Proceedings of a Workshop
OVERVIEW OF THE WORKSHOP1
In 2011, the National Cancer Policy Forum (NCPF) of the National Academies of Sciences, Engineering, and Medicine held a workshop on the role of obesity in cancer survival and recurrence.2 Workshop presentations and discussions examined the epidemiological evidence demonstrating that the risk of developing many cancers increases for individuals who are overweight and obese, as well as observational data that excess body weight can worsen outcomes for cancer survivors, including an increased risk of cancer progression, recurrence, and mortality (IOM, 2012). Workshop speakers also examined the biological mechanisms underlying the obesity–cancer link; potential interventions to counter or prevent the effects of obesity and restore energy balance;3 how clinicians could advise patients with cancer
___________________
1 The planning committee’s role was limited to planning the workshop. This Proceedings of a Workshop was prepared by the rapporteurs as a factual account of what occurred at the workshop. Statements, recommendations, and opinions expressed are those of individual presenters and participants and are not necessarily endorsed or verified by the National Academies of Sciences, Engineering, and Medicine. They should not be construed as reflecting any group consensus.
2 See http://www.nap.edu/catalog/13348 (accessed April 5, 2017).
3 Energy balance is determined by energy intake from diet, which can be influenced by ingestion and absorption, balanced by the total calories expended. In general, approximately 60–75 percent of energy is expended by maintaining the resting metabolic rate, 5–10 percent
regarding weight loss, diet, and exercise to reduce their risk of cancer progression or recurrence; and policy suggestions related to research, education, and dissemination of the findings on obesity and cancer (IOM, 2012).
Since the 2011 workshop, there have been a number of developments in the field, said Wendy Demark-Wahnefried, professor and chair of nutrition sciences at the University of Alabama at Birmingham. For example, the American Society of Clinical Oncology (ASCO) issued a position statement and established an initiative to reduce the impact of obesity on cancer (Ligibel et al., 2014). Research on obesity and cancer has also continued to advance. For example, Pamela Goodwin, professor of medicine at the University of Toronto, reported that a recent review found sufficient evidence to conclude that obesity and excess weight are linked to the development of 13 cancers: breast (postmenopausal), colorectal, endometrial, esophageal (adenocarcinoma), gallbladder, gastric, kidney (renal cell), liver, meningioma, multiple myeloma, ovarian, pancreatic, and thyroid cancers (Lauby-Secretan et al., 2016).
At the same time, Demark-Wahnefried noted that there is a need for an even greater focus on addressing excess body weight, poor diet, and inadequate physical activity among cancer survivors. She stressed that the number of cancer survivors is growing rapidly: In 2016, there were more than 15.5 million cancer survivors in the United States; by 2026, this number is expected to reach 20 million. Demark-Wahnefried noted that adult cancer survivors are more likely than the general population to have obesity, experience fatigue, have reduced muscle mass and strength, and to develop heart disease, stroke, osteoporosis, and metabolic syndrome (Greenlee et al., 2016; Henderson et al., 2014). They are also at risk for secondary cancers. Some of these health risks might be mitigated by interventions that are designed to promote weight management (weight maintenance or weight loss), increase physical activity, and improve diet, she said. Yet, studies show that more than half of cancer survivors are insufficiently active and have suboptimal diets (Blanchard et al., 2008; Ford et al., 2014; Niu et al., 2015; Zhang et al., 2012). Jennifer Ligibel, director of the Leonard P. Zakim Center for Integrative Therapies at the Dana-Farber Cancer Institute, said a survey of cancer survivors found that less than half (30–47 percent) engage
___________________
is expended as the energy needed to digest food, and 15–30 percent is expended through physical activity. Weight maintenance occurs when there is energy balance, or when energy input is equal to energy expenditure. Energy imbalances result in weight gains or losses (IOM, 2012).
in recommended levels of physical activity (Blanchard et al., 2008). Cancer survivors are also more likely to report greater sedentary behavior compared to individuals without cancer (Kim et al., 2013), noted Crystal Denlinger, chief of gastrointestinal medical oncology at the Fox Chase Cancer Center. Melinda Irwin, associate director for population sciences in the Yale Cancer Center and professor of epidemiology at the Yale School of Public Health, added that women with breast cancer tend to show marked reductions in cardiorespiratory fitness compared to sedentary women who have not been diagnosed with cancer, especially in the postadjuvant setting (Jones et al., 2012; Lakoski et al., 2013). Thus, there is considerable interest in evaluating and implementing interventions to promote weight management and physical activity among cancer survivors.
Given this interest, the NCPF held a second public workshop, Incorporating Weight Management and Physical Activity Throughout the Cancer Care Continuum, on February 13 and 14, 2017, in Washington, DC. The purpose of this workshop was to highlight the current evidence base, gaps in knowledge, and research needs on the associations among obesity, physical activity, weight management, and health outcomes for cancer survivors, as well as to examine the effectiveness of interventions for promoting physical activity and weight management among people living with or beyond cancer. Workshop sessions also reviewed the opportunities and challenges for providing weight management and physical activity interventions to cancer survivors.4
The workshop convened a number of stakeholders with a broad range of views and perspectives, including patients, researchers, clinicians, and insurers. This Proceedings of a Workshop chronicles the presentations and discussions at the workshop, which included the following:
- The evidence base for the impact of body weight and physical activity on cancer survivors;
- The effect of interventions to promote physical activity and weight management on health outcomes for cancer survivors and in other populations;
- How the available evidence base can be applied to patient care;
- Gaps in the evidence base;
___________________
4 While this workshop did include some discussion on the role of diet with respect to interventions for weight management and physical activity, this topic was addressed at length in a separate workshop, Examining Access to Nutrition Care in Outpatient Cancer Centers (NASEM, 2016).
- Examples of interventions for weight management and physical activity and the need to tailor interventions to meet the needs of diverse populations;
- Challenges in delivering weight management and physical activity interventions to cancer survivors and issues with infrastructure, program capacity, clinician referral, provision of care, and patient adherence; and
- Potential opportunities to improve weight management and physical activity interventions for cancer survivors.
The workshop proceedings chronicles a number of suggestions from individual participants for potential ways to improve the delivery of weight management and physical activity interventions for cancer survivors—these suggestions are discussed throughout the document and are summarized in Box 1. The workshop statement of task is included in Appendix A and the workshop agenda in Appendix B.5
Terminology and Concepts Discussed at the Workshop
Several speakers provided definitions for concepts and terminology used in workshop presentations and discussions. Goodwin defined obesity as having a body mass index (BMI6) of 30 or above (approximately 30 pounds overweight for a 5’4” tall adult). A person is defined as overweight if he or she has a BMI of 25 to < 30, normal weight if his or her BMI is 18.5 to < 25, and underweight if his or her BMI is < 18.5.
Marcas Bamman, professor and director of the Center for Exercise Medicine at the University of Alabama at Birmingham, provided definitions for physical activity, exercise, physical fitness, and sedentary behavior. Physical activity is defined as any movement that results in energy expenditure above resting levels, and encompasses exercise, sports, and physical activities completed as part of daily living, occupation, leisure, and active transportation (Garber et al., 2011).
In contrast, exercise is planned, structured, and repetitive activity with the objective to improve or maintain physical fitness (Garber et al., 2011).
___________________
5 The webcast and presentations from the workshop are available at http://nationalacademies.org/hmd/Activities/Disease/NCPF/2017-FEB-13.aspx (accessed April 5, 2017).
6 BMI is calculated as a person’s weight in kilograms divided by the square of his or her height in meters. See https://www.cdc.gov/obesity/adult/defining.html (accessed August 13, 2017).
Bamman defined physical fitness as the ability to carry out daily tasks with vigor, alertness, and without undue fatigue (Garber et al., 2011). He said that fitness is assessed through health- and skill-related attributes, including cardiorespiratory fitness (measured by maximal oxygen uptake during exercise—VO2max), muscular strength and endurance, body composition, flexibility, balance, agility, reaction time, and power. “Being more physically active is great, and we need to encourage that activity and measure it, but it does not necessarily translate into an improvement in fitness,” Bamman said, emphasizing that intensity and volume of exercise are important fitness determinants.
Bamman also described different types of exercise, including endurance or aerobic training, which includes repetitive exercises (e.g., running, walking, and swimming) that produce dynamic muscle contractions of large muscle groups for an extended period of time (Physical Activity Guidelines Advisory Committee, 2008). Another type of exercise is resistance or strength training (e.g., weight lifting and yoga), which is primarily designed to increase skeletal muscle strength, power, endurance, and mass (Physical Activity Guidelines Advisory Committee, 2008).
Bamman reported that the Department of Health and Human Services (HHS) recommendations for endurance (aerobic) training for the general population are 150 minutes per week of moderate-intensity exercise or 75 minutes of vigorous-intensity exercise, performed in episodes of at least 10 minutes, ideally spread throughout the week (Physical Activity Guidelines Advisory Committee, 2008). HHS recommendations for resistance (strength) training are to engage in strengthening exercises for each major muscle group 2 days per week (Physical Activity Guidelines Advisory Committee, 2008). Bamman added that most exercise studies measure the effects of endurance training rather than resistance training.
He defined sedentary behavior as activity that involves little or no movement, such as watching television or using a computer (Garber et al., 2011). Bamman emphasized that sedentary behavior, also referred to as physical inactivity, is critically important to health because it can affect nearly every organ system, and physical inactivity has been shown to double the relative risk of developing cardiovascular disease and diabetes (Booth and Laye, 2009; Wilmot et al., 2012).
EVIDENCE BASE FOR THE IMPACT OF BODY WEIGHT AND PHYSICAL ACTIVITY ON CANCER SURVIVORS
Excess weight and a lack of physical activity can contribute to the development of cancer and worsen outcomes for cancer survivors, a number of workshop participants reported. The growing evidence base linking obesity, a lack of physical activity, and worsened outcomes for cancer survivors, combined with the increasing number of cancer survivors who are overweight, obese, or insufficiently physically active, have led to research evaluating interventions for weight management and physical activity in cancer survivors. Many workshop participants described studies documenting how physical activity and weight management interventions can improve the well-being of patients with cancer. A number of speakers noted that it is much more difficult to evaluate the effect of these interventions on cancer outcomes, such as recurrence and mortality, but there is ongoing research that aims to collect this information.
Body Weight and Cancer
Goodwin noted that the United States is in the midst of an obesity epidemic, with obesity rates having doubled in most states between 1990 and 2010 (CDC, 2017). She added that obesity is also increasing worldwide. William Dietz, director of the Sumner M. Redstone Global Center on Prevention and Wellness at The George Washington University Milken Institute School of Public Health, added that while obesity rates have plateaued among U.S. children in recent years, these rates continue to increase among adults (Ogden et al., 2015). He noted that the increased prevalence of obesity in adults is largely due to an increase in obesity among women. Approximately 40 percent of all women—46 percent of Hispanic women and 57 percent of African American women—in the United States have obesity (Ogden et al., 2015).
Goodwin reported that in 2012, one-quarter of all obesity-related cancer cases globally—and more than one-third of cases in North America—could be attributed to the increase in BMI between 1982 and 2002 (Arnold et al., 2015). In addition, the prevalence of obesity is greater among cancer survivors compared to the general population (Greenlee et al., 2016).
Goodwin also said that obesity is associated with worsened cancer outcomes, including a modestly higher risk of recurrence or death in many common cancers. For example, a systematic review of breast cancer survi-
vors found that obesity is associated with a greater risk of overall mortality and cancer-specific mortality compared to women of normal weight (Chan et al., 2014). Being overweight or obese is also associated with a greater susceptibility to chemotherapy-induced cardiotoxicity for patients with breast cancer (Guenancia et al., 2016). Goodwin added that obesity is linked to shorter survival in patients with pancreatic and ovarian cancers (Bae et al., 2014; Shi et al., 2016). Patients with prostate cancer and obesity have a greater likelihood of recurrence than patients who are not obese (Hu et al., 2014), and patients with colorectal cancer and obesity have an increased risk of all-cause mortality, cancer-specific mortality, recurrence, and shorter disease-free survival, compared to patients with colorectal cancer who are normal weight (Doleman et al., 2016). In addition, children who are diagnosed with acute lymphoblastic leukemia or acute myeloid leukemia and have a high BMI have poorer event-free and overall survival rates (Orgel et al., 2016).
Although the association between obesity and worsened outcomes has been found in a broad range of cancers, Goodwin noted that there are exceptions. For example, in patients who have diffuse large cell lymphoma, esophageal cancer, lung cancer, and multiple myeloma, a lower BMI is linked to worsened cancer outcomes, potentially due to the poor prognosis associated with cancer-related weight loss (Beason et al., 2013; Carson et al., 2012; Fahey et al., 2015; Gupta et al., 2016; Jung et al., 2014; Weiss et al., 2014).
Goodwin said that obesity alters an individual’s physiology, including higher levels of inflammation, blood sugars, insulin, free fatty acids, and estrogen levels, as well as greater insulin resistance and altered tumor micro-environment (Goodwin and Stambolic, 2015). These physiologic and tissue changes can affect the development of cancer, and they may provide the biochemical signaling or cellular energy that fuels the proliferation of cancer cells and their invasion into other tissues (Goodwin and Stambolic, 2015).
For example, an analysis of tumor tissue from women with estrogen receptor-positive breast cancer found different gene expression signatures among women with obesity compared to women of normal weight. Women with obesity were more likely to have heightened expression of signaling pathways shown to play a role in many cancers, including activating the phosphatidylinositol-3-kinase (PI3K)/AKT/mammalian target of rapamycin (mTOR) signaling pathway (Fuentes-Mattei et al., 2014). “This is telling us that breast cancers in obese individuals are biologically different than the breast cancers in normal weight individuals,” Goodwin
noted. However, she said that it is unclear whether losing weight changes the outcomes from these cancers, and added that the link between obesity and cancer may not be a causal association. “I think this is a real association [between obesity and cancer],” Goodwin said, but she added that that even if the obesity–cancer link is causal, that does not necessarily mean that lifestyle changes targeting obesity will improve cancer outcomes. Ligibel added, “Although we do not know definitively whether metabolism, inflammation, or differences in immunity are responsible for the observed connections between obesity and cancer, we can see that there are biologically plausible mechanisms that could directly affect cancer outcomes in patients who are overweight, obese, and inactive.” Goodwin noted that well-conducted randomized controlled trials are needed to understand whether cancer outcomes can be altered through lifestyle interventions.
Physical Activity and Cancer
Physical activity, exercise, and cardiorespiratory fitness are linked to cancer outcomes, reported Bamman and Kerry Courneya, professor and Canada Research Chair in the Faculty of Physical Education and Recreation at the University of Alberta. High cardiorespiratory fitness has been linked to reductions in cancer risk, all-cause mortality, and cancer mortality, Bamman reported (Brunelli et al., 2009; Jones et al., 2010, 2012; Lakoski et al., 2015; Laukkanen et al., 2016; Schmid and Leitzmann, 2015). Compared to those in the lowest cardiorespiratory fitness category, people who have high cardiorespiratory fitness have a 45 percent lower relative risk of cancer mortality (Schmid and Leitzmann, 2015). In a review of 26 observational studies of patients with breast, colorectal, and prostate cancers, those who reported the highest level of physical activity had a 37 percent lower risk of cancer-specific mortality during the follow-up period compared to those reporting the lowest level of physical activity (Friedenreich et al., 2016). But Courneya pointed out the limitations of these observational studies, including that none were designed to detect the link between physical activity and cancer outcomes and that many lacked reliable and objective measures of physical activity and instead relied on self-reporting.
However, Courneya said that exercise has been found to act on the same biological pathways affecting tumor growth. In both animal and human studies, research has shown that exercise reduces inflammation, the production of estrogen, and insulin growth factor-1; it also increases protein production from the BRCA (BReast CAncer susceptibility) 1 and 2
genes, which have been found to play a role in the repair of damaged DNA (Thomas et al., 2016). Exercise has also been found to enhance immune responses and to affect the production of signaling molecules that regulate the cell cycle, which is often disrupted in cancer cells (Thomas et al., 2016). Bamman added that exercise has regenerative properties, including activating stem cells, creating new neurons, muscle, and bone tissue, and reducing fat deposits (Fiuza-Luces et al., 2013). “Lots of things are happening at the molecular and cellular levels that are very powerful,” he said.
Exercise may also improve cancer outcomes by influencing a patient’s response to chemotherapy, said Bamman. For example, a mouse modeling study found that aerobic exercise improved the delivery of chemotherapy to tumor tissues and inhibited tumor growth (Schadler et al., 2016). Another study found that exercise-trained mice had fewer cancerous cells crossing the blood–brain barrier (Wolff et al., 2015).
INTERVENTIONS FOR WEIGHT MANAGEMENT AND PHYSICAL ACTIVITY IN CANCER SURVIVORS
A number of speakers discussed previous and ongoing studies to assess the effects of physical activity and weight management interventions in cancer survivors. Many of the previously conducted studies have not generated adequate data to assess whether these interventions can improve cancer outcomes, such as reductions in mortality or cancer recurrence, Goodwin said. To ascertain the effects of interventions on cancer outcomes, a number of ongoing randomized controlled trials are currently being conducted.
However, current research has provided a number of insights about the role of weight management and physical activity interventions on other outcomes that are important to cancer survivors and the delivery of cancer care, including the effects on quality of life, fatigue, cancer-related morbidities, treatment completion, and chronic health conditions: “There is strong evidence that exercise and weight management improves quality of life, fitness, body weight, and sleep, and reduces fatigue, lymphedema, and joint pain. But the effects of exercise and weight management on bone mineral density, cognitive function, peripheral neuropathy, and cardiovascular disease biomarkers are uncertain,” said Irwin.
Fatigue and Quality of Life
Irwin discussed the effect of weight management and physical activity interventions on quality of life (emotional, mental, physical, and social well-being) and fatigue in cancer survivors. She said that cancer-related fatigue is the most frequent symptom that cancer survivors report. She defined fatigue as a distressing, persistent, and a subjective sense of physical, emotional, and/or cognitive tiredness or exhaustion related to cancer that is not proportional to recent activity and often interferes with usual functioning. A review of 85 exercise intervention studies in patients during and after cancer treatment found that exercise had favorable effects on fatigue, quality of life, physical function, and muscle strength (Schmitz et al., 2010b).7 A more recent meta-analysis from 34 trials found significant benefits of exercise—especially supervised exercise—on quality of life and physical functioning in patients with cancer (Buffart et al., 2017). “While the effect size was small, there was consistent evidence to support implementation of exercise as part of cancer care,” Irwin said. Another systematic review and meta-analysis of supervised exercise in breast cancer survivors also found reductions in cancer-related fatigue and improvements in overall quality of life, she added (Meneses-Echávez et al., 2015).
Irwin discussed the Rehabilitation Exercise for Health After Breast Cancer (REHAB) trial, which evaluated the effect of stationary bicycling three times per week for 15 weeks among postmenopausal breast cancer survivors following treatment. Compared to the control group, women in the bicycling intervention had significant reductions in fatigue and improvements in quality of life that were correlated with their improvements in cardiorespiratory fitness (Courneya et al., 2003).
She also reported on the Combined Aerobic and Resistance Exercise (CARE) trial, which evaluated weekly supervised exercise (25–30 minutes of aerobic exercise, 50–60 minutes of aerobic exercise, or 50–60 minutes of aerobic exercise and resistance training) in women with breast cancer who were undergoing chemotherapy treatment. The study found that the 50–60 minutes of aerobic exercise or combined aerobic exercise and resistance training may manage declines in physical functioning and treatment-related symptoms better than shorter exercise durations (Courneya et al., 2013).
Most research has assessed the effects of aerobic exercise or resistance training for patients with cancer, but Irwin also reported on a yoga interven-
___________________
7 Most of these studies were conducted in patients with breast cancer.
tion among breast cancer survivors who reported significant cancer-related fatigue. Compared to a group receiving health education, women in the yoga intervention had significant improvements in reduced fatigue and improved vigor, as assessed by the vigor subscale of the Multidimensional Fatigue Symptom Inventory (Bower et al., 2012). Irwin noted that providing individuals with different options for exercise could facilitate improvements in adherence.
Irwin and colleagues also completed a study of a 6-month, home-based, telephone-administered exercise intervention for patients with ovarian cancer who had just finished their first round of chemotherapy (Zhou et al., 2017). This study found that the exercise intervention improved quality of life and reduced fatigue compared to the control group. However, the intervention did not improve quality of life or reduce fatigue among the one-third of the women who experienced ovarian cancer recurrence while undergoing the intervention, Irwin noted.
Irwin said that the LIVESTRONG at the YMCA exercise program for cancer survivors (see Box 4) has also been found to improve quality of life and fatigue, especially among individuals who attended more than 20 sessions (Irwin et al., 2017).
Irwin reported on two studies that assessed the effects of combined diet and exercise interventions on quality of life and cancer-related symptoms. The Reach out to ENhancE Wellness (RENEW) trial evaluated a 12-month, home-based program of telephone counseling and mailed materials promoting exercise, improved diet quality, and modest weight loss, compared to a wait-list control group. The trial included survivors of breast, prostate, and colorectal cancer with BMIs ranging from 25 to 40. This study found that the intervention was linked to improvements of physical function and quality of life (Morey et al., 2009). “This is a really important finding because functional independence is critical for this older population,” Irwin stressed.
More recently, the Exercise and Nutrition to Enhance Recovery and Good Health for You (ENERGY) trial randomly assigned approximately 700 breast cancer survivors who were overweight or obese to either a group-based behavioral intervention with telephone counseling and tailored newsletters to support weight loss, or a control group with a less intensive intervention. The study found that the behavioral intervention led to clinically meaningful weight loss, and improved some aspects of quality of life, including physical functioning (Demark-Wahnefried et al., 2015; Rock et al., 2015).
Morbidities Associated with Cancer and Cancer Treatment
Cancer survivors often experience a number of health effects from cancer or its treatment, including bone loss, joint pain, lymphedema,8 sleep problems, and a decline in cognitive function. For example, Irwin noted that chemotherapies and hormonal therapies are associated with bone loss, osteoporosis, and risk of fractures (Lustberg et al., 2012), but few studies have assessed the effects of exercise on bone loss in patients with cancer. Irwin said that one of the largest studies conducted in this area found that step aerobics and circuit training exercises after adjuvant therapy prevented femoral neck bone loss among premenopausal breast cancer survivors, but not in postmenopausal women (Saarto et al., 2012).
Irwin said that joint pain occurs in up to 50 percent of breast cancer survivors taking aromatase inhibitors, and because of this side effect, many women do not continue therapy. A year-long program of resistance training and aerobic exercise significantly decreased joint pain in breast cancer survivors taking aromatase inhibitors compared to usual care (Irwin et al., 2015). “This is an important finding in that it might improve treatment adherence,” Irwin noted.
Irwin added that several studies have assessed the effects of resistance training on lymphedema in breast cancer survivors. Irwin reported that several studies have found that weight lifting does not increase the incidence of lymphedema in women at risk for lymphedema or exacerbate it in women with breast cancer-related lymphedema (Schmitz et al., 2009, 2010a).
Irwin said that cancer survivors often report problems with sleep, and one intervention study found that yoga improved sleep outcomes better than the standard of care among cancer survivors who had completed treatment (Mustian et al., 2013). Another intervention study involving yoga assessed self-reported cognitive functioning among cancer survivors; at a follow-up of 3 months, yoga participants reported significantly fewer problems with cognition compared to the wait-listed control group (Derry et al., 2015). A study also found that breast cancer survivors who are physically active performed better on cognitive tasks compared to cancer survivors who
___________________
8 Lymphedema is a condition where extra lymph fluid builds up in tissues and causes swelling if lymph vessels are blocked, damaged, or removed. Surgery, radiation, infection, or sometimes cancer itself can cause lymphedema. See https://www.cancer.gov/publications/dictionaries/cancer-terms?cdrid=45359 (accessed August 9, 2017) and https://www.cancer.org/treatment/treatments-and-side-effects/physical-side-effects/lymphedema/what-is-lymphedema.html (accessed August 9, 2017).
are inactive (Hartman et al., 2015). Irwin added that researchers are also evaluating the effect of an exercise intervention on cognitive functioning.
Chronic Disease Prevention in Cancer Survivors
Irwin said that cancer survivors are at risk for a number of chronic health conditions, such as cardiovascular disease and diabetes. For example, women with nonmetastatic breast cancer are at increased risk of cardiovascular disease compared with age-matched women without breast cancer (Hooning et al., 2007). Cardiovascular disease is also a leading cause of mortality in women with nonmetastatic breast cancer (Jones et al., 2016). One analysis found that nonmetastatic breast cancer survivors who met national guidelines for exercise had a 23 percent reduction in the risk of cardiovascular events compared to those who did not meet these guidelines (Jones et al., 2016). Irwin noted that this is an important finding, because some of these breast cancer survivors may have taken chemotherapies that are known to cause cardiotoxicity. “Exercise is important not only for cancer outcomes, but also for cardiovascular disease endpoints,” Irwin said.
Several large studies of weight loss interventions for breast cancer survivors have shown it is possible to achieve at least a 5 percent weight loss, Irwin reported (Befort et al., 2016; Goodwin et al., 2014; Rock et al., 2015). In one study, approximately 75 percent of the women receiving phone-based group counseling maintained at least a 5 percent weight loss 18 months later (Befort et al., 2016). Irwin added that weight loss and exercise interventions may help to reduce the risk of comorbidities. For example, early-stage breast cancer survivors in the ENERGY trial who received the weight loss intervention developed significantly fewer comorbidities than the control group at 1 year; however, there was no significant difference in comorbidities between the groups at 2 years (Sedjo et al., 2016).
The Nutrition and Exercise Study for Women (NEW) trial enrolled more than 400 women who were postmenopausal, sedentary, and either overweight or obese. Women were randomized to a moderate-intensity exercise intervention, a dietary caloric restriction intervention, a combination of both, or a control group (Foster-Schubert et al., 2012). The study found that lifestyle changes involving diet, exercise, or a combination of both can improve body weight in postmenopausal women. Other studies have also found that dietary weight loss, with or without exercise, improves some cancer-related metabolic and inflammatory biomarkers, Ligibel reported (Imayama et al., 2012; Mason et al., 2011).
The CHOICE trial enrolled postmenopausal breast cancer survivors with a BMI between 25 and 35 to a low-fat diet, a low-carbohydrate diet, or usual care for 6 months (Sedlacek et al., 2011; Thompson et al., 2012, 2015). This study found that cancer survivors enrolled in either type of diet had similar weight loss and reduced levels of fasting glucose. But Ligibel noted that biomarkers, such as fasting glucose, have not been validated for cancer outcomes.
Effects on Cancer Treatment Tolerability and Adherence
Several clinical trials have evaluated the role of exercise on cancer treatment tolerability and adherence, Irwin said. One study compared the effectiveness of a low-intensity, home-based, physical activity program and a moderate- to high-intensity program that combined supervised resistance and aerobic exercise to usual care on a number of outcomes in women undergoing adjuvant chemotherapy for breast cancer (van Waart et al., 2015). Women in the moderate- to high-intensity program required fewer chemotherapy dose adjustments compared to the other two groups. “Exercise could be very important in assisting patients to complete their chemotherapy,” Irwin noted.
