3
Mineral Carbonation to Produce Construction Materials
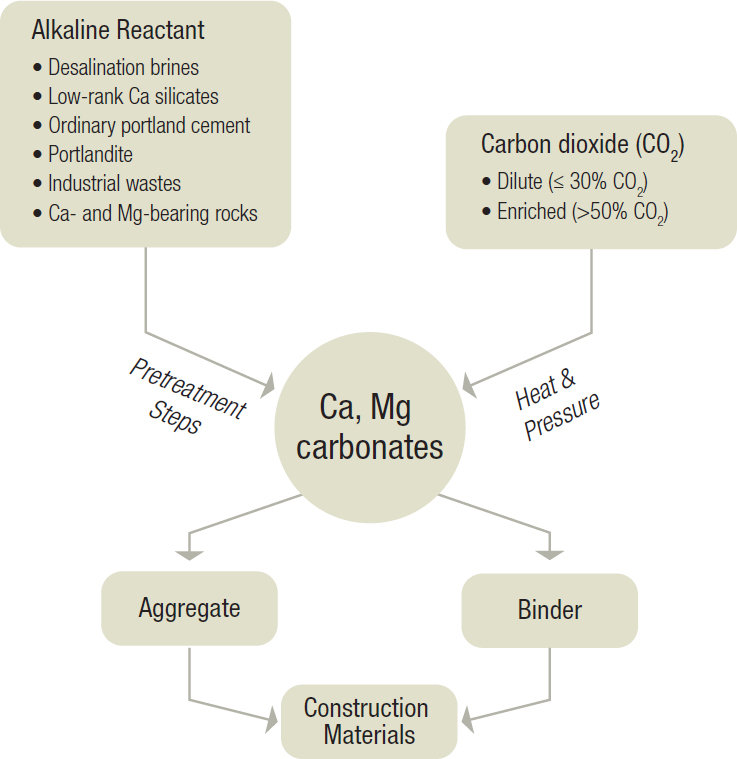
Mineral carbonation processes use waste carbon dioxide (CO2) to produce construction materials. Mineral carbonation offers an attractive route to CO2 utilization because (1) solid carbonates, the main products of mineral carbonation reactions, are already used in construction materials markets; (2) the chemistry involved in making carbonates based on calcium (Ca) and magnesium (Mg) is well known; (3) carbonation can consume large amounts of CO2 by chemically binding it into stable, long-lived mineral carbonates; and (4) the reaction of CO2 with alkaline solids is thermodynamically favored, thereby needing little, if any, extrinsic energy. This chapter assesses the current state of research in mineral carbonation, highlights pathways to convert CO2 to carbonates consisting of calcite (CaCO3), magnesite (MgCO3), or mixtures of the two (see Figure 3-1), and identifies market and environmental considerations relevant to the commercialization of mineral carbonation technologies.
Carbonates, commonly in a particulate or granular form, are predominately used in the production of cement and concrete, though they also find use in a variety of industrial applications, including in paper and food production.1 Depending on their end use, carbonates may be formed as nominally pure compounds or as mixtures with silicates, aluminates, and/or ferritic compositions. Natural carbonates are available from mined geological sources such as limestone quarries. In mineral carbonation, synthetic carbonates are made by contacting alkaline solids, often in an aqueous suspension, with carbon dioxide2 (Sanna et al., 2014; Vance et al., 2015). The conversion of CO2, a low-energy molecule, into solid mineral carbonates is one of only a few thermodynamically favorable reactions involving CO2 and can be accomplished at near-ambient temperatures. For this reason and because of the tremendous size of the con-
___________________
1 See https://pubchem.ncbi.nlm.nih.gov/compound/10112.
2 An example carbonation (CO2 mineralization) reaction involves the interaction of CO2 with portlandite to produce calcite and water: Ca(OH)2 + CO2 ↔ CaCO3 + H2O. The reaction is thermodynamically favored (ΔG = −74.61 kJ/mol) and exothermic (ΔH = −68 kJ/mol) and proceeds spontaneously under ambient conditions at relative humidities greater than 25 percent.
struction materials market (more than 50 billion tons of material such as concrete and mineral aggregates are produced annually3), mineral carbonation is considered to be among the largest and most energy-efficient routes for CO2 utilization. The market for construction materials produced through mineral carbonation using waste CO2 is expected to be further bolstered by the growing demand for “green” buildings and construction materials, for example, to meet and exceed design and construction standards such as LEED (Leadership in Energy and Environmental Design).4
___________________
3 See http://www.concreteconstruction.net/business/global-demand-for-construction-aggregates-to-exceed-48-billion-metric-tons-in-2015_o.
EMERGING TECHNOLOGIES FOR MINERAL CARBONATION
Mineral carbonation can be used to produce aggregates and binding agents suitable for use in construction materials. The process can involve a variety of approaches depending on the inputs used and the desired properties of the outputs. Some of these approaches are already employed commercially, suggesting the near-term potential for increased adoption of mineral carbonation in the large and growing construction materials market. More information on the existing commercial approaches is provided later in the chapter. However, a variety of factors influence the energy required and environmental impacts of these processes, which must be accounted for if mineral carbonation is to attain industrially relevant levels, thereby ensuring a net reduction in CO2 emissions.
Mineral Carbonation Products
The construction materials industry represents the primary market for products of mineral carbonation using waste CO2. Mineral carbonation can be used to make both aggregates and binding agents, which could potentially displace natural and existing synthetic sources of these important components of common construction materials.
Aggregates
Mineral aggregates, which range in size from micrometers to centimeters, are granular materials that form the dominant content of a concrete. By volume, mineral aggregates comprise roughly 60-80 percent of a typical concrete, with the rest consisting of a binding phase, or matrix. Currently, the vast majority of aggregates used globally are natural materials including sand, gravel and crushed rock, and other virgin materials mined from quarries, gravel pits, seabeds, and riverbeds. A smaller proportion are secondary and manufactured aggregates (Cresswell, 2007) including recycled concrete, by-products from industrial processes (e.g., blast furnace slag), and mineral aggregates that are purpose manufactured for use in concrete (e.g., geosynthetic aggregate and carbonate-cemented aggregate; Colangelo and Cioffi, 2013). Globally, pressures on natural aggregate resources have increased as a result of growing restrictions on mining activities and the depletion of quality aggregate sources in proximity to urban areas. These and other factors have driven an increased emphasis on secondary and manufactured aggregates; for instance, roughly 10 percent of aggregate produced, by mass, in Europe today consists of recycled or manufactured material, an approach which is promoted by limited landfill capacities and high landfill use fees in the region (ECO-SERVE Network, 2004). Mineral carbonation is expected to represent a promising avenue for aggregate production, as long as the synthetic products are economical and meet relevant quality and performance standards.
Concrete
Concrete is the dominant material used in the construction of buildings and other infrastructure. It consists of a mixture of aggregates (primarily sand and stone), water, chemical additives, and cement. Cement is the binding agent in concrete; upon reaction with water, cement forms a matrix that holds the solid components of concrete together, creating a synthetic rock that can be shaped as it hardens. Ordinary Portland cement (OPC), the most common type of cement, is produced from limestone, silica, clay, and iron compounds, along with other additives. While OPC has proved a strong and reliable building material over the past two centuries, its production and use have a large carbon footprint, generating approximately 0.7 tons5 of CO2 emissions per ton of cement produced and consumed globally.6 These CO2 emissions stem largely from the thermal decomposition of limestone (primarily CaCO3) to lime (CaO) and from fuel burning to provide the heat required to drive the formation of clinker phases in the kiln, a foundational step of OPC production. While the construction and cement sector has significantly reduced the CO2 impact of concrete production by improving kiln thermal efficiencies and combining cementitious materials such as fly ash and slags with OPC in the binder fraction (Snellings, 2016), these reductions have been more than offset by the global increase in cement consumption over the same period.
