4
Trends in Opioid Use, Harms, and Treatment
Not since the HIV/AIDS epidemic has the United States faced as devastating and lethal a health problem as the current crisis of opioid misuse and overdose and opioid use disorder (OUD). Current national trends indicate that each year more people die of overdoses—the majority of which involve opioid drugs—than died in the entirety of the Vietnam War, the Korean War, or any armed conflict since the end of World War II. Each day 90 Americans die prematurely from an overdose that involves an opioid (Rudd et al., 2016b), leaving families and friends bereft. The opioid epidemic’s toll is felt across the life span and in every sociodemographic group, but more heavily burdens vulnerable populations, such as those in economically depressed areas of the country. This chapter updates key statistics regarding use and misuse of prescription opioids, identifies risk factors for opioid-related harms, describes the recent increase in use of heroin and illicitly manufactured synthetic opioids and its relation to the prescription opioid epidemic, describes the impact of prescription opioids on illicit markets, reviews the current state of surveillance systems, and summarizes recent trends in treatment of OUD and use of naloxone to prevent overdose deaths. The committee selected these topics to discuss in particular for their relevance to the U.S. Food and Drug Administration’s (FDA’s) exercise of its authority to regulate pharmaceutical opioid products (analgesics, agonists, and antagonists). Each aspect of this chapter identifies considerations that should be taken into account when weighing the societal perspective and public health impact relevant to these products when they are being considered for new drug approval or during post-market surveillance.
TRENDS IN PRESCRIPTION OPIOID USE AND MISUSE
Medical prescriptions for opioids started to increase sharply in the mid-to late 1990s (NIDA, 2014). Shortly thereafter, nonmedical opioid use also started to increase markedly, reaching a peak of 2.7 million new users in 2002 (Kolodny et al., 2015). The annual number of new nonmedical users slowly declined to about 1.8 million in 2012 (SAMHSA, 2013b), but the overall pool of people continuing to use nonmedically is very large. From 1999 to 2011, hydrocodone use increased more than two-fold, oxycodone use more than five-fold (Jones, 2013b), and the mortality rate of opioid-related overdose almost four-fold (Chen et al., 2014). Overdose mortality is the most dramatic consequence of increased opioid use, but it is not the only one; rates of emergency room visits for nonmedical opioid use (SAMHSA, 2013a), neonatal abstinence syndrome (NAS) (Patrick et al., 2012), and OUD treatment admissions all have soared since 2002 (SAMHSA, 2010).
While death rates associated with opioid overdose have increased for virtually every population group, the rates are highest among males under age 50 (CDC, 2015a). In Massachusetts during the period 2013–2014, 76 percent of opioid overdose deaths occurred among people under the age of 50, and men aged 18 to 34 had opioid-related death rates nearly three times higher than those of women of the same age (Massachusetts Department of Public Health, 2016). Opioid-related death rates also were higher among those who had recently been released from prison, those who obtained opioid prescriptions from multiple pharmacies, and those who obtained prescription opioids in combination with other scheduled medications.
The age group with the greatest past-year nonmedical use of opioids is young adults aged 18 to 25, yet the greatest use (i.e., exposure) of prescription opioids is among adults aged 26 and older. Substance Abuse and Mental Health Services Administration (SAMHSA) data indicate that most people who report prescription opioid misuse in current cohorts initiated use in their early to late 20s, which may explain why prescription opioid mortality disproportionately affects adults aged 25 to 54 (CDC, 2016c). More recent data show an overlap in these age-related demographics with respect to current use of heroin and, more disturbingly, the coincident increase in overdose deaths caused by heroin and synthetic opioids other than methadone among people aged 15 and older (Rudd et al., 2016). It is important to acknowledge that data on overdose deaths may be subject to misclassification with respect to intent (i.e., whether the overdose was intentional or unintentional), especially for older, medically ill patients prescribed medications, whose deaths may not be followed up with toxicology testing and may not be referred to a medical examiner as a drug-involved or suspicious death. Misuse and aberrant opioid use behaviors also may manifest differently in older adults (Beaudoin et al., 2016; Henderson et
al., 2015), and given the aging U.S. population, the role of suicidal intent in prescription opioid poisoning in older adults is an area of active inquiry (Rocket et al., 2010; West et al., 2015).
The full extent of the public health consequences of prescription opioids is further complicated by the increased availability of heroin, which is less expensive than prescription opioids in the black market (DEA, 2013), and by the fact that so many who develop OUD from prescription opioids switch to heroin. In one study, about 80 percent of current heroin users reported that they began with prescription opioids (Muhuri et al., 2013). Therefore, the public health effects of prescription opioids and heroin are intertwined (Kolodny et al., 2015). Between 2001 and 2011, the rate of admission to treatment for OUD involving heroin doubled among non-Hispanic whites aged 20 to 34 (it stayed relatively constant for all other age groups among whites and for all age groups among non-Hispanic blacks), and the rate of heroin overdose deaths increased more than 2.5-fold among whites aged 18 to 44 (CDC, 2014; SAMHSA, 2013a). The cumulative effect is a 200 percent increase in opioid-involved overdoses from 2000 to 2014 (Rudd et al., 2016) concordant with increases in nonmedical prescription opioid use (Calcaterra et al., 2013; Cerdá et al., 2013; Kenan et al., 2013). In more recent years, national initiatives to reduce opioid prescribing have modestly decreased the number of prescription opioids dispensed (Dart et al., 2015). However, many people who otherwise would have been using prescription opioids have transitioned to heroin use, with a resulting three-fold increase in heroin-involved overdose deaths from 2010 to 2014 (Compton et al., 2016). Indeed, the overall frequency of heroin deaths has been accelerating since 2010 (see Figure 1-2 in Chapter 1).
Risk Factors for Prescription Opioid Misuse and Overdose
Despite the unsettling trends described above, a more nuanced examination indicates that not all prescription opioid medications confer similarly heightened risk. The causal pathways from the onset of pain to opioid exposure and to potential negative consequences such as misuse, drug seeking related to undertreatment of pain (Green and Chambers, 2015; Vadivelu et al., 2017), OUD, and overdose are difficult to disentangle, and represent an area of active research and investigation (Stumbo et al., 2017). Multiple post-marketing studies currently under way for extended-release (ER)/long-acting (LA) opioids (see Annex Table 6-1 in Chapter 6) may shed light on the timing and sequence of and precursors to the development of problem use and OUD and the incidence of nonfatal and fatal overdose among patients prescribed opioids for the treatment of chronic noncancer pain.
Characteristics of opioid medication and how they are prescribed can affect the risk of nonmedical use and other harms. Three key characteristics
of opioid medications that have been found to influence the risk of harms include the chemical compound, the formulation, and the intended route of administration. Also salient are the number of pills prescribed and dosage, as well as other prescribing patterns.
Chemical Compound
Neuropsychological experiments demonstrate that “likability,” and therefore “abuse liability,” is greater for some compounds than others. In seminal work by Comer and colleagues (2008) among a sample of patients dependent on heroin, laboratory experiments compared the likability of oxycodone, fentanyl, buprenorphine, and morphine with that of heroin. Findings indicated that across several validated subjective scales, oxycodone scored most favorably among participants, while buprenorphine scored lowest. Translating data from laboratory-based, controlled abuse liability studies to the community and clinic to examine possible increased risk is more challenging. However, several studies provide insight into “real-world” abuse liability and risk variation by compound. One means by which demand for a compound can be deduced is through street price. Taking availability into account, one recent study found that the street price of buprenorphine/naloxone was lower than that of buprenorphine single-entity and of methadone (Larance et al., 2015). Interestingly, these findings are congruent with those of the laboratory-based abuse liability studies noted earlier.
Another indicator of a compound’s risk is seen in mortality data. Unless the chemical entity is a novel one, it is difficult to differentiate branded from generic products as causal in an unintentional opioid poisoning death. Nevertheless, overdose death data show key compound-level trends, taking methadone as an example. Ray (2015) reports high overdose risk associated with use of methadone medications (for pain), and a 2017 analysis of methadone deaths and prescribing from 2007 to 2014 conducted by the U.S. Centers for Disease Control and Prevention (CDC) found that although methadone accounted for about 1 percent of all opioid prescriptions, overall methadone-related deaths accounted for 22.9 percent of all opioid-related mortality in 2014 (Faul et al., 2017). These findings have been replicated in other studies, suggesting that certain compounds are more likely to be misused and potentially lead to greater health consequences in the absence of preventive measures. Novel compounds, such as tapentadol (Nucynta), designed specifically to avoid tampering and reduce risk while achieving pain control, exhibit promising post-marketing epidemiologic data across a number of misuse and risk indicators (Butler et al., 2015; Dart et al., 2016; McNaughton et al., 2015), findings that warrant further examination in longitudinal studies.
Formulation
Another characteristic of a medication that may influence the risk of harm is its formulation, specifically whether it is an ER/LA or immediate-release (IR) formulation. The FDA’s Risk Evaluation and Mitigation Strategy (REMS) for ER/LA opioids anticipated that greater risks would be associated with opioids that increased the possible time of exposure through longer-time-release formulations. In fact, while further research is needed, available data show that ER/LA and IR formulations are associated with different types of elevated risk. ER/LA formulations are associated with increased risks of diagnosis of substance use disorder (SUD) and nonfatal and fatal opioid overdose (Braden et al., 2010; Miller et al., 2015; Zedler et al., 2014). However, limited data suggest that IR, short-acting opioid medications also may be associated with various morbidities and nonmedical use. Relative to ER/LA formulations, for example, these medications have been found to be indicated more often in poison center data as medications of misuse, and are associated with higher rates of nonfatal injury, including motor vehicle and pedestrian crashes and falls (Iwanicki et al., 2016). Moreover, an IR medication may be the first opioid of exposure over the course of one’s lifetime (SAMHSA, 2016a), given the routine use of these drugs following dental and surgical procedures, as discussed in Chapter 2. These data suggest that both ER/LA and IR opioids warrant measures to reduce risks that can arise with their use. Indeed, the FDA plans to expand its REMS program for opioids to include IR formulations (FDA, 2017b).
Combination opioid products, especially those coformulated with naloxone (e.g., Targaniq [oxycodone/naloxone] and Suboxone [buprenorphine/naloxone]) may be associated with lower rates of misuse and nonmedical use by other than intended routes of administration (i.e., by injection or insufflation) compared with their single-entity counterparts (Davis et al., 2013; Larance et al., 2015; Walsh et al., 2016). Although coformulations may help prevent misuse and OUD (Raffa et al., 2014), epidemiologic studies to explore these differences further are needed, and some such studies are under way (Degenhardt et al., 2015).
Route of Administration
A final characteristic that may elevate the risk of an opioid medication is its intended route of administration. Many preparations are used in ways other than prescribed and may be manipulated to extract the active pharmaceutical ingredient. For instance, pills may be crushed in the mouth, insufflated, smoked, or injected with few physical barriers to use, and a transdermal patch’s active pharmaceutical ingredients may be chewed, sucked, or extracted and prepared for injection. It is well substantiated that
drugs used by insufflation and injection, in particular, enter the bloodstream and hasten the opioid’s crossing of the blood–brain barrier, generating a faster onset of action, which in turn is associated with a greater risk of overdose and of developing OUD (EMCDDA, 2016).
Some prescription opioid preparations approved in recent years make crushing the pill more difficult or may be formulated to deter tampering. These abuse-deterrent formulations (ADFs) are reviewed more extensively in Chapter 5, but it is worth noting here that the level of tampering and prevalence of use by unintended routes associated with an opioid will influence its public health consequences. For example, a new and comprehensive analysis by Alpert and colleagues (2017) shows that the reformulation of OxyContin from a non-ADF to an ADF prescription opioid was linked to higher-than-expected rates of subsequent heroin use, especially in places with persistently high rates of opioid misuse. The authors estimate that up to 80 percent of the increase in heroin use could be attributed to the formulation change. Likewise, the ADF Opana ER (oxymorphone ER) has been associated with several injection-related harms, linked to the same ADF preparation applied to OxyContin. Because of these injection-related harms, in June 2017 the FDA requested that Opana ER be removed from the market by its manufacturer (FDA, 2017a).
In a retrospective 24-month cohort study based on National Poison Data System data, Copelan and colleagues (2017) found intentional misuse and suspected suicidal intent to be significantly lower among patients using a 7-day buprenorphine transdermal system/patch than among those taking other ER/LA opioid analgesics examined. On the other hand, data from a recent Australian study showed that, 2 years after the introduction of a buprenophine-naloxone film, levels of injection and diversion were comparable between the film and methadone and buprenorphine-naloxone tablets among out-of-treatment people who inject drugs (PWID), but levels of injection and diversion were lower for mono-buprenorphine than for the film, after adjusting for availability (Larance et al., 2015). The ADF film was found to be easier to administer, which impacted clinician time and workflow. These data suggest a need for caution in reliance on ADF products as a regulatory strategy for improving opioid safety and the importance of weighing the public health impacts of all decisions. Tracking the prevalence of the intended and unintended routes of administration of a drug can provide signals of compromised safety and harmful consequences at the individual and societal levels.
Number of Pills Prescribed and Dosage
Emerging literature since the Institute of Medicine (IOM) report Relieving Pain in America was issued (IOM, 2011) also suggests that potentially
modifiable features of the prescription itself are associated with harm. The greater the number of days for which a prescription is written and the higher the dosage, the greater is the risk exposure. Unfortunately, the literature lacks clear consensus on the number of days after which risk increases (i.e., the threshold). The CDC’s Guideline for Prescribing Opioids for Chronic Pain, released in 2016 (Dowell et al., 2016), urges prescribers to provide the lowest effective dosage and prescribe “no greater quantity than needed for the expected duration of pain severe enough to require opioids” (stating that “three days or less will often be sufficient”). Some states (e.g., Maine and Massachusetts) have recently legislated a supply limit for opioids prescribed for the treatment of noncancer pain, with far-reaching applications. (In Maine, the law limits the number of pills that can be prescribed to a 7-day supply within a 7-day period for acute pain and a 30-day supply within a 30-day period for chronic pain [Smith, 2016; Traynor, 2016], while Massachusetts imposes a 7-day supply limit for first-time prescriptions for adults and a 7-day limit at any time for minors.1) More research in this area could better inform policy makers, patients, and providers.
A concept related to that of number of days’ supply is daily morphine milligram equivalent (MME) dosing. Unlike the days’ supply literature, the literature on this topic presents a clear and consistent finding that risk of overdose increases as dose increases (i.e., a dose-response relationship) (Baumblatt et al., 2014; Bohnert et al., 2011, 2016; Dunn et al., 2010; Gomes et al., 2011; Liang and Turner, 2015; Paulozzi, 2012; Zedler et al., 2014). Based on several early findings, some authors concluded—erroneously—that a specific threshold or MME cutpoint value (e.g., >100, >50, or >20 MME) could signify the point of elevated risk, below which opioids are safe but above which risk rises. Based on the existing literature and analysis of large clinical datasets, however, the risk of overdose and OUD increases as a function of dose (i.e., dose-response relationship) at any given level of exposure greater than none.
The FDA’s required “abuse liability” studies attempt to anticipate and measure many of these drug-specific characteristics before a drug is approved. However, these studies are not designed to predict a fuller range of potentially harmful effects that one may want to consider in deciding whether to approve an opioid or other drug, such as unforeseen allergies, unanticipated side effects, co-use with other licit and illicit drugs, and ease of manipulation to prepare the product for misuse. For these effects, the current approach is to rely on post-marketing surveillance to capture, in a proactive, preventive way, the cumulative effects of drug-specific characteristics as the drugs are actually used or misused in the population. Given
___________________
1 See https://malegislature.gov/Bills/189/House/H4056 (accessed May 15, 2017).
heightened concerns about opioid misuse, OUD, overdose, and diversion, involving people who use drugs (or their representative organizations) in the review and discussion of post-marketing data may be informative.
Other Prescribing Patterns
Other patterns of prescribing and dispensing suggest additional risks for OUD and overdose. The timing of risk exposure, for instance, may contribute to iatrogenic overdose. Similar to the patterns of elevated risk of overdose mortality during the first 2 weeks after release from incarceration, circumstances defined by loss of tolerance (such as during hospitalization [Bird et al., 2016] or following detoxification [Strang et al., 2003]) or the establishment of tolerance, such as at the onset of treatment with opioid analgesics (Miller et al., 2015), all suggest that the timing of opioid exposure can affect patient safety and overdose risk. In addition to timing, obtaining opioids from multiple prescribers or multiple pharmacies and overlapping prescriptions have been associated with greater risk of overdose (Baumblatt et al., 2014; Hall et al., 2008; Yang et al., 2015). These patterns may ultimately reflect poor coordination of care for people with pain and OUD in the community rather than causal drivers of the epidemiology of nonmedical use of prescription opioids. In addition, a large body of health services literature indicates that a number of opioid analgesic prescribing behaviors contribute greatly to patient risk and prolonged opioid exposure. These include errors in MME calculations (e.g., during opioid rotation or conversion) (Paulozzi et al., 2009; Rich and Webster, 2011), underutilization of prescription drug monitoring programs (Starrels et al., 2011), and inconsistencies in monitoring of opioid use (Becker et al., 2011; Khalid et al., 2015), among others.
While the FDA-approved indications for use and labeling of opioids specify for whom and under what conditions the medications are intended to be used, prescribing and patient use patterns may differ from those envisioned at the time of approval. For instance, many opioid medications, such as IR products, are intended to be used to treat acute pain, such as postsurgical pain, over a short duration. However, a large proportion of patients continue to be treated with IR opioids far beyond the expected duration of healing (Bartels et al., 2016; Clarke et al., 2014), a phenomenon that could indicate failure to heal from an injury or surgery, progression or persistence of pain to a chronic state, opioid dependence, onset of OUD, poor product labeling, or something else entirely. Still other patients may be prescribed an ER/LA opioid to treat an acute pain condition, a practice that runs counter to recommendations of the CDC guideline and from professional organizations.
With respect to chronic pain, ER/LA opioids are approved for use
in the treatment of moderate to severe pain as may be needed to treat instances of failure to heal from injury or surgery or progression of acute to chronic pain, or in instances of treatment of other chronic conditions when moderate to severe pain occurs. As discussed in Chapter 2, however, there is a lack of long-term evidence (>1 year) from rigorous studies that opioid therapy is effective for improving pain and function for people with chronic noncancer pain (Dowell et al., 2016), while there is evidence that opioid therapy for chronic pain is associated with increased risk of OUD, overdose, and other adverse outcomes (Baldini et al., 2012; Chou et al., 2015; Dowell et al., 2016). For example, rates of iatrogenic OUD in studies in which OUD has been carefully diagnosed have averaged about 8 percent, while rates of iatrogenic misuse, OUD, and aberrant behaviors thought to be indicative of OUD have ranged from 15 to 26 percent (Volkow and McClellan, 2016). While the FDA does not regulate the practice of medicine, the committee recognizes the importance of prescribing practices in helping to curb opioid-related harms, and in Chapter 5 describes several interventions designed to promote more judicious prescribing.
One key aspect of opioid prescribing safety overseen by the FDA is drug–drug interactions, whereby concurrent use of certain medications may alter a patient’s risk. Certain medications are coprescribed more frequently based on the co-occurrence of pain with other conditions, and it is also widely observed that patients may co-use other drugs with opioids to achieve heightened or prolonged analgesic or euphoric effects.
The co-use of opioid medications with one class of drugs, benzodiazapines, has been well established in preclinical, clinical, and epidemiologic studies, and contributes to up to one-third of fatal opioid overdoses in the United States (Jones and McAnich, 2015). Biological data indicate that these two drug classes have synergistic effects in producing sedation and respiratory depression, increasing the risk of overdose and death. Studies of opioid and benzodiazepine co-use in humans have demonstrated an elevated risk of overdose, especially in the context of misuse (Park et al., 2015; Sun et al., 2017). A large case-cohort study of U.S. veterans treated for chronic pain with long-term opioid analgesics, for example, showed that the risk of death from drug overdose increased in a synergistic, dose-response fashion as daily benzodiazepine dose increased, with risk being independent of dosing schedule (Park et al., 2015; see Figure 4-1). The safety concerns related to co-use of opioids and benzodiazepines led the FDA to require boxed warnings and patient-focused medication guides providing information about the risks associated with the concurrent use of these medications for more than 400 opioid and benzodiazepine products (FDA, 2016). These concerns also led to a recommendation in the CDC guideline urging caution in co-use or mitigation of the risk of respiratory
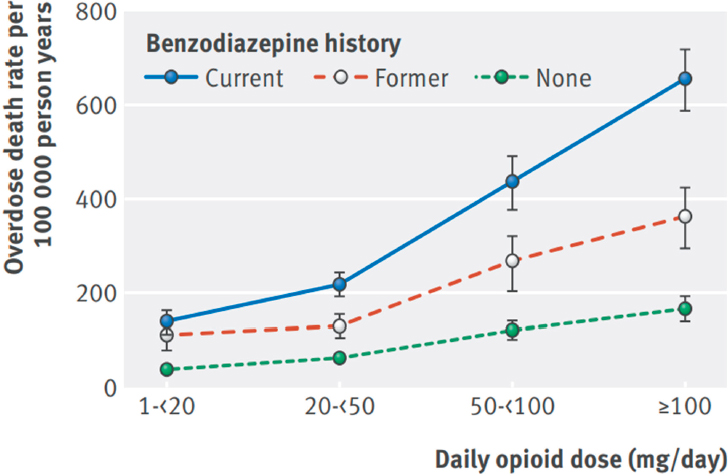
SOURCE: Park et al., 2015.
depression with naloxone for patients coprescribed benzodiazepines and opioids (Dowell et al., 2016).
Summary
The level and type of risk to a patient from a given opioid are influenced by specific features of the medication iself, including the compound, formulation (whether the medication is an ER/LA, IR, and/or combination product), and route of administration. How opioids are prescribed (e.g., with other medications, days for which prescribed) also may influence the risk of overdose. Available studies consistently demonstrate that the risk of overdose increases in a dose-response fashion with increasing MME. While the FDA abuse liability studies capture several features of drugs that influence the risk of harm, including mechanisms of misuse and diversion, post-marketing studies and surveillance data could help to identify a comprehensive range of potentially harmful effects.
Vulnerable Populations
This section reviews recent trends in OUD among three especially vulnerable populations—pregnant women and neonates, persons involved with the criminal justice system, and injection drug users.
Pregnant Women and Neonates
According to a study by Patrick and colleagues (2015), the proportion of babies born with NAS in the United States increased five-fold from 2000 to 2012, concurrently with a significant increase in opioid use and misuse among pregnant women. Subsequent studies have found that the incidence of NAS varies significantly among states, that the geographic variations in NAS are consistent with the variations in opioid pain prescriptions, and that the incidence of NAS and maternal opioid use increased disproportionately in rural relative to urban counties (Ko et al., 2016; Villapiano et al., 2017). Recent years have seen an unprecedented focus on NAS in the media; among policy makers; and among medical specialists in neonatology, pediatrics, and obstetrics. Strong disagreement among these interested groups is not uncommon as a result of poor understanding of and differences in opinion about the contexts and factors that affect NAS (Kaltenbach and Jones, 2016).
Recently the FDA has used the term “neonatal opioid withdrawal syndrome” on warning labels when referring to maternal use of opioids during pregnancy. It is understandable why this term is used on an FDA label pertaining to an opioid; however, the committee believes it is inappropriate for use in a clinical setting. When NAS occurs as a result of prenatal exposure to an opioid, it does so in various different contexts, and the presentation and severity are related to a number of factors in addition to maternal use of opioids. Accordingly, the discussion here uses the customary NAS terminology.
Although NAS was initially reported in 1865 as congenital morphism, with the first case of treatment reported in 1903, the focus of treatment and assessment over the past 50 years is based on work in the 1970s that established the definition of NAS and developed an instrument for measuring neonatal withdrawal. This work took place in response to the heroin epidemic and the resultant implementation of methadone pharmacotherapy for OUD (Jones and Fielder, 2015).
NAS generally is described as the occurrence of opioid withdrawal at birth after the discontinuation of prenatal opioid exposure. It is characterized by signs and symptoms of central nervous system irritability, including excessive crying, increased muscle tone, tremors, and sleep disturbances; gastrointestinal dysfunction, including poor feeding, vomiting,
and diarrhea; respiratory distress; and autonomic symptoms, including sweating, sneezing, and mottling (McQueen and Murphy-Oikonen, 2016). It is a temporary phenomenon that may or may not require treatment. In general, available data do not suggest an association between NAS in particular and long-term adverse developmental outcomes, regardless of whether the NAS was severe enough to require treatment.2 There is also no conclusive evidence that maternal dose is related to the severity of NAS (Cleary et al., 2010; Kaltenbach and Finnegan, 1986). In addition to factors discussed below, the presentation and severity of NAS are related to genetics (Wachman et al., 2013, 2014, 2015), maternal physiology (Jansson et al., 2007), and gestational age (Dysart et al., 2007; Gibson et al., 2017; Ruwanphthirana et al., 2015).