Courneya also discussed the Supervised Trial of Aerobic versus Resistance Training (START) trial, which compared strength training, aerobic exercise, and usual care in patients with breast cancer undergoing chemotherapy (Courneya et al., 2007). Although the study found that exercise did not significantly affect quality-of-life measures, it did improve chemotherapy completion rates (assessed as the average relative dose intensity for the originally planned regimen), body composition, physical fitness, and self-esteem without increasing the risk for lymphedema or serious adverse events (Courneya et al., 2007). After 8 years of follow-up, disease-free survival was 83 percent in the exercise groups compared to 76 percent in the usual care group (Courneya et al., 2014a). Because this was an exploratory analysis with a small number of patients, Courneya said that a definitive randomized phase III study is needed to confirm the findings.
Ongoing Studies Designed to Evaluate Weight Management and Physical Activity Interventions on Cancer Outcomes
Ligibel and Courneya described a number of studies that intend to assess how interventions affect cancer endpoints, such as disease recurrence and mortality (see Table 1).
| BWEL | CHALLENGE | DIANA-5 | INTERVAL-MCRPC | LIVES | SUCCESS C | |
|---|---|---|---|---|---|---|
| N | 3,136 | 962 | 1,241 | 866 | 1,040 | ~1,400 |
| Disease | Breast | Colon | Breast | Prostate | Ovarian | Breast |
| Stage | II–III | II–III | I–III | IV | II–IV | II–III |
| Intervention | 2-year weight loss | 3-year exercise | 4+-year medicinal diet and exercise | 2+-year exercise | 2-year diet and exercise | 2-year weight loss |
| Primary endpoint | IDFS | DFS | IDFS | OS | PFS | DFS |
| Correlative | Blood Tissue | Blood | Blood | Blood | Blood | Blood |
NOTE: BWEL = Breast Cancer WEight Loss; CHALLENGE = Colon Health and Life-Long Exercise Change; DFS = disease-free survival; DIANA = Diet and Androgens; IDFS = invasive disease-free survival; INTERVAL-MCRPC = INTense Exercise foR surVivAL Among Men with Metastatic Castrate-Resistant Prostate Cancer; LIVES = Lifestyle Intervention for Ovarian Cancer Enhanced Survival; OS = overall survival; PFS = progression-free survival.
SOURCE: Ligibel presentation, February 13, 2017.
The Breast Cancer WEight Loss9 (BWEL) study aims to enroll more than 3,000 women with stage II or III breast cancer who have a BMI of at least 27. These women will be randomized to a weight loss intervention group or control group that will receive education materials. The study uses an intensive intervention involving 42 calls with weight loss counselors over a 2-year time span, and is designed to foster a 10 percent weight loss. In addition to these phone calls, participants are also given a number of materials to facilitate behavior changes, including a workbook, wireless scale, food scale, and activity monitor. The endpoints the study will be evaluating include invasive disease-free survival, disease recurrence, overall survival, distant disease-free survival, comorbidities, death from any cause, weight change, and health behaviors. Ligibel said the study has opened at approximately 900 sites across the United States, and researchers expect to activate the trial in Canadian centers soon. Ligibel added that a Spanish language version of the intervention is also planned.
Courneya reported on the Colon Health and Life-Long Exercise Change (CHALLENGE) trial, the first phase III trial to evaluate the effects of exercise on disease-free survival in colon cancer survivors (Courneya et al., 2014b). Patients with high-risk stage II or III colon cancer who have received chemotherapy within the past 2 to 6 months are randomly assigned to an experimental arm to receive 3 years of behavioral support and an exercise guidebook or to a control arm to receive general health education materials about physical activity and nutrition. The behavioral support intervention consists of biweekly face-to-face sessions and supervised exercise for the first 6 months, followed by reduced interventions in subsequent months. Bernardine Pinto, professor and associate dean for research in the College of Nursing at the University of South Carolina, said that this is an intensive and complex intervention that includes access to a fitness facility, contact with staff, written materials, 17 different behavioral change techniques, and individualized tailoring to participants’ needs. A 1-year feasibility analysis found that the intervention group was doing substantially more physical activity than the control group, and that the level of physical activity is similar to levels associated with improved outcomes in past observational studies of patients with colon cancer (Courneya et al., 2016). The trial has accrued more than 525 of the 962 patients needed, at more than 40 centers worldwide, and the researchers planned to conduct an interim efficacy analysis in 2017, Courneya said.
___________________
9 See https://clinicaltrials.gov/ct2/show/NCT02750826 (accessed April 26, 2017).
Ligibel said that the Italian Diet and Androgens (DIANA)-5 trial enrolled more than 1,000 women with early-stage breast cancer (Villarini et al., 2012). These women were randomized to usual care or to an intervention that included the Mediterranean diet and increased physical activity. Researchers are currently following participants for disease outcomes, Ligibel reported.
Courneya reported on the INTense Exercise foR surVivAL Among Men with Metastatic Castrate-Resistant Prostate Cancer (INTERVAL-MCRPC)10 trial. This trial is the first phase III randomized controlled study examining whether high-intensity aerobic exercise, resistance training, and psychosocial support increases overall survival in men with metastatic, hormone therapy-resistant prostate cancer compared to a control group receiving only psychosocial support. The intervention group will receive an intense and supervised exercise routine three times per week the first month, twice per week for the next 7 or 8 months, and then once per week for the remainder of the year. After the first year, participants will then transition to a home-based, unsupervised program and monthly visits to the clinic. Ligibel stressed that this is one of the few studies examining the effect of lifestyle interventions on outcomes in patients with advanced cancer.
Ligibel also reported on the Lifestyle Intervention for Ovarian Cancer Enhanced Survival (LIVES) trial, which will enroll patients with stage II, III, and IV ovarian cancer who have completed primary chemotherapy at least 6 weeks prior to enrollment and have no evidence of disease (Thomson et al., 2016). The women will be randomized to a 2-year program structured to improve diet and physical activity, or to a health education control group. Both interventions involve telephone calls, print materials, text messages, emails, forums, and mail, and are grounded in social cognitive theory and use motivational interviewing. The primary endpoint of the trial is progression-free survival.
The German SUCCESS C trial, which is assessing the role of anthracycline-free chemotherapy in women with early-stage breast cancer, has an embedded lifestyle intervention program for women who have a BMI between 24 and 40. The embedded analysis will evaluate an intensive lifestyle intervention program on disease-free survival and will assess the predictive role of cancer-associated and obesity-related biomarkers for the prediction of disease recurrence and survival (Rack et al., 2010). Researchers have not yet reported the effect of the lifestyle intervention, Ligibel said.
___________________
10 See https://clinicaltrials.gov/ct2/show/NCT02730338 (accessed April 26, 2017).
Courneya also described the Alberta Moving Beyond Breast CancER (AMBER) study, the first prospective cohort study to examine the role of physical activity and health-related fitness in breast cancer survivorship (Courneya et al., 2012). Women are recruited for this study at the time of diagnosis and are followed through cancer treatment and up to 5 years post-diagnosis. Objective and self-reported measurements of physical activity are collected, as well as blood samples, information on health-related fitness, lymphedema, patient-reported outcomes, and sedentary behavior. Rather than using BMI, the study collects information on body composition using a dual-energy, x-ray absorptiometry scan to assess the overall percentage of body fat, total lean body mass, total fat mass, and bone mineral density. Courneya said that more than 1,000 patients with breast cancer have already enrolled in the AMBER study; he hopes accrual will be completed by the end of 2018, and that the researchers will have 5-year follow-up data by 2023. Participants will be followed for an additional 5 years to collect information on disease outcomes, Courneya said.
“These trials will clearly provide definitive evidence regarding the impact of increased physical activity and weight loss in some of the most common malignancies,” Ligibel said. She noted that most of the randomized controlled trials included numerous enrollment sites, which “can increase the diversity of patients and help us feel confident that these studies can provide some real-world information” about how these interventions could work in diverse community practice settings. She added that some of these trials will be large enough to perform subset analyses, such as comparisons among women with hormone receptor-positive or hormone receptor-negative breast cancer.
However, Ligibel said that the trial results may not be generalizable to all cancer survivors. For example, Suzanne Dixon, clinical liaison at Abbott Nutrition, said that metabolically, patients with breast and prostate cancer are quite different from patients with other types of cancer. But Ligibel also said it is probably not feasible to conduct randomized controlled trials of these interventions in every type of cancer, especially as cancers are increasingly being subdivided into numerous molecular subtypes. “Our biggest challenge will be thinking about how we can take the information we gain from these trials and generalize them to other malignancies,” Ligibel said.
Ligibel noted that these trials are collecting serial blood samples or other biospecimens that can be assessed for relevant biomarkers. Biomarker data could be helpful in developing future studies, especially if certain biomarkers could be validated as surrogate endpoints, Ligibel stressed. “If we
were able to demonstrate the way in which a lifestyle intervention reduces the risk of cancer recurrence by lowering estrogen, lowering insulin, or having a favorable effect on inflammation, then we could do smaller trials that would look at the impact of these interventions on these intermediate [endpoints], and we would be better able to compare different interventions and doses,” Ligibel said.
Courneya said that in a study of patients with colorectal cancer, those whose tumors had lost expression of p27, a cyclin-dependent kinase inhibitor, did not appear to benefit from physical activity intervention. However, individuals whose tumors expressed p27 and who were physically active had a 68 percent improvement in colon cancer-specific mortality compared to those with p27 expression who were not physically active (Meyerhardt et al., 2009). He noted, “This precision medicine approach could be fairly helpful at indicating for whom there could be a protective association between exercise and survival.”
Demark-Wahnefried noted the complexity of biomarker analyses: for example, an increase in one biomarker predicting a beneficial effect might be accompanied by heightened levels of another biomarker considered to predict an adverse effect. Andrew Dannenberg, associate director of cancer prevention at the Sandra and Edward Meyer Cancer Center at Weill Cornell Medical College, agreed, and noted the complexity of fat remodeling in response to exercise—he said it depends on the dose of exercise and the amount of weight loss achieved, and the timing of biomarker assessment matters. However, Dannenberg said that conducting such biomarker analyses in response to exercise and weight management interventions will be very informative over time. Goodwin and Turkan Gardenier, director of research at Pragmatica Corporation, noted that these analyses might also identify environmental factors that affect gene expression and increase the likelihood of a person developing cancer or having a recurrence. “People with different genetic makeups may respond in different ways to the same environmental changes,” Goodwin said. “This is part of the unraveling we need to do, and why biospecimens are collected. So, if an effect is seen, we can begin to unravel what [aspect of] the environment or the patient’s response to the intervention led to a benefit,” she added.
A few workshop participants emphasized that better biomarkers are needed to measure fitness and metabolic health, and described the challenges of using BMI as a biomarker. Goodwin noted a study that found that a subset of individuals who are obese or overweight but who are metabolically healthy have only a modest increased risk of developing cardiovascular
disease, compared to individuals who are metabolically unhealthy, regardless of their weight (Kramer et al., 2013). Dixon said, “What is important is their metabolic health. I may have patients who [are] obese, but have great metabolic profiles, with normal blood pressure, blood sugar levels, and cholesterol profiles. They may be physically active as well. So is that a patient with whom I should intervene with healthy lifestyle interventions?” Dannenberg added, “There are a lot of people who have a BMI of 24 and are getting a pat on the back, who in fact are hyperadipose with evidence of metabolic abnormality. I would argue that many of these individuals 10 to 20 years later will show up in the clinic with type 2 diabetes, heart disease, and cancer, and there should be a big push toward early detection and intervention in that cohort.”
Interventions for Weight Management and Physical Activity in Other Populations
Several speakers discussed the role of weight management and physical activity interventions in other populations and how these interventions may be relevant to cancer survivors. Several speakers noted that adherence to a specific diet and the total calories consumed are more important for weight loss than the type of diet (e.g., low-carbohydrate or low-fat diet). For example, Caroline Apovian, director of the Center for Nutrition and Weight Management at Boston Medical Center, discussed the Preventing Overweight Using Novel Dietary Strategies (POUNDS LOST) study, which randomized 800 participants to 4 diets (low-fat, average-protein; low-fat, high-protein; high-fat, average-protein; or high-fat, high-protein). Each participant’s diet represented a deficit of 750 calories per day from baseline (Sacks et al., 2009). Apovian said the reduced calorie intake led to weight loss, and that the diets were equally successful in promoting clinically meaningful weight loss and the maintenance of weight loss over the course of 2 years (Sacks et al., 2009).
Apovian added that a meta-analysis of more than 100 studies, none of which were controlled, found weight loss was associated with reduced caloric intake and increased diet duration; there was insufficient evidence to recommend for or against reducing carbohydrate consumption (Bravata et al., 2003). Another study that randomized participants to popular diet programs (Atkins, Ornish, Weight Watchers, and Zone) found that adherence, rather than type of diet, predicted successful weight loss (Dansinger et al., 2005). Apovian added that five recent meta-analyses have also found that adherence
was most strongly associated with weight loss (Ajala et al., 2013; Bueno et al., 2013; Hu et al., 2012; Johnston et al., 2014; Wycherley et al., 2012). She added that protein intake in older adults is particularly important for preventing the loss of muscle mass (Katsanos et al., 2006; Paddon-Jones and Rasmussen, 2009; Wolfe, 2006) and evidence suggests that a high-protein diet increases satiety (Dhillon et al., 2016; Leidy et al., 2015).
Studies in patients with diabetes have found that modest weight loss is achievable with a low-calorie diet and moderate physical activity, and this weight loss is linked to a reduction in biomarkers associated with diabetes and cardiovascular disease, Apovian said. In the Look AHEAD (Action for Health in Diabetes) study, almost 40 percent of participants following the diet and exercise regimen lost at least 10 percent of their body weight in 1 year (Look AHEAD Research Group, 2014). At 8 years, approximately 27 percent of the intervention group maintained a weight loss greater than 10 percent, Apovian reported (Look AHEAD Research Group, 2014).
Bamman said that trials of exercise interventions conducted in a wide range of populations have found that the frequency and intensity of exercise are most influential in terms of patient outcomes. In the Look AHEAD trial, patients with type 2 diabetes who increased their levels of cardiorespiratory fitness the most also showed the most improvement in their blood sugar levels (Jakicic et al., 2013). However, some study participants saw a decline in their cardiorespiratory fitness over the course of this study. “It matters how you respond, and that inter-individual response heterogeneity is really important,” Bamman said. “Exercise is like any other therapy from the standpoint that there are really robust responders to your treatment and there are those folks who do not get the same robust response. . . . We have to figure out what works best for [each] individual,” he said.
Apovian also reported on the Diabetes Prevention Program (DPP) trial that randomized participants at high risk of developing diabetes to an intensive lifestyle intervention, the drug metformin, or a placebo. Apovian said that at an average follow-up of 2.8 years, the incidence of diabetes was reduced by 58 percent with the lifestyle intervention and by 31 percent with metformin, compared with a placebo (Diabetes Prevention Program Research Group, 2002). Fifteen years later, although most participants had regained their weight, the cumulative diabetes incidence was still 27 percent lower in the lifestyle intervention group compared to the placebo group; for every 2.2 pounds of weight lost, there was a 16 percent reduction in the risk of developing diabetes, even if that weight was regained (Diabetes Prevention Program Research Group, 2015). “It is better to have lost weight
and regained it than never to have lost it at all,” Apovian said, quoting one of the principal investigators of this trial.
Both the DPP and the Look AHEAD trials incorporated physical activity into their intervention, but physical activity was not found to result in significant weight loss; however, it did help with weight loss maintenance, Apovian said. Those who exercised for 300 minutes per week maintained weight losses nearly three times as great as those who exercised for 150 minutes or less (Wadden et al., 2012), she said. Apovian added that people who are obese and experience weight loss have lower resting metabolic rates than those of the same weight who never were obese, which requires people who were formerly obese to work harder during exercise to get the same metabolic benefit. “This might be the reason why exercise is so important for [this population],” Apovian said. Demark-Wahnefried added that exercise preserves lean body mass, which is correlated with resting metabolic rates. “When you lose lean body mass, you no longer need as many calories to fuel your body, and that is probably one of the big reasons that it is very important to exercise while you are losing weight, so that you can keep that lean body mass” she said.
Apovian emphasized the importance of providing a behavioral intervention in addition to diet to help participants avoid regaining weight, citing a number of studies that found long-term weight loss can be achieved when it is accompanied by long-term, regular support (Elmer et al., 2006; Tate et al., 2001, 2003; Wadden, 1993).
EVIDENCE GAPS
Ligibel noted that there are still many unanswered questions: “We know that there are these links between inactivity, obesity, and poor outcomes. But what we do not know is if you take a patient who is diagnosed with cancer and is obese or not exercising regularly and you change that, will that actually lower their risk of recurrence and help them to live longer? If so, what dose of intervention is needed, and how long do people need to do it for? Is it something like chemotherapy where people need to do this for a number of months after their diagnosis, or is it something that people need to do for years? We do not have answers to these questions from the data currently available,” she said. She added that it is also not known which lifestyle interventions are most important for cancer outcomes. “Is it weight, exercise, [or] diet? Is it the same for all people, or does it vary by disease or by other characteristics of the patient?” she asked.
Christie Befort, associate professor in the Department of Preventive Medicine and Public Health at the University of Kansas Medical Center, and Irwin noted that more studies should be conducted to assess interventions for survivors of more diverse types of cancer. To date, a majority of studies have been completed in women with breast cancer, and these findings may not be generalizable to people with different types of cancer.
Irwin added that the completed studies on weight management and exercise interventions in cancer survivors have limitations. She said that a number of the studies did not control well for potential sources of confounding, such as whether patients assigned to exercise or weight loss interventions received more attention than what was given to other participants. In addition, most studies have not included patients with metastatic cancer. She suggested that more studies are needed to address cancer treatment–related symptoms or conditions, such as peripheral neuropathy and bone density loss, as well as additional studies on whether lifestyle interventions can help alleviate adverse side effects of cancer treatment and improve adherence to treatment regimens. Irwin and Pinto also emphasized the need to conduct more studies that include patients with advanced cancers throughout the entire cancer care continuum.
Melinda Stolley, professor of medicine and associate director for population health at the Medical College of Wisconsin, reported that researchers have conducted very few studies on lifestyle interventions with African American or Hispanic cancer survivors (Chung et al., 2016; Conlon et al., 2015; Djuric et al., 2009; Greenlee et al., 2015, 2016; Rossi et al., 2015; Sheppard et al., 2016). To improve the generalizability of research on lifestyle interventions for cancer survivors, Stolley noted that “we certainly have our work cut out for us.” Stolley said that there is a lack of research that includes racially and ethnically diverse male cancer survivors, Alaskan Native, Native American, and Pacific Islander populations, as well as immigrant and refugee populations (see section on tailoring interventions to special populations).
APPLYING THE EVIDENCE BASE TO PATIENT CARE
A number of workshop participants discussed whether the evidence base is currently strong enough to recommend that cancer survivors increase their physical activity, improve their diets, and if needed, maintain or lose weight. Goodwin suggested that clinicians be upfront regarding the purpose of recommending these lifestyle interventions for cancer survivors: “Are we
recommending them for general health benefits, a reduction in treatment toxicity, or are we saying to our patients that if they lose weight, they will have better [cancer] outcomes? I firmly believe we do not have the data yet to say the latter, but we do have the data to say these interventions will improve the quality of life and general health of our patients.” Goodwin stressed, “We need to convey the sense of urgency. We do not need to wait for every last study; [we need] to start with what we have for now. In the future, new evidence will change what we are doing, and we may want to also recommend interventions that impact recurrence and survival. But for now, there is an urgency to move forward, [and] identify, access, and fund programs that will support the outcomes [based on the] data we already have.” Goodwin added that there is enough data to recommend lifestyle interventions for improving quality of life, fatigue, and the joint symptoms associated with aromatase inhibitors in patients with breast cancer.
Goodwin cautioned that what appears to be true from observational studies may not hold up in randomized controlled studies: “We sometimes know the answer and we are right. I truly hope in this area we are right in terms of cancer outcomes, but we do not know that yet. We need to be as respectful of the lifestyle area as we are of the drug, radiation, and surgery area[s], and require strong evidence.” Courneya added that recurrence is a primary concern of cancer survivors, and some cancer survivors are willing to forego quality-of-life benefits for small improvements in survival. Goodwin added that some patients find it burdensome to adopt lifestyle changes, and many breast cancer survivors feel guilty when they do not lose weight or do not become more physically active, and worry that by not making these lifestyle changes, their cancer will recur. “We have no evidence right now to say that is the case, and we need to be respectful of the women who either will not or do not want to make those changes,” she said. Courneya agreed that more data needs to be collected on recurrence and cancer outcomes: “Once you roll this out to 15 million cancer survivors, I do not think you can roll it back. . . . Even though we are all very keen on lifestyle interventions, we have to be open to the possibility that a vigorous exercise intervention program for some cancer types may worsen outcomes. That is very important information for cancer patients to have,” he said. Dannenberg also agreed, noting there are still a number of unknowns about weight loss in cancer survivors, including how much weight loss over what period of time would be beneficial.
Joanne Buzaglo, senior vice president of research and training at the Cancer Support Community, stressed that she recognizes the importance
of building more evidence. “At the same time, we do not have to hold back from what we already know, which is that cancer survivors can benefit tremendously” from interventions and support to manage weight and be physically active, Buzaglo said. Dixon added, “Do not let the perfect be the enemy of the good. We know an awful lot, and doing nothing is not an option anymore.”
Kathryn Schmitz, professor of public health sciences and associate director of population sciences at The Pennsylvania State University’s College of Medicine, added that “We may not be ready to disseminate interventions aimed at reducing the risk of recurrence and death, but there is a lot to be gained by focusing on how these lifestyle interventions can enhance quality of life, survivorship from other illnesses, and general rehabilitation for our patients. There has been a burgeoning of information relating to the benefits of physical activity and weight loss in terms of cancer rehabilitation and recovery, symptom control, general health promotion, and reduction in risk of obesity-associated illnesses that many of our patients with cancer are dealing with.”
Pinto suggested that clinicians should recommend physical activity to cancer survivors, and pointed out that exercise is currently not usually part of a cancer treatment plan or survivorship care. “This is an area where there are multiple missed opportunities at different times and points after diagnosis,” she said. Schmitz suggested that clinicians tell cancer survivors to avoid inactivity. Dixon agreed, noting that just standing for 2 minutes a few times an hour can have health benefits (Beddhu et al., 2015). “This is something that might be a gateway for patients with cancer [along] the entire length of the continuum. They might not be able to go for a walk, but maybe they could stand up for 10 minutes once an hour.”
Larissa Nekhlyudov, associate professor of medicine at Harvard Medical School, clinical director of Internal Medicine for Cancer Survivors at the Dana-Farber Cancer Institute, and internist at the Brigham and Women’s Hospital, added that she tells her patients “Sitting is the new smoking,” and encourages them to wear a device that tracks their activity. Karen Basen-Engquist, professor of behavioral science and the director of the Center for Energy Balance in Cancer Prevention and Survivorship at The University of Texas MD Anderson Cancer Center, added, “There is a lot of good evidence that getting sedentary people up and walking around a bit does improve quality of life and physical functioning.”
Dixon noted that she encourages patients to walk daily and “just keep moving,” even if it does not result in weight loss because the goal is health
improvement. She added, many of “our patients are already obese, and exercise alone does not work for reducing weight—you have to have a dietary component. I have always told my patients ‘Diet for loss and exercise for maintenance’ and the data is bearing this out.” Dixon added, “We have to figure out where our patients are and meet them there. Maybe they are not there with the dietary component, but we have to reinforce the message that the goal is health, and not necessarily weight loss.” Dietz added, “A mantra consistent with the evidence would be ‘Diet for loss, exercise for life.’”
Kirsten Nyrop, research assistant professor of medicine at the University of North Carolina at Chapel Hill School of Medicine, suggested recommending that patients with cancer “walk safely and comfortably as much as they can and more than they are doing right now.” She noted that encouraging walking can be the gateway to a conversation about exercise between oncologists and their patients. Basen-Engquist suggested giving patients more choices about exercise and rather than prescribing a specific type or amount. She said, “We want patients to exercise or want them to address their weight, but we need to give them a little flexibility and some choices about how they do that.”
Buzaglo added, “Patients are looking for a good quality of life. They want to be engaged fully in their life and be able to function as active citizens and take care of their families and themselves. Fitness and weight management are key to that” (see Box 2).
At the same time, several speakers said that it was important to acknowledge the potential risks that lifestyle interventions could pose to patients undergoing cancer treatment. A few studies have suggested potential risks linked to weight loss or exercise in certain patients with cancer. “We need to be open to the possibility that there could be some cancer treatment side effects that are exacerbated by exercise,” Courneya said. Demark-Wahnefried said that she just completed a trial in which men with prostate cancer who lost more weight had higher tumor proliferation rates (Ki67) than those who had modest weight loss (Demark-Wahnefried et al., 2017), and she suggested that there may be a “sweet spot” for both exercise and weight loss. Courneya said results from one study, although not statistically significant, suggested that patients with colorectal cancer whose tumor did not express p27 might have slightly increased risk of worsened outcomes from exercise (Meyerhardt et al., 2009). He noted that although most animal studies have shown that exercise reduces tumor growth and metastases (Betof et al., 2015), a small number of animal studies have shown the opposite (e.g., Assi et al., 2017).
Courneya and Goodwin noted that some studies suggest weight loss is linked to adverse outcomes in certain cancers. Dixon noted the importance of distinguishing intentional from unintentional weight loss. She noted that the American Cancer Society (ACS) suggests that for people with cancer who are overweight or obese, modest weight loss in the context of a healthy diet and exercise may be beneficial, as long as it is closely monitored and does not affect cancer treatment. For people with cancer who may be malnourished and underweight when they are diagnosed or as a result of
cancer treatment, they may need help gaining or maintaining their weight (ACS, 2017). “We have a lot of work to do to figure out metabolically what is going on and to make sure we are not doing harm in some of these populations,” she said. Cynthia Thomson, leader of the Cancer Prevention and Control Program at The University of Arizona Cancer Center, added “We do not always know if the weight loss is intentional or unintentional, and even a patient’s assessment of this can be inaccurate.”
Examples of Intervention Components
A number of workshop participants described important elements to consider in weight management and physical activity interventions for cancer survivors, including optimal timing of the intervention; screening, risk assessment, and referral options; delivery methods; theoretical constructs to promote healthy behaviors and motivation; and tailoring programs to meet the diverse needs of cancer survivors.
Optimal Timing of Diet and Exercise Interventions
Many workshop participants discussed the optimal timing of lifestyle interventions in cancer survivors. Thomson pointed out that there may be a teachable moment after a cancer diagnosis, with oncology nurses reporting that patients are highly receptive to interventions targeting health behaviors during their cancer treatment (Karvinen et al., 2015). She added that one study found that the more time that has passed since diagnosis, the less healthy eating behaviors are (Bluethmann et al., 2015a). “We have a ways to go in terms of understanding timing, but we are learning,” Thomson added. Schmitz noted there are missed opportunities to provide these interventions in the intervals between a patient’s diagnosis and the start of treatment, or between surgery and adjuvant treatment. “There are opportunities for us to intervene on physical activity, weight control, and nutrition that we have not taken yet,” she said. Lisa Kennedy Sheldon, chief clinical officer at the Oncology Nursing Society (ONS), agreed and suggested recommending physical activity to manage the fatigue linked to cancer treatment. “Integrating physical activity during that teachable, vulnerable moment when people are undergoing treatment is a great way to start,” she said.
