1
Trends in Mortality, Disease, and Physiological Status in the Older Population
INTRODUCTION
Mortality and health trends in the last two to three decades relevant to the older population are the focus of this chapter. The emphasis is on research reporting trends relevant to the older population since 1980 in mortality, disease prevalence and incidence, and underlying physiological status. Trends in disability and functioning are treated in the chapter by V. Freedman.
In recent years there have been a number of disquieting trends, along with some positive changes, in mortality, life expectancy, disease presence, and physiological status in the United States. This chapter discusses those trends and how they might be explained both more narrowly in terms of proximate causes and more broadly by changes in American society. Most of the research on national trends reports trends in averages for the population or for significant subgroups of the population. In addition, this chapter highlights new theoretical approaches and data that are now becoming available for the study of mortality and health in aging individuals and that are likely to be increasingly incorporated into mortality and health outcome research in the coming decade. These approaches are aimed at explaining variability in individual aging at a more basic biological level; our belief is that future research on mortality risk and health change among aging individuals is likely to increasingly incorporate these mechanisms in the coming decades to explain differences among subgroups and changes over time.
RECENT TRENDS IN MORTALITY
Life expectancy continues to increase in the United States but at different rates for Black and White men and women (see Figure 1-1). Increases from 1980 to 2013 were relatively slow for White women, rising from 78.1 years to 81.4 years for an increase of 3.3 years over the 33-year period, or 1.2 months per year (Figure 1-1). For Black women, life expectancy increased from 72.5 years to 78.4 years, for an increase of 5.9 years over 33 years or 2.1 months per year. This was very similar to the increase among White men, from 70.7 years to 76.7, an increase of 6.0 years or 2.2 months per year. The greatest increase was experienced by Black men, rising from 63.8 years to 72.3 years, an increase of 8.5 years over the period or 3.1 months per year. So among these four population subgroups, White women have had the slowest increase in mortality over this period.
Whereas life expectancy for the general population had been increasing fairly steadily over recent decades, the last 2 years have seen a change. In December 2017, the Centers for Disease Control and Prevention reported a slight decline in life expectancy (0.1 year) for 2016, which followed a similar decline from 2014 to 2015 (Kochanek et al., 2017).
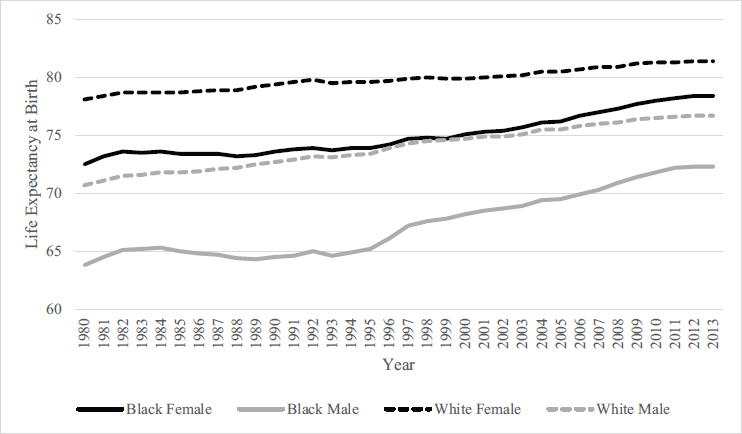
SOURCE: Chong et al. (2015).
Changing Differentials in Life Expectancy in the United States
Changes in mortality differentials between population subgroups have been substantial in recent years. The differential changes in life expectancy reported above result in narrower differences by gender and race at the end of the 33-year period shown in Figure 1-1. The difference in life expectancy at birth between White men and women declined from 7.4 years longer lives for women in 1980 to 4.7 years in 2013; for Black men and women, the greater life expectancy of females decreased from 8.7 to 6.1 years. As indicated above, life expectancy continued to increase for both Black and White women between 1980 and 2013, but for White women life expectancy increased only 1.2 months a year, while for Black men it increased 3.1 months a year. This resulted in reductions between 1980 and 2013 in life expectancy differentials by race at birth from 5.6 to 3.0 years for White versus Black women and from 6.9 to 4.4 years for White versus Black men—reductions over the 33 years of 46 percent for women and 36 percent for men. Results continue to confirm that Hispanic mortality is lower than African American mortality and fairly comparable to that of non-Hispanic Whites (Hummer and Hayward, 2015).
There is significant evidence that differentials in life expectancy by socioeconomic status (SES) have gotten wider in recent years (Olshansky et al., 2012). Meara et al. (2008) noted that the SES gap, as indicated by educational differentials, increased by about 30 percent between 1980 and 2001; this gap has continued to grow through 2010 (Sasson, 2016a, 2016b). Hayward and colleagues suggested that the shape of the association between educational attainment and U.S. adult life expectancy has changed over time, with mortality decline being concentrated among the most highly educated (Hayward et al., 2015; Montez et al., 2012).
Mortality trends by education have differed by gender. Mortality decline has occurred among men at almost all educational levels since 1980, but among women mortality has only declined for the highly educated. For women without a high school education, mortality actually increased during some periods (Montez et al., 2011; Sasson, 2016a). Figure 1-2 shows changes in life expectancy for four educational groups and four race-by-gender groups over a 20-year period. While a decrease in life expectancy only occurred among White women in the lowest educational group, in each of the other three educational groups White women experienced the smallest increases. On the other hand, the increases in life expectancy were substantial among all educational groups for Black men, although there was an increasing educational differential with increase in educational level even for Black men. Adding to the evidence of increasing life expectancy differentials by increasing SES, Chetty et al. (2016) have provided analysis by income levels that shows that higher income was associated with
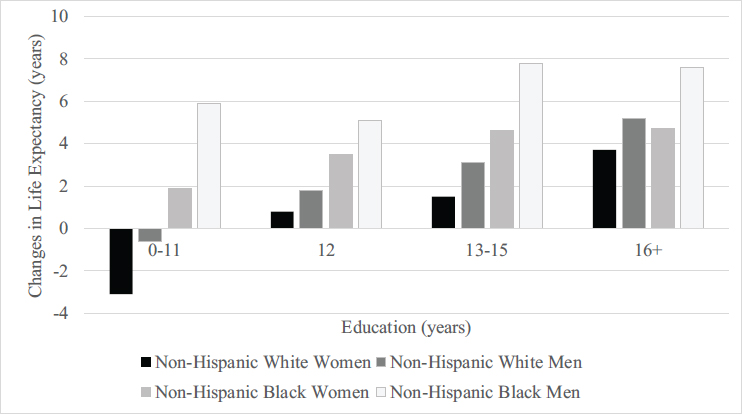
SOURCE: Data from Sasson (2016b).
greater longevity and that the differences in life expectancy across income groups increased from 2000 to 2014. Whereas different authors provide different estimates of the changes in life expectancy by SES group, there is no disagreement on the fact that life expectancy differentials by SES have increased in recent decades (Bound et al., 2015; Montez et al., 2016a).
Another recent focus of research on mortality in the United States has been on geographic differentials in life expectancy; this research has produced evidence of large spatial differentials across the country, as well as varying patterns of changes in these differentials. Murray et al. (2006) described life expectancy differentials in what they called “eight Americas”: eight groups of counties divided by location, density, race, income, and homicide rate. They found life expectancy differences that ranged up to 35 years across these groups and reported that between 1982 and 2001, the ordering of life expectancy among the eight Americas and the absolute difference between the advantaged and disadvantaged groups remained largely unchanged (Murray et al., 2006). Ezzati et al. (2008) pointed out that in many counties mortality actually increased for some groups during the last two decades of the 20th century. Chetty et al. (2016) examined state differences and changes in life expectancy and reported that for individuals with low incomes, life expectancy was lowest in Indiana, Nevada, and Oklahoma and highest in California, New York, and Vermont. They also reported, “the gap in life expectancy between the lowest and highest income quartiles decreased in some areas, such as areas within New Jersey
and Alabama, but increased by more than 3 years in other areas, such as areas within Florida” (Chetty et al., 2016, p. 20).
Explanations for Trends and Changing Differentials
Explanations for differences and trends in life expectancy can be done at many levels. There are proximate explanations in terms of composition, such as changing age patterns and causes of death—that is, which causes are increasing or decreasing. There are explanations in terms of changes or differences in risk factors for disease or death, such as smoking or obesity. Differences in health care availability and treatment offer another explanation. Then, there are larger-level policy-related factors and fundamental social causes, which may provide an even more upstream level of explanation (Woolf and Braveman, 2011).
