5
The Enabling Environment for Effective Partnerships in Global Health and the Sustainable Development Goals
Throughout the workshop series, participants acknowledge the importance of creating an enabling environment for multisectoral engagement in health and the Sustainable Development Goals
(SDGs). National regulatory authorities, and law and governance mechanisms protect individuals and communities by ensuring safety and efficacy of products and services, and incentivizing private-sector actions that align with the health needs of communities. Strong regulatory systems and laws help manage risks, promote stability and transparency, and provide guidance to private-sector companies. In addition to public-sector regulation, civil society has an important role in fostering an enabling environment for effective multisectoral engagement that improves that health and development status of communities.
This chapter summarizes presentations and discussions during the workshop series on regulation and accountability for creating an enabling environment for effective action on the SDGs, which includes strengthening national regulatory authorities, designing laws and regulation to improve the accountability of public–private partnerships (PPPs), and the increasing role of civil society.
REGULATORY SYSTEMS TO SUPPORT SUSTAINABLE DEVELOPMENT
Weak regulatory systems are an underlying threat to achieving many of the SDGs, as stated by Mary Lou Valdez from the U.S. Food and Drug Administration (FDA). In health, as well as other sectors, effective and efficient regulatory systems are often a necessary precursor for economic development and attracting private-sector investment. Regulatory authorities and systems can help to drive science-based approaches, data, and transparency for decision making and actions. Effective regulatory systems, suggested Valdez, are essential for the success and sustainability of global health investments from all stakeholders across civil society, industry, government, and international organizations. Although regulatory systems are not always seen as drivers for change, she emphasized that if a regulatory system is weak, many investments focused on the SDGs may not be able to achieve their intended effect. The cost of not strengthening these systems can be detrimental to any country’s overall viability and wellness, especially in the long term. Given the integral role of regulatory systems to undergird progress on the SDGs, Valdez led a multisectoral panel discussion to examine the roles the regulatory environment and PPPs play in strengthening regulatory systems in promoting public health, economic development, and sustainable investments to achieve the global goals.
Regulatory systems vary a great deal from country to country. In the United States, FDA includes 16,000 employees and covers a range of commodities—including pharmaceuticals, vaccines and biologics, veterinary products, food, animal feed, cosmetics, dietary supplements, medical
devices, tobacco products, and radiation-emitting equipment—that represent from $0.20 to $0.25 on the dollar of the U.S. gross domestic product. However, Valdez noted that there are national regulatory authorities that have a total of only 10 to 50 employees. Beyond personnel, Juergen Voegele from the World Bank noted the variability in the legal frameworks governing national regulatory authority, ranging from those with limited regulation or poorly designed laws to countries where there is overregulation but limited implementation.
Limited and weak regulatory systems can impede development. As Valdez suggested, effective and efficient regulatory systems are often a necessary precursor for economic development. For example, it is difficult for a country to attract investments in manufacturing if a regulatory authority is weak or lacking. Given the role of regulatory authorities in facilitating development, Valdez framed investments in regulatory systems in the context of global public goods, suggesting that there is a need for all stakeholders to participate in a systems approach to collectively addressing essential elements, such as regulatory systems, that underpin successful and sustainable investments. Against this backdrop, the panelists described the critical role that regulatory systems play in promoting development and increasing investments in support of the SDGs. The panelists acknowledged the complexity and challenges of regulatory systems strengthening and discussed possible solutions.
HOW REGULATORY SYSTEMS FOSTER DEVELOPMENT AND PROTECT CITIZENS
Juergen Voegele, World Bank
Building on the importance of regulatory systems as well as the high level of variability, Voegele suggested that achieving the SDGs across nations will not be possible without addressing the policy, legal, and regulatory environment in each country. While there are incentives for industry to invest, create jobs, and advance development in low- and middle-income countries (LMICs), there is the need to develop operating environments that are a conducive for private-sector engagement.
Voegele posited that the public sector could and should make investments in strengthening their operating environments to attract the private sector. However, rather than attempting to identify new public-sector financial resources for these investments, which will likely be difficult to find, Voegele suggested restructuring of existing subsidy programs as a possible opportunity to redirect current public funding toward strengthening the operating environment. From Voegele’s perspective, these subsidies should not be reduced but rather used differently to promote
investments in public goods aligned with the SDGs. These public goods outcomes would include strengthening regulatory systems and investments in research into long-term sustainable solutions.
