The bottleneck in effectively treating neurological and neurodegenerative diseases has been in delivery of drugs to the central nervous system (CNS), said Robert Thorne. There are currently three approved biologics that are delivered into the brain or spinal cord, he said. These are ziconotide, a peptide derived from a cone snail toxin that acts on calcium channels to relieve pain; nusinersen, an antisense oligonucleotide to treat infantile spinal muscular atrophy; and cerliponase alfa, an enzyme-replacement therapy for Batten disease, a rare inherited CNS disorder in children. All of these therapeutics are administered directly into the cerebrospinal fluid (CSF) either by injecting them directly into the lumbosacral subarachnoid space or into the ventricular space in the brain. By better understanding how these biologics are working, improvements may be possible that increase their effectiveness and better facilitate their delivery, said Thorne. Billions of dollars have also been spent on trials of immunotherapy drugs delivered systemically, but these trials have yielded disappointing results so far, said Thorne. Thus, he said the field is starting to appreciate that perhaps these antibodies are not reaching their appropriate targets within the CNS and that alternative delivery routes need to be explored further. Andrew Welchman of Wellcome concurred, noting that it is not enough to say that a treatment does not work—you have to know why. Quantifying how well the therapeutics have been delivered is thus very important but not well understood.
Delivering therapeutic agents to the brain by crossing the blood–brain barrier (BBB) can be accomplished in several different ways, according to Eric Schaeffer of Janssen Research & Development. One of the most active areas in industry uses “Trojan horses” (Pardridge, 2006) to ferry cargo across the brain capillary endothelium by engaging receptors on the endothelial cell surface, such as the transferrin or insulin receptor, according to Schaeffer. Viruses have also devised ways to cross the BBB, and thus they provide a powerful means of delivering therapeutic molecules to the brain, said Schaeffer. The BBB can also be temporarily disrupted with externally applied stimulation, such as ultrasound, he said. Finally, he described an approach that bypasses the BBB by delivering molecules intranasally or intrathecally. Developing any of these approaches into effective therapies for brain diseases will require technological advances in several areas, including the development of in vitro high-throughput screening assays, said Schaeffer. These approaches for traversing the BBB to deliver therapeutics to the CNS are addressed in more detail in the next sections.
CROSSING THE BBB
Several methods and technologies are being studied and implemented to deliver drugs across the BBB. Some methods exploit innate biological mechanisms in the BBB, while others employ external disruption or engineered transport of therapeutics.
The Trojan Horse Approach
According to Frank Walsh, the characteristics required of the shuttle in the Trojan horse approach include rapid uptake and efficient transfer of the cargo into the brain tissue, potency in therapeutic doses, ability to work with different types of cargo (such as antibodies or enzymes), safety, and translatability across rodent and human species. Many different transporters are in development as shuttles, including the transferrin receptor, the insulin receptor, and the low-density lipoprotein receptor-related protein 1 (LRP-1), said Walsh. These receptors enable transport of cargo to the brain tissue via a process called receptor-mediated transcytosis (Jones and Shusta, 2007). Through genetic engineering, antibodies or peptides that bind to these receptors are linked to the therapeutic molecule of interest, creating a fusion molecule capable of rapid brain uptake and delivery of the therapeutic molecule. Walsh’s company, Ossianix, for example, isolated cross-species binders to the transferrin receptor from synthetic libraries derived from primordial single-domain shark antibodies. These anti-transferrin receptor 1 variable new antigen receptors (anti-TfR1 VNARs) provided rapid, robust, and prolonged uptake into the brain tissue and neurons at therapeutic doses, said Walsh. As shown in Figure 3-1, this fusion protein bound to the monoclonal antibody rituximab enabled high, rapid transfer of the drug to the brain, as well as persistence for as long as 6 days. Balu Chakravarthy presented data from preclinical studies done with another compound that uses the Trojan horse approach. This therapeutic agent, KAL-ABP-BBB, comprises three components (see Figure 3-2): the “Trojan,” an antibody called FC5, which was derived from camelid and engages the receptor-mediated transcytosis process; an antibody fragment (Fc) that enhances serum half-life; and a therapeutic molecule called amyloid binding peptide (ABP), which is designed to bind to and clear amyloid.