Deaths from misuse of opioids have been noted as an increasingly important factor influencing the overall trend in life expectancy, especially for the White population (Case and Deaton, 2015; Ho, 2017). Ho (2017) has shown that mortality from drug overdose increased for men and women in all education groups, but more so for those with low education, and more so for non-Hispanic Whites. Her assessment is that mortality from drug overdose accounts for a sizable proportion of the increases in educational gradients in life expectancy, particularly at the prime adult ages (ages 30–60), where she estimates it accounts for 25 to 100 percent of the widening in educational gradients between 1992 and 2011 (Ho, 2017). The fact that opioid deaths occur at relatively young adult ages has been an important factor in overall life expectancy trends. The very recent decrease in life expectancy for the general population appears to reflect adverse changes in most of the major causes of mortality but with women experiencing more deterioration due to chronic diseases than men (Acciai and Firebaugh, 2017).
The decrease in the life expectancy differential by race can be attributed to the relative rate of decrease in mortality from homicide, HIV, unintentional injuries, diabetes, and heart disease (Harper et al., 2007, 2014). In recent years, the greater increase among Whites than Blacks in deaths from unintentional poisonings including drugs has reduced the race differential (Harper et al., 2012).
Meara et al. (2008) and Sasson (2016b) have emphasized a role for changes in smoking behavior as an explanation for the growing inequality by SES. Ho and Fenelon (2015) estimated that smoking accounts for half of the increase in the mortality gap by education between the 1980s and 2006 for White women but does not explain the increasing gap for White men. Smoking behavior is important in the relatively low mortality of Hispanics (Lariscy
et al., 2015). Montez and Zajacova (2013a) have also associated smoking and other behaviors to the poor trends among low-education women.
Race differences in life expectancy and mortality are generally assumed to primarily reflect SES composition of the population. A recent estimate is that socioeconomic and demographic characteristics account for 80 percent of the gap in life expectancy between Black and White males and 70 percent of the gap between Black and White females (Geruso, 2012). Recent work has suggested that better specification of SES might result in even fuller explanation (Do et al., 2012). In the case of Hispanics, the relatively low mortality of Hispanics tends to be confined to those of lower SES (Turra and Goldman, 2007) and the important compositional factor is that a large number are foreign born (Lariscy et al., 2015).
Avendano and Kawachi (2014) have noted that life expectancy is particularly low for regions in the U.S. South and Midwest. Examination of county differences in death rates by cause has found that regions differ in the causes of relatively high mortality. Cardiovascular-related mortality is higher among counties that border the southern half of the Mississippi River. Mortality rates from self-harm and violence were higher in counties in the Southwest, and mortality rates from chronic respiratory disease were highest in counties in eastern Kentucky and western West Virginia (Dwyer-Lindgren et al., 2016). Chetty et al. (2016) reported that most of the variation in life expectancy across geographic areas was related to differences in health behaviors, including smoking, obesity, and exercise. In examining county-level changes in mortality up to 2000, Ezzati et al. (2008) ascribed increases in county-level female mortality to increases in chronic diseases related to smoking, overweight and obesity, and high blood pressure.
Researchers who have examined a variety of differentials and trends return to fundamental social causes as the underlying explanation for multiple differentials and trends (Phelan and Link, 2015). Montez and colleagues examined trends by gender and differences by state and concluded that these patterns resulted from more fundamental social issues such as the lack of good paying jobs for women with limited education and the lack of cohesion resulting from an adverse policy environment (Montez and Zajacova, 2013a, 2013b; Montez et al., 2016b). Murray et al. (2006) concluded that county-level differences in life expectancy are not fully explained by race, income, or basic health care access and utilization, and they suggested broader underlying causes. Phelan and Link (2015) suggested that racism is a fundamental cause of the persistent difference in life expectancy between Blacks and Whites.
Trends in U.S. Life Expectancy Relative to Other Countries
There has also been significant research in recent years on how mortality rates and life expectancies in the United States compare to those of other countries (Crimmins et al., 2010a; National Research Council, 2011). This stream of work has tried to gain perspective on why the United States has the levels and trends in mortality that have been observed. The poor performance of the United States relative to peer countries has been noted in two National Research Council and Institute of Medicine studies (National Research Council, 2011; Institute of Medicine and National Research Council, 2013). The focus of the first study was the population age 50 and older; the focus of the second was on younger ages. Figure 1-3 extends and expands the comparisons shown in those studies to show the relative trend in life expectancy at birth from 1980 to 2015 for the 40 countries in the world with the highest life expectancy in 2015. The dark dashed line in the figure shows the continued relative decline of life expectancy in the United States relative to the other 39 countries. The United States falls from ranking 20th in 1980 to ranking 40th in 2015. The countries ranking from 30 to 39 in 2015 included Bermuda, the Channel Islands, Cyprus, Lebanon, Costa Rica, Puerto Rico, Cuba, Czech Republic, Brunei, and
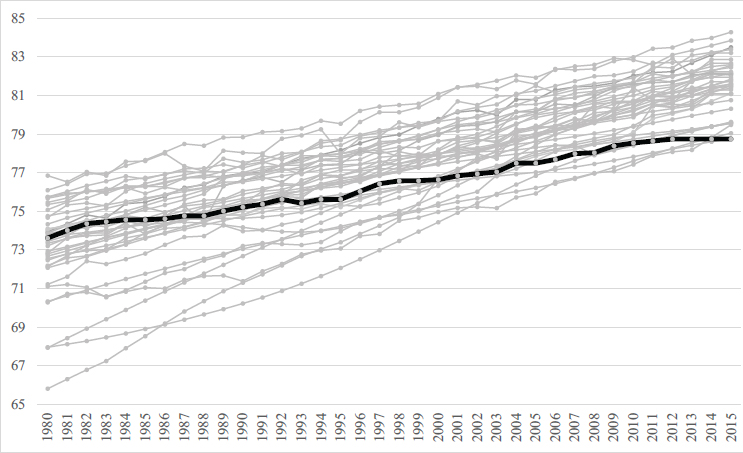
SOURCE: The World Bank Life Expectancy at Birth. Available: https://data.worldbank.org/indicator/SP.DYN.LE00.IN [April 2018].
Qatar. In 1980, most of these countries had lower life expectancies than the United States.
The poor performance of the United States in life expectancy is due to many conditions and diseases (Crimmins et al., 2010a; Glei et al., 2010; Institute of Medicine and National Research Council, 2013). It is worse at younger than older ages (Ho and Preston, 2010; Ho, 2013) and is not explained by racial/ethnic differences, as it characterizes the non-Hispanic White population as well as the general population (National Research Council, 2011). Potential explanations examined include individual behaviors, social integration and inequality, health care availability, environmental factors, policies, and social values (National Research Council, 2011; Institute of Medicine and National Research Council, 2013). Explanations differ markedly by age. For the older population, individual behaviors were a major contributor to the overall pattern and health care availability was not as important. Cohort patterns of both smoking and obesity play a major role in explaining the relative position of the U.S. older population (National Research Council, 2011; Preston and Stokes, 2011). For the population under age 50, the causes for the higher U.S. mortality are many, and they differ by age. They include infant and childhood conditions, accidents, chronic conditions, and violent deaths, particularly violent deaths related to firearms. At another level of explanation, causation is related to SES and the relatively large effect on mortality of low SES in the United States. Most analyses have concluded that the focus on immediate causes may not be appropriate for differentials that characterize most ages, many causes, and most social groups (Institute of Medicine and National Research Council, 2013; Avendano and Kawachi, 2014). Fundamental social and policy causes may be implicated (Link and Phelan, 1995).
RECENT TRENDS IN OTHER DIMENSIONS OF HEALTH
Changes in population health over the past century are more difficult to characterize succinctly than changes in life span. First of all, health has many dimensions, which do not necessarily change in the same way over time and which are not related to mortality change in the same way (Crimmins et al., 2010b). Crimmins and colleagues have characterized the morbidity process for populations as health change related to aging that begins with the physiological dysregulation indicated by a number of biological risk factors, which is followed by subsequent diagnosis of diseases, loss of functioning and disability, frailty, and death (Crimmins et al., 2010b). This section discusses recent trends in disease prevalence and incidence, as well as changes in physiological status associated with the beginning of the process of morbidity progression. These data are less available than those for mortality, and the varying availability of data results in differences in timing
of the trends and ages considered. The chapter by Freedman in this volume discusses trends in disability, functioning, and frailty—the later stages in morbidity progression.
