







Below is the uncorrected machine-read text of this chapter, intended to provide our own search engines and external engines with highly rich, chapter-representative searchable text of each book. Because it is UNCORRECTED material, please consider the following text as a useful but insufficient proxy for the authoritative book pages.
10 ASPHALT PRODUCTION According to the National Asphalt Pavement Association, of the 2.6 million miles of paved roads and highways in the United States, approximately 93% are paved with petroleum-based asphalt. Asphalt binder supplies are shrinking, as only a limited number of crude oils yield quality asphalt. The price of liquid asphalt has fluctuated dramatically within the past decade, ranging from $200 per ton in 2005 to $560 per ton in 2012. This is mainly because of the increasing cost of crude oil and the advent of coking technologies, which enabled refineries to decrease their production of asphalt (residua from crude oil refin- ing processes) by converting it into synthetic fuel. This development has led to a shortage of high-quality asphalt binders and an increase in their price independent of the crude oil price. U.S. refiners are running at maximum capacity (>90%) and no new refineries are planned in the near term. Existing refin- ery expansions must fill any gap but the expansion will focus on handling increased supplies of heavy crude oil and their con- version capabilities to meet light product demand. Early refineries were simply distillation units that processed the material boiling above 550°C and left approximately 40% of heavy crudes as residue (residuum), which was primarily sold as asphalt. The addition of various hydrocracking units allowed further processing of the residuum to produce more light products. These additions reduced the typical asphalt yield to 12%â15%. Currently, refineries are adding coker units that will convert the residuum to fuel oil and coke, and no asphalt is produced as a by-product. A refiner can decide whether to produce asphalt based on the relative price differential between light products and asphalt prices, the âcrack spread.â As the price of gasoline increases the resultant crack spread makes the production of asphalt uneconomical unless there is a corresponding increase in asphalt prices. Thus, asphalt can no longer be considered a by-product, but now it is a product that can be produced based on the market demand. The price of asphalt must keep pace with conversion feed values to encourage its production. In addition to the absolute price of crude oil, the production of asphalt is influenced by the light/heavy crude price differential, the light product crack spread, coking economics, and heavy crude availability. More heavy crude is becoming available and not all refineries have installed coker units, so the asphalt supply is ensured at the right price. A survey of asphalt usage prepared by the Asphalt Institute, based on responses from its 54 members, is summarized in Table 2. The total amount of asphalt used in 2013 was 19.1 million tons, of which 16.3 million tons went to paving applications. The categories of asphalt usages cited in the table are as follows: Asphalt cement (AC): A solid or semisolid asphalt that has not been modified by the addition of a low or intermediate boiling range solvent, emulsification, or the addition of inorganic fillers. It has sufficient quality and consistency for direct use in the manufacture