Speakers in the workshop’s third session explored existing technologies and new enabling technologies that could facilitate the efficient and cost-effective development of regenerative medicine therapy products that meet manufacturing and regulatory standards. Three panelists—Isabelle Rivière, the director of the cell therapy and cell engineering facility at Memorial Sloan Kettering Cancer Center; Rodney Rietze, the lead of current GMP (cGMP) process automation for cell and gene therapies at Novartis; and Philip Vanek, the general manager of cell therapy technologies at GE Healthcare—discussed opportunities for new technologies and manufacturing models that could increase efficiency and product quality, and they also considered novel and more precise technologies for in-process and final release testing. The panelists described the open and closed system models (i.e., systems in which cell cultures, respectively, are or are not exposed to the external environment during the manufacturing process) applicable to the manufacturing setting, and they discussed the feasibility of manufacturing therapies at the point of care. The discussion that followed focused on tools and models that could be developed and implemented to make the manufacturing process more efficient.
MODELS FOR MANUFACTURING CELL THERAPY PRODUCTS
Researchers at Sloan Kettering have helped develop a number of cell therapy platforms over the past 15 to 20 years, including CAR T cells for treating solid and hematologic cancers and platforms to genetically modify hematopoietic progenitor and stem cells for treating genetic deficiencies and cancer, Rivière said. More recently, she and her colleagues have worked on
producing cells derived from embryonic stem cells and iPSCs by differentiating them in various tissues for treating Parkinson’s disease and cancer. They also have experience with various genetic platforms for modifying embryonic stem cells and iPSCs that they are developing in collaborations and in consortia with academic and industry partners.
Focusing on her team’s experience manufacturing CAR T cells, Rivière described how they have taken advantage of the unit operations that are available for manufacturing these cells, breaking down the process into discrete steps, which has allowed them to develop a robust platform for manufacturing CAR T cells for a number of oncology indications (Hollyman et al., 2009; Przybylowski et al., 2006; Themeli et al., 2015), including acute lymphoblastic leukemia (see Figure 4-1 for the manufacturing process flow).
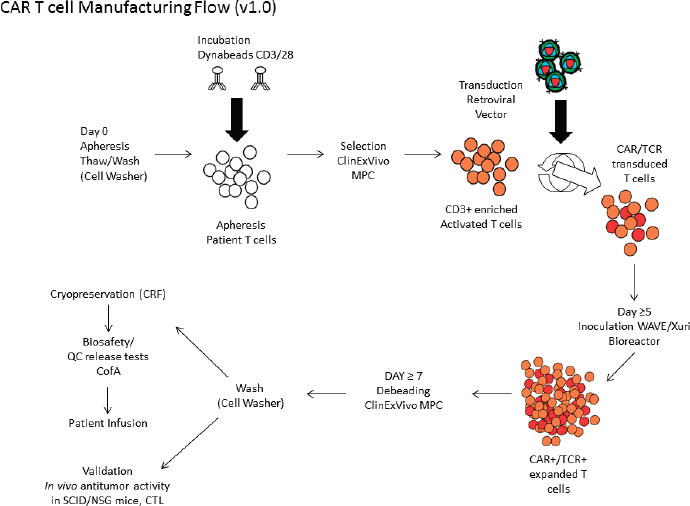
NOTE: CAR = chimeric antigen receptor; CofA = Certificate of Analysis; CRF = controlled rate freezer; CTL = cytotoxic T lymphocyte; MPC = magnetic particle concentrator; NSG = NOD scid gamma; QC = quality control; SCID = severe combined immunodeficiency; TCR = T cell receptor.
SOURCES: Isabelle Rivière, National Academies of Sciences, Engineering, and Medicine workshop presentation, June 26, 2017. Adapted from Figure 1: Scheme of the manufacturing process, in Themeli et al. (2015), and Figure 3: General schema of autologous T cell manufacturing in Hollyman et al. (2009).
One clinical trial using these CAR T cells been able to observe 77 to 90 percent of complete remission in patients with this disease (Park et al., 2016). Sloan Kettering researchers are conducting additional clinical trials in other leukemias, lymphomas, and solid tumors, and her team has used this process to produce more than 200 products, most of which have been infused into patients, she said. The team also uses this manufacturing scheme to make products for collaborators at other sites, and they have been able to transfer the manufactured products to their trial partners.
