New Materials for Emerging Desalination Technologies
BAOXIA MI
University of California, Berkeley
DESALINATION AS A SOLUTION TO WATER SHORTAGE
The global water shortage caused by dwindling fresh water resources and increasing water demand, and compounded by extreme climate conditions (less precipitation), has highlighted the importance of treating unconventional waters to ensure sustainable economic and societal growth in water-stressed regions (Shannon et al. 2008).
Desalination, a process that was originally defined as the removal of salts and minerals from saline water but now includes the treatment of brackish and wastewater, likely offers a long-term strategy for augmenting water supply. This technology is widely used in many parts of the world, especially the arid Middle East. For example, Israel has been heavily relying on wastewater reuse and seawater desalination to meet much of its water needs: 86 percent of its wastewater is recycled and 60 percent of its drinking water is produced by desalination. In sharp contrast, the numbers are only 7 percent and <1 percent, respectively, for California, which regularly suffers from severe drought (Stock et al. 2015).
State-of-the-art desalination technologies include (1) thermal processes such as multistage flash and multieffect distillation and (2) membrane-based processes such as reverse osmosis (RO) and electrodialysis. The RO technology, a hydraulic pressure-driven filtration process that removes contaminants from water mainly by size exclusion and charge repulsion, accounts for around 60 percent of the market thanks to its relative advantages in capital cost, energy consumption, and ease of operation. Many other processes—forward osmosis, membrane distillation, capacitive deionization, pressure-retarded osmosis, and enhanced solar evaporation—have recently emerged as attractive alternatives in view of their
promise for reducing operational energy consumption by using sustainable energy sources such as solar, geothermal, or waste heat.
Producing water by desalination at the current development stage is more expensive than treating conventional water sources. For example, the unit cost of RO seawater desalination in the United States is now about $2.0/m3 on average (it may go down to $1.1/m3 when the technology is scaled up), compared to a typical wholesale water price of $0.1 to $0.5/m3 (Wittholz 2008; WaterCAGov 1994). The high cost is mainly because desalination requires the removal of small, soluble contaminants (e.g., salts and inorganic/organic micropollutants such as pharmaceuticals and endocrine-disrupting compounds) that are generally not a concern in conventional water treatment. Additionally, the high salt concentration in seawater imposes a thermodynamic limit of 1.1 kWh/m3 as the theoretical minimum energy consumption at 50 percent recovery, significantly contributing to the cost of seawater desalination.
An important goal in improving desalination technology is to separate target contaminants from water more effectively and energy-efficiently. In particular, the development of high-performance desalination membranes using emerging two-dimensional (2D) nanomaterials may revolutionize membrane-based desalination technology (Stock et al. 2015).
NEW DESALINATION MEMBRANES MADE OF 2D NANOMATERIALS
Membranes made of conventional materials (e.g., polyamide) have inherent limitations in permeability, selectivity, chemical stability, and antifouling properties, severely affecting their separation performance in desalination. Recent advances in 2D nanomaterials offer an opportunity to help overcome these limitations through the fabrication of a new class of filtration membranes for desalination.
Emerging graphene-based nanomaterials possess a unique 2D structure and highly tunable physicochemical properties as well as exceptional mechanical, electrical, and biological characteristics, all of which can be advantageously leveraged to significantly improve the separation efficiency of desalination membranes (Mi 2014). Expected to be on par with carbon nanotube membranes (Holt et al. 2006) and biomimetic aquaporin membranes (Shen et al. 2014) in terms of separation capability, graphene-based membranes are much easier to scale up thanks to both the use of graphite as a low-cost raw material and membrane synthesis via facile, scalable routes.
There are two general types of graphene-based membranes, made via very different approaches and having fundamentally different separation mechanisms. The first is a porous graphene membrane made by punching nanometer pores through the ultrathin, super-strong, and impermeable graphene monolayer, as illustrated in Figure 1(a). With its precisely controlled size and manipulated
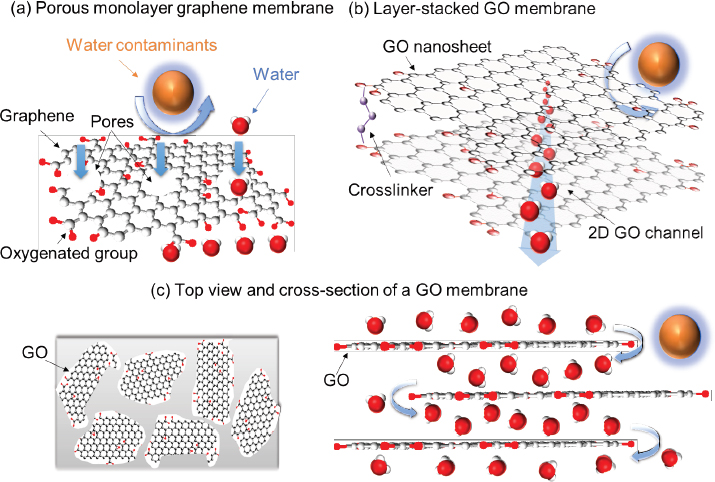
functional groups (which dictate the critical entrance properties) of the punched pores, the nanopore membrane allows only molecules smaller than the pores to permeate while larger molecules are rejected (Cohen-Tanugi and Grossman 2012; Suk and Aluru 2010).
