2
Committee Approach
The Statement of Task charges the committee with conducting a “comprehensive and systematic assessment and review of the literature” on the health effects of electronic cigarettes. The committee’s approach to this task was informed by published guidelines for conducting systematic reviews, as well as the approaches taken by prior National Academies committees (CRD, 2009; Higgins and Green, 2011; IOM, 2011a, pp. 10–24, 2011b, 2016, pp. 8–10; NASEM, 2017; NRC, 2014; OHAT, 2015; Sena et al., 2014; Whiting et al., 2016). Notably, the committee’s approach incorporated major attributes of systematic reviews. The committee systematically located, screened, and selected studies for review (including use of multiple databases to identify studies, predefined criteria to select studies for inclusion and exclusion, and systematically collecting data); evaluated individual studies for strengths and limitations; and synthesized findings into an assessment of the overall body of literature. The committee aims to be transparent about its process and thus describes its methods in this chapter and Appendix B with an eye to this goal.
The committee did not treat all bodies of evidence equally, and prioritized human studies (including studies on health effects as well as effects on combustible tobacco cigarette smoking initiation and cessation), which most relevantly bear on the committee’s charge for its most structured assessments. The chapter begins with brief overviews of the committee’s methods for identifying, reviewing, and assessing literature (more discussion of these methods is found in Appendix B). The chapter next describes the committee’s approach to assessing causality and integrating data from
human, animal, and in vitro studies. The chapter closes by presenting the standardized language the committee used to describe the weight of evidence assigned to its conclusions.
LITERATURE SEARCH
Working with a professional research librarian, the committee conducted a series of searches in six databases—PubMed, Scopus, World of Science, PsycINFO (ProQuest), MEDLINE (Ovid), and Embase (Ovid)—between February 1, 2017, and August 31, 2017,1 to identify all literature on e-cigarettes. In all databases, the committee used the following key terms: e-cigarette, e-cigarettes, electronic cigarette, electronic cigarettes, electronic nicotine delivery, electronic nicotine device, vape, vaping, and e-liquid.2 Searches in PubMed and MEDLINE also used the Medical Subject Headings (MeSH) term “electronic cigarettes.” Special searches further restricting the results from the original searches were conducted to more precisely identify literature on e-cigarettes and dependence outcomes as well as combustible tobacco smoking initiation and cessation outcomes. The committee’s literature search strategy is described comprehensively in Appendix B. After identifying literature, titles and abstracts of the search results were reviewed to identify studies for inclusion in the review. Inclusion criteria are listed in Appendix B. Studies that met the inclusion criteria were sorted by population (human, in vivo, animal, and in vitro) and outcomes for committee review and quality assessment.
LITERATURE REVIEW AND QUALITY ASSESSMENT
Health Effects Literature
For the assessment of studies on disease endpoints, in general, one committee member conducted an initial review of all literature identified pertaining to a set of outcomes. In its assessment of study strengths and limitations, the committee considered study design, elements of the design (e.g., sample size, setting, study population, exposure variables and methods of assessment, relevant controls or comparison groups, statistical methods, outcome measures assessed), other potential sources of
___________________
1 Due to e-pub ahead of print and online first articles, 2018 citations were captured. In addition, a few 2016 and 2017 studies may not have been captured due to lags and discrepancies in database indexing.
2 The committee excluded the term “e-liquid” from searches in Scopus and Web of Science, which are multidisciplinary databases, where the term produced results related to geothermal energy.
conflict of interest or bias,3 and study results. After the initial review, a full committee discussion evaluated the first assessment, with particular attention to the strengths and weaknesses of individual studies. More information on the committee’s qualitative assessment procedures can be found in Appendix B; special considerations for specific disease outcomes are discussed in Section II in the chapters on the relevant disease outcomes.
The committee used a modified approach to assess evidence from case studies, which are typically considered a weaker form of evidence. The committee looked for data on the patient (and patient characteristics where available), the exposure (including dose and other characteristics), and the conditions of the injury, accident, or other adverse outcome.
The committee also used a modified approach to assess in vivo animal and in vitro studies. The committee considered research design, conduct, analysis, and other sources of bias when assessing study strengths and weaknesses as it did for human studies.