Potential Ways to Optimize the Manufacturing Process
Despite these successes, Rivière and her collaborators have encountered several challenges to maintaining the robustness of the manufacturing platform. Some of these challenges, she explained, are inherent to the apheresis products, given that hospitals perform this step on many different devices. Patient apheresis products also vary according to the specific disease, the stage of the disease, and any previous chemotherapy a patient has received. One approach the Sloan Kettering team uses to decrease the variability of the incoming apheresis product is to start by selecting cell types that will create a more homogeneous starting material; however, Rivière said, a current limitation is that the product developers have yet to define the active cells within the CD3, CD4, and CD8 populations. While selecting subsets of cells and removing undesirable cell types is important for streamlining the manufacturing process, she said, the limited number and high cost of GMP antibodies used to select the starting material limits the applicability of this approach.
Another issue that Rivière’s team has encountered is the limited availability of reagents that are not encumbered by intellectual property concerns. At one point her group had to change the magnetic beads used to activate T cells because another user had exclusive rights to use the beads in a commercial application. Eventually, Rivière’s team modified the process to eliminate the use of magnetic beads altogether, which had the added benefit of simplifying the manufacturing process. They are now validating the new process, which is proving to be a challenge because they do not know the precise identity of the active ingredients in their cell products. For validation, Rivière and her colleagues are using a well-calibrated in vivo antitumor activity model, but it is a slow and tedious approach to assess comparability. Her hope is to develop an in vitro model that can be used to assess comparability and accelerate their current process, given that her team is also refining and improving other steps in the manufacturing process.
Working with equipment providers to develop better tools for the manufacturing process is something that her team would be interested in
doing, Rivière said. For example, the cell washers her team uses perform well, but the current process requires different cell washers to achieve the various desired parameters. Available cell separation techniques that do not require GMP antibodies are expensive today and are limited in the scope of the phenotypes they can select, something that Rivière said she hopes equipment manufacturers will address soon. Other pieces of equipment have only one manufacturer, raising concerns as to whether they will remain in business or if issues arise in their production chain.
Rivière offered several other lessons she has learned from her experiences transferring the manufacturing process to industry. For example, providing patient data to industry partners that would allow them to improve processes and develop new assays would be helpful, but her team has learned that it can be difficult to provide patient samples to industry partners, Rivière said, particularly if the consent forms signed by the patients at the time of treatment do not include permission to transfer their materials to other institutions. At her center, the transfer to industry of manufacturing data for analysis is relatively limited because of this reason. Thinking about transfer protocols and the type of data that need to be collected early in the manufacturing process could help ensure a more smooth transition from academia to industry, she said. The reverse transfer of manufacturing technology back to academia is also challenging because of intellectual property issues as well as the cost and time in implementing these changes at an academic center, she said. If the transfer of data and processes between industry and academia could be facilitated during Phase I clinical trials, Rivière said, we could accelerate the development of cell therapies in a much more efficient manner.
THE ROLE OF AUTOMATION IN MANUFACTURING
Automation is one key to producing a safe, robust, reproducible, and cost-effective therapy, Rietze said. What automation provides is the ability to control the process by removing the variability between operators and lowering the risk associated with variability, which should lead to fewer manufacturing failures and, ultimately, an increased rate of success and lower costs, he said. Focusing on analytics in the manufacturing process provides manufacturers with a deep understanding of the process and the opportunity for continual improvement of that process, Rietze said, describing how his group at Novartis went about automating and commercializing the CAR T cell production process that Bruce Levine and his collaborators developed at the University of Pennsylvania.
Designing a scalable and robust manufacturing process starts with the science and with understanding the product, how it is produced, and what mechanisms are available to control variability. Next, Rietze said, manufac-
turers must identify the key technologies needed to make the product and determine if there are off-the-shelf devices available to automate or remove variability in certain steps in the process. For example, an automated cell-washing device could replace centrifugation and get rid of the variability that comes from human involvement. In thinking about developing a process, he said, one should keep in mind that emerging technologies can play a role in automation. Even though it may require an investment in time and resources to integrate and validate a new technology, that investment can pay off if the technology can be used in other processes and in multiple unit operations. When his group was working on the CAR T cell process, Rietze said, they targeted specific unit operations for optimization to generate value and lower risk, and they started rethinking some of the analytics with an eye toward what assays would work best in an automated system.