The current public focus on NAS does not take into account the context in which it occurs. The context encompasses whether the opioid is a medication taken under the care of a health care provider (e.g., a woman receiving medication under the care of a physician for pain management, or a woman being treated by a physician for OUD with methadone or buprenorphine), or whether the woman is misusing pain medications with or without a prescription and/or using illicit opioids such as heroin. Even though the risk of NAS is comparable across contexts, the overall risk to the fetus and neonate differ between women taking medications under the care of a qualified health care provider and those misusing medications and/or using illicit drugs. In particular, in contrast with diverted medications and illicit drugs of unknown purity, source, and quantity, the treatment of pain or OUD with opioid medications occurs within the safety of known doses of FDA-approved medications that have been rigorously tested for safety and efficacy and obtained legally from a qualified pharmacy or dispensary. In the case of misuse and OUD involving black market prescription or illicit opioids such as heroin, in addition to the uncontrolled dose, quantity, and purity of the drugs, the pregnancy may be affected by stress, violence, and trauma surrounding illegal activity. Indeed, research shows that prenatal stress, depression, and trauma can influence birth outcomes and later development (Fatima et al., 2017; Su et al., 2015). Thus, although not altering the probability of NAS occurrence, shifting the opioid-exposed pregnancy from one that is untreated to one that is treated may improve overall health outcomes for both mother and baby.
The national and state data that have been used to report significant increases in NAS are based on hospital codes that do not differentiate between NAS occurring as a result of maternal opioid misuse and that due
___________________
2 Although some babies with NAS may have other risks, such as low birth weight and/or parents with suboptimal caregiving capacity due to SUD, which are known to be associated with increased risk for adverse developmental outcomes.
to the appropriate use of an opioid prescription. Additionally, the codes do not indicate whether an infant required treatment for NAS.
Complicating the understanding of NAS is that there are other medications that produce withdrawal symptoms similar to those associated with opioids and, when taken in conjunction with opioids, exacerbate NAS. When pregnant women receiving methadone or buprenorphine take selective serotonin reuptake inhibitors (SSRIs, i.e., antidepressants), for example, the SSRIs have been found to be related to both the presentation and treatment of NAS, with higher peak scores of NAS and higher doses of medication required for treatment (Jansson et al., 2010; Kaltenbach et al., 2012). A number of studies also have found that when pregnant women receiving methadone or buprenorphine take benzodiazepines, such concomitant use is related to prolonged length of treatment for NAS (Pritham et al., 2012; Seligman et al., 2008; Wachman et al., 2011). In addition, as noted earlier, this co-use of opioids and benzodiazepine increases the risk of overdose. Cigarette smoking also has been found to adversely affect NAS, including the total amount of medication required to treat it and the length of treatment (Jones et al., 2013).
With the exception of methadone and buprenorphine, no attention has been given to whether the incidence of signs and symptoms of NAS may differ by opioid. One study comparing the NAS profile before treatment or in the absence of treatment in infants exposed prenatally to methadone or buprenorphine found that the incidence of nasal stuffiness, sneezing, and loose stools was greater in the buprenorphine-exposed infants, whereas the methadone-exposed infants were found to have higher mean scores for hyperactive Moro reflex, disturbed and undisturbed tremors, failure to thrive, and excessive irritability (Gaalema et al., 2012). Such findings may explain reported differences in NAS incidence, severity, and treatment duration between methadone and buprenorphine. No information is available for other opioid pain medications regarding signs and symptoms of NAS, its incidence and severity, and the length of treatment. Importantly, little to no information is available regarding exposure to illicitly manufactured fentanyl or fentanyl analogs in pregnant women and its effect on the risk of fatal overdose; responsiveness to OUD treatment; the maternal medication-assisted treatment (MAT) dose; or NAS incidence, severity, or treatment duration.
The issue of assessment, which determines the diagnosis and severity of NAS and thus directs the course of treatment, is another area of misunderstanding. No objective, biological index or marker exists for the determination of NAS. Neonatal metabolic alterations such as hypocalcemia, hypoglycemia, hypomagnesemia, and hypothermia can mimic NAS and need to be ruled out before treatment for NAS is initiated. The most widely used assessment tool consists of 21 items with 31 possible scores (e.g.,
“mild tremors when disturbed” and “marked tremors when disturbed,” “loose stools” and “watery stools,” “hyperactive Moro reflex and markedly hyperactive Moro reflex”) (Finnegan and Kaltenbach, 1992). Making such distinctions requires extensive reliability training, and even with such training, it can be difficult to score some items with a high degree of accuracy. Additionally, neither the incremental validity of the differential weighting of the tool nor its sensitivity and specificity have been examined. Such limitations have led to calls to reexamine the assessment of NAS and the need for an objective measure derived from a rigorous psychometric approach (Jones and Fielder, 2015).
Although a standard of care for NAS has been developed over the past 50 years, aggregate data across several hospital/fellowship program surveys suggest significant variability in both diagnosis and treatment protocols (Jones and Fielder, 2015). Effectiveness evidence for medications used to treat NAS is limited. Currently, oral morphine solution and methadone are recommended by the American Academy of Pediatrics for the treatment of NAS (Hudak and Tan, 2012). Morphine has been found to have shortcomings under some dosing and weaning regimens, and no data from randomized controlled trials comparing methadone with morphine are currently available. Although not yet used in clinical settings, randomized controlled trial data comparing buprenorphine and morphine show buprenorphine to be more effective than morphine, requiring less medication and shorter length of treatment (Kraft et al., 2011). In a recent randomized trial involving 63 infants with NAS, those treated with buprenorphine had significantly shorter treatment duration compared with those treated with morphine. The median between-group difference in treatment duration was 13 days (Kraft et al., 2017).
Medication dose regimens for NAS are traditionally determined by the infant’s weight, but some institutions and research protocols use a symptom-based approach in which the dose is based on the severity of the infant’s symptoms. To date, no systematic studies have evaluated these differing regimens.
The lack of protocols has recently been identified as impacting the duration of NAS treatment, the length of inpatient stay, and the rate of adjunctive therapy. Other recent changes in hospital practices, such as supporting breastfeeding and integrating mothers as partners in care, have been found to decrease the need to treat NAS and reduce the length of hospital stay (Holmes et al., 2016).
It should be reemphasized that these data are specific to women maintained on methadone or buprenorphine for OUD. To the committee’s knowledge, no data specific to other opioid pain medications are available. Infants undergoing NAS would be assessed and treated the same, but mothers receiving opioids for chronic pain who wished to breastfeed would
require a safety evaluation, including type of medication, length of time on medication, and rapid increases in dose (Sachs, 2013).
The incidence of NAS in relation to the opioid epidemic has been identified as a major concern. Regrettably, strategies to address NAS are often punitive and excessive and applied disproportionately to vulnerable populations. The identification of NAS as fetal harm calls into question the ability to adequately parent their children for both women who use opioid medications as prescribed by their health care providers and those who misuse opioid medications or use illicit opioids (Terplan et al., 2015). Some state legislatures have required surveillance of NAS prevalence for both prescribed and illicit drugs. Judges and prosecutors have implemented punitive approaches with women who use both prescribed and nonprescribed opioids during pregnancy, including arrest, civil commitment, detention, prosecution, and loss of custody. The Child Abuse Prevention and Treatment Act of 20103 requires states to have policies and procedures in place for notifying child protective services about children affected by withdrawal symptoms from exposure to prenatal drugs, and the Comprehensive Addiction and Recovery Act of 2016 requires that a plan of safe care be implemented. Neither law differentiates among the highly varied contexts in which NAS occurs. While there may be situations that call for action to prevent child abuse and neglect, caution is warranted in designating NAS as a proxy for risk of abuse and neglect.
In summary, only by disentangling NAS due to the use of an opioid medication as prescribed by a health care provider from that due to misuse of these medications and/or the use of illicit opioids can prevention and treatment approaches for NAS be better refined. A more comprehensive response to NAS and treatment of OUD in pregnant women would be enabled by better understanding of the signs and symptoms of NAS for specific opioid medications and illicitly manufactured fentanyl and its analogs, including the development of an objective diagnostic tool, better understanding of the effectiveness of various medications and protocols for treatment of NAS, and the development of treatment protocols specifically for pregnant women using fentanyl.
Persons Involved with the Criminal Justice System
Another population heavily affected by the opioid epidemic and with unique risks consists of people within the criminal justice system. Drug-related crimes and seizures of illicit drugs point to a sharp rise in the opioid crisis. As the opioid epidemic shifts rapidly from prescription opioids to heroin, illicitly manufactured fentanyl, and other illicit drugs, more indi-
___________________
3 Public Law 93-247.
viduals, many of whom live with OUD, are coming into contact with the criminal justice system. Authors of a 2006 study analyzing data on arrests, incarcerations, and heroin use estimate that 24 to 36 percent of all people with OUD involving heroin pass through U.S. prisons and jails each year (Boutwell et al., 2006), although this figure may be different today owing to changes in the heroin-using population. People recently released from incarceration experience the highest risk of fatal opioid overdose of any subpopulation (Binswanger et al., 2007, 2011, 2013; Farrell and Marsden, 2008; Merrall et al., 2010) because of their loss of tolerance, social isolation, and extraordinarily high relapse rates. Examining data from the Arrestee Drug Abuse Monitoring II Program, Hunt and colleagues (2015) found that those with a history of heroin use had higher drug use and severity and higher rates of treatment utilization relative to those reporting use of other drugs. Only one-third (34 percent) of arrestees with drug use histories had received SUD treatment during their lifetime, and only 14 percent had obtained such treatment during the year prior to their arrest. Receipt of mental health treatment services also was extremely low in this population despite a high prevalence of mental health problems (Hunt et al., 2015).
As is the case for pregnant women with OUD, there are important opportunities to identify and treat people in the criminal justice system who are at risk of progressing to more severe OUD and overdose. However, the most effective evidence-based approaches for addressing OUD and reducing overdose risk (Connock et al., 2007) have historically been inaccessible to people who are incarcerated in the United States. The social, medical, and economic benefits of providing MAT in correctional settings have been well documented (Deck et al., 2009; Dolan et al., 2003; Heimer et al., 2006; Kerr et al., 2007; Kinlock et al., 2009; MacArthur et al., 2012; Mattick et al., 2009; McKenzie et al., 2012; Rich et al., 2015; Zaller et al., 2013). Although the World Health Organization (WHO, 2009) and SAMHSA (Miller and Hendrie, 2008) have strongly endorsed the use of MAT to treat OUD in criminal justice settings, there has been little to no implementation or routine use of MAT in U.S. jail and prison settings (Lee et al., 2015; Vestal, 2016).
National household-based surveys exclude people who are incarcerated and other institutionalized populations. Thus, trends in the epidemiology of opioid use and misuse, OUD, and overdose in this large, underserved, and particularly vulnerable population often are missed, as is the chance to provide lifesaving treatment and medications to a high-risk population at a high-risk point in time. When new medications are approved for the treatment of OUD and overdose, it will be important for those drugs to be made available to individuals who are incarcerated. In addition to the enormous potential public health benefit of doing so, people involved in the criminal justice system are in contact with community corrections and
thus could provide key surveillance data points, thereby improving postmarketing surveillance and public health data capacity.
In summary, OUD is prevalent in criminal justice settings, and improved access to effective treatments and collection of surveillance data with which to track opioid use and associated harms in these settings are needed. The status of surveillance systems for collecting data on drug use among individuals involved in the criminal justice system and other populations is discussed later in this chapter.
People Who Inject Drugs
PWID are subject not only to the harms related to the drug itself but also to the harms related to injection. In particular, PWID are at risk of abscesses, tissue infections, ulcers at the site of injection, and endocarditis (Smith et al., 2014), and those who share syringes and other injection equipment also are at risk of contracting bloodborne infections such as hepatitis C virus (HCV) and HIV.
HCV, which can cause liver scarring and liver cancer, is spread primarily through blood contact, with the primary risk factor in the United States being injection drug use. In 2014, there were an estimated 30,500 cases of acute HCV infection in the United States and an estimated 2.7 to 3.9 million people living with chronic HCV (CDC, 2016a). HCV is now responsible for nearly 20,000 deaths annually in the United States—more than the number due to 60 other infectious conditions combined (Ly et al., 2016). The number of acute HCV infections had been declining steadily in the United States but reversed course and began to increase in the mid-2000s; since 2005, the estimated number of acute infections has more than doubled (CDC, 2016b). This increase in infections has been particularly pronounced among young, nonurban white people (Suryaprasad et al., 2014). Between 2006 and 2012, there was an estimated 364 percent increase in HCV infection among people under age 30 in Kentucky, Tennessee, Virginia, and West Virginia, for a total of 1,377 reported cases (Zibbel et al., 2015). Among the 265 cases for which risk information was available, 73 percent of infected persons reported injection drug use (Zibbel et al., 2015). The authors of this study note that during the same period, there was a surge in the number of young people in these states seeking treatment for OUD related to use of prescription opioids and heroin, suggesting that “the increase in acute HCV infections in central Appalachia is highly correlated with the region’s epidemic of prescription opioid abuse and facilitated by an upsurge in the number of persons who inject drugs in these four states” (Zibbel et al., 2015, p. 457). An analysis of national surveillance data showed similar trends, with 75 percent of young persons newly infected with HCV reporting that they had ever injected drugs and 75 percent report-
ing that they had ever misused prescription opioids (Suryaprasad et al., 2014). The authors conclude that all “available information indicates that early prescription opioid abuse and addiction, followed by initiation to IDU [injection drug use], is fueling increases in HCV infection among young persons” (Suryaprasad et al., 2014, p. 1417).
HIV attacks a person’s immune system and can lead to infections, cancers, and death. It is spread primarily through sexual activity, but 6 percent (2,392) of new diagnoses in the United States in 2015 were attributable to injection drug use, and another 3 percent (1,202) were due to injection drug use in addition to male-to-male sexual contact (CDC, 2017a). It is estimated that more than 171,000 people in the United States are living with HIV that is attributable to injection drug use (CDC, 2017a). In general, HIV diagnoses among PWID are on the decline, down 48 percent between 2008 and 2014 (CDC, 2017a). However, an increase in injection drug use in nonurban areas and in new populations has created new challenges in monitoring and preventing HIV transmission. High-risk practices—sharing needles, syringes, and other injection equipment—have declined among black and Hispanic PWID, but have not declined among their white counterparts. Young (under 30 years) and new (injecting less than 5 years) PWID are more likely than other PWID to share equipment (CDC, 2017a). High-profile HIV outbreaks have been seen in areas that were previously considered low-risk for HIV. In southeast Indiana, for example, a region that normally saw about 5 new cases of HIV annually, 169 people were diagnosed with HIV in the first half of 2015 (Strathdee and Beyrer, 2015). Most of these people were young and white and lived in rural communities, and the infections were linked directly to the preparation of the newly reformulated ADF Opana ER (oxymorphone ER) for injection (Strathdee and Beyrer, 2015). This development represents a major shift. Since the beginning of the HIV epidemic in the United States, most PWID who became infected with HIV were black men older than 35 who lived in urban areas, and most infections were associated with the injection of street drugs, not prescription medications (Strathdee and Beyrer, 2015). Effective interventions for reducing harm associated with bloodborne disease have a strong evidence base and include the provision of new syringes and needles through syringe access programs and point-of-sale pharmacy access to this equipment (CDC, 2015b; Hagan et al., 2011; Logan and Deutsch, 2015); however, many states recently affected by HIV and HCV increases, including Indiana, do not provide legal access to safe injection equipment. Further discussion on policies related to injection equipment is included in Chapter 5.
New data presented by the CDC at a March 13–14, 2017, advisory committee meeting reviewing ADF Opana ER (oxymorphone ER) suggest that ADF strategies and specific formulation components common to the
ADF versions of OxyContin and Opana ER had harmful effects on PWID and drove outbreaks of HIV, HCV, and thrombotic thrombocytopenic purpura-like illness (TPP)4 in this population (Brooks, 2017). Data from quantitative (case-control) and qualitative (focus group and interview) studies were analyzed to understand how the characteristics of drugs—and their subsequent use—influenced risks of infection and TPP. Findings indicated that in these communities, which had endemic prescription opioid misuse (with little heroin use), diverted prescription opioids were used in multiple injection events per day. Oxymorphone (the active ingredient in Opana), which is 10 times more potent than the equivalent morphine dose, led to more intense withdrawal in people who had developed OUD involving use of the drug. Opana ER—like OxyContin ER—is formulated with a crush-resistant coating, which drove many users who had been snorting their Opana to inject the drug. The reformulation, however, required multiple steps to be prepared for injection, and the preparation methods used involved the use of more solvents, which ultimately diluted the injection so that more injections occurred during the same injection episode. Also unique to preparation of Opana ER ADF (compared with injection use of other prescription opioids or heroin, for instance) was the use of “rinse shots” to extract all possible drug from the leftover materials. The increased street cost of Opana ER in the community incentivized cooperation and collaboration among people injecting the drug, creating more opportunities for transmission of HIV and HCV (Brooks, 2017). Additional data reported from a 2011 outbreak of HCV in New York State traced transmission to injection of prescription opioids, and in this case, Opana ER and OxyContin ER were the two most frequently injected opioids. These three instances illustrate well the risks of specific drug characteristics and drugs developed to treat pain that can be expected to be misused, diverted, and repurposed.
In summary, PWID are vulnerable to harms related to drug use. It is predictable that new medications with abuse liability will be used by people with established patterns of injecting drugs. Tracking the toll of expected nonmedical use of specific products on the health of people who inject drugs is of public health importance. For new formulations of opioids and other drugs that may be manipulated and injected, it is prudent to anticipate and fully examine the possible harms to health that might occur via injection routes. Data on harms can be collected through surveillance, but ethnographic and qualitative research also is required to understand use behaviors. When harm arises, involving PWID and their health advocates
___________________
4 TPP is a rare but serious blood disorder characterized by microangiopathic hemolytic anemia and thrombocytopenia (low blood platelet count). Intravenous drug use is a known risk factor for TPP (CDC, 2013).
in interventions that affect them can improve public health outcomes. Harm to this population can be minimized and treatment entry improved through safe access to injection materials.
HEROIN USE AND ITS RELATION TO PRESCRIPTION OPIOID USE
It is now abundantly clear that heroin use and trends in illicit drug markets have a substantial influence on the public health impact of prescription opioid use and misuse and OUD. One cannot weigh the importance of new therapeutics without taking full account of unintended harm from diversion and transition to illicit opioid use.
Trends in Heroin Use
Heroin, also known as diamorphine, is a synthetic derivate of the opium plant that can produce intense feelings of euphoria. Its use by humans traces to 1874, when it was synthesized from morphine and subsequently marketed as a medication. Now considered an illegal drug with no medical applications in the United States, diamorphine is currently used in some countries in palliative care or as medication treatment for people with OUD who have not responded successfully to other opioid agonist therapies (Strang et al., 2015).
Data indicate that heroin use has been rising in the United States in recent years among both men and women, in most age groups, and across all income levels (see Figure 4-2). The CDC notes that some of the greatest increases have occurred in demographic groups with historically low rates of heroin use, including women, the privately insured, and people with higher incomes. Of note, heroin use among people aged 18 to 25 more than doubled in the past decade (Jones et al., 2015).
Concomitant with increased heroin use over the past decade have been increases in heroin-related overdose deaths, heroin-related emergency department visits, and help seeking through treatment admission for OUD. Heroin-related overdose deaths have more than quadrupled since 2010, totaling more than 12,989 in 2015. Demographically, the highest rate for heroin overdose death (7.0 per 100,000) in 2013 was among non-Hispanic whites aged 18 to 44, a demographic that one decade earlier had been heavily affected by nonmedical use of prescription opioids, as reviewed earlier in this chapter. Importantly, there are geographic differences in heroin overdose rates, with the greatest burden being exhibited in the Northeast (6.3 per 100,000) and Midwest (6.1 per 100,000) (see Figure 4-3).
Trends in heroin use among those entering treatment have changed radically and quickly. A study of patients entering SUD treatment programs for OUD involving heroin nationwide examined retrospective reports on
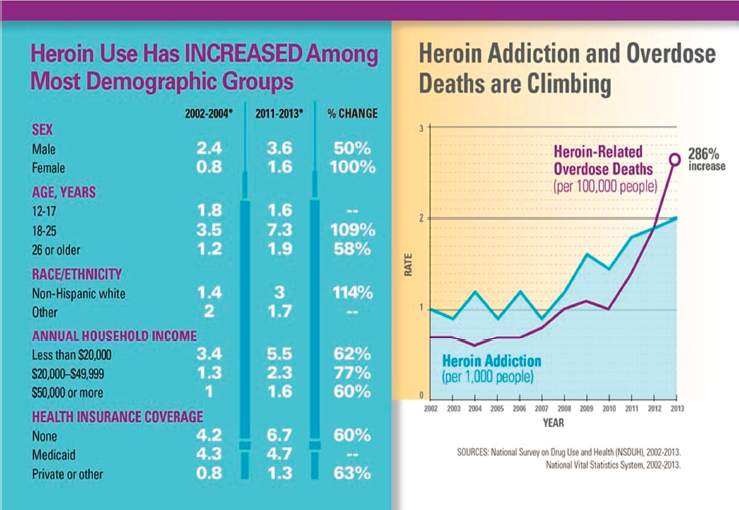
SOURCE: CDC, 2015c.
past drug use patterns (Cicero et al., 2014). Findings indicate significant changes in the profile of heroin users over the past several decades, from a previously predominantly inner-city, minority-centered problem to one that has more widespread geographic distribution. Users now comprise white men and women in their late 20s living outside of large urban areas who were introduced to opioids through prescription drugs and progressed to heroin in part because of its lower cost and greater availability (Cicero et al., 2014).
Interactions and Transitions from Prescription Opioids to Heroin
One of the most urgent concerns posed by the widespread increase in prescription opioid use and consequent misuse beginning around 2000 is how this epidemic is influencing current trends in the use of heroin and fentanyl and mortality due to overdose involving these drugs. A number of studies have yielded evidence strongly supporting the conclusion that the recent prescription opioid epidemic has resulted in a significant increase in domestic heroin use and associated overdose deaths (Al-Tayyib et al., 2017; Jones, 2013a; Muhuri et al., 2013). The rate of heroin overdose
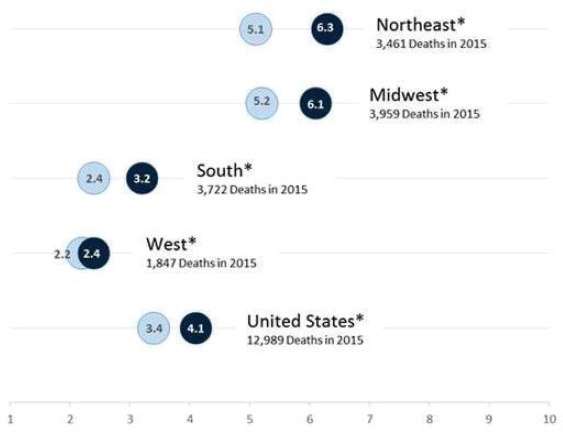
*Statistically significant at p <0.05 level.
SOURCE: Adapted from Rudd et al., 2016.
increased moderately from 2006 to 2010 but more than tripled from 2010 to 2014 for all age groups (see Figure 4-4), with the greatest increase occurring among those aged 25–34 (CDC, 2017b). Data for 2015 indicate that the rate of heroin overdose continued to climb, reaching a rate of 4.1 per 100,000 population, more than four times the rate in 2010 (Rudd et al., 2016). Furthermore, from 2007 to 2013, rates of past-year nonmedical use of or OUD involving heroin increased nearly 150 percent (Jones et al., 2015). While societal factors have certainly contributed to this trend, a major concern is how prescription opioids contributed to this problem both by serving as “gateway” drugs to heroin use (Muhuri et al., 2013) and by “squeezing the balloon” through focused efforts to reduce their misuse (e.g., the development of ADFs), leading to illicit sources and drugs such as heroin (Unick et al., 2013).
One issue to keep in mind in this discussion is the relative size of the heroin and prescription opioid epidemics. Heroin historically has attracted only a small number of chronic users in the United States. In terms of the number of people regularly using opioid medications (for pain or nonmedical reasons), the prescription opioid epidemic is many orders of magnitude larger than the endemic level of heroin use. This means that an unprecedented number of people are potentially vulnerable to meeting their
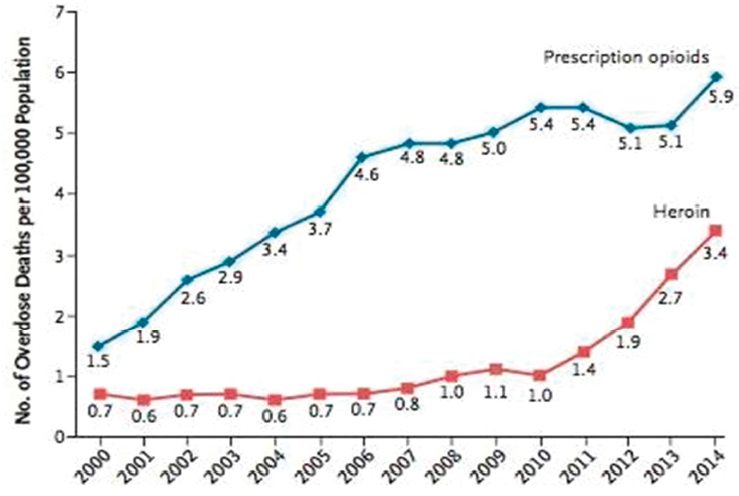
SOURCE: Adapted from Compton et al., 2016.
opioid use needs with heroin. Understanding how the dynamics of these two current epidemics overlap and the motives of people switching from pills to heroin is a critical challenge.
