6
Current Regulatory System for Biotech Trees and Other Methods Used to Address Forest Health
Consistent with the statement of task for this report, this chapter sets forth the U.S. federal regulatory system for any biotech trees as of 2018, including biotech trees developed to address forest health problems. It then analyzes whether that regulatory system, as currently constituted, evaluates the issues that are encompassed by this report’s definition of forest health and ecosystem service components when making regulatory decisions on those biotech trees. The chapter also sets forth the federal regulatory system for other human interventions that attempt to address forest health that are not a biotech tree (e.g., pesticides and biocontrol agents) and whether those regulatory systems make approval decisions that take into account the broad range of issues in this report’s definition of forest health.
Biotech trees developed to address forest health are regulated under the same statutes and regulations as any biotech plant, including commercial biotech trees (such as the virus-resistant papaya, nonbrowning apple, or cold-tolerant eucalyptus). That regulatory system was established in 1986 when the White House’s Office of Science and Technology Policy published the “Coordinated Framework for the Regulation of Biotechnology” (Coordinated Framework), which is not a statute or regulation but a federal policy statement that established principles on how the federal government regulates biotechnology products. The Coordinated Framework stated that different agencies in the federal government should apply existing statutes to biotechnology products (OSTP, 1986).1 The Coordinated Framework was partially updated in 1992 to provide further policy guidance to federal agencies and more comprehensively updated in 2017 to provide further clarity and transparency to the public and interested stakeholders (EOP, 2017).2 Under the Coordinated Framework, as many as three different federal agencies—the U.S. Department of Agriculture (USDA), the U.S.
___________________
1 The Coordinated Framework presents information about the agencies’ roles and responsibilities in several forms such as graphics that illustrate agency-specific roles and a table summarizing the responsibilities for different product categories.
2 A detailed discussion of the history of the Coordinated Framework and the policies it established can be found in the National Academies reports Genetically Engineered Crops: Experiences and Prospects (NASEM, 2016) and Preparing for Future Products of Biotechnology (NASEM, 2017). The controlling document is the 2017 version of the Coordinated Framework.
Environmental Protection Agency (EPA), and the U.S. Food and Drug Administration (FDA)—could regulate a biotech tree developed to address forest health.
It is important to note that biotechnological approaches have advanced since the Coordinated Framework was established as is acknowledged in the “National Strategy for Modernizing the Regulatory System for Biotechnology Products” (EOP, 2016), which the White House released in September 2016. The National Strategy mentioned that the federal government would provide additional guidance on how it will regulate products produced through new biotechnologies such as genome editing3 because the different federal statutes grant each agency authority to regulate specific products and activities or uses of those products, not the process by which they are produced. The application of the Coordinated Framework to specific products means that biotech trees and plants may be regulated by zero, one, two, or three or more agencies.4
U.S. DEPARTMENT OF AGRICULTURE
USDA is the first or primary agency that regulates some biotech plants. It regulates the import, interstate movement, transport, and environmental release of biotech plants that fall under the Code of Federal Regulations (CFR), specifically found at 7 CFR Part 340 and issued under the Plant Protection Act as amended December 23, 2004, 7 U.S.C. § 7701 et seq.5 Those regulations ensure that these plants are not “plant pests,” which are defined as “any living stage of a pest that can directly or indirectly injure, cause damage to, or cause disease in any plant or plant product” (7 CFR § 340.1). Biotech plants could become a plant pest if they carry the genes or DNA of species that have been determined to cause injury to other plants (e.g., when Agrobacterium-mediated transformation is used to introduce desired genes; Gelvin, 2003). USDA interprets that legal authority so that a biotech plant or tree is regulated if
- the biotech plant or tree has pests that are on USDA’s list of plant pests,
- the process of introducing the change in the plant or tree’s genome involves an organism on the list of plant pests (such as Agrobacterium-mediated transformation), or
- any of the introduced DNA (the gene, promotor, terminator, etc.) came from an organism on the list of plant pests (7 CFR § 340.1).
Under those regulations, any regulated biotech plants must be submitted to one of three oversight processes before that plant can be released into the environment. The first process, known as “notification,” is used for limited field trials of a biotech plant that meets certain eligibility criteria and field trial containment standards. The applicant provides USDA with a notification detailing the release, and USDA has 30 days to respond (USDA-APHIS, 2011). As many as 1,000 field trials, mostly for grains, fruits, and vegetables are authorized each year using this procedure. In 2008, USDA determined “based upon accumulated regulatory experience and currently available science” that it would no longer accept notifications for environmental releases lasting more than 1 year (USDA-APHIS, 2008). Thus, no biotech tree that falls under USDA’s jurisdiction can qualify
___________________
3 The National Strategy set forth a commitment by USDA, EPA, and FDA to clarify oversight of new biotechnology products, such as products produced using genome editing and genetically engineered insects.
4 The Coordinated Framework identifies agencies other than USDA, EPA, and FDA that would regulate different products of biotechnology. More discussion about other agencies can be found in Chapter 3 of the National Academies’ report Preparing for Future Products of Biotechnology (NASEM, 2017).
5 Available at https://www.aphis.usda.gov/plant_health/downloads/plant-protect-act.pdf. Accessed July 19, 2018. USDA has twice proposed revisions to 7 CFR Part 340, once in 2008 and again in 2017; both proposals were later withdrawn. Until 7 CFR Part 340 is changed, the regulatory system described in this chapter is what will be applied to trees developed with biotechnology.
for the notification process since it is virtually impossible to complete a field trial with a tree in less than 365 days.
The second process to get permission for a limited release of a biotech plant is the USDA “permitting” process. That process requires a more detailed application and a longer review time at USDA before the release is authorized (USDA-APHIS, 2017e). Permitting is not as common as the notification, although USDA has issued hundreds of permits since it began regulating biotech crops (USDA-APHIS, 2017b). Since 2008, all experiments with biotech trees that are regulated under 7 CFR Part 340, independent of whether those trees are developed to address forest health, require a permit before any release into the environment (USDA-APHIS, 2008).
The third process is a called a “petition for non-regulated status,” where a developer requests that USDA determine that there is no plant pest risk from the biotech plant, and it is no longer regulated (USDA-APHIS, 2016). The petition process is the primary path to commercialization, and more than 124 crops have been deregulated. As of 2018, three biotech fruit trees—transgenic papaya and plum6 and apple transformed via RNA interference—have successfully been granted nonregulated status, but no biotech plantation trees or trees with traits to address forest health had been granted that status. As the time the committee was writing its report, one petition for a eucalyptus tree (Eucalyptus urograndis) is pending but that tree is not being developed to address forest health (USDA-APHIS, 2018b).
To decide whether to grant a petition for nonregulated status, USDA’s regulatory review is limited to determining whether the biotech plant poses a “plant pest” risk. The risk assessment process (NASEM, 2016:475)
considers, among other things, whether the [genetically engineered (GE)] crop is more likely than its non-GE comparator to become invasive or weedy, to be more susceptible to pests or diseases, or to have greater effects on nontarget organisms. [USDA’s Animal and Plant Health Inspection Service (APHIS)] also considers the potential effects of gene flow to wild relatives and other organisms. In effect, APHIS uses the risk-assessment process to determine whether a GE crop is likely to pose a greater “plant pest” risk than a comparable conventionally bred crop variety.
