1
Introduction
Every system is perfectly designed to get the results it gets.
—Paul Batalden
Federal advice to the public on nutrition and diet is intended to reflect the state of the science and deliver the most reliable recommendations according to the best available evidence. This advice, presented in the Dietary Guidelines for Americans (DGA), underpins all federal nutrition policies and programs and is updated every 5 years. The process to create the guidelines is not a simple one, and it changes as the science evolves. Much has been accomplished to improve how the science is evaluated and translated into the DGA, such as the establishment of the Nutrition Evidence Library to conduct evidence-based reviews. The target population for the DGA will also expand in the 2020–2025 edition to include recommendations for all Americans by including pregnant women and children from birth to 2 years.
Despite the many accomplishments, recent challenges to federal nutrition guidance prompted Congress to question the process by which food and nutrition guidance is developed (Conaway, 2015; Hartzler et al., 2015). To address these complicated questions, Congress mandated a review of the entire process used to develop the DGA.1
___________________
1 Consolidated Appropriations Act, 2016, Public Law 114-113, 114th Cong. (December 18, 2015), 129 Stat. 2280–2281.
THE DIETARY GUIDELINES FOR AMERICANS
The DGA is a report that provides nutritional and dietary information to promote health and prevent disease (HHS/USDA, 2015). To help Americans make healthy food choices, the U.S. Department of Agriculture (USDA) and the U.S. Department of Health and Human Services (HHS) jointly review and update the guidelines every 5 years to reflect “the preponderance of the scientific and medical knowledge which is current at the time the report is prepared.”2 Since the first edition in 1980, the guidelines have served as the basis for all federal nutrition policies and nutrition assistance programs, as well as nutrition education programs (see Box 1-1). The process to develop the guidelines has evolved over time in an effort to develop gold standard guidelines.
The guidelines are developed through a multistep process developed by USDA and HHS. USDA and HHS receive input from a scientific advisory committee, other federal agencies, and the public (see Figure 1-1).
First, a charter is filed with Congress to establish a scientific advisory committee, known as the Dietary Guidelines Advisory Committee (DGAC). The advisory committee comprises nationally recognized experts responsible for independently evaluating the scientific evidence to inform revisions to the current policy or suggest new guidance. Its conclusions are submitted to the secretaries of USDA and HHS as the Scientific Report of the Dietary Guidelines Advisory Committee. The DGAC’s report serves as the scientific basis for the DGA, but its conclusions are advisory in nature only; the scientific report does not constitute draft policy.
The secretaries then solicit comments on the DGAC report from the public and other federal agencies. Next, the DGA writing team—made up of staff from USDA and HHS—collects, assesses, and reviews these comments as it develops the next edition of the DGA. The draft undergoes a series of internal departmental reviews, including reviews by more than 100 subject-matter experts from the federal government, and revisions prior to approval by the secretaries. Finally, the DGA is published publicly with the primary audience being policymakers and health professionals who then implement the guidelines through programs supported by federal, state, and local governments.
When the Scientific Report of the 2015 Dietary Guidelines Advisory Committee was released in February 2015, more than 29,000 written public comments were submitted. Only five of these public comments focused
___________________
2 National Nutrition Monitoring and Related Research Act of 1990, Public Law 101-445, 101st Cong. (October 22, 1990) 7 U.S.C. 5341, 104 Stat. 1042–1044. The departments are required to act within the National Nutrition Monitoring and Related Research Act of 1990, Agricultural Act of 2014, Federal Advisory Committee Act of 1972, and the Consolidated Appropriations Act of 2001 (“Data Quality Act”) (USDA/HHS, 2016a).
on the membership of the DGAC, stating that future DGACs should include food scientists, more registered dietitians, and other health professionals and practitioners (HHS, 2016). Critiques of the report itself and the process used—both in support of and against the conclusions—were raised for a wide range of topics. Comments questioning the report focused largely on the DGAC’s scope with some suggesting that it was too broad. Also questioned were aspects of the process it used to evaluate the scientific evidence. The DGAC was criticized for not having had the charge or expertise to make some of its recommendations, such as those related to sustainable diets and tax policy (Hartzler et al., 2015; Jack, 2016; Kovich, 2016; Merrigan et al., 2015). Questions were also raised regarding the evidence used and the comprehensiveness of the literature reviewed (Dabrowska, 2016; Heimowitz, 2016; Hentges, 2016; Mozzaffarian, 2016; Teicholz, 2015). Following an examination of these public comments, the House Committee on Agriculture held a hearing where the secretaries of USDA and HHS were asked to clarify, among other things, that the DGAC report was based in science and that sustainability concerns were outside the scope of the DGAC. Controversy arose again in January 2016 when the Dietary Guidelines for Americans 2015–2020, Eighth Edition, was released. Those critical of the document focused, among other things, on the process by which the DGAC’s scientific report was translated into the policy document (the DGA) (Walsh, 2016; Willett, 2016).