of bituminous pavements, roofing materials, or other industrial products. Outside the United States, the product is often called bitumen. Modified asphalt cement: An AC to which a performance-improving modifier and/or additive has been blended. Com- monly used modifiers include elastomers and plastics (polymers). Other additives include fillers, extenders, fibers, oxidants, antioxidants, hydrocarbons, polyphosphoric acid, and combinations. Normally, modifiers will improve the base ACâs grade by at least one level. Asphalts that are changed by the refining process, such as oxidation, are not considered modified for the purposes of this survey. Cutback asphalt (slow, medium, or rapid curing): Asphalt products produced by blending an asphalt with solvents such as naphtha, kerosene, No. 2 fuel oil, diesel oil, or other volatile solvents. Upon exposure to the atmosphere, the volatile product evaporates, leaving the asphalt. Also included under this heading are road oils, including residual asphalt oils, used as a dust palliative or other surface treatments of pavements. CHAPTER TWO LITERATURE REVIEW: CHEMICAL AND PHYSICAL CHARACTERIZATION OF ASPHALT BINDERS
11 TABLE 2 2013 U.S. ASPHALT USAGE IN SHORT TONS BY PADD DISTRICT PADD Districts Asphalt Cement % of Paving Asphalts Modified Asphalt Cement % of Paving Asphalts Cutback Asphalt Emulsified Asphalt % of Paving Asphalts Total Paving Asphalt Nonpaving Asphalt Total Asphalt Usage PADD I East Coast 4,541,850 88 377,833 7 5,085 222,109 4 5,146,877 897,320 6,044,197 PADD II Midwest 4,415,858 72 872,041 14 93,860 753,877 12 6,135,636 694,320 6,829,956 PADD III Gulf Coast 1,599,298 69 355,128 15 63,906 306,437 13 2,324,769 863,677 3,188,446 PADD IV Rocky Mountain 239,969 44 153,026 28 6,402 140,210 26 539,607 5,578 545,185 PADD V West Coast 1,771,847 83 175,720 8 37,589 152,917 7 2,138,073 387,285 2,525,358 Total U.S. tonnage 12,568,822 77 1,933,748 12 206,842 1,575,550 10 16,284,962 2,828,180 19,113,142 Source: This work compiled from www.eia.gov data. Emulsified asphalt: Emulsified asphalts consist of asphalt and water processed with emulsifying agents to produce a stable suspension of minute globules of asphalt in water; or alternatively, a suspension of minute globules of water in liquid asphalt. The listed figures are an estimate of the asphalt content and should not include water and other liquid additives that typically range between 30% and 45% by weight of mixture. Emulsified asphalts may be either anionic or cationic emulsions. Asphalt materials for nonpaving applications, primarily asphalt cements, emulsions, and fluxes used in the manufacture of roofing asphalts. Changes in geographic usage can be assessed when the usage is reported by Petroleum Administration for Defense Districts (PADDs) as defined by the U.S. Department of Energy. The PADD concept is used today for data collection purposes and to aid in understanding of supply and demand of domestic petroleum products. Figure 1 shows the current PADD delineation. FIGURE 1 Petroleum Administration for Defense Districts (PADDs) (Source: U.S. Energy Information Administration: http://www.eia.gov/petroleum/supply/monthly/pdf/append.pdf).