According to USDA, that review process culminates in a document, the Plant Pest Risk Assessment, which is the primary document used to determine whether to grant the petition. That document is not specifically required to address broad forest health concerns, only plant pest concerns. However, if forest health would be impacted because the biotech tree might have a plant pest concern, such as it could become invasive or have an impact on nontarget organisms, then those issues would be assessed in the Plant Pest Risk Assessment.7
In addition to addressing any plant pest issues with a biotech plant, any decisions by USDA to issue a permit for a field trial or to grant a petition for nonregulated status also must comply with the National Environmental Policy Act (NEPA), 42 U.S.C. § 4321 et seq.8 NEPA was established so that the government and the public would be aware of the environmental impact of government actions. Thus, NEPA requires federal agencies to assess the environmental impact of all major
___________________
6 Plum resistant to plum pox virus has been deregulated but had not been planted commercially in the United States as of 2018.
7 Although the Plant Pest Risk Assessment considers many things as described here and for which information is requested as part of the petition process as defined in 7 CFR Part 340.6, the regulations at 7 CFR Part 340 do not grant USDA authority to regulate under 7 CFR Part 340 on the basis of weediness. At the time the committee was writing its report, regulatory authority for 7 CFR Part 340 was based on the plant pest provisions of the Plant Protection Act, not the noxious weed provisions. Aspects of forest health could be addressed in the Plant Pest Risk Assessment or in the accompanying NEPA document.
8 Available at https://www.gpo.gov/fdsys/pkg/CFR-2017-title40-vol37/xml/CFR-2017-title40-vol37-sec1508-8.xml. Accessed July 19, 2018.
federal actions and make that assessment public. That analysis covers the effects on the “human environment,” which is interpreted comprehensively to include the natural and physical environment and the relationship of people to that environment (42 U.S.C. § 4332(c)). Effects (and impacts) include
ecological (such as the effects on natural resources and on the components, structures, and functioning of affected ecosystems), aesthetic, historic, cultural, economic, social, or health, whether direct, indirect, or cumulative. Effects may also include those resulting from actions which may have both beneficial and detrimental effects, even if on balance the agency believes that the effect will be beneficial. (40 CFR § 1508.8)9
For each permit or grant of nonregulated status by USDA, compliance with NEPA can consist of conducting an environmental assessment (EA) and a finding of no significant impact (FONSI). However, if the agency cannot make a FONSI determination after the completion of the EA, it must conduct a more detailed and time-consuming environmental impact statement (EIS) and a record of decision (ROD). The agency also can bypass an EA and directly conduct an EIS.10 Although agencies must go through the NEPA assessment process, NEPA does not give agencies any authority to make substantive decisions based on the results of their environmental assessment. In other words, NEPA requires an assessment of impacts but does not provide any basis for denying or modifying a government action based on that assessment. The EA or EIS provides information about impacts but does not change the government agency’s proposed action unless the statute under which the agency is carrying out its activity or action itself allows the agency to take into account the environmental analysis in making its decision.
USDA has received more than 21,000 requests under either the notification or permitting process to authorize field trials with a biotech organism that could be a potential plant pest. Of that amount, there have been approximately 1,329 notifications received by USDA for such trees and 1,191 have been acknowledged and allowed to proceed (see Figure 6-1). USDA has also received approximately 441 permit applications for biotech trees and issued 387 permits (see Figure 6-2). It has issued 220 permits to biotech nonfruit tree species and 167 permits for biotech fruit trees (USDA-APHIS, 2018a). For the permits issued for biotech trees, only 17 of them required an EA under NEPA, 5 fruit tree applications and 12 nonfruit tree applications (see Figure 6-3). Therefore, USDA completed an EA and FONSI for approximately 4 percent of its permit decisions for biotech trees. It should be noted that no field trial with a biotech forest tree (such as the transgenic chestnut) has required an EA for a field trial. All 12 EAs for nonfruit trees involved commercial plantation species (USDA-APHIS, 2017c).
As of May 2018, USDA had processed 124 petitions for nonregulated status for biotech plants that could pose a plant pest risk. To make decisions on those petitions and comply with NEPA, USDA generated 110 EAs with a FONSI and 8 EISs (the remaining 5 petitions had a FONSI that used an existing EA from a previous petition). Six of the 124 petitions involved tree species—5 for fruit trees and 1 for eucalyptus—and USDA carried out 4 EAs for the fruit trees (one petition received a FONSI using an EA from an earlier petition) and a draft EIS for the eucalyptus. Therefore, USDA carries out an EIS for a small minority of the petitions it receives, although the one nonfruit tree petition did require an EIS (USDA-APHIS, 2018b).
To the extent that USDA is going to assess forest health issues involving biotech trees, it is likely to occur in its compliance with NEPA (except for plant pest issues that address forest health, which are analyzed in a Plant Pest Risk Assessment). USDA has not completed EIS for a native bio-
___________________
9 Available at https://www.gpo.gov/fdsys/pkg/CFR-2017-title40-vol37/xml/CFR-2017-title40-vol37-sec1508-8.xml. Accessed July 19, 2018.
10 Permits and notifications can also comply with NEPA (7 CFR Part 372) through a categorical exclusion.
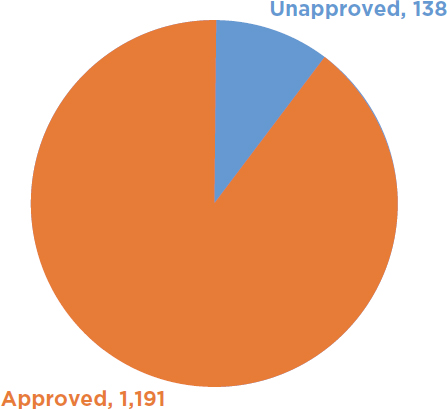
NOTE: Unapproved notifications are those submitted to USDA but that are not acknowledged and allowed to proceed.
SOURCE: Data from USDA-APHIS, 2018a.
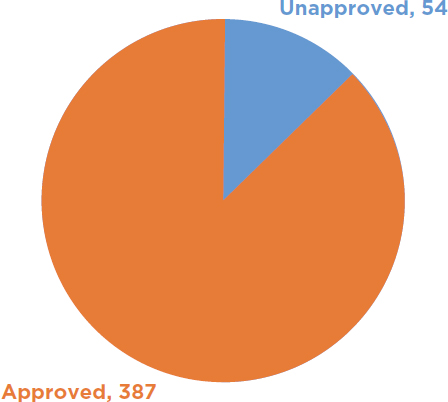
NOTE: Unapproved permits are those permit applications submitted to USDA but for which permits are not issued.
SOURCE: Data from USDA-APHIS, 2018a.
tech forest tree, but it did complete a draft EIS for a frost-tolerant eucalyptus tree (USDA-APHIS, 2017a) (see Box 6-1). Although for a commercial plantation tree, that draft EIS can provide some insight into what an EIS for a biotech tree developed for addressing forest health might cover.