Restructuring subsidies to promote public goods, he suggested, will require changing the narrative to reflect that subsidies can be powerful tools for positive change and creating win-win solutions for the public and the private sector. For example, globally there are significant subsidy programs in the energy and agriculture sectors. Voegele noted that currently a significant majority of the world subsidies in agriculture are directed to only a handful of crops, and this narrow focus on the types of crops is detrimental to nutritional outcomes. However, shifting to subsidizing diversified agriculture would be good for nutrition as well as reduce risks for farmers through diversification of their investments. Voegele emphasized that such a shift does not require changing longstanding policies regarding the allocation of subsidies to specific sectors but rather a strategic redirection that creates the win-win solution.
Shifting to the role of the private sector in investments in public goods such as regulatory systems strengthening, Voegele suggested that such investments are beneficial to companies, and platforms are needed to encourage their engagement. The World Bank is using its convening power to provide a platform for encouraging active collaboration among public and private stakeholders. The World Bank is bringing sectors together in the precompetitive space to develop systematic upstream strategies to address public goods of mutual benefit. As Valdez had acknowledged the need to do, this approach is moving to a systematic, structured, and multisectoral approach.
THE ROLE OF REGULATORY SYSTEMS IN ENABLING INDUSTRY INVESTMENTS AND MARKET GROWTH
Rajeev Venkayya, Takeda Pharmaceutical Company Limited
Given the success of public–private collaborations to address market failures that has occurred in several areas of global health, Rajeev Venkayya from Takeda Pharmaceutical Company Limited suggested the global health community should look to these successful examples to extract lessons about the interactions between the private and public sectors and regulations. These lessons might apply to gaps where this collaboration is not happening or has not been as successful. Three examples he noted as areas of success are biodefense, addressing some health needs in low-income countries, and responding to health emergencies such as pandemic outbreaks. In the case of biodefense, where incidents are low-probability but high consequence, Venkayya pointed to the creation of the
U.S. Biomedical Advanced Research and Development Authority, which serves as a central funding and coordination agency for research and development to address biodefense threats.
To address the historic market failures in LMICs, where there is a high need but market opportunities historically have been limited, there has been a proliferation of product development partnerships that are charged with the responsibility of developing one or more products, drugs, diagnostics, or vaccines against neglected diseases or diseases of developing countries. In terms of pandemic preparedness, where, as with biodefense, the probability in aggregate may be low but the consequences could be quite high, there is a renewed and increasing sense of ownership of governments to tackle this challenge in a more definitive way. The development of the Coalition for Epidemic Preparedness Innovations is a result of this renewed commitment. Within each of these three areas, Venkayya emphasized the critical need to engage the private sector to establish a sustainable framework for innovation. Such a framework can lead to commercialization of a product and its subsequent widespread availability.
When it comes to regulatory issues, Venkayya suggested that accountability for safety and efficacy of products is shared among four stakeholder groups: practitioners who administer the products, companies that produce them, the authorities responsible for evaluating safety and efficacy, and additional bodies or technical advisory groups that evaluate benefit or risk in specific populations. Speaking from a company perspective, Venkayya shared four elements industry looks for within a regulatory system to determine if it can be an effective partner in ensuring safety and efficacy:
- predictability in the process to secure regulatory approval in a given market
- capacity within the regulatory authority to swiftly evaluate safety and efficacy and issue a decision on whether the product will be approved
- a level of convergence of regulatory expectations across authorities
- in the event of emergencies, capacity to expedite processes as quickly as possible without compromising quality or safety
Venkayya touched on a few examples where there has been success in tackling challenges related to these four elements. On the second element, Article 58 in Europe is a mechanism that allows regulators in Europe to offer their expertise to inform reviews of products that address diseases outside of Europe. On the third element, he noted regional harmonization efforts, which Dan Hartman from the Bill & Melinda Gates Foundation
describes in more detail later in the chapter. Regarding the fourth element, he cited FDA’s response during the Ebola crisis as a positive example of the level of collaboration among industry parties, regulators, and other stakeholders to respond quickly in the event of an emergence of a pathogen with epidemic potential.