Concrete is most often used as ready-mix concrete or in the form of precast components. In the United States, ready-mix concrete, precast concrete, and concrete masonry comprise about 55, 25, and 15 percent of the overall concrete market, respectively.7 The differences in these market segments are broadly operational. In the typical application of ready-mix concrete, a slurry of concrete is transported by truck to a construction site, where it is placed into formwork staged with reinforcing steel8 and then allowed to harden into a structural component (i.e., floor, wall, beam, column, slab, etc.). Following hardening, the formwork is removed. In the case of precast concrete, a concrete slurry is cast into formwork or molds in the form of beams, columns, or slabs, often at a location far from construction sites. These components are then allowed to harden, following which the formwork is removed and the precast sections are transported to a construction site where they are assembled into buildings and infrastructure. Concrete is commonly classified by its compressive strength following 28 days of aging, which determines the load-bearing capacity of a given concrete formulation and the types of structural or nonstructural components it may be used for.
___________________
5 Direct carbon dioxide intensity of cement production.
6 See http://www.wbcsdcement.org/GNR-2015/world/GNR-Indicator_71DGWce-world-2015.html.
7 Estimated from the following sources: for bricks, blocks, and pavers, https://www.freedoniagroup.com/Bricks-Blocks-Pavers.html; for ready-mix concrete, https://www.statista.com/statistics/291597/revenue-of-ready-mix-concrete-manufacturing-in-the-us/; and for precast concrete, https://www.grandviewresearch.com/industry-analysis/precast-concrete-market.
8 Cementing binders and their derivatives like concrete present compressive strength that is superior to their tensile strength. For this reason, it is necessary to include either continuous (e.g., embedded steel rebar) or discontinuous (e.g., discrete fibers) reinforcement in such composites to enhance their tensile load-bearing performance.
Cement manufacturing plants are located in close proximity to the limestone quarries from which they draw their main feedstock, which are typically far from urban centers. Both limestone and cement are heavy, low-cost materials, and the cost of shipping over distances greater than about 250 miles, unless carried out over water, is often prohibitive. On the other hand, concrete, also a heavy, low-cost product, is often produced closer to the site of consumption. In particular, ready-mix concrete is typically made locally around the time of use from a mixture of cement, water, aggregates, and chemical additives. After the ingredients are mixed, the material can be transported, poured, or otherwise manipulated for only a few hours before it hardens. By contrast, cementing systems based on mineral carbonation show little if any hardening until all the mixed ingredients make contact with CO2, thereby offering greater control over the workability window. This is significant as the ease of controlling the rheology of such systems makes them potentially better suited for advanced (e.g., additive) manufacturing of structural components which feature superior strength-to-weight ratio, optimized topology, and complex geometries that could not be fabricated using existing casting or molding-based techniques.
Mineral Carbonation Processes
Mineral carbonation can be readily accomplished by contacting alkaline (mineral) solids, such as Ca- or Mg-rich materials, with CO2 in a fluid state or in solution. Depending on the types of alkaline solids used, these reactions can occur readily, even at ambient temperature and pressure. Carbonation often involves dissolution-precipitation reactions, specifically, dissolution of the elemental species from the reactant solid(s) and the solubilization of CO2 into the liquid phase (e.g., water) followed by the precipitation of carbonate mineral solids from a supersaturated solution. This process can result in the formation of many polymorphs of calcium carbonate (CaCO3), including amorphous calcium carbonate, vaterite, aragonite, and calcite. Calcite is the polymorph most often noted to exist at equilibrium (de Leeuw and Parker, 1998), though it is possible to direct the formation of one or multiple polymorphs (Chang et al., 2017a,b) by controlling the reaction conditions. In the presence of magnesium, magnesite (hydrated magnesium) carbonate phases may also form, although the formation of these types of carbonates is typically less preferred than their calcium-bearing variants (Pokrovsky, 1998). The need for alkaline reactants is due to the pH-dependent aqueous partitioning of dissolved carbon, in which carbonate ions (CO32−) are the dominant species at pH > 10.33 at 25°C as disclosed by a Bjerrum diagram (Morse and Mackenzie, 1990). Hence, increasing alkalinity results in increasing oversaturation of the solution with respect to CaCO3, favoring its precipitation. The precipitation of CaCO3 from aqueous carbonate is easier than formation from bicarbonate ions (HCO3−), which requires the deprotonation of the bicarbonate ion prior to the formation of CaCO3. The fact that the reaction generally requires caustic conditions in order to advance rapidly is considered the Achilles’ heel of the current paradigm of carbonation.
This approach of producing calcium and magnesium carbonates (particularly calcite, aragonite, and vaterite, which are polymorphs of CaCO3, and magnesite, MgCO3) from slurry-borne precursors is the basic pathway that can be exploited to produce mineral aggregates and binders for concrete. Carbonation can be accomplished using reactants including residue brines (e.g., from water desalination; Dindi et al., 2015), low-rank calcium silicates (Bukowski and Berger, 1979), hydrated lime (i.e., portlandite, Ca(OH)2; Moorehead, 1986), coal combustion residues (e.g., fly ash; Montes-Hernandez et al., 2009), and minerals and rocks such as serpentines and basalt (Oelkers et al., 2008). The abundance of Ca or Mg in the substrate is not a sufficient indicator for carbonation. Rather, the mobility and accessibility of Ca or Mg present (i.e., the reactivity of the solids) and the avoidance of surface passivation are critical factors that determine whether a given substrate may be suitable for carbonation processing or not (see Wei et al., 2018), which determines the degree of carbonation ultimately achieved.
The introduction of carbon dioxide into the “cementing formulation” can be plausibly accomplished by the following two pathways: (1) injecting carbon dioxide into the fresh concrete over a short period or (2) exposing preformed components of structural components to vapor-phase carbon dioxide, in dilute or concentrated form on the order of hours, within reactors. Expectedly, these pathways result in different levels of carbon dioxide uptake. For example, carbon dioxide injection into fresh concrete results in uptake that is limited by the overall rate of carbon dioxide reaction with calcium and magnesium, which in turn may be limited by the solubility of carbon dioxide in alkaline aqueous solution (i.e., ≤ 0.01 g CO2 per gram of cementitious components). On the other hand, the exposure of preformed components results in carbon dioxide uptake that is limited by the nature of the reactant used and potentially the geometry of the body, which may result in carbon dioxide transport limitations such that the carbon dioxide uptake typically ranges between 0.05 and 0.50 g CO2 per gram of cementitious components.
The following sections briefly describe a variety of approaches and possible inputs to mineral carbonation.
Residue Brines
Desalination operations, which provide potable water from seawater, and other operations such as oil and gas production generate large quantities of brines that are enriched in magnesium and calcium. When CO2 in a vapor state is injected into such brines at ambient temperature and pressure, hydrated carbonate products form spontaneously, as long as sufficient pH buffering is provided. While this approach can result in the formation of a wide range of hydrous calcium and magnesium carbonates, kinetic controls on precipitation reactions appear to often favor the formation of nesquehonite (Mg(HCO3)(OH)•2H2O) at the expense of other reaction products (Mignardi et al., 2011). This approach has been used,
for example, to produce nesquehonite-based products that yield comparatively low compressive strengths, on the order of 8 MPa (Glasser et al., 2016).