Not all biotech plants or trees are regulated under 7 CFR Part 340, which only applies to potential “plant pests.” Since around 2010, USDA has utilized its “Am I regulated” procedure to
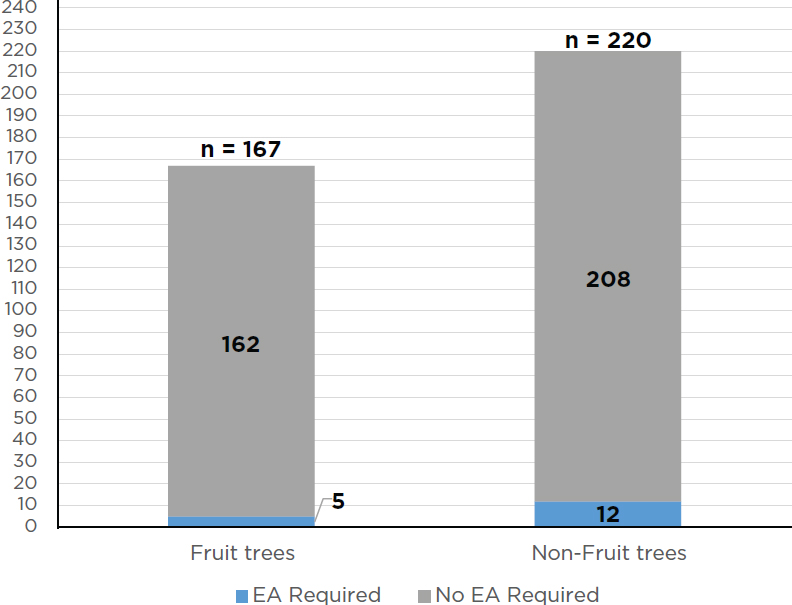
SOURCE: Data from USDA-APHIS, 2018a.
identify approximately 68 biotech plants that do not involve plant pests and need not comply with 7 CFR Part 340. That includes biotech plants developed using biolistics as well as many plants whose genomes were altered with genome-editing techniques such as zinc finger nucleases, transcription activator-like effector nucleases, and the clustered regularly interspaced short palindromic repeat (CRISPR)-Cas9 nuclease system. It also includes genome-edited plants that use Agrobacterium to introduce the genome-editing cassette as long as the final product contains no introduced genetic material. At the time the committee was writing its report, countries around the world were in the process of determining how agricultural products produced using genome-editing techniques would be regulated, if at all. The European Court of Justice ruled in July 2018 that those products would be considered “GMOs” under the European Union’s GMO Directive 2001/18 and would not qualify for an exemption from the substantive requirements of that Directive as is the case for products produced with chemical mutagenesis or irradiation. Thus, this decision means that genome-edited agricultural products will be regulated very differently in the European Union than how many genome-edited crops are regulated at USDA, which could have significant trade implications.11 As of August 2018, USDA has received one “Am I regulated” request for a biotech tree, a loblolly pine, and found that it did not fall within USDA’s oversight because the added genetic material did not involve any plant pest and the transfer of genetic material was achieved by biolistics (USDA-APHIS, 2018c). Unless the regulations in 7 CFR Part 340 are revised, it is safe to assume that there will be future biotech trees, including trees developed to address forest health, that will not
___________________
11 Judgment of 25 July 2018, Confédération paysanne and others, C-528/16, EU:C:2018:583.
be required to obtain a permit or the granting of nonregulated status before that tree is released into the environment.
In conclusion, not all biotech trees developed to address forest health will be regulated by USDA under its current regulations at 7 CFR Part 340 under the authority of the plant pest provisions of the Plant Protection Act. If a biotech tree falls within USDA’s legal mandate involving “plant pests,” then under the regulations it will require a permit for field trials that exceed 1 year as it would not qualify for the quicker “notification” process. It will also need to petition for nonregulated status where the agency will conduct a Plant Pest Risk Assessment and comply with NEPA and other relevant laws, executive orders, and treaties. USDA’s review of any application under its plant pest authority might include some aspects of forest health. That review would be due to the unique biological characteristics of trees and the characteristics of the genetic modifications done to those trees if related to plant pest risk and not because USDA specifically considers the full suite of forest health issues in its Plant Protection Act decision process. USDA only considers forest health issues in the Plant Pest Risk Assessment if those issues are also “plant pest” issues, such as weediness or impacts on nontarget organisms. USDA may consider some forest health issues in its compliance with NEPA, but that analysis is much more likely when USDA conducts an EIS and not the less burdensome EA. However, USDA has only conducted an EIS on a few of the petitions it has granted and only one on a biotech tree (out of six applications). Similarly, USDA only conducts an EA for a small number of permit applications and has only conducted such an EA on approximately 4 percent of the biotech trees it has permitted. Therefore, since the primary way that USDA’s oversight under the Plant Protection Act considers forest health issues is through compliance with NEPA, and the historical rate of conducting an EIS and EA for a biotech tree is very low, then without substantial revision to regulatory statues, guidelines, or practice, most biotech trees will not be comprehensively evaluated for their impacts on forest health, as defined in this report. In addition, the NEPA process is procedural, so it does not provide USDA with any authority to address forest health impacts that are identified in an EA or EIS.
U.S. ENVIRONMENTAL PROTECTION AGENCY
EPA regulates some biotech plants under the Federal Insecticide, Fungicide, and Rodenticide Act (FIFRA, 7 U.S.C. § 135 et seq.) and the Federal Food, Drug, and Cosmetic Act.12 Under FIFRA, EPA regulates the sale, distribution, and use of pesticides through a premarket mandatory registration process, and plants that have a protectant incorporated into them using biotechnology (called “plant incorporated protectants” or PIPs) are captured under regulations found in 40 CFR Parts 152 and 174. According to those regulations, “a pesticide is a substance or mixture intended to prevent, destroy, repel or mitigate any pest” (40 CFR § 152.3),13 and a PIP “is a pesticidal substance intended to be produced and used in a living plant and the genetic material necessary for its production” (40 CFR § 174.3).14 For those products, “registration requires an evidence-based premarket review in which product sponsors submit evidence to demonstrate that the product will not cause unreasonable adverse effects on the environment under its proposed conditions of use” (NASEM, 2017:90). The scope of the evaluation of a PIP by EPA is no different from its evaluation of more traditional chemical or biological pesticides. EPA addresses environmental impacts as
___________________
12 EPA’s role under the Federal Food, Drug, and Cosmetic Act involves determining a tolerance level for any pesticide residues that would remain on human food. This portion of EPA’s mandate has little applicability for biotech forest trees, and so it is not discussed in detail in this chapter.
13 Available at https://www.gpo.gov/fdsys/granule/CFR-2012-title40-vol25/CFR-2012-title40-vol25-sec152-3. Accessed July 20, 2018.
14 Available at https://www.gpo.gov/fdsys/granule/CFR-2013-title40-vol25/CFR-2013-title40-vol25-sec174-3. Accessed July 20, 2018.
well as impacts on humans and other species (such as insects and aquatic organisms). As stated in more detail below, that assessment covers some ecological aspects of forest health but misses the less quantitative ecosystem services a healthy forest provides (see section “Regulation of the Use of Conventional Pesticides to Address Forest Health,” below).
If a biotech tree is a PIP under EPA regulations—for example, if a Bacillus thuringiensis (Bt) gene were added to a poplar to confer protection against some insect pests—then it would be regulated under FIFRA. As of 2018, EPA has reviewed and registered two biotech fruit trees under its PIP process: a honeysweet plum with a plum pox viral coat protein gene and a papaya tree with a papaya ringspot virus coat protein gene (EPA, 2010, 2015, 2017). A review of the EPA decision documents for those two products does not reveal any special environmental considerations or data requirements solely because the protectant is integrated into a tree species instead of a grain or vegetable crop. In fact, the documents supporting those two registration decisions involve less data and a shorter overall assessment than for Bt crops. However, those two examples are not necessarily predictive about how EPA would review a protectant in a forest tree. It should also be noted that when EPA registers a pesticide, there is continuing oversight responsibility and the potential that the pesticide’s product registration will need to be renewed after a specified number of years or the use of the product will become illegal. This ongoing responsibility is different from USDA’s oversight because, once the nonregulated petition is granted, USDA has no oversight role going forward under its regulations at 7 CFR Part 340. EPA has stated that this continuing oversight could raise specific challenges for forest trees with incorporated protectants because if the PIP’s registration is not renewed, the biotech trees could have migrated throughout the landscape and could not easily be recalled or eliminated.
