5
An Integrated Impact Assessment Framework
Having defined forest health and described the threats facing North American forests (see Chapter 2), examined the options available for mitigating these threats (see Chapter 3), and elucidated the ecological, economic, social, and ethical considerations that accompany the use of biotechnology in forests (see Chapter 4), this chapter turns to the process of evaluating the risks and benefits of interventions to address forest health. The committee has been tasked with identifying the information and analysis needed to inform a decision framework for using biotechnology in trees.
Any decision framework for assessing the potential impacts of introducing a biotech tree on forest health will need to enable evaluation of trade-offs between positive, negative, and neutral impacts and incorporate sources of uncertainty associated with those evaluations. This impact assessment approach integrates assessment of potential benefits within a traditional risk assessment framework, which includes evaluation of the potential degree and probability of harm and how to manage that harm (i.e., minimize or control adverse impacts) (NRC, 1996; EPA, 1998). In this context, the risk of loss of ecosystem services over part, or all, of a species’ range is weighed against the potential to recover ecosystem services across that range with and without the biotech intervention. Therefore, both the benefits and trade-offs of those benefits with any risk should be included in the impact assessment of a biotech tree in this context (versus, for example, assessment of biotech trees engineered only for commercial products). Such a framework could be used to evaluate any intervention aimed at improving forest health, including the use of selectively bred trees, pesticide use, or biological control.
IMPACT ASSESSMENT FRAMEWORK
The characteristics of forest health that are central to an impact assessment framework are contained within the term’s definition in Chapter 2: structure, composition, processes, function, productivity, and resilience. However, given that the definition establishes a linkage between changes in ecosystems and in services that affect humans, an impact assessment framework should
seek to establish causal linkages between them. The Millennium Ecosystem Assessment noted a lack of information on the details of these connections, except perhaps when considering food and water. Gaps exist, particularly with respect to regulating, cultural, and supporting services, many of which are not traded in markets (MEA, 2005). The committee notes especially the challenges of incorporating nonmarket cultural ecosystem services, such as aesthetic experiences (Cooper et al., 2016). The committee also recognizes that the assessment of impacts to ecosystem services does not take into account noninstrumental, intrinsic values (see Chapters 2 and 4). Although assessing the impact of biotech trees on these intrinsic values is not incorporated into this decision framework, the committee recognizes the importance of these values and the likelihood that they will be prioritized by some stakeholders. In Chapter 7, the committee recommends further research into a framework that takes these values into account.
Fully informed assessment of the impacts of deploying biotech trees would include consideration of other options to protect forest health, including that of taking no action at all (see Chapter 3). For example, without intervention to control emerald ash borer (whether it be by selective breeding, pesticides, biocontrol, or biotechnology), many species of ash are certain to decline and, potentially, to face extinction. How will forest health be affected in this case? The roles that ash plays in the ecosystem may not be completely lost with no intervention, as maples and other tree species may have functional overlap in some ecosystems. However, other impacts of the loss of the tree may not be remedied. For example, as noted in Chapter 4, black ash (Fraxinus nigra) has cultural value for some Native American communities (Poland et al., 2017), and its disappearance from the forest would have consequences for cultural ecosystem services. Organisms dependent on ash or on its effects on soils and other aspects of ecosystem structure and function may decline regardless of its replacement by other trees. If the potential impacts of a biotech tree on forest health are considered only in isolation, the likely consequences of taking no action may not be fully appreciated. Historically, tree species have recovered from forest pests that drove them close to extinction, as in the case of eastern hemlock (Tsuga canadensis) during the mid-Holocene (Booth et al., 2012). However, at the time the committee was writing its report, forests were experiencing an increase in pest introductions (Lovett et al., 2016) and being affected by other stressors, such as climate change, pollution, and landscape fragmentation (Hansen et al., 2001; Dukes et al., 2009; Rogers et al., 2017). Thus, recovery dynamics possible in the past should not necessarily be expected to occur in the future.
Furthermore, any restoration process requires consideration of silvicultural practices to restore trees to the landscape across their impacted range. Whether or not biotech seeds or seedlings are the propagules involved, access to appropriate sites and site preparation may have impacts on other areas of forest health. If planting into a closed canopy forest will require canopy openings, impacts of that disturbance and any maintenance of openings until saplings are sufficiently tall need to be assessed. For example, growth of American chestnut seedlings was enhanced under high light conditions and where competition was controlled with herbicides (Clark et al., 2012). Soil and canopy disturbance can increase susceptibility of other species to insect pests and pathogens, allow establishment of invasive plants, and increase soil erosion and carbon and water loss (e.g., Covington, 1981; Denslow et al., 1998; Mack et al., 2000; Gandhi and Herms, 2010). These unintended consequences can have impacts on ecosystem services that need inclusion in both the impact assessment and in any site management plan.
Ecosystem Services
As discussed in Chapter 2, ecosystem services are the goods and services that are of value to people, provided wholly, or in part, by ecosystems (Olander et al., 2015). They have been characterized by the Millennium Ecosystem Assessment as falling into the categories of provisioning,
regulating, supporting, and cultural (Shvidenko et al., 2005). In 2015, the U.S. Environmental Protection Agency (EPA) considered how ecosystem services could be classified to be useful in assessing how changes in ecosystems affect human welfare (EPA, 2015). Specifically, the interest was in understanding the consequences of human interventions in ecosystems. In the context of the committee’s report, that intervention would be the introduction of a biotech tree into a forest ecosystem with the intent of gene flow through minimally managed, silvicultural, and urban forest systems. In other words, the intent of the intervention is for traits conferring resistance to insect pests and pathogens to become widespread.
There are a number of approaches to classifying ecosystem services, but for the purpose of assessing the potential for biotechnology to protect forest health, perhaps the best approach is to consider them in the context of human-induced change. The EPA (2015) classification is meant to enable characterization of the impacts of change in forest structure and functions on the provision of ecosystem services. To be effective, any changes that occur to ecosystem services must be considered holistically, not individually; all need to be accounted for since all are important and interlinked.
The National Ecosystem Services Classification System (NESCS) (EPA, 2015) tracks elements of the environment through the products those elements produce, to the different ways those products are used or appreciated by humans, to the direct user or appreciator of the products. The classification system identifies the “hand-off” from ecosystems to human beings by specifying “environmental classes” and the “end-product classes” they provide (see Figure 5-1). The committee recognizes that the concept of ecosystem services has an anthropocentric focus on instrumental value and does not incorporate intrinsic value (NRC, 2005). As discussed above and in Chapter 2, the committee considers that further research is needed to develop a complementary conceptual framework that takes intrinsic values into account.
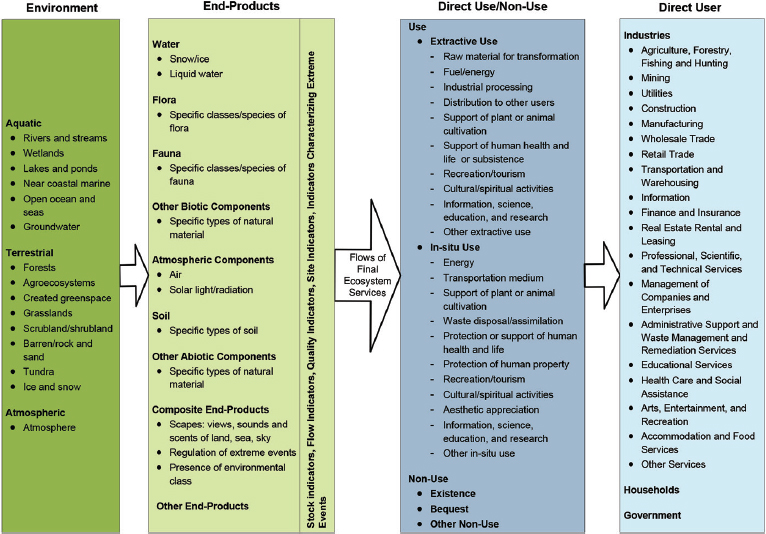
NOTE: Households as Direct Users include individuals, tribes, and communities for whom cultural/spiritual activities, aesthetic appreciation, and nonuse values may be significant.
SOURCE: EPA, 2015.
The NESCS (EPA, 2015) does capture the fact that not all ecosystem services are consumed by humans. Uses (consumptive and nonconsumptive) are situations in which a person actually uses a good (e.g., visits a forest), plans to use it, or entertains the possibility of using it (Pearce et al., 2006). In contrast, nonuse values are not derived from direct use or contact with ecosystems, but rather from appreciation of the existence of, say, tropical rain forests, or from a desire to preserve ecosystems for future generations (bequest value). A person could value the existence of a good (e.g., an endangered species) even though she or he may never see the good (Fisher and Turner, 2008; Cooper et al., 2016). Figure 5-1 shows the flow of services from the environment to the direct user or value for a wide range of ecosystems, including forest ecosystems.
The analysis of the impacts on human welfare based on change in ecosystem services involves identifying, quantifying, and valuing the services (EPA, 2015). The NESCS sets the stage for quantification and valuation of use and nonuse values by promoting the construction of an inclusive inventory of change. Particularly in the case of public goods associated with flows of ecosystem services, quantification may be challenging (e.g., what are the units in which scenic beauty or plant diversity is to be measured?). Valuation, as by assigning monetary values, is also complicated by the lack of market prices for services such as recharge of aquifers and clean air. At this stage, because of the challenges in quantification, cultural, aesthetic, and nonuse values may not be captured; however, the committee stresses the importance of their inclusion (see Box 5-1). There are a number of possible techniques for quantification and valuation, all of which can be problematic from a methodological standpoint (Nijnik and Miller, 2017). Ultimately, resolution of these difficulties would be needed to support impact analysis of the use of biotech trees in forests or other forest health interventions, but that analysis lies beyond the scope of this study.
