18
Harm Reduction
The adverse health consequences of combustible tobacco use have been documented extensively (HHS, 2014), and a major goal of tobacco control efforts is to reduce their health burden (Healthy People 2020, 2017). As described in the previous two chapters, primary strategies to achieve this goal are to prevent youth and young adults from starting smoking and to help current smokers quit expeditiously. Despite these efforts and in spite of successes to reduce initiation and increase cessation, a substantial portion of Americans still become regular smokers. Some of these regular smokers are unwilling to quit and, even among those who want to quit, some have serious difficulty quitting. For these populations who continue to expose themselves and others to harm from combustible tobacco use, it is appropriate to consider strategies that minimize or reduce but not eliminate harm from smoking.
The preceding chapters of this report describe the potential harms of e-cigarette use. Section I presents the evidence on known health effects of individual constituents present in e-cigarette liquids and aerosols. Section II presents the evidence on the health consequences of e-cigarettes on mental and physical health outcomes. By contrast to these earlier chapters, this chapter examines the potential harms of e-cigarettes relative to those of combustible tobacco cigarettes. In so doing, the committee applies a harm reduction approach. Broadly, harm reduction policies attempt to diminish the damaging effects of a particular behavior without aiming to eliminate the behavior itself. Thus, harm reduction policies and
interventions consider a broader set of outcomes than would be considered in an approach that aims solely to reduce prevalence.
Applied to tobacco, a harm reduction approach would aim to reduce but not eliminate tobacco-related health risks at the individual and population levels (IOM, 2001; Zeller et al., 2009). The most effective strategies are those that prevent initiation among non-users, promote cessation among current users, and prevent secondhand and thirdhand exposure among non-smokers. Indeed, cessation of any form of tobacco or nicotine-containing product use is currently considered the only guaranteed way to reduce tobacco-related health risks (IOM, 2001). A tobacco harm reduction approach also considers strategies that would reduce tobacco-related health risks while assuming continued use of tobacco or nicotine-containing products (i.e., those that reduce risks without reducing prevalence of tobacco use or exposure to nicotine). Because tobacco control efforts have focused on promoting abstinence from tobacco products, there is currently little evidence on the effectiveness of interventions to reduce harm from continued tobacco use (IOM, 2001). To date, most tobacco control interventions such as nicotine replacement therapy (NRT) are intended for short-term use and not considered long-term substitutes for smoking. A Cochrane review assessing such interventions concluded that using NRT could help smokers unwilling to quit to reduce their cigarette smoking in the short term and to quit in the longer term (Lindson-Hawley et al., 2016). Although tobacco harm reduction strategies could support smoking reduction, the health benefits of doing so remain unclear. Nevertheless, if e-cigarettes confer lower health risks compared with combustible tobacco cigarettes, encouraging use of this reduced-risk product rather than encouraging complete abstinence only could have public health benefits. As suggested by the Cochrane review’s conclusions, this approach might be especially salient for combustible tobacco cigarette smokers who are unable or unwilling to quit.
Although e-cigarettes have not been theoretically or experimentally proven to be safe, evidence reviewed in the previous chapters of this report suggest that they may be less harmful alternatives to combustible tobacco cigarettes in at least some scenarios. Some users perceive e-cigarettes to be less harmful than combustible tobacco cigarettes, although the proportion of U.S. adults who consider them to be as harmful as combustible tobacco cigarettes has increased over time (Majeed et al., 2016). These devices are also often advertised as less harmful products because they are believed to contain fewer and less toxic inhaled compounds than combustible tobacco cigarettes. If less harmful, their effectiveness at reducing tobacco-related harms will also depend on their reach. Because of their sensory and behavioral similarities to combustible tobacco cigarettes, e-cigarettes may be more appealing than NRT for both smoking cessation and tobacco
harm reduction, and therefore could have greater reach by attracting smokers who are unwilling or unsuccessful at using other approaches. Consequently, e-cigarettes could be an appropriate tool for tobacco harm reduction. Because the efficacy of e-cigarettes to actually reduce harm remains unclear, some researchers have raised concerns about using e-cigarettes for tobacco harm reduction. These concerns include the promotion of continued use of a substance with its own potential health risks (including uncertainty about long-term health risks), the possibility of increasing use of conventional tobacco products through both initiation and relapse, and concerns about questionable terms of engagement with the tobacco industry (McKee and Capewell, 2015).
The extent of risk reduction when transitioning from combustible tobacco cigarette smoking to e-cigarette use will depend on whether an individual switches completely or in part. Those who switch completely would be expected to accrue greater reductions compared with those who continue any smoking. Furthermore, due to the health risks of combustible tobacco smoke from even low levels of use, e-cigarette use among those who continue to smoke (concurrent e-cigarette and combustible tobacco cigarette use, i.e., dual use) may only confer benefits if dual use is merely a transitional state, after which a user transitions completely to e-cigarettes (i.e., quits combustible tobacco cigarettes). E-cigarette use would likely confer little long-term reduction in tobacco-related harm if an individual returns (relapses) to smoking only combustible tobacco cigarettes; it would be more worrisome if e-cigarettes increased likelihood of relapse.
In assessing the harm reduction potential of e-cigarettes among users, the committee therefore considered effects in two populations: smokers who switched to e-cigarette use alone and smokers who use e-cigarettes concurrently with combustible tobacco cigarettes. For smokers who transition completely to e-cigarette use, the committee sought to determine the effects on their health risk profile. The committee uses the term “health risk profile” to capture the various individual health effects and outcomes (e.g., cardiovascular, respiratory, oral) taken together. Overall risk profile is a cumulative product of various individual health risks. In other words, whereas the chapters in Section II examine effects on individual health outcomes from e-cigarette use among naïve users as well as current and former smokers, this chapter focuses on aggregate health effects. For dual users, the committee also sought to determine the reduction in the health risk profile. However, because the greatest reductions in health risk are expected to occur if they eventually quit combustible tobacco cigarette smoking, the committee sought to determine the influence of dual use on subsequent smoking cessation.
The committee also considered effects on non-smokers passively
exposed to e-cigarette aerosols compared with combustible tobacco smoke. Exposure of bystanders to environmental combustible tobacco smoke is associated with an increased risk of respiratory and cardiovascular conditions. Smoking cessation among household members is the only effective way of reducing risks from secondhand and thirdhand exposure to potentially toxic chemicals among passive smokers. Similar to the harm reduction strategy among active smokers, changing smoking practices by switching to e-cigarettes in indoor environments may reduce but not eliminate passive exposure to combustible tobacco smoke constituents, especially when smoking cessation is not possible. The committee therefore sought to determine the effectiveness of substituting combustible tobacco cigarettes with e-cigarettes on changes in health effects and exposure to toxicants among non-smokers passively exposed to e-cigarette aerosols compared with tobacco smoke.
EVIDENCE REVIEW: LEVELS OF EVIDENCE AVAILABLE
The committee drew upon comparisons of evidence from several points along the causal pathway between e-cigarettes or combustible tobacco cigarettes and health outcomes. These include evidence on the exposure to toxicants present in e-cigarette aerosols compared with those in cigarette smoke; nicotine and toxicant exposures in e-cigarette users as an intermediate outcome; and comparisons of effects on any health outcome from e-cigarette use compared with combustible tobacco cigarette smoking. Because multiple studies were available comparing e-cigarettes with combustible tobacco cigarettes for each step in the causal pathway, in this section, the committee discusses evidence by establishing likely exposures from the two products and moves down the causal chain to show how these exposures might shape health effects differently. The committee begins by drawing upon studies examining individual constituents discussed in Chapter 5 and highlights studies that directly compared emissions from e-cigarette devices and combustible tobacco cigarettes. The committee then reviews evidence on nicotine and other toxicant exposures among combustible tobacco cigarette smokers who completely switched to e-cigarettes. The optimal study design would include baseline data collection from subjects at the time of combustible tobacco cigarette use and follow-up evaluation after complete and partial substitution of combustible tobacco cigarettes with e-cigarettes. The committee also reviewed studies with cross-sectional designs, when exposure to nicotine and toxicants was compared among e-cigarette users, combustible tobacco cigarette smokers, and dual users of both products. The committee proceeds by discussing evidence comparing e-cigarette and combustible tobacco cigarette use on health outcomes. The optimal study
design should include baseline collection of clinically relevant health outcomes from subjects at the time of combustible tobacco cigarette use and follow-up evaluation after sufficient time of complete and partial substitution of combustible cigarettes with e-cigarettes. The committee also reviewed cross-sectional studies, when clinically relevant health outcomes were compared among e-cigarette users, combustible tobacco cigarette smokers, and dual users of both products. The committee then turns to supporting evidence on toxicity from animal and in vitro studies.
Epidemiological studies and randomized controlled trials (RCTs) evaluating the long-term health effects of exclusive e-cigarette use as well as reducing the frequency and number of combustible tobacco cigarettes smoked with concurrent combustible tobacco cigarette smoking and e-cigarette use (dual use) would provide the strongest evidence of harm reduction in these populations of users. As in the section on health effects (Section II), in their absence, the committee looks further up the causal pathway to epidemiological evidence on intermediate outcomes, including biomarkers of disease as well as evidence on exposure levels (i.e., studies that look at changes and differences in biomarkers levels). Furthermore, given that the greatest health benefits are likely to occur if dual use represents a temporary transitional stage before combustible tobacco cigarette users transition to e-cigarette use alone, the committee draws upon evidence of e-cigarette studies examining whether dual use increases likelihood of smoking cessation.
For studies on the effects of switching from combustible tobacco cigarettes to e-cigarettes to reduce harm from passive exposures, RCTs, and longitudinal observational studies that follow household members of combustible tobacco cigarette smokers who switch to e-cigarettes completely would provide the strongest evidence. In their absence, the committee draws inferences from studies that examine exposure to toxicants from passive exposure to e-cigarette aerosols compared with combustible tobacco smoke.
EVIDENCE REVIEW: METHODS
The committee identified four studies comparing toxicant levels in e-cigarette aerosols and combustible tobacco cigarette smoke (Goniewicz et al., 2014; Margham et al., 2016; Tayyarah and Long, 2014; Williams et al., 2013). One study compared potentially toxic substances from different e-cigarette models and combustible tobacco cigarettes based on published literature (Goniewicz et al., 2014). Three laboratory studies compared e-cigarette aerosols to combustible tobacco cigarette smoke (Margham et al., 2016; Tayyarah and Long, 2014; Williams et al., 2013).
To assess effects in smokers who transition completely from combus-
tible tobacco cigarettes to e-cigarettes, the committee identified studies that prospectively evaluated changes in health outcomes. Because the committee was interested in the overall health risk from e-cigarette use, the committee included literature on all health outcomes among former smokers who use e-cigarettes. Only studies that included e-cigarette exposure as the treatment condition and combustible tobacco cigarette exposure as a positive control were included. Studies that calculated potential risk levels from known risks of toxicants in e-cigarettes and combustible tobacco cigarettes from other settings (e.g., environmental or occupational exposures) were excluded. The committee identified both longitudinal observational studies and crossover experimental studies (n = 15 total). Because a fair number of studies met these criteria, the committee excluded cross-sectional studies and case reports, which are weaker study designs, from this analysis. The committee also drew on analogous data from in vivo animal and in vitro studies comparing the toxicity of e-cigarettes and combustible tobacco cigarettes. The committee limited studies to those that used the same models and experiment settings to expose cells or animals to both e-cigarette aerosols and combustible tobacco cigarette smoke. As with human studies, each study included in this review assessed harmful effects of e-cigarette aerosols with combustible tobacco smoke as a positive control. The committee identified 27 in vitro studies (Anderson et al., 2016; Anthérieu et al., 2017; Aufderheide and Emura, 2017; Aug et al., 2015; Azzopardi et al., 2016; Banerjee et al., 2017; Barber et al., 2016; Breheny et al., 2017; Carson et al., 2017; Cervellati et al., 2014; Farsalinos et al., 2013; Fields et al., 2017; Haswell et al., 2017; Hom et al., 2016; Leigh et al., 2016; Misra et al., 2014; Moses et al., 2017; Neilson et al., 2015; Putzhammer et al., 2016; Romagna et al., 2013; Rubenstein et al., 2015; Scheffler et al., 2015; Shen et al., 2016; Taylor et al., 2016; Teasdale et al., 2016; Thorne et al., 2016, 2017) and 5 in vivo animal studies (Larcombe et al., 2017; Palpant et al., 2015; Parker and Rayburn, 2017; Ponzoni et al., 2015; Rau et al., 2017).
To assess whether e-cigarettes may reduce harm among dual users, the committee sought to identify studies on the health risk profile in dual users, including exposure to nicotine and toxicants, intermediate outcomes, and distal health outcomes. Only studies that aimed for direct comparison of the outcomes between groups of smokers and e-cigarette users were included in the review. The studies included in the review should have recruited and measured outcomes at the same time, using the same analytical methods and clinical tools. Studies that measured outcomes only in e-cigarette users and compared results with historical data collected in different studies were excluded. The committee identified one study reporting nicotine exposure (Jorenby et al., 2017) and two studies reporting dependence symptoms (Jorenby et al., 2017; Loukas et al., 2016).