In addition, the single-carbon-atom thickness (~0.3 nm) of this super-strong membrane is almost three orders of magnitude less than the thickness (typically a few hundred nanometers) of traditional desalination membranes, significantly improving the water permeability, which is inversely proportional to membrane thickness. Challenges in making such a monolayer graphene membrane include the enormous difficulties of preparing a large-area, defect-free monolayer graphene sheet and creating high-density pores of controllable, relatively uniform sizes on the graphene sheet.
The second type of graphene-based membrane is made of mass-producible graphene oxide (GO) nanosheets. As illustrated in Figure 1(b), the unique 2D structure of GO nanosheets makes it possible to synthesize a membrane via a simple, scalable layer-stacking technique (Hu and Mi 2013, 2014). The nanochannels formed between the layer-stacked GO nanosheets, functionally equivalent to
nanopores in the monolayer graphene membrane, provide a zigzag water transport path while rejecting unwanted ions and molecules that are larger than the inter-GO-layer spacing (Figure 1c). Simulation and experimental evidence indicate that, because of the very large slip length (i.e., low friction) of water molecules on a graphene surface, water can flow at an extremely high rate in the planar graphene nanochannels (Kannam et al. 2012; Nair et al. 2012), a property that could lead to the formation of highly permeable membranes for desalination.
The layer-stacking synthesis approach also enhances the adjustability of the spacing and functionalities of GO nanosheets to optimize membrane permeability and selectivity. Moreover, the 2D carbon-walled channel surface yields stronger carbon-organic interactions and thus hinders the diffusion of organic molecules in the membrane. As a result, the GO membrane can efficiently remove neutral organic contaminants (Zheng and Mi 2016), a unique feature when compared to traditional polymeric RO membranes, which are typically charged and have a relatively poor removal rate for neutral molecules.
Graphene-based nanomaterials can also be used to modify existing membranes for improved performance or multifunctionality. For example, the semiconducting property of GO nanosheets and their composites (e.g., GO–titanium dioxide) makes GO photoactive under both ultraviolet and visible lights, a useful property for developing photocatalytic membranes (Gao et al. 2014). And GO nanosheet assembly is a convenient way to form a dense barrier layer on the porous side of a traditional asymmetric membrane for fouling control in pressure-retarded osmosis, a desalination-related, energy-production process whose advancement has been hindered by the accumulation of foulants in the porous membrane support (Hu et al. 2016).
MAJOR CHALLENGES IN GO MEMBRANE DEVELOPMENT
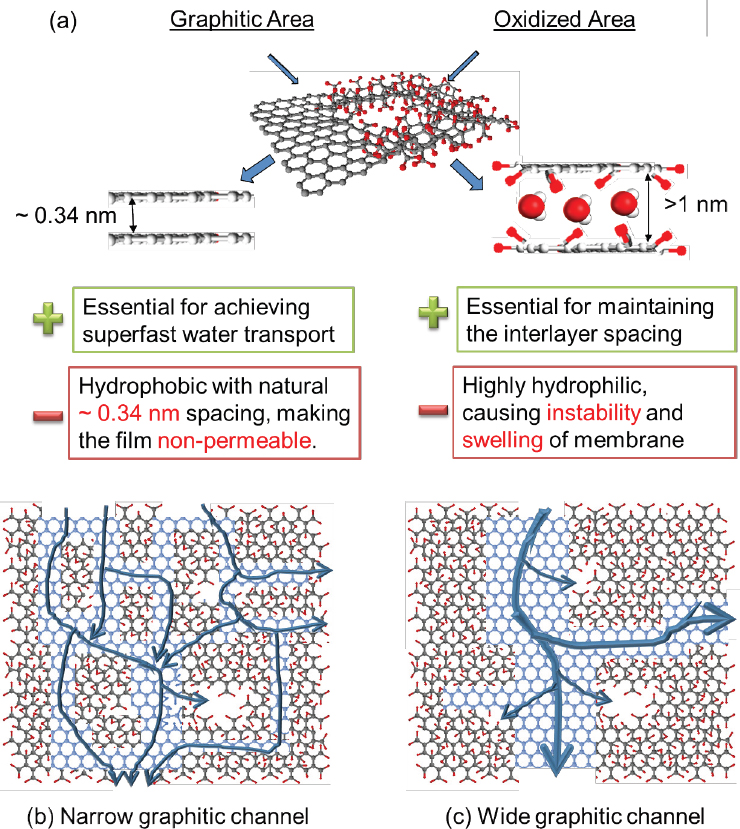
The high water permeability of a GO membrane relies on the hypothetical existence of a continuous, nearly frictionless path for water flow in the extremely smooth graphitic (i.e., nonoxidized) regions of GO nanochannels. But heavily oxidized GO regions, which represent a large portion of the GO basal plane, do not provide a frictionless pathway and could significantly affect water flow.