Smoking Transitions Literature
The largest body of evidence was available on questions of e-cigarette use in relation to combustible tobacco cigarette smoking transitions (initiation and cessation). Not only are epidemiological studies available, but high-quality systematic reviews and meta-analyses also exist. Rather than replicating the efforts of these existing reviews, the committee began by assessing the quality of the existing reviews and then examined additional literature not included in the reviews. This supplemental literature was published after the search dates of the reviews or the committee judged that they contributed in some other way to the committee’s ability to draw causal inferences about the relationship between e-cigarette use and subsequent smoking behavior. The committee’s approach to causality is detailed in the next section. Methods to assess reviews were adapted from published guidelines and prior National Academies committees (NASEM, 2017; Whiting et al., 2016). Assessment of primary literature followed methods described above for assessment of literature on the
___________________
3 The committee recognizes a range of non-scientific influences on research, including but not limited to the research sponsorship and source of employment. The committee also acknowledges particular concerns in literature on the health effects of tobacco products due to the tobacco industry’s past involvement in manipulating evidence to support their interests. For completeness, the committee documented the source of research sponsorship (including the provision of e-cigarette products for use in trials), noting whether each study was funded by industry, a federal research agency, or other (e.g., university or foundation), or was not stated, as well as other industry participation in a table available as an online supplement at https://www.nap.edu/catalog/24952.
effects of e-cigarettes on health outcomes. Additional details and special considerations regarding the committee’s approach to assessing causality for these combustible tobacco cigarette smoking outcomes are provided in Chapters 16 and 17.
The committee did not systematically or comprehensively review the health effects of known constituents and contaminants of e-cigarette devices or their refill solutions (e.g., nicotine, humectants, and certain metals). Because many of these constituents have been widely studied in other settings, the committee draws on existing bodies of evidence to describe the known health risks of these constituent parts.
APPROACH TO ASSESSING CAUSALITY
The committee faced some unique issues given the very recent introduction of e-cigarettes and limited empirical evidence for assessing their health effects. While there is a general consensus that high-quality epidemiological studies backed by solid toxicology and other mechanistic biological evidence provide the strongest basis for making firm inferences regarding causality, that simply does not exist for these devices. With only a few exceptions, the epidemiological literature is quite limited, and even where it is strongest (assessing short-term cardiovascular and respiratory effects), it does not address the etiology of chronic diseases. In other cases, such as cancer and reproductive health, there is simply no credible epidemiological research to consider.
Given this challenge, the committee drew upon indirect evidence based on knowledge of the health effects of some of the constituents of e-cigarette products, notably nicotine and humectants. While the nature of the devices makes the inferences based on analogy speculative, it does provide one line of evidence relevant to assessment of health effects of e-cigarettes. Another important source of evidence is from toxicology and other evidence with implications for biological mechanisms of e-cigarettes. The certainty, magnitude, and health relevance of these pathways bear on the value of such information for making causal inferences. For example, in vivo animal evidence may be more pertinent to inferences regarding human health effects than in vitro findings. Nevertheless, this toxicological and mechanistic literature provides evidence supporting the plausibility of various mechanisms by which e-cigarette exposure could influence health.
Tying these diverse threads of indirectly relevant evidence together to draw a summary conclusion is necessarily somewhat subjective, bringing together the knowledge and judgment of the committee as a whole to reach a consensus. To provide comparable inferences across the full array of health concerns, the committee modified approaches used in
other National Academies reports and published guidelines on evidence synthesis (e.g., IOM, 2011a; NRC, 2007, 2014) to reach conclusions based on human evidence, animal evidence, and their integration.
Conceptual Framework
The committee developed a conceptual framework illustrating potential causal pathways by which e-cigarettes could affect health to help integrate and present evidence on known and likely e-cigarette exposures, potential mechanisms, intermediate outcomes, and disease endpoints. Figure 2-1 presents a simplified schematic of a generic plausible pathway between e-cigarettes and a health outcome. The committee presents modified frameworks applied to specific exposures, mechanisms, and
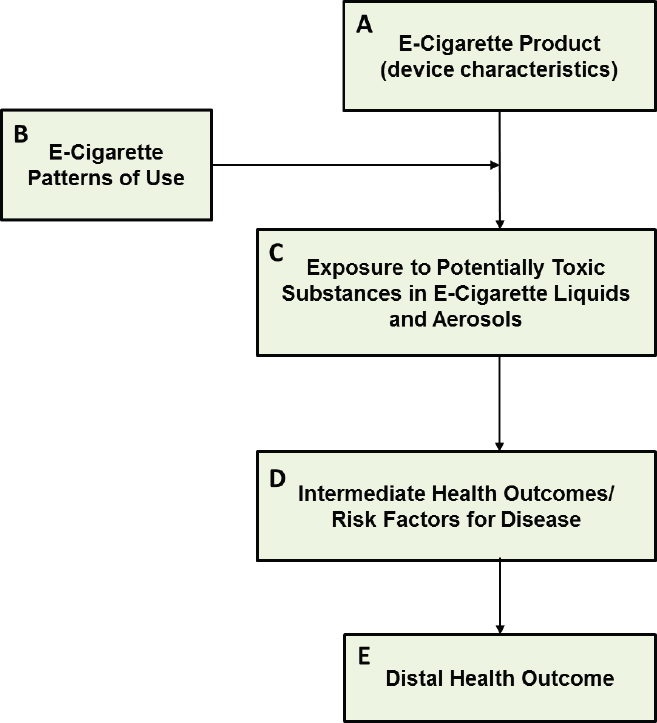
outcomes that represent plausible disease pathways for specific disease outcomes in Section II.