Low-Rank Calcium Silicates
Low-rank calcium silicates (CaO/SiO2 < 2, molar ratio) are well known to carbonate, especially in the presence of water and elevated CO2 concentrations. This reaction, often carried out under near-autoclave conditions, can be used to carbonate a range of otherwise slightly hydraulic low-calcium silicates including rankinite (Ca3SiO7) and wollastonite (Ca3SiO5). The carbonation of low-rank silicates in such a manner yields intermixed calcium carbonate and amorphous silica, which serve as effective binding agents (Bukowski and Berger, 1979). However, to realize favorable reaction kinetics, carbonation reactions in these systems need to be carried out using concentrated CO2; otherwise, the kinetics are typically too slow for practical exploitation.
Ordinary Portland Cement (OPC)
While low-rank silicates can be carbonated, the constituents of anhydrous OPC (CaO/SiO2 ≥ 2, molar ratio) are much more resistant to carbonation. This is because, in the presence of water, there exists a competition between hydration and carbonation, with hydration being the preferred pathway (Young et al., 1974). Even so, under ambient conditions, the reaction products of OPC hydration, for example, Ca(OH)2 and nonstoichiometric Cx-S-Hy phases (calcium-silicate hydrate, often, C = CaO, S = SiO2, H = H2O, x = 1.7, and y = 4.0), carbonate, albeit slowly, in the presence of atmospheric CO2 (0.4% CO2). However, due to the fine porosity and water saturation prevalent in hydrated OPC, carbonation of hydrated OPC and traditional concrete proceeds very slowly under ambient conditions due to the hindered diffusion of CO2 (Kashef-Haghighi and Ghoshal, 2013). As a result, it can take years for just the first few centimeters of a concrete to carbonate. Despite this slow rate, the tremendous volume of concrete emplaced worldwide can serve, over time, as a considerable sink for CO2 (Brady et al., 1996; Galan et al., 2010; Possan et al., 2017). In addition, crushed cementitious construction and demolition wastes in the form of fine particulates could achieve substantial, although inadvertent, CO2 uptake.
Portlandite
The carbonation of hydrated lime (portlandite, Ca(OH)2, often called slaked lime) has been practiced over millennia (Carran et al., 2012). Hydrated lime mortars take up atmospheric CO2 at ambient conditions over long periods of exposure, resulting in the formation of calcium carbonate. This process, while far too slow to be practical as a route for CO2 utilization (similar to the carbonation of hydrated OPC and traditional concrete under ambient
conditions), can be greatly accelerated under suitable conditions of temperature, pressure, CO2 concentration, and relative humidity (which dictates the water saturation state in the pore structure of solids prior to and during carbonation reactions). Similar to atmospheric carbonation, accelerated carbonation of portlandite also results in the formation of a monophasic CaCO3 product (often calcite) with robust cementation properties. The substantial tendency of portlandite to carbonate allows for accelerated carbonation to be accomplished even using relatively dilute streams of CO2 (≥ 5% CO2)—for example, using flue gas emitted by coal, natural gas, or cement plants—as long as suitable conditions of relative humidity and temperature can be maintained. Furthermore, portlandite offers among the highest CO2 uptake per unit mass (0.59 g CO2 per gram of portlandite), and its fast reaction kinetics allow near-complete carbonation to be achieved over the course of hours (Vance et al., 2015).
Industrial Wastes
Industrial (alkaline) wastes such as fly ash, slags, mine tailings, cement kiln dust, and air pollution control residues constitute the by-products of coal combustion, metal processing and mining, OPC production, and waste incineration, respectively. Such residues span a diversity of compositions as a function of the (1) parent coal composition, (2) ore-refining process, (3) ore composition, (4) waste make-up, and/or (5) combustion processes. However, in common, each of these industrial wastes features intrinsic alkalinity (e.g., they are mixtures of Ca and Mg with Al, Si, and alkalis) that can be neutralized by reaction with CO2. As an example, fly ashes can be broadly characterized as being Ca rich (Class C) or Ca poor (Class F) following standardized classifications.9 While Class C fly ashes are derived from the combustion of lignite or subbituminous coals, Class F fly ashes are derived from the combustion of bituminous or anthracitic coal. Recently, Wei et al. demonstrated an approach for fly ash carbonation wherein slurries of Ca-rich fly ash and water following exposure to dilute, vapor-phase CO2 offer robust cementation by the in situ formation of calcite (Wei et al., 2018). The carbonation of Ca-rich fly ashes is rapid, and the strength achieved is sufficient for structural construction. The extent of carbonation in such systems is critically linked to their mobile (readily reactive) Ca content. As a result, Ca-poor fly ashes offer little if any carbonation and strength gain compared to their Ca-rich counterparts. In fly ashes and other alkaline wastes the Ca and Mg present is not all mobile; as a result, such wastes often display carbonation extents that are substantially inferior to those inferred from their simple oxide (CaO or MgO) compositions (Monkman and Shao, 2006).
___________________
9 ASTM C618: Standard Specification for Coal Fly Ash and Raw or Calcined Natural Pozzolan for Use in Concrete. ASTM International.
Uptake Potential for Mineral Carbonation
Assuming a need for an alternative to OPC at today’s production levels, the amount of carbon dioxide that can be utilized—by mineral carbonation—can be established by considering the ability of diverse alkaline solids (e.g., fly ash, blast furnace slag, portlandite, and low-rank silicates) to react with carbon dioxide, thereby producing alkaline carbonates. A range of carbon dioxide uptake can be established by considering fly ashes and portlandite. Fly ashes feature a terminal carbon dioxide uptake of ≤ 0.05 g CO2 per gram of fly ash, and portlandite features a CO2 terminal uptake of 0.54 g CO2 per gram of portlandite over time scales of tens of hours. These uptakes suggest global carbon dioxide utilization levels ranging between 0.2 billion tons for fly ash10 and 2.2 billion tons for portlandite,11 if sufficient quantities of these materials were available. Since carbon dioxide mineralization resulting in cementation is expected to be achieved using a diversity of alkaline precursors, with a range of carbon dioxide uptake capacities, a rough estimate of approximately 1 billion tons of carbon dioxide on an annual basis is reasonable.
Carbonation of Ca- and Mg-Bearing Rocks
The formation of natural carbonate-bearing sedimentary rocks occurs through geological processes involving the weathering of rocks on the Earth’s surface by their reaction with water, releasing divalent ions such as Ca and Mg, which subsequently combine with CO2 to form carbonate minerals either through biogenic or abiogenic mechanisms. In certain localities rich in ultramafic and mafic rocks, such as ophiolitic formations, hyperalkaline Ca-rich ground-waters take up CO2 upon reaching the surface (for example, in springs), rapidly precipitating calcium carbonate in the form of travertine deposits (Kelemen and Matter, 2008). These processes can be mimicked in an accelerated manner by using rocks that are rich in Ca and Mg (e.g., basalt, gabbro, peridotite, and serpentinite) as carbonation reactants. However, because of their much lower reactivity than industrially produced materials, natural crystalline rocks require an initial pretreatment process before carbonation. These processes, which can be mechanical (e.g., ultrafine grinding) or thermal (e.g., exposure to temperatures above 600°C to remove chemically bound water), require large amounts of energy (Penner et al., 2004).