HOW REGULATORY SYSTEMS PROTECT AND SUSTAIN CONTRIBUTIONS
Dan Hartman, Bill & Melinda Gates Foundation
Recognizing the importance of strong and predictable regulatory systems for attracting private-sector investments in global health, multisectoral partnerships have been developed that target optimizing the regulatory systems through which products must go to be developed, registered, procured, and distributed in low-income countries. Dan Hartman from the Gates Foundation described these initiatives, stating that a key consideration in the design of these initiatives has been identifying sustainable solutions that will have the greatest impact in the shortest time with the least amount of human and financial resources.
As Valdez acknowledged earlier, regulatory systems vary greatly from country to country. Hartman described the fundamental medical product environmental challenges that underlie these country regulatory system variances. In most high-income countries, the regulatory process moves products from the point of development to patient access in a closed, highly regulated, proscribed system that assures quality and safety (see Figure 5-1). Conversely, in many low-income countries (LICs), current national medical product access processes, of which regulatory is a part, are complex and limited in the ability to assure the quality and safety of the medicines and products that patients can access. These medi-
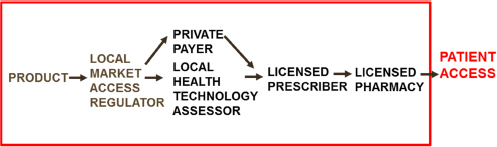
SOURCE: As presented by Dan Hartman on June 24, 2016.
cal product access systems are unregulated to a great degree with many different ways that products move from a manufacturer to a patient (see Figure 5-2).
In high-income countries (HICs), quality-assured medical product access generally requires only the marketing authorization of the medicines regulator in the HIC and, in some HICs, a payment decision by the responsible authority. In contrast, in most LICs, quality-assured medical product access (for products for HIV [human immunodeficiency virus], TB [tuberculosis], malaria, human reproductive products, etc.) generally requires three steps prior to patient access through national programs. First there is product registration (either for local use and/or export) in the country of manufacture or by a World Health Organization (WHO) recognized “stringent regulatory authority.” Large multination procurers and United Nations (UN) procurement agencies then require WHO prequalification to know which manufacturers are producing quality-assured versions of the product. If the product has been authorized by a stringent regulatory authority, WHO uses an abbreviated assessment process to assure that the product labeling, storage, etc., meets the conditions of use in the LIC (conditions that the stringent authorities would not consider during their assessment for their populations). Finally, the product needs to be registered by the local national regulatory authority (NRA) in order to be used legally in that jurisdiction. When examining this process, Hartman pointed out that Gates Foundation research revealed areas of redundancy, non-value-added activities, and long “down time” between these three steps often adding up to four to seven years from application to first regulator to authorization in the focus LIC. Hartman emphasized that creating impact within the 15 year timeframe of the 2030 Agenda is not feasible in such a system, especially when it comes to patient access for new products being developed now to address medical issues specific to LICs.
Within the context of this existing regulatory environment, the Gates Foundation sought to develop with its partners an approach to creating an optimized system through which a quality dossier could get to local registration (first registration to registration in LIC) in at least 50 percent less time than the baseline of the Gates Foundation research in 2012−2013. To achieve this goal, the Gates Foundation is supporting work with regulatory partners in three main areas: value-added activities (increasing the use of reliance on the work products of trusted agencies to inform decisions), manufacturer inputs (to help assure a quality dossier submitted initially and not something one builds over time during the regulatory assessment process), and decreasing complexity (emphasizing regional approaches to product regulation through harmonized standards and joint assessment procedures within a regional network of NRAs) (see Figure 5-3).