According to the NESCS, the effects of introducing a biotech tree would be traced to end-products (second column in Figure 5-1) and then mapped to direct uses and nonuses (see Figure 5-1, column 3) and ultimately to direct users or appreciators (see Figure 5-1, column 4). Box 5-2 describes how these effects would be traced and mapped for one of the case study trees, American chestnut. The NESCS provides an inventory of anticipated changes on which citizens and government decision makers can focus attention. Consideration of the consequences of the introduction of a biotech tree would require information not only about biotic and abiotic impacts on forest ecosystem structure and function, but also about the effect of the introduction on the availability and character of the breadth of ecosystem goods and services valued by members of society.
The impacts of changes to ecosystem services may be beneficial or adverse, may vary in size, and may differ across different members of society. Knowledge about these impacts provides the foundation for judgments about the potential risks and benefits of introducing a biotech tree. However, the causal linkages among changes in forest ecosystems, services, and human benefits are not always well understood by the ecologists, economists, and social scientists who must collaborate on the assessment. In 2018, the National Ecosystem Services Partnership, based at Duke University, published the Federal Resource Management and Ecosystem Services Guidebook (National Ecosystem Services Partnership, 2018). It provides practical advice for incorporating an ecosystems approach into resource management. As summarized in a presentation to the committee (Johnston, 2018), there are three basic steps in ecosystems services analysis that must be taken considering the specific circumstances of each intervention:
- Scoping to establish conceptual linkages among actions, ecological systems, and ecosystem services and values to different groups;
- Assessment and quantification to formalize causal chains, identify ecosystem service indicators and ecological production functions, and quantify changes in ecosystem services; and
- Valuation to quantify the effects on benefits (or value) realized by different beneficiary groups.
Risk Assessment Used in Tandem with Ecosystem Service Assessments
Ecological risk assessment uses science-based methods to evaluate the likelihood of adverse ecological effects resulting from particular stressors or actions (EPA, 1998). The assessment begins with problem formulation, in which risk assessors identify assessment endpoints and develop an analysis plan. Endpoints are typically biological or physical components (e.g., abundance of species) within the environment that may be affected by the proposed action. For example, EPA (1998:26903) defines an assessment endpoint as “an explicit expression of the environmental value to be protected, operationally defined as an ecological entity and its attributes.” Particular endpoints are situationally identified based on ecological relevance, susceptibility (sensitivity to the probable exposure), and significance to management goals (EPA, 1998). The choice of appropriate endpoints is crucial in ensuring that an assessment is useful for risk managers in making effective and defensible decisions (EPA, 1998).
In 2003, EPA released a set of conventional generic ecological assessment endpoints (conventional endpoints) that are broadly applicable in many ecological contexts and harmonized with environmental legislation and EPA policy (Munns et al., 2016). Conventional endpoints are designed to directly protect ecosystem function and biodiversity and to provide science-based proxies upon which an assessment (and decisions) can be based. Proxies are necessary because not all organisms and processes in an ecosystem can be studied or monitored (Suter, 2000). Protections recommended by risk assessments focus on protecting specific aspects and organisms within an environment, but they also indirectly support and protect many ecosystem services important to humans. As such, these endpoints address many of the ecosystem services relevant to assessment of forest health. As stated above, this approach can be adopted as part of an impact assessment framework but would need refinement to incorporate the full suite of services intended in the committee’s definition of forest health.
When considering impact assessment for the use of biotechnology in forests, links between specific forest protections and their effects on important ecosystem services should be made explicit. As suggested above, the advantage of bringing ecosystem services into impact assessments is that it opens up the possibility of including a broader range of values and making the connection between the protection of forests and human well-being clear for the public, stakeholders, and policy makers (Munns et al., 2016).
Efforts to incorporate ecosystem services directly into risk assessment frameworks have been made, both to implement broader protections and to garner better public and policy maker understanding and support (EPA, 2015). EPA has adopted this approach, defining ecosystem services as the outputs of ecological functions or processes that contribute to social welfare or have the potential to do so in the future (EPA, 2006). In 2015, EPA began explicitly encouraging the incorporation of ecosystem services into risk frameworks and released a list of ecosystem service generic assessment endpoints (ecosystem service endpoints). Because the definitions of these endpoints are intentionally broad in guidance, they must be specifically defined in individual assessments (EPA, 2015).
It is important to note that conventional and ecosystem service endpoints are complementary rather than competing concepts (EPA, 2015; Munns et al., 2016; see Table 5-1). The consideration of conventional endpoints may be legally required, and they represent the ecologically important and susceptible entities and attributes that require protection under current laws and regulations (see Chapter 6). However, although EPA considers conventional endpoints sufficient for use in risk assessment (EPA, 2015), the complexities associated with biotechnology suggest that ecosystem service endpoints should be a required component of a broader impact assessment. This approach is consistent with EPA’s recommendation that ecosystem service endpoints be added in situations that need better monitoring of the effects of proposed actions on those ecosystem attributes that
TABLE 5-1 Comparison of Selected Ecological and Ecosystem Service Endpoints
| Conventional Ecological Assessment Endpoint | Generic Ecological Assessment Endpoint | Possible Generic Ecosystem Services Endpoint |
|---|---|---|
| Population abundance Population size and structure Presence/absence of game species Taxa richness |
Population and abundance | Food production |
| Plant community uptake and deposition of pollutants | Ecosystem function | Air purification (for breathing and visibility) |
| Plant community net production Carbon sequestration |
Ecosystem function | Climate stabilization |
| Water retention | Ecosystem function | Flood and storm surge regulation |
| Wilderness quality Endangered species and habitat area and quality |
Area or quality of ecosystem or special place | Provision of aesthetic, scientific, recreational, educational, cultural, medical, genetic, ornamental, and spiritual resources |
SOURCE: Adapted from Table 3 in EPA, 2016a.
directly influence humans and where benefits accruing to humans from implementing protections that are not immediately obvious (EPA, 2015).
The use of ecosystem service endpoints alone may not afford sufficient protection of a forest ecosystem from the effects of intervention, because it could create a situation with perverse incentives when, in fact, the opposite was intended by the decision framework. For example, some ecosystem services may increase as an ecosystem is increasingly exploited (at least to a point), whereas some ecosystem services may decrease when protections are implemented. Therefore, the use of both conventional and ecosystem service endpoints, in tandem, may work best to produce a framework that accounts for both human use and nonuse ecosystem services and long-term ecosystem function.
EPA has considered how conventional risk assessment endpoints can be related to generic ecological assessment endpoints and then to possible generic ecosystem service assessment endpoints (EPA, 2016a). Although not customized to the current setting of development of a biotech tree, this overview may be helpful in understanding how risk assessment and ecosystem services can be integrated for use in forest ecosystems.
The incorporation of ecosystem service endpoints, however, is not straightforward. Conventional endpoints are based on precedent, regulations, and established practices and usually have recognized metrics for assessment (EPA, 2015). They are specifically defined for each individual framework, and elements that are not applicable can be dropped and novel endpoints added when appropriate (EPA, 2015). In contrast, ecosystem service endpoints often lack foundational bases and functional metrics. Some quantitative methods exist for their estimation, but additional research will need to be conducted before they can be put into common use (Kremen, 2005; EPA, 2016a). In addition, the complexity of using ecosystem service endpoints is increased because a wide range of nonuses are taken into account (EPA, 2016a). Given that this committee’s definition of forest health includes both ecological and human dimensions, explicit incorporation of both dimensions should be included in assessments of the risks, benefits, and costs of a forest health intervention, including the development and release of biotech trees.
TOOLS AVAILABLE TO INFORM AN IMPACT ASSESSMENT FRAMEWORK
Little information exists on the impacts of releasing biotech trees into minimally managed or unmanaged systems, even for those efforts with impacts likely to be similar to those of trees developed through selective breeding (Hoenicka and Fladung, 2006). Therefore, this section discusses the types of quantitative and qualitative data that will be needed, how the data can be generated, and how they can be used to inform the impact assessment approach proposed. In the case of forest trees, data on gene flow, establishment, and any potential impact of the modified tree species on the remaining forest ecosystem are critical to assessing any potential benefits and risks of implementing this technique. In many cases, the requisite information will take years to decades to compile, and often a combination of approaches will be needed to fulfill all the demands of a multidimensional assessment framework. For example, field trials evaluating biotech trees can provide information about the growth and resistance of the trees, but they can take years to develop and alone will not be sufficient for assessing the impacts of releasing the biotech tree to address forest health. However, data from field trials can be combined with data derived from other types of plant releases to parameterize simulation models to inform impact assessment. Similar modeling approaches can allow inclusion of gene flow and climatic tolerances. Models should be reassessed as specific data are obtained from field trials, allowing adaptive management of the risk assessment process. Additionally, the results of surveys and stakeholder engagement as described in Chapter 4 and Box 5-1 will provide information about human values and concerns associated with specific products of biotechnology.
Field Trials
The long-lived nature of tree species not only complicates the development and deployment of releasing biotech trees to maintain or improve forest health as described in Chapter 3, but it also makes the assessment of potential impacts difficult. Although modeling can help address the long temporal and large spatial scales involved (see the next section, “Scenario Modeling”), the question of how long to monitor the potential impacts of a genetic change in a long-lived organism and its surrounding ecosystem remains uncertain. Furthermore, even if critical unexpected impacts emerge, reversing continued gene flow will likely be impossible.