12 It is well understood that crude oil sourcing for U.S. refineries varies over time, but in general it can be assumed that the sources for a given PADD are consistent. PADD 1 refineries process crude oil shipped from all over the world, including Saudi Arabia and Venezuela. PADD 2 and PADD 4 depend primarily on crude oil produced and moved by pipeline from Canada supplemented by crude from PADD 3 as well as production from Rocky Mountain state sources. PADD 3 is the largest refin- ing region and obtains crude oil from the Gulf Coast outer continental shelf, Mexico, Venezuela, and the rest of the world. Currently, permitting issues stalling construction of the Keystone pipeline are forcing rail transport of Canadian syncrude (from oil sands) to PADD 3 refineries. PADD 5 obtains crude oil primarily from Alaska (by tanker) and California (Kern River Valley), and through imports. Data compiled in 2013 are summarized in Table 2. Asphalt cement constitutes more than 80% of the paving asphalt installed in the East and West Coasts districts. Modified asphalts and emulsified asphalts are used more extensively in the Midwest, Gulf Coast, and Rocky Mountain districts. Overall, 77% of the total tonnage applied is asphalt cement, 12% is modi- fied asphalts, and 10% is emulsified asphalts. Data from the U.S. Energy Information Administration show the trends in net production of asphalt and road oil by refiner- ies and blenders. The data are reported in annual thousand barrels of asphalt, so it is difficult to compare production directly with the asphalt usage reported in tons above. However, the trends in production can be observed in Figure 2. In the past 6 years, maximum production occurred in 2010. Asphalt production decreased steadily through 2014, but the trend was reversed in 2015. The largest increase in production occurred in the Louisiana Gulf Coast region of PADD 3. FIGURE 2 Refinery and blender net production of asphalt and road oil (Source: U.S. Energy Information Administration. This work was compiled from data at http://www.eia.gov/petroleum/supply). According to a study published by the Freedonia Group, demand for asphalt in paving applications is forecast to advance 3.1% annually through 2019, to 19.6 million tons, based on improving economic conditions and a pressing need to repair and expand the nationâs infrastructure. Among asphalt products, asphalt emulsions will see the fastest growth, boosted by interest in on-site recycling since asphalt emulsions can be used to recycle older pavements while minimizing asphalt consumption. Asphalt cement will remain the leading paving material used in the United States owing to the prevalence of hot-mix and warm-mix asphalts in paving jobs. These products are favored because of their moderate cost and solid performance proper- ties, such as durability and good drainage (Freedonia Group 2015). ASPHALT COMPOSITION The terms asphalt and bitumen are often used interchangeably to mean both natural and manufactured forms of the substance. Asphalt (or asphalt cement) is the carefully refined product derived from selected crude oils. Outside the United States, the prod- uct is often called bitumen. Asphalt is no longer just the residua from crude oil refining. It is now known as an asphalt binder and is part of an engineered system. For asphalt that is less suitable for paving applications, modifiers, such as polymers, adhesion
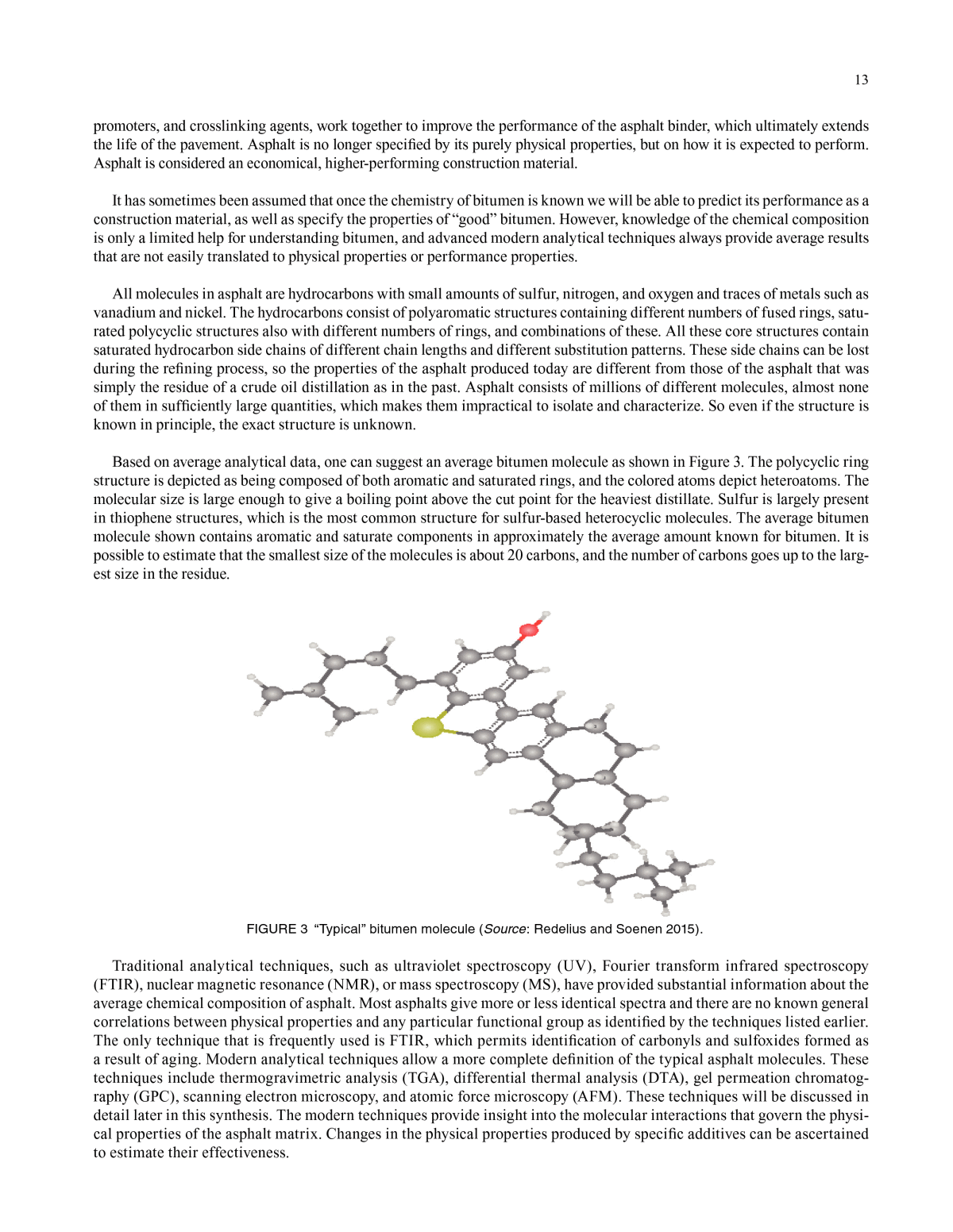