Other comments were made on February 19, 2016, when USDA invited 40 stakeholders to voice support or concern for the process for developing the DGA. Ten professional organizations were represented, as well as 18 members of the food industry, and 12 individuals with various background and professional associations. Specific to the membership issue of the DGAC, conflict of interest was raised as a prime area of concern among stakeholders who commented on the selection process, with views ranging from no potential members having any ties to the food industry at all, to acceptance of past conflicts of interest. There was general agreement among commenters that the composition of the DGAC should be diversified and expanded to include additional expertise including government, food industry, academia, nongovernmental organizations, and consumer representatives.3 It was noted that no single sector should have undue influence on the selection process. Some stakeholders supported the current selection process, and others offered suggestions such as publicizing selection criteria and any relevant conflicts of interest, as well as using public nominations for selecting the advisory committee (USDA, 2016b).
___________________
3 Specific areas of expertise called for, mentioned by more than three stakeholders, included pregnancy and birth to 24 months, registered dietitians, food industry, consumer behavior, food systems, food technology, nutrition, pediatrics.
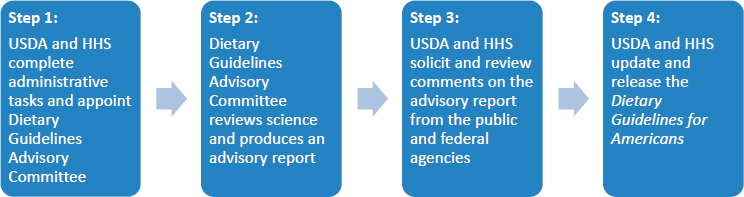
SOURCE: Abstracted from USDA, 2016a.
EVALUATION BY THE NATIONAL ACADEMIES OF SCIENCES, ENGINEERING, AND MEDICINE
In response to concerns raised about the process to produce the 2015–2020 DGA, Congress directed USDA to engage with the National Academies of Sciences, Engineering, and Medicine (the National Academies) to appoint a committee to conduct a comprehensive evaluation of the processes used to establish the DGA (see Box 1-2 for the statement of task). Importantly, the committee is not evaluating the substance of the guidelines or their use; its charge is to assess the process. The questions in the statement of task are to be divided and addressed in two reports.
This current report responds to the first part of the task: “How the advisory committee selection process can be improved to provide more transparency, eliminate bias, and include committee members with a range of viewpoints,” for the purpose of informing the 2020 cycle, which is scheduled to begin in early 2017. The phrase “eliminate bias” is interpreted to mean “minimize bias,” as complete elimination of bias may not be possible; this reframing of the charge was accepted by USDA (USDA/HHS, 2016b).
The findings and conclusions contained herein respond only to the first question in the statement of task; remaining questions will be answered in a second report. Although most of the evidence and analysis related to the other questions will be reserved for the second report, some issues related to the second report had to be included in the present report when needed to address the advisory committee selection process. For example, the Agricultural Act of 2014 requested that the DGA expand to include people across the life span, adding guidance for pregnant women and children from birth to 24 months. Although this expansion is included in the statement of task under parts 2–4, not part 1, this significant change to the DGA—and by extension to the DGAC—may affect the composition
or structure of the DGAC. As part of an overall, comprehensive review of the process to update the DGA, additional findings and recommendations about the selection process may be made as part of this committee’s second report. Additionally, some of the questions relevant to the selection process, such as how specific priority areas are determined and how the DGAC’s conclusions are considered in the final DGA, will be explored in this committee’s second report.
Committee Methods
The National Academies appointed 14 members to the Committee to Review the Process to Update the Dietary Guidelines for Americans to respond to a congressional request.4 To assess the advisory committee selection process, this National Academies committee met in person once and convened in closed session three times. Its discussions also benefited from engaging with the public by holding two open information-gathering sessions (see Appendix B). One public comment session was held, where members of the public were invited to address the committee; those who did make a statement included representatives of industry, professional organizations, and advocacy groups. Additionally, the committee solicited input from the public about what they believed to be the most important change that could be made to the selection process. Statements and comments were received by this National Academies committee from a variety of perspectives (see Appendix B). All statements were considered over the course of the committee’s deliberations.
Chapter 2 of this report reviews the current process for establishing and operating the DGAC, which is required to act within the Federal Advisory Committee Act. The chapter describes the relevant provisions of the act, and how they affect the establishment of the charter, bylaws, and operating procedures.
The committee sought to ground its work in as much evidence as possible. An evaluation was conducted of the processes for convening other advisory committees. The goal of this search was to identify promising practices and to learn from the wide variation of processes used. A full discussion of this search is presented in Chapter 3.
This National Academies committee also reviewed the published literature to explore the role of conflicts of interest in the development of advisory committees and guidelines (see Appendix A for literature search methods). The ultimate goal of limiting and managing conflicts of interest is to develop a trustworthy process and create reliable guidelines. Ideally,
___________________
4 Consolidated Appropriations Act, 2016, Public Law 114-113, 114th Cong. (December 18, 2015), 129 Stat. 2280–2281.
the guidelines, which have been developed with minimal bias, will be trustworthy, leading people to follow the recommendations put forth, which subsequently would lead to improved health outcomes. Management strategies are discussed in more detail in Chapter 4, as well as other opportunities to build trust with the public.