Prescription Opioids as a Gateway
The gateway theory of the movement of prescription opioid users to heroin is predicated on the fact that opioid medications produce the same neuropharmacologic effects as heroin, so the substances are natural substitutes. Use of both heroin and prescription opioids involves tolerance, cross-tolerance, and withdrawal. Yet heroin is, on balance, more potent than the most common low-dose prescription opioids (e.g., codeine, Vicodin, Percocet). This is true of even fairly low-purity (<30 percent) heroin, but has become even more evident with recent increases in heroin purity rates in some cities (Gray, 2014). The implication is that as people become tolerant to a dose (i.e., level) of opioid medication and no longer feel the desired effects of the drug, they may use heroin and thereby feel more intensely and rapidly effects that pills once may have produced. As discussed in Chapter 3, anyone consistently using these medications is likely to experi-
ence tolerance, which may lead to taking opioid medications in amounts greater than prescribed (Webster and Webster, 2005).
Moreover, initial use of opioids to treat pain may shift to chronic use. In an analysis of linked health care claims, Shah and colleagues (2017) found that the probability of long-term prescription opioid use increased markedly in the initial period of therapy, especially after 5 days or 1 month. Over this initial course of care, tolerance develops and can, if the patient is not tapered off the drug and cared for safely, lead to dependence and OUD. While other factors may influence the transition to heroin use, the point is that the risk of this transition is great for people prescribed opioids, and those initially prescribed the drugs for longer periods or in larger doses (i.e., ER opioids) tend to stay on opioids.
For many people who misuse opioids, switching to heroin also involves an associated transition to a more potent route of administration—e.g., injecting—either before or in conjunction with initiation of heroin use. It is true that most prescription opioids are swallowed, but depending on their formulation (and the knowledge of the person misusing) they also can be sniffed, smoked, chewed, sucked, or injected. In the United States, heroin is most commonly injected—the fastest route of administration—which introduces a host of additional public health consequences (discussed earlier regarding PWID). Heroin (along with fentanyl) is more potent than opioid analgesics (NIDA, 2016), and the potency of opioid analgesics is influenced by the route of administration. The differences in potency and onset of effects among orally ingested opioid medications, snorted or injected prescription opioids, and injected heroin places a person making the switch away from oral routes at much higher risk for overdose. Moreover, to someone tolerant to and misusing prescription opioids, ER opioid formulations and heroin offer a much more rapid onset of effects relative to prescription IR formulations. In this manner, ER opioids and heroin can reset the reward pathway, giving people who make this switch a powerful incentive to continue using them. Efforts to make ER opioid formulations less accessible and/or “abuse-deterrent” and black market efforts to make heroin more readily available, then, may tilt the reward mechanism in favor of seeking heroin.
It is important to acknowledge that an overwhelming majority of people who use prescription opioids do not continue to use them chronically (Shah et al., 2017), and so are not at risk of switching to using heroin. However, for those that do use chronically and then move to heroin through this pathway, the movement is typically one-way. Once a person has begun using heroin consistently, returning to a pattern of primary use of prescription opioids is unlikely for a variety of reasons, including heightened scrutiny by health care providers and the relative expense (see below for discussion of opioid markets) (DEA, 2013). Chronic users of heroin seldom
consume prescription opioids and typically do so only to delay withdrawal when heroin is episodically unavailable; when informally seeking to reduce their heroin intake; or, more recently, when protecting themselves against fentanyl-contaminated heroin.
Further promoting such transitions to heroin among persons previously using prescription opioids is the financial incentive for switching, since heroin is considerably cheaper than street-available pain medications (DEA, 2013). In locations where both illicit prescription opioids and heroin are available, drug users consistently report that prices are lower for heroin. This price difference has always existed. Heroin also has a much lower initial market entry price than that of opioid pills for new users (e.g., a bag of heroin sells for $10, while a pill might cost $20), but few people start with heroin because its use is stigmatized.
Market Effects and the Transition to Heroin
Differences in drug prices are complex and often a consequence of how the markets operate. For instance, the supply of legal prescription opioids is controlled and can therefore be restricted—for example, when a pill mill is shut down or an opioid is reformulated with abuse-deterrent properties (see discussion on OxyContin reformulation below and related discussion in Chapter 5). These medications also are sold in what can be described as a secondary market, meaning the drug is first diverted from some legitimate source to be resold illegally, which is costly and raises the price. As discussed further in the next section, these markets are now growing. Even within expanding markets for counterfeit opioid medications and illicitly manufactured synthetic opioids, moreover, the latter products remain less expensive to purchase than most opioid analgesics, both diverted and counterfeit.
Part of the reason for the price difference between illicit prescription opioids and heroin is that heroin supplies coming into the United States are largely unrestricted (other than by the sorts of supply-related control measures that may restrict opioid medications). In many places where heroin is sold, sales are well-organized and have the support of an established black market infrastructure. Therefore, all other things being equal, once a person starts using heroin, acquiring it consistently may become easier and less expensive relative to pills. As tolerance increases and if OUD progresses, evidence-based treatment may be the only intervention able to disrupt this cycle.
The important regional variations in the numbers of people switching to injection use and to heroin from prescription opioids noted earlier reflect such market factors. One reason especially high rates of prevalence of prescription opioid use did not immediately lead to extensive heroin use
in rural communities may be that heroin was not yet as entrenched and available in these locations. For instance, consistently low rates of heroin use have been seen in a cohort of rural Appalachian injectors in Hazard, Kentucky, even after reformulation of OxyContin and Opana (Havens et al., 2014). But more recent state and local data on overdose deaths, treatment entry, and arrests indicate that heroin is now surging in these same areas. The substantial delay in heroin uptake in these areas may be linked to shifts in drug trafficking patterns, localized interventions to reduce the supply of diverted opioid medications, or changes in the social structure created alongside the pill-based economy (Jonas et al., 2012).
Quantifying the Degree of Overlap
Although a number of factors have prompted people to move from use of opioid medications to use of heroin, quantifying precisely how many people have made this switch is difficult. Yet a number of studies suggest that an alarming overlap has occurred, and is still occurring, between these two epidemics. Authors of a national study of people who use heroin (Cicero et al., 2014) note that an important demographic shift has occurred in recent years. Over the past 50 years, the population of people using heroin has transformed to mirror the population of people using and misusing prescribed opioids. People who use heroin now are primarily younger and non-Hispanic white. Those who have an OUD involving heroin today are very different from their counterparts only 10 years ago, but much more like the people affected by the prescription opioid epidemic. In asking whether people who use heroin begin doing so before or after using prescription opioids, these authors identified a complete reversal from the 1960s: almost all people who initiated heroin use in the 1960s started with heroin, whereas almost all those who began using heroin in the 2000s began with the use of prescription opioids (Cicero et al., 2014).
One large cohort study and a number of regional studies confirmed that a majority of people who had recently started using heroin began by misusing opioid medications. In the first published study on this topic, Siegal and colleagues (2003) found that 50 percent of young persons (aged 18–33) in Ohio who had recently started using heroin reported first having misused opioid medications, primarily OxyContin. A number of similar studies yielded a similar finding, although rates of prior opioid misuse varied. A large study of illicit and prescription drug misuse in young urban people in New York and Los Angeles in 2008 and 2009 found that 73 percent had a lifetime history of obtaining a prescription for opioids and initiated prescription misuse at a younger age relative to use of heroin, suggesting that nonmedical opioid misuse may serve as a gateway to initiation of heroin use (Lankenau et al., 2012). Studies of heroin users in San Diego (Pollini et
al., 2011), Seattle (Peavy et al., 2012), and New York City (Mateu-Gelabert et al., 2015) found that 40 percent, 39 percent, and 77 percent of heroin users, respectively, were users of nonmedical opioids before initiating heroin use. In a more recent sample of PWID in Denver, 32 percent reported being “hooked” on prescription opioids before injecting, and the primary drug they injected was heroin (Al-Tayyib et al., 2017). Finally, in a large, matched cohort of aging U.S. veterans who reported no previous history of nonmedical prescription opioid or illicit opioid use, Banerjee and colleagues (2016) found that nonmedical use of prescription opioids was associated positively and independently with subsequent initiation of heroin use.
An analysis of data from the National Survey on Drug Use and Health (NSDUH), the only nationally representative study of self-reported drug use behavior in the United States, supports the conclusions of the above cohort and regional studies, although it is important to note that household surveys have unavoidable limitations for use in assessing high-frequency use of drugs such as heroin (Caulkins et al., 2015b). Using NSDUH data pooled from 2002 through 2011, Muhuri and colleagues (2013) noted that, among individuals aged 12–49, four of every five recent heroin initiates (79.5 percent) (i.e., those who had initiated heroin use within the past 12 months) were previous self-reported users for purposes of nonmedical pain relief (NMPR) (see Figure 4-5).
The analysis by Muhuri and colleagues (2013), which included approximately 609,000 respondents at risk for heroin initiation and 524,000 respondents at risk for NMPR use, is notable because it found that only a small percentage (3.9 percent) of NMPR users initiated heroin within 5 years after first using NMPR. The NSDUH, however, is a household-based sample that excludes institutionalized populations, homeless individuals, and others, and thus likely underestimates these outcomes. The small incidence rate also is deceptive because of the large annual number of new heroin users it represents. As others have noted, “given the large number of nonmedical users, even a small percentage who initiate heroin use translates into several hundred thousand new heroin users” (Compton et al., 2016, p. 158). Applying the 3.9 percent incidence rate to the 25 million Americans who ever initiated NMPR use between 2002 and 2011 (SAMHSA, 2012) indicates that the prescription opioid epidemic created nearly 1 million new heroin users in this 10-year time frame, or roughly 100,000 annually. Given underreporting, the correct number may be considerably higher still.5
___________________
5 It is important to note that until 2015, the NSDUH instrument posed questions regarding “misuse” in terms of two behaviors: using the medication in ways other than prescribed and using it for the way it makes one feel. In 2015 the latter query was eliminated. Because of this change, estimates of misuse from the NSDUH before and after the change was made are not entirely comparable.
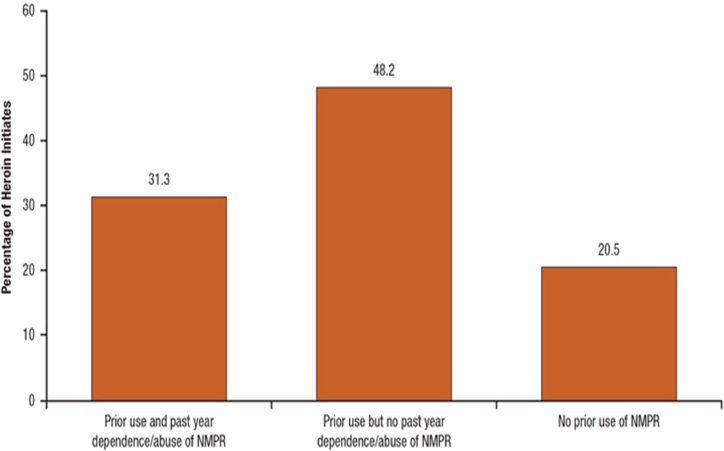
NOTES: Past-year NMPR users are those who had initiated NMPR use prior to initiation of heroin use in the past 12 months. Past-year NMPR users who initiated NMPR use subsequent to initiation of heroin use in the past 12 months are not included. Dependence or abuse is based on self-reported problems and definitions found in the Diagnostic and Statistical Manual of Mental Disorders, fourth edition (DSM-IV).
SOURCE: Muhuri et al., 2013.
Alarmingly, data from other sources are consistent with this projection. The most recent estimate from a RAND Corporation report prepared for the Office of National Drug Control Policy (ONDCP) suggests there were 1.5 million chronic heroin users in the United States in 2010 (the latest year estimated) (Kilmer et al., 2014). Based on this “high” projection, 400,000 more chronic heroin users existed in 2010 than in 2002. The estimated number of chronic heroin users remained fairly stable between 2000 and 2007, but from 2007 to 2010 increased 25 percent (see Figure 4-6). During 2007–2010, the rate of new chronic heroin users was >100,000 annually, keeping in mind that these calculations are conservative because they are based on the noted underestimates of the rate of initiation of heroin use from the NSDUH. Based on these estimates, starting from 2010 and assuming 100,000 new heroin users annually, the prescription opioid epidemic could at least double the number of heroin users in the United States by 2025.
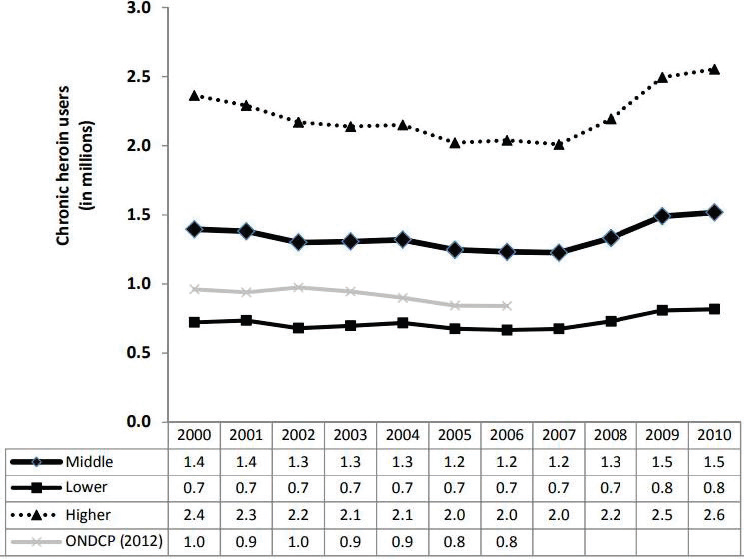
NOTE: ONDCP = Office of National Drug Control Policy.
SOURCE: Kilmer et al., 2014.
A preponderance of evidence suggests that the major increase in prescription opioid use beginning in the late 1990s has served as a gateway to increased heroin use. Two questions remain: How costly, in terms of heroin mortality, has this connection been? and What does this mean if prescription opioid supplies are curtailed? As in the findings cited above, the year 2010 is an important turning point for addressing these issues.
Marketed aggressively in a campaign that began in 2000, OxyContin—developed by Purdue Pharma in 1996 and the most popular opioid medication in history—is widely regarded as the drug that initiated the current opioid medication misuse epidemic. A critical factor in the initial epidemic was that many people were able to misuse OxyContin by crushing, dissolving, and injecting the drug. All routes of administration were available, and presumably, early in this epidemic, many individuals who misused the drug were particularly vulnerable to using heroin (if locally or regionally available) because they had progressed beyond barriers posed by injection. This trend in OxyContin misuse progressed unabated until August 2010, when at the request of the FDA, an ADF of OxyContin was introduced, after which it became more difficult for people to crush, snort, and inject
the drug. Yet the reformulation of OxyContin to an ADF led some users to abandon the drug entirely (e.g., for treatment), while others moved to other drugs or routes of administration and still others switched to heroin. Cicero and Ellis (2015) found that 33 percent of nonmedical OxyContin users had adapted to the OxyContin ADF by using other drugs, and 70 percent of that group had switched to heroin.
The importance of OxyContin and the change to its ADF formulation offered Alpert and colleagues (2017) an opportunity to conduct a unique analysis to assess how this policy influenced both opioid medication misuse and heroin mortality. Notably, using NSDUH data (again noting the limitations of this household survey described earlier) and comparing states with high and low rates of OxyContin misuse, the authors found that before 2010, no correlation existed between trends in heroin mortality and opioid misuse; death rates for heroin during this time period were stable. By contrast, in the years after the reformulation (2010–2013), “each additional percentage point of pre-reformulation OxyContin misuse is associated with a relative decrease in OxyContin misuse of 0.8 percentage points and an additional 2.5 heroin deaths per 100,000 through 2013” (Alpert et al., 2017, p. 5). In other words, the reformulation decreased opioid medication misuse as intended but substantially increased heroin mortality. This finding led the authors to conclude that for each percentage point reduction in misuse of OxyContin generated by its reformulation, there was an increase in heroin-related deaths of 3.1 per 100,000. When the authors applied their calculation to increased heroin mortality rates between 2010 and 2014, 80 percent of the increase in those rates was explained by OxyContin’s reformulation. As noted by the authors, the reformulation of OxyContin to an ADF had different short- and long-term outcomes. In the short term, the change increased heroin-related overdose deaths, while in the long term it reduced (or at least leveled) prescription opioid misuse, which could potentially reduce heroin deaths down the road (Alpert et al., 2017).
Finally, increases in the numbers of individuals who use heroin over the past decade of the prescription opioid epidemic entail important independent dynamics. With more new heroin users entering the market every year, it has become much easier for people to start using heroin directly, without first using prescription opioids. Thus, in addition to individuals who formerly misused prescription opioids, individuals whose heroin use began recently include those who were not influenced by the gateway effect of prescription opioid medications. As a result, heroin may become much more mainstream, appearing to have crossed a threshold that has historically restricted its popularity, so that the movement to direct use of heroin is occurring in the context of a social contagion fueled by the many heroin users produced by the prescription opioid epidemic. In short, the demographic shift in heroin use among persons who are rural, white, and
geographically isolated as well as those who are suburban, young, white, more educated, and from middle-class backgrounds may be facilitating the popularity of heroin by slowly eroding long-standing stigmas that have prevented people from using this drug in the past. The potential waves of new heroin users naïve to opioids are particularly alarming and may explain why heroin and synthetic opioids (fentanyl) have been increasing exponentially the numbers of heroin-related overdose deaths since 2010. Thus, in addition to initiating and continuing to directly feed the current heroin epidemic by facilitating people’s switch to heroin, the prescription opioid epidemic may have mutated into a new and independent heroin epidemic.
Summary
The prescription opioid and heroin epidemics are intertwined. One of the consequences of increased prescribing of opioid analgesics has been increases in the use of heroin; in associated overdose deaths; and in the incidence of HIV, HCV, and other injection-related harms. In addition to prescription opioids serving as a gateway to use of heroin, market forces and efforts designed to reduce harms associated with use of prescription opioid medications (e.g., ADFs) may be contributing to increased heroin use. And given the comparatively small population of heroin users relative to that of prescription opioid users, there is currently an unprecedented potential market for heroin use.
ILLICIT OPIOID MARKETS
While it is reasonable to presume for many prescription medicines that consumption is limited substantially to those to whom the drugs were prescribed, this is not the case for all medications, including prescription opioids. Prescription opioids may be diverted (e.g., through resale, theft, or other means) to illicit markets that are the proximate cause of considerable harm (OUD and overdose). Furthermore, these markets for diverted prescription opioids interact with purely illegal markets for opioids that are not supplied through the U.S. health care system (Unick et al., 2013), as well as with the dark web of vibrant online drug cryptomarkets (Aldridge and Décary-Hétu, 2016). Traditionally, markets for purely illegal opioids pertained primarily to heroin, but they have been expanding to encompass new psychoactive substances, most recently and infamously synthetic opioids such as fentanyl and its analogs (e.g., acetyl fentanyl, ocfentanil, carfentanyl) that are packaged and sold in bulk from abroad to drug trafficking organizations or even as counterfeit pills made to look like popularly diverted prescription opioid medications. Thus, part and parcel of creating the supply of prescription opioids for treatment of chronic
pain are increases in the supply to and demand for black markets for opioids, with all of their attendant harms, including violence, corruption, and incarceration.
History of Illicit Opioid Markets
Prescription opioids did not create the black markets for illegal opioids. The illicit opioid markets already had a long history in the United States. In fact, their prominence is reflected in the very names of such institutions as the Bureau of Narcotics and Dangerous Drugs (the predecessor of today’s U.S. Drug Enforcement Administration [DEA]) and in the fact that “narc” is a slang term for a drug enforcement officer. However, large-scale misuse of prescription opioids created new demand that substantially reinvigorated, expanded, and diversified those markets.
The illegal opioid markets saw ebbs and flows before the expansion of prescription opioid misuse. A surge of use occurred after World War II, but it had been largely contained by the 1960s (President’s Commission on Law Enforcement and Administration of Justice, 1967). Another, larger epidemic of heroin use took place in the late 1960s and early to mid-1970s, but that, too, was quelled by a combination of interventions on the demand side (early deployment of methadone) and supply side (Turkish poppy ban and breaking of the “French Connection”) (DuPont, 1971, 1973, 1974; DuPont and Greene, 1973; Kaplan, 1983).
The heroin market was not completely stable between the mid-1970s and mid-1990s. The source of supply shifted markedly, from Mexico to Southwest Asia to Southeast Asia to South America (DEA, 2016b, p. 47). Heroin purity rose between the 1980s and 1990s, and purity-adjusted prices fell sharply (DEA, 2016c). But initiation was low, and use had remained substantially confined to an aging group of mostly men in major urban centers, predominantly in the Northeast and Southwest. Notably, availability was quite limited in most small cities and rural areas.
The heroin market was revived in the mid-1990s by a new source of initiation in the form of people whose opioid misuse had started with prescription opioids who transitioned to cheaper, and riskier, black market opioids (see Figure 4-7). This influx changed the demographic composition of the user base (Cicero et al., 2014; Muhuri et al., 2013), roughly doubled initiation into heroin use, and much more than doubled demand because all of these new initiates were experienced opioid users.
The effects can be seen not only on initiation but also on the ages of those seeking treatment. Among those in the Treatment Episodes Data Set (TEDS) records as seeking treatment for heroin as their primary drug of use, in 1993 two-thirds were between the ages of 30 and 44. Twenty years later, in 2012, that proportion had fallen to one-third. The absolute numbers had
SOURCE: Committee analysis of 2003–2014 National Survey on Drug Use and Health (NSDUH) data.
declined by 20 percent (from 124,000 to 98,000), whereas the corresponding numbers for those under the age of 30 had grown by 150 percent (from 49,000 to 124,000) (SAMHSA, 2013b).
A further shift in supply occurred as well, with heroin produced in Mexico eclipsing that produced in South America. Seizures of heroin along the Southwest border of the United States began to increase sharply after 2007 (DEA, 2016b, p. 46). Importantly, retail distribution expanded into smaller cities and rural areas and to parts of the country, such as the Midwest, that previously had had lower availability. It is unclear whether that expansion in availability was demand-driven (supply reached out to where the new users lived), supply-driven (heroin distribution from other countries piggy-backing on networks that already had broad geographic reach for the delivery of cocaine and methamphetamine), or both.
In the past few years, two “new” and potentially very important product forms—fentanyl and counterfeit opioid pills—have proliferated in North American black markets for illegal opioids. The word “new” is in quotes
because little that happens in black markets is truly unprecedented (Baum, 1985). Rather, what is new is that these products are becoming common, not exceptional. For example, the DEA (2016a) reports that before 2014, the National Forensic Laboratory Information System (NFLIS) recorded more than 1,000 fentanyl exhibits only in a single year—2006, when a fentanyl “crisis” was associated with production tracked primarily to a clandestine lab in Toluca, Mexico. Yet by 2015, NFLIS recorded 13,002 fentanyl exhibits, more than 8 times the 1,594 exhibits observed during the 2006 crisis.
Present-Day Illicit Opioid Markets
Today’s illicitly manufactured fentanyl may have multiple sources that are diversifying and expanding. Much illicitly manufactured fentanyl is reputedly produced in the same areas (and perhaps even the very same factories) that produce legal medications for distribution by pharmaceutical companies (DEA, 2016a). Black market drugs often move through complex pathways, but the DEA believes a common pathway is bulk shipments from China to drug trafficking organizations in Mexico and thence across the Southwest border, although some of the drugs may also be produced in Mexico. The fentanyl may be sold straight up at retail, but also is mixed into heroin as an extender and increasingly into other drugs such as cocaine. This practice is facilitated because trafficking organizations now distribute various powdered forms of heroin, not just the traditional “black tar” heroin, which cannot as easily be adulterated with fentanyl.
An economic incentive exists for trafficking organizations to “extend” heroin with fentanyl or to sell fentanyl outright. Fentanyl is thought to be 25 to 50 times more potent than heroin (DEA, 2016d). As a synthetic opioid, it is more economically appealing than natural opioids such as heroin. The Western States Information Network (WSIN) (2016) reports kilogram prices of heroin ranging from $17,000 to $30,000 for Mexican brown and $20,000 to $46,000 for Colombian white (which is also distributed by Mexican drug trafficking organizations), two forms that can readily be cut with fentanyl (Southeast Asian heroin is somewhat more expensive, but has a very minor market share in the United States, especially in the West). While fentanyl manufactured in a lab could be purchased at prices below that of heroin per kilo (DEA, 2016a), fentanyl’s potency allows it to be diluted more and still deliver a dangerous dose. In this way, a kilo of drug can be multiplied into 10 to 20 kilos or more of drug for street sale with the addition of fentanyl products. There exist at present only anecdotal reports of wholesale fentanyl prices, but the DEA (2016a, p. 8) cites instances of a distributor selling fentanyl for $3,500 per kilogram, while the DEA’s Miami Field Division reports that fentanyl could be purchased for $1,700 per kilogram (DEA, 2016a, p. 8).
The other factor that affects relative price is competition and the presence of substitute products. As with many new synthetic psychoactive products, manipulation of fentanyl contributes to the creation and proliferation of fentanyl analog products in the illegal drug trade and cryptomarkets (Quintana et al., 2017) and a ready source of replacement chemicals.
If fentanyl in wholesale markets costs about one-tenth as much as heroin but is 10–25 times as potent on a pure milligram basis, then heroin “per unit of intoxication” from the customer’s perspective is 10–25 times more expensive for drug traffickers. Thus, there is an incentive to adulterate heroin (and other drugs) with fentanyl to reduce the costs of materials.