However, Pinto noted that exercise intervention trials have better patient retention when they are offered after cancer treatment rather than during treatment (Speck et al., 2010). But the optimal time to intervene might depend on the outcomes that patients and their clinicians hope to achieve. For example, an intervention can be offered soon after diagnosis to improve recovery from surgery, or it could be offered during treatment to reduce treatment-related toxicities and improve adherence to treatment regimens. In the post-treatment setting, the goal of interventions may be to improve long-term health outcomes, such as lowering the risk of developing other chronic conditions. She added that exercise may also be provided in the palliative care context, to help relieve certain symp-
toms and to improve mental health or quality of life. “There are multiple places in the cancer care continuum for exercise interventions, but the [goals and] outcomes are going to be different,” Pinto said. She added, “there are going to be times when . . . people are receptive, and sometimes they are less so,” noting that even if patients were not ready to embark on an exercise program during treatment, they may be more motivated to do so at a later time if they develop late effects from treatment.
Demark-Wahnefried noted that many women with breast cancer gain weight during the course of their chemotherapy, and advice about small change, such as replacing sweetened soft drinks with water, could be beneficial. “There are some simple messages that a [clinician] could deliver. We need to seize the moment,” she said. Denlinger agreed: “You have to give those messages early and often in order to make changes.”
Patient Screening, Assessment, Triage, and Referral
Basen-Engquist stated that clinicians will need to screen cancer survivors to determine whether they could benefit from lifestyle intervention programs, and clinicians will also need to assess the risks of different intervention options for individual patients. Basen-Engquist suggested using an approach analogous to the 5As model that clinicians use to connect people to smoking cessation programs.11 The components of the 5As model are:
- Ask patients about their smoking behavior.
- Advise patients on how smoking cessation could improve their health.
- Assess patients’ willingness to quit smoking.
- Assist patients to find the medicines, programs, and other resources to quit smoking.
- Arrange for follow-up to see if patients were successful in quitting, and if not, to identify what additional help they may need.
Basen-Engquist noted that many clinicians do not use this approach when counseling patients about weight management and physical activity. For example, although obesity is routinely assessed in patients with cancer
___________________
11 See https://www.uspreventiveservicestaskforce.org/Page/Document/RecommendationStatementFinal/tobacco-use-in-adults-and-pregnant-women-counseling-and-interventions1 (accessed April 26, 2017).
by measuring their height and weight, clinicians may not advise patients about how excess weight can influence health outcomes. It is also rare for clinicians to ask cancer survivors about their exercise habits, she said. One study found that less than one-third of patients with colorectal cancer surveyed remembered receiving advice about physical activity, but those who did recall such advice were more likely to be active and follow physical activity recommendations, Basen-Engquist reported (Fisher et al., 2015). Another study in patients with early-stage cancer found that approximately one-third of clinical oncology visits included a discussion about physical activity (Nyrop et al., 2016). “It appears to be helpful to patients if their clinicians talk about physical activity, but [patients] are not getting [much of this information] from their clinicians at this point,” Basen-Engquist said.
When assessing which interventions may be helpful to a specific patient with cancer, Basen-Engquist noted that clinicians should focus on matching an exercise routine that is best suited to a patient’s physical condition and goals. She said there are various risk stratification schemes that can help clinicians determine which patients need a structured and/ or supervised exercise program and which patients can safely participate in an independent, home-based exercise program. Denlinger added that the latest guidelines on physical activity for cancer survivors from the National Comprehensive Cancer Network (NCCN) includes a risk assessment tool for physical activity-induced adverse events, including an algorithm for how best to implement physical activity recommendations based on that risk assessment, patients’ current or prior exercise behavior, and how well they tolerate exercise (Denlinger and Ligibel, 2013). In addition, she said that clinicians need to consider a cancer survivor’s comorbidities, late- and long-term effects from cancer and its treatment, and how that might impede his or her ability to do certain types of exercise. For example, neuropathy-induced balance issues can hamper certain physical activities (IOM, 2006; Yabroff et al., 2004).
Basen-Engquist developed matrices to assist clinicians in determining the risks posed by different exercise and weight management programs based on a patient’s current health and the level of risk associated with these interventions (see Tables 2A and 2B). She said that clinicians can use these tools to determine what interventions might be appropriate, as well as how much supervision patients might need.
“We talk about triage in medical care, and [we] should think about using those same principles in how we deploy resources in behavior change, weight management, and physical activity,” said Kathleen Wolin, chief
executive officer of ScaleDown. Elizabeth Eakin, director of the Cancer Prevention Research Centre at the University of Queensland, provided a triage model for population-based screening of cancer survivors for weight management and physical activity interventions (see Figure 1). This model stratifies interventions based on the setting of care, the training of health care professionals involved in the intervention, and whether the intervention is supervised, as well as a cancer survivor’s time since diagnosis and current symptoms and comorbidities. In general, clinicians can refer cancer survivors to home-based, community-based, or clinic-based lifestyle intervention programs, said Basen-Engquist (see section on delivery methods).
Schmitz said that improvements to triage tools are needed, such as helping clinicians determine when transitions between different settings of care are needed (e.g., a community-based program versus a clinic-based program). “We need to do a better job of deciding how to triage people from one place to another,” she said. Schmitz added, “We do not do a good job of recognizing when somebody has an issue that [could be addressed with] physical therapy. That is a gaping hole that we have to fill at this point.” G. Stephen Morris, associate professor in the Department of Physical Therapy at Wingate University and president of the Oncology Section of the American Physical Therapy Association (APTA), described the experience of a breast cancer survivor who still reported pain and a compromised shoulder range of motion 15 years after completing her cancer treatment. “This shows we need to develop survivorship programs so these survivors can be directed to the clinicians who are best able to recognize and treat lingering or newly emerging adverse effects of cancer and its treatment,” he said.
Christopher Barnes, graduate assistant in the Department of Physical Therapy and Athletic Training at the University of Utah, said that at his university, physical therapists evaluate and triage patients based on their physical activity and mobility and they also coordinate outpatient and inpatient care. “Physical therapists already have the skills to evaluate patients and are interested in coordinating with other treatment teams and other providers. I suggest everybody find physical therapy partners to work with because physical therapists have this large skill set that is being underutilized right now as far as exercise dosing, and doing these triaging activities that do not come as naturally or are not in the purview of other providers,” he said.
Befort also suggested that weight management and physical activity programs be better coordinated with clinical care. In a telephone-based weight loss program among 210 rural breast cancer survivors, she said that 69 percent reported a new or worsening medical condition, with 47 percent
| Least Restrictive Alternative | ||||
|---|---|---|---|---|
| Risk Level of Survivor | Level IV: Survivor with problematic symptoms and/or refractory impairments | Consult with physician/rehabilitation professional prior to exercise in home-based or community program | Cancer rehabilitation | Cancer rehabilitation |
| Level III: Survivor with cancer- or cancer treatment–related impairments, uncomplicated by other systemic problems | Community/home-based program | Consult with physician/ rehabilitation professional prior to exercise in home-based or community program | Supervised exercise program, cancer-specific after consult with physician/rehabilitation professional | |
| Level II: No specific cancer- or cancer treatment–related impairments, but with comorbid or other conditions that may be exacerbated by exercise | Self-selected activity, self-monitoring | Community/home-based program following appropriate self-guided screening (e.g., American College of Sports Medicine/American Heart Association Questionnaires)a | Consult with physician/ rehabilitation professional prior to exercise in home-based or community program | |
| Level I: Post-treatment survivor, no/well-managed comorbidities, no cancer-specific morbidity | Self-selected activity, self-monitoring | Community/home-based program | Consult with physician/ rehabilitation professional prior to exercise in home-based or community program | |
| Reduce sedentary behavior, light intensity exercise | Moderate-intensity aerobic (e.g., walking), light-moderate resistance exercise | Vigorous intensity aerobic (e.g., running), moderate-vigorous resistance exercise | ||
| Risk Level of Activity Low |
||||
NOTE: Risk level is based on levels of impairment described by Alfano et al., 2016.
a See https://www.ncbi.nlm.nih.gov/pubmed/26473759 (accessed September 20, 2017).
SOURCES: Basen-Engquist presentation, February 13, 2017; Basen-Engquist, et al., 2017. Agenda for translating physical activity, nutrition, and weight management interventions for cancer survivors into clinical and community practice. Obesity 25:S9–S22. With permission from Wiley.
| Least Restrictive Alternative | ||||
|---|---|---|---|---|
| Risk Level of Survivor | Patient/survivors experiencing cachexia, severe sarcopenia | Consult with physician/dietitian | Dietitian counseling | Dietitian counseling |
| Morbidly obese (BMI ≥ 40, or ≥ 35 with obesity-related comorbidities) | Community/home-based program | Consult with physician/dietitian | Dietitian counseling | |
| Overweight or obese | Community/home-based program | Community/home-based program | Dietitian counseling | |
| BMI < 25 | Community/home-based program | Community/home-based program, short-term only, for weight gain prevention | Not recommended | |
| Healthy guidelines-based nutrition/eating behavior | Weight loss program, 1–2 pound(s) loss per week | Weight loss program, targeted loss > 2 pounds per week | ||
| Degree of Risk in Activity Low |
||||
NOTE: BMI = body mass index.
SOURCES: Basen-Engquist presentation, February 13, 2017; Basen-Engquist, et al., 2017. Agenda for translating physical activity, nutrition, and weight management interventions for cancer survivors into clinical and community practice. Obesity 25:S9–S22. With permission from Wiley.
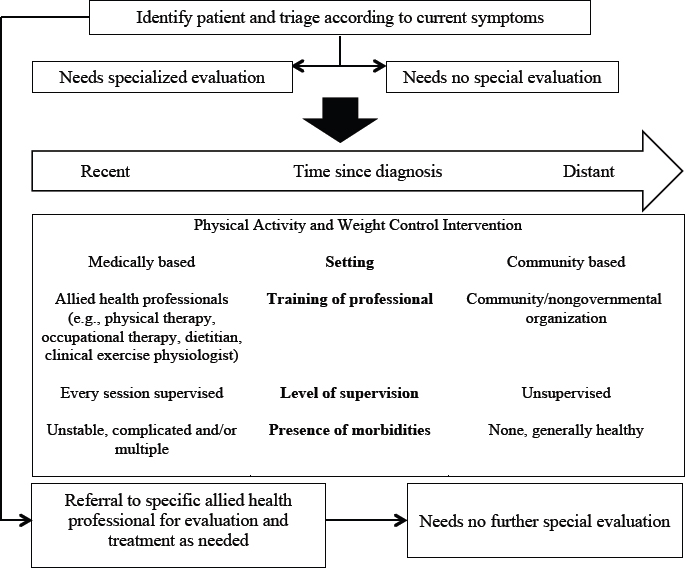
SOURCES: Eakin presentation, February 13, 2017; Translational Behavioral Medicine, Exercise for breast cancer survivors: Bridging the gap between evidence and practice, 1, 2011, 539–544, Hayes, S. C., K. Johansson, C. M. Alfano, and K. Schmitz, 2011. With permission of Springer.
reporting one or more conditions that were graded as moderate to severe. Of all 290 conditions reported 28 percent were possibly, probably, or definitely related to the intervention, including 35 cases of lymphedema.12 “Some amount of integration into clinical care is really essential as we try to implement these remote interventions to cancer survivors who probably carry a higher level of comorbidity,” Befort said.
Basen-Engquist noted that clinicians tend to overlook patient preferences and experiences when referring to exercise and weight management interventions. “We need to think not just about patient risk stratification,
___________________
12 Unpublished data.
but also what the patient’s interests and experiences are and make it a very patient-focused experience, rather than just, ‘This is what we think you should do,’” Basen-Engquist stressed. She noted that the American College of Sports Medicine (ACSM) recently revised its screening recommendations for exercise to encourage clinicians to consider a patient’s desired exercise intensity level (Riebe et al., 2015). Ligibel added that the best exercise for patients is the one that they will do. “Whatever programs we build have to be flexible,” she said.
Robert Boone, president and chief executive officer of the Medical Fitness Association, added that there is abundant anecdotal evidence in cancer wellness programs across the United States and in other countries that patients do better with personalized programs that are based on their health status, health risks, and personal goals. “True personalized medicine is a partnership with the patient within the continuum of care to look individually at what each person’s needs are,” he said. Dixon agreed that it is critical to individually tailor programs based on patient preferences. “We really need to meet patients where they are. . . . It [needs to be] an individualized approach,” she said.
Cost and location are two important factors that should be considered when making referrals to lifestyle intervention programs, Schmitz pointed out. “People will travel an hour or two to get high-quality curative cancer care, but they [may not] travel in order to go to a [recreational facility] that is too far away. They are going to want something really close to their home,” she said. Schmitz added that “Cost is a huge issue,” and noted that there are often high copays for physical therapy. In addition, clinicians often do not know which insurance plans will cover the various aspects of programs to which they refer patients.
Basen-Engquist added that lifestyle intervention programs should take a more active role to ensure that referred patients make it to these programs. “Many people are referred to programs by their health care system, but that does not mean they are all getting there.” As an analogy, she reported on a smoking cessation intervention that evaluated two different referral approaches: the first approach was to provide people who smoke with a referral card to a telephone-based quit line and encourage them to call; the second approach was to request permission for the quit line to proactively call the people who smoke (Vidrine et al., 2013). The more proactive referral approach resulted in a 13-fold increase in the number of people enrolling in smoking cessation treatment (Vidrine et al., 2013). “You have to be a little more active than just handing somebody a referral,” Basen-Engquist
said. Schmitz noted that for the Strength After Breast Cancer program (see Box 3), active follow-up of an oncologist’s referral by a physical therapist was critical to the adoption of the program by breast cancer survivors.
Approximately 70 percent of the women who received an active follow-up adopted the program, compared to only 35 percent of the women who received a paper referral.
Delivery Methods
A number of speakers described different methods of delivering interventions for weight management and physical activity, such as clinic-based, community-based, or home-based programs, programs facilitated through health information technology, and hybrid models. Basen-Engquist said that clinic-based programs provide more supervision and often are reimbursed, especially if they are considered rehabilitation programs; however, she said that nutrition programs are often not covered by insurance. Community-based programs tend to have more face-to-face interactions at community institutions, such as the YMCA (see Box 4). Home-based programs can include a variety of formats, such as print materials, videos, smartphone apps, wearables, social media interventions, and telephone and video-conference coaching.
Whether cancer survivors would benefit more from having a program tailored to their cancer-specific needs or a more broad-based weight management or physical activity program has not been sufficiently investigated yet, Basen-Engquist said, and the determination may depend on the health condition of the survivor, the risk level of the activity, and the survivor’s comfort level with that activity. Eakin compared two Australian lifestyle intervention programs (one designed for cancer survivors and the other for the general population) (see Box 5). She found similar outcomes for both programs, “which suggest that both types of programs can be safe and effective [for cancer survivors] when appropriate screening is conducted.” Eakin added that both types of programs will probably be needed to address the health-promotion needs of the growing population of cancer survivors, but that cancer-specific programs will always have an important role.
Thomson described advantages and disadvantages of diet interventions in cancer survivors by delivery method (see Table 3). She said that group counseling and one-on-one coaching tend to be more effective than telephone-based interventions. However, she noted that in-person interventions are more burdensome to patients and more costly to provide.
Pinto reported on the advantages and disadvantages of different methods of delivering exercise interventions. A meta-analysis of studies that evaluated how well various programs changed participants’ exercise
behaviors found that although the more structured programs produced larger effects, interventions delivered by phone or email were also effective (Bluethmann et al., 2015b). “More is not necessarily better and we have to keep this in mind when we are thinking about how costly these interventions are,” she said.
She noted that with clinic-based programs, investigators can conduct baseline exercise tests that indicate the most appropriate exercise regimens for participants. In off-site (or home-based) programs, Pinto said that these baseline assessments rarely occur, and they tend to prescribe lower-intensity physical activity, but they can often be longer than on-site
programs. Pinto added that most clinic-based programs are designed for groups and tend to select motivated individuals willing to overcome scheduling and travel issues. She contrasted this with off-site programs, in which individuals generally exercise on their own rather than as part of a group, that tend to reach a broader participant population. Pinto emphasized that delivery of the intervention is more consistent and patient adherence is better with clinic-based programs, but they tend to be more expensive than off-site programs.
Wolin noted that options for home-based intervention programs have expanded rapidly due to innovations in technology. “There are a lot of companies offering these telemedicine services, so the challenge is how to connect those resources to clinicians,” Wolin said. She described ScaleDown, a weight loss intervention delivered via text messaging synched with a smart scale (see Box 6). Befort and Stolley said there was a need for more studies to assess the effectiveness of web or mobile phone interventions and how they compare to interventions that involve face-to-face contact. “We have gotten into the mindset that we need to have remote, direct-to-consumer interventions, but if you talk to patients, a lot of them will tell you they really appreciate some level of face-to-face contact,” Befort said.
Ligibel noted that new technologies, including Fitbit and other wearable activity sensors, may better enable distance-based interventions. “They can connect health coaches with patients in ways that were never possible before. Coaches can see what patients are doing and can look at heart rate, blood pressure, and other data,” Ligibel said. She pointed out that Internet-based platforms such as Skype can be used to coach patients from afar and enable, for example, a trainer to supervise his or her patients at a distance and ensure they are not endangering themselves while exercising. “We also have to recognize the limitations of this technology and not think that it can take the place of the types of interventions shown to be successful at helping people incorporate behavior change,” Ligibel said. Home-based delivery may also be effective for maintenance of physical activity behavior change once patients have participated in initial in-person interventions, Ligibel noted.
Harrison praised the Skype-based intervention with an exercise physiologist he received at home. “This person whom I have never met was able to communicate with me, show me things to do, and observe my reactions to what she was saying. It was simple technology that works wonderfully and did not cost anything or take an awful lot of time. Skype made it very easy to stay engaged with her.” he said, adding that his exercise physiologist
TABLE 3 Delivery Modalities Using Diet with and Without Physical Activity for Weight Loss
| Delivery Mode | Advantages | Disadvantages | Cancer Type Targeted |
|---|---|---|---|
| Clinically-based/supervised (1:1 with or without groups) | Treatment fidelity Safety Integration with care delivery |
Cost Participant burden Access barriers |
Mixed |
| Community-based/diverse populations | Reach Dissemination |
Fidelity | Breast |
| Commercial programs | Availability Normalization Fidelity* |
Cost Access in rural areas |
Breast, Colorectal |
| Home-based | Privacy Cost savings |
Safety* | Breast, Prostate, Colorectal |
| Telephone/group phone | Cost savings Geographic reach Social support |
Less personal Safety* |
Breast |
| Technology-based | Reach/dissemination Cost savings Social support using social media |
Familiarity with technology, access IT support required Safety* |
Childhood Endometrial |
| Print materials | Reach/dissemination Cost savings |
Safety* | Breast, Prostate |
| Mixed-delivery modalities | Reach/dissemination Cost savings |
Safety* | Breast Endometrial |
NOTE: BMI = body mass index; DAMES = Daughters and Mothers Against Breast Cancer; ENERGY = Exercise and Nutrition to Enhance Recovery and Good Health for You; IT = information technology; LEAN = Lifestyle, Exercise, and Nutrition; LISA = Lifestyle Intervention in Adjuvant Treatment of Early Breast Cancer; RENEW = Reach out to ENhancE Wellness; STONE = Survivors Taking on Nutrition and Exercise; SUCCEED = Survivors of Uterine Cancer Empowered by Exercise and Healthy Diet; WW = Weight Watchers.
* The safety of these approaches has not been robustly evaluated, but generally these approaches are safe in a medically cleared patient sample.
SOURCE: Thomson presentation, February 13, 2017.
| Weight/BMI Change | Examples of Survivorship Interventions |
|---|---|
| Range 2–9 kg at 6 months; 5 kg at 12 months | CHOICE, Sedlacek et al., 2011; Campbell et al., 2012; Get Fit for the Fight, Swisher et al., 2015; SUCCEED, McCarroll et al., 2014; Travier et al., 2013; Harris et al., 2013 |
| −2.6 kg versus −1.5 kg at 12 months with active trial | Cocinar Para Su Salud, Greenlee et al., 2015; Moving Forward, Stolley et al., 2015 |
| −2.6 kg WW; −9.4 kg WW + 1:1; +0.85 kg control −3.3 (±3.5)% versus −1.8 (±2.9)% |
Weight Watchers, Djuric et al., 2002; Curves, Greenlee et al., 2013 |
| −2.06 kg versus −0.92 kg BMI at 12 months: −1.4 mom; −1.38 daughter |
RENEW, Morey et al., 2009; DAMES, Demark-Wahnefried et al., 2014 |
| −5.4% telephone versus −2.0% usual −4.8 kg telephone versus −1.7 kg usual −5.3 versus 0.7% (6 months)/−3.6 versus 0.4% (12 months) |
LEAN, Harrigan et al., 2016; Befort et al., 2014; LISA, Goodwin et al., 2014 |
| Over 14 years, less weight gain −6.4 kg at 6 months |
Fit4Life, Huang et al., 2014; McCarroll et al., 2015 |
| −0.3 versus +0.1 BMI No change |
FRESH START, Demark-Wahnefried et al., 2007; Park et al., 2016 |
| 6% versus 1.5% control (12 months) −3.5 kg versus +1.4 kg −0.8 kg versus +0.2 kg |
ENERGY, Rock et al., 2015; von Gruenigen et al., 2008; Stepping STONE, Sheppard et al., 2016 |
also communicated with him via email to make adjustments to his activities. Schmitz is currently developing an interactive tablet-based program for rural women with metastatic breast cancer. However, she pointed out that not all rural participants have access to Wi-Fi, and this program will use devices with cellular data plans. Basen-Engquist added she has started recruiting endometrial cancer survivors to a weight loss and physical activity program that uses information and coaching provided via Facetime on an iPad.13 This technology will also enable coaches to observe participants as they complete resistance training. However, Wolin cautioned that conducting lifestyle interventions through the use of programs like Skype can pose compliance challenges with the Privacy Rule promulgated under the Health Insurance Portability and Accountability Act of 1996 (HIPAA).
Promotion of Healthy Behaviors
Pinto said that many healthy lifestyle interventions are not based on any theoretical frameworks for inducing behavior change; however, of the interventions that are based on theoretical frameworks, social cognitive theory has been employed the most often (Loprinzi and Lee, 2014; Pinto and Ciccolo, 2011). In intervention studies that have used this framework, small to moderate improvements in physical activity and diet changes have been seen (Stacey et al., 2015). Pinto said that a key construct of this theory is perceived self-efficacy, or the confidence that a person can make changes in his or her own health habits. People who report higher self-efficacy tend to increase their physical activity faster than those with lower self-efficacy (Pinto and Ciccolo, 2011; Stacey et al., 2015). “Getting people to feel more confident about what they are doing and teaching them the skills to maintain it seem[s] to play a key role,” she said.
Pinto described additional factors contributing to successful intervention programs to promote healthy behaviors, including helping cancer survivors with goal setting and strategies for monitoring progress (e.g., wearables and scales). Wolin emphasized that self-monitoring is the cornerstone of successful behavior change, particularly for weight loss. Pinto added that providing individuals with social support and behavioral and cognitive skills are also evidence-based strategies, but these factors have not been well-studied in cancer survivors. Denlinger described additional
___________________
13 See https://www.smartpatients.com/trials/NCT02774759 (accessed April 26, 2017).
facilitators of health behavior changes (Aycinena et al., 2017; Baker et al., 2015; Beehler et al., 2014), such as:
- A positive perception of health behavior changes;
- Small, achievable goals;
- Self-motivation and readiness to change;
- Easy access to exercise facilities, healthy foods, or weight loss services;
- Supportive clinicians, family members, and friends;
- Hearing health messages from multiple clinicians; and
- Having a workout partner.
Thomson described approaches to help participants stay engaged in healthy behavior changes, including multimodal communications strategies, prizes associated with adherence challenges, self-monitoring, and reporting back of behavior changes (Delahanty et al., 2016; Goldberg and Kiernan, 2005; Warner et al., 2013).
Catherine Alfano, vice president of survivorship at the ACS, asked what can be done to empower patients to make healthy behavior changes. Wolin suggested repeated messaging strategies: “We cannot expect a single message in a clinical encounter to deeply resonate and engage the patient. That message has to be reinforced over and over again,” she said.
Scherezade Mama, assistant professor in kinesiology at The Pennsylvania State University, agreed that repeated messaging is important, and added that hearing this message from physicians is critical. “If we do not have the physician echoing the message, it [can] undermine the work that we are doing in the community,” she said. Buzaglo added that some research supports the notion that a message from a physician can prompt behavior change (Pool et al., 2014; Rose et al., 2013). Denlinger said that oncologist recommendations may help motivate patients to participate in healthy behaviors (Jones et al., 2004), but noted that these recommendations may not be sufficient for behavior change. A study comparing an oncologist’s recommendation to increase exercise, an oncologist’s recommendation to exercise plus a motivational package (exercise DVD, pedometer, education, and a diary), and usual care among breast and colorectal cancer survivors found that the oncologist’s recommendation alone did not improve the exercise levels (Park et al., 2015). However, participants who received the motivational package along with the oncologist’s recommendation did increase their exercise levels. “So for survivors, it may not be enough to just say you should do this. We may need to be doing more,” Denlinger said.
Ligibel agreed that messaging is important, and added that “we also need to have an infrastructure to help them do it.” She said that having convincing evidence that behavior changes will lower risk of cancer recurrence or mortality would make it easier to motivate and engage patients. “Until we really have a higher level of evidence, it is going to be hard to get the full level of engagement of both patients and providers that we need in order to help people make these changes,” she said. However, Denlinger noted that there is abundant high-quality evidence that diet and physical activity can improve outcomes in patients with prediabetes, yet many primary care clinicians do not make these recommendations to patients at risk for diabetes. “To assume that getting comparable evidence in the oncology setting is going to lead oncologists to act in a different way than primary care doctors act with regard to prediabetes is an assumption that is probably subject to some questions,” she said.
Matt Longjohn, national health officer and vice president at the Y,14 said that motivational interviewing is a “powerful tool” in helping people make small, sustainable, incremental changes. For example, if a patient said that she was only able to exercise for 5 minutes the previous week, instead of responding that she is not meeting the standard 150 minutes per week, the clinician could instead respond, “That is great. How did you get to 5 minutes and how might you get to 6?,” Longjohn said. Thomson said a systematic review found that motivational interviewing of cancer survivors was effective for a variety of lifestyle behaviors (Spencer and Wheeler, 2016). She added that while motivational interviewing is widely applied in telephone-based interventions, it has not been as widely used in the clinic, unless it is for a one-on-one counseling session. However, Thomson noted that motivational interviewing may not be effective in all populations. “You cannot necessarily take what works very effectively in non-Hispanic white women and directly translate it to other ethnic groups,” she said.
Tailoring Interventions to Special Populations
Befort, Demark-Wahnefried, and Stolley discussed the importance of tailoring interventions to meet the needs of diverse cancer survivors, including racial and ethnic minorities, children, older adults, and rural populations. The needs of adolescents and young adults were addressed in a previous NCPF workshop (IOM, 2013).
___________________
14 As of the time of publication, Matt Longjohn is no longer at the Y.
Racially and Ethnically Diverse Populations
Stolley said that some population subgroups in the United States have larger cancer burdens, as measured by cancer incidence, prevalence, mortality, and survivorship, and that worse outcomes are more common among low-income populations and racial/ethnic minorities (King et al., 2010; NCI, 2008). She added that there also is a higher incidence of obesity among cancer survivors who are Hispanic and African American compared to whites (Greenlee et al., 2016). In several studies, cancer survivors who are minorities have reported low adherence to physical activity and nutrition guidelines and high rates of obesity-related comorbidities. They also are more likely to report fair- to poor-health status compared to people who have not been diagnosed with cancer or non-minority cancer survivors (Ansa et al., 2016; Dennis Parker et al., 2013; Nayak et al., 2015; Nichols et al., 2009; Paxton et al., 2012; Stolley and Fitzgibbon, 1997; Tammemagi et al., 2005; Weaver et al., 2013a).