Therefore, field trials of biotech trees are a vital tool. Trials allow for an assessment of both the efficacy and the durability of resistance, and of the potential impact on the fitness of the tree species and on the ecosystems in which it will be released. The long-lived nature of trees and the varied ecosystems in which even a single tree species may occur mean the design of field trials will be more complicated and of longer duration than those currently implemented for agricultural crops. For example, many tree species do not produce significant quantities of pollen or seed for a decade or more. As in selective-breeding trials, resistance in any seedlings produced from open-pollinated seed orchards will need to be evaluated over variable environments (including areas of high disease or insect pressure) and over long time periods. Additionally, to increase the probability that resistance to the insect pest or pathogen will spread across forested systems, field trials need to be planted in environments similar to those that the tree species will encounter. As a result, confinement conferred through climatically or geographically inhospitable environments (McDougall et al., 2011) is not an option.
One approach for assessing the suitability of the biotechnological changes and minimizing escapes may be to develop field trials within the native range of the species, but in substantial isolation from other cultivated or wild individuals in the same species or genus. In cases where this is an option, methods for tracking gene flow from seed orchards, including use of sentinel trees within and at increasing distance from orchards and planting buffer trees at edges (Scorza et al., 2013)
may be useful. Simulation modeling of multiple generations will likely need to accompany field trials to better address the probability of gene flow over long time frames (Snow, 2002; Linacre and Ades, 2004; see also the next section).
However, in many cases isolation will not be feasible given the long distances that pollen can travel. It is important that approaches that allow flowering and pollen production be considered carefully given that spread is virtually impossible to contain once it occurs. One advantage of tree longevity is that trial stands can be followed through the sapling stage, with removal of propagated individuals before they flower or produce cones (Häggman et al., 2013) or at the first sign of reproduction, even though in such cases there can be at least some gene flow from a few precocious trees. Short-term trials would provide time to assess for traits that might result in a harmful impact when trees are released into minimally managed or unmanaged environments. Such management has been proposed for nonnative, potentially invasive species, such as Eucalyptus urograndis, grown for commercial purposes (Flory et al., 2012). If permitted under controlled conditions, field plantings would need to be conducted with sufficient assurances that plants would be removed or continuously managed once the research phase was completed. Effectiveness of this approach depends on the life history and management potential of the tree species involved. The ultimate question of how long to follow field trials for a tree species will vary by species characteristics and the genetic changes incorporated via biotechnology.
The need to be precautionary about pollen production is complicated because in some cases trials would need to be of longer duration to fully assess resistance. Trees may need to be on site for many years before a pathogen has sufficient impact to judge effectiveness of breeding or biotech interventions. For example, white pine blister rust can take 5–10 years to exhibit impacts (Kinloch et al., 2008; Sniezko et al., 2012), and ash trees may need to be of sufficient size to attract pests (Duan et al., 2017). In the case of white pine blister rust, increasing pathogen abundance via increasing local abundance of the alternate host (Ribes spp.) may accelerate colonization of trial plantations. Thus, impact assessment will need to weigh the risk of longer-term field trials on a case-by-case basis.
Field trials are important because the potential effects of gene flow from biotech trees need serious consideration. Individuals may be in the system for decades to centuries, long after people will recall the immediate concerns that the biotechnology was intended to address, but not before the full impacts of deployment occur. Ecological, social, and cultural issues may shift over time, and it will not be possible (at least with the currently understood state of the science) to reverse decisions made in the present to address a particular threat. Gene flow can also have economic repercussions. For example, if transgenes spread into certified forest operations, this could jeopardize the certification status and its associated value; the International Forest Stewardship Council does not currently certify genetically altered tree material (Auld and Bull, 2003). Some markets may be closed to biotech trees or those that are hybrids.
Field trials will be critical to provide information about any short-term impacts and the growth, form, and resistance of the trees, but they are unlikely to provide data on interactions with other forest biota and how the release could affect the entire ecosystem. If plantation trials are successful, dispersal, establishment, and growth into mixed-species stands should also be tested in small-scale pilot plantings. Such trials would both indicate the real feasibility of the intended restoration and help parameterize models that would allow evaluation not only of whether the timing of gene flow and restoration will exceed that of species loss from the system, but also of the potential ecological impacts of the release.
Scenario Modeling
Given the long-term nature of developing a biotech tree and the limitations of field trials to fully represent long-term impacts in a forest environment, scenario modeling may prove to be a valuable tool in an impact assessment framework. Impact assessment models that use available scientific data can be developed to assess potential impacts and population dynamics. These models can be used to integrate ecological, economic, and cultural considerations, including the benefits of the proposed actions and the consequences of not carrying them out. Synthesis of all available information and data-model integration will aid in making the most accurate and informed predictions of potential impacts. Modeling gene flow after the release will also aid assessment of the success and impacts across the landscape. In addition, modeling potential scenarios that include and track sources of uncertainty will allow quantification of the reliability of the assessments, estimation of the predictive capacity of the model, and identification of data needs. However, these models, which are quantitative in nature, will be challenged by the difficulty of including many of the qualitative impacts associated with cultural considerations.
Models that simulate spread, performance, and, in some cases, impact of introduced plants are commonly developed for invasive species (e.g., Bullock et al., 2008; Ibáñez et al., 2009). These models can address different components of the invasion, including movement across the landscape (LaFleur et al., 2009; Emry et al., 2011; Marco et al., 2011), performance under different environmental conditions (Buckley et al., 2003), impacts (Rinella and Luschei, 2007; Gómez-Aparicio et al., 2008), or performance under climate change scenarios (Beaumont et al., 2009; Bradley et al., 2010). Analogous approaches could be applied to modeling the spread of biotech trees (see Box 5-3).
Data Synthesis and Data-Model Integration
Data synthesis and data-model integration are common approaches used for enhancing model realism, explanatory insight, and predictive capability (Ibáñez et al., 2014; Dietze, 2017). Data-model integration techniques can merge physiological and demographic information gathered in field trials together with known species interactions and models of gene flow to provide a broad understanding of the introduced genotype’s performance under a variety of environmental conditions. Outputs from these models can provide critical information about the potential risks of releasing a new genotype into the forest by providing insight on not only the potential outcomes, but also the likelihood of the forecast.
Data synthesis and data-model integration can be implemented using multilevel, also known as hierarchical, Bayesian models (Clark, 2005). Hierarchical models can integrate diverse datasets and processes with the goal of developing predictive outcomes (Clark et al., 2010). They provide a venue for examining the complexity in a system (Clark and Gelfand, 2006). In addition, these models can incorporate new data as they become available, informing adaptive management of the biotech tree introduction. These techniques are also useful for generating forecasts under different scenarios, including climate scenarios, while still tracking the sources of uncertainty associated with the data, process, or predictor variables. Ongoing data assimilation into models will also be an important tool for continuous impact assessment and adaptive management. An iterative process of data assimilation will improve forecasts by reducing uncertainty in parameter estimates, improving model structure, and better identifying and quantifying sources of uncertainty (Luo et al., 2011).
Modeling Gene Flow
Given that biotech trees to protect forest health would be released into minimally managed or unmanaged environments, evaluation of their potential spread is critical in any impact assessment.
These trees would be planted with the goal of maximizing propagule and pollen spread in ecosystems to restore a species and protect or restore forest health. Along with data collected from field trials, modeling the rate of gene flow via pollen spread and seed dispersal is essential for understanding the potential spread of the introduced genes across the landscape and the necessary planting configuration. Because predicting the impact of biotech trees released into the environment is a complex and challenging undertaking, models can also help evaluate unintended consequences of the genetic modifications on nontarget organisms and ecosystem processes.
Gene flow is a multidimensional process that is influenced by the reproductive biology of the source and recipient organisms, the disturbance and management regimes in plantations and ecosystems, climate and other abiotic factors, and the direct and indirect effects of the genetic change (Savolainen et al., 2007; Ellstrand et al., 2013; Tsatsakis et al., 2017). Given the inherent stochasticity of many of the underlying processes, as well as the large spatial and temporal scales that must be considered, direct estimation of gene flow can be quite challenging, even for well-studied annual crops with wild relatives that have relatively limited dispersal of seeds and pollen. For example,
estimating transgene flow from cultivated maize to its wild relative teosinte has been highly controversial (Kinchy, 2012), due in part to the considerable methodological and sampling challenges inherent in detecting low levels of introgression over very large spatial scales (Agapito-Tenfen and Wickson, 2018). This problem is greatly magnified for trees, which are typically outcrossing, can disperse pollen and seeds over dozens or even hundreds of kilometers (Slavov et al., 2009; Williams, 2010), and may contribute pollen for centuries. This complexity means that rare, stochastic events can have outsized importance in determining gene flow rates, and these are notoriously difficult to measure (Nathan, 2006; Robledo-Arnuncio et al., 2014).
A partial solution to this problem is the use of simulation modeling to integrate all of the disparate factors that affect gene flow, thereby allowing analysis of the process at spatial and temporal scales that are appropriate for biotechnology impact assessment (Ellstrand et al., 2013). While spread of small edits in native genes are difficult to track in wild populations, models can be parameterized with gene flow data from marker genes and fitness differentials from results of confined experiments.