The committee identified no studies on the long-term health effects of using e-cigarettes concurrently with combustible tobacco cigarettes. However, because the greatest health benefits are likely to occur if smokers transition to e-cigarette use alone, the committee also identified studies on whether using e-cigarettes while also smoking combustible tobacco cigarettes is associated with greater likelihood of combustible tobacco cigarette smoking cessation. The committee identified two longitudinal observational studies (Etter and Bullen, 2014; Manzoli et al., 2015, 2017), one 26-day laboratory study (Jorenby et al., 2017), and one cross-sectional study (Loukas et al., 2016).
Finally, to assess whether changing smoking practices by switching to e-cigarettes in indoor environments may reduce passive exposure to combustible tobacco smoke constituents among non-smokers, the committee sought to identify studies examining changes in health effects and exposure to toxicants from passive exposure to e-cigarette aerosols compared with combustible tobacco smoke. Only studies that aimed for direct comparison of the environmental exposure levels between secondhand (and thirdhand) emissions from e-cigarettes and combustible tobacco cigarettes were included in the review. The same inclusion criteria applied to studies that evaluated biomarkers of exposure in persons passively exposed to aerosols emitted from e-cigarettes or environmental tobacco smoke. The studies included in the review should have used the same analytical methods to assess secondhand exposure from the two products. Studies that assessed secondhand exposure only in e-cigarette exposure conditions and compared results with historical data collected in different studies or with different methods were excluded from review. The committee identified no studies on intermediate or distal health outcomes related to passive exposures to e-cigarette aerosols compared with combustible tobacco smoke. In their absence, the committee identified four studies comparing passive exposures (Ballbè et al., 2014; Bush and Goniewicz, 2015; Czogała et al., 2014; Flouris et al., 2012).
EVIDENCE REVIEW: RESULTS
Comparison of Toxic Levels in E-Cigarettes and Combustible Tobacco Cigarettes
E-cigarette liquids can expose users to toxicants through solvents (propylene glycol [PG] and glycerol), flavorings, and other additives; heating and aerosolizing e-liquids can generate additional toxicants. Although some of these toxicants are also found in combustible tobacco cigarettes, they are generally at much lower levels in e-cigarettes. Carbon monoxide (CO), for example, is present in combustible tobacco smoke and
delivered to smokers, but is absent from e-cigarette aerosol (Vansickel et al., 2010). The physicochemical composition of an e-cigarette aerosol may be a key driver behind functional impairments. While e-cigarettes seem to be a promising harm reduction tool for smokers of combustible tobacco, evidence suggests that using these products could result in repeated inhalation of respiratory toxicants, irritants, and sensitizers. In other words, whereas evidence reviewed in Chapter 4 on nicotine and Chapter 5 on other constituents and metals in e-cigarettes examined effects of acute exposures from individual constituents, this chapter examines effects of periodic exposures over time to e-cigarette aerosols as a whole. To that end, this section reviews studies that directly compared emissions of harmful and potentially harmful chemicals from e-cigarette devices and from combustible tobacco cigarettes.
Reducing the quantity of tobacco a person uses and reducing the potentially toxic substances in tobacco products (such as in pharmaceutical nicotine and in potential reduced-exposure tobacco products) are strategies for reducing exposure to toxicants from smoking. Most studies evaluating toxicant levels in e-cigarettes have focused on liquid composition, showing that the levels of toxic chemicals present in liquids are far lower than in combustible tobacco cigarettes. Compared with combustible tobacco smoke, e-cigarette aerosol is simpler in composition and primarily composed of homogeneous particles with very low levels of volatile species suspended in air. Additionally, e-cigarette aerosol contains substantially lower levels of toxicants than combustible tobacco cigarette smoke.
Using data available in the literature, Goniewicz and colleagues (2014) compared the content of harmful substances among several models of e-cigarettes and combustible tobacco cigarettes. To compare levels of selected toxicants in e-cigarette aerosol and combustible tobacco cigarette mainstream smoke, the authors assumed that e-cigarette users take an average of 15 puffs during one session of product use, which would correspond to smoking one combustible tobacco cigarette. As shown in Table 18-1, levels of selected toxic compounds found in combustible tobacco cigarette smoke were from 9- to 450-fold higher than levels in e-cigarette aerosol. The results of the study support the proposition that the aerosol emitted from an e-cigarette is less injurious than the smoke from combustible tobacco cigarettes. Thus, one would expect that if a person switched from combustible tobacco cigarettes to e-cigarettes, exposure to toxic chemicals and related adverse health effects would be reduced. This hypothesis has been confirmed in several studies involving people using e-cigarette devices.
Margham and colleagues (2016) examined 150 chemicals emitted from an e-cigarette (Vype ePen), a reference combustible tobacco cigarette (Ky3R4F), and laboratory air (method blanks). Of the chemicals
| Toxic Compound | Combustible Tobacco Cigarette (µg in mainstream smoke) | E-Cigarette (µg per 15 puffs) | Average Ratio (combustible tobacco cigarette versus e-cigarette) |
|---|---|---|---|
| Formaldehyde | 1.6–52 | 0.20–5.61 | 9 |
| Acetaldehyde | 52–140 | 0.11–1.36 | 450 |
| Acrolein | 2.4–62 | 0.07–4.19 | 15 |
| Toluene | 8.3–70 | 0.02–0.63 | 120 |
| NNN | 0.0005–0.19 | 0.00008–0.00043 | 380 |
| NNK | 0.012–0.11 | 0.00011–0.00283 | 40 |
NOTE: NNK = 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone; NNN = N′-nitrosonornicotine.
SOURCE: Goniewicz et al., 2014.
examined, only 25 were detected; 104 chemicals were not detected and 21 were present due to laboratory background. Of those detected in the aerosol, the e-cigarette generated 16 (either in whole or in part), and 9 were present at levels too low to be quantified. The chemicals detected included e-liquid constituents (nicotine, PG, and glycerol), and eight thermal decomposition products of PG or glycerol. By contrast, approximately 100 chemicals were detected in mainstream combustible tobacco cigarette smoke. The authors concluded that, on a per-puff basis and depending on the regulatory list considered and the puffing method used, the toxicants emitted were from 82 to more than 99 percent lower in the e-cigarette aerosol than from the combustible tobacco cigarette smoke.
A study by Tayyarah and Long (2014) found that aerosol nicotine for the e-cigarette samples (blu and SKYCIG brands) was 85 percent lower than the nicotine yield for the combustible tobacco cigarettes. The authors also found that the mainstream combustible tobacco cigarette smoke contained approximately 1,500 times more harmful and potentially harmful constituents (HPHCs) compared with e-cigarette aerosol or room air. The deliveries of HPHCs tested for these e-cigarette products were more similar to the air blanks rather than to deliveries from combustible tobacco cigarettes; the HPHCs detected in combustible tobacco cigarette smoke were either not detected or detected at trace levels in the e-cigarette aerosols.
Williams and colleagues (2013) compared the concentrations of various metals in the aerosol generated from an e-cigarette cartomizer from a leading manufacturer (10 puffs) with the concentrations in the main-
stream smoke from one combustible tobacco cigarette. For 11 metals for which data were available for combustible tobacco cigarette smoke, the concentration of elements in the e-cigarette aerosol was higher for four elements (aluminum, iron, nickel, and sodium), within the combustible tobacco smoke range for five elements (chromium, copper, lead, magnesium, and manganese), and lower for two elements (potassium and zinc).
Synthesis
Although a limited number of laboratory studies compared emissions of harmful and potentially harmful chemicals from e-cigarette devices and from combustible tobacco cigarettes, laboratory studies reviewed above and in Chapter 5 overall found that an aerosol emitted from e-cigarettes is substantially less complex than tobacco smoke. Although several potentially toxic substances have been identified in e-cigarette aerosol, the amounts emitted from e-cigarettes under typical conditions of use are significantly lower compared with levels measured in combustible tobacco smoke. Thus, as the committee concluded in Chapter 5, there is substantial evidence that except for nicotine, under typical conditions of use, exposure to potentially toxic substances from e-cigarettes is significantly lower compared with combustible tobacco cigarettes (see Conclusion 5-3).
Harm Reduction in Smokers Who Switched from Combustible Tobacco Cigarettes to E-Cigarettes
Comparison of Nicotine and Toxicant Exposure
Whether e-cigarettes will substitute for combustible tobacco cigarettes depends, in part, on if they yield effects approximating the combustible tobacco cigarette effects thought to cause dependent cigarette use, including delivery of similar levels of nicotine. Potential harm reduction will be at least partially determined by the magnitude of reduction in exposure to harmful and potentially harmful toxicants as compared with exposure from tobacco smoking. Below, the committee reviews cross-sectional and longitudinal studies that compared exposure to nicotine and toxicants in tobacco smokers who substituted e-cigarettes for their combustible tobacco cigarettes.
Adriaens and colleagues (2014) conducted an RCT with 8 months follow-up on the effects on smoking behavior of providing e-cigarettes (Joyetech eGo-C and Kanger T2-CC brands) to combustible tobacco cigarette smokers. The researchers recruited participants who were willing to try a less harmful alternative, but did not intend to quit. Participants (n = 48) were randomized into two e-cigarette groups and one control
group. Over the first 2 months of the study during which laboratory studies were conducted, participants in the e-cigarette groups were provided with e-cigarettes to use, while participants in the control group continued smoking combustible tobacco cigarettes. After 8 weeks, the investigators also gave the control group e-cigarettes (same brands and types as those in the two e-cigarette groups). During the laboratory studies in the first 2-month period of the study, investigators examined craving and withdrawal symptoms in the lab. For 8 months after providing participants with e-cigarettes, the investigators assessed effects on smoking behavior, as well as benefits of and complaints about using an e-cigarette. At 3 months after the last lab session (5 months from the start of the study when the e-cigarette groups were given e-cigarettes; 3 months from when the control group was given e-cigarettes), 38 percent of participants across all groups (37 percent in the e-cigarette groups combined and 39 percent in the control group) reported complete abstinence from smoking, 6 percent showed a reduction in smoking of more than 80 percent, and another 10 percent showed a reduction of more than 50 percent. At the same time, 46 percent of participants were smoking 50 percent or more of their number of combustible tobacco cigarettes at baseline (including participants with missing data). Of note, in the control group, there were no significant changes in smoking behavior over the first 2 months of the study (during which they monitored behavior but were not provided e-cigarettes). At follow-up 6 months after the last laboratory session (8 months after the start of the study), 21 percent of participants across all groups (19 percent of the e-cigarette groups combined and 25 percent of the control group) reported complete abstinence from smoking, 15 percent showed a reduction in combustible tobacco cigarette use of more than 80 percent, and another 8 percent showed a reduction of more than 50 percent; 56 percent reported smoking 50 percent or more of their number of combustible tobacco cigarettes at baseline (including missing data). Reduction in smoking was consistent with decreased levels of expired CO. There were no significant differences in saliva cotinine levels among groups either during the laboratory sessions (during which the control group was smoking combustible tobacco cigarettes exclusively) or at follow-up. Together, these results suggest that e-cigarettes may help promote smoking reduction or cessation even among smokers who do not intend to quit, and also that e-cigarette users (both exclusive and dual) can self-titrate nicotine intake from e-cigarettes with some practice.
Cravo and colleagues (2016) conducted a randomized, parallel group clinical study to evaluate the safety profile of an e-cigarette product (2.0 percent nicotine, developed by Fontem Ventures B.V., Amsterdam, the Netherlands) in 420 smokers of combustible tobacco cigarettes switching to use the e-cigarette product for 12 weeks. Urine nicotine equivalents
decreased by up to 33.8 percent in e-cigarette product subjects and three biomarkers of exposure to toxicants known to be present in combustible tobacco cigarette smoke (benzene, acrolein, and 4-[methylnitrosamino]-1-[3-pyridyl]-1-butanone [NNK]) also decreased. The decrease in nicotine equivalents coincided with an increase in nicotine withdrawal symptoms, measured by a questionnaire, which subsided after 2 weeks.
A 5-day study randomized 105 clinically confirmed smokers into one of seven groups: three exclusive e-cigarette use groups (blu e-cigarettes in rechargeable tobacco, rechargeable cherry, and disposable cherry), three dual-use groups (one of the three blu e-cigarettes and continued combustible tobacco cigarette smoking), and one group that abstained from all nicotine and tobacco products (cessation) (D’Ruiz et al., 2016; O’Connell et al., 2016). The investigators assessed blood, urine, and exhaled breath biomarkers of exposure of toxicants believed to contribute to smoking-related disease at baseline and 5-day follow-up. They found that subjects switching to e-cigarettes (either partially or completely) had significantly lower levels (29–95 percent) of urinary biomarkers of exposure. All groups experienced significant decreases in exhaled CO (27–89 percent), and nicotine equivalents decreased by 25–40 percent. Dual users who reported that they substituted half of their daily combustible tobacco cigarette consumption with e-cigarettes experienced 7–38 percent reductions, but had increases (1–20 percent) in nicotine equivalents. Reductions were broadly proportional to the reduced numbers of combustible tobacco cigarettes smoked. At follow-up, blood nicotine biomarker levels were lower in the cessation (75–96 percent) and e-cigarette use groups (11–83 percent), but there were no significant reductions among dual users. All subjects experienced significant decreases in exhaled CO; reductions of 88–89 percent were observed among the cessation and exclusive e-cigarette use groups and from 27 percent to 32 percent among dual users. Exhaled nitric oxide (NO) increased (46–63 percent) in the cessation and e-cigarette groups, but there were only minimal changes among dual users (O’Connell et al., 2016).