Stolley suggested considering the environmental context of communities and how that influences their risk for obesity and cancer (see Figure 2). She noted that minority communities are more likely to face adverse living conditions that are associated with the risk of being overweight or obese, including high segregation, low neighborhood socioeconomic status, high crime rates and traffic density, high numbers of fast food restaurants, a lack of full-service supermarkets, and a lack of access to safe, affordable exercise options (Mellerson et al., 2010; Shariff-Marco et al., 2017; Zenk et al., 2005). “When we tell patients to eat healthy and exercise more, we need to think about where they are going to be doing these things,” Stolley said. In addition, she suggested using a biopsychosocial framework that integrates biological, behavioral, and community-based research to understand and address cancer disparities (Warnecke et al., 2008).
Cultural context is also important, she added, because it influences perceptions of disease, belief systems related to health, help-seeking behaviors, attitudes toward health care, and use of alternative therapies (Kreuter et al., 2003; Lim et al., 2009; Lindberg et al., 2013; Whitt-Glover et al., 2014). Stolley suggested that lifestyle interventions should be culturally appropriate; for example, culturally appropriate music should be offered for exercise sessions and educational materials should be offered in the dominant language of the community. Stolley also suggested researchers involve the community when developing an intervention program and recruit program staff who are representative of the communities they serve (Kreuter et al.,
NOTE: CVD = cardiovascular disease.
SOURCES: Stolley presentation, February 13, 2017; Golden, S. H., A. Ferketich, J. Boyington, S. Dugan, E. Garroutte, P. G. Kaufmann, J. Krok, A. Kuo, A. N. Ortega, T. Purnell, and S. Srinivasan. Transdisciplinary cardiovascular and cancer health disparities training: Experiences of the Centers for Population Health and Health Disparities. American Journal of Public Health 105(Suppl 3):S395–S402, 2015, American Public Health Association.
2003). She added that cultural competency training is needed for clinicians and researchers working with diverse cancer survivors. She also suggested incorporating hands-on learning opportunities in interventions, such as demonstrations of how to prepare traditional foods in healthier ways. To encourage attendance, activities could be scheduled in conjunction with other community, religious, or social events. Stolley added that opportunities for interventions to engage friends and family as social support can aid behavioral change and maintenance of these behaviors. She also suggested creating links between community organizations and clinical practices providing cancer survivorship care, because survivors want resources in their own communities (Crookes et al., 2016; Stolley et al., 2006; Whitt-Glover et al., 2014).
Basen-Engquist asked how general intervention programs could be tailored to meet the needs of diverse populations. Stolley noted that the same basic weight loss intervention could be used in different populations but could be adapted depending on the needs of the population being served. “You could have the same basic weight loss intervention for a 40-year-old with three kids at home and an [older adult], but you are going to contextualize that differently for those populations. You have to tailor it to each individual and what works for them,” she said.
Lucile Adams-Campbell, associate director for minority health and health disparities research at the Georgetown Lombardi Comprehensive Cancer Center, pointed out that such contextualizing can be difficult to achieve because there is a lack of diversity in the health care community. “We do not have diverse clinicians coming down the pipeline, and that will continue unless we can figure out how to solve that problem,” she said. Adams-Campbell added that diversity is also needed in academia.
Tasha Moses, president and managing principal at Strategic Management Services, raised the question of how to continue engaging minority populations who participate in research. “Have you looked at some social service organizations of churches or other types of organizations in the community that you could potentially partner with and bring them on board to continue the work?” she asked. Adams-Campbell said that when she first started conducting research at Georgetown University, she set up a community-based office “so we are part of the fabric of the community and do not give the impression of doing helicopter science, which is the biggest negative thing you can ever do to minority communities.” She engages with the community through community advisory boards and her patient navigation program, which help recruit patients for many studies. “Having community people sitting at the table and hiring them enables us to be totally engaged with the community,” she said, adding that sometimes the tables are turned and the community comes to her asking that she conduct research on a topic of concern to its members. “We must really serve the community,” she said.
Befort added that a few of the women who participated in her studies are now are on her patient advisory board. “We are trying to keep that patient activation and advocacy going in our region,” she said. Stolley agreed, and noted that it is important to disseminate research findings within the communities where the research is conducted. “We have a responsibility to disseminate the results of our studies, and not just through medical journals.” She noted that with the help of her community advisory panel, she distributes newsletters and holds semi-annual town hall meetings
at libraries across Chicago that are attended by the women who participated in her studies and their families or other interested community members (Stolley et al., 2015). At those meetings, “we talk about what we have found and what we hope to do,” she said. Stolley added that her next step is to conduct a dissemination and implementation study in public recreational systems in Chicago and Milwaukee because “these recreational systems are very eager to bring this program on.”
Children and Older Adults
Demark-Wahnefried noted that older adults (65 years or older) comprise 62 percent of all cancer survivors; with the aging of the population, earlier diagnosis, and improvements in treatments, she said that the older adult cancer survivor subpopulation will continue to grow (NCI, 2016). At the same time, there are almost 400,000 childhood cancer survivors, many of whom have long lives ahead of them. She noted that lifestyle interventions need to be tailored to both ends of the age spectrum, especially given the prevalence of suboptimal diets and insufficient physical activity among both populations (40–70 percent of childhood cancer survivors and 52–85 percent of older adult cancer survivors have suboptimal diets; 54–84 percent of childhood cancer survivors and 53–70 percent of older adult cancer survivors are insufficiently physically active) (Blanchard et al., 2008; Ford et al., 2014; Niu et al., 2015; Zhang et al., 2012).
Demark-Wahnefried said that up to 71 percent of older adult cancer survivors are overweight or obese. The prevalence of obesity has not increased among childhood cancer survivors except among those diagnosed with brain cancer or acute lymphoblastic leukemia (Huang et al., 2014; Wilson et al., 2015). However, childhood cancer survivors are more likely to have a lack of muscle mass and strength (sarcopenia) compared to their peers (Henderson et al., 2014). “Even though we are not seeing obesity by BMI standards, we are seeing adverse body composition change” among childhood cancer survivors, Demark-Wahnefried said. In addition, the risk of sarcopenia increases with age, and cancer and its treatment can exacerbate sarcopenia in older adult cancer survivors, Demark-Wahnefried said (Henderson et al., 2014; Muscaritoli et al., 2010). She added that physical activity can reduce the risk of sarcopenia, but weight loss can worsen it (Henderson et al., 2014; Muscaritoli et al., 2010).
Demark-Wahnefried defined frailty as an insufficient physiologic reserve that is associated with increased susceptibility to diseases and dis-
abilities (Buchner and Wagner, 1992; Henderson et al., 2014). She noted that cancer and its treatment exacerbate the decline into frailty (Henderson et al., 2014). In addition, Demark-Wahnefried said, “We all become frail over time, and if you follow a poor diet, smoke, or are inactive, you can exacerbate that decline into frailty.” Although frailty is often associated with someone of slight build, people who are overweight or obese can also be frail. For example, a study of breast cancer survivors found that the odds of frailty increased among those with higher BMIs (Bennett et al., 2013). Lifestyle interventions that promote muscle growth and weight control can help mitigate sarcopenia and frailty among older adult and childhood cancer survivors, Demark-Wahnefried said.
She stressed that clinicians need to give special consideration to childhood and older adult cancer survivors when devising weight management and exercise interventions. For example, children who are on stringent diets may be susceptible to stunting and the development of sarcopenia. Thus, for children, clinicians recommend “growing into their weight,” or more behavioral approaches, such as substituting water for soda, adopting slower rates of eating, or avoiding distracted eating (Barlow, 2007). For children with significant obesity, a diet with a modest energy deficit is generally recommended (e.g., one that will promote weight loss of approximately a half a pound per week), Demark-Wahnefried noted. Similarly in older adults, concerns of sarcopenia and functional declines have led to recommendations for low-intensity diet interventions (e.g., energy restriction of 500 to 750 calories per day, rather than the 500 to 1,000 calorie-deficit diets), and combining those diets with resistance training (Villareal et al., 2005).
Apovian noted that the American Academy of Pediatrics recommends weight management in children based on their BMI, diet, physical activity, family history, and other health conditions (Ireland et al., in press). She stressed that lifestyle interventions that involve the entire family are more successful in pediatric populations. Dietz said that the Agency for Healthcare Research and Quality recommends a family-based, multicomponent behavioral treatment approach with medical oversight for treating obesity in children (Wilfley et al., 2017).
Demark-Wahnefried said the ACSM recommends that older adults perform resistance training two to three times per week, noting that physical activity has been found to increase strength and physical function in this population (Klepin et al., 2013). Apovian stressed that exercise in older adults is important not just for weight loss and weight maintenance, but also to preserve lean body mass. This requires resistance training, she said,
adding that the ACSM recommends participation in a regular exercise program as an effective intervention to reduce or prevent a number of functional declines associated with aging (Mazzeo et al., 1998). One small study found that over a 12-year period, seven healthy men lost approximately 15 percent of their muscle strength/area on average, but that almost all of that could be regained by completing an intensive 12-week resistance training program (Roubenoff, 2000).
For children, the Council on Sports Medicine and Fitness recommends engagement in strength training under supervision and within a sports curriculum (McCambridge and Stricker, 2008). Demark-Wahnefried said few studies have assessed the effects of resistance training in children or older adult cancer survivors. One review found trends suggesting that exercise improved body composition, flexibility, cardiorespiratory fitness, muscle strength, and health-related quality of life among childhood cancer survivors (Braam et al., 2016). But Demark-Wahnefried said, “We cannot say definitively that exercise is helpful for this group.” Another study found that light-intensity physical activity is helpful for older adult cancer survivors (Blair et al., 2014).
To tailor weight management and physical activity interventions for childhood and older adult cancer survivors, Demark-Wahnefried said that remote programs or programs with transportation support are critically needed. Demark-Wahnefried also suggested using low-literacy materials with a large font size, volume controls, and buttons that are large enough to press easily. Both of these groups can also benefit from having caregivers help in delivering the intervention. She said that game- and play-based interventions are appropriate for pediatric populations, whereas the older adults prefer holistic programs that have meaning and involve others. Interventions for both populations should be tailored to the treatment-related conditions they have, she said.
Demark-Wahnefried said that a small study of a weight management intervention in childhood survivors of acute lymphoblastic leukemia showed the intervention to be safe and effective in preventing weight gain (Huang et al., 2014). She also said the RENEW study found that a diet and exercise intervention in older survivors of colorectal, breast, and prostate cancer helped limit the decline in physical function (Demark-Wahnefried et al., 2012; Morey et al., 2009).
Rural Populations
Befort noted that approximately 20 percent of the U.S. population resides in a rural location,15 including 2.8 million cancer survivors (U.S. Census Bureau, 2015; Weaver et al., 2013b). Compared to the U.S. population, rural residents are more likely to be non-Hispanic white (78 percent of rural residents versus 64 percent of the U.S. population), but there are some areas of the country with high proportions of rural minorities, including Hispanics in the Southwest and African Americans in the southern part of the United States, she said. Befort said that rural residents also tend to be older, have a lower income, and have less education than their urban counterparts (U.S. Census Bureau, 2016). She added that rural residents tend to share some common cultural elements, including conservatism, self-reliance, and an orientation toward work, family, and religion (Slama, 2004). In addition, she said rural residents tend to have lower income and are more likely to rely on self-employment (Goetz, 2008), so it can be more financially burdensome for them to take time off of work to see health care professionals.
People who reside in rural areas have higher rates of cancer mortality compared to urban residents across all regions in the United States; in addition, Befort said that cancer mortality rates have declined more slowly in rural counties than in metropolitan counties (Garcia et al., 2017). Rural cancer survivors report poorer health status, more psychological distress, higher rates of depression and anxiety, and greater gaps in understanding about cancer and the effects of cancer treatment (Burris and Andrykowski, 2010; Weaver et al., 2013b). They also report high levels of unmet support needs (Wilson et al., 2000).
Access to cancer care, including survivorship care, is especially challenging in rural America; Befort noted that approximately 3 percent of medical oncologists (Kirkwood et al., 2014), 16 percent of radiation oncologists (Lewis and Sunshine, 2007), and 3 percent of social workers practice in
___________________
15 According to the U.S. Census Bureau, the “urban-rural classification is fundamentally a delineation of geographical areas, identifying both individual urban areas and the rural areas of the nation. The Census Bureau’s urban areas represent densely developed territory, and encompass residential, commercial, and other non-residential urban land uses. . . . The Census Bureau identifies two types of urban areas: Urbanized Areas (UAs) of 50,000 or more people; Urban Clusters (UCs) of at least 2,500 and less than 50,000 people. ‘Rural’ encompasses all population, housing, and territory not included within an urban area.” See https://www.census.gov/geo/reference/urban-rural.html/geo/reference/urban-rural.html (accessed August 31, 2017).
rural areas (Center for Workforce Studies, 2006). Consequently, rural residents typically have to travel farther and longer to receive specialty cancer care (Onega et al., 2008).
Physical activity levels tend to be suboptimal in rural cancer survivors. In one study of rural breast cancer survivors, 54 percent reported sedentary behaviors, and only 19 percent reported meeting physical activity recommendations (Rogers et al., 2009). Most reported they preferred to exercise at home or outdoors, and only one out of five wanted to go to an exercise facility (Rogers et al., 2009), probably because of the distance to reach one, Befort noted. She also said that, “There are just not the same cultural norms for leisure time physical activity, walking for exercise, or walking for transportation in rural areas.”
Obesity is also more prevalent in rural settings and rural Americans tend to eat a diet higher in fat and are less likely to exercise (Befort et al., 2012). The diets of rural cancer survivors also tend to include more sugar-sweetened beverages and have higher amounts of sweets and starchy vegetables, whereas urban cancer survivors have a broader range of components in their diet, including nuts, grains, and vegetables, Befort reported (Miller et al., 2012).
In a survey, approximately half of rural breast cancer survivors who were overweight or obese reported that they wanted to lose weight, but two-thirds reported doing so without assistance (Befort et al., 2011). Thus, Befort said, there is a need for lifestyle interventions among rural cancer survivors, and added that remote-based interventions are essential to maximize reach into rural communities, due to challenges with access to health care services, travel time, and financial barriers. She noted that only one trial has been published of a lifestyle intervention conducted exclusively in a rural setting (Befort et al., 2015). In this study, rural breast cancer survivors were enrolled into a telephone-based intervention delivered via weekly conference calls. Those in the intervention group achieved a 12.9 percent weight loss, with a 10.6 percent weight loss maintained at 18 months (Befort et al., 2016; Fazzino et al., 2016). While the intervention was exclusively home-based, Befort noted that many participants made arrangements to meet in person with a care provider, indicating that some face-to-face contact may enhance intervention efficacy.
EXAMPLES OF CHALLENGES IN IMPLEMENTING LIFESTYLE INTERVENTIONS
A number of participants at the workshop described some of the challenges to implementing lifestyle interventions in clinical practice, including factors associated with clinicians and patients, as well as from infrastructure and capacity.
Clinician Factors
Although guidelines from groups such as the ACS and NCCN are relatively straightforward regarding physical activity and weight management recommendations for cancer survivors (Rock et al., 2012), Denlinger noted that they can be difficult to implement within the oncology practice setting. “They are in theory very easy to talk about and endorse, but I can tell you as a clinician, that is not always the case [in practice],” Denlinger said.
For example, Denlinger conducted a patient chart review of adherence to NCCN disease-specific survivorship guidelines for colorectal cancer and non-small cell lung cancer, and found that clinicians documented physical activity and healthy diet counseling in fewer than 20 percent of colorectal cancer survivors and approximately 30 percent of lung cancer survivors (Denlinger et al., 2015). In a survey, more than half of oncologists said that physical activity is beneficial, important, and safe during cancer treatment; however, during the previous month, oncologists reported recommending exercise to less than one-third of their patients (Jones et al., 2005). A survey of cancer survivors found that few (10 percent) reported discussions or recommendations for diet or exercise with their clinicians, Denlinger said (Sabatino et al., 2007).
She pointed out that oncologists often have many things to discuss during a 15- to 20-minute office visit, especially early in the cancer care trajectory when oncologists are working with patients to determine a treatment plan, assess and address the side effects of cancer treatment, and provide psychosocial support. Even during survivorship care visits, Denlinger said, there are competing demands on time, including discussions about cancer screening and surveillance, and late- and long-term adverse effects of cancer and its treatment. Nekhlyudov agreed that primary care clinicians taking care of cancer survivors lack the time needed to discuss healthy lifestyles with their patients.
Pinto noted that due to lack of time and/or expertise, there are multiple missed opportunities to discuss healthy lifestyles throughout the cancer care continuum. Goodwin pointed out that the reluctance of clinicians to counsel patients on lifestyle interventions “might reflect that they have to do many things, so they focus on what they are good at, and they are not good at this. I do not think they are resistant to having these discussions, but they need someone to refer to,” she said.
Nyrop added that clinicians vary in terms of their willingness to have a conversation with their patients about physical activity or weight management. She suggested that further research be conducted to understand what clinicians are willing to recommend, even if it is a very brief suggestion that patients consider increasing physical activity. Pinto agreed that patients need to hear from clinicians that healthy behaviors are important; likewise, she said that clinicians need resources and programs to which they can refer their patients. “This takes the burden off the provider, but at least the message is heard by the patients,” Pinto said. Stolley agreed that “if we can offer oncologists something to refer their patients to, they will be very eager for that because patients are asking them for information and resources.”
Dietz added that clinicians need information about the available lifestyle intervention programs in their community. “It is a real barrier if we do not know what the community resources are to which we can refer patients,” he said. Denlinger agreed, noting that local resources can be hard to find. Ligibel suggested that clinicians be provided with a list of resources or programs that could help patients with weight management and physical activity. Schmitz agreed, and reiterated that more work is needed to improve the referral and triage process to ensure that patients are referred to an appropriate program that will meet their needs.
“Given there is such a diversity of programs and approaches that could be appropriate for a [cancer] survivor, how does a particular oncology clinician know where to refer a patient, especially if he or she is based in a tertiary cancer center that serves patients coming from a very broad geographic area?” asked Basen-Engquist. She suggested having consistent standards for a variety of programs so oncologists can be comfortable and confident in referring their patients to specific programs.
Dietz added that lack of expertise in obesity, weight management, and physical activity extends to primary care clinicians. He said that fewer than half of those surveyed knew that the recommended level of physical activity was 150 minutes per week; only one-third knew that multiple types of
diets could be used for weight loss.16 In addition, few knew that the U.S. Preventive Services Task Force (USPSTF) recommends between 12 and 26 counseling sessions for the treatment of adults with obesity. “This lack of knowledge about treatment . . . is a general and pervasive problem,” Dietz said.
Patient-Centered Terminology for People with Obesity
Dietz said that many clinicians do not use appropriate language when discussing obesity and weight management with their patients. He emphasized that clinicians should recognize that obesity is a disease and not an identity. He said referring to people as patients with obesity (as opposed to obese patients) is less stigmatizing and “moves us away from blaming the patient. . . . We have to recognize that people do not decide to develop obesity. The obesity epidemic did not start 20 years ago because everyone decided to overeat and become less active. Those are consequences of the environment, and framing obesity as something other than an issue of personal responsibility relieves the patient of that responsibility, and enables the clinician to see obesity in a broader context that they might address.”
Dietz added that clinicians should use patient-centered terminology when discussing obesity and its management in order to mitigate the influence of stigma and discrimination. For example, clinicians could use the term “unhealthy weight” as opposed to “obesity,” as well as substituting “improved nutrition and physical activity” for “diet and exercise.” Dietz added that weight discrimination can have negative effects on health. One study found that people who experienced weight discrimination were 2.5 times more likely to become obese at follow-up and people who were obese at baseline were 3 times more likely to remain obese at follow-up (Sutin and Terracciano, 2013).
Dixon added that clinicians should acknowledge that losing weight is difficult, and noted that if losing weight was easy, two-thirds of the population would not be overweight or obese. “So step one with your patients is to acknowledge the difficulty of losing weight,” and provide them with the tools and support to succeed, Dixon said.
___________________
16 Manuscript submitted.
Patient Factors
Cancer survivors may also have competing priorities that interfere with weight management and physical activity, Nekhlyudov noted. “If the patient is struggling to pay his rent, the last thing on his agenda is weight loss,” she said. Denlinger said that cancer survivors’ other responsibilities can be a major barrier to exercise and weight management, as can poor access to safe exercise facilities, healthy food options, and nutritional expertise (Beehler et al., 2014). Schmitz pointed out that the cost of lifestyle intervention programs can also be a significant challenge for cancer survivors, especially if they are not covered by insurance.
Denlinger said that clinicians may also recommend unrealistic lifestyle changes or may overload cancer survivors with too many recommendations for changes at once. Dietz pointed out that clinicians need to recognize that “just because something is a priority on our part, that does not mean it is a priority on the patient’s part. We have to understand the contextual [factors] . . . in this patient’s environment.”
Denlinger said that the effects of cancer treatment can alter a patient’s ability to participate in diet and exercise changes and may make clinicians hesitant to recommend them. Buzaglo noted that patients often underestimate the side effects from their cancer treatment, such as neuropathy, that can be a barrier to adhering to physical activity guidelines.
Christopher Cogle, professor of medicine at the University of Florida College of Medicine, noted that many cancer survivors can experience strong, vacillating emotions that influence physical activity and weight management, including elation, anxiety, and depression. Dietz responded that physical activity is recommended for people with depression, and Rachel Ballard, director of Prevention Research Coordination at the National Institutes of Health (NIH), added that it is difficult for people to adopt healthy behaviors if their depression goes untreated.
Capacity and Infrastructure
A lack of program capacity or infrastructure can also make it challenging to implement lifestyle interventions. Befort said, “We need to continually think about capacity in rural clinical settings and potentially some of the downsides if we start moving more toward direct-to-consumer remote interventions.” Dietz added that even in non-rural areas, the capacity of clinicians to treat patients with obesity is insufficient given the current
prevalence of the condition. If you limit the range of obesity to those with a BMI of 35 or above, Dietz estimated that each primary care clinician would have approximately 164 patients with obesity. “There is a mismatch of the disease burden and provider capacity. . . . We need to think about care extenders or alternative modes of care delivery, such as the modification of the DPP, or the delivery of care through electronic methods,” Dietz stressed.
Elaine Trujillo, a nutritionist at the NCI, added that though there are nutrition services at cancer centers, there is a lack of nutritionists in outpatient care settings, including community oncology practices. Denlinger added that most patients with cancer are treated in community settings where they are less likely to have access to nutrition services. “Being able to refer people to a nutritionist would be a start, but a nutritionist who has expertise in working with oncology patients would be ideal,” Denlinger said.
POTENTIAL OPPORTUNITIES TO IMPROVE LIFESTYLE INTERVENTIONS FOR CANCER SURVIVORS
A number of workshop participants suggested opportunities to promote healthy lifestyles among cancer survivors, including
- integrating clinical and community-based programs;
- increasing patient awareness about the associations among obesity, physical activity, and patient outcomes, as well as the availability of lifestyle intervention programs;
- improving insurance coverage for lifestyle interventions;
- improving clinician education, standards, and resources related to lifestyle interventions;
- creating collaborations among organizations and clinicians; and
- improving research.
Integrating Clinical and Community-Based Programs and Resources
Dietz suggested utilizing a framework that integrates clinical and community-based systems of care in order to deliver weight management and physical activity interventions to cancer survivors (Dietz et al., 2015) (see Figure 3). This framework is centered on the patient and emphasizes individual empowerment and engagement. Dietz said, “Unless these community systems are supportive of what the care system is trying to achieve, we are not
SOURCES: Dietz presentation, February 14, 2017; Dietz et al., 2015.
going to succeed.” Community systems include a supportive environment and social norms, as well as resources and services. According to Dietz, the benefits of an integrated system would include improved patient and family engagement, better investments in the upstream determinants of health, support for value-based care, and the potential for improved patient outcomes and lower health care costs. “Providers need to be advocates for improved community services and resources,” Dietz said.
Eakin noted that collaboration among Australia’s state health programs and cancer councils was essential to the implementation and delivery of the two healthy lifestyle programs she discussed. She suggested advocating for such community-based programs in the United States that leverage funding from multiple sources. Stolley also stressed the importance of recognizing a community’s assets and resources—such as community gardens, Y programs, public recreation systems and programs, and community health workers—when considering lifestyle intervention programs. She advocated for improved access to and support of community-based programs for weight management and physical activity.
Increasing Patient Awareness
A number of workshop participants suggested that cancer survivors should be better informed about the associations among weight, physical activity, and patient outcomes, as well as the availability of lifestyle interventions programs. Ligibel said that a nationally representative household survey found that when asked about the most serious health effects of obesity and being overweight, a majority of respondents mentioned heart disease and diabetes; however, only 7 percent mentioned cancer (Tompson et al., 2013). To counter this lack of awareness, ASCO developed educational materials17 for clinicians to provide to their patients. Kennedy Sheldon also suggested improving messaging about why diet and exercise are important for people at risk for cancer, as well as for cancer survivors. She said that ONS created a national campaign called Get Up, Get Moving18 that encourages oncology nurses to implement evidence-based changes in practice and specifically to recommend physical activity to patients dur-
___________________
17 See https://www.asco.org/practice-guidelines/resources-patients (accessed April 25, 2017).
18 See https://www.ons.org/practice-resources/get-up-get-moving (accessed April 5, 2017).
ing cancer treatment. This campaign provides a number of educational resources for both cancer nurses and patients.
Dietz reported on the Why Weight?19 guide for health care professionals, developed by the Strategies to Overcome and Prevent Obesity Alliance. This guide is designed to help encourage patient-centered conversations about obesity and includes information on the role of stigma and weight bias, the importance of accommodation, and how to initiate conversations with nonjudgmental language such as, “Are you concerned about your weight?” rather than, “You have a weight problem and you must lose weight.” The guide also helps health care professionals understand the challenges patients face, the importance of shared decision making, and appropriate language and communication strategies to use, Dietz reported.
Dixon stated, “I am a huge fan of educating physicians [and] nurses about nutrition because there is a lot that those clinicians can do to get the ball moving forward, and if the patient hears it from three different clinicians and then gets a referral to a dietitian, they are more likely to be interested in that intervention and more receptive to it.” In addition, Demark-Wahnefried noted that both childhood and older adult cancer survivors are generally unaware of their increased risk for adverse health effects of cancer and its treatment—including sarcopenia and frailty—and clinicians need to increase awareness among these patient populations.
Insurance Coverage for Lifestyle Interventions
A number of workshop participants suggested that patient access to weight management and physical activity programs could be improved through better insurance coverage. Don Bradley, director of The Practical Playbook at Duke University, noted that private and governmental payers of health insurance plans consider multiple factors when deciding what benefits to cover, including consumer/employer demand for a service, cost of the benefit, evidence for effectiveness and efficiency of the service, the ability to administer the benefit consistently and fairly, presence of state or federal mandates for a service, and how the benefit will affect the marketability and adverse selection of a health plan.
Bradley noted that some insurers provide coverage for obesity treatment because the evidence base for treatment is growing (Garvey et al., 2016; Jensen et al., 2014; USPSTF, 2012, 2017). However, the services that
___________________
19 See http://whyweightguide.org (accessed on April 5, 2017).
are covered and how the benefit is implemented in a health benefit plan remains highly variable. He pointed out that insurers are required under the Patient Protection and Affordable Care Act (ACA) to cover recommendations from the USPSTF with an A or B grade, including the recommendation that clinicians screen their patients for obesity and either offer or refer patients with obesity to behavioral interventions.20 But he noted that not every health plan is subject to the mandates of the ACA, and these guidelines are not specific in terms of how a health plan is to provide the services. “It is there but not [very] clear from a regulatory standpoint,” he said.