Chakravarthy and colleagues first demonstrated that the fusion molecule is transported across the BBB in vitro. This was followed by studies in mice, which showed that the drug is transferred to the brain in a time- and
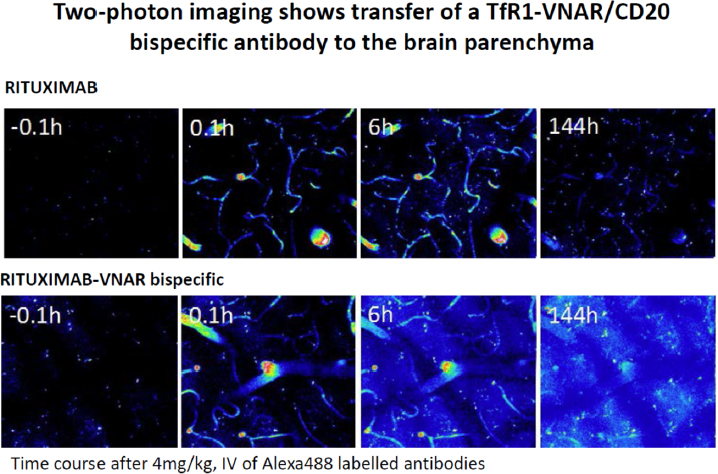
SOURCE: Presentation by Walsh, September 8, 2017.
dose-dependent manner and that it reduces amyloid levels in the cortex and hippocampus. CSF studies showed a similar reduction in amyloid beta (Aβ) levels, suggesting that CSF Aβ levels could be used as a surrogate marker of target engagement.
Chakravarthy and colleagues next repeated these studies in a rat model, which, because of its larger size, allowed them to conduct longitudinal structural and functional imaging studies. These studies showed a significant reduction in amyloid load, an increase in hippocampal volume, restoration of neuron connectivity, and no microhemorrhages. To assess how well the technology translated into larger animals, the researchers next moved to a dog model. Dogs naturally accumulate amyloid beta in their brains as they age and have measurable levels of CSF Aβ. After treatment with KAL-ABP-BBB, the dogs showed a pharmacokinetic profile, CSF to serum ratio, and decreases in CSF Aβ similar to the rat. The next step will be to test the compound in primates, said Chakravarthy.
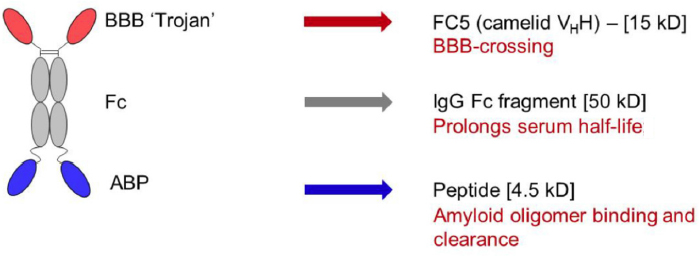
SOURCE: Presentation by Chakravarthy, September 8, 2017.
Ruben Boado, cofounder of ArmaGen, described two Trojan horse therapeutics designed to treat mucopolysaccharidosis (MPS) types I and II, rare genetic diseases caused by deficiency of lysosomal enzymes that break down polysaccharide molecules called glycosaminoglycans (GAGs). Buildup of GAGs causes progressive multisystem dysfunction in children (Neufield and Muenzer, 2001). The most severe form of MPS I, known as Hurler syndrome, leads to profound neurocognitive decline (Shapiro et al., 2015). Patients with MPS II, also known as Hunter syndrome, may also develop neurological symptoms, including cognitive impairment (Whiteman and Kimura, 2017). Enzyme replacement therapy is the standard treatment. However, these enzymes do not cross the BBB, said Boado, so while they can be beneficial for relief of systemic symptoms they have little or no effect on CNS-related symptoms (Boado and Pardridge, 2017; Boado et al., 2013).