Factors Affecting Carbonation Dynamics
A number of factors affect carbonation dynamics and CO2 uptake of these mineral carbonation approaches. First, carbonation reactions, while often unaffected by flue gas contaminants such as particulate matter and acid gases (e.g., SOx and NOx), are sensitive to the presence and state (liquid or vapor) of moisture, which may be present or liberated over the
___________________
10 0.05 g CO2 /g fly ash × 4.1 billion tons = 0.2 billion tons of fly ash.
11 0.54 g CO2 /g portlandite × 4.1 billion tons = 2.2 billion tons of portlandite.
course of carbonation. This is because water, although appropriate for accelerating carbonation reactions on mineral surfaces, when condensed within pores retards the diffusive transport of CO2 (and hence carbonation kinetics) toward the reactants. Therefore, it is important to control liquid water saturation levels within microstructures, such as when carbonation is carried out in precast components. Second, the solubility of CO2 decreases with increasing temperature, reducing the concentration of CO2 in the liquid and promoting precipitation of calcite by decreasing its solubility. Hence, the temperatures over which carbonation reactions are carried out need to be considered and controlled. Third, as is typical for fluid-solid reactions, the surface area of the reactants (particle size) affects reaction kinetics. Although fine particles can accelerate kinetics, their production is energy intensive due to the need for grinding. In addition, their use may complicate slurry processing, due to their tendency to agglomerate, and result in refined microstructures that show retarded liquid and vapor transport.
All of the approaches described above result in the formation of calcium carbonate polymorphs (aragonite, vaterite, calcite; Chang et al., 2017a,b) and in certain cases hydrous silica (Ashraf and Olek, 2016), or hydrated calcium silicates, as the reaction products that glue the composite together, thereby imparting strength (Fernández Bertoz et al., 2004). These products are stable under ambient conditions of temperature, pressure, and CO2 concentration over geological time scales, as evidenced by the widespread deposits of limestone in nature.
Market Considerations
Construction materials including OPC, concrete, and mineral aggregates constitute the largest material flows in the world, second only to water. Each year, nearly 30 billion tons of concrete (World Business Council, 2009) are generated globally from a production base of 4.1 billion tons of OPC (USGS, 2017) and nearly 52 billion tons of mineral aggregates12 (which, beyond their use in concrete, find use in engineered barriers and road base applications; Tepordei, 1999). Given the scale of these markets, mineral carbonation products may have the potential to utilize up to 1 gigaton (Gt) of CO2 annually (Global CO2 Initiative, 2016) if they were to replace existing products. The large production volumes offer substantial economies of scale for conventionally produced products, resulting in costs on the order of $50 per ton for concrete (Villere, 2015), $100 per ton for OPC, and $10 per ton for aggregates (USGS, 2018). Taken together, these numbers define a cost basis for both existing and emerging construction materials. They also highlight some of the challenges that any new product might face: competition with well-entrenched products with low costs that can be produced worldwide.
In general, concrete, OPC, and aggregates incorporate elements (e.g., Si, O, Al, and Fe) that are abundant in the Earth’s crust (≥3 mass percent), with minor distributions of Ca, the alkalis K and Na, and Mg (Fleischer, 1953). This abundance ensures that the raw materials
___________________
12 See https://www.prnewswire.com/news-releases/the-freedonia-group-world-construction-aggregates-to-reach-517-billion-metric-tons-300241261.html.
required for production, such as limestone and clays, are (1) well dispersed geographically, (2) economical to extract, and (3) available in quantities large enough to satisfy prevailing and anticipated demand over long time scales. There is, for example, sufficient limestone in the Earth’s crust to support construction demands for more than 100,000 years (Biernacki et al., 2017). In addition, demolition wastes are also present in large volumes; while construction and demolition wastes may be recycled for use as road base and for limited use as concrete aggregates, the vast majority of these wastes continue to be landfilled (EPA, 2016).
While the construction sector offers large material flows cumulatively, the manufacture of construction materials is a localized activity. For example, there are 5,500 ready-mix concrete plants,13 hundreds of precast concrete plants,14 and nearly 100 OPC production plants (Portland Cement Association, 2013) in the United States alone. This localization and fragmentation implies that if mineral carbonation is to be used to produce construction materials that can replace existing materials, (1) CO2 will need to be consumed in many discrete locations to produce a range of materials and products, and (2) the logistics of CO2 access and transportation of manufactured products will be critical for economic viability.
There are substantial efforts under way to produce construction materials via mineral carbonation. Most are being undertaken by startup companies and precommercial entities, which fall into two groupings: (1) those producing binding agents in concrete (examples include Solidia Technologies, Carbon Upcycling UCLA, CarbonCure, and Carbstone Innovation) and (2) those producing synthetic aggregates (examples include Carbon8 Systems and Blue Planet):
- Solidia Technologies15 is using low-rank silicate(s) as the reactant for carbonate mineralization. While low-rank silicates, which feature a CO2 uptake of up to 0.38 g CO2 per gram of reactant, can be produced in existing OPC production kilns, their carbonation processing so far involves purified CO2, secured from industrial suppliers, at superambient conditions. This renders the approach well suited for ex situ production, similar to precast concrete. The reaction products, which include intermixed CaCO3 and SiO2 (hydrous silica), offer robust cementing behavior.
- Carbon Upcycling UCLA16 is using portlandite and industrial wastes as the primary reactants for carbonate mineralization. The CaCO3 formed in situ serves as the cementation agent. While portlandite, which features CO2 uptake on the order of 0.59 g CO2 per gram of portlandite, can be produced in existing OPC production kilns, its carbonation processing, while compatible with dilute CO2 waste streams
___________________
13 See https://www.nrmca.org/concrete/data.asp.
14 Estimated from the number of certified precast plants by National Precast Concrete Association: https://precast.org/certification/, Precast/Prestressed Concrete Institute: https://www.pci.org, etc.
15 See http://solidiatech.com.
- (>5% CO2 v/v), requires the use of slightly above-ambient reaction temperatures. Therefore, the approach is suited for co-located ex situ production, for example, alongside a fossil-fuel power plant or an OPC production plant, which serve as a source of solid reactants, waste CO2, and/or heat.
- CarbonCure17 injects pure CO2 into ready-mix concrete formed during initial mixing. The injected CO2 reacts with either unreacted Ca, liberated from the dissolution of OPC or from OPC replacement materials such as slag or fly ash. The reaction products are formed in situ, then blended with the binder, thereby forming carbonate compounds that contribute to increased and somewhat accelerated strength gain as compared with traditional concrete. The CO2 uptake is approximately ≤ 0.01 g CO2 per gram of reactants, following Henry’s law. The approach enables reduction in cement use, thus offering the benefit of CO2 avoidance by reducing the need for traditional OPC production. This approach is currently being implemented across numerous ready-mix concrete plants in the United States.
- Carbstone Innovation18 is using slags produced from iron and steel manufacturing as the alkaline substrate for mineral carbonation. The process involves contacting fine slag particulates in an aqueous suspension with enriched CO2 resulting in the formation of CaCO3 as the cementing agent. While the CO2 uptake of slags varies as a function of their composition, it is anticipated that CO2 uptake on the order of 0.10 g CO2 per gram of slag can be realized. The approach is best suited for ex situ production alongside a source of both slags and waste CO2. It has been used to produce a diversity of preformed products.