Anand Parekh, chief medical advisor at the Bipartisan Policy Center, added that, “Payers are all over the map when it comes to coverage of many of these types of nonpharmacologic and nonsurgical interventions” for obesity treatment. Despite behavioral counseling for obesity being recommended by the USPSTF and part of the ACA mandate that certain plans cover this benefit without cost sharing, the ways in which plans are translating this recommendation into a benefit design vary. Some are outsourcing obesity preventive services to health behavioral consulting companies while others are covering telephone-based nutrition counseling, but “there is not a single standard approach that is being taken,” Parekh said. For example, he said that a survey of fee-for-service Medicaid programs found that 8 states and the District of Columbia cover all obesity-related preventive care CPT codes; 12 states and the District of Columbia cover all obesity-related behavioral consult CPT codes; and 15 states and the District of Columbia cover all obesity-related nutritional consult CPT codes (Petrin et al., 2014). “Most of the states are not covering many of these services,” he noted.
Elizabeth Kraft, senior clinical officer at Anthem Blue Cross Colorado and Nevada, said that insurers are increasingly focused on achieving health care value for their constituents, which has resulted in a shift away from fee-for-service reimbursement and toward new payment and care delivery models. “Regardless of how practitioners are organized or whether they call themselves an accountable care organization, independent practice association, or integrated health network, we are beginning to shift and share the risk and responsibility of patient care [among insurers and providers] so that the best care is rewarded and payment is based on value, not volume,” she said.
___________________
20 See https://www.uspreventiveservicestaskforce.org/Page/Document/RecommendationStatementFinal/obesity-in-adults-screening-and-management (accessed May 3, 2017) and https://www.uspreventiveservicestaskforce.org/Page/Document/RecommendationStatementFinal/obesity-in-children-and-adolescents-screening1 (accessed May 3, 2017).
She reported on the Enhanced Personal Health Care Program in which more than 80 percent of primary care practitioners in Colorado participate.21 Practices have the opportunity to share in the savings gained by providing high-quality care and reducing the total cost of their patients’ care, such as reducing emergency room visits and avoiding inpatient admissions. For example, Anthem made it easy for participating clinicians to refer patients to the DPP, but as Kraft noted, they were already eager to do so because they recognized the shared savings garnered from participation in the program, in addition to the benefits of improving their patients’ health.
Anthem has also created a Cancer Care Quality Program,22 the goal of which is to transform cancer care by supporting evidence-based, patient-centered, and value-conscious decisions in partnership with oncologists that improve health outcomes for each member. To create this program, Anthem reviewed available protocols for each cancer type and selected a subset of them based on efficacy, toxicity, and cost. Based on their review, Anthem created preferred “cancer care pathways.” For oncology practices that choose to use one of the pathways, or to have their patients participate in a precision medicine clinical trial, Anthem provides enhanced reimbursement to cover the costs of treatment planning and care coordination. Kraft added that it will be beneficial for Anthem to share information with participating practices about the importance of weight management, exercise, and strength building. She noted that such information sharing could be incorporated into care coordination, or care managers could be made aware of this information so they can make appropriate referrals.
Parekh suggested devising quality metrics for lifestyle interventions. Clinicians could be reimbursed when their patients meet specific targets, regardless of the intervention used to achieve weight loss goals or other health targets. But Parekh noted that there is a lack of validated quality measures for obesity; the National Quality Forum has only endorsed four screening metrics and has not endorsed any outcome metrics. “While there are endorsed measures related to BMI screening and assessment, there really are not any measures when it comes to addressing weight gain at the population or the plan level, and there are no patient-reported outcome measures,” he said. However, he said currently there is a lack of consensus on what those measures should be (e.g., whether it should be percent body weight
___________________
21 See https://www.pcpcc.org/initiative/enhanced-personal-health-care-program-colorado (accessed April 25, 2017).
22 See https://anthem.aimoncology.com (accessed May 3, 2017).
loss, and if so, what percentage is optimal). Population-based measures, such as obesity prevalence, are difficult to track accurately at the patient level with claims data, he noted. A framework for value-based obesity care also is yet to be determined. “Should you reward a lowering of the BMI that does not meet the target?” he asked.
Darshak Sanghavi, chief medical officer and senior vice president of translation at OptumLabs, noted that, “Everybody thinks that healthy weight is a good thing . . . and yet nobody pays for it. Nobody puts their money where their mouth is.” He said that it is expensive and time consuming to provide effective weight loss interventions, and the benefits of that weight loss are not seen for many years “so the people who pay for the care are not the people who are benefiting from the care,” and he added, “This is a business and political problem, not a medical problem. How do you create a business case for investments in weight management so that it is compelling enough to get people to actually invest in it?”
He then pointed out that innovations in insurance coverage are often pioneered at the federal level with demonstration projects showing cost savings without a sacrifice in quality care, and that leads to coverage through Medicare and Medicaid. Private insurers often then follow the lead of the Centers for Medicare & Medicaid Services (CMS). Although such a demonstration project led to CMS coverage of the DPP, Sanghavi noted that the project was a nonrandomized study of less than 3 years duration in only 1,000 people, in whom diabetes incidence was not assessed. But when he and his colleagues tracked the expenses of participants in the study and compared those to that of a matched control group, there was a savings of about $1,500 per patient in the intervention group compared to the control group during the first 15 months. As a result, the coverage decision for the DPP was actuarially certified, which is challenging to accomplish.
An analysis conducted by the CMS Office of the Actuary found that if the DPP were expanded with coverage, CMS costs would actually increase over time because the number of diabetes-related premature deaths would be reduced and those patients would incur more Medicare costs in the long run (CMS, 2016). “They were saying that one could not certify a preventive benefit if it made you live longer because then it would cost more,” Sanghavi said. But after some negotiations and advocacy on the part of Sanghavi and his colleagues, he said that CMS recognized that “future life should not be counted as a cost and is contrary to the integrity and values of who we are at CMS.” This principle is critical to consider when evaluating the value of investments in long-term prevention and in population health, he said.
Parekh noted that compared to pharmacologic interventions, much less funding is available for research on lifestyle interventions. In addition, there is no analogous Food and Drug Administration (FDA) approval of safety and efficacy, which leaves insurers in a quandary over what lifestyle interventions to cover. He suggested convening private and public payers to develop a standard benefit design for obesity prevention and treatment interventions.23 However, initial meetings with a dozen state Medicaid directors have surfaced a number of challenges in covering these interventions, including member retention length and turnover (i.e., the cycle of individuals coming and going in plans), lack of evidence on return on investment, the need for partnerships with community partners, which some clinical entities do not have, the lack of senior leadership support, and the need for data tracking beyond claims data.
When considering reimbursement for these interventions, Bradley asked, “Why don’t we look at obesity much as we do smoking?” Sanghavi responded by noting that even though Medicaid has a mandate to cover smoking cessation treatment, most states still do not. He added that CMS is considering smoking prevalence (by county) as a quality metric for value-based hospital reimbursements. “The only way to get [uptake and] long-term benefits is to measure things at the community level and hold people accountable for those community indicators,” he said.
Parekh noted lessons learned for cancer survivorship care and weight management related to insurance benefits, saying that interventions have to be evidence based, a broad array of clinicians need to be reimbursed for providing the benefit, and patient cost sharing can be a deterrent to uptake. He pointed out that although Medicare covers weight management interventions, the uptake is low, mainly because only physicians or nurse practitioners can be reimbursed. Dixon encouraged advocating for insurers to reimburse care provided by physical therapists, dietitians, and other providers of lifestyle interventions. “It is cheaper and more effective to refer to a dietitian, exercise physiologist, or physical therapist to get that comprehensive plan in place for weight management, body composition change, and obesity prevention, rather than have a physician do it, because on an hourly basis physicians are a much more expensive resource,” she said.
___________________
23 Standardization of benefit design helps consumers “effectively compare such elements as deductibles [and] co-payments for doctor and emergency room visits, hospital stays, and prescription drugs.” See https://ccf.georgetown.edu/2013/02/05/the-goldilocks-plan-getting-benefit-design-just-right-for-insurance-exchanges (accessed June 2, 2017).
Parekh also suggested incentivizing the referring clinician. For example, CMS recently launched a new Cardiac Rehabilitation Incentive Payment Model,24 which provides payment to cardiologists and cardiac surgeons who refer individuals to a cardiac rehabilitation program.
Payal Martin, public health advocate and analyst at Payal Shah Martin, LLC, asked if a mandate was needed for insurers to reimburse weight loss or physical activity programs. Kraft responded that the most important determinant is the evidence base for an intervention. She added that there is the need to have “assurances and guardrails as to exactly what the program is and that what you are paying for is legitimate, and clinicians are recommending it at the behest of the patient and not selling products, which potentially can happen.” Schmitz pointed out that coverage for cardiac rehabilitation was implemented without evidence that it reduced the recurrence of a heart attack, mainly due to studies showing that men who received the rehab returned to work sooner. “So the idea that we need a single trial such as the BWEL study to show efficacy to get coverage for this is holding things to a very different standard,” she said. “Could getting people back to work quicker be the basis for reimbursing cancer rehabilitation like it was for cardiac rehabilitation?” she asked.
Sanghavi responded that there are several ways for CMS to cover new interventions. CMS can make a national coverage determination,25 which requires a high bar for evidence of benefit. Alternatively, CMS can make local coverage determinations26 or CMS can evaluate an intervention in a CMS demonstration project, and if effectiveness is demonstrated, have it certified for broader expansion, he said. Kraft added that providing coverage for an intervention because it hastens the patient’s return to work is appropriate for those who are employed, but not all individuals have their insurance covered by employers. “How do you then create a value proposition for those who are buying insurance just for themselves? That group of individuals is extremely price sensitive,” Kraft said.
___________________
24 See https://innovation.cms.gov/initiatives/cardiac-rehabilitation (accessed May 3, 2017).
25 See https://www.cms.gov/Medicare/Coverage/DeterminationProcess/Downloads/FR08072013.pdf (accessed June 2, 2017).
26 See https://www.cms.gov/Medicare/Coverage/DeterminationProcess/LCDs.html (accessed June 2, 2017).
Clinician Education, Resources, and Standards
There was a great deal of discussion about the need to better inform clinicians about the value of physical activity and weight management for cancer survivors. Several workshop participants suggested providing referral resources for clinicians and standardizing weight management and exercise prescriptions. Ligibel pointed out that currently there is no information about weight management, nutrition, or physical activity in the medical oncology curriculum.
Goodwin said it was not realistic to expect the oncologist to deliver a lifestyle intervention or to know in detail what the intervention is going to be. But she added that an oncologist should have a high-level understanding of the benefits of the intervention. Nekhlyudov also noted the need for primary care clinicians to understand the benefits of weight management and physical activity for their cancer survivors, and suggested providing education and outreach to the primary care community.
Kennedy Sheldon said that when it comes to weight management, the focus of many practicing oncology nurses today is still to prevent patients from losing weight during treatment. “We need to educate nurses better, particularly those in cancer care or those working with cancer survivors, who need to get the message out that weight control is important, not just for reducing the risk of recurrences, but also of second primary cancers,” Kennedy Sheldon said. She noted that about 130,000 nurses work in cancer care in the United States, but incorporating physical activity and weight management into cancer treatment and survivorship is not covered in oncology nursing training or included on certification exams.
Parekh reported on the 2012 Bipartisan Policy Center (BPC) publication Lots to Lose, which focused on policies and environmental changes to tackle the obesity epidemic. The BPC recommended a focus on improving clinician competencies regarding nutrition, physical activity, and weight management, as well as coverage policies for both obesity prevention and treatment (Bipartisan Policy Center’s Nutrition and Physical Activity Initiative, 2012).
Dietz reported on an effort to develop interprofessional competencies27 for the prevention and treatment of obesity, which involved more
___________________
27 The competencies were developed by the Provider Training and Education Workgroup of the Integrated Clinical and Social Systems for the Prevention and Management of Obesity Innovation Collaborative, an ad hoc activity associated with the Roundtable on Obesity Solutions at the National Academies of Sciences, Engineering, and Medicine (the National Academies). The responsibility for the content rests with the Innovation Collaborative and not with the National Academies. This product does not necessarily represent the views of any one organization, the roundtable, or the National Academies and has not been subjected to the review procedures of, nor is it a publication or product of, the National Academies.
than 20 organizations convened by a subgroup of the Innovation Collaborative on Integrated Clinical and Social Systems for the Prevention and Management of Obesity, which is an ad hoc activity associated with the National Academies Roundtable on Obesity Solutions (see Box 7). These competencies include understanding the framework of obesity as a disease, recognizing the disparities and inequities in obesity prevention and care, and awareness of approaches to mitigate weight bias and the stigma linked to obesity. In addition to knowing strategies for patient care related to obesity, the competencies also include knowing how to use patient-centered communication. Dietz suggested the competencies could be incorporated into health care professional curricula and continuing education. “If these get incorporated into the certification of providers, then these competencies will be adapted, adopted, and discussed in medical schools and schools of nursing, physical therapy, nutrition, and social work,” Dietz said.
Apovian noted that the Obesity Society developed an obesity medicine certification and has been certifying physicians and other health care providers for the past 5 years. Oncologists interested in developing obesity medicine for their practices could undergo such certification, she said. In addition, the Obesity Society helped the Academy of Nutrition and Dietetics develop their own certification exam for obesity treatment for dietitians, as well as certification exams for physical therapists and exercise physiologists. It expects to provide certification exams for nurse practitioners and physician assistants in the future. “These exams will help educate doctors and other health care providers to treat obesity,” she said.
Schmitz suggested using continuing education and licensure to train clinicians about the benefits of exercise for cancer survivors. Bamman agreed, adding that such training could include the distinction between resistance and aerobic exercise, the goals of each, and how they differ from the goals of a weight management program. Denlinger suggested also finding novel educational opportunities within disease-specific meetings or via maintenance of certification.
Schmitz pointed out that the ACSM certifies exercise professionals, and in collaboration with ACS, has developed a specific certification for exercise professionals to design and administer fitness assessments and exercise
programs specific to a person’s cancer diagnosis, treatment, and current recovery status.28 There are now 400 exercise professionals who have been certified to care for patients with cancer. “That is not enough, so we need to do some more work on that,” she said. She added that the ACSM developed an educational resource for clinicians called Exercise is Medicine.29
Morris pointed out that physical therapists have been treating cancer survivors for almost 50 years, and in 1983, the APTA recognized the
___________________
28 See https://certification.acsm.org/acsm-cancer-exercise-trainer (accessed June 2, 2017).
29 See http://exerciseismedicine.org (accessed April 25, 2017).
unique role physical therapists play in oncology rehabilitation by creating the Oncology Section, a professional home for therapists who treat cancer survivors.30 Today, the Oncology Section has about 1,200 members and has as its current mission the advancement of physical therapy practice for persons affected by cancer and chronic illness by maximizing movement and wellness across the lifespan, he said. The Oncology Section provides educational opportunities for its members, as well as for the general public.
Recently, the House of Delegates of the APTA determined that oncology rehabilitation required a sufficiently unique skill set and knowledge base, and that sufficient data existed to support the recognition of oncology rehabilitation as a specialty practice area within the broader arena of physical therapy. Consequently, by 2019, physical therapists who have demonstrated skills and knowledge in the area of oncology rehabilitation will be able to identify themselves as being specialists in this field.31 Morris pointed out that over the past 6 years, the Oncology Section has made a concerted effort to identify appropriate outcome measures that have both the evidence and biometrics to be useful to clinicians in assessing the physical function of cancer survivors. These efforts have resulted in more than 25 publications, and the Oncology Section is developing clinical practice guidelines to further assist clinicians in providing better care for cancer survivors.
Standards for implementing weight management and physical activity programs are also needed, Longjohn suggested. He said the DPP is a good example of a program with quality standards, noting that the program included science-based information that formed a set of quality indicators for training, marketing, and other implementation efforts to ensure that decisions about individual participation, patient referral, and health insurance benefits are based on accurate, reliable, and trustworthy information.
Clinician Referral Resources
Denlinger suggested developing and maintaining a web-based list of local resources related to weight management and physical activity to which clinicians could refer their patients. “I would love to have some sort of interface, either with my EHR [electronic health record] or something I could easily pull up on the Internet in which I could put in my zip code, press a
___________________
30 See http://oncologypt.org/about-oncology-section/index.cfm (accessed June 2, 2017).
31 See http://oncologypt.org/specialization/index.cfm (accessed June 2, 2017).
button, and be given a list of weight loss and physical activity resources that offer cancer survivor-oriented programs that I could put into the patient instruction section of the after-visit summary that I hand to patients,” she said. However, Dietz pointed out that when he hired a group of students to map such resources that are a part of or near The University of Chicago, he found that it was difficult to generate and maintain an accurate list of active sites.
Alfano responded that the ACS is considering how to work with partners to create a registry for clinical services and resources that cancer survivors may need during their survivorship care, including nutrition, physical activity, and weight management programs; cancer rehabilitation; and psychosocial, palliative, and other specialty care. She noted that this registry would be patterned after the Palliative Care Registry.32 Alfano said that clinicians and programs could add and maintain their information on the survivorship registry at their own initiative and there would be no policing of the registry to ensure accuracy or completeness. “The idea is [to] let all the different kinds of services autopopulate into the registry according to a matrix we give them,” she said.
Denlinger added that when she was planning the Survivorship Program at the Fox Chase Cancer Center, she did a resource assessment and was surprised at how many resources within her own campus described themselves as being a survivorship resource. “There were organizations that I was not necessarily thinking about as a survivorship resource that did provide survivorship care. You run the risk of potentially either not catching everybody engaged in survivorship care or potentially finding some resources that are not as survivorship oriented as they should be. But overall, you probably would be surprised at what is actually in your backyard,” she said.
Denlinger suggested a number of cancer survivorship, weight management, and physical activity resources for clinicians (see Box 8). Boone added that a not-for-profit organization called the Coalition for the Registration of Exercise Professionals maintains the Registry of Exercise Professionals,33 an internationally recognized registry of exercise professionals in the United States, including their credentials and certifications.
___________________
32 See https://registry.capc.org (accessed September 14, 2017).
33 See http://customer.usreps.org/USREPS/USREPSWcm/Membership/Directory/Shared_Content/Directory.aspx (accessed April 25, 2017).
Collaborations and Partnerships
A number of workshop participants suggested that collaborations between organizations as well as partnerships between clinicians could further efforts to improve weight management and physical activity in cancer survivors. Ligibel noted that ASCO hosted a meeting in 2016 that convened many organizations focused on obesity as a central or peripheral aspect of their mission to discuss what obesity-related initiatives each has developed or is interested in developing, and how ASCO could partner with them to make people more aware of the links between cancer and obesity and ways to help motivate patients to manage their weight.34 “We wanted to know what groups like the Obesity Society and the American Heart Association (AHA) were doing and how we could partner with them,” she said, noting that for many patients, the message that obesity is related to cancer is more motivating than the link to diabetes or heart disease. “There is a real desire for organizations to partner with oncology groups to hopefully reinvigorate those conversations,” Ligibel said. Buzaglo agreed that it would be worthwhile to have the AHA involved in such collaborations, as well as patient advocates. Apovian also suggested the Obesity Society “can align and collaborate with oncology to promote obesity medicine so we can get more obesity treatment services covered by third-party payers and for nonphysician clinicians.”
Schmitz reported that the ACSM has a Cancer Interest Group,35 and she is trying to convene a roundtable of representatives from the ACSM, APTA, ASCO, American Academy of Physical Medicine and Rehabilitation, ONS, and other organizations to generate a position statement about physical activity for cancer survivors. “The potential for the ACSM to join hands with other organizations to try to effect change and move things forward on a policy standpoint is very real,” Schmitz said.
Boone noted that partnerships on the clinician level are also important to develop and asked how nonphysician professionals, such as exercise physiologists, nutritionists, and physical therapists, can partner with physicians. “We have found that it is difficult to get the door cracked open so we can even talk to practitioners about what it is that we can offer their patients. How do we jointly work toward meeting the best needs of
___________________
34 See https://www.asco.org/advocacy-policy/asco-in-action/asco-convenes-obesity-work-group-meeting-multidisciplinary (accessed June 2, 2017).
35 See http://www.acsm.org/membership/special-interests/interest-groups (accessed April 25, 2017).
patients?” he asked. Dixon agreed this is an issue, stating, “I always used to say to oncologists, ‘you would not send your patients to a nutritionist like me for their chemotherapy dosing or their radiation protocols, and I would not expect my patients to get nutrition counseling from you.’ So collaboration is key.”
Ligibel responded that for many communities there is a lack of exercise or nutrition professionals with which to forge relationships. Even if such relationships are established, a lack of insurance coverage for the services they provide can prevent patients from following up on referrals. “We need to create an infrastructure where we could send somebody to a program that would provide the support they need [and to ensure that] they were not completely responsible for paying for,” she said.
Eakin suggested that another key to success is collaborating early in the development of the program with partners and stakeholders, including patients, clinicians, and advocacy organizations. “The earlier we involve them, the better,” she said, noting that to develop the Healthy Living after Cancer program, she met with colleagues at cancer councils throughout Australia over a 2-year period to assess their cancer survivorship needs, the programming currently offered, and their willingness to adopt an integrated telephone-based program.
Improving Research
A number of workshop participants suggested strategies to improve research, including collaborations through research networks, development of biomarkers and appropriate endpoints, engagement of diverse research personnel and study participants, and ensuring that studies are pragmatic and patient centered.
National Research Networks and Resources
Bamman described the National Exercise Clinical Trials Network,36 a 73-site research network that is led by a partnership among the ACSM, the University of Alabama at Birmingham, and the NIH Clinical and Translational Science Award Consortium. “Within this network there is a lot of expertise and interest in cancer-focused trials,” he said. Bamman also described the Rehabilitation Research Resource to Enhance Clinical
___________________
36 See https://www.uab.edu/medicine/nextnet (accessed April 26, 2017).
Trials Center,37 funded by the NIH as a national resource that can help researchers design and conduct clinical trials that will fill key evidence gaps in medical rehabilitation, including exercise interventions. He noted that cancer rehabilitation is at the forefront of medical rehabilitation from the NIH’s perspective.
Developing Biomarkers
Thomson suggested validating and measuring cancer-associated biomarkers of prognosis, such as vascular endothelial growth factor or E-selectin levels (Linkov et al., 2012), or determining prognostic scores like the Glasgow Prognostic Score that could standardize outcomes collected across studies (Simmons et al., 2017). She also suggested assessing genetic markers of responsiveness, such as patatin-like phospholipase 3, in study populations (Shen et al., 2015). Biomarkers that can indicate mechanisms and mediators of action are also useful, as are repeated measurements to indicate trends, Thomson said. Pinto agreed, adding “We need to be thinking about what the mechanisms are—is exercise influencing cancer outcomes by working through an insulin pathway or immune functioning, or is it endocrine related?” Thomson also called for objective biomarker measures of lifestyle behavior change, as well as measures that can indicate an intervention’s effect on body composition (Caan and Kroenke, 2017).
Bamman suggested measuring biomarkers to examine why some patients respond more to a given exercise intervention than others. He referred to the NIH Common Fund’s Molecular Transducers of Physical Activity Consortium,38 which is assessing proteomics, genomics, epigenomics, and metabolomics for approximately 2,700 healthy adults randomized to either resistance or aerobic training. “It is going to be a really powerful resource for all of us,” he noted.
Improving Measures and Endpoints
Pinto emphasized the importance of identifying noncancer causes of death, especially cardiovascular outcomes, in addition to cancer outcomes. She also suggested assessing whether healthy behaviors are maintained over
___________________
37 See https://react.center (accessed April 26, 2017).
38 See https://commonfund.nih.gov/MolecularTransducers/overview (accessed April 26, 2017).
time. Irwin also emphasized the importance of following participants for longer periods of time to assess the long-term effects of the interventions, because the effects of a given intervention often attenuate over time.
Several participants called for high-quality, randomized controlled trials of lifestyle interventions that blind the assessor to the randomization status, and include intention-to-treat analyses and other efforts to reduce bias. Pinto also suggested that studies document the safety of an intervention and plan for maintenance of behavior change.
Ligibel added that the “dose” of an intervention should also be measured (i.e., how many telephone calls are needed to be effective in a phone-based coaching or counseling intervention), because knowing that information may help make interventions more cost effective. She also stressed that for a disease like breast cancer, which has a long natural history, it may take 5 years or more to determine the cost effectiveness of interventions if they affect such outcomes as recurrence, but within a shorter frame of time, other aspects of cost effectiveness can be measured, such as the impact of an intervention on a patient’s return to work or productivity.
Thomson stressed that the caloric restriction of a given diet should be measured, in addition to the type of diet evaluated (e.g., a diet that is low-fat versus low-carb). Pinto said that when providing exercise interventions, the dose of exercise given needs to be reported so that outcomes can be linked to specific amounts of physical activity. “We have to think about the minimal dose of exercise needed because that will have immediate bearing on the feasibility of implementing these interventions to reach a greater number of cancer survivors,” she said. As an example, she cited a trial comparing three different exercise doses (Courneya et al., 2013).
Bamman added that for an exercise trial, it is important to consider the mode, intensity, frequency, and volume of the exercise. He also suggested that exercise interventions be designed based on expected outcomes and to consider variability by age, with older individuals often requiring a different frequency of exercise than younger people to receive the same benefit. He noted that a trial with the goal of improving morbidity or mortality risk may have a different exercise prescription than one to prevent muscle loss during chemotherapy or to restore muscle mass in survivors.
Data on Economic Outcomes and Implementation
Dixon said more studies should collect health economic data that can reveal the cost-effectiveness of various lifestyle interventions. She added that
some of her colleagues have shared their data with the chief executive officers of local insurers, and that it has convinced insurers to start covering care provided by a dietitian. “As health care providers and academic researchers, [we] need to think beyond our silos and work with health economists to show there is a value in providing this care. Because we all know that the bottom line is all about whether this care saves money,” she said.
Dixon added “I used to marvel that we would give someone a drug that would maybe buy them 3 months of life and cost $100,000, but we would not refer them to a dietitian, exercise physiologist, or physical therapist, even though these are [often] very cost-effective interventions. There are data already to support that, but we need to generate more data and share it. In addition to providing something that resonates with the patient population, it has to resonate with the insurers so we can get over this hump of ‘Well, it is not covered so I cannot recommend it.’” Longjohn also noted the importance of health economic analyses: “It is unbelievable how effective it can be to go to a chief medical officer of a health plan and show the data and say ‘This is something that we can do together to benefit your employees and your insured population.’ There is a lot of opportunity there.”
Stolley suggested that studies include measures of the implementation process and outcomes to inform scalability and sustainability, especially those relevant to community settings. Befort suggested that the NIH offer grants for research with effectiveness-implementation hybrid designs.
Longjohn added that multiple types of research—efficacy trials, validation studies in different contexts, translational studies, scaling and operational practice-based evidence studies, and cost-effectiveness research—need to be conducted, and should be done simultaneously rather than sequentially. “All of these are important and needed for us to get where we want to go,” he said.
Increasing Diversity
Several workshop participants discussed the importance of increasing diversity of research personnel for lifestyle intervention studies. Thomson noted that studies demonstrating effective weight loss—and more specifically, weight management in cancer survivors—have included psychologists or other behavioral scientists, biostatisticians, and a wide array of clinicians, including oncologists, dietitians, exercise physiologists, and health coaches. She added that some intervention studies would benefit from greater involvement of adaptive design specialists and software engineers early in the research process.