Boado said that for MPS I, ArmaGen generated a protein called AGT-181, which fused the missing enzyme, iduronidase (IDUA), with an antibody that targets the insulin receptor. The fusion protein has dual targeting effects for entry into the brain via the human insulin receptor and the mannose-6-phosphate receptor, which targets peripheral tissues (Boado et al., 2009, 2013). As shown in Figure 3-3, both IDUA and AGT-181 are taken up throughout peripheral tissue, but only the fusion protein enters the brain. Preliminary results from a phase II clinical study indicate that AGT-181 improves shoulder range of motion and reduces GAG buildup throughout the body, said Boado. In the CNS, AGT-181 improved or stabilized test scores across multiple cognitive domains and reduced
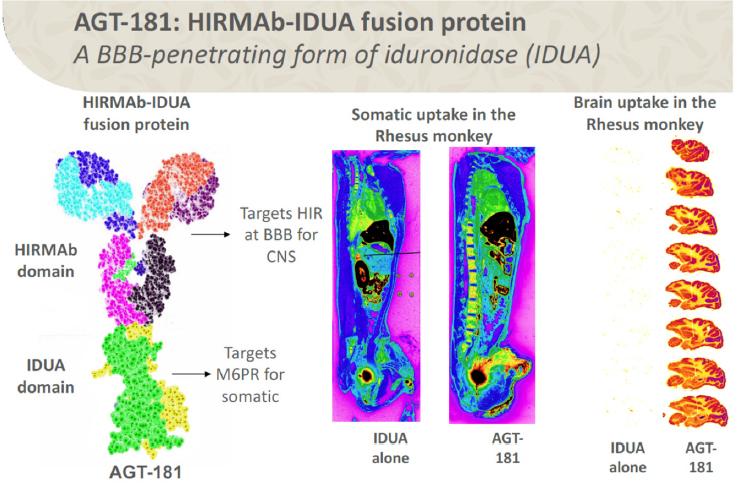
SOURCES: Presentation by Boado, September 8, 2017. Reprinted with permission from Boado, R. J., and Pardridge, W. M. 2017. Brain and Organ Uptake in the Rhesus Monkey in Vivo of Recombinant Iduronidase Compared to an Insulin Receptor Antibody–Iduronidase Fusion Protein. Molecular Pharmaceutics 14(4):1271–1277. Copyright 2017 American Chemical Society.
brain atrophy, he said. He added that there is also evidence that the compound induces immune tolerance to IDUA, in contrast to treatment with enzyme replacement therapy, which can sometimes elicit a severe immune reaction. ArmaGen is also testing a similar compound for the treatment of MPS II, with similar results in early clinical trials, said Boado.
Cells and Exosomes: Harnessing Inflammatory Processes
A different approach to traversing the BBB uses immune cells, such as monocytes and macrophages, which are capable of infiltrating the brain in neuroinflammatory diseases such as Parkinson’s disease (PD). Alexander Kabanov, Mescal Swaim Ferguson Distinguished Professor at
the University of North Carolina at Chapel Hill, said that this technique was originally used to deliver antiretroviral therapy to the brain in a rodent model of human immunodeficiency virus (HIV)-associated brain disease (Dou et al., 2009). His lab took this approach further by using macrophages to deliver therapeutic “nanozymes” (small artificial enzymes) to inflammatory sites in the brains of PD mouse models (Zhao et al., 2011), reasoning that antioxidant enzymes would suppress the inflammatory response and protect neurons. Indeed, neuroinflammation was reduced, and neuroprotective responses increased, said Kabanov (Brynskikh et al., 2010).
Kabanov and colleagues have also shown that genetically modified macrophages can be used to transfer therapeutic genes into the brain (Haney et al., 2013). They transfected macrophages with the gene for glial cell derived neurotrophic factor and showed that these cells produced anti-inflammatory, neuroprotective, and improved behavioral effects in a PD mouse model (Zhao et al., 2014). They went on to show that the enzymes were packaged into extracellular vesicles, called exosomes, secreted by the macrophages. These exosomes increase the persistence of the enzymes in the blood and facilitate the uptake of the nanozyme by neurons, said Kabanov (Haney et al., 2012). His lab has also shown that the uptake of exosomes is increased in the presence of brain inflammation, and that uptake is mediated, at least in part, by a protein on the exosome surface called lymphocyte function-associated antigen 1 (LFA-1), which binds to an intercellular adhesion molecule 1 (ICAM-1) on the surface of endothelial cells (Yuan et al., 2017). Kabanov suggested that understanding these mechanisms more fully may enable development of additional strategies to deliver to the brain therapeutic proteins, as well as DNA, small interfering RNA (siRNA), and messenger RNA (mRNA).
Viral Vectors
Viruses provide yet another versatile and powerful strategy to traverse the BBB and deliver proteins and genes to the brain, said Viviana Gradinaru. Adenoassociated viruses (AAVs) have been used in clinical trials to safely deliver therapeutic genes, via direct injection, to the brains of people with PD, which she said suggested the feasibility of this approach (Kaplitt et al., 2007). Gradinaru, however, said she wanted to use AAV as a vector to deliver multicolored gene labels to the brain as a way of mapping brain circuits, and she wanted to introduce these viral vectors systemically, which would require traversing the BBB. A paper published
in 2009 showed that AAV9, when injected intravenously into neonatal mice, efficiently transduced neurons throughout the brain, although the efficiency was greatly reduced in adult mice (Foust et al., 2009). To overcome this roadblock, she and her team created libraries of AAV capsids (the outer coat of the viral particle) with different characteristics, and then selected those that made it through the BBB and efficiently transduced neurons and astrocytes in the brain (Deverman et al., 2016) (see Figure 3-4). Gradinaru said that by using different gene regulatory elements, it is also possible to target specific cells or regions of the brain for gene delivery (Chan et al., 2017).