The prevalence of some diseases has declined in the older population in recent decades. For instance, fewer persons have bronchitis and emphysema; reductions in smoking are assumed to be the cause. On the other hand, the prevalence of diabetes has been increasing steadily since before the 1980s. This trend reflects improved survival among diabetics, as well as increased incidence due to increasing numbers of overweight persons. The prevalence of certain other chronic diseases—cancer, heart disease, and stroke—has also increased from the 1970s through the 1990s (Crimmins, 2015a). This may in part reflect improved diagnosis, but it certainly reflects increased survival among those who have the conditions (Crimmins and Saito, 2000). As we make progress in defeating mortality by delaying death among those who have disease, the prevalence of disease increases in the population. Trends in this century indicate that the prevalence of heart disease and stroke may have declined somewhat after 2000, whereas cancer and diabetes prevalences continue to increase (see Figure 1-4).
Trends in incidence are better indicators than prevalence trends of the current health changes in the population, but data on incidence of health change are not as available. Since 2000, incidence of heart disease and stroke may be beginning to decline, which would explain part of the change
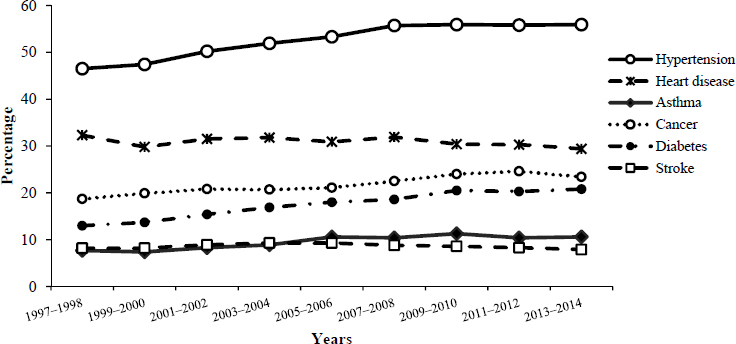
SOURCE: Centers for Disease Control and Prevention. Health, United States, 2015 (Tbl. 17b). Available: https://www.cdc.gov/nchs/data/hus/hus15.pdf [February 2018].
in prevalence. This has been reported from recent trends in incidence of acute myocardial infarction, which indicate that the increasing trend in incidence may have ended around 1990, followed by a period of stability up to 2000, after which there have been marked reductions of 20 to 40 percent (Chen et al., 2010; Rosamond et al., 2012; Talbott et al., 2013; Wang et al., 2012; Yeh et al., 2010). Stroke incidence, indicated by hospitalizations, is also reported to have peaked around 1997 for those age 65 and older and to have now declined somewhat (Fang et al., 2014). Examination of disease onset in two cohorts in the Health and Retirement Study (HRS) showed that those born from 1943 to 1953 experienced a lower rate of heart attack than those who were born in 1931–1941; however, the rates of overall heart disease and stroke did not appear to differ (Crimmins and Levine, 2015). It is possible that we are beginning to see the beginning of reductions in incidence in some conditions that could reflect improvement in innate health of older persons, but more data over a longer period and for the whole country are needed to make this a general conclusion.
The onset of a number of diseases important in old age increase exponentially with age, including the diseases discussed above (St. Sauver et al., 2015). Exponential increase is also a characteristic of loss of physical and cognitive functioning. This has led to growing recognition that aging is a process that often involves multimorbidity and to a belief that the explanations of disease increase and deterioration in functional aging may be common across outcomes (Kennedy et al., 2014). Two-thirds of older persons in the United States have two or more chronic diseases; for those over 85 the fraction is more than 80 percent (Espeland et al., 2017; Salive, 2013). Multimorbidity is more common among those of lower education, those who have experienced financial hardship as a child, and African Americans (Tucker-Seeley et al., 2011; St. Sauver et al., 2015). This might lead to an expectation of different outcomes for any one disease for these groups who differ in overall disease burden. Multimorbidity is also more prevalent in the United States than in other countries (Crimmins et al., 2010a). The incorporation of multimorbidity into research reflects advances in empirical clarity, theoretical developments, and interventional potentials.
TRENDS IN PHYSIOLOGICAL STATUS
Physiological deterioration and dysregulation is an upstream aspect of health that may precede disease diagnosis and mortality and may indicate a stage for initial medical or behavioral intervention in order to delay the progression to morbidity and mortality. Physiological dysregulation has been indicated by markers of the functioning of many systems and organs. Since cardiovascular diseases are among the most important sources of mortality and morbidity, we first examine trends in two markers of car-
diovascular health: blood pressure and cholesterol. We present trends over the longest period we can, more than 60 years, because the consistent pattern of change over this long period indicates that these are very long-term trends. As shown in Figures 1-5 and 1-6, for almost 60 years the United States has experienced remarkable declines in measured blood
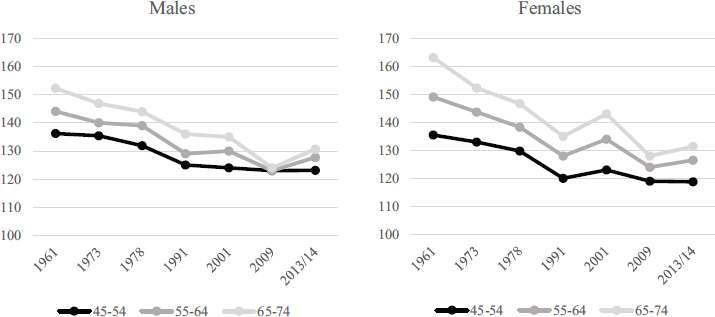
SOURCES: Kumanyika et al. (1998): 1960–1980; National Health and Nutrition Examination Survey: 1998–1994, 1999–2002, 2007–2010, 2013–2014.
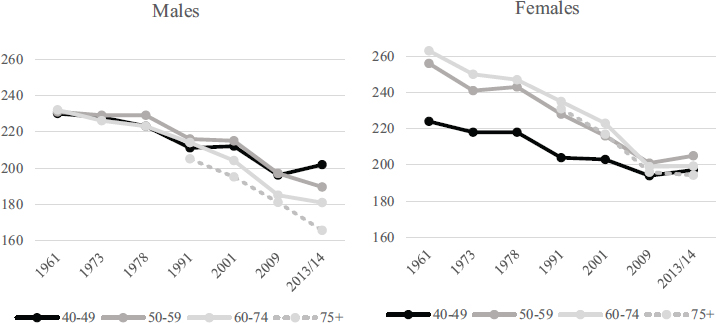
SOURCES: Carroll et al. (2005): 1960–1978; National Health and Nutrition Examination Survey: 1998–1994, 1999–2002, 2007–2010, 2013–2014.
pressure and cholesterol (Crimmins et al., 2010a; Crimmins, 2015a; Ong et al., 2013). The use of effective antihypertensives and statins has reached high levels and is one of the major reasons for the current low levels of hypertension and high cholesterol (Crimmins, 2015a). However, dietary changes, reduced smoking, and perhaps improved early-life development and healthier lives may have also played a role in these declines (Finch and Crimmins, 2004).
While prevalences of high blood pressure and elevated cholesterol have become lower over time, obesity has increased (Alley et al., 2010). There is also some evidence of increase in adverse levels of biomarkers related to obesity and diabetes, such as plasma glucose (Beltrán-Sánchez et al., 2013).
In attempting to summarize change over time in physiological status, researchers have examined summary indicators. The change over time in the average number of cardiovascular-metabolic measures for three dates, spanning almost 20 years from about 1990 to 2010, shows that the average number of risk factors measured at clinically defined high-risk levels has been reduced in the past two decades for ages above 50 years (Crimmins, 2015a). Other researchers have reported small reductions in the prevalence of metabolic syndrome, which includes a set of biomarkers related to metabolism, in the most recent decade (Beltrán-Sánchez et al., 2013). In addition, a recent study of change from around 1990 to 2010 in “biological age” based on eight indicators of physiology has shown that at a given chronological age “biological age” has been reduced, with the largest reductions in the oldest age group (Levine and Crimmins, 2018).