As illustrated in Figure 2(a), a GO nanosheet is composed of three distinct regions: graphitic, oxidized, and defect, illustrated by a hole in the middle. The graphitic region typically occupies less than half of the total area of a GO nanosheet prepared using the Hummers method (Hummers and Offeman 1958; Marcano et al. 2010). Because graphitic regions even with the same overall area ratio could be distributed quite differently in GO nanosheets, as illustrated in Figure 2(b,c), the resulting water transport paths, boundary effects, and membrane separation capabilities can be dramatically different.
The microstructure of GO nanochannels as well as the associated water and molecular transport mechanisms are not clearly understood. Efforts are needed to
precisely control the size of GO nanochannels, characterize the transport length and channel width, and build mechanistic models to correlate such characteristics to membrane performance.
Controlling the interlayer spacing in a GO membrane is another critical challenge to the manufacture of effective desalination membranes. Studies have shown that it is relatively straightforward to construct a membrane with GO inter-
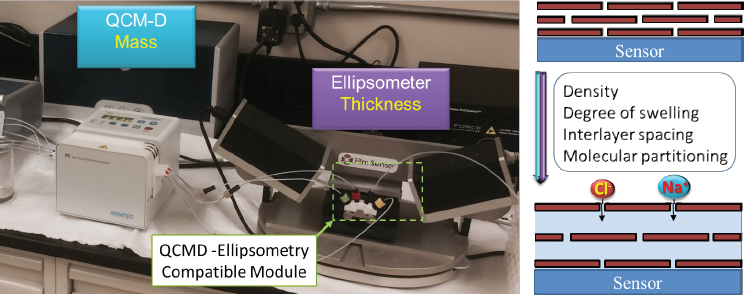
layer spacing of more than 1 nm (Hu and Mi 2013, 2014; Zheng and Mi 2016). But it becomes challenging to reduce the spacing to less than 0.8 nm (a critical value for desalination membranes to achieve high removal of sodium chloride by size exclusion) because the oxidized region in GO starts to create strong hydration forces and charge repulsion that cause membrane swelling and thus increase interlayer spacing. To accurately quantify the degree of swelling and interlayer spacing, a protocol has recently been established to simultaneously measure the mass of GO thin film by quartz crystal microbalance with dissipation (QCM-D) and film thickness by ellipsometer (Figure 3). A GO film can swell to about three times its size when it changes from dry to wet. Potential strategies to overcome such swelling include creating short covalent bonds, crosslinking out of aqueous solution, and inserting appropriately sized spacers between GO layers.
CONCLUDING REMARKS
Notwithstanding the challenges, 2D graphene-based nanomaterials hold great promise for revolutionizing membrane-based desalination technology, thanks to their advantages over traditional materials in enabling the synthesis of a desalination membrane via simple, scalable layer-stacking techniques and in the flexible manipulation of membrane permeability and selectivity for target contaminants. Other 2D materials (e.g., zeolite, molybdenum disulfide), with unique configurations that could help control interlayer spacing, are also attracting research interest in making high-performance membranes (Kang et al. 2014).