Prices in illegal markets adjust slowly, perhaps because of poor information flows, but they are competitive, and in the long run prices tend to fall in parallel with production costs, at least if one understands costs broadly to include compensation for the various risks involved in distributing drugs (Caulkins and Reuter, 2010; Reuter and Kleiman, 1986). One should not be surprised, then, if over the next half-dozen years, fentanyl continues to displace heroin in illegal opioid markets, and its prices continue to fall, perhaps very substantially.
A related phenomenon is the selling of counterfeit prescription opioid pills, often laced with or containing only fentanyl. The logic for the fentanyl adulteration is compelling. Fentanyl, as noted, is cheaper than heroin, and heroin is cheaper than prescription opioids, so fentanyl-laced counterfeit pills are markedly cheaper than are diverted pharmaceuticals. That this is so is not really surprising, given that production costs for many pharmaceuticals are just a tiny fraction of their sales price in the United States.
Pressing pills is not difficult. Pill presses are not regulated and can be purchased openly in some countries. (It is illegal to bring presses into the United States without notifying the DEA, but criminal organizations ignore that law or do the pressing in other countries.) The DEA (2016a, p. 9) cites prices of under $1,000 for a press that can produce 5,000 pills per hour and die molds selling for a little over $100, so the equipment costs are negligible given that pills often sell for $20 apiece at retail, and perhaps $6.50 per counterfeit pill in bulk. And while it may be difficult to meet the exacting standards for legal pharmaceutical pills, it is not difficult to make counterfeit pills that are potent and indistinguishable from true pharmaceutical pills to the casual observer. Moreover, the street-based purchase environment for the illicit drug consumer often is not conducive to thorough inspection of pills to verify indicia, color, weight, and shape (Green et al., 2015a). Counterfeit pills may serve a purpose for suppliers as well: they may be a relatively safe means of transporting some of the most potent fentanyl analogs (e.g., carfentanyl), and may be perceived as a more economically efficient and controlled dosing mechanism than powdered fentanyl or contaminated illicit powder drugs (if the fentanyl quantity contained in the pill is known to the
supplier or purchaser) (Green and Gilbert, 2016). The proliferation of a counterfeit prescription opioid market into the foreseeable future is likely.
Whether the trafficker is pressing it into pills, dividing it to sell outright, or using it to adulterate other powdered illicit drugs, fentanyl’s chemical properties leave little room for error. Its potency means that very small quantities can be lethal, and it is sometimes difficult for black market producers to mix and dilute powders with sufficient precision to avoid inadvertently selling quantities that contain a lethal dose. (It is easier to reliably dilute and prepare fentanyl solutions, which can be delivered via metered dose, either intranasally or intravenously, as is typically performed by anesthesiologists in hospitals.)
Again, while prices in illegal markets do not always arbitrage away price gaps swiftly, they tend to do so over time. So as with fentanyl displacing heroin, one can envision counterfeit pills displacing diverted pharmaceutical pills in the coming years, at least for those who have developed OUD. It will be important to track the public health implications of the fentanyl and counterfeit market displacements on the symptoms, prevalence, and severity of OUD.
Smaller-Scale Diversion to Illicit Markets
Thus far, this section has been addressing traditional black markets that involve long distribution chains through which organized criminal groups connect users to (mostly) overseas production. There exists another form of illegal market in which smaller quantities of prescribed medications are diverted and sometimes even sold. This is a sort of retail-to-retail distribution more akin to heavy cannabis users growing their own and selling to other users on the side.
It has long been understood that prescription drugs get diverted into illegal markets in multiple ways (Inciardi et al., 2007), but solid estimates of the relative magnitude of these channels are lacking, for reasons that also have long been understood (Inciardi et al., 2009). It appears that most of the diversion is carried out by individuals who receive prescriptions lawfully rather than through robberies of pharmacies or delivery trucks and other diversion from the legal, wholesale supply chain.
To understand why, it is important to get a sense of scale. It has been estimated that the United States consumes 39,487 defined daily doses (DDDs)6 of opioids per million inhabitants per day (Häuser et al., 2016).
___________________
6 DDD refers to “the assumed average maintenance dose per day for a drug used for its main indication in adults.” It does not necessarily correspond to the recommended or prescribed daily dose for a given patient, which will often differ from the DDD based on such characteristics as age and weight, as well as pharmacokinetic considerations (WHO, 2003).
Multiplying by the U.S. population of 320 million and by 365 days per year indicates that there are approximately 4.6 billion DDDs of opioids per year in the United States.
Respondents to the 2014 NSDUH self-reported 564 million days of use of prescription pain relievers that were not prescribed for them or were taken “for the experience or feeling it caused.” As an aside, the majority (61 percent) of those days was among respondents who self-reported enough problems with drugs or alcohol to be judged as meeting the criteria of the Diagnostic and Statistical Manual of Mental Disorders, fourth edition (DSM-IV), for abuse or dependence on drugs or alcohol, and 43 percent was among respondents who met those criteria specifically for “abuse or dependence on prescription pain relievers.”
Surveys, moreover, can underestimate drug consumption as a result of respondents’ social desirability concerns or inability to recall, among other reasons. Even for alcohol, it has been found that survey self-reports account for only about half of the alcohol known to be sold based on tax records (Cook, 2007). Thus, the 564 million self-reported days in the NSDUH may correspond to more like 1 billion actual days. If the average dose per day for NSDUH respondents equals the DDDs underpinning the 39,487 DDDs per million figure, then dividing that 1 billion by the 4.6 billion DDDs posited above, one might speculate that very roughly 20 to 25 percent of prescription opioids in the United States are used nonmedically.
The DEA (2016b, p. 34) reports that in recent years, distributors in the United States disbursed 12–15 billion dosage units of opioid narcotics to retail-level purchasers, suggesting that total diversion is on the order of 2.5–4.0 billion dosage units. By contrast, the DEA (2016b) reports that in the entire country in 2015, only 9.1 million dosage units of opioid narcotics were lost to diversion from the supply chain (e.g., from robberies of pharmacies), while another 1.9 million dosage units were “lost in transit.” Those are small numbers compared with the 12–15 billion dosage units disbursed to the retail level and the speculation of something like 2.5–4.0 billion units diverted.
A small number of high-volume, corrupt prescribers can provide substantial supply. ProPublica, for example, reported on Medicare’s top 20 OxyContin prescribers for 2010.7 The 12 prescribers who were charged, were fined, and/or had their medical licenses revoked wrote 17,000 OxyContin prescriptions and more than 56,000 prescriptions for narcotics of all kinds in 2010. Those are prescriptions, not dosage units, and there are many more than just a dozen corrupt doctors. Still, it is not clear that a handful of extreme prescribers can account for a number of dosage units in the billions.
___________________
7 See https://projects.propublica.org/checkup/oxycontin (accessed January 30, 2017).
There is slightly better information from the other direction on where people obtained the analgesics they used nonmedically. It is clear from the NSDUH and other sources that many people who use prescription analgesics nonmedically obtain them for free from friends or family, and it is believed that in turn, most of those friends and family obtained those drugs from a single doctor (DEA, 2016b; Hughes et al., 2016; Kennedy-Hendricks et al., 2016). However, for drugs, and for that matter many other consumer goods, a minority of heavy users account for a disproportionate share of consumption. In the 2014 NSDUH, two-thirds of those answering the question about where they most recently had obtained pain relievers for nonmedical use reported use on 50 or fewer days in the past year (i.e., less than weekly), and those users accounted for just 14 percent of the self-reported days of use. To the extent that frequent users also tend to use more per day of use, their share of market demand was even smaller. Conversely, the 8 percent of those respondents who said they had used on 180 or more days in the past year (so every other day or more often) accounted for almost half of the days of use, and presumably well more than half of the consumption. This means that statistics based on numbers of users can differ sharply from those based on a measure related more closely to market demand. For example, people who reported in the 2014 NSDUH that they had obtained nonmedical analgesics most recently by purchasing them—whether from a friend, relative, dealer, or other stranger—tended to be heavy users. So even though they represented just 14 percent of respondents who had used analgesics for nonmedical reasons, they accounted for 25 percent of the self-reported days of use (SAMHSA, 2014).8
It is worth noting as well that some people who had acquired the drugs most recently by some relatively innocuous means may also have purchased them or obtained them by fraud at other times. Respondents who reported use within the past 30 days account for the majority of days of use, and the NSDUH asks respondents to “Please enter all of the ways that you got the prescription pain relievers you used in the past 30 days.” In 2014, fully 39 percent of those individuals reporting days of use indicated that they had bought the drugs at some point in the past month, from a dealer, friend or relative, or the Internet. Another 5 percent denied purchasing but admitted to other illegal behavior (stealing, obtaining fake prescriptions, or taking from a friend or relative without asking), and a further 5 percent had neither bought nor scammed, but had obtained from multiple doctors. Based on these findings, perhaps roughly half of current nonmedical consumption is among people who engage in such tactics at least some of the time. To be clear, this does not mean that half of nonmedical analgesics are obtained using these tactics. Even among the 500,000 respondents who reported
___________________
8 Committee calculations. Variable ANLLTS2 = 6 or 8.
buying from drug dealers, 20 percent said they also had obtained in the past month from a single doctor.
This pattern is not new. Figure 4-8 shows that if anything, the proportion of current demand attributable to people who buy analgesics for nonmedical use at least occasionally has been greater in previous years.
Furthermore, all of these statistics apply to those who responded to the questions on this household survey, and household surveys fail badly at capturing the behavior of most problematic users. Caulkins and colleagues (2015a), for example, observe that the NSDUH suggests there were only 60,000 daily or near-daily heroin users in the United States, whereas Kilmer and colleagues’ (2014) more comprehensive estimate, drawing on the Arrestee Drug Abuse Monitoring (ADAM) system, among other sources, puts the figure closer to 1,000,000.
If the people who fell outside the NSDUH’s sampling frame were unwilling to complete the survey, skipped these questions, or did not respond truthfully were more heavily involved in diversion relative to those who answered the survey questions forthrightly, then the extent of diversion may be even greater than is suggested by this discussion. Omitted and
SOURCE: Committee analysis of 2005–2014 National Survey on Drug Use and Health (NSDUH) data.
untruthful responses by individuals within the NSDUH’s sampling frame also are a potential source of bias. What is clear is that the scale of diversion is sufficient to enable such organizations as StreetRx.com (Dasgupta et al., 2013) and WSIN (2016, p. 27) to quote black market prices with high geographic specificity not only for such staples as oxycodone, methadone, and hydromorphone (Dialaudid) tablets, but also for buprenorphine (with and without naloxone) and 25, 50, 75, and 100 mcg/hour fentanyl patches.
Trends indicate that for more than a decade, opioid-related harms, including OUD and unintentional overdose, have been growing problems across the country (Calcaterra et al., 2013; Paulozzi, 2012) and the world (EMCDDA, 2015a,b), and are now negating indicators of public health advances and altering both life expectancy (Olshansky et al., 2012) and the very demography of the American populace. The implications of these extensive illicit markets for the evaluation of post-marketing or other policy interventions for prescription opioids, given the current paucity of surveillance capacities (discussed in the section on surveillance below), cannot be overstated.
Summary and Recommendation
Several distinct, well-established markets for opioids exist with overlapping demand in the United States that are likely to persist for the foreseeable future. The products they supply include opioids prescribed, dispensed, and used by patients as medically intended; those prepared as a prescription but not used as intended, including opioids dispensed and misused, as well as those that are diverted before being dispensed (i.e., diverted from lawful channels of commercial distribution, such as wholesalers and pharmacies); and those supplied by drug trafficking organizations, mostly from international sources. Conditions appear ripe for fentanyl and counterfeit prescription pills to continue to spread, with potential effects not only on heroin and other illicit drug markets but also on markets for diverted prescription drugs. These markets are both well established and likely to persist for the foreseeable future. The committee recommends that, in designing and implementing policies and programs pertaining to prescribing of, access to, and use of prescription opioids, the U.S. Food and Drug Administration, other agencies within the U.S. Department of Health and Human Services, state agencies, and other stakeholders consider the potential effects of these interventions on illicit markets—including both the diversion of prescription opioids from lawful sources and the effect of increased demand for illegal opioids such as heroin among users of prescription opioids—and take appropriate steps to mitigate those effects (Recommendation 4-1).
THE CURRENT STATE OF SURVEILLANCE SYSTEMS
Since the IOM report Relieving Pain in America (IOM, 2011) was issued, a remarkable loss of publicly available data sources on drug-related trends has occurred. Four major publicly funded data sources (discussed later in this section) were phased out during this period, and only one has been replaced with a new system; still others remain in validation stages for redesign. In the void created by the defunding of these data sources, proprietary and specialized post-marketing surveillance systems have gained immense importance. The Researched Abuse, Diversion and Addiction-Related Surveillance (RADARS®) System and the National Addictions Vigilance Intervention and Prevention Program (NAVIPPRO) are two such multimodal data systems. They provide product-level real-time post-marketing surveillance at cost to the pharmaceutical industry, which then uses these data to respond to the FDA REMS and other FDA-related post-marketing reports and inquiries.
RADARS originated as part of Purdue Pharma’s risk management activities and was subsequently incorporated into the Rocky Mountain Poison and Drug Center, a division of the Denver Health and Hospital Authority. Its real-time, product-specific data collection includes a survey of key informants across the country, a survey of methadone treatment program attendees, analysis of news and social media mentions, drug diversion investigator surveys, a college student survey, street price analysis, and poison control reports. NAVIPPRO operates a similar system, with real-time data collection via a version of the well-known Addiction Severity Index (ASI), amended to collect product-level information about misuse, route of administration, and drug source. NAVIPPRO is a proprietary dataset owned by Inflexxion, Inc., which created the system through a series of Small Business Innovation Research (SBIR) grants from the National Institute on Drug Abuse (NIDA). NAVIPPRO includes data collected from a national sample of both adults and young adults attending substance use treatment centers. The data are compiled for analysis together with poison control data and text-based analysis of drug-related online message boards and chatter from drug-use discussion forums. Although both systems have published extensively on their creation, validation, and product-level analyses and are used by pharmaceutical companies, they have not been widely used by public health practitioners and researchers. Sources that report drug-related data are catalogued in Appendix C; those no longer operating since 2011 to date are discussed immediately below.
The Drug Abuse Warning Network (DAWN) was a public health surveillance system created in 1972 that monitored drug-related hospital emergency department visits (DAWN-ED) in order to report on the impact of drug use in metropolitan areas and nationally. While DAWN was never
designed to be nationally representative, the system generated estimates at the metropolitan area level and was later used to produce nationwide estimates. In addition, the system was expanded to encompass drug-related deaths investigated by medical examiners or coroners (DAWN-ME) in a selected sample of metropolitan areas. After 2003, DAWN included a real-time data access portal called DAWN Live. The site facilitated quicker access to data for participating sites and public health organizations, with clear indicators of reporting completeness and attendant caveats.
The agent (i.e., product and compound)-level specificity of the data reported in DAWN meant that the pharmaceutical industry and the public had access to product-level information and could compare product impacts, including morbidity and mortality trends, interactively. DAWN was initially overseen by the DEA, then NIDA, and finally SAMHSA, but both DAWN-ED and DAWN-ME were discontinued in 2011 (SAMHSA, 2016b). Thus, this resource was unavailable as the opioid epidemic unfolded. In retrospect, the product-level detail in DAWN could have informed decision makers across institutions of the nature and challenge of the prescription opioid and illicit drug crises.
In researching the reasons for the defunding of DAWN, the committee learned of several factors, including frustrations with the sampling frame, incompleteness of data, concerns among industry about the product-level data, cost, and the lack of representation of small-town and suburban communities. In the absence of DAWN, it has become more difficult to track drug-related emergency department visits (Rowe et al., 2016). SAMHSA’s new Emergency Department Surveillance System (SEDSS) is intended to serve as the new source of data on drug-related emergency department visits, and will combine aspects of DAWN with the National Center on Health Statistics’ (NCHS’s) National Hospital Care Survey. The timeliness of reporting, geographic specificity, and product-level details of the new system are unknown.
In 2014, two additional key data sources were phased out. First, funding for the Arrestee Drug Abuse Monitoring survey (ADAM II), which had been funded since 2007 by ONDCP, was cut for budgetary reasons (before 2007, an earlier version of the system had been housed in the National Institute of Justice) (Kilmer and Caulkins, 2014; NIJ, 2014). ADAM collected self-reported data and biological samples from arrestees admitted to booking facilities, inquiring about drug use trends and street prices and examining their urinalysis results. The value of the ADAM data was evident in information on trends of illicit drugs other than marijuana, which generated strikingly different estimates from those extrapolated from the NSDUH (Caulkins, 2015a; Kilmer et al., 2014). These data were useful for policy makers, law enforcement, and treatment resource planners. To date, this data source has not been replaced or reinvigorated.
Also phased out was NIDA’s Community Epidemiology Work Group (CEWG), a network of local experts in drug-related topics, which had met semiannually to report on drug trends and emerging issues in sentinel sites from 1976 to 2014. The CEWG experts created metrics and indicators of drug use trends, collaborated on annual reports, and conducted field research on emerging trends. The CEWG was replaced by the National Drug Early Warning System (NDEWS) (NIDA, 2015), which coordinates a listserv, hosts webinars, tracks online media mentions of various drug-related terms and trends, and convenes a virtual network of sentinel sites that conduct local area data collection as requested. Only 3 years into its existence, the NDEWS is not equal to its predecessor in terms of representation, participation, and reach; however, its role and purpose continue to evolve, providing a crucial platform for questions and discussion related to drug use trends for its online and invited membership.
Notably, few of the public and proprietary datasets that have collected self-reported data from people who use drugs have asked respondents about their overdose history. Those that have inquired about overdoses have tended to employ wording that conflates unintentional and intentional (i.e., suicide attempt) overdose or failed to specify or ask separately about overdose on opioids (heroin, pain medication, or MAT medications). More recent efforts to better apply and report emergency department International Classification of Diseases (ICD) E-codes in order to standardize and improve the reporting of hospital-treated overdoses are laudable, but will underestimate the true rate of nonfatal overdose in a community. Capturing the many nonfatal overdose experiences in which the person is not transported to the hospital requires a valid and reliable direct inquiry encompassing all people who use these drugs.
It has been said that one cannot see what one does not count. The absence of agent-specific, real-time, drug-related data has contributed to the severity of the current opioid crisis. The timing of these data losses exacerbated the inability to detect changes in misuse and mortality driven by prescription opioids, and it continues to hinder the nation’s capacity to track illicit drug trends and their public health consequences. Cost-effective and nimble data collection systems may be reliable and even timely, but need to be examined rigorously for validity. More critically, the pervasiveness and lethality of illicit synthetic drugs heighten the need to capture agent-level information and concurrent and subsequent drug-using behaviors.
As discussed in Chapters 2 and 3, gaps exist in the reporting of data that can be used to accurately describe the epidemiology of pain and OUD in the United States, including how these conditions relate to one another and how often they co-occur. This chapter has reviewed the interrelated nature of the prescription and illicit opioid epidemics and the limitations of current salient surveillance systems. Closing these data gaps would improve
understanding of pain, OUD, and overlapping illicit use, and enable more effective and measurable policy interventions. The committee recommends that the Substance Abuse and Mental Health Services Administration, the U.S. Food and Drug Administration, the National Institutes of Health, and the U.S. Centers for Disease Control and Prevention collaborate to identify best practices and reporting formats that portray the epidemiology of both pain and opioid use disorder accurately, objectively, and in relation to one another (Recommendation 4-2).
The committee recommends that the National Institute on Drug Abuse and the U.S. Centers for Disease Control and Prevention invest in data collection and research relating to population-level opioid use patterns and consequences, especially nonmedical use of prescription opioids and use of illicit opioids, such as heroin and illicitly manufactured fentanyl (Recommendation 4-3). The research proposed in Recommendation 4-3 could include transitions to and cessation of use of heroin and fentanyl; motivations for use; social determinants underpinning misuse and illicit use; and differences arising by sex, gender, race, and ethnicity.
RECENT DEVELOPMENTS IN PHARMACEUTICAL TREATMENT OF OPIOID USE DISORDER
This section highlights the use of pharmacotherapies in the treatment of OUD, with an emphasis on new research and treatment approaches that have emerged since the 2011 IOM report was issued. A review of current trends in access to, utilization of, and outcomes of treatment services is presented in Chapter 5.
The Centrality of Pharmacotherapies in Treatment of Opioid Use Disorder
Medications are central to the treatment of OUD. The three medications approved by the FDA for treatment of OUD are methadone, buprenorphine, and naltrexone (see Table 4-1). There continues to be some debate in the field regarding whether, and under what circumstances, use of these medications should be regarded as necessary or sufficient, a debate that is reflected in the terms used to refer to treatment with these medications. For example, recovery community advocates encourage the use of the term “medication-assisted recovery” to describe the combination of pharmacotherapy and counseling and/or recovery work that they believe patients should undergo. They argue that remission of SUD achieved through use of medication alone is not genuine because without counseling, the person may not have achieved the interpersonal and spiritual changes deemed necessary for lasting recovery. The assumption is that only by participating
TABLE 4-1 Characteristics of Medications for the Treatment of Opioid Use Disorder
| Characteristic | Methadone | Buprenorphine | Naltrexone |
|---|---|---|---|
| Selected Brands | Dolophine, Methadose | Subutex,* Suboxone, Zubsolv | Depade, Revia, Vivitrol |
| Class | Agonist (fully activates opioid receptors) | Partial agonist (activates opioid receptors but produces a diminished response even with full occupancy) | Antagonist (blocks the opioid receptors and interferes with the rewarding and analgesic effects of opioids) |
| Use and Effects | Taken once per day orally to reduce opioid cravings and withdrawal symptoms | Taken orally or sublingually (usually once per day) to relieve opioid cravings and withdrawal symptoms | Taken daily orally or monthly by injection to diminish the reinforcing effects of opioids (potentially extinguishing the association between conditioned stimuli and opioid use) |
| Advantages | High strength and efficacy as long as oral dosing (which slows brain uptake and reduces euphoria) is adhered to; excellent option for patients who have no response to other medications | Eligible to be prescribed by certified physicians, nurse practitioners, and physician assistants, which eliminates the need to visit specialized treatment clinics and thus widens availability; lower risk of overdose | Not addictive or sedating and does not result in physical dependence; a recently approved depot injection formulation, Vivitrol, eliminates the need for daily dosing |
| Disadvantages | Mostly available through approved outpatient treatment programs, which patients must visit daily; respiratory depression; abuse liability | Subutex* has measurable abuse liability; Suboxone diminishes this risk by including naloxone, an antagonist that induces withdrawal if the drug is injected; for Subutex and Suboxone, withdrawal in patients dependent on methadone or short-acting prescription opioids | Poor patient compliance with the oral form (but Vivitrol should improve compliance); initiation requires attaining prolonged (e.g., 7-day) abstinence, during which withdrawal, relapse, and early dropout may occur; overdose fatality due to self-discontinuation and hypersensitized µ opioid receptors |
*Subutex (a single-agent buprenorphine product) is no longer on the market in the United States. However, multiple other generic single-agent buprenorphine products are available.
SOURCE: Adapted from Volkow et al., 2014.
in regular counseling and adjunctive treatment services can people attain signficant recovery achievements. As an alternative, WHO uses the term “psychosocially assisted” pharmacotherapy, to capture the central role of medications in the treatment of OUD (WHO, 2009). It is both critical and convenient for the purposes of this report that the most effective approaches for treating OUD are those within the purview of the FDA.
The committee has chosen to use the acronym MAT to refer to the use of pharmacotherapies in treatment of OUD. As explained in Box 1-2 in Chapter 1, MAT may be defined to refer either to “medically assisted treatment” (use of medications in combination with counseling and behavior therapies to treat OUD) or to “medication for addiction treatment” (implying that medication may be used alone, but need not be). The committee has chosen to use MAT to embrace this ambiguity instead of opting for one definition or the other. For purposes of this report, the only material scientific conclusion is that medications should play a central (if not exclusive) role in treatment of OUD, a view strongly supported by the scientific literature.
A 2009 Cochrane systematic review found that opioid agonist treatment without counseling is more effective than being waitlisted for treatment or receiving psychosocial treatment with or without placebo (Mattick et al., 2009). These findings were affirmed by recent results from the NIDA Clinical Trials Network’s Prescription Opioid Addiction Treatment Study (POATS), in which a randomized controlled trial examined buprenorphine-naloxone treatment of varying durations and counseling of varying intensities among patients dependent on prescription opioids. It was found that patients receiving individual counseling for OUD in conjunction with the medication (weekly 45- to 60-minute sessions with a trained mental health or substance abuse professional) showed no additional benefit over those receiving standard medical management (15- to 20-minute visits with a physician certified to prescribe the medication) (Weiss et al., 2011). Similarly, a study of more intensive counseling in the setting of office-based buprenorphine prescribing compared with medication only showed no superior patient outcomes (Fiellin et al., 2006). On the other hand, one study in a veteran population showed superior outcomes for patients receiving methadone coupled with counseling compared with medication-only treatment (McLellan et al., 1993).