NEW APPROACHES TO INCORPORATING PHYSIOLOGICAL STATUS AND EPIGENETIC MARKERS IN MODELS OF AGING HEALTH OUTCOMES
The discussion above of population health trends has demonstrated the use of population-level data to portray a picture of health differences and trends in the U.S. general population or in subgroups of that population. This work tends to focus on change in the mean or prevalence in the population. However, much of the research effort in recent years reflects a focus on individual “aging” or health changes with age, where the aim is to predict variability in who “ages” later and who “ages” at an earlier chronological age. This work follows individuals over time as they age and links prior risk factors and life circumstances thought to affect the process of aging health change with subsequent health outcomes. Over recent decades the empirical models of individual health change indicating the factors affecting the level of risk have incorporated variables from multiple domains, becoming increasingly multidisciplinary, with increasing explanatory power (Crimmins and Seeman, 2004; Institute of Medicine
and National Research Council, 2013). Most of the population differentials discussed above would be included in a list of social and demographic variables that influence health, but a full explanatory model for most researchers would now also include economic, psychological, policy, and context variables as mediating or moderating variables. In addition, the last two decades have seen much greater incorporation of the biological pathways that explain how social variables “get under the skin” to affect age-related downstream health outcomes such as disease, disability and functioning loss, and death. Figure 1-7 is a heuristic model that includes a selection of the independent variables that represent social, economic, psychological, and policy influences and the growing biological mechanisms posited as being mechanisms that influence “aging.” This model is presented to illustrate the typical multidisciplinary model that is tested in studies of individual aging. This section focuses on the growing interest in the biological mechanisms intervening between social factors and health outcomes, as these mediators are thought to clarify the biological pathways that explain why health changes with age.
An interest in summarizing biological risk, such as the measures mentioned above in summarizing recent change in physiological status, initially developed as a way of incorporating multiple physiological and sometimes behavioral risk factors into a succinct indicator of relative risk for spe-
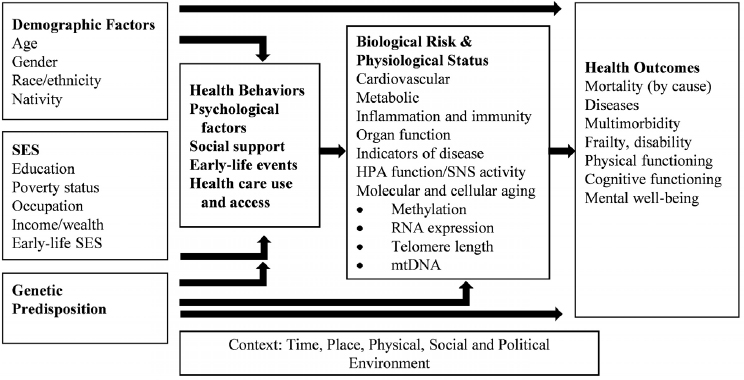
NOTES: Demographic, socioeconomic status earlier in life, and genetic factors are determined prior to health behaviors, psychological factors, current social support, health care use and access. Biological risk and physiological status represent the biological pathways through which the prior variables affect health outcomes.
TABLE 1-1 Summary Indicators of Physiological Dysregulation and Molecular and Cellular Changes with Age
| Multisystem-Variable Physiological Dysregulation with Age | Framingham risk score (Wilson et al., 1998) |
| Allostatic load (Seeman et al., 1997) | |
| Biological age (Levine, 2013) | |
| Pace of aging (Belsky et al., 2015) | |
| Age-related homeostatic dysregulation (Cohen et al., 2015) | |
| Molecular and Cellular Changes with Age | Telomere length (Blackburn et al., 2006) |
| Biological clock–DNA methylation (Horvath, 2013; Hannum et al., 2013; Weidner et al., 2014) | |
| Gene expression–RNA (Holly et al., 2013; Peters et al., 2015) | |
| Proteomics (Menni et al., 2015) | |
| Metabolomics (Menni et al., 2015; Hertel et al., 2016) |
cific health outcomes. This began with the Framingham risk score, which developed an indicator of the 10-year likelihood of having a cardiac event (Wilson et al., 1998) based on both behaviors and clinical indicators of cardiovascular risk (see Table 1-1). Expanding on this approach, Seeman et al. (1997) developed an indicator of allostatic load that reflected the growing belief that multiple systems contribute to the overall level of physiological dysregulation that characterizes aging; this indicator included cardiovascular, metabolic, neuroendocrine, and inflammatory markers of risk. The allostatic load concept emphasized that small changes in a number of systems could result in adverse health outcomes. It also recognized that the challenges to homeostasis increase with age and with deterioration of physiology. Allostatic load has been linked to a number of subsequent health outcomes including loss of functioning ability, cognitive loss, disease, and mortality (Seeman et al., 2001), as well as being associated with SES and race (Geronimus et al., 2006).
The last decade has seen exponential growth in the number of population studies including biological or physiological measures, as well growth in the type of markers included. Many studies now include the individual measures discussed above—for example, blood pressure, cholesterol, measured glucose levels, obesity, and/or inflammation. But a far greater number of indicators are now available in many national and population-representative studies. Recent research has compared the value of these measures in predicting health outcomes to the predictive value of more standard measures and to the value of behavioral and psychological measures (Cornman et al., 2017; Goldman et al., 2016; McClintock et al., 2016). Cross-national research has also demonstrated that biological measures
are context specific and not universal across aging populations (Crimmins, 2015b; Rehkopf et al., 2016).
The increase in measures has encouraged continued refinement and development of summary indicators of physiological aging. Gruenewald et al. (2006) have used a more sophisticated recursive partitioning approach, along with novel markers of aging, in defining the risk of allostatic load. Levine (2013) introduced a measure of biological age based on values of a set of 10 markers of physiological regulation of multiple systems and organs. This measure, which has the desirable characteristic of being in units of age (years), has been shown to change over time (Levine and Crimmins, 2018), to be related to social and behavioral factors, and to partly explain race differences in mortality (Levine and Crimmins, 2014). Using a somewhat similar but expanded approach, Belsky et al. (2015) developed a measure they call “pace of aging,” which has been related to relevant health outcomes and change in those outcomes—for a middle-aged population. Cohen et al. (2015) represent another approach; they combined 43 indicators into a measure they called “age-related homeostatic dysregulation.” Belsky et al. (2015) compared “biological age,” the “pace of aging,” and “age-related homeostatic dysregulation” in the Dunedin, New Zealand, middle-aged population and found that these measures have a correlation of 0.4–0.6, which indicates they are related but not the same measure.
The last few years have brought the development of measures of biological aging at the molecular and cellular level (see lower half of Table 1-1). Telomere length has been suggested as a summary measure of biological aging and could reflect all of the types of systems in the physiological measures (Blackburn et al., 2006). In practice, telomere length has been inconsistently related to outcomes of aging (Blackburn et al., 2015; Glei et al., 2016; Mather et al., 2011). The National Institute on Aging and the National Institute of Environmental Health Sciences recently held a workshop to try to understand some of the confusion surrounding telomere work in populations (National Institutes of Health, 2017).
Summary measures built with molecular and cellular markers could reflect change that is prior to some of the physiological and clinical measures contained in the above physiological measures. This would include epigenetic markers of DNA methylation as well as RNA expression. To date, three “biological clocks” have been defined, based on DNA methylation (Horvath, 2013; Hannum et al., 2013; Weidner et al., 2014). These clocks are based on different numbers of largely overlapping markers, and their values are related to each other in the study of the middle-aged population in Dunedin, New Zealand (Belsky et al., 2017) and generally indicate whether someone is aging more rapidly or slowly than his/her chronological cohort. Belsky et al. (2017) compared these three measures and again found modest correlation. They also found low correlation between these measures and both the physiology-based measures and telomere length. In
addition, the statistical correlations with health outcomes in middle age of all measures were relatively modest. Additional summary measures based on RNA expression, proteomics, and metabolomics have been proposed even more recently (Table 1-1). Jylhävä et al. (2017), in a review of existing literature, compared the links between mortality and the “biological age” measure, the “biological clock” measure, the measure based on metabolomics, and telomere length. They concluded that mortality appears best predicted by the “biological age” and “biological clock” constructs and was not well predicted by telomere length. Continued refinement of measures of this type, and development of data and links between health outcomes and DNA methylation, RNA expression, proteomics, and metabolomics, are likely to be extensive in the coming decade.
INTEGRATING GENETIC RISK INTO RESEARCH ON HEALTH AND AGING
Not mentioned as yet in this discussion is the recent expansion of research on the role of DNA in affecting health outcomes of older populations. This role is indicated in Figure 1-7 as “Genetic Predisposition.” The growth of DNA-based data on large populations in this century offers new approaches to determining how innate genetic characteristics both directly affect aging health and interact with the environment and behaviors of individuals. The technological developments that have allowed large-scale throughput of DNA samples have resulted in data with information on millions of single-nucleotide polymorphisms for millions of people. While this progress builds on research over many years using twin and familial samples, it also offers a new approach to determining genetic effects on health and aging. Unlike the RNA-based measures and the DNA methylation measures discussed above, these genetic characteristics are fixed and not changed by environments.