In addition, 2D nanomaterials can be innovatively constructed into a 3D
structure and thus function as a nanosized reactor to further enhance membrane selectivity and minimize membrane fouling (Jiang et al. 2015). Finally, it is worth noting that 2D nanomaterials are finding potential applications in nonmembrane-based desalination technologies. For example, 2D graphene material–enabled thin films may be used to enhance solar evaporation (Ghasemi et al. 2014) and thus help desalinate water by using sustainable solar energy.
ACKNOWLEDGMENTS
This article is based on work supported by the National Science Foundation under grant no. CBET-1565452. The opinions expressed are those of the author and do not necessarily reflect those of the sponsor.
REFERENCES
Cohen-Tanugi D, Grossman JC. 2012. Water desalination across nanoporous graphene. Nano Letters 12:3602–3608.
Gao Y, Hu M, Mi B. 2014. Membrane surface modification with tiO2-graphene oxide for enhanced photocatalytic performance. Journal of Membrane Science 455:349–356.
Ghasemi H, Ni G, Marconnet AM, Loomis J, Yerci S, Miljkovic N, Chen G. 2014. Solar steam generation by heat localization. Nature Communications 5.
Holt J, Park H, Wang Y, Stadermann M, Artyukhin A, Grigoropoulos C, Noy A, Bakajin O. 2006. Fast mass transport through sub-2-nanometer carbon nanotubes. Science 312:1034–1037.
Hu M, Mi B. 2013. Enabling graphene oxide nanosheets as water separation membranes. Environmental Science & Technology 47:3715–3723.
Hu M, Mi B. 2014. Layer-by-layer assembly of graphene oxide membranes via electrostatic interaction. Journal of Membrane Science 469:80–87.
Hu M, Zheng S, Mi B. 2016. Organic fouling of graphene oxide membranes and its implications for membrane fouling control in engineered osmosis. Environmental Science & Technology 50:685–693.
Hummers WS, Offeman RE. 1958. Preparation of graphitic oxide. Journal of the American Chemical Society 80:1339–1339.
Jiang Y, Wang W-N, Liu D, Nie Y, Li W, Wu J, Zhang F, Biswas P, Fortner JD. 2015. Engineered crumpled graphene oxide nanocomposite membrane assemblies for advanced water treatment processes. Environmental Science & Technology 49:6846–6854.
Kang Y, Emdadi L, Lee MJ, Liu D, Mi B. 2014. Layer-by-layer assembly of zeolite/polyelectrolyte nanocomposite membranes with high zeolite loading. Environmental Science & Technology Letters 1:504–509.
Kannam SK, Todd BD, Hansen JS, Daivis PJ. 2012. Slip length of water on graphene: Limitations of non-equilibrium molecular dynamics simulations. Journal of Chemical Physics 136.
Marcano DC, Kosynkin DV, Berlin JM, Sinitskii A, Sun ZZ, Slesarev A, Alemany LB, Lu W, Tour JM. 2010. Improved synthesis of graphene oxide. ACS Nano 4:4806–4814.
Mi B. 2014. Graphene oxide membranes for ionic and molecular sieving. Science 343:740–742.
Nair RR, Wu HA, Jayaram PN, Grigorieva IV, Geim AK. 2012. Unimpeded permeation of water through helium-leak-tight graphene-based membranes. Science 335:442–444.
Shannon MA, Bohn PW, Elimelech M, Georgiadis JG, Marinas BJ, Mayes AM. 2008. Science and technology for water purification in the coming decades. Nature 452:301–310.
Shen Y-X, Saboe PO, Sines IT, Erbakan M, Kumar M. 2014. Biomimetic membranes: A review. Journal of Membrane Science 454:359–381.
Stock S, Bott M, Carroll J, Escamilla F. 2015. Solutions to California’s water crisis from half a world away. NBC Bay Area, November 3. Online at http://www.nbcbayarea.com/investigations/Surviving-the-Drought-Solutions-to-California-Water-Crisis-From-Israel-339638362.html.
Suk ME, Aluru NR. 2010. Water transport through ultrathin graphene. Journal of Physical Chemistry Letters 1:1590–1594.
WaterCAGov. Bulletin 166-4, Urban Water Use in California. August 1994. Online at http://www.water.ca.gov/historicaldocs/irwm/b166-1994/ch4.html.
Wittholz MK, O’Neill BK, Colby CB, Lewis D. 2008. Estimating the cost of desalination plants using a cost database. Desalination 229:10–20.
Zheng S, Mi B. 2016. Silica-crosslinked graphene oxide membrane and its unique capability in removing neutral organic molecules from water. Environmental Science: Water Research & Technology 2:717–725.