There is some dispute about the proper time frame for such models. On one hand, simulating biotech tree establishment over many generations enables assessment of the combined effects of genetic drift and selection on abundance of resistance alleles in unmanaged or minimally managed ecosystems (Chapman and Burke, 2006; Meirmans et al., 2009). However, with trees this would require simulations that run for millennia, and the relevance of such models for near-term ecological risk assessment is questionable. Another approach is to use more detailed, spatially explicit models that run on annual time steps to allow explicit simulation of disturbance and management processes within a realistic landscape and time context (Kuparinen and Schurr, 2007; DiFazio et al., 2012). Such models allow comparison of different management and disturbance scenarios and identification of the factors that have the largest impact on spread of genes; this enables more effective prioritization of research and monitoring efforts to improve the accuracy of predictions of gene flow and evaluation of impacts.
Modeling the timing and distance of gene flow and seed dispersal could help identify the likelihood that the desired spread of the species will occur within a time frame sufficient to allow species persistence. Successful development of resistant genotypes will not confer successful species restoration if the scale of reproduction and dispersal suggests that resistant genotypes will not spread across the range of the species. Including factors associated with climate change into models may also indicate where restoration might be most successfully accomplished (Häggman et al., 2013). If changing environmental conditions suggest that the species requires planting beyond its historically native range, additional risk factors associated with this range expansion would require consideration (Aitken and Whitlock, 2013).
Limited Representation of Qualitative Impacts
Modeling is limited by its ability to incorporate factors such as social, political, cultural, and ethical issues related to the use of biotech trees to improve forest health. These factors would be incorporated if the NESCS (EPA, 2015) were implemented (see section “Impact Assessment Framework,” above). Refinement of the ecosystem services endpoints may address some aspects (such as recreational use of a forest), but others will not be amenable to quantitative modeling (see Box 5-1).
Predictive risk assessment and modeling of the potential for sufficient spatial dispersal of the genetic change over a temporal scale to restore or recover the species should also be assessed, either within, or in analyses complementary to, those involved in assessing impacts on conventional endpoints. The predictive risk assessment would elucidate the potential for inadvertent negative genetic, ecological, or health traits to be inextricably associated with the intended positive genetic modification. If the genetic change involved has already been assessed for a crop plant species or
for the species of interest in another country (e.g., biotech Populus nigra with insect resistance in China [FAO, 2010]), the results may inform elements of the impact assessment, as would results from selective-breeding efforts (Häggman et al., 2013). Risk assessments developed for agricultural biotech crops could help identify some of the information and analysis needs for an impact assessment framework, with the caveat that agricultural fields are much more intensively managed than noncommercial forests. Additional differences include the issues that agricultural fields are not usually viewed as “natural” environments or wilderness and that biotech crops are not planted with the intention of spread and persistence, as is intended for a biotech tree for forest health improvement.
Modeling can help elucidate the scale of potential impacts over time (e.g., Vose et al., 2015). Potential harmful invasiveness (i.e., large-scale exclusion of other native species) of the tree would be included here and might involve a modification of the USDA PERAL risk assessment (see Box 5-3). Results from confined growth trials could also inform this assessment. Although separate risk assessments for field trials and unconfined distribution are often required for other uses of biotechnology, in this case both elements could be considered simultaneously.
Incorporating Uncertainty
Given the critical role of uncertainty analysis in risk assessment (Hayes et al., 2007), explicit evaluation of this factor should be included in the impact assessment framework (Häggman et al., 2013). Uncertainty may result from missing data, data variability, bias in the data collected, or external stochastic processes (e.g., environmental conditions). Identifying the sensitivity of the impact assessment framework to these types of uncertainty will influence both confidence in the results and the effectiveness of decision making.
There are numerous methods to evaluate uncertainty. Probabilistic models can incorporate uncertainty at all stages of the analysis (Mantyniemi et al., 2013). Even for deterministic model outputs, potential uncertainty can be assessed through expert judgment, model emulation, sensitivity analysis, temporal and spatial variability in the model output, use of multiple models, and use of statistical approaches (Uusitalo et al., 2015). As with data synthesis and data-model integration, hierarchical Bayesian models provide a useful framework for incorporating the different sources of uncertainty by representing the system as a network of components that include observed data, underlying process models, errors in parameter estimates, and models of parameter interdependence (Clark, 2005; Dietze, 2017).
Comparing outputs from different modeling approaches is a technique to offset uncertainties in the predictions (Millar et al., 2007) because they focus on different aspects of species’ performances that are complementary. For example, niche models and process-based models can be combined to better predict future species performance and distribution under climate change (Morin and Thuiller, 2009; Mellert et al., 2015). Scenario modeling can also be used to assess impact of, and responses to, environmental stochasticity. Furthermore, these predictions can be improved with data as these become available over time, allowing for verification of the forecasts, improvement of management practices, and validation and reevaluation of the models. Hierarchical Bayesian models can accommodate complex processes within a spatiotemporal context and still track the uncertainty associated with each component (Wikle, 2015). These methods also allow tracking uncertainty in any forecasts generated by modeling outcomes under potential scenarios.
Impact assessment models that use available scientific information can be developed to assess potential benefits, risks, and dynamics. These assessments would integrate ecological, economic, and cultural considerations, including the impacts of the proposed actions and the consequences of not carrying them out. Synthesis of all available information will aid with making the most accurate and informed predictions of potential risks (Dietze, 2017). In addition, modeling potential scenarios that include and track sources of uncertainty will allow quantification of the reliability
of the assessments, estimation of the predictive capacity of the model, and identification of data needs. However, as described above, the processes of establishment and spread of forest trees are complex and occur over large spatial and temporal spans, so even estimating the uncertainty in key parameters is challenging. A potential solution to this problem is the use of adaptive management.
USE OF ADAPTIVE MANAGEMENT
The concept of adaptive management was formalized in the 1970s as a flexible approach to natural resource management that uses data collected during implementation of a management action to evaluate the efficacy of that action and make any needed modifications (Holling, 1978; Walters and Hilborn, 1978). Multiple types of adaptive management have been identified, ranging from active to passive. As defined by Williams (2011:1371) “active adaptive management actively pursues the reduction of uncertainty through management interventions, whereas passive adaptive management focuses on resource objectives, with learning a useful but unintended by-product of decision making.” Because active adaptive management is designed to identify the actions best able to meet management objectives, experimental (or quasi-experimental) comparisons of management applications are employed. Monitoring of existing management efforts to conduct the assessment is more passive and generally less expensive. Because biotech trees would require new field trials (if permitted), establishing active adaptive management efforts may be less costly than using this approach for other purposes.
There is increasing interest in using adaptive management coupled with ecosystem services as a new way forward in natural resources management in the United States (Epanchin-Niell et al., 2018). The overall framework is depicted in Figure 5-2. The process begins with a “deliberative phase” during which the problem is framed by engaging the stakeholders, identifying endpoints, developing models, and deciding on an initial course of action. The procedures for defining endpoints in an ecosystem services context and developing models are discussed above (see section “Risk Assessment Used in Tandem with Ecosystem Service Assessments,” above). Next is the
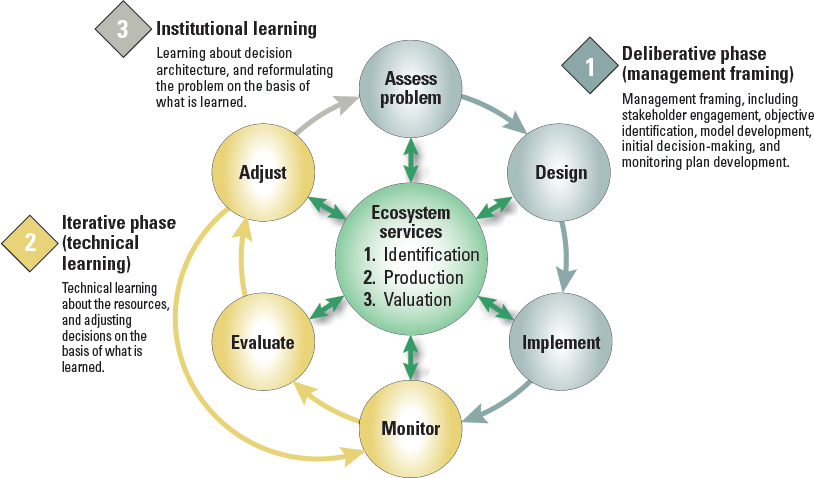
SOURCE: Epanchin-Niell et al., 2018.
“iterative phase” during which the initial management actions are implemented, and outcomes are monitored to provide data about the efficacy of the management actions relative to the defined endpoints. The final phase is “institutional learning,” which requires an administrative process to evaluate the data from the technical learning phase and recalibrate endpoints and refine management as needed. This process is intended to be open-ended and iterative, where the learning cycle would continuously improve the solutions to the problem. Adaptive management has been suggested as an effective framework for managing exotic tree introductions for biofuel production (Lorentz and Minogue, 2015) as well as for release of biotech trees into the environment (Strauss et al., 2010).
The current U.S. biotechnology regulatory system accommodates adaptive management approaches associated with field trials, although this may be limited for forest trees due to restrictions on flowering (Strauss et al., 2015).1 The committee was not asked to make recommendations about the regulatory process, but it was tasked with identifying how adaptive management can be used to enable realistic testing and assessment of biotechnology approaches for mitigating threats to forest health.