With the device selected, the next step is to think about the manufacturing facility. For example, automated closed systems allow for processing to occur in rooms with less stringent classifications thereby reducing the burden on the facility’s clean room and the need for biosafety cabinets, Rietze said. At the same time, automating one step in a process might eliminate the need for several individuals to perform a unit operation, but given how long it takes to leave and enter a clean room, workers might need something else to do while waiting to perform the next manual operation without entering and exiting the clean room, creating new challenges in workforce management. In the end, “the development and integration of automation changes your manufacturing, changes your process flow and your footprint, and it reduces your overhead and changes how you train your staff,” Rietze said. The ultimate goal of automation is to enable a global supply of a product by moving from a manufacturing facility that may require more space for equipment down to a small space, thereby improving the efficiency of the manufacturing process, and then ultimately moving toward a device capable of manufacturing these products in a point-of-care device, he added.
Rietze described the roadmap (see Figure 4-2) of how he and his team developed the commercial version of the academic CAR T cell process. The first step was to define the highest need for each unit operation, and he did that by surveying each of the key stakeholders in his company, including its process operators, its analytical group, and management. These survey questions fell in the areas of safety, process, product, and cost. From the answers, his team generated a heat map showing that the highest needs were at the beginning and end of the process.
At that point the Novartis team identified and engaged providers to develop a novel device that reflected stakeholder needs. They built a project team and defined a formal user requirement specification (URS), an important step that involves defining what the new automated devices need to do, the environment in which they will do it, and the criteria to judge
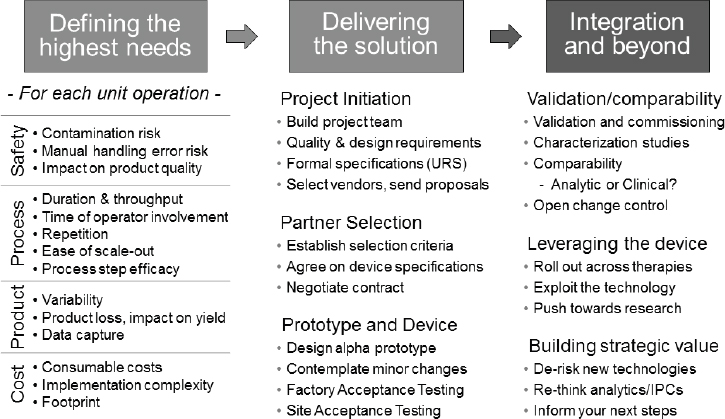
NOTE: IPC = in-process control; URS = user requirement specification.
SOURCE: Rodney Rietze, National Academies of Sciences, Engineering, and Medicine workshop presentation, June 26, 2017.
whether the devices are successful. The team sent the URS to a number of vendors, selected one, and then moved through the device-manufacturing process, which involved building alpha and beta prototypes and testing them in-house. The final step before the integration of the devices into the manufacturing process was to conduct product characterization studies to determine if the devices changed the product.
It is important to begin to use the device in early research (as new products are being developed for the product pipeline) to enable the use of the same system that will ultimately manufacture those products as a means of lowering the barrier for process transfer, Rietze said. They also rethought the analytics and in-process controls since the device was able to do the same operation reproducibly.
The eventual device that the Novartis team settled on was named FlowSPA (Flow Sample Prep Automation, Acquisition, and Analysis), and it has the ability to automate flow cytometry-based analytics. This device, made by combining pre-existing technologies, starts with a liquid-handling platform to which the team added a flow analyzer to automate data acquisition from the liquid handler. A liquid handler reduces the high variability of manual pipetting, which allowed the team to use smaller volumes of
antibodies and yielded more accurate analytics, Rietze said. The team used publicly available data to develop a computer-controlled gating process for the cytometer, which made it possible to automate the analysis of the cytometer data. The idea is that the automation is not only about the cost of goods or a reduction in throughput, but also about a reduction in hands-on time, increased quality, and an increased consistency and integrity of the data that are generated, Rietze said.