U.S. FOOD AND DRUG ADMINISTRATION
FDA regulates biotech plants if they produce food for humans or feed for animals. FDA uses its food-safety authority under the Federal Food, Drug, and Cosmetic Act (FFDCA, 21 U.S.C. § 301 et seq.) to oversee the safety of all foods, including foods derived from biotech crops. Under this authority, FDA requires mandatory approvals of food additives but determined in a policy statement issued in 1992 that biotech food crops are generally not “food additives” requiring approval. Instead, FDA set up a voluntary consultation process by which biotech crop developers can share food safety data with FDA, and the agency can identify any deficiencies in the developer’s food and feed safety risk assessment (FDA, 1992). As of 2018, approximately 150 biotech crops had completed the FDA voluntary consultation process (FDA, 2018). Products that have completed the voluntary consultation process include biotech apple, plum, and papaya fruit trees but no forest tree species. However, it is anticipated that biotech forest trees that have edible portions, such as the transgenic chestnut, could voluntarily participate in the FDA consultation process (see Box 6-2). The FDA voluntary review process is limited to food and feed safety issues of a biotech plant. They do not address any aspects of this report’s definition of forest health.
STAKEHOLDER CRITICISM OF FEDERAL OVERSIGHT OF BIOTECH PLANTS AND TREES
There have been many criticisms voiced by different stakeholders about the Coordinated Framework and the oversight performed by EPA, FDA, and USDA. The committee specifically heard from stakeholders about their criticisms of that regulatory system as it applies to biotech trees developed to address forest health (Campbell, 2018; Costanza, 2018; Petermann, 2018; Strauss, 2018). It is outside the scope of this report to analyze the current federal oversight of biotech plants and trees and make observations or recommendations about that oversight. That type of analysis
was conducted (not specific to biotech trees) in two recent National Academies studies, and those reports identified recommendations for improving the regulatory system that would impact biotech tree oversight if those recommendations were implemented (NASEM, 2016, 2017).
MOVEMENT OF BIOTECH TREES DEVELOPED TO ADDRESS FOREST HEALTH ACROSS NATIONAL BORDERS
A unique aspect of a biotech tree developed to address forest health is that it will result in an unconfined release into the environment that is meant to propagate, spread, and persist without human oversight and control; this is significantly different from previously developed biotech trees, which are meant to be grown in orchards or plantations, and biotech crops, which are grown on managed farms. Consequently, biotech trees released to address forest health could cross national borders, raising the possibility of a biotech plant being approved for release in one country but not approved in the other country (such as between the United States and Canada). Does the U.S. regulatory system account for this cross-border movement, and how would it deal with the release of a biotech tree that migrated from another country?
When USDA obtains a petition for nonregulated status of a biotech plant, including a biotech tree, it is required to make the petition available to the public and seek comment as well as publicly announce its decision. This process, as well as the Trilateral Technical Working Group of agricultural biotechnology regulators from Canada, Mexico, and the United States would ensure that the neighboring countries are aware of any potential biotech trees that could migrate across borders (Doley, 2018). In addition, while the Plant Pest Risk Assessment need not consider cross-border impacts, USDA’s compliance with NEPA also requires that USDA comply with Executive Order 12114 on Environmental Effects Abroad of Major Federal Actions (Carter, 1979).15 Therefore, USDA’s EA or EIS would need to analyze any environmental effects of migration of the approved biotech tree across the U.S. border, although USDA has no authority to base its decision to grant or deny the petition on that analysis. That analysis, as discussed above, might include some aspects of forest health but likely will not comprehensively cover the topic. Although there are no examples of any detailed analysis required by Executive Order 12114 (Carter, 1979) for biotech plants or trees by USDA, an example of this type of analysis can be found in the NEPA compliance documents for FDA’s decision to approve the fast-growing transgenic salmon, which involves production in Canada and Panama (FDA, 2017).
If a biotech tree were introduced in Canada and migrated across the border to the United States and the tree was a product that falls within the regulatory authority of USDA under the Plant Protection Act, then USDA could impose quarantine or other mitigation activities to address the illegally growing biotech trees. USDA could also require the developer to submit a petition for nonregulated status or on its own without any document submitted by the developer conduct a Plant Pest Risk Assessment and environmental analysis under NEPA to determine whether the illegal activity should be allowed under the Plant Protection Act. Similar activities by the Canadian regulators might occur if the biotech tree were approved in the United States and migrated to Canada.
Similar issues could arise in relation to the sovereign boundaries of tribal nations within the United States. For example, Native American Haudenosaunee territories are within the historic range of the American chestnut tree, and the environmental release of a transgenic chestnut might eventually cross their sovereign borders. While consultation processes are formally in place between the U.S. federal government and tribal nations, they have not been uniformly successful in practice, and it is unclear how a difference in policy over a transgenic tree would be negotiated and managed (Barnhill-Dilling, 2018; Patterson, 2018).
___________________
15 See also the National Environmental Policy Act of 1969, as amended. Available at https://www.whitehouse.gov/sites/whitehouse.gov/files/ceq/NEPA_full_text.pdf. Accessed July 19, 2018.
The above actions—being aware of cross-border approvals, analyzing environmental impacts, and addressing migrating biotech trees through quarantine or after-the-fact approval—only apply to biotech trees that fall within the mandates of U.S. regulators (and Canadian regulators across the border). As stated earlier in this chapter, some biotech trees may not involve plant pests and fall outside USDA’s legal authority. If that is the case, those trees could be released in the United States without any federal oversight or notice to neighboring countries or tribal nations that they might cross sovereign borders. Similarly, a biotech tree planted in Canada that migrates to the United States might not violate USDA’s biotech regulations under the Plant Protection Act, if it does not involve “plant pests.” However, if it has a PIP incorporated in it, then it might be in violation of EPA’s regulations under FIFRA.
CURRENT REGULATORY SYSTEM FOR OTHER INTERVENTIONS TO ADDRESS FOREST HEALTH
Independent of whether biotech trees are developed and deployed to address forest health issues, there are and will continue to be other methods utilized to address forest health concerns. This includes the use of pesticides, biological control agents, and introduction of tree varieties with resilience characteristics produced through selective-breeding methods. It is helpful to compare how these interventions are regulated before they are adopted to identify differences in the evaluation processes and to determine how the federal regulatory system considers forest health, if at all.
Regulation of the Use of Conventional Pesticides to Address Forest Health
EPA regulates the use of all pesticides, including pesticides used in forests, using three laws: FIFRA, FFDCA, and the Pesticide Registration Improvement Act (7 U.S.C. § 136 et seq.). Any pesticide—which can include conventional pesticides (such as a chemical pesticide) as well as biopesticides (such as a microbial agent)—can obtain a registration from EPA if the agency determines that the pesticide will have no “unreasonable adverse effects on the environment” (FIFRA, 7 U.S.C. § 136(bb)) and there is a “reasonable certainty that no harm [to humans] will result from aggregate exposure [dietary, drinking water and residential exposure] to the pesticide residue” (FFDCA, 21 U.S.C. § 408(c)). Furthermore, FIFRA defines “unreasonable adverse effects on the environment” to mean “any unreasonable risk to man or the environment, taking into account the economic, social and environmental costs and benefits of the use of any pesticide” (7 U.S.C. § 136(bb)).