- Carbon8 Systems19 is using heterogeneous air pollution control (APC) residues as the alkaline reagent for the production of carbonate aggregates. The process involves contacting APC residues with pure CO2 supplied by industrial vendors within a formulation containing other binders and fillers where the formation of CaCO3 allows for the creation of carbonate cemented aggregates. The approach yields CO2 uptake around 0.12 g CO2 per gram of solid reactants and is well suited for jurisdictions with high landfill use costs. The approach has achieved commercial operations in United Kingdom based on its ability to encapsulate and isolate APC residues in a carbonate matrix.
- Blue Planet20 is using alkaline rock and industrial wastes as the primary reactants for carbonate mineralization. The process involves the formation of synthetic layers of CaCO3 over a substrate, resulting in the formation of synthetic carbonate aggregates in a manner analogous to their formation in nature. The process is thought to be
___________________
17 See https://www.carboncure.com.
18 See https://www.carbstoneinnovation.be/en/.
19 See http://c8s.co.uk/.
- compatible with the use of dilute CO2 waste streams and is therefore well suited for co-location alongside fossil-fuel power plants or OPC production plants that could serve as a source of solid reactants and/or waste CO2.
Each of these approaches exploits the thermodynamic ease of mineral carbonation and the potential for cementation offered by calcium carbonate. While these efforts are in the early stage, they highlight the potential for CO2 utilization in the construction materials markets. However, the scalability and market viability of these approaches are affected by a diversity of factors, including (1) the purity and the availability of CO2, (2) the availability of low-cost alkaline reagents and/or facilities for their manufacture at scale, (3) the low-cost, commoditized nature of the existing analogous products, OPC and concrete, (4) restrictive building codes and standards wherein compliance is often a function of the material composition (e.g., OPC-based chemistries) rather than their engineering performance, and (5) the net amount of CO2 utilization that can be achieved.
Net Greenhouse Gas Emissions
Ultimately, a goal of mineral carbonation is to utilize CO2 in waste gas that would otherwise be emitted into the atmosphere. As a result, it is critical to consider whether mineral carbonation processes produce a net reduction in CO2 emissions once all the relevant material and energy balances are considered. Mineral carbonation approaches need to be carefully evaluated using consistent methods for life-cycle assessments (see Chapter 8) to allow clear estimation of net CO2 utilization that is achieved. Such analyses are important to evaluate pathways that may yield the largest levels of CO2 utilization, thus reducing the accumulation of CO2 in the atmosphere.
A RESEARCH AGENDA FOR MINERAL CARBONATION
The expanded use of mineral carbonation depends on addressing technical, economic, and market-acceptance criteria. If carbonated materials are to contribute to CO2 management, each of these factors must be addressed. For example, a technical solution that does not fulfill acceptance criteria or that cannot achieve the tremendous scale required to fulfill construction demands is unlikely to significantly impact industrial practices. Similarly, a product will not succeed if it does not fulfill the relevant engineering requirements. An additional challenge is the physical handling of carbon dioxide. The following sections outline key barriers to commercialization of mineral carbonation technologies along with key areas in which further research could help to overcome those barriers.
Barriers to Commercialization
The development of commercially viable carbonation technologies is impacted by gaps in our understanding of carbonation processes, technical challenges facing various carbonation approaches, and a variety of economic and social factors affecting the potential costs and market penetration of mineral carbonation technologies.
Chemical and Physical Barriers to Carbonation
While carbonation reactions often proceed readily under certain conditions, this is often due to favorable thermodynamics rather than design. Table 3-1 summarizes key barriers to mineral carbonation approaches using various precursors. Carbonation is commonly impeded by one of three factors: (1) competitive reactions, (2) the formation of surface-passivating films, and (3) the presence of water. The first factor is often a function of the tendency of solids to hydrate versus carbonate. As a result, Ca-rich solids that are highly hydratable (e.g., MIII-Ca3SiO5, alite; Taylor, 1997) typically show poor potential to carbonate vis-à-vis fully hydrated Ca-rich solids (e.g., Ca(OH)2). The second factor, passivating films, refers to dense films of calcite that form on carbonating surfaces and hinder further contact between the reactant solid and CO2. The formation of such physical barriers is a key reason why solids such as CaO (lime, a highly hydratable solid) carbonate only fractionally (often 10 percent or less at nonisothermal conditions; Montes-Hernandez et al., 2012), following which the reaction is arrested (Nikulshina et al., 2007). The third factor relates to the fact that water favors carbonation when adsorbed on surfaces (Beruto and Botter, 2000) but inhibits carbonation when
TABLE 3-1 Summary of key chemical and physical barriers associated with selected mineral carbonation inputs.
| Input | Key Barriers |
|---|---|
| Mature OPC-based concrete | At ambient conditions under mass transfer (diffusion) control, it takes years for carbonation to occur. |
| Fresh OPC-based concrete | Reaction rates are slow at ambient conditions. Carbon dioxide concentration in the slurry is limited. Moreover, carbonation is impeded in the presence of moisture. |
| Low-rank calcium silicates | A high concentration of CO2 is needed to achieve sufficiently fast reaction kinetics. |
| Hydrated lime | In dilute CO2, favorable reaction conditions require the maintenance of slightly above-ambient temperature conditions. |
| Industrials wastes (e.g., fly ash, slags, etc.) | The heterogeneity of these wastes results in broad variations in reaction kinetics, with their CO2 uptake being substantially lower than that estimated from their bulk oxide composition. |
condensed within the pores of a micro- or mesoporous solid (Burkan Isgor and Razaqpur, 2004; Goracci et al., 2017). This is especially relevant to the case of precast sections that are contacted with CO2 vapors. Since they are produced from slurries or are dry cast in dense suspensions, wherein mixtures of the solids are intermixed with water for lubricity, such sections feature substantial amounts of water trapped within their pores. Water trapping hinders the transport of vapor-phase CO2 into the microstructure, even when CO2 may be solubilized into the pore liquid. This effect becomes more prominent with the size of the sections such that larger components may take longer to carbonate than smaller components following a typical t1/2 (“diffusive”) scaling for CO2 penetration (Bouquet et al., 2009).
Barriers to Carbonation Processing
Two major issues that complicate carbonation processing are (1) the need for alkaline precursors that are progressively neutralized following contact with CO2 and (2) the need for sustained elevated pH levels in solution to promote carbonate precipitation. These requirements are problematic because, first, the production of the alkaline precursors and reactants typically requires high-temperature activation. For example, the production of portlandite (Ca(OH)2) requires the thermal desorption of CO2 from limestone (i.e., at T ≈ 800°C, p = 1 bar) to produce lime (CaO), which is subsequently hydrated to form portlandite.21 The portlandite thus formed, following contact with CO2, carbonates to re-form calcite. Considering that the mineralized CO2 released from the initially decarbonated limestone can be recaptured (but is not currently), this high-temperature process is, at best, CO2 neutral when viewed from the perspective of the mineralized CO2. While this is indeed less CO2 intensive than the traditional pathway of OPC production, the development of truly CO2-neutral or -negative pathways requires the development of new scalable low-temperature or hydrothermal routes for producing alkaline precursors, such as from waste streams containing Ca and Mg. Second, because the solubility of CaCO3 diminishes with increasing pH and CO2’s dissolution in water induces acidity (by the formation of carbonic acid, in equilibrium with dissolved CO2, pKa = 6.35), there is a need for alkaline buffering to sustain the precipitation of carbonates. Therefore, increasing the pH is the key means of accelerating carbonation rates. Therefore, it is necessary to develop chemical additives that can enhance carbonation rates by affecting reaction controls, whether kinetic or thermodynamic. This includes enhancing growth rates (for example, by epitaxial matching; Gehrke et al., 2005), facilitating surface dehydration to ease ion attachment or other interactions with adsorbed water (Ricci et al., 2017), and surface complexation that promotes subsequent adsorption and heterogeneous growth (Brady et al., 1996). These approaches could improve carbonation rates while reducing the extent of basicity that is needed to sustain fast mineral carbonation kinetics.