The central importance of medication treatment is further affirmed for patients with prescription OUD in a recent evidence synopsis by Nielsen and colleagues (2017, p. 967), who found that “long-term maintenance of opioid agonists is associated with less prescription opioid use and better adherence to medication and psychological therapies for opioid dependence compared with opioid taper or psychological treatments alone.” In addition, no differences in efficacy were observed between methadone and
buprenorphine maintenance therapies (Nielsen et al., 2017). While the studies across this literature were not exhaustive in the psychological therapies tested, and therefore should not be construed as suggesting that all such approaches are ineffective, the data consistently indicate clinical utility and improvements in quality of life for people with OUD who receive medication treatment.
Instistence on provision of counseling is an important factor in access to buprenorphine. According to state regulations and accrediting standards, opioid treatment programs are required to provide a minimum of counseling services each month. Yet the literature shows that counseling may help engage people in their recovery, but may not be necessary or effective beyond the provider–patient clinical sessions. The inability to provide the recommended OUD treatment services alongside prescription buprenorphine does not indicate inferior treatment, and withholding prescription buprenorphine from a patient with OUD if these services are unavailable, as may be the case as a result of insurance companies’ prior authorization requirements for buprenorphine, may be lethal.
Data from studies of methadone treatment programs provide a compelling rationale for medication-only treatment when this is the only available option. Schwartz and colleagues (2012), for example, compared mortality rates among patients with OUD treated with methadone in a treatment program providing counseling services with similar patients on a waitlist for the program treated only with medication (i.e., interim dosing) and with waitlisted patients not receiving interim dosing. Mortality rates were comparably reduced for patients receiving MAT with or without supportive counseling, but were significantly higher among patients who received no medication (Schwartz et al., 2012). In a randomized trial, patients receiving MAT without counseling also showed lower HIV risk behaviors, suggesting that this approach could reduce the risk of bloodborne virus transmission (Wilson et al., 2010). A recent systematic review of interim methadone dosing studies concluded that this approach helped bridge gaps due to treatment shortages, improved patient outcomes, and warranted expansion to assess generalizability (Sigmon, 2015). And in a small randomized pilot study, participants assigned to interim dosing with buprenorphine combined with technology-assisted components to support adherence showed a statistically significant reduction in the use of illicit opioids and intravenous drugs compared with waitlist controls, indicating that interim therapy may be suitable when treatment options are limited. The authors note that additional studies with larger samples and longer follow-up periods are needed (Sigmon et al., 2016).
Notably, other countries that provide pharmacotherapies to treat patients with OUD do not impose counseling and psychotherapy as a requirement for receipt of treatment; indeed, the provision of medication
in combination with counseling is not common. In the United Kingdom, for example, pharmacotherapies are dispensed daily or less frequently to patients through community pharmacies, and patients with OUD are managed by general practitioner–assisted teams of SUD treatment specialists (NICE, 2007). Counseling and psychological therapies may be used, but are not a condition or expectation for receipt of medication.
The literature is consistent in finding that the longer a person with OUD is treated and maintained on medication for the disorder, the better are their health outcomes. This consistent finding argues against the application of a tapering approach, a detoxification model, and the expectation that short-term courses of therapy can treat OUD effectively. It further supports a long-term, maintenance model of provision of pharmacotherapy and the need for a more diverse product environment for FDA-approved medications for treatment of OUD. In fact, short-term treatment for OUD, especially in the case of abstinence-based treatment, but also with medications, is associated with increased mortality risk (Woody et al., 2008).
The following subsections briefly describe the medications available for treatment of OUD, whose characteristics are summarized in Table 4-1.
Methadone
Response to methadone appears to be dose related. Mean response at 1 year is approximately 60 percent, but differs based on a host of patient factors and adherence to evidence-based dosing practices (Bart, 2012). Methadone is a full opioid agonist that was invented in Germany in the late 1930s for use during World War II as a cheaper and easier-to-manufacture analgesic alternative to the opioids available at the time (Strang and Tober, 2003). It was approved for use in the United States shortly after the end of the war and started being used to treat opioid withdrawal within 1 year (Isbell et al., 1947). A few decades later, in the 1960s, it began to be investigated for maintenance therapy for OUD (Dole and Nyswander, 1965). For reasons that may have to do with its antagonism at the NMDA (N-methyl-d-aspartate) receptor, tolerance does not increase for methadone the way it often does for other opioids (Davis and Inturrisi, 1999). This feature, along with its low cost, makes methadone an ideal medication for long-term maintenance therapy for OUD.
In the 50 years since first being used to treat OUD, methadone has been the subject of hundreds of studies evaluating its efficacy and safety. Several large-scale studies in the 1970s and 1980s showed that 25–45 percent of people with OUD who were treated with methadone remained drug-free after 1 year (Hubbard and Marsden, 1986; IOM, 1995; Sells et al., 1979). Modern reviews confirm these findings, and observe further that retention
in treatment is greater for people on methadone than for those in treatment who are not receiving pharmacotherapy (Mattick et al., 2009).
Methadone’s safety also has been well established, having been documented extensively for at least 40 years (Kreek, 1973). While methadone can, like all opioids, lead to respiratory depression, most cases of overdose involving methadone stem not from its use to treat OUD but its less tightly regulated use as a pain medication (SAMHSA, 2007). Among patients with OUD, it has been shown that more intensive monitoring of medication dosing is associated with decreased mortality (Bart, 2012; Strang et al., 2010).
Buprenorphine
Buprenorphine was the first opioid medication to become available in the United States since 1914 that could be used for OUD maintenance treatment in primary care settings. FDA approval of buprenorphine came in 2002. Since that time, several forms of buprenorphine have been approved, as a single entity or formulated in combination with naloxone to protect against tampering (see Box 4-1), in pill form and as sublingual film, and in varying flavors. A systematic review of 16 randomized controlled trials on the efficacy of buprenorphine found that it is associated with improved outcomes compared with placebo for individuals and pregnant women with OUD (Thomas et al., 2014).
The Drug Addiction Treatment Act of 2000 (DATA 2000) broadened the types of clinical settings where MAT for OUD could be provided. In the two decades prior to its passage, only opioid treatment programs could dispense Schedule III–V medications used to treat OUD. DATA 2000 specified that qualified providers are permitted to dispense or prescribe specifically approved Schedule III, IV, and V narcotic medications (medications with a lower risk for misuse, such as buprenorphine) in settings other than an opioid treatment program (SAMHSA, 2017b).
While expanding the types of health professionals and the places where people with OUD could find treatment, DATA 2000 also specified a cap on the number of patients per prescriber who could be treated, as well as the requirements of providers who opted to provide office-based treatment. Providers must apply to SAMHSA to provide buprenorphine treatment beyond a 30-patient limit for up to 100 patients with OUD (SAMHSA, 2017a). In 2016, two changes aimed at improving access to buprenorphine treatment were announced. First, providers who have prescribed buprenorphine to 100 patients for at least 1 year can apply to increase their patient limit to 275 (SAMHSA, 2017a). Second, the 2016 Comprehensive Addiction and Recovery Act extended buprenorphine prescribing privileges to physician assistants and nurse practitioners for 5 years (until October
of 2021) (ASAM, 2017), with rigorous training requirements in place to ensure consistent and careful prescribing.
Importantly, DATA 2000 did not require prescribers with a waiver to prescribe buprenorphine for OUD to provide other treatment services (i.e., counseling, group therapy) as well. Rather, the act states only that it is recommended that such services be provided or coordinated. While many providers prescribing buprenorphine are SUD specialists, and many oth-
ers recognize the importance of ensuring coordination of SUD treatment services, many do no more than prescribe medication. As discussed earlier, many believe that optimal care for OUD involves providing medication accompanied by supportive counseling and other treatment services.
Since buprenorphine may be dispensed within an office-based practice and methadone can be dispensed only within an opioid treatment facility, buprenorphine has the potential to provide better access to treatment. Many areas of the country have limited numbers of opioid treatment facilities or facilities that lack the capacity to meet demand (see Figure 4-9). Additionally, although methadone regulations require that opioid treatment facilities give priority to pregnant women, facilities are not always compliant. Preference for an office-based system of care also often makes buprenorphine preferable since the requirements for onsite dosing differ significantly from those for an opioid treatment facility. However, the delivery system for buprenorphine functions well below capacity. A recent study found that the majority of physicians with waivers to prescribe buprenorphine were doing so well below the limits allowed by law, with fewer than 10 percent prescribing to at least 75 patients (Stein et al., 2016). How this gap impacts special populations such as pregnant women is unknown, but anecdotally,
SOURCE: SAMHSA, 2016b.
many pregnant women report they were discharged from care for OUD upon becoming pregnant, and many prescribers report being unwilling to provide care to pregnant women. It is possible that for many pregnant women with OUD, the context and structural challenges of receiving MAT contribute substantially to the severity of NAS. (See the section below on pharmacotherapies for treatment of pregnant women with OUD.)
In addition, significant disparities in the use of buprenorphine have been documented. A recent review of the literature found that buprenorphine patients are largely white, are employed full time, are seeking treatment for heroin or prescription OUD, are treated in private physician practices, and pay out of pocket or are privately insured (Duncan et al., 2015). Furthermore, a study in New York City found neighborhood-level disparities, with the highest buprenorphine prescription rates being in high-income residential areas with low percentages of African American and Hispanic residents (Hansen et al., 2016). The authors note that these disparities may be attributable to buprenorphine marketing to the private sector (primary care physicians represent 65 percent of buprenorphine maintenance providers) and perceptions that this form of treatment is most appropriate for employed patients. Despite increased numbers of buprenorphine providers, moreover, 43 percent of U.S. counties had no buprenorphine-waivered physicians as of 2011 (Stein et al., 2015). The authors argue that because of buprenorphine’s greater effectiveness relative to methadone in the treatment of OUD; its suitability for varying therapeutic settings, including public health care systems; and its additional advantages (e.g., less required oversight, potential to reduce stigma, increase in treatment of comorbid health and psychiatric conditions), its accessibility in such settings should be promoted as a first line of treatment.
Naltrexone
Naltrexone is a µ opioid receptor antagonist, and when formulated as naltrexone ER has been shown to be safe and effective in treating OUD. Accordingly, the FDA approved the naltrexone ER product in 2011. The evidence for oral naltrexone’s effects on craving in OUD is less clear than that for its effects on craving in alcohol addiction (Bart, 2012), and the oral formulation is not recommended or widely used for treating OUD. However, the long-acting form of naltrexone, which is implanted under the skin, is more effective than a daily pill because it eliminates problems with adherence (Comer et al., 2006; Krupitsky and Blokhina, 2010; Krupitsky et al., 2012). Patients using long-acting naltrexone are three times more likely than those using oral naltrexone to remain relapse-free after 6 months (Krupitsky et al., 2012).
Some have questioned the findings of pivotal efficacy studies of naltrexone and raised additional safety concerns about naltrexone ER related to overdose (Wolfe et al., 2011). A meta-analysis of cost and utilization outcomes between naltrexone ER and other pharmacotherapies for treatment of OUD found that patients with OUD taking naltrexone ER had lower inpatient substance misuse–related utilization relative to those taking other agents, and had $8,170 lower total costs relative to those taking methadone (Hartung et al., 2014). With respect to clinical outcomes, however, it is unclear whether naltrexone ER is as effective as methadone and buprenorphine in reducing the risk of fatal overdose and other drug-related health and quality-of-life outcomes. Lee and colleagues’ (2015) study of outcomes in jail-initiated naltrexone ER found reductions in opioid use and increased abstinence, while findings on secondary outcomes suggested lower risk of overdose compared with controls. Another trial examined naltrexone ER compared with treatment as usual for the prevention of opioid relapse among individuals in the criminal justice system. No overdoses occurred in the naltrexone group compared with seven in the usual treatment group. Individuals assigned to naltrexone ER also had significantly lower rates of relapse than those in the usual treatment group (43 percent versus 64 percent) (Lee et al., 2015). While promising, these findings have not been replicated in other populations and settings.
Other Alternatives
In Europe, Canada, and Australia, other opioids have been used successfully for opioid maintenance treatment to reduce the risks of injection of illicit opioids. For example, several trials using slow-release morphine (Ferri et al., 2013), heroin (Ferri et al., 2011), and hydromorphone (Oviedo-Joekes et al., 2016) for patients who had not done well with methadone showed positive outcomes (Strang et al., 2015).
Prescription of heroin also is integrated into the treatment systems of several European countries (Uchtenhagen, 2010). Supervised injectable heroin (SIH, or diamorphine) may be an effective treatment for heroin dependence refractory to standard treatment, although it is less safe than methadone maintenance treatment and therefore requires more clinical attention to manage safety issues (Strang et al., 2015). A systematic review and meta-analysis identified six randomized clinical trials of SIH and concluded that among patients with OUD involving heroin, those receiving SIH compared with control groups (most often receiving methadone maintenance treatment) demonstrated better outcomes with respect to greater reduction in use of illicit heroin (Strang et al., 2015).
Pharmacotherapies for Treatment of Women with Opioid Use Disorder Who Are Pregnant
The use of MAT for the treatment of women with OUD who are pregnant has a long history, beginning with the implementation of methadone pharmacotherapy in the late 1960s. Initially, the FDA mandated methadone-assisted withdrawal for pregnant women, but it quickly reversed this decision following the occurrence of adverse pregnancy events (Blinick et al., 1969; Jones et al., 1999). Currently, questions often arise about exposure of the fetus to the medication as the newborn may experience withdrawal that requires treatment, and there have been calls recently for pregnant women wth OUD to be withdrawn from all opioids, including treatment medications. However, the risk of withdrawal is deemed much less important than the benefits of treatment. The 1993 and 2004 SAMHSA Treatment Improvement Protocols for OUD, the 1997 National Institutes of Health Consensus Panel on Effective Medical Treatment of Opioid Addiction, the 2012 American College of Obstetricians and Gynecologists and American Society of Addiction Medicine Joint Opinion, the WHO 2014 Guidelines for the Identification and Management of Substance Use and Substance Use Disorders in Pregnancy, and the 2016 SAMHSA Collaborative Approach to the Treatment of Pregnant Women with Opioid Use Disorders all recommend MAT for pregnant women as the standard of care. The underlying principle behind the use of MAT during pregnancy is that it prevents erratic maternal opioid levels and protects the fetus from repeated episodes of withdrawal. In addition, it ensures that the woman is engaged in the health care system and promotes prenatal care, which results in healthier outcomes for both mother and infant (Kaltenbach et al., 1998).
The emergence of the implementation of methadone pharmacotherapy for pregnant women with OUD coincided closely with the creation of NIDA. One of NIDA’s first endeavors was to fund a number of research demonstration projects in 1974 implementing treatment programs for pregnant women with OUD. This research provided the foundation for the model of care that emerged in the 1980s. Another major contributor to the development of treatment for this population was the funding source created by SAMHSA’s Pregnant and Postpartum Women’s project, initiated in the early 1990s, which is still part of the agency’s portfolio.
The treatment options that exist today are an extension of the original model, which began with the premise that services for pregnant women must be comprehensive, to include not only treatment of OUD but also obstetrical, medical, and psychiatric care. Research has shown that women with SUD, including OUD, have a complex array of biopsychosocial problems that must be addressed if treatment is to be successful and recovery sustained (Comfort and Kaltenbach, 1999).
The framework for treatment is grounded in the premise that the treatment should be woman-centered (i.e., responsive to the specific needs of the individual); trauma-informed (i.e., recognizing the role of trauma and violence in the lives of women); strengths-based (i.e., focusing on strengths rather than deficits); and culturally competent (i.e., acknowledging the role of culture, ethnicity, race, racism, and sexual orientation) (SAMHSA, 2009). The treatment approach should be multidisciplinary and include pharmacotherapy with methadone, buprenorphine, or buprenorphine-naloxone. Initiation of naltrexone currently is not recommended in pregnant women.
At present, the field is waiting for existing recommendations to reflect new data. The current recommendation that the combination product buprenorphine-naloxone not be used was published in 2004 and was based on a lack of data on infant exposure to naloxone. And although there have been no salient randomized controlled trial data to date, several studies have shown no difference in infant outcomes between the single-entity and combination products, with the latter being used by many providers (Debelak et al., 2013; Lund et al., 2013; Wiegand et al., 2015). In addition to pharmacotherapy supports, a multidisciplinary approach would involve not only obstetrical, medical, and psychiatric services but also individual and family therapy, trauma services, case management, parent–child services, and liaison relationships with the department of human services. Treatment modalities encompass traditional levels of care, including outpatient, intensive outpatient, and women and children residential care.
Although the efficacy of comprehensive treatment for pregnant women with OUD has been well established, the number of programs available to provide such services is extremely limited. Nationally, there exist only 20 residential treatment programs for pregnant and parenting women funded under the SAMHSA portfolio, and of those, only three provide treatment specific to OUD. Among the 1,450 opioid treatment programs (see Figure 4-9), it is estimated that no more than 12 programs provide specialized treatment for pregnant women. Moreover, treatment for pregnant women often is fragmented and may be impeded when collaboration is lacking among the opioid treatment facility, obstetrician, pediatrician, and hospital.
In light of these limited services, newer models of collaboration among multiple systems of care have emerged within the past few years to provide comprehensive care to pregnant women with OUD. Excellent examples of collaboration among the state, medical providers, and treatment providers are the Vermont Children and Recovering Mothers (CHARM) Collaborative and the Ohio Maternal Opiate Medical Support (MOMS) project. CHARM involves 10 organizations, including hospitals, treatment providers, state agencies, maternal and child health programs, and the visiting nurse association aimed at providing comprehensive care coordination for
pregnant women with OUD.9 The MOMS project, funded by the state of Ohio, employs a maternity care home (MCH) model in four sites across the state. Each site is unique, but all utilize the MCH team-based care delivery model, which emphasizes coordination of community services and treatment for OUD, including pharmacotherapy, case management, and prenatal care.10 Additionally, a new model based on Project ECHO (Extension for Community Healthcare Outcomes)11 is currently being examined to provide support and improve care in treatment programs for pregnant and postpartum women with SUD. Project ECHO is based on an approach in which telemonitoring utilizes case-based learning to focus on best practices. The ECHO model is based on a hub-and-spoke knowledge-sharing network led by a team of “experts” using video conferencing to conduct virtual clinics with community providers.
Other treatment matters to be addressed for this vulnerable population are centered on the medications used. Since the FDA approved buprenorphine in 2002, there have been two medications to use in treating pregnant women with OUD. The two have different benefits and disadvantages, but the basic tenets of treatment are the same.
The efficacy criterion for the choice of medication for pregnant women with OUD (i.e., methadone or buprenorphine) has not yet been established. However, data from a multisite randomized controlled trial that compared maternal and infant outcomes among women maintained on methadone with those of women maintained on buprenorphine often are cited as a determining factor. The study found that, although there was no difference in the number of infants that required treatment for NAS, infants exposed prenatally to buprenorphine required 89 percent less morphine to treat NAS, spent 58 percent less time in the hospital being medicated for NAS, and spent 43 percent less time in the hospital overall relative to infants exposed prenatally to methadone (Jones et al., 2010).
A systematic review and meta-analysis of 12 studies, including the above-cited randomized controlled trial, found that infants explosed prenatally to buprenorphine had better outcomes than methadone-exposed infants with respect to treatment duration, morphine dose, birth weight, length, and head circumference (Brogley et al., 2014). These findings have led some practitioners to recommend always that buprenorphine rather than methadone be used for pregnant women. Ideally, however, treatment
___________________
9 See SAMHSA’s Collaborative Approach to the Treatment of Pregnant Women with Opioid Use Disorders (SAMHSA, 2016c) for a detailed description.
10 See www.momsohio.org for further information.
11 Project ECHO, developed at the University of New Mexico to address hepatitis C, is now used throughout the United States and other countries to address 40 different subject areas. There are 14 institutions in the United States conducting pain management ECHOs. See http://echo.unm.edu for more information.
will be based on what is best for both the mother and child; each woman’s medical, psychological, and substance use history must be considered in any treatment decision. As a partial agonist, for example, buprenorphine may not be as effective as methadone for certain women. Without data to guide decisions, however, the current recommendation is that women with OUD who are naïve to agonist treatment may be good candidates for buprenorphine. If women do not respond to buprenorphine, transfer to methadone can easily be initiated. In any case, it is recommended that women successfully stabilized on methadone or buprenorphine who become pregnant remain on their current medication (Jones et al., 2012). And the 2012 Joint Opinion of the American College of Obstetricians and Gynecologists and American Society of Addiction Medicine recommends the use of either methadone or buprenorphine.
Although withdrawal often is cited as a way to reduce NAS, there is no evidence based on an intention-to-treat analysis that withdrawal without medication is beneficial to the mother, fetus, or infant. In addition, limited data suggest that infant treatment outcomes with buprenorphine may be similar to those of withdrawal. A long history of concern regarding withdrawal during pregnancy also merits consideration. Adverse fetal events that occurred in the 1970s as a result of withdrawing pregnant women from methadone led to recommendations that withdrawal be initiated only in the second trimester because of safety concerns, such as fetal demise in the first trimester of pregnancy and prematurity in the third trimester. In the 1990s, however, research indicated that with appropriate fetal monitoring, women could be withdrawn safely at anytime during pregnancy (Jarvis and Schnoll, 1994). Yet the question is not whether withdrawal can be done safely but whether it should be done at all. A summary of the recent literature on medication-assisted withdrawal during pregnancy indicates that it can be safe and may be associated with less NAS and improved birth weights. When given a choice, however, approximately 50 percent of women choose medication treatment rather than withdrawal, and among those who are undergoing withdrawal, the risk of relapse is high (Bell et al., 2016; Dashe et al., 1988; Jones et al., 2008; Lund et al., 2012; Stewart et al., 2013). A recent commentary by Jones and colleagues (2017) speaks to the lack of evidence supporting a clear benefit of medication-assisted withdrawal for the maternal–infant dyad, as it increases the risk of poor treatment engagement and relapse for the mother and does not improve the health of or significantly reduce the occurance of NAS in the infant. The WHO 2014 Guidelines for the Identification and Management of Substance Use and Substance Use Disorders in Pregnancy, the 2012 American College of Obstetricians and Gynecologists and American Society of Addiction Medicine Opinion No. 524: Opioid Abuse, Dependence, and Addiction in Pregnancy, and SAMHSA’s 2016 A Collaborative Approach to the Treatment
of Pregnant Women with Opioid Use Disorders all recommend treatment rather than withdrawal because of the high rate of relapse that places the fetus at additional risk.
Access to care for pregnant women with OUD also is driven by state policies. Office-based provision of buprenorphine is covered by Medicaid in all states and the District of Columbia, but provision of methadone is covered only in 31 states and the District of Columbia. A recent study by Angelotta and colleagues (2016) found that fewer than 50 percent of pregnant women with OUD received MAT. The most important factors associated with lack of MAT were referral source, geographic location, Medicaid funding for methadone, and state laws permitting child abuse charges for illicit drug use in pregancy. Pregnant women referred to treatment by the criminal justice system were the least likely to receive MAT, especially in states with prenatal child abuse laws (Angelotta et al., 2016). As might be expected, lack of Medicaid coverge also was a factor, but there was a high correlation as well between lack of Medicaid funding for methadone and state prenatal child abuse laws. Absent better coordination between medical standards of care and public policy at both the national and state levels, the provision of effective treatment for this at-risk population will continue to be fragmented at best.
Summary
Three underutilized, efficacious medications are available for the treatment of OUD. Few new products for treatment of OUD have entered the market, although several new modes of medication delivery have emerged. Even for special populations such as pregnant and postpartum women, medication therapy is the standard of care. Expected side effects of opioid exposure in utero, such as NAS, can be treated and symptoms abated with no current evidence of long-term effects.
TRENDS IN TREATMENT OF OPIOID OVERDOSE WITH NALOXONE
The term “overdose” is used to describe the poisoning event that occurs when opioid exposure results in respiratory depression, morbidity, or mortality. The onset of respiratory depression caused by exposure to opioids may progress to severe, life-threatening symptoms within a matter of minutes to hours depending on a number of factors, including the drug involved (e.g., rapid-onset medications such as fentanyl), the presence of other drugs in the individual’s system, the route of administration (i.e., injection hastens delivery of opioids to the bloodstream and speeds crossing of the blood–brain barrier, bringing on respiratory depression, among
other physiological reactions), and the individual’s health condition (e.g., a respiratory condition or metabolic distrubance can worsen symptoms more rapidly) (EMCDDA, 2016). Therefore, although a single large dose can cause severe respiratory depression and death, overdoses occur at varying opioid doses in individuals with compromised breathing, metabolic conditions, or altered opioid tolerance (Sporer, 1999), and even at therapeutic levels when used in combination with other central nervous system depressants such as benzodiazepines (as reviewed above) or alcohol.
Use of Naloxone to Treat Overdose
Naloxone, a synthetic N-allyl derivative of oxymorphone and an opioid antagonist, was first synthesized in 1961 by Jack Fishman and investigated by Harold Blumberg. The discovery was the first of its kind, an antagonist with the ability to avoid agonistic activity through prevention or elimination of agonistic narcotic binding. Also related to its antagonistic activity, naloxone uniquely reverses opioid-induced respiratory depression and may precipitate withdrawal. Naloxone was approved by the FDA in 1971 as a diagnostic and therapeutic agent for the treatment of opioid-induced respiratory depression and is currently on the WHO Model List of Essential Medicines (WHO, 2015).