Before issuing a pesticide registration, EPA conducts a risk assessment, which has a human safety component and an ecological effects component. For conventional, outdoor pesticides that involve a potential food product, this assessment involves a complete suite of tests for pesticide active ingredients, including but not limited to toxicological tests for acute, subchronic, and chronic toxicity, carcinogenicity, neurotoxicity, and immunotoxicity and ecological effects on surrogate species for aquatic, terrestrial, and avian species as well as honeybees (40 CFR Part 158, “Data Requirements for Pesticides”).16 A similar process occurs for biochemical and microbial pesticides except that, because those products are naturally found in the environment, there is a tiered testing scheme. Under that testing scheme, if no adverse effects are found in lower tiers, there is no need for testing in higher tiers.17 According to EPA, there are no special regulations or data requirements specific to pesticides used in a forest, and they are assessed under the same procedures and
___________________
16 Pesticides that are designed for use on (or produced in) plants or animals, whose byproducts are not part of the human food supply, generally do not require the carcinogenic and other long-term bioassays listed. Available at https://www.gpo.gov/fdsys/granule/CFR-2012-title40-vol25/CFR-2012-title40-vol25-part158. Accessed July 20, 2018.
17 If a biopesticide is imported into the United States, it also requires a permit issued by USDA under the Plant Protection Act.
standards as pesticides used in other contexts. However, it is clear that the process for registering any pesticide is extensive and requires addressing several different safety questions with the generation of safety data.
Based on the information provided by EPA, the registration process for pesticides used in forests does not formally take forest health into consideration. However, some portions of their ecological assessment (ecological exposure and toxicity) require data and analysis that address areas that fall within the definition of forest health:
- The assessment of ecological effects on aquatic, terrestrial, and avian species as well as honeybees;
- The assessment of environmental fate of the pesticide in various media and its persistence; and
- Testing and assessing the impacts on nontarget organisms.
EPA is not required to conduct an EIS under NEPA when it registers a pesticide, and so the risk assessment is the only avenue for considering forest health parameters. However, the FIFRA definition of “unreasonable adverse effects on the environment” could allow EPA to integrate social and economic impacts involving forest health into its decision process.
Other agencies, such as the U.S. Forest Service, have their own policies and requirements for the use of pesticides for land they manage. According to the U.S. Forest Service, pesticides are one component of Integrated Pest Management “to prevent, control, or manage unwanted native plants, animals, and pathogens, and non-native invasive species on all areas of the National Forest System” (USDA-FS, 2013). To use a pesticide on national forest lands, the pesticide use must be reviewed and approved. The form used for the approval asks for standard information when using a pesticide (e.g., pesticide product, application information, treatment area), but no information about potential ecosystem service impacts. The pesticide approval process includes approving the application and complying with NEPA (USDA-FS, 2013).
The U.S. Forest Service states that it places “high priority on human and ecological health and safety” and that any use of pesticides “must be based on analysis of effectiveness, specificity, environmental impact, economic efficiency and human exposure” (USDA-FS, 2018a). Thus, the U.S. Forest Service conducts a human health and ecological risk assessment (HERA) for commonly used pesticides. According to the U.S. Forest Service, “these documents are used to determine the probability of adverse effects to humans, wildlife, and the environment from the use of pesticides” (USDA-FS, 2018b). The U.S. Forest Service states that it is required to do a HERA and cannot rely solely on the EPA safety demonstration under FIFRA. According to the U.S. Forest Service, EPA often considers many forestry pesticide uses to be minor so the U.S. Forest Service must evaluate “the project-specific rates, spectrum of target and non-target organisms, and specialized exposure scenarios” in its HERA (USDA-FS, 2018c). Therefore, the U.S. Forest Service does evaluate the forest-specific impacts of pesticides for potential human and environmental impacts, which are a part of forest health and the ecosystem services a forest provides. However, the HERA is primarily a quantitative, classic risk assessment and does not encompass many ecosystem services, such as cultural services.
Regulation of Biological Control Agents to Address Forest Health
Biological control agents are another method to address the impacts of insect pests and pathogens on forest health. USDA regulates biocontrol agents under the “plant pest” provisions of the Plant Protection Act, which is the same program that regulates certain biotech plants and trees. A PPQ 526 permit is required “for the importation, interstate movement and environmental release of
plant pests (plant feeding insects, mites, snails, slugs, and plant pathogenic bacteria, viruses, fungi, etc.), biological control organisms of plant pests and weeds, bees, parasitic plants and Federally listed noxious weeds” (USDA-APHIS, 2017d). Thus, all research involving a potential biocontrol agent that might be used in a forest, as well as the release of that biocontrol agent into the environment, requires a permit from USDA. The research permits are designed to ensure that the biocontrol agent does not escape and persist in the environment, and so the research is conducted in a highly controlled setting. The release permits are granted only after a thorough review and assessment of potential impacts in the environment. In addition to the plant pest analysis, USDA also conducts a biological assessment to comply with the Endangered Species Act and either an EA or an EIS to comply with NEPA. The scope of the NEPA analysis is the whole continental United States, and it considers cumulative effects. Compliance with NEPA for the release of a biocontrol agent is similar to complying with NEPA for a biotech plant. The analysis covers only some aspects of forest health and ecosystem services. According to USDA, it can take more than a decade to identify, rear, and test biocontrol agents intended to control invasive plants or insects (Rose, 2018).
Assisted Migration or Habitat Restoration of Tree Varieties
Another method to address forest health issues is to plant existing tree varieties in new locations or to restore the existing habitat with selectively bred, genetically improved tree varieties that are resilient to the threat. The planting of trees can occur in one of three different forest settings: (1) federally owned forests; (2) state-owned forests; or (3) privately owned lands. The ownership of the land is the critical factor in determining the regulatory oversight for those plantings.
For privately owned lands, the private landowner has no significant restrictions limiting what trees can be planted on his or her property unless the planting of the tree may harm a listed endangered species or its habitat, in which case the Endangered Species Act would apply (Shirey and Lamberti, 2010). In addition, the Plant Protection Act only applies if the plant species is either a noxious weed or plant pest or if the tree that is being moved or planted harbors a plant pest or a noxious weed. Thus, even an endangered tree can be planted outside its normal range if it is done on private land. This circumstance arises in the case of the endangered Florida torreya (Torreya taxifolia). Members of the Torreya Guardians18 are openly planting these trees in nonnative environments in 12 states and the District of Columbia,19 and those actions do not violate any federal law. Nor is there any requirement to assess the impact of those new species on the forest health of the private lands where they are being planted. This circumstance would also apply to a biotech forest tree that did not fall within the regulatory authority of USDA (not a plant pest) or EPA (not a pesticide). For a biotech tree that is regulated by USDA, once the regulatory process has been satisfactorily completed, the biotech tree could be planted on privately owned land without triggering any other regulatory requirements or government policies. For a biotech tree regulated by EPA, EPA oversight continues after registration and conditions could be placed on how the tree is planted in the environment. It is anticipated, for example, that the biotech American chestnut tree will be reintroduced through plantings on private lands.
It is unclear whether planting a nonnative tree species or planting a new variety of an existing tree on federal lands requires any special permit or regulatory review that assess the impact of the introduction on forest health. The U.S. Forest Service has a definition of “assisted migration,” which is a “management approach whereby resource managers physically move species (individuals, seeds, etc.) to new locations assessed suitable under changed climate conditions” (USDA-FS, n.d.). However, there does not seem to be any specific policy on the use of assisted migration on Forest
___________________
18 See http://www.torreyaguardians.org.
19 The District of Columbia, Florida, Georgia, Indiana, Massachusetts, Michigan, New Hampshire, North Carolina, Ohio, Oregon, Pennsylvania, Tennessee, and Wisconsin.