Large population studies have been used to determine genetic markers for a number of risk factors for health outcomes such as body mass index (Locke et al., 2015) but also socioeconomic risk factors such as educational attainment (Rietveld et al., 2013; Okbay et al., 2016). Much work has used large agglomerations of data to produce megasamples, which can be used to discover genes related to health outcomes such as longevity (Broer et al., 2015), Alzheimer’s disease (Lambert et al., 2013), and cognitive ability (Kirkpatrick et al., 2014). Social scientists have used the data to investigate how genetic characteristics might change over time in the population, how life cycle–social influences interact with time trends in behaviors as well as with the genome to affect health, and how genetic characteristics of social networks can influence health outcomes (Conley et al., 2016; Domingue et al., 2018; Liu and Guo, 2015). A significant amount of work is now
ongoing to investigate heterogeneous genetic risk in populations that could be related to differential effects of adverse social events and circumstances (Domingue et al., 2017). The genomic links with the downstream biomarkers discussed above will increasingly be integrated into investigations of aging health and longevity (Deelen et al., 2013).
Social scientists have benefited from the development of polygenic risk scores (PRSs): scores built to reflect the influence of many individual genes that affect complex health outcomes (Belsky and Israel, 2014; Dudbridge, 2013). These scores are quite complex to define initially, as they tend to be based on large samples and investigate millions of genetic markers, but once determined, they provide a fairly easy way for social scientists to integrate genetics into analyses. Large population studies such as HRS are providing already-derived PRSs for complex traits to data users. These have the additional characteristic of not being highly restricted data, since they do not identify individuals as do the raw DNA data. As an example, we have used the PRSs provided by HRS to demonstrate that there are statistically significant relationships between the PRS for a number of outcomes and the gender, education, and age of individuals in the HRS sample (see Table 1-2).
TABLE 1-2 Significant Differences in Polygenic Risk Scores in White Health and Retirement Study Sample by Sex, Education, and Age
| Female/male | Educationa | Ageb | |
|---|---|---|---|
| Alzheimer’s Disease | |||
| General Cognition | High | ||
| Body Mass Index | Low | Younger | |
| Waist Circumference | Low | ||
| Waist-to-Hip Ratio | |||
| Height | High | Older | |
| Mean Arterial Pressure | |||
| Educational Attainment | High | Older | |
| Ever Smoker | Low | Younger | |
| Depressive Symptoms | Low | ||
| Major Depressive Disorder | Low | ||
| Neuroticism | Low | ||
| Schizophrenia | +Female | Low | |
| Subjective Well-being |
a High = education ≥ 12 years; Low = education ≤ 11 years.
b Younger = polygenic risk scores are higher for younger people; Older = polygenic risk scores are higher for older people.
These associations merely indicate that genetic risk is associated in a given direction with the characteristic; causation is not implied. There are almost no associations between the PRSs in HRS and gender; only the PRS for schizophrenia indicates a higher risk for females. The PRS associations with age shown in Table 1-2 probably reflect survival selection or possibly that smoking and obesity are correlated with earlier age at death, whereas the height and educational attainment PRSs are associated with later age at death. The large number of PRS associations with education may reflect many pathways. Our point here is that a relationship exists between educational attainment as a broadly used social variable and genetic risk for a large number of outcomes. If analyses want to control for related risks, this is a good use of the PRSs. The relatively new availability of genetic data should allow this influence on health outcomes to be better represented in future research. It should also help clarify who is most at risk from genetic factors that, at this point, are immutable.
SUMMARY AND IMPLICATIONS FOR FUTURE TRENDS IN MORTALITY AND FOR HEALTH AND RESEARCH ON LONGEVITY AND AGING HEALTH
Recent trends are a mixture, some favorable and some unfavorable, and one’s classification of them into favorable or unfavorable may depend on whether the focus of one’s interest is on relative or absolute differences. U.S. life expectancy has increased, albeit slowly, until the last couple of years. This could be viewed as a favorable trend, until one looks at the progress of other countries, and the U.S. trend seems relatively unfavorable by comparison. The prevalences of most diseases, with the exception of those clearly related to tobacco smoking, have been increasing for decades. This seems clearly to be an unfavorable trend, until one realizes that it is in large part due to the reduction of mortality. Long-term reductions in prevalence of hypertension and high cholesterol are a positive health trend, but they have been accompanied by an increase in obesity and metabolic dysregulation related to diabetes. So changes in physiological dysregulation consist of both positive and negative trends.
Could we have predicted the recently observed changing health differentials observed in recent years? That men and women would become more similar in longevity is not surprising, given that women and men are behaving more similarly. When women behave more like men, for instance by smoking more and taking illegal drugs, their health outcomes are likely to become more like those of men because this behavioral component of the mortality difference will be reduced. On the other hand, the extremely adverse trends among women of lower educational attainment might not have been predicted. As indicated above, numerous researchers have con-
cluded that the policies and social fabric of American society have affected women’s health more adversely than men’s. This may be more characteristic of early adult life, when women are often single parents and sole supporters of children, but researchers have also increasingly clarified the lifelong health effects of early life hardships such as these. Increasing the relative rate of mortality decline for women may require social changes that are not on the immediate horizon.
The greater relative improvement of mortality and health among African Americans was somewhat surprising, given that it occurred in a period when socioeconomic differences in mortality, as well as in many other aspects of life, were widening in the general population. Whereas the relative size of the Black/White difference in life expectancy was affected by trends in deaths from violence and drugs, there also was some contribution from the reduction in deaths from chronic conditions. One factor that has not been considered in explaining the greater relative improvement in life expectancy for African Americans is cohort factors. It is possible that life circumstances of African Americans now reaching old age improved earlier in life with changes in civil rights, and these changed circumstances have resulted in improved mortality, relative to earlier cohorts of their peers or to Whites in the same cohort, in later life. If this is the explanation for the observed changes in life expectancy trends and differentials, the effect may play out in the future only as long as the relevant life circumstances improve over the life cycle.
Policies that are likely to reduce socioeconomic, racial, and geographic differentials in health and mortality in the United States seem unlikely to change in the near future. If anything, current policies such as rescinding the expansion of health insurance availability or having large geographic differentials in availability of support for health insurance for those of low SES may cause continued growth in the differentials in health and mortality between lower and higher socioeconomic groups. SES may thus become an even stronger predictor of health and longevity in the future.
For the U.S. population as a whole, the outlook for future morbidity and longevity depends on whether the onset and progression of the morbidity process are delayed and whether interventions at early points in the process are both feasible and implemented. One possibility is that the prevalence of disease could continue to increase but with reduced links to disability and death, as has occurred recently. Another possibility is that more and earlier intervention actions could be undertaken to attempt to prevent the changes in physiology that occur with aging. The reductions in cholesterol and blood pressure are a prototype for this alternative. The last few decades of work have shown that many biological factors, in addition to cholesterol and blood pressure, affect the rate of aging. The focus on “biological aging” and developing measures of aging
that capture the process that begins with the beginning of life promises the best hope for eventually intervening in, and delaying, the process of health change with age. Certainly, better understanding of individual differences in susceptibility to poor health outcomes, as well as individual differences in genetic influences of behaviors and life circumstances, suggests that delaying aging health change may become more personalized and targeted in the future.
The integration of research on aging health across disciplines has been very promising in clarifying the complex pathways through which life circumstances operate. The multidisciplinary model of health and mortality outlined in Figure 1-7 has developed over the past four decades, with each decade expanding the model while improving understanding of observed differences in aging health. Numerous social and behavioral sciences were natural allies in building this model, but they have been increasingly joined by the biological sciences, so that the view of health and age is at once more similar across disciplines yet more complex than it was a decade ago. The coming decade will see much expansion in relevant research, as research clarifies how life circumstances “get under the skin” biologically. This will add important answers as we continue to address basic questions about how socioeconomic, gender, race, and geographic differences in health and mortality arise. The basic questions that are the point of social science investigation of health and mortality have been the same for more than a century, but the kinds of answers that can be provided have expanded steadily.
The rapid development of data and methods is likely to continue. A wealth of longitudinal cohorts has increased focus on health change with age, seeing it as a process with different timing across individuals and groups. The development of multivariable measures such as multimorbidity, frailty, polygenic risk scores, biological risk, pace of aging, and biological clocks have helped capture the complexity and the multisystem nature of the process. Much of the reward in improved understanding that can result from these developments is still promise; but the near future is likely to bring rapid increases in understanding. Results are only beginning to be integrated into health care so that outcomes for individuals are maximized. Optimizing health for aging populations may require social and policy changes that do not depend on scientific progress.
REFERENCES
Acciai, F., and Firebaugh, G. (2017). Why did life expectancy decline in the United States in 2015? A gender-specific analysis. Social Science and Medicine, 190, 174–180.