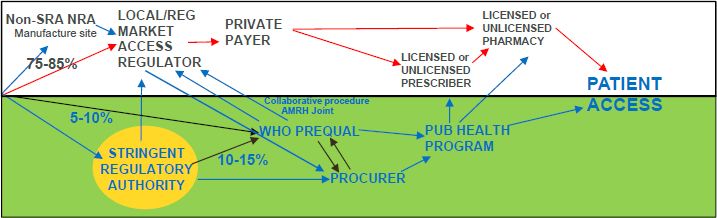
NOTES: AMRH = African Medicines Regulatory Harmonization; NRA = national regulatory authority; SRA = stringent regulatory authority; WHO = World Health Organization. The upper box represents ways patients may access products with minimal product quality assurance. The lower box represents ways patients pay access products with assured product quality assurance. Percentages represent percent of medicines that generally follow that route to get to patients in low-income countries.
SOURCE: As presented by Dan Hartman on June 24, 2016.
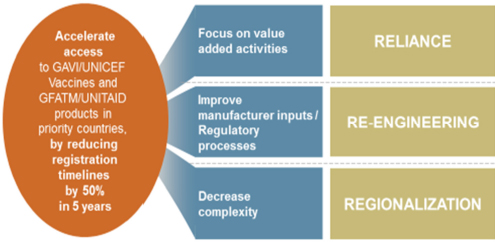
NOTE: GAVI = Gavi, the Vaccine Alliance; GFATM = Global Fund to Fight AIDS, Tuberculosis and Malaria; UNICEF = United Nations Children’s Fund; WHO = World Health Organization.
SOURCE: As presented by Dan Hartman on June 24, 2016.
Hartman shared how the Gates Foundation is tackling the third area, decreasing complexity, by supporting regional harmonization of regulatory processes. The objective of the harmonization initiatives is to pool resources together and to support sustainable capacity building that focuses on the skills and processes needed to regulate within a network of NRAs. Using the example of the African Medicines Regulatory Harmonisation (AMRH), Hartman noted that the goal is to improve the fragmented regulatory system for product registration in Africa by changing from a country-focused approach to a regional approach, using established regional economic communities that are optimized and collaborative. The AMRH partnership was launched in the East African Community (EAC) countries in 2012 and includes the NRAs in those jurisdictions, the regional bloc Secretariat, WHO (providing technical support), New Partnerships for Africa’s Development (providing overall project management and advocacy support), and funding support from the Gates Foundation, the United Kingdom’s Department for International Development, The U.S. President’s Emergency Plan for AIDS Relief, the Swiss Agency for Development and Cooperation through a World Bank trust fund. The partnership focuses on harmonizing and streamlining technical requirements and processes for product registration, which should lead to increased and timely product access. From a regional product registration perspective in East Africa, companies are now able to
submit a single dossier and have it reviewed simultaneously by six EAC regulatory authorities.
Hartman noted that there are initiatives under way to develop similar regional regulatory processes in other regions (including other regional economic communities in western and southern Africa, the Caribbean, and southeastern Asia). Based on the experience from the harmonization process in East Africa, Hartman shared several elements that he feels were necessary for the success of the AMRH partnership that can likely be applied in forging successful partnerships for regional regulatory approaches in these other regions as well as global health partnerships more broadly. The elements are building regional ownership, trust, aligning with government and other in-country stakeholders, transparency, accountability, clear and measurable goals, and approaching with a spirit of genuine collaboration to help get needed quality-assured products to patients as quickly as possible.
Shifting Regulatory Philosophy
The panelists and other workshop participants discussed the evolving philosophy around the role of regulation and how it has led to such initiatives described by Hartman, Venkayya, and Voegele. Derek Yach from The Vitality Group commented that regulatory philosophy is changing toward a focus on regulation to solve social problems in a way that also mobilizes market forces to be a part of the solution to solving these problems. There is a shift from punitive regulation to regulation that incentivizes better policies. Voegele agreed with this shifting regulatory philosophy and provided an example from Mexico to demonstrate it. He suggested the changes to the Mexican regulatory system were initiated by the appointment of an economist to lead the regulatory authority. That appointee shifted the focus toward recognition of the importance of the regulatory authority for economic growth, and within 5 years Voegele noted that there was a dramatic shift to mobilizing markets, increasing competition, and focusing on high quality (Valenzuela, 2016). By adding these things together, both the manufacturing and quality of pharmaceuticals in Mexico is increasing, the price of products is decreasing, and, he suggested, the health of the population will increase.