Using a longitudinal within-subjects observational design, Goniewicz and colleagues (2017), evaluated the effects of switching from combustible tobacco cigarettes to e-cigarettes on nicotine delivery and exposure to selected carcinogens and toxicants. The authors measured metabolites of nicotine and major carcinogens and toxicants present in combustible tobacco smoke and combustible tobacco smoke exposure biomarkers (including NNK, 1,3-butadiene, crotonaldehyde, acrolein, benzene, acrylamide, acrylonitrile, ethylene oxide, propylene oxide, naphthalene, fluorene, phenanthrene, and pyrene) in the urine samples of 20 smokers collected before and after switching to pen-style M201 e-cigarettes for 2 weeks. One week after participants switched from combustible
tobacco cigarettes to e-cigarettes, levels of total nicotine and some polycyclic aromatic hydrocarbon metabolites did not change, but all other biomarkers significantly decreased (p < 0.05). The greatest reductions were seen in metabolites of 1,3-butadiene, benzene, and acrylonitrile. Total 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol, known as NNAL (a metabolite of a tobacco-specific nitrosamine [TSNA], NNK), declined by 57 percent after 1 week and 64 percent after 2 weeks. Levels of 3-hydroxy-fluorene declined by 46 percent after 1 week and 34 percent after 2 weeks. Results from this study are shown in Figure 18-1.
Hecht and colleagues (2015) analyzed urine samples from 28 e-cigarette users who had not smoked combustible tobacco cigarettes for at least 2 months for toxicant and carcinogen metabolites, including 1-hydroxy-pyrene (1-HOP), NNAL and its glucuronides (total NNAL), 3-hydroxy-propylmercapturic acid (3-HPMA, the primary metabolite of acrolein), 2-hydroxypropylmercapturic acid (2-HPMA), 3-hydroxy-1-methylpropyl-mercapturic acid (HMPMA), S-phenylmercapturic acid (SPMA), nicotine,
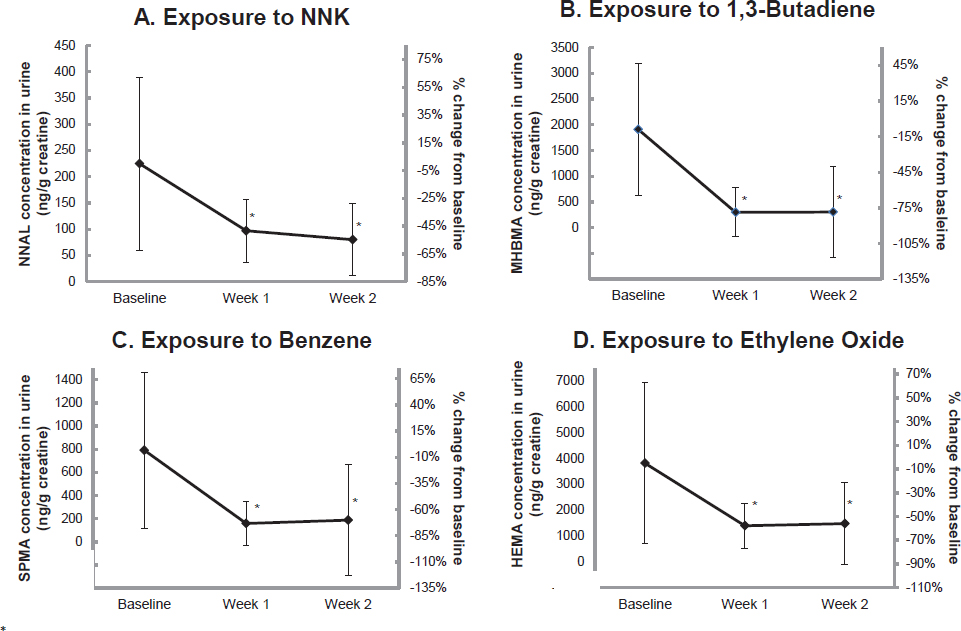
NOTES: *Denotes statistically significant differences from baseline according to repeated-measure analysis of variance (p < 0.05). HEMA= 2-hydroxyethylmercapturic acid; MHBMA = 2-hydroxy-3-buten-1-yl-mercapturic acid; NNAL = 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol; NNK = 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone; SPMA = S-phenylmercapturic acid.
SOURCE: Goniewicz et al., 2017.
and cotinine. The authors compared these samples to previous analyses of combustible tobacco cigarette smokers’ urine using similar, validated methods. These comparisons showed that samples from e-cigarette users had significantly lower levels of 1-HOP, total NNAL, 3-HPMA, 2-HPMA, HMPMA, and SPMA. However, levels of nicotine and cotinine in e-cigarette users were significantly lower compared with combustible tobacco cigarette smokers in one study but not another.
McRobbie and colleagues (2015) measured exposure to CO, urinary cotinine (as a nicotine metabolite), and urinary 3-HPMA before and after 4 weeks of e-cigarette use (Green Smoke cigalike device, labeled 2.4 percent nicotine) in 40 smokers. Four weeks after quitting, 33 participants reported using e-cigarettes, 48 percent reported abstaining from combustible tobacco cigarettes and exclusively using e-cigarettes in the week prior, and 52 percent reported using both e-cigarettes and combustible tobacco cigarettes (dual use). CO levels were significantly reduced among e-cigarette–only users (80 percent decrease) and dual users (52 percent decrease). Cotinine levels also declined, but to a lesser extent (17 percent decrease among exclusive e-cigarette users and 44 percent decrease among dual users). Mean 3-HPMA levels decreased 79 percent among exclusive e-cigarette users and by 60 percent in dual users.
Pulvers and colleagues (2016) enrolled 40 combustible tobacco cigarette smokers (with 1 year or more of smoking) interested in switching to e-cigarettes in a 4-week observational study. The researchers provided participants with an e-Go C non-variable battery e-cigarette and refillable atomizers in a choice of eight flavors with 12 mg or 24 mg nicotine and measured urinary cotinine, NNAL, and eight volatile organic compounds (VOCs) that are known toxic constituents of combustible tobacco cigarettes at baseline and week 4. All participants with follow-up data (92.5 percent) reported using the study e-cigarette. At week 2, 40 percent reported abstaining from combustible tobacco cigarettes. At week 4, 15 percent had remained abstinent. At 4 weeks, there were no significant changes in nicotine intake (p = 0.90), but CO (p < 0.001), NNAL (p < 0.01), and metabolites of benzene (p < 0.01) and acrylonitrile (p = 0.001) decreased significantly. Smokers switching exclusively to e-cigarettes for at least half of the study period demonstrated significant reductions in metabolites of ethylene oxide (p = 0.03) and acrylamide (p < 0.01).
Shahab and colleagues (2017) compared biomarkers of exposure to nicotine and potentially toxic and carcinogenic chemicals among exclusive combustible tobacco cigarette smokers, former smokers with long-term exclusive e-cigarette use, former smokers with long-term exclusive NRT use, long-term dual users of both combustible tobacco cigarettes and e-cigarettes, and long-term users of both combustible tobacco cigarettes and NRT (n = 36 or 37 per group; total n = 181). Participants provided
urine and saliva samples, which were analyzed for biomarkers of nicotine, TSNAs, and VOCs. There were no differences in salivary or urinary biomarkers of nicotine intake after controlling for confounders. Levels of metabolites of TSNAs (including NNAL) and VOCs (including metabolites of the toxicants acrolein, acrylamide, acrylonitrile, 1,3-butadiene, and ethylene oxide) were significantly lower among exclusive e-cigarette and exclusive NRT users than for exclusive combustible tobacco cigarette smokers, dual combustible tobacco cigarette–e-cigarette users, and dual combustible tobacco cigarette–NRT users. The e-cigarette–only users had significantly lower NNAL levels than all other groups. Levels of TSNA and VOC metabolites were similar among combustible tobacco cigarette–only, dual combustible tobacco cigarette–NRT users, and dual combustible tobacco cigarette–e-cigarette users. Results from this study are shown in Figure 18-2.
van Staden and colleagues (2013) used a single group within-subject design to examine effects of switching from combustible tobacco cigarettes to e-cigarettes (Twisp brand) for 2 weeks among 13 participants (median age = 38 years, range = 23–46, median cigarettes per day = 20, range = 12–30). At baseline (before using e-cigarettes) and 2-week follow-up, the researchers measured arterial carboxyhaemoglobin (COHb), venous COHb, and venous cotinine levels and asked the participants to complete a questionnaire reporting perceptions of their health and
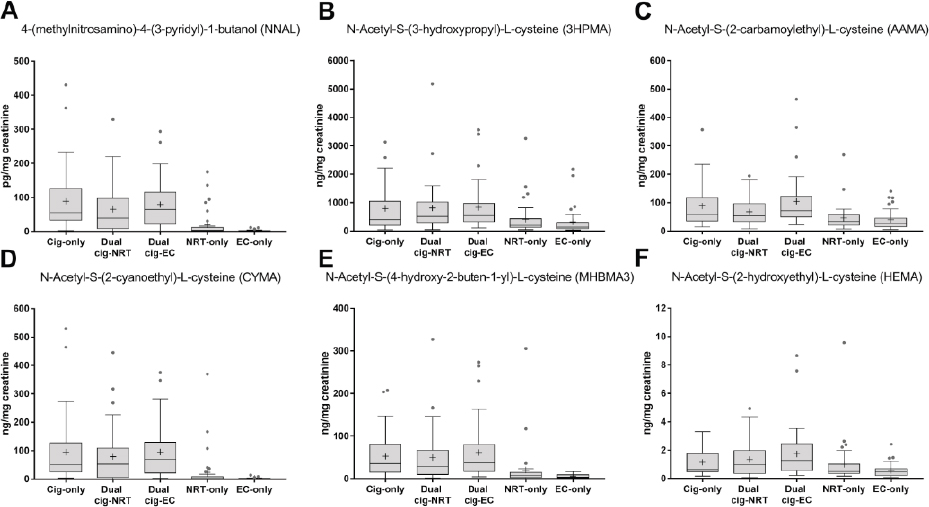
NOTE: EC = e-cigarette; NRT = nicotine replacement therapy.
SOURCE: Shahab et al., 2017.
lifestyle. After 2 weeks of e-cigarette use, COHb levels were significantly lower (percent mean ± SD for both arterial COHb and venous COHb after 2 weeks of e-cigarette use compared with baseline [arterial: 4.66 ± 1.99 at baseline versus 2.46 ± 1.35 at 2-week follow-up, p = 0.014; venous: 4.37 ± 2.1 at baseline versus 2.50 ± 1.23 at follow-up, p = 0.018]). The authors also found a significant decrease in cotinine levels (p = 0.001) and a significant increase in oxygen saturation (p = 0.002). Most participants reported perceiving improvements in their health and lifestyle measures.
A cross-sectional study by Martin and colleagues (2016) compared immune gene expression profiles in superficial nasal scrape biopsies collected from non-smokers (n = 13), combustible tobacco cigarette smokers (n = 14), and e-cigarette users (n = 12), and analyzed them using the nCounter human immunology V2 expression panel. The researchers determined smoking status by taking smoking histories and from a 3- to 4-week smoking diary, which was biochemically confirmed with serum cotinine and urinary NNAL. Results showed that all genes with decreased expression in combustible tobacco cigarette smokers (n = 53) were also decreased in e-cigarette smokers. Moreover, compared with combustible tobacco cigarette smokers, e-cigarette users showed more gene expression changes and stronger levels of suppression in a gene-by-gene comparison. In genes common with those changed in smokers, e-cigarette users showed greater suppression, especially for expression of transcription factors. For example, EGR1 was functionally associated with decreased expression of 18 target genes in e-cigarette users compared with only 5 target genes in combustible tobacco cigarette smokers.
Synthesis Several cross-sectional and longitudinal studies compared exposure to nicotine and toxicants in smokers who substituted e-cigarettes for their combustible tobacco cigarettes. All studies showed that smokers who substituted their tobacco cigarettes with e-cigarettes had significantly reduced levels of biomarkers of exposure to potentially toxic chemicals. Nicotine intake from e-cigarette devices among ex-smokers who were experienced e-cigarette users was comparable to that from tobacco cigarettes. Except for nicotine, exposure to potentially toxic substances from using e-cigarettes was significantly lower compared with smoking combustible tobacco cigarettes.
Conclusion 18-1. There is conclusive evidence that completely substituting e-cigarettes for combustible tobacco cigarettes reduces users’ exposure to numerous toxicants and carcinogens present in combustible tobacco cigarettes.
