5
Service Delivery
Part of the challenge of eliminating hepatitis B and C in the United States is that the people suffering from or at risk for the infections are often not engaged in care and can be difficult to reach. Many hepatitis B patients are born abroad; they may not be comfortable with the U.S. health system or with English-speaking providers (Derose et al., 2009; Hacker et al., 2015). Hepatitis C is common among people who inject drugs, for whom hepatitis treatment is often not a high priority and who feel discriminated against in medical settings (Grebely et al., 2015; Mehta et al., 2011; Treloar et al., 2010). Both conditions are associated with feelings of shame and anxiety, which can be antithetical to pursuing treatment. For these reasons, the national hepatitis elimination strategy must give as much attention to the delivery of essential services as the services themselves. This piece of the strategy, what the World Health Organization (WHO) describes as “the how,” considers steps that can be taken to make viral hepatitis a priority, support efficient care, and reach patients who might otherwise slip through the cracks (WHO, 2016). Overseeing such efforts should be the responsibility of the central coordinating office described in Chapter 2. This chapter discusses steps that could help improve the reach of essential hepatitis services, thereby hastening the end of hepatitis B and C in the United States.
ENCOURAGING COMPLIANCE AMONG PROVIDERS
The previous chapter discusses essential interventions for elimination of viral hepatitis in the United States. As crucial as these interventions are,
they can easily be neglected in the routine practice of medicine. National survey data suggest that fewer than half of Asian Americans are tested for hepatitis B virus (HBV), for example (Hu et al., 2013). The Advisory Committee on Immunization Practices (ACIP) recommends that all infants be vaccinated against HBV before leaving the hospital, but only 72.4 percent are immunized within 3 days of birth (95 percent confidence interval [CI] 70.9 to 73.9), with state and local coverage varying from fewer than half in Vermont to nearly 90 percent in North Dakota (Hill et al., 2015; Mast et al., 2005).
There is often a gap between the practice of medicine as recommended by experts and what actually happens. Closing this gap is of concern to the National Committee for Quality Assurance (NCQA), which in the 1990s, devised a set of indicators meant to hold health plans accountable for results (Marwick, 1997; NCQA, n.d.-b). These indicators, called HEDIS,1 are now instrumental for monitoring performance in 90 percent of American health plans (NCQA, n.d.-a). HEDIS indicators are standardized, as is the method for collecting and verifying the HEDIS data that pertain to “the most pressing clinical areas” (NCQA, 2016a). Viral hepatitis is surely one of these areas, and the addition of a few relevant HEDIS indicators would greatly aid the national elimination effort.
Recommendation 5-1: The National Committee for Quality Assurance should establish measures to monitor compliance with viral hepatitis screening guidelines and hepatitis B vaccine birth dose coverage and include the new measures in the Healthcare Effectiveness Data and Information Set.
HEDIS measures compel a certain attention from providers and health plan managers because they are used to evaluate performance (NCQA, 2012). Their role in changing clinical practice can be pronounced, especially for common conditions for which there is an effective treatment but wide variability in care (Eddy et al., 2008). HEDIS measures have improved the management of hypertension (Jaffe et al., 2013). Mathematical models indicate that compliance with HEDIS for diabetes mellitus and cardiovascular disease would have prevented almost 2 million myocardial infarctions, 800,000 strokes, and 100,000 cases of end-stage renal disease between 1995 and 2005 alone (Eddy et al., 2008).
At the same time, HEDIS indicators put a burden on the health system. Emphasizing a short list of clinical actions risks creating a hierarchy wherein some services matter more than other equally meaningful ones. There are also logistical constraints. Some health plans do not have enough
___________________
1 Officially, the Healthcare Effectiveness Data and Information Set.
members eligible for a given service to allow for a valid comparison of compliance (McGlynn, 1997). In selecting HEDIS indicators, NCQA considers feasibility, or the trade-off between comprehensiveness and practicality required to track an indicator (McGlynn, 1997). The indicator’s scientific soundness is also taken into account, as is its relevance, with preference “given to those areas where better performance will enhance the health of the population” (McGlynn, 1997, p. 15). With these criteria in mind, the committee suggests screening and prevention measures that would benefit from NCQA’s attention.
Adherence to Screening Guidelines
HBV and hepatitis C virus (HCV) are, for the most part, clinically silent infections. Fewer than a third of chronic hepatitis B patients in the United States are aware of their condition (Lin et al., 2007); about half of those with chronic hepatitis C are (Denniston et al., 2012). Diagnosis is an obvious prerequisite to any kind of service delivery. To this end, the U.S. Preventive Services Task Force recommends HBV screening for everyone at high risk of chronic infection, including people born in HBV-endemic countries, and all pregnant women during the first trimester of pregnancy (USPSTF, 2014, 2015). Similarly, the task force recommends that people at high risk of HCV infection be screened, as should anyone born between 1945 and 1965 (USPSTF, 2013). NCQA has HEDIS measures on screening for breast, cervical, and colorectal cancers, as well as chlamydia screening in young women and lead screening in children (NCQA, 2016b). Inclusion in HEDIS makes screening more of a priority for health plan managers. In order to improve chlamydia screening in adolescent girls, Kaiser of Northern California devised a system of team meetings and clinical tools to improve their screening rates (Shafer et al., 2002). About 46 percent of a sample of 37 Pennsylvania health plans implemented reminder and tracking systems in response to the introduction of HEDIS measures for colorectal screening, eliciting a 54 percentage point increase in the screening rate (Sarfaty and Myers, 2008).
A point-of-care test could facilitate wider screening. A recent systematic review and meta-analysis concluded that reliable, affordable point-of-care tests for HCV core antigen have a sensitivity and specificity greater than 90 and 98 percent respectively (Freiman et al., 2016). Core antigen tests are likely to be cheaper than traditional nucleic acid tests and may, therefore, facilitate wider use (Freiman et al., 2016). The Food and Drug Administration has not approved any point of care HBsAg tests for use in the United States, although some of the tests that are used in other countries have been found to be highly accurate (Gish et al., 2014; Njai et al., 2015; Shivkumar et al., 2012). Simple to use, point of care assays for HBsAg are used in
HBV-endemic countries, but manufacturers have little incentive to seek market authorization for such a product in the United States (Lin et al., 2008; WHO, 2001). It is possible that the addition of a HEDIS indicator on hepatitis screening would encourage manufacturers to reconsider this position. Regardless of what assay is used, an increase in screening would translate into vastly fewer undiagnosed viral hepatitis patients.
The Hepatitis B Vaccine Birth Dose
NCQA includes various measures of child and adolescent immunization as HEDIS indicators, including full immunization against HBV during the first 2 years of life (NCQA, 2009). HEDIS and Centers for Disease Control and Prevention (CDC) evaluations indicate that over 90 percent of children meet this goal, though misclassification is not uncommon (Bundy et al., 2012). This indicator does not take into account the relative importance of the timing of the first dose, however. In 2016, ACIP recommended all infants receive the first dose of the hepatitis B vaccine within 24 hours of birth (Chitnis, 2016; Jenco, 2016). Children born to HBsAg+ women or to women who have never been tested for HBV require vaccination within 12 hours of birth, and others within 24 hours (CDC, 2016b,d; Chitnis, 2016; Mast et al., 2005). The proportion of children receiving the vaccine in the first 3 days of life has improved steadily, from 53.2 percent in 2007 (95 percent CI: 51.9 to 54.5) to 72.4 percent in 2014 (95 percent CI: 70.9 to 73.9) (CDC, 2012a; Hill et al., 2015).
Starting the hepatitis B vaccine series at birth is associated with better odds of completing the three-dose series on time (Yusuf et al., 2000). Full immunization, in turn, conveys lifelong immunity to 95 percent of those vaccinated (CDC, 2016c). Emphasizing early vaccination would show a commitment to ending the vertical transmission of HBV. The risk of chronic hepatitis B is highest among those infected in early life. Up to 90 percent of people exposed to HBV in infancy and 30 percent of those exposed in early childhood develop chronic hepatitis B (Zhao and Murphy, 2013). Birth dose immunization is the first step in preventing these infections. By emphasizing the hepatitis B birth dose, as well as the completion of the three-dose series, NCQA would direct the attention of hospital and health plan administrators to this essential intervention.
REACHING PATIENTS
The viral hepatitis patient is at the center of any elimination campaign. As time passes, meeting the goals set out in Chapter 2 will depend on diagnosing and treating progressively more challenging patients. A system to manage viral hepatitis patients must include the uninsured and
underinsured, people born abroad, those with mental and behavioral health problems, as well as people in prison and in unstable housing. Integrated, comprehensive primary and specialty care will be essential to the elimination effort (HHS, 2017). Making better use of primary care is one way to support this goal.
Building Capacity in Primary Care
One of the limitations of the hepatitis C models discussed in Chapter 2 is the assumption, based on published estimates, that only 260,000 hepatitis C patients could be treated every year between now and 2030. The model draws attention to this bottleneck, and the trade-off between reducing incidence of new infections or a greater reduction in liver-related deaths. Both would be possible if more patients could be treated every year. To this end, primary care providers need to be in a position to take on hepatitis C patients.
Primary care providers often build trusting relationships with their patients over the course of years (Baron, 2010). Some evidence suggests that certain kinds of patients, those with a substance use disorder for example, have better success in primary care than in specialist clinics (Bruggmann and Grebely, 2015; Bruggmann and Litwin, 2013). Primary care is also an efficient way to provide services, one the American Academy of Family Physicians describes as “the first point of entry and continuing focal point for all needed health services” (AAFP, n.d.). Viral hepatitis services should be no exception. But traditional models of primary care do not, for the most part, leave providers with much room to take on more work. Over 80 percent of primary care physicians responding to the 2016 Survey of America’s Physicians described their practice as overextended or at full capacity (The Physicians Foundation, 2016).
Treating viral hepatitis in primary care also poses risks that providers in small practices may be reluctant to accept. Hepatitis patients are usually healthy; only about 20 to 25 percent of chronic hepatitis C patients will develop cirrhosis after two or three decades of infection (Hepatitis C Online, 2016). Similarly, more than half of all chronic hepatitis B patients will never develop any life-threatening complications (McMahon, 2009). But in a subset of patients with either infection, progression to cirrhosis and cancer can be quick. There is no good way to distinguish one kind of patient from the other early on (Hepatitis C Online, 2016; McMahon, 2009). If fibrosis progresses quickly, or if a patient develops cirrhosis, management becomes much more complicated. Cirrhotics need frequent hospitalization—about three times per person per year—usually for hepatic encephalopathy, fluid overload, and gastrointestinal hemorrhage (Ge and Runyon, 2016; Volk et al., 2012). Although such events are rare, their management may exceed
the resources of solo practitioners in small towns or rural areas. As a result, viral hepatitis care remains out of reach for millions of patients in rural and underserved communities (Mellinger and Volk, 2013).
The University of New Mexico’s ECHO2 program aimed to reduce this disparity and make treatment for complex diseases more accessible in rural and underserved areas (Project ECHO, n.d.-d) (see Box 5-1). The program started in 2003, when hepatitis C was treated with pegylated interferon, a drug with serious side effects that required complicated medical management (Arora et al., 2011). Even so, primary care providers in the ECHO
___________________
2 Officially, Extension for Community Healthcare Outcomes.
program saw no higher rates of serious adverse events than specialists at the University of New Mexico did (Arora et al., 2011).
