescriptions in the United States: The potential role of vitamin D. J Allergy Clin Immunol 120(1):131-136.
Campos, E., N. Shimojo, Y. Inoue, T. Arima, S. Suzuki, M. Tomiita, T. Matsuura, A. Hata, Y. Suzuki, M. Aoyagi, and Y. Kohno. 2007. No association of polymorphisms in the 5’ region of the CD14 gene and food allergy in a Japanese population. Allergol Int 56(1):23-27.
Campos, E. J., N. Shimojo, Y. Suzuki, Y. Mashimo, T. Arima, T. Matsuura, Y. Inoue, A. Yamaide, M. Tomiita, K. Fujii, A. Hata, and Y. Kohno. 2008. IL-10 gene polymorphism, but not TGF-beta1 gene polymorphisms, is associated with food allergy in a Japanese population. Pediatr Allergy Immunol 19(8):716-721.
Cantorna, M. T., Y. Zhu, M. Froicu, and A. Wittke. 2004. Vitamin D status, 1,25-dihydroxyvitamin D3, and the immune system. Am J Clin Nutr 80(6 Suppl): 1717S-1720S.
Chen, S., G. P. Sims, X. X. Chen, Y. Y. Gu, S. Chen, and P. E. Lipsky. 2007. Modulatory effects of 1,25-dihydroxyvitamin D3 on human B cell differentiation. J Immunol 179(3):1634-1647.
Chen, T. K., J. H. Lee, H. H. Yu, Y. H. Yang, L. C. Wang, Y. T. Lin, and B. L. Chiang. 2012. Association between human IL-10 gene polymorphisms and serum IL-10 level in patients with food allergy. J Formos Med Assoc 111(12):686-692.
Cuello-Garcia, C. A., J. L. Brozek, A. Fiocchi, R. Pawankar, J. J. Yepes-Nunez, L. Terracciano, S. Gandhi, A. Agarwal, Y. Zhang, and H. J. Schunemann. 2015. Probiotics for the prevention of allergy: A systematic review and meta-analysis of randomized controlled trials. J Allergy Clin Immunol 136(4):952-961.
Cuello-Garcia, C. A., A. Fiocchi, R. Pawankar, J. J. Yepes-Nunez, G. P. Morgano, Y. Zhang, K. Ahn, S. Al-Hammadi, A. Agarwal, S. Gandhi, K. Beyer, W. Burks, G. W. Canonica, M. Ebisawa, R. Kamenwa, B. W. Lee, H. Li, S. Prescott, J. J. Riva, L. Rosenwasser, H. Sampson, M. Spigler, L. Terracciano, A. Vereda, S. Waserman, H. J. Schunemann, and J. L. Brozek. 2016. World Allergy Organization-McMaster University Guidelines for Allergic Disease Prevention (GLAD-P): Prebiotics. World Allergy Organ J 9:10.
de Silva, D., M. Geromi, S. Halken, A. Host, S. S. Panesar, A. Muraro, T. Werfel, K. Hoffmann-Sommergruber, G. Roberts, V. Cardona, A. E. Dubois, L. K. Poulsen, R. Van Ree, B. Vlieg-Boerstra, I. Agache, K. Grimshaw, L. O’Mahony, C. Venter, S. H. Arshad, and A. Sheikh. 2014. Primary prevention of food allergy in children and adults: Systematic review. Allergy 69(5):581-589.
Depner, M., M. J. Ege, J. Genuneit, J. Pekkanen, M. Roponen, M. R. Hirvonen, J. C. Dalphin, V. Kaulek, S. Krauss-Etschmann, J. Riedler, C. Braun-Fahrlander, C. Roduit, R. Lauener, P. I. Pfefferle, J. Weber, and E. von Mutius. 2013. Atopic sensitization in the first year of life. J Allergy Clin Immunol 131(3):781-788.
DGAC (Dietary Guidelines Advisory Committee). 2015. Scientific Report of the 2015 Dietary Guidelines Advisory Committee: Advisory Report to the Secretary of Health and Human Services and Secretary of Agriculture. U.S. Department of Agriculture, Agriculture Research Service, Washington DC.
Dharmage, S. C., A. J. Lowe, M. C. Matheson, J. A. Burgess, K. J. Allen, and M. J. Abramson. 2014. Atopic dermatitis and the atopic march revisited. Allergy 69(1):17-27.
Dominguez-Bello, M. G., E. K. Costello, M. Contreras, M. Magris, G. Hidalgo, N. Fierer, and R. Knight. 2010. Delivery mode shapes the acquisition and structure of the initial microbiota across multiple body habitats in newborns. Proc Natl Acad Sci U S A 107(26):11971-11975.
Dowhower Karpa, K., I. M. Paul, J. A. Leckie, S. Shung, N. Carkaci-Salli, K. E. Vrana, D. Mauger, T. Fausnight, and J. Poger. 2012. A retrospective chart review to identify perinatal factors associated with food allergies. Nutr J 11:87.
Du Toit, G., Y. Katz, P. Sasieni, D. Mesher, S. J. Maleki, H. R. Fisher, A. T. Fox, V. Turcanu, T. Amir, G. Zadik-Mnuhin, A. Cohen, I. Livne, and G. Lack. 2008. Early consumption of peanuts in infancy is associated with a low prevalence of peanut allergy. J Allergy Clin Immunol 122(5):984-991.
Du Toit, G., G. Roberts, P. H. Sayre, M. Plaut, H. T. Bahnson, H. Mitchell, S. Radulovic, S. Chan, A. Fox, V. Turcanu, and G. Lack. 2013. Identifying infants at high risk of peanut allergy: The Learning Early About Peanut Allergy (LEAP) screening study. J Allergy Clin Immunol 131(1):135-143.
Du Toit, G., G. Roberts, P. H. Sayre, H. T. Bahnson, S. Radulovic, A. F. Santos, H. A. Brough, D. Phippard, M. Basting, M. Feeney, V. Turcanu, M. L. Sever, M. Gomez Lorenzo, M. Plaut, and G. Lack. 2015. Randomized trial of peanut consumption in infants at risk for peanut allergy. N Engl J Med 372(9):803-813.
Du Toit, G., P. H. Sayre, G. Roberts, M. L. Sever, K. Lawson, H. T. Bahnson, H. A. Brough, A. F. Santos, K. M. Harris, S. Radulovic, M. Basting, V. Turcanu, M. Plaut, and G. Lack. 2016. Effect of avoidance on peanut allergy after early peanut consumption. N Engl J Med 374(15):1435-1443.
Dunstan, J. A., T. A. Mori, A. Barden, L. J. Beilin, A. L. Taylor, P. G. Holt, and S. L. Prescott. 2003. Fish oil supplementation in pregnancy modifies neonatal allergen-specific immune responses and clinical outcomes in infants at high risk of atopy: A randomized, controlled trial. J Allergy Clin Immunol 112(6):1178-1184.
Dunstan, J., C. West, S. McCarthy, J. Metcalfe, S. Meldrum, W. Oddy, M. Tulic, N. D’Vaz, and S. Prescott. 2012. The relationship between maternal folate status in pregnancy, cord blood folate levels, and allergic outcomes in early childhood. Allergy: Eur J Allergy Clin Immunol 67(1):50-57.
Ege, M. J., I. Herzum, G. Buchele, S. Krauss-Etschmann, R. P. Lauener, M. Roponen, A. Hyvarinen, D. A. Vuitton, J. Riedler, B. Brunekreef, J. C. Dalphin, C. Braun-Fahrlander, J. Pekkanen, H. Renz, E. von Mutius. 2008. Prenatal exposure to a farm environment modifies atopic sensitization at birth. J Allergy Clin Immunol 122(2):407-412.