In Figure 2-1, the e-cigarette products used are shown at the top of the causal chain (Box A). Although e-cigarette devices and their components are not exposures per se, characteristics of e-cigarette products (e.g., nicotine concentration, power, etc.) influence the quantity and level of potential toxic substances emitted from the device. This emission, moderated by how the device is used (Box B), in turn influences users’ exposure to potentially toxic substances in e-cigarette aerosols (i.e., what is inhaled, Box C). These exposures influence intermediate health outcomes (Box D) proximally before affecting disease endpoints more distally (Box E). Intermediate outcomes include biomarkers of exposure, mechanisms, and biomarkers or risk factors of disease; they also capture short-term effects of e-cigarettes on organ systems, such as short-term increases in blood pressure. Distal disease consequences relevant to e-cigarette exposure examined in this report include cardiovascular, respiratory, oral, cancer, developmental and reproductive, and dependence outcomes.
Because the committee is primarily concerned about distal health outcomes, evidence on the effects of e-cigarette exposure on these outcomes is most relevant for the committee’s assessment of the health effects of e-cigarettes. In the absence of high-quality epidemiological evidence on these outcomes, the committee drew upon data further up the causal chain as additional evidence supportive of hypothesized disease pathways. Thus, to assess a given health risk, after examining evidence on long-term health outcomes, the committee looked to literature on intermediate or short-term outcomes, mechanisms, modes of action, and exposures from which it could draw inferences about potential health risk.
The committee considered data from humans to be most relevant for assessing human health risks of e-cigarettes, whereas additional animal data provide supporting evidence. For example, evidence of short-term effects of e-cigarette aerosol exposure from animal studies was considered weaker evidence compared with evidence of similar effects in humans. Although useful for hypothesis generation and critical for understanding mechanisms of health outcomes, because of important differences such as those pertaining to dose, duration of exposure, and changes in particles and constituents with aging, the relevance of in vitro data for establishing human health risk is uncertain.
Evidence Synthesis
The committee’s assessment of data aimed to establish causation between e-cigarettes and a given health endpoint, not merely a statistical association. In the absence of high-quality epidemiological studies,
the committee took into account several considerations to draw causal inferences from the evidence available. These considerations draw from criteria typically used to interpret and establish causation based on epidemiological data, and which are adapted from the approach taken in the 2014 Surgeon General’s report on smoking and health (HHS, 2014; Hill, 1965). First, the committee sought to identify the strength of an association between e-cigarettes and any given outcome. The committee began by identifying a statistical association or a point estimate of an effect. Relevant studies to determine such an association include experimental and observational studies examining an e-cigarette exposure and a health outcome. In general, randomized controlled trials (RCTs) provide the strongest evidence, but in some cases are infeasible for ethical reasons—for example, assessing risks of e-cigarette use among never smokers. Where randomized studies were not possible, prospective longitudinal studies provided the next strongest evidence. Well-documented case reports and case series provide evidence for the committee’s conclusion on injuries and poisonings. After establishing an association, the committee then considered the magnitude (strength) of the association, whereby effects of greater magnitude were considered stronger evidence than evidence of smaller effects. Relatedly, the committee looked for evidence of a dose–response relationship, meaning that increases in health risk correspond with increases in exposure. Across multiple studies, the committee considered the consistency of an observed association—for example, the replication of findings across multiple studies, especially those with different designs or populations, or those conducted by different investigators. Evidence from multiple epidemiological studies would provide the strongest evidence of a consistent effect.
The committee took into account several considerations of particular importance to assessing observational studies, including temporality and specificity. Establishing a temporal relationship, or that e-cigarette exposure occurred before the outcome was particularly relevant for effects on combustible tobacco cigarette smoking initiation, where reverse causation is plausible; temporality is less relevant for health effects because the likelihood that a disease endpoint or even an intermediate outcome would cause an individual to use cigarettes is unlikely. Establishing temporality is especially important in assessing observational data; longitudinal studies with multiple follow-up periods provide the strongest evidence of temporal precedence, whereas cross-sectional studies are considered weaker because they cannot exclude the possibility of reverse causation. Specificity of observed relationships describes whether the association was unique to e-cigarette exposure. For observational studies, statistically controlling for potential confounders could increase confidence in a
specific effect. For RCTs, because both known and unknown confounders are randomized, they would affect all trial arms equally.