There are also transferable lessons from the management of other chronic conditions in primary care. Like hepatitis B and C, depression is a serious, chronic health problem, especially for older adults, many of whom have problems with adherence to treatment (Unützer et al., 2002b). Since 1999, the University of Washington’s IMPACT3 program has worked with primary care providers to improve outcomes among elderly people with depression and other complex health needs (Unützer et al., 2002b). IMPACT teams can include family doctors, nurses, pharmacists, social workers, be-
___________________
3 Officially, Improving Mood–Promoting Access to Collaborative Treatment.
havioral health specialists, and various occupational and physical therapists (Hern et al., 2013; Tracy et al., 2013). The program makes good use of the internet for data management and monitoring treatment response over time (Unützer et al., 2002a,b). Patient records are kept on a secure, encrypted website in a database that is programmed to remind clinicians about followup or possible lapses in treatment. This feature is thought to account for the program’s high rate of keeping patient assessments on schedule (Unützer et al., 2002a). The data management system encourages ongoing monitoring of both process and outcome measures, something that would be equally useful in viral hepatitis care.
Recommendation 5-2: The American Association for the Study of Liver Diseases and the Infectious Diseases Society of America should partner with primary care providers and their professional organizations to build capacity to treat hepatitis B and C in primary care. The program should set up referral systems for medically complex patients.
There can be no elimination of viral hepatitis without trained providers who can manage the infections from the acute phase to cure (with HCV) or lifelong suppression (with HBV). Large-scale replication of the ECHO program’s success is possible, especially with support from IDSA4 and AASLD5. The program should give attention to both training and technological tools to ease collaboration. There is precedent for this level of collaboration in the management of other chronic diseases, and it is something that the Health Resources and Services Administration (HRSA) has shown support for, as with its AIDS Education and Training Centers, a feature of the Ryan White program, discussed later in this chapter. In an effort to ensure standard training across the country, IDSA and AASLD might consider developing a joint guideline, with a training curriculum, for primary care providers to use. It is generally easier for both patients and primary care providers to treat hepatitis C without a time-consuming referral process. Primary care providers have an incentive to learn new skills, especially when these are skills they can use to help patients who have problems accessing specialty care. Specialists, for their part, may find participation in a collaborative program not only an interesting educational project, but rewarding, as it allows them to reach more patients then they would in their regular practice. The rapid expansion of ECHO programs to 110 medical centers in 21 countries is evidence of the demand for such collaboration.
For the most part, the medicines used to treat viral hepatitis today have mild side effects (CDC, 2016e; VA, 2016); primary care doctors routinely
___________________
4 Officially, the Infectious Diseases Society of America.
5 Officially, the American Association for the Study of Liver Diseases.
write prescriptions for far more toxic treatments. Perhaps for this reason, specialists are often not clear how much involvement is sought or appropriate in collaboration with primary care providers (Mellinger and Volk, 2013). In the ECHO program, primary providers have predictable, weekly teleconferences with teams of specialists at an academic medical center to confer on problems such as management of cirrhosis (Arora et al., 2007). Such consults, made possible with Skype and other videoconferencing programs, help avoid ambiguity and keep lines of communication open among the involved providers.
Using phone and video conferencing (called telehealth or telemedicine) can facilitate the management of hepatitis in primary care, but telehealth laws and reimbursement practices vary widely by state (Klink et al., 2015). Of 1,557 family doctors responding to a Robert Graham Center survey, 89 percent agreed that telehealth can improve their patients’ access to care, but reimbursement, liability, and training were seen as barriers to its use (Klink et al., 2015). Collaborations for viral hepatitis treatment would need to take this into account, setting up consultation networks within single states, for example. In addition to providing a system for teaching and communication, technology allows primary care and specialty providers a way to share information, similar to the internet-based clinical information system that IMPACT provides (Patel et al., 2014; Unützer et al., 2002a).
The primary goal of conferencing with specialists should be training primary care providers to manage most of the patients themselves. To this end, telehealth, as modeled in the ECHO program (that is, more a teaching tool then a patient management one) should be encouraged. As mentioned earlier, cirrhotic patients are complicated. Adherence to practice guidelines for treatment of cirrhotics is generally better among specialists (Kanwal et al., 2012; Mellinger and Volk, 2013). Provider training would need to clarify the signs and symptoms of cirrhosis and ensure that primary care providers have a way to confirm diagnosis and refer appropriately.
An information system accessible to both specialists and primary providers (similar to the one used in the IMPACT example) helps track patients over time and facilitates shared decision making (Kvamme et al., 2001). Compatible electronic systems also encourage communication between providers, who may be separated by considerable distance (Kvamme et al., 2001; Patel et al., 2014). Electronic records alone will not necessarily improve communication, however. Even in the Department of Veterans Affairs, which uses a unified patient record system, analysis of patients’ records suggests unexplained breaks in communication about 10 percent of the time (Singh et al., 2011). AASLD and IDSA should ensure that their training gives clear guidelines on everyone’s role in the collaboration, including expectations for responsiveness (Mellinger and Volk, 2013).
Clarifying roles will apply not just to the specialists and the primary
care provider. Success in managing complex conditions in primary care depends on the cooperation of teams of health workers (Thornton et al., 2016). Educating patients on treatment adherence has long been the role of pharmacists, one that became better and more deliberately integrated into hepatitis C treatment when pegylated interferon was the standard treatment (Spooner, 2011). Nurses are also well positioned for patient counseling, and are often better at it than doctors, especially as it pertains to chronic diseases, such as hepatitis B, and behavioral change, which can be part of hepatitis C care (Bodenheimer and Bauer, 2016). Team-based care also requires support from new types of health professionals, such as care coordinators, who can have more of the routine responsibility for the logistics of clinic visits and for check-ins to help avoid hospitalization (Ge and Runyon, 2016). With more staff working to manage any patient’s case, it becomes important to organize patient information and have it accessible to the care team, as in the IMPACT program.
Inclusion of Special Populations
Viral hepatitis often affects people who are hard to reach: people who were born abroad, who are underinsured, who have substance use disorders and may have other mental health problems, and who are or have been imprisoned (NASEM, 2016). Hepatitis B and C cannot be eliminated without reaching these populations, something that requires extra effort. This effort is often unrewarded. The time and resources needed to coordinate care for such patients is not accurately captured in the Medicare and Medicaid reimbursement system, which has a bias toward in-patient care and paying for clinical services (Martyn and Davis, 2014). This imbalance could undermine the viral hepatitis elimination program.
The people with the most serious need for health care, including those who are poor or have behavioral health problems, rarely have a single office to coordinate their services (Blumenthal and Abrams, 2016; Druss and Walker, 2011). Without a single entity responsible for managing care, the process can disintegrate. Case management may be the key to avoiding such problems (Johnson et al., 2016). More holistic care keeps high-risk patients engaged with the health system over time (Martyn and Davis, 2014). The right strategy to achieve such care may vary by setting and by the particular group. In any case, bringing hepatitis services to challenging populations will be an integral part of hepatitis elimination, and as such it warrants more explicit attention from the federal and state agencies involved, as well as from various local and community organizations.
The Ryan White Comprehensive AIDS Resources Emergency Act of 1990 (hereafter, the Ryan White Act) was passed in response to a similar problem with HIV services in uninsured and underinsured people (HRSA,
2016a). HRSA works with state and local health departments to administer the program, tailoring services to meet local needs (HRSA, 2015). The Ryan White Act provides a key safety net for vulnerable groups (Sood et al., 2014). It also provides states with grants to improve the organization of HIV care and support services (HRSA, 2016a). The specific services supported are chosen by state or territorial authorities in consideration of local needs, but can include case management, transportation, and language interpretation (HRSA, 2016c). A system of the same flexibility and breadth would be needed to reach the marginalized populations suffering from viral hepatitis.
Recommendation 5-3: The Department of Health and Human Services should work with states to build a comprehensive system of care and support for special populations with hepatitis B and C on the scale of the Ryan White system.
The Ryan White program gives states incentives to reach vulnerable patient groups, something that would greatly benefit viral hepatitis elimination efforts. The committee recognizes that building a parallel program with the reach of the Ryan White Act for viral hepatitis might not be feasible; at best it is not within the control of any health department. It may, therefore, be most efficient to try to build outreach activities for viral hepatitis onto existing Ryan White programs, albeit using separate funding for services for HIV-negative people.
The Ryan White Act already reaches viral hepatitis patients who also have HIV, the vast majority of whom have a history of injection drug use (Grebely et al., 2013; Thomas et al., 2011). In Rhode Island, for example, an HIV clinic used Ryan White funds to improve adherence to hepatitis C treatment among people who inject drugs (Taylor, 2005). Some states use their Ryan White program to treat substance use disorder (Arkansas Department of Health, n.d.; Baltimore City Health Department, n.d.; Fischer, 2012; Honeck and Dolansky, 2011; Kansas Department of Health and Environment, n.d.; West Virginia Department of Health and Human Resources, n.d.). Between 2011 and 2014, HRSA sponsored 29 programs to treat hepatitis C in Ryan White clients (Doshi and Tinsley, 2015). In all of these instances, however, the beneficiaries have both HIV and viral hepatitis. Currently the terms of the Ryan White Act and HRSA’s AIDS Drug Assistance program are clear that funding may not be used for broader prevention activities or for services for people not infected with HIV (HRSA, n.d.-a).
It would take time for the Department of Health and Human Services (HHS) to implement a Ryan White like program for hepatitis B and C. In the meantime, the CDC and HRSA would do well to build more flexibility
into their grant structure to allow state and local jurisdictions to support viral hepatitis prevention and linkage to care. Especially when it comes to testing and counseling, states should be allowed to build on existing infrastructure developed for HIV.
There are also viral hepatitis patient groups who do not overlap with the Ryan White population. Federally qualified health centers (FQHCs) may be an efficient way to reach these groups. The Bureau of Primary Health Care awards FQHC designation to organizations that offer comprehensive health services to underserved populations with a sliding fee scale (HRSA, n.d.-b). The HHS report The Community Response to Viral Hepatitis highlighted the work of FQHCs in educating and screening high-risk groups, as well as in developing strategies to ensure patients are retained in care (HHS, 2016). The centers also have good reach into rural areas, as shown in Figure 5-1. Box 5-2 describes the hepatitis and substance use programming of an FQHC that works with homeless people in Boston.
Between 2008 and 2016 the CDC supported state viral hepatitis prevention coordinators in five cities (Chicago, Houston, Los Angeles, New York, and Philadelphia) and all states except South Dakota (CDC, 2012b).
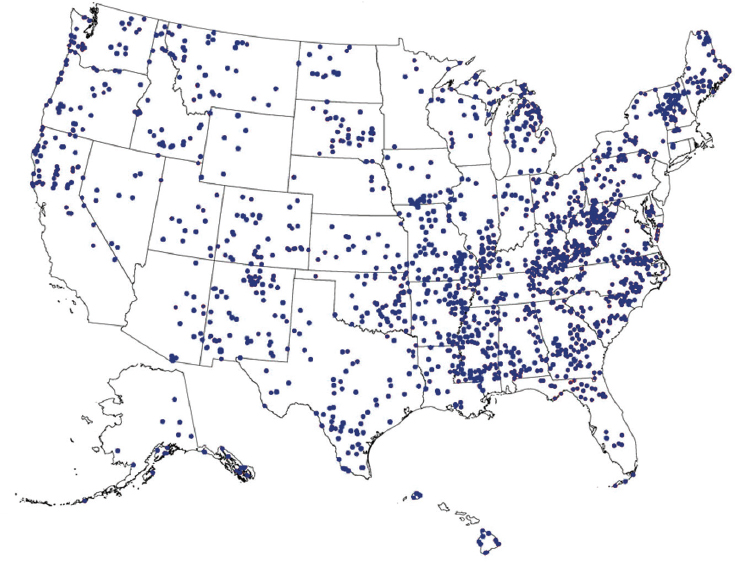
NOTE: Alaska and Hawaii not to scale.