___________________
21 See https://www3.epa.gov/ttnchie1/ap42/ch11/final/c11s17.pdf (accessed October 10, 2018).
Availability and Suitability of CO2 Streams
While the pressure, temperature, and CO2 concentration required to achieve optimal reaction conditions for mineral carbonation remain largely unknown, it is clear that, for most alkaline reactants, kinetics is favored by increasing CO2 concentration (Cui et al., 2015; Sun et al., 2008). There exist significant industrial sources of concentrated CO2 streams which may present opportunities for carbon dioxide utilization (see Chapter 2). However, the vast majority of waste CO2 streams are dilute (≤ 25% CO2 v/v). Thus, for most alkaline feedstocks there is a need for either low-cost CO2 capture systems or the ability to handle large volumes of gas streams containing components other than CO2. A further complication is that dilute CO2 is most often available distant from major construction markets (cities). This may necessitate (1) the co-location of manufacturing facilities for carbonated materials alongside CO2 emissions sites, which may also offer waste heat that favors the advancement of chemical reactions, or (2) transporting CO2 to production and consumption centers. These aspects may impose additional costs related to CO2 capture and/or transport, especially since current CO2 distribution pipeline networks are limited; in the United States, for example, these exist primarily in Texas and Oklahoma (DOE, 2015) (see Figure 7-3). Broadly, in light of the low profit margins of the commoditized construction materials sector, it appears prerequisite to maximize the use of dilute CO2 streams. While concentrated and purified CO2 is supplied by vendors for a fee, dilute, unprocessed CO2 could conceivably be sourced at little or no cost, or at presumably the equivalent of a landfill tipping fee, so long as it could be consumed in proximity to its site of production.
Costs and Abundance of Precursors and Mineral Carbonation Products
OPC and coal combustion wastes are produced at industrial scales worldwide. As such, global production of these materials is currently on the order of 4.1 billion and 1 billion tons on an annual basis for OPC and coal combustion wastes, respectively, with pricing on the order of $100 per ton, and $10 to $100 per ton (depending on location and the material’s suitability for use in traditional concrete22), respectively. Reactants appropriate for carbonation, including portlandite and wollastonite (CaSiO3, a low-rank silicate), are currently produced at a global level approaching 350 million tons (USGS, 2017) and 0.8 million tons, respectively.23 In principle, OPC, hydrated lime, and wollastonite can all be produced using similar facilities (such as typical OPC plants), as long as suitable raw materials are available in the local vicinity. As such, based on volume scaling and the availability of raw materials, it might be possible to produce portlandite and wollastonite at cost parity to OPC, and with considerably reduced
___________________
22 For example, compare the prices of crystalline versus ground granulated blast furnace slag (U.S. Geological Survey Mineral Commodities Summary, https://minerals.usgs.gov/minerals/pubs/commodity/iron_&_steel_slag/mcs-2018-fesla.pdf) and of compliant versus noncompliant fly ash (Concrete Construction – Fly Ash, http://www.concreteconstruction.net/_view-object?id=00000153-8b9c-dbf3-a177-9fbd58630000).
23 See https://minerals.usgs.gov/minerals/pubs/commodity/wollastonite/mcs-2017-wolla.pdf.
CO2 intensity due to the reduced synthesis temperatures needed (Zulumyan et al., 2015) and their lower molar content of CaO per unit mass. While coal combustion wastes are currently readily available, this may change with a transition away from coal-fired electricity generation. It is unlikely that production of carbonated aggregates will be produced at cost parity compared to quarrying. Therefore, absent suitable incentives, carbonated binders may offer a more market-viable proposition than carbonated aggregates.
Shipping of construction materials, whether as constituents or as preformed elements, is expensive on account of their substantial bulk. Thus, their transport is cost effective only over a few hundred miles, if by road. While rail or over-water transport may be more cost effective in some cases, it typically requires shipment of much larger quantities of product. These logistical limitations suggest carbonation technologies may be most effectively implemented at multiple discrete sites rather than at large centralized operations, reducing the economies of scale.
Construction Codes and Standards
In general, it is desirable that a new product achieves performance equivalence, or ideally performance benefits, vis-à-vis a product that it is intended to displace. While this argument is logical for the vast majority of applications in construction, its application in the context of OPC-based products may differ. This is because the construction sector has gained empirical confidence in the use of OPC and traditional concrete as construction materials, and construction industry standards are notoriously slow to change (see Chapter 9). Furthermore, construction standards and codes are often jurisdictional (e.g., city by city or state by state). This results in fragmented compliance and acceptance standards that may inhibit or delay the market entry and adoption of new products. A further complication is that construction standards are often prescriptive rather than performance based; as such, they often define the compositions of materials that can be used. Pending external forcing through legislation (e.g., see AB262: Buy Clean California Act24 and AB32: Global Warming Solutions Act25), preferential government purchasing programs, or the imposition of CO2 taxes and penalties, this is a substantial challenge that has to be overcome before the use of carbonated materials can become widespread within the construction sector. Among other elements, this requires moving to a system of harmonized performance-based standards, which will accelerate the adoption and acceptance of new materials for construction applications and new tools that can model and predict long-term performance of new materials.
___________________
24 See https://leginfo.legislature.ca.gov/faces/billNavClient.xhtml?bill_id=201720180AB262.
Priority Research Areas
Mineral carbonation offers great potential for CO2 utilization in the short to medium term. While the technology for mineral carbonation processes is farther along than for many chemical and biological utilization pathways, further research and development is needed to address key barriers in mineral carbonation technologies, processing, and market adoption. Priority research areas include controlling carbonation reactions, process design, accelerating carbonation and crystal growth, green synthesis routes for alkaline reactants, structure-property relationships, analytical and characterization tools, and construction methodologies.
Controlling Carbonation Reactions
Research is needed to understand the fundamental chemical features that control the relative rates of carbonation and hydration. This could lead to improved selection of alkaline solids and reaction conditions.
Process Design
Research is needed to integrate mineral carbonation processes with existing carbon dioxide capture technologies. This could lead to improved process performance and ensure optimal carbon dioxide conversion rates and energy use efficiencies.
Accelerating Carbonation and Crystal Growth
Research is needed to develop additives for enhanced carbon dioxide solubility or structure-directing agents that accelerate particle growth. This could accelerate carbonation reactions such as crystal growth rates in solution beyond what is achieved simply by increasing the pH.
Green Synthesis Routes for Alkaline Reactants
Research is needed to develop energy- and carbon dioxide–efficient pathways and processes for producing alkaline solids that can be readily carbonated and do not require high-temperature activation. This could lead to energy- and carbon dioxide–efficient pathways.