OPPORTUNITIES FOR LAW, GOVERNANCE, AND REGULATORY DESIGN IN IMPROVING ACCOUNTABILITY OF PUBLIC−PRIVATE PARTNERSHIPS
Roger S. Magnusson, University of Sydney Law School
Roger Magnusson from the University of Sydney Law School opened his presentation by asking how the siloed interests of government, industry, and civil society can be transformed into a productive collaboration with strong accountability for advancing the health-focused SDGs. Bridging interests to create effective PPPs, Magnusson suggested, depends on robust law, governance, and regulatory design, which create opportunities and strengthen accountability for PPPs as they accelerate national sustainable development initiatives. Focusing on the role of law and regulatory design in managing PPPs, Magnusson proposed three scenarios for PPPs in the space of advancing the SDGs: (1) win-win in which value is created both for health and for shareholders; (2) inadequate alignment between business incentives and the incentives needed to achieve health goals; and (3) businesses with net impact that is harmful to health. Magnusson explained these three scenarios and the role of law and regulatory design within each of them.
The first of the three scenarios is the win-win situation, wherein the public and private sectors partner in such a way that they create value for both society and shareholders. Drawing from Yach’s presentation from the Forum on Public−Private Partnerships for Global Health and Safety’s December 2015 workshop, “Exploring Shared Value in Global Health and Safety,” Magnusson noted three areas in which businesses can generate shared value: workplace wellness programs, meeting market demand for healthier products, and addressing health risks in the company’s consumer base (NASEM, 2016).
To achieve the win-win scenario, transparent and independently adjudicated laws must govern businesses so they can, as Magnusson elaborated, operate with stability and predictability, which in turn motivates businesses to invest in the lives and communities of employees and customers. Magnusson argued that respect for the rule of law and for human rights creates an enabling environment for effective partnerships. Secondly, to foster the win-win scenario, Magnusson raised the concept of “framework legislation” for the health-focused SDG targets as a strategy for supporting development of effective PPPs. According to Magnusson, such legislation could create a mandate for national surveillance of risk factors and a process for setting national targets and indicators that are ambitious but realistic and context appropriate. Other elements of the framework, Magnusson elaborated, could include highly visible moni-
toring and reporting processes; a national cross-ministerial governance mechanism to encourage an all-of-government response to the health SDGs, and a formal mechanism for encouraging engagement between civil society, the private sector, and government that effectively manages any conflicts of interest.
Illustrating the formation of national cross-ministerial governance mechanisms, Magnusson provided the example of national AIDS commissions formed outside of a ministry of health that coordinate action between a wide range of ministries. Magnusson described how in some countries, UN theme groups, sometimes including members from government representatives and leading civil society organizations, facilitated collaborations between countries themselves and their international partners. These theme groups were convened by the UN resident representative in each country. Moreover, Magnusson drew from previous work of the Ad Hoc Working Group on Implementation Monitoring and Accountability for WHO’s Commission on Ending Childhood Obesity to recommend appointing national or regional rapporteurs for the health-focused targets of the SDGs (WHO, 2016). Rapporteurs could facilitate the development of win-win partnerships through acting as liaisons between civil society, government, and business, being a champion for partnering with business in accelerating the SDGs, and providing a channel for shadow reporting, thereby increasing accountability of PPPs.
Accountability is vital if PPPs are to be effective mechanisms to advance the health-focused targets of the SDGs; therefore, Magnusson urged that accountability should permeate every stage in the development and implementation of a partnership. To illustrate the ubiquity of accountability, he presented the action cycle (see Figure 5-4), developed to address the gap between stated intentions and results, and divided it into three steps: planning, doing, and assurance of action. Accountability is integrated throughout all components of the cycle. Mutual accountability is key; entities within and outside of the government hold the government accountable, and similarly, entities within and outside of businesses monitor and evaluate the company’s performance. Civil society, Magnusson emphasized, plays a key role through the free media in order to hold partnerships, both on the public and private sides, accountable. Added to this, he noted, is the notion that an empowered civil society is essential for effective partnerships.