Adverse reactions and consequential events associated with naloxone are well established in the literature. Serious complications (seizure, pulmonary edema, asystole, cardiac arrest) following naloxone administration are reportedly rare (occurring in between 0.3 and 1.6 percent of individuals) (Buajordet et al., 2004; Osterwalder, 1996; Yealy et al., 1990) and could be related to the overdose itself as opposed to the naloxone. Opioid withdrawal symptoms (confusion, headache, nausea or vomiting, aggressiveness, tachycardia, sweating, and tremor) are expected in opioid-dependent persons (Buajordet et al., 2004; Osterwalder, 1996; Terman, 2012; Yealy et al., 1990). Also reported in postoperative patients are hypotension, hypertension, ventricular tachycardia and fibrillation, and pulmonary edema. Naloxone is light-sensitive, is recommended to be stored at room temperature, and typically has a shelf life of 18 to 24 months. It is safe, effective, and nonaddictive and lacks contraindications except for a possible rare allergic reaction (Hardmann et al., 2001; Sporer, 1999, 2003).
While use of naloxone over the past 40 years has been primarily by trained health professionals in research, hospital, and prehospital settings, community activism since the late 1990s on the part of harm reduction organizations and people who use drugs has moved it to the forefront of efforts to address the opioid crisis. As of 2014, 136 opioid overdose prevention and response programs collectively managed 644 naloxone distribution sites throughout the United States, distributing naloxone kits to 152,283 lay-
persons and reporting 26,463 overdose reversals (between 1996 and 2014) (Wheeler et al., 2015). In addition to the pharmacologic and extensive clinical application literature, the evidence base for expanded community access to naloxone is growing. Data show that educating and providing naloxone to people who are at risk of witnessing or experiencing overdose is associated with reduced heroin consumption (Seal et al., 2005), fewer opioid-related emergency department visits (Coffin et al., 2016), and a 30–45 percent decrease in opioid overdose death rates at the community and individual levels (Bird et al., 2016; Walley et al., 2013). Increasing the availability of naloxone, therefore, is a central component of population-level efforts to prevent opioid overdose deaths, as illustrated by the U.S. Department of Health and Human Services having identified access to naloxone as one of three main strategies for addressing the national opioid epidemic (HHS, 2016).
In the United States, naloxone is available only by prescription, although many states and locales have implemented innovative models of expanded community access to naloxone, such as standing orders (whereby pharmacists are permitted to offer the medication broadly under a prescriber’s order and according to the prescriber’s stipulations); collaborative pharmacy practice agreements (whereby pharmacists are permitted to manage the medication on behalf of a prescriber after fulfilling certain training and documentation requirements); or other regulatory changes (Green et al., 2015b) designed to enable more first responders and laypersons to obtain naloxone from community organizations or pharmacies, to carry the medication, and to use it to reverse a witnessed overdose. Additional laws and policies aimed at providing broader access to naloxone at low or no cost to people at risk of opioid overdose are emerging across the country (see Chapter 5 for discussion of these policies). In addition, the trusted, privileged, and critical access to people who use drugs afforded by these programs is particularly important as the opioid epidemic becomes dominated more by illicit than by prescribed opioids.
Naloxone is a known and established medication. Its generic status has meant that the FDA would consider novel delivery devices or alternative routes of administration along the 505(b)(2) regulatory pathway (discussed in Chapter 6). Indeed, the past 2 years has seen entry into the U.S. market of two new, FDA-approved naloxone products. Patients now can choose among prescribed naloxone products, allowing them to factor in their living situation; type of opioid of exposure; comfort level with syringes; and other factors, such as preference for little to no instruction or voice-activated instructions upon administration. Across all products and access points, instructions stress that training a family member, friend, or caregiver to use naloxone is recommended.
The cost of naloxone is a key consideration for most people (Beletsky et al., 2009) and a major impediment for the branded naloxone products.
The community-based and volunteer capacity of many naloxone distribution programs depends on innovations in pricing, donations, billing, and other distribution factors to sustain low- or no-cost naloxone. It is unclear whether the emergence of multiple new naloxone products will benefit patients, family members, and community-based programs. Unless covered by insurance, the out-of-pocket cost of $40 to $150 for naloxone makes it inaccessible for most people, especially if it is being administered in larger quantities or more frequently in the presence of potent opioids such as fentanyl. Prescription formulary coverage of the different prescription naloxone products varies, but with time and increasing demand (Jones et al., 2016), greater coverage is expected. Indeed, public and private insurers increasingly include naloxone in their formularies, thereby creating a sustainable and accessible source of the medication through medical and pharmacy routes. When naloxone is covered by insurance, its uptake improves, and states such as Rhode Island that have instituted both statewide pharmacy access to naloxone and broad insurance coverage of multiple products have seen the emergence of sustainable models of naloxone access as a complement to community-based programs. However, the new products, and increasingly the generic ones as well, are beyond the financial reach of most community-based programs, many of which have had to rely on small grants or donations. In the face of unprecedented numbers of opioid overdoses and the infiltration of fentanyl into the illicit drug supply, the FDA and other federal agencies would be well advised to take steps to ensure that organizations and institutions with privileged access to those with high overdose risk have free (or lowest-cost) naloxone so as to maximize the reach and sustainability of their efforts. Examples of such steps include novel pricing, alternative models, or price controls.
Finally, several FDA public meetings have considered the prospects and requirements for making naloxone an over-the-counter (OTC) product. A first public meeting in 2012 featured presentations from researchers in naloxone and overdose, the FDA, and others on the state of the science and regulatory requirements for an OTC naloxone product. Absent a branded product, few to no current naloxone manufacturers were willing or able to undertake the studies necessary to achieve that status. Three years after this initial public meeting, a new FDA-approved naloxone product was available, joined by another the following year. At this time, no naloxone product has attained OTC status, and in the meantime, as discussed above, states have greatly expanded access to naloxone through pharmacies, emergency departments, community-based organizations, and first responders using various implementation models. Research is needed to understand the impact and reach of these models. Given the variety of settings in which naloxone providers and programs operate and the unique access of many programs to populations at high risk of overdose, it is unclear how an
OTC naloxone product would improve the accessibility and availability of naloxone at the community level.
Summary
Medication to treat a pernicious side effect of opioid exposure and overdose is available, and two new FDA-approved medications join several generic naloxone products. The provision of naloxone to overdose victims by health professionals in the prehospital setting is the standard of care, and in response to rising community overdose rates, community-based programs and first responder agencies have adopted this protocol for treating opioid overdose. Mechanisms for increasing naloxone prescribing and dispensing and equipping of first responders, and possibly enabling direct patient access (e.g., an OTC status), are warranted, but are impeded by high and unpredictable costs for the medication.
SUMMARY AND RECOMMENDATIONS
While it is unrealistic to expect that the diversion and misuse of pain medications can be entirely eradicated, the effects of these drugs on public health need to be acknowledged, tracked, and mitigated. The interrelated nature of the prescription and illicit opioid epidemics means that one cannot be addressed separately from the other. Moreover, there are both iatrogenic and predictable consequences of opioid exposure at the individual patient and societal levels that can be anticipated and actively mitigated. The downstream effects and societal impact of these intertwined epidemics require consideration by the FDA and other agencies with authority to affect the flow of prescription opioid medications and illicit opioids before, during, and after the introduction of new, similar opioid products into the marketplace. Important research gaps exists in such areas as surveillance; ethnographic studies of drug use behaviors; epidemiologic studies of exposure, natural histories describing transitions in routes of administration and use, and risk of new illicitly manufactureed synthetic opioids; evolving OUD treatment trajectories; changes in opioid markets; and measurement of the impact of use of opioids, particularly heroin and illicit fentanyl, on society and the economy.
Recommendation 4-1. Consider potential effects on illicit markets of policies and programs for prescription opioids. In designing and implementing policies and programs pertaining to prescribing of, access to, and use of prescription opioids, the U.S. Food and Drug Administration, other agencies within the U.S. Department of Health and Human Services, state agencies, and other stakeholders should consider the
potential effects of these interventions on illicit markets—including both the diversion of prescription opioids from lawful sources and the effect of increased demand for illegal opioids such as heroin among users of prescription opioids—and take appropriate steps to mitigate those effects.
Recommendation 4-2. Improve reporting of data on pain and opioid use disorder. The Substance Abuse and Mental Health Services Administration, the U.S. Food and Drug Administration, the National Institutes of Health, and the U.S. Centers for Disease Control and Prevention should collaborate to identify best practices and reporting formats that portray the epidemiology of both pain and opioid use disorder accurately, objectively, and in relation to one another.
Recommendation 4-3. Invest in data and research to better characterize the opioid epidemic. The National Institute on Drug Abuse and the U.S. Centers for Disease Control and Prevention should invest in data collection and research relating to population-level opioid use patterns and consequences, especially nonmedical use of prescription opioids and use of illicit opioids, such as heroin and illicitly manufactured fentanyl.
REFERENCES
Aldridge, J., and D. Décary-Hétu. 2016. Hidden wholesale: The drug diffusing capacity of online drug cryptomarkets. International Journal on Drug Policy 35:7-15.
Alpert, A., D. Powell, and R.L. Pacula. 2017. Supply-side drug policy in the presence of substitutes: Evidence from the introduction of abuse-deterrent opioids. NBER Working Paper 23031. http://www.nber.org/papers/w23031 (accessed February 28, 2017).
Al-Tayyib, A.A., S. Koester, and P. Riggs. 2017. Prescription opioids prior to injection drug use: Comparisons and public health implications. Addictive Behaviors 65:224-228.
Angelotta, C., C.J. Weiss, J.W. Angelotta, and R.A. Friedman. 2016. A moral or medical problem? The relationship between legal penalties and treatment practices for opioid use disorders in pregnant women. Women’s Health Issues 26(6):595-601.
ASAM (American Society of Addiction Medicine). 2017. Summary of the Comprehensive Addiction and Recovery Act. http://www.asam.org/advocacy/issues/opioids/summary-of-the-comprehensive-addiction-and-recovery-act (accessed March 1, 2017).
Baldini, A., M. Von Korff, and E.H. Lin. 2012. A review of potential adverse effects of long-term opioid therapy: A practitioner’s guide. Primary Care Companion for CNS Disorders 14(3).
Banerjee, G., E.J. Edelman, D.T. Barry, W.C. Becker, M. Cerda, S. Crystal, J.R. Gaither, A.J. Gordon, K.S. Gordon, R.D. Kerns, S.S. Martins, D.A. Fiellin, and B.D. Marshall. 2016. Non-medical use of prescription opioids is associated with heroin initiation among U.S. veterans: A prospective cohort study. Addiction 111(11):2021-2031.
Bart, G. 2012. Maintenance medication for opiate addiction: The foundation of recovery. Journal of Addictive Diseases 31(3):207-225.
Bartels, K., L.M. Mayes, C. Dingmann, K.J. Bullard, C.J. Hopfer, and I.A. Binswanger. 2016. Opioid use and storage patterns by patients after hospital discharge following surgery. PLoS One 11(1):e0147972.
Baum, R.M. 1985. New variety of street drugs poses growing problem. Chemical and Engineering News 63(36):7-16.
Baumblatt, J.A.G., C. Wiedeman, J.R. Dunn, W. Schaffner, L.J. Paulozzi, and T.F. Jones. 2014. High-risk use by patients prescribed opioids for pain and its role in overdose deaths. JAMA Internal Medicine 174(5):796-801.
Beaudoin, F.L., R.C. Merchant, and M.A. Clark. 2016. Prevalence and detection of prescription opioid misuse and prescription opioid use disorder among emergency department patients 50 years of age and older: Performance of the Prescription Drug Use Questionnaire, patient version. American Journal of Geriatric Psychiatry 24(8):627-636.
Becker, W.C., J.L. Starrels, M. Heo, X. Li, M.G. Weiner, and B.J. Turner. 2011. Racial differences in primary care opioid risk reduction strategies. Annals of Family Medicine 9(3):219-225.
Beletsky, L., S. Burris, and A. Kral. 2009. Closing death’s door: Action steps to facilitate emergency opioid drug overdose reversal in the U.S. https://papers.ssrn.com/sol3/papers.cfm?abstract-id=1437163 (accessed March 1, 2017).
Bell, J., C.V. Towers, M.D. Hennessy, C. Heitzman, B. Smith, and K. Chattin. 2016. Detoxification from opiates during pregnancy. American Journal of Obstetrics and Gynecology 215(3):374.e1-6.
Binswanger, I.A., M.F. Stern, R.A. Deyo, P.J. Heagerty, A. Cheadle, J.G. Elmore, and T.D. Koepsell. 2007. Release from prison—A high risk of death for former inmates. New England Journal of Medicine 356(2):157-165.
Binswanger, I.A., P.J. Blatchford, R.G. Lindsay, and M.F. Stern. 2011. Risk factors for all-cause overdose and early deaths after release from prison in Washington state. Drug and Alcohol Dependence 117(1):1-6.
Binswanger, I.A., P.J. Blatchford, S.R. Mueller, and M.F. Stern. 2013. Mortality after prison release: Opioid overdose and other causes of death, risk factors, and time trends from 1999 to 2009. Annals of Internal Medicine 159(9):592-600.
Bird, S.M., A. McAuley, S. Perry, and C. Hunter. 2016. Effectiveness of Scotland’s national naloxone programme for reducing opioid-related deaths: A before (2006-10) versus after (2011-13) comparison. Addiction 111(5):883-891.
Blinick, G., R.W. Wallach, and E. Jerez. 1969. Pregnancy in narcotic addicts treated by medical withdrawal. The methadone detoxification program. American Journal of Obstetrics and Gynecology 105(7):997-1003.
Bohnert, A.S., M. Valenstein, M.J. Bair, D. Ganoczy, J.F. McCarthy, M.A. Ilgen, and F.C. Blow. 2011. Association between opioid prescribing patterns and opioid overdose-related deaths. Journal of the American Medical Association 305(13):1315-1321.
Bohnert, A.S.B., J.E. Logan, D. Gnoczy, and D. Dowell. 2016. A detailed exploration into the association of prescribed opioid dosage and prescription opioid overdose deaths among patients with chronic pain. Medical Care 54(5):435-441.
Boutwell, A.E., A. Nijhawan, N. Zaller, and J. Rich. 2006. Arrested on heroin: A national opportunity. Journal of Opioid Management 3(6):328-332.
Braden, J.B., J. Russo, M.Y. Fan, M.J. Edlund, B.C. Martin, A. DeVries, and M.D. Sullivan. 2010. Emergency department visits among recipients of chronic opioid therapy. Archives of Internal Medicine 170(16):328-332.
Brogly, S.B., K.A. Saia, A.Y. Walley, H.M. Du, and P. Sebastian. 2014. Prenatal buprenorphine versus methadone exposure and neonatal abstinence syndrome: Systematic review and meta-analysis. American Journal of Epidemiology 180(7):673-686.
Brooks, J.T. 2017. CDC outbreak investigations involving OPANA®ER. https://www.fda.gov/downloads/AdvisoryCommittees/CommitteesMeetingMaterials/Drugs/AnestheticAndAnalgesicDrugProductsAdvisoryCommittee/UCM547237.pdf (accessed April 23, 2017).
Buajordet, I., A.C. Naess, D. Jacobsen, and O. Brors. 2004. Adverse events after naloxone treatment of episodes of suspected acute opioid overdose. European Journal of Emergency Medicine 11(1):1923.
Butler, S.F., E.C. McNaughton, and R.A. Black. 2015. Tapentadol abuse potential: A postmarketing evaluation using a sample of individuals evaluated for substance abuse treatment. Pain Medicine 16(1):119-130.
Calcaterra, S., J. Glanz, and I.A. Binswanger. 2013. National trends in pharmaceutical opioid related overdose deaths compared to other substance related overdose deaths: 1999–2009. Drug and Alcohol Dependence 131(3):263-270.
Caulkins, J.P., and P. Reuter. 2010. How drug enforcement affects drug prices. http://faculty.publicpolicy.umd.edu/sites/default/files/reuter/files/Drug%20Enforcement%20and%20Drug%20Price.pdf (accessed February 28, 2017).
Caulkins, J.P., B. Kilmer, P.H. Reuter, and G. Midgette. 2015a. Cocaine’s fall and marijuana’s rise: Questions and insights based on new estimates of consumption and expenditures in U.S. drug markets. Addiction 110(5):728-736.
Caulkins, J.P., J. Sussell, B. Kilmer, and A. Kasunic. 2015b. How much of the cocaine market are we missing? Insights from respondent-driven sampling in a mid-sized American city. Drug and Alcohol Dependence 147:190-195.
CDC (U.S. Centers for Disease Control and Prevention). 2013. Thrombotic Thrombocytopenic Purpura (TTP)–like illness associated with intravenous Opana ER abuse—Tennessee, 2012. https://www.cdc.gov/mmwr/preview/mmwrhtml/mm6201a1.htm (accessed June 13, 2017).
CDC. 2014. Quickstats: Rates of drug poisoning deaths involving heroin, by selected age and racial/ethnic groups—United States, 2002 and 2011. Morbidity and Mortality Weekly Report 63(27):595.
CDC. 2015a. National Center for Health Statistics. Multiple cause of death 1999-2014 on CDC WONDER online database, released 2015. Data are from the Multiple Cause of Death Files, 1999-2014, as compiled from data provided by the 57 vital statistics jurisdictions through the Vital Statistics Cooperative Program. http://wonder.cdc.gov/mcd-icd10.html (accessed May 25, 2017).
CDC. 2015b. CDC statement on syringe services programs—December 21, 2015. https://www.cdc.gov/nchhstp/newsroom/2015/syringe_service_statement.html (accessed January 5, 2017).
CDC. 2015c. Today’s Heroin Epidemic Infographics. https://www.cdc.gov/vitalsigns/heroin/infographic.html#graphic (accessed January 22, 2017).
CDC. 2016a. Hepatitis C FAQs for the public. https://www.cdc.gov/hepatitis/hcv/cfaq.htm (accessed April 21, 2017).
CDC. 2016b. Hepatitis C information. Statistics and surveillance. https://www.cdc.gov/hepatitis/hcv/statisticshcv.htm (accessed April 21, 2017).
CDC. 2016c. Prescription opioid overdose data. https://www.cdc.gov/drugoverdose/data/overdose.html (accessed April 23, 2017).
CDC. 2017a. HIV and injection drug use. https://www.cdc.gov/hiv/risk/idu.html (accessed April 21, 2017).
CDC. 2017b. QuickStats: Rates of drug overdose deaths involving heroin, by selected age groups—United States, 2006–2015. Morbidity and Mortality Weekly Report 65(52):1497.
Cerdá, M., Y. Ransome, K.M. Keyes, K.C. Koenen, M. Tracy, K.J. Tardiff, D. Vlahov, and S. Galea. 2013. Prescription opioid mortality trends in New York City, 1990–2006: Examining the emergence of an epidemic. Drug and Alcohol Dependence 132(1):53-62.
Chen, L.H., H. Hedegaard, and M. Warner. 2014. Drug-poisoning deaths involving opioid analgesics: United States, 1999–2011. NCHS Data Brief 166. Hyattsville, MD: National Center for Health Statistics.
Chou, R., J.A. Turner, E.B. Devine, R.N. Hansen, S.D. Sullivan, I. Blazina, T. Dana, C. Bougatsos, and R.A. Deyo. 2015. The effectiveness and risks of long-term opioid therapy for chronic pain: A systematic review for a National Institutes of Health Pathways to Prevention Workshop. Annals of Internal Medicine 162(4):276-286.
Cicero, T.J., and M.S. Ellis. 2015. Abuse-deterrent formulations and the prescription opioid abuse epidemic in the United States: Lessons learned from OxyContin. 2015. JAMA Psychiatry 72(5):424-430.
Cicero, T.J., M.S. Ellis, H.L. Surratt, and S.P. Kurtz. 2014. The changing face of heroin use in the United States: A retrospective analysis of the past 50 years. JAMA Psychiatry 71(7):821-826.
Clarke, H., H. Soneji, D.K. Ko, L. Yun, and D.N. Wijeysundera. 2014. Rates and risk factors for prolonged opioid use after major surgery: Population based cohort study. British Medical Journal 348:g1251.
Cleary, B.J., J. Donelly, J. Strawbridge, P.J. Gallagher, T. Fahey, M. Clarke, and D.J. Murphy. 2010. Methadone dose and neonatal abstinence syndrome—systematic review and metaanalysis. Addiction 105(12):2071-2084.
Coffin, P.O., E. Behar, C. Rowe, G. M. Santos, D. Coffa, M. Bald, and E. Vittinghoff. 2016. Nonrandomized intervention study of naloxone coprescription for primary care patients receiving long-term opioid therapy for pain. Annals of Internal Medicine 165(4):245-252.
Comer, S.D., M.A. Sullivan, E. Yu, J.L. Rothenberg, H.D. Kleber, K. Kampman, C. Dackis, and C.P. O’Brien. 2006. Injectable, sustained-release naltrexone for the treatment of opioid dependence: A randomized, placebo-controlled trial. Archives of General Psychiatry 63(2):210-218.
Comer, S.D., M.A. Sullivan, R.A. Whittington, S.K. Vosburg, and W.J. Kowalczyk. 2008. Relative abuse liability of prescription opioids compared to heroin in morphine-maintained heroin abusers. Neuropsychopharmacology 33(5):1179-1191.
Comfort, M.L., and K. Kaltenbach. 1999. Bio-psychosocial characteristics and treatment outcomes of pregnant cocaine dependent women in residential and outpatient substance abuse treatment. Journal of Psychoactive Drugs 30(3):279-289.
Compton, W.M., C.M. Jones, and G.T. Baldwin. 2016. Relationship between nonmedical prescription-opioid use and heroin use. New England Journal of Medicine 374(2):154-163.
Connock, M., A. Juarez-Garcia, S. Jowett, E. Frew, Z. Liu, R. Taylor, A. Fry-Smith, E. Day, N. Lintzeris, and T. Roberts. 2007. Methadone and buprenorphine for the management of opioid dependence: A systematic review and economic evaluation. Health Technology Assessment 11(9):1-171, iii-iv.
Cook, P.J. 2007. Paying the tab: The costs and benefits of alcohol control. Princeton, NJ: Princeton University Press.
Coplan, P.M., N.E. Sessler, V. Harikrishnan, R. Singh, and C. Perkel. 2017. Comparison of abuse, suspected suicidal intent, and fatalities related to the 7-day buprenorphine transdermal patch versus other opioid analgesics in the National Poison Data System. Postgraduate Medicine 129(1):55-61.
Dart, R.C., H.L. Surratt, T.J. Cicero, M.W. Parrino, S.G. Severtson, B. Bucher-Bartelson, and J.L. Green. 2015. Trends in opioid analgesic abuse and mortality in the United States. New England Journal of Medicine 372(3):241-248.
Dart, R.C., H.L. Surrat, M.C. LeLait, Y. Stivers, V.S. Bebarta, C.C. Freifeld, J.S. Brownstein, J.J. Burke, S.P. Kurtz, and N. Dasgupta. 2016. Diversion and illicit sale of extended release Tapentadol in the United States. Pain Medicine 17(8):1490-1496.
Dasgupta, N., C. Freifeld, J.S. Brownstein, C.M. Menone, H.L. Surratt, L. Poppish, J.L. Green, E.J. Lavonas, and R.C. Dart. 2013. Crowdsourcing black market prices for prescription opioids. Journal of Medical Internet Research 15(8):e178.
Dashe, J.S., G.L. Jackson, D.A. Olscher, E.H. Zane, and G.D. Wendel. 1998. Opioid detoxification in pregnancy. Obstetrics and Gynecology 92(5):854-858.
Davis, A.M., and C.E. Inturrisi. 1999. d-Methadone blocks morphine tolerance and N-methyl-d-aspartate-induced hyperalgesia. Journal of Pharmacology and Experimental Therapeutics 289(2):1048-1053.
Davis, M., H.W. Goforth, and P. Gamier. 2013. Oxycodone combined with opioid receptor antagonists: Efficacy and safety. Expert Opinion on Drug Safety 12(3):389-402.
DEA (U.S. Drug Enforcement Administration). 2013. National drug threat assessment summary, 2013. https://www.dea.gov/resource-center/DIR-017-13%20NDTA%20Summary%20final.pdf (accessed April 23, 2017).
DEA. 2016a. Counterfeit prescription pills containing fentanyl: A global threat. https://content.govdelivery.com/attachments/USDOJDEA/2016/07/22/file_attachments/590360/fentanyl%2Bpills%2Breport.pdf (accessed February 28, 2017).
DEA. 2016b. 2016 national drug threat assessment summary. https://www.dea.gov/resource-center/2016%20NDTA%20Summary.pdf (accessed April 23, 2017).
DEA. 2016c. National heroin threat assessment summary—updated. http://www.emsi.org/webfm_send/220 (accessed February 28, 2017).
DEA. 2016d. Public safety alert from Drug Enforcement Administration counterfeit hydrocodone tablets containing fentanyl. https://www.dea.gov/divisions/sf/2016/sf040116.shtml (accessed June 12, 2017).
Debelak, K., W.R. Morrone, K.E. O’Grady, and H.E. Jones. 2013. Buprenorphine + naloxone in the treatment of opioid dependence during pregnancy: Initial patient care and outcome data. American Journal on Addictions 22(3):252-254.
Deck, D., W. Wiitala, B. McFarland, K. Campbell, J. Mullooly, A. Krupski, and D. McCarty. 2009. Medicaid coverage, methadone maintenance, and felony arrests: Outcomes of opiate treatment in two states. Journal of Addictive Diseases 28(2):89-102.