Structure-Property Relationships
Research is needed to develop physical and instrumental assessment methods, improved modeling, and performance-based criteria for product properties. This could improve predictions of structure-property relations and increase the durability, viability, and acceptance of carbonated solids.
Analytical and Characterization Tools
Research is needed to develop new analytical tools for studying carbonation reactions in dense and viscous suspensions, as well as the evolution of microstructure across length scales. This could lead to new scientific tools to characterize mineral carbonation technologies.
Construction Methodologies
Research is needed to develop new material formulations with novel properties and to advance the use of additive manufacturing to construct components with superior strength-to-weight ratio, optimized topology, and more complex geometries compared to what can be made with existing construction methods. This could enable new categories of carbon utilization products.
FINDINGS AND RECOMMENDATIONS
Finding 3-1 CO2 utilization through the production of carbonate-based construction materials (binding agents, mineral aggregates, and concrete) appears to be closest to application at commercial scale compared to other emerging CO2 utilization technologies.
Finding 3-2 The engineering properties, performance, and long-term chemical durability of mineral carbonation–based construction materials need to be better established for such materials to gain acceptance as substitutes for today’s base materials.
Finding 3-3 The purity, pressure, and temperature of CO2 sources affect the suitability of a given CO2 waste stream for a given mineral carbonation pathway. However, such criteria remain undefined for the vast majority of mineral carbonation technologies.
Finding 3-4 Mineral carbonation and carbonate formation may be accomplished by a multiplicity of pathways. To better understand such pathways at the molecular level it is necessary to develop improved tools for in situ analysis, characterization, and computational modeling to describe the rheology of concentrated suspensions, the reaction kinetics of carbonation processes, and the evolution of microstructures during reaction and/or phase transformations.
Finding 3-5 Given the very large material flows in the construction sector, this industry offers a tremendous potential for CO2 utilization. However, matching and integrating CO2 from widely distributed sources with a diversity of potential
consumers and construction product types and markets poses logistical and commercial challenges.
Finding 3-6 The production of construction materials via mineral carbonation has the potential to consume ~1 Gt of CO2 annually. However, to do so, such construction materials must be able to displace existing products that are currently produced globally at low cost and in large volumes.
Recommendation 3-1 Researchers should continue research efforts to control carbonation reactions by understanding the fundamental chemical features that govern relative rates of carbonation and hydration.
Recommendation 3-2 Researchers should integrate mineral carbonation processes with existing carbon dioxide capture technologies.
Recommendation 3-3 Researchers should continue to develop additives for enhanced carbon dioxide solubility or structure-directing agents which will help accelerate carbonation and crystal growth.
Recommendation 3-4 Researchers should focus on energy- and carbon dioxide–efficient pathways and processes for producing alkaline solids that can be readily carbonated and do not require high-temperature activation.
Recommendation 3-5 Researchers should increase efforts in developing physical and instrumentation assessment methods, improved modeling, and performance-based criteria for product properties to improve predictions of structure-property relations which will increase durability, viability, and acceptance of carbonated solids.
Recommendation 3-6 Researchers should increase attention to developing new analytical tools for studying carbonation reactions in dense and viscous suspensions, as well as the evolution of microstructure across length scales.
Recommendation 3-7 Researchers should continue research efforts to develop new material formulations with novel properties and to advance the use of additive manufacturing to construct components with superior strength-to-weight ratio, optimized topology, and more complex geometries.
Recommendation 3-8 Critical chemical, physical, and market-acceptance data should be acquired, analyzed, and disseminated by researchers to enable the widespread use of mineral carbonation–based binding agents and aggregates at scale and over broad geographic areas.
REFERENCES
Ashraf, W., and J. Olek. 2016. Carbonation behavior of hydraulic and non-hydraulic calcium silicates: Potential of utilizing low-lime calcium silicates in cement-based materials. Journal of Materials Science 51(13):6173-6191.
Beruto, D. T., and R. Botter. 2000. Liquid-like H2O adsorption layers to catalyze the Ca(OH)2/CO2 solid–gas reaction and to form a non-protective solid product layer at 20°C. Journal of the European Ceramic Society 20(4):497-503.
Biernacki, J. J., J. W. Bullard, G. Sant, K. Brown, F. P. Glasser, S. Jones, T. Ley, R. Livingston, L. Nicoleau, J. Olek, et al. 2017. Cements in the 21st century: Challenges, perspectives, and opportunities. Journal of the American Ceramic Society 100(7):2746-2773.
Bouquet, E., G. Leyssens, C. Schönnenbeck, and P. Gilot. 2009. The decrease of carbonation efficiency of CaO along calcination–carbonation cycles: Experiments and modelling. Chemical Engineering Science 64(9):2136-2146.
Brady, P. V., J. L. Krumhansl, and H. W. Papenguth. 1996. Surface complexation clues to dolomite growth. Geochimica et Cosmochimica Acta 60(4):727-731.
Bukowski, J. M., and R. L. Berger. 1979. Reactivity and strength development of CO2 activated non-hydraulic calcium silicates. Cement and Concrete Research 9(1):57-68.
Burkan Isgor, O., and A. G. Razaqpur. 2004. Finite element modeling of coupled heat transfer, moisture transport and carbonation processes in concrete structures. Cement and Concrete Composites 26(1):57-73.
Carran, D., J. Hughes, A. Leslie, and C. Kennedy. 2012. A short history of the use of lime as a building material beyond Europe and North America. International Journal of Architectural Heritage 6(2):117-146.
Chang, R., D. Choi, M. H. Kim, and Y. Park. 2017a. Tuning crystal polymorphisms and structural investigation of precipitated calcium carbonates for CO2 mineralization. ACS Sustainable Chemistry & Engineering 5(2):1659-1667.
Chang, R., S. Kim, S. Lee, S. Choi, M. Kim, and Y. Park. 2017b. Calcium carbonate precipitation for CO2 storage and utilization: A review of the carbonate crystallization and polymorphism. Frontiers in Energy Research 5(17). doi: 10.3389/fenrg.2017.00017.
Colangelo, F., and R. Cioffi. 2013. Use of cement kiln dust, blast furnace slag and marble sludge in the manufacture of sustainable artificial aggregates by means of cold bonding pelletization. Materials 6(8):3139-3159.
Cresswell, D. 2007. MIRO Characterisation of Mineral Wastes, Resources and Processing Technologies—Integrated Waste Management for the Production of Construction Material. WRT 177/WR0115.
Cui, H., W. Tang, W. Liu, Z. Dong, and F. Xing. 2015. Experimental study on effects of CO2 concentrations on concrete carbonation and diffusion mechanisms. Construction and Building Materials 93:522-527.
de Leeuw, N. H., and S. C. Parker. 1998. Surface structure and morphology of calcium carbonate polymorphs calcite, aragonite, and vaterite: An atomistic approach. Journal of Physical Chemistry B 102(16):2914-2922.
Dindi, A., D. V. Quang, and M. R. M. Abu-Zahra. 2015. Simultaneous carbon dioxide capture and utilization using thermal desalination reject brine. Applied Energy 154:298-308.
DOE (U.S. Department of Energy) National Energy Technology Laboratory. 2015. A Review of the CO2 Pipeline Infrastructure in the U.S. DOE/NETL-2014/1681.
ECO-SERVE Network. 2004. Baseline Report for the Aggregate and Concrete Industries in Europe, Cluster 3: Aggregate and Concrete Production.