Service lands (Millar, 2015). It also has a Native Plant Materials Policy, and its Forest Service Manual has a chapter about “Vegetation Ecology” (USDA-FS, 2008). Those two policy documents give primary consideration to the planting of “genetically appropriate native plant materials” and restrict nonnative plant material use to limited conditions, such as emergency conditions to protect basic resource values, when native plant material is not available, or in highly altered plant communities, such as road cuts or log landings.
In 2016, the U.S. Forest Service finalized its Ecosystems Restoration Policy (USDA-FS, 2016:24785) to provide “broad guidance for restoring ecosystems on National Forest System lands so that they are self-sustaining and, if subject to disturbances or environmental change, have the ability to reorganize and renew themselves.” It specifies a science-based approach that the forest can be resilient for multiple uses. In particular, the policy states that when developing plans that include restoration, the U.S. Forest Service should include
- Factors such as the following:
- public values and desires;
- natural range of variation;
- ecological integrity;
- current and likely future ecological capabilities;
- a range of climate and other environmental change projections;
- the best available scientific information; and
- detrimental human uses.
- technical and economic feasibility to achieve desired future conditions.
- ecological, social, and economic sustainability….
- the social, economic, and ecological influences of restoration activities at multiple scales. (USDA-FS, 2016:24792)
Thus, this policy seems to require that the U.S. Forest Service consider a number of factors that fall within this report’s broad definition of forest health when deciding about restoring ecosystems affected by natural disturbances and climate change. Depending on how it is implemented, it could result in an analysis covering more comprehensively many of the ecosystem services provided by forests.
For lands managed by the U.S. Department of the Interior’s Fish & Wildlife Service (FWS), “there are no written guidelines or policy for introductions” within the historical range, augmentation of an existing population, and translocation (Johnson et al., 2013; Communication from the U.S. Fish & Wildlife Service, Washington, DC, to the National Academies of Sciences, Engineering, and Medicine’s Committee on the Potential for Biotechnology to Address Forest Health, March 2018). However, the planting of the trees would need to comply with the Endangered Species Act and the Migratory Bird Treaty Act. For National Wildlife Refuges, nonnative communities are not allowed unless there is no feasible alternative. In addition, the FWS has a Biological Integrity, Diversity and Environmental Health policy (FWS, 2001) for maintaining and restoring lands and waters in the National Wildlife Refuge System. That policy states that if there are lost or degraded areas, they should be restored to first mimic historic conditions and only use nonnative plant communities if there is no feasible alternative to accomplish the refuge’s purposes. The policy does allow for the use of genetically modified organisms but only if they are essential to accomplish refuge purposes and their use is approved by the Regional Chief (FWS, 2001).20
___________________
20 In addition to the U.S. Forest Service and the U.S. Fish & Wildlife Service, there are other federal agencies that manage federal lands, such as the National Park Service, the Bureau of Land Management, and the U.S. Department of Defense. The committee did not investigate the policies of those agencies for how they would address different interventions to address a forest health issue.
CONCLUSIONS AND RECOMMENDATIONS
Biotech trees developed to address forest health are regulated under the same statutes and regulations as any biotech plant. Forest health is not accounted for in the regulations for the use of biotechnology or for other approaches to mitigating forest tree insect pests or pathogens.
Conclusion: The current regulatory framework for biotech plants applies to biotech forest trees and does not impose any additional or different requirements for trees than other plants.
The Coordinated Framework for the Regulation of Biotechnology, which was established in 1986, specified that oversight of biotechnology products would be carried out using existing legislative statutes. The same statutes utilized by EPA, FDA, and USDA to regulate biotech plants apply to biotech trees, including biotech trees designed to address forest health. There are no specific regulations or policies that those agencies apply to biotech trees.
Conclusion: The current regulatory framework that applies to biotech trees that are developed to address forest health encapsulates very few elements of the committee’s comprehensive definition of forest health.
If a biotech forest tree falls within the legal mandate of USDA, EPA, and/or FDA, then they regulate the tree the same as other biotech plants. USDA will conduct the necessary scientific risk analysis to ensure that the biotech tree is not a plant pest or noxious weed. EPA will carry out the different risk analyses to register the biotech tree’s pesticidal component, such as impacts on the environment as well as impacts on humans and other species. If a biotech tree has a food or feed component, such as a biotech chestnut, FDA will review any voluntary submission from the developer to alert them to any potential food or feed safety concerns. While some of the regulatory assessment procedures by USDA or EPA may cover a few aspects of forest health (such as impacts on nontarget species or impacts on soil or groundwater), those regulatory procedures do not consider most aspects of forest health when regulating a biotech tree.
Conclusion: If a regulatory agency is required to comply with the National Environmental Policy Act when regulating a biotech tree, then some components of forest health will be analyzed.
NEPA requires that the federal government assess the environmental impact of its actions. To the extent that an agency regulating a biotech tree is required to conduct an EA or an EIS to comply with NEPA, some components of forest health will be analyzed before the government regulatory process is completed. That analysis is supposed to cover the “human environment,” which can include “ecological, aesthetic, historic, cultural, economic, social or health” effects. An analysis under NEPA must also comply with various Executive Orders, which encompass aspects of forest health (such as impacts on minority and low-income populations or consultations with tribal governments). However, out of the three agencies, only USDA undertakes the analysis required by NEPA for biotech plants. In addition, USDA cannot alter its regulatory decision based on the NEPA findings.
Conclusion: USDA only carries out a NEPA analysis—environmental assessment and/or environmental impact statement—for a small subset of biotech trees.
A review of the regulatory activity by USDA for biotech trees found that much biotech research (1,191 regulatory requests) has been carried out under USDA’s notification process, which does
contain an individual NEPA compliance assessment document. For biotech trees that have required a USDA permit to conduct research, only 4 percent of the 387 permits for biotech fruit and nonfruit trees required an EA and none required an EIS. For petitions to USDA for nonregulated status, for a large majority of those requests, USDA conducted an EA and not an EIS. USDA has completed 8 EISs and a draft EIS for its oversight of biotech plants out of approximately 124 petitions, and only the draft EIS has been for a biotech tree (a frost-tolerant eucalyptus). Therefore, only a small fraction of USDA’s oversight of biotech trees involves a NEPA analysis.
Conclusion: As is the case with other biotech plants, some biotech trees could become commercial products without any oversight by the three regulatory agencies.
The three major statutes that regulate biotech plants do not necessarily require oversight for all biotech plant applications. If USDA determines that a biotech tree does not fall within its legal mandate to regulate potential “plant pests,” then the agency does not have any regulatory authority over the tree and does not carry out any assessment, and the tree can be released into the environment without USDA’s approval. In 2014, USDA made such a determination for a biotech loblolly pine and could make such determinations in the future for biotech trees where a gene was introduced using biolistics, if modifications to the genome are made with genome editing, or if no Agrobacterium DNA remains in the final product. EPA only has regulatory oversight if the tree produces a pesticide, and many biotech trees will not produce a pesticide. FDA’s process is voluntary and only applies to plants that have food products or feed products, and many biotech forest trees may not have a food and/or feed use.
Conclusion: There are mechanisms in place to alert neighboring countries about biotech forest trees that could enter their territory, but biotech trees could migrate across a national border without notice if the biotech tree is not regulated in the country of origin.
If a biotech tree is regulated by USDA and/or EPA, before a decision is made, notice is given both to the public as well as to neighboring countries. Similarly, if Canada regulates a biotech tree, federal regulators are made aware of their actions. The public notice and coordination mechanisms allow for proper compliance with regulatory requirements in the country in which the biotech tree might migrate. However, if the biotech tree is not regulated in the country of origin, then it could migrate to a neighboring country, potentially in noncompliance with that country’s regulations.