Alley, D.E., Lloyd, J., and Shardell, M. (2010). Can obesity account for cross-national differences in life expectancy trends? In National Research Council, International Differences in Mortality at Older Ages: Dimensions and Sources (pp. 164–192). E.M. Crimmins, S.H. Preston, and B. Cohen (Eds.). Panel on Understanding Cross-National Health Differences among High-Income Countries. Washington, DC: The National Academies Press.
Avendano, M., and Kawachi, I. (2014). Why do Americans have shorter life expectancy and worse health than do people in other high-income countries? Annual Review of Public Health, 35, 307–325.
Belsky, D.W., and Israel, S. (2014). Integrating genetics and social science: Genetic risk scores. Biodemography and Social Biology, 60(2), 137–155.
Belsky, D.W., Caspi, A., Houts, R., Cohen, H.J., Corcoran, D.L., Danese, A., Harrington, H., Israel, S., Levine, M.E., Schaefer, J.D., et al. (2015). Quantification of biological aging in young adults. Proceedings of the National Academy of Sciences of the United States of America, 112, E4104–E4110.
Belsky, D.W., Moffitt, T.E., Cohen, A.A., Corcoran, D.L., Levine, M.E., Prinz, J.A., Schaefer, J., Sugden, K., Williams, B., Poulton, R., et al. (2017). Eleven telomere, epigenetic clock, and biomarker-composite quantifications of biological aging: Do they measure the same thing? American Journal of Epidemiology, 1–11. doi: 10.1093/aje/kwx346.
Beltrán-Sánchez, H., Harhay, M.O., Harhay, M.M., and McElligott, S. (2013). Prevalence and trends of metabolic syndrome in the adult U.S. population, 1999–2010. Journal of the American College of Cardiology, 62, 697–703.
Blackburn, E.H., Greider, C.W., and Szostak, J.W. (2006). Telomeres and telomerase: The path from maize, Tetrahymena and yeast to human cancer and aging. Nature Medicine, 2(10), 1133–1138.
Blackburn, E.H., Epel, E.S., and Lin, J. (2015). Human telomere biology: A contributory and interactive factor in aging, disease risks, and protection. Science, 350, 1193–1198.
Bound, J., Geronimus, A.T., Rodriguez, J.M., and Waidmann, T.A. (2015). Measuring recent apparent declines in longevity: The role of increasing educational attainment. Health Affairs, 34(12), 2167–2173.
Broer, L., Buchman, A., Deelen, J., Evans, D., Faul, J., Lunetta, K., Sebastiani, P., Smith, J.A., Smith, A.V., Tanaka, T., et al. (2015). GWAS of longevity in CHARGE consortium confirms APOE and FOXO3 candidacy. Journal of Gerontology Series A: Biological Sciences and Medical Sciences, 70(1), 110–118.
Carroll, M., Lacher, D., Sorlie, P., Cleeman, J., Gordon, D., Wolz, M., Grundy, S., and Johnson, C. (2005). Trends in serum lipids and lipoproteins of adults, 1960–2002. Journal of the American Medical Association, 294, 1773–1781.
Case, A., and Deaton, A. (2015). Rising morbidity and mortality in midlife among white non-Hispanic Americans in the 21st century. Proceedings of the National Academy of Sciences of the United States of America, 112, 15078–15083.
Chen, J., Normand, S.L.T., Wang, Y., Drye, E.E., Schreiner, G.C., and Krumholz, H.M. (2010). Recent declines in hospitalizations for acute myocardial infarction for Medicare fee-for-service beneficiaries. Circulation, 121, 1322–1328.
Chetty, R., Stepner, M., Abraham, S., Lin, S., Scuderi, B., Turner, N., Bergeron, A., and Cutler, D. (2016). The association between income and life expectancy in the United States, 2001–2014. Journal of the American Medical Association, 315, 1750–1766.
Chong, Y., Tejada Vera, B., Lu, L., Anderson, R.N., Arias, E., and Sutton, P.D. (2015). Deaths in the United States, 1900–2013. Hyattsville, MD: National Center for Health Statistics.
Cohen, A.A., Milot, E., Li, Q., Bergeron, P., Poirier, R., Dusseault-Belanger, F., Fülöp, T., Leroux, M., Legault, V., Metter, J.E., et al. (2015). Detection of a novel, integrative aging process suggests complex physiological integration. PLoS One, 10(3), e0116489.
Conley, D., Laidley, T., Boardman, J., and Domingue, B. (2016). Changing polygenic penetrance on phenotypes in the 20th century among adults in the U.S. Population. Scientific Reports, 6, 30348. doi: 10.1038/srep30348.
Cornman, J.C., Glei, D.A., Goldman, N., and Weinstein, M. (2017). Physiological dysregulation, frailty, and risk of mortality among older adults. Research on Aging, 39(8), 911–933. doi: 10.1177/0164027516630794.
Crimmins, E. M. (2015a). Lifespan and healthspan: Past, present, and promise. The Gerontologist, 55, 901–911.
Crimmins, E.M. (2015b). Physiological differences across populations reflecting early life and later life nutritional status and later life risk for chronic disease. Journal of Population Aging, 8, 51–69.
Crimmins, E.M., and Levine, M. (2015). Current status of research on trends in morbidity, healthy life expectancy, and the compression of morbidity. In M. Kaeberlein and G. Martin (Eds), Handbook of the Biology of Aging (Ch. 18; pp. 495–505). St. Louis, MO: Elsevier Science.
Crimmins, E.M., and Saito, Y. (2000). Change in the prevalence of diseases among older Americans: 1984–1994. Demographic Research, 3, 1–20.
Crimmins, E.M., and Seeman, T.E. (2004). Integrating biology into the study of health disparities. Population and Development Review, 30, 89–107.
Crimmins, E.M., Garcia, K., and Kim, J.K. (2010a). Are international differences in health similar to international differences in life expectancy? In National Research Council, International Differences in Mortality at Older Ages: Dimensions and Sources (pp. 68–101). Washington, DC: The National Academies Press.
Crimmins, E.M., Kim, J.K., and Vasunilashorn, S. (2010b). Biodemography: New approaches to understanding trends and differences in population health and mortality. Demography, 47, S41–S64.
Deelen, J., Beekman, M., Capri, M., Franceschi, C., and Slagboom, P.E. (2013). Identifying the genomic determinants of aging and longevity in human population studies: Progress and challenges. Bioessays, 35(4), 386–396.
Do, D.P., Frank, R., and Finch, B.K. (2012). Does SES explain more of the black/white health gap than we thought? Revisiting our approach toward understanding racial disparities in health. Social Science & Medicine, 74, 1385–1393.
Domingue, B.W., Liu, H., Okbay, A., and Belsky, D.W. (2017). Genetic heterogeneity in depressive symptoms following the death of a spouse: Polygenic score analysis of the U.S. Health and Retirement Study. American Journal of Psychiatry, 174(10), 963–970.
Domingue, B.W., Belsky, D.W., Fletcher, J.M., Conley, D., Boardman, J.D., and Harris, K.M. (2018). The social genome of friends and schoolmates in the National Longitudinal Study of Adolescent to Adult Health. Proceedings of the National Academy of Sciences of the United States of America, 115(4), 702–707. doi: 10.1073/pnas.1711803115.
Dudbridge, F. (2013). Power and predictive accuracy of polygenic risk scores. PLoS Genetics, 9(3), e1003348. doi: https://doi.org/10.1371/journal.pgen.1003348.
Dwyer-Lindgren, L., Bertozzi-Villa, A., Stubbs, R.W., Morozoff, C., Kutz, M.J., Huynh, C., and Flaxman, A.D. (2016). U.S. county-level trends in mortality rates for major causes of death, 1980–2014. Journal of the American Medical Association, 316, 2385–2401.
Espeland, M.A., Crimmins, E.M., Grossardt, B.R., Crandall, J.P., Gelfond, J.A., Harris, T.B., Kritchevsky, S.B., Manson, J.E., Robinson, J.G., Rocca, W.A., et al. (2017). Clinical trials targeting aging and age-related multimorbidity. Journals of Gerontology Series A: 72, 355–361.
Ezzati, M., Friedman, A.B., Kulkarni, S.C., and Murray, C.J. (2008). The reversal of fortunes: Trends in county mortality and cross-county mortality disparities in the United States. PLoS Medicine, 5, 557–568.
Fang, M.C., Perraillon, M.C., Ghosh, K., Cutler, D.M., and Rosen, A.B. (2014). Trends in stroke rates, risk, and outcomes in the United States, 1988 to 2008. The American Journal of Medicine, 127, 608–615.
Finch, C.E., and Crimmins, E.M. (2004). Inflammatory exposure and historical changes in human life-spans. Science, 305, 1736–1739.