Health Risk Profile
In the same 8-month follow-up study by Adriaens and colleagues (2014) described in the harm reduction section above of smokers not intending to quit (n = 48), who were randomized to three e-cigarette–only groups, three dual-use groups, and a control group (cessation from nicotine and tobacco for 8 weeks, followed by switching to the same type of e-cigarettes as the e-cigarette groups), the authors also assessed benefits of and complaints about using e-cigarettes and smoking combustible tobacco cigarettes. Participants reported these benefits and complaints in online diaries. Adverse events included in the reported complaints included dry mouth/throat, mouth/throat irritation, dizziness, headache, nausea, increased heart rate/palpitations, and increased weight. Participants also reported concerns about health risks. Benefits included pleasant sensation when inhaling, improved breathing, pleasant taste when inhaling, less coughing or sore throat, improved health and fitness, improved taste and smell, less unpleasant smells, and improved sleep. E-cigarette users were also asked about the pleasure of e-cigarette use, decreased desire for combustible tobacco cigarettes, fresher breath, and a device that can be used in more places. At the beginning of the study, the control group reported more complaints about their combustible tobacco cigarettes than the e-cigarette groups did about their e-cigarettes, but this difference disappeared at follow-up. This change may be attributable to the fact that the control group switched to the e-cigarettes after 8 weeks. Participants in the e-cigarette groups also reported more benefits experienced from the e-cigarette than the control group experienced from combustible tobacco cigarettes. Results also showed an increase in experienced benefits among e-cigarette users over the course of the study, which may reflect a learning effect.
In a single-blind, crossover study, Carnevale and colleagues (2016) compared the effects of e-cigarettes and combustible tobacco cigarettes on oxidative stress and endothelial cell function in healthy adult smokers (n = 20) and non-smokers (n = 20). Participants were matched for age and sex. First, all subjects smoked combustible tobacco cigarettes. After 1 week, the subjects switched to smoking an e-cigarette with the same labeled nicotine content as contained in the combustible tobacco cigarettes. Immediately before and after smoking, blood samples were drawn and markers of oxidative stress, nitric oxide bioavailability, and vitamin E levels were measured. Flow-mediated dilation (FMD), a marker of endothelial function in humans, was also measured. Levels of soluble NOX2-derived peptide and 8-iso-prostaglandin F2α and a significant decrease in nitric oxide bioavailability, vitamin E levels, and FMD increased significantly after both e-cigarette use and combustible tobacco smoking. Generalized estimating equation analysis confirmed that smoking affected all markers of oxida-
tive stress and FMD and showed that the biological effects of e-cigarettes compared with combustible tobacco cigarettes on vitamin E levels and FMD were not statistically different. However, e-cigarettes showed a lesser impact than combustible tobacco cigarettes on levels of soluble NOX2derived peptide, 8-iso-prostaglandin F2α, and nitric oxide bioavailability.
Cibella and colleagues (2016) conducted a 1-year randomized controlled trial to evaluate changes in spirometric indexes and respiratory symptoms. Participants (n = 300) were smokers invited to substitute their combustible tobacco cigarettes with e-cigarettes wholly (to quit) or in part (to reduce their smoking), and were given e-cigarettes with 2.4 percent, 1.8 percent, or 0 percent nicotine. Participants were classified as quitters (those who completely switched from combustible tobacco cigarettes to e-cigarettes), reducers (those who substituted some of their smoking with e-cigarettes and reduced their combustible tobacco cigarette consumption), and failures (those with no changes in smoking). The authors found no significant differences in spirometric indexes (forced expiratory volume [FEV1], forced vital capacity [FVC], and FEV1/FVC ratio) among the groups, except for forced expiratory flow 25–75 percent, which significantly increased over the time among those who quit smoking (85.7 ± 15.6 percent at baseline to 100.8 ± 14.6 percent at follow-up, p = 0.034). Among all participants, 43.1 percent reported having cough or phlegm and 34.8 percent reported shortness of breath at baseline. The prevalence of these symptoms decreased substantially over follow-up visits. No participants who reduced their smoking by substituting some of their smoking with e-cigarette use reported shortness of breath at any follow-up visit (week 12, 24, and 52), and participants who switched completely reported neither symptom at any follow-up.
Cravo and colleagues (2016) conducted a randomized, parallel group clinical study to evaluate the safety profile of an e-cigarette product (2.0 percent nicotine, developed by Fontem Ventures B.V., Amsterdam, the Netherlands) in 420 smokers of combustible tobacco cigarettes switching to use the e-cigarette product for 12 weeks. During the study, no clinically significant product-related findings were observed in terms of vital signs, electrocardiogram, lung function tests, and standard clinical laboratory parameters. Adverse events (AEs) reported by e-cigarette product subjects were more frequent during the first week after switching to the e-cigarette product. The frequency of AEs decreased thereafter, and out of a total of 1,515 reported AEs, 495 were judged as being related to nicotine withdrawal symptoms. The most frequently stated AEs were headache, sore throat, desire to smoke, and cough reported by 47.4 percent, 27.8 percent, 27.5 percent, and 17.0 percent of subjects, respectively.
D’Ruiz and colleagues (2015) conducted a randomized, partially single-blinded, crossover study on the nicotine pharmacokinetics, effects
on smoking urge, tolerability of, and AEs from using e-cigarettes compared with combustible tobacco cigarettes. Thirty-eight adult smokers (averaging 10 or more cigarettes per day, biochemically confirmed with urine cotinine and CO) were given two of the study e-cigarette products (non-menthol and menthol flavors with 2.4 percent nicotine, glycerol-based e-liquid) to practice using the devices for a 7-day at-home period. Of these 38 participants, 24 were randomly selected for enrollment in the 11-day trial, during which they were randomized to one of six product usage sequences. Products used were a combustible tobacco cigarette or a commercially available, rechargeable e-cigarette (3.7 nominal volts, 3-Ω resistance) containing one of five e-liquids:
- Commercial product in classic tobacco flavor with 1.6 percent nicotine in a 50 percent glycerol/20 percent PG base;
- Commercial product in classic tobacco flavor with 1.6 percent nicotine in a 75 percent glycerol base;
- Non-commercial product in classic tobacco flavor with 2.4 percent nicotine in a 75 percent glycerol base;
- Non-commercial product in classic tobacco flavor with 2.4 percent nicotine in a 50 percent glycerol/20 percent PG base; and
- Non-commercial product in menthol flavor with 75 percent glycerol base.
On product use days, subjects participated in 90-minute exposure sessions (30 minutes controlled followed by 1 hour ad lib use). None of the participants reported serious AEs or discontinued the study owing to AEs. Eighteen of the 38 total subjects provided with a study product reported minor AEs such as cough (20 reports by 11 subjects, more commonly among e-cigarette users than smokers), throat irritation (8 reports by 5 subjects), headache (6 reports by 5 subjects), and dizziness (5 reports by 4 subjects). All of the AEs resolved without sequelae.
The authors also investigated the acute effects of e-cigarettes on blood pressure and heart rate compared with the effects of combustible tobacco cigarette smoking (Yan and D’Ruiz, 2015). On product use days, they measured participants’ (n = 23) systolic and diastolic blood pressure approximately 30 minutes before the controlled session and approximately 20 minutes after the end of the ad lib session. The heart rate and systolic and diastolic blood pressure were significantly elevated after use of Marlboro cigarettes, but the elevation was less after use of most of the e-cigarettes.
In another study, D’Ruiz and colleagues (2017) measured cardiovascular physiology (systolic and diastolic blood pressure and heart rate), pulmonary function (FVC, FEV1, and exhaled CO and NO), and AEs in 105 clinically confined subjects who were randomized into groups that
either completely or partially switched from combustible tobacco cigarettes to e-cigarettes (blu) or completely discontinued using tobacco and nicotine products altogether. Use of the e-cigarettes for 5 days under the various study conditions did not lead to higher blood pressure or heart rate values, negative respiratory health outcomes, or serious adverse health events. Reductions in blood pressure and heart rate vital signs were observed in most of the participants who either ceased tobacco and nicotine product use altogether or switched completely to using e-cigarettes. Pulmonary function tests showed small but non-statistically significant improvements in FVC and FEV1 measurements in most use groups. Statistically significant (p < 0.05) benefits associated with smoking reduction were also noted in exhaled CO and NO levels. All study products were well tolerated.
Flouris and colleagues (2012) evaluated the acute effect of e-cigarette use and combustible tobacco cigarette smoking on complete blood count (CBC) markers in smokers (n = 15, eight men, averaging 15 or more cigarettes per day). Subjects participated in three 30-minute experimental sessions (e-cigarette use, combustible tobacco cigarette smoking, and control) in random order, with a minimum wash-out period of 7 days. For the e-cigarette session, the authors provided an e-cigarette device (Giant, Nobacco, G.P., Greece) containing tobacco-flavored e-liquid with 11 mg/ml nominal nicotine concentration in a base of greater than 60 percent PG and instructed participants to take a number of puffs (calculated for each participant based on his or her combustible tobacco cigarette consumption). Participants smoked two cigarettes of their own brand for the smoking session and smoked a sham (unlit) cigarette of their own brand for the control session. Blood samples were collected before, immediately after, and 1 hour after each experimental session. The authors found that CBC indexes remained unchanged during the control session and the e-cigarette exposure sessions (p > 0.05), whereas combustible tobacco cigarette smoke exposure increased white blood cell, lymphocyte, and granulocyte counts for at least 1 hour (p < 0.05).
From the same experimental setup, the researchers (Poulianiti et al., 2016) also examined the acute effects of e-cigarette use and combustible tobacco cigarette smoking on selected redox status markers. The researchers assessed total antioxidant capacity (TAC), catalase activity (CAT), and reduced glutathione (GSH) in the blood samples collected prior to, immediately after, and 1 hour after exposure. Results showed that TAC, CAT, and GSH remained similar to baseline levels immediately after and 1 hour after exposure (p > 0.05) to e-cigarette, combustible tobacco cigarette, and control conditions.
In a number of studies, Polosa and colleagues (2014a,b, 2016b,c) retrospectively assessed changes in respiratory and asthma symptoms (changes
in spirometry data, airway hyperresponsiveness [AHR], asthma exacerbations, and subjective asthma control) from baseline (prior to switching) over 1-year follow-up (with visits at 6 and 12 months). Participants were 18 asthmatic smokers (10 single users, 8 dual users) who switched to e-cigarettes (Polosa et al., 2014a). Overall spirometry data, asthma control, and AHR improved significantly among both exclusive combustible tobacco cigarette smokers and dual users at baseline (Polosa et al., 2014a). Figure 18-3 shows the results for FEV1 at the four assessment points. Participants reported fewer asthma exacerbations, but the reduction did not reach statistical significance. They also reported no severe AEs.
Polosa and colleagues (2016a) reviewed medical records to evaluate changes in resting blood pressure and blood pressure control among hypertensive current and former smokers who reported using e-cigarettes daily at two consecutive visits (n = 43) compared with a control group of combustible tobacco cigarette smokers (n = 46). The authors observed a marked reduction in combustible tobacco cigarette smoking in those who had switched to e-cigarettes (both completely and partly), whereas no such change was observed in the control group. Among e-cigarette users, smoking reduction was associated with significant reductions in median
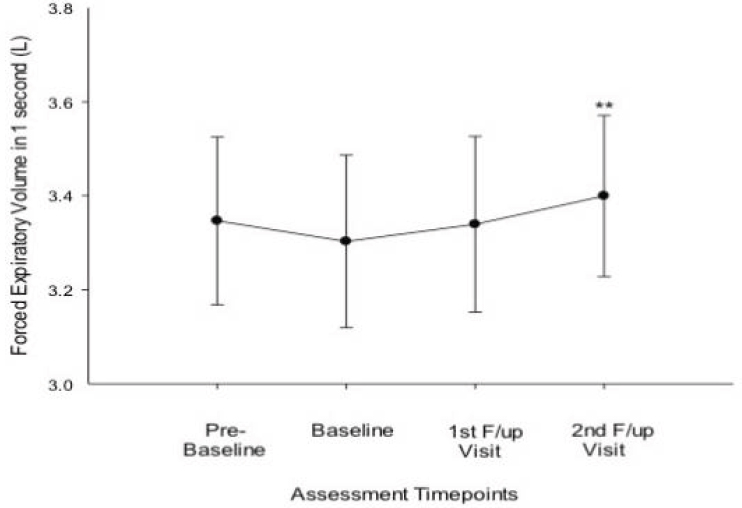
NOTES: All data expressed as mean, and error bars are standard error of the mean. ** = p ≤ 0.01.
SOURCE: Polosa et al., 2014a.
(25th, 75th centile) systolic blood pressure (140 [134.5, 144] to 130 [123.5, 138.5] mmHg; p < 0.001) and diastolic blood pressure (86 [78, 90] to 80 [74.5, 90] mmHg; p = 0.006) at 12-month follow-up compared with baseline. No significant changes were observed in the control group. Figure 18-4 illustrates the changes in diastolic blood pressure among e-cigarette users and controls (combustible tobacco cigarette smokers).