An important unresolved question is whether gene therapy vectors will behave differently when confronted with a compromised BBB, such as in multiple sclerosis, inflammation, or brain tumors, said Gradinaru. She noted that a compromised BBB is not necessarily leakier because of fibrous tissue that could prevent the passage of small molecules. This will require more testing, she said.
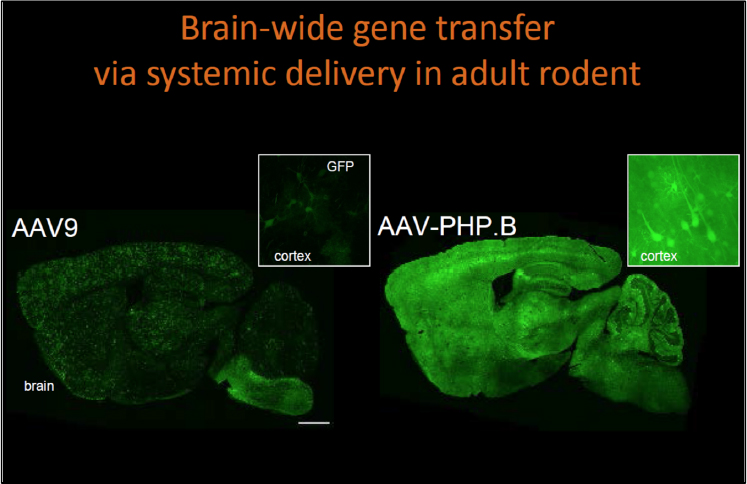
SOURCE: Presentation by Gradinaru, September 8, 2017.
Focused Ultrasound
Focused ultrasound is yet another approach that has shown promise in disrupting the BBB to facilitate drug entry into the brain, said Alexandra Golby. As a neurosurgeon specializing in brain tumor surgery, Golby became interested in this technology because of the urgent need to improve treatments for brain tumors, which are difficult to resect owing to their infiltrative nature. And even though brain tumors may be associated with a disrupted BBB, they are less permeable to drugs, which limits efficacy, said Golby (Lockman et al., 2010).
While questions remain about the exact mechanisms by which focused ultrasound disrupts the BBB, Golby said the effects appear to be largely facilitated by microbubbles creating oscillations and vibrations that enhance the permeability of the brain-tumor barrier (Park et al., 2017). She noted that BBB disruption can occur in the absence of microbubbles; the microbubbles facilitate this and allow it to happen at lower power, thus minimizing damage.
The microbubbles may also carry a payload into the tumor, including chemotherapy agents, antibodies, nanoparticles, neuroprotective agents, and viruses, she added. The delivery of these agents has been advanced through the efforts of Nathan McDannold and others, who have fine tuned the magnitude of the BBB opening and the restoration of its closure, said Golby (Marty et al., 2012; Park et al., 2012). Indeed, she said it may be advantageous to close the BBB relatively quickly to protect the brain from toxins.
Golby described preclinical studies in a rat glioma model that showed that focused ultrasound increased both drug delivery and retention (Park et al., 2017) and improved survival (Aryal et al., 2013). Studies in macaque monkeys further showed that focused ultrasound can be targeted to open the BBB in a restricted area without causing tissue damage or functional impairment, she said (McDannold et al., 2012).
An Israeli company, Insightec, has received Food and Drug Administration (FDA) approval for a focused ultrasound device to do thermal ablations for movement disorders, said Golby. She added that the device can readily perform BBB opening as well. However, infrastructure requirements for the procedure are high: presently this procedure must be performed in an MRI scanner with a stereotactic head frame and a water bath for ultrasound transmission and cooling the scalp. Preoperative imaging is
also needed to determine the exact bone thickness so the beams can be focused accurately. Golby suggested that for some indications, unfocused ultrasound may be useful, although this approach is less well developed.
BYPASSING THE BBB
While many mechanisms for traversing the BBB are under development, researchers seeking improved drug delivery to the CNS are also investigating methods that bypass the BBB altogether.
Intrathecal Injection
Intrathecal infusions can effectively deliver drugs to the subarachnoid space; however, since there is little penetration beyond the brain surface their use has been limited, according to Golby. Thorne said he has been trying to overcome problems with distribution of therapeutic molecules, such as antibodies. One approach Thorne has taken, in collaboration with Danica Stanimirovic, is to use single-domain antibodies, which are able to diffuse more readily in the brain. They have also tried CSF co-infusions of therapeutic molecules with substances such as mannitol that enhance distribution by increasing the access of some therapeutics to the perivascular spaces, leading to deeper brain penetration and more widespread distribution overall within the CNS.