Automating the analytics also allowed the Novartis team to develop a more in-depth understanding of the product and of the patient material it receives. This, in turn, improved consistency across hundreds of patients, Rietze said. Analytic automation also creates the possibility of more closely integrating the manufacturing process and the clinical process by breaking down data silos, he said.
The field of cell-based therapies is evolving, as are the analytics and devices, Rietze said. This evolution is leading to shorter manufacturing timelines and non-destructive and in-process analytics that remove the need for humans to “touch” the product. People involved in the field should understand that the patients affect the cells they provide, which in turn affects the manufacturing process and the drugs that the patients ultimately receive.
SCALING PERSONALIZED CELL THERAPY MANUFACTURING
Tool providers, or companies who develop enabling technologies such as bioprocessing equipment for manufacturing cellular therapies, have a different perspective than therapy manufacturers, Vanek said, because they have to be agnostic as to cell type or therapy and instead build platforms with broad applicability. When building a tool, it is important to identify the needs of the end user, he added. For cell-based therapies, those needs include operational excellence, compliance, reproducibility, and the ability to make a safe and effective product. One challenge, he said, is that companies come to GE Healthcare and ask for very broad solutions for their problems that also leave open the possibility of optimizing each unit operation. Everyone wants a future technology today, Vanek said, and that is a challenge for tool providers.
Tool providers must also take into account how the market will likely evolve, Vanek said, so to help forecast where technology needs to be in order to keep up with that evolution, he and his colleagues conduct market analyses to understand where CAR T cell therapy and gene therapies are going. For example, he said, projections indicate that the number of patients treatable with these therapies will increase and that the cost per dose will have to come down from its current cost of goods of greater than $100,000 per dose. The key, he said, is to not focus on the cost per dose,
but rather focus on managing risk, biology, and data. “The rest will come with scale and better integration and automation,” he said. New and better therapies will be coming, and the number of patients treated with these technologies will increase, and both of these factors create challenges for tool providers. The demand for integration of analytics, automation, and embedded sensors as well as tools for data management and handoff also creates challenges for tool providers, he added.
Market research is key when defining the company’s investment priorities, Vanek said, explaining that “it takes years and a huge investment to bring new platforms to the market.” While U.S. programs will account for most of the new products, Asian and Pacific counties are expected to grow their cell therapy programs significantly, he said.
Refining investment priorities is important because it can take 3 to 7 years and $1 million to $25 million (or more) to take a new platform from idea to product (see Figure 4-3). Platform development occurs in three broad phases: research, early development, and late development and launch. The research phase, characterized by what he called an “innovation loop,” is a period when companies are hesitant to invest because it is costly and high risk. Late development and launch, and its product care loop, is when companies are more likely to spend money because development at that stage largely iterates on a given theme. Closing the gap requires that companies look at all of the derivatives of technology, such as company spinouts, new intellectual property, and out-licensing, that can fund the innovation loop.
An important piece of the development process is determining what drives the cost of each unit operation. The costs for reagents and consumables are predicted to fall as they move into large-scale production to meet growing demand, Vanek said, and that is an area in which GE Healthcare is investing. While capital expenditures for hardware do represent a cost to a business, consumables are the real drivers of the cost of a therapy because they are not produced at scale today, he said. Labor is also a big part of the cost today, but as the industry automates, simplifies, and combines unit operations, those costs are expected to fall. The lack of optimization of manufacturing capacity adds to the overall cost, too. Costs should fall as researchers improve the effectiveness of these therapies, which should reduce the number of cells needed per dose. A move from autologous to allogeneic cells, with the potential for manufacturing at scale, would also have a significant beneficial effect on cost, Vanek said.