In another study, Polosa and colleagues (2016c) retrospectively reviewed medical records of current and former smokers with chronic obstructive pulmonary disease (COPD) who reported using e-cigarettes daily on three visits (baseline, 12-month follow-up, and 24-month follow-up) to examine changes in respiratory outcomes. Regularly smoking COPD patients (n = 24) were included as a reference group. Among exclusive e-cigarette users, the authors observed a significant reduction in COPD exacerbations, with their mean (± SD) severity score decreasing from 2.3 (±1) at baseline to 1.8 (±1; p = 0.002) at 12-month follow-up and 1.4 (± 0.9; p < 0.001) at 24-month follow-up (see Figure 18-5). The authors
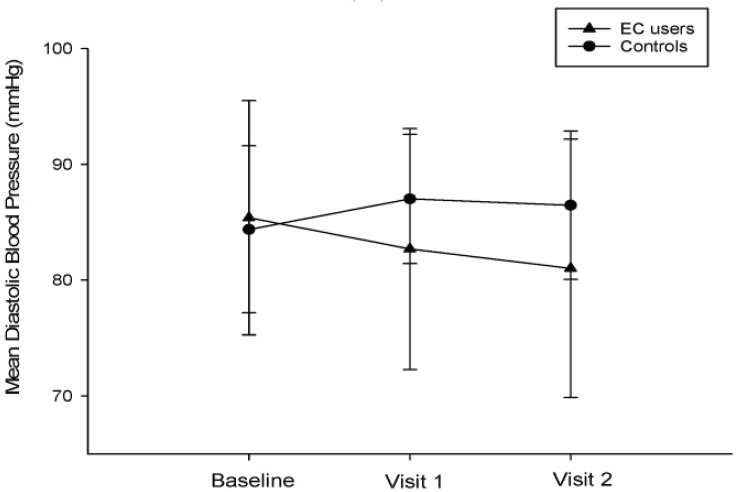
NOTES: All data expressed as mean, and error bars are standard error of the mean. EC = e-cigarette; mmHg = millimeters of mercury.
SOURCE: Polosa et al., 2016a.
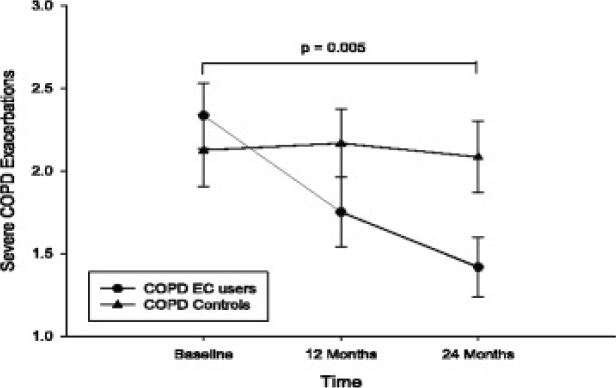
NOTES: All data expressed as mean, and error bars are standard deviation of th mean. The p-value is an overall comparison of both groups over the 24-mont period. COPD = chronic obstructive pulmonary disease; EC = e-cigarette.
SOURCE: Polosa et al., 2016c.
also observed a significant reduction in COPD exacerbations among dual users. In addition, COPD symptoms and ability to perform physical activities improved at both follow-ups among e-cigarette users, whereas no changes were observed among the control group (exclusive smokers).
Szołtysek-Bołdys and colleagues (2014) examined the effects of e-cigarettes on acute cardiovascular outcomes compared with combustible tobacco smoking. Participants were smokers (n = 15 women) who smoked five or more combustible tobacco cigarettes per day for at least 2 years. The authors measured arterial stiffness (stiffness index and reflection index), systolic and diastolic blood pressure, and heart rate before and after smoking a combustible tobacco cigarette (filtered, “slim” cigarette with manufacturer-defined 0.7 mg nicotine) and using an e-cigarette (Ego-3, Volish Ltd., Poland with Crystal 2 clearomizer, 2.4-Ω heating coil, 3.4-V battery and e-liquid with 24 mg/ml nicotine). The authors observed no significant changes in arterial stiffness before and after smoking a combustible tobacco cigarette (stiffness index: 6.75 m/s [95% CI = 6.66–6.85] after versus 6.56 m/s [95% CI = 6.46–6.65] before, p = 0.0056; reflection index: 54.0 percent [95% CI = 51.5–56.7] after versus 49.6 percent [95% CI = 47.5–51.8] after, p = 0.010). They observed no significant changes in arterial stiffness after e-cigarette use, compared with before use. Systolic
and diastolic blood pressure and heart rate increased after use of both products, but the changes did not meet statistical significance.
Tatullo and colleagues (2016) conducted a clinical observational pilot study involving 110 smokers who switched to e-cigarettes. Smokers were divided into two groups, according to the number of years of combustible tobacco cigarette smoking: group 1 (less than 10 years of combustible tobacco cigarette smoking) and group 2 (more than 10 years). Patients were subjected to oral examinations to investigate plaque index, bleeding index, and papillary bleeding index. A questionnaire was distributed to self-assess the variations of some parameters of general health and to self-assess the need to smoke combustible tobacco cigarettes. At the end of the study, authors registered a progressive improvement in the periodontal indexes, as well as in the general health perception.
In a pilot study, Wadia and colleagues (2016) compared the gingival health (bleeding on probing to assess gingival inflammation) among 20 established smokers who substituted e-cigarettes for smoking combustible tobacco cigarettes for 2 weeks. The authors found a statistically significant increase in gingival inflammation after 2 weeks of using e-cigarettes instead of their usual combustible tobacco cigarettes.
Toxicity in In Vitro and Animal Studies
The committee reviewed in vitro and animal studies that directly compared effects of exposure to aerosols from e-cigarettes to effects of combustible tobacco cigarette smoke exposure. Table 18-2 compares characteristics of in vitro studies on the toxic effect of e-cigarettes compared with combustible tobacco cigarette studies. Table 18-3 compares characteristics of in vivo animal studies on the toxic effect of e-cigarettes compared with combustible tobacco cigarette studies. As the tables illustrate, the majority of studies (21 of 27 in vitro studies and 3 of 5 in vivo animal studies) favored e-cigarettes as products less harmful than combustible tobacco cigarettes. No studies found e-cigarettes to be more harmful than combustible tobacco cigarettes.
Synthesis
The health effects of using e-cigarettes are still not well understood, but current evidence points to e-cigarettes being less harmful than combustible tobacco cigarettes. All but one of the human studies reviewed showed significant short-term improvements in health outcomes in smokers who switched from combustible tobacco cigarettes to e-cigarettes. Although most of the reviewed studies included relatively small numbers of subjects, the health improvement after this transition
| Reference | Study Measures and Outcomes | Harmful Effects of E-Cigarettes Versus Combustible Tobacco Cigarettes | ||
|---|---|---|---|---|
| Favors E-Cigarettes as Less Harmful | Similar Harm | Favors Combustible Tobacco Cigarettes as Less Harmful | ||
| Anderson et al., 2016 | Reactive oxygen species, DNA damage, cell viability, and markers of programmed cell death pathways in HUVECs | ✔ | ||
| Anthérieu et al., 2017 | Viability, oxidative stress, and secretion of inflammatory mediators by human bronchial epithelial BEAS-2B cells | ✔ | ||
| Aufderheide and Emura, 2017 | Morphological alterations (histopathology) of differentiated immortalized primary NHBE cells (CL-1548) | ✔ | ||
| Aug et al., 2015 | Metabolome of HBECs | ✔ | ||
| Azzopardi et al., 2016 | Cytotoxicity test with H292 human bronchial epithelial cells | ✔ | ||
| Banerjee et al., 2017 | Transcriptomic perturbations in lung epithelial tissue (MucilAir) | ✔ | ||
| Barber et al., 2016 | Inflammatory processes, viability, density, and metabolic activity of HUVECs | ✔ | ||
| Breheny et al., 2017 | Carcinogenic potential with cell transformation assays in mouse fibroblast cells (Bhas 42) | ✔ | ||
| Reference | Study Measures and Outcomes | Harmful Effects of E-Cigarettes Versus Combustible Tobacco Cigarettes | ||
|---|---|---|---|---|
| Favors E-Cigarettes as Less Harmful | Similar Harm | Favors Combustible Tobacco Cigarettes as Less Harmful | ||
| Carson et al., 2017 | Ciliary function and secretion, cellular and exogenous NO concentrations in airway epithelial cultures from non-smoking human subjects | ✔ | ||
| Cervellati et al., 2014 | Cytotoxicity, ultrastructural morphology, and pro-inflammatory cytokines in skin (HaCaT) and lung (A549) cells | ✔ | ||
| Farsalinos et al., 2013 | Cytotoxicity in myocardial cells (H9c2) | ✔ | ||
| Fields et al., 2017 | Viability, barrier integrity, and gene promoter/expression regulation in human airway culture (EpiAirway) | ✔ | ||
| Haswell et al., 2017 | Transcriptional response in differentiated reconstituted human airway epithelia (MucilAir) | ✔ | ||
| Hom et al., 2016 | Platelets from healthy volunteers (n = 50) were exposed to combustible tobacco smoke extracts and e-cigarette aerosol extracts, and changes in platelet activation, adhesion, aggregation, and inflammation were evaluated | ✔ | ||
| Leigh et al., 2016 | Cell viability, metabolic activity, and release of inflammatory mediators (cytokines) in H292 human bronchial epithelial cells | ✔ | ||
| Misra et al., 2014 | Cytotoxicity, mutagenicity, genotoxicity, and inflammatory responses in human lung epithelial carcinoma cells (A549) | ✔ | ||
| Moses et al., 2017 | Gene expression profiling in primary HBEC cells | ✔ | ||
| Neilson et al., 2015 | Cytotoxicity in human 3D reconstructed airway tissues | ✔ | ||
| Putzhammer et al., 2016 | Cell death induction, proliferation rates, occurrence of intracellular reactive oxygen species, cell morphology in HUVECs | ✔ | ||
| Romagna et al., 2013 | Cell viability in murine fibroblasts (3T3) | ✔ | ||
| Rubenstein et al., 2015 | Kupffer cell complement receptor expression, oxidative stress production, cytokine release and viability, and density | ✔ | ||
| Scheffler et al., 2015 | Cell viability and histology of immortalized normal human bronchial epithelial cell (NHBE48) | ✔ | ||
| Shen et al., 2016 | Transcriptomes of differentiated human bronchial epithelial cells | ✔ | ||
| Taylor et al., 2016 | Oxidative stress, apoptotic and necrotic responses in human bronchial epithelial cells | ✔ | ||
| Teasdale et al., 2016 | Stress response in human coronary artery endothelial cells | ✔ | ||
| Thorne et al., 2016 | Mutagenicity test with Salmonella typhimurium strains TA98 and TA100 | ✔ | ||
| Thorne et al., 2017 | Induction of double-strand DNA damage in vitro using human lung epithelial cells (BEAS-2Bs) | ✔ |
NOTE: HBEC = human bronchial epithelial cell; HUVEC = human umbilical vein endothelial cell; NHBE = normal human bronchial epithelial.
| Reference | Study Measures and Outcomes | Harmful Effects of E-Cigarettes Versus Combustible Tobacco Cigarettes | ||
|---|---|---|---|---|
| Favors E-Cigarettes as Less Harmful | Similar Harm | Favors Combustible Tobacco Cigarettes as Less Harmful | ||
| Larcombe et al., 2017 | Pulmonary inflammation, lung volume, lung mechanics, and responsiveness to methacholine were measured in female BALB/c mice exposed for 8 weeks to tobacco smoke or one of four types of e-cigarette aerosol | ✔ | ||
| Palpant et al., 2015 | Developmental effects in vivo with zebrafish (Danio rerio) and cardiac differentiation of human embryonic stem cells | ✔ | ||
| Parker et al., 2017 | Developmental toxicities using the FETAX | ✔ | ||
| Ponzoni et al., 2015 | Body weight, food intake, and the signs of mecamylamine-precipitated and spontaneous withdrawal episodic memory and emotional responses in male BALB/c mice | ✔ | ||
| Rau et al., 2017 | Skin flap survival (microcirculation and perfusion) in rats | ✔ | ||
NOTE: FETAX = frog embryo teratogenesis assay–Xenopus.
from smoking combustible tobacco cigarettes to using e-cigarettes was consistent across all studies and was observed for respiratory, cardiovascular, and oral health outcomes. In several studies, smokers also self-reported improvement in health after switching to e-cigarettes. Although in vitro and animal studies that compared acute effects of exposure to e-cigarette aerosols with effects caused by combustible tobacco smoke provided mixed results, the majority of studies favored e-cigarettes as less harmful products than combustible tobacco cigarettes. Moreover, although some studies found similar harm from e-cigarettes, no studies found that e-cigarettes were more harmful than combustible tobacco cigarettes among combustible tobacco cigarette smokers who switched to exclusive e-cigarette use. E-cigarettes might be considered as a harm reduction tool for tobacco smokers if their efficacy in reducing health risk is supported by epidemiological studies and proven in well-performed epidemiological studies and RCTs.