Intranasal Delivery
Thorne has also explored using intranasal delivery to increase distribution of therapeutic antibodies throughout the CNS. He noted that many therapeutics (such as calcitonin for osteoporosis) are already approved and available that use intranasal administration to achieve systemic distribution since the nasal passages are highly vascularized. But his research has done much to expand further on how the intranasal route may be used to target the CNS specifically. He and others have shown in rodent models that intranasally delivered molecules are transported across the nasal epithelia to reach brain entry pathways associated with the olfactory and trigeminal nerves in the nasal mucosa, bypassing the BBB to achieve higher levels of macromolecules (e.g., labeled dextrans, antibodies, and antibody fragments) in perivascular spaces compared to intra-arterial administration (Lochhead et al., 2015). However, the olfactory mucosa in humans covers
a much smaller area than in rodents, so further testing in primates and humans is necessary to determine the feasibility of this approach. Thorne and colleagues have tested intranasal administration of beta interferon—a drug approved for relapsing remitting multiple sclerosis—in cynomolgus monkeys and showed “fairly widespread distribution” with localization to appropriate brain regions (i.e., the basal ganglia); however, he acknowledged that this effect may not be completely generalizable so it will likely be necessary to examine on a case-by-case basis the distribution and CNS target concentrations of other drugs administered intranasally (Thorne et al., 2008). The intranasal route has also been used to administer exosomes, said Kabanov.
MODELING THE BBB
Development of novel biological and computational models of the BBB can contribute to facilitating translational research and understanding the dynamics of therapeutics once they are delivered to their nervous system target.
Spheroids as In Vitro Screening Tools
In vitro models of the BBB facilitate the discovery and development of new drugs to treat CNS diseases, said Choi-Fong Cho. Cho described a new platform for drug discovery using multicellular spheroids composed of all the important cells of the BBB: astrocytes in the core surrounded by pericytes and endothelial cells on the surface (Cho et al., 2017). She said these spheroids are extremely cost-effective and easily reproducible. Because they are created from human endothelial cells, they can provide extra support for moving a preclinical program into human clinical trials, she said. They are characterized by the expression of tight junctions and the function of efflux pumps, and certain receptors (such as the LRP-1 receptor) can mediate transcytosis of their ligands from the surface to the interior of the spheroid, enabling screening and prediction of molecules that can cross the BBB, she said. The platform is a high-throughput tool for studying molecular movement from the surface to the interior, enabling screening for molecules that can cross the BBB, Cho added.
Her lab has also used the model to test for BBB penetrating compounds. Compared to the commonly used transwell models of the BBB (Hatherell et al., 2011), the spheroid model is superior at reproducing and
maintaining essential BBB properties and functions, said Cho. Her research showed a high level of angiopep-2 transport through their transwell insert over time, but the transwell insert also saw high penetration of the control (scrambled) peptide (Cho et al., 2017), highlighting the inherently leaky nature of the transwell system. The leakiness of the transwell system has been a long-standing problem with the model, and she said their findings are in line with previously published reports. Eric Schaeffer commented that other labs have reported good results using the transwell model and suggested that understanding the differences between these systems could provide valuable insight. Steven Hyman suggested that using human-induced pluripotent stem cells could avoid potential problems associated with immortalized cells.
Translational Pharmacokinetics Models
Mathematical models may also be useful to translate pharmacokinetics data from animal studies and predict how a compound will behave in humans, according to Dhaval Shah, assistant professor in the Department of Pharmaceutical Sciences at the State University of New York at Buffalo. His rationale for this approach is based on two beliefs: first, that molecules will behave similarly in humans and animals, and second, that the fundamental behavior of a system can be characterized mathematically. He suggested that the decision-making process for clinical translation would become more reliable by relying on mathematical models rather than one’s hunch or gut feeling.
Shah and colleagues performed microdialysis experiments in rats, combined with sensitive enzyme-linked immunosorbent assay (ELISA) assays to understand and quantify the main determinants for protein therapeutics disposition, such as antibody-drug conjugates (Khot et al., 2017). They then used these data to develop a translational mathematical model. This model suggests, for example, that the brain–cerebrospinal fluid barrier may play an important role in transferring antibody molecules into the CSF, and that antibody concentrations vary among different compartments of the CSF circulatory system. Shah suggested that better understanding this process could help drug developers better understand how much drug is actually reaching targets in the brain and predict appropriate dosing.