Eggesbo, M., G. Botten, H. Stigum, P. Nafstad, and P. Magnus. 2003. Is delivery by cesarean section a risk factor for food allergy? J Allergy Clin Immunol 112(2):420-426.
Eggesbo, M., G. Botten, H. Stigum, S. O. Samuelsen, B. Brunekreef, and P. Magnus. 2005. Cesarean delivery and cow milk allergy/intolerance. Allergy 60(9):1172-1173.
Faa, G., C. Gerosa, D. Fanni, S. Nemolato, P. van Eyken, and V. Fanos. 2013. Factors influencing the development of a personal tailored microbiota in the neonate, with particular emphasis on antibiotic therapy. J Matern Fetal Neonatal Med 26(Suppl 2):35-43.
Falth-Magnusson, K., and N. I. Kjellman. 1987. Development of atopic disease in babies whose mothers were receiving exclusion diet during pregnancy—A randomized study. J Allergy Clin Immunol 80(6):868-875.
FAO/WHO (Food and Agriculture Organization/World Health Organization). 2001. Evaluation of allergenicity of genetically modified foods. Report of a Joint FAO/WHO Expert Consultation on Allergenicity of Foods Derived from Biotechnology. Rome: Food and Agricultural Organization/World Health Organization.
Fasano, A. 2000. Regulation of intercellular tight junctions by zonula occludens toxin and its eukaryotic analogue zonulin. Ann N Y Acad Sci 915:214-222.
Feeney, M., G. Du Toit, G. Roberts, P. H. Sayre, K. Lawson, H. T. Bahnson, M. L. Sever, S. Radulovic, M. Plaut, and G. Lack. 2016. Impact of peanut consumption in the LEAP Study: Feasibility, growth, and nutrition. J Allergy Clin Immunol 138(4):1108-1118.
Fleischer, D. M., J. M. Spergel, A. H. Assa’ad, and J. A. Pongracic. 2013. Primary prevention of allergic disease through nutritional interventions. J Allergy Clin Immunol Pract 1(1):29-36.
Fleischer, D. M., S. Sicherer, M. Greenhawt, D. Campbell, E. Chan, A. Muraro, S. Halken, Y. Katz, M. Ebisawa, L. Eichenfield, H. Sampson, G. Lack, G. Du Toit, G. Roberts, H. Bahnson, M. Feeney, J. Hourihane, J. Spergel, M. Young, A. As’aad, K. Allen, S. Prescott, S. Kapur, H. Saito, I. Agache, C. A. Akdis, H. Arshad, K. Beyer, A. Dubois, P. Eigenmann, M. Fernandez-Rivas, K. Grimshaw, K. Hoffman-Sommergruber, A. Host, S. Lau, L. O’Mahony, C. Mills, N. Papadopoulos, C. Venter, N. Agmon-Levin, A. Kessel, R. Antaya, B. Drolet, and L. Rosenwasser. 2015. Consensus communication on early peanut introduction and the prevention of peanut allergy in high-risk infants. J Allergy Clin Immunol 136(2):258-261.
Fooladi, A. A., S. Khani, H. M. Hosseini, S. F. Mousavi, E. M. Aghdam, and M. R. Nourani. 2013. Impact of altered early infant gut microbiota following breastfeeding and delivery mode on allergic diseases. Inflamm Allergy Drug Targets 12(6):410-418.
Frazier, A. L., C. A. Camargo, Jr., S. Malspeis, W. C. Willett, and M. C. Young. 2014. Prospective study of peripregnancy consumption of peanuts or tree nuts by mothers and the risk of peanut or tree nut allergy in their offspring. JAMA Pediatr 168(2):156-162.
Furuhjelm, C., K. Warstedt, J. Larsson, M. Fredriksson, M. F. Bottcher, K. Falth-Magnusson, and K. Duchen. 2009. Fish oil supplementation in pregnancy and lactation may decrease the risk of infant allergy. Acta Paediatr 98(9):1461-1467.
Furuhjelm, C., K. Warstedt, M. Fageras, K. Falth-Magnusson, J. Larsson, M. Fredriksson, and K. Duchen. 2011. Allergic disease in infants up to 2 years of age in relation to plasma omega-3 fatty acids and maternal fish oil supplementation in pregnancy and lactation. Pediatr Allergy Immunol 22(5):505-514.
Galli, S. J., P. Starkl, T. Marichal, and M. Tsai. 2016. Mast cells and IgE in defense against venoms: Possible “good side” of allergy? Allergol Int 65(1):3-15.
Gaudieri, S., M. Lucas, A. Lucas, E. McKinnon, H. Albloushi, A. Rauch, J. di Iulio, D. Martino, S. L. Prescott, and M. K. Tulic. 2012. Genetic variations in IL28B and allergic disease in children. PLoS One 7(1):e30607.
Goldberg, M., E. Eisenberg, A. Elizur, N. Rajuan, M. Rachmiel, A. Cohen, G. Zadik-Mnuhin, and Y. Katz. 2013. Role of parental atopy in cow’s milk allergy: A population-based study. Ann Allergy Asthma Immunol 110(4):279-283.
Goldman, A. S. 2000. Modulation of the gastrointestinal tract of infants by human milk. Interfaces and interactions. An evolutionary perspective. J Nutr 130(2S Suppl):426S-431S.
Goodman, R. E., M. Ebisawa, F. Ferreira, H. A. Sampson, R. van Ree, S. Vieths, J. L. Baumert, B. Bohle, S. Lalithambika, J. Wise, and S. L. Taylor. 2016. AllergenOnline: A peer-reviewed, curated allergen database to assess novel food proteins for potential cross-reactivity. Molec Nutr Food Res 60(5):1183-1198.
Gottgens, B. 2012. Genome-scale technology driven advances to research into normal and malignant haematopoiesis. Scientifica (Cairo) 2012:437956.
Granell, R., A. J. Henderson, D. M. Evans, G. D. Smith, A. R. Ness, S. Lewis, T. M. Palmer, and J. A. Sterne. 2014. Effects of BMI, fat mass, and lean mass on asthma in childhood: A Mendelian randomization study. PLoS Med 11(7):e1001669.
Greer, F. R., S. H. Sicherer, and A. W. Burks. 2008. Effects of early nutritional interventions on the development of atopic disease in infants and children: The role of maternal dietary restriction, breastfeeding, timing of introduction of complementary foods, and hydrolyzed formulas. Pediatrics 121(1):183-191.
Grimshaw, K. E., K. Allen, C. A. Edwards, K. Beyer, A. Boulay, L. B. van der Aa, A. Sprikkelman, S. Belohlavkova, M. Clausen, R. Dubakiene, E. Duggan, M. Reche, L. V. Marino, P. Norhede, L. Ogorodova, A. Schoemaker, A. Stanczyk-Przyluska, Z. Szepfalusi, E. Vassilopoulou, S. H. Veehof, B. J. Vlieg-Boerstra, M. Wjst, and A. E. Dubois. 2009. Infant feeding and allergy prevention: A review of current knowledge and recommendations. A EuroPrevall state of the art paper. Allergy 64(10):1407-1416.
Grimshaw, K. E. C., J. Maskell, E. M. Oliver, R. C. G. Morris, K. D. Foote, E. N. C. Mills, B. M. Margetts, and G. Roberts. 2014. Diet and food allergy development during infancy: Birth cohort study findings using prospective food diary data. J Allergy Clin Immunol 133(2):511-519.
Gunaratne, A. W., M. Makrides, and C. T. Collins. 2015. Maternal prenatal and/or postnatal n-3 long chain polyunsaturated fatty acids (LCPUFA) supplementation for preventing allergies in early childhood. Cochrane Database Syst Rev 7:CD010085.