Conclusion: Forest health also is not considered in the regulation of nonbiotech products designed to address forest health problems, such as biological control agents, pesticides, and assisted migration.
Other interventions to address forest health, such as pesticides, biological control agents, and assisted migration, also may require federal government review and oversight before deployment. When reviewing the regulatory processes for those interventions and the risk analysis conducted by the regulatory agency, the assessments or reviews conducted do not do a better job of incorporating forest health and ecosystem services into their analysis than the assessments conducted for biotech trees.
Conclusion: Some federal agencies have policies for the assisted migration of trees and/or the planting of biotech trees on federal lands, while private landowners can plant nonnative and biotech trees without violating any federal laws or policies.
The U.S. Forest Service has some policies that apply to assisted migration and the planting of species to restore forest ecosystems. Those policies seem to consider some aspects of forest health in deciding whether to introduce or reintroduce a tree species. FWS has no policy for introductions within their historical range, but it does have a policy for maintaining and restoring lands and waters in the National Wildlife Refuge System. Private landowners can plant virtually any commercially available tree on their lands, whether a native species, a nonnative species, or a biotech tree.
Recommendation: Regulatory agencies should explore ways to incorporate into their regulatory oversight responsibilities the ability to assess the impact on ecosystem services for biotech and nonbiotech products developed for improving forest health.
REFERENCES
Barnhill-Dilling, S.K. 2018. Engineered Governance: Public Engagement as a Path to Inclusive Environmental Governance in Genetically Engineered Trees. Ph.D. dissertation, North Carolina State University. Available at https://repository.lib.ncsu.edu/handle/1840.20/35317. Accessed November 21, 2018.
Campbell, F.T. 2018. Biotechnology? OK, But So Many More Issues Need to Be Resolved. Webinar presentation to the National Academies of Sciences, Engineering, and Medicine’s Committee on the Potential for Biotechnology to Address Forest Health. Available at http://nas-sites.org/dels/files/2018/02/Faith-Campbell-Presentation.pdf. Accessed July 27, 2018.
Carter, J. 1979. Exec. Order No. 12114. Environmental effects abroad of major federal actions. Federal Register 44:1957. Available at https://www.archives.gov/federal-register/codification/executive-order/12114.html. Accessed July 20, 2018.
Clinton, W.J. 1994. Exec. Order No. 12898: Federal actions to address environmental justice in minority populations and low-income populations. Federal Register 59(32):7629–7633. Available at https://www.archives.gov/files/federalregister/executive-orders/pdf/12898.pdf. Accessed July 19, 2018.
Clinton, W.J. 1997. Exec. Order No. 13045: Protection of children from environmental health risks and safety risks. Federal Register 62(78):19885–1988. Available at https://www.gpo.gov/fdsys/pkg/FR-1997-04-23/pdf/97-10695.pdf. Accessed July 19, 2018.
Clinton, W.J. 2000. Exec. Order No. 13175: Consultation and coordination with Indian tribal governments. Federal Register 65(218): 67249–67252. Available at https://www.gpo.gov/fdsys/pkg/FR-2000-11-09/pdf/00-29003.pdf. Accessed December 12, 2018.
Clinton, W.J. 2001. Exec. Order No. 13,186: Responsibilities of federal agencies to protect migratory birds. Federal Register 66(11):3853–3856. Available at https://www.energy.gov/sites/prod/files/nepapub/nepa_documents/RedDont/Req-EO13186migratorybirds.pdf. Accessed July 19, 2018.
Costanza, A. 2018. Regulation of GE Trees in the U.S. and Beyond. Webinar presentation to the National Academies of Sciences, Engineering, and Medicine’s Committee on the Potential for Biotechnology to Address Forest Health. Available at http://nas-sites.org/dels/files/2018/02/Adam-Costanza-Presentation.pdf. Accessed July 27, 2018.
Doley, B. 2018. Unintentional International Movement of GE Trees: Regulatory Response Scenarios. Webinar presentation to the National Academies of Sciences, Engineering, and Medicine’s Committee on the Potential for Biotechnology to Address Forest Health. Available at http://nas-sites.org/dels/files/2018/02/Bill-Doley-Presentation.pdf. Accessed July 30, 2018.
EOP (Executive Office of the President). 2016. National Strategy for Modernizing the Regulatory System for Biotechnology Products. Available at https://obamawhitehouse.archives.gov/sites/default/files/microsites/ostp/biotech_national_strategy_final.pdf. Accessed December 12, 2018.
EOP. 2017. Modernizing the Regulatory System for Biotechnology Products: An Update to the Coordinated Framework for the Regulation of Biotechnology. Available at https://obamawhitehouse.archives.gov/sites/default/files/microsites/ostp/2017_coordinated_framework_update.pdf. Accessed July 19, 2018.
EPA (U.S. Environmental Protection Agency). 2010. Biopesticides Registration Action Document: Coat Protein Gene of Plum Pox Virus. Available at https://www3.epa.gov/pesticides/chem_search/reg_actions/registration/decision_PC-006354_7-May-10.pdf. Accessed July 19, 2018.
EPA. 2015. Proposed Registration Decision for the New Active Ingredient Papaya Ringspot Virus Resistance Gene (Papaya Ringspot Virus Coat Protein Gene) in X17-2 Papaya (OECD Unique Identifier: UFL-X17CP-6). Available at https://www.regulations.gov/document?D=EPA-HQ-OPP-2015-0418-0005. Accessed July 27, 2018.
EPA. 2017. Current and Previously Registered Section 3 Plant-Incorporated Protectant (PIP) Registrations. Available at https://www.epa.gov/ingredients-used-pesticide-products/current-and-previously-registered-section-3-plant-incorporated. Accessed July 27, 2018.
FDA (U.S. Food and Drug Administration). 1992. Statement of Policy—Foods Derived from New Plant Varieties. Available at https://www.fda.gov/Food/GuidanceRegulation/GuidanceDocumentsRegulatoryInformation/Biotechnology/ucm096095.htm. Accessed July 19, 2018.
FDA. 2017. AquAdvantage Salmon – Response to Public Comments on the Environmental Assessment. Available at https://www.fda.gov/AnimalVeterinary/DevelopmentApprovalProcess/GeneticEngineering/GeneticallyEngineeredAnimals/ucm466220.htm. Accessed July 20, 2018.
FDA. 2018. Biotechnology Consultations on Food from GE Plant Varieties. Available at https://www.accessdata.fda.gov/scripts/fdcc/?set=Biocon. Accessed July 19, 2018.
FWS (U.S. Fish & Wildlife Service). 2001. 601 FW 3: Biological Integrity, Diversity, and Environmental Health. Available at https://www.fws.gov/policy/601fw3.pdf. Accessed July 30, 2018.
Gelvin, S.B. 2003. Agrobacterium-mediated plant transformation: The biology behind the “gene-jockeying” tool. Microbiology and Molecular Biology Reviews 67(1):16–37.
Johnson, R., S. Boyce, L. Brandt, V. Erickson, L. Iverson, G. Kujawa, and B. Tkacz. 2013. Policy and Strategy Considerations for Assisted Migration on USDA Forest Service Lands. Proceedings of the 60th Annual Western International Forest Disease Work Conference, October 8–12, 2012, Tahoe City, CA. Available at https://www.fs.fed.us/nrs/pubs/jrnl/2013/nrs_2013_johnson_001.pdf. Accessed July 27, 2018.