SOURCES: HRSA, 2016b; Rural Health Information Hub, 2016.
The coordinators would be an invaluable part of any effort to bring viral hepatitis services to a wider population. In Hawaii, for example, the coordinator’s office developed hepatitis B education materials in the Chuukese, Ilocano, Marshallese, Samoan, and Tongan languages (CDC, 2012b). In Maine, they supported free immunization sites at sexually transmitted disease clinics, prisons, a clinic for homeless people, and five FQHCs (CDC, 2012b). However, in 2016, the CDC redirected the program that supported the viral hepatitis prevention coordinators toward improving testing and linkage at federally qualified health centers and safety-net hospitals and
their affiliated clinics, and away from specific support of a hepatitis coordinator position.
Behavioral health problems often contribute to the challenge of reaching people with hepatitis, making it important to support them with a range of services, such as appointment reminders, help with insurance forms, transportation to clinics, and addiction counseling. Barriers to accessing and continuing in care fall into four main categories: logistical and child care; coordination, meaning the overall difficulty of navigating the health system; individual, referring to the patient’s own mental and physical health problems that can impede functioning; and systemic, such as poverty and stigma (Broeckaert and Challacombe, 2015).
Defining the Special Populations for Viral Hepatitis Elimination
Some patients are less accessible than others. While it is not always easy to predict who will need more attention or supportive services, there are certain populations whose relative isolation and high burden of viral hepatitis make them essential targets for any hepatitis elimination campaign. Some situations worthy of concentrated outreach are described below (in no particular order).
Cultural and language barriers People born in HBV-endemic countries account for about 95 percent of new cases of chronic hepatitis B in the United States (Hu, 2008; Mitchell et al., 2011). Among the 929 Asian Americans in the NHANES6 study, the prevalence of chronic hepatitis B is around 3 percent, 10 times higher than in the general population (Roberts et al., 2016). Among women of childbearing age, the prevalence of chronic hepatitis B was 8.9 percent among people born in Asia, compared to 0.14 percent among Asian Americans born in the United States (Smith et al., 2012). For comparison, an estimated 0.71 percent of African Americans, 0.08 percent of whites, and 0.03 percent of Hispanics have chronic hepatitis B (Smith et al., 2012). By some estimates, Asian Americans comprise more than half of the country’s chronic hepatitis B cases (CDC, 2013). Nevertheless, only 35 to 60 percent of Asian Americans have been screened for HBV (CDC, 2013; Cohen et al., 2008; Hutton et al., 2007; Roberts et al., 2016; Sarpel et al., 2016).
Screening for HBV at Asian American cultural events is a strategy to identify more infected people, but one that has yielded mixed results (Hyun et al., 2016; Woo et al., 2013). Some evidence suggests that connecting the HBsAg+ patient to care is the bigger challenge (Hyun et al., 2016; Tran, 2009). Hepatitis B carries a pronounced stigma in some Asian communities
___________________
6 Officially, the National Health and Nutrition Examination Survey.
(Tran, 2009; Yoo et al., 2012). In a discussion of barriers to hepatitis B treatment, Tram Tran described a Taoist preference for the natural course of things and a complementary Hmong belief in predestination and “indifference toward suffering” (Tran, 2009). She also cited the isolation of not having an English speaking family member older than 14 in a household (Tran, 2009). By this measure, almost half of Vietnamese immigrant households and more than one-third of Chinese, Hmong, Korean, and Taiwanese ones are isolated (Tran, 2009).
Health workers from the same ethnic and language groups as the target patients can help expand the reach of hepatitis B services. Such health workers have been shown to improve knowledge of hepatitis B among Chinese and Cambodian Americans, but with less effect on testing behavior (Taylor et al., 2009, 2013). Other research among Asian Americans has shown that reminders from lay health workers can motivate vaccination and linkage to care (Hyun et al., 2016; Juon et al., 2016). An ongoing randomized trial is exploring the use of patient navigators and mobile messaging to improve care for Asian Americans with chronic hepatitis B (Chak et al., 2016).
Homeless or unstable housing People who live on the street or in shelters, single room occupancy facilities, or transitional housing even occasionally are at elevated risk of death, about a three to nine times higher risk after controlling for age (Baggett et al., 2013; Hibbs et al., 1994; Zivanovic et al., 2015). At any given time, there are over half a million such people in the United States, though the biannual survey used to count them is biased in ways that invariably underestimate the true population (The Economist, 2016; The National Alliance to End Homelessness, 2016). Homeless people have twice the odds (as compared to housed people) of having their medical needs go untreated (Lebrun-Harris et al., 2013).
A meta-analysis of data from seven countries on the burden of infectious disease in homeless people found hepatitis C to be more common than HIV or tuberculosis (Beijer et al., 2012). In the United States, almost a third of homeless people are thought to have HCV antibody, though published estimates range from 7.5 to 52.5 percent (Edlin et al., 2015). A study in Los Angeles found that only about 30 percent of homeless, HCV-infected persons had been diagnosed and informed of their infection, though other research from the same group suggested the same proportion diagnosed as in the general population (Gelberg et al., 2012; Stein et al., 2012).
Managing hepatitis care for people in unstable housing is challenging, but recent promising examples in Boston suggest it is possible. Boston Health Care for the Homeless, the FQHC described in Box 5-2 recently developed a protocol for treating hepatitis C in its clients (Barocas et al., 2017). After 12 weeks, 97 percent had sustained virologic response (Barocas et al., 2017). The program made use of care coordinators and
nurses to manage the patients’ paperwork, appointments, and adherence to treatment (Beiser, 2016). Outreach workers can also be helpful in building connections with this population and encouraging the use of services (Zlotnick et al., 2013). Provider teams may also find mobile phones useful for staying in contact with transient patients (Asgary et al., 2016; McInnes et al., 2014).
Lack of stable housing can be both a cause and an effect of substance use, itself a risk factor for HBV and HCV infection (Aidala et al., 2005; Tompsett et al., 2013; Zivanovic et al., 2015). Outreach to people in unstable housing may, therefore, overlap with harm reduction and substance use treatment programs.
Ongoing substance use disorders and mental health problems The Substance Abuse and Mental Health Services Administration estimates that 43.6 million American adults have a mental illness that interferes with life activities; for about 4 percent of them the illness is considered severe, meaning it substantially limits life activities (SAMHSA, 2015). Substance use is also common, affecting 21.5 million Americans older than 12, about 8 percent of the population (SAMHSA, 2015). People diagnosed with a drug disorder often have comorbid mental health problems; compared to someone without a drug problem, they are about twice as likely to have mood (e.g, bipolar, major depressive disorder) or anxiety disorders (e.g, phobias, panic disorder) (Conway et al., 2006; NIDA, 2010).
Injection drug use is the behavioral health problem most closely associated with hepatitis B and C (CDC, 2016f). In the United States, almost three-quarters of people who inject drugs have HCV antibody; older estimates put the prevalence of HBsAg in the same group around 12 percent, roughly consistent with worldwide estimates (Grebely et al., 2015; Nelson et al., 2011). People who inject drugs can be challenging patients. They often have logistical problems making appointments, and some distrust the health system (Taylor, 2005). There is evidence that these patients do better in primary care than specialty clinics, especially if the providers treat them with compassion and respect and make an effort to build trust (Reimer and Haasen, 2009). Patient management may also require the collaboration of social workers and different kinds of providers (Bruggmann and Grebely, 2015; Bruggmann and Litwin, 2013; Hill et al., 2008).
One promising strategy for reaching this group is to treat hepatitis C in addiction clinics, involving a hepatologist or infectious disease specialist when necessary (Bruggmann and Grebely, 2015; Bruggmann and Litwin, 2013; Reimer and Haasen, 2009). Even in the days of pegylated interferon therapy, a far worse-tolerated course of treatment than direct-acting antivirals, methadone maintenance and addiction clinics had good success at treating HCV infection (Harris et al., 2010; Wilkinson et al., 2009).
Though a recent WHO review found the quality of the evidence regarding the effectiveness of most strategies to prevent viral hepatitis in people who inject drugs to be low, it encouraged syringe exchange and HBV immunization, especially with a high-dose, accelerated vaccine regimen (Walsh et al., 2014).
As discussed in Chapter 4, the opioid epidemic is drawing attention to substance use problems, including those in rural areas and small towns, and among people younger than 35, who are difficult to engage in care (Altarum Institute, 2013). Reaching these patients, and all people with substance use and mental health problems, requires sensitivity and effort; organizations working closely with the target patients may be in the best position to facilitate this.
Recently incarcerated Prisoners are often poor, and many have a history of substance use disorder. Data from the Bureau of Justice Statistics suggest a median annual income for recently incarcerated men of less than $20,000 in 2014 dollars, for recently incarcerated women of less than $14,000 (Rabuy and Kopf, 2015). During incarceration, inmates are dropped from Medicaid. A 2014 survey of medical directors found that fewer than 20 percent follow CDC recommendations for discharge planning of inmates (Solomon et al., 2014). It can take months after release to re-enroll even for the most motivated beneficiary (Solomon et al., 2014). Easing the ability of prisoners to re-enroll in Medicaid or private insurance can smooth the transition from prison life.
More than 90 percent of prisoners re-enter civilian society (Bushway, 2006; Neate, 2016; Rich et al., 2014). Release is a time of heightened risk for the inmate. In the first 2 weeks after release, the former prisoner’s risk of death is almost 13 times higher than that of controls of the same age, sex, and race (Binswanger et al., 2007). Among people with a history of injection drug use (the same patients at highest risk of hepatitis B and C), death from overdose is about 12 times more likely (Binswanger et al., 2007). Viral hepatitis and accompanying substance use problems should be addressed as much as possible in the highly structured prison environment, for reasons discussed later in this chapter. Nevertheless, most prisoners, especially those with hepatitis B, will need help to ensure continued medical care.
Underinsured The Commonwealth Fund estimates that 31 million Americans are underinsured, meaning that their out-of-pocket health costs are more than 10 percent of their household income (or 5 percent of household income for people under 200 percent of the federal poverty line) (Collins et al., 2015). This includes a fifth of people with insurance from their employers, and slightly more than a fifth of Medicaid beneficiaries (Collins et al., 2015). But by far the most represented group among the
underinsured is Medicare beneficiaries (people over 65 or disabled), who account for more than 40 percent of the total (Collins et al., 2015). This group has considerable burden of hepatitis B and C; the CDC estimates that people 55 and older account for over a quarter of chronic hepatitis B infections and almost 40 percent of those with hepatitis C (CDC, 2016f).