13 promoters, and crosslinking agents, work together to improve the performance of the asphalt binder, which ultimately extends the life of the pavement. Asphalt is no longer specified by its purely physical properties, but on how it is expected to perform. Asphalt is considered an economical, higher-performing construction material. It has sometimes been assumed that once the chemistry of bitumen is known we will be able to predict its performance as a construction material, as well as specify the properties of âgoodâ bitumen. However, knowledge of the chemical composition is only a limited help for understanding bitumen, and advanced modern analytical techniques always provide average results that are not easily translated to physical properties or performance properties. All molecules in asphalt are hydrocarbons with small amounts of sulfur, nitrogen, and oxygen and traces of metals such as vanadium and nickel. The hydrocarbons consist of polyaromatic structures containing different numbers of fused rings, satu- rated polycyclic structures also with different numbers of rings, and combinations of these. All these core structures contain saturated hydrocarbon side chains of different chain lengths and different substitution patterns. These side chains can be lost during the refining process, so the properties of the asphalt produced today are different from those of the asphalt that was simply the residue of a crude oil distillation as in the past. Asphalt consists of millions of different molecules, almost none of them in sufficiently large quantities, which makes them impractical to isolate and characterize. So even if the structure is known in principle, the exact structure is unknown. Based on average analytical data, one can suggest an average bitumen molecule as shown in Figure 3. The polycyclic ring structure is depicted as being composed of both aromatic and saturated rings, and the colored atoms depict heteroatoms. The molecular size is large enough to give a boiling point above the cut point for the heaviest distillate. Sulfur is largely present in thiophene structures, which is the most common structure for sulfur-based heterocyclic molecules. The average bitumen molecule shown contains aromatic and saturate components in approximately the average amount known for bitumen. It is possible to estimate that the smallest size of the molecules is about 20 carbons, and the number of carbons goes up to the larg- est size in the residue. FIGURE 3 âTypicalâ bitumen molecule (Source: Redelius and Soenen 2015). Traditional analytical techniques, such as ultraviolet spectroscopy (UV), Fourier transform infrared spectroscopy (FTIR), nuclear magnetic resonance (NMR), or mass spectroscopy (MS), have provided substantial information about the average chemical composition of asphalt. Most asphalts give more or less identical spectra and there are no known general correlations between physical properties and any particular functional group as identified by the techniques listed earlier. The only technique that is frequently used is FTIR, which permits identification of carbonyls and sulfoxides formed as a result of aging. Modern analytical techniques allow a more complete definition of the typical asphalt molecules. These techniques include thermogravimetric analysis (TGA), differential thermal analysis (DTA), gel permeation chromatog- raphy (GPC), scanning electron microscopy, and atomic force microscopy (AFM). These techniques will be discussed in detail later in this synthesis. The modern techniques provide insight into the molecular interactions that govern the physi- cal properties of the asphalt matrix. Changes in the physical properties produced by specific additives can be ascertained to estimate their effectiveness.