Degenhardt, L., R. Bruno, R. Ali, N. Lintzeris, M. Farrell, and B. Larance. 2015. The introduction of a potentially abuse deterrent oxycodone formulation: Early findings from the Australian National Opioid Medications Abuse Deterrence (NOMAD) study. Drug and Alcohol Dependence 151:56-67.
Dolan, K.A., J. Shearer, M. MacDonald, R.P. Mattick, W. Hall, and A.D. Wodak. 2003. A randomised controlled trial of methadone maintenance treatment versus wait list control in an Australian prison system. Drug and Alcohol Dependence 72(1):59-65.
Dole, V.P., and M. Nyswander. 1965. Medical treatment for diacetylmorphine (heroin) addiction. A clinical trial with methadone hydrochloride. Journal of the American Medical Association 193:646-650.
Dowell, D., T.M. Haegerich, and R. Chou. 2016. CDC guideline for prescribing opioids for chronic pain—United States, 2016. Morbidity and Mortality Weekly Report 65(RR-1):1-49.
Duncan, L.G., S. Mendoza, and H. Hansen. 2015. Buprenorphine maintenance for opioid dependence in public sector healthcare: Benefits and barriers. Journal of Addiction Medicine and Therapeutic Science 1(2):31-36.
Dunn, K.M., K.W. Saunders, C.M. Rutter, C.J. Banta-Green, J.O. Merrill, M.D. Sullivan, C.M. Weisner, M.J. Silverberg, C.I. Campbell, B.M. Psaty, and M. VonKorff. 2010. Opioid prescriptions for chronic pain and overdose: A cohort study. Annals of Internal Medicine 152(2):85-92.
DuPont, R.L. 1971. Profile of a heroin-addiction epidemic. New England Journal of Medicine 285(6):320-324.
DuPont, R.L. 1973. Coming to grips with an urban heroin addiction epidemic. Journal of the American Medical Association 223(1):46-48.
DuPont, R.L. 1974. The rise and fall of heroin addiction. Natural History 83(6):66-71.
DuPont, R.L., and M.H. Greene. 1973. The dynamics of a heroin addiction epidemic. Science 181(4101):716-722.
Dysart, K., H. Hsieh, K. Kaltenbach, and J.S. Greenspan. 2007. Sequela of preterm versus term infants born to mothers on a methadone maintenance program: Differential course of neonatal abstinence syndrome. Journal of Perinatal Medicine 35(4):344-346.
EMCDDA (European Monitoring Centre for Drugs and Drug Addiction). 2015a. European drug report 2015: Trends and developments. Lisbon, Portugal: EMCDDA.
EMCDDA. 2015b. Preventing overdose deaths in Europe. Lisbon, Portugal: EMCDDA.
EMCDDA. 2016. Preventing opioid overdose deaths with take-home naloxone. Lisbon, Portugal: EMCDDA.
Farrell, M., and J. Marsden. 2008. Acute risk of drug-related death among newly released prisoners in England and Wales. Addiction 103(2):251-255.
Fatima, M., S. Srivastav, and A.C. Mondal. 2017. Prenatal stress and depression associated neuronal development in neonates. International Journal of Developmental Science 60:1-7.
Faul., M., M. Bohm, and C. Alexander. 2017. Methadone prescribing and overdose and the association with Medicaid preferred drug list policies—United States, 2007–2014. Morbidity and Mortality Weekly Report 66:320-323.
FDA (U.S. Food and Drug Administration). 2016. FDA requires strong warnings for opioid analgesics, prescription opioid cough products, and benzodiazepine labeling related to serious risks and death from combined use. https://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm518697.htm (accessed March 2, 2017).
FDA. 2017a. FDA news release: FDA requests removal of Opana ER for risks related to abuse. https://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm562401.htm?source=govdelivery&utm_medium=email&utm_source=govdelivery (accessed June 8, 2017).
FDA. 2017b. Risk Evaluation and Mitigation Strategy (REMS) for extended-release and long-acting opioid analgesics. https://www.fda.gov/Drugs/DrugSafety/InformationbyDrugClass/ucm163647.htm (accessed June 10, 2017).
Ferri, M., M. Davoli, and C.A. Perucci. 2011. Heroin maintenance for chronic heroin-dependent individuals. Cochrane Database of Systematic Reviews 12:CD003410.
Ferri, M., S. Minozzi, A. Bo, and L. Amato. 2013. Slow-release oral morphine as maintenance therapy for opioid dependence. Cochrane Database of Systematic Reviews 6:CD009879.
Fiellin, D.A., M.V. Pantalon, M.C. Chawarski, B.A. Moore, L.E. Sullivan, P.G. O’Connor, and R.S. Schottenfeld. 2006. Counseling plus buprenorphine-naloxone maintenance therapy for opioid dependence. New England Journal of Medicine 355:365-374.
Finnegan, L.P., and K. Kaltenbach. 1992. Neonatal abstinence syndrome. In Primary pediatric care, 2nd ed., edited by R.A. Hoekelman, S.B. Friedman, and N.M. Nelson. St. Louis, MO: Mosby. Pp. 1367-1378.
Gaalema, D.E., T.L. Scott, S.H. Heil, M.G. Coyle, K. Kaltenbach, G.J. Badger, A.M. Arria, S.M. Stine, P.R. Martin, and H.E. Jones. 2012. Differences in the profile of neonatal abstinence syndrome signs in methadone—versus buprenorphine—exposed infants. Addiction 107(Suppl. 1):53-62.
Gibson, K.S., S. Stark, D. Kumar, and J. Bailit. 2017. Relationship between gestational age and the severity of neonatal abstinence syndrome. Addiction 112(4):711-716.
Gomes, T., M.M. Mamdani, I.A. Dhalla, J.M. Paterson, and D.N. Juurlink. 2011. Opioid dose and drug-related mortality in patients with nonmalignant pain. Archives of Internal Medicine 171:686-691.
Gray, E. 2014. Heroin gains popularity as cheap doses flood the U.S. http://time.com/4505/heroin-gains-popularity-as-cheap-doses-flood-the-u-s (accessed March 1, 2017).
Green, M.S., and R.A. Chambers. 2015. Pseudoaddiction: Fact or fiction? An investigation of the medical literature. Current Addiction Reports 2(4):310-317.
Green, T.C., and M. Gilbert. 2016. Invited commentary: Counterfeit medications and fentanyl. JAMA Internal Medicine 176(10):1555-1557.
Green, T.C., C. Griffel, T. Dailey, P. Garg, E. Thorley, C. Kaczmarsky, and S.F. Butler. 2015a. How did you know you got the right pill? Prescription opioid identification and measurement error in the abuse deterrent formulation era. Addiction Science & Clinical Practice 10(Suppl. 1):A16.
Green, T.C., E.F. Dauria, J. Bratberg, C.S. Davis, and A.Y. Walley. 2015b. Orienting patients to greater opioid safety: Models of community pharmacy-based naloxone. Harm Reduction Journal 12:25.
Hagan, H., E.R. Pouget, and D.C. Des Jarlais. 2011. A systematic review and meta-analysis of interventions to prevent hepatitis C virus infection in people who inject drugs. Journal of Infectious Diseases 204(1):74-83.
Hall, A.J., J.E. Logan, R.L. Toblin, J.A. Kaplan, J.C. Kraner, D. Bixler, A.E. Crosby, and L.J. Paulozzi. 2008. Patterns of abuse among unintentional pharmaceutical overdose fatalities. Journal of the American Medical Association 300(22):2613-2620.
Hansen, H., C. Siegel, J. Wanderling, and D. DiRocco. 2016. Buprenorphine and methadone treatment for opioid dependence by income, ethnicity and race of neighborhoods in New York City. Drug and Alcohol Dependence 164:14-21.
Hardman, J.G., L.E. Limberd, and A.G. Gilman, Eds. 2001. Goodman and Gilman’s the pharmacologic basis of therapeutics. 10th ed. New York: McGraw-Hill.
Hartung, D.M., D. McCarty, R. Fu, K. Wiest, M. Chalk, and D.R. Gastfriend. 2014. Extended-release naltrexone for alcohol and opioid dependence: A meta-analysis of healthcare utilization studies. Journal of Substance Abuse Treatment 47(2):113-121.
Häuser, W., F. Petzke, L. Radbruch, and T.R. Tölle. 2016. The opioid epidemic and the long-term opioid therapy for chronic noncancer pain revisited: A transatlantic perspective. Pain Management 6(3):249-263.
Havens, J.R., C.G. Leukefeld, A.M. DeVeaugh-Geiss, P. Coplan, and H.D. Chilcoat. 2014. The impact of a reformulation of extended-release oxycodone designed to deter abuse in a sample of prescription opioid abusers. Drug and Alcohol Dependence 139:9-17.
Heimer, R., H. Catania, R.G. Newman, J. Zambrano, A. Brunet, and A.M. Ortiz. 2006. Methadone maintenance in prison: Evaluation of a pilot program in Puerto Rico. Drug and Alcohol Dependence 83(2):122-129.
Henderson, A.W., K.M. Babu, R.C. Merchant, and F.L. Beaudoin. 2015. Prescription opioid use and misuse among older adult Rhode Island hospital emergency department patients. Rhode Island Medical Journal 98(3):28-31.
HHS (U.S. Department of Health and Human Services). 2016. Opioids Factsheet. https://www.hhs.gov/sites/default/files/Factsheet-opioids-061516.pdf (accessed January 11, 2017).
Holmes, A.V., E.C. Atwood, B. Whalen, J. Beliveau, J.D. Jarvis, and S.L. Ralston. 2016. Rooming-in to treat neonatal abstinence syndrome: Improved family centered care and lower cost. Pediatrics 137(6).
Hubbard, R.L., and M.E. Marsden. 1986. Relapse to use of heroin, cocaine, and other drugs in the first year after treatment. NIDA Research Monograph 72:157-166.
Hudak, M.L., and R.C. Tan. 2012. Neonatal drug withdrawal. Pediatrics 129(2):e540-e560.
Hughes, A., M.R. Williams, R.N. Lipari, J. Bose, E.A.P. Copello, and L.A. Kroutil. 2016. Prescription drug use and misuse in the United States: Results from the 2015 National Survey on Drug Use and Health. https://www.samhsa.gov/data/sites/default/files/NSDUH-FFR2-2015/NSDUH-FFR2-2015.htm (accessed August 14, 2017).
Hunt, E., R.H. Peters, and J. Kremling. 2015. Behavioral health treatment history among persons in the justice system: Findings from the Arrestee Drug Abuse Monitoring II Program. Psychiatric Rehabilitation Journal 38(1):7-15.
Inciardi, J.A., H.L. Surratt, S.P. Kurtz, and T.J. Cicero. 2007. Mechanisms of prescription drug diversion among drug-involved club- and street-based populations. Pain Medicine 8(2):171-183.
Inciardi, J.A., H.L. Surratt, T.J. Cicero, S.P. Kurtz, S.S. Martin, and M.W. Parrino. 2009. The “black box” of prescription drug diversion. Journal of Addictive Diseases 28(4):332-347.
IOM (Institute of Medicine). 1995. Federal regulation of methadone treatment. Washington, DC: National Academy Press.
IOM. 2011. Relieving pain in America: A blueprint for transforming prevention, care, education, and research. Washington, DC: National Academy Press.
Isbell, H., A. Wikler, N.E. Eddy, J.L. Wilson, and C.F. Moran. 1947. Tolerance and addiction liability of 6-dimethylanimo-4-4-diphenylheptanone-3 (Methadon). Journal of the American Medical Association 135(14):888-894.
Iwanicki, J.L., S.G. Severtson, H. McDaniel, A. Rosenblum, C. Fong, T.J. Cicero, M.S. Ellis, S.P. Kurtz, M.E. Butttram, and R.C. Dart. 2016. Abuse and diversion of immediate release opioid analgesics as compared to extended release formulations in the United States. PLoS One 11(12):e0167499.
Jansson, L.M., J.A. Dipietro, A. Elko, and M. Velez. 2007. Maternal vagal tone changes in response to methadone associated with neonatal abstinence syndrome severity in exposed infants. Journal of Maternal-Fetal and Neonatal Medicine 20(9):677-685.
Jansson, L.M., J.A. Diepietro, A. Elko, and M. Velez. 2010. Infant autonomic functioning and neonatal abstinence. Drug and Alcohol Dependence 109(1-3):198-204.
Jarvis, M.A., and S.H. Schnoll. 1994. Methadone treatment during pregnancy. Journal of Psychoactive Drugs 26(2):151-161.
Jonas, A.B., A.M. Young, C.B. Oser, C.G. Leukefeld, and J.R. Havens. 2012. OxyContin® as currency: OxyContin® use and increased social capital among rural Appalachian drug users. Social Science & Medicine 74(10):1602-1609.
Jones, C.M. 2013a. Heroin use and heroin use risk behaviors among nonmedical users of prescription opioid pain relievers—United States, 2002–2004 and 2008–2010. Drug and Alcohol Dependence 132(1-2):95-100.
Jones, C.M. 2013b. Trends in the distribution of selected opioids by state, US, 1999–2011. Presented at National Meeting Safe States Alliance, Baltimore, MD, June 6.
Jones, C.M., and J.K. McAninch. 2015. Emergency department visits and overdose deaths from combined use of opioids and benzodiazepines. American Journal of Preventive Medicine 49(4):493-501.
Jones, C.M., J. Logan, R.M. Gladden, and M.K. Bohm. 2015. Vital signs: Demographic and substance use trends among heroin users—United States, 2002–2013. Morbidity and Mortality Weekly Report 64(26):719-725.
Jones, C.M., P.G. Lurie, and W.M. Compton. 2016. Increase in naloxone prescriptions dispensed in U.S. retail pharmacies since 2013. American Journal of Public Health 106(4):689-690.
Jones, H.E., and A. Fielder. 2015. Neonatal abstinence syndrome: Historical perspective, current focus, future directions. Prevention Medicine 80:12-17.
Jones, H.E., M.L. Velez, M.E. McCaul, and D.S. Svikis. 1999. Special treatment issues for women. In Methadone treatment for opioid dependence, edited by E.C. Strain and M. Stizer. Baltimore, MD: John Hopkins University Press. Pp. 251-280.
Jones, H.E., K.E. O’Grady, D. Malfi, and M. Tuten. 2008. Methadone maintenance vs. methadone taper during pregnancy: Maternal and neonatal outcomes. American Journal on Addictions 17(5):372-386.
Jones, H.E., K. Kaltenbach, S.H. Heil, S.M. Stine, M.G. Coyle, A.M. Arria, K.E. O’Grady, P. Selby, P.R. Martin, and G. Fischer. 2010. Neonatal abstinence syndrome after methadone or buprenorphine exposure. New England Journal of Medicine 363(24):2320-2331.
Jones, H.E., L.P. Finnegan, and K. Kaltenbach. 2012. Methadone and buprenorphine for the management of opioid dependence in pregnancy. Drugs 72(6):747-757.
Jones, H.E., S.H. Heil, M. Tuten, M.S. Chisolm, J.M. Foster, K.E. O’Grady, and K. Kaltenbach. 2013. Cigarette smoking in opioid dependent pregnant women: Neonatal and maternal outcomes. Drug and Alcohol Dependence 131(3):271-277.
Jones, H.E., M. Terplan, and M. Meyer. 2017. Medically assisted withdrawal (detox): Considering the mother–infant dyad. Journal of Addiction Medicine 11(2):90-92.
Kaltenbach, K., and L.P. Finnegan. 1986. Neonatal abstinence: Pharmacotherapy and developmental outcome. Neurobehavioral Toxicology and Teratology 8(4):353-355.
Kaltenbach, K., and H.E. Jones. 2016. Neonatal abstinence syndrome: Presentation and treatment considerations. Journal of Addiction Medicine 10(4):217-233.
Kaltenbach, K., V. Berghella, and L.P. Finnegan. 1998. Opioid dependence during pregnancy: Effects and management. Obstetrics and Gynecology Clinics of North America 25(1):139-151.
Kaltenbach, K., A. Holbrook, M. Coyle, S.H. Heil, A.L. Salisbury, S. Stine, P.R. Martin, and H.E. Jones. 2012. Predicting treatment for neonatal abstinence syndrome in infants born to women maintained on opioid agonist medication. Addiction 107(Suppl. 1):45-52.
Kaplan, J. 1983. Heroin: The hardest drug. Chicago, IL: University of Chicago Press.
Kenan, K., K.A. Mack, and L. Paulozzi. 2012. Trends in prescriptions for oxycodone and other commonly used opioids in the United States, 2000–2010. Open Medicine 6(2):41-47.
Kennedy-Hendricks, A., A. Gielen, E. McDonald, E.E. McGinty, W. Shields, and C.L. Barry. 2016. Medication sharing, storage, and disposal practices for opioid medications among U.S. adults. JAMA Internal Medicine 176(7):1027-1029.
Kerr, T., N. Fairbairn, M. Tyndall, D. Marsh, K. Li, J. Montaner, and E. Wood. 2007. Predictors of non-fatal overdose among a cohort of polysubstance-using injection drug users. Drug and Alcohol Dependence 87(1):39-45.
Khalid, L., J.M. Liebschutz, Z. Xuan, S. Dossabhov, Y. Kim, D. Crooks, C. Shanahan, A. Lange, O. Heymann, and K.E. Lasser. 2015. Adherence to prescription opioid monitoring guidelines among residents and attending physicians in the primary care setting. Pain Medicine 16(3):480-487.
Kilmer, B., and J. Caulkins. 2014. Hard drugs demand solid understanding. USA Today, March 8. https://www.usatoday.com/story/opinion/2014/03/08/heroin-abuse-hoffman-research-column/6134337 (accessed April 12, 2017).
Kilmer, B., S. Everingham, J. Caulkins, G. Midgette, R. Pacula, P. Reuter, R. Burnes, B. Han, and R. Lundberg. 2014. What America’s users spend on illegal drugs: 2000-2010. http://atforum.com/documents/wausid_results_report.pdf (accessed February 28, 2017).
Kinlock, T.W., M.S. Gordon, R.P. Schwartz, T.T. Fitzgerald, and K.E. O’Grady. 2009. A randomized clinical trial of methadone maintenance for prisoners: Results at 12 months postrelease. Journal of Substance Abuse Treatment 37(3):277-285.
Ko, J.Y., S.W. Patrick, V.T. Tong, R. Patel, J.N. Lind, and W.D. Barfield. 2016. Incidence of neonatal abstinence syndrome-28 states, 1999–2013. Morbidity and Mortality Weekly Report 65(31):799-802.
Kolodny, A., D.T. Courtwright, C.S. Hwang, P. Kreiner, J.L. Eadie, T.W. Clark, and G.C. Alexander. 2015. The prescription opioid and heroin crisis: A public health approach to an epidemic of addiction. Annual Review of Public Health 36:559-574.
Kraft, W.K., K. Dysart, J.S. Greenspan, E. Gibson, K. Kaltenbach, and M.E. Ehrlich. 2011. Revised dose schema of sublingual buprenorphine in the treatment of neonatal opioid abstinence syndrome. Addiction 106(3):574-580.
Kraft, W.K., S.C. Adenivi-Jones, I. Chervoneya, J.S. Greenspan, D. Abatemarco, K. Kaltenbach, and M.E. Ehrlich. 2017. Buprenorphine for the treatment of the neonatal abstinence syndrome. New England Journal of Medicine 376:2341-2348.
Kreek, M.J. 1973. Medical safety and side effects of methadone in tolerant individuals. Journal of the American Medical Association 223(6):665-668.
Krupitsky, E.M., and E.A. Blokhina. 2010. Long-acting depot formulations of naltrexone for heroin dependence: A review. Current Opinion in Psychiatry 23(3):210-214.
Krupitsky, E., E. Avartau, E. Blokhina, E. Verbitskaya, V. Wahlgren, M. Tsoy-Podosenin, N. Bushara, A. Burakov, D. Masalov, T. Romanova, A. Tyurina, V. Palatkin, T. Slavina, A. Pecoraro, and G.E. Woody. 2012. Randomized trial of long-acting sustained-release naltrexone implant vs. oral naltrexone or placebo for preventing relapse to opioid dependence. Archives of General Psychiatry 69(9):973-981.
Lankenau, S.E., S.M. Schrager, K. Silva, A. Kocejevic, J.J. Bloom, C. Wong, and E. Iverson. 2012. Misuse of prescription and illicit drugs among high-risk young adults in Los Angeles and New York. Journal of Public Health Research 1(1):22-30.
Larance, B., R. Mattick, R. Ali, N. Lintzeris, R. Jenkinson, N. White, I. Kihas, R. Cassidy, and L. Degenhardt. 2015. Diversion and injection of buprenorphine-naloxone films two years post-introduction in Australia. Drug and Alcohol Review 35:83-91.
Lee, J.D., R. McDonald, E. Grossman, J. McNeely, E. Laska, J. Rotrosen, and M.N. Gourevitch. 2015. Opioid treatment at release from jail using extended-release naltrexone: A pilot proof-of-concept randomized effectiveness trial. Addiction 110(6):1008-1014.
Liang, Y., and B.J. Turner. 2015. Assessing risk for drug overdose in a national cohort: Role for both daily and total opioid dose? The Journal of Pain 16(4):313-325.
Logan, K., and S. Deutsch. 2015. Room for improvement in the New York State pharmacy-based syringe access program. Columbia Medical Review 1(1):40-50.
Lund, I.O., H. Fitzimons, M. Tuten, M.S. Chisolm, and H.E. Jones. 2012. Comparing methadone and buprenorphine maintenance with medication assisted withdrawal for the treatment of opioid dependence in pregnancy. Substance Abuse Rehabilitation 3(Suppl. 1):17-25.
Lund, I.O., G. Fischer, G.K. Welle-Strand, K.E. O’Grady, K. Debelak, W.R. Morrone, and H.E. Jones. 2013. A comparison of buprenorphine + naloxone to buprenorphine and methadone in the treatment of opioid dependence during pregnancy: Maternal and neonatal outcomes. Substance Abuse Research and Treatment 7:61-74.
Ly, K.N., E.M. Hughes, R.B. Jiles, and S.D. Holmberg. 2016. Rising mortality associated with hepatitis C virus in the United States, 2003–2013. Clinical Infectious Diseases 62(10):1287-1288.
MacArthur, G.J., S. Minozzi, N. Martin, P. Vickerman, S. Deren, J. Bruneau, L. Degenhardt, and M. Hickman. 2012. Opiate substitution treatment and HIV transmission in people who inject drugs: Systematic review and meta-analysis. British Medical Journal 345:e5945.
Massachusetts Department of Public Health. 2016. An assessment of opioid-related deaths in Massachusetts (2013–2014). Boston, MA: Massachusetts Department of Public Health.
Mateu-Gelabert, P., H. Guarino, L. Jessell, and A. Teper. 2015. Injection and sexual HIV/HCV risk behaviors associated with nonmedical use of prescription opioids among young adults in New York City. Journal of Substance Abuse Treatment 48:13-20.
Mattick, R.P., C. Breen, J. Kimber, and M. Davoli. 2009. Methadone maintenance therapy versus no opioid replacement therapy for opioid dependence. Cochrane Database of Systematic Reviews 3:CD002209.
McKenzie, M., N. Zaller, S.L. Dickman, T.C. Green, A. Parihk, P.D. Friedmann, and J.D. Rich. 2012. A randomized trial of methadone initiation prior to release from incarceration. Substance Abuse 33(1):19-29.
McLellan, A.T., I.O. Arndt, D.S. Metzger, G.E. Woody, and C.P. O’Brien. 1993. The effects of psychosocial services in substance abuse treatment. Journal of the American Medical Association 269:1953-1959.
McNaughton, E.C., R.A. Black, S.E. Weber, and S.F. Butler. 2015. Assessing abuse potential of new analgesic medications following market release: An evaluation of Internet discussion of tapentadol abuse. Pain Medicine 16(1):131-140.
McQueen, K., and J. Murphy-Oikonen. 2016. Neonatal abstinence syndrome. New England Journal of Medicine 375:2468-2479.
Merrall, E.L., A. Kariminia, I.A. Binswanger, M.S. Hobbs, M. Farrell, J. Marsden, S.J. Hutchinson, and S.M. Bird. 2010. Meta-analysis of drug-related deaths soon after release from prison. Addiction 105(9):1545-1554.
Miller, M., C.W. Barber, S. Leatherman, J. Fonda, J.A. Hermos, K. Cho, and D.R. Gagnon. 2015. Prescription opioid duration of action and the risk of unintentional overdose among patients receiving opioid therapy. JAMA Internal Medicine 175(4):608-615.
Miller, T., and D. Hendrie. 2008. Substance abuse prevention dollars and cents: A cost-benefit analysis. DHHS Publication (SMA) 07-4298. Rockville, MD: Center for Substance Abuse Prevention, Substance Abuse and Mental Health Services Administration.
Muhuri, P., J. Gfroerer, and M.C. Davies. 2013. Associations of nonmedical pain reliever use and initiation of heroin use in the United States. CBHSQ Data Review 2013 (August). https://www.samhsa.gov/data/sites/default/files/DR006/DR006/nonmedical-pain-reliever-use-2013.htm (accessed May 25, 2017).
NICE (National Institute for Health and Care Excellence). 2007. Methadone and buprenorphine for the management of opioid dependence. https://www.nice.org.uk/guidance/ta114/resources/methadone-and-buprenorphine-for-the-management-of-opioid-dependencepdf-82598072878789 (accessed May 1, 2017).