PPPs, Magnusson argued, often reflect volunteerism rather than regulation in that businesses engaging in partnerships negotiate based on their interests and priorities, whereas under regulation, they submit to controls placed upon them. To illustrate the significance of this point, Magnusson moved to the scenario for PPPs that is the opposite of the win-win scenario: that is, where the business of the private-sector partner is harmful
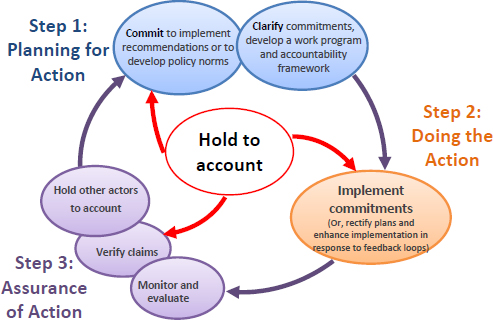
SOURCES: As presented by Roger Magnusson on October 27, 2016. Diagram was developed by the Working Group on Implementation, Monitoring and Accountability (WGIMA) in support of the work of the WHO Commission on Ending Childhood Obesity, with input from Roger Magnusson. Information on WGIMA can be found at http://www.who.int/end-childhood-obesity/commissioners/en (accessed May 5, 2017).
to health. Businesses harmful to health may include those that market harmful products, damage the environment, ignore the health and welfare of their suppliers and employees, or fail to prevent workplace accidents. He emphasized that confronting harmful products and practices using law and regulation are vital and may be far more beneficial to public health than soft diplomacy, whereas businesses may be attracted to partnerships for reputational improvement, legitimation, and potential access to policy makers, which thereby presents them with an opportunity to shift policy to the business’s advantage.
Magnusson offered examples that illustrate how partnering with businesses rather than imposing controls on them could have opportunity costs to health advancement. For example, if a country sought to increase affordable access to an essential drug, it might either engage in negotiations with the manufacturer for a voluntary license, or it might issue a compulsory license and seek to make greater use of flexibilities contained within the
Trade-Related Aspects of Intellectual Property Rights agreement. Depending on circumstances, the price or access achieved through negotiations with the pharmaceutical company might be lower than the result that could have been achieved through regulation, as company interests are able to, perhaps heavily, influence the result. Magnusson concluded that in developing partnerships, the public sector should be acutely aware of the company’s interests and its capacity to extract concessions (explicit or implicit) from governments, as PPPs with companies whose market incentives are misaligned with the public’s health can have significant opportunity costs.
Magnusson then described the scenario for PPPs wherein the partnered business is not necessarily harmful to health but lacks adequate alignment between the incentives that create economic value and the incentives that are needed to achieve the health goals, which results in underperformance and poses the risk of investments in health being lightweight or simply public relations exercises. Ultimately, Magnusson suggested that if partnered businesses consistently fail to achieve their stated goals, in ways that jeopardize the achievement of national health goals, governments may need to enhance the regulatory environment around PPPs and be ready to introduce more muscular controls. As a solution for addressing stagnation in partnerships, Magnusson introduced the concept of “regulatory scaffolds,” or the incremental strengthening of different components of a partnership. As defined in Table 5-1, the components of regulation fall under three main headings: the content of regulation, regulatory processes, and enforcement, and they include matters such as the goals and terms of the partnership or initiative, administration, monitoring and review, and incentives for compliance.
Even when health goals are pursued through voluntary partnerships, there may still be room for governments to intervene in underperforming partnerships in order to strengthen performance, such as through setting objectively verifiable targets, ensuring the partnership faces systematic and independent monitoring, and providing economic incentives for businesses to improve performance. Magnusson noted that improving private-sector performance also depends heavily on independent assessments and reviews of partnerships regarding their progress toward stated goals, as well as on continuous public reporting of results made accessible to civil society.
In closing, Magnusson reemphasized the significant opportunities that PPPs pose for creating social and business value and achieving the health focused SDGs, and he reiterated how partnerships pose risks if formed between businesses whose goals are not properly aligned with health goals. For his final point, Magnusson emphasized the importance of public-sector intervention throughout the components of the regulatory process in order to strengthen underperforming partnerships.