Hand, S., C. Darke, J. Thompson, C. Stingl, S. Rolf, K. P. Jones, and B. H. Davies. 2004. Human leucocyte antigen polymorphisms in nut-allergic patients in South Wales. Clin Exp Allergy 34(5):720-724.
Hansen, C. H., D. S. Nielsen, M. Kverka, Z. Zakostelska, K. Klimesova, T. Hudcovic, H. Tlaskalova-Hogenova, and A. K. Hansen. 2012. Patterns of early gut colonization shape future immune responses of the host. PLoS One 7(3):e34043.
Hersoug, L. G., and A. Linneberg. 2007. The link between the epidemics of obesity and allergic diseases: Does obesity induce decreased immune tolerance? Allergy 62(10):1205-1213.
Hollingsworth, J. W., S. Maruoka, K. Boon, S. Garantziotis, Z. Li, J. Tomfohr, N. Bailey, E. N. Potts, G. Whitehead, D. M. Brass, and D. A. Schwartz. 2008. In utero supplementation with methyl donors enhances allergic airway disease in mice. J Clin Invest 118(10):3462-3469.
Hong, X., and X. Wang. 2014. Epigenetics and development of food allergy (FA) in early childhood. Curr Allergy Asthma Rep 14(9):460.
Hong, X., G. Wang, X. Liu, R. Kumar, H. J. Tsai, L. Arguelles, K. Hao, C. Pearson, K. Ortiz, A. Bonzagni, S. Apollon, L. Fu, D. Caruso, J. A. Pongracic, R. Schleimer, P. G. Holt, H. Bauchner, and X. Wang. 2011. Gene polymorphisms, breast-feeding, and development of food sensitization in early childhood. J Allergy Clin Immunol 128(2):374-381.
Hong, X., K. Hao, C. Ladd-Acosta, K. D. Hansen, H. J. Tsai, X. Liu, X. Xu, T. A. Thornton, D. Caruso, C. A. Keet, Y. Sun, G. Wang, W. Luo, R. Kumar, R. Fuleihan, A. M. Singh, J. S. Kim, R. E. Story, R. S. Gupta, P. Gao, Z. Chen, S. O. Walker, T. R. Bartell, T. H. Beaty, M. D. Fallin, R. Schleimer, P. G. Holt, K. C. Nadeau, R. A. Wood, J. A. Pongracic, D. E. Weeks, and X. Wang. 2015. Genome-wide association study identifies peanut allergy-specific loci and evidence of epigenetic mediation in US children. Nat Commun 6:6304.
Hong, X., C. Ladd-Acosta, K. Hao, B. Sherwood, H. Ji, C. A. Keet, R. Kumar, D. Caruso, X. Liu, G. Wang, Z. Chen, Y. Ji, G. Mao, S. O. Walker, T. R. Bartell, Z. Ji, Y. Sun, H. J. Tsai, J. A. Pongracic, D. E. Weeks, and X. Wang. 2016. Epigenome-wide association study links site-specific DNA methylation changes with cow’s milk allergy. J Allergy Clin Immunol 138(3):908-911.
Horimukai, K., K. Morita, M. Narita, M. Kondo, H. Kitazawa, M. Nozaki, Y. Shigematsu, K. Yoshida, H. Niizeki, K. Motomura, H. Sago, T. Takimoto, E. Inoue, N. Kamemura, H. Kido, J. Hisatsune, M. Sugai, H. Murota, I. Katayama, T. Sasaki, M. Amagai, H. Morita, A. Matsuda, K. Matsumoto, H. Saito, and Y. Ohya. 2014. Application of moisturizer to neonates prevents development of atopic dermatitis. J Allergy Clin Immunol 134(4):824-830.
Hu, Y., J. Chen, and H. Li. 2010. Comparison of food allergy prevalence among Chinese infants in Chongqing, 2009 versus 1999. Pediatr Int 52(5):820-824.
Iho, S., F. Kura, H. Sugiyama, T. Takahashi, and T. Hoshino. 1985. The role of monocytes in the suppression of PHA-induced proliferation and IL 2 production of human mononuclear cells by 1,25-dihydroxyvitamin D3. Immunol Lett 11(5-6):331-336.
Ivakhnenko, O., and S. Nyankovskyy. 2013. Effect of the specific infant formula mixture of oligosaccharides on local immunity and development of allergic and infectious disease in young children: Randomized study. Pediatria Polska 88(5):398-404.
Jarvis, D., S. Chinn, J. Potts, and P. Burney. 2002. Association of body mass index with respiratory symptoms and atopy: Results from the European Community Respiratory Health Survey. Clin Exp Allergy 32(6):831-837.
Jones, A. C., E. A. Miles, J. O. Warner, B. M. Colwell, T. N. Bryant, and J. A. Warner. 1996. Fetal peripheral blood mononuclear cell proliferative responses to mitogenic and allergenic stimuli during gestation. Pediatr Allergy Immunol 7(3):109-116.
Jones, A. P., D. Palmer, G. Zhang, and S. L. Prescott. 2012. Cord blood 25-hydroxyvitamin D3 and allergic disease during infancy. Pediatrics 130(5):e1128-e1135.
Jones, B., and J. Chen. 2006. Inhibition of IFN-gamma transcription by site-specific methylation during T helper cell development. EMBO J 25(11):2443-2452.
Katz, Y., N. Rajuan, M. R. Goldberg, E. Eisenberg, E. Heyman, A. Cohen, and M. Leshno. 2010. Early exposure to cow’s milk protein is protective against IgE-mediated cow’s milk protein allergy. J Allergy Clin Immunol 126(1):77-82.
Kelly, D., T. King, and R. Aminov. 2007. Importance of microbial colonization of the gut in early life to the development of immunity. Mutat Res 622(1-2):58-69.
Klemens, C. M., D. R. Berman, and E. L. Mozurkewich. 2011. The effect of perinatal omega-3 fatty acid supplementation on inflammatory markers and allergic diseases: A systematic review. BJOG 118(8):916-925.
Kong, X. Y., Y. Yang, J. Guan, and R. Z. Wang. 2014. Probiotics’ preventive effect on pediatric food allergy: A meta-analysis of randomized controlled trials. Chin Med Sci J 29(3):144-147.
Koplin, J. J., and K. J. Allen. 2013. Optimal timing for solids introduction—why are the guidelines always changing? Clin Exp Allergy 43(8):826-834.
Koplin, J. J., N. J. Osborne, M. Wake, P. E. Martin, L. C. Gurrin, M. N. Robinson, D. Tey, M. Slaa, L. Thiele, L. Miles, D. Anderson, T. Tan, T. D. Dang, D. J. Hill, A. J. Lowe, M. C. Matheson, A. L. Ponsonby, M. L. Tang, S. C. Dharmage, and K. J. Allen. 2010. Can early introduction of egg prevent egg allergy in infants? A population-based study. J Allergy Clin Immunol 126(4):807-813.
Koplin, J. J., S. C. Dharmage, A. L. Ponsonby, M. L. Tang, A. J. Lowe, L. C. Gurrin, N. J. Osborne, P. E. Martin, M. N. Robinson, M. Wake, D. J. Hill, and K. J. Allen. 2012a. Environmental and demographic risk factors for egg allergy in a population-based study of infants. Allergy 67(11):1415-1422.
Koplin, J. J., M. L. Tang, P. E. Martin, N. J. Osborne, A. J. Lowe, A. L. Ponsonby, M. N. Robinson, D. Tey, L. Thiele, D. J. Hill, L. C. Gurrin, M. Wake, S. C. Dharmage, and K. J. Allen. 2012b. Predetermined challenge eligibility and cessation criteria for oral food challenges in the HealthNuts population-based study of infants. J Allergy Clin Immunol 129(4):1145-1147.