Thomson also suggested including a health economist on the research team “because the cost of what we are doing is an important outcome.” Pinto agreed, emphasizing that determining cost effectiveness is key to insurance coverage for lifestyle interventions. Ya-Chen Tina Shih, chief of the section of cancer economics and policy in the Department of Health Services Research at The University of Texas MD Anderson Cancer Center, suggested involving health economists when first planning a trial. “It is too late once the trial is done because you lose the opportunity to collect data,” she said, and added that there are several validated tools researchers can use to collect economic information in clinical trials. She also noted that health economists tend to consult with payers about what types of data they need; often, she said, payers prefer budget impact modeling over cost-effectiveness analysis. Longjohn added that federal agencies prefer the scoring metrics of the Congressional Budget Office.
Thomson said that it is critical to include the input of cancer survivors in the development of lifestyle intervention study designs and implementation strategies. To make research more effective, she suggested using focus groups or in-depth interviews with the target population to acquire general knowledge and understanding, including what people’s interests are, what recruitment models might work, what can be expected for adherence, preferred delivery modes, and potential barriers and ways to overcome them. “Right now we tend to try to fix things once the intervention has started. While there still will be some of that adaptation, it is important that we get as much up-front information [as possible] from this formative research,” Thomson said. She added that community-based research should engage with community health workers and institutional partners.
Befort, Demark-Wahnefried, Stolley, and Thomson suggested enrolling a broader representation of cancer survivors who are racially and ethnically diverse, children, older adults, and those living in rural areas. Pinto agreed, noting that in addition to expanding the diversity of study participants, studies should also include participants with fatigue or quality-of-life issues the interventions are designed to target. Thomson noted that most studies of weight loss interventions that have demonstrated an effect have enrolled the “worried well,” or cancer survivors who already are healthier at baseline than the general population. She called for expanding the diversity of trial participants by including cancer survivors who have diet or activity limitations, or those on medications for preventing or managing comorbid conditions.
Irwin suggested more research to assess the impact of interventions on treatment side effects, such as loss in bone density, simultaneously with
chemotherapy or other cancer treatments. Assessing cancer- or treatment-related symptoms is also critical to ensure that they will not interfere with participants’ ability to adhere to a given intervention. Thomson noted that in the LIVES trial for patients with ovarian cancer, there was a high prevalence of symptoms such as neuropathy, sleepiness, and depression, all of which would influence participants’ ability to adhere to an exercise intervention. She suggested integrating this information into interventions, as the RENEW trial did. This study showed that despite participants having substantial symptoms, it was possible to work with them and have a positive impact on weight loss (Kenzik et al., 2015).
Stolley suggested that researchers conduct more comparative effectiveness research to compare two or more interventions, because these studies might be more appealing to patients than those that use a placebo or no intervention. She also stressed the need for collaboration with churches, community health centers, and other local organizations to enhance recruitment and retention of racially and ethnically diverse volunteers, who have traditionally been underrepresented in clinical trials.
Befort said, “I would like to advocate that rurality be designated as an underrepresented group officially. This would help us start to use a common definition that will make it easier to make comparisons across studies.” She added that rural populations should be included in enrollment targets and catchment area needs assessments. More research is needed on the determinants of health behaviors in rural settings, she said. “I would love to see some planned rural–urban subgroup analyses in some of the larger studies that are being done,” she said.
Thomson noted that frequently clinical trials are underpowered to conduct subgroup analyses on various subpopulations of interest because not enough consideration is given a priori to the size of various populations needed to conduct these subanalyses. “We know that one size does not fit all, and we should be moving into studying what works for whom under what circumstances and for which outcomes,” she said.
Developing More Pragmatic and Patient-Centered Trials
Pinto suggested developing pragmatic trials that can be implemented in the current health care system. “Sometimes an intensive intervention may work but may not be something that can be offered to many patients,” she said. Longjohn suggested partnering with community resources to conduct pragmatic trials.
“Patients should be given choices, and there should be flexibility as to where, when, and how they will exercise,” Pinto said. She also suggested making research more patient centered by focusing on outcomes most relevant to patients, such as relief of fatigue and pain, and longer survival. She noted a systematic review that indicated patients want programs that will help them with meaningful goals focused on symptom management and functional independence (Granger et al., 2017). “We need to listen to our patients,” she said. Denlinger suggested more research on ways to overcome challenges that both patients and their clinicians experience.
WRAP-UP
Demark-Wahnefried quoted cancer survivor Michael Douglas, who said “Cancer did not bring me to my knees. It brought me to my feet,” and added, “That is our mission—to get cancer survivors back on their feet and hopefully back to where they were before their diagnosis.” She stressed, “We need to provide physical activity and weight management as a part of the standard of care,” and noted that these interventions need to be personalized. “We still do not know whether weight management and physical activity will reduce cancer recurrence or metastasis, but we do know about other benefits [from these interventions] for patients with cancer,” Demark-Wahnefried added. She suggested making improvements in clinician education and research, and applying current knowledge to screen more patients and triage them to appropriate lifestyle interventions, as well as helping empower patients to meet their goals. She also reiterated the need for referrals and the importance of reimbursing nonphysician clinicians, as well as certifying competencies on weight management and physical activity for all clinicians. “It has been a wonderful conference, but now the real work begins,” she said. “The situation is urgent, and there are definitely some messages we can put out right now that would really improve the health of the cancer survivor community,” she concluded.
REFERENCES
Ackermann, R. T., E. A. Finch, E. Brizendine, H. Zhou, and D. G. Marrero. 2008. Translating the diabetes prevention program into the community: The DEPLOY pilot study. American Journal of Preventive Medicine 35(4):357–363.
ACS (American Cancer Society). 2017. What does the American Cancer Society recommend about body weight? https://www.cancer.org/cancer/cancer-causes/diet-physical-activity/body-weight-and-cancer-risk/acs-recommendations.html (accessed August 31, 2017).
Ajala, O., P. English, and J. Pinkney. 2013. Systematic review and meta-analysis of different dietary approaches to the management of type 2 diabetes. American Journal of Clinical Nutrition 97(3):505–516.
Alfano, C. M., A. L. Cheville, and K. Mustian. 2016. Developing high-quality cancer rehabilitation programs: A timely need. American Society of Clinical Oncology Educational Book 35:241–249.
Ansa, B., W. Yoo, M. Whitehead, S. Coughlin, and S. Smith. 2016. Beliefs and behaviors about breast cancer recurrence risk reduction among African American breast cancer survivors. International Journal of Environmental Research and Public Health 13(1):46.
Arnold, M., N. Pandeya, G. Byrnes, A. G. Renehan, G. A. Stevens, M. Ezzati, J. Ferlay, J. J. Miranda, I. Romieu, R. Dikshit, D. Forman, and I. Soerjomataram. 2015. Global burden of cancer attributable to high body-mass index in 2012: A population-based study. Lancet Oncology 16(1):36–46.
Assi, M., F. Derbré, L. Lefeuvre-Orfila, D. Saligaut, N. Stock, M. Ropars, and A. Rébillard. 2017. Maintaining a regular physical activity aggravates intramuscular tumor growth in an orthotopic liposarcoma model. American Journal of Cancer Research 7(5):1037–1053.
Aycinena, A. C., C. Valdovinos, K. D. Crew, W. Y. Tsai, J. M. Mata, R. Sandoval, D. Hershman, and H. Greenlee. 2017. Barriers to recruitment and adherence in a randomized controlled diet and exercise weight loss intervention among minority breast cancer survivors. Journal of Immigrant and Minority Health 19(1):120–129.
Bae, H. S., H. J. Kim, J. H. Hong, J. K. Lee, N. W. Lee, and J. Y. Song. 2014. Obesity and epithelial ovarian cancer survival: A systematic review and meta-analysis. Journal of Ovarian Research 7:41.
Baker, A. M., K. C. Smith, K. I. Coa, K. J. Helzlsouer, L. E. Caulfield, K. S. Peairs, L. D. Shockney, and A. C. Klassen. 2015. Clinical care providers’ perspectives on body size and weight management among long-term cancer survivors. Integrative Cancer Therapies 14(3):240–248.
Baker, R. C., and D. S. Kirschenbaum. 1993. Self-monitoring may be necessary for successful weight control. Behavior Therapy 24(3):377–394.
Barlow, S. E. 2007. Expert committee recommendations regarding the prevention, assessment, and treatment of child and adolescent overweight and obesity: Summary report. Pediatrics 120(Suppl 4):S164–S192.
Basen-Engquist, K., C. M. Alfano, M. Maitin-Shepard, C. A. Thomson, K. H. Schmitz, B. M. Pinto, K. Stein, D. S. Zucker, K. L. Syrjala, E. Fallon, C. Doyle, and W. Demark-Wahnefried. 2017. Agenda for translating physical activity, nutrition, and weight management interventions for cancer survivors into clinical and community practice. Obesity 25:S9–S22.
Beason, T. S., S.-H. Chang, K. M. Sanfilippo, S. Luo, G. A. Colditz, R. Vij, M. H. Tomasson, J. F. Dipersio, K. Stockerl-Goldstein, A. Ganti, T. Wildes, and K. R. Carson. 2013. Influence of body mass index on survival in veterans with multiple myeloma. Oncologist 18(10):1074–1079.
Beddhu, S., G. Wei, R. L. Marcus, M. Chonchol, and T. Greene. 2015. Light-intensity physical activities and mortality in the United States general population and CKD subpopulation. Clinical Journal of the American Society of Nephrology 10(7):1145–1153.
Beehler, G. P., A. E. Rodrigues, M. A. Kay, M. T. Kiviniemi, and L. Steinbrenner. 2014. Perceptions of barriers and facilitators to health behavior change among veteran cancer survivors. Military Medicine 179(9):998–1005.
Befort, C. A., H. Austin, and J. Klemp. 2011. Weight control needs and experiences among rural breast cancer survivors. Psycho-Oncology 20(10):1069–1075.
Befort, C. A., N. Nazir, and M. G. Perri. 2012. Prevalence of obesity among adults from rural and urban areas of the United States: Findings from NHANES (2005–2008). Journal of Rural Health 28(4):392–397.
Befort, C. A., J. R. Klemp, C. Fabian, M. G. Perri, D. K. Sullivan, K. H. Schmitz, F. J. Diaz, and T. Shireman. 2014. Protocol and recruitment results from a randomized controlled trial comparing group phone-based versus newsletter interventions for weight loss maintenance among rural breast cancer survivors. Contemporary Clinical Trials 37(2):261–271.
Befort, C. A., L. Bennett, D. Christifano, J. R. Klemp, and H. Krebill. 2015. Effective recruitment of rural breast cancer survivors into a lifestyle intervention. Psychooncology 24:487–490.
Befort, C. A., J. R. Klemp, D. K. Sullivan, T. Shireman, F. J. Diaz, K. Schmitz, M. G. Perri, and C. Fabian. 2016. Weight loss maintenance strategies among rural breast cancer survivors: The Rural Women Connecting for Better Health Trial. Obesity 24(10):2070–2077.
Beidas, R. S., B. Paciotti, F. Barg, A. R. Branas, J. C. Brown, K. Glanz, A. DeMichele, L. DiGiovanni, D. Salvatore, and K. H. Schmitz. 2014. A hybrid effectiveness-implementation trial of an evidence-based exercise intervention for breast cancer survivors. Journal of the National Cancer Institute Monograph 2014(50):338–345.
Bennett, J. A., K. M. Winters-Stone, J. Dobek, and L. M. Nail. 2013. Frailty in older breast cancer survivors: Age, prevalence, and associated factors. Oncology Nursing Forum 40(3):E126–E134.
Betof, A. S., C. D. Lascola, D. Weitzel, C. Landon, P. M. Scarbrough, G. R. Devi, G. Palmer, L. W. Jones, and M. W. Dewhirst. 2015. Modulation of murine breast tumor vascularity, hypoxia, and chemotherapeutic response by exercise. Journal of the National Cancer Institute 107(5):djv040.
Bipartisan Policy Center’s Nutrition and Physical Activity Initiative. 2012. Lots to lose: How America’s health and obesity crisis threatens our economic future. https://bipartisanpolicy.org/wp-content/uploads/sites/default/files/5023_BPC_NutritionReport_FNL_Web.pdf (accessed April 25, 2017).
Blair, C. K., M. C. Morey, R. A. Desmond, H. J. Cohen, R. Sloane, D. C. Snyder, and W. Demark-Wahnefried. 2014. Light-intensity activity attenuates functional decline in older cancer survivors. Medicine and Science in Sports and Exercise 46(7):1375–1383.
Blanchard, C. M., K. S. Courneya, and K. Stein. 2008. Cancer survivors’ adherence to lifestyle behavior recommendations and associations with health-related quality of life: Results from the American Cancer Society’s SCS-II. Journal of Clinical Oncology 26(13):2198–2204.
Bluethmann, S. M., K. Basen-Engquist, S. W. Vernon, M. Cox, K. P. Gabriel, S. A. Stansberry, C. L. Carmack, J. A. Blalock, and W. Demark-Wahnefried. 2015a. Grasping the “teachable moment”: Time since diagnosis, symptom burden and health behaviors in breast, colorectal and prostate cancer survivors. Psycho-Oncology 24(10):1250–1257.
Bluethmann, S. M., S. W. Vernon, K. P. Gabriel, C. C. Murphy, and L. K. Bartholomew. 2015b. Taking the next step: A systematic review and meta-analysis of physical activity and behavior change interventions in recent post-treatment breast cancer survivors. Breast Cancer Research and Treatment 149(2):331–342.
Booth, F. W., and M. J. Laye. 2009. Lack of adequate appreciation of physical exercise’s complexities can pre-empt appropriate design and interpretation in scientific discovery. Journal of Physiology 587(Pt 23):5527–5539.
Bower, J. E., D. Garet, B. Sternlieb, P. A. Ganz, M. R. Irwin, R. Olmstead, and G. Greendale. 2012. Yoga for persistent fatigue in breast cancer survivors: A randomized controlled trial. Cancer 118(15):3766–3775.
Braam, K. I., P. van der Torre, T. Takken, M. A. Veening, E. van Dulmen-den Broeder, and G. J. L. Kaspers. 2016. Physical exercise training interventions for children and young adults during and after treatment for childhood cancer. Cochrane Database of Systematic Reviews (4):CD008796.
Bradley, D.W., Dietz, W.H., and the Provider Training and Education Workgroup. 2017. Provider Competencies for the Prevention and Management of Obesity. Washington, DC: Bipartisan Policy Center. https://bipartisanpolicy.org/library/providercompetencies-for-the-prevention-and-management-of-obesity (accessed August 30, 2017).
Bravata, D. M., L. Sanders, J. Huang, H. M. Krumholz, I. Olkin, C. D. Gardner, and D. M. Bravata. 2003. Efficacy and safety of low-carbohydrate diets: A systematic review. JAMA 289(14):1837–1850.
Brown, J. C., and K. H. Schmitz. 2015. Weight lifting and physical function among survivors of breast cancer: A post hoc analysis of a randomized controlled trial. Journal of Clinical Oncology 33(19):2184–2189.
Brunelli, A., R. Belardinelli, M. Refai, M. Salati, L. Socci, C. Pompili, and A. Sabbatini. 2009. Peak oxygen consumption during cardiopulmonary exercise test improves risk stratification in candidates to major lung resection. Chest 135(5):1260–1267.
Buchner, D. M., and E. H. Wagner. 1992. Preventing frail health. Clinics in Geriatric Medicine 8(1):1-17.
Bueno, N. B., I. S. V. de Melo, S. L. de Oliveira, and T. da Rocha Ataide. 2013. Very-low-carbohydrate ketogenic diet v. low-fat diet for long-term weight loss: A meta-analysis of randomised controlled trials. British Journal of Nutrition 110(7):1178–1187.
Buffart, L. M., J. Kalter, M. G. Sweegers, K. S. Courneya, R. U. Newton, N. K. Aaronson, P. B. Jacobsen, A. M. May, D. A. Galvão, M. J. Chinapaw, K. Steindorf, M. L. Irwin, M. M. Stuiver, S. Hayes, K. A. Griffith, A. Lucia, I. Mesters, E. van Weert, H. Knoop, M. M. Goedendorp, N. Mutrie, A. J. Daley, A. McConnachie, M. Bohus, L. Thorsen, K.-H. Schulz, C. E. Short, E. L. James, R. C. Plotnikoff, G. Arbane, M. E. Schmidt, K. Potthoff, M. van Beurden, H. S. Oldenburg, G. S. Sonke, W. H. van Harten, R. Garrod, K. H. Schmitz, K. M. Winters-Stone, M. J. Velthuis, D. R. Taaffe, W. van Mechelen, M.-J. Kersten, F. Nollet, J. Wenzel, J. Wiskemann, I. M. Verdonck-de Leeuw, and J. Brug. 2017. Effects and moderators of exercise on quality of life and physical function in patients with cancer: An individual patient data meta-analysis of 34 RCTs. Cancer Treatment Reviews 52:91–104.
Burris, J. L., and M. Andrykowski. 2010. Disparities in mental health between rural and nonrural cancer survivors: A preliminary study. Psycho-Oncology 19(6):637–645.
Caan, B. J., and C. H. Kroenke. 2017. Next steps in understanding the obesity paradox in cancer. Cancer Epidemiology Biomarkers & Prevention 26(1):12.
Campbell, K. L., C. L. Van Patten, S. E. Neil, A. A. Kirkham, C. C. Gotay, K. A. Gelmon, and D. C. McKenzie. 2012. Feasibility of a lifestyle intervention on body weight and serum biomarkers in breast cancer survivors with overweight and obesity. Journal of the Academy of Nutrition and Dietetics 112(4):559–567.
Carson, K. R., N. L. Bartlett, J. R. McDonald, S. Luo, A. Zeringue, J. Liu, Q. Fu, S.-H. Chang, and G. A. Colditz. 2012. Increased body mass index is associated with improved survival in United States veterans with diffuse large b-cell lymphoma. Journal of Clinical Oncology 30(26):3217–3222.
CDC (Centers for Disease Control and Prevention). 2017. Adult obesity prevalence maps. https://www.cdc.gov/obesity/data/prevalence-maps.html (accessed April 25, 2017).
Center for Workforce Studies. 2006. Assuring the sufficiency of a frontline workforce project: A national study of licensed social workers. http://workforce.socialworkers.org/studies/nasw_06_execsummary.pdf (accessed August 25, 2017).
Chan, D. S. M., A. R. Vieira, D. Aune, E. V. Bandera, D. C. Greenwood, A. McTiernan, D. Navarro Rosenblatt, I. Thune, R. Vieira, and T. Norat. 2014. Body mass index and survival in women with breast cancer—systematic literature review and meta-analysis of 82 follow-up studies. Annals of Oncology 25(10):1901–1914.
Chung, S., S. Zhu, E. Friedmann, C. Kelleher, A. Kozlovsky, K. W. Macfarlane, K. H. R. Tkaczuk, A. S. Ryan, and K. A. Griffith. 2016. Weight loss with mindful eating in African American women following treatment for breast cancer: A longitudinal study. Supportive Care in Cancer 24(4):1875–1881.
CMS (Centers for Medicare & Medicaid Services). 2016. Certification of Medicare Diabetes Prevention Program. https://www.cms.gov/Research-Statistics-Data-and-Systems/Research/ActuarialStudies/Downloads/Diabetes-Prevention-Certification-2016-03-14.pdf (accessed May 3, 2017).
Conlon, B. A., M. Kahan, M. Martinez, K. Isaac, A. Rossi, R. Skyhart, J. Wylie-Rosett, and A. Moadel-Robblee. 2015. Development and evaluation of the curriculum for BOLD (Bronx Oncology Living Daily) healthy living: A diabetes prevention and control program for underserved cancer survivors. Journal of Cancer Education 30(3):535–545.
Courneya, K. S., J. R. Mackey, G. J. Bell, L. W. Jones, C. J. Field, and A. S. Fairey. 2003. Randomized controlled trial of exercise training in postmenopausal breast cancer survivors: Cardiopulmonary and quality of life outcomes. Journal of Clinical Oncology 21(9):1660–1668.
Courneya, K. S., R. J. Segal, J. R. Mackey, K. Gelmon, R. D. Reid, C. M. Friedenreich, A. B. Ladha, C. Proulx, J. K. H. Vallance, K. Lane, Y. Yasui, and D. C. McKenzie. 2007. Effects of aerobic and resistance exercise in breast cancer patients receiving adjuvant chemotherapy: A multicenter randomized controlled trial. Journal of Clinical Oncology 25(28):4396–4404.
Courneya, K. S., J. K. Vallance, S. N. Culos-Reed, M. L. McNeely, G. J. Bell, J. R. Mackey, Y. Yasui, Y. Yuan, C. E. Matthews, D. C. W. Lau, D. Cook, and C. M. Friedenreich. 2012. The Alberta Moving Beyond Breast CancER (AMBER) cohort study: A prospective study of physical activity and health-related fitness in breast cancer survivors. BMC Cancer 12(1):525.
Courneya, K. S., D. C. McKenzie, J. R. Mackey, K. Gelmon, C. M. Friedenreich, Y. Yasui, R. D. Reid, D. Cook, D. Jespersen, C. Proulx, L. B. Dolan, C. C. Forbes, E. Wooding, L. Trinh, and R. J. Segal. 2013. Effects of exercise dose and type during breast cancer chemotherapy: Multicenter randomized trial. Journal of the National Cancer Institute 105(23):1821–1832.
Courneya, K. S., R. J. Segal, D. C. McKenzie, H. Dong, K. Gelmon, C. M. Friedenreich, Y. Yasui, R. D. Reid, J. J. Crawford, and J. R. Mackey. 2014a. Effects of exercise during adjuvant chemotherapy on breast cancer outcomes. Medicine and Science in Sports and Exercise 46(9):1744–1751.
Courneya, K. S., J. Vardy, S. Gill, D. Jonker, P. O’Brien, C. M. Friedenreich, H. Dhillon, R. K. S. Wong, R. M. Meyer, J. J. Crawford, K. L. Campbell, H. Prapavessis, C. O’Callaghan, J. Turner, L. M. Spencer, H. P. van der Ploeg, D. Tu, and C. M. Booth. 2014b. Update on the colon health and life-long exercise change trial: A phase III study of the impact of an exercise program on disease-free survival in colon cancer survivors. Current Colorectal Cancer Reports 10(3):321–328.
Courneya, K. S., J. L. Vardy, C. J. Callaghan, C. M. Friedenreich, K. L. Campbell, H. Prapavessis, J. J. Crawford, P. Brien, H. M. Dhillon, D. J. Jonker, N. S. Chua, S. Lupichuk, M. S. Sanatani, S. Gill, R. M. Meyer, S. Begbie, T. Bonaventura, M. E. Burge, J. Turner, D. Tu, and C. M. Booth. 2016. Effects of a structured exercise program on physical activity and fitness in colon cancer survivors: One year feasibility results from the challenge trial. Cancer Epidemiology Biomarkers & Prevention 25(6):969.
Crookes, D. M., R. C. Shelton, P. Tehranifar, C. Aycinena, A. O. Gaffney, P. Koch, I. R. Contento, and H. Greenlee. 2016. Social networks and social support for healthy eating among Latina breast cancer survivors: Implications for social and behavioral interventions. Journal of Cancer Survivorship 10(2):291–301.
Dansinger, M. L., J. Gleason, J. L. Griffith, H. P. Selker, and E. J. Schaefer. 2005. Comparison of the Atkins, Ornish, Weight Watchers, and zone diets for weight loss and heart disease risk reduction: A randomized trial. JAMA 293(1):43–53.
Delahanty, L. M., M. Riggs, S. S. Klioze, R. D. Chew, R. D. England, and A. Digenio. 2016. Maximizing retention in long-term clinical trials of a weight loss agent: Use of a dietitian support team. Obesity Science & Practice 2(3):256–265.
Demark-Wahnefried, W., E. C. Clipp, I. M. Lipkus, D. Lobach, D. Clutter Snyder, and R. Sloane. 2007. Main outcomes of the FRESH START trial: A sequentially tailored, diet and exercise mailed print intervention among breast and prostate cancer survivors. Journal of Clinical Oncology 25(19):2709–2718.
Demark-Wahnefried, W., M. C. Morey, R. Sloane, D. C. Snyder, P. E. Miller, T. J. Hartman, and H. J. Cohen. 2012. Reach out to enhance wellness home-based diet-exercise intervention promotes reproducible and sustainable long-term improvements in health behaviors, body weight, and physical functioning in older, overweight/obese cancer survivors. Journal of Clinical Oncology 30(19):2354–2361.
Demark-Wahnefried, W., L. W. Jones, D. C. Snyder, R. Sloane, G. G. Kimmick, D. C. Hughes, H. Badr, P. E. Miller, L. Burke, and I. Lipkus. 2014. Daughters and mothers against breast cancer (DAMES): Main outcomes of a randomized controlled trial of weight loss in overweight mothers with breast cancer and their overweight daughters. Cancer 120(16):2522–2534.
Demark-Wahnefried, W., G. A. Colditz, C. L. Rock, R. L. Sedjo, J. Liu, K. Y. Wolin, H. Krontiras, T. Byers, B. Pakiz, B. A. Parker, M. Naughton, A. Elias, and P. A. Ganz. 2015. Quality of life outcomes from the exercise and nutrition enhance recovery and good health for you (ENERGY)-randomized weight loss trial among breast cancer survivors. Breast Cancer Research and Treatment 154(2):329–337.
Demark-Wahnefried, W., S. Rais-Bahrami, R. A. Desmond, J. B. Gordetsky, G. R. Hunter, E. S. Yang, M. Azrad, A. D. Fruge, Y. Tsuruta, L. A. Norian, R. Segal, and W. E. Grizzle. 2017. Presurgical weight loss affects tumour traits and circulating biomarkers in men with prostate cancer. British Journal of Cancer 117(9):1303–1313.
Denlinger, C. S., and J. A. Ligibel. 2013. NCCN presents new guidelines for survivorship. Journal of the National Comprehensive Cancer Network 11(5 Suppl):640–644.
Denlinger, C. S., K. Filchner, M. A. O’Grady, E. Sein, E. Slater, M. Slifker, S. Litwin, H. Borghaei, and S. J. Cohen. 2015. Adherence to NCCN survivorship guidelines in non-small cell lung cancer (NSCLC) and colorectal cancer (CRC). Journal of Clinical Oncology 32(15 Suppl):e20593.
Dennis Parker, E. A., V. B. Sheppard, and L. Adams-Campbell. 2013. Compliance with national nutrition recommendations among breast cancer survivors in “Stepping STONE.” Integrative Cancer Therapies 13(2):114–120.
Derry, H. M., L. M. Jaremka, J. M. Bennett, J. Peng, R. Andridge, C. Shapiro, W. B. Malarkey, C. F. Emery, R. Layman, E. Mrozek, R. Glaser, and J. K. Kiecolt-Glaser. 2015. Yoga and self-reported cognitive problems in breast cancer survivors: A randomized controlled trial. Psycho-Oncology 24(8):958–966.
Dhillon, J., B. A. Craig, H. J. Leidy, A. F. Amankwaah, K. Osei-Boadi Anguah, A. Jacobs, B. L. Jones, J. B. Jones, C. L. Keeler, C. E. M. Keller, M. A. McCrory, R. L. Rivera, M. Slebodnik, R. D. Mattes, and R. M. Tucker. 2016. The effects of increased protein intake on fullness: A meta-analysis and its limitations. Journal of the Academy of Nutrition and Dietetics 116(6):968–983.