TABLE 5-1 Components of Regulatory Design and Accompanying Forms of Government Intervention
| Component of Regulation | Form of Government Intervention |
|---|---|
| Regulatory or policy framework | Determine an overarching policy framework and objectives |
| Content of Regulation | |
| Goals of voluntary initiatives | Clearly identify the goals of self-regulation; set objectively verifiable targets or performance indicators to be achieved within a defined timeframe |
| Terms of voluntary initiatives | Define key terms and definitions underpinning voluntary schemes |
| Regulatory Processes | |
| Administration | Provide for administration of the scheme by an independent body representing a wide range of interests |
| Monitoring | Ensure that the scheme includes systematic and independent monitoring |
| Review | Ensure that there is regular, independent, and structured review of the scheme’s overall operation |
| Enforcement | |
| Incentives for compliance | Provide incentives that give participants an economic incentive to comply |
| Deterring noncompliance | Take steps to deter noncompliance at both company and industry levels |
SOURCES: As presented by Roger Magnusson on October 27, 2016. Adapted from Magnusson and Reeve, 2015.
In response to Magnusson’s presentation, Jo Ivey Boufford of The New York Academy of Medicine acknowledged a dichotomy that had been present throughout the workshop series discussions: regulations and legal frameworks are often seen as obstacles to forming effective partnerships, but at the same time, as Magnusson presented, concerns exist over the lack of legal frameworks and the rule of law governing PPPs. She requested examples of either the strengthening of or relaxing of regulations to form productive partnerships. Magnusson responded by introducing the theory of responsive regulation, which links regulation to performance. Magnusson underlined how achieving the SDGs relies on concrete, measurable goals to govern partnerships and on the government’s readiness to take action against underperformance in partnerships. The government, Boufford noted, also has the responsibility to establish role clarity and set clear expectations from the onset of a partnership.
CIVIL SOCIETY ENGAGEMENT: PERSPECTIVE FROM SAVE THE CHILDREN
Beck Smith, Save the Children
Addressing the role of civil society in enhancing actions to advance the SDGs, Beck Smith focused her presentation on her experience at Save the Children UK. She noted Save the Children’s optimism surrounding the achievement of SDGs, but she acknowledged that process can impede progress. A focus for Save the Children has been ensuring that accountability mechanisms are fit for purpose from the start. Smith honed in on the 2030 Agenda pledge to leave no one behind, the wording and inclusive focus for which Save the Children had lobbied. She underlined the implicit understanding that the SDGs cannot be achieved unless the global community reaches the furthest behind first and mentioned Save the Children’s related global campaign, Every Last Child, which focuses on the most excluded children, defined as those facing both poverty and discrimination.
Introducing four levels with which to conceptualize accountability, Smith outlined the individual, regional, national, and international levels of accountability for progress on the SDGs. At the individual level, Smith asserted, public information campaigns raising awareness of the development agenda are necessary in order to familiarize the public with the advocacy levers they possess and how they can hold their decision makers and governments accountable. The national level of accountability, Smith continued, entails the full and meaningful implementation of the goals by the nation with the development and implementation of a people-centered, transparent accountability framework. This framework would allow civil society to monitor, evaluate, and hold governments accountable for the progress on the development agenda. Smith explained that accountability at the regional level is necessary in order to monitor best practices and share them between countries. Accountability at the international level, she continued, should be embedded in the development agenda from the start, and Smith hoped for a high level of ambition for accountability at this level.
To illustrate how civil society can facilitate progress on the goals and develop advocacy agendas, Smith turned to Save the Children’s work on the global indicator framework developed by the Inter-Agency and Expert Group on SDG Indicators (IAEG-SDGs) at the 47th session of the UN Statistical Commission held in March 2016. She discussed indicators under Goal 3, “Ensure healthy lives and promote well-being for all at all ages,” that measure universal health coverage, and focused on indicator 3.8.2, which had been revised to measure “The number of people covered
by health insurance or a public health system per 1,000 population.” In contrast the original indicator, Smith explained, had meaningfully measured financial protection under universal health coverage,1 but the indicator measured health insurance coverage without regard for financial security related to health care expenses. Save the Children and other civil society organizations, along with support from WHO and the World Bank, together advocated for financial security to be reintegrated into the indicator. Smith explained that civil society organizations were in agreement on the indicator, and this enabled effective coordination for advocacy mechanisms, including starting social media campaigns and writing letters to the IAEG-SDGs to demonstrate concern. Smith explained that by working together toward a common goal, sharing intelligence, and collaborating with WHO and the World Bank, civil society organizations were able to push their agenda forward, and indicator 3.8.2 was put up for consultation, with support from 22 member states, at the last meeting of the IAEG-SDGs in Geneva in late 2016.