Koplin, J. J., R. L. Peters, A. L. Ponsonby, L. C. Gurrin, D. Hill, M. L. Tang, S. C. Dharmage, and K. J. Allen. 2014. Increased risk of peanut allergy in infants of Asian-born parents compared to those of Australian-born parents. Allergy 69(12):1639-1647.
Koplin, J. J., N. H. Suaini, P. Vuillermin, J. A. Ellis, M. Panjari, A. L. Ponsonby, R. L. Peters, M. C. Matheson, D. Martino, T. Dang, N. J. Osborne, P. Martin, A. Lowe, L. C. Gurrin, M. L. Tang, M. Wake, T. Dwyer, J. Hopper, S. C. Dharmage, and K. J. Allen. 2016. Polymorphisms affecting vitamin D-binding protein modify the relationship between serum vitamin D (25[OH]D3) and food allergy. J Allergy Clin Immunol 137(2):500-506.
Kozyrskyj, A. 2015. Infant gut microbial markers of food sensitization at age 1. Presented at Comittee Workshop, August 31, 2015. Washington, DC.
Kramer, M. S., and R. Kakuma. 2012. Maternal dietary antigen avoidance during pregnancy or lactation, or both, for preventing or treating atopic disease in the child. Cochrane Database Syst Rev 9:Cd000133.
Kumar, R., F. Ouyang, R. E. Story, J. A. Pongracic, X. Hong, G. Wang, C. Pearson, K. Ortiz, H. Bauchner, and X. Wang. 2009. Gestational diabetes, atopic dermatitis, and allergen sensitization in early childhood. J Allergy Clin Immunol 124(5):1031-1038.
Kusunoki, T., I. Okafuji, T. Yoshioka, M. Saito, R. Nishikomori, T. Heike, M. Sugai, A. Shimizu, and T. Nakahata. 2005. SPINK5 polymorphism is associated with disease severity and food allergy in children with atopic dermatitis. J Allergy Clin Immunol 115(3):636-638.
Lack, G. 2012. Update on risk factors for food allergy. J Allergy Clin Immunol 129(5): 1187-1197.
Lack, G., D. Fox, K. Northstone, and J. Golding. 2003. Factors associated with the development of peanut allergy in childhood. N Engl J Med 348(11):977-985.
Langley-Evans, S. 1997. Fetal programming of immune function and respiratory disease. Clin Exp Allergy 27(12):1377-1379.
Lau, M. Y., S. C. Dharmage, J. A. Burgess, A. J. Lowe, C. J. Lodge, B. Campbell, and M. C. Matheson. 2014. CD14 polymorphisms, microbial exposure and allergic diseases: A systematic review of gene-environment interactions. Allergy 69(11):1440-1453.
Lauritzen, L., T. M. Kjaer, M. B. Fruekilde, K. F. Michaelsen, and H. Frokiaer. 2005. Fish oil supplementation of lactating mothers affects cytokine production in 2 1/2-year-old children. Lipids 40(7):669-676.
Li, M., J. B. Liu, Q. Liu, M. Yao, R. Cheng, H. Xue, H. Zhou, and Z. Yao. 2012. Interactions between FLG mutations and allergens in atopic dermatitis. Arch Dermatol Res 304(10):787-793.
Li, M., M. Wang, and S. M. Donovan. 2014. Early development of the gut microbiome and immune-mediated childhood disorders. Semin Reprod Med 32(1):74-86.
Liao, S. L., S. H. Lai, K. W. Yeh, Y. L. Huang, T. C. Yao, M. H. Tsai, M.C. Hua, and J. L. Huang. 2014. Exclusive breastfeeding is associated with reduced cow’s milk sensitization in early childhood. Pediatr Allergy Immunol 25(5):456-461.
Lilja, G., A. Dannaeus, K. Falth-Magnusson, V. Graff-Lonnevig, S. G. Johansson, N. I. Kjellman, and H. Oman. 1988. Immune response of the atopic woman and foetus: Effects of high- and low-dose food allergen intake during late pregnancy. Clin Allergy 18(2):131-142.
Litonjua, A. A., and S. T. Weiss. 2007. Is vitamin D deficiency to blame for the asthma epidemic? J Allergy Clin Immunol 120(5):1031-1035.
Litonjua, A. A., and S. T. Weiss. 2008. Reply. J Allergy Clin Immunol 121(4):1066.
Liu, X., T. H. Beaty, P. Deindl, S. K. Huang, S. Lau, C. Sommerfeld, M. D. Fallin, W. H. Kao, U. Wahn, and R. Nickel. 2004. Associations between specific serum IgE response and 6 variants within the genes IL4, IL13, and IL4RA in German children: The German Multicenter Atopy Study. J Allergy Clin Immunol 113(3):489-495.
Liu, X., S. Zhang, H. J. Tsai, X. Hong, B. Wang, Y. Fang, X. Liu, J. A. Pongracic, and X. Wang. 2009. Genetic and environmental contributions to allergen sensitization in a Chinese twin study. Clin Exp Allergy 39(7):991-998.
Liu, X., G. Wang, X. Hong, D. Wang, H. J. Tsai, S. Zhang, L. Arguelles, R. Kumar, H. Wang, R. Liu, Y. Zhou, C. Pearson, K. Ortiz, R. Schleimer, P. G. Holt, J. Pongracic, H. E. Price, C. Langman, and X. Wang. 2011. Gene-vitamin D interactions on food sensitization: A prospective birth cohort study. Allergy 66(11):1442-1448.
Liu, X., L. Arguelles, Y. Zhou, G. Wang, Q. Chen, H. J. Tsai, X. Hong, R. Liu, H. E. Price, C. Pearson, S. Apollon, N. Cruz, R. Schleimer, C. B. Langman, J. A. Pongracic, and X. Wang. 2013. Longitudinal trajectory of vitamin D status from birth to early childhood in the development of food sensitization. Pediatr Res 74(3):321-326.
Lodge, C. J., K. J. Allen, A. J. Lowe, and S. C. Dharmage. 2013. Overview of evidence in prevention and aetiology of food allergy: A review of systematic reviews. Int J Environ Res Public Health 10(11):5781-5806.
Lodge, C. J., D. J. Tan, M. X. Lau, X. Dai, R. Tham, A. J. Lowe, G. Bowatte, K. J. Allen, and S. C. Dharmage. 2015. Breastfeeding and asthma and allergies: A systematic review and meta-analysis. Acta Paediatr 104(467):38-53.
Loo, E. X., G. V. Llanora, Q. Lu, M. M. Aw, B. W. Lee, and L. P. Shek. 2014. Supplementation with probiotics in the first 6 months of life did not protect against eczema and allergy in at-risk Asian infants: A 5-year follow-up. Int Arch Allergy Immunol 163(1):25-28.
Lowe, A. J., C. S. Hosking, C. M. Bennett, K. J. Allen, C. Axelrad, J. B. Carlin, M. J. Abramson, S. C. Dharmage, and D. J. Hill. 2011. Effect of a partially hydrolyzed whey infant formula at weaning on risk of allergic disease in high-risk children: A randomized controlled trial. J Allergy Clin Immunol 128(2):360-365.
Lowe, A. J., S. C. Dharmage, K. J. Allen, M. L. Tang, and D. J. Hill. 2013. The role of partially hydrolyzed whey formula for the prevention of allergic disease: Evidence and gaps. Expert Rev Clin Immunol 9(1):31-41.
Luccioli, S., Y. Zhang, L. Verrill, M. Ramos-Valle, and E. Kwegyir-Afful. 2014. Infant feeding practices and reported food allergies at 6 years of age. Pediatrics 134(Suppl 1):S21-S28.