14 FTIR is one of the more important methods for fingerprinting asphalt materials and quantifying the distribution of asphalt components. By determining the various chemical functional groups in the binder, an understanding of its origin and history can be obtained. The FTIR method is an efficient technique for identifying polymer additives in a binder. Determination of polymer content is essential for quality control and quality assurance during the processing and application of polymer- modified asphalts (PMAs). TGA and differential scanning calorimetry (DSC) are used to characterize petroleum bitumens and their chromatographic fractions, including the glass transition temperature and the percentage of crystalline phases. The advent of modulated DSC provides new insight on asphalt microstructure. The development of bitumen microstructure and the calculations of the entropy and enthalpy of transitions suggest that bitumen is a structured amorphous phase containing a small crystalline phase. Proton nuclear magnetic resonance (1H NMR) spectroscopy has emerged as a very powerful and versatile tool for bitumen characterization. Using 1H and 13C NMR can yield information on average structural parameters of asphalt and asphaltenes, such as percentages of aromatic carbons, aliphatic carbons, bridged carbons, methyl carbons, ring carbons, naphthenic car- bons, paraffinic chain lengths, and other parameters. The specific environment of the different types of hydrogens and carbons can be defined (Betancourt Cardozo et al. 2016). GPC is a separation method that takes advantage of differences in the size of molecular structures of asphalt components. The procedure separates these components based on their molecular size (hydrodynamic volume). The polymer and asphalt components of polymer-modified asphalt cements can be separated completely. GPCâs ability to separate mixtures by molecu- lar size rather than by some complex property such as solubility or absorptivity is one of the great advantages of the technique. This feature has made GPC a useful alternate technique for fractionating complicated mixtures such as crude oil residua, asphalts, and asphaltenes and a vital contributor to understanding asphalt mixes (Altgelt 1965; Dickie and Yen 1967; Snyder 1969; Yapp et al. 1991). The chromatograms are used to predict aging viscosity, aging index, viscosity number, and penetra- tion. GPC is an excellent tool for measuring polymer content in modified binders on a routine basis. When NMR and GPC information is combined, possible structures for asphalt and mechanisms of aging can be suggested. One school of thought is that asphalt binder may be considered as a colloidal or micellar system. The hydrocarbon insoluble components, asphaltenes and resins, are dispersed in a hydrocarbon blend. Many different types of molecular interactions contribute to the stability of the dispersion. Because asphalt consists of nonpolar hydrocarbons, the dominating interactions are London dispersive interactions resulting from temporary dipoles caused by variations in the electrons surrounding the molecules. Also contributing are polar interactions and hydrogen bonding interactions that are due to the content of the more electronegative elements nitrogen and oxygen. There are also piâpi interactions, which for nonpolar molecules involve aro- matic components of the molecules. Quantification of these interactions is difficult since the moleculesâ exact structure is not known. Each type of interaction occurs in concert with the other types of interactions described previously. The presence of a solvent may exert a strong effect on the relative strength of the interactions (Redelius and Soenen 2015). The overall behavior of asphalt cement is controlled by the compatibility and the relationships of the different components in this macroscopically homogeneous mixture rather than by the quantitative amount of any single component (Petersen 1984). ASPHALTENE STRUCTURES Asphaltenes are the insoluble fraction precipitated from a toluene solution of asphalt binder by a nonpolar solvent such as pentane or heptane. The precipitating solvent affects the quantity of asphaltenes precipitated. The nature of the asphaltene molecules in the precipitate depends on the precipitating solvent; greater amounts of a precipitant are produced by pentane. In general, the asphaltenes are defined as the mixture of materials precipitated by heptane. The asphaltene molecules are very complex and exhibit a very high tendency to associate into molecular clusters. The amount and characteristics of asphaltenes vary from one asphalt source to another. They play a significant role as viscosity builders in the rheology of asphalt binders. The composition of an asphaltene fraction has been the subject of debate for the past 30 years (Petersen 1984; Goodrich et al. 1986). The complexity of the fraction and the tendency for the asphaltene molecules to associate has stimulated an extensive debate on their structures. Virtually all asphaltene chemical properties, except elemental composition, had been the subject of debate; their molecular weight (MW) had been estimated at values spanning six orders of magnitude (Mullins et al. 2008; Yarranton et al. 2013). MW determination has been, and still is, a challenging problem in asphaltene chemistry. The source of complications is attributable to three basic properties of asphaltene, namely, compositional variance, size polydis- persity, and, most important, the high propensity of the covalent asphaltene molecules to form molecular aggregates (Strausz