EPA (U.S. Environmental Protection Agency). 2016. Advancing Sustainable Materials Management: 2016 Recycling Economic Information (REI) Report. Available at https://www.epa.gov/smm/recycling-economic-information-rei-report (accessed October 10, 2018).
Fernández Bertos, M., S. J. R. Simons, C. D. Hills, and P. J. Carey. 2004. A review of accelerated carbonation technology in the treatment of cement-based materials and sequestration of CO2. Journal of Hazardous Materials 112(3):193-205.
Fleischer, M. 1953. Recent Estimates of the Abundances of the Elements in the Earth’s Crust. U.S. Geological Survey.
Galan, I., C. Andrade, P. Mora, and M. A. Sanjuan. 2010. Sequestration of CO2 by concrete carbonation. Environmental Science & Technology 44(8):3181-3186.
Gehrke, N., H. Cölfen, N. Pinna, M. Antonietti, and N. Nassif. 2005. Superstructures of calcium carbonate crystals by oriented attachment. Crystal Growth & Design 5(4):1317-1319.
Glasser, F. P., G. Jauffret, J. Morrison, J.-L. Galvez-Martos, N. Patterson, and M. S.-E. Imbabi. 2016. Sequestering CO2 by mineralization into useful nesquehonite-based products. Frontiers in Energy Research 4(3). doi: 10.3389/fenrg.2016.00003.
The Global CO2 Initiative. 2016. Carbon Dioxide Utilization (CO2U): ICEF Roadmap 1.0.
Goracci, G., M. Monasterio, H. Jansson, and S. Cerveny. 2017. Dynamics of nano-confined water in Portland cement: Comparison with synthetic C-S-H gel and other silicate materials. Scientific Reports 7(1):8258.
Kashef-Haghighi, S., and S. Ghoshal. 2013. Physico–chemical processes limiting CO2 uptake in concrete during accelerated carbonation curing. Industrial & Engineering Chemistry Research 52(16):5529-5537.
Kelemen, P. B., and J. Matter. 2008. In situ carbonation of peridotite for CO2 storage. Proceedings of the National Academy of Sciences of the United States of America 105(45):17295-17300.
Mignardi, S., C. De Vito, V. Ferrini, and R. F. Martin. 2011. The efficiency of CO2 sequestration via carbonate mineralization with simulated wastewaters of high salinity. Journal of Hazardous Materials 191(1):49-55.
Montes-Hernandez, G., R. Pérez-López, F. Renard, J. M. Nieto, and L. Charlet. 2009. Mineral sequestration of CO2 by aqueous carbonation of coal combustion fly-ash. Journal of Hazardous Materials 161(2):1347-1354.
Montes-Hernandez, G., R. Chiriac, F. Toche, and F. Renard. 2012. Gas–solid carbonation of Ca(OH)2 and CaO particles under non-isothermal and isothermal conditions by using a thermogravimetric analyzer: Implications for CO2 capture. International Journal of Greenhouse Gas Control 11:172-180.
Monkman, S., and Y. Shao. 2006. Assessing the carbonation behavior of cementitious materials. Journal of Materials in Civil Engineering 18(6):768-776.
Moorehead, D. R. 1986. Cementation by the carbonation of hydrated lime. Cement and Concrete Research 16(5):700-708.
Morse, J. W., and F. T. Mackenzie. 1990. Geochemistry of Sedimentary Carbonates. Amsterdam: Elsevier.
Nikulshina, V., M. E. Gálvez, and A. Steinfeld. 2007. Kinetic analysis of the carbonation reactions for the capture of CO2 from air via the Ca(OH)2–CaCO3–CaO solar thermochemical cycle. Chemical Engineering Journal 129(1):75-83.
Oelkers, E. H., S. R. Gislason, and J. Matter. 2008. Mineral carbonation of CO2. Elements 4(5):333-337.
Penner, L., W. O’Connor, D. Dahlin, S. Gerdemann, and G. Rush. 2004. Mineral Carbonation: Energy Costs of Pretreatment Options and Insights Gained from Flow Loop Reaction Studies. DOE/ARC–2004-042.
Pokrovsky, O. S. 1998. Precipitation of calcium and magnesium carbonates from homogeneous supersaturated solutions. Journal of Crystal Growth 186(1):233-239.
Portland Cement Association. 2013. U.S. Portland Cement Industry: Plant Information Summary. Skokie, IL: Portland Cement Association.
Possan, E., W. A. Thomaz, G. A. Aleandri, E. F. Felix, and A. C. P. dos Santos. 2017. CO2 uptake potential due to concrete carbonation: A case study. Case Studies in Construction Materials 6:147-161.
Ricci, M., W. Trewby, C. Cafolla, and K. Voïtchovsky. 2017. Direct observation of the dynamics of single metal ions at the interface with solids in aqueous solutions. Scientific Reports 7:43234.
Sanna, A., M. Uibu, G. Caramanna, R. Kuusik, and M. Maroto-Valer. 2014. A review of mineral carbonation technologies to sequester CO2. Chemical Society Reviews 43(23):8049-8080.
Snellings, R. 2016. Assessing, understanding and unlocking supplementary cementitious materials. RILEM Technical Letters 1:50-55.
Sun, J., M. F. Bertos, and S. J. Simons. 2008. Kinetic study of accelerated carbonation of municipal solid waste incinerator air pollution control residues for sequestration of flue gas CO2. Energy & Environmental Science 1(3):370-377.
Taylor, H. F. W. 1997. Cement Chemistry. London: Thomas Telford.
Tepordei, V. 1997. Natural Aggregates—Foundation of America’s Future. U.S. Geological Survey Fact Sheet 144-97.
USGS (U.S. Geological Survey). 2017. Mineral commodity summaries 2017: U.S. Geological Survey, 202 pp., https://doi.org/10.3133/70180197.
USGS. 2018. Mineral Commodity Summaries 2018. Reston, VA: U.S. Geological Survey, 200 pp., doi: 10.3133/70194932.
Vance, K., G. Falzone, I. Pignatelli, M. Bauchy, M. Balonis, and G. Sant. 2015. Direct carbonation of Ca(OH)2 using liquid and supercritical CO2: Implications for carbon-neutral cementation. Industrial & Engineering Chemistry Research 54(36):8908-8918.
Villere, P. 2015. 2015 Industry Data Survey. The Concrete Producer. Available at https://www.concreteconstruction.net/producers/2015-industry-data-survey_o (accessed September 11, 2018).
Wei, Z., B. Wang, G. Falzone, E. C. La Plante, M. U. Okoronkwo, Z. She, T. Oey, M. Balonis, N. Neithalath, L. Pilon, et al. 2018. Clinkering-free cementation by fly ash carbonation. Journal of CO2 Utilization 23:117-127.
World Business Council for Sustainable Development. 2009. The Cement Sustainability Initiative. Available at https://www.wbcsd.org/Sector-Projects/Cement-Sustainability-Initiative (accessed October 10, 2018).
Young, J. F., R. L. Berger, and J. Breese. 1974. Accelerated curing of compacted calcium silicate mortars on exposure to CO2. Journal of the American Ceramic Society 57(9):394-397.
Zulumyan, N., A. Mirgorodski, A. Isahakyan, H. Beglaryan, A. Gabrielyan, and A. Terzyan. 2015. A low-temperature method of the β-wollastonite synthesis.Journal of Thermal Analysis and Calorimetry 122(1):97-104.
This page intentionally left blank.
