Ma, J., L. Xiao, and S. B. Knowles. 2010. Obesity, insulin resistance and the prevalence of atopy and asthma in US adults. Allergy 65(11):1455-1463.
Madore, A. M., V. T. Vaillancourt, Y. Asai, R. Alizadehfar, M. Ben-Shoshan, D. L. Michel, A. L. Kozyrskyj, A. Becker, M. Chan-Yeung, A. E. Clarke, P. Hull, D. Daley, A. J. Sandford, and C. Laprise. 2013. HLA-DQB1*02 and DQB1*06:03P are associated with peanut allergy. Eur J Hum Genet 21(10):1181-1184.
Magdelijns, F. J., M. Mommers, J. Penders, L. Smits, and C. Thijs. 2011. Folic acid use in pregnancy and the development of atopy, asthma, and lung function in childhood. Pediatrics 128(1):e135-144.
Mailhol, C., F. Giordano-Labadie, V. Lauwers-Cances, A. Ammoury, C. Paul, and F. Rance. 2014. Point prevalence and risk factors for food allergy in a cohort of 386 children with atopic dermatitis attending a multidisciplinary dermatology/paediatric allergy clinic. Eur J Dermatol 24(1):63-69.
Makrides, M., R. A. Gibson, A. J. McPhee, C. T. Collins, P. G. Davis, L. W. Doyle, K. Simmer, P. B. Colditz, S. Morris, L. G. Smithers, K. Willson, and P. Ryan. 2009. Neurodevelopmental outcomes of preterm infants fed high-dose docosahexaenoic acid: A randomized controlled trial. JAMA 301(2):175-182.
Makrides, M., R. A. Gibson, A. J. McPhee, L. Yelland, J. Quinlivan, P. Ryan, and Domino Investigative Team. 2010. Effect of DHA supplementation during pregnancy on maternal depression and neurodevelopment of young children: A randomized controlled trial. JAMA 304(15):1675-1683.
Marichal, T., P. Starkl, L. L. Reber, J. Kalesnikoff, H. C. Oettgen, M. Tsai, M. Metz, and S. J. Galli. 2013. A beneficial role for immunoglobulin E in host defense against honeybee venom. Immunity 39(5):963-975.
Marrs, T., K. D. Bruce, K. Logan, D. W. Rivett, M. R. Perkin, G. Lack, and C. Flohr. 2013. Is there an association between microbial exposure and food allergy? A systematic review. Pediatr Allergy Immunol 24(4):311-320.
Martin, P. E., J. J. Koplin, J. K. Eckert, A. J. Lowe, A. L. Ponsonby, N. J. Osborne, L. C. Gurrin, M. N. Robinson, D. J. Hill, M. L. K. Tang, S. C. Dharmage, and K. J. Allen. 2013. The prevalence and socio-demographic risk factors of clinical eczema in infancy: A population-based observational study. Clin Exp Allergy 43(6):642-651.
Martin, P. E., J. K. Eckert, J. J. Koplin, A. J. Lowe, L. C. Gurrin, S. C. Dharmage, P. Vuillermin, M. L. Tang, A. L. Ponsonby, M. Matheson, D. J. Hill, and K. J. Allen. 2015. Which infants with eczema are at risk of food allergy? Results from a population-based cohort. Clin Exp Allergy 45(1):255-264.
Martin, R., A. J. Nauta, K. Ben Amor, L. M. Knippels, J. Knol, and J. Garssen. 2010. Early life: Gut microbiota and immune development in infancy. Benef Microbes 1(4):367-382.
Martino, D., J. E. Joo, A. Sexton-Oates, T. Dang, K. Allen, R. Saffery, and S. Prescott. 2014. Epigenome-wide association study reveals longitudinally stable DNA methylation differences in CD4+ T cells from children with IgE-mediated food allergy. Epigenetics 9(7):998-1006.
Matsui, E. C., and W. Matsui. 2009. Higher serum folate levels are associated with a lower risk of atopy and wheeze. J Allergy Clin Immunol 123(6):1253-1259.
McAndrew, F., J. Thompson, L. Fellows, A. Large, M. Speed, and M. J. Renfrew. 2012. Infant feeding survey. Leeds, England: Health and Social Care Information Centre, IFF Research.
McGowan, E. C., G. R. Bloomberg, P. J. Gergen, C. M. Visness, K. F. Jaffee, M. Sandel, G. O’Connor, M. Kattan, J. Gern, and R. A. Wood. 2015. Influence of early-life exposures on food sensitization and food allergy in an inner-city birth cohort. J Allergy Clin Immunol 135(1):171-178.
McLoughlin, R. M., and K. H. Mills. 2011. Influence of gastrointestinal commensal bacteria on the immune responses that mediate allergy and asthma. J Allergy Clin Immunol 127(5):1097-1107; quiz 1108-1109.
Metsala, J., A. Lundqvist, M. Kaila, M. Gissler, T. Klaukka, and S. M. Virtanen. 2010. Maternal and perinatal characteristics and the risk of cow’s milk allergy in infants up to 2 years of age: A case-control study nested in the Finnish population. Am J Epidemiol 171(12):1310-1316.
Metsala, J., A. Lundqvist, L. J. Virta, M. Kaila, M. Gissler, and S. M. Virtanen. 2013. Mother’s and offspring’s use of antibiotics and infant allergy to cow’s milk. Epidemiology 24(2):303-309.
Mullins, R. J., S. Clark, and C. A. Camargo, Jr. 2009. Regional variation in epinephrine autoinjector prescriptions in Australia: More evidence for the vitamin D-anaphylaxis hypothesis. Ann Allergy Asthma Immunol 103(6):488-495.
Mullins, R. J., S. Clark, and C. A. Camargo, Jr. 2010. Regional variation in infant hypoallergenic formula prescriptions in Australia. Pediatr Allergy Immunol 21(2 Pt 2):e413-e420.
Mullins, R. J., S. Clark, C. Katelaris, V. Smith, G. Solley, and C. A. Camargo, Jr. 2011. Season of birth and childhood food allergy in Australia. Pediatr Allergy Immunol 22(6):583-589.
Muraro, A., S. Halken, S. H. Arshad, K. Beyer, A. E. Dubois, G. Du Toit, P. A. Eigenmann, K. E. Grimshaw, A. Hoest, G. Lack, L. O’Mahony, N. G. Papadopoulos, S. Panesar, S. Prescott, G. Roberts, D. de Silva, C. Venter, V. Verhasselt, A. C. Akdis, and A. Sheikh. 2014. Primary prevention of food allergy. Allergy 69(5):590-601.
Muto, T., A. Fukuoka, K. Kabashima, S. F. Ziegler, K. Nakanishi, K. Matsushita, and T. Yoshimoto. 2014. The role of basophils and proallergic cytokines, TSLP and IL-33, in cutaneously sensitized food allergy. Int Immunol 26(10):539-549.
Myers, J. M. B., N. Wang, G. K. LeMasters, D. I. Bernstein, T. G. Epstein, M. A. Lindsey, M. B. Ericksen, R. Chakraborty, P. H. Ryan, M. S. Villareal, J. W. Burkle, J. E. Lockey, T. Reponen, and G. K. K. Hershey. 2010. Genetic and environmental risk factors for childhood eczema development and allergic sensitization in the CCAAPS Cohort. J Invest Derm 130(2):430-437.
Myles, I. A. 2014. Fast food fever: Reviewing the impacts of the Western diet on immunity. Nutr J 13:61.
Neeland, M. R., D. J. Martino, and K. J. Allen. 2015. The role of gene-environment interactions in the development of food allergy. Expert Rev Gastroenterol Hepatol 9(11):1317-1318.