Geronimus, A.T., Hicken, M., Keene, D., and Bound, J. (2006). “Weathering” and age patterns of allostatic load scores among Blacks and Whites in the United States. American Journal of Public Health, 96(5), 826–833.
Geruso, M. (2012). Black-White disparities in life expectancy: How much can the standard SES variables explain? Demography, 49, 553–574.
Glei, D.A., Meslé, F., and Vallin, J. (2010). Diverging trends in life expectancy at age 50: A look at causes of death. In National Research Council, International Differences in Mortality at Older Ages: Dimensions and Sources (pp. 1–38). E.M. Crimmins, S.H. Preston, and B. Cohen (Eds.), Panel on Understanding Cross-National Health Differences Among High-Income Countries. Washington, DC: The National Academies Press.
Glei, D.A., Goldman, N., Risques, R.A., Rehkopf, D.H., Dow, W.H., Rosero-Bixby, L., and Weinstein, M. (2016). Predicting survival from telomere length versus conventional predictors: A multinational population-based cohort study. PLoS One, 11(4), 1–18. doi: 10.1371/journal.pone.0152486.
Goldman, N., Glei, D.A., and Weinstein, M. (2016). What matters most for predicting survival? A multinational population-based cohort study. PLoS One, 11(7), 1–11. doi: 10.1371/journal.pone.0159273.
Gruenewald, T.L., Seeman, T.E., Ryff, C.D., Karlamangla, A.S., and Singer, B.H. (2006). Combinations of biomarkers predictive of later life mortality. Proceedings of the National Academy of Sciences of the United States of America, 103(38), 14158–14163.
Hannum, G., Guinney, L., Zhao, L., Zhang, G., Hughes, S., Sadda, B., Klotzle, B., Bibikova, M., Fan, J., Gao, Y., et al. (2013). Genome-wide methylation profiles reveal quantitative views of human aging rates. Molecular Cell, 49, 359–367.
Harper, S., Lynch, J., Burris, S., and Smith, G.D. (2007). Trends in the Black-White life expectancy gap in the United States, 1983–2003. Journal of the American Medical Association, 297, 1224–1232.
Harper, S., Rushani, D., and Kaufman, J.S. (2012). Trends in the Black-White life expectancy gap, 2003–2008. Journal of the American Medical Association, 307, 2257–2259.
Harper, S., MacLehose, R.F., and Kaufman, J.S. (2014). Trends in the Black-White life expectancy gap among U.S. states, 1990–2009. Health Affairs, 33, 1375–1382.
Hayward, M.D., Hummer, R.A., and Sasson, I. (2015). Trends and group differences in the association between educational attainment and U.S. adult mortality: Implications for understanding education’s causal influence. Social Science & Medicine, 127, 8–18.
Hertel, J., Friedrich, N., Wittfeld, K., Pietzner, M., Budde, K., van Der Auwera, S., Lohmann, T., Teumer, A., Völzke, H., Nauck, M., and Grabe, H.J. (2016). Measuring biological age via metabonomics: The metabolic age score. Journal of Proteome Research, 15, 400–410.
Ho, J.Y. (2013). Mortality under age 50 accounts for much of the fact that U.S. life expectancy lags that of other high-income countries. Health Affairs, 32, 459–467.
Ho, J.Y. (2017). The contribution of drug overdose to educational gradients in life expectancy in the United States, 1992–2011. Demography, 54, 1175–1202. doi: 10.1007/s13524-017-0565-3.
Ho, J.Y., and Fenelon, A. (2015). The contribution of smoking to educational gradients in U.S. life expectancy. Journal of Health and Social Behavior, 56, 307–322.
Ho, J.Y., and Preston, S.H. (2010). U.S. mortality in an international context: Age variations. Population and Development Review, 36, 749–773.
Holly, A.C., Melzer, C.D., Pilling, L.C., Henley, W., Hernandez, D.G., Singleton, A.B., Bandinelli, S., Guralnik, J.M., Ferrucci, L. and Harries, L.W. (2013). Towards a gene expression biomarker set for human biological age. Aging Cell, 12(2), 324-326.
Horvath, S. (2013). DNA methylation age of human tissues and cell types. Genome Biology, 14, 1-19.
Hummer, R.A., and Hayward, M.D. (2015). Hispanic older adult health and longevity in the United States: Current patterns and concerns for the future. Daedalus, 144, 20–30.
Institute of Medicine and National Research Council. (2013). U.S. Health in International Perspective: Shorter Lives, Poorer Health. S.H. Woolf and L. Aron (Eds.), Panel on Understanding Cross-National Health Differences Among High-Income Countries, Committee on Population. Washington, DC: The National Academies Press.
Jylhävä, J., Pedersen, N.L., and Hägg, S. (2017). Biological age predictors. EBioMedicine, 21, 29–36. doi: 10.1016/j.ebiom.2017.03.046.
Kennedy, B.K., Berger, S.L., Brunet, A., Campisi, J., Cuervo, A.M., Epel, E.S., Franceschi, C., Lithgow, G.J., Morimoto, R.I., Pessin, J.E., et al. (2014). Geroscience: linking aging to chronic disease. Cell, 159(4), 709–713. doi: 10.1016/j.cell.2014.10.039.
Kirkpatrick, R.M., McGue, M., Iacono, W.G., Miller, M.B., and Basu, S. (2014). Results of a “GWAS plus”: General cognitive ability is substantially heritable and massively polygenic. PLoS One, 9(11), 1–13. doi: 10.1371/journal.pone.0112390.
Kochanek, D.K., Murphy, S.L., Xu, J., and Arias, E. (2017). Mortality in the United States, 2016 (NCHS Data Brief. No. 293). Hyattsville, MD: National Center for Health Statistics.
Kumanyika, S.K., Landis, J.R., Matthews-Cook, Y.L., Almy, S.L., and Boehmer, S.J. (1998). Systolic blood pressure trends in U.S. adults between 1960 and 1980: Influence of anti-hypertensive drug therapy. American Journal of Epidemiology, 148, 528–538.
Lambert, J.C., Ibrahim-Verbaas, C.A., Harold, D., Naj, A.C., Sims, R., Bellenguez, C., Jun, G., DeStefano, A.L., Bis, J.C., Beecham, G.W., et al. (2013). Meta-analysis of 74,046 individuals identifies 11 new susceptibility loci for Alzheimer’s disease. Nature Genetics, 45(12), 1452–1458. doi: 10.1038/ng.2802.
Lariscy, J.T., Hummer, R.A., and Hayward, M.D. (2015). Hispanic older adult mortality in the United States: New estimates and an assessment of factors shaping the Hispanic paradox. Demography, 52, 1–14.
Levine, M.E. (2013). Modeling the rate of senescence: Can estimated biological age predict mortality more accurately than chronological age? The Journals of Gerontology Series A: Biological Science and Medical Science, 68(6), 667–674.
Levine, M.E., and Crimmins, E.M. (2014). Evidence of accelerated aging among African Americans and its implications for mortality. Social Science and Medicine, 118, 27–32.
Levine, M.E., and Crimmins, E.M. (2018). Is 60 the new 50? Examining changes in biological age over the past two decades. Demography, 55(2), 387–402.
Link, B., and Phelan, J. (1995). Social conditions as fundamental causes of disease. Journal of Health and Social Behavior, (Special Issue), 80–94.
Liu, H., and Guo, G. (2015). Lifetime socioeconomic status, historical context, and genetic inheritance in shaping body mass in middle and late adulthood. American Sociological Review, 80, 705–737.
Locke, A.E., Kahali, B., Berndt, S.I., Justice, A.E., Pers, T.H., Day, F.R., Powell, C., Vedantam, S., Buchkovich, M.L., Yang, J., Croteau-Chonka, D.C., et al. (2015). Genetic studies of body mass index yield new insights for obesity biology. Nature, 518(7538), 197.
Mather, K.A., Jorm, A.F., Parslow, R.A., and Christensen, H. (2011). Is telomere length a biomarker of aging? A review. The Journals of Gerontology Series A: Biological Science and Medical Science, 66(2), 202–213.
McClintock, M.K., Dale, W., Laumann, E.O., and Waite, L. (2016). Empirical redefinition of comprehensive health and well-being in the older adults of the United States. Proceedings of the National Academy of Sciences of the United States of America, 113(22), E3071–E3080.
Meara, E.R., Richards, S., and Cutler, D.M. (2008). The gap gets bigger: Changes in mortality and life expectancy, by education, 1981–2000. Health Affairs, 27, 350–360. doi: 10.1377/hlthaff.27.2.350.