Concerning the transition of manufacturing from the clinical scale to a commercial scale, there are times when it is easier to implement change, Vanek said, including early on when the cost of change is not that high. As a trial moves into Phase II or Phase III, the costs can be more substantial. His company’s philosophy is to develop technologies that are scalable so
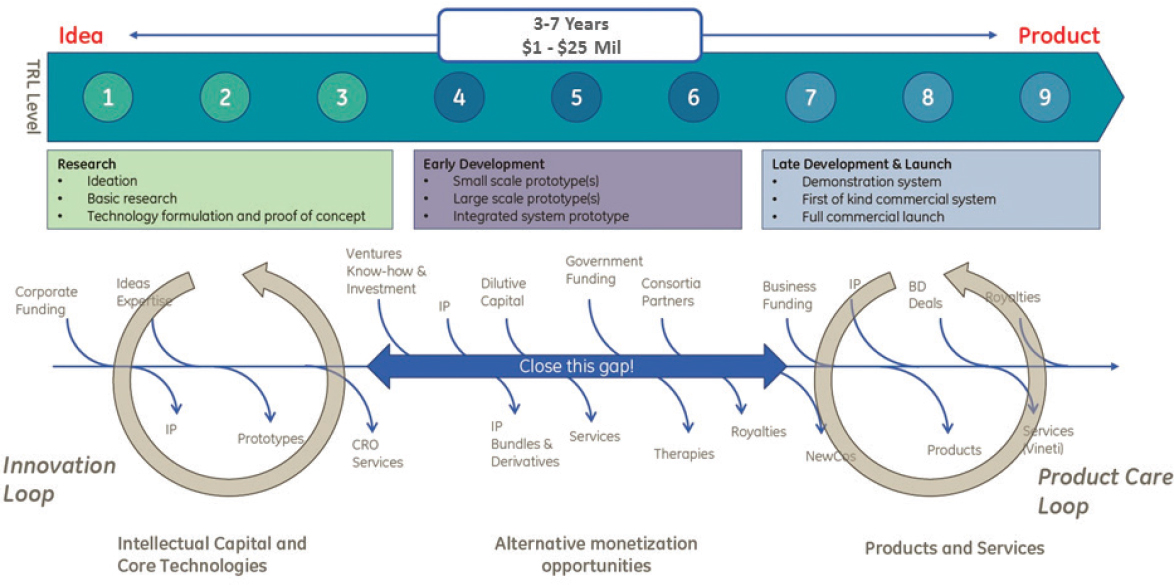
NOTE: BD = business development; CRO = contract research organization; IP = intellectual property; TRL = technology readiness level.
SOURCE: Philip Vanek, National Academies of Sciences, Engineering, and Medicine workshop presentation, June 26, 2017.
that they can be introduced early in the development process and grow in scale as demand increases, he said. By looking across the different therapies currently undergoing research and development, the company has tried to identify common subroutines of activity for which tools can be developed that would be translatable across the production of multiple types of therapies and at different scales. However, he acknowledged, that approach does not solve the problems of data portability and data integration.
While he is not a proponent of taking a complex, multistep process and oversimplifying it, Vanek said, he does see the need for some automation and simplification. At the end of the day, GE Healthcare either invents new technologies or acquires them from other inventors, he said, and in either case, data integration is still an issue. All of the components must communicate to make the complete process work. Toward that end, he said, the company has built and made a significant investment in technologies to power digital connectivity.
DISCUSSION
During the session panel discussion, speakers and workshop participants highlighted some of the challenges and opportunities for using new technologies and automating processes to enable a robust manufacturing platform for cellular therapies. Individual participants identified some of the financial considerations for device and tool companies as well as the technical challenges in the manufacturing process for academic and industry partnerships. The logistics and feasibility of point-of-care manufacturing, which takes a scaled-down and decentralized approach to manufacturing and providing therapies to patients, were also discussed.
Utilizing Technologies to Optimize the Manufacturing Process
Building on the comments heard throughout the day on automation, Rietze highlighted some of the logistical considerations regarding automated manufacturing. When identifying whether a manufacturing process being used in the development of a new device is ready for automation, it is crucial to know the process thoroughly and to think about automation at the beginning, Rietze said. From a practical perspective, most commercial firms are going to receive a process from an academic institute, in which case the “sweet spot” would be around the time Phase II trials start. By that time, there should be a deeper understanding of the manufacturing process without automation.