Millar, C. 2015. Assisted Migration for Forest Regeneration: Examples from Canada and a Bit on USFS Policy. Presentation at the meeting of the Consortium for Integrated Climate Research in Western Mountains on Assisted Migration Reforestation in Sierra Nevada Ecosystems. Available at https://www.fs.fed.us/psw/cirmount/meetings/AM2015/Millar_AM2015.pdf. Accessed July 30, 2018.
NASEM (National Academies of Sciences, Engineering, and Medicine). 2016. Genetically Engineered Crops: Experiences and Prospects. Washington, DC: The National Academies Press.
NASEM. 2017. Preparing for Future Products of Biotechnology. Washington, DC: The National Academies Press.
OSTP (Office of Science and Technology Policy). 1986. Coordinated Framework for Regulation of Biotechnology. Available at https://www.aphis.usda.gov/brs/fedregister/coordinated_framework.pdf. Accessed July 19, 2018.
Patterson, N. 2018. Haudenosaunee & the TG chestnut. Webinar presentation to the National Academies of Sciences, Engineering, and Medicine’s Committee on the Potential for Biotechnology to Address Forest Health, March 2.
Petermann, A. 2018. Genetically Engineered Trees. Webinar presentation to the National Academies of Sciences, Engineering, and Medicine’s Committee on the Potential for Biotechnology to Address Forest Health. Available at http://nas-sites.org/dels/files/2018/02/Anne-Petermann-Presentation.pdf. Accessed July 27, 2018.
Rose, R. 2018. USDA, Animal and Plant Health Inspection Service, Plant Protection and Quarantine: Biological Control Permitting Overview. Webinar presentation to the National Academies of Sciences, Engineering, and Medicine’s Committee on the Potential for Biotechnology to Address Forest Health. Available at http://nas-sites.org/dels/files/2018/02/Robyn-Rose-Presentation.pdf. Accessed July 27, 2018.
Shirey, P.D., and G.A. Lamberti. 2010. Assisted colonization under the U.S. Endangered Species Act. Conservation Letters 3(1):45–52.
Strauss, S. 2018. Why Biotech Solutions are Needed to Address Forest Health. Presentation to the National Academies of Sciences, Engineering, and Medicine’s Committee on the Potential for Biotechnology to Address Forest Health. Available at http://nas-sites.org/dels/files/2017/11/Steve-Strauss-Presentation.pdf. Accessed July 27, 2018.
USDA-APHIS (U.S. Department of Agriculture’s Animal and Plant Health Inspection Service). 2008. Perennials Statement. Available at https://www.aphis.usda.gov/brs/pdf/BRS_Perennials_Statement.pdf. Accessed July 19, 2018.
USDA-APHIS. 2011. USDA-APHIS Biotechnology Regulatory Services User Guide: Notification. Available at https://www.aphis.usda.gov/biotechnology/downloads/notification_guidance_0311.pdf. Accessed July 19, 2018.
USDA-APHIS. 2013. Questions and Answers: APHIS’ Notice of Intent to Prepare an Environmental Impact Statement for Freeze Tolerant Eucalyptus Lines. Available at https://www.aphis.usda.gov/publications/biotechnology/2013/faq_brs_combined_petitions.pdf. Accessed July 19, 2018.
USDA-APHIS. 2016. USDA-APHIS Biotechnology Regulatory Services Request to Extend Nonregulated Status from a Previous Determination: Extension Guidance for Developers. Available at https://www.aphis.usda.gov/brs/aphisdocs/guidance_ext_nonreg.pdf. Accessed July 19, 2018.
USDA-APHIS. 2017a. ArborGen, Inc. Petition (11-019-01p) for Determination of Non-regulated Status for Freeze Tolerant Eucalyptus Lines FTE 427 and FTE 435: Draft Environmental Impact Statement—April 2017. Available at https://www.aphis.usda.gov/brs/aphisdocs/11_01901p_peis.pdf. Accessed July 19, 2018.
USDA-APHIS. 2017b. Check Status: Search APHIS BRS Permits and Notification Data. Available at https://www.aphis.usda.gov/aphis/ourfocus/biotechnology/permits-notifications-petitions/sa_permits/ct_status. Accessed July 19, 2018.
USDA-APHIS. 2017c. Permits with Environmental Assessments. Available at https://www.aphis.usda.gov/aphis/ourfocus/biotechnology/reports/ea-table. Accessed July 19, 2018.
USDA-APHIS. 2017d. Regulated Organism and Soil Permits. Available at https://www.aphis.usda.gov/aphis/ourfocus/planthealth/import-information/permits/regulated-organism-and-soil-permits. Accessed July 20, 2018.
USDA-APHIS. 2017e. USDA-APHIS Biotechnology Regulatory Services Permit User’s Guide with Special Guidance for ePermits. Available at https://www.aphis.usda.gov/biotechnology/downloads/permit_guidance.pdf. Accessed July 19, 2018.
USDA-APHIS. 2018a. BRS Interstate/Release and Release Permits and Notifications. Available at https://www.aphis.usda.gov/aphis/ourfocus/biotechnology/permits-notifications-petitions/sa_permits/status-update/release-permits. Accessed July 19, 2018.
USDA-APHIS. 2018b. Petitions for Determination of Nonregulated Status. Available at https://www.aphis.usda.gov/aphis/ourfocus/biotechnology/permits-notifications-petitions/petitions/petition-status. Accessed July 19, 2018.
USDA-APHIS. 2018c. Regulated Article Letters of Inquiry. Available at https://www.aphis.usda.gov/aphis/ourfocus/biotechnology/am-i-regulated/regulated_article_letters_of_inquiry/regulated_article_letters_of_inquiry. Accessed July 19, 2018.
USDA-FS (U.S. Department of Agriculture’s Forest Service). 2008. Forest Service Manual, National Headquarters (WO), Washington, DC. FSM 2000—National Forest Resource Management. Chapter 2070—Vegetation Ecology. Available at https://www.fs.usda.gov/Internet/FSE_DOCUMENTS/fsbdev3_066309.pdf. Accessed July 30, 2018.
USDA-FS. 2013. Forest Service Manual, National Headquarters (WO), Washington, DC. FSM 2100—Environmental Management. Chapter 2150—Pesticide Use Management and Coordination. Available at https://www.fs.fed.us/dirindexhome/fsm/2100/wo_2150.doc. Accessed July 20, 2018.
USDA-FS. 2016. Ecosystem Restoration Policy. Federal Register 81(81):24785–24793. Available at https://www.gpo.gov/fdsys/search/pagedetails.action?granuleId=2016-09750&packageId=FR-2016-04-27&acCode=FR&collectionCode=FR. Accessed July 30, 2018.
USDA-FS. 2018a. Pesticide Management & Coordination. Available at https://www.fs.fed.us/foresthealth/protecting-forest/integrated-pest-management/pesticide-management. Accessed July 20, 2018.
USDA-FS. 2018b. Pesticide Management & Coordination: Health & Safety. Available at https://www.fs.fed.us/foresthealth/protecting-forest/integrated-pest-management/pesticide-management/pesticide-health-safety.shtml. Accessed July 20, 2018.
USDA-FS. 2018c. Pesticide Management & Coordination: Risk Assessments. Available at https://www.fs.fed.us/foresthealth/protecting-forest/integrated-pest-management/pesticide-management/pesticide-risk-assessments.shtml. Accessed July 20, 2018.
USDA-FS. n.d. Climate Change Resource Center: Management Options FAQs. Available at https://www.fs.usda.gov/ccrc/climate-basics/climate-faq/management-options. Accessed July 30, 2018.




