15 et al. 2008). A whole gamut of methods has been applied for the MW determination of asphaltene over the decades, including chemical, physical, absolute, relative, equilibrium, and nonequilibrium methods, but a consensus of the appropriate MW value has not been reached. Nevertheless, an understanding of asphaltenes is essential because they have a significant impact on many physical and chemical properties of crude oils and asphalts. Many diverse studies have converged on a simple, hierarchical model of the molecular and colloidal structure of asphaltenes: the modified Yen model (also known as the YenâMullins model). The structural hierarchy in Figure 4 shows the extent of aggregation of the polycyclic aromatic hydrocarbon (PAH), which is the asphaltene monomer. The predominant asphaltene molecular architecture contains a single, moderately large PAH with peripheral alkanes (~1.5 nm). Asphaltene molecules form nanoaggregates with aggregation numbers of approximately six, with a single disordered PAH stack (~2 nm). These asphaltene nanoaggregates can form clusters with aggregation numbers of approximately eight (5 nm). FIGURE 4 The modified Yen model (or the YenâMullins model): polycyclic aromatic hydrocarbon monomer (left), nanoaggregate (center), and clusters (right) (Source: Mullins 2011). It was unknown whether asphaltene molecules contain predominantly one PAH (the island architecture) or many cross- linked PAHs (the archipelago architecture) (Strausz et al. 1992). Molecular diffusion measurements, especially from time- resolved fluorescence depolarization, helped to resolve these molecular properties of asphaltenes and provide evidence for the island molecular architecture. Petroleum asphaltenes consist of approximately 40% to 45% aromatic carbon; the remainder is aliphatic, as shown by 13C NMR. Alkane chains are an average of four to five carbons long, as has been determined by integrated infrared (IR) spectroscopy and NMR studies. Observations of active hydrogen in asphaltene (Gould and Wiehe 2007) show that naphthenic rings are fused to aromatic rings. The aromatic ring system is also where almost all the nitrogen (~1% by mass) is located, as shown by X-ray absorp- tion near-edge structure; both basic pyridinic nitrogen and acidic pyrrolic nitrogen are present (Mitra-Kirtley et al. 1997). Sulfur X-ray absorption near-edge structure shows that most of the several percent sulfur consists of thiophene- and sulfide-type groups and generally a small fraction of sulfoxide (George and Gorbaty 1989). The proposed PAH asphaltene monomers shown in Figure 5 have an MW of 750 Da, and a single fused aromatic ring system per molecule (island molecular architecture) (Mullins 2011). FIGURE 5 Typical asphaltene molecular architecture (Source: Mullins 2011).
16 A variety of experimental techniques were applied to a single source asphaltene sample at the same experimental condi- tions to reveal the possible size distributions of asphaltene monomers and aggregates (Yarranton et al. 2013). The asphaltene sample was divided into solubility cuts by selective precipitation in a solution of toluene diluted with various concentrations of heptane. Asphaltene self-association was assessed through a combination of density, vapor pressure osmometry (VPO), elemental analysis, Fourier transform-ion cyclotron resonance mass spectrometry, and time-resolved fluorescence emission spectra measurements performed on each cut. The physical dimensions of the asphaltenes were assessed using small angle X-ray scattering (SAXS), dynamic light scattering (DLS), membrane diffusion, Rayleigh scattering, and nanofiltration mea- surements. Molecular and nanoaggregate dimensions were also investigated through a combination of interfacial tension, interfacial adsorption, and surface force measurements. All of the measurements indicated that approximately 90 weight percent of the asphaltenes are self-associated. Ultrahigh resolution spectrometry suggests that the nonassociated asphaltenes are smaller and more aromatic than bulk asphaltenes, indicating that the associating species are larger and less aromatic. On the basis of VPO, the average monomer MW was approximately 850 g/mole, while the MW of the nanoaggregates spanned a range