Negoro, T., K. Orihara, T. Irahara, H. Nishiyama, K. Hagiwara, R. Nishida, H. Takagi, K. Satoh, Y. Yamamoto, S. Shimizu, T. Hagiwara, M. Ishii, T. Tanioka, Y. Nakano, K. Takeda, I. Yoshimura, Y. Iikura, and T. Tobe. 2006. Influence of SNPs in cytokine-related genes on the severity of food allergy and atopic eczema in children. Pediatr Allergy Immunol 17(8):583-590.
Newberry, S. J., M. Chung, M. Booth, M. A. Maglione, A. M. Tang, C. E. O’Hanlon, D. D. Wang, A. Okunogbe, C. Huang, A. Motala, M. Timmer, W. Dudley, R. Shanman, T. R. Coker, and P. G. Shekelle. 2016. Omega-3 fatty acids and maternal and child health: An updated systematic review. Evidence Report/Technology Assessment No. 224. AHRQ Publication No. 16-E003-EF. Rockville, MD: Agency for Healthcare Research and Quality.
NHMRC (National Health and Medical Research Council). 2013. Infant feeding guidelines. Canberra, Australia: National Health and Medical Research Council. https://www.eatforhealth.gov.au/guidelines (accessed November 14, 2016).
NHS (National Health Service). 2015a. Pregnancy and baby: Food allergies in babies. http://www.nhs.uk/conditions/pregnancy-and-baby/pages/food-allergies-in-children.aspx (accessed June 23, 2016).
NHS. 2015b. Pregnancy and baby: Your baby’s first solid foods. http://www.nhs.uk/conditions/pregnancy-and-baby/pages/solid-foods-weaning.aspx (accessed July 16, 2016).
Nordlee, J. A., S. L. Taylor, J. A. Townsend, L. A. Thomas, and R. K. Bush. 1996. Identification of a Brazil-nut allergen in transgenic soybeans. N Engl J Med 334(11):688-692.
Norizoe, C., N. Akiyama, T. Segawa, H. Tachimoto, H. Mezawa, H. Ida, and M. Urashima. 2014. Increased food allergy and vitamin D: Randomized, double-blind, placebo-controlled trial. Pediatr Int 56(1):6-12.
Noti, M., B. S. Kim, M. C. Siracusa, G. D. Rak, M. Kubo, A. E. Moghaddam, Q. A. Sattentau, M. R. Comeau, J. M. Spergel, and D. Artis. 2014. Exposure to food allergens through inflamed skin promotes intestinal food allergy through the thymic stromal lymphopoietinbasophil axis. J Allergy Clin Immunol 133(5):1390-1399.
Okupa, A. Y., R. F. Lemanske, Jr., D. J. Jackson, M. D. Evans, R. A. Wood, and E. C. Matsui. 2013. Early-life folate levels are associated with incident allergic sensitization. J Allergy Clin Immunol 131(1):226-228.
Osborn, D. A., and J. Sinn. 2006. Formulas containing hydrolyzed protein for prevention of allergy and food intolerance in infants. Cochrane Database Syst Rev(4):CD003664.
Osborn, D. A., and J. Sinn. 2009. Formulas containing hydrolysed protein for prevention of allergy and food intolerance in infants (Review). Cochrane Database Syst Rev (1): CD003664.
Osborne, N. J., O. C. Ukoumunne, M. Wake, and K. J. Allen. 2012. Prevalence of eczema and food allergy is associated with latitude in Australia. J Allergy Clin Immunol 129(3):865-867.
Ouyang, F., R. Kumar, J. Pongracic, R. E. Story, X. Liu, B. Wang, H. Xing, X. Liu, Z. Li, W. Zhang, Y. Fang, S. Zhang, X. Xu, and X. Wang. 2009. Adiposity, serum lipid levels, and allergic sensitization in Chinese men and women. J Allergy Clin Immunol 123(4):940-948.
Oxelius, V. A., R. Krueger, S. Ahlstedt, T. Keil, S. Lau, and U. Wahn. 2015. Innate IgG molecules and innate B cells expressed by immunoglobulin constant heavy G chain (Fcγ) genetic marker genes are involved in the ‘allergic march’ of IgE sensitization in children. Int Arch Allergy Immunol 166(1):25-29.
Pabst, O., and A. M. Mowat. 2012. Oral tolerance to food protein. Mucosal Immunol 5(3):232-239.
Palm, N. W., R. K. Rosenstein, S. Yu, D. D. Schenten, E. Florsheim, and R. Medzhitov. 2013. Bee venom phospholipase A2 induces a primary type 2 response that is dependent on the receptor ST2 and confers protective immunity. Immunity 39(5):976-985.
Palmer, D. J., T. Sullivan, M. S. Gold, S. L. Prescott, R. Heddle, R. A. Gibson, and M. Makrides. 2012. Effect of n-3 long chain polyunsaturated fatty acid supplementation in pregnancy on infants’ allergies in first year of life: Randomised controlled trial. BMJ 344:e184.
Palmer, D. J., J. Metcalfe, M. Makrides, M. S. Gold, P. Quinn, C. E. West, R. Loh, and S. L. Prescott. 2013. Early regular egg exposure in infants with eczema: A randomized controlled trial. J Allergy Clin Immunol 132(2):387-392.
Panjari, M., J. J. Koplin, S. C. Dharmage, R. L. Peters, L. C. Gurrin, S. M. Sawyer, V. McWilliam, J. K. Eckert, D. Vicendese, B. Erbas, M. C. Matheson, M. L. Tang, J. Douglass, A. L. Ponsonby, T. Dwyer, S. Goldfeld, and K. J. Allen. 2016. Nut allergy prevalence and differences between Asian-born children and Australian-born children of Asian descent: A state-wide survey of children at primary school entry in Victoria, Australia. Clin Exp Allergy 46(4):602-609.
Pele, F., E. Bajeux, H. Gendron, C. Monfort, F. Rouget, L. Multigner, J. F. Viel, and S. Cordier. 2013. Maternal fish and shellfish consumption and wheeze, eczema and food allergy at age two: A prospective cohort study in Brittany, France. Environ Health 12:102.
Perkin, M. R., K. Logan, A. Tseng, B. Raji, S. Ayis, J. Peacock, H. Brough, T. Marrs, S. Radulovic, J. Craven, C. Flohr, and G. Lack. 2016. Randomized trial of introduction of allergenic foods in breast-fed infants. N Engl J Med 374(18):1733-1743.
Peters, R. L., S. C. Dharmage, L. C. Gurrin, J. J. Koplin, A. L. Ponsonby, A. J. Lowe, M. L. Tang, D. Tey, M. Robinson, D. Hill, H. Czech, L. Thiele, N. J. Osborne, and K. J. Allen. 2014. The natural history and clinical predictors of egg allergy in the first 2 years of life: A prospective, population-based cohort study. J Allergy Clin Immunol 133(2):485-491.
Peters, R. L., K. J. Allen, S. C. Dharmage, C. J. Lodge, J. J. Koplin, A. L. Ponsonby, M. Wake, A. J. Lowe, M. L. Tang, M. C. Matheson, and L. C. Gurrin. 2015. Differential factors associated with challenge-proven food allergy phenotypes in a population cohort of infants: A latent class analysis. Clin Exp Allergy 45(5):953-963.
Poole, J. A., K. Barriga, D. Y. Leung, M. Hoffman, G. S. Eisenbarth, M. Rewers, and J. M. Norris. 2006. Timing of initial exposure to cereal grains and the risk of wheat allergy. Pediatrics 117(6):2175-2182.
Praveen, P., F. Jordan, C. Priami, and M. J. Morine. 2015. The role of breast-feeding in infant immune system: A systems perspective on the intestinal microbiome. Microbiome 3:41.