The 2017 National Academies report Preparing for Future Products of Biotechnology suggested a tiered risk assessment framework that incorporates adaptive management (NASEM, 2017). Under such a system, new biotech products are initially assessed and assigned to “bins” that are defined by the novelty and complexity of the product. Each bin then follows a different risk analysis procedure. For example, a native tree that is engineered to have increased expression of a cisgene might fall into the “familiar or noncomplex” bin. In contrast, a native tree engineered with a synthetic gene that confers levels of insect tolerance not seen in nature might fall into the “unfamiliar or complex” bin, triggering a more extensive external input process, as well as interactions between responsible federal agencies and expert advisory panels. A native tree engineered with a gene from an unrelated species introduced through Agrobacterium might fall somewhere in the middle because such modification is familiar, having been conducted for many years in agricultural crops, but trees with such modifications have been released only in agricultural settings rather than in minimally managed environments.
To illustrate how adaptive management might be applied in a tiered risk framework, biotech trees that fall into the lower risk bins would be good candidates for adaptive management. Such trees could be established in large field trials and allowed to flower under a streamlined permitting system. As part of the permitting process, a monitoring plan would be developed to ensure that data could be collected to reduce the uncertainty in critical parameter estimates, such as growth rates, age of flowering, gene flow distances, establishment rates, stability of resistance, occurrence of unanticipated pleiotropic effects of the genetic modification, and assessment of impacts on key ecosystem services. The data could then be used to refine simulation models to obtain more precise prediction of potential outcomes. These analyses could then be used to propose increasingly larger environmental releases until the trees are either discontinued or deregulated by USDA-APHIS. Similar adaptive management could be employed for biotech trees falling in other bins in a tiered framework. This stepwise approach may be the only practical way to obtain realistic data on gene flow and impacts at the spatial and temporal scales that are needed for proper impact assessment for biotech trees.
In 2008, the U.S. Forest Service modified its procedures to integrate adaptive management into their compliance with the National Environmental Policy Act (NEPA) in 36 CFR Part 220 (USDA-FS, 2008). This adjustment allowed the U.S. Forest Service to implement initial management practices when there was some uncertainty about their impacts on desired endpoints, moni-
___________________
1 The U.S. biotechnology regulatory system for biotech plants as it relates to forest health is discussed in the next chapter. Additional detail on the regulatory system can be found in Chapter 9 of Genetically Engineered Crops: Experiences and Prospects (NASEM, 2016).
tor the effects of the management practices, and then alter those practices as needed. The initial environmental assessment carried out to comply with NEPA has to clearly identify adjustments that could be made in response to monitoring in the adaptive management context.
A good example of implementation of adaptive management by the U.S. Forest Service is provided by the Black Hills Mountain Pine Beetle Response Project (USDA-FS, 2012). This project was focused on mitigating the impacts of the mountain pine beetle (Dendroctonus ponderosae) in the Black Hills National Forest, where an epidemic outbreak of the mountain pine beetle was devastating hundreds of thousands of acres of ponderosa pine (Pinus ponderosa) forest. The public called for mitigating impacts of the beetles through stand management and treatment of infested trees. The main goals were to maintain biodiversity, reduce fire risks from the large amount of accumulated fuel from dead trees, and preserve the scenic beauty of the area. The proposed management intervention was an integrated pest management plan that used a decision tree (see Figure 5-3) to choose among multiple options for stand thinning, treatment of infested trees by burning or chip-
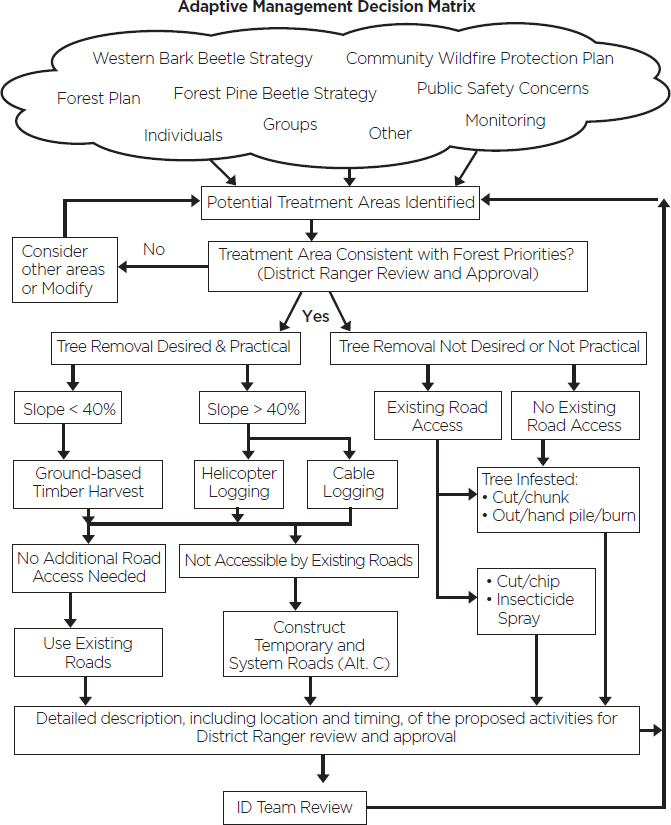
SOURCE: USDA-FS, 2012.
ping, judicious use of insecticides on high value trees, and use of semiochemicals2 to attract beetles to specific trees where they could be destroyed. These latter three treatments were to be conducted on a small scale initially, coupled with monitoring and assessment to determine their efficacy. These treatments would be scaled up or abandoned depending on the outcome. The project included an effectiveness monitoring plan that is implemented by project leaders and interdisciplinary resource specialists on 10 percent of the treated sites annually. The plan was developed and implemented through extensive consultation with state agencies, state government officials, tribes, conservation groups, and an advisory board comprising 16 stakeholders. As of 2018, the beetle epidemic had ended in most parts of the forest. The most recent monitoring report stated that efforts to mitigate the risk of fire to local communities from the beetle-killed trees continued and scenery objectives were on track to be met, but concerns about loss of habitat diversity in the forest prompted recommendations for approaches that would respond adaptively to those concerns (USDA-FS, 2018).
This example illustrates the value of adaptive management in a situation where there are multiple, potentially competing endpoints and uncertainty about the impacts of management interventions. Such circumstances could also be the case for impact assessment of biotech trees modified to improve resistance to insects or diseases. There will be multiple stakeholders with potentially competing interests, and the magnitude and complexity of the problem virtually ensure a great deal of uncertainty about potential impacts of such a tree on ecosystem services. The adaptive management approach allows testing of interventions coupled with careful monitoring to both reduce the risk of unanticipated consequences and to refine the approaches to management based on learning from the initial field trials.
For example, in the case of American chestnut engineered with the oxalate oxidase transgene to resist chestnut blight, if regulators decide that the overall ecological risk is relatively low (e.g., due to extensive experience with the gene involved and the method of the genetic modification and the historic presence of the American chestnut in native forests), a plan might be developed to release engineered chestnuts on a limited basis on a small land area in consultation with stakeholders and informed by extensive simulation modeling. The plots would be monitored for key parameters that were determined by modeling to be important factors controlling the spread of chestnut. These would likely include factors such as blight resistance, growth rate, seed production, seed dispersal, seedling establishment, seedling survival, wildlife usage, soil characteristics, and overall stand biodiversity. The models could be updated with this new information and used, again in consultation with stakeholders, to either scale up the release or to terminate the established trees.
CONCLUSIONS AND RECOMMENDATIONS
Using an impact assessment framework that builds on the National Ecosystem Services Classification System developed by EPA would help those who may develop, approve, and live with biotech trees to minimize the risks of using biotechnology in forest trees while increasing the benefits to forest health. It would help identify the ecosystem services, including those related to cultural and societal values, that are important for addressing forest health within the context of each tree species being modified. Using data from field trials and results from models, impacts on the ecosystem could also be forecasted and evaluated. Adaptive management strategies to adjust the type of resistance introduced or planting decisions would also help minimize risks while increasing benefits.
Conclusion: An integrated impact assessment framework that combines ecological risk assessment with consideration of ecosystem services would provide a way to evaluate impacts of
___________________
2 Semiochemicals are chemical substances or mixtures that carry information between organisms and cause changes in organisms’ behavior. In pest management, they are used to draw insects to traps for monitoring or removal purposes.
introduction of a biotech tree both on the forest functions and on the ecosystem services provided. Societal and cultural values need to be incorporated into this approach.
Impact assessment integrates assessment of potential benefits within a traditional risk assessment framework, weighing the risk of loss of ecosystem services over part, or all, of a species’ range against the potential to recover ecosystem services across that range with and without the biotech intervention. Bringing ecosystem services into impact assessments improves the ability to take into account social and cultural values, which are difficult to quantify and therefore often omitted from impact assessments. It also makes the connection between the protection of forests and human well-being clearer for the public, stakeholders, and policy makers.
Recommendation: Federal agencies should continue efforts to improve the incorporation of all components of ecosystem services into the integrated impact assessment.
Conclusion: Field trials are an important tool to gather data on biotech trees in terms of gene flow, the durability and effectiveness of resistance, seed generation and dispersal, genetic fitness, and some impacts on the ecosystems into which the trees are planted.
Evaluating the success of an introduced resistance trait in trees can be difficult because of their long life spans. Furthermore, success has many dimensions: resistance to the target pest, conversion to the next generation, fitness in the environment, and lack of adverse effects on other species in the environment. Field trials create conditions for observing and measuring the degree of success in these different categories. The long-lived nature of trees and the varied ecosystems in which even a single tree species may occur mean that field trials will need to take place in many locations for a single species. Field trials are also important because of the potential effects of gene flow from biotech trees, and they also will provide information about the feasibility of using biotech trees with resistance to accomplish species restoration or preservation goals.