A second accountability mechanism Smith provided as an example of Save the Children’s work in this area was its participation in the High-Level Political Forum (HLPF), the annual July meeting at the UN in New York where countries volunteer to be reviewed on their progress on the SDGs. Smith spoke of Save the Children’s involvement in the HLPF in 2016. Save the Children organized several side events and convened a platform with the Rockefeller Center, the UN Foundation, and academics from the London School of Hygiene & Tropical Medicine; they then asked the question of how the private sector could support universal health coverage to help deliver on the SDGs’ promise to leave no one behind. Convening a platform with civil society organizations and the private sector was an effective mechanism for addressing the challenge of universal health coverage, as the private sector is increasingly involved in development initiatives and is a principle funder of these projects.
Smith shared a project on which Save the Children collaborated with Bond, an umbrella organization for nongovernmental organizations (NGOs) in the United Kingdom, and the World Wildlife Fund that aims to ensure that accountability, the HLPF, and country reporting on the SDGs are processes for which the standard is raised each year to provide for increasingly effective monitoring. To assess how effectively countries are reporting, the secretary-general’s voluntary reporting guidelines, a set of guidelines put forth by the secretary-general of the UN for how countries should structure their progress reports, use a “traffic light” system to indicate whether countries had filled the criteria in their
___________________
1 Indicator 3.8.2 previously read, “Fraction of the population protected against catastrophic or impoverishing out-of-pocket health expenditure.”
reports. Smith summarized conclusions from the assessment, including, for example, how China opted out of most guidelines whereas Georgia used the guidelines as a backbone for its report. Moreover, page lengths of the reports differ; for example, Uganda released a 100-page report, while Switzerland published a 30-page summary. Differences in report lengths render comparison difficult and demonstrate a large disparity in the amount of information being made public between countries on their progress. Smith concluded that the assessment underlined the importance of having consistency in country reporting so country progress may be fairly compared and contrasted, and best practices identified.
In concluding, Smith provided examples of recently emerged civil society coalitions. Smith noted that civil society must form strong, cohesive coalitions in order to provide weight and draw attention to the issues at hand, and she drew attention to Together 2030, a coalition between World Vision and Site Savers, Centro de Pensamiento Estratégico Internacional (Colombia), the Justice, Development and Peace Commission (Nigeria), the Social Enterprise Network (Philippines), and Save Matabeleland (Zimbabwe), as a good example of an organized coalition that aims to build the capacity of civil society engagement in the sustainable development agenda. Together 2030 coordinates indicator consultation responses and produces webinars that educate individuals about the SDGs and raise awareness about how they might engage with the 2030 Agenda. Smith highlighted other strong coalitions using such strategies for engaging civil society as concentrating efforts around a specific goal, reaching and engaging smaller NGOs and giving them a platform to have their voices heard, and raising the prominence of the agenda.
REFERENCES
Magnusson, R., and B. Reeve. 2015. Food Reformulation, Responsive Regulation, and “Regulatory Scaffolding”: Strengthening Performance of Salt Reduction Programs in Australia and the United Kingdom. Nutrients 7:5281-5308.
NASEM (National Academies of Sciences, Engineering, and Medicine). 2016. Exploring Shared Value in Global Health and Safety: Workshop Summary. Washington, DC: The National Academies Press.
Valenzuela, P. 2016. Mexico’s Reforms and the Prospects for Growth. Mexico Institute. https://www.wilsoncenter.org/sites/default/files/mexicos_reforms_and_the_prospects_for_growth_final.pdf (accessed May 23, 2017).
WHO (World Health Organization). 2016. Report of the Commission on Ending Childhood Obesity. http://apps.who.int/iris/bitstream/10665/204176/1/9789241510066_eng.pdf?ua=1 (accessed April 12, 2017).


