Diabetes Prevention Program Research Group. 2002. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. New England Journal of Medicine 346(6):393–403.
Diabetes Prevention Program Research Group. 2015. Long-term effects of lifestyle intervention or metformin on diabetes development and microvascular complications over 15-year follow-up: The Diabetes Prevention Program Outcomes Study. Lancet Diabetes & Endocrinology 3(11):866–875.
Dietz, W. H., L. S. Solomon, N. Pronk, S. K. Ziegenhorn, M. Standish, M. M. Longjohn, D. D. Fukuzawa, I. U. Eneli, L. Loy, N. D. Muth, E. J. Sanchez, J. Bogard, and D. W. Bradley. 2015. An integrated framework for the prevention and treatment of obesity and its related chronic diseases. Health Affairs 34(9):1456–1463.
Djuric, Z., N. M. DiLaura, I. Jenkins, L. Darga, C. K. L. Jen, D. Mood, E. Bradley, and W. M. Hryniuk. 2002. Combining weight-loss counseling with the Weight Watchers plan for obese breast cancer survivors. Obesity Research 10(7):657–665.
Djuric, Z., J. Mirasolo, L. Kimbrough, D. R. Brown, L. K. Heilbrun, L. Canar, R. Venkatranamamoorthy, and M. S. Simon. 2009. A pilot trial of spirituality counseling for weight loss maintenance in African American breast cancer survivors. Journal of the National Medical Association 101(6):552–564.
Doleman, B., K. T. Mills, S. Lim, M. D. Zelhart, and G. Gagliardi. 2016. Body mass index and colorectal cancer prognosis: A systematic review and meta-analysis. Techniques in Coloproctology 20(8):517–535.
Eakin, E. G., S. C. Hayes, M. R. Haas, M. M. Reeves, J. L. Vardy, F. Boyle, J. E. Hiller, G. D. Mishra, A. D. Goode, M. Jefford, B. Koczwara, C. M. Saunders, W. Demark-Wahnefried, K. S. Courneya, K. H. Schmitz, A. Girgis, K. White, K. Chapman, A. G. Boltong, K. Lane, S. McKiernan, L. Millar, L. O’Brien, G. Sharplin, P. Baldwin, and E. L. Robson. 2015. Healthy Living after Cancer: A dissemination and implementation study evaluating a telephone-delivered healthy lifestyle program for cancer survivors. BMC Cancer 15(1):992.
Elmer, P. J., E. Obarzanek, W. M. Vollmer, D. Simons-Morton, V. J. Stevens, D. R. Young, P. H. Lin, C. Champagne, D. W. Harsha, L. P. Svetkey, J. Ard, P. J. Brantley, M. A. Proschan, T. P. Erlinger, and L. J. Appel. 2006. Effects of comprehensive lifestyle modification on diet, weight, physical fitness, and blood pressure control: 18-month results of a randomized trial. Annals of Internal Medicine 144(7):485–495.
Fahey, P. P., K.-A. Mallitt, T. Astell-Burt, G. Stone, and D. C. Whiteman. 2015. Impact of pre-diagnosis behavior on risk of death from esophageal cancer: A systematic review and meta-analysis. Cancer Causes and Control 26(10):1365–1373.
Fazzino, T. L., N. J. Sporn, and C. A. Befort. 2016. A qualitative evaluation of a group phone-based weight loss intervention for rural breast cancer survivors: Themes and mechanisms of success. Supportive Care in Cancer 24(7):3165–3173.
Fisher, A., K. Williams, R. Beeken, and J. Wardle. 2015. Recall of physical activity advice was associated with higher levels of physical activity in colorectal cancer patients. BMJ Open 5(4):e006853.
Fiuza-Luces, C., N. Garatachea, N. A. Berger, and A. Lucia. 2013. Exercise is the real polypill. Physiology 28(5):330–358.
Ford, J. S., M. Barnett, and R. Werk. 2014. Health behaviors of childhood cancer survivors. Children 1(3):355–373.
Foster-Schubert, K. E., C. M. Alfano, C. R. Duggan, L. Xiao, K. L. Campbell, A. Kong, C. Bain, C. Y. Wang, G. Blackburn, and A. McTiernan. 2012. Effect of diet and exercise, alone or combined, on weight and body composition in overweight-to-obese postmenopausal women. Obesity 20(8):1628–1638.
Friedenreich, C. M., H. K. Neilson, M. S. Farris, and K. S. Courneya. 2016. Physical activity and cancer outcomes: A precision medicine approach. Clinical Cancer Research 22(19):4766–4775.
Fuentes-Mattei, E., G. Velazquez-Torres, L. Phan, F. Zhang, P.-C. Chou, J.-H. Shin, H. H. Choi, J.-S. Chen, R. Zhao, J. Chen, C. Gully, C. Carlock, Y. Qi, Y. Zhang, Y. Wu, F. J. Esteva, Y. Luo, W. L. McKeehan, J. Ensor, G. N. Hortobagyi, L. Pusztai, W. Fraser Symmans, M.-H. Lee, and S.-C. Jim Yeung. 2014. Effects of obesity on transcriptomic changes and cancer hallmarks in estrogen receptor–positive breast cancer. Journal of the National Cancer Institute 106(7):dju158.
Garber, C. E., B. Blissmer, M. R. Deschenes, B. A. Franklin, M. J. Lamonte, I. M. Lee, D. C. Nieman, and D. P. Swain. 2011. American College of Sports Medicine position stand. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: Guidance for prescribing exercise. Medicine and Science in Sports and Exercise 43(7):1334–1359.
Garcia, M. C., M. Faul, G. Massetti, C. C. Thomas, Y. Hong, U. E. Bauer, and M. F. Iademarco. 2017. Reducing potentially excess deaths from the five leading causes of death in the rural United States. Morbidity and Mortality Weekly Report 66(2):1–7.
Garvey, W. T., J. I. Mechanick, E. M. Brett, A. J. Garber, D. L. Hurley, A. M. Jastreboff, K. Nadolsky, R. Pessah-Pollack, and R. Plodkowski. 2016. American association of clinical endocrinologists and american college of endocrinology comprehensive clinical practice guidelines for medical care of patients with obesity. Endocrine Practice 22(Suppl 3):1–203.
Goetz, S. J. 2008. Self-employment in rural America: The new economic reality. Rural Realities 2(3):1–13.
Goldberg, J. H., and M. Kiernan. 2005. Innovative techniques to address retention in a behavioral weight-loss trial. Health Education Research 20(4):439–447.
Golden, S. H., A. Ferketich, J. Boyington, S. Dugan, E. Garroutte, P. G. Kaufmann, J. Krok, A. Kuo, A. N. Ortega, T. Purnell, and S. Srinivasan. 2015. Transdisciplinary cardiovascular and cancer health disparities training: Experiences of the Centers for Population Health and Health Disparities. American Journal of Public Health 105(Suppl 3):S395–S402.
Goodwin, P. J., and V. Stambolic. 2015. Impact of the obesity epidemic on cancer. Annual Review of Medicine 66:281–296.
Goodwin, P. J., R. J. Segal, M. Vallis, J. A. Ligibel, G. R. Pond, A. Robidoux, G. L. Blackburn, B. Findlay, J. R. Gralow, S. Mukherjee, M. Levine, and K. I. Pritchard. 2014. Randomized trial of a telephone-based weight loss intervention in postmenopausal women with breast cancer receiving letrozole: The LISA Trial. Journal of Clinical Oncology 32(21):2231–2239.
Granger, C. L., B. Connolly, L. Denehy, N. Hart, P. Antippa, K.-Y. Lin, and S. M. Parry. 2017. Understanding factors influencing physical activity and exercise in lung cancer: A systematic review. Supportive Care in Cancer 25(3):983–999.
Greenlee, H. A., K. D. Crew, J. M. Mata, P. S. McKinley, A. G. Rundle, W. Zhang, Y. Liao, W. Y. Tsai, and D. L. Hershman. 2013. A pilot randomized controlled trial of a commercial diet and exercise weight loss program in minority breast cancer survivors. Obesity 21(1):65–76.
Greenlee, H., A. O. Gaffney, A. C. Aycinena, P. Koch, I. Contento, W. Karmally, J. M. Richardson, E. Lim, W.-Y. Tsai, K. Crew, M. Maurer, K. Kalinsky, and D. L. Hershman. 2015. ¡Cocinar para su salud!: Randomized controlled trial of a culturally-based dietary intervention among Hispanic breast cancer survivors. Journal of the Academy of Nutrition and Dietetics 115(5):709–723.
Greenlee, H., Z. Shi, C. L. Sardo Molmenti, A. Rundle, and W. Y. Tsai. 2016. Trends in obesity prevalence in adults with a history of cancer: Results from the U.S. National Health Interview Survey, 1997 to 2014. Journal of Clinical Oncology 34(26):3133–3140.
Guenancia, C., A. Lefebvre, D. Cardinale, A. F. Yu, S. Ladoire, F. Ghiringhelli, M. Zeller, L. Rochette, Y. Cottin, and C. Vergely. 2016. Obesity as a risk factor for anthracyclines and trastuzumab cardiotoxicity in breast cancer: A systematic review and meta-analysis. Journal of Clinical Oncology 34(26):3157–3165.
Gupta, A., K. Majumder, N. Arora, H. G. Mayo, P. P. Singh, M. S. Beg, R. Hughes, S. Singh, and D. H. Johnson. 2016. Premorbid body mass index and mortality in patients with lung cancer: A systematic review and meta-analysis. Lung Cancer 102:49–59.
Harrigan, M., B. Cartmel, E. Loftfield, T. Sanft, A. B. Chagpar, Y. Zhou, M. Playdon, F. Li, and M. L. Irwin. 2016. Randomized trial comparing telephone versus in-person weight loss counseling on body composition and circulating biomarkers in women treated for breast cancer: The lifestyle, exercise, and nutrition (LEAN) study. Journal of Clinical Oncology 34(7):669–676.
Harris, M. N., D. L. Swift, V. H. Myers, C. P. Earnest, N. M. Johannsen, C. M. Champagne, B. D. Parker, E. Levy, K. C. Cash, and T. S. Church. 2013. Cancer survival through lifestyle change (CASTLE): A pilot study of weight loss. International Journal of Behavioral Medicine 20(3):403–412.
Hartman, S. J., C. R. Marinac, L. Natarajan, and R. E. Patterson. 2015. Lifestyle factors associated with cognitive functioning in breast cancer survivors. Psycho-Oncology 24(6):669–675.
Hayes, S. C., K. Johansson, C. M. Alfano, and K. Schmitz. 2011. Exercise for breast cancer survivors: Bridging the gap between evidence and practice. Translational Behavioral Medicine 1(4):539–544.
Henderson, T. O., K. Ness, and H. J. Cohen. 2014. Accelerated aging among cancer survivors: From pediatrics to geriatrics. American Society of Clinical Oncology 2014 Educational Book e423–e430.
Hooning, M. J., A. Botma, B. M. P. Aleman, M. H. A. Baaijens, H. Bartelink, J. G. M. Klijn, C. W. Taylor, and F. E. van Leeuwen. 2007. Long-term risk of cardiovascular disease in 10-year survivors of breast cancer. Journal of the National Cancer Institute 99(5):365–375.
Hu, M.-B., H. Xu, P.-D. Bai, H.-W. Jiang, and Q. Ding. 2014. Obesity has multifaceted impact on biochemical recurrence of prostate cancer: A dose–response meta-analysis of 36,927 patients. Medical Oncology 31(2):829.
Hu, T., K. T. Mills, L. Yao, K. Demanelis, M. Eloustaz, W. S. Yancy, T. N. Kelly, J. He, and L. A. Bazzano. 2012. Effects of low-carbohydrate diets versus low-fat diets on metabolic risk factors: A meta-analysis of randomized controlled clinical trials. American Journal of Epidemiology 176(Suppl 7):S44–S54.
Huang, J. S., L. Dillon, L. Terrones, L. Schubert, W. Roberts, J. Finklestein, M. C. Swartz, G. J. Norman, and K. Patrick. 2014. Fit4life: A weight loss intervention for children who have survived childhood leukemia. Pediatric Blood & Cancer 61(5):894–900.
Imayama, I., C. M. Ulrich, C. M. Alfano, C. Wang, L. Xiao, M. H. Wener, K. L. Campbell, C. Duggan, K. E. Foster-Schubert, A. Kong, C. E. Mason, C. Y. Wang, G. L. Blackburn, C. E. Bain, H. J. Thompson, and A. McTiernan. 2012. Effects of a caloric restriction weight loss diet and exercise on inflammatory biomarkers in overweight/ obese postmenopausal women: A randomized controlled trial. Cancer Research 72(9):2314–2326.
IOM (Institute of Medicine). 2006. From cancer patient to cancer survivor: Lost in transition. Washington, DC: The National Academies Press.
IOM. 2012. The role of obesity in cancer survival and recurrence: Workshop summary. Washington, DC: The National Academies Press.
IOM. 2013. Identifying and addressing the deeds of adolescents and young adults with cancer: Workshop summary. Washington, DC: The National Academies Press.
Ireland, K., P. Newby, E. Keefe, A. Meyers, A. Lim-Miller, and C. Lenders. In press. Prevention and treatment of pediatric obesity. In Encyclopedia of human biology, 3rd ed, edited by R. Dulbecco and J. Abelson. Oxford: Elsevier.
Irwin, M. L., B. Cartmel, C. P. Gross, E. Ercolano, F. Li, X. Yao, M. Fiellin, S. Capozza, M. Rothbard, Y. Zhou, M. Harrigan, T. Sanft, K. Schmitz, T. Neogi, D. Hershman, and J. Ligibel. 2015. Randomized exercise trial of aromatase inhibitor-induced arthralgia in breast cancer survivors. Journal of Clinical Oncology 33(10):1104–1111.
Irwin, M. L., B. Cartmel, M. Harrigan, F. Li, T. Sanft, L. Shockro, K. O’Connor, N. Campbell, S. M. Tolaney, E. L. Mayer, R. Yung, R. A. Freedman, A. H. Partridge, and J. A. Ligibel. 2017. Effect of the LIVESTRONG at the YMCA exercise program on physical activity, fitness, quality of life, and fatigue in cancer survivors. Cancer 123(7):1249–1258.
Jakicic, J. M., C. M. Egan, A. N. Fabricatore, S. A. Gaussoin, S. P. Glasser, L. A. Hesson, W. C. Knowler, W. Lang, J. G. Regensteiner, P. M. Ribisl, D. H. Ryan, and the Look AHEAD Research Group. 2013. Four-year change in cardiorespiratory fitness and influence on glycemic control in adults with type 2 diabetes in a randomized trial: The Look Ahead Trial. Diabetes Care 36(5):1297–1303.
Jensen, M. D., D. H. Ryan, C. M. Apovian, J. D. Ard, A. G. Comuzzie, K. A. Donato, F. B. Hu, V. S. Hubbard, J. M. Jakicic, R. F. Kushner, C. M. Loria, B. E. Millen, C. A. Nonas, F. X. Pi-Sunyer, J. Stevens, V. J. Stevens, T. A. Wadden, B. M. Wolfe, S. Z. Yanovski, H. S. Jordan, K. A. Kendall, L. J. Lux, R. Mentor-Marcel, L. C. Morgan, M. G. Trisolini, J. Wnek, J. L. Anderson, J. L. Halperin, N. M. Albert, B. Bozkurt, R. G. Brindis, L. H. Curtis, D. DeMets, J. S. Hochman, R. J. Kovacs, E. M. Ohman, S. J. Pressler, F. W. Sellke, W. K. Shen, S. C. Smith, Jr., and G. F. Tomaselli. 2014. 2013 AHA/ACC/TOS guideline for the management of overweight and obesity in adults: A report of the American College of Cardiology/American Heart Association task force on practice guidelines and the obesity society. Circulation 129(25 Suppl 2):S102–S138.
Johnston, B. C., S. Kanters, K. Bandayrel, and et al. 2014. Comparison of weight loss among named diet programs in overweight and obese adults: A meta-analysis. JAMA 312(9):923–933.
Jones, L. W., K. S. Courneya, A. S. Fairey, and J. R. Mackey. 2004. Effects of an oncologist’s recommendation to exercise on self-reported exercise behavior in newly diagnosed breast cancer survivors: A single-blind, randomized controlled trial. Annals of Behavioral Medicine 28(2):105–113.
Jones, L. W., K. S. Courneya, C. Peddle, and J. R. Mackey. 2005. Oncologists’ opinions towards recommending exercise to patients with cancer: A Canadian National Survey. Supportive Care in Cancer 13(11):929–937.
Jones, L. W., D. Watson, J. E. Herndon 2nd, N. D. Eves, B. E. Haithcock, G. Loewen, L. Kohman. 2010. Peak oxygen consumption and long-term all-cause mortality in nonsmall cell lung cancer. Cancer 116(20):4825–4832.
Jones, L. W., K. S. Courneya, J. R. Mackey, H. B. Muss, E. N. Pituskin, J. M. Scott, W. E. Hornsby, A. D. Coan, J. E. Herndon, P. S. Douglas, and M. Haykowsky. 2012. Cardiopulmonary function and age-related decline across the breast cancer survivorship continuum. Journal of Clinical Oncology 30(20):2530–2537.
Jones, L. W., L. A. Habel, E. Weltzien, A. Castillo, D. Gupta, C. H. Kroenke, M. L. Kwan, C. P. Quesenberry, Jr., J. Scott, B. Sternfeld, A. Yu, L. H. Kushi, and B. J. Caan. 2016. Exercise and risk of cardiovascular events in women with nonmetastatic breast cancer. Journal of Clinical Oncology 34(23):2743–2749.
Jung, S.-H., D.-H. Yang, J.-S. Ahn, S.-S. Lee, S.-Y. Ahn, Y.-K. Kim, H. J. Kim, and J.-J. Lee. 2014. Decreased body mass index is associated with poor prognosis in patients with multiple myeloma. Annals of Hematology 93(5):835–840.
Karvinen, K., B. Bruner, and T. Truant. 2015. The teachable moment after cancer diagnosis: Perceptions from oncology nurses. Oncology Nursing Forum 42(6):602–609.
Katsanos, C. S., H. Kobayashi, M. Sheffield-Moore, A. Aarsland, and R. R. Wolfe. 2006. A high proportion of leucine is required for optimal stimulation of the rate of muscle protein synthesis by essential amino acids in the elderly. American Journal of Physiology-Endocrinology and Metabolism 291(2):E381–E387.
Kenzik, K. M., M. C. Morey, H. J. Cohen, R. Sloane, and W. Demark-Wahnefried. 2015. Symptoms, weight loss, and physical function in a lifestyle intervention study of older cancer survivors. Journal of Geriatric Oncology 6(6):424–432.
Kim, R. B., A. Phillips, K. Herrick, M. Helou, C. Rafie, M. S. Anscher, R. B. Mikkelsen, and Y. Ning. 2013. Physical activity and sedentary behavior of cancer survivors and noncancer individuals: Results from a national survey. PLoS ONE 8(3):e57598.
King, D., P. Miranda, B. Gor, R. Fuchs-Young, J. Chilton, R. Hajek, I. Torres-Vigil, M. A. Hernández-Valero, S. A. Snipes, and L. Jones. 2010. Addressing cancer health disparities using a global biopsychosocial approach. Cancer 116(2):264–269.
Kirkwood, M. K., S. S. Bruinooge, M. A. Goldstein, D. F. Bajorin, and M. P. Kosty. 2014. Enhancing the American Society of Clinical Oncology workforce information system with geographic distribution of oncologists and comparison of data sources for the number of practicing oncologists. Journal of Oncology Practices 10(1):32–38.
Klepin, H. D., S. G. Mohile, and S. Mihalko. 2013. Exercise for older cancer patients: Feasible and helpful? Interdisciplinary Topics in Gerontology 38:146–157.
Kramer, C. K., B. Zinman, and R. Retnakaran. 2013. Are metabolically healthy overweight and obesity benign conditions?: A systematic review and meta-analysis. Annals of Internal Medicine 159(11):758–769.
Kreuter, M. W., S. N. Lukwago, R. D. Bucholtz, E. M. Clark, and V. Sanders-Thompson. 2003. Achieving cultural appropriateness in health promotion programs: Targeted and tailored approaches. Health Education and Behavior 30(2):133–146.
Lakoski, S. G., C. E. Barlow, G. J. Koelwyn, W. E. Hornsby, J. Hernandez, L. F. DeFina, N. B. Radford, S. M. Thomas, J. E. Herndon, J. Peppercorn, P. S. Douglas, and L. W. Jones. 2013. The influence of adjuvant therapy on cardiorespiratory fitness in early-stage breast cancer seven years after diagnosis: The Cooper Center Longitudinal Study. Breast Cancer Research and Treatment 138(3):909–916.
Lakoski, S. G., B. L. Willis, C. E. Barlow, D. Leonard, A. Gao, N. B. Radford, S. W. Farrell, P. S. Douglas, J. D. Berry, L. F. DeFina, and L. W. Jones. 2015. Midlife cardiorespiratory fitness, incident cancer, and survival after cancer in men: The Cooper Center Longitudinal Study. JAMA Oncology 1(2):231–237.
Lauby-Secretan, B., C. Scoccianti, D. Loomis, Y. Grosse, F. Bianchini, and K. Straif. 2016. Body fatness and cancer—viewpoint of the IARC working group. New England Journal of Medicine 375(8):794–798.
Laukkanen, J. A., F. Zaccardi, H. Khan, S. Kurl, S. Y. Jae, and R. Rauramaa. 2016. Long-term change in cardiorespiratory fitness and all-cause mortality. Mayo Clinic Proceedings 91(9):1183–1188.
Lawler, S., G. Maher, M. Brennan, A. Goode, M. M. Reeves, and E. Eakin. 2017. Get healthy after breast cancer—examining the feasibility, acceptability and outcomes of referring breast cancer survivors to a general population telephone-delivered program targeting physical activity, healthy diet and weight loss. Supportive Care in Cancer 25(6):1953–1962.
Leidy, H. J., P. M. Clifton, A. Astrup, T. P. Wycherley, M. S. Westerterp-Plantenga, N. D. Luscombe-Marsh, S. C. Woods, and R. D. Mattes. 2015. The role of protein in weight loss and maintenance. American Journal of Clinical Nutrition 101(6):1320S–1329S.
Lewis, R. S., and J. H. Sunshine. 2007. Radiation oncologists in the United States. International Journal of Radiation Oncology, Biology, and Physics 69(2):518–527.
Ligibel, J. A., C. M. Alfano, K. S. Courneya, W. Demark-Wahnefried, R. A. Burger, R. T. Chlebowski, C. J. Fabian, A. Gucalp, D. L. Hershman, M. M. Hudson, L. W. Jones, M. Kakarala, K. K. Ness, J. K. Merrill, D. S. Wollins, and C. A. Hudis. 2014. American Society of Clinical Oncology position statement on obesity and cancer. Journal of Clinical Oncology 32(31):3568–3574.
Lim, J. W., P. Gonzalez, M. F. Wang-Letzkus, and K. T. Ashing-Giwa. 2009. Understanding the cultural health belief model influencing health behaviors and health-related quality of life between Latina and Asian-American breast cancer survivors. Supportive Care in Cancer 17(9):1137–1147.
Lindberg, N. M., V. J. Stevens, and R. O. Halperin. 2013. Weight-loss interventions for Hispanic populations: The role of culture. Journal of Obesity 6.
Linkov, F., G. L. Maxwell, A. S. Felix, Y. Lin, D. Lenzner, D. H. Bovbjerg, A. Lokshin, M. Hennon, J. M. Jakicic, B. H. Goodpaster, and J. P. DeLany. 2012. Longitudinal evaluation of cancer-associated biomarkers before and after weight loss in renew study participants: Implications for cancer risk reduction. Gynecologic Oncology 125(1):114–119.
Look AHEAD Research Group. 2014. Eight-year weight losses with an intensive lifestyle intervention: The Look AHEAD study. Obesity 22(1):5–13.
Loprinzi, P. D., and H. Lee. 2014. Rationale for promoting physical activity among cancer survivors: Literature review and epidemiologic examination. Oncology Nursing Forum 41(2):117–125.
Lustberg, M. B., R. E. Reinbolt, and C. L. Shapiro. 2012. Bone health in adult cancer survivorship. Journal of Clinical Oncology 30(30):3665–3674.
Mason, C., K. E. Foster-Schubert, I. Imayama, A. Kong, L. Xiao, C. Bain, K. L. Campbell, C. Y. Wang, C. R. Duggan, C. M. Ulrich, C. M. Alfano, G. L. Blackburn, and A. McTiernan. 2011. Dietary weight loss and exercise effects on insulin resistance in postmenopausal women. American Journal of Preventive Medicine 41(4):366–375.
Mazzeo, R. S., P. Cavanagh, W. J. Evans, M. Fiatarone, J. Hagberg, E. McAuley, and J. Startzell. 1998. American College of Sports Medicine: Position stand on exercise and physical activity for older adults. Medicine and Science in Sports and Exercise 30:992–1008.
McCambridge, T. M., and P. R. Stricker. 2008. Strength training by children and adolescents. Pediatrics 121(4):835–840.
McCarroll, M. L., S. Armbruster, H. E. Frasure, M. D. Gothard, K. M. Gil, and M. B. Kavanagh. 2014. Self-efficacy, quality of life, and weight loss in overweight/obese endometrial cancer survivors (SUCCEED): A randomized controlled trial. Gynecologic Oncology 132(2):397–402.
McCarroll, M. L., S. Armbruster, R. J. Pohle-Krauza, A. M. Lyzen, S. Min, and D. W. Nash. 2015. Feasibility of a lifestyle intervention for overweight/obese endometrial and breast cancer survivors using an interactive mobile application. Gynecologic Oncology 137(2):508–515.
Mellerson, J., H. Landrine, Y. Hao, I. Corral, L. Zhao, and D. L. Cooper. 2010. Residential segregation and exercise among a national sample of Hispanic adults. Health Place 16(3):613–615.
Meneses-Echávez, J. F., E. González-Jiménez, and R. Ramírez-Vélez. 2015. Effects of supervised exercise on cancer-related fatigue in breast cancer survivors: A systematic review and meta-analysis. BMC Cancer 15(1):77.
Meyerhardt, J. A., S. Ogino, G. J. Kirkner, A. T. Chan, B. Wolpin, K. Ng, K. Nosho, K. Shima, E. L. Giovannucci, M. Loda, and C. S. Fuchs. 2009. Interaction of molecular markers and physical activity on mortality in patients with colon cancer. Clinical Cancer Research 15(18):5931–5936.
Miller, P. E., M. C. Morey, T. J. Hartman, D. C. Snyder, R. Sloane, H. J. Cohen, and W. Demark-Wahnefried. 2012. Dietary patterns differ between urban and rural older, long-term survivors of breast, prostate, and colorectal cancer and are associated with body mass index. Journal of the Academy of Nutrition and Dietetics 112(6):824–831.
Morey, M. C., D. C. Snyder, R. Sloane, H. J. Cohen, B. Peterson, T. J. Hartman, P. Miller, D. C. Mitchell, and W. Demark-Wahnefried. 2009. Effects of home-based diet and exercise on functional outcomes among older, overweight long-term cancer survivors: Renew: A randomized controlled trial. JAMA 301(18):1883–1891.