Before automation can be built into the manufacturing process, there are other challenges that researchers can address. Workshop participants discussed the importance of identifying what makes a product therapeuti-
cally active early in the development process in order to be able to optimize production and reduce spending. For example, one participant said, it is likely that, at least with CAR T cells, that only a few clones, not billions of cells, are therapeutically active, and if those cells can be identified, the manufacturing process and automation models will change drastically. Researchers need to identify those cells at a much earlier stage before industry devotes a tremendous amount of energy and money building tools and technologies, another participant said. Rietze agreed, and acknowledged that the current process for CAR T manufacturing will likely evolve as the technology advances. To reflect the reality that manufacturing is likely to change as additional products are developed, Novartis moved its process development group back into the company’s basic research institutes so that process and product are being developed together. Data will drive the development of new processes that rely on early product characterization during the discovery phase, Rietze said. GE Healthcare’s approach to development is to design scalable manufacturing platforms that will solve a problem regardless of scale, Vanek said.
From a financial perspective, Vanek said, capital expenditures are not the most important cost driver in the long run when developing a new tool. According to the modeling his group has conducted, when production reaches a scale of tens of thousands or hundreds of thousands of patients at a time, the overall capital expenditure shrinks in comparison to the consumable demand on a per-patient basis. That is not to say that the operational expenditures related to repairs and maintenance and idle capacity are inconsequential, but if manufacturing facilities can be designed with efficiency in mind to support more than one indication, those costs get absorbed across multiple products.
Engineers have the ability to unitize a process, gather the available data, and create integrative solutions, a workshop participant noted, and an important part of the tool development process is identifying those areas in the development pathway where current tools are not sufficient and need updating. One of those areas, Vanek said, is the characterization and qualification of starting cells. Addressing that bottleneck may require the development of more efficient processes of cell selection and of in-process smart devices and sensors that enable machines to become smarter on their own. Investments made in developing novel platforms such as microfluidics that are very high risk compared to existing technologies that have been around for decades (and in many cases are off patent) is challenging, Rietze said, but sharing the risk for product development could help move the field forward. There are very few systems that can be used in the laboratory to expand cells that will then be scalable for commercial application, Rivière said. Researchers and manufacturers need to better understand how to identify the active cells in order to reduce dose size, she added, and eventu-
ally the manufacturers need to think about how to make pseudo-universal products derived from iPSCs, which would eliminate the need to use patient materials.
Manufacturing at the Point-of-Care
Manufacturing at the point-of-care versus in a centralized location is a viable and important option for orphan diseases, Rivière said. Unless the analytics are automated too, there will need to be a huge amount of training and standard operating procedures that will need to be transferred from one center to another. For diseases with a large number of patients, she said, hospitals should focus their efforts on the clinical trials and leave manufacturing to biotechnology or pharmaceutical companies.
One problem with point-of-care manufacturing is that it makes multicenter trials difficult because of the need to validate and demonstrate comparability across sites. The requirement to have centralized analytical laboratories is another obstacle for point-of-care manufacturing in multicenter clinical trials, unless the analytics are integrated and fully automated. Automation is expensive, however, and, as Rivière pointed out, there are limited funds available to develop manufacturing automation and analytics for point-of-care applications. Furthermore, there are issues to address before point-of-care manufacturing becomes feasible, including building the necessary infrastructure and identifying resources. Automation could eventually provide an answer to this issue, she said, and researchers are working on accomplishing that feat (Roh et al., 2016). There is some confusion about what CQAs need to be monitored in an automated process, Rivière said (see Chapter 5 for further discussion on CQAs). Some in the field hope that the large datasets now being collected from patients who have received some of the first commercialized products will provide insights about what attributes are critical to measure in order to monitor the manufacturing process.
Several things have to happen for point-of-care manufacturing to become realistic, Vanek said. First, the dose would have to be of the right size in order to be able to be manufactured at that scale, and the time to produce that dose would have to be relatively short, he said. Second, the understanding of data analysis and cell characterization would have to reach a level sufficient to enable automation of the manufacturing process.
This page intentionally left blank.