Finally, Chapter 5 discusses the need to continuously update the selection process as new evidence becomes available.
REFERENCES
Conaway, M. 2015. The science in our diet. U.S. News & World Report. http://www.usnews.com/opinion/articles/2015/10/05/why-the-science-behind-the-dietary-guidelines-matters (accessed October 3, 2017).
Dabrowska, A. 2016. Dietary Guidelines for Americans: Frequently asked questions. R44360. Washington, DC: Congressional Research Service.
FDA (U.S. Food and Drug Administration). 2016. Changes to the Nutrition Facts label. http://www.fda.gov/Food/GuidanceRegulation/GuidanceDocumentsRegulatoryInformation/LabelingNutrition/ucm385663.htm (accessed October 3, 2017).
Hartzler, V., M. Conaway, J. Walorski, K. Noem, and D. Webster. 2015. Letter from Members of Congress to the Honorable Tom Vilsack, Secretary of Agriculture and the Honorable Sylvia Mathews Burwell, Secretary of Health and Human Services (March 31, 2015). http://agriculture.house.gov/uploadedfiles/ag_dietaryguidelineslettertosecsvilsackburwell.pdf (accessed October 3, 2017).
Heimowitz, C. 2016 (unpublished). Comments presented at USDA Dietary Guidelines for Americans listening sessions: Atkins Nutritionals. Washington, DC, February 19, 2016.
Hentges, E. 2016 (unpublished). Comments presented at USDA Dietary Guidelines for Americans listening sessions: International Life Sciences Institute. Washington, DC, February 19, 2016.
HHS (U.S. Department of Health and Human Services). 2016. Public comments on the scientific report of the 2015 Dietary Guidelines Advisory Committee. https://health.gov/dietaryguidelines/dga2015/comments/advisory-report-summary.asp (accessed October 4, 2017).
HHS/USDA (U.S. Department of Health and Human Services/U.S. Department of Agriculture). 2015. Dietary Guidelines for Americans 2015–2020: Eighth edition. https://health.gov/dietaryguidelines/2015/guidelines (accessed October 4, 2017).
Jack, M. 2016 (unpublished). Comments presented at USDA Dietary Guidelines for Americans listening sessions: American Beverage Association. Washington, DC, February 19, 2016.
Kovich, D. 2016 (unpublished). Comments presented at USDA Dietary Guidelines for Americans listening sessions: National Pork Producers Council. Washington, DC, February 19, 2016.
Massachusetts Department of Public Health. 2012. Massachusetts State Agency Food Standards: Requirements and recommendations. http://www.mass.gov/eohhs/docs/dph/mass-in-motion/eo509-state-agency-food-standards.pdf (accessed October 4, 2017).
Massachusetts Executive Office of Health and Human Services. 2016. Tools and resources for implementation of Executive Order 509. http://www.mass.gov/eohhs/gov/departments/dph/programs/community-health/mass-in-motion/about-mim/components/tools-and-resources-for-executive-order-509.html (accessed October 4, 2017).
Merrigan, K., T. Griffin, P. Wilde, K. Robien, J. Goldberg, and W. Dietz. 2015. Designing a sustainable diet. Science 350(6257):165-166.
Mozzaffarian, D. 2016 (unpublished). Comments presented at USDA Dietary Guidelines for Americans listening sessions. Washington, DC, February 19, 2016.
Teicholz, N. 2015. The scientific report guiding the US dietary guidelines: Is it scientific? British Medical Journal 351:h4962.
USDA (U.S. Department of Agriculture). 2016a (unpublished). Understanding the committee’s charge: A discussion with the sponsor. Presentation to the Committee to Review the Process to Update the Dietary Guidelines for Americans.
USDA. 2016b. Dietary Guidelines for Americans Listening Sessions: Transcript. https://www.cnpp.usda.gov/sites/default/files/dietary_guidelines_for_americans/ListeningSessionTranscript-2-19-16.pdf (accessed October 4, 2017).
USDA/HHS (U.S. Department of Agriculture/U.S. Department of Health and Human Services). 2016a (unpublished). Dietary Guidelines for Americans: Process brief, sections 1–3. Prepared for the Committee to Review the Process to Update the Dietary Guidelines for Americans.
USDA/HHS. 2016b (unpublished). HMD follow-up questions for USDA. Response to Committee to Review the Process to Update the Dietary Guidelines for Americans.
Walsh, D. 2016 (unpublished). Comments presented at USDA Dietary Guidelines for Americans listening sessions: Snack Food Association. Washington, DC, February 19, 2016.
Washington State Department of Health. 2014. Healthy nutrition guidelines: Implementation guide for agencies, sites and vendors. Healthy Communities. (DOH 340-224).
Willett, W. 2016 (unpublished). Comments presented at USDA Dietary Guidelines for Americans listening sessions. Washington, DC, February 19, 2016.