Menni, C., Kiddle, S.J., Mangino, M., Viñuela, A., Psatha, M., Steves, C., Sattlecker, M., Buil, A., Newhouse, S., Nelson, S., et al. (2015). Circulating proteomic signatures of chronological age. The Journals of Gerontology Series A: Biological Science and Medical Science, 70, 809–816.
Montez, J.K., and Zajacova, A. (2013a). Trends in mortality risk by education level and cause of death among U.S. White women from 1986 to 2006. American Journal of Public Health, 103(3), 473–479.
Montez, J.K., and Zajacova, A. (2013b). Explaining the widening education gap in mortality among U.S. White women. Journal of Health and Social Behavior, 54(2), 166–182.
Montez, J.K., Hummer, R.A., Hayward, M.D., Woo, H., and Rogers, R.G. (2011). Trends in the educational gradient of U.S. adult mortality from 1986 through 2006 by race, gender, and age group. Research on Aging, 33, 145–171.
Montez, J.K., Hummer, R.A., and Hayward, M.D. (2012). Educational attainment and adult mortality in the United States: A systematic analysis of functional form. Demography, 49, 315–336.
Montez, J.K., Sasson, I., and Hayward, M.D. (2016a). Declining U.S. life expectancy, 1990–2010. Health Affairs, 35, 550.
Montez, J.K., Zajacova, A., and Hayward, M.D. (2016b). Explaining inequalities in women’s mortality in U.S. states. SSM-Population Health, 2, 561–571.
Murray, C.J., Kulkarni, S.C., Michaud, C., Tomijima, N., Bulzacchelli, M.T., Iandiorio, T.J., and Ezzati, M. (2006). Eight Americas: Investigating mortality disparities across races, counties, and race-counties in the United States. PLoS Medicine, 3, 1513–1524.
National Institutes of Health. (2017). Telomeres as Sentinels for Environmental Exposures, Psychosocial Stress, and Disease Susceptibility. A Workshop Co-sponsored by the National Institute of Environmental Health Sciences and the National Institute on Aging. Research Triangle Park, NC: National Institute on Aging.
National Research Council. (2011). Explaining Divergent Trends in Longevity in High-Income Countries. E. Crimmins, S.H. Preston, and B. Cohen (Eds.), Panel on Understanding Divergent Trends in Longevity in High-Income Countries, Committee on Population, Division of Behavioral and Social Sciences and Education. Washington, DC: The National Academies Press. doi: https://doi.org/10.17226/13089.
Okbay, A., Beauchamp, J.P., Fontana, M.A., Lee, J.J., Pers, T.H., Rietveld, C.A., Turley, P., Chen, G.B., Emilsson, V., Meddens, S.F.W., et al. (2016). Genome-wide association study identifies 74 loci associated with educational attainment. Nature, 533(7604), 539-542.
Olshansky, S.J., Antonucci, T., Berkman, L., Binstock, R.H., Boersch-Supan, A., Cacioppo, J.T., Carnes, B.A., Carstensen, L.L., Fried, L.P., Goldman, D.P., et al. (2012). Differences in life expectancy due to race and educational differences are widening, and many may not catch up. Health Affairs, 31, 1803–1813.
Ong, K.L., Allison, A., Cheung, B.M., Wu, B.J., Barter, P.J., and Rye, K.A. (2013). Trends in C-reactive protein levels in U.S. adults from 1999 to 2010. American Journal of Epidemiology, 177, 1430–1442. doi: http://dx.doi.org/10.1093/aje/kws443.
Peters, M.J., Joehanes, R., Pilling, L.C., Schurmann, C., Conneely, K.N., Powell, J., Reinmaa, E., Sutphin, G.L., Zhernakova, A., Schramm, K., et al. (2015). The transcriptional landscape of age in human peripheral blood. Nature Communications, 6, 8570.
Phelan, J.C., and Link, B.G. (2015). Is racism a fundamental cause of inequalities in health? Annual Review of Sociology, 41, 311–330. doi: https://doi.org/10.1146/annurev-soc-073014-112305.
Preston, S.H., and Stokes, A. (2011). Contribution of obesity to international differences in life expectancy. American Journal of Public Health, 101, 2137–2143. doi: 10.2105/AJPH.2011.300219.
Rehkopf, D.H., Rosero-Bixby, L., and Dow, W.H. (2016). A cross-national comparison of twelve biomarkers finds no universal biomarkers of aging among individuals aged 60 and older. Vienna Yearbook of Population Research, 14, 255–277.
Rietveld, C.A., Medland, S.E., Derringer, J., Yang, J., Esko, T., Martin, N.W., Westra, H.J., Shakhbazov, K., Abdellaoui, A., Agrawal, A., et al. (2013). GWAS of 126,559 individuals identifies genetic variants associated with educational attainment. Science, (2013), 1–11.
Rosamond, W.D., Chambless, L.E., Heiss, G., Mosley, T.H., Coresh, J., Whitsel, E., Wagenknecht, L., Hanyu, N., and Folsom, A.R. (2012). Twenty-two year trends in incidence of myocardial infarction, CHD mortality, and case-fatality in four U.S. communities, 1987 to 2008. Circulation, 125, 1848–1857.
Salive, M.E. (2013). Multimorbidity in older adults. Epidemiologic Reviews, 35, 75–83. doi: 10.1093/epirev/mxs009.
Sasson, I. (2016a). Diverging trends in cause-specific mortality and life years lost by educational attainment: Evidence from United States vital statistics data, 1990-2010. PLoS One, 11, 1–16.
Sasson, I. (2016b). Trends in life expectancy and lifespan variation by educational attainment: United States, 1990–2010. Demography, 53, 269–293.
Seeman, T.E., Singer, B.H., Rowe, J.W., Horwitz, R.I., and McEwen, B.S. (1997). Price of adaptation—Allostatic load and its health consequences. MacArthur studies of successful aging. Archives of Internal Medicine, 157, 2259–2268.
Seeman, T.E., McEwen, B.S., Rowe, J.W., and Singer, B.H. (2001). Allostatic load as a marker of cumulative biological risk: MacArthur studies of successful aging. Proceedings of the National Academy of Sciences of the United States of America, 98(8), 4770–4775.
St. Sauver, J.L., Boyd, C.M., Grossardt, B.R., Bobo, W.V., Rutten, L.J.F., Roger, V.L., Ebbert, J.O., Therneau, T.M., Yawn, B.P., and Rocca, W.A. (2015). Risk of developing multimorbidity across all ages in an historical cohort study: Differences by sex and ethnicity. The BMJ, 5, 1–13. doi: 10.1136/bmjopen-2014-006413.
Talbott, E.O., Rager, J.R., Brink, L.L., Benson, S.M., Bilonick, R.A., Wu, W.C., and Han, Y.Y. (2013). Trends in acute myocardial infarction hospitalization rates for U.S. states in the CDC tracking network. PLoS One, 8, 1–8.
Tucker-Seeley, R.D., Li, Y., Sorensen, G., and Subramanian, S.V. (2011). Lifecourse socioeconomic circumstances and multimorbidity among older adults. BMC Public Health, 11, 1–9.
Turra, C.M., and Goldman, N. (2007). Socioeconomic differences in mortality among U.S. adults: Insights into the Hispanic paradox. The Journals of Gerontology Series B: Psychological Sciences and Social Sciences, 62, S184–S192.
Wang, O.J., Wang, Y., Chen, J., and Krumholz, H.M. (2012). Recent trends in hospitalization for acute myocardial infarction. The American Journal of Cardiology, 109, 1589–1593.
Weidner, C.I., Lin, Q., Koch, C., Eisele, L., Beier, F., Ziegler, P., Bauerschlag, D., Jöckel, K., Erbel, R., Mühleisen, T.W., et al. (2014). Aging of blood can be tracked by DNA methylation changes at just three CpG sites. Genome Biology, 15, R24.
Wilson, P.W., D’Agostino, R.B., Levy, D., Belanger, A.M., Silbershatz, H., and Kannel, W.B. (1998). Prediction of coronary heart disease using risk factor categories. Circulation, 97(18), 1837–1847. doi: 10.1161/01.CIR.97.18.1837.
Woolf, S.H., and Braveman, P. (2011). Where health disparities begin: The role of social and economic determinants—and why current policies may make matters worse. Health Affairs, 30, 1852–1859.
Yeh, R.W., Sidney, S., Chandra, M., Sorel, M., Selby, J.V., and Go, A.S. (2010). Population trends in the incidence and outcomes of acute myocardial infarction. New England Journal of Medicine, 362, 2155–2165.
This page intentionally left blank.




