Conclusion: Modeling efforts will be essential to address the large spatial and temporal scales and stochastic nature of biotech tree impact assessment.
Impact assessment models that use available scientific data can integrate ecological, economic, and cultural considerations, including the benefits of the proposed actions and the consequences of not carrying them out. Synthesis of all available information, data-model integration, and modeling gene flow will aid with making the most accurate and informed predictions of potential impacts. Modeling potential scenarios that include and track sources of uncertainty will allow quantification of the reliability of the assessments, estimation of the predictive capacity of the model, and identification of data needs.
Recommendation: Modeling and other approaches should be developed to address questions about biotech tree gene flow, dispersal, establishment, performance, and impact that are precluded where flowering of field trial material is restricted.
Recommendation: Models for tree biotech impact assessments should identify, quantify, and account for sources of uncertainty.
Conclusion: Iterative decision making is required, such that impact assessments are continually modified with improvements in knowledge gained through on-the-ground experience with biotech tree development, testing, and deployment because of the uncertainty associated with
predictions of the impacts of release of biotech trees into minimally managed or unmanaged environments.
As field trials return more data and models improve, the decisions based on those tools will need to be continually adjusted to ensure that steps being taken to address forest health maximize benefits and minimize risks. Impacts are likely to vary across biotech tree species and will need case-by-case evaluation.
Recommendation: An adaptive management approach to forest health should be used to ensure continued learning and address impacts both to the environment and society.
Recommendation: Impact assessment should be a continuous and iterative process.
REFERENCES
Agapito-Tenfen, S.Z., and F. Wickson. 2018. Challenges for transgene detection in landraces and wild relatives: Learning from 15 years of debate over GM maize in Mexico. Biodiversity and Conservation 27(3):539–566.
Aitken, S.N., and M.C. Whitlock. 2013. Assisted gene flow to facilitate local adaptation to climate change. Annual Review of Ecology, Evolution, and Systematics 44:367–388.
Aukema, J.E., D.G. McCullough, B. Von Holle, A.M. Liebhold, K. Britton, and S.J. Frankel. 2010. Historical accumulation of nonindigenous forest pests in the continental United States. BioScience 60(11):886–897.
Auld, G., and G.Q. Bull. 2003. The institutional design of forest certification standards initiatives and its influence on the role of science: The case of forest genetic resources. Journal of Environmental Management 69(1):47–62.
Barnhill-Dilling, S.K. 2018. Engineered Governance: Public Engagement as a Path to Inclusive Environmental Governance in Genetically Engineered Trees. Ph.D. dissertation, North Carolina State University. Available at https://repository.lib.ncsu.edu/handle/1840.20/35317. Accessed November 21, 2018.
Beaumont, L.J., R.V. Gallagher, W. Thuiller, P.O. Downey, M.R. Leishman, and L. Hughes. 2009. Different climatic envelopes among invasive populations may lead to underestimations of current and future biological invasions. Diversity and Distributions 15(3):409–420.
Beaumont, N.J., R. Mongruel, and T. Hooper. 2017. Practical application of the Ecosystem Service Approach (ESA): Lessons learned and recommendations for the future. International Journal of Biodiversity Science, Ecosystem Services, and Management 13(3):68–78.
Bertrand, P., Z. Pirtle, and D. Tomblin. 2017. Participatory technology assessment for Mars mission planning: Public values and rationales. Space Policy 42(Supplement C):41–53.
Booth, R.K., S. Brewer, M. Blaauw, T.A. Minckley, and S.T. Jackson. 2012. Decomposing the mid-Holocene Tsuga decline in eastern North America. Ecology 93(8):1841–1852.
Bradley, B., D. Wilcove, and M. Oppenheimer. 2010. Climate change increases risk of plant invasion in the Eastern United States. Biological Invasions 12(6):1855–1872.
Buckley, Y.M., D.T. Briese, and M. Rees. 2003. Demography and management of the invasive plant species Hypericum perforatum. II. Construction and use of an individual-based model to predict population dynamics and the effects of management strategies. Journal of Applied Ecology 40:494–507.
Bullock, J.M., R.F. Pywell, and S.J. Coulson-Phillips. 2008. Managing plant population spread: Prediction and analysis using a simple model. Ecological Applications 18(4):945–953.
Caton, B.P., A.L. Koop, L. Fowler, L. Newton, and L. Kohl. 2018. Quantitative uncertainty analysis for a weed risk assessment system. Risk Analysis 38(9):1972–1987.
Chapman, M.A., and J.M. Burke. 2006. Letting the gene out of the bottle: The population genetics of genetically modified crops. New Phytologist 170(3):429–443.
Clark, J.S. 2005. Why environmental scientists are becoming Bayesians. Ecology Letters 8(1):2–14.
Clark, J.S., and A.E. Gelfand. 2006. A future for models and data in ecology. Trends in Ecology & Evolution 21(7):375–380.
Clark, J.S., D. Bell, C. Chu, B. Courbaud, M. Dietze, M. Hersh, J. HilleRisLambers, I. Ibáñez, S. LaDeau, S. McMahon, J. Metcalf, J. Mohan, E. Moran, L. Pangle, S. Pearson, C. Salk, Z. Shen, D. Valle, and P. Wyckoff. 2010. High-dimensional coexistence based on individual variation: A synthesis of evidence. Ecological Monographs 80(4):569–608.
Clark, S., H. McNab, D. Loftis, and S. Zarnoch. 2012. American chestnut growth and survival five years after planting in two silvicultural treatments in the southern Appalachians, USA. Forests 3(4):1017–1033.
Cooper, N., E. Brady, H. Steen, and R. Bryce. 2016. Aesthetic and spiritual values of ecosystems: Recognising the ontological and axiological plurality of cultural ecosystem “services.” Ecosystem Services 21(Part B):218–229.
Covington, W.W. 1981. Changes in forest floor organic matter and nutrient content following clear cutting in northern hardwoods. Ecology 62(1):41–48.
Delborne, J.A., A.R. Binder, L. Rivers, J.C. Barnes, S.K. Barnhill-Dilling, D. George, A. Kokotovich, and J. Sudweeks. 2018. Biotechnology, the American Chestnut Tree, and Public Engagement: Workshop Report. North Carolina State University, Genetic Engineering and Society Center. Available at https://research.ncsu.edu/ges/files/2018/10/Biotech-American-Chestnut-Public-Engagement-2018.pdf. Accessed October 29, 2018.
Denslow, J.S., A.M. Ellison, and R.E. Sanford. 1998. Treefall gap size effects on above-and below-ground processes in a tropical wet forest. Journal of Ecology 86(4):597–609.
Dietrich, W. 1993. The Final Forest: The Battle for the Last Great Trees of the Pacific Northwest. New York: Penguin Books.
Dietz, T. 2013. Bringing values and deliberation to science communication. Proceedings of the National Academy of Sciences of the United States of America 110(Suppl 3):14081–14087.
Dietze, M.C. 2017. Ecological Forecasting. Princeton, NJ: Princeton University Press.
DiFazio, S.P., S. Leonardi, G.T. Slavov, S.L. Garman, W.T. Adams, and S.H. Strauss. 2012. Gene flow and simulation of transgene dispersal from hybrid poplar plantations. New Phytologist 193(4):903–915.
Dillman, D.A., J.D. Smyth, and L.M. Christian. 2014. Internet, Phone, Mail, and Mixed-Mode Surveys: The Tailored Design Method. Hoboken, NJ: Wiley.
Dryzek, J.S., and A. Tucker. 2008. Deliberative innovation to different effect: Consensus conferences in Denmark, France, and the United States. Public Administration Review 68(5):864–876.
Duan, J.J., L.S. Bauer, and R.G. Van Driesche. 2017. Emerald ash borer biocontrol in ash saplings: The potential for early stage recovery of North American ash trees. Forest Ecology and Management 394:64–72.
Dukes, J.S., J. Pontius, D. Orwig, J.R. Garnas, V.L. Rodgers, N. Brazee, B. Cooke, K.A. Theoharides, E.E. Stange, R. Harrington, J. Ehrenfeld, J. Gurevitch, M. Lerdau, K. Stinson, R. Wick, and M. Ayres. 2009. Responses of insect pests, pathogens, and invasive plant species to climate change in the forests of northeastern North America: What can we predict? Canadian Journal of Forest Research-Revue Canadienne De Recherche Forestiere 39(2):231–248.
Ellison, A.M., M.S. Bank, B.D. Clinton, E.A. Colburn, K. Elliott, C.R. Ford, D.R. Foster, B.D. Kloeppel, J.D. Knoepp, G.M. Lovett, J. Mohan, D.A. Orwig, N.L. Rodenhouse, W.V. Sobczak, K.A. Stinson, J.K. Stone, C.M. Swan, J. Thompson, B. Von Holle, and J.R. Webster. 2005. Loss of foundation species: Consequences for the structure and dynamics of forested ecosystems. Frontiers in Ecology and the Environment 3(9):479–486.
Ellstrand, N.C., P. Meirmans, J. Rong, D. Bartsch, A. Ghosh, T.J. de Jong, P. Haccou, B.-R. Lu, A.A. Snow, C.N. Stewart, Jr., J.L. Strasburg, P.H. van Tienderen, K. Vrieling, and D. Hooftman. 2013. Introgression of crop alleles into wild or weedy populations. Annual Review of Ecology, Evolution, and Systematics 44:325–345.
Emry, D.J., H.M. Alexander, and M.K. Tourtellot. 2011. Modelling the local spread of invasive plants: Importance of including spatial distribution and detectability in management plans. Journal of Applied Ecology 48(6):1391–1400.