Underinsured people are more likely to forgo medical care when they need it (Collins et al., 2015; McCarthy, 2015). Forgoing hepatitis care may be particularly tempting, as HBV and HCV infections are both clinically silent until their late stages (Chen and Morgan, 2006; Post et al., 2011). The underinsured are also a less easily distinguishable group then some other high-risk patients. It is not usually obvious which patients in a practice are near poverty, on a fixed income, or have a high deductible health plan. On the other hand, outreach to underinsured patients is at least possible. By definition, all of them have some contact with the health system. With this in mind, a medical social worker or patient navigator might determine which patients in a practice were vulnerable to forgoing care and act to prevent it.
The Opportunity to Treat Incarcerated Patients
CDC estimates from the early 2000s put the prevalence of hepatitis C in jails and prisons between 12 and 35 percent, and the prevalence of chronic HBV infection between 1 and 3.7 percent (Weinbaum et al., 2003). A 2015 survey by the American Correctional Association concluded that relatively few prison systems collect the information necessary to measure prevalence, but among those that do, estimates of hepatitis C prevalence range from 8 to 10 percent on the low end and 17 percent at the high end (Maurer and Gondles, 2015). A recent, small survey in Wisconsin is representative of the lower end of this distribution, finding about 8 percent of inmates had chronic hepatitis C, and 6 percent had chronic HBV infection (Stockman et al., 2016). The northeast may have a higher disease burden. Research among Pennsylvania inmates between 2004 and 2012 found about 18 percent had HCV antibody (Larney et al., 2014). The authors did not report prevalence of chronic hepatitis C, but given spontaneous cure rates of one-fifth to one-third, a Pennsylvania prison prevalence of about 12 to 15 percent is plausible. Similarly, a 2014 study of New York City jail records found a 20 percent prevalence of HCV antibody, suggesting a similar 14 to 16 percent prevalence of chronic hepatitis C (Akiyama et al., 2016). All this is broadly consistent with Lancet estimates putting the prevalence of chronic hepatitis B in North American prisoners between 0.3 and 3.1 percent, and HCV antibody between 13.1 and 17.7 percent (Dolan et al., 2016).
Even at the low end of the prevalence distribution, prisoners bear a
disproportionate burden of viral hepatitis. Unprotected sex and needle sharing, both risk factors for viral hepatitis, are common in correctional facilities (Macalino et al., 2004; Rubin, 2016). Therefore, prisons are sometimes seen as an amplifying reservoir for viral hepatitis (Macalino et al., 2004). The irony of this problem is that correctional facilities are, at the same time, ideal venues in which to test and vaccinate against hepatitis B and to test for and treat hepatitis C. Directly observed therapy is the norm and risk of drug diversion is low. Prisoners are disproportionately poor and male, a group often out of contact with the health system (Fox et al., 2005; Rabuy and Kopf, 2015; Weinbaum et al., 2005).
A 6-week course of ledipasvir-sofosbuvir has been shown effective at treating acute hepatitis C and well-tolerated by even the sickest patients (Deterding et al., 2016). Treating hepatitis C in its earliest stages reduces the duration of the disease and its associated morbidities, and could help limit the spread of hepatitis C in high-risk populations such as prisoners (Deterding et al., 2016). Still, only half of prison systems responding to an American Correctional Association survey reported having a clinical guideline regarding treatment of hepatitis C (Maurer and Gondles, 2015). The success of the elimination effort may well depend on reaching imprisoned patients, more than 90 percent of whom re-enter the general population (Bushway, 2006; Neate, 2016; Rich et al., 2014). Returning these inmates to their communities vaccinated against HBV and cured of hepatitis C would be an invaluable step toward elimination.
The expense of testing, vaccination, and treatment may be a limiting facor for state correctional officers (Maurer and Gondles, 2015; Weinbaum et al., 2005). Strategies to defray the costs of treatment in prisons, especially at the state and local levels, are discussed in Chapter 6.
Recommendation 5-4: The criminal justice system should screen, vaccinate, and treat hepatitis B and C in correctional facilities according to national clinical practice guidelines.
Having sufficient staff to test prisoners for viral hepatitis and, depending on the result, manage prevention or treatment programs can be a problem for the correctional system. Less competitive salaries and the challenges of the work environment make it difficult to recruit health professionals to prison jobs; 90 percent of respondents to a 2016 survey of state and federal prisons and large jails reported such problems (Gondles et al., 2017). The Department of Justice review found adequate medical staffing in only about 25 percent of federal prisons (DOJ, 2016). Correctional facilities usually fill gaps by contracting with local providers (Ellis, 2009). Most state prisons also use a combination of on- and off-site providers (Chari et al., 2016). There are no comprehensive data on providers in local prisons and jails, but
the use of local providers and correctional medical corporations appears to be common in jails (Shalev et al., 2011). For-profit companies may also provide about half of medical care for state and local correctional facilities (Von Zielbauer, 2005).
It is not realistic to expect any jail or prison to employ the full range of health providers their inmates may need. Off-site provider visits are necessary, but add expense and logistical complications; a guard must accompany an inmate traveling to a clinic, for example. Telehealth for specialist visits to inmates is a particularly useful strategy in correctional facilities (Schiff, 2014). Only 30 of the 45 states responding to the 2016 National Survey of Prison Health Care reported using telemedicine, however (Chari et al., 2016). Concerns about malpractice and the legality of using providers licensed in other states may be preventing some correctional health officers from using telemedicine (NCCHC, 2016). The National Commission on Correctional Health Care position supports telemedicine in correctional facilities, allowing that all providers must be properly licensed in the inmate’s state, and that alternative arrangements be made for urgent and emergency care and for in-person exams when necessary (NCCHC, 2016).
The previous section discussed the efficient use of telemedicine to support primary care providers in managing hepatitis patients. The same can be done to support primary care providers who manage viral hepatitis patients in correctional facilities. The use of telemedicine is consistent with recommendations from the national commission, and a modest majority of state prisons are already moving toward phone and video health consultations (NCCHC, 1998; Schaenman et al., 2013). Viral hepatitis elimination may provide an impetus to make better use of these services for incarcerated patients.
Hepatitis B in Correctional Facilities
Prisons and jails have a constant rotation of inmates, sometimes in close quarters. The mixing of people and opportunities for disease transmission make immunization a priority for correctional health officers (Sequera et al., 2013). Hepatitis B can be a particular risk, motivating the British government’s target of 80 percent of inmates being vaccinated against HBV in the first month of incarceration (Sequera et al., 2013; U.K. Department of Health, 2009). Fewer than half of British prisons reported meeting this target in a recent survey, however (U.K. Government, 2011).
Immunization in jails and prisons has proven challenging in the United States as well. A 2000 survey of 35 states and the Federal Bureau of Prisons found only Texas and Michigan routinely offered hepatitis B immunization to prisoners (Charuvastra et al., 2001). Twenty-six prison systems said they would vaccinate more widely if the vaccine were free, though nine
states indicated that free hepatitis B vaccine would not be enough because of staffing and infrastructure problems (Charuvastra et al., 2001). More recent survey data suggest that about two-thirds of state prisons (covering about 84 percent of state inmates) have a hepatitis B vaccination policy; in most the policy is to vaccinate at the inmate’s request or because of a risk factor for HBV acquisition (BJS, 2004). Even these policies may be observed mostly in the breach. Of the 190 prisons reporting a universal hepatitis B immunization policy, 80 percent had not vaccinated anyone in the year before the survey (BJS, 2004; Weinbaum et al., 2005).
The 2016 National Survey of Prison Care found that 32 of 45 participating states offered hepatitis B testing to incoming inmates, but 21 of those tested only if the inmate had had a clinical indication for it (Chari et al., 2016). Wider attention to testing could draw attention to the related question of immunization. If many inmates are shown to be vulnerable to infection, prison health officers might be able to make a stronger case to their state authorities for support of hepatitis B immunization.
Hepatitis C in Correctional Facilities
In 2003, the CDC recommended screening all inmates with a history of injection drug use for hepatitis C (Macalino et al., 2005; Weinbaum et al., 2003). In prisons with a relatively lower prevalence of HCV, screening only inmates who report certain risk factors may be adequate. Research in Wisconsin found that screening inmates with a history of injection drug use and those born between 1945 and 1965 would correctly identify about 92 percent of patients with a history of HCV infection (Stockman et al., 2016). In higher prevalence settings, opt-out screening may be necessary. In Rhode Island, over 65 percent of male and 44 percent of female inmates with a history of HCV infection did not report any prior injection drug use (Macalino et al., 2005). In California, where 43 percent of inmates reported a history of injection drug use, other notable risks for HCV infection were having a sexual relationship with a man who injects drugs and age, with older people and those imprisoned longer having elevated risk of infection (Fox et al., 2005). Pennsylvania researchers also found that screening inmates based only on risk factors for infection would have missed about three-quarters of people with HCV infection (Kuncio et al., 2015).
Concerns about adequacy of follow-up may have prevented correctional officials from pursuing wider screening in the past (Kuncio et al., 2015). Such concerns remain challenging, but the drug purchasing arrangements discussed in Chapter 6 should give prison systems access to an affordable, steady drug supply. The use of telemedicine to link inmates to specialists will also be helpful when necessary, and evidence suggests that
primary care providers have no higher rate of adverse events in treating hepatitis C than specialists do (Baker et al., 2014; Ho et al., 2015, 2016).
REFERENCES
AAFP (American Academy of Family Physicians). n.d. Primary care. http://www.aafp.org/about/policies/all/primary-care.html (accessed October 27, 2016).
AHRQ (Agency for Healthcare Research and Quality). n.d. Project ECHO: Bringing specialty care to rural New Mexico. https://healthit.ahrq.gov/ahrq-funded-projects/transforming-healthcare-quality-through-health-it/project-echo-bringing (accessed November 2, 2016).
Aidala, A., J. E. Cross, R. Stall, D. Harre, and E. Sumartojo. 2005. Housing status and HIV risk behaviors: Implications for prevention and policy. AIDS and Behavior 9(3):251-265.
Akiyama, M. J., F. Kaba, Z. Rosner, H. Alper, R. S. Holzman, and R. MacDonald. 2016. Hepatitis C screening of the “birth cohort” (born 1945–1965) and younger inmates of New York City jails. American Journal of Public Health 106(7):1276-1277.
Altarum Institute. 2013. Hepatitis C virus infection in young persons who inject drugs. Altarum Institute. https://www.aids.gov/pdf/hcv-and-young-pwid-consultation-report.pdf (accessed December 29, 2016).
Arkansas Department of Health. n.d. HIV/Ryan White Part B services program. http://www.healthy.arkansas.gov/programsServices/infectiousDisease/hivStdHepatitisC/Pages/HIVServices.aspx (accessed October 25, 2016).
Arora, S., C. M. Geppert, S. Kalishman, D. Dion, F. Pullara, B. Bjeletich, G. Simpson, D. C. Alverson, L. B. Moore, D. Kuhl, and J. V. Scaletti. 2007. Academic health center management of chronic diseases through knowledge networks: Project ECHO. Academic Medicine 82(2):154-160.
Arora, S., K. Thornton, G. Murata, P. Deming, S. Kalishman, D. Dion, B. Parish, T. Burke, W. Pak, J. Dunkelberg, M. Kistin, J. Brown, S. Jenkusky, M. Komaromy, and C. Qualls. 2011. Outcomes of treatment for hepatitis C virus infection by primary care providers. New England Journal of Medicine 364(23):2199-2207.
Arora, S., K. Thornton, M. Komaromy, S. Kalishman, J. Katzman, and D. Duhigg. 2014. Demonopolizing medical knowledge. Academic Medicine 89(1):30-32.