Conclusion 18-2. There is substantial evidence that completely switching from regular use of combustible tobacco cigarettes to e-cigarettes results in reduced short-term adverse health outcomes in several organ systems.
Harm Reduction in Smokers Who Use E-Cigarettes Concurrently with Combustible Tobacco Cigarettes (Dual Users)
Health Risk Profile and Smoking Cessation
People who smoke combustible tobacco cigarettes may switch to other tobacco products (e.g., chewing tobacco) or use products concurrently (dual users) when attempting to quit smoking (Messer et al., 2015; Popova and Ling, 2013). This, however, is not a proven method for combustible tobacco cigarette cessation, conceivably because nicotine dependence persists while using these other products (Dunbar et al., 2016; Popova and Ling, 2013). For example, a recent study using data from the 2010–2011 Tobacco Use Supplement to the Current Population Survey found that although dual users were more likely to attempt to quit than those who smoked only combustible tobacco cigarettes, they reverted back to smoking more quickly (Messer et al., 2015). The study found no significant difference in the proportion of dual users and exclusive combustible tobacco cigarette smokers who abstained from combustible tobacco cigarettes in the last 30 days (Messer et al., 2015). In this sample, lower combustible tobacco cigarette consumption was the best predictor of abstinence from combustible tobacco cigarette smoking (Messer et al., 2015). These results have implications for clinicians advising patients
attempting to quit smoking combustible tobacco cigarettes (Dunbar et al., 2016).
Etter and Bullen (2014) used longitudinal Internet surveys to assess changes in tobacco use among e-cigarette users (including those using other tobacco products concurrently) over 12 months between 2011 and 2013. The authors recruited participants through e-cigarette and smoking cessation websites. In the recruited cohort, e-cigarette and tobacco use was assessed at baseline (n = 733), after 1 month (n = 477), and after 1 year (n = 367). Among dual users of e-cigarettes and combustible tobacco cigarettes at baseline, 22 percent reported abstaining from smoking in the previous 7 days after 1 month and 46 percent after 1 year. Dual users who were still smoking at follow-up reported a temporary reduction in combustible tobacco cigarette smoking at 1 month (average cigarettes per day declined from 11.3 to 6.0, p = 0.006). However, results showed no changes in smoking between baseline and 1-year follow-up.
Jorenby and colleagues (2017) conducted a 26-day study examining tobacco use behaviors among dual users (n = 74) compared with exclusive combustible tobacco cigarette smokers (n = 74). Subjects participated in 1 week of ad lib use, 1 week of 75 percent combustible tobacco smoking reduction (dual users were free to use their e-cigarettes as they wished), followed by another week of ad lib use, and finally 3 days of abstinence. The authors also measured CO and urinary nicotine and cotinine. Results showed that combustible tobacco cigarette consumption did not differ between dual users and exclusive smokers during ad lib periods. However, dual users quadrupled their e-cigarette use during smoking reduction periods. Dual users were significantly more likely to maintain 100 percent reduction (97.1 percent versus 81.2 percent). Nicotine levels were higher among women dual users.
Loukas and colleagues (2016) examined patterns of tobacco and e-cigarette use, quit attempts, and dependence symptoms among college students (n = 5,468, age 18–29). The study found that poly-tobacco product use is associated with some indicators of dependence, but not with smoking cessation attempts.
Manzoli and colleagues (2015, 2017) evaluated e-cigarette efficacy and safety at 12 and 24 months using data from a prospective cohort study of 1,355 subjects, including 343 users of e-cigarettes only and 319 dual users of tobacco and e-cigarettes. Most dual users at baseline abandoned e-cigarettes and continued to smoke tobacco. At 12 months, 21.9 percent of dual users quit combustible tobacco smoking while 20.5 percent of those who only smoked combustible tobacco cigarettes quit smoking. After 24 months, 26.0 percent of dual users quit smoking while 23.1 percent of those who only smoke combustible tobacco cigarettes quit smoking.
Synthesis
Dual use of combustible tobacco cigarettes and e-cigarettes is highly prevalent among adults and youth; however, there is limited evidence about dual users’ patterns of use and smoking cessation attempts. The studies reviewed show that, on average, dual users do not smoke fewer combustible tobacco cigarettes than those who smoke only combustible tobacco cigarettes; however, among dual users, e-cigarettes may help maintain smoking reduction. There is a lack of evidence on exposure levels to nicotine and toxicants and health outcomes among dual users who do not reduce combustible tobacco cigarette use. It is very unlikely that those smokers who do not reduce smoking after initiating e-cigarette use will reduce health risks of smoking and they may also be exposed to additional adverse health effects of e-cigarettes. A better understanding of the patterns and differing contexts of dual use of e-cigarettes and combustible tobacco cigarettes is needed to inform public policy on adult and youth e-cigarette use and combustible tobacco cigarette smoking.
Conclusion 18-3. There is no available evidence whether or not long-term e-cigarette use among smokers (dual use) changes morbidity or mortality compared with those who only smoke combustible tobacco cigarettes.
Conclusion 18-4. There is insufficient evidence that e-cigarette use changes short-term adverse health outcomes in several organ systems in smokers who continue to smoke combustible tobacco cigarettes (dual users).
Harm Reduction from Passive Exposure to E-Cigarette Aerosol Compared with Combustible Tobacco Cigarette Smoke Among Non-Users
As described in the committee’s discussion of secondhand exposures to e-cigarette aerosol compared with ambient air (see Chapter 3), the Surgeon General and the World Health Organization Framework Convention on Tobacco Control have indicated that there is no risk-free level of exposure to secondhand tobacco smoke (HHS, 2006; WHO, 2003). Additionally, just as quitting smoking is the only guaranteed way to reduce tobacco-related harms, eliminating exposure from indoor spaces is the most effective intervention to prevent secondhand tobacco smoke exposure. Due to the involuntary nature of secondhand exposure, such strategies are particularly important to reduce exposures to vulnerable populations, such as children, pregnant women, the elderly, and individuals with cardiorespiratory disease. Because eliminating exposure to
passive combustible tobacco cigarette smoke in the indoor environment has been the traditional focus of tobacco control, whether replacing smoking by e-cigarette use in indoor environments is a possible strategy to reduce risk of health effects among those involuntarily exposed to secondhand smoke is an effective harm reduction strategy is unknown. This may be especially important to reduce harm for non-smoking household members of smokers who are unable or unwilling to quit using other evidence-based smoking cessation methods. In this section, the committee reviews evidence on whether changing smoking practices by switching to e-cigarettes in indoor environments may reduce passive exposure to combustible tobacco smoke constituents. The committee did not identify evidence on clinically relevant health outcomes of passive exposure to e-cigarettes. In the absence of such direct literature, the committee draws upon four studies assessing exposure among non-smokers to emissions from e-cigarettes compared with combustible tobacco cigarettes.
In the same study described earlier in the chapter regarding effects of active e-cigarette use on CBC markers compared with effects of active smoking, Flouris and colleagues (2012) also evaluated the effect of passive exposure on CBC markers in 15 never smokers. Never smokers underwent three 30-minute experimental exposure sessions: a control session, a passive combustible tobacco cigarette exposure session, and a passive e-cigarette exposure session (Giant brand, Nobacco G.P., Greece filled with tobacco-flavored nicotine-containing 11-mg/ml solution). CBC indexes remained unchanged during the control session and the passive e-cigarette exposure sessions (p > 0.05). By contrast, passive combustible tobacco smoke exposure increased white blood cell, lymphocyte, and granulocyte counts for at least 1 hour in never smokers (p < 0.05). The authors also examined effects on antioxidant response, and found no changes in TAC, CAT, and GSH before, immediately after, and 1 hour after exposure to any condition (e-cigarette aerosol, combustible tobacco smoke, and control) (Poulianiti et al., 2016).
Ballbè and colleagues (2014) conducted an observational study to characterize passive exposure to nicotine from e-cigarettes and combustible tobacco cigarettes among non-smokers (n = 54) from home settings with different tobacco use conditions. Twenty-five participants lived in homes with smokers, 5 lived with nicotine-containing e-cigarette users, and 24 lived in homes with no combustible tobacco cigarette smokers or e-cigarette users (control homes). All participants passively exposed to e-cigarettes reported more than 2 hours of exposure per day, while 17 of the 25 participants passively exposed to tobacco smoke reported less than 2 hours of exposure per day. Airborne nicotine at home and biomarkers of nicotine exposure (salivary and urinary cotinine) were measured. Airborne nicotine was significantly higher (5.7 times) in homes with smokers
than in homes with e-cigarette users. Airborne nicotine (geometric means [geometric standard deviation, GSD]) was 0.74 µg/m3 (GSD = 4.05) in homes with smokers and 0.13 µg/m3 (GSD = 2.4) in homes with e-cigarette users. Salivary cotinine concentrations were also significantly higher among non-smokers passively exposed to tobacco smoke compared with those passively exposed to e-cigarettes. Salivary cotinine was 0.38 ng/ml (GSD = 2.34) in the smokers’ homes compared with 0.19 ng/ml (GSD = 2.17) in the e-cigarettes users’ homes.
Czogała and colleagues (2014) compared secondhand exposure among aerosols from three e-cigarette models (Colins Age with Camel High cartomizer with 11 mg nicotine [Colins Poland], Dekang 510 Pen with SGC Regular cartridge with 18 mg nicotine [Ecigars Polska], and Mild M201 Pen with Marlboro cartridge with 19 mg nicotine [Mild Poland]) and combustible tobacco smoke generated by a smoking machine and by five dual users. Nicotine was measured over 1-hour exposure using gas chromatography with nitrogen–phosphorus detector following active sampling on XAD-4 sorption tubes (SKC Inc.) according to the National Institute of Occupational Safety and Health reference method 2551. Results showed that e-cigarettes are a source of secondhand exposure to nicotine, but not CO, and VOCs. The average concentration of airborne nicotine over 1 hour from smoking combustible tobacco cigarettes was 10 times higher than from e-cigarettes (31.60 ± 6.91 versus 3.32 ± 2.49 µg/m3, respectively; p = 0.0081; see Figure 18-6). Similarly, the mean PM2.5 concentration from tobacco smoke was seven times higher compared with that from e-cigarettes (819.3 ± 228.6 versus 151.7 ± 86.8 µg/m3, respectively; p = 0.0081). The number of aerosol particles (PM2.5) generated directly by the e-cigarette user was higher than generated by a smoking machine, suggesting that examining aerosols exhaled by users may be more appropriate than those produced by smoking machines.
In a pilot study, Bush and Goniewicz (2015) measured nicotine on the household surfaces in homes of e-cigarette users (n = 8), combustible tobacco cigarette smokers (n = 6), and non-users of either product (n = 8) in western New York. The e-cigarette users estimated that they puffed on their own devices from 50 to 500 times per day in their home and reported that the nicotine concentration in their e-liquids ranged from 10 to 15 mg/ml. Investigators took surface wipe samples from the floor, wall, and window. They then extracted nicotine from the wipes and analyzed the extract using gas chromatography. Results showed that detectable levels of nicotine were found on surfaces of half of the e-cigarette users’ homes, whereas it was found on surfaces in all of the smokers’ homes. Additionally, in homes of e-cigarette users where nicotine was found on surfaces, the nicotine levels were significantly lower than in combustible tobacco
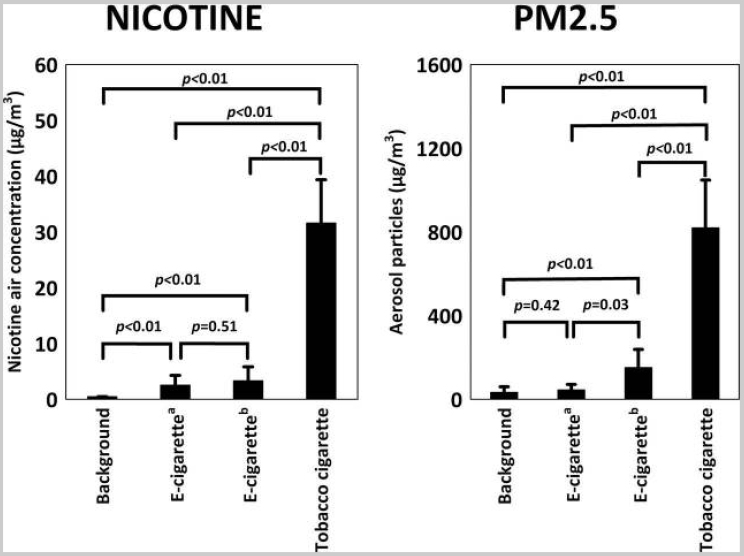
a Aerosol generated with smoking machine (Study 1).
b Aerosol exhaled by users (Study 2).
NOTE: PM2.5 = particulate matter 2.5 micrometers or less in diameter.
SOURCE: Czogała et al., 2014.
cigarette smokers’ homes (average concentration 7.7 ± 17.2 versus 1,303 ± 2,676 µg/m2; p < 0.05).