EPA (U.S. Environmental Protection Agency). 1998. Guidelines for Ecological Risk Assessment. Federal Register 63(93):26846–26924.
EPA. 2006. Ecological Benefits Assessment Strategic Plan. Washington, DC: EPA.
EPA. 2015. National Ecosystem Services Classification System (NESCS): Framework Design and Policy Application. Washington, DC: EPA.
EPA. 2016a. Ecosystem Services as Assessment Endpoints in Ecological Risk Assessment. EPA/100/F/004. Washington, DC: EPA.
EPA. 2016b. EJ 2020 Action Agenda: The U.S. EPA’s Environmental Justice Strategic Plan for 2016–2020. Washington, DC: EPA.
Epanchin-Niell, R.S., J.W. Boyd, M.K. Macauley, L. Scarlett, C.D. Shapiro, and B.K. Williams. 2018. Integrating Adaptive Management and Ecosystem Services Concepts to Improve Natural Resource Management: Challenges and Opportunities. Reston, VA: U.S. Geological Survey.
FAO (Food and Agriculture Organization). 2010. Global Forest Resources Assessment 2010. Rome, Italy: FAO.
Fisher, B, and R.K. Turner. 2008. Ecosystem service: Classification for valuation. Biological Conservation 141(5):1167–1169.
Flory, S.L., K.A. Lorenz, D.R. Gordon, and L.E. Sollenberger. 2012. Experimental approaches for evaluating the invasion risk of biofuel crops. Environmental Research Letters 7(4):045904.
Gandhi, K.J., and D.A. Herms. 2010. Direct and indirect effects of alien insect herbivores on ecological processes and interactions in forests of eastern North America. Biological Invasions 12(2):389–405.
Gómez-Aparicio, L., C.D. Canham, and P.H. Martin. 2008. Neighbourhood models of the effects of the invasive Acer platanoides on tree seedling dynamics: Linking impacts on communities and ecosystems. Journal of Ecology 96(1):78–90.
Gordon, D.R., D.A. Onderdonk, A.M. Fox, and R.K. Stocker. 2008. Consistent accuracy of the Australian weed risk assessment system across varied geographies. Diversity and Distributions 14(2):234–242.
Guston, D.H. 1999. Evaluating the first U.S. consensus conference: The impact of the citizens’ panel on telecommunications and the future of democracy. Science, Technology and Human Values 24(4):451–482.
Häggman, H., A. Raybould, A. Borem, T. Fox, L. Handley, M. Hertzberg, M.Z. Lu, P. Macdonald, T. Oguchi, G. Pasquali, and L. Pearson. 2013. Genetically engineered trees for plantation forests: Key considerations for environmental risk assessment. Plant Biotechnology Journal 11(7):785–798.
Hansen, A.J., R.R. Neilson, V.H. Dale, C.H. Flather, L.R. Iverson, D J. Currie, S. Shafer, R. Cook, and P.J. Bartlein. 2001. Global change in forests: Responses of species, communities, and biomes. BioScience 51(9):765–779.
Hayes, K.R., H.M. Regan, and M.A. Burgman. 2007. Introduction to the concepts and methods of uncertainty analysis. Pp. 188–208 in Environmental Risk Assessment of Genetically Modified Organisms. Volume 3. Methodologies for Transgenic Fish in Developing Countries, A.R. Kapuscinski, K.R. Hayes, S. Li, and G. Dana, eds. Wallingford, UK: CAB International.
Hoenicka, H., and M. Fladung. 2006. Biosafety in Populus spp. and other forest trees: From non-native species to taxa derived from traditional breeding and genetic engineering. Trees 20(2):131–144.
Holling, C.S. 1978. Adaptive Environmental Assessment and Management. Chichester, UK: John Wiley & Sons.
Ibáñez, I., J.A. Silander, A.M. Wilson, N. LaFleur, N. Tanaka, and I. Tsuyama. 2009. Multivariate forecasts of potential distributions of invasive plant species. Ecological Applications 19(2):359–375.
Ibáñez, I., J.M. Diez, L.P. Miller, J.D. Olden, C.J.B. Sorte, D.M. Blumenthal, B.A. Bradley, C.M. D’Antonio, J.S. Dukes, R.I. Early, E.D. Grosholz, and J.J. Lawler. 2014. Integrated assessment of biological invasions. Ecological Applications 24(1):25–37.
Johnston, R.J. 2018. Forest Health and Biotechnology within an Ecosystem Services Framework. Presentation to National Academies of Sciences, Engineering, and Medicine’s Committee on the Potential for Biotechnology to Address Forest Health, February 8, Washington, DC.
Joss, S. 1998. Danish consensus conferences as a model of participatory technology assessment: An impact study of consensus conferences on Danish parliament and Danish public debate. Science and Public Policy 25(1):2–22.
Keane, R.M., and M.J. Crawley. 2002. Exotic plant invasions and the enemy release hypothesis. Trends in Ecology & Evolution 17(4):164–170.
Kinchy, A.J. 2012. Seeds, Science, and Struggle: The Global Politics of Transgenic Crops. Cambridge, MA: MIT Press.
Kinloch, B.B., Jr., D.A. Davis and D. Burton. 2008. Resistance and virulence interactions between two white pine species and blister rust in a 30-year field trial. Tree Genetics & Genomes 4(1):65–74.
Kleinman, D.L., M. Powell, J. Grice, J. Adrian, and C. Lobes. 2007. A toolkit for democratizing science and technology policy: The practical mechanics of organizing a consensus conference. Bulletin of Science, Technology & Society 27(2):154–169.
Kleinman, D.L., J.A. Delborne, and A.A. Anderson. 2011. Engaging citizens: The high cost of citizen participation in high technology. Public Understanding of Science 20(2):221–240.
Koop, A.L., L. Fowler, L.P. Newton, and B.P. Caton. 2012. Development and validation of a weed screening tool for the United States. Biological Invasions 14(2):273–294.
Kowarik, I. 1995. Time lags in biological invasions with regard to the success and failure of alien species. Pp. 15–38 in Plant Invasions—General Aspects and Special Problems, P. Pysek, M. Rejmanek, and M. Wade, eds. Amsterdam, The Netherlands: SPB Academic.
Kremen, C. 2005. Managing ecosystems services: What do we need to know about their ecology? Ecology Letters 8(5):468–479.
Kuparinen, A., and F.M. Schurr. 2007. Assessing the risk of gene flow from genetically modified trees carrying mitigation transgenes. Biological Invasions 10(3):281–290.
LaFleur, N., C. Merow, M. Rubega, and J. Silander. 2009. Predicting the rate of spread for a bird-dispersed invasive plant using simulation modeling. Integrative and Comparative Biology 49:E96.
Linacre, N.A., and P.K. Ades. 2004. Estimating isolation distances for genetically modified trees in plantation forestry. Ecological Modeling 179(3):247–257.
Lorentz, K.A., and P.J. Minogue. 2015. Exotic Eucalyptus plantations in the southeastern US: Risk assessment, management and policy approaches. Biological Invasions 17(6):1581–1593
Lovett, G.M., M. Weiss, A.M. Liebhold, T.P. Holmes, B. Leung, K.F. Lambert, D.A. Orwig, F.T. Campbell, J. Rosenthal, D.G. McCullough, R. Wildova, M.P. Ayres, C.D. Canham, D.R. Foster, S.L. LaDeau, and T. Weldy. 2016. Nonnative forest insects and pathogens in the United States: Impacts and policy options. Ecological Applications 26(5):1437–1455.
Luo, Y., K. Ogle, C. Tucker, S. Fei, C. Gao, S. LaDeau, J.S. Clark, and D.S. Schimel. 2011. Ecological forecasting and data assimilation in a data-rich era. Ecological Applications 21(5):1429–1442.
Mack, R.N., D. Simberloff, W.M. Lonsdale, H. Evans, M. Clout, and F.A. Bazzaz. 2000. Biotic invasions: Causes, epidemiology, global consequences, and control. Ecological Applications 10(3):689–710.
Mantyniemi, S., L. Uusitalo, H. Peltonen, P. Haapasaari, and S. Kuikka. 2013. Integrated, age-structured, length-based stock assessment model with uncertain process variances, structural uncertainty, and environmental covariates: Case of Central Baltic herring. Canadian Journal of Fisheries and Aquatic Sciences 70(9):1317–1326.
Marco, D.E., M.A. Montemurro, and S.A. Cannas. 2011. Comparing short and long-distance dispersal: modelling and field case studies. Ecography 34(4):671–682.
Maron, J.L., and M. Vilà. 2001. When do herbivores affect plant invasion? Evidence for the natural enemies and biotic resistance hypotheses. Oikos 95(3):361–373.
McDougall, K.L., A.A. Khuroo, L.L. Loope, C.G. Parks, A. Pauchard, Z.A. Reshi, I. Rushworth, and C. Kueffer. 2011. Plant invasions in mountains: Global lessons for better management. Mountain Research and Development 31(4):380–387.
MEA (Millennium Ecosystem Assessment). 2005. Ecosystems and Human Well-Being: Synthesis. Washington, DC: Island Press.
Meirmans, P.G., J. Bousquet, and N. Isabel. 2009. A metapopulation model for the introgression from genetically modified plants into their wild relatives. Evolutionary Applications 2(2):160–171.
Mellert, K.H., V. Deffner, H. Kuchenhoff, and C. Kolling. 2015. Modeling sensitivity to climate change and estimating the uncertainty of its impact: A probabilistic concept for risk assessment in forestry. Ecological Modelling 316:211–216.