of at least 30,000 g/mole with an average on the order of 10,000 to 20,000 g/mol. SAXS and DLS gave MWs 10 times larger. However, these techniques are known to be unreliable for asphaltene MW measurement (Mullins et al. 2008). The discrepancies demonstrate that the final judgment on asphaltene structure remains in doubt. The physical dimensions of the nanoaggregates were less than 20 nm based on nanofiltration and with average diameters of 5 to 9 nm based on diffusion and Rayleigh scattering. SAXS and DLS showed average diameters of 14 nm and indicated that the nanoaggregates had loose structures. Film studies were consistent with the lower MWs and dimensions. The asphaltene monolayers swell by a factor of 4 in the presence of a solvent. The most consistent interpretation of the data is that asphaltenes form a highly polydisperse distribution of loosely structured (porous or low fractal dimension) nanoaggregates (Yarranton et al. 2013). ASPHALT MORPHOLOGY Lesueur has published a detailed review of the current asphalt structure concepts that defines links between chemistry, structure, and mechanical properties in a framework of an updated colloidal picture of asphalt (Lesueur 2009). Asphalt is considered a colloidal dispersion of asphaltene micelles in the maltenes. The resinsâthat is, the polar components of the maltenesâwere thought to stabilize the asphaltene micelles. Extensive analytical procedures support this model. A more pre- cise description of this process is starting to arise. SAXS and small angle neutron scattering (SANS) confirm that asphaltenes form micelles in organic solvents in asphalt (Yen 1992). The diffusion pattern observed in SAXS or SANS experiments disap- peared once the asphaltenes were removed from the asphalt. The colloidal model is also consistent with results obtained by thermal analysis. Asphalt undergoes a glass transition at a temperature very close to that of its aromatics moieties. This strongly suggests that the asphaltenes exist as dispersed solid particles and do not directly participate in the glass transition. The asphaltenes extend the span of the glass transition range, implying that at least some molecules of this family might contribute to the glass transition when mixed with maltenes (Claudy et al. 1992a). Recently researchers combined atomic resolution imaging using atomic force microscopy and molecular orbital imaging using scanning tunneling microscopy to study more than 100 asphaltene molecules derived from coal and petroleum (Schuler et al. 2015). The complexity and range of asphaltene polycyclic aromatic hydrocarbons are established in detail. Aromatic hydrocarbon (PAH) moieties of asphaltene comprise the primary site of intermolecular interaction in the asphalt colloids. The petroleum asphalt shown in Figure 6 is from a single crude oil. Sample PA1 has a large PAH core with two side chains attached. PA2 shows a PAH that has a side chain of about 20 Ã in length, The PAH in PA3 contains three five-membered rings, which are identified in the image. The asphaltene molecules consist of a central aromatic core with peripheral alkane chains. In some cases, this central core is divided into several distinct PAHs connected by a single bond, which proves the presence of archipelago-type molecules. Nevertheless, a single aromatic core with peripheral alkanes is the dominant asphaltene molecu- lar architecture, proving the main aspects proposed by the YenâMullins model (Mullins 2011). All this evidence makes it difficult to deny the colloidal nature of bitumen. The most convincing evidence is that which comes from diffusion experiments. Scattering techniques highlighted an elementary structure consisting of diffusive particles with a of radius 2â8 nm. Such particle size is reminiscent of the crystal size of âpureâ asphaltenes observed by
17 AFM in a pure petroleum fraction and concurs with the earlier description of asphaltene micelles as being made of a few individual molecules. FIGURE 6 Atomic force microscopy (AFM) Laplace filtered images of petroleum asphaltenes (Source: Schuler et al. 2015). Identifying molecular structures provides a foundation for understanding all aspects of petroleum science from colloi- dal structure and interfacial interactions to petroleum thermodynamics, enabling a first-principles approach to optimizing resource utilization. Particularly, the findings contribute to a long-standing debate about asphaltene molecular. architecture. AFM can be a powerful complementary tool to rheology and spectroscopy for characterizing asphalts. It makes it possible to compare the mechanical properties of pure and modified bitumen. The contributions of the AFM studies to the understanding of asphalt morphologies will be detailed in the section on instrumental analysis. When polymers are added to binders, the equilibrium situation is a macroscopic phase separation of the two phases (Lesueur 2009). The polymer