Asgary, R., B. Sckell, A. Alcabes, R. Naderi, A. Schoenthaler, and G. Ogedegbe. 2016. Rates and predictors of uncontrolled hypertension among hypertensive homeless adults using New York City shelter-based clinics. Annals of Family Medicine 14(1):41-46.
Baggett, T., S. Hwang, J. O’Connell, B. Porneala, E. Stringfellow, and J. Orav. 2013. Mortality among homeless adults in Boston: Shifts in causes of death over a 15-year period. JAMA Internal Medicine 173(3):189-195.
Baker, D., M. Alavi, A. Erratt, S. Hill, A. Balcomb, R. Hallinan, S. Siriragavan, D. Richmond, J. Smart, J. Keats, N. Doong, P. Marks, J. Grebely, and G. J. Dore. 2014. Delivery of treatment for hepatitis C virus infection in the primary care setting. European Journal of Gastroenterology & Hepatology 26(9):1003-1009.
Baltimore City Health Department. n.d. Ryan White program. http://health.baltimorecity.gov/hiv-std-services/ryan-white-program (accessed October 25, 2016).
Barocas, J. A., M. Beiser, C. Leon, M. Ingemi, P. McCabe, L. Cardoso, B. P. Linas, and J. O’Connell. 2017. Real-world outcomes of HCV treatment in homeless and marginally housed adults. PowerPoint presentation at Conference on Retroviruses and Opportunistic Infections in Seattle, WA, February 13-16, 2017. Received February 23, 2017. Available by request from the National Academies of Sciences, Engineering, and Medicine Public Access Records Office. For more information, email PARO@nas.edu.
Baron, R. J. 2010. What’s keeping us so busy in primary care? A snapshot from one practice. New England Journal of Medicine 362(17):1632-1636.
Beijer, U., A. Wolf, and S. Fazel. 2012. Prevalence of tuberculosis, hepatitis C virus, and HIV in homeless people: A systematic review and meta-analysis. The Lancet Infectious Diseases 12(11):859-870.
Beiser, M. 2016 (unpublished). Enabling hepatitis C cure for individuals experiencing homelessness. PowerPoint presentation, Boston Health Care for the Homeless Program, June 2, 2016. https://www.nhchc.org/wp-content/uploads/2016/06/enabling-hepatitis-c-cure-for-individuals-experiencing-homelessness-beiser-1.pdf (accessed February 23, 2017).
Binswanger, I. A., M. F. Stern, R. A. Deyo, P. J. Heagerty, A. Cheadle, J. G. Elmore, and T. D. Koepsell. 2007. Release from prison—A high risk of death for former inmates. New England Journal of Medicine 356(2):157-165.
BJS (Bureau of Justice Statistics). 2004. Hepatitis testing and treatment in state prisons. Bureau of Justice Statistics Special Report NCJ 199173C. http://www.bjs.gov/content/pub/pdf/httsp.pdf (accessed September 23, 2016).
Blumenthal, D., and M. K. Abrams. 2016. Tailoring complex care management for high-need, high-cost patients. JAMA 316(16):1657-1658.
Bodenheimer, T., and L. Bauer. 2016. Rethinking the primary care workforce—An expanded role for nurses. New England Journal of Medicine 375(11):1015-1017.
Boston Health Care for the Homeless Program. n.d.-a. Hepatitis C services. https://www.bhchp.org/specialized-services/hepc-consult-service (accessed December 7, 2016).
Boston Health Care for the Homeless Program. n.d.-b. History. https://www.bhchp.org/about/history (accessed December 7, 2016).
Broeckaert, L., and L. Challacombe. 2015. Does multidisciplinary care improve health outcomes among people living with HIV and/or HCV? A review of the evidence. Prevention in Focus. Fall. http://www.catie.ca/en/pif/fall-2015/does-multidisciplinary-care-improve-health-outcomes-among-people-living-hiv-andor-hcv- (accessed November 14, 2016).
Bruggmann, P., and J. Grebely. 2015. Prevention, treatment and care of hepatitis C virus infection among people who inject drugs. International Journal on Drug Policy 26(Suppl 1):S22-S26.
Bruggmann, P., and A. H. Litwin. 2013. Models of care for the management of hepatitis C virus among people who inject drugs: One size does not fit all. Clinical Infectious Diseases 57(Suppl 2):S56-S61.
Bundy, D. G., B. S. Solomon, J. M. Kim, and M. R. Miller. 2012. Accuracy and usefulness of the HEDIS childhood immunization measures. Pediatrics 129(4):648-656.
Bushway, S. D. 2006. The problem of prisoner (re)entry. Review of When prisoners come home: Parole and prisoner reentry, Joan Petersilia; But they all come back: Facing the challenges of prisoner reentry, Jeremy Travis; Prisoner reentry and crime in America, Jeremy Travis, Christy Visher. Contemporary Sociology 35(6):562-565.
CDC (Centers for Disease Control and Prevention). 2012a. National, state, and local area vaccination coverage among children aged 19-35 months—United States, 2011. Morbidity and Mortality Weekly Report 61:689-696.
CDC. 2012b. Viral hepatitis prevention initiative: 5-years of accomplishments 2008-2012: Putting viral hepatitis on the map. PS 08-801. CDC, National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention.
CDC. 2013. Asian Americans and hepatitis B. https://www.cdc.gov/features/aapihepatitisb (accessed January 11, 2017).
CDC. 2016a. Chronic liver disease/cirrhosis mortality by state: 2014. http://www.cdc.gov/nchs/pressroom/sosmap/liver_disease_mortality/liver_disease.htm (accessed November 2, 2016).
CDC. 2016b. Hepatitis B. http://www.cdc.gov/vaccines/pubs/pinkbook/hepb.html (accessed November 3, 2016).
CDC. 2016c. Hepatitis B: Hepatitis B vaccine. http://www.cdc.gov/vaccines/pubs/pinkbook/hepb.html#vaccine (accessed November 7, 2016).
CDC. 2016d. Perinatal transmission. http://www.cdc.gov/hepatitis/hbv/perinatalxmtn.htm (accessed November 3, 2016).
CDC. 2016e. Possible side-effects from vaccines: Hepatitis B vaccine side-effects. http://www.cdc.gov/vaccines/vac-gen/side-effects.htm#hepb (accessed November 7, 2016).
CDC. 2016f. Viral hepatitis surveillance: United States, 2014. http://www.cdc.gov/hepatitis/statistics/2014surveillance/pdfs/2014hepsurveillancerpt.pdf (accessed September 22, 2016).
Chak, E. W., S. Sarkar, and C. Bowlus. 2016. Improving healthcare systems to reduce healthcare disparities in viral hepatitis. Digestive Diseases and Sciences 61(10):2776-2783.
Chari, K. A., A. E. Simon, C. J. DeFrances, and L. Maruschak. 2016. National survey of prison health care: Selected findings. National Health Statistics Reports (96):1-23.
Charuvastra, A., J. Stein, B. Schwartzapfel, A. Spaulding, E. Horowitz, G. Macalino, and J. D. Rich. 2001. Hepatitis B vaccination practices in state and federal prisons. Public Health Reports 116(3):203-209.
Chen, S. L., and T. R. Morgan. 2006. The natural history of hepatitis C virus (HCV) infection. International Journal of Medical Sciences 3(2):47-52.
Chitnis, D. 2016. ACIP approves change to hepatitis B vaccination guidelines. Pediatric News, MDedge. http://www.mdedge.com/pediatricnews/article/116025/vaccines/acip-approves-change-hepatitis-b-vaccination-guidelines (accessed November 21, 2016).
Cohen, C., A. A. Evans, W. T. London, J. Block, M. Conti, and T. Block. 2008. Underestimation of chronic hepatitis B virus infection in the United States of America. Journal of Viral Hepatitis 15(1):12-13.
Collins, S. R., P. W. Rasmussen, S. Beutel, and M. M. Doty. 2015. The problem of under-insurance and how rising deductibles will make it worse. Findings from the Commonwealth Fund Biennial Health Insurance Survey, 2014. Issue Brief (Commonwealth Fund) 13:1-20.
Conway, K. P., W. Compton, F. S. Stinson, and B. F. Grant. 2006. Lifetime comorbidity of DSM-IV mood and anxiety disorders and specific drug use disorders: Results from the National Epidemiologic Survey on Alcohol and Related Conditions. Journal of Clinical Psychiatry 67(2):247-257.
Denniston, M. M., R. M. Klevens, G. M. McQuillan, and R. B. Jiles. 2012. Awareness of infection, knowledge of hepatitis C, and medical follow-up among individuals testing positive for hepatitis C: National Health and Nutrition Examination Survey 2001-2008. Hepatology 55(6):1652-1661.
Derose, K. P., B. W. Bahney, N. Lurie, and J. J. Escarce. 2009. Review: Immigrants and health care access, quality, and cost. Medical Care Research and Review 66(4):355-408.
Deterding, K., C. D. Spinner, E. Schott, T. M. Welzel, G. Gerken, H. Klinker, U. Spengler, J. Wiegand, J. S. Zur Wiesch, A. Pathil, M. Cornberg, A. Umgelter, C. Zollner, S. Zeuzem, A. Papkalla, K. Weber, S. Hardtke, H. von der Leyen, A. Koch, D. von Witzendorff, M. P. Manns, and H. Wedemeyer. 2016. Ledipasvir plus sofosbuvir fixed-dose combination for 6 weeks in patients with acute hepatitis C virus genotype 1 monoinfection (HepNet Acute HCV IV): An open-label, single-arm, phase 2 study. The Lancet Infectious Diseases 17(2):215-222.
DOJ (Department of Justice). 2016. Review of the Federal Bureau of Prisons’ medical staffing challenges. DOJ, Office of the Inspector General, Evaluation and Inspections Division 16-02.
Dolan, K., A. L. Wirtz, B. Moazen, M. Ndeffo-mbah, A. Galvani, S. A. Kinner, R. Courtney, M. McKee, J. J. Amon, L. Maher, M. Hellard, C. Beyrer, and F. L. Altice. 2016. Global burden of HIV, viral hepatitis, and tuberculosis in prisoners and detainees. Lancet 388(10049):1089-1102.
Doshi, R. K., and M. Tinsley. 2015. Hepatitis C initiative in Ryan White clinics—Findings from a special program of national significance. https://blog.aids.gov/2015/10/hepatitis-c-initiative-in-ryan-white-clinics-findings-from-a-special-program-of-national-significance.html (accessed October 19, 2016).
Druss, B. G., and E. R. Walker. 2011. Mental disorders and medical comorbidity. Research synthesis report no. 21. The Synthesis Project and Robert Wood Johnson Foundation.
Eddy, D. M., L. G. Pawlson, D. Schaaf, B. Peskin, A. Shcheprov, J. Dziuba, J. Bowman, and B. Eng. 2008. The potential effects of HEDIS performance measures on the quality of care. Health Affairs 27(5):1429-1441.
Edlin, B. R., B. J. Eckhardt, M. A. Shu, S. D. Holmberg, and T. Swan. 2015. Toward a more accurate estimate of the prevalence of hepatitis C in the United States. Hepatology 62(5):1353-1363.
Ellis, A. 2009. BOP health care: What you (and your clients) need to know. Criminal Justice 23(4).