Synthesis
The committee identified a limited number of studies that compared secondhand exposure to e-cigarette emissions to combustible tobacco cigarette smoke. The committee did not identify any long-term studies comparing health effects resulting from passive exposure to secondhand aerosol from e-cigarettes with effects in non-smokers passively exposed to tobacco smoke. In general, the studies reviewed show that using an e-cigarette in indoor environments may involuntarily expose non-users to nicotine and particulates, but at lower levels compared with exposure to secondhand tobacco smoke from combustible tobacco cigarettes. Second-
hand exposure to toxic tobacco-specific combustion products is substantially reduced from e-cigarettes compared with combustible tobacco cigarettes. Of note, the effects of these reduced exposures on health remain unknown. Due to the involuntary nature of secondhand exposure and because even low levels of particulate matter may confer health risks, vulnerable populations such as children, pregnant women, the elderly, and individuals with cardiorespiratory diseases may still be at special risk.
Conclusion 18-5. There is moderate evidence that secondhand exposure to nicotine and particulates is lower from e-cigarettes compared with combustible tobacco cigarettes.
REFERENCES
Adriaens, K., D. Van Gucht, P. Declerck, and F. Baeyens. 2014. Effectiveness of the electronic cigarette: An eight-week Flemish study with six-month follow-up on smoking reduction, craving and experienced benefits and complaints. International Journal of Environmental Research & Public Health 11(11):11220–11248.
Anderson, C., A. Majeste, J. Hanus, and S. Wang. 2016. E-cigarette aerosol exposure induces reactive oxygen species, DNA damage, and cell death in vascular endothelial cells. Toxicological Sciences 154(2):332–340.
Anthérieu, S., A. Garat, N. Beauval, M. Soyez, D. Allorge, G. Garçon, and J. M. Lo-Guidice. 2017. Comparison of cellular and transcriptomic effects between electronic cigarette vapor and cigarette smoke in human bronchial epithelial cells. Toxicology in Vitro 45(Part 3):417–425.
Aufderheide, M., and M. Emura. 2017. Phenotypical changes in a differentiating immortalized bronchial epithelial cell line after exposure to mainstream cigarette smoke and e-cigarette vapor. Experimental and Toxicologic Pathology 69(6):393–401.
Aug, A., S. Altraja, K. Kilk, R. Porosk, U. Soomets, and A. Altraja. 2015. E-cigarette affects the metabolome of primary normal human bronchial epithelial cells. PLoS ONE 10(11):e0142053. https://doi.org/10.1371/journal.pone.0142053 (accessed February 7, 2018).
Azzopardi, D., K. Patel, T. Jaunky, S. Santopietro, O. M. Camacho, J. McAughey, and M. Gaça. 2016. Electronic cigarette aerosol induces significantly less cytotoxicity than tobacco smoke. Toxicology Mechanisms and Methods 26(6):477–491.
Ballbè, M., J. M. Martínez-Sánchez, X. Sureda, M. Fu, R. Pérez-Ortuño, J. A. Pascual, E. Saltó, and E. Fernández. 2014. Cigarettes vs. e-cigarettes: Passive exposure at home measured by means of airborne marker and biomarkers. Environmental Research 135:76–80.
Banerjee, A., L. E. Haswell, A. Baxter, A. Parmar, D. Azzopardi, S. Corke, D. Thorne, J. Adamson, J. Mushonganono, M. Gaça, and E. Minet. 2017. Differential gene expression using RNA sequencing profiling in a reconstituted airway epithelium exposed to conventional cigarette smoke or electronic cigarette aerosols. Applied In Vitro Toxicology 3(1):84–98.
Barber, K. E., B. Ghebrehiwet, W. Yin, and D. A. Rubenstein. 2016. Endothelial cell inflammatory reactions are altered in the presence of e-cigarette extracts of variable nicotine. Cellular and Molecular Bioengineering 10(1):124–133.
Breheny, D., O. Oke, K. Pant, and M. Gaça. 2017. Comparative tumor promotion assessment of e-cigarette and cigarettes using the in vitro Bhas 42 cell transformation assay. Environmental and Molecular Mutagenesis 58(4):190–198.
Bush, D., and M. L. Goniewicz. 2015. A pilot study on nicotine residues in houses of electronic cigarette users, tobacco smokers, and non-users of nicotine-containing products. International Journal of Drug Policy 26(6):609–611.
Carnevale, R., S. Sciarretta, F. Violi, C. Nocella, L. Loffredo, L. Perri, M. Peruzzi, A. G. M. Marullo, E. De Falco, I. Chimenti, V. Valenti, G. Biondi-Zoccai, and G. Frati. 2016. Acute impact of tobacco vs. electronic cigarette smoking on oxidative stress and vascular function. Chest 150(3):606–612.
Carson, J. L., L. Zhou, L. Brighton, K. H. Mills, H. Zhou, I. Jaspers, and M. Hazucha. 2017. Temporal structure/function variation in cultured differentiated human nasal epithelium associated with acute single exposure to tobacco smoke or e-cigarette vapor. Inhalation Toxicology 29(3):137–144.
Cervellati, F., X. M. Muresan, C. Sticozzi, R. Gambari, G. Montagner, H. J. Forman, C. Torricelli, E. Maioli, and G. Valacchi. 2014. Comparative effects between electronic and cigarette smoke in human keratinocytes and epithelial lung cells. Toxicology in Vitro 28(5):999–1005.
Cibella, F., D. Campagna, P. Caponnetto, M. D. Amaradio, M. Caruso, C. Russo, D. W. Cockcroft, and R. Polosa. 2016. Lung function and respiratory symptoms in a randomized smoking cessation trial of electronic cigarettes. Clinical Science 130(21):1929–1937.
Cravo, A. S., J. Bush, G. Sharma, R. Savioz, C. Martin, S. Craige, and T. Walele. 2016. A randomised, parallel group study to evaluate the safety profile of an electronic vapour product over 12 weeks. Regulatory Toxicology and Pharmacology 81 (Supplement 1):S1–S14. https://doi.org/10.1016/j.yrtph.2016.10.003 (accessed February 7, 2018).
Czogała, J., M. L. Goniewicz, B. Fidelus, W. Zielinska-Danch, M. J. Travers, and A. Sobczak. 2014. Secondhand exposure to vapors from electronic cigarettes. Nicotine & Tobacco Research 16(6):655–662.
D’Ruiz, C. D., D. W. Graff, and X. S. Yan. 2015. Nicotine delivery, tolerability and reduction of smoking urge in smokers following short-term use of one brand of electronic cigarettes. BMC Public Health 15:991. https://doi.org/10.1186/s12889-015-2349-2 (accessed February 7, 2018).
D’Ruiz, C. D., D. W. Graff, and E. Robinson. 2016. Reductions in biomarkers of exposure, impacts on smoking urge and assessment of product use and tolerability in adult smokers following partial or complete substitution of cigarettes with electronic cigarettes. BMC Public Health 16(1):543.
D’Ruiz, C. D., G. O’Connell, D. W. Graff, and X. S. Yan. 2017. Measurement of cardiovascular and pulmonary function endpoints and other physiological effects following partial or complete substitution of cigarettes with electronic cigarettes in adult smokers. Regulatory Toxicology and Pharmacology 87:36–53.
Dunbar, M. S., W. G. Shadel, J. S. Tucker, and M. O. Edelen. 2016. Use of and reasons for using multiple other tobacco products in daily and nondaily smokers: Associations with cigarette consumption and nicotine dependence. Drug and Alcohol Dependence 168:156–163.
Etter, J. F., and C. Bullen. 2014. A longitudinal study of electronic cigarette users. Addictive Behaviors 39(2):491–494.
Farsalinos, K. E., G. Romagna, E. Allifranchini, E. Ripamonti, E. Bocchietto, S. Todeschi, D. Tsiapras, S. Kyrzopoulos, and V. Voudris. 2013. Comparison of the cytotoxic potential of cigarette smoke and electronic cigarette vapour extract on cultured myocardial cells. International Journal of Environmental Research and Public Health 10(10):5146–5162.
Fields, W., A. Maione, B. Keyser, and B. Bombick. 2017. Characterization and application of the Vitrocell VC1 smoke exposure system and 3D EpiAirway models for toxicological and e-cigarette evaluations. Applied In Vitro Toxicology 3(1):68–83.
Flouris, A. D., K. P. Poulianiti, M. S. Chorti, A. Z. Jamurtas, D. Kouretas, E. O. Owolabi, M. N. Tzatzarakis, A. M. Tsatsakis, and Y. Koutedakis. 2012. Acute effects of electronic and tobacco cigarette smoking on complete blood count. Food and Chemical Toxicology 50(10):3600–3603.
Goniewicz, M. L., J. Knysak, M. Gawron, L. Kosmider, A. Sobczak, J. Kurek, A. Prokopowicz, M. Jablonska-Czapla, C. Rosik-Dulewska, C. Havel, P. Jacob, and N. Benowitz. 2014. Levels of selected carcinogens and toxicants in vapour from electronic cigarettes. Tobacco Control 23(2):133–139.
Goniewicz, M. L., M. Gawron, D. M. Smith, M. Peng, P. Jacob, 3rd, and N. L. Benowitz. 2017. Exposure to nicotine and selected toxicants in cigarette smokers who switched to electronic cigarettes: A longitudinal within-subjects observational study. Nicotine & Tobacco Research 19(2):160–167.
Haswell, L. E., A. Baxter, A. Banerjee, I. Verrastro, J. Mushonganono, J. Adamson, D. Thorne, M. Gaça, and E. Minet. 2017. Reduced biological effect of e-cigarette aerosol compared to cigarette smoke evaluated in vitro using normalized nicotine dose and RNA-seq-based toxicogenomics. Scientific Reports 7(1):888. https://doi.org/10.1038/s41598-017-00852-y (accessed February 7, 2018).
Healthy People 2020. 2017. Tobacco: Overview and impact. https://www.healthypeople.gov/2020/leading-health-indicators/2020-lhi-topics/Tobacco (accessed November 29, 2017).
Hecht, S. S., S. G. Carmella, D. Kotandeniya, M. E. Pillsbury, M. Chen, B. W. Ransom, R. I. Vogel, E. Thompson, S. E. Murphy, and D. K. Hatsukami. 2015. Evaluation of toxicant and carcinogen metabolites in the urine of e-cigarette users versus cigarette smokers. Nicotine & Tobacco Research 17(6):704–709.
HHS (U.S. Department of Health and Human Services). 2006. The health consequences of involuntary exposure to tobacco smoke: A report of the Surgeon General. Atlanta, GA: U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Disease Prevention and Health Promotion, Office on Smoking and Health.
HHS. 2014. The health consequences of smoking—50 years of progress: A report of the Surgeon General. Atlanta, GA: U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Disease Prevention and Health Promotion, Office on Smoking and Health.
Hom, S., L. Chen, T. Wang, B. Ghebrehiwet, W. Yin, and D. A. Rubenstein. 2016. Platelet activation, adhesion, inflammation, and aggregation potential are altered in the presence of electronic cigarette extracts of variable nicotine concentrations. Platelets 27(7):694–702.
IOM (Institute of Medicine). 2001. Clearing the smoke: Assessing the science base for tobacco harm reduction. Washington, DC: National Academy Press.
Jorenby, D. E., S. S. Smith, M. C. Fiore, and T. B. Baker. 2017. Nicotine levels, withdrawal symptoms, and smoking reduction success in real world use: A comparison of cigarette smokers and dual users of both cigarettes and e-cigarettes. Drug and Alcohol Dependence 170:93–101.
Larcombe, A. N., M. A. Janka, B. J. Mullins, L. J. Berry, A. Bredin, and P. J. Franklin. 2017. The effects of electronic cigarette aerosol exposure on inflammation and lung function in mice. American Journal of Physiology: Lung Cellular and Molecular Physiology 313(1):L67–L79.
Leigh, N. J., R. I. Lawton, P. A. Hershberger, and M. L. Goniewicz. 2016. Flavourings significantly affect inhalation toxicity of aerosol generated from electronic nicotine delivery systems (ENDS). Tobacco Control 25:ii81–ii87. https://doi.org/10.1136/tobaccocontrol-2016-053205 (accessed February 7, 2018).
Lindson-Hawley, N., J. Hartmann-Boyce, T. R. Fanshawe, R. Begh, A. Farley, and T. Lancaster. 2016. Interventions to reduce harm from continued tobacco use. Cochrane Database of Systematic Reviews 10:CD005231.
Loukas, A., S. Chow, K. E. Pasch, X. Li, J. T. Hinds, III, C. N. Marti, M. B. Harrell, M. R. Creamer, and C. L. Perry. 2016. College students’ polytobacco use, cigarette cessation, and dependence. American Journal of Health Behavior 40(4):514–522.
Majeed, B. A., S. R. Weaver, K. R. Gregory, C. F. Whitney, P. Slovic, T. F. Pechacek, and M. P. Eriksen. 2016. Changing perceptions of harm of e-cigarettes among U.S. adults, 2012–2015. American Journal of Preventive Medicine 52(3):331–338.