Prescott, S. L., C. Macaubas, B. J. Holt, T. B. Smallacombe, R. Loh, P. D. Sly, and P. G. Holt. 1998. Transplacental priming of the human immune system to environmental allergens: Universal skewing of initial T cell responses toward the Th2 cytokine profile. J Immunol 160(10):4730-4737.
Prescott, S. L., P. Smith, M. Tang, D. J. Palmer, J. Sinn, S. J. Huntley, B. Cormack, R. G. Heine, R. A. Gibson, and M. Makrides. 2008. The importance of early complementary feeding in the development of oral tolerance: Concerns and controversies. Pediatr Allergy Immunol 19(5):375-380.
Prince, B. T., M. J. Mandel, K. Nadeau, and A. M. Singh. 2015. Gut microbiome and the development of food allergy and allergic disease. Pediatr Clin North Am 62(6):1479-1492.
Pyrhonen, K., S. Nayha, L. Hiltunen, and E. Laara. 2013. Caesarean section and allergic manifestations: Insufficient evidence of association found in population-based study of children aged 1 to 4 years. Acta Paediatr 102(10):982-989.
Reichel, H., H. P. Koeffler, A. Tobler, and A. W. Norman. 1987. 1 alpha,25-Dihydroxyvitamin D3 inhibits gamma-interferon synthesis by normal human peripheral blood lymphocytes. Proc Natl Acad Sci U S A 84(10):3385-3389.
Roduit, C., R. Frei, M. Depner, B. Schaub, G. Loss, J. Genuneit, P. Pfefferle, A. Hyvarinen, A. M. Karvonen, J. Riedler, J. C. Dalphin, J. Pekkanen, E. von Mutius, C. Braun-Fahrlander, and R. Lauener. 2014. Increased food diversity in the first year of life is inversely associated with allergic diseases. J Allergy Clin Immunol 133(4):1056-1064.
Roider, E., T. Ruzicka, and J. Schauber. 2013. Vitamin D, the cutaneous barrier, antimicrobial peptides and allergies: Is there a link? Allergy Asthma Immunol Res 5(3):119-128.
Rudders, S. A., J. A. Espinola, and C. A. Camargo, Jr. 2010. North-south differences in US emergency department visits for acute allergic reactions. Ann Allergy Asthma Immunol 104(5):413-416.
Saarinen, K. M., K. Juntunen-Backman, A. L. Jarvenpaa, P. Kuitunen, L. Lope, M. Renlund, M. Siivola, and E. Savilahti. 1999. Supplementary feeding in maternity hospitals and the risk of cow’s milk allergy: A prospective study of 6209 infants. J Allergy Clin Immunol 104(2 Pt 1):457-461.
Sampson, H. A., R. Gerth van Wijk, C. Bindslev-Jensen, S. Sicherer, S. S. Teuber, A. W. Burks, A. E. Dubois, K. Beyer, P. A. Eigenmann, J. M. Spergel, T. Werfel, and V. M. Chinchilli. 2012. Standardizing double-blind, placebo-controlled oral food challenges: American Academy of Allergy, Asthma & Immunology-European Academy of Allergy and Clinical Immunology PRACTALL consensus report. J Allergy Clin Immunol 130(6):1260-1274.
Senechal, H., S. Geny, F. X. Desvaux, M. Busson, C. Mayer, Y. Aron, J. P. Oster, J. C. Bessot, G. Peltre, G. Pauli, and E. Swierczewski. 1999. Genetics and specific immune response in allergy to birch pollen and food: Evidence of a strong, positive association between atopy and the HLA class II allele HLA-DR7. J Allergy Clin Immunol 104(2 Pt 1):395-401.
Shek, L. P., M. F. Chong, J. Y. Lim, S. E. Soh, and Y. S. Chong. 2012. Role of dietary long-chain polyunsaturated fatty acids in infant allergies and respiratory diseases. Clin Dev Immunol 2012:730568.
Sicherer, S. H., T. J. Furlong, H. H. Maes, R. J. Desnick, H. A. Sampson, and B. D. Gelb. 2000. Genetics of peanut allergy: A twin study. J Allergy Clin Immunol 106(1 Pt 1):53-56.
Siegel, A. M., K. D. Stone, G. Cruse, M. G. Lawrence, A. Olivera, M. Y. Jung, J. S. Barber, A. F. Freeman, S. M. Holland, M. O’Brien, N. Jones, C. G. Nelson, L. B. Wisch, H. H. Kong, A. Desai, O. Farber, A. M. Gilfillan, J. Rivera, and J. D. Milner. 2013. Diminished allergic disease in patients with STAT3 mutations reveals a role for STAT3 signaling in mast cell degranulation. J Allergy Clin Immunol 132(6):1388-1396.
Simopoulos, A. P. 2002. The importance of the ratio of omega-6/omega-3 essential fatty acids. Biomed Pharmacother 56(8):365-379.
Simpson, E. L., J. R. Chalmers, J. M. Hanifin, K. S. Thomas, M. J. Cork, W. H. McLean, S. J. Brown, Z. Chen, Y. Chen, and H. C. Williams. 2014. Emollient enhancement of the skin barrier from birth offers effective atopic dermatitis prevention. J Allergy Clin Immunol 134(4):818-823.
Starkl, P., T. Marichal, N. Gaudenzio, L. L. Reber, R. Sibilano, M. Tsai, and S. J. Galli. 2016. IgE antibodies, FceRIα, and IgE-mediated local anaphylaxis can limit snake venom toxicity. J Allergy Clin Immunol 137(1):246-257.
Stelmach, I., M. Bobrowska-Korzeniowska, K. Smejda, P. Majak, J. Jerzynska, W. Stelmach, K. Polanska, W. Sobala, J. Krysicka, and W. Hanke. 2014. Risk factors for the development of atopic dermatitis and early wheeze. Allergy Asthma Proc 35(5):382-389.
Strachan, D. P. 1989. Hay fever, hygiene, and household size. BMJ 299(6710):1259-1260.
Strid, J., J. Hourihane, I. Kimber, R. Callard, and S. Strobel. 2005. Epicutaneous exposure to peanut protein prevents oral tolerance and enhances allergic sensitization. Clin Exp Allergy 35(6):757-766.
Sudo, N., S. Sawamura, K. Tanaka, Y. Aiba, C. Kubo, and Y. Koga. 1997. The requirement of intestinal bacterial flora for the development of an IgE production system fully susceptible to oral tolerance induction. J Immunol 159(4):1739-1745.
Tan, H. T., J. A. Ellis, J. J. Koplin, M. C. Matheson, L. C. Gurrin, A. J. Lowe, P. E. Martin, T. D. Dang, M. Wake, M. L. Tang, A. L. Ponsonby, S. C. Dharmage, and K. J. Allen. 2012a. Filaggrin loss-of-function mutations do not predict food allergy over and above the risk of food sensitization among infants. J Allergy Clin Immunol 130(5):1211-1213.
Tan, T. H., J. A. Ellis, R. Saffery, and K. J. Allen. 2012b. The role of genetics and environment in the rise of childhood food allergy. Clin Exp Allergy 42(1):20-29.
Tey, D., K. J. Allen, R. L. Peters, J. J. Koplin, M. L. K. Tang, L. C. Gurrin, A.-L. Ponsonby, A. J. Lowe, M. Wake, and S. C. Dharmage. 2014. Population response to change in infant feeding guidelines for allergy prevention. J Allergy Clin Immunol 133(2):476-484.