Fischer, L. 2012. Colorado’s Ryan White screening, brief intervention, and referral to treatment collaborative project to address substance use in HIV/AIDS case management and health-care settings. Addiction Science & Clinical Practice 7(Suppl 1):A73.
Fox, R. K., S. L. Currie, J. Evans, T. L. Wright, L. Tobler, B. Phelps, M. P. Busch, and K. A. Page-Shafer. 2005. Hepatitis C virus infection among prisoners in the California state correctional system. Clinical Infectious Diseases 41(2):177-186.
Freiman, J. M., T. M. Tran, S. G. Schumacher, L. F. White, S. Ongarello, J. Cohn, P. J. Easterbrook, B. P. Linas, and C. M. Denkinger. 2016. Hepatitis C antigen testing for diagnosis of hepatitis C virus infection: A systematic review and meta-analysis. Annals of Internal Medicine 165(5):345-355.
Gaeta, J., B. Bock, and M. Takach. 2016. Providing a safe space and medical monitoring to prevent overdose deaths. Health Affairs Blog, August 31. http://healthaffairs.org/blog/2016/08/31/providing-a-safe-space-and-medical-monitoring-to-prevent-overdose-deaths (accessed December 7, 2016).
Ge, P. S., and B. A. Runyon. 2016. Treatment of patients with cirrhosis. New England Journal of Medicine 375(8):767-777.
Gelberg, L., M. J. Robertson, L. Arangua, B. D. Leake, G. Sumner, A. Moe, R. M. Andersen, H. Morgenstern, and A. Nyamathi. 2012. Prevalence, distribution, and correlates of hepatitis C virus infection among homeless adults in Los Angeles. Public Health Reports 127(4):407-421.
Gish, R. G., J. A. Gutierrez, N. Navarro-Cazarez, K. Giang, D. Adler, B. Tran, S. Locarnini, R. Hammond, and S. Bowden. 2014. A simple and inexpensive point-of-care test for hepatitis B surface antigen detection: Serological and molecular evaluation. Journal of Viral Hepatitis 21(12):905-908.
Gondles, E. F., K. F. Maurer, and A. Bell. 2017. A major challenge for corrections: National survey findings identify challenges in recruiting and retaining correctional health care professionals. Corrections Today January/February:20-27. Article courtesy of the American Correctional Association (accessed January 4, 2016).
Grebely, J., M. Oser, L. E. Taylor, and G. J. Dore. 2013. Breaking down the barriers to hepatitis C virus (HCV) treatment among individuals with HCV/HIV coinfection: Action required at the system, provider, and patient levels. Journal of Infectious Diseases 207(Suppl 1):S19-S25.
Grebely, J., G. Robaeys, P. Bruggmann, A. Aghemo, M. Backmund, J. Bruneau, J. Byrne, O. Dalgard, J. J. Feld, M. Hellard, M. Hickman, A. Kautz, A. Litwin, A. R. Lloyd, S. Mauss, M. Prins, T. Swan, M. Schaefer, L. E. Taylor, and G. J. Dore. 2015. Recommendations for the management of hepatitis C virus infection among people who inject drugs. International Journal on Drug Policy 26(10):1028-1038.
Hacker, K., M. Anies, B. L. Folb, and L. Zallman. 2015. Barriers to health care for undocumented immigrants: A literature review. Risk Management and Health Care Policy 8:175-183.
Harris, K. A., Jr., J. H. Arnsten, and A. H. Litwin. 2010. Successful integration of hepatitis C evaluation and treatment services with methadone maintenance. Journal of Addiction Medicine 4(1):20-26.
Hepatitis C Online. 2016. Natural history of hepatitis C infection. http://www.hepatitisc.uw.edu/pdf/evaluation-staging-monitoring/natural-history/core-concept/all (accessed November 2, 2016).
Hern, T., A. Burke Valeras, J. Banker, and G. Riebe. 2013. Collaborative partnerships within integrated behavioral health and primary care. In Integrated behavioral health in primary care: Evaluating the evidence, identifying the essentials, edited by M. R. Talen and A. Burke Valeras. New York: Springer. Pp. 209-227.
HHS (Department of Health and Human Services). 2016. The community response to viral hepatitis: Contributions toward achieving the goals of the viral hepatitis action plan. HHS, Office of the Assistant Secretary for Health, Office of HIV/AIDS and Infectious Disease Policy.
HHS. 2017. National viral hepatitis action plan 2017-2020. HHS, Office of the Assistant Secretary for Health, Office of HIV/AIDS and Infectious Disease Policy. https://www.hhs.gov/sites/default/files/National%20Viral%20Hepatitis%20Action%20Plan%202017-2020.pdf (accessed February 24, 2017).
Hibbs, J., L. Benner, L. Klugman, R. Spencer, I. Macchia, and A. Mellinger. 1994. Mortality in a cohort of homeless adults in Philadelphia. New England Journal of Medicine 331(5):304-309.
Hill, W. D., G. Butt, M. Alvarez, and M. Krajden. 2008. Capacity enhancement of hepatitis C virus treatment through integrated, community-based care. Canadian Journal of Gastroenterology & Hepatology 22(1):27-32.
Hill, H. A., L. D. Elam-Evans, D. Yankey, J. A. Singleton, and M. Kolasa. 2015. National, state, and selected local area vaccination coverage among children aged 19–35 months—United States, 2014. Morbidity and Mortality Weekly Report 64(33):889-896.
Ho, S. B., N. Bräu, R. Cheung, L. Liu, C. Sanchez, M. Sklar, T. E. Phelps, S. G. Marcus, M. M. Wasil, A. Tisi, L. Huynh, S. K. Robinson, A. L. Gifford, S. M. Asch, and E. J. Groessl. 2015. Integrated care increases treatment and improves outcomes of patients with chronic hepatitis C virus infection and psychiatric illness or substance abuse. Clinical Gastroenterology and Hepatology 13(11):2005-2014.
Ho, S. B., A. Dollarhide, H. Thorisdottir, J. Michelsen, C. Perry, D. Kravetz, A. Herrin, L. Carlson, S. Hadley, D. Montoya, S. Robinson, C. Sanchez, E. Enrique, and E. Groessl. 2016. A primary care-based collaborative hepatitis C clinic: Clinical structure and virologic outcomes with direct acting antiviral therapy. Open Medicine Journal 3(Suppl 1: M4):70-78.
Honeck, J., and T. Dolansky. 2011. The Ryan White HIV drug assistance program: A vital part of Ohio’s public health infrastructure. State Budgeting Matters 7(8). http://www.communitysolutions.com/assets/docs/State_Budgeting_Matters/sbmv7n8ryanwhitereport100711.pdf (accessed October 25, 2016).
HRSA (Health Resources and Services Administration). 2015. Ryan White HIV/AIDS program: Part B manual. http://hab.hrsa.gov/sites/default/files/hab/Global/habpartbmanual2013.pdf (accessed December 8, 2016).
HRSA. 2016a. About the Ryan White HIV/AIDS program. https://hab.hrsa.gov/about-ryan-white-hivaids-program/about-ryan-white-hivaids-program (accessed December 8, 2016).
HRSA. 2016b. Federally qualified health centers (FQHC) and Federal Office of Rural Health Policy (FORHP) rural health areas. https://datawarehouse.hrsa.gov/ExportedMaps/ORHP/HGDWMapGallery_ORHP_FQHC.pdf (accessed December 9, 2016).
HRSA. 2016c. Part B: Grants to states & territories. https://hab.hrsa.gov/about-ryan-white-hivaids-program/part-b-grants-states-territories (accessed December 8, 2016).
HRSA. n.d.-a. Ryan White HIV/AIDS program services: Eligible individuals & allowable uses of funds. Policy clarification notice (PCN) #16-02. http://hab.hrsa.gov/sites/default/files/hab/Global/service_category_pcn_16-02_final.pdf (accessed October 25, 2016).
HRSA. n.d.-b. What are federally qualified health centers (FQHCs)? http://www.hrsa.gov/healthit/toolbox/RuralHealthITtoolbox/Introduction/qualified.html (accessed December 8, 2016).
Hu, D. J. 2008. Issues related to the prevention and control of hepatitis B virus (HBV) infection in the U.S. Paper presented at Institute of Medicine Roundtable on the Prevention and Control of Viral Hepatitis Infection, Washington, DC, December 4.
Hu, D. J., J. Xing, R. A. Tohme, Y. Liao, H. Pollack, J. W. Ward, and S. D. Holmberg. 2013. Hepatitis B testing and access to care among racial and ethnic minorities in selected communities across the United States, 2009-2010. Hepatology 58(3):856-862.
Hutton, D. W., D. Tan, S. K. So, and M. L. Brandeau. 2007. Cost-effectiveness of screening and vaccinating Asian and Pacific Islander adults for hepatitis B. Annals of Internal Medicine 147(7):460-469.
Hyun, C. S., W. R. Ventura, S. S. Kim, S. Yoon, and S. Lee. 2016. A community-based hepatitis B linkage-to-care program: A case study on Asian Americans chronically infected with hepatitis B virus. Hepatology, Medicine and Policy 1(1).
Jaffe, M. G., G. A. Lee, J. D. Young, S. Sidney, and A. S. Go. 2013. Improved blood pressure control associated with a large-scale hypertension program. JAMA 310(7):699-705.
Jenco, M. 2016. ACIP updates recommendations on HPV, HepB, MenB vaccines. American Academy of Pediatrics (AAP) News. http://www.aappublications.org/news/2016/10/20/ACIP102016 (accessed November 21, 2016).
Johnson, N. J., M. Ip, D. D. Munoz, and F. Laraque. 2016. Using qualitative evaluation to strengthen hepatitis B and C health promotion by patients navigators for hard-to-reach populations. https://nphic.confex.com/cdc/nphic16/webprogram/Paper36897.html (accessed November 14, 2016).
Juon, H. S., C. Strong, F. Kim, E. Park, and S. Lee. 2016. Lay health worker intervention improved compliance and hepatitis B vaccination in Asian Americans: Randomized controlled trial. PLoS One 11(9):e0162683.
Kansas Department of Health and Environment. n.d. The Kansas Ryan White Part B program. http://www.kdheks.gov/sti_hiv/ryan_white_care.htm#MHSA (accessed October 19, 2016).
Kanwal, F., J. R. Kramer, P. Buchanan, S. M. Asch, Y. Assioun, B. R. Bacon, J. Li, and H. B. El-Serag. 2012. The quality of care provided to patients with cirrhosis and ascites in the Department of Veterans Affairs. Gastroenterology 143(1):70-77.
Klink, K., M. Coffman, M. Moore, A. Jetty, S. Petterson, and A. Bazemore. 2015. Family physicians and telehealth: Findings from a national survey. Washington, DC: Robert Graham Center.
Kuncio, D. E., E. C. Newbern, M. H. Fernandez-Vina, B. Herdman, C. C. Johnson, and K. M. Viner. 2015. Comparison of risk-based hepatitis C screening and the true seroprevalence in an urban prison system. Journal of Urban Health 92(2):379-386.
Kvamme, O. J., F. Olesen, and M. Samuelson. 2001. Improving the interface between primary and secondary care: A statement from the European Working Party on Quality in Family Practice (EQuiP). Quality in Health Care 10(1):33-39.
Larney, S., M. K. Mahowald, N. Scharff, T. P. Flanigan, C. G. Beckwith, and N. D. Zaller. 2014. Epidemiology of hepatitis C virus in Pennsylvania state prisons, 2004–2012: Limitations of 1945–1965 birth cohort screening in correctional settings. American Journal of Public Health 104(6):e69-e74.