Manzoli, L., M. E. Flacco, M. Fiore, C. La Vecchia, C. Marzuillo, M. R. Gualano, G. Liguori, G. Cicolini, L. Capasso, C. D’Amario, S. Boccia, R. Siliquini, W. Ricciardi, and P. Villari. 2015. Electronic cigarettes efficacy and safety at 12 months: Cohort study. PLoS ONE 10(6):e0129443. https://doi.org/10.1371/journal.pone.0129443 (accessed February 7, 2018).
Manzoli, L., M. E. Flacco, M. Ferrante, C. La Vecchia, R. Siliquini, W. Ricciardi, C. Marzuillo, P. Villari, and M. Fiore. 2017. Cohort study of electronic cigarette use: Effectiveness and safety at 24 months. Tobacco Control 26(3):284–292.
Margham, J., K. McAdam, M. Forster, C. Liu, C. Wright, D. Mariner, and C. Proctor. 2016. Chemical composition of aerosol from an e-cigarette: A quantitative comparison with cigarette smoke. Chemical Research in Toxicology 29(10):1662–1678.
Martin, E. M., P. W. Clapp, M. E. Rebuli, E. A. Pawlak, E. Glista-Baker, N. L. Benowitz, R. C. Fry, and I. Jaspers. 2016. E-cigarette use results in suppression of immune and inflammatory-response genes in nasal epithelial cells similar to cigarette smoke. American Journal of Physiology: Lung Cellular and Molecular Physiology 311(1):L135–L144.
McKee, M., and S. Capewell. 2015. Evidence about electronic cigarettes: A foundation built on rock or sand? BMJ 351:h4863. https://doi.org/10.1136/bmj.h4863 (accessed February 7, 2018).
McRobbie, H., A. Phillips, M. L. Goniewicz, K. M. Smith, O. Knight-West, D. Przulj, and P. Hajek. 2015. Effects of switching to electronic cigarettes with and without concurrent smoking on exposure to nicotine, carbon monoxide, and acrolein. Cancer Prevention Research 8(9):873–878.
Messer, K., M. Vijayaraghavan, M. M. White, Y. Shi, C. Chang, K. P. Conway, A. Hartman, M. J. Schroeder, W. M. Compton, and J. P. Pierce. 2015. Cigarette smoking cessation attempts among current U.S. smokers who also use smokeless tobacco. Addictive Behaviors 51:113–119.
Misra, M., R. D. Leverette, B. T. Cooper, M. B. Bennett, and S. E. Brown. 2014. Comparative in vitro toxicity profile of electronic and tobacco cigarettes, smokeless tobacco and nicotine replacement therapy products: E-liquids, extracts and collected aerosols. International Journal of Environmental Research & Public Health 11(11):11325–11347.
Moses, E., T. Wang, S. Corbett, G. R. Jackson, E. Drizik, C. Perdomo, C. Perdomo, E. Kleerup, D. Brooks, G. O’Connor, S. Dubinett, P. Hayden, M. E. Lenburg, and A. Spira. 2017. Molecular impact of electronic cigarette aerosol exposure in human bronchial epithelium. Toxicological Sciences 155(1):248–257.
Neilson, L., C. Mankus, D. Thorne, G. Jackson, J. DeBay, and C. Meredith. 2015. Development of an in vitro cytotoxicity model for aerosol exposure using 3D reconstructed human airway tissue; application for assessment of e-cigarette aerosol. Toxicology in Vitro 29(7):1952–1962.
O’Connell, G., D. W. Graff, and C. D. D’Ruiz. 2016. Reductions in biomarkers of exposure (BOE) to harmful or potentially harmful constituents (HPHCs) following partial or complete substitution of cigarettes with electronic cigarettes in adult smokers. Toxicology Mechanisms and Methods 26(6):443–454.
Palpant, N. J., P. Hofsteen, L. Pabon, H. Reinecke, and C. E. Murry. 2015. Cardiac development in zebrafish and human embryonic stem cells is inhibited by exposure to tobacco cigarettes and e-cigarettes. PLoS ONE 10(5):e0126259. https://doi.org/10.1371/journal.pone.0126259 (accessed February 7, 2018).
Parker, T. T., and J. Rayburn. 2017. A comparison of electronic and traditional cigarette butt leachate on the development of Xenopus laevis embryos. Toxicology Reports 4:77–82.
Polosa, R., J. Morjaria, P. Caponnetto, M. Caruso, S. Strano, E. Battaglia, and C. Russo. 2014a. Effect of smoking abstinence and reduction in asthmatic smokers switching to electronic cigarettes: Evidence for harm reversal. International Journal of Environmental Research and Public Health 11(5):4965–4977.
Polosa, R., J. B. Morjaria, P. Caponnetto, D. Campagna, C. Russo, A. Alamo, M. D. Amaradio, and A. Fisichella. 2014b. Effectiveness and tolerability of electronic cigarette in real-life: A 24-month prospective observational study. Internal and Emergency Medicine 9(5):537–546.
Polosa, R., J. B. Morjaria, P. Caponnetto, E. Battaglia, C. Russo, C. Ciampi, G. Adams, and C. M. Bruno. 2016a. Blood pressure control in smokers with arterial hypertension who switched to electronic cigarettes. International Journal of Environmental Research and Public Health 13(11):E1123. https://doi.org/10.3390/ijerph13111123 (accessed February 7, 2018).
Polosa, R., J. B. Morjaria, P. Caponnetto, M. Caruso, D. Campagna, M. D. Amaradio, G. Ciampi, C. Russo, and A. Fisichella. 2016b. Persisting long term benefits of smoking abstinence and reduction in asthmatic smokers who have switched to electronic cigarettes. Discovery Medicine 21(114):99–108.
Polosa, R., J. B. Morjaria, P. Caponnetto, U. Prosperini, C. Russo, A. Pennisi, and C. M. Bruno. 2016c. Evidence for harm reduction in COPD smokers who switch to electronic cigarettes. Respiratory Research 17(1):166. https://doi.org/10.1186/s12931-016-0481-x (accessed February 7, 2018).
Ponzoni, L., M. Moretti, M. Sala, F. Fasoli, V. Mucchietto, V. Lucini, G. Cannazza, G. Gallesi, C. N. Castellana, F. Clementi, M. Zoli, C. Gotti, and D. Braida. 2015. Different physiological and behavioural effects of e-cigarette vapour and cigarette smoke in mice. European Neuropsychopharmacology 25(10):1775–1786.
Popova, L., and P. M. Ling. 2013. Alternative tobacco product use and smoking cessation: A national study. American Journal of Public Health 103(5):923–930.
Poulianiti, K., C. Karatzaferi, A. D. Flouris, I. G. Fatouros, Y. Koutedakis, and A. Z. Jamurtas. 2016. Antioxidant responses following active and passive smoking of tobacco and electronic cigarettes. Toxicology Mechanisms and Methods 26(6):455–461.
Pulvers, K., A. S. Emami, N. L. Nollen, D. R. Romero, D. R. Strong, N. L. Benowitz, and J. S. Ahluwalia. 2016. Tobacco consumption and toxicant exposure of cigarette smokers using electronic cigarettes. Nicotine & Tobacco Research 20(2):206–214.
Putzhammer, R., C. Doppler, T. Jakschitz, K. Heinz, J. Forste, K. Danzl, B. Messner, and D. Bernhard. 2016. Vapours of U.S. and E.U. market leader electronic cigarette brands and liquids are cytotoxic for human vascular endothelial cells. PLoS ONE 11(6):e0157337. https://doi.org/10.1371/journal.pone.0157337 (accessed February 7, 2018).
Rau, A. S., V. Reinikovaite, E. P. Schmidt, L. Taraseviciene-Stewart, and F. W. Deleyiannis. 2017. Electronic cigarettes are as toxic to skin flap survival as tobacco cigarettes. Annals of Plastic Surgery 79(1):86–91.
Romagna, G., E. Allifranchini, E. Bocchietto, S. Todeschi, M. Esposito, and K. E. Farsalinos. 2013. Cytotoxicity evaluation of electronic cigarette vapor extract on cultured mammalian fibroblasts (ClearStream-LIFE): Comparison with tobacco cigarette smoke extract. Inhalation Toxicology 25(6):354–361.
Rubenstein, D. A., S. Hom, B. Ghebrehiwet, and W. Yin. 2015. Tobacco and e-cigarette products initiate Kupffer cell inflammatory responses. Molecular Immunology 67(2):652–660.
Scheffler, S., H. Dieken, O. Krischenowski, and M. Aufderheide. 2015. Cytotoxic evaluation of e-liquid aerosol using different lung-derived cell models. International Journal of Environmental Research and Public Health 12(10):12466–12474.
Shahab, L., M. L. Goniewicz, B. C. Blount, J. Brown, A. McNeill, K. U. Alwis, J. Feng, L. Wang, and R. West. 2017. Nicotine, carcinogen, and toxin exposure in long-term e-cigarette and nicotine replacement therapy users: A cross-sectional study. Annals of Internal Medicine 166(6):390–400.
Shen, Y., M. J. Wolkowicz, T. Kotova, L. Fan, and M. P. Timko. 2016. Transcriptome sequencing reveals e-cigarette vapor and mainstream-smoke from tobacco cigarettes activate different gene expression profiles in human bronchial epithelial cells. Scientific Reports 6:23984. https://doi.org/10.1038/srep23984 (accessed February 7, 2018).
Szołtysek-Bołdys, I., A. Sobczak, W. Zieli ska-Danch, A. Bartoń, B. Koszowski, and L. Kośmider. 2014. Influence of inhaled nicotine source on arterial stiffness. Przegląd lekarski 71(11):572–575.
Tatullo, M., S. Gentile, F. Paduano, L. Santacroce, and M. Marrelli. 2016. Crosstalk between oral and general health status in e-smokers. Medicine (United States) 95(49):e5589. https://doi.org/10.1097/MD.0000000000005589 (accessed February 7, 2018).
Taylor, M., T. Carr, O. Oke, T. Jaunky, D. Breheny, F. Lowe, and M. Gaça. 2016. E-cigarette aerosols induce lower oxidative stress in vitro when compared to tobacco smoke. Toxicology Mechanisms and Methods 26(6):465–476.
Tayyarah, R., and G. A. Long. 2014. Comparison of select analytes in aerosol from e-cigarettes with smoke from conventional cigarettes and with ambient air. Regulatory Toxicology and Pharmacology 70(3):704–710.
Teasdale, J. E., A. C. Newby, N. J. Timpson, M. R. Munafo, and S. J. White. 2016. Cigarette smoke but not electronic cigarette aerosol activates a stress response in human coronary artery endothelial cells in culture. Drug and Alcohol Dependence 163:256–260.
Thorne, D., I. Crooks, M. Hollings, A. Seymour, C. Meredith, and M. Gaça. 2016. The mutagenic assessment of an electronic-cigarette and reference cigarette smoke using the Ames assay in strains TA98 and TA100. Mutation Research—Genetic Toxicology and Environmental Mutagenesis 812:29–38.
Thorne, D., S. Larard, A. Baxter, C. Meredith, and M. Gaça. 2017. The comparative in vitro assessment of e-cigarette and cigarette smoke aerosols using the γ-H2AX assay and applied dose measurements. Toxicology Letters 265:170–178.
van Staden, S. R., M. Groenewald, R. Engelbrecht, P. J. Becker, and L. T. Hazelhurst. 2013. Carboxyhaemoglobin levels, health and lifestyle perceptions in smokers converting from tobacco cigarettes to electronic cigarettes. South African Medical Journal. Suid-Afrikaanse Tydskrif Vir Geneeskunde 103(11):865–868.
Vansickel, A. R., C. O. Cobb, M. F. Weaver, and T. E. Eissenberg. 2010. A clinical laboratory model for evaluating the acute effects of electronic “cigarettes”: Nicotine delivery profile and cardiovascular and subjective effects. Cancer Epidemiology, Biomarkers & Prevention 19(8):1945–1953.
Wadia, R., V. Booth, H. F. Yap, and D. L. Moyes. 2016. A pilot study of the gingival response when smokers switch from smoking to vaping. British Dental Journal 221(11):722–726.
WHO (World Health Organization). 2003. WHO framework convention on tobacco control. Geneva, Switzerland: World Health Organization.
Williams, M., A. Villarreal, K. Bozhilov, S. Lin, and P. Talbot. 2013. Metal and silicate particles including nanoparticles are present in electronic cigarette cartomizer fluid and aerosol. PLoS ONE 8(3):e57987. https://doi.org/10.1371/journal.pone.0057987 (accessed February 7, 2018).
Yan, X. S., and C. D’Ruiz. 2015. Effects of using electronic cigarettes on nicotine delivery and cardiovascular function in comparison with regular cigarettes. Regulatory Toxicology and Pharmacology 71(1):24–34.
Zeller, M., D. Hatsukami, and Strategic Dialogue on Tobacco Harm Reduction Group. 2009. The strategic dialogue on tobacco harm reduction: A vision and blueprint for action in the US. Tobacco Control 18(4):324–332.










