Muscaritoli, M., S. D. Anker, J. Argiles, Z. Aversa, J. M. Bauer, G. Biolo, Y. Boirie, I. Bosaeus, T. Cederholm, P. Costelli, K. C. Fearon, A. Laviano, M. Maggio, F. Rossi Fanelli, S. M. Schneider, A. Schols, and C. C. Sieber. 2010. Consensus definition of sarcopenia, cachexia and pre-cachexia: Joint document elaborated by special interest groups (SIG) “cachexia-anorexia in chronic wasting diseases” and “nutrition in geriatrics.” Clinical Nutrition 29(2):154–159.
Mustian, K. M., L. K. Sprod, M. Janelsins, L. J. Peppone, O. G. Palesh, K. Chandwani, P. S. Reddy, M. K. Melnik, C. Heckler, and G. R. Morrow. 2013. Multicenter, randomized controlled trial of yoga for sleep quality among cancer survivors. Journal of Clinical Oncology 31(26):3233–3241.
NASEM (National Academies of Sciences, Engineering, and Medicine). 2016. Examining access to nutrition care in outpatient cancer centers: Proceedings of a workshop. Washington, DC: The National Academies Press.
Nayak, P., R. J. Paxton, H. Holmes, H. Thanh Nguyen, and L. S. Elting. 2015. Racial and ethnic differences in health behaviors among cancer survivors. American Journal of Preventive Medicine 48(6):729–736.
NCI (National Cancer Institute). 2008. Cancer health disparities. https://www.cancer.gov/about-nci/organization/crchd/cancer-health-disparities-fact-sheet#q5 (accessed June 29, 2017).
NCI. 2016. Statistics. https://cancercontrol.cancer.gov/ocs/statistics/statistics.html (accessed September 13, 2017).
Nichols, H. B., A. Trentham-Dietz, K. M. Egan, L. Titus-Ernstoff, M. D. Holmes, A. J. Bersch, C. N. Holick, J. M. Hampton, M. J. Stampfer, W. C. Willett, and P. A. Newcomb. 2009. Body mass index before and after breast cancer diagnosis: Associations with all-cause, breast cancer, and cardiovascular disease mortality. Cancer Epidemiology, Biomarkers and Prevention 18(5):1403–1409.
Niu, C., L. Eng, X. Qiu, X. Shen, O. Espin-Garcia, Y. Song, D. Pringle, M. Mahler, O. Halytskyy, R. Charow, C. Lam, R. M. Shani, J. Villeneuve, K. Tiessen, M. C. Brown, P. Selby, D. Howell, J. M. Jones, W. Xu, G. Liu, and S. M. H. Alibhai. 2015. Lifestyle behaviors in elderly cancer survivors: A comparison with middle-age cancer survivors. Journal of Oncology Practice 11(4):e450–e459.
Nyrop, K. A., A. M. Deal, G. R. Williams, E. J. Guerard, M. Pergolotti, and H. B. Muss. 2016. Physical activity communication between oncology providers and patients with early-stage breast, colon, or prostate cancer. Cancer 122(3):470–476.
Ogden, C. L., M. D. Carroll, C. D. Fryar, and K. M. Flegal. 2015. Prevalence of obesity among adults and youth: United States, 2011–2014. NCHS (National Center for Health Statistics) Data Brief No. 219.
Onega, T., E. J. Duell, X. Shi, D. Wang, E. Demidenko, and D. Goodman. 2008. Geographic access to cancer care in the U.S. Cancer 112(4):909–918.
Orgel, E., J. M. Genkinger, D. Aggarwal, L. Sung, M. Nieder, and E. J. Ladas. 2016. Association of body mass index and survival in pediatric leukemia: A meta-analysis. American Journal of Clinical Nutrition 103(3):808–817.
Paddon-Jones, D., and B. B. Rasmussen. 2009. Dietary protein recommendations and the prevention of sarcopenia: Protein, amino acid metabolism and therapy. Current Opinion in Clinical Nutrition and Metabolic Care 12(1):86–90.
Park, C. L., D. Cho, A. L. Salner, and E. Dornelas. 2016. A randomized controlled trial of two mail-based lifestyle interventions for breast cancer survivors. Supportive Care in Cancer 24(7):3037–3046.
Park, J.-H., J. Lee, M. Oh, H. Park, J. Chae, D.-I. Kim, M. Lee, Y. J. Yoon, C. Lee, N. K. Kim, L. W. Jones, S. I. Kim, S. H. Park, and J. Y. Jeon. 2015. The effect of oncologists’ exercise recommendation on the level of exercise and quality of life in breast and colorectal cancer survivors: Randomized controlled trial. Cancer 121(16):2740–2748.
Paxton, R. J., K. L. Phillips, L. A. Jones, S. Chang, W. C. Taylor, K. S. Courneya, and J. P. Pierce. 2012. Associations among physical activity, body mass index, and health-related quality of life by race/ethnicity in a diverse sample of breast cancer survivors. Cancer 118(16):4024–4031.
Petrin, C., K. Prakash, S. Kahan, and C. Gallagher. 2014. Medicaid fee-for-service treatment of obesity interventions. http://stopobesityalliance.org/wp-content/themes/stopobesityalliance/pdfs/Medicaid%20FFS%20Treatment%20of%20Obesity%20Interventions%202014.pdf (accessed May 3, 2017).
Physical Activity Guidelines Advisory Committee. 2008. 2008 physical activity guidelines for Americans. https://health.gov/paguidelines/pdf/paguide.pdf (accessed June 16, 2017).
Pinto, B. M., and J. T. Ciccolo. 2011. Physical activity motivation and cancer survivorship. In Physical activity and cancer, edited by K. S. Courneya and C. M. Friedenreich. Berlin, Heidelberg: Springer Berlin Heidelberg. Pp. 367–387.
Pool, A. C., J. L. Kraschnewski, L. A. Cover, E. B. Lehman, H. L. Stuckey, K. O. Hwang, K. I. Pollak, and C. N. Sciamanna. 2014. The impact of physician weight discussion on weight loss in US adults. Obesity Research & Clinical Practice 8(2):e131–e139.
Rack, B., U. Andergassen, J. Neugebauer, J. Salmen, P. Hepp, H. Sommer, W. Lichtenegger, K. Friese, M. W. Beckmann, D. Hauner, H. Hauner, and W. Janni. 2010. The German SUCCESS C study—the first European lifestyle study on breast cancer. Breast Care 5(6):395–400.
Riebe, D., B. A. Franklin, P. D. Thompson, C. E. Garber, G. P. Whitfield, M. Magal, and L. S. Pescatello. 2015. Updating ACSM’s recommendations for exercise preparticipation health screening. Medicine and Science in Sports and Exercise 47(11):2473–2479.
Rock, C. L., C. Doyle, W. Demark-Wahnefried, J. Meyerhardt, K. S. Courneya, A. L. Schwartz, E. V. Bandera, K. K. Hamilton, B. Grant, M. McCullough, T. Byers, and T. Gansler. 2012. Nutrition and physical activity guidelines for cancer survivors. CA: A Cancer Journal for Clinicians 62(4):242–274.
Rock, C. L., S. W. Flatt, T. E. Byers, G. A. Colditz, W. Demark-Wahnefried, P. A. Ganz, K. Y. Wolin, A. Elias, H. Krontiras, J. Liu, M. Naughton, B. Pakiz, B. A. Parker, R. L. Sedjo, and H. Wyatt. 2015. Results of the exercise and nutrition to enhance recovery and good health for you (ENERGY) trial: A behavioral weight loss intervention in overweight or obese breast cancer survivors. Journal of Clinical Oncology 33(28):3169–3176.
Rogers, L. Q., S. J. Markwell, S. Verhulst, E. McAuley, and K. S. Courneya. 2009. Rural breast cancer survivors: Exercise preferences and their determinants. Psycho-Oncology 18(4):412–421.
Rose, S. A., P. S. Poynter, J. W. Anderson, S. M. Noar, and J. Conigliaro. 2013. Physician weight loss advice and patient weight loss behavior change: A literature review and meta-analysis of survey data. International Journal of Obesity (2005) 37(1):118–128.
Rossi, A., A. Moadel-Robblee, C. E. Garber, D. Kuo, G. Goldberg, M. Einstein, and N. Nevadunsky. 2015. Physical activity for an ethnically diverse sample of endometrial cancer survivors: A needs assessment and pilot intervention. Journal of Gynecologic Oncology 26(2):141–147.
Roubenoff, R. 2000. Sarcopenia and its implications for the elderly. European Journal of Clinical Nutrition 54(Suppl 3):S40–S47.
Saarto, T., H. Sievänen, P. Kellokumpu-Lehtinen, R. Nikander, L. Vehmanen, R. Huovinen, H. Kautiainen, S. Järvenpää, H. M. Penttinen, M. Utriainen, A. S. Jääskeläinen, A. Elme, J. Ruohola, T. Palva, H. Vertio, M. Rautalahti, M. Fogelholm, R. Luoto, and C. Blomqvist. 2012. Effect of supervised and home exercise training on bone mineral density among breast cancer patients. A 12-month randomised controlled trial. Osteoporosis International 23(5):1601–1612.
Sabatino, S. A., R. J. Coates, R. J. Uhler, L. A. Pollack, L. G. Alley, and L. J. Zauderer. 2007. Provider counseling about health behaviors among cancer survivors in the United States. Journal of Clinical Oncology 25(15):2100–2106.
Sacks, F. M., G. A. Bray, V. J. Carey, S. R. Smith, D. H. Ryan, S. D. Anton, K. McManus, C. M. Champagne, L. M. Bishop, N. Laranjo, M. S. Leboff, J. C. Rood, L. de Jonge, F. L. Greenway, C. M. Loria, E. Obarzanek, and D. A. Williamson 2009. Comparison of weight-loss diets with different compositions of fat, protein, and carbohydrates. New England Journal of Medicine 360(9):859–873.
Schadler, K. L., N. J. Thomas, P. A. Galie, D. H. Bhang, K. C. Roby, P. Addai, J. E. Till, K. Sturgeon, A. Zaslavsky, C. S. Chen, and S. Ryeom. 2016. Tumor vessel normalization after aerobic exercise enhances chemotherapeutic efficacy. Oncotarget 7(40):65429–65440.
Schmid, D., and M. F. Leitzmann. 2015. Cardiorespiratory fitness as predictor of cancer mortality: A systematic review and meta-analysis. Annals of Oncology 26(2):272–278.
Schmitz, K. H., R. L. Ahmed, A. Troxel, A. Cheville, R. Smith, L. Lewis-Grant, C. J. Bryan, C. T. Williams-Smith, and Q. P. Greene 2009. Weight lifting in women with breast-cancer–related lymphedema. New England Journal of Medicine 361(7):664–673.
Schmitz, K. H., R. L. Ahmed, A. B. Troxel, A. Cheville, L. Lewis-Grant, R. Smith, C. J. Bryan, C. T. Williams-Smith, and J. Chittams. 2010a. Weight lifting for women at risk for breast cancer-related lymphedema: A randomized trial. JAMA 304(24):2699–2705.
Schmitz, K. H., K. S. Courneya, C. Matthews, W. Demark-Wahnefried, D. A. Galvao, B. M. Pinto, M. L. Irwin, K. Y. Wolin, R. J. Segal, A. Lucia, C. M. Schneider, V. E. von Gruenigen, and A. L. Schwartz. 2010b. American College of Sports Medicine roundtable on exercise guidelines for cancer survivors. Medicine and Science in Sports and Exercise 42(7):1409–1426.
Sedjo, R. L., S. W. Flatt, T. Byers, G. A. Colditz, W. Demark-Wahnefried, P. A. Ganz, K. Y. Wolin, A. Elias, H. Krontiras, J. Liu, M. Naughton, B. Pakiz, B. A. Parker, H. Wyatt, and C. L. Rock. 2016. Impact of a behavioral weight loss intervention on comorbidities in overweight and obese breast cancer survivors. Supportive Care in Cancer 24(8):3285–3293.
Sedlacek, S. M., M. C. Playdon, P. Wolfe, J. N. McGinley, M. R. Wisthoff, E. A. Daeninck, W. Jiang, Z. Zhu, and H. J. Thompson. 2011. Effect of a low fat versus a low carbohydrate weight loss dietary intervention on biomarkers of long term survival in breast cancer patients (“choice”): Study protocol. BMC Cancer 11:287.
Shariff-Marco, S., J. Von Behren, P. Reynolds, T. H. Keegan, A. Hertz, M. L. Kwan, J. M. Roh, C. Thomsen, C. H. Kroenke, C. Ambrosone, L. H. Kushi, and S. L. Gomez. 2017. Impact of social and built environment factors on body size among breast cancer survivors: The pathways study. Cancer Epidemiology, Biomarkers and Prevention 26(4):505–515.
Shen, J., G. L.-H. Wong, H. L.-Y. Chan, R. S.-M. Chan, H.-Y. Chan, W. C.-W. Chu, B. H.-K. Cheung, D. K.-W. Yeung, L. S. Li, M. M.-M. Sea, J. Woo, and V. W.-S. Wong. 2015. PNPLA3 gene polymorphism and response to lifestyle modification in patients with nonalcoholic fatty liver disease. Journal of Gastroenterology and Hepatology 30(1):139–146.
Sheppard, V. B., J. Hicks, K. Makambi, A. Hurtado-de-Mendoza, W. Demark-Wahnefried, and L. Adams-Campbell. 2016. The feasibility and acceptability of a diet and exercise trial in overweight and obese black breast cancer survivors: The Stepping STONE study. Contemporary Clinical Trials 46:106–113.
Shi, Y.-Q., J. Yang, P. Du, T. Xu, X.-H. Zhuang, J.-Q. Shen, and C.-F. Xu. 2016. Effect of body mass index on overall survival of pancreatic cancer: A meta-analysis. Medicine 95(14):e3305.
Simmons, C. P. L., D. C. McMillan, K. McWilliams, T. A. Sande, K. C. Fearon, S. Tuck, M. T. Fallon, and B. J. Laird. 2017. Prognostic tools in patients with advanced cancer: A systematic review. Journal of Pain and Symptom Management 53(5):962–970.
Slama, K. 2004. Rural culture is a diversity issue. Minnesota Psychologist 9–11.
Speck, R. M., K. S. Courneya, L. C. Masse, S. Duval, and K. H. Schmitz. 2010. An update of controlled physical activity trials in cancer survivors: A systematic review and meta-analysis. Journal of Cancer Survivorship 4(2):87–100.
Spencer, J. C., and S. B. Wheeler. 2016. A systematic review of motivational interviewing interventions in cancer patients and survivors. Patient Education and Counseling 99(7):1099–1105.
Stacey, F. G., E. L. James, K. Chapman, K. S. Courneya, and D. R. Lubans. 2015. A systematic review and meta-analysis of social cognitive theory-based physical activity and/or nutrition behavior change interventions for cancer survivors. Journal of Cancer Survivorship 9(2):305–338.
Steinberg, D. M., D. F. Tate, G. G. Bennett, S. Ennett, C. Samuel-Hodge, and D. S. Ward. 2013. The efficacy of a daily self-weighing weight loss intervention using smart scales and email. Obesity 21(9):1789–1797.
Stolley, M. R., and M. L. Fitzgibbon. 1997. Effects of an obesity prevention program on the eating behavior of African American mothers and daughters. Health Education and Behavior 24(2):152–164.
Stolley, M. R., L. K. Sharp, A. M. Wells, N. Simon, and L. Schiffer. 2006. Health behaviors and breast cancer: Experiences of urban African American women. Health Education and Behavior 33(5):604–624.
Stolley, M. R., G. Fantuzzi, C. Arroyo, P. Sheean, L. Schiffer, R. Campbell, and B. Gerber. 2015. Study design and protocol for moving forward: A weight loss intervention trial for African-American breast cancer survivors. BMC Cancer 15(1):1018.
Sutin, A. R., and A. Terracciano. 2013. Perceived weight discrimination and obesity. PLoS ONE 8(7):e70048.
Swisher, A. K., J. Abraham, D. Bonner, D. Gilleland, G. Hobbs, S. Kurian, M. A. Yanosik, and L. Vona-Davis. 2015. Exercise and dietary advice intervention for survivors of triple-negative breast cancer: Effects on body fat, physical function, quality of life, and adipokine profile. Supportive Care in Cancer 23(10):2995–3003.
Tammemagi, C., D. Nerenz, C. Neslund-Dudas, C. Feldkamp, and D. Nathanson. 2005. Comorbidity and survival disparities among black and white patients with breast cancer. JAMA 294(14):1765–1772.
Tate, D. F., R. R. Wing, and R. A. Winett. 2001. Using Internet technology to deliver a behavioral weight loss program. JAMA 285(9):1172–1177.
Tate, D. F., E. H. Jackvony, and R. R. Wing. 2003. Effects of internet behavioral counseling on weight loss in adults at risk for type 2 diabetes: A randomized trial. JAMA 289(14):1833–1836.
Thomas, R. J., S. A. Kenfield, and A. Jimenez. 2016. Exercise-induced biochemical changes and their potential influence on cancer: A scientific review. British Journal of Sports Medicine 51:640–644.
Thompson, H. J., S. M. Sedlacek, D. Paul, P. Wolfe, J. N. McGinley, M. C. Playdon, E. A. Daeninck, S. N. Bartels, and M. R. Wisthoff. 2012. Effect of dietary patterns differing in carbohydrate and fat content on blood lipid and glucose profiles based on weight-loss success of breast-cancer survivors. Breast Cancer Research 14(1):R1.
Thompson, H. J., S. M. Sedlacek, M. C. Playdon, P. Wolfe, J. N. McGinley, D. Paul, and S. G. Lakoski. 2015. Weight loss interventions for breast cancer survivors: Impact of dietary pattern. PLoS ONE 10(5):e0127366.
Thomson, C. A., T. E. Crane, A. Miller, D. O. Garcia, K. Basen-Engquist, and D. S. Alberts. 2016. A randomized trial of diet and physical activity in women treated for stage II–IV ovarian cancer: Rationale and design of the lifestyle intervention for ovarian cancer enhanced survival (LIVES): An NRG oncology/gynecologic oncology group (GOG-225) study. Contemporary Clinical Trials 49:181–189.
Tompson, T., J. Benz, J. Agiesta, K. H. Brewer, L. Bye, R. Reimer, and D. Junius. 2013. Obesity in the United States: Public perceptions. http://www.apnorc.org/PDFs/Obesity/AP-NORC-Obesity-Research-Highlights.pdf (accessed August 27, 2017).
Travier, N., A. Fonseca-Nunes, C. Javierre, E. Guillamo, L. Arribas, I. Peiró, G. Buckland, F. Moreno, A. Urruticoechea, G. R. Oviedo, A. Roca, L. Hurtós, V. Ortega, M. Muñoz, L. Garrigós, B. Cirauqui, S. del Barco, A. Arcusa, M. A. Seguí, J. M. Borràs, C. A. Gonzalez, and A. Agudo. 2013. Effect of a diet and physical activity intervention on body weight and nutritional patterns in overweight and obese breast cancer survivors. Medical Oncology 31(1):783.
U.S. Census Bureau. 2015. 2010 census urban and rural classification and urban area criteria. https://www.census.gov/geo/reference/ua/urban-rural-2010.html (accessed April 20, 2017).
U.S. Census Bureau. 2016. Measuring America: Our changing landscape. https://www.census.gov/library/visualizations/2016/comm/acs-rural-urban.html?CID=acs-rural-urban (accessed August 27, 2017).
USPSTF (U.S. Preventive Services Task Force). 2012. Obesity in adults: Screening and management https://www.uspreventiveservicestaskforce.org/Page/Document/UpdateSummaryFinal/obesity-in-adults-screening-and-management (accessed August 28, 2017).
USPSTF. 2017. Obesity in children and adolescents: Screening. https://www.uspreventiveservicestaskforce.org/Page/Document/RecommendationStatementFinal/obesity-in-children-and-adolescents-screening1 (accessed August 28, 2017).
van Waart, H., M. M. Stuiver, W. H. van Harten, E. Geleijn, J. M. Kieffer, L. M. Buffart, M. de Maaker-Berkhof, E. Boven, J. Schrama, M. M. Geenen, J. M. Meerum Terwogt, A. van Bochove, V. Lustig, S. M. van den Heiligenberg, C. H. Smorenburg, J. A. J. H. Hellendoorn-van Vreeswijk, G. S. Sonke, and N. K. Aaronson. 2015. Effect of low-intensity physical activity and moderate- to high-intensity physical exercise during adjuvant chemotherapy on physical fitness, fatigue, and chemotherapy completion rates: Results of the paces randomized clinical trial. Journal of Clinical Oncology 33(17):1918–1927.
Vidrine, J., S. Shete, Y. Cao, A. Greisinger, P. Harmonson, B. Sharp, L. Miles, S. M. Zbikowski, and D. W. Wetter. 2013. Ask-advise-connect: A new approach to smoking treatment delivery in health care settings. JAMA Internal Medicine 173(6):458–464.
Villareal, D. T., C. M. Apovian, R. F. Kushner, and S. Klein. 2005. Obesity in older adults: Technical review and position statement of the American Society for Nutrition and NAASO, the Obesity Society. American Journal of Clinical Nutrition 82(5):923–934.
Villarini, A., P. Pasanisi, A. Traina, M. P. Mano, B. Bonanni, S. Panico, C. Scipioni, R. Galasso, A. Paduos, M. Simeoni, E. Bellotti, M. Barbero, G. Macellari, E. Venturelli, M. Raimondi, E. Bruno, G. Gargano, G. Fornaciari, D. Morelli, E. Seregni, V. Krogh, and F. Berrino. 2012. Lifestyle and breast cancer recurrences: The DIANA-5 trial. Tumori 98(1):1–18.
von Gruenigen, V. E., K. S. Courneya, H. E. Gibbons, M. B. Kavanagh, S. E. Waggoner, and E. Lerner. 2008. Feasibility and effectiveness of a lifestyle intervention program in obese endometrial cancer patients: A randomized trial. Gynecologic Oncology 109(1):19–26.
Wadden, T. A. 1993. Treatment of obesity by moderate and severe caloric restriction. Results of clinical research trials. Annals of Internal Medicine 119(7 Pt 2):688–693.
Wadden, T. A., V. L. Webb, C. H. Moran, and B. A. Bailer. 2012. Lifestyle modification for obesity: New developments in diet, physical activity, and behavior therapy. Circulation 125(9):1157–1170.
Warnecke, R. B., A. Oh, N. Breen, S. Gehlert, E. Paskett, K. L. Tucker, N. Lurie, T. Rebbeck, J. Goodwin, J. Flack, S. Srinivasan, J. Kerner, S. Heurtin-Roberts, R. Abeles, F. L. Tyson, G. Patmios, and R. A. Hiatt. 2008. Approaching health disparities from a population perspective: The National Institutes of Health Centers for Population Health and Health Disparities. American Journal of Public Health 98(9):1608–1615.
Warner, E. T., R. E. Glasgow, K. M. Emmons, G. G. Bennett, S. Askew, B. Rosner, and G. A. Colditz. 2013. Recruitment and retention of participants in a pragmatic randomized intervention trial at three community health clinics: Results and lessons learned. BMC Public Health 13(1):192.
Weaver, K. E., R. E. Foraker, C. M. Alfano, J. H. Rowland, N. K. Arora, K. M. Bellizzi, A. S. Hamilton, I. Oakley-Girvan, G. Keel, and N. M. Aziz. 2013a. Cardiovascular risk factors among long-term survivors of breast, prostate, colorectal, and gynecologic cancers: A gap in survivorship care? Journal of Cancer Survivorship 7(2):253–261.
Weaver, K. E., A. M. Geiger, L. Lu, and L. D. Case. 2013b. Rural-urban disparities in health status among US cancer survivors. Cancer 119(5):1050–1057.
Weiss, L., T. Melchardt, S. Habringer, A. Boekstegers, C. Hufnagl, D. Neureiter, G. Hopfinger, R. Greil, and A. Egle. 2014. Increased body mass index is associated with improved overall survival in diffuse large b-cell lymphoma. Annals of Oncology 25(1):171–176.
Whitt-Glover, M. C., S. K. Kumanyika, and D. Haire-Joshu. 2014. Introduction to the special issue on achieving healthy weight in black American communities. Obesity Reviews 15:1–4.
Wilfley, D. E., A. E. Staiano, M. Altman, J. Lindros, A. Lima, S. G. Hassink, W. H. Dietz, S. Cook, and The Improving Access and Systems of Care for Evidence-Based Childhood Obesity Treatment Conference Workgroup. 2017. Improving access and systems of care for evidence-based childhood obesity treatment: Conference key findings and next steps. Obesity 25(1):16–29.
Wilmot, E. G., C. L. Edwardson, F. A. Achana, M. J. Davies, T. Gorely, L. J. Gray, K. Khunti, T. Yates, and S. J. Biddle. 2012. Sedentary time in adults and the association with diabetes, cardiovascular disease and death: Systematic review and meta-analysis. Diabetologia 55(11):2895–2905.
Wilson, C. L., W. Liu, J. J. Yang, G. Kang, R. P. Ojha, G. Neale, D. K. Srivastava, J. G. Gurney, M. M. Hudson, L. L. Robison, and K. K. Ness. 2015. Genetic and clinical factors associated with obesity among adult survivors of childhood cancer: A report from the St. Jude lifetime cohort. Cancer 121(13):2262–2270.
Wilson, S. E., M. R. Andersen, and H. Meischke. 2000. Meeting the needs of rural breast cancer survivors: What still needs to be done? Journal ofWomen’s Health & Gender-Based Medicine 9(6):667–677.
Wolfe, R. R. 2006. The underappreciated role of muscle in health and disease. The American Journal of Clinical Nutrition 84(3):475–482.
Wolff, G., S. J. Davidson, J. K. Wrobel, and M. Toborek. 2015. Exercise maintains blood–brain barrier integrity during early stages of brain metastasis formation. Biochemical and Biophysical Research Communications 463(4):811–817.
Wycherley, T. P., L. J. Moran, P. M. Clifton, M. Noakes, and G. D. Brinkworth. 2012. Effects of energy-restricted high-protein, low-fat compared with standard-protein, low-fat diets: A meta-analysis of randomized controlled trials. American Journal of Clinical Nutrition 96(6):1281–1298.
Yabroff, K. R., W. F. Lawrence, S. Clauser, W. W. Davis, and M. L. Brown. 2004. Burden of illness in cancer survivors: Findings from a population-based national sample. Journal of the National Cancer Institute 96(17):1322–1330.
Zenk, S. N., A. J. Schulz, B. A. Israel, S. A. James, S. Bao, and M. L. Wilson. 2005. Neighborhood racial composition, neighborhood poverty, and the spatial accessibility of supermarkets in metropolitan Detroit. American Journal of Public Health 95(4):660–667.
Zhang, F. F., E. Saltzman, A. Must, and S. K. Parsons. 2012. Do childhood cancer survivors meet the diet and physical activity guidelines? A review of guidelines and literature. International Journal of Child Health and Nutrition 1(1):44–58.
Zhou, Y., B. Cartmel, L. Gottlieb, E. A. Ercolano, F. Li, M. Harrigan, R. McCorkle, J. A. Ligibel, V. E. von Gruenigen, R. Gogoi, P. E. Schwartz, H. A. Risch, and M. L. Irwin. 2017. Randomized trial of exercise on quality of life in women with ovarian cancer: Women’s activity and lifestyle study in connecticut (WALC). Journal of the National Cancer Institute 109(12):djx072.














































































