Lebrun-Harris, L. A., T. P. Baggett, D. M. Jenkins, A. Sripipatana, R. Sharma, A. S. Hayashi, C. A. Daly, and Q. Ngo-Metzger. 2013. Health status and health care experiences among homeless patients in federally supported health centers: Findings from the 2009 patient survey. Health Services Research 48(3):992-1017.
Lin, S. Y., E. T. Chang, and S. K. So. 2007. Why we should routinely screen Asian American adults for hepatitis B: A cross-sectional study of Asians in California. Hepatology 46(4):1034-1040.
Lin, Y. H., Y. Wang, A. Loua, G. J. Day, Y. Qiu, E. C. Nadala, Jr., J. P. Allain, and H. H. Lee. 2008. Evaluation of a new hepatitis B virus surface antigen rapid test with improved sensitivity. Journal of Clinical Microbiology 46(10):3319-3324.
Macalino, G. E., D. Vlahov, S. Sanford-Colby, S. Patel, K. Sabin, C. Salas, and J. D. Rich. 2004. Prevalence and incidence of HIV, hepatitis B virus, and hepatitis C virus infections among males in Rhode Island prisons. American Journal of Public Health 94(7):1218-1223.
Macalino, G. E., D. Dhawan, and J. D. Rich. 2005. A missed opportunity: Hepatitis C screening of prisoners. American Journal of Public Health 95(10):1739-1740.
Martyn, H., and K. Davis. 2014. Care coordination for people with complex care needs in the U.S.: A policy analysis. International Journal of Care Coordination 17(3-4):93-98.
Marwick, C. 1997. NCQA: Quality through evaluation. JAMA 278(19):1555-1556.
Mast, E. E., H. S. Margolis, A. E. Fiore, E. W. Brink, S. T. Goldstein, S. A. Wang, L. A. Moyer, B. P. Bell, and M. J. Alter. 2005. A comprehensive immunization strategy to eliminate transmission of hepatitis B virus infection in the United States. Recommendations of the Advisory Committee on Immunization Practices (ACIP). Part 1: Immunization of infants, children, and adolescents. Morbidity and Mortality Weekly Report 54(RR-16):1-31.
Maurer, K., and E. F. Gondles. 2015. Hepatitis C in correctional settings: Challenges and opportunities. Coalition of Correctional Health Authorities and American Correctional Association 2(1).
McCarthy, M. 2015. A quarter of U.S. adults with health insurance are underinsured, report finds. BMJ 350:h2786.
McGlynn, E. A. 1997. Six challenges in measuring the quality of health care. Health Affairs 16(3):7-21.
McInnes, D. K., B. A. Petrakis, A. L. Gifford, S. R. Rao, T. K. Houston, S. M. Asch, and T. P. O’Toole. 2014. Retaining homeless veterans in outpatient care: A pilot study of mobile phone text message appointment reminders. American Journal of Public Health 104(Suppl 4):S588-S594.
McMahon, B. J. 2009. The natural history of chronic hepatitis B virus infection. Hepatology 49(5 Suppl):S45-S55.
Mehta, S. H., J. Astemborski, G. D. Kirk, S. A. Strathdee, K. E. Nelson, D. Vlahov, and D. L. Thomas. 2011. Changes in blood-borne infection risk among injection drug users. Journal of Infectious Diseases 203(5):587-594.
Mellinger, J. L., and M. L. Volk. 2013. Multidisciplinary management of patients with cirrhosis: A need for care coordination. Clinical Gastroenterology and Hepatology 11(3):217-223.
Mitchell, T., G. L. Armstrong, D. J. Hu, A. Wasley, and J. A. Painter. 2011. The increasing burden of imported chronic hepatitis B—United States, 1974-2008. PLoS One 6(12):e27717.
Mitruka, K., K. Thornton, S. Cusick, C. Orme, A. Moore, R. A. Manch, T. Box, C. Carroll, D. Holtzman, and J. W. Ward. 2014. Expanding primary care capacity to treat hepatitis C virus infection through an evidence-based care model—Arizona and Utah, 2012-2014. Morbidity and Mortality Weekly Report 63(18):393-398.
NASEM (National Academies of Sciences, Engineering, and Medicine). 2016. Eliminating the public health problem of hepatitis B and C in the United States: Phase one report. Washington, DC: The National Academies Press.
NCCHC (National Commission on Correctional Health Care). 1998. National Commission on Correctional Health Care position statement. Use of telemedicine technology in correctional facilities. Journal of Correctional Health Care 5(1):103-111.
NCCHC. 2016. Telemedicine technology in correctional facilities. National Commission on Correctional Health Care. http://www.ncchc.org/telemedicine-technology-in-correctional-facilities (accessed September 23, 2016).
NCQA (National Committee for Quality Assurance). 2009. Childhood immunization status. http://www.ncqa.org/portals/0/Childhood%20Immunization%20Status.pdf (accessed November 3, 2016).
NCQA. 2012. The value of requiring health plans to report NCQA’s HEDIS quality measures. http://www.ncqa.org/portals/0/Public%20Policy/ValueinRequiringHEDIS_8.15.12.pdf (accessed November 3, 2016).
NCQA. 2016a. HEDIS & quality measurement. http://store.ncqa.org/index.php/performance-measurement.html (accessed November 3, 2016).
NCQA. 2016b. Summary table of measures, product lines and changes. http://www.ncqa.org/Portals/0/HEDISQM/HEDIS2017/HEDIS%202017%20Volume%202%20List%20of%20Measures.pdf?ver=2016-06-27-135433-350 (accessed November 7, 2016).
NCQA. n.d.-a. HEDIS®and Quality Compass®. http://www.ncqa.org/hedis-quality-measurement/what-is-hedis (accessed November 3, 2016).
NCQA. n.d.-b. HEDIS®measures. http://www.ncqa.org/hedis-quality-measurement/hedis-measures (accessed October 31, 2016).
Neate, R. 2016. Welcome to Jail Inc: How private companies make money off U.S. prisons. Guardian, June 16. https://www.theguardian.com/us-news/2016/jun/16/us-prisons-jail-private-healthcare-companies-profit (accessed December 28, 2016).
Nelson, P. K., B. M. Mathers, B. Cowie, H. Hagan, D. Des Jarlais, D. Horyniak, and L. Degenhardt. 2011. Global epidemiology of hepatitis B and hepatitis C in people who inject drugs: Results of systematic reviews. Lancet 378(9791):571-583.
NIDA (National Institute on Drug Abuse). 2010. Research report series. Comorbidity: Addiction and other mental illnesses. NIH publication number 10-5771. National Institutes of Health, NIDA.
Njai, H. F., Y. Shimakawa, B. Sanneh, L. Ferguson, G. Ndow, M. Mendy, A. Sow, G. Lo, C. Toure-Kane, J. Tanaka, M. Taal, U. D’Alessandro, R. Njie, M. Thursz, and M. Lemoine. 2015. Validation of rapid point-of-care (POC) tests for detection of hepatitis B surface antigen in field and laboratory settings in the Gambia, Western Africa. Journal of Clinical Microbiology 53(4):1156-1163.
Patel, K., M. Darling, K. Samuels, and McClellan. 2014. Transforming rural health care: High-quality, sustainable access to specialty care. Health Affairs Blog, December 5. http://healthaffairs.org/blog/2014/12/05/transforming-rural-health-care-high-quality-sustainable-access-to-specialty-care (accessed December 9, 2016).
Pfeiffer, S. 2016. Overwhelmed by overdoses, clinic offers a room for highs. Boston Globe, April 26. https://www.bostonglobe.com/business/2016/04/25/overwhelmed-overdose-epidemic-health-clinic-offers-room-for-supervised-highs/vQ61K3jao0vXNUPHK0iQhP/story.html (accessed December 7, 2016).
Post, S. E., N. K. Sodhi, C. H. Peng, K. Wan, and H. J. Pollack. 2011. A simulation shows that early treatment of chronic hepatitis B infection can cut deaths and be cost-effective. Health Affairs 30(2):340-348.
Project ECHO. n.d.-a. About ECHO. http://echo.unm.edu/about-echo/ (accessed November 2, 2016).
Project ECHO. n.d.-b. CDC and western states consortium. http://echo.unm.edu/initiatives/cdc-western-states-consortium (accessed October 31, 2016).
Project ECHO. n.d.-c. Funders. http://echo.unm.edu/about-echo/funders (accessed October 31, 2016).
Project ECHO. n.d.-d. Our story. http://echo.unm.edu/about-echo/our-story (accessed November 2, 2016).
Rabuy, B., and D. Kopf. 2015. Prisons of poverty: Uncovering the pre-incarceration incomes of the imprisoned. Prison Policy Initiative. http://www.prisonpolicy.org/reports/income.html (accessed September 23, 2016).
Reimer, J., and C. Haasen. 2009. Need-adapted HCV-treatment setting for injection drug users. Lancet 373(9681):2090-2091.
Rich, J. D., R. Chandler, B. A. Williams, D. Dumont, E. A. Wang, F. S. Taxman, S. A. Allen, J. G. Clarke, R. B. Greifinger, C. Wildeman, F. C. Osher, S. Rosenberg, C. Haney, M. Mauer, and B. Western. 2014. How health care reform can transform the health of criminal justice-involved individuals. Health Affairs 33(3):462-467.
Roberts, H., D. Kruszon-Moran, K. N. Ly, E. Hughes, K. Iqbal, R. B. Jiles, and S. D. Holmberg. 2016. Prevalence of chronic hepatitis B virus (HBV) infection in U.S. households: National Health and Nutrition Examination Survey (NHANES), 1988-2012. Hepatology 63(2):388-397.
Rubin, R. 2016. U.S. prisons missing opportunities to tackle HIV in inmates. Lancet 388(10049):1041.
Rural Health Information Hub. 2016. Nonmetropolitan federally qualified health centers. https://www.ruralhealthinfo.org/rural-maps/mapfiles/federally-qualified-health-centers.jpg (December 8, 2016).
SAMHSA (Substance Abuse and Mental Health Services Administration). 2015. Behavioral health trends in the United States: Results from the 2014 National Survey on Drug Use and Health. Rockville, MD: SAMHSA.
Sarfaty, M., and R. E. Myers. 2008. The effect of HEDIS measurement of colorectal cancer screening on insurance plans in Pennsylvania. American Journal of Managed Care 14(5):277-282.
Sarpel, D., E. Baichoo, and D. T. Dieterich. 2016. Chronic hepatitis B and C infection in the United States: A review of current guidelines, disease burden and cost effectiveness of screening. Expert Review of Anti-Infective Therapy 14(5):511-521.
Schaenman, P., E. Davies, R. Jordan, and R. Chakraborty. 2013. Opportunities for cost savings in corrections without sacrificing service quality: Inmate health care. Washington, DC: Urban Institute.
Schiff, M. 2014. Examining state prison health care spending: Cost drivers and policy approaches. Health Affairs Blog, November 4. http://healthaffairs.org/blog/2014/11/04/examining-state-prison-health-care-spending-cost-drivers-and-policy-approaches (accessed September 23, 2016).
Sequera, V. G., A. L. Garcia-Basteiro, and J. M. Bayas. 2013. The role of vaccination in prisoners’ health. Expert Review of Vaccines 12(5):469-471.