is swollen by the light aromatic components from the parent bitumen, and the polymer-rich phase (PRP) occupies between 4 to 10 times the volume of added polymer, especially for styrene-butadiene diblock copolymer (SB) and ethylene-vinyl acetate copolymer (EVA). The continuous phase is an asphaltene-rich phase. Polymer/binder compat- ibility is indeed a dynamic concept; compatible systems are those with a slow phase separation (creaming rate). The creaming rate is a function of the relative densities of the microphases; the larger the density difference and the larger the phase sizes, the faster the creaming rate. To prevent phase separation, a few stabilization mechanisms are developed. Adding a crosslinking agent to the PMA under agitation allows the PMA to slightly crosslink, preventing PRP droplets from coalescing. The freez- ing of the equilibrium droplet size under agitation yields a very low particle size and favors stability. High crosslink density is desired because it would result in less swelling. Because each asphalt has its own particular chemical composition, ways to predict whether a particular polymer will be compatible with a given asphalt are not well defined, so the formulator usually relies on laboratory experiments rather than on theoretical predictions. In all cases, and even if the polymer has a potentially compatible chemistry, the formulation of PMAs requires knowledge of the chemical properties of the initial bitumen. Critical binder properties can be identified; that is, high asphaltene content decreases polymer/asphalt compatibility and the aromatic- ity of the maltenes needs to fall between certain values to reach a good level of compatibility. The importance of polyaromaticity on the elastic properties of bituminous binders was studied (Soenen and Redelius 2014). The size of the polyaromatic structures appears to play a crucial role. Observations indicate that larger conjugated aromatic structures provide stronger piâpi interactions between asphaltene stacks, which are known to be the main mecha- nism responsible for the formation of clusters at the nanoscale. The larger extent of these interactions relates to the elastic behavior at longer loading times or up to higher temperatures, while smaller aromatic structures determine more the elastic
18 behavior at short loading times or at low temperatures. In addition to aromatic interactions, natural wax can, upon crystalliza- tion, also induce increased elastic effects, especially at low frequencies. A large variety of binders was investigated: bitumen from straight distillation, visbreaking, solvent deasphalting, and oxidation were included in the sample set. Average levels of aromaticity were determined by FTIR and by refractive index measurements. Chromatography combined with UV-visible absorption spectroscopy was used as an indicator of the average size of the aromatic structures. Rheological properties were determined using a dynamic shear rheometer, in a temperature range from 0 up to 90°C. Good relationships were observed between the phase angle measurements, at specific test conditions of frequency and temperature, and ultraviolet-visible absorption levels at specific wavelengths (Soenen and Redelius 2014). Changes in the stacking behavior of asphaltene units are investigated in the presence of hexadecanamide, a representative amide-type additive. Molecular dynamics simulations and experiments using high-resolution transmission electron micros- copy and X-ray powder diffraction support results obtained from rigorous quantum mechanical calculations through a high quantum level of density functional-dispersion correction approach. Based on this multiscale bottom-up study, interaction of the amide-type binder with asphaltenes disturbs the uniformity of the Ï density throughout the aromatic region and creates some polarization in this region. This alteration of the Ï system over the aromatic zone disturbs the eventual piâpi interactions between asphaltene stacks. Disturbing these interactions alters the stacking distance, the corresponding binding energy, and ultimately the extent of clustering of asphaltene units. Any change in the clustering of asphaltene affects the rheology and morphological properties of asphalt, which in turn alters the asphaltâs performance, including but not limited to its resistance to fatigue and low-temperature cracking (Mousavi et al. 2016). A number of factors and variables influence the chemical composition and ultimately the engineering properties of a binder used in pavement applications. It is important that the users most closely working with the binders have a strong working knowledge of the relationship between binder chemistry and engineering properties. In addition, the relationship between standard process control actions and final chemical composition needs to be understood so that correlation to physical per- formance can be made.