Togias, A., S. F. Cooper, M. L. Acebal, A. Assa’ad, L. Baker, L. A. Beck, J. Block, C. Byrd-Bredbenner, E. S. Chan, L. F. Eichenfield, D. M. Fleischer, G. J. Fuchs III, G. T. Furuta, M. J. Greenhawt, R. S. Gupta, M. Habich, S. M. Jones, K. Keaton, A. Muraro, M. Plaut, L. J. Rosenwasser, D. Rotrosen, H. A. Sampson, L. Schneider, S. H. Sicherer, R. Sidbury, J. Spergel, D. R. Stukus, C. Venter, and J. A. Boyce. 2017. Addendum guidelines for the prevention of peanut allergy in the United States: Report of the National Institute of Allergy and Infectious Diseases–sponsored expert panel. Allergy Asthma Clin Immunol 13(1):1-20.
Torgerson, T. R., A. Linane, N. Moes, S. Anover, V. Mateo, F. Rieux-Laucat, O. Hermine, S. Vijay, E. Gambineri, N. Cerf-Bensussan, A. Fischer, H. D. Ochs, O. Goulet, and F. M. Ruemmele. 2007. Severe food allergy as a variant of IPEX syndrome caused by a deletion in a noncoding region of the FOXP3 gene. Gastroenterology 132(5):1705-1717.
Tsai, H. J., R. Kumar, J. Pongracic, X. Liu, R. Story, Y. Yu, D. Caruso, J. Costello, A. Schroeder, Y. Fang, H. Demirtas, K. E. Meyer, M. R. O’Gorman, and X. Wang. 2009. Familial aggregation of food allergy and sensitization to food allergens: A family-based study. Clin Exp Allergy 39(1):101-109.
Van Gysel, D., E. Govaere, K. Verhamme, E. Doli, and F. De Baets. 2009. Body mass index in Belgian schoolchildren and its relationship with sensitization and allergic symptoms. Pediatr Allergy Immunol 20(3):246-253.
Vassallo, M. F., A. Banerji, S. A. Rudders, S. Clark, and C. A. Camargo, Jr. 2010. Season of birth and food-induced anaphylaxis in Boston. Allergy 65(11):1492-1493.
Venkataraman, D., N. Soto-Ramirez, R. J. Kurukulaaratchy, J. W. Holloway, W. Karmaus, S. L. Ewart, S. H. Arshad, and M. Erlewyn-Lajeunesse. 2014. Filaggrin loss-of-function mutations are associated with food allergy in childhood and adolescence. J Allergy Clin Immunol 134(4):876-882.
Visness, C. M., S. J. London, J. L. Daniels, J. S. Kaufman, K. B. Yeatts, A. M. Siega-Riz, A. H. Liu, A. Calatroni, and D. C. Zeldin. 2009. Association of obesity with IgE levels and allergy symptoms in children and adolescents: Results from the National Health and Nutrition Examination Survey 2005-2006. J Allergy Clin Immunol 123(5):1163-1169.
von Berg, A., B. Filipiak-Pittroff, U. Kramer, E. Link, C. Bollrath, I. Brockow, S. Koletzko, A. Grubl, J. Heinrich, H. E. Wichmann, C. P. Bauer, D. Reinhardt, and D. Berdel. 2008. Preventive effect of hydrolyzed infant formulas persists until age 6 years: Long-term results from the German Infant Nutritional Intervention Study (GINI). J Allergy Clin Immunol 121(6):1442-1447.
von Berg, A., B. Filipiak-Pittroff, U. Kramer, B. Hoffmann, E. Link, C. Beckmann, U. Hoffmann, D. Reinhardt, A. Grubl, J. Heinrich, H. E. Wichmann, C. P. Bauer, S. Koletzko, and D. Berdel. 2013a. Allergies in high-risk schoolchildren after early intervention with cow’s milk protein hydrolysates: 10-Year results from the German Infant Nutritional Intervention (GINI) study. J Allergy Clin Immunol 131(6):1565-1573.
von Berg, A., S. Koletzko, B. Filipiak-Pittroff, J. Heinrich, C. P. Bauer, U. Kramer, B. Hoffmann, and D. Berdel. 2013b. The German Infant Nutritional Intervention (GINI) study and formulation issues. Reply. J Allergy Clin Immunol 132(3):770-771.
Wall, R., R. P. Ross, G. F. Fitzgerald, and C. Stanton. 2010. Fatty acids from fish: The anti-inflammatory potential of long-chain omega-3 fatty acids. Nutr Rev 68(5):280-289.
Wawro, N., J. Heinrich, E. Thiering, J. Kratzsch, B. Schaaf, B. Hoffmann, I. Lehmann, C. P. Bauer, S. Koletzko, A. von Berg, D. Berdel, and J. Linseisen. 2014. Serum 25(OH)D concentrations and atopic diseases at age 10: Results from the GINIplus and LISAplus birth cohort studies. BMC Pediatr 14:286.
Weisse, K., S. Winkler, F. Hirche, G. Herberth, D. Hinz, M. Bauer, S. Roder, U. Rolle-Kampczyk, M. von Bergen, S. Olek, U. Sack, T. Richter, U. Diez, M. Borte, G. I. Stangl, and I. Lehmann. 2013. Maternal and newborn vitamin D status and its impact on food allergy development in the German LINA cohort study. Allergy 68(2):220-228.
West, C. E., J. Dunstan, S. McCarthy, J. Metcalfe, N. D’Vaz, S. Meldrum, W. H. Oddy, M. K. Tulic, and S. L. Prescott. 2012. Associations between maternal antioxidant intakes in pregnancy and infant allergic outcomes. Nutrients 4(11):1747-1758.
West, C. E., M. L. Hammarstrom, and O. Hernell. 2013. Probiotics in primary prevention of allergic disease—Follow-up at 8-9 years of age. Allergy 68(8):1015-1020.
West, C. E., H. Renz, M. C. Jenmalm, A. L. Kozyrskyj, K. J. Allen, P. Vuillermin, and S. L. Prescott. 2015. The gut microbiota and inflammatory noncommunicable diseases: Associations and potentials for gut microbiota therapies. J Allergy Clin Immunol 135(1):3-13.
Wetzig, H., R. Schulz, U. Diez, O. Herbarth, B. Viehweg, and M. Borte. 2000. Associations between duration of breast-feeding, sensitization to hens’ eggs and eczema infantum in one and two year old children at high risk of atopy. Int J Hyg Environ Health 203(1):17-21.
WHO (World Health Organization). 2003. Prevention of allergy and allergic asthma. Based on the WHO/WAO meeting on the prevention of allergy and allergic asthma. Geneva, Switzerland: WHO.
WHO. 2016. Infant and young child feeding. http://who.int/mediacentre/factsheets/fs342/en (accessed June 5, 2016).
Wjst, M. 2008. Allergy risk of vitamin D supplements has been described in various settings. J Allergy Clin Immunol 121(4):1065-1066; author reply 1066.
Wjst, M., and S. Dold. 1999. Genes, factor X, and allergens: What causes allergic diseases? Allergy 54(7):757-759.
Woo, J. G., A. Assa’ad, A. B. Heizer, J. A. Bernstein, and G. K. Hershey. 2003. The –159 C→T polymorphism of CD14 is associated with nonatopic asthma and food allergy. J Allergy Clin Immunol 112(2):438-444.
Xu, B., M. R. Jarvelin, and J. Pekkanen. 2000. Body build and atopy. J Allergy Clin Immunol 105(2 Pt 1):393-394.
Zhang, Y., F. Collier, G. Naselli, R. Saffery, M. L. Tang, K. J. Allen, A. L. Ponsonby, L. C. Harrison, and P. Vuillermin. 2016. Cord blood monocyte-derived inflammatory cytokines suppress IL-2 and induce nonclassic “TH2-type” immunity associated with development of food allergy. Sci Transl Med 8(321):321ra8.
























































