Shafer, M. A., K. P. Tebb, R. H. Pantell, C. J. Wibbelsman, J. M. Neuhaus, A. C. Tipton, S. B. Kunin, T. H. Ko, D. M. Schweppe, and D. A. Bergman. 2002. Effect of a clinical practice improvement intervention on chlamydial screening among adolescent girls. JAMA 288(22):2846-2852.
Shalev, N., M. A. Chiasson, J. F. Dobkin, and G. Lee. 2011. Characterizing medical providers for jail inmates in New York state. American Journal of Public Health 101(4):693-698.
Shivkumar, S., R. Peeling, Y. Jafari, L. Joseph, and N. P. Pai. 2012. Rapid point-of-care first-line screening tests for hepatitis B infection: A meta-analysis of diagnostic accuracy (1980-2010). American Journal of Gastroenterology 107(9):1306-1313.
Singh, H., A. Esquivel, D. F. Sittig, D. Murphy, H. Kadiyala, R. Schiesser, D. Espadas, and L. A. Petersen. 2011. Follow-up actions on electronic referral communication in a multispecialty outpatient setting. Journal of General Internal Medicine 26(1):64-69.
Smith, E. A., L. Jacques-Carroll, T. Y. Walker, B. Sirotkin, and T. V. Murphy. 2012. The national perinatal hepatitis B prevention program, 1994-2008. Pediatrics 129(4):609-616.
Solomon, L., B. T. Montague, C. G. Beckwith, J. Baillargeon, M. Costa, D. Dumont, I. Kuo, A. Kurth, and J. D. Rich. 2014. Survey finds that many prisons and jails have room to improve HIV testing and coordination of postrelease treatment. Health Affairs 33(3):434-442.
Sood, N., T. Juday, J. Vanderpuye-Orgle, L. Rosenblatt, J. A. Romley, D. Peneva, and D. P. Goldman. 2014. HIV care providers emphasize the importance of the Ryan White program for access to and quality of care. Health Affairs 33(3):394-400.
Spooner, L. M. 2011. The expanding role of the pharmacist in the management of hepatitis C infection. Journal of Managed Care Pharmacy 17(9):709-712.
St. Joseph’s Hospital and Medical Center. n.d. Liver/hepatology. http://www.dignityhealth.org/stjosephs/research-and-education/research/research-areas/liver-or-hepatology (accessed November 8, 2016).
Stein, J. A., R. M. Andersen, M. Robertson, and L. Gelberg. 2012. Impact of hepatitis B and C infection on health services utilization in homeless adults: A test of the Gelberg-Andersen behavioral model for vulnerable populations. Health Psychology 31(1):20-30.
Stockman, L. J., J. Greer, R. Holzmacher, B. Dittmann, S. A. Hoftiezer, L. E. Alsum, A. Prieve, R. P. Westergaard, S. M. Guilfoyle, and J. M. Vergeront. 2016. Performance of risk-based and birth-cohort strategies for identifying hepatitis C virus infection among people entering prison, Wisconsin, 2014. Public Health Reports 131(4):544-551.
Taylor, L. E. 2005. Delivering care to injection drug users coinfected with HIV and hepatitis C virus. Clinical Infectious Diseases 40(Suppl 5):S355-S361.
Taylor, V. M., T. G. Hislop, S. P. Tu, C. Teh, E. Acorda, M. P. Yip, E. Woodall, and Y. Yasui. 2009. Evaluation of a hepatitis B lay health worker intervention for Chinese Americans and Canadians. Journal of Community Health 34(3):165-172.
Taylor, V. M., N. J. Burke, C. Sos, H. H. Do, Q. Liu, and Y. Yasui. 2013. Community health worker hepatitis B education for Cambodian American men and women. Asian Pacific Journal of Cancer Prevention 14(8):4705-4709.
The Economist. 2016. How many homeless people are there in America? February 22. http://www.economist.com/blogs/democracyinamerica/2016/02/counting-street-sleepers (accessed October 27, 2016).
The National Alliance to End Homelessness. 2016. The state of homelessness in America. Washington, DC: Homelessness Research Institute.
The Physicians Foundation. 2016. 2016 survey of America’s physicians: Practice patterns & perspectives. The Physicians Foundation.
Thomas, D. L., D. Leoutsakas, T. Zabransky, and M. S. Kumar. 2011. Hepatitis C in HIV-infected individuals: Cure and control, right now. Journal of the International AIDS Society 14:22.
Thornton, K., P. Deming, B. Struminger, M. Sedillo, E. Castillo, M. Komaromy, S. Zalud-Cerrato, and S. Arora. 2016. Project ECHO: A revolutionary approach to expanding access to modern treatments for hepatitis C. Current Hepatology Reports 15(3):178-186.
Tompsett, C. J., S. E. Domoff, and P. A. Toro. 2013. Peer substance use and homelessness predicting substance abuse from adolescence through early adulthood. American Journal of Community Psychology 51(3-4):520-529.
Tracy, C. S., S. H. Bell, L. A. Nickell, J. Charles, and R. E. Upshur. 2013. The IMPACT clinic: Innovative model of interprofessional primary care for elderly patients with complex health care needs. Canadian Family Physician 59(3):e148-e155.
Tran, T. T. 2009. Understanding cultural barriers in hepatitis B virus infection. Cleveland Clinic Journal of Medicine 76(Suppl 3):S10-S13.
Treloar, C., J. Newland, J. Rance, and M. Hopwood. 2010. Uptake and delivery of hepatitis C treatment in opiate substitution treatment: Perceptions of clients and health professionals. Journal of Viral Hepatitis 17(12):839-844.
U.K. Department of Health. 2009. Guidance notes: Prison health performance and quality indicators. http://webarchive.nationalarchives.gov.uk/20140714084352/http://www.hpa.org.uk/webc/HPAwebFile/HPAweb_C/1232006593707 (accessed September 23, 2016).
U.K. Government. 2011. Prison health performance and quality indicators: Annual report 2011. https://www.gov.uk/government/uploads/system/uploads/attachment_data/file/215077/dh_133381.pdf (accessed September 23, 2016).
Unützer, J., Y. Choi, I. A. Cook, and S. Oishi. 2002a. A web-based data management system to improve care for depression in a multicenter clinical trial. Psychiatric Services 53(6):671-673, 678.
Unützer, J., W. Katon, C. M. Callahan, J. W. Williams, Jr., E. Hunkeler, L. Harpole, M. Hoffing, R. D. Della Penna, P. H. Noel, E. H. Lin, P. A. Arean, M. T. Hegel, L. Tang, T. R. Belin, S. Oishi, and C. Langston. 2002b. Collaborative care management of late-life depression in the primary care setting: A randomized controlled trial. JAMA 288(22):2836-2845.
USPSTF (U.S. Preventive Services Task Force). 2013. Final recommendation statement: Hepatitis C: Screening. https://www.uspreventiveservicestaskforce.org/Page/Document/RecommendationStatementFinal/hepatitis-c-screening (accessed December 8, 2016).
USPSTF. 2014. Final update summary: Hepatitis B virus infection: Screening, 2014. https://www.uspreventiveservicestaskforce.org/Page/Document/UpdateSummaryFinal/hepatitis-b-virus-infection-screening-2014 (accessed November 7, 2016).
USPSTF. 2015. Final update summary: Hepatitis B in pregnant women: Screening. https://www.uspreventiveservicestaskforce.org/Page/Document/UpdateSummaryFinal/hepatitis-b-in-pregnant-women-screening (accessed November 7, 2016).
VA (Department of Veterans Affairs). 2016. Hepatitis C medications: A review and update for patients. http://www.hepatitis.va.gov/products/patient/treatment-update.asp#S11X (accessed November 3, 2016).
Volk, M. L., R. S. Tocco, J. Bazick, M. O. Rakoski, and A. S. Lok. 2012. Hospital readmissions among patients with decompensated cirrhosis. American Journal of Gastroenterology 107(2):247-252.
Von Zielbauer, P. 2005. As health care in jails goes private, 10 days can be a death sentence. New York Times, February 27. http://www.nytimes.com/2005/02/27/nyregion/as-health-care-in-jails-goes-private-10-days-can-be-a-death.html?_r=0 (accessed December 27, 2016).
Walsh, N., A. Verster, M. Rodolph, and E. A. Akl. 2014. WHO guidance on the prevention of viral hepatitis B and C among people who inject drugs. International Journal on Drug Policy 25(3):363-371.
Weinbaum, C., R. Lyerla, and H. S. Margolis. 2003. Prevention and control of infections with hepatitis viruses in correctional settings. Morbidity and Mortality Weekly Report 52(RR-01):1-33.
Weinbaum, C. M., K. M. Sabin, and S. S. Santibanez. 2005. Hepatitis B, hepatitis C, and HIV in correctional populations: A review of epidemiology and prevention. AIDS 19(Suppl 3):S41-S46.
West Virginia Department of Health and Human Resources. n.d. Ryan White eligibility: WV Ryan White Part B state direct services. http://www.dhhr.wv.gov/oeps/std-hiv-hep/HIV_AIDS/caresupport/Pages/RyanWhiteEligibility.aspx (accessed October 25, 2016).
WHO (World Health Organization). 2001. Hepatitis B surface antigen assays: Operational characteristics (phase I). Report 1. Geneva, Switzerland: WHO.
WHO. 2016. Global health sector strategy on viral hepatitis, 2016-2021: Towards ending viral hepatitis. Geneva, Switzerland: WHO. http://apps.who.int/iris/bitstream/10665/246177/1/WHO-HIV-2016.06-eng.pdf (accessed July 19, 2016).
Wilkinson, M., V. Crawford, A. Tippet, F. Jolly, J. Turton, E. Sims, M. Hekker, J. Dalton, R. Marley, and G. R. Foster. 2009. Community-based treatment for chronic hepatitis C in drug users: High rates of compliance with therapy despite ongoing drug use. Alimentary Pharmacology & Therapeutics 29(1):29-37.
Woo, G. A., M. A. Hill, M. D. de Medina, and E. R. Schiff. 2013. Screening for hepatitis B virus and hepatitis C virus at a community fair: A single-center experience. Gastroenterology & Hepatology 9(5):293-299.
Yoo, G. J., T. Fang, J. Zola, and W. M. Dariotis. 2012. Destigmatizing hepatitis B in the Asian American community: Lessons learned from the San Francisco Hep B free campaign. Journal of Cancer Education 27(1):138-144.
Yusuf, H. R., D. Daniels, P. Smith, V. Coronado, and L. Rodewald. 2000. Association between administration of hepatitis B vaccine at birth and completion of the hepatitis B and 4:3:1:3 vaccine series. JAMA 284(8):978-983.
Zhao, Z., and T. V. Murphy. 2013. Which newborns missed the hepatitis B birth dose vaccination among U.S. children? Preventive Medicine 57(5):613-617.
Zivanovic, R., M. Milloy, K. Hayashi, H. Dong, C. Sutherland, T. Kerr, and E. Wood. 2015. Impact of unstable housing on all-cause mortality among persons who inject drugs. BMC Public Health 15:106.
Zlotnick, C., S. Zerger, and P. B. Wolfe. 2013. Health care for the homeless: What we have learned in the past 30 years and what’s next. American Journal of Public Health 103(Suppl 2):S199-S205.
This page intentionally left blank.


