Finally, the committee looked for coherence across the body of evidence. For example, the committee draws analogies from exposure to e-liquid constituents as well as other tobacco products. The committee also uses animal and in vitro data as well as evidence on intermediate outcomes to establish the biological plausibility of a hypothesized disease pathway. Evidence of effects from animal and in vitro populations that were similar to and in the same direction as observed effects in human populations would be coherent with human studies. Therefore, if such research provided evidence of potential mechanisms or otherwise supporting biological plausibility, the committee considered it to bolster in vivo animal or epidemiological studies. However, animal or in vitro evi-
dence was not necessary to draw causal conclusions. Additionally, the committee did not consider null, mixed, or negative in vitro findings to downgrade findings from robust, high-quality animal or human studies.
Additional considerations regarding these factors specific to organ systems or smoking transitions are discussed in the relevant chapters of Sections II and III.
CONCLUSIONS
Informed by reports of previous Institute of Medicine and National Academies committees (IOM, 2011a, 2016; NASEM, 2017), the committee developed standardized language to categorize the weight of evidence as described in the committee’s conclusions. Box 2-1 presents the conclusion categories and describes the types of evidence that correspond to each conclusion category. Stronger evidence implies that observed associations between e-cigarette use and a given outcome are more likely to be causal, whereas weaker evidence is less supportive of causality. Of note, conclusions of moderate, substantial, conclusive, or limited evidence describe a direction of effect (i.e., increased risk of a health outcome), whereas conclusions of no available or insufficient evidence do not imply a direction. The level of evidence does not indicate the size, magnitude, or importance of the effect. The committee notes that the framework is a guide, but that a great deal of expert judgment—in the evaluation of individual studies and in bodies of evidence—is always involved.
REFERENCES
CRD (Centre for Reviews and Dissemination). 2009. Systematic reviews: CRD’s guidance for undertaking reviews in health care. https://www.york.ac.uk/crd/SysRev/!SSL!/WebHelp/SysRev3.htm (accessed November 27, 2017).
HHS (U.S. Department of Health and Human Services). 2014. The health consequences of smoking—50 years of progress: A report of the Surgeon General. Atlanta, GA: U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health.
Higgins, J. P. T., and S. Green, eds. 2011. Cochrane handbook for systematic reviews of interventions version 5.1.0. The Cochrane Collaboration. http://www.handbook.-5-1-cochrane.org (accessed November 27, 2017).
Hill, A. B. 1965. The environment and disease: Association or causation? Proceedings of the Royal Society of Medicine 58:295–300.
IOM (Institute of Medicine). 2011a. Adverse effects of vaccines: Evidence and causality. Washington, DC: The National Academies Press.
IOM. 2011b. Finding what works in health care: Standards for systematic reviews. Washington, DC: The National Academies Press.
IOM. 2016. Veterans and Agent Orange: Update 2014. Washington, DC: The National Academies Press.
NASEM (National Academies of Sciences, Engineering, and Medicine). 2017. The health effects of cannabis and cannabinoids: The current state of evidence and recommendations for research. Washington, DC: The National Academies Press.
NRC (National Research Council). 2007. Toxicity testing in the 21st century: A vision and a strategy. Washington, DC: The National Academies Press.
NRC. 2014. Review of EPA’s Integrated Risk Information System (IRIS) process. Washington, DC: The National Academies Press.
OHAT (National Toxicology Program Office of Health Assessment and Translation). 2015. Handbook for conducting a literature-based health assessment using OHAT approach for systematic review and evidence integration. https://ntp.niehs.nih.gov/ntp/ohat/pubs/handbookjan2015_508.pdf (accessed November 27, 2017).
Sena, E. S., G. L. Currie, S. K. McCann, M. R. Macleod, and D. W. Howells. 2014. Systematic reviews and meta-analysis of preclinical studies: Why perform them and how to appraise them critically. Journal of Cerebral Blood Flow and Metabolism 34(5):737–742.
Whiting, P., J. Savovic, J. P. Higgins, D. M. Caldwell, B. C. Reeves, B. Shea, P. Davies, J. Kleijnen, R. Churchill, and R. Group. 2016. ROBIS: A new tool to assess risk of bias in systematic reviews was developed. Journal of Clinical Epidemiology 69:225–234.