NIDA (National Institute on Drug Abuse). 2014. America’s addiction to opioids: Heroin and prescription drug abuse. https://www.drugabuse.gov/about-nida/legislative-activities/testimony-to-congress/2016/americas-addiction-to-opioids-heroin-prescription-drug-abuse (accessed February 11, 2017).
NIDA. 2015. Community Epidemiology Work Group (CEWG). https://www.drugabuse.gov/about-nida/organization/workgroups-interest-groups-consortia/community-epidemiology-work-group-cewg (accessed April 12, 2017).
NIDA. 2016. What is fentanyl? https://www.drugabuse.gov/publications/drugfacts/fentanyl (accessed June 13, 2017).
Nielsen, S., B. Larance, and N. Lintzeris. 2017. Opioid agonist treatment for patients with dependence on prescription opioids. Journal of the American Medical Association 317(9): 967-968.
NIJ (National Institute of Justice). 2014. NIJ’s drugs and crime research: Arrestee drug abuse monitoring programs. https://www.nij.gov/topics/drugs/markets/adam/pages/welcome.aspx (accessed April 12, 2017).
Olshansky, S.J., T. Antonucci, L. Berkman, R.H. Binstock, A. Boersch-Supan, J.T. Cacioppo, B.A. Carnes, L.L. Carstensen, L.P. Fried, D.P. Goldman, J. Jackson, M. Kohli, J. Rother, Y. Zheng, and J. Rowe. 2012. Differences in life expectancy due to race and educational differences are widening, and many may not catch up. Health Affairs 31(8):1803-1813.
Osterwalder, J.J. 1996. Naloxone—for intoxications with intravenous heroin and heroin mixtures—harmless or hazardous? A prospective clinical study. Journal of Toxicology: Clinical Toxicology 34(4):409-416.
Oviedo-Joekes, E., D. Guh, S. Brissette, K. Merchand, S. MacDonald, K. Lock, S. Harrison, A. Jonmohamed, A.H. Anis, M. Krausz, D.C. Marsh, and M.T. Schechter. 2016. Hydromorphone compared with diacetylmorphine for long-term opioid dependence: A randomized clinical trial. JAMA Psychiatry 73(5):447-455.
Park, T.W., R. Saitz, D. Ganoczy, M.A. Ilgen, and A.S.B. Bohnert. 2015. Benzodiazepine prescribing patterns and deaths from drug overdose among U.S. veterans receiving opioid analgesics: Case-cohort study. British Medical Journal 350:h2698.
Patrick, S.W., R.E. Schumacher, B.D. Benneyworth, E.E. Krans, J.M. McAllister, and M.M. Davis. 2012. Neonatal abstinence syndrome and associated health care expenditures: United States, 2000-2009. Journal of the American Medical Association 307(18): 1934-1940.
Patrick, S.W., M.M. Davis, C.U. Lehman, and W.O. Cooper. 2015. Increasing incidence and geographic distribution of neonatal abstinence syndrome: United States, 2009-2012. Journal of Perinatology 35:650-655.
Paulozzi, L.J. 2012. Prescription drug overdoses: A review. Journal of Safety Research 43(4):283-289.
Paulozzi, L.J., J.E. Logan, A.J. Hall, E. McKinstry, J.A. Kaplan, and A.E. Crosby. 2009. A comparison of drug overdose deaths involving methadone and other opioid analgesics in West Virginia. Addiction 104(9):1541-1548.
Peavy, K.M., C.J. Banta-Green, S. Kingston, M. Hanrahan, J.O. Merrill, and P.O. Coffin. 2012. “Hooked on” prescription-type opiates prior to using heroin: Results from a survey of syringe exchange clients. Journal of Psychoactive Drugs 44(3):259-265.
Pollini, R.A., C.J. Banta-Green, J. Cuevas-Mota, M. Metzner, E. Teshale, and R.S. Garfein. 2011. Problematic use of prescription-type opioids prior to heroin use among young heroin injectors. Substance Abuse and Rehabilitation 2(1):173-180.
President’s Commission on Law Enforcement and Administration of Justice. 1967. The challenge of crime in a free society. Washington, DC: U.S. Government Printing Office.
Pritham, U.A., J.A. Paul, and M.J. Hayes. 2012. Opioid dependency in pregnancy and length of stay for neonatal abstinence syndrome. Journal of Obstetric, Gynecologic and Neonatal Nursing 41(20):180-190.
Quintana, P., M. Ventura, M. Grifell, A. Palma, L. Galindo, I. Fornis, C. Gil, X. Carbon, F. Caudevilla, M. Farre, and M. Torrens. 2017. The hidden web and the fentanyl problem: Detection of ocfentanil as an adulterant in heroin. International Journal on Drug Policy 40:78-83.
Raffa, R.B., R. Taylor, and J.V. Pergolizzi. 2014. Sequestered naltrexone in sustained release morphine or oxycodone: A way to inhibit illicit use? Expert Opinion on Drug Safety 13(2):181-190.
Ray, W.A., C.P. Chung, K.T. Murray, W.O. Cooper, K. Hall, and C.M. Stein. 2015. Out-of-hospital mortality among patients receiving methadone for noncancer pain. Out-of-hospital mortality among patients receiving methadone for noncancer pain. JAMA Internal Medicine 175(3):420-427.
Reuter, P., and M. Kleiman. 1986. Risks and prices: An economic analysis of drug enforcement. In Crime and justice: A review of research, Vol. 7, edited by M. Tonry and N. Morris. Chicago, IL: University of Chicago Press. Pp. 289-340.
Rich, B.A., and L.R. Webster. 2011. A review of forensic implications of opioid prescribing with examples from malpractice cases involving opioid-related overdose. Pain Medicine 12(Suppl. 2):S59-S65.
Rich, J.D., M. McKenzie, S. Larney, J.B. Wong, L. Tran, J. Clarke, A. Noska, M. Reddy, and N. Zaller. 2015. Methadone continuation versus forced withdrawal on incarceration in a combined U.S. prison and jail: A randomised, open-label trial. Lancet 386(9991):350-359.
Rockett, I.R., G. Hobbs, D. De Leo, S. Stack, J.L. Frost, A.M. Ducatman, N.D. Kapusta, and R.L. Walker. 2010. Suicide and unintentional poisoning mortality trends in the United States, 1987–2006: Two unrelated phenomena? BMC Public Health 10:705.
Rowe, C., E. Vittinghoff, G.M. Santos, E. Behar, C. Turner, and C.O. Coffin. 2016. Performance measures of diagnostic codes for detecting opioid overdose in the emergency department. Academic Emergency Medicine 24(4):475-483.
Rudd, R.A., N. Aleshire, J.E. Zibbell, and R.M. Gladden. 2016a. Increases in drug and opioid overdose deaths—United States, 2000–2014. Morbidity and Mortality Weekly Report 64(50-51):1378-1382.
Rudd, R.A., P. Seth, F. David, and L. Scholl. 2016b. Increases in drug and opioid-involved overdose deaths—United States, 2010–2015. Morbidity and Mortality Weekly Report 65:1445-1452.
Ruwanpathirana, R., M.E. Abedel-Latif, L. Burns, J. Chen, F. Craig, K. Lui, and J.L. Oei. 2015. Prematurity reduces the severity and need for treatment of neonatal abstinence syndrome. Acta Paediatrica 104(5):e188-e194.
Sachs, H.C. 2013. The transfer of drugs and therapeutics into human breast milk. Pediatrics 132(3):e796-e809.
SAMHSA (Substance Abuse and Mental Health Services Administration). 2007. Methadone mortality: A reassessment—report of the meeting and follow-up activities, July 2007. Rockville, MD: U.S. Department of Health and Human Services.
SAMHSA. 2009. Substance abuse treatment addressing the specific needs of women. A treatment improvement protocol. TIP 51. http://store.samhsa.gov/shin/content/SMA13-4426/SMA13-4426.pdf (accessed March 7, 2017).
SAMHSA. 2010. Treatment Episode Data Set: 2007. Discharges from substance abuse treatment services. HHS Publication SMA 10-4479. Rockville, MD: SAMHSA.
SAMHSA. 2012. Results from the 2011 National Survey on Drug Use and Health: Summary of national findings. HHS Publication SMA 12-4713, NSDUH Series H-44. Rockville, MD: SAMHSA.
SAMHSA. 2013a. Results from the 2012 National Survey on Drug Use and Health: Summary of national findings. HHS Publication SMA 13-4795, NSDUH Series H-46. Rockville, MD: SAMHSA.
SAMHSA. 2013b. Treatment Episode Data Set (TEDS): 2001–2011. State admissions to substance abuse treatment services. Rockville, MD: SAMHSA. https://www.samhsa.gov/data/sites/default/files/TEDS2011St_Web/TEDS2011St_Web/TEDS2011St_Web.pdf (accessed May 25, 2017).
SAMHSA. 2014. National Survey on Drug Use and Health. NSDUH-2014. http://datafiles.samhsa.gov/study/national-survey-drug-use-and-health-nsduh-2014-nid13618 (accessed January 10, 2017).
SAMHSA. 2016a. 2015 National Survey on Drug Use and Health: Detailed tables. Rockville, MD: SAMHSA. https://www.samhsa.gov/data/sites/default/files/NSDUH-DetTabs-2015/NSDUH-DetTabs-2015/NSDUH-DetTabs-2015.pdf (accessed April 7, 2017).
SAMHSA. 2016b. About emergency department data. https://www.samhsa.gov/data/emergency-department-data-dawn/about (accessed January 18, 2017).
SAMHSA. 2016c. Collaborative approach to the treatment of pregnant women with opioid use disorders. https://ncsacw.samhsa.gov/files/Collaborative_Approach_508.pdf (accessed March 1, 2017).
SAMHSA. 2017a. Apply to increase patient limits. https://www.samhsa.gov/medication-assisted-treatment/buprenorphine-waiver-management/increase-patient-limits (accessed June 12, 2017).
SAMHSA. 2017b. Buprenorphine waiver management. https://www.samhsa.gov/medication-assisted-treatment/buprenorphine-waiver-management (accessed March 1, 2017).
Sarpatwari, A., M.S.S. Sinha, and A.S. Kesselheim. 2017. The opioid epidemic: Fixing a broken pharmaceutical market. Harvard Law and Policy Review 11:463-484.
Schwartz, R.P., S.M. Kelly, K.E. O’Grady, D. Gandhi, and J.H. Jaffe. 2012. Randomized trial of standard methadone treatment compared to initiating methadone without counseling: 12-month findings. Addiction 107:943-952.
Seal, K.H., R. Thawley, L. Gee, J. Bamberger, A.H. Kral, D. Ciccarone, M. Downing, and B.R. Edlin. 2005. Naloxone distribution and cardiopulmonary resuscitation training for injection drug users to prevent heroin overdose death: A pilot intervention study. Journal of Urban Health 82(2):303-311.
Seligman, N.S., N. Salva, E.J. Hayes, K.C. Dysart, E.C. Pequinot, and J.K. Baxter. 2008. Predicting length of treatment for neonatal abstinence syndrome in methadone exposed infants. American Journal of Obstetrics and Gynecology 199(4):396.e1-e7.
Sells, S., R. Demaree, and C. Hornick. 1979. Comparative effectiveness of drug abuse treatment modalities. Washington, DC: U.S. Department of Health, Education, and Welfare, Public Health Service.
Shah, A., C.J. Hayes, and B.C. Martin. 2017. Characteristics of initial prescription episodes and likelihood of long-term opioid use—United States, 2006–2015. Morbidity and Mortality Weekly Report 66(10):265-269.
Siegal, H.A., R.G. Carlson, D.R. Kenne, and M.G. Swora. 2003. Probable relationship between opioid abuse and heroin use. American Family Physician 67(5):942-945.
Sigmon, S.C. 2015. Interim treatment: Bridging delays to opioid treatment access. Prevention Medicine 80:32-36.
Sigmon, S.C., T.A. Ochalek, A.C. Meyer, B. Hruska, S.H. Bell, G.J. Badger, G. Rose, J.R. Brooklyn, R.P. Schwartz, B.A. Moore, and S.T. Higgins. 2016. Interim buprenorphine vs. waiting list for opioid dependence. New England Journal of Medicine 375(25):2504-2505.
Smith, G. 2016. Maine’s new opioid prescribing law & the opioid crisis: Implications for providers. https://www.mainemed.com/sites/default/files/content/2016_Opioid_Law_c488%20_PowerPoint_9-7-2016.pdf (accessed August 14, 2017).
Smith, M.E., N. Robinowitz, P. Chaulk, and K.E. Johnson. 2014. Self-care and risk reduction habits in older injection drug users with chronic wounds: A cross-sectional study. Harm Reduction Journal 11:28.
Sporer, K.A. 1999. Acute heroin overdose. Annals of Internal Medicine 130(7):584-590.
Sporer, K.A. 2003. Strategies for preventing heroin overdose. British Medical Journal 326(7386):442-444.
Starrels, J.L., W.C. Becker, M.G. Weiner, X. Li, M. Heo, and B.J. Turner. 2011. Low use of opioid risk reduction strategies in primary care even for high risk patients with chronic pain. Journal of General Internal Medicine 26(9):958-964.
Stein, B.D., A.J. Gordon, A.W. Dick, R.M. Burns, R.L. Pacula, C.M. Farmer, D.L. Leslie, and M. Sobrero. 2015. Supply of buprenorphine waivered physicians: The influence of state policies. Journal of Substance Abuse Treatment 48(1):104-111.
Stein, B.D., M. Sorbero, A.W. Dick, R.L. Pacula, R.M. Burns, and A.J. Gordon. 2016. Physician capacity to treat opioid use disorder with buprenorphine-assisted treatment. Journal of the American Medical Association 316(11):1211-1212.
Stewart, R.D., D.B. Nelson, E.H. Adhikari, D.D. McIntire, S.W. Roberts, J.S. Dashe, and J.S. Sheffield. 2013. The obstetrical and neonatal impact of maternal opioid detoxification in pregnancy. American Journal of Obstetrics and Gynecology 209(3):267.e1-e5.
Strang, J., and G. Tober. 2003. Methadone matters: Evolving community methadone treatment of opiate addiction. Boca Raton, FL: CRC Press.
Strang, J., J. McCambridge, D. Best, T. Beswick, J. Bearn, S. Rees, and M. Gossop. 2003. Loss of tolerance and overdose mortality after inpatient opiate detoxification: Follow up study. British Medical Journal 326(7396):959-960.
Strang, J., W. Hall, M. Hickman, and S.M. Bird. 2010. Impact of supervision of methadone consumption on deaths related to methadone overdose (1993–2008): Analyses using OD4 index in England and Scotland. British Medical Journal 341:c4851.
Strang, J., T. Groshkova, A. Uchtenhagen, W. van den Brink, C. Haasen, M.T. Schechter, N. Lintzeris, J. Bell, A. Pirona, E. Oviedo-Joekes, R. Simon, and N. Metrebian. 2015. Heroin on trial: Systematic review and meta-analysis of randomised trials of diamorphine-prescribing as treatment for refractory heroin addiction. British Journal of Psychiatry 207(1):5-14.
Strathdee, S.A., and C. Beyrer. 2015. Threading the needle—How to stop the HIV outbreak in rural Indiana. New England Journal of Medicine 373:397-399.
Stumbo, S.P., B.J. Yarborough, D. McCarty, C. Weisner, and C.A. Green. 2017. Patient-reported pathways to opioid use disorders and pain-related barriers to treatment engagement. Journal of Substance Abuse Treatment 73:45-54.
Su, Q., H. Zhang, Y. Zhang, H. Zhang, D. Ding, J. Zeng, Z. Zhu, and H. Li. 2015. Maternal stress in gestation: Birth outcomes and stress-related hormone response of the neonates. Pediatrics & Neonatology 56(6):376-381.
Sun, E.C., A. Dexit, K. Humphreys, B.D. Darnall, L.C. Baker, and S. Mackey. 2017. Association between concurrent use of prescription opioids and benzodiazepines and overdose: Retrospective analysis. British Medical Journal 356:j760.
Suryaprasad, A.G., J.Z. White, F. Xu, B.A. Eichler, J. Hamilton, A. Patel, S.B. Hamdounia, D.R. Church, K. Barton, C. Fisher, K. Macomber, M. Stanley, S.M. Guilfoyle, K. Sweet, S. Liu, K. Igbal, R. Tohme, U. Sharapov, B.A. Kupronis, J.W. Ward, and S.D. Holmberg. 2014. Emerging epidemic of hepatitis C virus infections among young nonurban persons who inject drugs in the United States, 2006–2012. Clinical Infectious Diseases 59(10):1411-1419.
Terman, G. 2012. Naloxone: Effects and side effects. Paper presented at Role of Naloxone in Opioid Overdose Fatality Prevention; Request for Comments; Public Workshop, Silver Spring, MD.
Terplan, M., A. Kennedy-Hendricks, and M. Chisolm. 2015. Prenatal substance use: Exploring assumptions of maternal unfitness. Substance Abuse 9(Suppl. 2):1-4.
Thomas, C.P., C.A. Fullerton, M. Kim, L. Montejano, D.R. Lyman, R.H. Dougherty, A.S. Daniels, S.S. Ghose, and M.E. Delphin-Rittmon. 2014. Medication-assisted treatment with buprenorphine: Assessing the evidence. Psychiatric Services 65(2):158-170.
Traynor, K. 2016. Maine enacts statewide limits on opioid prescribing. American Journal of Health System Pharmacy 73(12):854-856.
Uchtenhagen, A. 2010. Heroin-assisted treatment in Switzerland: A case study in policy change. Addiction 105(1):29-37.
Unick, G.J., D. Rosenblum, S. Mars, and D. Ciccarone. 2013. Intertwined epidemics: National demographic trends in hospitalizations for heroin- and opioid-related overdoses, 1993–2009. PLoS One 8(2):e54496.
Vadivelu, N., A.M. Kai, V. Kodumudi, R. Zhu, and R. Hines. 2017. Pain management of patients with substance abuse in the ambulatory setting. Current Pain and Headache Reports 21(2):9.
Vestal, C. 2016. At Rikers Island, a legacy of medication-assisted opioid treatment. Stateline: The Pew Charitable Trusts. http://www.pewtrusts.org/en/research-and-analysis/blogs/stateline/2016/05/23/at-rikers-island-a-legacy-of-medication-assisted-opioid-treatment (accessed February 27, 2017).
Villapiano, N.L.G., T.N.A. Winkelman, K.B. Kozhimannil, M.M. Davis, and S.W. Patrick. 2017. Rural and urban differences in neonatal abstinence syndrome and maternal opioid use, 2004–2013. JAMA Pediatrics 171(2):194-196.
Volkow, N.D., and A.T. McLellan. 2016. Opioid abuse in chronic pain—Misconceptions and mitigation strategies. New England Journal of Medicine 374:1253-1263.
Volkow, N.D., T.R. Frieden, P.S. Hyde, and S.S. Cha. 2014. Medication-assisted therapies—tackling the opioid-overdose epidemic. New England Journal of Medicine 370(22): 2063-2066.
Wachman, E.M., P.K. Newby, J. Vreeland, J. Byun, A. Bonganzi, and H. Baucher. 2011. The relationship between maternal opioid agonists and psychiatric medications on length of hospitalization for neonatal abstinence. Journal of Addiction Medicine 5(4):293-299.
Wachman, E.M., M.J. Hayes, M.S. Brown, J. Paul, W.K. Harvey, N. Terrin, G.S. Huggins, J.V. Aranda, and J.M. Davis. 2013. Association of OPRM1 and COMT single-nucleotide polymorphisms with hospital length of stay and treatment of neonatal abstinence syndrome. Journal of the American Medical Association 309(17):1821-1827.
Wachman, E.M., M.J. Hayes, B.M. Lester, N. Terin, M.S. Brown, D.A. Neilson, and J.M. Davis. 2014. Epigenetic variation in the mu-opioid receptor gene in infants with neonatal abstinence syndrome. Journal of Pediatrics 165(3):472-478.
Wachman, E.M., M.J. Hayes, R. Sherva, M.S. Brown, J.M. Davis, L.A. Farrer, and D.A. Nielsen. 2015. Variations in opioid receptor genes in neonatal abstinence syndrome. Drug and Alcohol Dependence 155:253-259.
Walley, A.Y., Z. Xuan, H.H. Hackman, E. Quinn, M. Doe-Simkins, A. Sorensen-Alawad, S. Ruiz, and A. Ozonoff. 2013. Opioid overdose rates and implementation of overdose education and nasal naloxone distribution in Massachusetts: Interrupted time series analysis. British Medical Journal 346:f174.
Walsh, S.L., P.A. Nuzzo, S. Babalonis, V. Casselton, and M.R. Lofwall. 2016. Intranasal buprenorphine along and in combination with naloxone: Abuse liability and reinforcing efficacy in physically dependent opioid abusers. Drug and Alcohol Dependence 162:190-198.
Webster, L.R., and R.M. Webster. 2005. Predicting aberrant behaviors in opioid-treated patients: Preliminary validation of the Opioid Risk Tool. Pain Medicine 6(6):432-442.
Weiss, R.D., J.S. Potter, D.A. Fiellin, M. Byrne, H.S. Connery, and W. Dickenson. 2011. Adjunctive counseling during brief and extended buprenorphine-naloxone treatment for prescription opioid dependence: A 2-phase randomized controlled trial. Archives of General Psychiatry 68:1238-1246.
West, N.A., S.G. Severtson, J.L. Green, and R.C. Dart. 2015. Trends in abuse and misuse of prescription opioids among older adults. Drug and Alcohol Dependence 149:117-121.
Wheeler, E., T. Jones, M. Gilbert, and P. Davidson. 2015. Opioid overdose prevention programs providing naloxone to laypersons—United States, 2014. Morbidity and Mortality Weekly Report 64(23):631-635.
WHO (World Health Organization). 2003. Introduction to drug utilization research. http://apps.who.int/medicinedocs/pdf/s4876e/s4876e.pdf (accessed March 1, 2017).
WHO. 2009. Guidelines for the psychosocially assisted pharmacological treatment of opioid dependence. http://www.who.int/substance_abuse/publications/opioid_dependence_guidelines.pdf (accessed February 27, 2017).
WHO. 2015. WHO model list of essential medicines. 19th list (April 2015) (amended November 2015). http://www.who.int/medicines/publications/essentialmedicines/en (accessed March 1, 2017).
Wiegand, S.L., E.M. Stringer, A.M. Stuebe, H.E. Jones, C. Seashore, and J. Thorp. 2015. Buprenorphine and naloxone comparison with methadone treatment in pregnancy. Obstetrics and Gynecology 125(2):363-368.
Wilson, M.E., R.P. Schwartz, K.E. O’Grady, and J.H. Jaffe. 2010. Impact of interim methadone maintenance on HIV risk behaviors. Journal of Urban Health 87(4):586-591.
Wolfe, D., M.P. Carrieri, N. Dasgupta, A. Wodak, R. Newman, and R.D. Bruce. 2011. Concerns about injectable naltrexone for opioid dependence. Lancet 377(9776):1468-1470.
Woody, G.E., S.A. Poole, G. Subramaniam, K. Dugosh, M. Bogenschutz, P. Abbott, A. Patkar, M. Publicker, K. McCain, J.S. Potter, R. Forman, V. Vetter, L. McNicholas, J. Blaine, K.G. Lynch, and P. Fudala. 2008. Extended vs short-term buprenorphine-naloxone for treatment of opioid-addicted youth: A randomized trial. Journal of the American Medical Association 300(17):2003-2011.
WSIN (Western States Information Network). 2016. Drug price and purity guide, 2016. Sacramento, CA: WSIN.
Yang, Z., B. Wilsey, M. Bohm, M. Weyrich, K. Roy, D. Ritley, C. Jones, and J. Melnikow. 2015. Defining risk of prescription opioid overdose: Pharmacy shopping and overlapping prescriptions among long-term opioid users in Medicaid. The Journal of Pain 16(5):445-453.
Yealy, D.M., P.M. Paris, R.M. Kaplan, M.B. Heller, and S.E. Marini. 1990. The safety of prehospital naloxone administration by paramedics. Annals of Emergency Medicine 19(8):902-905.
Zaller, N., M. McKenzie, P.D. Friedmann, T.C. Green, S. McGowan, and J.D. Rich. 2013. Initiation of buprenorphine during incarceration and retention in treatment upon release. Journal of Substance Abuse Treatment 45(2):222-226.
Zedler, B., L. Xie, L. Wang, A. Joyce, C. Vick, F. Kariburyo, P. Rajan, O. Baser, and L. Murrelle. 2014. Risk factors for serious prescription opioid-related toxicity or overdose among Veterans Health Administration patients. Pain Medicine 15(11):1911-1929.
Zibbell, J.E., K. Iqbal, R.C. Patel, A. Suryaprasad, K.J. Sanders, L. Moore-Moravian, J. Serrecchia, S. Blankenship, J.W. Ward, and D. Hotzman. 2015. Increases in Hepatitis C virus infection related to injection drug use among persons aged ≤30 Years—Kentucky, Tennessee, Virginia, and West Virginia, 2006–2012. Morbidity and Mortality Weekly Report 64(17):453-458.
This page intentionally left blank.
















































