Millar, C.I., N.L. Stephenson, and S.L. Stephens. 2007. Climate change and forests of the future: Managing in the face of uncertainty. Ecological Applications 17(8):2145–2151.
Morin, X., and W. Thuiller. 2009. Comparing niche- and process-based models to reduce prediction uncertainty in species range shifts under climate change. Ecology 90(5):1301–1313.
Munns, W.R. Jr., A.W. Rea, G.W. Suter, L. Martin, L. Blake-Hedges, T. Crik, C. Davis, G. Ferreira, S. Jordan, M. Mahoney, and M.G. Barron. 2016. Ecosystem services as assessment endpoints for ecological assessment. Integrated Environmental Assessment and Management 12(3):522–528.
NASEM (National Academies of Sciences, Engineering, and Medicine). 2016. Genetically Engineered Crops: Experiences and Prospects. Washington, DC: The National Academies Press.
NASEM. 2017. Preparing for Future Products of Biotechnology. Washington, DC: The National Academies Press.
Nathan, R. 2006. Long-distance dispersal plants. Science 313(5788):786–788.
National Ecosystem Services Partnership. 2018. Federal Resource Management and Ecosystem Services Online Guidebook. Available at http://www.nespguidebook.com. Accessed July 14, 2018.
Nijnik, M., and D. Miller. 2017. Valuation of ecosystem services: Paradox or Pandora’s box for decision-makers? One Ecosystem 2:e14808.
NRC (National Research Council). 1996. Understanding Risk: Informing Decisions in a Democratic Society. Washington, DC: National Academy Press.
NRC. 2005. Valuing Ecosystem Services: Toward Better Environmental Decision-making. Washington, DC: The National Academies Press.
Olander, L., R.J. Johnston, H. Tallis, J. Kagan, L. Maguire, S. Polasky, D. Urban, J. Boyd, L. Wainger, and M. Palmer. 2015. Best Practices for Integrating Ecosystem Services into Federal Decision Making. Durham, NC: National Ecosystem Services Partnership, Duke University.
Paillet, F.L. 2002. Chestnut: History and ecology of a transformed species. Journal of Biogeography 29(10–11):1517–1530.
Pearce, D., G. Atkinson, and S. Mourato. 2006. Cost Benefit Analysis and the Environment: Recent Developments. Paris, France: Organisation for Economic Co-operation and Development.
Pheloung, P.C., P.A. Williams, and S.R. Halloy. 1999. A weed risk assessment model for use as a biosecurity tool evaluating plant introductions. Journal of Environmental Management 57(4):239–251.
Poland, T.M., M.R. Emery, T. Ciaramitaro, E. Pigeon, and A. Pigeon. 2017. Emerald ash borer, black ash, and Native American basketmaking; Invasive insects, forest ecosystems and cultural practices. Pp. 127–140 in Biodiversity, Conservation, and Environmental Management in the Great Lakes Basin, E. Freedman and M. Meuzil, eds. Abingdon, UK: Routledge.
Rask, M., and R. Worthington. 2015. Governing Biodiversity through Democratic Deliberation. New York: Routledge.
Rask, M., R. Worthington, and M. Lammi, eds. 2012. Citizen Participation in Global Environmental Governance. London, UK: Earthscan Publications.
Reinhart, K.O., A. Packer, W.H. van der Putten, and K. Clay. 2003. Plant-soil biota interactions and spatial distribution of black cherry in its native and invasive ranges. Ecology Letters 6(12):1046–1050.
Rinella, M.J., and E.C. Luschei. 2007. Hierarchical Bayesian methods estimate invasive weed impacts at pertinent spatial scales. Biological Invasions 9(5):545–558.
Robledo-Arnuncio, J.J., E.K. Klein, H.C. Muller-Landau, and L. Santamariá. 2014. Space, time and complexity in plant dispersal ecology. Movement Ecology 2(1):16.
Rogers, B.M., P. Jantz, and S.J. Goetz. 2017. Vulnerability of eastern US tree species to climate change. Global Change Biology 23(8):3302–3320.
Rowe, G., and L.J. Frewer. 2005. A typology of public engagement mechanisms. Science, Technology and Human Values 30(2):251–290.
Savolainen, O., T. Pyhäjärvi, and T. Knürr. 2007. Gene flow and local adaptation in trees. Annual Review of Ecology, Evolution, and Systematics 38:595–619.
Schneider, J., and J. Delborne. 2012. Seeking the spotlight: World wide views and the U.S. media context. Pp. 241–260 in Citizen Participation in Global Environmental Governance, M. Rask, R. Worthington, and M. Lammi, eds. London, UK: Earthscan.
Sclove, R. 2010a. Why the polls on climate change are wrong, March 18. Available at https://www.huffingtonpost.com/richard-sclove-phd/why-the-polls-on-climate_b_331896.html. Accessed December 4, 2018.
Sclove, R. 2010b. Reinventing technology assessment. Issues in Science and Technology 27(1):34–38.
Scorza, R., A.B. Kriss, A.M. Callahan, K. Webb, M. Demuth, and T. Gottwald. 2013. Spatial and temporal assessment of pollen- and seed-mediated gene flow from genetically engineered plum Prunus domestica. PLoS One 8(10):e75291.
Shvidenko, A., C.V. Barber, and R. Persson. 2005. Forest and woodland systems. Pp. 587–621 in Ecosystems and Human Well-being: Current State and Trends, Vol 1, R. Hassan, R. Scholes, and N. Ash, eds. Washington, DC: Island Press.
Simberloff, D. 2011. How common are invasion-induced ecosystem impacts? Biological Invasions 13(5):1255–1268.
Slavov, G.T., S. Leonardi, J. Burczyk, W.T. Adams, S.H. Strauss, and S.P. DiFazio. 2009. Extensive pollen flow in two ecologically contrasting populations of Populus trichocarpa. Molecular Ecology 18(2):357–373.
Sniezko, R.A., R. Danchok, J. Hamlin, A. Kegley, S. Long, and J. Mayo. 2012. White pine blister rust resistance of 12 western white pine families at three field sites in the Pacific Northwest. Pp. 356–367 in Proceedings of the Fourth International Workshop on the Genetics of Host–Parasite Interactions in Forestry: Disease and Insect Resistance in Forest Trees, R.A. Sniezko, A.D. Yanchuk, J.T. Kliejunas, K.M. Palmieri, J.M. Alexander, and S.J. Frankel, tech. cords. Albany, CA: USDA-FS.
Snow, A.A. 2002. Transgenic crops—why gene flow matters. Nature Biotechnology 20(6):542.
Strauss, S.H., A. Costanza, and A. Séguin. 2015. Genetically engineered trees: Paralysis from good intentions. Science 349(6250):794–795.
Strauss, S.H., D.L. Kershen, J.H. Bouton, T.P. Redick, H. Tan, and R.A. Sedjo. 2010. Far-reaching deleterious impacts of regulations on research and environmental studies of recombinant DNA-modified perennial biofuel crops in the United States. BioScience 60(9):729–741.
Suter, G.W., II. 2000. Generic assessment endpoints are needed for ecological risk assessments. Risk Analysis 20(2):173–178.
Tsatsakis, A.M., M.A. Nawaz, D. Kouretas, G. Balias, K. Savolainen, V.A. Tutelyan, K.S. Golokhvast, J.D. Lee, S.H. Yang, and G. Chung. 2017. Environmental impacts of genetically modified plants: A review. Environmental Research 156:818–833.
USDA-FS (U.S. Department of Agriculture’s Forest Service). 2008. National Environmental Policy Act Procedures. Federal Register 73(143):43084–43099.
USDA-FS. 2012. Mountain Pine Beetle Response Project: Record of Decision. Available at https://www.fs.usda.gov/nfs/11558/www/nepa/81228_FSPLT2_291923.pdf. Accessed November 24, 2018.
USDA-FS. 2018. Mountain Pine Beetle Response Project Monitoring Report FY 2016 and 2017. Available at https://www.fs.usda.gov/nfs/11558/www/nepa/81228_FSPLT3_4273675.pdf. Accessed November 24, 2018.
Uusitalo, L., A. Lehikoinen, I. Helle, and K. Myrberg. 2015. An overview of methods to evaluate uncertainty of deterministic models in decision support. Environmental Modelling & Software 63:24–31.
Vanclay, F. 2003. International principles for social impact assessment. Impact Assessment and Project Appraisal 21(1):5–12.
Vaske, J.J. 2008. Survey Research and Analysis: Applications in Parks, Recreation and Human Dimensions. State College, PA: Venture.
Vose, J.M., C.F. Miniat, G. Sun, and P.V. Caldwell. 2015. Potential implications for expansion of freeze-tolerant eucalyptus plantations on water resources in the southern United States. Forest Science 61(3):509–521.
Walters, C.J., and R Hilborn. 1978. Ecological optimization and adaptive management. Annual Review of Ecology and Systematics 9(1):157–188.
Wikle, C.K. 2015. Hierarchical models for uncertainty quantification: An overview. In Handbook of Uncertainty Quantification, R. Ghanem, D. Higdon, and D. Owhadi, eds. Switzerland: Springer International.
Williams, B.K. 2011. Passive and active adaptive management: Approaches and an example. Journal of Environmental Management 92(5):1371–1378.
Williams, C.G. 2010. Long-distance pine pollen still germinates after meso-scale dispersal. Reproductive Biology 97(5):846–855.
This page intentionally left blank.


























